Chimeric Virus-Like Particles of Prawn Nodavirus Displaying Hepatitis B Virus Immunodominant Region: Biophysical Properties and Cytokine Response
Abstract
1. Introduction
2. Results
2.1. Generation of Recombinant Baculovirus Harbouring the MrNV Capsid Protein-HBV ‘a’ Determinant (Nc-aD) Coding Sequence
2.2. Expression, Purification and Quantification of the Nc-aD-Sf9 VLPs
2.3. Dynamic Light Scattering (DLS) Analysis
2.4. Circular Dichroism (CD) Spectroscopy
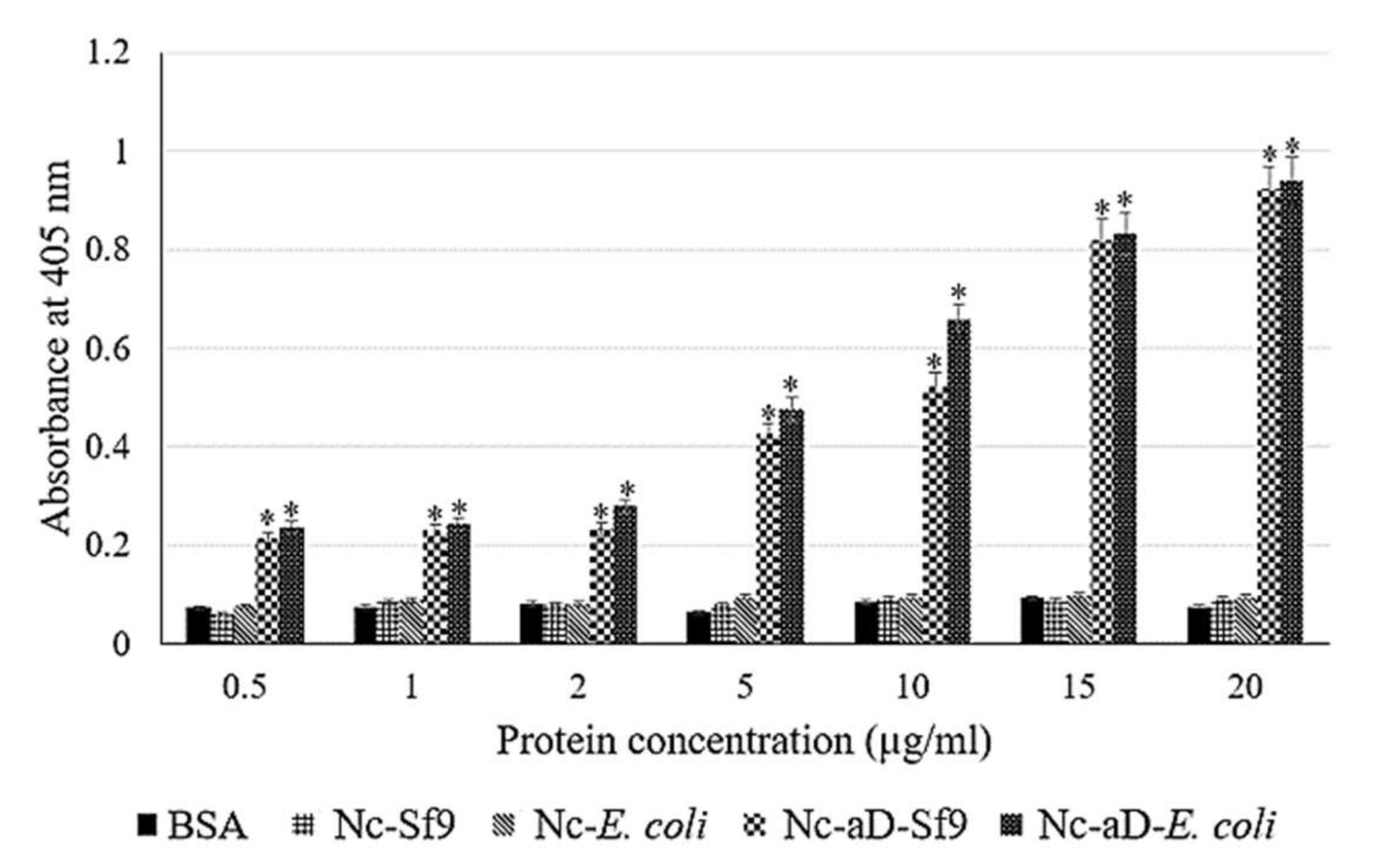
2.5. Enzyme-Linked Immunosorbent Assay (ELISA) of Nc-aD VLPs
2.6. Multiplex ELISA for Cytokine Quantification
3. Discussion
4. Materials and Methods
4.1. Plasmid Construction
4.2. Cell Line and Transfection
4.3. Production of Nc-aD in Sf9 Cells
4.4. Purification of Chimeric Nc-aD-Sf9 VLPs with Sucrose Density Gradient Ultracentrifugation
4.5. Production of Nc-aD and Nc in E. coli, and Nc in Sf9
4.6. SDS-PAGE and Western Blotting
4.7. Dynamic Light Scattering (DLS)
4.8. Circular Dichroism (CD) Spectroscopy
4.9. Enzyme-Linked Immunosorbent Assay (ELISA)
4.10. In Vivo Studies on BALB/c Mice
4.11. Multiplex ELISA for Cytokine Quantification
4.12. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Yong, C.Y.; Yeap, S.K.; Goh, Z.H.; Ho, K.L.; Omar, A.R.; Tan, W.S. Induction of humoral and cell-mediated immune responses by hepatitis B virus epitope displayed on the virus-like particles of prawn nodavirus. Appl. Environ. Microbiol. 2015, 81, 882–889. [Google Scholar] [CrossRef]
- Yong, C.Y.; Yeap, S.K.; Ho, K.L.; Omar, A.R.; Tan, W.S. Potential recombinant vaccine against influenza- A virus based on m2e displayed on nodaviral capsid nanoparticles. Int. J. Nanomed. 2015, 10, 2751–2763. [Google Scholar] [CrossRef]
- Yong, C.Y.; Yeap, S.K.; Omar, A.R.; Tan, W.S. Advances in the study of nodavirus. Peer J. 2017, 5, e3841. [Google Scholar] [CrossRef] [PubMed]
- Hanapi, U.F.; Yong, C.Y.; Goh, Z.H.; Alitheen, N.B.; Yeap, S.K.; Tan, W.S. Tracking the virus-like particles of Macrobrachium rosenbergii nodavirus in insect cells. Peer J. 2017, 5, e2947. [Google Scholar] [CrossRef] [PubMed]
- Goh, Z.H.; Tan, S.G.; Bhassu, S.; Tan, W.S. Virus-like particles of Macrobrachium rosenbergii nodavirus produced in bacteria. J. Virol. Methods 2011, 175, 74–79. [Google Scholar] [CrossRef]
- Kueh, C.L.; Yong, C.Y.; Masoomi Dezfooli, S.; Bhassu, S.; Tan, S.G.; Tan, W.S. Virus-like particle of Macrobrachium rosenbergii nodavirus produced in Spodoptera frugiperda (Sf9) cells is distinctive from that produced in Escherichia coli. Biotechnol. Prog. 2016, 33, 548–557. [Google Scholar] [CrossRef]
- Somrit, M.; Watthammawut, A.; Chotwiwatthanakun, C.; Ounjai, P.; Suntimanawong, W.; Weerachatyanukul, W. C-terminal domain on the outer surface of the Macrobrachium rosenbergii nodavirus capsid is required for Sf9 cell binding and internalization. Virus Res. 2017, 227, 41–48. [Google Scholar] [CrossRef]
- Goh, Z.H.; Mohd, N.A.S.; Tan, S.G.; Bhassu, S.; Tan, W.S. RNA-binding region of Macrobrachium rosenbergii nodavirus capsid protein. J. Gen. Virol. 2014, 95, 1919–1928. [Google Scholar] [CrossRef]
- Ong, H.K.; Yong, C.Y.; Tan, W.S.; Yeap, S.K.; Omar, A.R.; Razak, M.A.; Ho, K.L. An influenza A vaccine based on the extracellular domain of matrix 2 protein protects BALB/c mice against H1N1 and H3N2. Vaccines 2019, 7, 91. [Google Scholar] [CrossRef]
- Tan, W.S.; Ho, K.L. Phage display creates innovative applications to combat hepatitis B virus. World J. Gastroenterol. 2014, 20, 11650–11670. [Google Scholar] [CrossRef]
- Hyakumura, M.; Walsh, R.; Thaysen-Andersen, M.; Kingston, N.J.; La, M.; Lu, L.; Lovrecz, G.; Packer, N.H.; Locarnini, S.; Netter, H.J. Modification of asparagine-linked glycan density for the design of hepatitis B virus virus-like particles with enhanced immunogenicity. J. Virol. 2015, 89, 11312–11322. [Google Scholar] [CrossRef]
- Venkatakrishnan, B.; Zlotnick, A. The structural biology of hepatitis B virus: Form and function. Annu. Rev. Virol. 2016, 3, 429–451. [Google Scholar] [CrossRef] [PubMed]
- Gerlich, W.H. Do we need better hepatitis B vaccines? Indian J. Med. Res. 2017, 145, 414–419. [Google Scholar] [CrossRef]
- Tai, C.; Wu, J.; Chen, H.; Ni, Y.; Hsu, H.; Chang, M. The impact of hepatitis B vaccine failure on long-term natural course of chronic hepatitis B virus infection in hepatitis B e antigen–seropositive children. J. Immunol. 2017, 216, 662–669. [Google Scholar] [CrossRef]
- Cai, X.; Zheng, W.; Pan, S.; Zhang, S.; Xie, Y.; Guo, H.; Wang, G.; Li, Z.; Luo, M.A. Virus-like particle of the hepatitis B virus preS antigen elicits robust neutralizing antibodies and T cell responses in mice. Antiviral Res. 2018, 149, 48–57. [Google Scholar] [CrossRef]
- Netter, H.J.; Woo, W.; Tindle, R.; Macfarlan, R.I.; Gowans, E.J. Immunogenicity of recombinant HBsAg/HCV particles in mice pre-immunised with hepatitis B virus-specific vaccine. Vaccine 2003, 21, 2692–2697. [Google Scholar] [CrossRef]
- Wei, S.; Lei, Y.; Yang, J.; Wang, X.; Shu, F.; Wei, X.; Lin, F.; Li, B.; Cui, Y.; Zhang, H.; et al. Neutralization effects of antibody elicited by chimeric HBV S antigen viral-like particles presenting HCV neutralization epitopes. Vaccine 2018, 36, 2273–2281. [Google Scholar] [CrossRef] [PubMed]
- Ryu, C.J.; Gripon, P.; Park, H.R.; Park, S.S.; Kim, Y.K.; Guguen-Guillouzo, C.; Yoo, O.J.; Hyo, J.H. In vitro neutralization of hepatitis B virus by monoclonal antibodies against the viral surface antigen. J. Med. Virol. 1997, 52, 226–233. [Google Scholar] [CrossRef]
- Ninyio, N.N.; Ho, K.L.; Ong, H.K.; Yong, C.Y.; Chee, H.Y.; Hamid, M.; Tan, W.S. Immunological analysis of the hepatitis B virus ‘a’ determinant displayed on chimeric virus-like particles of Macrobrachium rosenbergii nodavirus capsid protein produced in Sf9 cells. Vaccines 2020, 8, 275. [Google Scholar] [CrossRef]
- Tan, G.H.; Yusoff, K.; Seow, H.F.; Tan, W.S. Antigenicity and immunogenicity of the immunodominant region of hepatitis B surface antigen displayed on bacteriophage T7. J. Med. Virol. 2005, 77, 475–480. [Google Scholar] [CrossRef]
- Howard, C.R.; Allison, L.M.C. Hepatitis B surface antigen variation and protective immunity. Intervirology 1995, 38, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Reed, J.; Reed, T.A. A set of constructed type spectra for the practical estimation of peptide secondary structure from circular dichroism. Anal. Biochem. 1997, 254, 36–40. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, F.; Carinhas, N.; Carrondo, M.J.T.; Alves, P.M. Insect cells as a production platform of complex virus-like particles. Expert Rev. Vaccines 2013, 12, 225–236. [Google Scholar] [CrossRef] [PubMed]
- Ho, K.L.; Gabrielsen, M.; Beh, P.L.; Kueh, C.L.; Thong, Q.X.; Streetley, J.; Tan, W.S.; Bhella, D. Structure of the Macrobrachium rosenbergii nodavirus: A new genus within the Nodaviridae? PLoS Biol. 2018, 16, 3000038. [Google Scholar] [CrossRef]
- Lanford, R.E.; Luckow, V.; Kennedy, R.C.; Dreesman, G.R.; Notvall, L.; Summers, M.D. Expression and characterization of hepatitis B virus surface antigen polypeptides in insect cells with a baculovirus expression system. J. Virol. 1989, 63, 1549–1557. [Google Scholar] [CrossRef]
- Eshaghi, M.; Tan, W.S.; Ong, S.T.; Yusoff, K. Purification and characterization of Nipah virus nucleocapsid protein produced in insect cells. J. Clin. Microb. 2005, 43, 3172–3177. [Google Scholar] [CrossRef]
- Krol, E.; Brzuska, G.; Szewczyk, B. Production and biomedical application of flavivirus-like particles. Trends Biotechnol. 2019, 37, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Pushko, P.; Tumpey, T.M.; Bu, F.; Knell, J.; Robinson, R.; Smith, G. Influenza virus-like particles comprised of the HA, NA, and M1 proteins of H9N2 influenza virus induce protective immune responses in BALB / c mice. Vaccine 2005, 23, 5751–5759. [Google Scholar] [CrossRef]
- Pabisch, S.; Feichtenschlager, B.; Kickelbick, G.; Peterlik, H. Effect of interparticle interactions on size determination of zirconia and silica based systems- A comparison of SAXS, DLS, BET, XRD and TEM. Chem. Phys. Lett. 2012, 521, 91–97. [Google Scholar] [CrossRef]
- Chong, L.C.; Ganesan, H.; Yong, C.Y.; Tan, W.S.; Ho, K.L. Expression, purification and characterization of the dimeric protruding domain of Macrobrachium rosenbergii nodavirus capsid protein expressed in Escherichia coli. PLoS ONE 2019, 14, e0211740. [Google Scholar] [CrossRef]
- Cao, J.; Luo, S.; Xiong, Y. The variability of amino acid sequences in hepatitis B virus. Virol. Sin. 2019, 34, 42–49. [Google Scholar] [CrossRef] [PubMed]
- Facchiano, A.M.; Colonna, G.; Ragone, R. Helix stabilizing factors and stabilization of thermophilic proteins: An X-ray based study. Protein Eng. 1998, 11, 753–760. [Google Scholar] [CrossRef]
- Vogt, G.; Argos, P. Protein thermal stability: Hydrogen bonds or internal packing? Fold. Des. 1997, 2, 40–46. [Google Scholar] [CrossRef]
- Zepeda-Cervantes, J.; Ramírez-Jarquín, J.O.; Vaca, L. Interaction between virus-like particles (VLPs) and pattern recognition receptors (PRRs) from dendritic cells (DCs): Toward better engineering of VLPs. Front. Immunol. 2020, 11, 1–22. [Google Scholar] [CrossRef]
- Bornhorst, J.A.; Falke, J.J. Purification of proteins using polyhistidine affinity tags. Methods Enzym. 2010, 2000, 245–254. [Google Scholar] [CrossRef]
- Ho, K.L.; Kueh, C.L.; Beh, P.L.; Tan, W.S.; Bhella, D. Cryo-electron microscopy structure of the Macrobrachium rosenbergii nodavirus capsid at 7 angstroms resolution. Sci. Rep. 2017, 7, 1–8. [Google Scholar] [CrossRef]
- Xia, Y.; Protzer, U. Control of hepatitis B virus by cytokines. Viruses 2017, 9, 18. [Google Scholar] [CrossRef] [PubMed]
- Guidotti, L.G.; Ishikawa, T.; Hobbs, M.V.; Matzke, B.; Schreiber, R.; Chisari, F.V. Intracellular inactivation of the hepatitis B virus by inflammatory cytokines. Immunity 1996, 39, 25–36. [Google Scholar] [CrossRef]
- Lin, S.J.; Shu, P.Y.; Chang, C.; Ng, A.K.; Hu, C. IL-4 suppresses the expression and the replication of hepatitis B virus in the hepatocellular carcinoma cell line Hep3B. J. Immunol. 2003, 171, 4708–4716. [Google Scholar] [CrossRef] [PubMed]
- Cheng, X.; Xia, Y.; Serti, E.; Block, P.D.; Chung, M.; Chayama, K.; Rehermann, B.; Liang, T.J. Hepatitis B virus evades innate immunity of hepatocytes but activates macrophages during infection. Hepatology 2017, 66, 1779–1793. [Google Scholar] [CrossRef]
- Lang, P.A.; Recher, M.; Honke, N.; Scheu, S.; Borkens, S.; Gailus, N.; Krings, C.; Meryk, A.; Kulawik, A.; Cervantes-Barragan, L.; et al. Tissue macrophages suppress viral replication and prevent severe immunopathology in an interferon-I-dependent manner in mice. Hepatology 2010, 52, 25–32. [Google Scholar] [CrossRef]
- Shen, Z.Y.; Zheng, W.P.; Liu, T.; Yang, Y.; Song, H.L. Effects of dendritic cells from hepatitis B virus transgenic mice-stimulated autologous lymphocytes on hepatitis B virus replication: A study on the impact of specific sensitized effector cells on in vitro virus replication. Viral Immunol. 2015, 28, 85–92. [Google Scholar] [CrossRef]
- Escriou, V.; Ciolina, C.; Helbling-Leclerc, A.; Wils, P.; Scherman, D. Cationic lipid-mediated gene transfer: Analysis of cellular uptake and nuclear import of plasmid DNA. Cell Biol. Toxicol. 1998, 14, 95–104. [Google Scholar] [CrossRef] [PubMed]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Yang, J.T.; Wu, C.C.; Martinez, H.M. Calculation of protein conformation from circular dichroism. Methods Enzymol. 1986, 130, 208–269. [Google Scholar] [CrossRef] [PubMed]

| Temperature (°C) | α-Helix (%) | β-Sheet (%) | β-Turn (%) | Random Coil (%) | Total (%) |
|---|---|---|---|---|---|
| 20.0 | 17.9 | 43.6 | 5.7 | 32.8 | 100.0 |
| 30.0 | 20.6 | 48.8 | 1.9 | 28.7 | 100.0 |
| 40.0 | 26.1 | 40.8 | 2.4 | 30.7 | 100.0 |
| 50.0 | 33.5 | 5.9 | 8.0 | 52.6 | 100.0 |
| 60.0 | 52.5 | 0.0 | 2.8 | 44.6 | 100.0 |
| 70.0 | 25.2 | 0.3 | 28.0 | 46.4 | 100.0 |
| Primer | Primer Sequences | Ta (°C) | Function |
|---|---|---|---|
| Nc-aD-F Nc-aD-R | 5′-GGGGGCCATGGGGATGGCCCTTAACATCAAG-3′ 5′-CCCCCTCGAGTTAATGATGATGATGATGATGG-3′ | 72 | Amplification of Nc-aD gene from recombinant pTrcHis-TARNA2 plasmid |
| Nc-aD/pFastBac HT C-F Nc-aD/pFastBac HT C-R | 5′-CCATGGCGTACTACCATCAC-3′ 5′-GTAAAACCTCTACAAATGTGG-3′ | 59 | To detect the Nc-aD gene in the recombinant pFastBac HT C vector |
| pUC/M13-F pUC/M13-R | 5′-CCCAGTCACGACGTTGTAAAACG-3′ 5′-AGCGGATAACAATTTCACACAGG-3′ | 55 | To detect the Nc-aD gene in the recombinant bacmid DNA prior to transfection into Sf9 cells |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ninyio, N.N.; Ho, K.L.; Yong, C.Y.; Chee, H.Y.; Hamid, M.; Ong, H.K.; Mariatulqabtiah, A.R.; Tan, W.S. Chimeric Virus-Like Particles of Prawn Nodavirus Displaying Hepatitis B Virus Immunodominant Region: Biophysical Properties and Cytokine Response. Int. J. Mol. Sci. 2021, 22, 1922. https://doi.org/10.3390/ijms22041922
Ninyio NN, Ho KL, Yong CY, Chee HY, Hamid M, Ong HK, Mariatulqabtiah AR, Tan WS. Chimeric Virus-Like Particles of Prawn Nodavirus Displaying Hepatitis B Virus Immunodominant Region: Biophysical Properties and Cytokine Response. International Journal of Molecular Sciences. 2021; 22(4):1922. https://doi.org/10.3390/ijms22041922
Chicago/Turabian StyleNinyio, Nathaniel Nyakaat, Kok Lian Ho, Chean Yeah Yong, Hui Yee Chee, Muhajir Hamid, Hui Kian Ong, Abdul Razak Mariatulqabtiah, and Wen Siang Tan. 2021. "Chimeric Virus-Like Particles of Prawn Nodavirus Displaying Hepatitis B Virus Immunodominant Region: Biophysical Properties and Cytokine Response" International Journal of Molecular Sciences 22, no. 4: 1922. https://doi.org/10.3390/ijms22041922
APA StyleNinyio, N. N., Ho, K. L., Yong, C. Y., Chee, H. Y., Hamid, M., Ong, H. K., Mariatulqabtiah, A. R., & Tan, W. S. (2021). Chimeric Virus-Like Particles of Prawn Nodavirus Displaying Hepatitis B Virus Immunodominant Region: Biophysical Properties and Cytokine Response. International Journal of Molecular Sciences, 22(4), 1922. https://doi.org/10.3390/ijms22041922