A Two-in-One Strategy: Target and Nontarget Site Mechanisms Both Play Important Role in IMI-Resistant Weedy Rice
Abstract
1. Introduction
2. Results
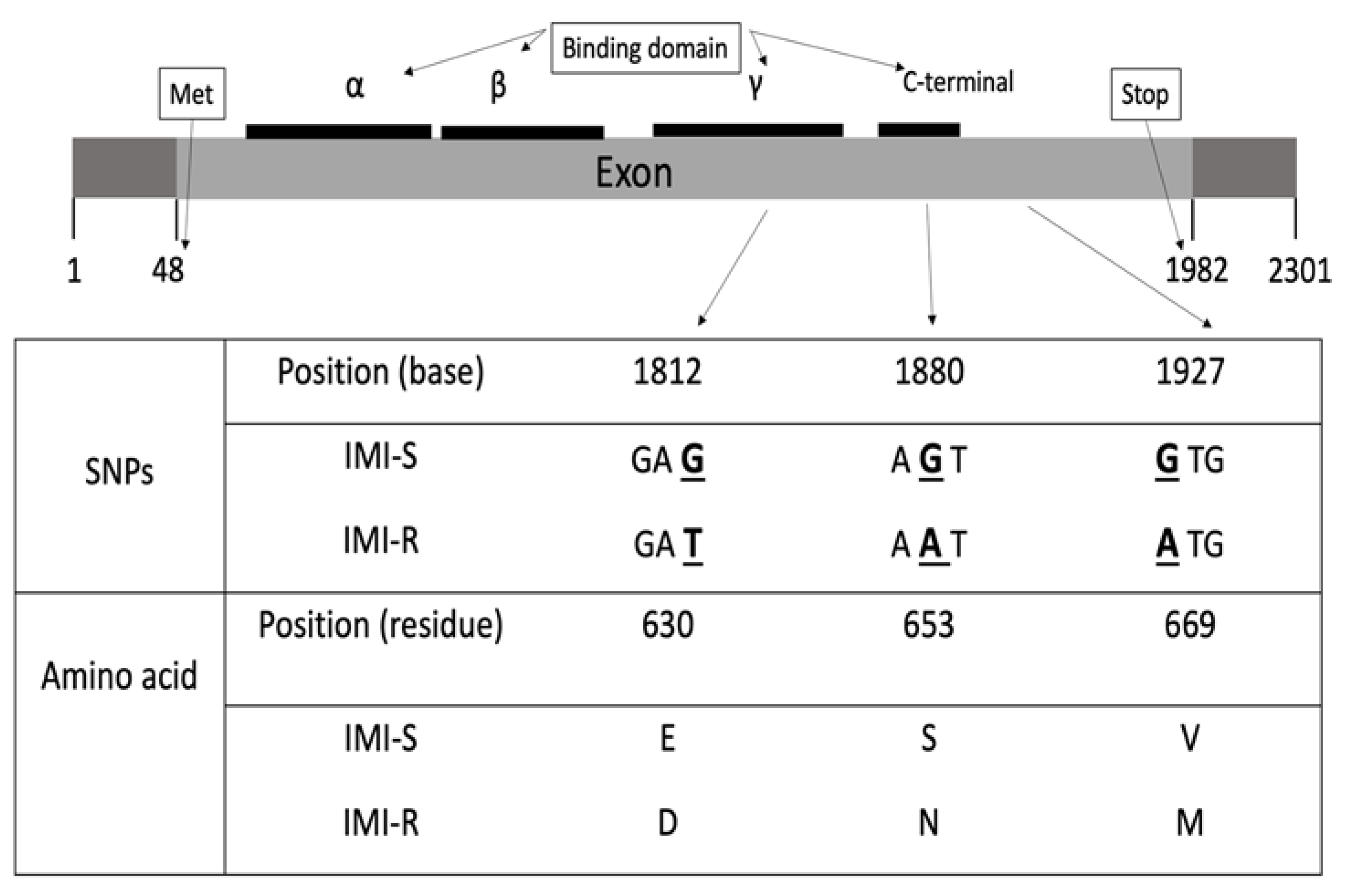
2.1. ALS-Coding Gene Sequence Analysis
2.2. Functional Mutation Analysis
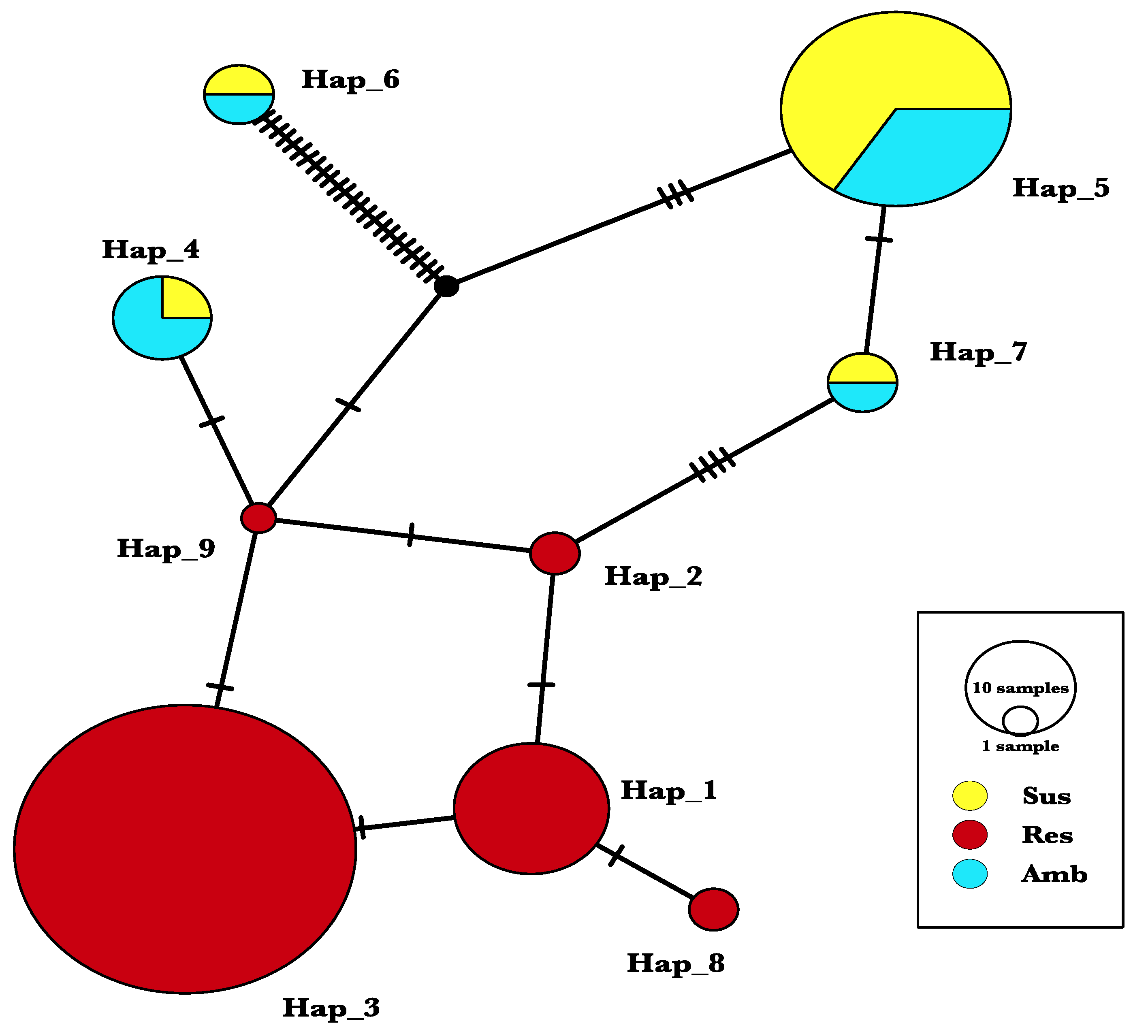
2.3. Haplotype Analysis
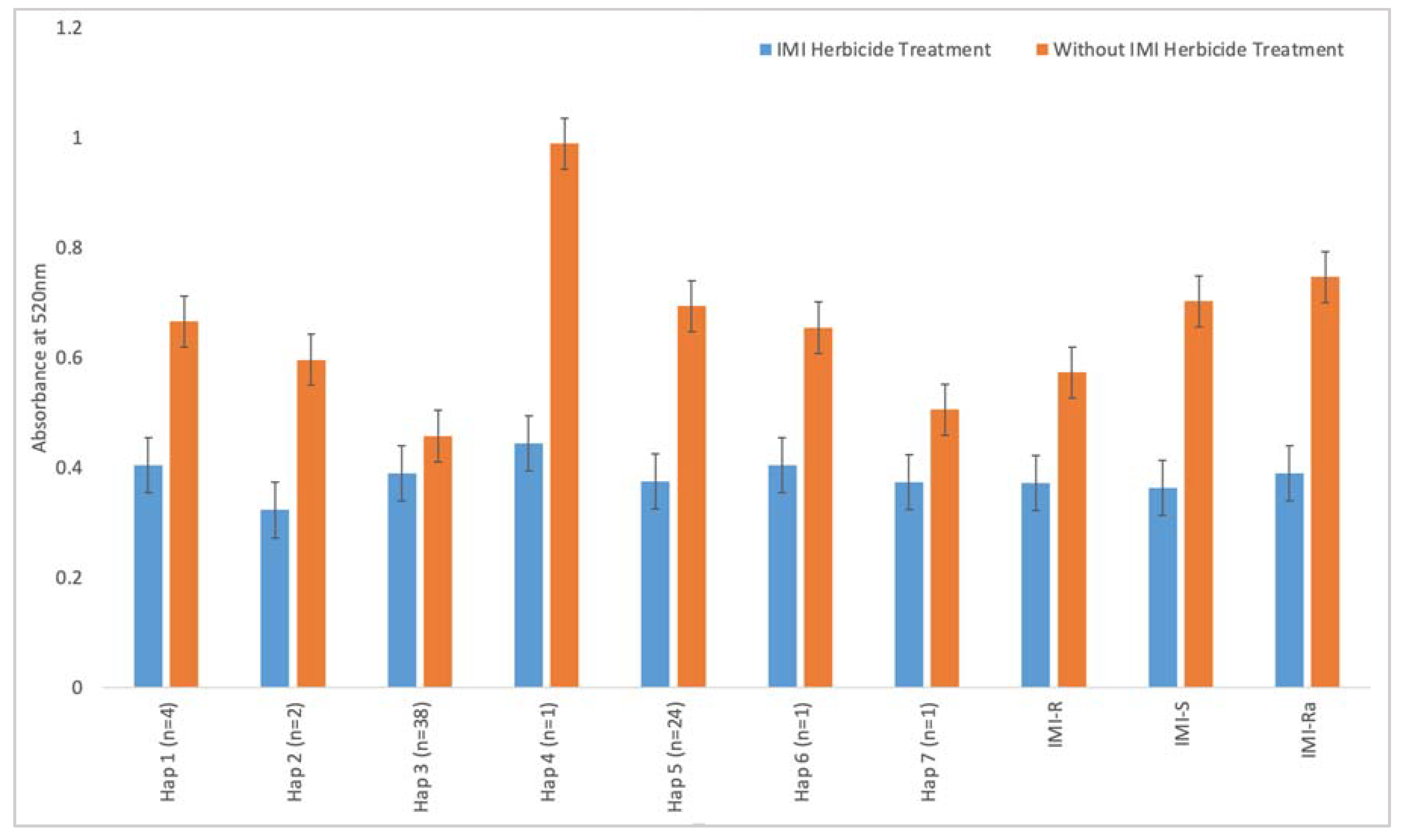
2.4. Enzyme Colorimetric Assay
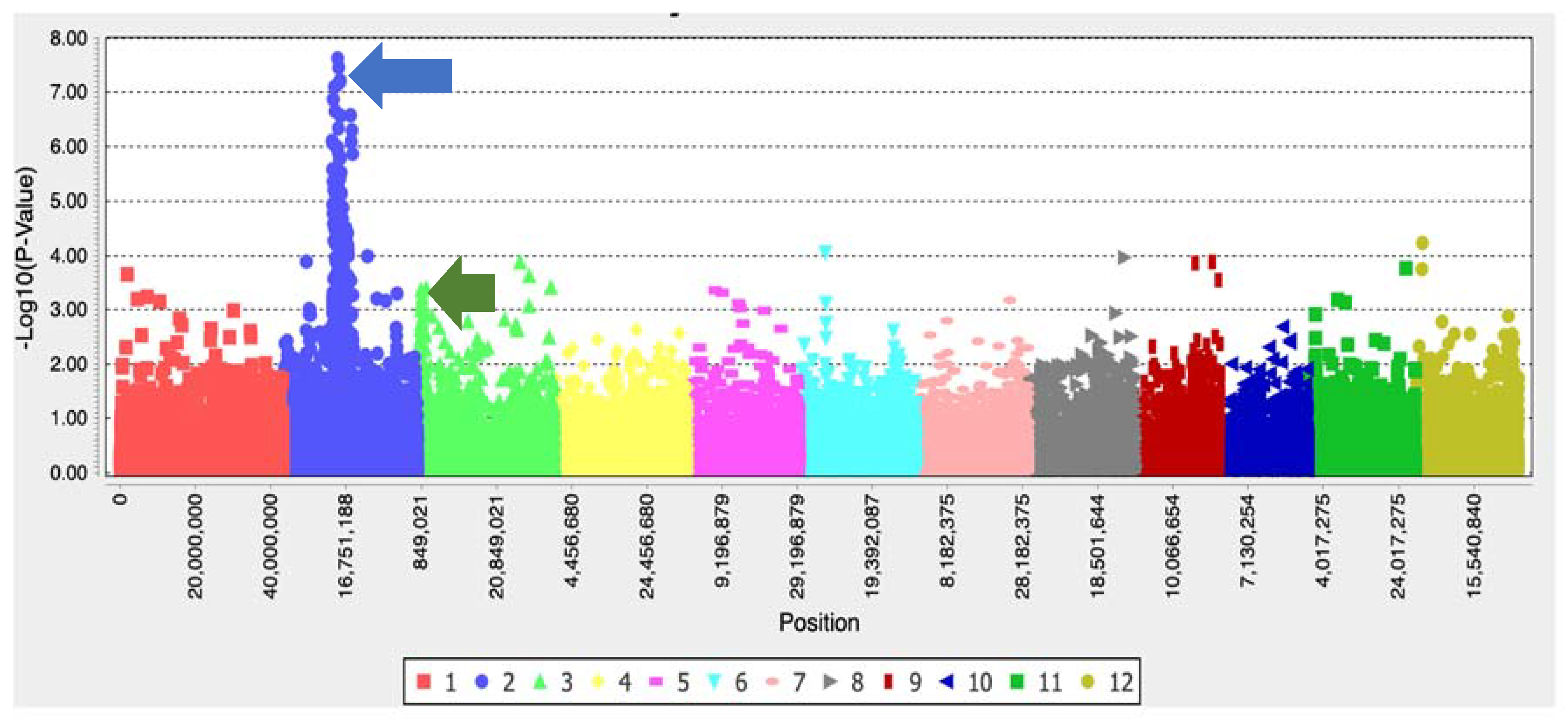
2.5. Genome-Wide Association Studies (GWAS)
3. Discussion
3.1. Functional Mutation of the ALS Gene and Gene Flow from Clearfield to Weedy Rice
3.2. ALS Enzyme Activity
3.3. Ambiguous Resistance of IMI-Ra
4. Materials and Methods
4.1. Plant Materials
4.2. Genomic DNA Samples
4.3. Gene Amplification and Sequence Analysis
4.4. Enzyme Extraction
4.5. Colorimetric Enzyme Activity Assay
4.6. Genotyping-by-Sequencing (GBS) and Genome-Wide Association Studies (GWAS)
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Délye, C. Unravelling the genetic bases of non-target-site-based resistance (NTSR) to herbicides: A major challenge for weed science in the forthcoming decade. Pest Manag. Sci. 2012, 69, 176–187. [Google Scholar] [CrossRef]
- Hatami, Z.M.; Gherekhloo, J.; Rojano-Delgado, A.M.; Osuna, M.D.; Alcántara, R.; Fernández, P.; Sadeghipour, H.R.; De Prado, R. Multiple Mechanisms Increase Levels of Resistance in Rapistrum rugosum to ALS Herbicides. Front. Plant Sci. 2016, 7, 169. [Google Scholar] [CrossRef]
- The Imidazolinone Herbicides (1991); Informa UK Limited: London, UK, 2017.
- Croughan, T. Clearfield Rice: It’s Not A GMO. LSU AgCenter. 2004. Available online: https://www.lsuagcenter.com/portals/communications/publications/agmag/archive/2003/fall/clearfield-rice-its-not-a-gmo (accessed on 28 October 2020).
- Qiu, J.; Jia, L.; Wu, D.; Weng, X.; Chen, L.; Sun, J.; Chen, M.; Mao, L.; Jiang, B.; Ye, C.; et al. Diverse genetic mechanisms underlie worldwide convergent rice feralization. Genome Biol. 2020, 21, 1–11. [Google Scholar] [CrossRef]
- Yang, Q.; Deng, W.; Li, X.; Yu, Q.; Bai, L.; Zheng, M.Q. Target-site and non-target-site based resistance to the herbicide tribenuron-methyl in flixweed (Descurainia sophia L.). BMC Genom. 2016, 17, 1–13. [Google Scholar] [CrossRef]
- Rajguru, S.N.; Roma-Burgos, N.; Shivrain, V.K.; Stewart, J.M. Mutations in the red rice ALS gene associated with resistance to imazethapyr. Weed Sci. 2005, 53, 567–577. [Google Scholar] [CrossRef]
- Shivrain, V.K.; Roma-Burgos, N.; Sales, M.A.; Kuk, Y.I. Polymorphisms in the ALS gene of weedy rice (Oryza sativa L.) accessions with differential tolerance to imazethapyr. Crop. Prot. 2010, 29, 336–341. [Google Scholar] [CrossRef]
- Singh, V.; Singh, S.; Black, H.; Boyett, V.; Basu, S.; Gealy, D.; Gbur, E.; Pereira, A.; Scott, R.C.; Caicedo, A.; et al. Introgression of Clearfield™ rice crop traits into weedy red rice outcrosses. Field Crop. Res. 2017, 207, 13–23. [Google Scholar] [CrossRef]
- Roso, A.C.; Jr, A.M.; Delatorre, C.A.; Menezes, V.G. Regional scale distribution of imidazolinone herbicide-resistant alleles in red rice (Oryza sativa L.) determined through SNP markers. Field Crop. Res. 2010, 119, 175–182. [Google Scholar] [CrossRef]
- Ruzmi, R.; Ahmad-Hamdani, M.S.; Bakar, B.B. Prevalence of herbicide-resistant weed species in Malaysian rice fields: A review. Weed Biol. Manag. 2017, 17, 3–16. [Google Scholar] [CrossRef]
- Chua, E. Farmers Go Against the Grain. The Star 2018. Available online: https://www.thestar.com.my/news/nation/2018/01/16/farmers-go-against-the-grain-weed-interbreeding-sees-clearfields-future-grow-cloudy (accessed on 2 March 2020).
- Engku, A.K.; Norida, M.; Juraimi, A.S.; Rafii, M.Y.; Abdullah, S.N.A.; Alam, M.A. Gene flow from Clearfield®;rice to weedy rice under field conditions. Plant Soil Environ. 2016, 62, 16–22. [Google Scholar] [CrossRef]
- Singh, S.; Singh, V.; Salas-Perez, R.A.; Bagavathiannan, M.V.; Lawton-Rauh, A.; Roma-Burgos, N. Target-site mutation accumulation among ALS inhibitor-resistant Palmer amaranth. Pest Manag. Sci. 2019, 75, 1131–1139. [Google Scholar] [CrossRef]
- Deng, W.; Yang, Q.; Zhang, Y.; Jiao, H.; Mei, Y.; Li, X.; Zheng, M.Q. Cross-resistance patterns to acetolactate synthase (ALS)-inhibiting herbicides of flixweed (Descurainia sophia L.) conferred by different combinations of ALS isozymes with a Pro-197-Thr mutation or a novel Trp-574-Leu mutation. Pestic. Biochem. Physiol. 2017, 136, 41–45. [Google Scholar] [CrossRef]
- Han, B.; Xue, Y. Genome-wide intraspecific DNA-sequence variations in rice. Curr. Opin. Plant Biol. 2003, 6, 134–138. [Google Scholar] [CrossRef]
- Powles, S.B.; Yu, Q. Evolution in Action: Plants Resistant to Herbicides. Annu. Rev. Plant Biol. 2010, 61, 317–347. [Google Scholar] [CrossRef]
- McCourt, J.A.; Pang, S.S.; King-Scott, J.; Guddat, L.W.; Duggleby, R.G. Herbicide-binding sites revealed in the structure of plant acetohydroxyacid synthase. Proc. Natl. Acad. Sci. USA 2006, 103, 569–573. [Google Scholar] [CrossRef]
- Garcia, M.D.; Nouwens, A.; Lonhienne, T.G.; Guddat, L.W. Comprehensive understanding of acetohydroxyacid synthase inhibition by different herbicide families. Proc. Natl. Acad. Sci. USA 2017, 114, E1091–E1100. [Google Scholar] [CrossRef]
- Wright, A.A.; Rodriguez-Carres, M.; Sasidharan, R.; Koski, L.; Peterson, D.G.; Nandula, V.K.; Ray, J.D.; Bond, J.A.; Shaw, D.R. Multiple Herbicide–Resistant Junglerice (Echinochloa colona): Identification of Genes Potentially Involved in Resistance through Differential Gene Expression Analysis. Weed Sci. 2018, 66, 347–354. [Google Scholar] [CrossRef]
- Riar, D.S.; Norsworthy, J.K.; Srivastava, V.; Nandula, V.; Bond, J.A.; Scott, R.C. Physiological and Molecular Basis of Acetolactate Synthase-Inhibiting Herbicide Resistance in Barnyardgrass (Echinochloa crus-galli). J. Agric. Food Chem. 2013, 61, 278–289. [Google Scholar] [CrossRef]
- Liu, X.; Huang, M.; Fan, B.; Buckler, E.S.; Zhang, Z. Iterative Usage of Fixed and Random Effect Models for Powerful and Efficient Genome-Wide Association Studies. PLoS Genet. 2016, 12, e1005767. [Google Scholar] [CrossRef]
- Shaner, D.L. Role of Translocation as A Mechanism of Resistance to Glyphosate. Weed Sci. 2009, 57, 118–123. [Google Scholar] [CrossRef]
- Délye, C.; Pernin, F.; Scarabel, L. Evolution and diversity of the mechanisms endowing resistance to herbicides inhibiting acetolactate-synthase (ALS) in corn poppy (Papaver rhoeas L.). Plant Sci. 2011, 180, 333–342. [Google Scholar] [CrossRef]
- Singh, B.K.; Stidham, M.A.; Shaner, D.L. Assay of acetohydroxyacid synthase. Anal. Biochem. 1988, 171, 173–179. [Google Scholar] [CrossRef]
- Vigueira, C.C.; Qi, X.; Song, B.-K.; Li, L.; Caicedo, A.L.; Jia, Y.; Olsen, K.M. Call of the wild rice: Oryza rufipogon shapes weedy rice evolution in Southeast Asia. Evol. Appl. 2018, 12, 93–104. [Google Scholar] [CrossRef] [PubMed]
- Bradbury, P.J.; Zhang, Z.; Kroon, D.E.; Casstevens, T.M.; Ramdoss, Y.; Buckler, E.S. TASSEL: Software for association mapping of complex traits in diverse samples. Bioinformatics 2007, 23, 2633–2635. [Google Scholar] [CrossRef]
- Li, H.; Durbin, R. Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics 2009, 25, 1754–1760. [Google Scholar] [CrossRef]
| Nucleotide Sequences at Positions 1879–1881 | Rice Accession |
|---|---|
| AGT | IMI-S weedy rice (n = 18); IMI-Ra weedy rice (n = 12): RDA6, RDA4, RKI2, RSK6, RSK9, RTA, RDA4a, RDA5a, RPA4, RTAa, RDE2a, RPC3 |
| AAT | Clearfield rice CL2; IMI-R weedy rice (n = 46) |
| Nucleotide Position | Nucleotide Change | Domain | Altered Amino Acid | Amino Acid Group Change |
|---|---|---|---|---|
| 1239 | GCG-GCA | - | Neutral | - |
| 1754 | AGT-AGC | - | Neutral | - |
| 1812 | GAT-GAG | Gamma | Asp630Glu | - |
| 1880 | AGT-AAT | C-terminal | Ser653Asn | - |
| 1927 | GTG-ATG | - | Val669Met | - |
| Spearman Correlation Test | |||
|---|---|---|---|
| Phenotype | Genotype | ||
| Correlation Coefficient | Phenotype | 1.000 | 0.062 * |
| Genotype | 0.062 * | 1.000 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yean, R.-A.; Dilipkumar, M.; Rahman, S.; Song, B.-K. A Two-in-One Strategy: Target and Nontarget Site Mechanisms Both Play Important Role in IMI-Resistant Weedy Rice. Int. J. Mol. Sci. 2021, 22, 982. https://doi.org/10.3390/ijms22030982
Yean R-A, Dilipkumar M, Rahman S, Song B-K. A Two-in-One Strategy: Target and Nontarget Site Mechanisms Both Play Important Role in IMI-Resistant Weedy Rice. International Journal of Molecular Sciences. 2021; 22(3):982. https://doi.org/10.3390/ijms22030982
Chicago/Turabian StyleYean, Ru-Ann, Masilamany Dilipkumar, Sadequr Rahman, and Beng-Kah Song. 2021. "A Two-in-One Strategy: Target and Nontarget Site Mechanisms Both Play Important Role in IMI-Resistant Weedy Rice" International Journal of Molecular Sciences 22, no. 3: 982. https://doi.org/10.3390/ijms22030982
APA StyleYean, R.-A., Dilipkumar, M., Rahman, S., & Song, B.-K. (2021). A Two-in-One Strategy: Target and Nontarget Site Mechanisms Both Play Important Role in IMI-Resistant Weedy Rice. International Journal of Molecular Sciences, 22(3), 982. https://doi.org/10.3390/ijms22030982





