An Adult Mouse Model of Dilated Cardiomyopathy Caused by Inducible Cardiac-Specific Bis Deletion
Abstract
1. Introduction
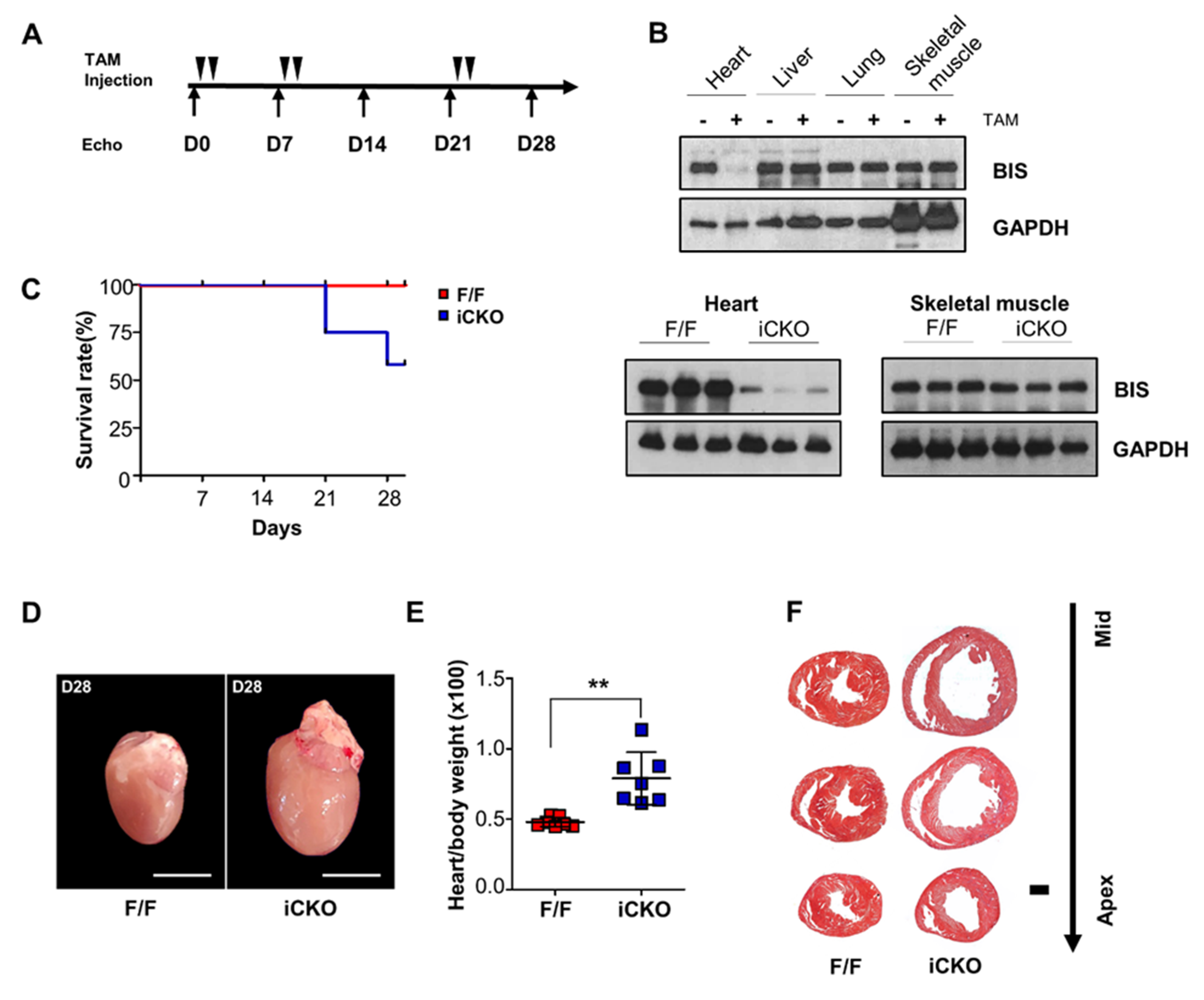
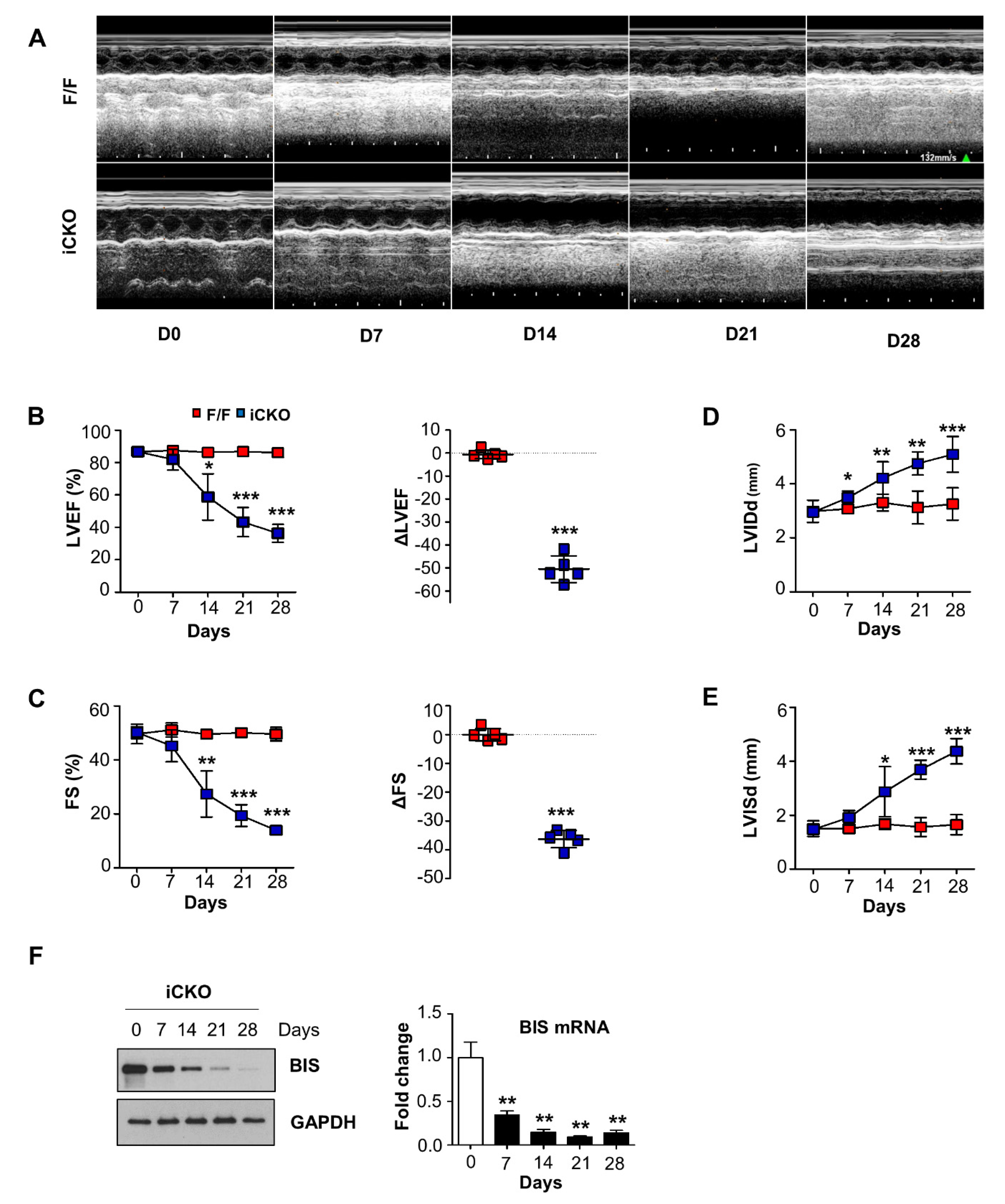
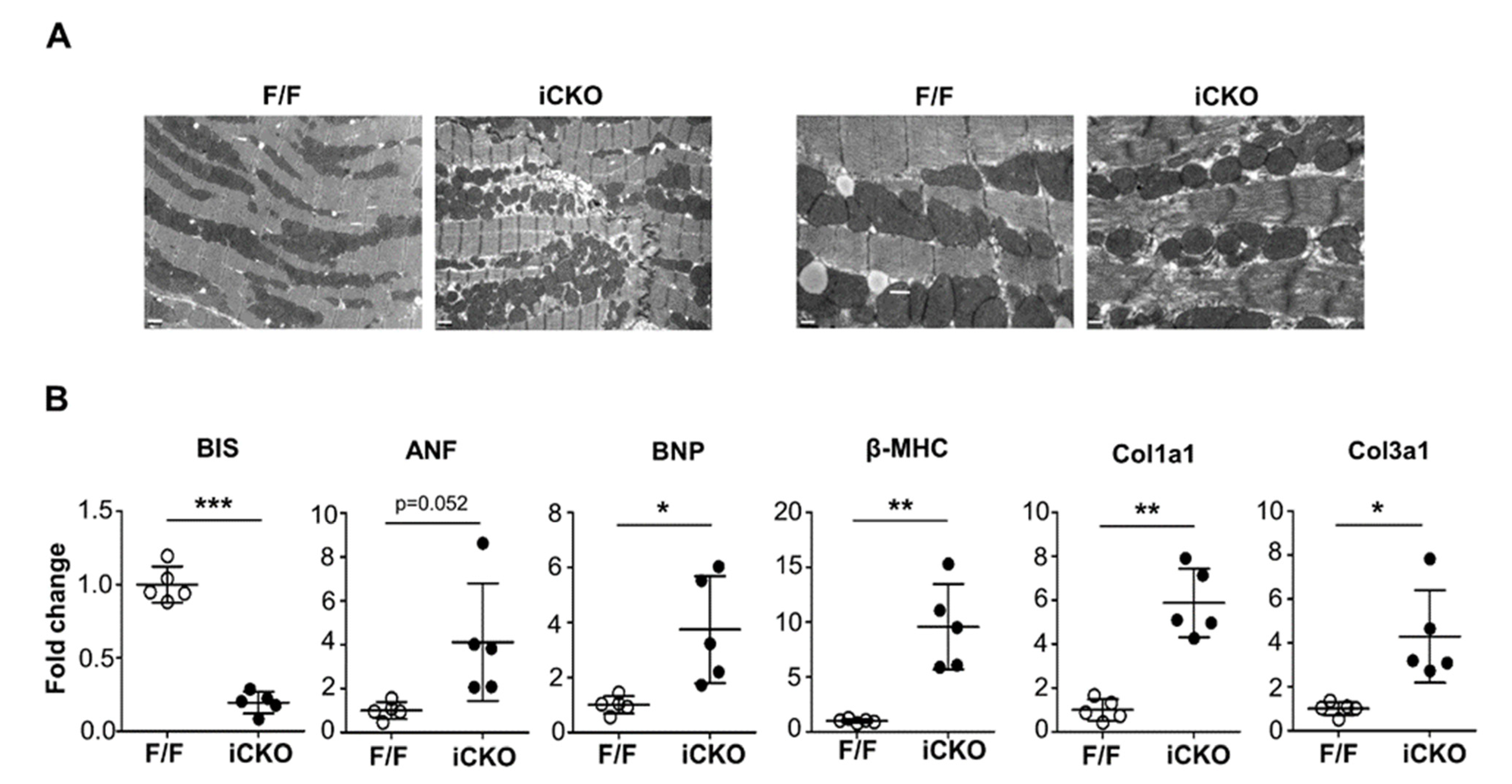
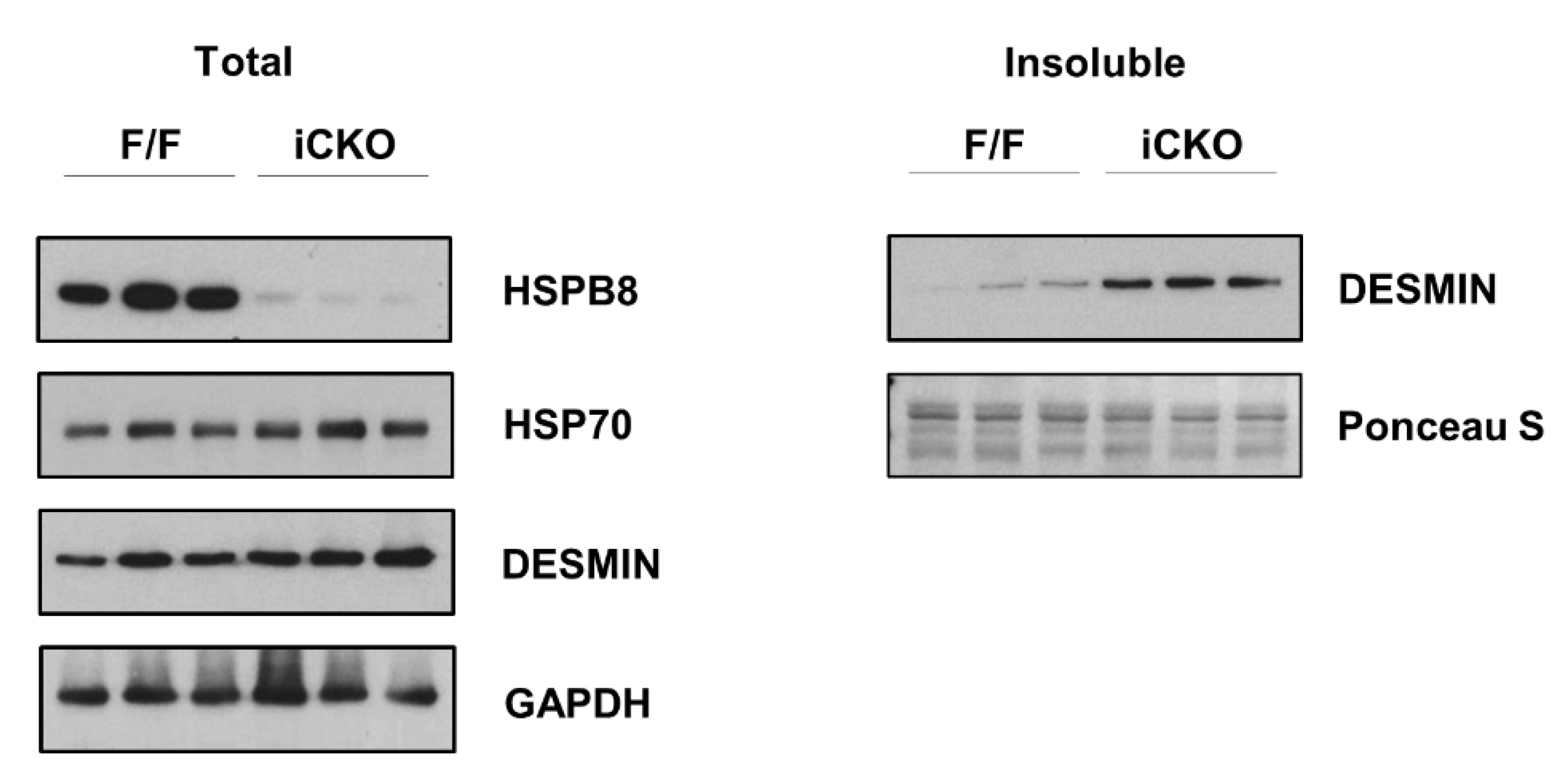
2. Results
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Echocardiography
4.3. Histology and Electron Microscopy
4.4. Western Blotting Assay
4.5. Quantitative Real-Time PCR (qRT–PCR)
4.6. Statistics
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lee, J.H.; Takahashi, T.; Yasuhara, N.; Inazawa, J.; Kamada, S.; Tsujimoto, Y. Bis, a Bcl-2-binding protein that synergizes with Bcl-2 in preventing cell death. Oncogene 1999, 18, 6183–6190. [Google Scholar] [CrossRef] [PubMed]
- De Marco, M.; Basile, A.; Iorio, V.; Festa, M.; Falco, A.; Ranieri, B.; Pascale, M.; Sala, G.; Remondelli, P.; Capunzo, M.; et al. Role of BAG3 in cancer progression: A therapeutic opportunity. Semin. Cell Dev. Biol. 2018, 78, 85–92. [Google Scholar] [CrossRef] [PubMed]
- Behl, C. Breaking BAG: The Co-Chaperone BAG3 in Health and Disease. Trends Pharmacol. Sci. 2016, 37, 672–688. [Google Scholar] [CrossRef] [PubMed]
- Sturner, E.; Behl, C. The Role of the Multifunctional BAG3 Protein in Cellular Protein Quality Control and in Disease. Front. Mol. Neurosci. 2017, 10. [Google Scholar] [CrossRef] [PubMed]
- Dominguez, F.; Cuenca, S.; Bilinska, Z.; Toro, R.; Villard, E.; Barriales-Villa, R.; Ochoa, J.P.; Asselbergs, F.; Sammani, A.; Franaszczyk, M.; et al. Dilated Cardiomyopathy Due to BLC2-Associated Athanogene 3 (BAG3) Mutations. J. Am. Coll. Cardiol. 2018, 72, 2471–2481. [Google Scholar] [CrossRef] [PubMed]
- Gandhi, P.U.; Gaggin, H.K.; Belcher, A.M.; Harisiades, J.E.; Basile, A.; Falco, A.; Rosati, A.; Piscione, F.; Januzzi, J.L., Jr.; Turco, M.C. Analysis of BAG3 plasma concentrations in patients with acutely decompensated heart failure. Clin. Chim. Acta 2015, 445, 73–78. [Google Scholar] [CrossRef] [PubMed]
- De Marco, M.; D’Auria, R.; Rosati, A.; Vitulano, G.; Gigantino, A.; Citro, R.; Piscione, F.; Zilinski, J.; Januzzi, J.L., Jr.; Turco, M.C. BAG3 protein in advanced-stage heart failure. JACC Heart Fail 2014, 2, 673–675. [Google Scholar] [CrossRef] [PubMed]
- Youn, D.Y.; Lee, D.H.; Lim, M.H.; Yoon, J.S.; Lim, J.H.; Jung, S.E.; Yeum, C.E.; Park, C.W.; Youn, H.J.; Lee, J.S.; et al. Bis deficiency results in early lethality with metabolic deterioration and involution of spleen and thymus. Am. J. Physiol. Endocrinol. Metab. 2008, 295, E1349–E1357. [Google Scholar] [CrossRef][Green Version]
- Fang, X.; Bogomolovas, J.; Wu, T.B.; Zhang, W.; Liu, C.Z.; Veevers, J.; Stroud, M.J.; Zhang, Z.Y.; Ma, X.L.; Mu, Y.X.; et al. Loss-of-function mutations in co-chaperone BAG3 destabilize small HSPs and cause cardiomyopathy. J. Clin. Investig. 2017, 127, 3189–3200. [Google Scholar] [CrossRef] [PubMed]
- Myers, V.D.; Tomar, D.; Madesh, M.; Wang, J.; Song, J.; Zhang, X.Q.; Gupta, M.K.; Tahrir, F.G.; Gordon, J.; McClung, J.M.; et al. Haplo-insufficiency of Bcl2-associated athanogene 3 in mice results in progressive left ventricular dysfunction, beta-adrenergic insensitivity, and increased apoptosis. J. Cell Physiol. 2018, 233, 6319–6326. [Google Scholar] [CrossRef] [PubMed]
- Sohal, D.S.; Nghiem, M.; Crackower, M.A.; Witt, S.A.; Kimball, T.R.; Tymitz, K.M.; Penninger, J.M.; Molkentin, J.D. Temporally regulated and tissue-specific gene manipulations in the adult and embryonic heart using a tamoxifen-inducible Cre protein. Circ. Res. 2001, 89, 20–25. [Google Scholar] [CrossRef] [PubMed]
- Tsikitis, M.; Galata, Z.; Mavroidis, M.; Psarras, S.; Capetanaki, Y. Intermediate filaments in cardiomyopathy. Biophys. Rev. 2018, 10, 1007–1031. [Google Scholar] [CrossRef] [PubMed]
- Arndt, V.; Dick, N.; Tawo, R.; Dreiseidler, M.; Wenzel, D.; Hesse, M.; Furst, D.O.; Saftig, P.; Saint, R.; Fleischmann, B.K.; et al. Chaperone-Assisted Selective Autophagy Is Essential for Muscle Maintenance. Curr. Biol. 2010, 20, 143–148. [Google Scholar] [CrossRef] [PubMed]
- Koitabashi, N.; Bedja, D.; Zaiman, A.L.; Pinto, Y.M.; Zhang, M.L.; Gabrielson, K.L.; Takimoto, E.; Kass, D.A. Avoidance of Transient Cardiomyopathy in Cardiomyocyte-Targeted Tamoxifen-Induced MerCreMer Gene Deletion Models. Circ. Res. 2009, 105, 12–15. [Google Scholar] [CrossRef] [PubMed]
- Halliday, B.P.; Gulati, A.; Ali, A.; Newsome, S.; Lota, A.; Tayal, U.; Vassiliou, V.S.; Arzanauskaite, M.; Izgi, C.; Krishnathasan, K.; et al. Sex- and age-based differences in the natural history and outcome of dilated cardiomyopathy. Eur. J. Heart Fail. 2018, 20, 1392–1400. [Google Scholar] [CrossRef]
- Hong, J.; Park, J.S.; Lee, H.; Jeong, J.; Yun, H.H.; Kim, H.Y.; Ko, Y.G.; Lee, J.H. Myosin heavy chain is stabilized by BCL-2 interacting cell death suppressor (BIS) in skeletal muscle. Exp. Mol. Med. 2016, 48, 1–9. [Google Scholar] [CrossRef]
- Lee, S.; Lee, D.H.; Park, B.W.; Kim, R.; Hoang, A.D.; Woo, S.K.; Xiong, W.; Lee, Y.J.; Ban, K.; Park, H.J. In vivo transduction of ETV2 improves cardiac function and induces vascular regeneration following myocardial infarction. Exp. Mol. Med. 2019, 51, 1–14. [Google Scholar] [PubMed]
- Yano, M.; Nakamuta, S.; Wu, X.J.; Okumura, Y.; Kido, H. A novel function of 14-3-3 protein: 14-3-3 zeta is a heat-shock-related molecular chaperone that dissolves thermal-aggregated proteins. Mol. Biol. Cell 2006, 17, 4769–4779. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yun, H.H.; Jung, S.Y.; Park, B.W.; Ko, J.S.; Yoo, K.; Yeo, J.; Kim, H.L.; Park, H.J.; Youn, H.J.; Lee, J.H. An Adult Mouse Model of Dilated Cardiomyopathy Caused by Inducible Cardiac-Specific Bis Deletion. Int. J. Mol. Sci. 2021, 22, 1343. https://doi.org/10.3390/ijms22031343
Yun HH, Jung SY, Park BW, Ko JS, Yoo K, Yeo J, Kim HL, Park HJ, Youn HJ, Lee JH. An Adult Mouse Model of Dilated Cardiomyopathy Caused by Inducible Cardiac-Specific Bis Deletion. International Journal of Molecular Sciences. 2021; 22(3):1343. https://doi.org/10.3390/ijms22031343
Chicago/Turabian StyleYun, Hye Hyeon, Soon Young Jung, Bong Woo Park, Ji Seung Ko, Kyunghyun Yoo, Jiyoung Yeo, Hong Lim Kim, Hun Jun Park, Ho Joong Youn, and Jeong Hwa Lee. 2021. "An Adult Mouse Model of Dilated Cardiomyopathy Caused by Inducible Cardiac-Specific Bis Deletion" International Journal of Molecular Sciences 22, no. 3: 1343. https://doi.org/10.3390/ijms22031343
APA StyleYun, H. H., Jung, S. Y., Park, B. W., Ko, J. S., Yoo, K., Yeo, J., Kim, H. L., Park, H. J., Youn, H. J., & Lee, J. H. (2021). An Adult Mouse Model of Dilated Cardiomyopathy Caused by Inducible Cardiac-Specific Bis Deletion. International Journal of Molecular Sciences, 22(3), 1343. https://doi.org/10.3390/ijms22031343




