Association of Sleep Architecture and Physiology with Depressive Disorder and Antidepressants Treatment
Abstract
1. Introduction
2. The Physiology of Sleep
3. The Regulation of Sleep
4. Polysomnography—A Non-Invasive Tool for Sleep Evaluation
5. Sleep Disturbances Associated with Depressive Disorder
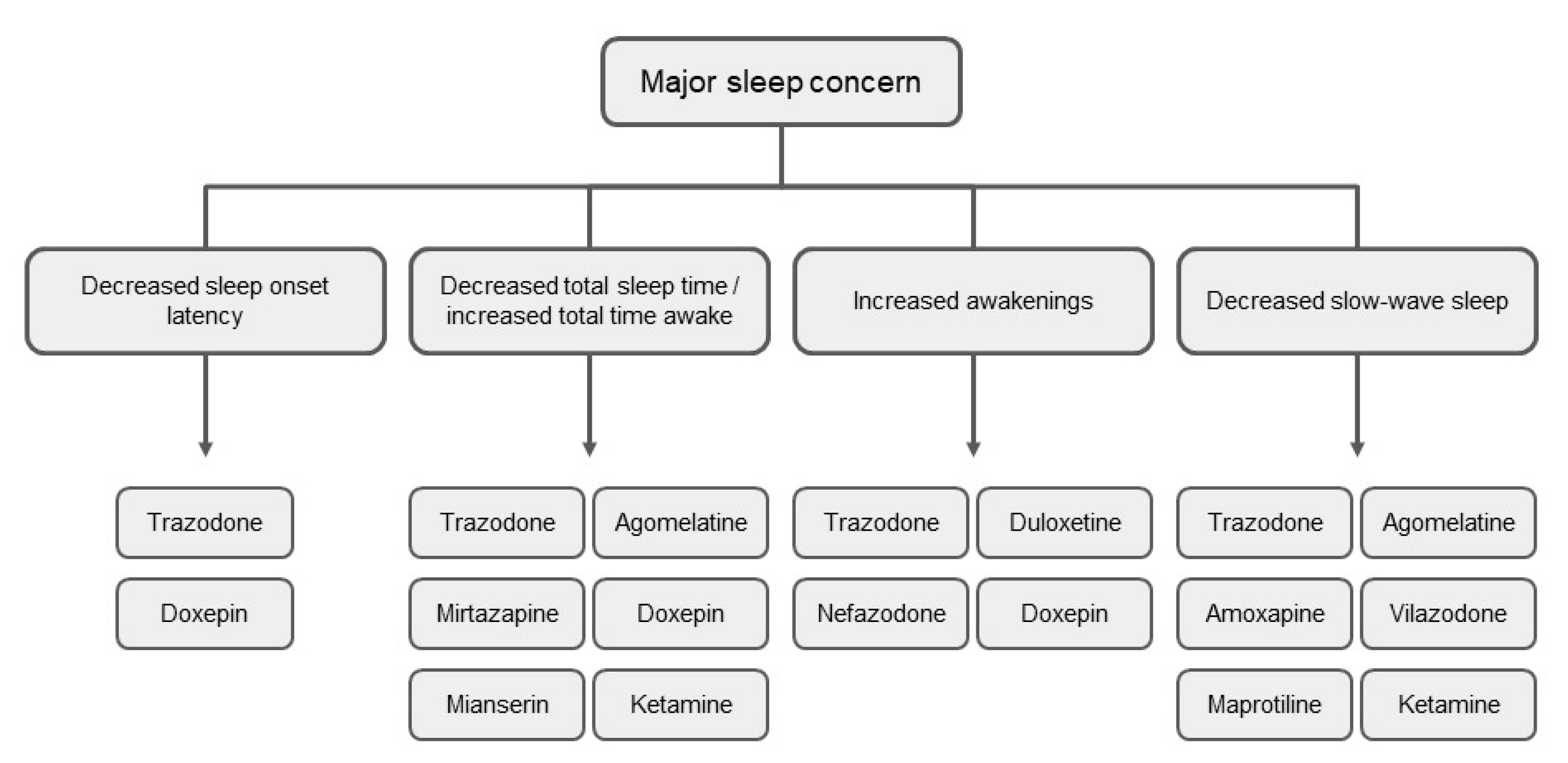
6. The Effects of Antidepressants on Sleep
6.1. Tricyclic Antidepressants (TCA)
6.2. Tetracyclic Antidepressants (TeCA)
6.3. Inhibitors of Monoaminoxidase (MAOI)
6.4. Selective Serotonin Reuptake Inhibitors (SSRI) and Serotonin Norepinephrine Reuptake Inhibitors (SNRI)
6.5. Dopamine Noradrenaline Reuptake Inhibitors (DNRI)
6.6. Serotonin Antagonist and Reuptake Inhibitors (SARI)
6.7. Dopamine Reuptake Inhibitor (DARI)
6.8. Noradrenaline Reuptake Inhibitors (NARI)
6.9. Novel Antidepressants
7. Depression and Sleep in Children and Adolescents
8. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- James, S.L.; Abate, D.; Abate, K.H.; Abay, S.M.; Abbafati, C.; Abbasi, N.; Abbastabar, H.; Abd-Allah, F.; Abdela, J.; Abdelalim, A.; et al. Global, regional, and national incidence, prevalence, and years lived with disability for 354 Diseases and Injuries for 195 countries and territories, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2018, 392, 1789–1858. [Google Scholar] [CrossRef]
- Hidaka, B.H. Depression as a disease of modernity: Explanations for increasing prevalence. J. Affect. Disord. 2012, 140, 205–214. [Google Scholar] [CrossRef] [PubMed]
- Ménard, C.; Hodes, G.E.; Russo, S.J. Pathogenesis of depression: Insights from human and rodent studies. Neuroscience 2016, 321, 138–162. [Google Scholar] [CrossRef]
- Paykel, E.S. Treatment of depression. The relevance of research for clinical practice. Br. J. Psychiatry 1989, 155, 754–763. [Google Scholar] [CrossRef] [PubMed]
- Greden, J.F. Unmet need: What justifies the search for a new antidepressant? J. Clin. Psychiatry 2002, 63, 3–7. [Google Scholar] [PubMed]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; American Psychiatric Association: Washington, DC, USA, 2013; ISBN 0-89042-555-8. [Google Scholar]
- WHO. International Classification of Diseases, 11th ed.; WHO: Geneva, Switzerland, 2019. [Google Scholar]
- Punnoose, A.R.; Golub, R.M.; Burke, A.E. Insomnia. JAMA J. Am. Med. Assoc. 2012, 307, 2653. [Google Scholar] [CrossRef]
- Ohayon, M.M.; Shapiro, C.M.; Ohayon, M.M.; Shapiro, C.M. Sleep disturbances and psychiatric disorders associated with posttraumatic stress disorder in the general population. Compr. Psychiatry 2000, 41, 469–478. [Google Scholar] [CrossRef]
- Wichniak, A.; Wierzbicka, A.; Walęcka, M.; Jernajczyk, W. Effects of Antidepressants on Sleep. Curr. Psychiatry Rep. 2017, 19, 63. [Google Scholar] [CrossRef]
- Abad, V.C.; Guilleminault, C. Sleep and psychiatry. Dialogues Clin. Neurosci. 2005, 7, 291–303. [Google Scholar]
- Kaplan, K.A.; Harvey, A.G. Hypersomnia across mood disorders: A review and synthesis. Sleep Med. Rev. 2009, 13, 275–285. [Google Scholar] [CrossRef]
- Freeman, D.; Sheaves, B.; Waite, F.; Harvey, A.G.; Harrison, P.J. Sleep disturbance and psychiatric disorders. Lancet Psychiatry 2020, 7, 628–637. [Google Scholar] [CrossRef]
- Van Moffaert, M.M.M.P. Sleep disorders and depression: The “chicken and egg” situation. J. Psychosom. Res. 1994, 38, 9–13. [Google Scholar] [CrossRef]
- Ostatnikova, D. Laboratory Guide to Medical Physiology; Comenius University: Bratislava, Slovakia, 2018; ISBN 978-80-223-4499-9. [Google Scholar]
- WHO Technical Meeting on Sleep and Health. Available online: https://www.euro.who.int/__data/assets/pdf_file/0008/114101/E84683.pdf (accessed on 7 January 2021).
- Carskadon, M.A.; Dement, W.C. Normal Human Sleep: An Overview. In Principles and Practice of Sleep Medicine; Kryger, M.H., Roth, T., Dement, W.C., Eds.; Elsevier Saunders: St. Louis, MI, USA, 2011; pp. 16–26. [Google Scholar]
- Guyton, A.C.; Hall, J.E. TEXTBOOK of Medical Physiology, 11th ed.; Elsevier Saunders: Philadelphia, PA, USA, 2006; ISBN 0-7216-0240-1. [Google Scholar]
- Maurovich-Horvat, E.; Pollmächer, T.Z.; Sonka, K. The Effects of Sleep and Sleep Deprivation on Metabolic, Endocrine and Immune Parameters. Prague Med. Rep. 2008, 109, 275–285. [Google Scholar] [PubMed]
- Holshoe, J.M. Antidepressants and sleep: A review. Perspect. Psychiatr. Care 2009, 45, 191–197. [Google Scholar] [CrossRef]
- Leshner, A.I. National Institutes of Health State of the Science Conference statement on manifestations and management of chronic insomnia in adults, June 13–15, 2005. Sleep 2005, 28, 1049–1057. [Google Scholar]
- Sun, Y.; Shi, L.; Bao, Y.; Sun, Y.; Shi, J.; Lu, L. The bidirectional relationship between sleep duration and depression in community-dwelling middle-aged and elderly individuals: Evidence from a longitudinal study. Sleep Med. 2018, 52, 221–229. [Google Scholar] [CrossRef]
- Mallampalli, M.P.; Carter, C.L. Exploring sex and gender differences in sleep health: A society for women’s health research report. J. Womens Health 2014, 23, 553–562. [Google Scholar] [CrossRef]
- Iber, C. The AASM Manual for the Scoring of Sleep and Associated Events: Rules, Terminology and Technical Specifications; American Academy of Sleep Medicine: Westchester, IL, USA, 2007. [Google Scholar]
- Berry, R.B.; Brooks, R.; Gamaldo, C.; Harding, S.M.; Lloyd, R.M.; Quan, S.F.; Troester, M.T.; Vaughn, B.V. AASM scoring manual updates for 2017 (version 2.4). J. Clin. Sleep Med. 2017, 13, 665–666. [Google Scholar] [CrossRef]
- Harris, C.D. Neurophysiology of sleep and wakefulness. Respir. Care Clin. N. Am. 2005, 11, 567–586. [Google Scholar]
- Weber, F.; Dan, Y. Circuit-based interrogation of sleep control. Nature 2016, 538, 51–59. [Google Scholar] [CrossRef]
- Schmidt, C.; Peigneux, P.; Cajochen, C. Age-related changes in sleep and circadian rhythms: Impact on cognitive performance and underlying neuroanatomical networks. Front. Neurol. 2012. [Google Scholar] [CrossRef]
- Riemann, D.; Hohagen, F.; Bahro, M.; Berger, M. Sleep in depression: The influence of age, gender and diagnostic subtype on baseline sleep and the cholinergic REM induction test with RS 86. Eur. Arch. Psychiatry Clin. Neurosci. 1994, 243, 279–290. [Google Scholar] [CrossRef]
- Armitage, R. The effects of antidepressants on sleep in patients with depression. Can. J. Psychiatry 2000, 45, 803–809. [Google Scholar] [CrossRef]
- Wichniak, A.; Wierzbicka, A.; Jernajczyk, W. Sleep as a biomarker for depression. Int. Rev. Psychiatry 2013, 25, 632–645. [Google Scholar] [CrossRef]
- Pandi-Perumal, S.R.; Kramer, M. (Eds.) Sleep and Mental Illness; Cambridge University Press: New York, NY, USA, 2010. [Google Scholar]
- Ford, A.T.; Fong, P.P. The effects of antidepressants appear to be rapid and at environmentally relevant concentrations. Environ. Toxicol. Chem. 2016, 35, 794–798. [Google Scholar] [CrossRef] [PubMed]
- Kay, D.B.; Dombrovski, A.Y.; Buysse, D.J.; Reynolds, C.F.; Begley, A.; Szanto, K. Insomnia is associated with suicide attempt in middle-aged and older adults with depression. Int. Psychogeriatr. 2016, 28, 613–619. [Google Scholar] [CrossRef] [PubMed]
- Monaca, C.; Boutrel, B.; Hen, R.; Hamon, M.; Lle Adrien, J. 5-HT 1A/1B Receptor-Mediated Effects of the Selective Serotonin Reuptake Inhibitor, Citalopram, on Sleep: Studies in 5-HT 1A and 5-HT 1B Knockout Mice. Neuropsychopharmacology 2003, 28, 850–856. [Google Scholar] [CrossRef] [PubMed]
- Gillin, J.C.; Jernajczyk, W.; de Valladares-Neto, D.C.; Golshan, S.; Lardon, M.; Stahl, S.M. Inhibition of REM sleep by ipsapirone, A 5HT1A agonist, in normal volunteers. Psychopharmacology 1994, 116, 433–436. [Google Scholar] [CrossRef]
- Landolt, H.P.; Kelsoe, J.R.; Rapaport, M.H.; Gillin, J.C. Rapid tryptophan depletion reverses phenelzine-induced suppression of REM sleep. J. Sleep Res. 2003, 12, 13–18. [Google Scholar] [CrossRef] [PubMed]
- Argyropoulos, S.V.; Wilson, S.J. Sleep disturbances in depression and the effects of antidepressants. Int. Rev. Psychiatry 2005, 17, 237–245. [Google Scholar] [CrossRef]
- Schmid, D.A.; Wichniak, A.; Uhr, M.; Ising, M.; Brunner, H.; Held, K.; Weikel, J.C.; Sonntag, A.; Steiger, A. Changes of Sleep Architecture, Spectral Composition of Sleep EEG, the Nocturnal Secretion of Cortisol, ACTH, GH, Prolactin, Melatonin, Ghrelin, and Leptin, and the DEX-CRH Test in Depressed Patients during Treatment with Mirtazapine. Neuropsychopharmacology 2006, 31, 832–844. [Google Scholar] [CrossRef] [PubMed]
- Lam, R.W. Sleep disturbances and depression: A challenge for antidepressants. Int. Clin. Psychopharmacol. 2006, 21, S25–S29. [Google Scholar] [CrossRef] [PubMed]
- Steiger, A.; Pawlowski, M. Depression and sleep. Int. J. Mol. Sci. 2019, 20, 607. [Google Scholar] [CrossRef]
- Haas, H.; Panula, P. The role of histamine and the tuberomamillary nucleus in the nervous system. Nat. Rev. Neurosci. 2003, 4, 121–130. [Google Scholar] [CrossRef] [PubMed]
- Hicks, J.A.; Argyropoulos, S.V.; Rich, A.S.; Nash, J.R.; Bell, C.J.; Edwards, C.; Nutt, D.J.; Wilson, S.J. Randomised controlled study of sleep after nefazodone or paroxetine treatment in out-patients with depression. Br. J. Psychiatry 2002, 180, 528–535. [Google Scholar] [CrossRef] [PubMed]
- Rush, A.J.; Armitage, R.; Gillin, J.C.; Yonkers, K.A.; Winokur, A.; Moldofsky, H.; Vogel, G.W.; Kaplita, S.B.; Fleming, J.B.; Montplaisir, J.; et al. Comparative effects of nefazodone and fluoxetine on sleep in outpatients with major depressive disorder. Biol. Psychiatry 1998, 44, 3–14. [Google Scholar] [CrossRef]
- Jindal, R.D.; Friedman, E.S.; Berman, S.R.; Fasiczka, A.L.; Howland, R.H.; Thase, M.E. Effects of Sertraline on Sleep Architecture in Patients with Depression. J. Clin. Psychopharmacol. 2003, 23, 540–548. [Google Scholar] [CrossRef] [PubMed]
- Ehlers, C.L.; Havstad, J.W.; Kupfer, D.J. Estimation of the time course of slow-wave sleep over the night in depressed patients: Effects of clomipramine and clinical response. Biol. Psychiatry 1996, 39, 171–181. [Google Scholar] [CrossRef]
- Kupfer, D.J.; Frank, E.; Mceachran, A.B.; Grochocinski, V.J. Delta Sleep Ratio: A Biological Correlate of Early Recurrence in Unipolar Affective Disorder. Arch. Gen. Psychiatry 1990, 47, 1100–1105. [Google Scholar] [CrossRef]
- Wang, Y.-Q.; Li, R.; Zhang, M.-Q.; Zhang, Z.; Qu, W.-M.; Huang, Z.-L. The Neurobiological Mechanisms and Treatments of REM Sleep Disturbances in Depression. Curr. Neuropharmacol. 2015, 13, 543–553. [Google Scholar] [CrossRef]
- DeMartinis, N.; Winokur, A. Effects of Psychiatric Medications on Sleep and Sleep Disorders. CNS Neurol. Disord. Drug Targets 2008, 6, 17–29. [Google Scholar] [CrossRef] [PubMed]
- Sateia, M.J.; Buysse, D.J.; Krystal, A.D.; Neubauer, D.N.; Heald, J.L. Clinical Practice Guideline for the Pharmacologic Treatment of Chronic Insomnia in Adults: An American academy of sleep medicine clinical practice guideline. J. Clin. Sleep Med. 2017, 13, 307–349. [Google Scholar] [CrossRef] [PubMed]
- Sonntag, A.; Rothe, B.; Guldner, J.; Yassouridis, A.; Holsboer, F.; Steiger, A. Trimipramine and imipramine exert different effects on the sleep EEG and on nocturnal hormone secretion during treatment of major depression. Depression 1996, 4, 1–13. [Google Scholar] [CrossRef]
- Dunleavy, D.L.; Brezinova, V.; Oswald, I.; Maclean, A.W.; Tinker, M. Changes during weeks in effects of tricyclic drugs on the human sleeping brain. Br. J. Psychiatry 1972, 120, 663–672. [Google Scholar] [CrossRef]
- Nicholson, A.N.; Pascoe, P.A.; Turner, C. Modulation of sleep by trimipramine in man. Eur. J. Clin. Pharmacol. 1989, 37, 145–150. [Google Scholar] [CrossRef]
- Ware, J.C.; Brown, F.W.; Moorad, P.J.; Pittard, J.T.; Cobert, B. Effects on sleep: A double-blind study comparing trimipramine to imipramine in depressed insomniac patients. Sleep 1989, 12, 537–549. [Google Scholar] [CrossRef]
- Gursky, J.T.; Krahn, L.E. The effects of antidepressants on sleep: A review. Harv. Rev. Psychiatry 2000, 8, 298–306. [Google Scholar] [CrossRef]
- Höchli, D.; Riemann, D.; Zulley, J.; Berger, M. Is there a relationship between response to total sleep deprivation and efficacy of clomipramine treatment in depressed patients? Acta Psychiatr. Scand. 1986, 74, 190–192. [Google Scholar] [CrossRef][Green Version]
- Kupfer, D.J.; Perel, J.M.; Pollock, B.G.; Nathan, R.S.; Grochocinski, V.J.; Wilson, M.J.; McEachran, A.B. Fluvoxamine versus desipramine: Comparative polysomnographic effects. Biol. Psychiatry 1991, 29, 23–40. [Google Scholar] [CrossRef]
- Shain, B.N.; Naylor, M.; Shipley, J.E.; Alessi, N. Imipramine effects on sleep in depressed adolescents: A preliminary report. Biol. Psychiatry 1990, 28, 459–462. [Google Scholar] [CrossRef]
- Shipley, J.E.; Kupfer, D.J.; Griffin, S.J.; Dealy, R.S.; Coble, P.A.; McEachran, A.B.; Grochocinski, V.J.; Ulrich, R.; Perel, J.M. Comparison of effects of desipramine and amitriptyline on EEG sleep of depressed patients. Psychopharmacology 1985, 85, 14–22. [Google Scholar] [CrossRef] [PubMed]
- Mendlewicz, J.; Kempenaers, C.; de Maertelaer, V. Sleep EEG and amitryptiline treatment in depressed inpatients. Biol. Psychiatry 1991, 30, 691–702. [Google Scholar] [CrossRef]
- Staner, L.; Kerkhofs, M.; Detroux, D.; Leyman, S.; Linkowski, P.; Mendlewicz, J. Acute, subchronic and withdrawal sleep EEG changes during treatment with paroxetine and amitriptyline: A double-blind randomized trial in major depression. Sleep 1995, 18, 470–477. [Google Scholar] [CrossRef] [PubMed]
- Wilson, S.J.; Bailey, J.E.; Alford, C.; Nutt, D.J. Sleep and daytime sleepiness the next day following single night-time dose of fluvoxamine, dothiepin and placebo in normal volunteers. J. Psychopharmacol. 2000, 14, 378–386. [Google Scholar] [CrossRef] [PubMed]
- Eugene, A.R. Association of sleep among 30 antidepressants: A population-wide adverse drug reaction study, 2004–2019. PeerJ 2020, 8, e8748. [Google Scholar] [CrossRef] [PubMed]
- Maudhuit, C.; Jolas, T.; Lainey, E.; Hamon, M.; Adrien, J. Effects of acute and chronic treatment with amoxapine and cericlamine on the sleep-wakefulness cycle in the rat. Neuropharmacology 1994, 33, 1017–1025. [Google Scholar] [CrossRef]
- Loas, G. Implications of 5-HT2 receptors in the increase of slow wave sleep in healthy volunteers under amoxapine. Encephale 1991, 3, 423–425. [Google Scholar]
- Brebbia, D.R.; Branchey, M.H.; Pyne, E.; Watson, J.; Brebbia, A.F.; Simpson, G. The effects of amoxapine on electroencephalographic stages of sleep in normal human subjects. Psychopharmacologia 1975, 45, 1–7. [Google Scholar] [CrossRef]
- Nicholson, A.N.; Pascoe, P.A. 5-Hydroxytryptamine and noradrenaline uptake inhibition: Studies on sleep in man. Neuropharmacology 1986, 25, 1079–1083. [Google Scholar] [CrossRef]
- Polc, P.; Haefely, W.; Schneeberger, J. Effect of tricyclic and tetracyclic antidepressants on the sleep-wakefulness cycle of cats. In Sleep; Koella, W.P., Levin, P., Eds.; Karger: Basel, Switzerland, 1976; pp. 380–382. [Google Scholar]
- Mendlewicz, J.; Dunbar, G.C.; Hoffman, G. Changes in sleep EEG architecture during the treatment of depressed patients with mianserin. Acta Psychiatr. Scand. 1985, 72, 26–29. [Google Scholar] [CrossRef]
- Maeda, Y.; Hayashi, T.; Furuta, H.; Kim, Y.; Morikawa, K.; Ishiguro, N.; Ueno, K.; Sano, J.; Yamaguchi, N. Effects of mianserin on human sleep. Neuropsychobiology 1990, 24, 198–204. [Google Scholar] [CrossRef] [PubMed]
- Schittecatte, M.; Dumont, F.; Machowski, R.; Cornil, C.; Lavergne, F.; Wilmotte, J. Effects of mirtazapine on sleep polygraphic variables in major depression. Neuropsychobiology 2002, 46, 197–201. [Google Scholar] [CrossRef] [PubMed]
- Shen, J.; Chung, S.A.; Kayumov, L.; Moller, H.; Hossain, N.; Wang, X.; Deb, P.; Sun, F.; Huang, X.; Novak, M.; et al. Polysomnographic and symptomatological analyses of major depressive disorder patients treated with mirtazapine. Can. J. Psychiatry 2006, 51, 27–34. [Google Scholar] [CrossRef] [PubMed]
- Wilson, S.J.; Nutt, D.J. SSRIs and sleep in man. In Sleep and Sleep Disorders: A Neuropsychopharmacological Approach; Lader, M.H., Cardinali, D.P., Pandi-Perumal, S.R., Eds.; Springer: Boston, MA, USA, 2006; pp. 269–273. ISBN 9780387276823. [Google Scholar]
- Wyatt, R.J.; Fram, D.H.; Kupfer, D.J.; Snyder, F. Total Prolonged Drug-Induced REM Sleep Suppression in Anxious-Depressed Patients. Arch. Gen. Psychiatry 1971, 24, 145–155. [Google Scholar] [CrossRef] [PubMed]
- Landolt, H.P.; Raimo, E.B.; Schnierow, B.J.; Kelsoe, J.R.; Rapaport, M.H.; Gillin, J.C. Sleep and sleep electroencephalogram in depressed patients treated with phenelzine. Arch. Gen. Psychiatry 2001, 58, 268–276. [Google Scholar] [CrossRef] [PubMed]
- Blois, R.; Gaillard, J.M. Effects of moclobemide on sleep in healthy human subjects. Acta Psychiatr. Scand. 1990, 82, 73–75. [Google Scholar] [CrossRef]
- Monti, J.M.; Alterwain, P.; Monti, D. The effects of moclobemide on nocturnal sleep of depressed patients. J. Affect. Disord. 1990, 20, 201–208. [Google Scholar] [CrossRef]
- Steiger, A.; Benkert, O.; Holsboer, F. Effects of Long-Term Treatment with the MAO-A Inhibitor Moclobemide on Sleep EEG and Nocturnal Hormonal Secretion in Normal Men. Neuropsychobiology 1994, 30, 101–105. [Google Scholar] [CrossRef]
- Jindal, R.D.; Fasiczka, A.L.; Himmelhoch, J.M.; Mallinger, A.G.; Thase, M.E. Effects of tranylcypromine on the sleep of patients with anergic bipolar depression. Psychopharmacol. Bull. 2003, 37, 118–126. [Google Scholar]
- Feige, B.; Voderholzer, U.; Riemann, D.; Dittmann, R.; Hohagen, F.; Berger, M. Fluoxetine and sleep EEG: Effects of a single dose, subchronic treatment, and discontinuation in healthy subjects. Neuropsychopharmacology 2002, 26, 246–258. [Google Scholar] [CrossRef]
- van Bemmel, A.L.; van den Hoofdakker, R.H.; Beersma, D.G.M.; Bouhuys, A.L. Changes in sleep polygraphic variables and clinical state in depressed patients during treatment with citalopram. Psychopharmacology 1993, 113, 225–230. [Google Scholar] [CrossRef] [PubMed]
- Trivedi, M.H.; Rush, A.J.; Armitage, R.; Gullion, C.M.; Grannemann, B.D.; Orsulak, P.J.; Roffwarg, H.P. Effects of fluoxetine on the polysomnogram in outpatients with major depression. Neuropsychopharmacology 1999, 20, 447–459. [Google Scholar] [CrossRef]
- Luthringer, R. A double-blind, placebo-controlled evaluation of the effects of orally administered venlafaxine on sleep in inpatients with major depression. Psychopharmacol. Bull. 1996, 32, 637–646. [Google Scholar] [PubMed]
- Chalon, S.; Pereira, A.; Lainey, E.; Vandenhende, F.; Watkin, J.G.; Staner, L.; Granier, L.A. Comparative effects of duloxetine and desipramine on sleep EEG in healthy subjects. Psychopharmacology 2005, 177, 1–10. [Google Scholar] [CrossRef]
- Kluge, M.; Schüssler, P.; Steiger, A. Duloxetine increases stage 3 sleep and suppresses rapid eye movement (REM) sleep in patients with major depression. Eur. Neuropsychopharmacol. 2007, 17, 527–531. [Google Scholar] [CrossRef]
- Lemoine, P.; Faivre, T. Subjective and polysomnographic effects of milnacipran on sleep in depressed patients. Hum. Psychopharmacol. Clin. Exp. 2004, 19, 299–303. [Google Scholar] [CrossRef]
- Rahmadi, M.; Narita, M.; Yamashita, A.; Imai, S.; Kuzumaki, N.; Suzuki, T. Sleep disturbance associated with an enhanced orexinergic system induced by chronic treatment with paroxetine and milnacipran. Synapse 2011, 65, 652–657. [Google Scholar] [CrossRef]
- Krystal, A.; Thase, M.; Tucker, V.; Goodale, E. Bupropion HCL and Sleep in Patients with Depression. Curr. Psychiatry Rev. 2007, 3, 123–128. [Google Scholar] [CrossRef]
- Nofzinger, E.A.; Berman, S.; Fasiczka, A.; Miewald, J.M.; Meltzer, C.C.; Price, J.C.; Sembrat, R.C.; Wood, A.; Thase, M.E. Effects of bupropion SR on anterior paralimbic function during waking and REM sleep in depression: Preliminary findings using [18F]-FDG PET. Psychiatry Res. Neuroimaging 2001, 106, 95–111. [Google Scholar] [CrossRef]
- Ott, G.E.; Rao, U.; Nuccio, I.; Lin, K.M.; Poland, R.E. Effect of bupropion-SR on REM sleep: Relationship to antidepressant response. Psychopharmacology 2002, 165, 29–36. [Google Scholar] [CrossRef]
- Nofzinger, E.A.; Reynolds, C.F.; Thase, M.E.; Frank, E.; Jennings, J.R.; Fasiczka, A.L.; Sullivan, L.R.; Kupfer, D.J. REM sleep enhancement by bupropion in depressed men. Am. J. Psychiatry 1995, 152, 274–276. [Google Scholar] [CrossRef] [PubMed]
- Schramm, P.J.; Poland, R.E.; Rao, U. Bupropion response on sleep quality in patients with depression: Implications for increased cardiovascular disease risk. Eur. Neuropsychopharmacol. 2014, 24, 207–214. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Mouret, J.; Lemoine, P.; Minuit, M.P.; Benkelfat, C.; Renardet, M. Effects of trazodone on the sleep of depressed subjects—A polygraphic study. Psychopharmacology 1988, 95. [Google Scholar] [CrossRef] [PubMed]
- Montgomery, I.; Oswald, I.; Morgan, K.; Adam, K. Trazodone enhances sleep in subjective quality but not in objective duration. Br. J. Clin. Pharmacol. 1983, 16, 139–144. [Google Scholar] [CrossRef] [PubMed]
- van Bemmel, A.L.; Havermans, R.G.; van Diest, R. Effects of trazodone on EEG sleep and clinical state in major depression. Psychopharmacology 1992, 107, 569–574. [Google Scholar] [CrossRef]
- Yamadera, H.; Nakamura, S.; Suzuki, H.; Endo, S. Effects of trazodone hydrochloride and imipramine on polysomnography in healthy subjects. Psychiatry Clin. Neurosci. 1998, 52, 439–443. [Google Scholar] [CrossRef]
- Sharpley, A.L.; Walsh, A.E.S.; Cowen, P.J. Nefazodone-A novel antidepressant-May increase REM sleep. Biol. Psychiatry 1992, 31, 1070–1073. [Google Scholar] [CrossRef]
- Sharpley, A.L.; Williamson, D.J.; Attenburrow, M.E.J.; Pearson, G.; Sargent, P.; Cowen, P.J. The effects of paroxetine and nefazodone on sleep: A placebo controlled trial. Psychopharmacology 1996, 126, 50–54. [Google Scholar] [CrossRef] [PubMed]
- Ware, J.C.; Rose, F.V.; McBrayer, R.H. The acute effects of nefazodone, trazodone and buspirone on sleep and sleep-related penile tumescence in normal subjects. Sleep 1994, 17, 544–550. [Google Scholar] [CrossRef][Green Version]
- Bramanti, P.; Bianchi, L.; Benedetto, M.; Ricci, R.M.; Scribano, M.A.; Di Perri, R. Study of the hypnic effect of amineptine evaluation by means of polygraphy and tests. Prog. Neuropsychopharmacol. Biol. Psychiatry 1985, 9, 157–165. [Google Scholar] [CrossRef]
- Di Perri, R.; Maill, F.; Bramanti, P. The effects of amineptine on the mood and nocturnal sleep of depressed patients. Prog. Neuropsychopharmacol. Biol. Psychiatry 1987, 11, 65–70. [Google Scholar] [CrossRef]
- Ferini-Strambi, L.; Manconi, M.; Castronovo, V.; Riva, L.; Bianchi, A. Effects of reboxetine on sleep and nocturnal cardiac autonomic activity in patients with dysthymia. J. Psychopharmacol. 2004, 18, 417–422. [Google Scholar] [CrossRef] [PubMed]
- Wilson, W.H.; Freemon, F.R.; Ban, T.A.; Petrie, W.M.; Clinton, C.L. Acute effects of viloxazine HCl and flurazepam when given alone and in combination on sleep EEG: A double-blind interaction study with normals. Pavlov. J. Biol. Sci. Off. J. Pavlov. 1980, 15, 68–73. [Google Scholar] [CrossRef]
- Březinová, V.; Adam, K.; Chapman, K.; Oswald, I.; Thomson, J. Viloxazine, sleep, and subjective feelings. Psychopharmacology 1977, 55, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Salva, M.A.Q.; Vanier, B.; Laredo, J.; Hartley, S.; Chapotot, F.; Moulin, C.; Lofaso, F.; Guilleminault, C. Major depressive disorder, sleep EEG and agomelatine: An open-label study. Int. J. Neuropsychopharmacol. 2007, 10, 691–696. [Google Scholar] [CrossRef]
- Quera-Salva, M.A.; Lemoine, P.; Guilleminault, C. Impact of the novel antidepressant agomelatine on disturbed sleep-wake cycles in depressed patients. Hum. Psychopharmacol. 2010, 25, 222–229. [Google Scholar] [CrossRef]
- Quera-Salva, M.A.; Hajak, G.; Philip, P.; Montplaisir, J.; Keufer-Le Gall, S.; Laredo, J.; Guilleminault, C. Comparison of agomelatine and escitalopram on nighttime sleep and daytime condition and efficacy in major depressive disorder patients. Int. Clin. Psychopharmacol. 2011, 26, 252–262. [Google Scholar] [CrossRef]
- Ahnaou, A.; Huysmans, H.; Biermans, R.; Manyakov, N.V.; Drinkenburg, W.H.I.M. Ketamine: Differential neurophysiological dynamics in functional networks in the rat brain. Transl. Psychiatry 2017, 7, e1237. [Google Scholar] [CrossRef]
- Duman, R.S.; Shinohara, R.; Fogaça, M.V.; Hare, B. Neurobiology of rapid-acting antidepressants: Convergent effects on GluA1-synaptic function. Mol. Psychiatry 2019, 24, 1816–1832. [Google Scholar]
- Duncan, W.C.; Ballard, E.D.; Zarate, C.A. Ketamine-Induced Glutamatergic Mechanisms of Sleep and Wakefulness: Insights for Developing Novel Treatments for Disturbed Sleep and Mood. In Handbook of Experimental Pharmacology; Springer: New York, NY, USA, 2019; Volume 253, pp. 337–358. [Google Scholar]
- Duncan, W.C.; Zarate, C.A. Ketamine, sleep, and depression: Current status and new questions. Curr. Psychiatry Rep. 2013, 15. [Google Scholar] [CrossRef]
- Gassaway, M.M.; Rives, M.L.; Kruegel, A.C.; Javitch, J.A.; Sames, D. The atypical antidepressant and neurorestorative agent tianeptine is a μ-opioid receptor agonist. Transl. Psychiatry 2014, 4. [Google Scholar] [CrossRef] [PubMed]
- Samuels, B.A.; Nautiyal, K.M.; Kruegel, A.C.; Levinstein, M.R.; Magalong, V.M.; Gassaway, M.M.; Grinnell, S.G.; Han, J.; Ansonoff, M.A.; Pintar, J.E.; et al. The Behavioral Effects of the Antidepressant Tianeptine Require the Mu-Opioid Receptor. Neuropsychopharmacology 2017, 42, 2052–2063. [Google Scholar] [CrossRef] [PubMed]
- Murck, H.; Nickel, T.; Künzel, H.; Antonijevic, I.A.; Schill, J.; Zobel, A.; Steiger, A.; Sonntag, A.; Holsboer, F. State markers of depression in sleep EEG: Dependency on drug and gender in patients treated with tianeptine orparoxetine. Neuropsychopharmacology 2003, 28, 348–358. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.-M.; Han, C.; Lee, S.-J.; Patkar, A.A.; Masand, P.S.; Pae, C.-U. Vilazodone for the Treatment of Depression: An Update. Chonnam Med. J. 2016, 52, 91. [Google Scholar] [CrossRef]
- Murck, H.; Frieboes, R.M.; Antonijevic, I.A.; Steiger, A. Distinct temporal pattern of the effects of the combined serotonin-reuptake inhibitor and 5-HT1A agonist EMD 68843 on the sleep EEG in healthy men. Psychopharmacology 2001, 155, 187–192. [Google Scholar] [CrossRef]
- Florea, I.; Loft, H.; Danchenko, N.; Rive, B.; Brignone, M.; Merikle, E.; Jacobsen, P.L.; Sheehan, D.V. The effect of vortioxetine on overall patient functioning in patients with major depressive disorder. Brain Behav. 2017, 7, 622. [Google Scholar] [CrossRef]
- Wilson, S.; Højer, A.M.; Buchberg, J.; Areberg, J.; Nutt, D.J. Differentiated effects of the multimodal antidepressant vortioxetine on sleep architecture: Part 1, a pharmacokinetic/pharmacodynamic comparison with paroxetine in healthy men. J. Psychopharmacol. 2015, 29, 1085–1091. [Google Scholar] [CrossRef]
- Liguori, C.; Ferini-Strambi, L.; Izzi, F.; Mari, L.; Manfredi, N.; D’Elia, A.; Mercuri, N.B.; Placidi, F. Preliminary evidence that vortioxetine may improve sleep quality in depressed patients with insomnia: A retrospective questionnaire analysis. Br. J. Clin. Pharmacol. 2019, 85, 240–244. [Google Scholar] [CrossRef]
- Emslie, G.J.; Armitage, R.; Weinberg, W.A.; John Rush, A.; Mayes, T.L.; Hoffmann, R.F. Sleep polysomnography as a predictor of recurrence in children and adolescents with major depressive disorder. Int. J. Neuropsychopharmacol. 2001, 4, 159–168. [Google Scholar] [CrossRef]
- Rao, U. Sleep disturbances in pediatric depression. Asian J. Psychiatr. 2011, 4, 234–247. [Google Scholar] [CrossRef]
- Bertocci, M.A.; Dahl, R.E.; Williamson, D.E.; Iosif, A.M.; Birmaher, B.; Axelson, D.; Ryan, N.D. Subjective sleep complaints in pediatric depression: A controlled study and comparison with EEG measures of sleep and waking. J. Am. Acad. Child Adolesc. Psychiatry 2005, 44, 1158–1166. [Google Scholar] [CrossRef] [PubMed]
- Dahl, R.E.; Puig-Antich, J.; Ryan, N.D.; Nelson, B.; Dachille, S.; Cunningham, S.L.; Trubnick, L.; Klepper, T.P. EEG sleep in adolescents with major depression: The role of suicidality and inpatient status. J. Affect. Disord. 1990, 19, 63–75. [Google Scholar] [CrossRef]
- Forbes, E.E.; Bertocci, M.A.; Gregory, A.M.; Ryan, N.D.; Axelson, D.A.; Birmaher, B.; Dahl, R.E. Objective sleep in pediatric anxiety disorders and major depressive disorder. J. Am. Acad. Child Adolesc. Psychiatry 2008, 47, 148–155. [Google Scholar] [CrossRef] [PubMed]
- Arana-Lechuga, Y.; Nuñez-Ortiz, R.; Terán-Pérez, G.; Castillo-Montoya, C.; Jiménez-Anguiano, A.; Gonzalez-Robles, R.; Castro-Roman, R.; Velázquez-Moctezuma, J. Sleep-EEG patterns of school children suffering from symptoms of depression compared to healthy controls. World J. Biol. Psychiatry 2008, 9, 115–120. [Google Scholar] [CrossRef]
- Shahid, A.; Khairandish, A.; Gladanac, B.; Shapiro, C. Peeking into the minds of troubled adolescents: The utility of polysomnography sleep studies in an inpatient psychiatric unit. J. Affect. Disord. 2012, 139, 66–74. [Google Scholar] [CrossRef]
- Singareddy, R.; Krishnamurthy, V.B.; Vgontzas, A.N.; Fernandez-Mendoza, J.; Calhoun, S.L.; Shaffer, M.L.; Bixler, E.O. Subjective and objective sleep and self-harm behaviors in young children: A general population study. Psychiatry Res. 2013, 209, 549–553. [Google Scholar] [CrossRef]
- Boafo, A.; Armitage, R.; Greenham, S.; Tavakoli, P.; Dale, A.; Nixon, A.; Lafrenière, A.; Ray, L.B.; De Koninck, J.; Robillard, R. Sleep architecture in adolescents hospitalized during a suicidal crisis. Sleep Med. 2019, 56, 41–46. [Google Scholar] [CrossRef]
- Goetz, R.R.; Wolk, S.I.; Coplan, J.D.; Ryan, N.D.; Weissman, M.M. Premorbid polysomnographic signs in depressed adolescents: A reanalysis of EEG sleep after longitudinal follow-up in adulthood. Biol. Psychiatry 2001, 49, 930–942. [Google Scholar] [CrossRef]
- Armitage, R.; Emslie, G.; Rintelmann, J. The effect of fluoxetine on sleep EEG in childhood depression: A preliminary report. Neuropsychopharmacology 1997, 17, 241–245. [Google Scholar] [CrossRef]
| Sleep Stage | Characteristic Features |
|---|---|
| N1 |
|
| N2 |
|
| N3 |
|
| REM |
|
| Parameter | Definition | Normal values | |
|---|---|---|---|
| in Young Adult Patients | in Elderly Patients | ||
| Parameters of sleep continuity | |||
| Sleep latency | The length of time that it takes to accomplish the transition from full wakefulness to sleep after the lights are turned off. | <30 min | <45 min |
| Total sleep time (TST) | The total time spent asleep during the sleep episode. | >6.5 h | >6 h |
| Sleep efficiency (SE) | Percentage of time spent asleep of the total time spent in bed. | >90% | >85% |
| Wake after sleep onset (WASO) | The length of time spent awake after the sleep onset. | <30 min | <30 min |
| Parameters of sleep depth | |||
| Total and relative amounts of stage N3 | Duration of N3 expressed as minutes or percentage relative to TST. | 20–25% | 10% |
| Delta sleep ratio (DSR) | The ratio of SWS in the first NREM period to SWS in the second. | >1.1 | >1.1 |
| Parameters of REM sleep | |||
| REM latency | The length of time that it takes from onset of sleep to the start of the first REM episode. | <65 min | <50 min |
| Total and relative amounts of stage REM | Duration of REM expressed as minutes or percentage relative to TST. | 20–25% | 20–25% |
| REM density | Measure of the frequency of rapid eye movements during REM sleep expressed as movements per minute. | 9–12 movements per min | 9–12 movements per min |
| Antidepressant Class | Drugs | REM Sleep | ROL | SWS | Sleep Continuity |
|---|---|---|---|---|---|
| TCA (sedative) | amitriptyline, doxepin | ↓ | ↑ | ↑ | ↑ |
| trimipramine | 0/↑ | 0/↓ | ↑ | ↑ | |
| TCA (activating) | desipramine, imipramine, nortriptyline, protriptyline | ↓ | ↑ | ↓ | ↓ |
| clomipramine | ↓↓↓ | ↑↑↑ | ↓ | ↓ | |
| TeCA | amoxapine | ↓ | ↑ | 0/↑ | 0 |
| maprotiline | 0/↓ | 0 | ↑ | 0 | |
| mianserin | ↓ | ↑ | ↑ | ↑ | |
| mirtazapine | 0 | 0/↑ | ↑ | ↑ | |
| MAOI (reversible) | moclobemide | ↓ | ↑ | 0/↑ | ↑ |
| MAOI (irreversible) | phenelzine, tranylcypromine | ↓↓↓ | ↑↑↑ | 0 | 0/↓ |
| SSRI | citalopram, escitalopram, fluoxetine, fluvoxamine, paroxetine, sertraline | ↓ | ↑ | 0 | ↓ |
| SNRI | duloxetine, venlafaxine | ↓ | ↑ | ↑ | ↓ |
| milnacipran | 0 | 0/↑ | 0 | 0/↓ | |
| DNRI | bupropion | 0 | 0 | 0/↑ | 0/↓ |
| SARI | trazodone | 0/↓ | 0/↑ | ↑ | ↑ |
| nefazodone | 0/↑ | 0 | ↑ | ↑ | |
| DARI | amineptine | ↑ | 0/↓ | ↓ | ↓ |
| NARI | atomoxetine, reboxetine, viloxazine | ↓ | ↑ | 0/↓ | 0/↓ |
| Novel drugs | agomelatine | 0 | 0 | ↑ | ↑ |
| ketamine | ↓ | ↑ | ↑ | ↑ | |
| tianeptine | 0 | 0 | 0 | 0 | |
| vilazodone | ↓ | ↑ | ↑ | ↓ | |
| vortioxetine | ↓ | ↑ | 0 | 0/↓ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hutka, P.; Krivosova, M.; Muchova, Z.; Tonhajzerova, I.; Hamrakova, A.; Mlyncekova, Z.; Mokry, J.; Ondrejka, I. Association of Sleep Architecture and Physiology with Depressive Disorder and Antidepressants Treatment. Int. J. Mol. Sci. 2021, 22, 1333. https://doi.org/10.3390/ijms22031333
Hutka P, Krivosova M, Muchova Z, Tonhajzerova I, Hamrakova A, Mlyncekova Z, Mokry J, Ondrejka I. Association of Sleep Architecture and Physiology with Depressive Disorder and Antidepressants Treatment. International Journal of Molecular Sciences. 2021; 22(3):1333. https://doi.org/10.3390/ijms22031333
Chicago/Turabian StyleHutka, Peter, Michaela Krivosova, Zuzana Muchova, Ingrid Tonhajzerova, Andrea Hamrakova, Zuzana Mlyncekova, Juraj Mokry, and Igor Ondrejka. 2021. "Association of Sleep Architecture and Physiology with Depressive Disorder and Antidepressants Treatment" International Journal of Molecular Sciences 22, no. 3: 1333. https://doi.org/10.3390/ijms22031333
APA StyleHutka, P., Krivosova, M., Muchova, Z., Tonhajzerova, I., Hamrakova, A., Mlyncekova, Z., Mokry, J., & Ondrejka, I. (2021). Association of Sleep Architecture and Physiology with Depressive Disorder and Antidepressants Treatment. International Journal of Molecular Sciences, 22(3), 1333. https://doi.org/10.3390/ijms22031333






