Changes Induced by Mind–Body Intervention Including Epigenetic Marks and Its Effects on Diabetes
Abstract
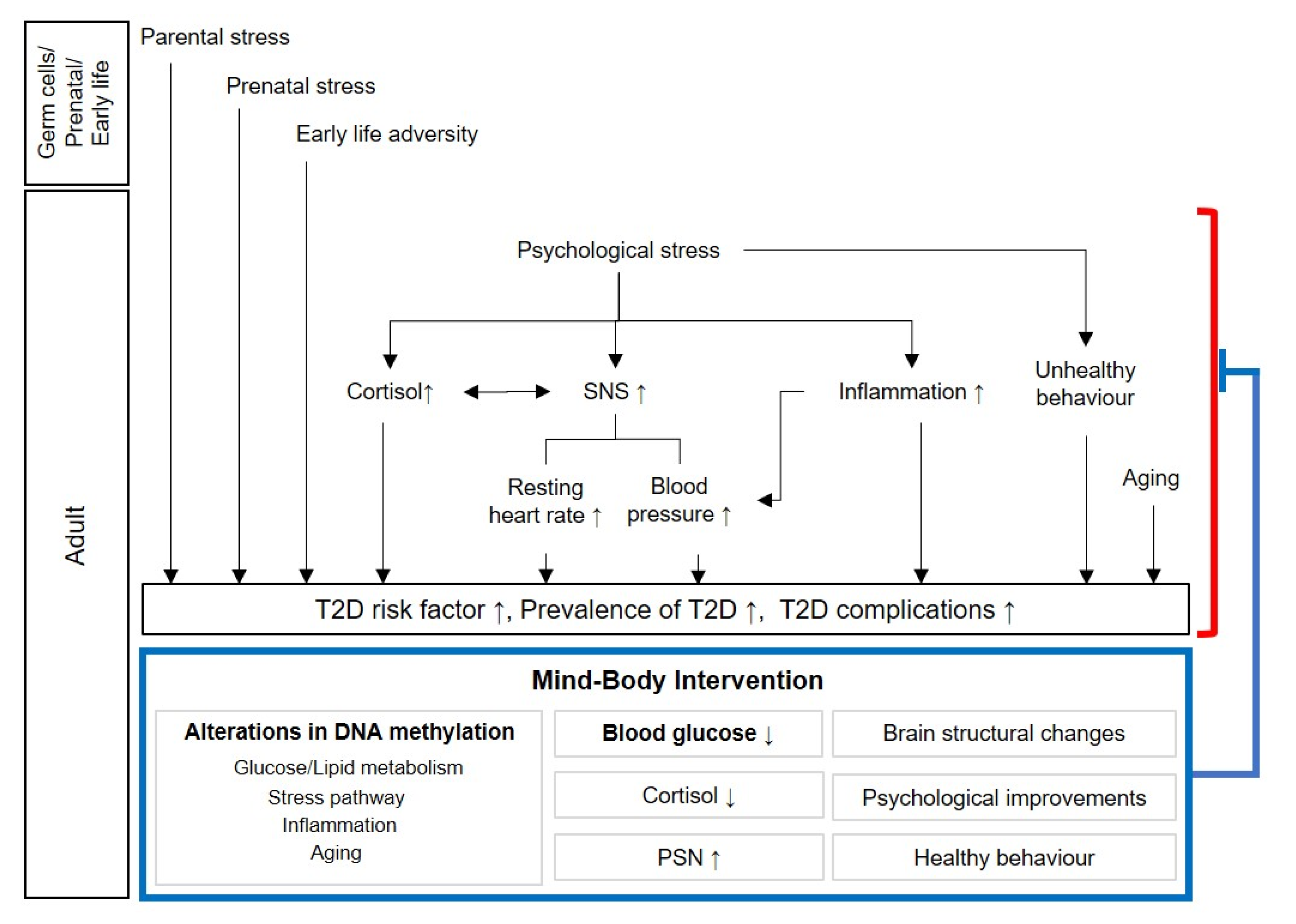
1. Introduction
1.1. Epigenetics
1.2. Epigenetic Changes and Diabetes
1.2.1. Diabetes-Related Epigenetic Changes in Parents, and During Prenatal and Early Life
1.2.2. Psychological Stress and Type 2 Diabetes
Psychological Factors Related to Type 2 Diabetes
Cortisol and Type 2 Diabetes
Autonomic Nervous System and Type 2 Diabetes
Inflammation and Type 2 Diabetes
Complications
1.2.3. Aging and Type 2 Diabetes
1.2.4. Lifestyle and Type 2 Diabetes
1.3. Types of Mind–Body Intervention and Their Effects
2. Epigenetic Changes Induced by Mind–Body Intervention and Their Effects on Diabetes
2.1. Effects of Mind–Body Intervention on Diabetes
2.1.1. Effects of Moving Meditation on Diabetes
Effects of Tai Chi on Diabetes
Effects of Qigong on Diabetes
Effects of Yoga on Diabetes
2.1.2. Effects of Combined Practices (Sitting Meditation and Moving Meditation) on Diabetes
Mindfulness-Based Intervention
Buddhist Walking Meditation
Brain Education Meditation
2.1.3. The Effects of Other Practices on Diabetes-Related Factors
2.2. Potential Mechanism for Diabetes-Related Effects of Mind–Body Intervention
2.2.1. Changes in Epigenetic Modifications Related to Glucose/Lipid Metabolism and Inflammation through Mind–Body Intervention
2.2.2. Reduction of Psychological Stress, A Risk Factor of Type 2 Diabetes, through Mind–Body Intervention
Cortisol Secretion and Glycemic Control through Mind–Body Intervention
Autonomic Nervous System Changes and Glycemic Control through Mind–Body Intervention
Inflammation Reduction and Glycemic Control through Mind–Body Intervention
2.2.3. Delayed Epigenetic Age through Mind–Body Intervention and Its Relation to Type 2 Diabetes
2.2.4. Glycemic Control through Lifestyle Changes with Mind–Body Intervention
2.3. Epigenetic Changes Induced by Non-Pharmacological Interventions in Addition to Mind–Body Interventions and Their Effects on Diabetes
3. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cavalli, G.; Heard, E. Advances in epigenetics link genetics to the environment and disease. Nature 2019, 571, 489–499. [Google Scholar] [CrossRef] [PubMed]
- Rönn, T.; Volkov, P.; Davegårdh, C.; Dayeh, T.; Hall, E.; Olsson, A.H.; Nilsson, E.; Tornberg, A.; Dekker Nitert, M.; Eriksson, K.F.; et al. A six months exercise intervention influences the genome-wide DNA methylation pattern in human adipose tissue. PLoS Genet. 2013, 9, e1003572. [Google Scholar] [CrossRef] [PubMed]
- Barrès, R.; Yan, J.; Egan, B.; Treebak, J.T.; Rasmussen, M.; Fritz, T.; Caidahl, K.; Krook, A.; O’Gorman, D.J.; Zierath, J.R. Acute exercise remodels promoter methylation in human skeletal muscle. Cell Metab. 2012, 15, 405–411. [Google Scholar] [CrossRef] [PubMed]
- Aristizabal, M.J.; Anreiter, I.; Halldorsdottir, T.; Odgers, C.L.; McDade, T.W.; Goldenberg, A.; Mostafavi, S.; Kobor, M.S.; Binder, E.B.; Sokolowski, M.B.; et al. Biological embedding of experience: A primer on epigenetics. Proc. Natl. Acad. Sci. USA 2020, 117, 23261–23269. [Google Scholar] [CrossRef]
- Hall, E.; Volkov, P.; Dayeh, T.; Bacos, K.; Rönn, T.; Nitert, M.D.; Ling, C. Effects of palmitate on genome-wide mRNA expression and DNA methylation patterns in human pancreatic islets. BMC Med. 2014, 12, 103. [Google Scholar] [CrossRef]
- Hall, E.; Dekker Nitert, M.; Volkov, P.; Malmgren, S.; Mulder, H.; Bacos, K.; Ling, C. The effects of high glucose exposure on global gene expression and DNA methylation in human pancreatic islets. Mol. Cell. Endocrinol. 2018, 472, 57–67. [Google Scholar] [CrossRef]
- Davegårdh, C.; García-Calzón, S.; Bacos, K.; Ling, C. DNA methylation in the pathogenesis of type 2 diabetes in humans. Mol. Metab. 2018, 14, 12–25. [Google Scholar] [CrossRef]
- Zhou, Z.; Sun, B.; Li, X.; Zhu, C. DNA methylation landscapes in the pathogenesis of type 2 diabetes mellitus. Nutr. Metab. 2018, 15, 47. [Google Scholar] [CrossRef]
- Ng, S.F.; Lin, R.C.; Laybutt, D.R.; Barres, R.; Owens, J.A.; Morris, M.J. Chronic high-fat diet in fathers programs beta-cell dysfunction in female rat offspring. Nature 2010, 467, 963–966. [Google Scholar] [CrossRef]
- Wu, L.; Lu, Y.; Jiao, Y.; Liu, B.; Li, S.; Li, Y.; Xing, F.; Chen, D.; Liu, X.; Zhao, J.; et al. Paternal psychological stress reprograms hepatic gluconeogenesis in offspring. Cell Metab. 2016, 23, 735–743. [Google Scholar] [CrossRef]
- Vaiserman, A.; Koliada, A.; Lushchak, O. Developmental programming of aging trajectory. Ageing Res. Rev. 2018, 47, 105–122. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Trejo, A.; Ortiz-López, M.G.; Zambrano, E.; Granados-Silvestre Mde, L.; Méndez, C.; Blondeau, B.; Bréant, B.; Nathanielsz, P.W.; Menjivar, M. Developmental programming of neonatal pancreatic β-cells by a maternal low-protein diet in rats involves a switch from proliferation to differentiation. Am. J. Physiol. Endocrinol. Metab. 2012, 302, E1431–E1439. [Google Scholar] [CrossRef] [PubMed]
- Pettitt, D.J.; Baird, H.R.; Aleck, K.A.; Bennett, P.H.; Knowler, W.C. Excessive obesity in offspring of Pima Indian women with diabetes during pregnancy. N. Engl. J. Med. 1983, 308, 242–245. [Google Scholar] [CrossRef] [PubMed]
- Pettitt, D.J.; Aleck, K.A.; Baird, H.R.; Carraher, M.J.; Bennett, P.H.; Knowler, W.C. Congenital susceptibility to NIDDM. Role of intrauterine environment. Diabetes 1988, 37, 622–628. [Google Scholar] [CrossRef] [PubMed]
- Roseboom, T.; de Rooij, S.; Painter, R. The Dutch famine and its long-term consequences for adult health. Early Hum. Dev. 2006, 82, 485–491. [Google Scholar] [CrossRef]
- Entringer, S.; Buss, C.; Wadhwa, P.D. Prenatal stress, telomere biology, and fetal programming of health and disease risk. Sci. Signal. 2012, 5, pt12. [Google Scholar] [CrossRef]
- Burgueño, A.L.; Juárez, Y.R.; Genaro, A.M.; Tellechea, M.L. Prenatal stress and later metabolic consequences: Systematic review and meta-analysis in rodents. Psychoneuroendocrinology 2020, 113, 104560. [Google Scholar] [CrossRef]
- Peña, C.J.; Monk, C.; Champagne, F.A. Epigenetic effects of prenatal stress on 11β-hydroxysteroid dehydrogenase-2 in the placenta and fetal brain. PLoS ONE 2012, 7, e39791. [Google Scholar]
- Nemoda, Z.; Szyf, M. Epigenetic alterations and prenatal maternal depression. Birth Defects Res. 2017, 109, 888–897. [Google Scholar] [CrossRef]
- Oberlander, T.F.; Weinberg, J.; Papsdorf, M.; Grunau, R.; Misri, S.; Devlin, A.M. Prenatal exposure to maternal depression, neonatal methylation of human glucocorticoid receptor gene (NR3C1) and infant cortisol stress responses. Epigenetics 2008, 3, 97–106. [Google Scholar] [CrossRef]
- Mulligan, C.; D’Errico, N.; Stees, J.; Hughes, D. Methylation changes at NR3C1 in newborns associate with maternal prenatal stress exposure and newborn birth weight. Epigenetics 2012, 7, 853–857. [Google Scholar] [CrossRef] [PubMed]
- Berends, L.M.; Ozanne, S.E. Early determinants of type-2 diabetes. Best Pract. Res. Clin. Endocrinol. Metab. 2012, 26, 569–580. [Google Scholar] [CrossRef] [PubMed]
- Estampador, A.C.; Franks, P.W. Precision medicine in obesity and type 2 diabetes: The relevance of early-life exposures. Clin. Chem. 2018, 64, 130–141. [Google Scholar] [CrossRef] [PubMed]
- Bansal, A.; Simmons, R.A. Epigenetics and developmental origins of diabetes: Correlation or causation? Am. J. Physiol. Endocrinol. Metab. 2018, 315, E15–E28. [Google Scholar] [CrossRef]
- Chen, M.; Lacey, R.E. Adverse childhood experiences and adult inflammation: Findings from the 1958 British birth cohort. Brain Behav. Immun. 2018, 69, 582–590. [Google Scholar] [CrossRef]
- Lacey, R.E.; Kumari, M.; Bartley, M. Social isolation in childhood and adult inflammation: Evidence from the National Child Development Study. Psychoneuroendocrinology 2014, 50, 85–94. [Google Scholar] [CrossRef]
- Tsalamandris, S.; Antonopoulos, A.S.; Oikonomou, E.; Papamikroulis, G.A.; Vogiatzi, G.; Papaioannou, S.; Deftereos, S.; Tousoulis, D. The role of inflammation in diabetes: Current concepts and future perspectives. Eur. Cardiol. 2019, 14, 50–59. [Google Scholar] [CrossRef]
- Hackett, R.A.; Steptoe, A. Type 2 diabetes mellitus and psychological stress—A modifiable risk factor. Nature Rev. Endocrinol. 2017, 13, 547. [Google Scholar] [CrossRef]
- Dallman, M.F.; Strack, A.M.; Akana, S.F.; Bradbury, M.J.; Hanson, E.S.; Scribner, K.A.; Smith, M. Feast and famine: Critical role of glucocorticoids with insulin in daily energy flow. Front. Neuroendocrinol. 1993, 14, 303–347. [Google Scholar] [CrossRef]
- Rod, N.H.; Kristensen, T.S.; Lange, P.; Prescott, E.; Diderichsen, F. Perceived stress and risk of adult-onset asthma and other atopic disorders: A longitudinal cohort study. Allergy 2012, 67, 1408–1414. [Google Scholar] [CrossRef]
- Gonzalez, J.S.; Peyrot, M.; McCarl, L.A.; Collins, E.M.; Serpa, L.; Mimiaga, M.J.; Safren, S.A. Depression and diabetes treatment nonadherence: A meta-analysis. Diabetes Care 2008, 31, 2398–2403. [Google Scholar] [CrossRef] [PubMed]
- Mezuk, B.; Eaton, W.W.; Albrecht, S.; Golden, S.H. Depression and type 2 diabetes over the lifespan: A meta-analysis. Diabetes Care 2008, 31, 2383–2390. [Google Scholar] [CrossRef] [PubMed]
- Demakakos, P.; Zaninotto, P.; Nouwen, A. Is the association between depressive symptoms and glucose metabolism bidirectional? Evidence from the English longitudinal study of ageing (ELSA). Psychosom. Med. 2014, 76, 555. [Google Scholar] [CrossRef] [PubMed]
- Rotella, F.; Mannucci, E. Depression as a risk factor for diabetes: A meta-analysis of longitudinal studies. J. Clin. Psychiatry 2013, 74, 31–37. [Google Scholar] [CrossRef] [PubMed]
- Golden, S.H.; Williams, J.E.; Ford, D.E.; Yeh, H.-C.; Sanford, C.P.; Nieto, F.J.; Brancati, F.L. Anger temperament is modestly associated with the risk of type 2 diabetes mellitus: The atheroslcerosis risk in communities study. Psychoneuroendocrinology 2006, 31, 325–332. [Google Scholar] [CrossRef]
- Abraham, S.; Shah, N.G.; Roux, A.D.; Hill-Briggs, F.; Seeman, T.; Szklo, M.; Schreiner, P.J.; Golden, S.H. Trait anger but not anxiety predicts incident type 2 diabetes: The multi-ethnic study of atherosclerosis (MESA). Psychoneuroendocrinology 2015, 60, 105–113. [Google Scholar] [CrossRef]
- Yi, J.P.; Vitaliano, P.P.; Smith, R.E.; Yi, J.C.; Weinger, K. The role of resilience on psychological adjustment and physical health in patients with diabetes. Br. J. Health Psychol. 2008, 13, 311–325. [Google Scholar] [CrossRef]
- Tsenkova, V.K.; Love, G.D.; Singer, B.H.; Ryff, C.D. Socioeconomic status and psychological well-being predict cross-time change in glycosylated hemoglobin in older women without diabetes. Psychosom. Med. 2007, 69, 777–784. [Google Scholar] [CrossRef]
- Gong, S.; Miao, Y.-L.; Jiao, G.-Z.; Sun, M.-J.; Li, H.; Lin, J.; Luo, M.-J.; Tan, J.-H. Dynamics and correlation of serum cortisol and corticosterone under different physiological or stressful conditions in mice. PLoS ONE 2015, 10, e0117503. [Google Scholar] [CrossRef]
- Karatsoreos, I.N.; Bhagat, S.M.; Bowles, N.P.; Weil, Z.M.; Pfaff, D.W.; McEwen, B.S. Endocrine and physiological changes in response to chronic corticosterone: A potential model of the metabolic syndrome in mouse. Endocrinology 2010, 151, 2117–2127. [Google Scholar] [CrossRef]
- Fransson, L.; Franzén, S.; Rosengren, V.; Wolbert, P.; Sjöholm, Å.; Ortsäter, H. b-cell adaptation in a mouse model of glucocorticoid-induced metabolic syndrome. J. Endocrinol. 2013, 219, 231–241. [Google Scholar] [CrossRef] [PubMed]
- McEwen, B.S. Protective and damaging effects of stress mediators: Central role of the brain. Dialogues Clin. Neurosci. 2006, 8, 367. [Google Scholar] [PubMed]
- Di Dalmazi, G.; Pagotto, U.; Pasquali, R.; Vicennati, V. Glucocorticoids and type 2 diabetes: From physiology to pathology. J. Nutr. Metab. 2012, 2012, 525093. [Google Scholar] [CrossRef] [PubMed]
- Newell-Price, J.; Bertagna, X.; Grossman, A.B.; Nieman, L.K. Cushing’s syndrome. Lancet 2006, 367, 1605–1617. [Google Scholar] [CrossRef]
- Clore, J.; Thurby-Hay, L. Glucocorticoid-induced hyperglycemia. Endocr. Pract. 2009, 15, 469–474. [Google Scholar] [CrossRef]
- Hackett, R.A.; Kivimäki, M.; Kumari, M.; Steptoe, A. Diurnal cortisol patterns, future diabetes, and impaired glucose metabolism in the Whitehall II cohort study. J. Clin. Endocrinol. Metab. 2016, 101, 619–625. [Google Scholar] [CrossRef]
- Atlas, D.; International Diabetes Federation. IDF Diabetes Atlas, 7th ed.; International Diabetes Federation: Brussels, Belgium, 2015. [Google Scholar]
- Emdin, C.A.; Anderson, S.G.; Woodward, M.; Rahimi, K. Usual blood pressure and risk of new-onset diabetes: Evidence from 4.1 million adults and a meta-analysis of prospective studies. J. Am. College Cardiol. 2015, 66, 1552–1562. [Google Scholar] [CrossRef]
- Aune, D.; ó Hartaigh, B.; Vatten, L.J. Resting heart rate and the risk of type 2 diabetes: A systematic review and dose–response meta-analysis of cohort studies. Nutr. Metab. Cardiovasc. Dis. 2015, 25, 526–534. [Google Scholar] [CrossRef]
- Licht, C.M.; Vreeburg, S.A.; van Reedt Dortland, A.K.; Giltay, E.J.; Hoogendijk, W.J.; DeRijk, R.H.; Vogelzangs, N.; Zitman, F.G.; de Geus, E.J.; Penninx, B.W.; et al. Increased sympathetic and decreased parasympathetic activity rather than changes in hypothalamic-pituitary-adrenal axis activity is associated with metabolic abnormalities. J. Clin. Endocrinol. Metab. 2010, 95, 2458–2466. [Google Scholar] [CrossRef]
- Thayer, J.F.; Sternberg, E. Beyond heart rate variability: Vagal regulation of allostatic systems. Ann. N. Y. Acad. Sci. 2006, 1088, 361–372. [Google Scholar] [CrossRef]
- Donath, M.Y.; Shoelson, S.E. Type 2 diabetes as an inflammatory disease. Nat. Rev. Immunol. 2011, 11, 98–107. [Google Scholar] [CrossRef] [PubMed]
- Grossmann, V.; Schmitt, V.H.; Zeller, T.; Panova-Noeva, M.; Schulz, A.; Laubert-Reh, D.; Juenger, C.; Schnabel, R.B.; Abt, T.G.; Laskowski, R. Profile of the immune and inflammatory response in individuals with prediabetes and type 2 diabetes. Diabetes Care 2015, 38, 1356–1364. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Bao, W.; Liu, J.; Ouyang, Y.Y.; Wang, D.; Rong, S.; Xiao, X.; Shan, Z.L.; Zhang, Y.; Yao, P.; et al. Inflammatory markers and risk of type 2 diabetes: A systematic review and meta-analysis. Diabetes Care 2013, 36, 166–175. [Google Scholar] [CrossRef] [PubMed]
- Sieu, N.; Katon, W.; Lin, E.H.; Russo, J.; Ludman, E.; Ciechanowski, P. Depression and incident diabetic retinopathy: A prospective cohort study. General Hosp. Psychiatry 2011, 33, 429–435. [Google Scholar] [CrossRef] [PubMed]
- Iversen, M.M.; Tell, G.S.; Espehaug, B.; Midthjell, K.; Graue, M.; Rokne, B.; Berge, L.I.; Østbye, T. Is depression a risk factor for diabetic foot ulcers? 11-years follow-up of the Nord-Trøndelag Health Study (HUNT). J. Diabetes Complicat. 2015, 29, 20–25. [Google Scholar] [CrossRef]
- Novak, M.; Mucsi, I.; Rhee, C.M.; Streja, E.; Lu, J.L.; Kalantar-Zadeh, K.; Molnar, M.Z.; Kovesdy, C.P. Increased risk of incident chronic kidney disease, cardiovascular disease, and mortality in patients with diabetes with comorbid depression. Diabetes Care 2016, 39, 1940–1947. [Google Scholar] [CrossRef]
- Scherrer, J.F.; Garfield, L.D.; Chrusciel, T.; Hauptman, P.J.; Carney, R.M.; Freedland, K.E.; Owen, R.; True, W.R.; Lustman, P.J. Increased risk of myocardial infarction in depressed patients with type 2 diabetes. Diabetes Care 2011, 34, 1729–1734. [Google Scholar] [CrossRef]
- Lin, E.H.; Rutter, C.M.; Katon, W.; Heckbert, S.R.; Ciechanowski, P.; Oliver, M.M.; Ludman, E.J.; Young, B.A.; Williams, L.H.; McCulloch, D.K. Depression and advanced complications of diabetes: A prospective cohort study. Diabetes Care 2010, 33, 264–269. [Google Scholar] [CrossRef]
- Park, M.; Katon, W.J.; Wolf, F.M. Depression and risk of mortality in individuals with diabetes: A meta-analysis and systematic review. General Hosp. Psychiatry 2013, 35, 217–225. [Google Scholar] [CrossRef]
- Agardh, E.; Lundstig, A.; Perfilyev, A.; Volkov, P.; Freiburghaus, T.; Lindholm, E.; Ronn, T.; Agardh, C.D.; Ling, C. Genome-wide analysis of DNA methylation in subjects with type 1 diabetes identifies epigenetic modifications associated with proliferative diabetic retinopathy. BMC Med. 2015, 13, 182. [Google Scholar] [CrossRef]
- Bell, C.G.; Finer, S.; Lindgren, C.M.; Wilson, G.A.; Rakyan, V.K.; Teschendorff, A.E.; Akan, P.; Stupka, E.; Down, T.A.; Prokopenko, I.; et al. Integrated genetic and epigenetic analysis identifies haplotype-specific methylation in the FTO type 2 diabetes and obesity susceptibility locus. PLoS ONE 2010, 5, e14040. [Google Scholar] [CrossRef] [PubMed]
- Nakatochi, M.; Ichihara, S.; Yamamoto, K.; Naruse, K.; Yokota, S.; Asano, H.; Matsubara, T.; Yokota, M. Epigenome-wide association of myocardial infarction with DNA methylation sites at loci related to cardiovascular disease. Clin. Epigenet. 2017, 9, 54. [Google Scholar] [CrossRef] [PubMed]
- Spazzafumo, L.; Olivieri, F.; Abbatecola, A.M.; Castellani, G.; Monti, D.; Lisa, R.; Galeazzi, R.; Sirolla, C.; Testa, R.; Ostan, R.; et al. Remodelling of biological parameters during human ageing: Evidence for complex regulation in longevity and in type 2 diabetes. Age (Dordr) 2013, 35, 419–429. [Google Scholar] [CrossRef] [PubMed]
- Geesaman, B.J. Genetics of aging: Implications for drug discovery and development. Am. J. Clin. Nutr. 2006, 83, 466S–469S. [Google Scholar] [CrossRef]
- Fraga, M.F.; Ballestar, E.; Paz, M.F.; Ropero, S.; Setien, F.; Ballestar, M.L.; Heine-Suner, D.; Cigudosa, J.C.; Urioste, M.; Benitez, J.; et al. Epigenetic differences arise during the lifetime of monozygotic twins. Proc. Natl. Acad. Sci. USA 2005, 102, 10604–10609. [Google Scholar] [CrossRef]
- Bysani, M.; Perfilyev, A.; de Mello, V.D.; Ronn, T.; Nilsson, E.; Pihlajamaki, J.; Ling, C. Epigenetic alterations in blood mirror age-associated DNA methylation and gene expression changes in human liver. Epigenomics 2017, 9, 105–122. [Google Scholar] [CrossRef]
- Wu, Y.; Ding, Y.; Tanaka, Y.; Zhang, W. Risk factors contributing to type 2 diabetes and recent advances in the treatment and prevention. Int. J. Med. Sci. 2014, 11, 1185–1200. [Google Scholar] [CrossRef]
- Bailey, D.P.; Hewson, D.J.; Champion, R.B.; Sayegh, S.M. Sitting time and risk of cardiovascular disease and diabetes: A systematic review and meta-analysis. Am. J. Prev. Med. 2019, 57, 408–416. [Google Scholar] [CrossRef]
- McEwen, B.S.; Stellar, E. Stress and the individual: Mechanisms leading to disease. Arch. Intern. Med. 1993, 153, 2093–2101. [Google Scholar] [CrossRef]
- Bhattacharyya, K.K.; Hueluer, G.; Meng, H.; Hyer, K. Mind-body practices in U.S. adults: Prevalence and correlates. Complement. Ther. Med. 2020, 52, 102501. [Google Scholar] [CrossRef]
- Wahbeh, H.; Elsas, S.M.; Oken, B.S. Mind-body interventions: Applications in neurology. Neurology 2008, 70, 2321–2328. [Google Scholar] [CrossRef] [PubMed]
- Black, D.S.; Slavich, G.M. Mindfulness meditation and the immune system: A systematic review of randomized controlled trials. Ann. N. Y. Acad. Sci. 2016, 1373, 13–24. [Google Scholar] [CrossRef] [PubMed]
- Wallace, R.K. Physiological effects of transcendental meditation. Science 1970, 167, 1751–1754. [Google Scholar] [CrossRef] [PubMed]
- Benson, H.; Beary, J.F.; Carol, M.P. The relaxation response. Psychiatry 1974, 37, 37–46. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, D.Z. Zen Koan as a Means of Attaining Enlightenment; Tuttle Publishing: North Clarendon, VT, USA, 2011. [Google Scholar]
- Creswell, J.D. Mindfulness Interventions. Annu. Rev. Psychol. 2017, 68, 491–516. [Google Scholar] [CrossRef] [PubMed]
- Feuerstein, G. The Yoga Tradition: Its History, Literature, Philosophy and Practice; SCB Distributors: Gardena, CA, USA, 2012. [Google Scholar]
- Kabat-Zinn, J. Mindfulness-based stress reduction (MBSR). Constr. Hum. Sci. 2003, 8, 73–107. [Google Scholar]
- Bower, J.E.; Crosswell, A.D.; Stanton, A.L.; Crespi, C.M.; Winston, D.; Arevalo, J.; Ma, J.; Cole, S.W.; Ganz, P.A. Mindfulness meditation for younger breast cancer survivors: A randomized controlled trial. Cancer 2015, 121, 1231–1240. [Google Scholar] [CrossRef]
- Robert-McComb, J.J.; Cisneros, A.; Tacón, A.; Panike, R.; Norman, R.; Qian, X.P.; McGlone, J. The effects of mindfulness-based movement on parameters of stress. Int. J. Yoga Ther. 2015, 25, 79–88. [Google Scholar] [CrossRef]
- Prakhinkit, S.; Suppapitiporn, S.; Tanaka, H.; Suksom, D. Effects of Buddhism walking meditation on depression, functional fitness, and endothelium-dependent vasodilation in depressed elderly. J. Altern. Complement. Med. 2014, 20, 411–416. [Google Scholar] [CrossRef]
- Lee, I. The Power Brain: Five Steps to Upgrading Your Brain Operating System; Best Life Media: Gilbert, AZ, USA, 2016. [Google Scholar]
- Lee, I. Brain Wave Vibration: Getting Back into the Rhythm of a Happy, Healthy Life; Best Life Media: Gilbert, AZ, USA, 2009. [Google Scholar]
- Abbott, R.; Lavretsky, H. Tai Chi and Qigong for the treatment and prevention of mental disorders. Psychiatr. Clinics N. Am. 2013, 36, 109–119. [Google Scholar] [CrossRef]
- Chételat, G.; Lutz, A.; Arenaza-Urquijo, E.; Collette, F.; Klimecki, O.; Marchant, N. Why could meditation practice help promote mental health and well-being in aging? Alzheimer’s Res. Ther. 2018, 10, 57. [Google Scholar] [CrossRef] [PubMed]
- Ni, Y.; Ma, L.; Li, J. Effects of mindfulness-based stress reduction and mindfulness-based cognitive therapy in people with diabetes: A systematic review and meta-analysis. J. Nurs. Scholarsh. 2020, 52, 379–388. [Google Scholar] [CrossRef] [PubMed]
- Ni, W.J.; Zhou, H.; Ding, H.H.; Tang, L.Q. Berberine ameliorates renal impairment and inhibits podocyte dysfunction by targeting the phosphatidylinositol 3-kinase-protein kinase B pathway in diabetic rats. J. Diabetes Investig. 2020, 11, 297–306. [Google Scholar] [CrossRef]
- Meng, D.; Chunyan, W.; Xiaosheng, D.; Xiangren, Y. The effects of Qigong on type 2 diabetes mellitus: A systematic review and meta-analysis. Evid. Based Complement. Altern. Med. eCAM 2018, 2018, 8182938. [Google Scholar] [CrossRef] [PubMed]
- Xia, T.W.; Yang, Y.; Li, W.H.; Tang, Z.H.; Li, Z.R.; Qiao, L.J. Different training durations and styles of tai chi for glucose control in patients with type 2 diabetes: A systematic review and meta-analysis of controlled trials. BMC Complement. Altern. Med. 2019, 19, 63. [Google Scholar] [CrossRef]
- Chao, M.; Wang, C.; Dong, X.; Ding, M. The effects of tai chi on type 2 diabetes mellitus: A meta-analysis. J. Diabetes Res. 2018, 2018, 7350567. [Google Scholar] [CrossRef]
- Pascoe, M.C.; Thompson, D.R.; Ski, C.F. Yoga, mindfulness-based stress reduction and stress-related physiological measures: A meta-analysis. Psychoneuroendocrinology 2017, 86, 152–168. [Google Scholar] [CrossRef]
- Thind, H.; Lantini, R.; Balletto, B.L.; Donahue, M.L.; Salmoirago-Blotcher, E.; Bock, B.C.; Scott-Sheldon, L.A.J. The effects of yoga among adults with type 2 diabetes: A systematic review and meta-analysis. Prev. Med. 2017, 105, 116–126. [Google Scholar] [CrossRef]
- Cui, J.; Yan, J.H.; Yan, L.M.; Pan, L.; Le, J.J.; Guo, Y.Z. Effects of yoga in adults with type 2 diabetes mellitus: A meta-analysis. J. Diabetes Investig. 2017, 8, 201–209. [Google Scholar] [CrossRef]
- Kumar, V.; Jagannathan, A.; Philip, M.; Thulasi, A.; Angadi, P.; Raghuram, N. Role of yoga for patients with type II diabetes mellitus: A systematic review and meta-analysis. Complement. Ther. Med. 2016, 25, 104–112. [Google Scholar] [CrossRef]
- Li, C.; Xu, D.; Hu, M.; Tan, Y.; Zhang, P.; Li, G.; Chen, L. A systematic review and meta-analysis of randomized controlled trials of cognitive behavior therapy for patients with diabetes and depression. J. Psychosom. Res. 2017, 95, 44–54. [Google Scholar] [CrossRef] [PubMed]
- Uchendu, C.; Blake, H. Effectiveness of cognitive-behavioural therapy on glycaemic control and psychological outcomes in adults with diabetes mellitus: A systematic review and meta-analysis of randomized controlled trials. Diabet. Med. 2017, 34, 328–339. [Google Scholar] [CrossRef] [PubMed]
- Liubaoerjijin, Y.; Terada, T.; Fletcher, K.; Boule, N.G. Effect of aerobic exercise intensity on glycemic control in type 2 diabetes: A meta-analysis of head-to-head randomized trials. Acta Diabetol. 2016, 53, 769–781. [Google Scholar] [CrossRef] [PubMed]
- Boule, N.G.; Haddad, E.; Kenny, G.P.; Wells, G.A.; Sigal, R.J. Effects of exercise on glycemic control and body mass in type 2 diabetes mellitus: A meta-analysis of controlled clinical trials. JAMA 2001, 286, 1218–1227. [Google Scholar] [CrossRef] [PubMed]
- Lora-Pozo, I.; Lucena-Anton, D.; Salazar, A.; Galan-Mercant, A.; Moral-Munoz, J.A. Anthropometric, cardiopulmonary and metabolic benefits of the high-intensity interval training versus moderate, low-intensity or control for type 2 diabetes: Systematic review and meta-analysis. Int. J. Environ. Res. Public Health 2019, 16, 4524. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Miller, Y.D.; Burton, N.W.; Brown, W.J. A preliminary study of the effects of Tai Chi and Qigong medical exercise on indicators of metabolic syndrome, glycaemic control, health-related quality of life, and psychological health in adults with elevated blood glucose. Br. J. Sports Med. 2010, 44, 704–709. [Google Scholar] [CrossRef]
- Xia, T.; Yang, Y.; Li, W.; Tang, Z.; Huang, Q.; Li, Z.; Guo, Y. Meditative movements for patients with type 2 diabetes: A systematic review and meta-analysis. Evid. Based Complement. Altern. Med. eCAM 2020, 2020, 5745013. [Google Scholar] [CrossRef]
- Yang, H.; Wu, X.; Wang, M. Effect of conventional medical treatment plus Qigong exercise on type 2 diabetes mellitus in Chinese patients: A meta-analysis. J. Tradit. Chin. Med. Chung i Tsa Chih Ying Wen Pan 2018, 38, 167–174. [Google Scholar]
- Harkness, E.; Macdonald, W.; Valderas, J.; Coventry, P.; Gask, L.; Bower, P. Identifying psychosocial interventions that improve both physical and mental health in patients with diabetes: A systematic review and meta-analysis. Diabetes Care 2010, 33, 926–930. [Google Scholar] [CrossRef]
- Ismail, K.; Winkley, K.; Rabe-Hesketh, S. Systematic review and meta-analysis of randomised controlled trials of psychological interventions to improve glycaemic control in patients with type 2 diabetes. Lancet (Lond. Engl.) 2004, 363, 1589–1597. [Google Scholar] [CrossRef]
- Gainey, A.; Himathongkam, T.; Tanaka, H.; Suksom, D. Effects of Buddhist walking meditation on glycemic control and vascular function in patients with type 2 diabetes. Complement. Ther. Med. 2016, 26, 92–97. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H.; Hwang, S.M.; Kang, D.H.; Yang, H.J. Brain education-based meditation for patients with hypertension and/or type 2 diabetes: A pilot randomized controlled trial. Medicine 2019, 98, e15574. [Google Scholar] [CrossRef] [PubMed]
- Sung, M.K.; Lee, U.S.; Ha, N.H.; Koh, E.; Yang, H.J. A potential association of meditation with menopausal symptoms and blood chemistry in healthy women: A pilot cross-sectional study. Medicine 2020, 99, e22048. [Google Scholar] [CrossRef] [PubMed]
- Moritz, S.; Göritz, A.S.; Schmotz, S.; Weierstall-Pust, R.; Gehlenborg, J.; Gallinat, J.; Kühn, S. Imaginal retraining decreases craving for high-calorie food in overweight and obese women: A randomized controlled trial. Transl. Psychiatry 2019, 9, 319. [Google Scholar] [CrossRef] [PubMed]
- Melo, K.C.B.; Araújo, F.S.; Cordeiro Júnior, C.C.M.; de Andrade, K.T.P.; Moreira, S.R. Pilates method training: Functional and blood glucose responses of older women with type 2 diabetes. J. Strength Cond. Res. 2020, 34, 1001–1007. [Google Scholar] [CrossRef] [PubMed]
- Gapp, K.; Bohacek, J.; Grossmann, J.; Brunner, A.M.; Manuella, F.; Nanni, P.; Mansuy, I.M. Potential of environmental enrichment to prevent transgenerational effects of paternal trauma. Neuropsychopharmacol. Off. Publ. Am. Coll. Neuropsychopharmacol. 2016, 41, 2749–2758. [Google Scholar] [CrossRef]
- McCreary, J.K.; Erickson, Z.T.; Hao, Y.; Ilnytskyy, Y.; Kovalchuk, I.; Metz, G.A. Environmental intervention as a therapy for adverse programming by ancestral stress. Sci. Rep. 2016, 6, 37814. [Google Scholar] [CrossRef]
- Venditti, S.; Verdone, L.; Reale, A.; Vetriani, V.; Caserta, M.; Zampieri, M. Molecules of silence: Effects of meditation on gene expression and epigenetics. Front. Psychol. 2020, 11, 1767. [Google Scholar] [CrossRef]
- Chaix, R.; Fagny, M.; Cosin-Tomás, M.; Alvarez-López, M.; Lemee, L.; Regnault, B.; Davidson, R.J.; Lutz, A.; Kaliman, P. Differential DNA methylation in experienced meditators after an intensive day of mindfulness-based practice: Implications for immune-related pathways. Brain Behav. Immun. 2020, 84, 36–44. [Google Scholar] [CrossRef]
- Kaliman, P.; Alvarez-López, M.J.; Cosín-Tomás, M.; Rosenkranz, M.A.; Lutz, A.; Davidson, R.J. Rapid changes in histone deacetylases and inflammatory gene expression in expert meditators. Psychoneuroendocrinology 2014, 40, 96–107. [Google Scholar] [CrossRef]
- Chaix, R.; Alvarez-López, M.J.; Fagny, M.; Lemee, L.; Regnault, B.; Davidson, R.J.; Lutz, A.; Kaliman, P. Epigenetic clock analysis in long-term meditators. Psychoneuroendocrinology 2017, 85, 210–214. [Google Scholar] [CrossRef] [PubMed]
- García-Campayo, J.; Puebla-Guedea, M.; Labarga, A.; Urdánoz, A.; Roldán, M.; Pulido, L.; de Morentin, X.M.; Perdones-Montero, Á.; Montero-Marín, J.; Mendioroz, M. Epigenetic response to mindfulness in peripheral blood leukocytes involves genes linked to common human diseases. Mindfulness 2018, 9, 1146–1159. [Google Scholar] [CrossRef]
- Mendioroz, M.; Puebla-Guedea, M.; Montero-Marín, J.; Urdánoz-Casado, A.; Blanco-Luquin, I.; Roldán, M.; Labarga, A.; García-Campayo, J. Telomere length correlates with subtelomeric DNA methylation in long-term mindfulness practitioners. Sci. Rep. 2020, 10, 4564. [Google Scholar] [CrossRef] [PubMed]
- Bishop, J.R.; Lee, A.M.; Mills, L.J.; Thuras, P.D.; Eum, S.; Clancy, D.; Erbes, C.R.; Polusny, M.A.; Lamberty, G.J.; Lim, K.O. Methylation of FKBP5 and SLC6A4 in relation to treatment response to mindfulness based stress reduction for posttraumatic stress disorder. Front. Psychiatry 2018, 9, 418. [Google Scholar] [CrossRef] [PubMed]
- Harkess, K.N.; Ryan, J.; Delfabbro, P.H.; Cohen-Woods, S. Preliminary indications of the effect of a brief yoga intervention on markers of inflammation and DNA methylation in chronically stressed women. Transl. Psychiatry 2016, 6, e965. [Google Scholar] [CrossRef]
- Ren, H.; Collins, V.; Clarke, S.J.; Han, J.S.; Lam, P.; Clay, F.; Williamson, L.M.; Andy Choo, K.H. Epigenetic changes in response to tai chi practice: A pilot investigation of DNA methylation marks. Evid. Based Complement. Altern. Med. eCAM 2012, 2012, 841810. [Google Scholar] [CrossRef]
- Elsenbruch, S.; Langhorst, J.; Popkirowa, K.; Muller, T.; Luedtke, R.; Franken, U.; Paul, A.; Spahn, G.; Michalsen, A.; Janssen, O.E.; et al. Effects of mind-body therapy on quality of life and neuroendocrine and cellular immune functions in patients with ulcerative colitis. Psychother. Psychosom. 2005, 74, 277–287. [Google Scholar] [CrossRef]
- Rosenkranz, M.A.; Davidson, R.J.; Maccoon, D.G.; Sheridan, J.F.; Kalin, N.H.; Lutz, A. A comparison of mindfulness-based stress reduction and an active control in modulation of neurogenic inflammation. Brain Behav. Immun. 2013, 27, 174–184. [Google Scholar] [CrossRef]
- Fritsch, K.; Finke, J.; Grüllich, C. Suppression of granzyme B activity and caspase-3 activation in leukaemia cells constitutively expressing the protease inhibitor 9. Ann. Hematol. 2013, 92, 1603–1609. [Google Scholar] [CrossRef]
- Spinelli, C.; Wisener, M.; Khoury, B. Mindfulness training for healthcare professionals and trainees: A meta-analysis of randomized controlled trials. J. Psychosom. Res. 2019, 120, 29–38. [Google Scholar] [CrossRef]
- Ng, Q.X.; Venkatanarayanan, N.; Loke, W.; Yeo, W.S.; Lim, D.Y.; Chan, H.W.; Sim, W.S. A meta-analysis of the effectiveness of yoga-based interventions for maternal depression during pregnancy. Complement. Ther. Clin. Pract. 2019, 34, 8–12. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Y.; Xie, X.; Cheng, A.S.K. Qigong or tai chi in cancer care: An updated systematic review and meta-analysis. Curr. Oncol. Rep. 2019, 21, 48. [Google Scholar] [CrossRef] [PubMed]
- Fox, K.C.; Nijeboer, S.; Dixon, M.L.; Floman, J.L.; Ellamil, M.; Rumak, S.P.; Sedlmeier, P.; Christoff, K. Is meditation associated with altered brain structure? A systematic review and meta-analysis of morphometric neuroimaging in meditation practitioners. Neurosci. Biobehav. Rev. 2014, 43, 48–73. [Google Scholar] [CrossRef] [PubMed]
- So, W.W.Y.; Cai, S.; Yau, S.Y.; Tsang, H.W.H. The neurophysiological and psychological mechanisms of Qigong as a treatment for depression: A systematic review and meta-analysis. Front. Psychiatry 2019, 10, 820. [Google Scholar] [CrossRef]
- Gathright, E.C.; Salmoirago-Blotcher, E.; DeCosta, J.; Balletto, B.L.; Donahue, M.L.; Feulner, M.M.; Cruess, D.G.; Wing, R.R.; Carey, M.P.; Scott-Sheldon, L.A.J. The impact of transcendental meditation on depressive symptoms and blood pressure in adults with cardiovascular disease: A systematic review and meta-analysis. Complement. Ther. Med. 2019, 46, 172–179. [Google Scholar] [CrossRef]
- Wu, Y.; Johnson, B.T.; Acabchuk, R.L.; Chen, S.; Lewis, H.K.; Livingston, J.; Park, C.L.; Pescatello, L.S. Yoga as antihypertensive lifestyle therapy: A systematic review and meta-analysis. Mayo Clin. Proc. 2019, 94, 432–446. [Google Scholar] [CrossRef]
- Park, S.H.; Han, K.S. Blood pressure response to meditation and yoga: A systematic review and meta-analysis. J. Altern. Complement. Med. 2017, 23, 685–695. [Google Scholar] [CrossRef]
- Morgan, N.; Irwin, M.R.; Chung, M.; Wang, C. The effects of mind-body therapies on the immune system: Meta-analysis. PLoS ONE 2014, 9, e100903. [Google Scholar] [CrossRef]
- Creswell, J.D.; Irwin, M.R.; Burklund, L.J.; Lieberman, M.D.; Arevalo, J.M.; Ma, J.; Breen, E.C.; Cole, S.W. Mindfulness-based stress reduction training reduces loneliness and pro-inflammatory gene expression in older adults: A small randomized controlled trial. Brain Behav. Immun. 2012, 26, 1095–1101. [Google Scholar] [CrossRef]
- Black, D.S.; Cole, S.W.; Irwin, M.R.; Breen, E.; St Cyr, N.M.; Nazarian, N.; Khalsa, D.S.; Lavretsky, H. Yogic meditation reverses NF-kappaB and IRF-related transcriptome dynamics in leukocytes of family dementia caregivers in a randomized controlled trial. Psychoneuroendocrinology 2013, 38, 348–355. [Google Scholar] [CrossRef]
- Campion, J.; Milagro, F.I.; Goyenechea, E.; Martinez, J.A. TNF-alpha promoter methylation as a predictive biomarker for weight-loss response. Obesity (Silver Spring) 2009, 17, 1293–1297. [Google Scholar] [CrossRef] [PubMed]
- Horvath, S. DNA methylation age of human tissues and cell types. Genome Biol. 2013, 14, R115. [Google Scholar] [CrossRef] [PubMed]
- Hannum, G.; Guinney, J.; Zhao, L.; Zhang, L.; Hughes, G.; Sadda, S.; Klotzle, B.; Bibikova, M.; Fan, J.B.; Gao, Y.; et al. Genome-wide methylation profiles reveal quantitative views of human aging rates. Mol. Cell 2013, 49, 359–367. [Google Scholar] [CrossRef] [PubMed]
- Yi, S.W.; Park, S.; Lee, Y.H.; Park, H.J.; Balkau, B.; Yi, J.J. Association between fasting glucose and all-cause mortality according to sex and age: A prospective cohort study. Sci. Rep. 2017, 7, 8194. [Google Scholar] [CrossRef]
- Group, D.S. Age- and sex-specific prevalences of diabetes and impaired glucose regulation in 13 European cohorts. Diabetes Care 2003, 26, 61–69. [Google Scholar] [CrossRef]
- Qiao, Q.; Hu, G.; Tuomilehto, J.; Nakagami, T.; Balkau, B.; Borch-Johnsen, K.; Ramachandran, A.; Mohan, V.; Iyer, S.R.; Tominaga, M.; et al. Age- and sex-specific prevalence of diabetes and impaired glucose regulation in 11 Asian cohorts. Diabetes Care 2003, 26, 1770–1780. [Google Scholar] [CrossRef]
- Rod, N.H.; Grønbaek, M.; Schnohr, P.; Prescott, E.; Kristensen, T.J. Perceived stress as a risk factor for changes in health behaviour and cardiac risk profile: A longitudinal study. J. Intern. Med. 2009, 266, 467–475. [Google Scholar] [CrossRef]
- Zou, L.; Yeung, A.; Li, C.; Wei, G.X.; Chen, K.W.; Kinser, P.A.; Chan, J.S.M.; Ren, Z. Effects of meditative movements on major depressive disorder: A systematic review and meta-analysis of randomized controlled trials. J. Clin. Med. 2018, 7, 195. [Google Scholar] [CrossRef]
- Breedvelt, J.J.F.; Amanvermez, Y.; Harrer, M.; Karyotaki, E.; Gilbody, S.; Bockting, C.L.H.; Cuijpers, P.; Ebert, D.D. The Effects of meditation, yoga, and mindfulness on depression, anxiety, and stress in tertiary education students: A meta-analysis. Front. Psychiatry 2019, 10, 193. [Google Scholar] [CrossRef]
- Lv, J.; Liu, Q.; Zeng, X.; Oei, T.P.S.; Liu, Y.; Xu, K.; Sun, W.; Hou, H.; Liu, J. The effect of four Immeasurables meditations on depressive symptoms: A systematic review and meta-analysis. Clin. Psychol. Rev. 2020, 76, 101814. [Google Scholar] [CrossRef]
- Tricco, A.C.; Ivers, N.M.; Grimshaw, J.M.; Moher, D.; Turner, L.; Galipeau, J.; Halperin, I.; Vachon, B.; Ramsay, T.; Manns, B.; et al. Effectiveness of quality improvement strategies on the management of diabetes: A systematic review and meta-analysis. Lancet (Lond. Engl.) 2012, 379, 2252–2261. [Google Scholar] [CrossRef]
- Norris, S.L.; Nichols, P.J.; Caspersen, C.J.; Glasgow, R.E.; Engelgau, M.M.; Jack, L.; Isham, G.; Snyder, S.R.; Carande-Kulis, V.G.; Garfield, S.; et al. The effectiveness of disease and case management for people with diabetes. A systematic review. Am. J. Prev. Med. 2002, 22, 15–38. [Google Scholar] [CrossRef]
- Naumova, O.Y.; Lee, M.; Koposov, R.; Szyf, M.; Dozier, M.; Grigorenko, E.L. Differential patterns of whole-genome DNA methylation in institutionalized children and children raised by their biological parents. Dev. Psychopathol. 2012, 24, 143–155. [Google Scholar] [CrossRef] [PubMed]
- Quach, A.; Levine, M.E.; Tanaka, T.; Lu, A.T.; Chen, B.H.; Ferrucci, L.; Ritz, B.; Bandinelli, S.; Neuhouser, M.L.; Beasley, J.M.; et al. Epigenetic clock analysis of diet, exercise, education, and lifestyle factors. Aging 2017, 9, 419–446. [Google Scholar] [CrossRef]
- Chien, W.T.; Chow, K.M.; Chong, Y.Y.; Bressington, D.; Choi, K.C.; Chan, C.W.H. The role of five facets of mindfulness in a mindfulness-based psychoeducation intervention for people with recent-onset psychosis on mental and psychosocial health outcomes. Front. Psychiatry 2020, 11, 177. [Google Scholar] [CrossRef]
- Tovote, K.A.; Fleer, J.; Snippe, E.; Peeters, A.C.; Emmelkamp, P.M.; Sanderman, R.; Links, T.P.; Schroevers, M.J. Individual mindfulness-based cognitive therapy and cognitive behavior therapy for treating depressive symptoms in patients with diabetes: Results of a randomized controlled trial. Diabetes Care 2014, 37, 2427–2434. [Google Scholar] [CrossRef]
- Roberts, S.; Keers, R.; Lester, K.J.; Coleman, J.R.; Breen, G.; Arendt, K.; Blatter-Meunier, J.; Cooper, P.; Creswell, C.; Fjermestad, K.; et al. HPA axis related genes and response to psychological therapies: Genetics and epigenetics. Depress. Anxiety 2015, 32, 861–870. [Google Scholar] [CrossRef]
- Brody, G.H.; Yu, T.; Chen, E.; Beach, S.R.; Miller, G.E. Family-centered prevention ameliorates the longitudinal association between risky family processes and epigenetic aging. J. Child Psychol. Psychiatry Allied Discip. 2016, 57, 566–574. [Google Scholar] [CrossRef]
- Perrin, N.E.; Davies, M.J.; Robertson, N.; Snoek, F.J.; Khunti, K. The prevalence of diabetes-specific emotional distress in people with Type 2 diabetes: A systematic review and meta-analysis. Diabet. Med. J. Br. Diabet. Assoc. 2017, 34, 1508–1520. [Google Scholar] [CrossRef]
- Fabre, O.; Ingerslev, L.R.; Garde, C.; Donkin, I.; Simar, D.; Barres, R. Exercise training alters the genomic response to acute exercise in human adipose tissue. Epigenomics 2018, 10, 1033–1050. [Google Scholar] [CrossRef]
- Nitert, M.D.; Dayeh, T.; Volkov, P.; Elgzyri, T.; Hall, E.; Nilsson, E.; Yang, B.T.; Lang, S.; Parikh, H.; Wessman, Y.; et al. Impact of an exercise intervention on DNA methylation in skeletal muscle from first-degree relatives of patients with type 2 diabetes. Diabetes 2012, 61, 3322–3332. [Google Scholar] [CrossRef] [PubMed]
- Steinhubl, S.R.; Wineinger, N.E.; Patel, S.; Boeldt, D.L.; Mackellar, G.; Porter, V.; Redmond, J.T.; Muse, E.D.; Nicholson, L.; Chopra, D.; et al. Cardiovascular and nervous system changes during meditation. Front. Hum. Neurosci. 2015, 9, 145. [Google Scholar] [CrossRef] [PubMed]
- Manchanda, S.C.; Madan, K. Yoga and meditation in cardiovascular disease. Clin. Res. Cardiol. 2014, 103, 675–680. [Google Scholar] [CrossRef] [PubMed]
- Denham, J.; O’Brien, B.J.; Harvey, J.T.; Charchar, F.J. Genome-wide sperm DNA methylation changes after 3 months of exercise training in humans. Epigenomics 2015, 7, 717–731. [Google Scholar] [CrossRef] [PubMed]
| Meta-Analysis | Study Type | Participants | Intervention | Duration, Frequency, Intervention Length | Control | Number of Trials (Number of Participants) | Outcomes (Compared to the Control) | |
|---|---|---|---|---|---|---|---|---|
| Ni et al., 2020 (Journal of Nursing Scholarship) [87] | RCT | Patients with types 1 and type 2 diabetes | Mind–body intervention | Mindfulness-based intervention (MBSR, MBCT) | 90–150 min/session, 1 session/week, 8–9 weeks | Usual care, waitlist, health education without any mindful component | 6 studies (n = 578) | ↓HbA1c |
| Ni et al., 2020 (J Diabetes Investing) [88] | RCT | Patients with types 1 and type 2 diabetes | Mindfulness-based intervention (MBSR, mindful eating intervention, MBCT, MBCT + MBSR) | 30–150 min/session, 1–7 sessions/week, 8–12 weeks | Usual care, Smart choices DSME-based intervention, waitlist, CBT | 7 studies (n = 665) | ↓HbA1c | |
| Meng et al., 2018 [89] | RCT | Patients with type 2 diabetes | Qigong | NR, 20–90 min/session, 1–5 sessions/week, 2–12 months | No exercise | 17 studies (n = 966) | ↓FBG | |
| Other aerobic exercises | 5 studies (n = 389) | = FBG (p = 0.07) | ||||||
| Antiresistance exercise | 1 study (n = 22) | ↓FBG | ||||||
| No exercise | 16 studies (n = 834) | ↓HbA1c | ||||||
| Other aerobic exercises | 5 studies (n = 389) | ↓HbA1c | ||||||
| Antiresistance exercise | 1 study (n = 22) | = HbA1c | ||||||
| No exercise | 6 studies (n = 330) | ↓PPBG | ||||||
| Other aerobic exercises | 1 study (n = 122) | ↓PPBG | ||||||
| Xia et al., 2019 [90] | RCT | Patients with type 2 diabetes | Tai chi | 30–120 min/session, 2–14 sessions/week, 2–6 months | Usual care, standard treatment, any kind of exercise | 13 studies (n = 616) | ↓FBG | |
| 9 studies (n = 517) | ↓HbA1c | |||||||
| 7 studies (n = 343) | ↓TC | |||||||
| 8 studies (n = 359) | ↓TG | |||||||
| 6 studies (n = 290) | = HDL-C | |||||||
| 6 studies (n = 290) | = LDL-C | |||||||
| 6 studies (n = 296) | ↓BMI | |||||||
| Chao et al., 2018 [91] | RCT | Patients with type 2 diabetes | Tai chi | 15–60 min/session, 2–7 sessions/week, 1–6 months | Non-exercise | 10 studies (n = 489) | ↓FBG | |
| Other aerobic exercise | 7 studies (n = 342) | = FBG | ||||||
| Non-exercise | 7 studies (n = 293) | ↓HbA1c | ||||||
| Other aerobic exercise | 7 studies (n = 372) | = HbA1c | ||||||
| Non-exercise | 5 studies (n = 162) | ↓PPBG | ||||||
| Other aerobic exercise | 3 studies (n = 84) | = PPBG | ||||||
| Xia et al., 2020 [87] | RCT | Patients with type 2 diabetes | Meditative movements (tai chi or qigong or yoga) | NR, 10–120 min/session, 2–7 sessions/week, 6–36 weeks | Any type of control group | 19 studies (n = 1505) | ↓FBG | |
| 15 studies (n = 1116) | ↓HbA1c | |||||||
| 5 studies (n = 624) | ↓PPBG | |||||||
| 12 studies (n = 1110) | ↓TC | |||||||
| 8 studies (n = 844) | ↓LDL-C | |||||||
| 10 studies (n = 991) | ↓TG | |||||||
| 9 studies (n = 938) | ↑HDL-C | |||||||
| 11 studies (n = 915) | =BMI | |||||||
| Pascoe et al., 2017 [92] | RCT | All population | Yoga w/wo MBSR | 45–120 min/sessions, 1–7 sessions/week, 2–14 months | Active controls (exercise, physical activity, health education, social support, stretching, progressive muscle relaxation, other counselling/therapy) | 7 studies (n = 534) | ↓FBG | |
| 6 studies (n = 389) | ↓TC | |||||||
| 6 studies (n = 389) | ↓LDL-C | |||||||
| 7 studies (n = 560) | = TG | |||||||
| 7 studies (n = 560) | = HDL-C | |||||||
| Thind et al., 2017 [93] * | RCT, non-RCT | Patients with type 2 diabetes | Yoga (not specified, hatha, Sudarshan kriya) | 50–240 min/session, total 12~182 h (<1–26 weeks) | Usual care, waitlist, exercise only, exercise plus lifestyle education | 18 studies (n = 2212) | ↓HbA1c | |
| 21 studies (n = 2081) | ↓FBG | |||||||
| 14 studies (n = 1473) | ↓PPBG | |||||||
| 16 studies (n = 1880) | ↑HDL-C | |||||||
| 16 studies (n = 1838) | ↓LDL-C | |||||||
| 16 studies (n = 1895) | ↓TC | |||||||
| 14 studies (n = 1790) | ↓TG | |||||||
| 9 studies (n = 1260) | ↓BMI | |||||||
| Cui et al., 2016 [94] | RCT | Patients with type 2 diabetes | Yoga (hatha, asana, pranaya, Sudarshan kriya, shavasana) | 30–120 min/session, 1–7 sessions/week, 15 days–9 months | Usual care, physical exercises, life style education, brisk walking, waitlist | 9 studies (n = 805) | ↓FBG | |
| 7 studies (n = 718) | ↓HbA1c | |||||||
| 4 studies (n = 527) | ↓PPBG | |||||||
| 5 studies (n = 618) | ↓TC | |||||||
| 4 studies (n = 588) | ↑HDL-C | |||||||
| 5 studies (n = 618) | ↓LDL-C | |||||||
| 4 studies (n = 588) | =TG | |||||||
| Kumar et al., 2016 [95] | RCT | Patients with type 2 diabetes | Yoga (asana, pranayama, hatha, relaxation, diaphragmatic breathing in supine position) | 30–120 min/session, 1–7 sessions/week, 40 days–6 months | Usual care, walking, education | 17 studies (n = 1358) | ↓FBG | |
| 9 studies (n = 659) | ↓PPBG | |||||||
| 13 studies (n = 1097) | ↓HbA1c | |||||||
| Li et al., 2017 [96] | RCT | Diabetes patients (type1 or 2) with clinically relevant depression | CBT | CBT or a therapy based on CBT | NR, 45–90 min/sessions, 1–2 sessions/week, 2–12 months | Usual care, diabetes education, usual care, waitlist | 7 studies (n = 759) | = HbA1c |
| 5 studies (n = 303) short-term effect | = HbA1c | |||||||
| 6 studies (n = 705) long-term effect | = HbA1c | |||||||
| 3 studies (n = 175) | ↓FBG | |||||||
| Uchendu et al., 2017 [97] | RCT | Patients with Type 1 or Type 2 diabetes | CBT | 30–120 min/sessions, 1–2 sessions/week, 6–16 weeks | Non-CBT | 16 studies (n = 1375) | ↓HbA1c | |
| Liubaoerjijin et al., 2016 [98] | RCT | Patients with type 2 diabetes | Exercise | Higher intensity training (walking/cycling/running/treadmill/XC ski) | NR, 15–60 min/session, 3–6 sessions/week, 12–25 weeks | Lower intensity training (Walking/Cycling/Treadmill/XC ski) | 8 studies (n = 233) | ↓HbA1c |
| = FBG | ||||||||
| Boule et al., 2001 [99] | RCT, CCT | Patients with type 2 diabetes | Exercise | 40–90 min/sessions, 2–6 sessions/week, 8–22 weeks | Non-exercise | 11 studies (n = 310) | ↓HbA1c | |
| Exercise with diet | 30–45 min/session, 3–3.5 sessions/week, 13–52 weeks | Non-exercise, Non-diet | 3 studies (n = 142) | ↓HbA1c | ||||
| Lora–Pozo et al., 2019 [100] | RCT | Patients with type 2 diabetes | High-intensity interval training (64–90% VO2max or 77–95% heart rate max) | 21–60 min/session, 3–5 sessions/week, 12–16 weeks | Non-exercise | 2 studies (n = 43) | ↓HbA1c | |
| 30–60 min/session, 2–5 sessions/week, 12–16 weeks | Moderate-intensity training | 4 studies (n = 105) | = HbA1c | |||||
| 60–83 min/session, 2–5 sessions/week, 16–48 weeks | Low-intensity training | 2 studies (n = 312) | ↓HbA1c |
| Ref | Study Type | Intervention | Control | Sample | Changes in Epigenetic Marks | Differentially Methylated Area | Related Functions | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Participants | Protocol | Duration | Participants | Protocol | Duration | ||||||
| Kaliman et al., 2014 [115] | L | Experienced meditators (n = 19) (a daily meditation practice spanning a minimum of 3 years, ≥30 min/day, ≥3 intensive retreats lasting 5 or more days) | Intensive meditation practice (a day-long session of the MBSR, which is routinely used in North-American hospitals) | 8 h | People with no meditation experience (n = 21) | Leisure activity (reading, watching documentaries or playing computer games, and walking) | 8 h | PBMC |
| ||
| Chaix et al., 2020 [114] | Experienced meditators (n = 17) (Same participant pools of Kaliman et al., 2014) | People with no meditation experience (n = 17) (Same participant pools of Kaliman et al., 2014.) |
| ACADM, CPT1A, HSD17B4 | Fatty acid metabolism | ||||||
| SAP18, EIF1B, NCBP2 | RNA transport | ||||||||||
| APITD1, ERCC1 | DNA repair | ||||||||||
| KLF15 | Glucose homeostasis, stress response, inflammation | ||||||||||
| EGR1 | DNA damage, immunity, inflammatory responses | ||||||||||
| SP3 | DNA damage, immunity, hematopoiesis, expression regulation of anti-inflammatory molecules such as IL-10 and COX-2 | ||||||||||
| SP4 | Inflammatory and neuropathic persistent pain states, dendrite patterning, neurotransmission | ||||||||||
| EGR2 | Involved in immunity and inflammatory processes, as well as various neuropathies | ||||||||||
| Chaix et al., 2017 [116] | C | Experienced meditators (n = 17) (Same participant pools of Kaliman et al., 2014) | - | - | People with no meditation experience (n = 17) (Same participant pools of Kaliman et al., 2014) | - | - | PBMC |
| - | - |
| García–Campayo et al., 2018 [117] | C | Experienced mindfulness meditators (n = 17) (≥ 10 years in total, ≥ 60 min/day) | - | - | Healthy relatives and friends of the meditators who had a similar lifestyle (n = 17) | - | - | PBMC |
| Meis3, Mafk | Glucose homeostasis |
| APOB, APOC2, HRH1, PTCH1, CLEC11A, NCOR | Lipid metabolism | ||||||||||
| TNFα, NF-kB, Nrf2 | Inflammation | ||||||||||
| Mendioroz et al., 2020 [118] | C | Experienced meditators (n = 17) (Same participant pools of García–Campayo et al., 2017) | - | - | Healthy relatives and friends of the meditators who had a similar lifestyle (n = 17) (Same participant pools of García–Campayo et al., 2017) | - | - | PBMC |
| GPR31 | Tumorigenesis, extravasation, and metastasis |
| SERPINB9 | Inhibition of apoptosis, inflammation, insulin resistance in coronary atherosclerosis | ||||||||||
| Intergenic CpG island within the subtelomeric region of chromosome 4 short arm | - | ||||||||||
| Bishop et al., 2018 [119] | L | PTSD patients of MBSR responder (n = 11) | MBSR | 9 weeks | PTSD patients of MBSR non-responder (n = 11) | MBSR | 9 weeks | PBMC | ↓Methylation in responders | FKBP5 | Stress-related pathway (glucocorticoid receptor regulation) |
| ↑Methylation in non-responders | |||||||||||
| Harkess et al., 2016 [120] | L,C | Women reporting psychological distress (≥ 16 on Kessler Psychological Distress Scale) (n = 15) | Yoga | 8 weeks | Women reporting psychological distress (≥ 26 on Kessler Psychological Distress Scale) (n = 11) | Control (waitlist) | 8 weeks | PBMC | ↓Methylation (cross-sectional: Post-intervention) | TNF | Inflammation |
| Ren et al., 2012 [121] | C | Women tai chi practitioners (n = 237) (≥ 3 years) | - | - | Women with no practice of tai chi (n = 263) | - | - | Saliva | Significantly slow age-related methylation dynamics in tai chi group compared to the control group in six age-related CpG marks | Age-related CpGs (Rad50_2, 17P_7, G6PD_6, G6PD_7, Rad50_10, Xp13_1) | Aging |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, H.-J.; Koh, E.; Sung, M.-K.; Kang, H. Changes Induced by Mind–Body Intervention Including Epigenetic Marks and Its Effects on Diabetes. Int. J. Mol. Sci. 2021, 22, 1317. https://doi.org/10.3390/ijms22031317
Yang H-J, Koh E, Sung M-K, Kang H. Changes Induced by Mind–Body Intervention Including Epigenetic Marks and Its Effects on Diabetes. International Journal of Molecular Sciences. 2021; 22(3):1317. https://doi.org/10.3390/ijms22031317
Chicago/Turabian StyleYang, Hyun-Jeong, Eugene Koh, Min-Kyu Sung, and Hojung Kang. 2021. "Changes Induced by Mind–Body Intervention Including Epigenetic Marks and Its Effects on Diabetes" International Journal of Molecular Sciences 22, no. 3: 1317. https://doi.org/10.3390/ijms22031317
APA StyleYang, H.-J., Koh, E., Sung, M.-K., & Kang, H. (2021). Changes Induced by Mind–Body Intervention Including Epigenetic Marks and Its Effects on Diabetes. International Journal of Molecular Sciences, 22(3), 1317. https://doi.org/10.3390/ijms22031317






