Molecular Iodine Has Extrathyroidal Effects as an Antioxidant, Differentiator, and Immunomodulator
Abstract
1. Introduction
1.1. Safety Concentration
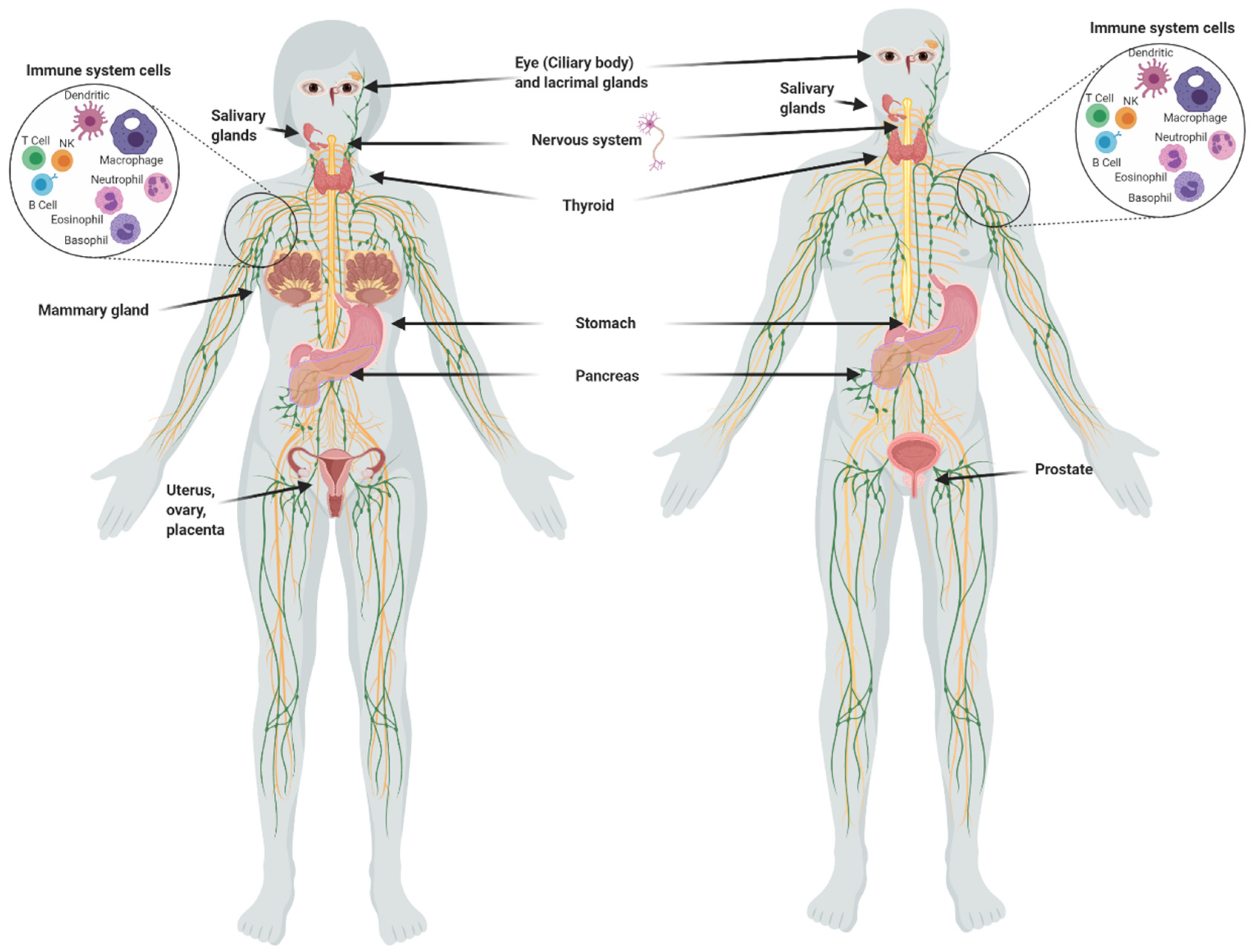
1.2. Iodine in Normal Tissues
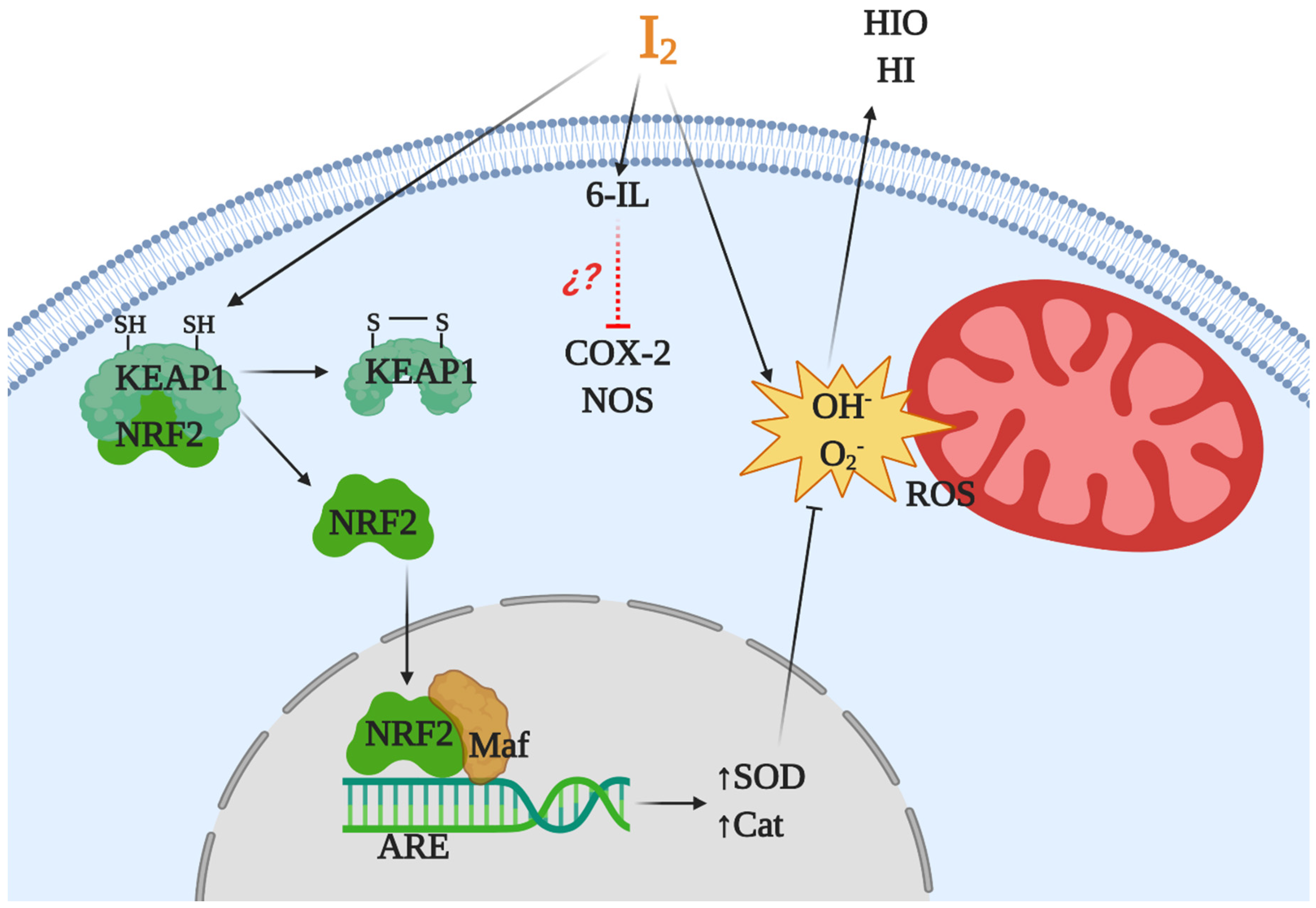
1.3. Antioxidant Effects
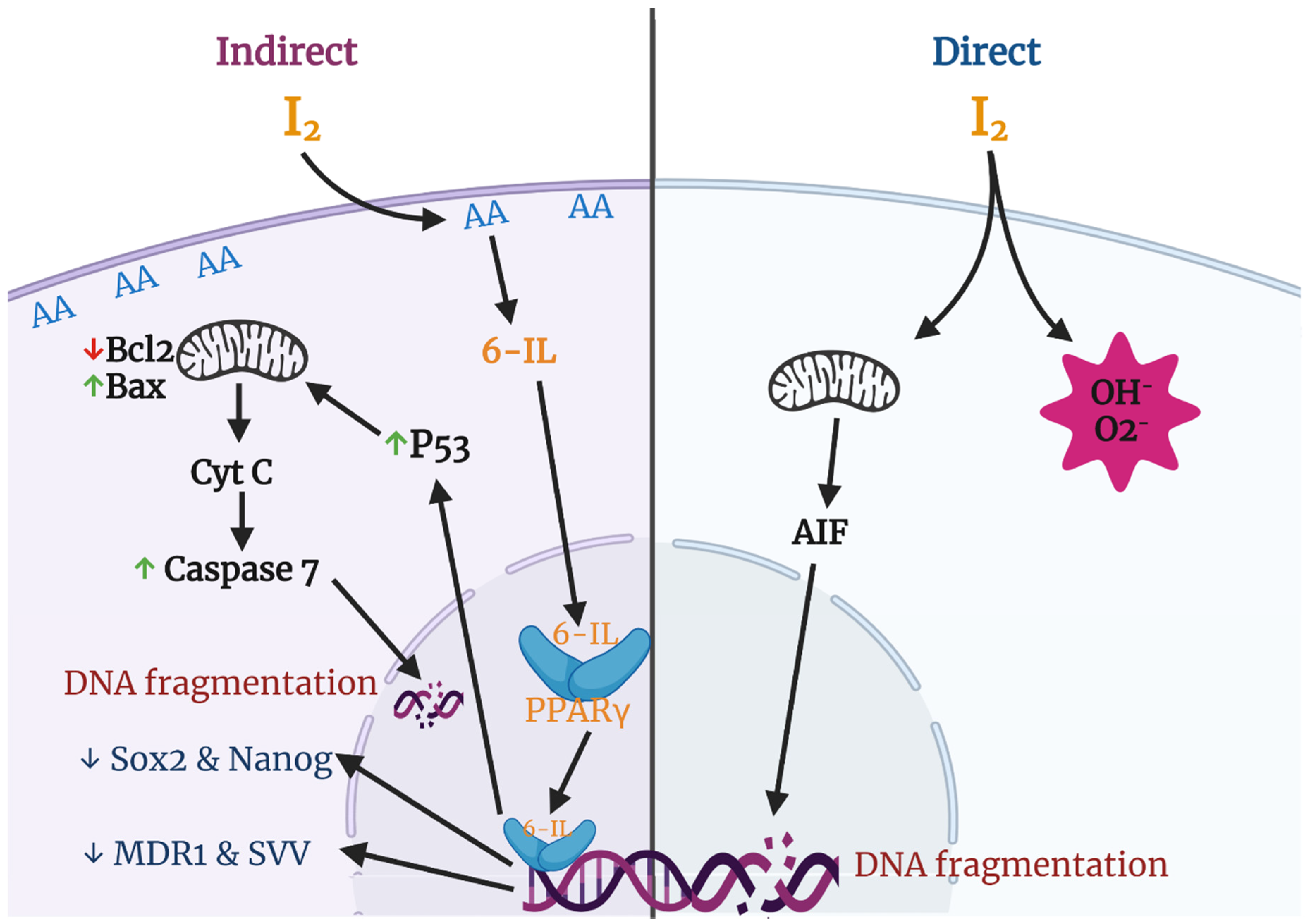
1.4. Antiproliferative and Apoptotic Actions
1.5. Effects on Cellular Differentiation
1.6. Immune Modulator
2. Discussion/Conclusions
- as an ancestral antioxidant by combining or competing with free radicals for membrane lipids, proteins, and DNA, increasing the expression or activity of antioxidant enzymes, or inactivating proinflammatory pathways stabilizing cellular redox status;
- as an inducer of antiproliferative, differentiation or apoptotic mechanisms by modulating mitochondrial potential or forming iodolipids and activating nuclear receptors;
- as an immune modulator acting directly on specific immune cells; and
- as a constituent part of thyroid hormones.
Funding
Acknowledgments
Conflicts of Interest
References
- De la Vieja, A.; Santisteban, P. Role of iodide metabolism in physiology and cancer. Endocr. Relat. Cancer 2018, 25, R225–R245. [Google Scholar] [CrossRef]
- Venturi, S. Evolutionary significance of iodine. Curr. Chem. Biol. 2011, 5, 155–162. [Google Scholar]
- Cann, S.A.; Van Netten, J.P.; Van Netten, C. Hypothesis: Iodine, selenium and the development of breast cancer. Cancer Causes Control 2000, 11, 121–127. [Google Scholar] [CrossRef]
- Smyth, P.P.A. Role of iodine in antioxidant defence in thyroid and breast disease. BioFactors 2003, 19, 121–130. [Google Scholar] [CrossRef]
- Venturi, S.; Venturi, M. Iodine in Evolution of Salivary Glands and in Oral Health. Nutr. Health 2009, 20, 119–134. [Google Scholar] [CrossRef]
- Aceves, C.; Anguiano, B.; Delgado, G. The Extrathyronine Actions of Iodine as Antioxidant, Apoptotic, and Differentiation Factor in Various Tissues. Thyroid 2013, 23, 938–946. [Google Scholar] [CrossRef]
- Juvenal, G.J.; Thomasz, L.; Oglio, R.; Perona, M.; Pisarev, M.A.; Rossich, L.; Salvarredi, L. Thyroid: Iodine beyond the thyronines. Curr. Chem. Biol. 2011, 5, 163–167. [Google Scholar]
- Torremante, E.P.; Rosner, H. Antiproliferative effects of molecular iodine in cancers. Curr. Chem. Biol. 2011, 5, 168–176. [Google Scholar]
- Bilal, M.Y.; Dambaeva, S.; Kwak-Kim, J.; Gilman-Sachs, A.; Beaman, K.D. A role for iodide and thyroglobulin in modulating the function of human immune cells. Front. Immunol. 2017, 8, 1573. [Google Scholar] [CrossRef]
- Moore, K.; Thomas, A.; Harding, K.G. Iodine released from the wound dressing iodosorb modulates the secretion of cytokines by human macrophages responding to bacterial lipopolysaccharide. Int. J. Biochem. Cell Biol. 1997, 29, 163–171. [Google Scholar] [CrossRef]
- Thrall, K.D.; Bull, R. Differences in the distribution of iodine and iodide in the Sprague-Dawley rat. Fundament. Appl. Toxicol. 1990, 15, 75–81. [Google Scholar] [CrossRef]
- Sherer, T.T.; Thrall, K.D.; Bull, R.J. Comparison of toxicity induced by iodine and iodide in male and female rats. J. Toxicol. Environ. Health Part A 1991, 32, 89–101. [Google Scholar] [CrossRef] [PubMed]
- García-Solís, P.; Alfaro, Y.; Anguiano, B.; Delgado, G.; Guzman, R.C.; Nandi, S.; Aceves, C. Inhibition of N-methyl-N-nitrosourea-induced mammary carcinogenesis by molecular iodine (I2) but not by iodide (I−) treatment: Evidence that I2 prevents cancer promotion. Mol. Cell. Endocrinol. 2005, 236, 49–57. [Google Scholar] [CrossRef] [PubMed]
- Eskin, B.A.; Grotkowski, C.E.; Connolly, C.P.; Ghent, W.R. Different tissue responses for iodine and iodide in rat thyroid and mammary glands. Biol. Trace Elem. Res. 1995, 49, 9–19. [Google Scholar] [CrossRef]
- Farebrother, J.; Zimmermann, M.B.; Andersson, M. Excess iodine intake: Sources, assessment, and effects on thyroid function. Ann. N. Y. Acad. Sci. 2019, 1446, 44–65. [Google Scholar] [CrossRef]
- WHO. Progress towards the Elimination of Iodine Deficiency Disorders (IDD). 1999. Available online: https://apps.who.int/iris/handle/10665/65931 (accessed on 1 September 2020).
- Backer, H.; Hollowell, J. Use of iodine for water disinfection: Iodine toxicity and maximum recommended dose. Environ. Health Perspect. 2000, 108, 679–684. [Google Scholar] [CrossRef]
- Bürgi, H. Iodine excess. Best Pract. Res. Clin. Endocrinol. Metab. 2010, 24, 107–115. [Google Scholar] [CrossRef]
- Alfaro, Y.; Delgado, G.; Cárabez-Trejo, A.; Anguiano, B.; Aceves, C. Iodine and doxorubicin, a good combination for mammary cancer treatment: Antineoplastic adjuvancy, chemoresistance inhibition, and cardioprotection. Mol. Cancer 2013, 12, 45. [Google Scholar] [CrossRef]
- Winkler, R.; Griebenow, S.; Wonisch, W. Effect of iodide on total antioxidant status of human serum. Cell Biochem. Funct. 2000, 18, 143–146. [Google Scholar] [CrossRef] [PubMed]
- Ghent, W.R.; Eskin, B.A.; Low, D.A.; Hill, L.P. Iodine replacement in fibrocystic disease of the breast. Can. J. Surg. 1993, 36, 453–460. [Google Scholar]
- Kessler, J.H. The Effect of Supraphysiologic Levels of Iodine on Patients with Cyclic Mastalgia. Breast J. 2004, 10, 328–336. [Google Scholar] [CrossRef] [PubMed]
- Anguiano, B.; Ledezma, O.; Juárez, M.; Nunez, F.; Aceves, C. Therapeutic effect of iodine on human benign prostatic hyperplasia. In Proceedings of the 14th International Thyroid Congress, Paris, France, 11–16 September 2010. [Google Scholar]
- Kessler, J. Are there Side Effects when Using Supraphysiologic Levels of Iodine in Treatment Regimens? Compr. Handb. Iodine 2009, 801, 801–810. [Google Scholar] [CrossRef]
- Anguiano, B.; García-Solís, P.; Delgado, G.; Velasco, C.A. Uptake and Gene Expression with Antitumoral Doses of Iodine in Thyroid and Mammary Gland: Evidence that Chronic Administration Has No Harmful Effects. Thyroid 2007, 17, 851–859. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Vega, A.; Vega-Riveroll, L.; Ayala, T.; Peralta, G.; Torres-Martel, J.M.; Rojas, J.; Mondragón, P.; Domínguez, A.; De Obaldía, R.; Avecilla-Guerrero, C.; et al. Adjuvant Effect of Molecular Iodine in Conventional Chemotherapy for Breast Cancer. Randomized Pilot Study. Nutrients 2019, 11, 1623. [Google Scholar] [CrossRef] [PubMed]
- Olvera-Caltzontzin, P.; Delgado, G.; Aceves, C.; Anguiano, B. Iodine Uptake and Prostate Cancer in the TRAMP Mouse Model. Mol. Med. 2013, 19, 409–416. [Google Scholar] [CrossRef]
- Zambrano-Estrada, X.; Landaverde-Quiroz, B.; Dueñas-Bocanegra, A.A.; De Paz-Campos, M.A.; Hernández-Alberto, G.; Solorio-Perusquia, B.; Trejo-Mandujano, M.; Pérez-Guerrero, L.; Delgado-González, E.; Anguiano, B.; et al. Molecular iodine/doxorubicin neoadjuvant treatment impair invasive capacity and attenuate side effect in canine mammary cancer. BMC Vet. Res. 2018, 14, 87. [Google Scholar] [CrossRef]
- Yeğinsu, A.; Karamustafaoglu, A.; Ozugurlu, F.; Etikan, I. Iodopovidone pleurodesis does not effect thyroid function in normal adults. Interact. Cardiovasc. Thorac. Surg. 2007, 6, 563–564. [Google Scholar] [CrossRef] [PubMed]
- Bogazzi, F.; Tomisti, L.; Bartalena, L.; Aghini-Lombardi, F.; Martino, E. Amiodarone and the thyroid: A 2012 update. J. Endocrinol. Investig. 2012, 35, 340–348. [Google Scholar]
- Rhee, C.M.; Bhan, I.; Alexander, E.K.; Brunelli, S.M. Association Between Iodinated Contrast Media Exposure and Incident Hyperthyroidism and Hypothyroidism. Arch. Intern. Med. 2012, 172, 153. [Google Scholar] [CrossRef]
- Michikawa, T.; Inoue, M.; Shimazu, T.; Sawada, N.; Iwasaki, M.; Sasazuki, S.; Yamaji, T.; Tsugane, S.; Japan Public Health Center-Based Prospective Study Group. Seaweed consumption and the risk of thyroid cancer in women: The Japan Public Health Center-based Prospective Study. Eur. J. Cancer Prev. 2012, 21, 254–260. [Google Scholar] [CrossRef]
- Nagataki, S. The Average of Dietary Iodine Intake due to the Ingestion of Seaweeds is 1.2 mg/day in Japan. Thyroid 2008, 18, 667–668. [Google Scholar] [CrossRef] [PubMed]
- Arroyo-Helguera, O.; Anguiano, B.; Delgado, G.; Aceves, C. Uptake and antiproliferative effect of molecular iodine in the MCF-7 breast cancer cell line. Endocr. Relat. Cancer 2006, 13, 1147–1158. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Küpper, F.C.; Schweigert, N.; Gall, E.A.; Legendre, J.-M.; Vilter, H.; Kloareg, B. Iodine uptake in Laminariales involves extracellular, haloperoxidase-mediated oxidation of iodide. Planta 1998, 207, 163–171. [Google Scholar] [CrossRef]
- Anguiano, B.; Aceves, C. Iodine in mammary and prostate pathologies. Curr. Chem. Biol. 2011, 5, 177–182. [Google Scholar]
- Kamangar, F.; Dores, G.M.; Anderson, W.F. Patterns of Cancer Incidence, Mortality, and Prevalence Across Five Continents: Defining Priorities to Reduce Cancer Disparities in Different Geographic Regions of the World. J. Clin. Oncol. 2006, 24, 2137–2150. [Google Scholar] [CrossRef] [PubMed]
- Zava, T.T.; Zava, D.T. Assessment of Japanese iodine intake based on seaweed consumption in Japan: A literature-based analysis. Thyroid Res. 2011, 4, 14. [Google Scholar] [CrossRef]
- Küpper, F.C.; Carpenter, L.J.; McFiggans, G.B.; Palmer, C.J.; Waite, T.J.; Boneberg, E.-M.; Woitsch, S.; Weiller, M.; Abela, R.; Grolimund, D.; et al. Iodide accumulation provides kelp with an inorganic antioxidant impacting atmospheric chemistry. Proc. Natl. Acad. Sci. USA 2008, 105, 6954–6958. [Google Scholar] [CrossRef]
- Berking, S.; Czech, N.; Gerharz, M.; Herrmann, K.; Hoffmann, U.; Raifer, H.; Sekul, G.; Siefker, B.; Sommerei, A.; Vedder, F. A newly discovered oxidant defence system and its involvement in the development of Aurelia aurita (Scyphozoa, Cnidaria): Reactive oxygen species and elemental iodine control medusa formation. Int. J. Dev. Biol. 2005, 49, 969–976. [Google Scholar] [CrossRef]
- Miller, A.E.M.; Heyland, A. Iodine accumulation in sea urchin larvae is dependent on peroxide. J. Exp. Biol. 2012, 216, 915–926. [Google Scholar] [CrossRef]
- Elstner, E.; Adamczyk, R.; Kröner, R.; Furch, A. The uptake of potassium iodide and its effect as an antioxidant in isolated rabbit eyes. Int. J. Ophthalmol. 1985, 191, 122–126. [Google Scholar]
- Cocchi, M.; Venturi, S. Iodide, antioxidant function and omega-6 and omega-3 fatty acids: A new hypothesis of biochemical cooperation? Prog. Nutr. 2000, 2, 15–19. [Google Scholar]
- Tseng, Y.-C.L.; Latham, K.R. Iodothyronines: Oxidative deiodination by hemoglobin and inhibition of lipid peroxidation. Lipids 1984, 19, 96–102. [Google Scholar] [CrossRef] [PubMed]
- Turner, K.B.; Khayat, G.B. Studies on the prevention of cholesterol atherosclerosis in rabbits. The influence of thyroidectomy upon the protective action of potassium iodide. J. Exp. Med. 1993, 58, 127. [Google Scholar] [CrossRef] [PubMed]
- Zhao, S.-J.; Ye, Y.; Sun, F.-J.; Tian, E.-J.; Chen, Z.-P. The Impact of Dietary Iodine Intake on Lipid Metabolism in Mice. Biol. Trace Elem. Res. 2010, 142, 581–588. [Google Scholar] [CrossRef] [PubMed]
- Herter-Aeberli, I.; Cherkaoui, M.; El Ansari, N.; Rohner, R.; Stinca, S.; Chabaa, L.; Von Eckardstein, A.; Aboussad, A.; Zimmermann, M. Iodine Supplementation Decreases Hypercholesterolemia in Iodine-Deficient, Overweight Women: A Randomized Controlled Trial. J. Nutr. 2015, 145, 2067–2075. [Google Scholar] [CrossRef]
- Rodríguez-Castelán, J.; Delgado-González, E.; Rodríguez-Benítez, E.; Castelán, F.; Cuevas-Romero, E.; Aceves, C. SAT-561 Protective Effect of Moderated Dose of Iodine in Pancreatic Alterations during Hypothyroidism. J. Endocr. Soc. 2019, 3, SAT-561. [Google Scholar] [CrossRef]
- Gärtner, R. Autoregulation of thyroid growth and function by iodine: Independent regulation of the thyroid gland by iodocompounds. In Comprehensive Handbook of Iodine: Nutritional, Biochemical, Pathological and Therapeutic Aspects, 1st ed.; Academic Press/Elsevier: Cambridge, MA, USA, 2009; pp. 243–247. [Google Scholar]
- Quintero-García, M.; Delgado-González, E.; Sánchez-Tusie, A.; Vázquez, M.; Aceves, C.; Anguiano, B. Iodine prevents the increase of testosterone-induced oxidative stress in a model of rat prostatic hyperplasia. Free Radic. Biol. Med. 2018, 115, 298–308. [Google Scholar] [CrossRef]
- Greenwald, M.B.-Y.; Frušić-Zlotkin, M.; Soroka, Y.; Ben-Sasson, S.; Bianco-Peled, H.; Kohen, R. A novel role of topical iodine in skin: Activation of the Nrf2 pathway. Free Radic. Biol. Med. 2017, 104, 238–248. [Google Scholar] [CrossRef]
- Paunkov, A.; Chartoumpekis, D.V.; Ziros, P.G.; Sykiotis, G.P. A Bibliometric Review of the Keap1/Nrf2 Pathway and its Related Antioxidant Compounds. Antioxidants 2019, 8, 353. [Google Scholar] [CrossRef]
- Vitale, M.; Di Matola, T.; D’Ascoli, F.; Salzano, S.; Bogazzi, F.; Fenzi, G.; Rossi, G. Iodide excess induces apoptosis in thyroid cells through a p53-independent mechanism involving oxidative stress. Endocrinology 2000, 141, 598–605. [Google Scholar] [CrossRef]
- Zhang, L.; Sharma, S.; Zhu, L.X.; Kogai, T.; Hershman, J.M.; Brent, G.A.; Dubinett, S.M.; Huang, M. Nonradioactive iodide effectively induces apoptosis in genetically modified lung cancer cells. Cancer Res. 2003, 63, 5065–5072. [Google Scholar] [PubMed]
- Hanahan, D.; Weinberg, R.A. Hallmarks of Cancer: The Next Generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [PubMed]
- Teas, J.; Harbison, M.L.; Gelman, R.S. Dietary seaweed (Laminaria) and mammary carcinogenesis in rats. Cancer Res. 1984, 44, 2758–2761. [Google Scholar] [PubMed]
- Funahashi, H.; Imai, T.; Tanaka, Y.; Tsukamura, K.; Hayakawa, Y.; Kikumori, T.; Mase, T.; Itoh, T.; Nishikawa, M.; Hayashi, H.; et al. Wakame Seaweed Suppresses the Proliferation of 7,12-Dimethylbenz(a)-anthracene-induced Mammary Tumors in Rats. Jpn. J. Cancer Res. 1999, 90, 922–927. [Google Scholar] [CrossRef]
- Yamamoto, I.; Maruyama, H.; Moriguchi, M. The effect of dietary seaweeds on 7,12-dimethylbenz[a]anthracene-induced mammary tumorigenesis in rats. Cancer Lett. 1987, 35, 109–118. [Google Scholar] [CrossRef]
- Kato, N. Suppressive effect of iodine preparations on proliferation of DMBA-induced breast cancer in rat. J. Jpn. Soc. Cancer Ther. 1994, 29, 582. [Google Scholar]
- Soriano, O.; Delgado, G.; Anguiano, B.; Petrosyan, P.; Molina-Servín, E.D.; Gonsebatt, M.E.; Aceves, C. Antineoplastic effect of iodine and iodide in dimethylbenz[a]anthracene-induced mammary tumors: Association between lactoperoxidase and estrogen-adduct production. Endocr. Relat. Cancer 2011, 18, 529–539. [Google Scholar] [CrossRef]
- Løvstad, R.A. A kinetic study on the lactoperoxidase catalyzed oxidation of estrogens. BioMetals 2006, 19, 587–592. [Google Scholar] [CrossRef]
- Cavalieri, E.L.; Stack, D.E.; Devanesan, P.D.; Todorovic, R.; Dwivedy, I.; Higginbotham, S.; Johansson, S.L.; Patil, K.D.; Gross, M.L.; Gooden, J.K.; et al. Molecular origin of cancer: Catechol estrogen-3,4-quinones as endogenous tumor initiators. Proc. Natl. Acad. Sci. USA 1997, 94, 10937–10942. [Google Scholar] [CrossRef]
- Cavalieri, E.L.; Rogan, E.G. Depurinating estrogen-DNA adducts in the etiology and prevention of breast and other human cancers. Future Oncol. 2010, 6, 75–91. [Google Scholar] [CrossRef]
- Shrivastava, A.; Tiwari, M.; Sinha, R.A.; Kumar, A.; Balapure, A.K.; Bajpai, V.K.; Sharma, R.; Mitra, K.; Tandon, A.; Godbole, M.M. Molecular Iodine Induces Caspase-Independent Apoptosis in Human Breast Carcinoma Cells Involving the Mitochondria-mediated Pathway. J. Biol. Chem. 2006, 281, 19762–19771. [Google Scholar] [CrossRef]
- Aceves, C.; García-Solís, P.; Omar, A.-H.; Vega-Riveroll, L.; Delgado, G.; Anguiano, B. Antineoplastic effect of iodine in mammary cancer: Participation of 6-iodolactone (6-IL) and peroxisome proliferator-activated receptors (PPAR). Mol. Cancer 2009, 8, 33. [Google Scholar] [CrossRef]
- Chen, X.; Duan, N.; Zhang, C.; Zhang, W. Survivin and Tumorigenesis: Molecular Mechanisms and Therapeutic Strategies. J. Cancer 2016, 7, 314–323. [Google Scholar] [CrossRef]
- Yang, C.F.; Shen, H.M.; Ong, C.N. Intracellular Thiol Depletion Causes Mitochondrial Permeability Transition in Ebselen-Induced Apoptosis. Arch. Biochem. Biophys. 2000, 380, 319–330. [Google Scholar] [CrossRef]
- Upadhyay, G.; Singh, R.; Sharma, R.; Balapure, A.K.; Godbole, M.M. Differential action of iodine on mitochondria from human tumoral- and extra-tumoral tissue in inducing the release of apoptogenic proteins. Mitochondrion 2002, 2, 199–210. [Google Scholar] [CrossRef]
- Boeynaems, J.; Hubbard, W. Transformation of arachidonic acid into an iodolactone by the rat thyroid. J. Biol. Chem. 1980, 255, 9001–9004. [Google Scholar] [CrossRef]
- Dugrillon, A.; Uedelhoven, W.M.; Pisarev, M.A.; Bechtner, G.; Gärtner, R. Identification of δ-Iodolactone in Iodide Treated Human Goiter and Its Inhibitory Effect on Proliferation of Human Thyroid Follicles. Horm. Metab. Res. 1994, 26, 465–469. [Google Scholar] [CrossRef]
- Wang, D.; Dubois, R.N. Role of prostanoids in gastrointestinal cancer. J. Clin. Investig. 2018, 128, 2732–2742. [Google Scholar] [CrossRef]
- Desai, S.J.; Prickril, B.; Rasooly, A. Mechanisms of Phytonutrient Modulation of Cyclooxygenase-2 (COX-2) and Inflammation Related to Cancer. Nutr. Cancer 2018, 70, 350–375. [Google Scholar] [CrossRef]
- Arroyo-Helguera, O.; Rojas, E.; Delgado, G.; Aceves, C. Signaling pathways involved in the antiproliferative effect of molecular iodine in normal and tumoral breast cells: Evidence that 6-iodolactone mediates apoptotic effects. Endocr. Relat. Cancer 2008, 15, 1003–1011. [Google Scholar] [CrossRef]
- Aranda, N.; Sosa, S.; Delgado, G.; Aceves, C.; Anguiano, B. Uptake and antitumoral effects of iodine and 6-iodolactone in differentiated and undifferentiated human prostate cancer cell lines. Prostate 2012, 73, 31–41. [Google Scholar] [CrossRef]
- Rösner, H.; Möller, W.; Groebner, S.; Torremante, P. Antiproliferative/cytotoxic effects of molecular iodine, povidone-iodine and Lugol’s solution in different human carcinoma cell lines. Oncol. Lett. 2016, 12, 2159–2162. [Google Scholar] [CrossRef]
- Nuñez-Anita, R.; Arroyo-Helguera, O.; Cajero-Juárez, M.; López-Bojorquez, L.; Aceves, C. A complex between 6-iodolactone and the peroxisome proliferator-activated receptor type gamma may mediate the antineoplasic effect of iodine in mammary cancer. Prostaglandins Other Lipid Mediat. 2009, 89, 34–42. [Google Scholar] [CrossRef]
- Reka, A.K.; Kurapati, H.; Narala, V.R.; Bommer, G.T.; Chen, J.; Standiford, T.J.; Keshamouni, V.G. Peroxisome Proliferator-Activated Receptor-γ Activation Inhibits Tumor Metastasis by Antagonizing Smad3-Mediated Epithelial-Mesenchymal Transition. Mol. Cancer Ther. 2010, 9, 3221–3232. [Google Scholar] [CrossRef]
- Samid, D.; Wells, M.; Greene, M.E.; Shen, W.; Palmer, C.N.; Thibault, A. Peroxisome proliferator-activated receptor γ as a novel target in cancer therapy: Binding and activation by an aromatic fatty acid with clinical antitumor activity. Clin. Cancer Res. 2000, 6, 933–941. [Google Scholar]
- Reka, A.K.; Goswami, M.T.; Krishnapuram, R.; Standiford, T.J.; Keshamouni, V.G. Molecular cross-regulation between PPAR-γ and other signaling pathways: Implications for lung cancer therapy. Lung Cancer 2011, 72, 154–159. [Google Scholar] [CrossRef]
- Dijkstra, B.; Prichard, R.S.; Lee, A.; Kelly, L.M.; Smyth, P.P.A.; Crotty, T.; McDermott, E.W.; Hill, A.D.K.; O’Higgins, N.J. Changing patterns of thyroid carcinoma. Ir. J. Med. Sci. 2007, 176, 87–90. [Google Scholar] [CrossRef]
- Maso, L.D.; Bosetti, C.; La Vecchia, C.; Franceschi, S. Risk factors for thyroid cancer: An epidemiological review focused on nutritional factors. Cancer Causes Control 2008, 20, 75–86. [Google Scholar] [CrossRef]
- Knobel, M.; Medeiros-Neto, G. Relevance of iodine intake as a reputed predisposing factor for thyroid cancer. Arq. Bras. Endocrinol. Metabol. 2007, 51, 701–712. [Google Scholar] [CrossRef]
- Mendieta, I.; Nuñez-Anita, R.E.; Nava-Villalba, M.; Zambrano-Estrada, X.; Delgado-González, E.; Anguiano, B.; Aceves, C. Molecular iodine exerts antineoplastic effects by diminishing proliferation and invasive potential and activating the immune response in mammary cancer xenografts. BMC Cancer 2019, 19, 261. [Google Scholar] [CrossRef]
- Olivo-Vidal, Z.E.; Rodríguez, R.C.; Arroyo-Helguera, O. Iodine Affects Differentiation and Migration Process in Trophoblastic Cells. Biol. Trace Elem. Res. 2015, 169, 180–188. [Google Scholar] [CrossRef]
- Bigoni-Ordóñez, G.D.; Ortiz-Sánchez, E.; Rosendo-Chalma, P.; Valencia-González, H.A.; Aceves, C.; García-Carrancá, A. Molecular iodine inhibits the expression of stemness markers on cancer stem-like cells of established cell lines derived from cervical cancer. BMC Cancer 2018, 18, 928. [Google Scholar] [CrossRef]
- Bontempo, A.; Ugalde-Villanueva, B.; Delgado-González, E.; Rodríguez, Á.L.; Aceves, C. Molecular iodine impairs chemoresistance mechanisms, enhances doxorubicin retention and induces downregulation of the CD44+/CD24+ and E-cadherin+/vimentin+ subpopulations in MCF-7 cells resistant to low doses of doxorubicin. Oncol. Rep. 2017, 38, 2867–2876. [Google Scholar] [CrossRef]
- Stone, O.J. The role of the primitive sea in the natural selection of iodides as a regulating factor in inflammation. Med. Hypotheses 1988, 25, 125–129. [Google Scholar] [CrossRef]
- Ullberg, S.; Ewaldsson, B. Distribution of Radio-Iodine Studied by Whole-Body Autoradiography. Acta Radiol. Ther. Phys. Biol. 1964, 2, 24–32. [Google Scholar] [CrossRef]
- Venturi, S.; Venturi, M. Iodine, thymus, and immunity. Nutrition 2009, 25, 977–979. [Google Scholar] [CrossRef]
- Miyachi, Y.; Niwa, Y. Effects of potassium iodide, colchicine and dapsone on the generation of polymorphonuclear leukocyte-derived oxygen intermediates. Br. J. Dermatol. 1982, 107, 209–214. [Google Scholar] [CrossRef]
- Honma, K.; Saga, K.; Onodera, H.; Takahashi, M. Potassium iodide inhibits neutrophil chemotaxis. Acta Derm. Venereol. 1990, 70, 247–249. [Google Scholar]
- Beukelman, C.; Berg, A.V.D.; Hoekstra, M.; Uhl, R.; Reimer, K.; Mueller, S. Anti-inflammatory properties of a liposomal hydrogel with povidone-iodine (Repithel®) for wound healing in vitro. Burns 2008, 34, 845–855. [Google Scholar] [CrossRef]
- Klebanoff, S.J.; Kettle, A.J.; Rosen, H.; Winterbourn, C.C.; Nauseef, W.M. Myeloperoxidase: A front-line defender against phagocytosed microorganisms. J. Leukoc. Biol. 2013, 93, 185–198. [Google Scholar] [CrossRef]
- Zhao, D.; Lim, C.-P.; Miyanaga, K.; Tanji, Y. Iodine from bacterial iodide oxidization by Roseovarius spp. inhibits the growth of other bacteria. Appl. Microbiol. Biotechnol. 2012, 97, 2173–2182. [Google Scholar] [CrossRef] [PubMed]
- Bigliardi, P.L.; Alsagoff, S.A.L.; El-Kafrawi, H.Y.; Pyon, J.-K.; Wa, C.T.C.; Villa, M.A. Povidone iodine in wound healing: A review of current concepts and practices. Int. J. Surg. 2017, 44, 260–268. [Google Scholar] [CrossRef] [PubMed]
- Marani, L.; Venturi, S.; Masala, R. Role of iodine in delayed immune response. Ir. J. Med. Sci. 1985, 21, 864. [Google Scholar]
- Costa, R.O.; De Macedo, P.M.; Carvalhal, A.; Bernardes-Engemann, A.R. Use of potassium iodide in Dermatology: Updates on an old drug. An. Bras. Dermatol. 2013, 88, 396–402. [Google Scholar] [CrossRef]
- Cuenca-Micó, O. Efectos del Yodo Molecular/Quimioterapia en los Patrones de Metilación de Células Inmunes Asociadas a Tumores de Cáncer Mamario. Ph.D. Thesis, UNAM Institute of Neurobiology (Instituto de Neurobiología UNAM), Juriquilla, Querétaro, Mexico, 2020. [Google Scholar]
- Luongo, D.; Bergamo, P.; Rossi, M. Effects of conjugated linoleic acid on growth and cytokine expression in Jurkat T cells. Immunol. Lett. 2003, 90, 195–201. [Google Scholar] [CrossRef]
- Doloff, J.C.; Waxman, D.J. Transcriptional profiling provides insights into metronomic cyclophosphamide-activated, innate immune-dependent regression of brain tumor xenografts. BMC Cancer 2015, 15, 375. [Google Scholar] [CrossRef]
- Wang, Z.; Lu, Q. Epigenetic Alterations in Cellular Immunity: New Insights into Autoimmune Diseases. Cell. Physiol. Biochem. 2017, 41, 645–660. [Google Scholar] [CrossRef]
- Gan, Z.; Wei, W.; Wu, J.; Zhao, Y.; Zhang, L.; Wang, T.; Gan, Z. Resveratrol and Curcumin Improve Intestinal Mucosal Integrity and Decrease m6A RNA Methylation in the Intestine of Weaning Piglets. ACS Omega 2019, 4, 17438–17446. [Google Scholar] [CrossRef]

| Individuals with Underlying Thyroid Disease |
|---|
| Graves’s disease |
| Hashimoto thyroiditis |
| Euthyroid with a history of subacute thyroiditis |
| Euthyroid with a history of postpartum thyroiditis |
| Euthyroid with a history of type 2 amiodarone–induced thyroiditis |
| Euthyroid with post-hemithyroidectomy |
| Euthyroid after interferon-γ therapy |
| Individuals with a family history of goiter or thyroiditis |
| Individuals with chronic iodine deficiency |
| Fetuses, preterm neonates, and newborn infants exposed to high doses of iodine through the placenta and milk |
| Elderly people with subclinical hypothyroidism |
| Patients taking medications such as expectorants or amiodarone that contains high concentrations of iodine |
| Patients with certain nonthyroidal disease such as chronic dialysis and cystic fibrosis, especially those taking sulfisoxazole. |
| Patients taking lithium |
| Source of Iodine | Iodine Dose (mg/day) | Treatment Time | Chemical Form of Iodine | Effects on Thyroid Function | Ref |
|---|---|---|---|---|---|
| Iodopovidone (5% solution) mouthwash using 2–4 mL | 14–28 | Days-weeks | I2 | Values remain within normal range | [17,18,29] |
| Amiodarone | Months-years | Iodide | [15,30] | ||
| 1 tablet (100 mg) | 3 | Thyrotoxicosis (2%) Hyperthyroidism (1%) | |||
| 1 tablet (600 mg) | 21 | Hypothyroidism (2–10%) | |||
| Iodinated contrast medium (200 mL/dose) | 7–10 | One dose | Iodide | Hyperthyroidism or Hypothyroidism (1–2%) | [15,31] |
| Seaweed Blended brown seaweed (1 bowl, 250 mL soup) | 1–3 | Weeks-months | Iodide, I2 | Normal values or transient subclinical hypothyroidism (2–10%) | [32,33] |
| High level of consumption (>6 g seaweed/day) | >20 | Risk of papillary thyroid cancer (1–10%) | |||
| KI supplements Water solution (5–15 mg) | >2 | Days-weeks | Iodide | Transient subclinical hypothyroidism, | [15,17,18] |
| 1 tablet (50 mg) | >30 | Thyrotoxicosis (2–10%) TPOAb, TgAb (6–20%) | |||
| Purified water solutions (8 mg/L per tablet) 1 tablet | 1–5 | Months-years | I2 | Normal values | [15,17,18] |
| 4 tablets | 10–32 | Transient hypothyroidism and goiter TPOAb, TgAb (3–16%) | |||
| Aqueous I2 solution I2 water solution; Lugol’s solution, or (1–2 tablets (3 mg per tablet) | 1–6 | Months-years | I2, I2-iodide | Values remain within normal range | [17,18,21,22,26] |
| 3–4 tablets (3 mg per tablet) | 9–12 | Transient subclinical hypothyroidism, headache, sinusitis, diarrhea acne (6–20%) | |||
| Mix yodica | 1–3 | Values remain within normal range |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aceves, C.; Mendieta, I.; Anguiano, B.; Delgado-González, E. Molecular Iodine Has Extrathyroidal Effects as an Antioxidant, Differentiator, and Immunomodulator. Int. J. Mol. Sci. 2021, 22, 1228. https://doi.org/10.3390/ijms22031228
Aceves C, Mendieta I, Anguiano B, Delgado-González E. Molecular Iodine Has Extrathyroidal Effects as an Antioxidant, Differentiator, and Immunomodulator. International Journal of Molecular Sciences. 2021; 22(3):1228. https://doi.org/10.3390/ijms22031228
Chicago/Turabian StyleAceves, Carmen, Irasema Mendieta, Brenda Anguiano, and Evangelina Delgado-González. 2021. "Molecular Iodine Has Extrathyroidal Effects as an Antioxidant, Differentiator, and Immunomodulator" International Journal of Molecular Sciences 22, no. 3: 1228. https://doi.org/10.3390/ijms22031228
APA StyleAceves, C., Mendieta, I., Anguiano, B., & Delgado-González, E. (2021). Molecular Iodine Has Extrathyroidal Effects as an Antioxidant, Differentiator, and Immunomodulator. International Journal of Molecular Sciences, 22(3), 1228. https://doi.org/10.3390/ijms22031228





