Abstract
Mechanisms involved in the de-acclimation of herbaceous plants caused by warm periods during winter are poorly understood. This study identifies the genes associated with this mechanism in winter barley. Seedlings of eight accessions (four tolerant and four susceptible to de-acclimation cultivars and advanced breeding lines) were cold acclimated for three weeks and de-acclimated at 12 °C/5 °C (day/night) for one week. We performed differential expression analysis using RNA sequencing. In addition, reverse-transcription quantitative real-time PCR and enzyme activity analyses were used to investigate changes in the expression of selected genes. The number of transcripts with accumulation level changed in opposite directions during acclimation and de-acclimation was much lower than the number of transcripts with level changed exclusively during one of these processes. The de-acclimation-susceptible accessions showed changes in the expression of a higher number of functionally diverse genes during de-acclimation. Transcripts associated with stress response, especially oxidoreductases, were the most abundant in this group. The results provide novel evidence for the distinct molecular regulation of cold acclimation and de-acclimation. Upregulation of genes controlling developmental changes, typical for spring de-acclimation, was not observed during mid-winter de-acclimation. Mid-winter de-acclimation seems to be perceived as an opportunity to regenerate after stress. Unfortunately, it is competitive to remain in the cold-acclimated state. This study shows that the response to mid-winter de-acclimation is far more expansive in de-acclimation-susceptible cultivars, suggesting that a reduced response to the rising temperature is crucial for de-acclimation tolerance.
Keywords:
de-acclimation; freezing tolerance; barley; climate change; RNAseq; gene expression; oxidoreductase 1. Introduction
Under global warming, it might be considered that winter hardiness will be less critical for future crop production. However, this assumption is invalid, as the only parameters likely to change will be the predominant factors that influence the overwintering of plants locally. Climate change scenarios predict that weather conditions will become unstable, and in most cases, not typical for the season [1]. In a moderate climate zone, freezing tolerance is most important for a plant’s survival in winter. Therefore, a large body of winter hardiness-oriented research has focused on this trait. Different genes associated with freezing tolerance have been identified in many species, and the mechanisms influencing their expression have been widely studied [2,3]. In comparison, limited information is available on tolerance to de-acclimation, and the studies that have been conducted have predominantly investigated woody species [4,5].
Susceptibility to de-acclimation during winter is a complex trait. At least two types of de-acclimation with potentially distinct genetic and physiological bases can be distinguished. (1) The highest degree of freezing tolerance is attained in most plants in mid-winter. Subsequently, freezing tolerance decreases gradually. This “passive” (i.e., independent of environmental conditions) de-acclimation is connected mainly with the vegetative/reproductive transition and is widely described as the relationship between cold acclimation ability and vernalization requirements. However, it may also be associated with the decrease in organic compounds accumulated by the plant before winter and the plant’s general weakening. This type of de-acclimation is irreversible. (2) Plants also tend to de-acclimate as a result of mid-winter warm spell [1]. This “active” (in the sense of suggested reception of environmental signals) type of de-acclimation can be reversible or irreversible depending on various factors [6]. De-acclimation is unfavorable for the plant only when in spring, or after a warm period in winter, the temperature decreases rapidly to freezing temperatures [7]. Various future weather simulation models predict an increase in mean winter temperatures, which will probably cause an increase in yield loss caused by de-acclimation. Thus, tolerance to de-acclimation or ability for rapid re-acclimation will likely be critical for winter hardiness in the future [1].
Winter barley shows a relatively weak cold acclimation capability [8,9], and, in consequence, low winter hardiness, which limits large-scale production of the crop despite increasing interest from the beer industry in winter barley cultivars. The genetic basis of freezing tolerance in winter barley has been studied previously by many research groups, for example [10,11,12,13,14]. Certain genes involved in the process of cold hardening in winter barley have been identified [15,16], but the “active” de-acclimation process remains undissected.
The aim of this study was to identify genes associated with response to de-acclimation in winter barley. We assumed that mid-winter de-acclimation is not a process simply reverse to cold acclimation, and therefore, new genes associated only with active de-acclimation could be dissected.
2. Results
Eight previously studied (Wójcik-Jagła and Rapacz, unpublished), cold-acclimated barley accessions (four tolerant and four susceptible to de-acclimation) were subjected to de-acclimation treatment that mimicked a mid-winter warm spell (i.e., active de-acclimation). We performed differential expression analysis using RNA sequencing (RNAseq) followed by reverse-transcription quantitative real-time PCR (RT-qPCR) and enzyme activity analyses to explore the genetic basis of the response to active de-acclimation in barley.
From the differential gene expression analysis followed by comparisons using Venn diagrams, many differentially expressed genes (DEGs) were detected in various comparisons. It is emphasized that the following numbers are based on DEGs common to four accessions from each group of de-acclimation-tolerant and -susceptible genotypes.
In barley accessions tolerant to de-acclimation, 698 genes (397 upregulated and 301 downregulated) were differentially expressed between cold acclimation (CA-21) and the control (CA-0 (C)) at false discovery rate (FDR) < 0.05 and 430 genes (259 upregulated and 171 downregulated) were significant at FDR < 0.01. With regard to accessions susceptible to de-acclimation, we identified 1082 DEGs (680 upregulated and 402 downregulated) between CA-21 and CA-0 (C) with FDR < 0.05 and 747 (494 upregulated and 253 downregulated) with FDR < 0.01 (Figure 1).
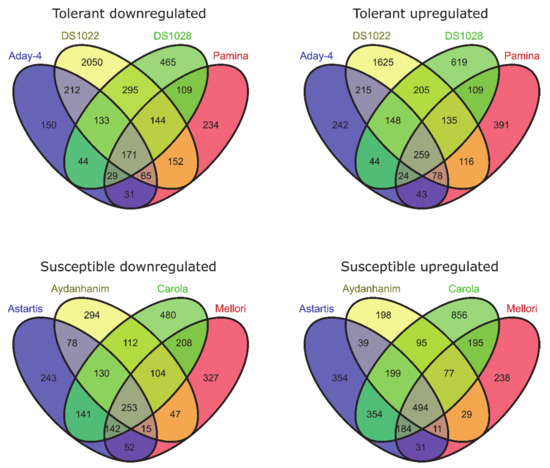
Figure 1.
Differentially expressed genes (DEGs) between cold-acclimated (CA-21) and control (CA-0 (C), before cold acclimation) barley accessions (log2FC = 2, false discovery rate (FDR) < 0.01).
Two hundred and thirteen DEGs (114 upregulated and 99 downregulated) were identified between de-acclimated (DA-28) and CA-21 for de-acclimation-tolerant accessions at FDR < 0.05, of which 115 genes (49 upregulated and 66 downregulated) were significant at FDR < 0.01. With regard to de-acclimation-susceptible accessions, 789 genes (382 upregulated and 407 downregulated) were differentially expressed in response to de-acclimation at FDR < 0.05 and 475 genes (230 upregulated and 245 downregulated) at FDR < 0.01 (Figure 2).
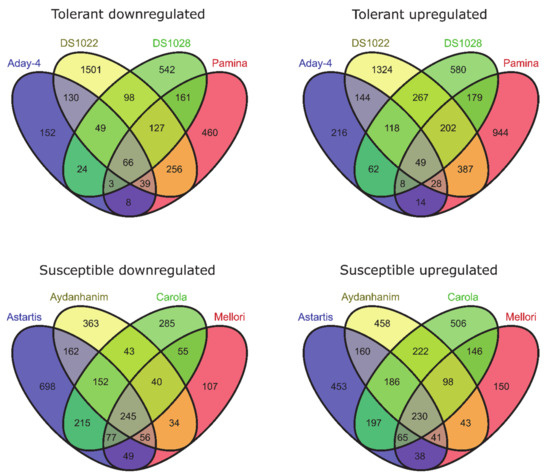
Figure 2.
DEGs between de-acclimated (DA-28) and cold-acclimated (CA-21) barley accessions (log2FC = 2, FDR < 0.01).
When compared between DA-28 and CA-0 (C) for tolerant barley accessions, 118 genes were differentially expressed (97 upregulated and 21 downregulated) at FDR < 0.05 and 57 (48 upregulated and 9 downregulated) at FDR < 0.01 (Figure 3). With respect to susceptible accessions, the same comparison identified 125 DEGs (95 upregulated and 30 downregulated) at FDR < 0.05, of which 59 (46 upregulated and 13 downregulated) were significant at FDR < 0.01 (Figure 3).
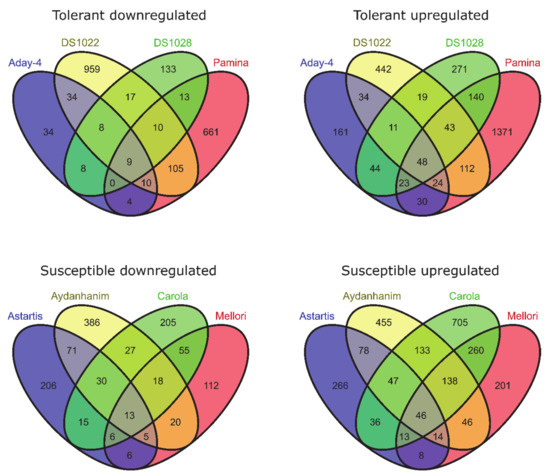
Figure 3.
DEGs between de-acclimated (DA-28) and control (C0, before cold-acclimation) barley accessions (log2FC = 2, FDR < 0.01).
To identify genes for which expression changed owing to de-acclimation only (different from DEGs also associated with cold acclimation but regulated in the opposite direction during de-acclimation), we compared common DEGs for CA-0 (C) vs. CA-21 and DA-28 vs. CA-21. Ninety-nine DEGs (25 downregulated and 74 upregulated) were specific only to conditions mimicking de-acclimation during mid-winter warm spell in de-acclimation-tolerant barley accessions (FDR < 0.05), of which 49 genes (33 up- and 16 downregulated) were significant at FDR < 0.01 (Figure 4). In addition, 343 genes (121 downregulated and 222 upregulated) were differentially expressed explicitly after de-acclimation in susceptible barley accessions (FDR < 0.05), of which 204 genes (137 upregulated and 67 downregulated) were significant at FDR < 0.01 (Figure 4). Only 54 DEGs (19 upregulated and 35 downregulated, FDR < 0.05) specific only to de-acclimation during mid-winter warm periods were common to both de-acclimation-tolerant and -susceptible barley accessions, of which seven genes (five upregulated and two downregulated) were significant at FDR < 0.01 (Figure 5).
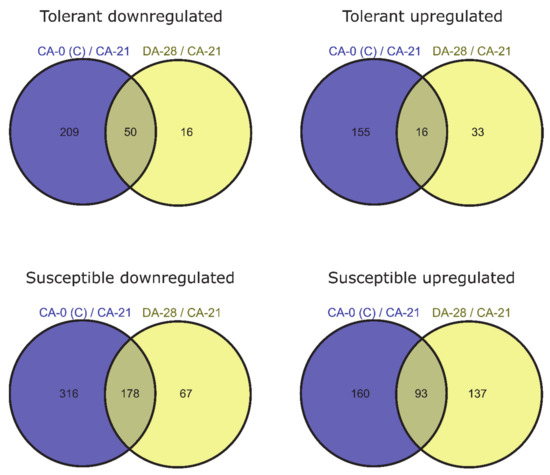
Figure 4.
DEGs specific only to de-acclimation (yellow) during mid-winter warm periods (log2FC = 2, FDR < 0.01).
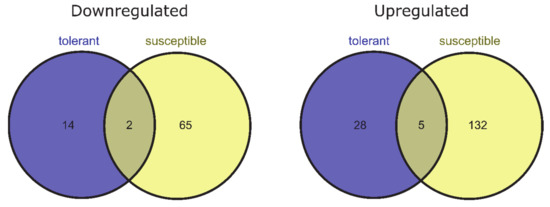
Figure 5.
DEGs specific only to de-acclimation during mid-winter warm periods in susceptible and tolerant to de-acclimation barley accessions (log2FC = 2, FDR < 0.01).
Gene ontology (GO) enrichment analysis revealed significant GO terms for DEGs specific only to de-acclimation (Table 1 and Table S1, Figures S1–S6). The susceptible group of accessions showed a much more functionally diverse spectrum of genes for which expression was changed in response to temperature rise. A considerable number of DEGs from both tolerant and susceptible accessions was directly or indirectly associated with photosynthesis. Thirty-nine sequences were associated only with phosphorylation (GO:0016310), and 27 were associated with phosphate-containing compound metabolic processes (GO:0006796), eight with ion transmembrane transport (GO:0034220), and 41 were significantly associated with either ATP binding (GO:0005524) or ATP metabolic processes (GO:0046034; Table 1).

Table 1.
Functional groups of significant (FDR < 0.05) GO terms for differentially expressed genes specific only to de-acclimation.
A total of 224 sequences of DEGs specific to de-acclimation (FDR < 0.01) were successfully annotated with either characterized or uncharacterized proteins. In addition, the most similar sequences were identified using the BLAST algorithm. Thirty-six sequences were annotated only in de-acclimation-tolerant barley accessions, whereas 181 sequences were characteristic solely of de-acclimation-susceptible accessions. Seven annotated sequences were common to de-acclimation-susceptible and -tolerant accessions (Table 2).

Table 2.
Annotation of selected differentially expressed barley gene sequences specific only to de-acclimation. Sequences common for both tolerant and susceptible to de-acclimation barley accessions are in italics, N/A—not available.
Five annotated DEGs were selected for further verification in an RT-qPCR experiment. The selected sequences were annotated with characterized proteins. The average (for four accessions) log2 fold change in their expression between CA-21 and DA-28 was at least 4.0 or −4.0. In addition, the function of the annotated protein was a crucial consideration in the selection process. The chosen sequences were annotated to proteins belonging to four groups: Stress response—antioxidative enzymes (peroxidase and catalase), stress response—heat shock proteins (sHSP domain-containing protein), stress response—freezing tolerance-related proteins (CBF 14), and proteins involved in structural functions of cell walls and membranes (LRRNT_2 domain-containing protein/polygalacturonase (PGU) inhibitor-like).
The transcription profiles of Peroxidase did not show a consistent pattern among the studied de-acclimation-tolerant and -susceptible barley accessions (Figure 6). Increase in transcript copy number after de-acclimation (DA-28) was observed in the accessions Aday-4, Astartis, Aydanhanim, Carola, and Mellori, of which the latter four were classified as de-acclimation-susceptible in a previous study (data not published). The increase in Peroxidase transcript accumulation was preceded by an initial decrease at DA-23 in most cases (Figure 6). The expression profiles of Catalase were almost identical for half of the tested barley accessions, namely, Aday-4, DS1022, Pamina (de-acclimation tolerant), and Carola (de-acclimation susceptible) (Figure 6). In these accessions, a slight increase in Catalase expression was observed at the beginning of de-acclimation, and a distinct increase was detected after one week under de-acclimating conditions followed by a dramatic decrease in transcript copy number during re-acclimation to cold. An increase in expression of Catalase at DA-28 in relation to CA-21 was also observed in DS1028, Astartis (de-acclimation tolerant), and Aydanhanim (de-acclimation susceptible). In these accessions, a more significant (or equally high) number of copies of the Catalase gene was detected at DA-23 (Figure 6).
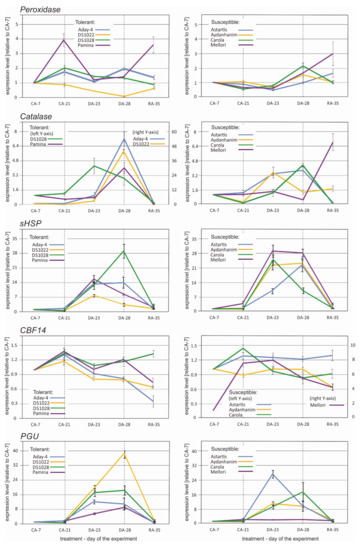
Figure 6.
Expression profiles of peroxidase, catalase, sHSP, CBF14, and PGU inhibitor-like genes during acclimation to cold (CA-7), after 3-week cold acclimation (CA-21), during de-acclimation (DA-23), after 7-day de-acclimation (DA-28), and during re-acclimation to cold (RA-35) in tolerant (left) and susceptible (right) to de-acclimation barley accessions. The de-acclimation period is indicated between the vertical dashed lines.
Accumulation of sHSP transcripts was distinctly higher at DA-23 and DA-28 in relation to that of CA-21 in all tested barley accessions, regardless of their tolerance to de-acclimation (Figure 6). However, expression drastically decreased after one week of re-acclimation in all accessions. Three types of expression patterns were distinguishable for sHSP: The same level of sHSP transcripts at the DA-23 and DA-28 time points (Aday-4, Astartis, and Mellori), an abrupt increase in expression at the beginning of de-acclimation followed by a slight decrease after seven days of de-acclimation (Pamina, Carola, and DS1022), and a gradual increase in sHSP transcript accumulation from the beginning of de-acclimation and peaking after seven days of de-acclimation (Aydanhanim and DS1028) (Figure 6). The expression of cbf14 did not change or slightly decreased at the DA-23 and DA-28 time points in relation to CA-21 in all tested barley accessions (Figure 6).
Higher accumulation of PGU inhibitor-like transcripts during and after de-acclimation in relation to CA-21 was observed in all tested barley accessions except Mellori (Figure 6). In Mellori, the transcript level did not change in response to de-acclimation. Three patterns of expression of the PGU inhibitor-like protein-coding gene were observed among the remaining seven accessions: A significant increase in transcript level at DA-23 with the level maintained after seven days of de-acclimation (Aday-4, Astartis, and DS1028), a gradual increase in transcript level starting from DA-23 with the peak at DA-28 (Pamina, Carola, and DS1022), and a significant increase in transcript level at DA-23 with reduced accumulation of transcripts observed after completion of de-acclimation (Aydanhanim) (Figure 6).
An apparent increase in ascorbate peroxidase activity after de-acclimation (DA-28) compared with that under cold acclimation (CA-21) was observed in five (Aday-4, DS1022, Pamina, Astartis, and Mellori) of the eight tested barley accessions (Figure 7). In four of the former accessions, ascorbate peroxidase activity decreased or remained unchanged at the beginning of de-acclimation (DA-23). In Astartis ascorbate peroxidase activity had already started to increase at DA-23. No changes in the activity of this enzyme owing to de-acclimation were observed in DS1028. In Aydanhanim the activity rose at DA-23, but drastically decreased after seven days of de-acclimation (DA-28). The pattern of changes in ascorbate peroxidase activity caused by de-acclimation in Carola was the opposite to that observed in Aydanhanim –activity decreased significantly at DA-23 and at DA-28 returned to a level similar to that recorded at CA-21 (Figure 7).
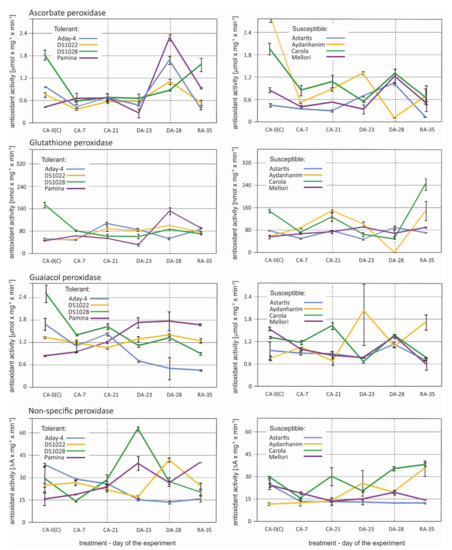
Figure 7.
Changes in antioxidant activity of peroxidases: Ascorbate, glutathione, guaiacol, and nonspecific peroxidase in six time points—before cold acclimation (CA-0 (C)), during acclimation to cold (CA-7), after 3-week cold acclimation (CA-21), during de-acclimation (DA-23), after 7-day de-acclimation (DA-28), and during re-acclimation to cold (RA-35) in tolerant (left) and susceptible (right) to de-acclimation barley accessions. The de-acclimation period is indicated between the vertical dashed lines.
An increase in glutathione peroxidase activity after de-acclimation (DA-28) in relation to that of cold-acclimated plants (CA-21) was observed in three tested barley accessions—DS1022, DS1028, and Pamina—which were all classified as tolerant to de-acclimation in previous experiments (data not published) (Figure 7). In Pamina, this increase in activity was most distinct and was preceded by a decrease in activity at the beginning of de-acclimation (DA-23). In Astartis, the glutathione peroxidase activity decreased initially during de-acclimation but returned to the CA-21 level after seven days of de-acclimation. In Mellori, a slight initial increase in activity was observed at DA-23, followed by a decrease leading to the same level of activity recorded at CA-21. In Aydanhanim, Aday-4, and Carola, glutathione peroxidase activity decreased during and after de-acclimation compared with cold-acclimated plants. The decrease was most drastic in Aydanhanim (Figure 7).
Changes in guaiacol peroxidase activity caused by de-acclimation showed different patterns among the barley accessions (Figure 7). In Aday-4, DS1028, and Carola, activity was lower during and after de-acclimation compared with that recorded for cold-acclimated plants. In DS1028 and Carola, activity rose at DA-28 compared with that at DA-23, but did not attain the level of activity observed after cold acclimation (CA-21). In Astartis and Mellori, a slight decrease in guaiacol peroxidase activity was observed at the beginning of de-acclimation but was followed by a considerable increase after one week of de-acclimation, attaining higher activity than that observed in cold-acclimated plants. In Aydanhanim, DS1022, and Pamina, the guaiacol peroxidase activity was higher during (DA-23) and after (DA-28) de-acclimation than after cold acclimation (CA-21). In DS1022 and Pamina, the activities recorded at the DA-23 and DA-28 time points were similar, whereas in Aydanhanim, the guaiacol peroxidase activity at DA-28 was distinctly lower than that at DA-23 (Figure 7).
The pattern of nonspecific peroxidase activity differed among all of the tested barley accessions, but some similarities were observed (Figure 7). The activity increased initially during de-acclimation in DS1028 and Pamina, then decreased to a level similar to that recorded for cold-acclimated plants after seven days of de-acclimation. The profile of changes caused by de-acclimation was similar for Aydanhanim, but the decrease at DA-28 was smaller, but the activity remained higher at DA-28 than in CA-21. In Mellori nonspecific peroxidase activity gradually increased owing to de-acclimation and decreased rapidly during re-acclimation to cold. In Carola and DS1022, the initial decrease in nonspecific peroxidase activity observed at DA-23 was followed by a rapid increase at DA-28, resulting in higher activity than that recorded in CA-21. In Aday-4 a decrease in nonspecific peroxidase activity during and after de-acclimation was observed. No changes in nonspecific peroxidase activity caused by de-acclimation were observed for Astartis (Figure 7).
The profile of changes in formate dehydrogenase activity caused by de-acclimation was similar for five barley accessions (Figure 8). In Astartis, Aydanhanim, Carola, DS1028, and Pamina, activity increased considerably in the initial stage of de-acclimation (DA-23) and decreased rapidly after seven days of de-acclimation. The decrease led to activity lower than that observed in CA-21 in four of the accessions. In Aday-4 and Mellori, the formate dehydrogenase activity was lower during and after de-acclimation compared with that of cold-acclimated plants. The activity remained low also during re-acclimation to cold. In DS1022, formate dehydrogenase activity increased during and after de-acclimation, and the trend towards increase continued during re-acclimation (Figure 8).
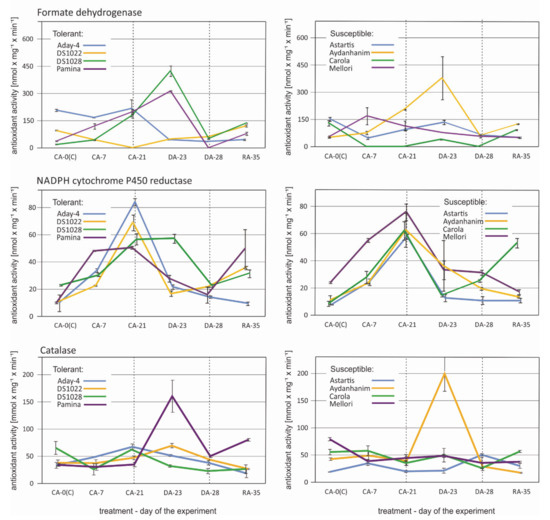
Figure 8.
Changes in antioxidant activity of selected enzymes: Formate dehydrogenase, NADPH cytochrome P450 reductase, and catalase in six time points—before cold acclimation (K), during acclimation to cold (CA-0 (C)), during acclimation to cold (CA-7), after 3-week cold acclimation (CA-21), during de-acclimation (DA-23), after 7-day de-acclimation (DA-28), and during re-acclimation to cold (RA-35)in tolerant (left) and susceptible (right) to de-acclimation barley accessions. The de-acclimation period is indicated between the vertical dashed lines.
The typical pattern of change in NADPH cytochrome P450 reductase activity was a significant increase in response to cold acclimation (CA-21) in all tested barley accessions (Figure 8). In some accessions (Carola, Mellori, and Pamina), the increase was notable at the beginning of cold acclimation (CA-7). In DS1028, the activity remained high at the beginning of de-acclimation and decreased rapidly by the end of de-acclimation treatment. In the remaining accessions, NADPH cytochrome P450 reductase activity decreased abruptly in the initial stage of de-acclimation (DA-23). A slight increase in activity by the end of de-acclimation was observed in Carola and DS1022, and this trend continued during re-acclimation to cold (Figure 8).
Four accessions, namely, Aydanhanim, Carola, DS1022, and Pamina, displayed an increase in catalase activity induced by de-acclimation (DA-23) followed by a substantial decrease after one week of de-acclimation (DA-28; Figure 8). This pattern was much more pronounced in Aydanhanim, DS1022, and Pamina than in Carola. Astartis also showed an increase in catalase activity caused by de-acclimation, but only by the end of the treatment (DA-28). Mellori was the only cultivar to show no response in catalase activity to de-acclimation. Aday-4 and DS1028 showed a steady decrease in catalase caused activity by de-acclimation treatment (Figure 8).
3. Discussion
Limited information is available on the molecular control of the response to de-acclimation in herbaceous plants. To the best of our knowledge, only one previous study has examined control at the DNA level using genome-wide association mapping [17], and that study was performed on a dicotyledonous species. Furthermore, few proteomic studies have explored changes associated with de-acclimation [18,19]. The majority of transcriptomic analyses, which represent the most common molecular investigations of de-acclimation, have used Arabidopsis thaliana as the experimental material [20,21,22,23,24]. Arabidopsis is a model plant with limited relevance to cereals. The conditions used for cold acclimation and de-acclimation in previous studies are not entirely relevant to the field conditions under which cereals are grown. Studies of other plant species, including grasses, also have employed a broad range of approaches to de-acclimation treatments [6,25,26,27,28,29,30,31,32,33,34,35]. De-acclimation conditions applied in previous studies often more closely resemble spring warming than mid-winter warm spell, using equal night and day lengths or longer days/shorter nights sometimes accompanied by relatively high temperatures [6,25,28,35]. Moreover, most of these studies describe physiological and biochemical changes caused by de-acclimation in herbaceous plants, but not their molecular background.
In the only previous study of the molecular background of changes caused by de-acclimation in barley available to date, microRNAs isolated during de-acclimation were identified [28]. In that study, the most significant number of differentially expressed microRNAs was observed on the sixth day of cold de-acclimation, which corresponds to seven days of de-acclimation in the present RNAseq analysis. Although drawing deductions from the applied de-acclimation treatment, the previous study is actually concerned with spring-type de-acclimation events, and one of the two barley cultivars studied is a spring cultivar. Nevertheless, some of the results reported are similar to those obtained in the current study. MicroRNAs targeting two peroxidases and 15 other oxidoreductases were detected in the winter barley cultivar Nure [28], which is in agreement with four peroxidase- and seven other oxidoreductase-coding transcripts identified in the present study. In addition, C-repeat binding factor (CBF), late embryogenesis abundant (LEA), and auxin response protein-encoding genes identified in the present study were previously recognized as targeted by microRNAs that were differentially expressed in response to de-acclimation [28].
In the current study, a large number of transcripts were associated with the response to de-acclimation both in de-acclimation-tolerant and -susceptible barley accessions under FDR < 0.05. A considerable number of these transcripts remained significant at FDR < 0.01. In contrast to some studies [18], in which most of the changes detected during de-acclimation were simply opposite to changes observed during acclimation to cold, the present study showed that a higher number of transcripts are characteristic only to de-acclimation or only to cold acclimation than are common to both responses (but are regulated in the opposite direction) (Figure 4). Furthermore, we showed that most DEGs associated specifically with de-acclimation in barley differs between de-acclimation-susceptible and -tolerant accessions (Figure 5). The DEG analysis also revealed a substantially higher number of de-acclimation-induced expression changes in de-acclimation-susceptible accessions than in de-acclimation-tolerant cultivars (Figure 5). These findings may indicate that the deciding factor determining the survival of frost events after a mid-winter warm period is not mechanisms that confer tolerance, but rather an insensitivity to temperature rise, which triggers a set of metabolic or developmental changes in de-acclimation-susceptible accessions associated with up- and downregulation of genes. The differences in the number of DEGs associated with de-acclimation and the scarcity of common transcripts suggest that de-acclimation-tolerant and-susceptible genotypes exhibit distinct genetic responses to mid-winter active de-acclimation, which could select de-acclimation-tolerant genotypes.
Curiously, in the present study, no significant GO enrichment terms in the “cell component” category were identified. Previous reports focusing on a cell component, namely, the plasma membrane, have examined aspects of plant de-acclimation [18]. The present GO analysis identified photosynthesis-related molecular functions and biological processes as the most highly enriched categories. The role of photosynthesis in response to de-acclimation has been discussed previously by several authors [6,21,22,30], and transcripts of photosynthesis-related genes have been identified in several transcriptomic studies [20,22,23,36]. However, the present GO enrichment analysis did not entirely correspond to the results of a more thorough bioinformatic analysis leading to the annotation of specific genes (Table 2).
Although the gene annotation performed in the present study also revealed a large number of DEGs involved in photosynthesis or associated with chloroplasts, a distinct overrepresentation of genes encoding oxidoreductases, especially peroxidases, was noted, which the GO analysis did not reveal. It was previously suggested that redox enzymes might play a crucial role in the de-acclimation response [4,27], thus associating the susceptibility to freezing after a warm period, with reduced tolerance to oxidative stress. This suggestion would imply the downregulation rather than upregulation of genes encoding antioxidant enzymes, but the results from the present DEG analysis revealed the upregulation of all genes from this group (Table 2). Similar results were obtained previously [21,22,28]. In the present study, the significant de-acclimation-related DEGs encoding oxidoreductases were only identified among the de-acclimation-susceptible barley cultivars. Given that these DEGs were also overrepresented compared with other annotated genes, the results indicate that susceptibility to mid-winter de-acclimation in barley is predominantly caused by overly rapid activation of defense mechanisms against reactive oxygen species. Perhaps excessively early mobilization of oxidative stress defense mechanisms results in the plant not responding sufficiently quickly to the real threat of freezing temperatures. This hypothesis is supported by most RT-qPCR and oxidoreductase activity results observed at the RA-35 (re-acclimation) time point in this study, wherein the transcript abundance and enzyme activity decreased rapidly after peaking during de-acclimation treatment (Figure 6 and Figure 7).
However, it was previously suggested that the upregulation of gene expression is followed by post-transcriptional suppression of antioxidants during de-acclimation, leading to reduction of stress tolerance in barley [28]. As already mentioned, a larger majority of DEGs associated with de-acclimation was detected in the susceptible group of cultivars compared with the de-acclimation-tolerant cultivars, and most of those DEGs were upregulated. This result suggests the superiority of genotypes that do not show a response to de-acclimation. The genotypes that de-acclimate may initiate post-stress recovery mechanisms, hence the upregulation of antioxidant enzyme-coding genes, but also other stress-related and cytoskeleton-related genes identified in the current study. The GO enrichment analysis also revealed upregulation of genes associated with ATP binding and protein phosphorylation (Table 1, Figure S3), which suggests these genes participate in post-transcriptional protein modifications. The changes may relate to protein reprogramming owing to the temperature rise [37,38,39]. This response also emphasizes the difference in perception of the two basic types of de-acclimation—active (mid-winter) and passive (spring). Spring warming events also involve recovery mechanisms, but predominantly trigger signals for plant development [4], and almost no plant development-related genes were annotated in the present study (Table 2). Furthermore, downregulation of genes involved in the control of ATP synthesis and energy coupled proton transport (Table 1, Figure S5) might reflect decreased energy demand, which normally increases during acclimation to cold. A lowered requirement for energy also suggests that no rapid growth/developmental processes are triggered in actively de-acclimated barley plants. The reason may relate to day length, which is markedly shorter in mid-winter than in early spring, and night temperatures, which are lower in winter compared with those in early spring.
Among annotated transcripts revealed to be associated with the de-acclimation response in the current study were LEA-coding genes (Table 2). Previous studies have noted an association of LEA proteins with de-acclimation [25,28,31]. Identification of auxin response protein-coding genes among the DEGs upregulated in de-acclimation-tolerant barley accessions in the present study is consistent with previous reports [20,22,23,28]. Many genes associated with stress response in plants were identified in the present study, including oxidoreductase-coding genes, heat shock protein-coding genes, pathogen response-associated genes (of which the core response is similar to the freezing stress response), and freezing stress-related genes, namely, CBFs. Genes belonging to all of these groups, especially CBF genes, were previously reported as associated with de-acclimation in herbaceous plants [20,22,23,24,28].
The results of the present RT-qPCR experiments confirmed the changes in expression of the selected genes associated with the response to de-acclimation in the majority of cases (Figure 6). No expected changes related to mid-winter de-acclimation were observed only in the cbf14 expression profile (Figure 6). This result may be associated with the specific time-dependent character of cbf gene expression, which usually peaks within the first 12–24 h of stress treatment [40]. There is a possibility that the timing of collection of samples for the RNAseq and RT-qPCR experiments differed sufficiently to affect the detection of their expression despite our careful efforts to repeat the experimental conditions. For the remainder of the selected genes, namely, peroxidase, catalase, sHSP, and PGU inhibitor-like coding genes, upregulation during and after seven days of de-acclimation was observed in most of the barley accessions irrespective of their tolerance to mid-winter de-acclimation (Figure 6). These results may partly reflect that the comparisons made for detecting differential transcripts using Venn diagrams [41] showed only DEGs common for all of the four de-acclimation-tolerant or four susceptible barley accessions. In addition, certain DEGs could also be expressed in some members of the other group. That was, indeed, the case for all of the RT-qPCR-tested genes where the gene identified as differentially expressed in response to de-acclimation in all of the four susceptible genotypes was also differentially expressed in one (cbf14), two (Peroxidase, Catalase, and sHSP), or three (PGU inhibitor-like) tolerant accessions (data not shown).
The overrepresentation of different types of oxidoreductase gene transcripts among the DEGs responsive to de-acclimation in barley showed the necessity for an enzyme activity analysis of certain selected oxidoreductases, mostly peroxidases, under the same conditions as those applied for the RT-qPCR experiment. The changes observed in the activity of the selected enzymes did not correspond or corresponded only partially to the changes in the number of accumulated transcripts of genes encoding peroxidases and catalase (Figure 7 and Figure 8). As described previously [42], the number of accumulated transcripts might not comply with the amount of accumulated protein for various reasons. Additional proteomic analysis would be helpful in the future to provide a comprehensive overview of the role of oxidoreductase enzymes in response to mid-winter de-acclimation in barley.
In conclusion, although certain portions of the response to mid-winter warm spell-induced de-acclimation are the reverse of the response to cold acclimation, the molecular backgrounds of these two processes’ predominantly differ. The present study provides novel evidence for the distinct molecular regulation of cold acclimation and de-acclimation. In addition, mid-winter active de-acclimation is regulated differently from that of passive spring de-acclimation, which is associated with developmental changes. De-acclimation in mid-winter is indicated to be perceived as an opportunity to regenerate after stress. Unfortunately, it is competitive to remain in the cold-acclimated state, which can be deduced from the majority of genes for which expression is activated under de-acclimation. Antioxidant enzymes and other oxidoreductases seem to play a crucial role in the process of active de-acclimation, but there is still insufficient evidence to link their abundance with the degree of barley tolerance to de-acclimation. Photosynthesis-related processes may be of fundamental importance during de-acclimation, as deduced from GO enrichment analysis, but unambiguous confirmation is required. Nonetheless, the present study demonstrates that the response to mid-winter de-acclimation is far more expansive in de-acclimation-susceptible cultivars, suggesting that the key to de-acclimation tolerance is a passive or muted response to the rise in temperature.
4. Materials and Methods
4.1. Plant Material and Growth Conditions
Four winter barley lines and cultivars tolerant to de-acclimation (Aday-4, DS1022, DS1028, and Pamina) and four de-acclimation-susceptible accessions (Aydanhanim, Astartis, Carola, and Mellori) selected previously (Wójcik-Jagła and Rapacz, unpublished) were used in this study. Seeds were sown in plastic pots (5 dm3, one genotype per pot and one pot per genotype, 12 seeds per genotype) filled with a mixture of universal garden soil substrate (Ekoziem, Jurkow, Poland) and sand (1:1, v/v). The pots were transferred to a growth chamber after sowing (darkness, 25 °C/17 °C [day/night]). Irradiance of 400 μmol m−2 s−1 (HPS lamps, SON-T+ AGRO, Philips, Brussels, Belgium) under a photoperiod of 12 h/12 h (light/dark) was provided when the seedlings started to emerge. The temperature was reduced to 15 °C/12 °C (day/night) 8 days after sowing. The plants were subjected to 3 weeks cold-hardening 20 days after sowing (4 °C/2 °C [day/night], photoperiod of 9 h/15 h [light/dark], and irradiance of 250 μmol m−2 s−1). After 3 weeks acclimation to cold, the plants were subjected to de-acclimation (7 days of 12 °C/5 °C [day/night]).
4.2. RNA Isolation
Leaves from each genotype were sampled before (CA-0 (C)) and after cold acclimation (CA-21), and after de-acclimation (DA-28) in three biological replicates (leaves from three different plants). Samples were immediately frozen in liquid nitrogen and stored at −80 °C until use. Total RNA was isolated from 72 leaf samples (0.03–0.05 g from the middle portion of the youngest fully developed leaf) using the RNeasy Plant Mini Kit (Qiagen, Hilden, Germany). The quantity and purity of RNA was checked using a UV-Vis Q5009 spectrophotometer (Quawell, San Jose, CA, USA). RNA integrity was tested using a 2100 Bioanalyzer (Agilent Technologies, Santa Clara, CA, USA).
4.3. RNA Sequencing and Differential Expression Analysis
Total RNA from the 72 samples was submitted to Genomed (Warsaw, Poland) for sequencing. The RNA was subjected to mRNA isolation using the NEBNext Poly(A) mRNA Magnetic Isolation Module (New England Biolabs Inc., Ipswich, MA, USA). The libraries were prepared with the NEBNext Ultra Directional RNA Library Prep Kit for Illumina (New England Biolabs) and sequenced using a HiSeq 4000 platform (Illumina, San Diego, CA, USA) in PE101 mode. Received raw sequence data were subjected to FastQC analysis to check the quality of reads and presence/absence of adapters [43]. The BAC-based barley reference sequence [44] was used to map the RNA-seq data. Read count and transcripts per million reads mapped data were determined using Kallisto version 0.43.0 software [45]. Differential expression analysis was performed using DeSeq2 [46] to compare the transcriptomes of control (pre-hardening), cold-acclimated, and de-acclimated plants. The FDR was primarily set as <0.05 so as not to overlook interesting but weakly significant interactions, and then reduced to <0.01 to simplify the selection of genes for further verification via RT-qPCR.
Obtained data sets were grouped and contrasted using Venn diagrams [41]. Comparisons were made for control vs. cold-acclimated (CA-0 (C)/CA-21), cold-acclimated vs. de-acclimated (CA-21/DA-28), and de-acclimated vs. control (DA-28/CA-0 (C)) for de-acclimation-tolerant and -susceptible accessions separately and also for common DEGs. The DEGs were then subjected to GO analysis using the AgriGo online toolkit with singular enrichment analysis [47] using the default settings (FDR < 0.05).
The Horvu sequences were annotated to specific proteins using the Uniprot database [48] and aligned to determine similarities with closely related species using the NCBI Blast tool [49].
4.4. Gene Expression Analysis
Five genes were selected for verification of their expression under de-acclimation treatment. The genes were selected on the basis of GO analysis, annotation, and the magnitude of expression changes in response to de-acclimation revealed by differential expression analysis. Primer and probe sequences (Table 3) were designed for these genes using Primer3Plus [50,51] based on consensus sequences (when more than one splicing variant was possible) derived from the EnsemblPlants.org database [52,53]. For the alignment of two splicing variants, the pairwise alignment tool Lalign [54] was used. In comparison, the multiple alignment tool Clustal Omega [55], as well as Kalign [56] were used for aligning three or more variants.

Table 3.
Primer and probe sequences in the expression analysis of selected candidate genes associated with tolerance to de-acclimation in winter barley.
RNA for gene expression analysis was isolated from leaves of the genotypes used for RNAseq using the aforementioned method. The growth, cold acclimation, and de-acclimation conditions were the same as described in Section 4.1, but the plants were also subjected to re-acclimation (same conditions as for cold acclimation but treated for 10 days). Leaves were sampled with three biological replications (leaves from three individual plants) at five time points: CA-7, during cold acclimation (1 week after moving the plants to the hardening conditions); CA-21, after cold acclimation; DA-23, during de-acclimation (2 days after moving the plants to the de-acclimating conditions), DA-28, after de-acclimation; and RA-35, during re-acclimation to cold (after seven days). To receive template cDNA the RNA was subjected to reverse transcription using the QuantiTect Reverse Transcription Kit (Qiagen, Hilden, Germany) reagent set. We used RT-qPCR analysis to determine changes in expression of the selected genes. The reactions were performed using a QuantStudio 3 Real-Time PCR System (Thermo Fisher Scientific, Waltham, MA, USA). Amplification was observed from the increase in fluorescence intensity of SYBRGreen (for reference genes [57]) and 6-carboxyfluorescein (FAM) from TaqMan MGB probes (for analyzed genes [58,59]). The reference genes were the ADP-ribosylation factor 1-like protein (ADP) and S-adenosylmethionine decarboxylase (sAMD) coding genes [11]. The reactions were conducted in three biological replicates for each genotype, each in three instrumental replicates. Each reaction contained 900 nM of each primer, approximately 35 ng cDNA template, and TaqMan™ Gene Expression Master Mix (Thermo Fisher Scientific, Waltham, MA, USA).
The relative level of expression of the analyzed genes was calculated using the modified standard curve method [60]. The expression level during cold hardening (CA-7) normalized in relation to the geometric mean of the internal standard genes’ copy number was used as a reference time point (number of gene copies = 1) for all of the other tested time points. The standard error was calculated for the geometric means of three instrumental replications × three biological replications × two reference genes.
4.5. Analysis of Oxidoreductase Activity
The samples for analysis of oxidoreductase activity were collected at the same time points as for the gene expression analysis plus an additional control time point, CA-0 (C), before cold acclimation. One sample consisted of one fully developed leaf, either the first, second, or third leaf, depending on the developmental stage attained at a particular time point. The weight of the leaves ranged from 0.12 to 0.59 g. Each line at each time point was represented by three biological replications (three leaves from three individual plants). The standard error was calculated for the mean of three repetitions at each time point. The activity of seven enzymes was measured: Ascorbate, glutathione, guaiacol, and nonspecific peroxidases, as well as catalase, formate dehydrogenase, and NADPH-cytochrome P450 reductase.
Leaves were homogenized in 50 mM Tris–HCl buffer (pH 7.8) supplemented with 1 mM EDTA-Na2, 3% polyvinylpyrrolidone, and 1% Protease Inhibitore Coctail (Merck, Darmstadt, Germany). Homogenization buffer was added in the proportion of 6 µL buffer per 1 mg of plant material. The homogenate was centrifuged at 12,000 g for 20 min at 4 °C. The supernatant was used for further analysis. Enzyme activity was measured spectrophotometrically using a Synergy 2 Microplate Reader (BioTek, Winooski, VT, USA). The activity of the enzymes was normalized to the amount of protein. Protein content was determined in accordance with Reference [61], using bovine serum albumin as a standard.
Ascorbate peroxidase activity was determined as the oxidation of ascorbic acid, in accordance with Reference [62]. The reaction mixture (114 µL) consisted of 100 µL buffer (50 mM Tris–HCl buffer, pH 7.8), 5 µL of 590 mM ascorbic acid, 5 µL of 19.6 mM H2O2, and 4 µL leaf extract. Enzyme activity was measured by monitoring the decline in absorbance at 290 nm (extinction coefficient of ascorbic acid was 2.8 mM−1 cm−1). The respective control reaction mixtures contained buffer instead of H2O2 solution. The ascorbate peroxidase activity was expressed as nmol ascorbate mg−1 protein min−1.
Glutathione peroxidase activity was assayed by measuring the decrease in NADPH concentration, in accordance with Reference [63]. The assay mixture (126 µL) contained 100 µL buffer (50 mM Tris–HCl, pH 7.0, 0.5 mM EDTA-Na2, and 1.6 mM NaN3), 5 µL of 19.52 mM reduced glutathione, 5 µL of 2.69 mM NADPH, 5 µL of 3 U mL−1 glutathione reductase, 5 µL of 4.5 mM H2O2, and 6 µL leaf extract. The absorbance was monitored at 340 nm (extinction coefficient of NADPH was 6.2 mM−1 cm−1). The control reaction was performed using the buffer instead of H2O2 solution. The glutathione peroxidase activity was expressed as nmol NADPH mg−1 protein min−1.
The activity of nonspecific peroxidases was measured using two methods exploiting different substrates. Nonspecific peroxidase 1 (Guaiacol peroxidase) activity was determined by measuring the formation of the conjugate of guaiacol, in accordance with Reference [64]. The reaction medium (98 µL) contained 88 µL buffer (0.1 M sodium phosphate buffer, pH 6.5), 4 µL of 2 mM guaiacol, 4 µL of 0.13 M H2O2, and 2 µL leaf extract. The kinetic evolution was measured at 436 nm (extinction coefficient of ascorbic acid was 26.6 mM−1 cm−1 for the conjugate). The guaiacol peroxidase activity was expressed as µmol tetraguaiacol mg−1 protein min−1. Nonspecific peroxidase 2 activity was measured using p-phenylenediamine (pPD) as the substrate, in accordance with Reference [65]. The reaction mixture (249.5 µL) contained 236.5 µL of buffer (0.3 M potassium phosphate buffer, pH 7.8, and 0.1 mM EDTA-Na2), 1.5 µL of 0.5% pPD, 10 µL of 9.8 M H2O2, and 1.5 µL leaf extract. The nonspecific peroxidase 2 activity was calculated as the change in absorbance at 485 nm (∆A) per minute and normalized to micrograms of protein.
Catalase activity was measured based on the rate of H2O2 decomposition at 240 nm (extinction coefficient of H2O2 was 0.04 mM−1 cm−1), in accordance with Reference [66]. The assay mixture (107.5 µL) was composed of 100 µL buffer (0.1 M sodium phosphate buffer, pH 6.5), 5 µL of 134.5 mM H2O2, and 2.5 µL leaf extract. The catalase activity was expressed as mmol H2O2 mg−1 protein min−1.
NADPH-cytochrome P450 reductase activity was determined by measuring the oxidation of cytochrome c (Cyt. c), in accordance with Reference [67]. The assay medium (200 µL) consisted of 166 µL buffer (0.3 M potassium phosphate buffer, pH 7.8, and 0.1 mM EDTA-Na2), 20 µL of 10 mM NADPH, 16 µL of 0.5 mM cytochrome c, and 16 µL leaf extract. The absorbance was monitored at 550 nm (extinction coefficient of the oxidized form of Cyt. c was 21 mM−1 cm−1). The NADPH-cytochrome P450 reductase activity was expressed as nmol Cyt. cox mg−1 protein min−1.
Formate dehydrogenase activity was determined by measuring the production of NADH, in accordance with Reference [68]. The reaction mixture (250 µL) contained 195 µL buffer (0.1 M sodium phosphate buffer, pH 6.5), 25 µL of 0.5 M sodium formate, 25 µL of 10 mM NAD+, and 5 µL leaf extract. The kinetic evolution was measured at 550 nm after incubation at 30 °C (extinction coefficient of NADH was 6.2 mM−1 cm−1). The formate dehydrogenase activity was expressed as nmol NADH mg−1 protein min−1.
Supplementary Materials
Supplementary Materials (Figures S1–S6, Table S1) can be found at https://www.mdpi.com/1422-0067/22/3/1057/s1.
Author Contributions
Conceptualization, M.W.-J.; methodology, M.W.-J., A.D.-G., A.F., P.K. and M.R.; software, A.D.-G.; validation, M.W.-J. and M.R.; formal analysis, M.W.-J. and A.D.-G.; investigation, M.W.-J., A.D.-G., A.F., and P.K.; resources, M.W.-J., A.D.-G., P.K. and M.R.; data curation, M.W.-J. and A.F.; writing—original draft preparation, M.W.-J.; writing—review and editing, A.D.-G., P.K. and M.R.; visualization, M.W.-J. and M.R.; supervision, M.R.; project administration, M.W.-J.; funding acquisition, M.W.-J. All authors have read and agreed to the published version of the manuscript.
Funding
This study was funded by National Science Centre in Poland, grant no 2016/21/D/NZ9/01318.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
Acknowledgments
We thank Fulvia Rizza from Council for Agricultural Research and Agricultural Economy Analysis (CREA) in Italy for providing seeds from 39 European cultivars of winter and facultative barley for this study, as well as Danko Hodowla Roślin Ltd. for providing seeds of winter barley’s Polish advanced breeding lines and cultivars. We also thank Robert McKenzie, from Edanz Group (https://en-author-services.edanz.com/ac), for editing a draft of this manuscript.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Abbreviations
| RNAseq | RNA sequencing |
| RT-qPCR | Quantitative (real-time) PCR performed on cDNA samples |
| CA | Cold acclimation |
| DA | Cold de-acclimation |
| RA | Re-acclimation to cold |
| FDR | False discovery rate |
| DEG | Differentially expressed gene |
| GO | Gene ontology |
| BLAST | Basic Local Alignment Search Tool |
| sHSP | Small Heat Shock Protein |
| PGU | PolyGalactoUronase |
| LEA | Late Embryogenesis Abundant |
References
- Rapacz, M.; Ergon, Å.; Höglind, M.; Jørgensen, M.; Jurczyk, B.; Østrem, L.; Rognli, O.A.; Tronsmo, A.M. Overwintering of herbaceous plants in a changing climate. Still more questions than answers. Plant Sci. 2014, 225, 34–44. [Google Scholar] [CrossRef] [PubMed]
- Galiba, G.; Vágújfalvi, A.; Li, C.; Soltész, A.; Dubcovsky, J. Regulatory genes involved in the determination of frost tolerance in temperate cereals. Plant Sci. 2009, 176, 12–19. [Google Scholar] [CrossRef]
- Thomashow, M.F. Role of Cold-Responsive Genes in Plant Freezing Tolerance. Plant Physiol. 1998, 118, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Kalberer, S.R.; Wisniewski, M.; Arora, R. Deacclimation and reacclimation of cold-hardy plants: Current understanding and emerging concepts. Plant Sci. 2006, 171, 3–16. [Google Scholar] [CrossRef]
- Pagter, M.; Arora, R. Winter survival and deacclimation of perennials under warming climate: Physiological perspectives. Physiol. Plant. 2013, 147, 75–87. [Google Scholar] [CrossRef]
- Rapacz, M. Regulation of frost resistance during cold de-acclimation and re-acclimation in oilseed rape. A possible role of PSII redox state. Physiol. Plant. 2002, 115, 236–243. [Google Scholar] [CrossRef]
- Gu, L.; Hanson, P.J.; Post, W.M.; Kaiser, D.P.; Yang, B.; Nemani, R.; Pallardy, S.G.; Meyers, T. The 2007 Eastern US Spring Freeze: Increased Cold Damage in a Warming World? Bioscience 2008, 58, 253–262. [Google Scholar] [CrossRef]
- Hömmö, L.M. Hardening of Some Winter Wheat (Triticum aestivum L.), Rye (Secale cereals L.), Triticale (× Triticosecale Wittmack) and Winter Barley (Hordeum vulgare L.) Cultivars During Autumn and the Final Winter Survival in Finland. Plant Breed. 1994, 112, 285–293. [Google Scholar] [CrossRef]
- Rizza, F.; Crosatti, C.; Stanca, A.M.; Cattivelli, L. Studies for assessing the influence of hardening on cold tolerance of barley genotypes. Euphytica 1994, 75, 131–138. [Google Scholar] [CrossRef]
- Tyrka, M.; Rapacz, M.; Fiust, A.; Wójcik-Jaglła, M. Quantitative trait loci mapping of freezing tolerance and photosynthetic acclimation to cold in winter two- and six-rowed barley. Plant Breed. 2015, 134, 271–282. [Google Scholar] [CrossRef]
- Fiust, A.; Rapacz, M. Downregulation of three novel candidate genes is important for freezing tolerance of field and laboratory cold acclimated barley. J. Plant Physiol. 2020, 244, 153049. [Google Scholar] [CrossRef] [PubMed]
- Jeknić, Z.; Pillman, K.A.; Dhillon, T.; Skinner, J.S.; Veisz, O.; Cuesta-Marcos, A.; Hayes, P.M.; Jacobs, A.K.; Chen, T.H.H.; Stockinger, E.J. Hv-CBF2A overexpression in barley accelerates COR gene transcript accumulation and acquisition of freezing tolerance during cold acclimation. Plant Mol. Biol. 2014, 84, 67–82. [Google Scholar] [CrossRef] [PubMed]
- Zhu, B.; Choi, D.W.; Fenton, R.; Close, T.J. Expression of the barley dehydrin multigene family and the development of freezing tolerance. Mol. Gen. Genet. Mgg 2000, 264, 145–153. [Google Scholar] [CrossRef] [PubMed]
- Giorni, E.; Crosatti, C.; Baldi, P.; Grossi, M.; Marè, C.; Stanca, A.M.; Cattivelli, L. Cold-regulated gene expression during winter in frost tolerant and frost susceptible barley cultivars grown under field conditions. Euphytica 1999, 106, 149–157. [Google Scholar] [CrossRef]
- Rapacz, M.; Wolanin, B.; Hura, K.; Tyrka, M. The effects of cold acclimation on photosynthetic apparatus and the expression of COR14b in four genotypes of barley (Hordeum vulgare) contrasting in their tolerance to freezing and high-light treatment in cold conditions. Ann. Bot. 2008, 101, 689–699. [Google Scholar] [CrossRef]
- Soltész, A.; Smedley, M.; Vashegyi, I.; Galiba, G.; Harwood, W.; Vágújfalvi, A. Transgenic barley lines prove the involvement of TaCBF14 and TaCBF15 in the cold acclimation process and in frost tolerance. J. Exp. Bot. 2013, 64, 1849–1862. [Google Scholar] [CrossRef]
- Horvath, D.P.; Zhang, J.; Chao, W.S.; Mandal, A.; Rahman, M. Genome-Wide Association Studies and Transcriptome Changes during Acclimation and Deacclimation in Divergent Brassica napus Varieties. Int. J. Mol. Sci. 2020, 21, 9148. [Google Scholar] [CrossRef]
- Miki, Y.; Takahashi, D.; Kawamura, Y.; Uemura, M. Temporal proteomics of Arabidopsis plasma membrane during cold- and de-acclimation. J. Proteom. 2019, 197, 71–81. [Google Scholar] [CrossRef]
- Nakaminami, K.; Matsui, A.; Nakagami, H.; Minami, A.; Nomura, Y.; Tanaka, M.; Morosawa, T.; Ishida, J.; Takahashi, S.; Uemura, M.; et al. Analysis of Differential Expression Patterns of mRNA and Protein During Cold-acclimation and De-acclimation in Arabidopsis. Mol. Cell Proteom. 2014, 13, 3602–3611. [Google Scholar] [CrossRef]
- Firtzlaff, V.; Oberländer, J.; Geiselhardt, S.; Hilker, M.; Kunze, R. Pre-exposure of Arabidopsis to the abiotic or biotic environmental stimuli “chilling” or “insect eggs” exhibits different transcriptomic responses to herbivory. Sci. Rep. 2016, 6, 1–13. [Google Scholar] [CrossRef]
- Juszczak, I.; Cvetkovic, J.; Zuther, E.; Hincha, D.K.; Baier, M. Natural variation of cold deacclimation correlates with variation of cold-acclimation of the plastid antioxidant system in Arabidopsis thaliana accessions. Front. Plant Sci. 2016, 7, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Oono, Y.; Seki, M.; Satou, M.; Iida, K.; Akiyama, K.; Sakurai, T.; Fujita, M.; Yamaguchi-Shinozaki, K.; Shinozaki, K. Monitoring expression profiles of Arabidopsis genes during cold acclimation and deacclimation using DNA microarrays. Funct. Integr. Genom. 2006, 6, 212–234. [Google Scholar] [CrossRef] [PubMed]
- Pagter, M.; Alpers, J.; Erban, A.; Kopka, J.; Zuther, E.; Hincha, D.K. Rapid transcriptional and metabolic regulation of the deacclimation process in cold acclimated Arabidopsis thaliana. BMC Genom. 2017, 18, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Zuther, E.; Juszczak, I.; Ping Lee, Y.; Baier, M.; Hincha, D.K. Time-dependent deacclimation after cold acclimation in Arabidopsis thaliana accessions. Sci. Rep. 2015, 5, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Borovik, O.A.; Pomortsev, A.V.; Korsukova, A.V.; Polyakova, E.A.; Fomina, E.A.; Zabanova, N.S.; Grabelnych, O.I. Effect of Cold Acclimation and Deacclimation on the Content of Soluble Carbohydrates and Dehydrins in the Leaves of Winter Wheat. J. Stress Physiol. Biochem. 2019, 15, 62–67. [Google Scholar]
- Rapacz, M.; Jurczyk, B.; Sasal, M. Deacclimation may be crucial for winter survival of cereals under warming climate. Plant Sci. 2017, 256, 5–15. [Google Scholar] [CrossRef]
- Rys, M.; Pociecha, E.; Oliwa, J.; Ostrowska, A.; Jurczyk, B.; Saja, D.; Janeczko, A. Deacclimation of winter oilseed rape-insight into physiological changes. Agronomy 2020, 10. [Google Scholar] [CrossRef]
- Chen, F.; He, J.; Jin, G.; Chen, Z.H.; Dai, F. Identification of novel microRNAs for cold deacclimation in barley. Plant Growth Regul. 2020, 92, 389–400. [Google Scholar] [CrossRef]
- Espevig, T.; Höglind, M.; Aamlid, T.S. Dehardening resistance of six turfgrasses used on golf greens. Env. Exp. Bot. 2014, 106, 182–188. [Google Scholar] [CrossRef]
- Hoffman, L.; DaCosta, M.; Scott Ebdon, J. Examination of cold deacclimation sensitivity of annual bluegrass and creeping bentgrass. Crop Sci. 2014, 54, 413–420. [Google Scholar] [CrossRef]
- Hoffman, L.; DaCosta, M.; Bertrand, A.; Castonguay, Y.; Ebdon, J.S. Comparative assessment of metabolic responses to cold acclimation and deacclimation in annual bluegrass and creeping bentgrass. Env. Exp. Bot. 2014, 106, 197–206. [Google Scholar] [CrossRef]
- Jørgensen, M.; Østrem, L.; Höglind, M. De-hardening in contrasting cultivars of timothy and perennial ryegrass during winter and spring. Grass Forage Sci. 2010, 65, 38–48. [Google Scholar] [CrossRef]
- Li, X.; Cai, J.; Liu, F.; Zhou, Q.; Dai, T.; Cao, W.; Jiang, D. Wheat plants exposed to winter warming are more susceptible to low temperature stress in the spring. Plant Growth Regul. 2015, 77, 11–19. [Google Scholar] [CrossRef]
- Rapacz, M.; Plazek, A.; Niemczyk, E. Frost de-acclimation of barley (Hordeum vulgare L.) and meadow fescue (Festuca pratensis Huds.). Relationship between soluble carbohydrate content and resistance to frost and the fungal pathogen Bipolaris sorokiniana (Sacc.) Shoem. Ann. Bot. 2000, 86, 539–545. [Google Scholar] [CrossRef]
- Rapacz, M. Cold-deacclimation of oilseed rape (Brassica napus var. oleifera) in response to fluctuating temperatures and photoperiod. Ann. Bot. 2002, 89, 543–549. [Google Scholar] [CrossRef]
- Nagano, A.J.; Kawagoe, T.; Sugisaka, J.; Honjo, M.N.; Iwayama, K.; Kudoh, H. Annual transcriptome dynamics in natural environments reveals plant seasonal adaptation. Nat. Plants 2019, 5, 74–83. [Google Scholar] [CrossRef]
- Chinnusamy, V.; Zhu, J.K. Epigenetic regulation of stress responses in plants. Curr. Opin. Plant Biol. 2009, 12, 133–139. [Google Scholar] [CrossRef]
- Millar, A.H.; Heazlewood, J.L.; Giglione, C.; Holdsworth, M.J.; Bachmair, A.; Schulze, W.X. The Scope, Functions, and Dynamics of Posttranslational Protein Modifications. Annu. Rev. Plant Biol. 2019, 70, 119–151. [Google Scholar] [CrossRef]
- Wang, Q.L.; Chen, J.H.; He, N.Y.; Guo, F.Q. Metabolic reprogramming in chloroplasts under heat stress in plants. Int. J. Mol. Sci. 2018, 19, 849. [Google Scholar] [CrossRef]
- Skinner, J.S.; Von Zitzewitz, J.; Szűcs, P.; Marquez-Cedillo, L.; Filichkin, T.; Amundsen, K.; Stockinger, E.J.; Thomashow, M.F.; Chen, T.H.H.; Hayes, P.M. Structural, functional, and phylogenetic characterization of a large CBF gene family in barley. Plant Mol. Biol. 2005, 59, 533–551. [Google Scholar] [CrossRef]
- Oliveros, J.C. Venny. An Interactive Tool for Comparing Lists with Venn Diagrams. 2007. Available online: https://bioinfogp.cnb.csic.es/tools/venny_old/venny.php (accessed on 2 April 2019).
- Wójcik-Jagła, M.; Rapacz, M.; Dubas, E.; Krzewska, M.; Kopeć, P.; Nowicka, A.; Ostrowska, A.; Malaga, S.; Żur, I. Candidate genes for freezing and drought tolerance selected on the basis of proteome analysis in doubled haploid lines of barley. Int. J. Mol. Sci. 2020, 21, 2062. [Google Scholar] [CrossRef] [PubMed]
- Braham Bioinformatics. FastQC. Available online: https://www.bioinformatics.babraham.ac.uk/projects/fastqc/ (accessed on 3 December 2018).
- Mascher, M.; Gundlach, H.; Himmelbach, A.; Beier, S.; Twardziok, S.O.; Wicker, T.; Radchuk, V.; Dockter, C.; Hedley, P.E.; Russell, J. A chromosome conformation capture ordered sequence of the barley genome. Nature 2017, 544, 427–433. [Google Scholar] [CrossRef] [PubMed]
- Bray, N.L.; Pimentel, H.; Melsted, P.; Pachter, L. Near-optimal probabilistic RNA-seq quantification. Nat. Biotechnol. 2016, 34, 525–527. [Google Scholar] [CrossRef] [PubMed]
- Love, M.; Anders, S.; Huber, W. Differential analysis of count data–the DESeq2 package. Genome Biol 2014, 15, 10–1186. [Google Scholar]
- AgriGo. Volume 2. Available online: http://systemsbiology.cau.edu.cn/agriGOv2/specises_analysis.php?&SpeciseID=23&latin=Hordeum_vulgare (accessed on 5 January 2019).
- UniProt. Available online: https://www.uniprot.org/uniprot/ (accessed on 10 March 2019).
- Basic Local Alignment Search Tool. Available online: https://blast.ncbi.nlm.nih.gov/Blast.cgi (accessed on 5 April 2019).
- Primer3Plus. Available online: https://primer3plus.com/cgi-bin/dev/primer3plus.cgi (accessed on 20 October 2019).
- Untergasser, A.; Cutcutache, I.; Koressaar, T.; Ye, J.; Faircloth, B.C.; Remm, M.; Rozen, S.G. Primer3--new capabilities and interfaces. Nucleic Acids Res. 2012, 40, e115. [Google Scholar] [CrossRef]
- EnsemblPlants. Available online: https://plants.ensembl.org/index.html (accessed on 5 September 2019).
- Howe, K.L.; Contreras-Moreira, B.; De Silva, N.; Maslen, G.; Akanni, W.; Allen, J.; Alvarez-Jarreta, J.; Barba, M.; Bolser, D.M.; Cambell, L.; et al. Ensembl Genomes 2020—enabling non-vertebrate genomic research. Nucleic Acids Res. 2020, 48, D689–D695. [Google Scholar] [CrossRef]
- EMBL-EBI. Lalign. Available online: https://www.ebi.ac.uk/Tools/psa/lalign/ (accessed on 2 September 2019).
- EMBL-EBI. Clustal Omega. Available online: https://www.ebi.ac.uk/Tools/msa/clustalo/ (accessed on 5 September 2019).
- EMBL-EBI. Kalign. Available online: https://www.ebi.ac.uk/Tools/msa/kalign/ (accessed on 5 September 2019).
- Bustin, S.A. Absolute quantification of mRNA using real-time reverse transcription polymerase chain reaction assays. J. Mol. Endocrinol. 2000, 25, 169–193. [Google Scholar] [CrossRef]
- Holland, P.M.; Abramson, R.D.; Watson, R.; Gelfand, D.H. Detection of specific polymerase chain reaction product by utilizing the 5’—3’exonuclease activity of Thermus aquaticus DNA polymerase. Proc. Natl. Acad. Sci. USA 1991, 88, 7276–7280. [Google Scholar] [CrossRef]
- Kutyavin, I.V.; Afonina, I.A.; Mills, A.; Gorn, V.V.; Lukhtanov, E.A.; Belousov, E.S.; Singer, M.J.; Walburger, D.K.; Lokhov, S.G.; Gall, A.A. 3′-minor groove binder-DNA probes increase sequence specificity at PCR extension temperatures. Nucleic Acids Res. 2000, 28, 655–661. [Google Scholar] [CrossRef]
- Pfaffl, M.W. A new mathematical model for relative quantification in real-time RT–PCR. Nucleic Acids Res. 2001, 29, e45. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Nakano, Y.; Asada, K. Hydrogen Peroxide is Scavenged by Ascorbate-specific Peroxidase in Spinach Chloroplasts. Plant Cell Physiol. 1981, 22, 867–880. [Google Scholar] [CrossRef]
- Wendel, A.B.T.M. Glutathione peroxidase. In Detoxication and Drug Metabolism: Conjugation and Related Systems; Jakoby, W.B., Ed.; Academic Press: Cambridge, MA, USA, 1981; pp. 325–333. ISBN 0076-6879. [Google Scholar]
- Harrach, B.D.; Fodor, J.; Pogány, M.; Preuss, J.; Barna, B. Antioxidant, ethylene and membrane leakage responses to powdery mildew infection of near-isogenic barley lines with various types of resistance. Eur. J. Plant Pathol. 2008, 121, 21–33. [Google Scholar] [CrossRef]
- Żur, I.; Dubas, E.; Golemiec, E.; Szechyńska-Hebda, M.; Gołębiowska, G.; Wędzony, M. Stress-related variation in antioxidative enzymes activity and cell metabolism efficiency associated with embryogenesis induction in isolated microspore culture of triticale (x Triticosecale Wittm.). Plant Cell Rep. 2009, 28, 1279–1287. [Google Scholar] [CrossRef] [PubMed]
- Aebi, H.B.T.M. Catalase in vitro. In Oxygen Radicals in Biological Systems; Kaplan, N., Colowick, N., Eds.; Academic Press: Cambridge, MA, USA, 1984; pp. 121–126. ISBN 0076-6879. [Google Scholar]
- Guengerich, F.P.; Martin, M.V.; Sohl, C.D.; Cheng, Q. Measurement of cytochrome P450 and NADPH–cytochrome P450 reductase. Nat. Protoc. 2009, 4, 1245–1251. [Google Scholar] [CrossRef]
- Quayle, J.R.B.T.M. Formate dehydrogenase. In Carbohydrate Metabolism; Wood, W.A., Ed.; Academic Press: Cambridge, MA, USA, 1966; Volume 9, pp. 360–364. ISBN 0076-6879. [Google Scholar]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).