Gut Microbiota Influence in Hematological Malignancies: From Genesis to Cure
Abstract
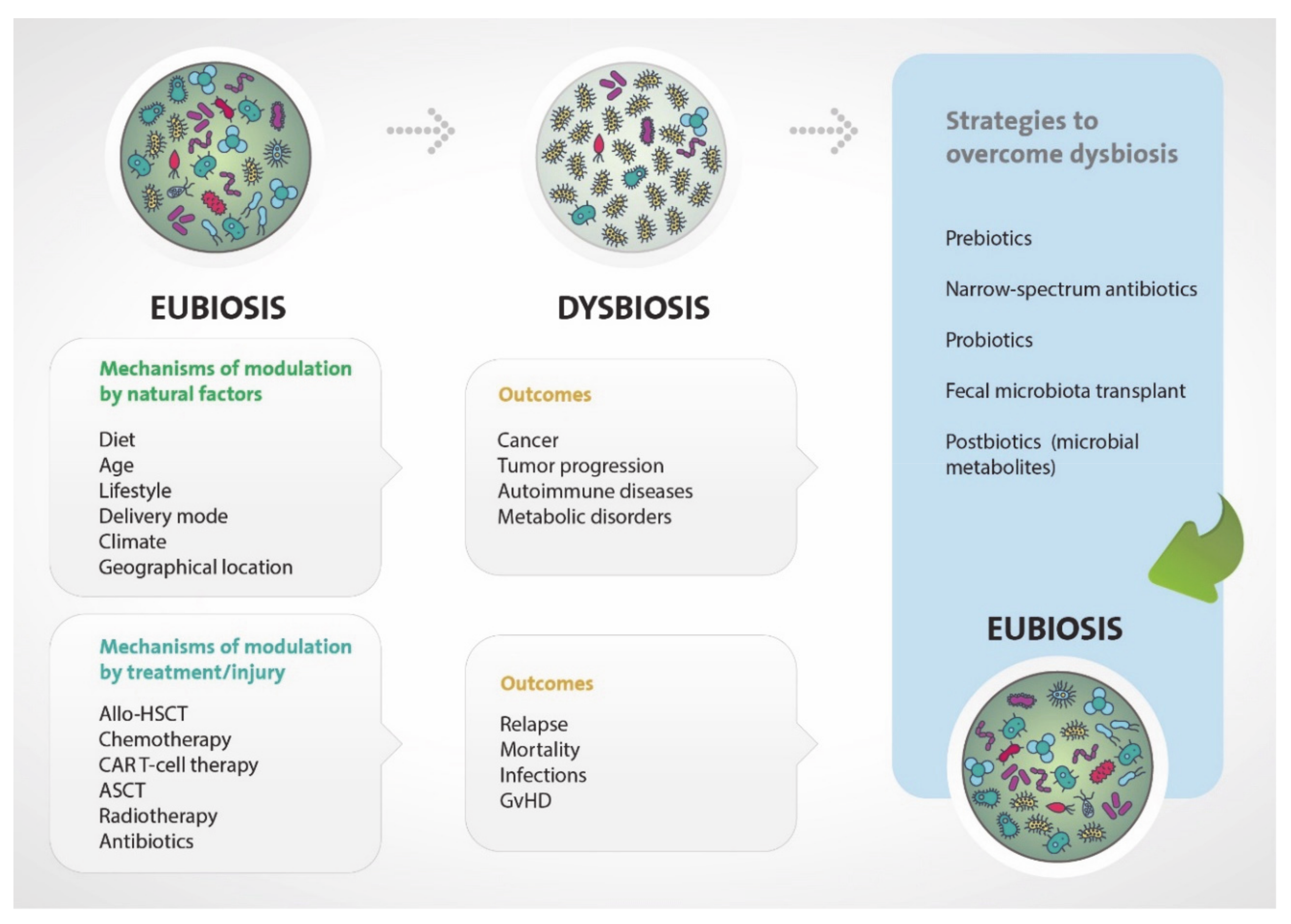
1. Introduction
2. Gut Microbiota in Hematological Malignancies
3. The Treatment of Hematological Malignancies and Microbiota
4. Allogeneic Stem Cell Transplantation
5. Autologous Stem Cell Transplant
6. Antibiotics and Acute Myeloid Leukemia
7. First Line Treatment in Multiple Myeloma
8. CAR T Cells
9. Outlook-Future Perspective
10. Concluding Remarks
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Helander, H.F.; Fändriks, L. Surface Area of the Digestive Tract-Revisited. Scand. J. Gastroenterol. 2014, 49, 681–689. [Google Scholar] [CrossRef] [PubMed]
- Sender, R.; Fuchs, S.; Milo, R. Are We Really Vastly Outnumbered? Revisiting the Ratio of Bacterial to Host Cells in Humans. Cell 2016, 164, 337–340. [Google Scholar] [CrossRef] [PubMed]
- Donaldson, G.P.; Lee, S.M.; Mazmanian, S.K. Gut Biogeography of the Bacterial Microbiota. Nat. Rev. Microbiol. 2016, 14, 20–32. [Google Scholar] [CrossRef]
- Ley, R.E.; Hamady, M.; Lozupone, C.; Turnbaugh, P.; Ramey, R.R.; Bircher, J.S.; Schlegel, M.L.; Tucker, T.A.; Schrenzel, M.D.; Knight, R.; et al. Evolution of Mammals and Their Gut Microbes. Science 2008, 320, 1647–1651. [Google Scholar] [CrossRef] [PubMed]
- Qin, J.; Li, R.; Raes, J.; Arumugam, M.; Burgdorf, K.S.; Manichanh, C.; Nielsen, T.; Pons, N.; Levenez, F.; Yamada, T.; et al. A Human Gut Microbial Gene Catalogue Established by Metagenomic Sequencing. Nature 2010, 464, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Dominguez-Bello, M.G.; Costello, E.K.; Contreras, M.; Magris, M.; Hidalgo, G.; Fierer, N.; Knight, R. Delivery Mode Shapes the Acquisition and Structure of the Initial Microbiota across Multiple Body Habitats in Newborns. PNAS 2010, 107, 11971–11975. [Google Scholar] [CrossRef]
- Ferretti, P.; Pasolli, E.; Tett, A.; Asnicar, F.; Gorfer, V.; Fedi, S.; Armanini, F.; Truong, D.T.; Manara, S.; Zolfo, M.; et al. Mother-to-Infant Microbial Transmission from Different Body Sites Shapes the Developing Infant Gut Microbiome. Cell Host Microbe 2018, 24, 133–145. [Google Scholar] [CrossRef]
- Jakobsson, H.E.; Abrahamsson, T.R.; Jenmalm, M.C.; Harris, K.; Quince, C.; Jernberg, C.; Björkstén, B.; Engstrand, L.; Andersson, A.F. Decreased Gut Microbiota Diversity, Delayed Bacteroidetes Colonisation and Reduced Th1 Responses in Infants Delivered by Caesarean Section. Gut 2014, 63, 559–566. [Google Scholar] [CrossRef]
- Stewart, C.J.; Ajami, N.J.; O’Brien, J.L.; Hutchinson, D.S.; Smith, D.P.; Wong, M.C.; Ross, M.C.; Lloyd, R.E.; Doddapaneni, H.; Metcalf, G.A.; et al. Temporal Development of the Gut Microbiome in Early Childhood from the TEDDY Study. Nature 2018, 562, 583–588. [Google Scholar] [CrossRef]
- Rothschild, D.; Weissbrod, O.; Barkan, E.; Kurilshikov, A.; Korem, T.; Zeevi, D.; Costea, P.I.; Godneva, A.; Kalka, I.N.; Bar, N.; et al. Environment Dominates over Host Genetics in Shaping Human Gut Microbiota. Nature 2018, 555, 210–215. [Google Scholar] [CrossRef]
- Jernberg, C.; Löfmark, S.; Edlund, C.; Jansson, J.K. Long-Term Ecological Impacts of Antibiotic Administration on the Human Intestinal Microbiota. ISME J. 2007, 1, 56–66. [Google Scholar] [CrossRef] [PubMed]
- Dethlefsen, L.; Huse, S.; Sogin, M.L.; Relman, D.A. The Pervasive Effects of an Antibiotic on the Human Gut Microbiota, as Revealed by Deep 16S RRNA Sequencing. PLoS Biol. 2008, 6, e280. [Google Scholar] [CrossRef] [PubMed]
- Maier, L.; Pruteanu, M.; Kuhn, M.; Zeller, G.; Telzerow, A.; Anderson, E.E.; Brochado, A.R.; Fernandez, K.C.; Dose, H.; Mori, H.; et al. Extensive Impact of Non-Antibiotic Drugs on Human Gut Bacteria. Nature 2018, 555, 623–628. [Google Scholar] [CrossRef] [PubMed]
- Dethlefsen, L.; Relman, D.A. Incomplete Recovery and Individualized Responses of the Human Distal Gut Microbiota to Repeated Antibiotic Perturbation. PNAS 2011, 108, 4554–4561. [Google Scholar] [CrossRef] [PubMed]
- Marra, F.; Marra, C.A.; Richardson, K.; Lynd, L.D.; Kozyrskyj, A.; Patrick, D.M.; Bowie, W.R.; Fitzgerald, J.M. Antibiotic Use in Children Is Associated with Increased Risk of Asthma. Pediatrics 2009, 123, 1003–1010. [Google Scholar] [CrossRef] [PubMed]
- Livanos, A.E.; Greiner, T.U.; Vangay, P.; Pathmasiri, W.; Stewart, D.; McRitchie, S.; Li, H.; Chung, J.; Sohn, J.; Kim, S.; et al. Antibiotic-Mediated Gut Microbiome Perturbation Accelerates Development of Type 1 Diabetes in Mice. Nat. Microbiol. 2016, 1, 16140. [Google Scholar] [CrossRef]
- Cho, I.; Yamanishi, S.; Cox, L.; Methé, B.A.; Zavadil, J.; Li, K.; Gao, Z.; Mahana, D.; Raju, K.; Teitler, I.; et al. Antibiotics in Early Life Alter the Murine Colonic Microbiome and Adiposity. Nature 2012, 488, 621–626. [Google Scholar] [CrossRef]
- Cox, L.M.; Yamanishi, S.; Sohn, J.; Alekseyenko, A.V.; Leung, J.M.; Cho, I.; Kim, S.; Li, H.; Gao, Z.; Mahana, D.; et al. Altering the Intestinal Microbiota during a Critical Developmental Window Has Lasting Metabolic Consequences. Cell 2014, 158, 705–721. [Google Scholar] [CrossRef]
- Suez, J.; Zmora, N.; Zilberman-Schapira, G.; Mor, U.; Dori-Bachash, M.; Bashiardes, S.; Zur, M.; Regev-Lehavi, D.; Ben-Zeev Brik, R.; Federici, S.; et al. Post-Antibiotic Gut Mucosal Microbiome Reconstitution Is Impaired by Probiotics and Improved by Autologous FMT. Cell 2018, 174, 1406–1423. [Google Scholar] [CrossRef]
- Hooper, L.V.; Wong, M.H.; Thelin, A.; Hansson, L.; Falk, P.G.; Gordon, J.I. Molecular Analysis of Commensal Host-Microbial Relationships in the Intestine. Science 2001, 291, 881–884. [Google Scholar] [CrossRef]
- Bäumler, A.J.; Sperandio, V. Interactions between the Microbiota and Pathogenic Bacteria in the Gut. Nature 2016, 535, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Ridaura, V.K.; Faith, J.J.; Rey, F.E.; Cheng, J.; Duncan, A.E.; Kau, A.L.; Griffin, N.W.; Lombard, V.; Henrissat, B.; Bain, J.R.; et al. Gut Microbiota from Twins Discordant for Obesity Modulate Metabolism in Mice. Science 2013, 341. [Google Scholar] [CrossRef] [PubMed]
- Andriessen, E.M.; Wilson, A.M.; Mawambo, G.; Dejda, A.; Miloudi, K.; Sennlaub, F.; Sapieha, P. Gut Microbiota Influences Pathological Angiogenesis in Obesity-driven Choroidal Neovascularization. EMBO Mol. Med. 2016, 8, 1366–1379. [Google Scholar] [CrossRef] [PubMed]
- Cummings, J.H.; Pomare, E.W.; Branch, W.J.; Naylor, C.P.; Macfarlane, G.T. Short Chain Fatty Acids in Human Large Intestine, Portal, Hepatic and Venous Blood. Gut 1987, 28, 1221–1227. [Google Scholar] [CrossRef] [PubMed]
- Macpherson, A.J.; Slack, E.; Geuking, M.B.; McCoy, K.D. The Mucosal Firewalls against Commensal Intestinal Microbes. Semin. Immunopathol. 2009, 31, 145–149. [Google Scholar] [CrossRef]
- Elinav, E.; Strowig, T.; Kau, A.L.; Henao-Mejia, J.; Thaiss, C.A.; Booth, C.J.; Peaper, D.R.; Bertin, J.; Eisenbarth, S.C.; Gordon, J.I.; et al. NLRP6 Inflammasome Regulates Colonic Microbial Ecology and Risk for Colitis. Cell 2011, 145, 745–757. [Google Scholar] [CrossRef]
- Garrett, W.S.; Lord, G.M.; Punit, S.; Lugo-Villarino, G.; Mazmanian, S.K.; Ito, S.; Glickman, J.N.; Glimcher, L.H. Communicable Ulcerative Colitis Induced by T-Bet Deficiency in the Innate Immune System. Cell 2007, 131, 33–45. [Google Scholar] [CrossRef]
- Vijay-Kumar, M.; Aitken, J.D.; Carvalho, F.A.; Cullender, T.C.; Mwangi, S.; Srinivasan, S.; Sitaraman, S.V.; Knight, R.; Ley, R.E.; Gewirtz, A.T. Metabolic Syndrome and Altered Gut Microbiota in Mice Lacking Toll-like Receptor 5. Science 2010, 328, 228–231. [Google Scholar] [CrossRef]
- Skelly, A.N.; Sato, Y.; Kearney, S.; Honda, K. Mining the Microbiota for Microbial and Metabolite-Based Immunotherapies. Nat. Rev. Immunol. 2019, 19, 305–323. [Google Scholar] [CrossRef]
- Franzosa, E.A.; Sirota-Madi, A.; Avila-Pacheco, J.; Fornelos, N.; Haiser, H.J.; Reinker, S.; Vatanen, T.; Hall, A.B.; Mallick, H.; McIver, L.J.; et al. Gut Microbiome Structure and Metabolic Activity in Inflammatory Bowel Disease. Nat. Microbiol. 2019, 4, 293–305. [Google Scholar] [CrossRef]
- Blacher, E.; Levy, M.; Tatirovsky, E.; Elinav, E. Microbiome-Modulated Metabolites at the Interface of Host Immunity. J. Immunol. 2017, 198, 572–580. [Google Scholar] [CrossRef] [PubMed]
- Arpaia, N.; Campbell, C.; Fan, X.; Dikiy, S.; van der Veeken, J.; de Roos, P.; Liu, H.; Cross, J.R.; Pfeffer, K.; Coffer, P.J.; et al. Metabolites Produced by Commensal Bacteria Promote Peripheral Regulatory T-Cell Generation. Nature 2013, 504, 451–455. [Google Scholar] [CrossRef] [PubMed]
- Smith, P.M.; Howitt, M.R.; Panikov, N.; Michaud, M.; Gallini, C.A.; Bohlooly-Y, M.; Glickman, J.N.; Garrett, W.S. The Microbial Metabolites, Short-Chain Fatty Acids, Regulate Colonic Treg Cell Homeostasis. Science 2013, 341, 569–573. [Google Scholar] [CrossRef] [PubMed]
- Nastasi, C.; Candela, M.; Bonefeld, C.M.; Geisler, C.; Hansen, M.; Krejsgaard, T.; Biagi, E.; Andersen, M.H.; Brigidi, P.; Ødum, N.; et al. The Effect of Short-Chain Fatty Acids on Human Monocyte-Derived Dendritic Cells. Sci. Rep. 2015, 5, 16148. [Google Scholar] [CrossRef]
- Uribe-Herranz, M.; Rafail, S.; Beghi, S.; Gil-de-Gómez, L.; Verginadis, I.; Bittinger, K.; Pustylnikov, S.; Pierini, S.; Perales-Linares, R.; Blair, I.A.; et al. Gut Microbiota Modulate Dendritic Cell Antigen Presentation and Radiotherapy-Induced Antitumor Immune Response. J. Clin. Invest. 2020, 130, 466–479. [Google Scholar] [CrossRef]
- Le Poul, E.; Loison, C.; Struyf, S.; Springael, J.-Y.; Lannoy, V.; Decobecq, M.-E.; Brezillon, S.; Dupriez, V.; Vassart, G.; Van Damme, J.; et al. Functional Characterization of Human Receptors for Short Chain Fatty Acids and Their Role in Polymorphonuclear Cell Activation. J. Biol. Chem. 2003, 278, 25481–25489. [Google Scholar] [CrossRef]
- Bauer, H.; Horowitz, R.E.; Levenson, S.M.; Popper, H. The Response of the Lymphatic Tissue to the Microbial Flora. Studies on Germfree Mice. Am. J. Pathol. 1963, 42, 471–483. [Google Scholar]
- Chung, H.; Pamp, S.J.; Hill, J.A.; Surana, N.K.; Edelman, S.M.; Troy, E.B.; Reading, N.C.; Villablanca, E.J.; Wang, S.; Mora, J.R.; et al. Gut Immune Maturation Depends on Colonization with a Host-Specific Microbiota. Cell 2012, 149, 1578–1593. [Google Scholar] [CrossRef]
- Mazmanian, S.K.; Liu, C.H.; Tzianabos, A.O.; Kasper, D.L. An Immunomodulatory Molecule of Symbiotic Bacteria Directs Maturation of the Host Immune System. Cell 2005, 122, 107–118. [Google Scholar] [CrossRef]
- Talham, G.L.; Jiang, H.-Q.; Bos, N.A.; Cebra, J.J. Segmented Filamentous Bacteria Are Potent Stimuli of a Physiologically Normal State of the Murine Gut Mucosal Immune System. Infect. Immun. 1999, 67, 1992–2000. [Google Scholar] [CrossRef]
- Macpherson, A.J.; Hunziker, L.; McCoy, K.; Lamarre, A. IgA Responses in the Intestinal Mucosa against Pathogenic and Non-Pathogenic Microorganisms. Microbes Infect. 2001, 3, 1021–1035. [Google Scholar] [CrossRef]
- Shroff, K.E.; Meslin, K.; Cebra, J.J. Commensal Enteric Bacteria Engender a Self-Limiting Humoral Mucosal Immune Response While Permanently Colonizing the Gut. Infect. Immun. 1995, 63, 3904–3913. [Google Scholar] [CrossRef] [PubMed]
- Ivanov, I.I.; de Llanos Frutos, R.; Manel, N.; Yoshinaga, K.; Rifkin, D.B.; Sartor, R.B.; Finlay, B.B.; Littman, D.R. Specific Microbiota Direct the Differentiation of Th17 Cells in the Mucosa of the Small Intestine. Cell Host Microbe 2008, 4, 337–349. [Google Scholar] [CrossRef] [PubMed]
- Atarashi, K.; Tanoue, T.; Oshima, K.; Suda, W.; Nagano, Y.; Nishikawa, H.; Fukuda, S.; Saito, T.; Narushima, S.; Hase, K.; et al. Treg Induction by a Rationally Selected Mixture of Clostridia Strains from the Human Microbiota. Nature 2013, 500, 232–236. [Google Scholar] [CrossRef]
- Round, J.L.; Mazmanian, S.K. Inducible Foxp3+ Regulatory T-Cell Development by a Commensal Bacterium of the Intestinal Microbiota. PNAS 2010, 107, 12204–12209. [Google Scholar] [CrossRef]
- Contijoch, E.J.; Britton, G.J.; Yang, C.; Mogno, I.; Li, Z.; Ng, R.; Llewellyn, S.R.; Hira, S.; Johnson, C.; Rabinowitz, K.M.; et al. Gut Microbiota Density Influences Host Physiology and Is Shaped by Host and Microbial Factors. eLife 2019, 8, e40553. [Google Scholar] [CrossRef]
- Belkaid, Y.; Naik, S. Compartmentalized and Systemic Control of Tissue Immunity by Commensals. Nat. Immunol. 2013, 14, 646–653. [Google Scholar] [CrossRef]
- Khosravi, A.; Yáñez, A.; Price, J.G.; Chow, A.; Merad, M.; Goodridge, H.S.; Mazmanian, S.K. Gut Microbiota Promotes Hematopoiesis Tocontrol Bacterial Infection. Cell Host Microbe 2014, 15, 374–381. [Google Scholar] [CrossRef]
- Clarke, T.B.; Davis, K.M.; Lysenko, E.S.; Zhou, A.Y.; Yu, Y.; Weiser, J.N. Recognition of Peptidoglycan from the Microbiota by Nod1 Enhances Systemic Innate Immunity. Nat. Med. 2010, 16, 228–231. [Google Scholar] [CrossRef]
- Hegazy, A.N.; West, N.R.; Stubbington, M.J.T.; Wendt, E.; Suijker, K.I.M.; Datsi, A.; This, S.; Danne, C.; Campion, S.; Duncan, S.H.; et al. Circulating and Tissue-Resident CD4+ T Cells With Reactivity to Intestinal Microbiota Are Abundant in Healthy Individuals and Function Is Altered During Inflammation. Gastroenterology 2017, 153, 1320–1337. [Google Scholar] [CrossRef]
- Lathrop, S.K.; Bloom, S.M.; Rao, S.M.; Nutsch, K.; Lio, C.-W.; Santacruz, N.; Peterson, D.A.; Stappenbeck, T.S.; Hsieh, C.-S. Peripheral Education of the Immune System by Colonic Commensal Microbiota. Nature 2011, 478, 250–254. [Google Scholar] [CrossRef] [PubMed]
- Horai, R.; Zárate-Bladés, C.R.; Dillenburg-Pilla, P.; Chen, J.; Kielczewski, J.L.; Silver, P.B.; Jittayasothorn, Y.; Chan, C.-C.; Yamane, H.; Honda, K.; et al. Microbiota-Dependent Activation of an Autoreactive T Cell Receptor Provokes Autoimmunity in an Immunologically Privileged Site. Immunity 2015, 43, 343–353. [Google Scholar] [CrossRef]
- Benckert, J.; Schmolka, N.; Kreschel, C.; Zoller, M.J.; Sturm, A.; Wiedenmann, B.; Wardemann, H. The Majority of Intestinal IgA+ and IgG+ Plasmablasts in the Human Gut Are Antigen-Specific. J. Clin. Invest. 2011, 121, 1946–1955. [Google Scholar] [CrossRef] [PubMed]
- Brusca, S.B.; Abramson, S.B.; Scher, J.U. Microbiome and Mucosal Inflammation as Extra-Articular Triggers for Rheumatoid Arthritis and Autoimmunity. Curr. Opin. Rheumatol. 2014, 26, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Lloyd-Price, J.; Arze, C.; Ananthakrishnan, A.N.; Schirmer, M.; Avila-Pacheco, J.; Poon, T.W.; Andrews, E.; Ajami, N.J.; Bonham, K.S.; Brislawn, C.J.; et al. Multi-Omics of the Gut Microbial Ecosystem in Inflammatory Bowel Diseases. Nature 2019, 569, 655–662. [Google Scholar] [CrossRef] [PubMed]
- Trompette, A.; Gollwitzer, E.S.; Yadava, K.; Sichelstiel, A.K.; Sprenger, N.; Ngom-Bru, C.; Blanchard, C.; Junt, T.; Nicod, L.P.; Harris, N.L.; et al. Gut Microbiota Metabolism of Dietary Fiber Influences Allergic Airway Disease and Hematopoiesis. Nat. Med. 2014, 20, 159–166. [Google Scholar] [CrossRef]
- Wen, L.; Ley, R.E.; Volchkov, P.V.; Stranges, P.B.; Avanesyan, L.; Stonebraker, A.C.; Hu, C.; Wong, F.S.; Szot, G.L.; Bluestone, J.A.; et al. Innate Immunity and Intestinal Microbiota in the Development of Type 1 Diabetes. Nature 2008, 455, 1109–1113. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.-J.; Ivanov, I.I.; Darce, J.; Hattori, K.; Shima, T.; Umesaki, Y.; Littman, D.R.; Benoist, C.; Mathis, D. Gut-Residing Segmented Filamentous Bacteria Drive Autoimmune Arthritis via T Helper 17 Cells. Immunity 2010, 32, 815–827. [Google Scholar] [CrossRef]
- Tilg, H.; Moschen, A.R. Microbiota and Diabetes: An Evolving Relationship. Gut 2014, 63, 1513–1521. [Google Scholar] [CrossRef]
- de Martel, C.; Georges, D.; Bray, F.; Ferlay, J.; Clifford, G.M. Global Burden of Cancer Attributable to Infections in 2018: A Worldwide Incidence Analysis. Lancet Glob. Health 2020, 8, e180–e190. [Google Scholar] [CrossRef]
- Zhang, H.; Sun, L. When Human Cells Meet Bacteria: Precision Medicine for Cancers Using the Microbiota. Am. J. Cancer Res. 2018, 8, 1157–1175. [Google Scholar] [PubMed]
- Rosshart, S.P.; Vassallo, B.G.; Angeletti, D.; Hutchinson, D.S.; Morgan, A.P.; Takeda, K.; Hickman, H.D.; McCulloch, J.A.; Badger, J.H.; Ajami, N.J.; et al. Wild Mouse Gut Microbiota Promotes Host Fitness and Improves Disease Resistance. Cell 2017, 171, 1015–1028.e13. [Google Scholar] [CrossRef] [PubMed]
- Song, X.; Gao, H.; Lin, Y.; Yao, Y.; Zhu, S.; Wang, J.; Liu, Y.; Yao, X.; Meng, G.; Shen, N.; et al. Alterations in the Microbiota Drive Interleukin-17C Production from Intestinal Epithelial Cells to Promote Tumorigenesis. Immunity 2014, 40, 140–152. [Google Scholar] [CrossRef] [PubMed]
- Zhan, Y.; Chen, P.-J.; Sadler, W.D.; Wang, F.; Poe, S.; Núñez, G.; Eaton, K.A.; Chen, G.Y. Gut Microbiota Protects against Gastrointestinal Tumorigenesis Caused by Epithelial Injury. Cancer Res. 2013, 73, 7199–7210. [Google Scholar] [CrossRef] [PubMed]
- Lakritz, J.R.; Poutahidis, T.; Mirabal, S.; Varian, B.J.; Levkovich, T.; Ibrahim, Y.M.; Ward, J.M.; Teng, E.C.; Fisher, B.; Parry, N.; et al. Gut Bacteria Require Neutrophils to Promote Mammary Tumorigenesis. Oncotarget 2015, 6, 9387–9396. [Google Scholar] [CrossRef]
- Rao, V.P.; Poutahidis, T.; Ge, Z.; Nambiar, P.R.; Boussahmain, C.; Wang, Y.Y.; Horwitz, B.H.; Fox, J.G.; Erdman, S.E. Innate Immune Inflammatory Response against Enteric Bacteria Helicobacter Hepaticus Induces Mammary Adenocarcinoma in Mice. Cancer Res. 2006, 66, 7395–7400. [Google Scholar] [CrossRef]
- Ertz-Archambault, N.; Keim, P.; Von Hoff, D. Microbiome and Pancreatic Cancer: A Comprehensive Topic Review of Literature. World J. Gastroenterol. 2017, 23, 1899–1908. [Google Scholar] [CrossRef]
- Dapito, D.H.; Mencin, A.; Gwak, G.-Y.; Pradere, J.-P.; Jang, M.-K.; Mederacke, I.; Caviglia, J.M.; Khiabanian, H.; Adeyemi, A.; Bataller, R.; et al. Promotion of Hepatocellular Carcinoma by the Intestinal Microbiota and TLR4. Cancer Cell 2012, 21, 504–516. [Google Scholar] [CrossRef]
- Gopalakrishnan, V.; Spencer, C.N.; Nezi, L.; Reuben, A.; Andrews, M.C.; Karpinets, T.V.; Prieto, P.A.; Vicente, D.; Hoffman, K.; Wei, S.C.; et al. Gut Microbiome Modulates Response to Anti–PD-1 Immunotherapy in Melanoma Patients. Science 2017, eaan4236. [Google Scholar] [CrossRef]
- Matson, V.; Fessler, J.; Bao, R.; Chongsuwat, T.; Zha, Y.; Alegre, M.-L.; Luke, J.J.; Gajewski, T.F. The Commensal Microbiome Is Associated with Anti-PD-1 Efficacy in Metastatic Melanoma Patients. Science 2018, 359, 104–108. [Google Scholar] [CrossRef]
- Routy, B.; Le Chatelier, E.; Derosa, L.; Duong, C.P.M.; Alou, M.T.; Daillère, R.; Fluckiger, A.; Messaoudene, M.; Rauber, C.; Roberti, M.P.; et al. Gut Microbiome Influences Efficacy of PD-1-Based Immunotherapy against Epithelial Tumors. Science 2017. [Google Scholar] [CrossRef] [PubMed]
- Sivan, A.; Corrales, L.; Hubert, N.; Williams, J.B.; Aquino-Michaels, K.; Earley, Z.M.; Benyamin, F.W.; Lei, Y.M.; Jabri, B.; Alegre, M.-L.; et al. Commensal Bifidobacterium Promotes Antitumor Immunity and Facilitates Anti–PD-L1 Efficacy. Science 2015, 350, 1084–1089. [Google Scholar] [CrossRef] [PubMed]
- Uribe-Herranz, M.; Bittinger, K.; Rafail, S.; Guedan, S.; Pierini, S.; Tanes, C.; Ganetsky, A.; Morgan, M.A.; Gill, S.; Tanyi, J.L.; et al. Gut Microbiota Modulates Adoptive Cell Therapy via CD8α Dendritic Cells and IL-12. JCI Insight 2018, 3. [Google Scholar] [CrossRef] [PubMed]
- Viaud, S.; Saccheri, F.; Mignot, G.; Yamazaki, T.; Daillère, R.; Hannani, D.; Enot, D.P.; Pfirschke, C.; Engblom, C.; Pittet, M.J.; et al. The Intestinal Microbiota Modulates the Anticancer Immune Effects of Cyclophosphamide. Science 2013, 342, 971–976. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, M.L.; Maier, I.; Dang, A.T.; Berry, D.; Liu, J.; Ruegger, P.M.; Yang, J.-I.; Soto, P.A.; Presley, L.L.; Reliene, R.; et al. Intestinal Bacteria Modify Lymphoma Incidence and Latency by Affecting Systemic Inflammatory State, Oxidative Stress, and Leukocyte Genotoxicity. Cancer Res. 2013, 73, 4222–4232. [Google Scholar] [CrossRef] [PubMed]
- Salas, C.; Niembro, A.; Lozano, V.; Gallardo, E.; Molina, B.; Sánchez, S.; Ramos, S.; Carnevale, A.; Pérez-Vera, P.; Rivera Luna, R.; et al. Persistent Genomic Instability in Peripheral Blood Lymphocytes from Hodgkin Lymphoma Survivors. Environ. Mol. Mutagen. 2012, 53, 271–280. [Google Scholar] [CrossRef] [PubMed]
- Martin, R.; Nauta, A.J.; Ben Amor, K.; Knippels, L.M.J.; Knol, J.; Garssen, J. Early Life: Gut Microbiota and Immune Development in Infancy. Benef. Microbes 2010, 1, 367–382. [Google Scholar] [CrossRef]
- Poppema, S. Immunobiology and Pathophysiology of Hodgkin Lymphomas. Hematol. Am. Soc. Hematol. Educ. Program 2005, 231–238. [Google Scholar] [CrossRef]
- Blumberg, R.; Powrie, F. Microbiota, Disease, and Back to Health: A Metastable Journey. Sci. Transl. Med. 2012, 4, 137rv7. [Google Scholar] [CrossRef]
- Cozen, W.; Yu, G.; Gail, M.H.; Ridaura, V.K.; Nathwani, B.N.; Hwang, A.E.; Hamilton, A.S.; Mack, T.M.; Gordon, J.I.; Goedert, J.J. Fecal Microbiota Diversity in Survivors of Adolescent/Young Adult Hodgkin Lymphoma: A Study of Twins. Br. J. Cancer 2013, 108, 1163–1167. [Google Scholar] [CrossRef]
- Shaffer, A.L.; Rosenwald, A.; Staudt, L.M. Lymphoid Malignancies: The Dark Side of B-Cell Differentiation. Nat. Rev. Immunol. 2002, 2, 920–932. [Google Scholar] [CrossRef] [PubMed]
- Pasqualucci, L.; Neumeister, P.; Goossens, T.; Nanjangud, G.; Chaganti, R.S.; Küppers, R.; Dalla-Favera, R. Hypermutation of Multiple Proto-Oncogenes in B-Cell Diffuse Large-Cell Lymphomas. Nature 2001, 412, 341–346. [Google Scholar] [CrossRef] [PubMed]
- Federico, A.; Morgillo, F.; Tuccillo, C.; Ciardiello, F.; Loguercio, C. Chronic Inflammation and Oxidative Stress in Human Carcinogenesis. Int. J. Cancer 2007, 121, 2381–2386. [Google Scholar] [CrossRef] [PubMed]
- Epeldegui, M.; Widney, D.P.; Martínez-Maza, O. Pathogenesis of AIDS Lymphoma: Role of Oncogenic Viruses and B Cell Activation-Associated Molecular Lesions. Curr. Opin. Oncol. 2006, 18, 444–448. [Google Scholar] [CrossRef]
- Illes, A.; Varoczy, L.; Papp, G.; Wilson, P.C.; Alex, P.; Jonsson, R.; Kovacs, T.; Konttinen, Y.T.; Zehert, M.; Nakken, B.; et al. Aspects of B-Cell Non-Hodgkin’s Lymphoma Development: A Transition from Immune-Reactivity to Malignancy. Scand. J. Immunol. 2009, 69, 387–400. [Google Scholar] [CrossRef]
- Pflug, N.; Kluth, S.; Vehreschild, J.J.; Bahlo, J.; Tacke, D.; Biehl, L.; Eichhorst, B.; Fischer, K.; Cramer, P.; Fink, A.-M.; et al. Efficacy of Antineoplastic Treatment Is Associated with the Use of Antibiotics That Modulate Intestinal Microbiota. Oncoimmunology 2016, 5, e1150399. [Google Scholar] [CrossRef]
- Iida, N.; Dzutsev, A.; Stewart, C.A.; Smith, L.; Bouladoux, N.; Weingarten, R.A.; Molina, D.A.; Salcedo, R.; Back, T.; Cramer, S.; et al. Commensal Bacteria Control Cancer Response to Therapy by Modulating the Tumor Microenvironment. Science 2013, 342, 967–970. [Google Scholar] [CrossRef]
- Gaffen, S.L.; Jain, R.; Garg, A.V.; Cua, D.J. The IL-23-IL-17 Immune Axis: From Mechanisms to Therapeutic Testing. Nat. Rev. Immunol. 2014, 14, 585–600. [Google Scholar] [CrossRef]
- Calcinotto, A.; Brevi, A.; Chesi, M.; Ferrarese, R.; Garcia Perez, L.; Grioni, M.; Kumar, S.; Garbitt, V.M.; Sharik, M.E.; Henderson, K.J.; et al. Microbiota-Driven Interleukin-17-Producing Cells and Eosinophils Synergize to Accelerate Multiple Myeloma Progression. Nat. Commun. 2018, 9, 4832. [Google Scholar] [CrossRef]
- Peled, J.U.; Devlin, S.M.; Staffas, A.; Lumish, M.; Khanin, R.; Littmann, E.R.; Ling, L.; Kosuri, S.; Maloy, M.; Slingerland, J.B.; et al. Intestinal Microbiota and Relapse After Hematopoietic-Cell Transplantation. J. Clin. Oncol. 2017, 35, 1650–1659. [Google Scholar] [CrossRef]
- Valdes, A.M.; Walter, J.; Segal, E.; Spector, T.D. Role of the Gut Microbiota in Nutrition and Health. BMJ 2018, 361, k2179. [Google Scholar] [CrossRef] [PubMed]
- Bai, L.; Zhou, P.; Li, D.; Ju, X. Changes in the Gastrointestinal Microbiota of Children with Acute Lymphoblastic Leukaemia and Its Association with Antibiotics in the Short Term. J. Med. Microbiol. 2017, 66, 1297–1307. [Google Scholar] [CrossRef] [PubMed]
- Nearing, J.T.; Connors, J.; Whitehouse, S.; Van Limbergen, J.; Macdonald, T.; Kulkarni, K.; Langille, M.G.I. Infectious Complications Are Associated With Alterations in the Gut Microbiome in Pediatric Patients With Acute Lymphoblastic Leukemia. Front. Cell. Infect. Microbiol. 2019, 9, 28. [Google Scholar] [CrossRef] [PubMed]
- van Vliet, M.J.; Tissing, W.J.E.; Dun, C.A.J.; Meessen, N.E.L.; Kamps, W.A.; de Bont, E.S.J.M.; Harmsen, H.J.M. Chemotherapy Treatment in Pediatric Patients with Acute Myeloid Leukemia Receiving Antimicrobial Prophylaxis Leads to a Relative Increase of Colonization with Potentially Pathogenic Bacteria in the Gut. Clin. Infect. Dis. 2009, 49, 262–270. [Google Scholar] [CrossRef] [PubMed]
- Kaysen, A.; Heintz-Buschart, A.; Muller, E.E.L.; Narayanasamy, S.; Wampach, L.; Laczny, C.C.; Graf, N.; Simon, A.; Franke, K.; Bittenbring, J.; et al. Integrated Meta-Omic Analyses of the Gastrointestinal Tract Microbiome in Patients Undergoing Allogeneic Hematopoietic Stem Cell Transplantation. Transl. Res. 2017, 186, 79–94. [Google Scholar] [CrossRef]
- Galloway-Peña, J.R.; Smith, D.P.; Sahasrabhojane, P.; Ajami, N.J.; Wadsworth, W.D.; Daver, N.G.; Chemaly, R.F.; Marsh, L.; Ghantoji, S.S.; Pemmaraju, N.; et al. The Role of the Gastrointestinal Microbiome in Infectious Complications during Induction Chemotherapy for Acute Myeloid Leukemia. Cancer 2016, 122, 2186–2196. [Google Scholar] [CrossRef]
- Galloway-Peña, J.R.; Smith, D.P.; Sahasrabhojane, P.; Wadsworth, W.D.; Fellman, B.M.; Ajami, N.J.; Shpall, E.J.; Daver, N.; Guindani, M.; Petrosino, J.F.; et al. Characterization of Oral and Gut Microbiome Temporal Variability in Hospitalized Cancer Patients. Genome Med. 2017, 9, 21. [Google Scholar] [CrossRef]
- Kelly, M.S.; Ward, D.V.; Severyn, C.J.; Arshad, M.; Heston, S.M.; Jenkins, K.; Martin, P.L.; McGill, L.; Stokhuyzen, A.; Bhattarai, S.K.; et al. Gut Colonization Preceding Mucosal Barrier Injury Bloodstream Infection in Pediatric Hematopoietic Stem Cell Transplantation Recipients. Biol. Blood Marrow Transpl. 2019, 25, 2274–2280. [Google Scholar] [CrossRef]
- Lehouritis, P.; Cummins, J.; Stanton, M.; Murphy, C.T.; McCarthy, F.O.; Reid, G.; Urbaniak, C.; Byrne, W.L.; Tangney, M. Local Bacteria Affect the Efficacy of Chemotherapeutic Drugs. Sci. Rep. 2015, 5, 14554. [Google Scholar] [CrossRef]
- Gyurkocza, B.; Rezvani, A.; Storb, R.F. Allogeneic Hematopoietic Cell Transplantation: The State of the Art. Expert Rev. Hematol. 2010, 3, 285–299. [Google Scholar] [CrossRef]
- Malard, F.; Huang, X.-J.; Sim, J.P.Y. Treatment and Unmet Needs in Steroid-Refractory Acute Graft-versus-Host Disease. Leukemia 2020, 34, 1229–1240. [Google Scholar] [CrossRef] [PubMed]
- DiMaggio, E. Acute Graft-versus-Host Disease: Emerging Insights and Updates into Detection, Prevention, and Treatment. Pharmacotherapy 2020, 40, 788–807. [Google Scholar] [CrossRef] [PubMed]
- Filipovich, A.H.; Weisdorf, D.; Pavletic, S.; Socie, G.; Wingard, J.R.; Lee, S.J.; Martin, P.; Chien, J.; Przepiorka, D.; Couriel, D.; et al. National Institutes of Health Consensus Development Project on Criteria for Clinical Trials in Chronic Graft-versus-Host Disease: I. Diagnosis and Staging Working Group Report. Biol. Blood Marrow Transpl. 2005, 11, 945–956. [Google Scholar] [CrossRef] [PubMed]
- Harris, A.C.; Ferrara, J.L.M.; Levine, J.E. Advances in Predicting Acute GVHD. Br. J. Haematol. 2013, 160, 288–302. [Google Scholar] [CrossRef] [PubMed]
- Jacobsohn, D.A.; Kurland, B.F.; Pidala, J.; Inamoto, Y.; Chai, X.; Palmer, J.M.; Arai, S.; Arora, M.; Jagasia, M.; Cutler, C.; et al. Correlation between NIH Composite Skin Score, Patient-Reported Skin Score, and Outcome: Results from the Chronic GVHD Consortium. Blood 2012, 120, 2545–2552, quiz 2774. [Google Scholar] [CrossRef] [PubMed]
- Shulman, H.M.; Cardona, D.M.; Greenson, J.K.; Hingorani, S.; Horn, T.; Huber, E.; Kreft, A.; Longerich, T.; Morton, T.; Myerson, D.; et al. NIH Consensus Development Project on Criteria for Clinical Trials in Chronic Graft-versus-Host Disease: II. The 2014 Pathology Working Group Report. Biol. Blood Marrow Transpl. 2015, 21, 589–603. [Google Scholar] [CrossRef] [PubMed]
- Ferrara, J.L.M.; Levine, J.E.; Reddy, P.; Holler, E. Graft-versus-Host Disease. Lancet 2009, 373, 1550–1561. [Google Scholar] [CrossRef]
- Zeiser, R.; Blazar, B.R. Acute Graft-versus-Host Disease-Biologic Process, Prevention, and Therapy. N. Engl. J. Med. 2017, 377, 2167–2179. [Google Scholar] [CrossRef]
- Martin, P.J.; Inamoto, Y.; Flowers, M.E.D.; Carpenter, P.A. Secondary Treatment of Acute Graft-versus-Host Disease: A Critical Review. Biol. Blood Marrow Transpl. 2012, 18, 982–988. [Google Scholar] [CrossRef]
- Taur, Y.; Xavier, J.B.; Lipuma, L.; Ubeda, C.; Goldberg, J.; Gobourne, A.; Lee, Y.J.; Dubin, K.A.; Socci, N.D.; Viale, A.; et al. Intestinal Domination and the Risk of Bacteremia in Patients Undergoing Allogeneic Hematopoietic Stem Cell Transplantation. Clin. Infect. Dis. 2012, 55, 905–914. [Google Scholar] [CrossRef]
- Taur, Y.; Jenq, R.R.; Ubeda, C.; van den Brink, M.; Pamer, E.G. Role of Intestinal Microbiota in Transplantation Outcomes. Best Pract. Res. Clin. Haematol. 2015, 28, 155–161. [Google Scholar] [CrossRef] [PubMed]
- Shono, Y.; van den Brink, M.R.M. Gut Microbiota Injury in Allogeneic Haematopoietic Stem Cell Transplantation. Nat. Rev. Cancer 2018, 18, 283–295. [Google Scholar] [CrossRef] [PubMed]
- Connell, M.S.; Wilson, R. The treatment of x-irradiated germfree cfw and c3h mice with isologous and homologous bone marrow. Life Sci. 1965, 4, 721–729. [Google Scholar] [CrossRef]
- Jones, J.M.; Wilson, R.; Bealmear, P.M. Mortality and Gross Pathology of Secondary Disease in Germfree Mouse Radiation Chimeras. Radiat. Res. 1971, 45, 577–588. [Google Scholar] [CrossRef] [PubMed]
- van Bekkum, D.W.; Roodenburg, J.; Heidt, P.J.; van der Waaij, D. Mitigation of Secondary Disease of Allogeneic Mouse Radiation Chimeras by Modification of the Intestinal Microflora. J. Natl. Cancer Inst. 1974, 52, 401–404. [Google Scholar] [CrossRef]
- Bealmear, P.M.; Mirand, E.A.; Holtermann, O.A. Modification of Graft-vs-Host Disease Following Bone Marrow Transplantation in Germfree Mice. Prog. Clin. Biol. Res. 1983, 132C, 409–421. [Google Scholar]
- Wade, A.C.; Luckert, P.H.; Tazume, S.; Niedbalski, J.L.; Pollard, M. Characterization of Xenogeneic Mouse-to-Rat Bone Marrow Chimeras. I. Examination of Hematologic and Immunologic Function. Transplantation 1987, 44, 88–92. [Google Scholar] [CrossRef]
- Lampert, I.A.; Moore, R.H.; Huby, R.; Cohen, J. Observations on the Role of Endotoxin in Graft-versus-Host Disease. Prog. Clin. Biol. Res. 1988, 272, 351–359. [Google Scholar]
- Heidt, P.J.; Vossen, J.M. Experimental and Clinical Gnotobiotics: Influence of the Microflora on Graft-versus-Host Disease after Allogeneic Bone Marrow Transplantation. J. Med. 1992, 23, 161–173. [Google Scholar]
- Hooper, L.V.; Littman, D.R.; Macpherson, A.J. Interactions between the Microbiota and the Immune System. Science 2012, 336, 1268–1273. [Google Scholar] [CrossRef]
- Round, J.L.; Mazmanian, S.K. The Gut Microbiota Shapes Intestinal Immune Responses during Health and Disease. Nat. Rev. Immunol. 2009, 9, 313–323. [Google Scholar] [CrossRef] [PubMed]
- Buckner, C.D.; Clift, R.A.; Sanders, J.E.; Meyers, J.D.; Counts, G.W.; Farewell, V.T.; Thomas, E.D. Protective Environment for Marrow Transplant Recipients: A Prospective Study. Ann. Intern. Med. 1978, 89, 893–901. [Google Scholar] [CrossRef] [PubMed]
- Petersen, F.B.; Buckner, C.D.; Clift, R.A.; Nelson, N.; Counts, G.W.; Meyers, J.D.; Thomas, E.D. Prevention of Nosocomial Infections in Marrow Transplant Patients: A Prospective Randomized Comparison of Systemic Antibiotics versus Granulocyte Transfusions. Infect. Control 1986, 7, 586–592. [Google Scholar] [CrossRef] [PubMed]
- Petersen, F.B.; Buckner, C.D.; Clift, R.A.; Lee, S.; Nelson, N.; Counts, G.W.; Meyers, J.D.; Sanders, J.E.; Stewart, P.S.; Bensinger, W.I. Laminar Air Flow Isolation and Decontamination: A Prospective Randomized Study of the Effects of Prophylactic Systemic Antibiotics in Bone Marrow Transplant Patients. Infection 1986, 14, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Storb, R.; Prentice, R.L.; Buckner, C.D.; Clift, R.A.; Appelbaum, F.; Deeg, J.; Doney, K.; Hansen, J.A.; Mason, M.; Sanders, J.E.; et al. Graft-versus-Host Disease and Survival in Patients with Aplastic Anemia Treated by Marrow Grafts from HLA-Identical Siblings. Beneficial Effect of a Protective Environment. N. Engl. J. Med. 1983, 308, 302–307. [Google Scholar] [CrossRef] [PubMed]
- Petersen, F.B.; Buckner, C.D.; Clift, R.A.; Nelson, N.; Counts, G.W.; Meyers, J.D.; Thomas, E.D. Infectious Complications in Patients Undergoing Marrow Transplantation: A Prospective Randomized Study of the Additional Effect of Decontamination and Laminar Air Flow Isolation among Patients Receiving Prophylactic Systemic Antibiotics. Scand. J. Infect. Dis. 1987, 19, 559–567. [Google Scholar] [CrossRef]
- Beelen, D.W.; Haralambie, E.; Brandt, H.; Linzenmeier, G.; Müller, K.D.; Quabeck, K.; Sayer, H.G.; Graeven, U.; Mahmoud, H.K.; Schaefer, U.W. Evidence That Sustained Growth Suppression of Intestinal Anaerobic Bacteria Reduces the Risk of Acute Graft-versus-Host Disease after Sibling Marrow Transplantation. Blood 1992, 80, 2668–2676. [Google Scholar] [CrossRef]
- Shono, Y.; Docampo, M.D.; Peled, J.U.; Perobelli, S.M.; Velardi, E.; Tsai, J.J.; Slingerland, A.E.; Smith, O.M.; Young, L.F.; Gupta, J.; et al. Increased GVHD-Related Mortality with Broad-Spectrum Antibiotic Use after Allogeneic Hematopoietic Stem Cell Transplantation in Human Patients and Mice. Sci. Transl. Med. 2016, 8, 339ra71. [Google Scholar] [CrossRef]
- Nishi, K.; Kanda, J.; Hishizawa, M.; Kitano, T.; Kondo, T.; Yamashita, K.; Takaori-Kondo, A. Impact of the Use and Type of Antibiotics on Acute Graft-versus-Host Disease. Biol. Blood Marrow Transpl. 2018, 24, 2178–2183. [Google Scholar] [CrossRef]
- Weber, D.; Hiergeist, A.; Weber, M.; Dettmer, K.; Wolff, D.; Hahn, J.; Herr, W.; Gessner, A.; Holler, E. Detrimental Effect of Broad-Spectrum Antibiotics on Intestinal Microbiome Diversity in Patients After Allogeneic Stem Cell Transplantation: Lack of Commensal Sparing Antibiotics. Clin. Infect. Dis. 2019, 68, 1303–1310. [Google Scholar] [CrossRef]
- Vossen, J.M.; Heidt, P.J.; van den Berg, H.; Gerritsen, E.J.; Hermans, J.; Dooren, L.J. Prevention of Infection and Graft-versus-Host Disease by Suppression of Intestinal Microflora in Children Treated with Allogeneic Bone Marrow Transplantation. Eur. J. Clin. Microbiol. Infect. Dis. 1990, 9, 14–23. [Google Scholar] [CrossRef] [PubMed]
- Beelen, D.W.; Elmaagacli, A.; Müller, K.D.; Hirche, H.; Schaefer, U.W. Influence of Intestinal Bacterial Decontamination Using Metronidazole and Ciprofloxacin or Ciprofloxacin Alone on the Development of Acute Graft-versus-Host Disease after Marrow Transplantation in Patients with Hematologic Malignancies: Final Results and Long-Term Follow-up of an Open-Label Prospective Randomized Trial. Blood 1999, 93, 3267–3275. [Google Scholar] [PubMed]
- Weber, D.; Oefner, P.J.; Dettmer, K.; Hiergeist, A.; Koestler, J.; Gessner, A.; Weber, M.; Stämmler, F.; Hahn, J.; Wolff, D.; et al. Rifaximin Preserves Intestinal Microbiota Balance in Patients Undergoing Allogeneic Stem Cell Transplantation. Bone Marrow Transpl. 2016, 51, 1087–1092. [Google Scholar] [CrossRef] [PubMed]
- Köhler, N.; Zeiser, R. Intestinal Microbiota Influence Immune Tolerance Post Allogeneic Hematopoietic Cell Transplantation and Intestinal GVHD. Front Immunol 2018, 9, 3179. [Google Scholar] [CrossRef] [PubMed]
- Taur, Y.; Jenq, R.R.; Perales, M.-A.; Littmann, E.R.; Morjaria, S.; Ling, L.; No, D.; Gobourne, A.; Viale, A.; Dahi, P.B.; et al. The Effects of Intestinal Tract Bacterial Diversity on Mortality Following Allogeneic Hematopoietic Stem Cell Transplantation. Blood 2014, 124, 1174–1182. [Google Scholar] [CrossRef] [PubMed]
- Jenq, R.R.; Taur, Y.; Devlin, S.M.; Ponce, D.M.; Goldberg, J.D.; Ahr, K.F.; Littmann, E.R.; Ling, L.; Gobourne, A.C.; Miller, L.C.; et al. Intestinal Blautia Is Associated with Reduced Death from Graft-versus-Host Disease. Biol. Blood Marrow Transpl. 2015, 21, 1373–1383. [Google Scholar] [CrossRef]
- Golob, J.L.; Pergam, S.A.; Srinivasan, S.; Fiedler, T.L.; Liu, C.; Garcia, K.; Mielcarek, M.; Ko, D.; Aker, S.; Marquis, S.; et al. Stool Microbiota at Neutrophil Recovery Is Predictive for Severe Acute Graft vs Host Disease After Hematopoietic Cell Transplantation. Clin. Infect. Dis. 2017, 65, 1984–1991. [Google Scholar] [CrossRef]
- Peled, J.U.; Gomes, A.L.C.; Devlin, S.M.; Littmann, E.R.; Taur, Y.; Sung, A.D.; Weber, D.; Hashimoto, D.; Slingerland, A.E.; Slingerland, J.B.; et al. Microbiota as Predictor of Mortality in Allogeneic Hematopoietic-Cell Transplantation. N. Engl. J. Med. 2020, 382, 822–834. [Google Scholar] [CrossRef]
- Gavriilaki, M.; Sakellari, I.; Anagnostopoulos, A.; Gavriilaki, E. The Impact of Antibiotic-Mediated Modification of the Intestinal Microbiome on Outcomes of Allogeneic Hematopoietic Cell Transplantation: Systematic Review and Meta-Analysis. Biol. Blood Marrow Transpl. 2020, 26, 1738–1746. [Google Scholar] [CrossRef]
- Jenq, R.R.; Ubeda, C.; Taur, Y.; Menezes, C.C.; Khanin, R.; Dudakov, J.A.; Liu, C.; West, M.L.; Singer, N.V.; Equinda, M.J.; et al. Regulation of Intestinal Inflammation by Microbiota Following Allogeneic Bone Marrow Transplantation. J. Exp. Med. 2012, 209, 903–911. [Google Scholar] [CrossRef]
- Fredricks, D.N. The Gut Microbiota and Graft-versus-Host Disease. J. Clin. Invest. 2019, 129, 1808–1817. [Google Scholar] [CrossRef]
- Gorshein, E.; Wei, C.; Ambrosy, S.; Budney, S.; Vivas, J.; Shenkerman, A.; Manago, J.; McGrath, M.K.; Tyno, A.; Lin, Y.; et al. Lactobacillus Rhamnosus GG Probiotic Enteric Regimen Does Not Appreciably Alter the Gut Microbiome or Provide Protection against GVHD after Allogeneic Hematopoietic Stem Cell Transplantation. Clin. Transpl. 2017, 31. [Google Scholar] [CrossRef] [PubMed]
- Biagi, E.; Zama, D.; Nastasi, C.; Consolandi, C.; Fiori, J.; Rampelli, S.; Turroni, S.; Centanni, M.; Severgnini, M.; Peano, C.; et al. Gut Microbiota Trajectory in Pediatric Patients Undergoing Hematopoietic SCT. Bone Marrow Transpl. 2015, 50, 992–998. [Google Scholar] [CrossRef] [PubMed]
- Laterza, L.; Rizzatti, G.; Gaetani, E.; Chiusolo, P.; Gasbarrini, A. The Gut Microbiota and Immune System Relationship in Human Graft-versus-Host Disease. Mediterr. J. Hematol. Infect. Dis. 2016, 8, e2016025. [Google Scholar] [CrossRef] [PubMed]
- Holler, E.; Butzhammer, P.; Schmid, K.; Hundsrucker, C.; Koestler, J.; Peter, K.; Zhu, W.; Sporrer, D.; Hehlgans, T.; Kreutz, M.; et al. Metagenomic Analysis of the Stool Microbiome in Patients Receiving Allogeneic Stem Cell Transplantation: Loss of Diversity Is Associated with Use of Systemic Antibiotics and More Pronounced in Gastrointestinal Graft-versus-Host Disease. Biol. Blood Marrow Transpl. 2014, 20, 640–645. [Google Scholar] [CrossRef]
- Stein-Thoeringer, C.; Peled, J.U.; Lazrak, A.; Slingerland, A.E.; Shono, Y.; Staffas, A.; Gomes, A.; Argyropoulos, K.; Docampo, M.; Xavier, J.; et al. 20-Domination of the Gut Microbiota with Enterococcus Species Early after Allogeneic Bone Marrow Transplantation Is an Important Contributor to the Development of Acute Graft-Versus-Host Disease (GHVD) in Mouse and Man. Biol. Blood Marrow Transpl. 2018, 24, S40–S41. [Google Scholar] [CrossRef]
- Eriguchi, Y.; Takashima, S.; Oka, H.; Shimoji, S.; Nakamura, K.; Uryu, H.; Shimoda, S.; Iwasaki, H.; Shimono, N.; Ayabe, T.; et al. Graft-versus-Host Disease Disrupts Intestinal Microbial Ecology by Inhibiting Paneth Cell Production of α-Defensins. Blood 2012, 120, 223–231. [Google Scholar] [CrossRef]
- Heimesaat, M.M.; Nogai, A.; Bereswill, S.; Plickert, R.; Fischer, A.; Loddenkemper, C.; Steinhoff, U.; Tchaptchet, S.; Thiel, E.; Freudenberg, M.A.; et al. MyD88/TLR9 Mediated Immunopathology and Gut Microbiota Dynamics in a Novel Murine Model of Intestinal Graft-versus-Host Disease. Gut 2010, 59, 1079–1087. [Google Scholar] [CrossRef]
- Stein-Thoeringer, C.K.; Nichols, K.B.; Lazrak, A.; Docampo, M.D.; Slingerland, A.E.; Slingerland, J.B.; Clurman, A.G.; Armijo, G.; Gomes, A.L.C.; Shono, Y.; et al. Lactose Drives Enterococcus Expansion to Promote Graft-versus-Host Disease. Science 2019, 366, 1143–1149. [Google Scholar] [CrossRef]
- Simms-Waldrip, T.R.; Sunkersett, G.; Coughlin, L.A.; Savani, M.R.; Arana, C.; Kim, J.; Kim, M.; Zhan, X.; Greenberg, D.E.; Xie, Y.; et al. Antibiotic-Induced Depletion of Anti-Inflammatory Clostridia Is Associated with the Development of Graft-versus-Host Disease in Pediatric Stem Cell Transplantation Patients. Biol. Blood Marrow Transpl. 2017, 23, 820–829. [Google Scholar] [CrossRef]
- Zwielehner, J.; Lassl, C.; Hippe, B.; Pointner, A.; Switzeny, O.J.; Remely, M.; Kitzweger, E.; Ruckser, R.; Haslberger, A.G. Changes in Human Fecal Microbiota Due to Chemotherapy Analyzed by TaqMan-PCR, 454 Sequencing and PCR-DGGE Fingerprinting. PLoS ONE 2011, 6, e28654. [Google Scholar] [CrossRef] [PubMed]
- Montassier, E.; Batard, E.; Massart, S.; Gastinne, T.; Carton, T.; Caillon, J.; Le Fresne, S.; Caroff, N.; Hardouin, J.B.; Moreau, P.; et al. 16S RRNA Gene Pyrosequencing Reveals Shift in Patient Faecal Microbiota during High-Dose Chemotherapy as Conditioning Regimen for Bone Marrow Transplantation. Microb. Ecol. 2014, 67, 690–699. [Google Scholar] [CrossRef] [PubMed]
- Schluter, J.; Peled, J.U.; Taylor, B.P.; Markey, K.A.; Smith, M.; Taur, Y.; Niehus, R.; Staffas, A.; Dai, A.; Fontana, E.; et al. The Gut Microbiota Is Associated with Immune Cell Dynamics in Humans. Nature 2020, 588, 303–307. [Google Scholar] [CrossRef] [PubMed]
- Swimm, A.; Giver, C.R.; DeFilipp, Z.; Rangaraju, S.; Sharma, A.; Ulezko Antonova, A.; Sonowal, R.; Capaldo, C.; Powell, D.; Qayed, M.; et al. Indoles Derived from Intestinal Microbiota Act via Type I Interferon Signaling to Limit Graft-versus-Host Disease. Blood 2018, 132, 2506–2519. [Google Scholar] [CrossRef] [PubMed]
- Weber, D.; Oefner, P.J.; Hiergeist, A.; Koestler, J.; Gessner, A.; Weber, M.; Hahn, J.; Wolff, D.; Stämmler, F.; Spang, R.; et al. Low Urinary Indoxyl Sulfate Levels Early after Transplantation Reflect a Disrupted Microbiome and Are Associated with Poor Outcome. Blood 2015, 126, 1723–1728. [Google Scholar] [CrossRef] [PubMed]
- Mathewson, N.D.; Jenq, R.; Mathew, A.V.; Koenigsknecht, M.; Hanash, A.; Toubai, T.; Oravecz-Wilson, K.; Wu, S.-R.; Sun, Y.; Rossi, C.; et al. Gut Microbiome-Derived Metabolites Modulate Intestinal Epithelial Cell Damage and Mitigate Graft-versus-Host Disease. Nat. Immunol. 2016, 17, 505–513. [Google Scholar] [CrossRef]
- Levine, J.E.; Huber, E.; Hammer, S.T.G.; Harris, A.C.; Greenson, J.K.; Braun, T.M.; Ferrara, J.L.M.; Holler, E. Low Paneth Cell Numbers at Onset of Gastrointestinal Graft-versus-Host Disease Identify Patients at High Risk for Nonrelapse Mortality. Blood 2013, 122, 1505–1509. [Google Scholar] [CrossRef]
- Ubeda, C.; Taur, Y.; Jenq, R.R.; Equinda, M.J.; Son, T.; Samstein, M.; Viale, A.; Socci, N.D.; van den Brink, M.R.M.; Kamboj, M.; et al. Vancomycin-Resistant Enterococcus Domination of Intestinal Microbiota Is Enabled by Antibiotic Treatment in Mice and Precedes Bloodstream Invasion in Humans. J. Clin. Invest. 2010, 120, 4332–4341. [Google Scholar] [CrossRef]
- Ubeda, C.; Bucci, V.; Caballero, S.; Djukovic, A.; Toussaint, N.C.; Equinda, M.; Lipuma, L.; Ling, L.; Gobourne, A.; No, D.; et al. Intestinal Microbiota Containing Barnesiella Species Cures Vancomycin-Resistant Enterococcus Faecium Colonization. Infect. Immun. 2013, 81, 965–973. [Google Scholar] [CrossRef] [PubMed]
- Legoff, J.; Resche-Rigon, M.; Bouquet, J.; Robin, M.; Naccache, S.N.; Mercier-Delarue, S.; Federman, S.; Samayoa, E.; Rousseau, C.; Piron, P.; et al. The Eukaryotic Gut Virome in Hematopoietic Stem Cell Transplantation: New Clues in Enteric Graft-versus-Host Disease. Nat. Med. 2017, 23, 1080–1085. [Google Scholar] [CrossRef]
- Laheij, A.M.G.A.; Raber-Durlacher, J.E.; Koppelmans, R.G.A.; Huysmans, M.-C.D.N.J.M.; Potting, C.; van Leeuwen, S.J.M.; Hazenberg, M.D.; Brennan, M.T.; von Bültzingslöwen, I.; Johansson, J.-E.; et al. Microbial Changes in Relation to Oral Mucositis in Autologous Hematopoietic Stem Cell Transplantation Recipients. Sci. Rep. 2019, 9, 16929. [Google Scholar] [CrossRef] [PubMed]
- Galloway-Peña, J.R.; Shi, Y.; Peterson, C.B.; Sahasrabhojane, P.; Gopalakrishnan, V.; Brumlow, C.E.; Daver, N.G.; Alfayez, M.; Boddu, P.C.; Khan, M.A.W.; et al. Gut Microbiome Signatures Are Predictive of Infectious Risk Following Induction Therapy for Acute Myeloid Leukemia. Clin. Infect. Dis. 2020, 71, 63–71. [Google Scholar] [CrossRef] [PubMed]
- Pianko, M.J.; Devlin, S.M.; Littmann, E.R.; Chansakul, A.; Mastey, D.; Salcedo, M.; Fontana, E.; Ling, L.; Tavitian, E.; Slingerland, J.B.; et al. Minimal Residual Disease Negativity in Multiple Myeloma Is Associated with Intestinal Microbiota Composition. Blood Adv. 2019, 3, 2040–2044. [Google Scholar] [CrossRef] [PubMed]
- Smith, M.; Littmann, E.; Slingerland, J.; Clurman, A.; Slingerland, A.E.; Taur, Y.; Peled, J.U.; Park, J.; O’Cearbhaill, R.; Mailankody, S.; et al. Intestinal Microbiome Analyses Identify Biomarkers for Patient Response to CAR T Cell Therapy. Biol. Blood Marrow Transpl. 2019, 25, S177. [Google Scholar] [CrossRef][Green Version]
- Muraro, P.A.; Martin, R.; Mancardi, G.L.; Nicholas, R.; Sormani, M.P.; Saccardi, R. Autologous Haematopoietic Stem Cell Transplantation for Treatment of Multiple Sclerosis. Nat. Rev. Neurol. 2017, 13, 391–405. [Google Scholar] [CrossRef]
- Passweg, J.R.; Baldomero, H.; Chabannon, C.; Basak, G.W.; Corbacioglu, S.; Duarte, R.; Dolstra, H.; Lankester, A.C.; Mohty, M.; Montoto, S.; et al. The EBMT Activity Survey on Hematopoietic-Cell Transplantation and Cellular Therapy 2018: CAR-T’s Come into Focus. Bone Marrow Transpl. 2020, 55, 1604–1613. [Google Scholar] [CrossRef]
- Zahid, U.; Akbar, F.; Amaraneni, A.; Husnain, M.; Chan, O.; Riaz, I.B.; McBride, A.; Iftikhar, A.; Anwer, F. A Review of Autologous Stem Cell Transplantation in Lymphoma. Curr. Hematol. Malig. Rep. 2017, 12, 217–226. [Google Scholar] [CrossRef]
- Al Hamed, R.; Bazarbachi, A.H.; Malard, F.; Harousseau, J.-L.; Mohty, M. Current Status of Autologous Stem Cell Transplantation for Multiple Myeloma. Blood Cancer J. 2019, 9, 44. [Google Scholar] [CrossRef]
- El Jurdi, N.; Filali-Mouhim, A.; Salem, I.; Retuerto, M.; Dambrosio, N.M.; Baer, L.; Lazarus, H.M.; Caimi, P.; Cooper, B.; Tomlinson, B.; et al. Gastrointestinal Microbiome and Mycobiome Changes during Autologous Transplantation for Multiple Myeloma: Results of a Prospective Pilot Study. Biol Blood Marrow Transpl. 2019, 25, 1511–1519. [Google Scholar] [CrossRef]
- Kusakabe, S.; Fukushima, K.; Maeda, T.; Motooka, D.; Nakamura, S.; Fujita, J.; Yokota, T.; Shibayama, H.; Oritani, K.; Kanakura, Y. Pre- and Post-Serial Metagenomic Analysis of Gut Microbiota as a Prognostic Factor in Patients Undergoing Haematopoietic Stem Cell Transplantation. Br. J. Haematol. 2020, 188, 438–449. [Google Scholar] [CrossRef]
- Montassier, E.; Gastinne, T.; Vangay, P.; Al-Ghalith, G.A.; Bruley des Varannes, S.; Massart, S.; Moreau, P.; Potel, G.; de La Cochetière, M.F.; Batard, E.; et al. Chemotherapy-Driven Dysbiosis in the Intestinal Microbiome. Aliment. Pharmacol. Ther. 2015, 42, 515–528. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez-García, G.; Rovira, M.; Magnano, L.; Rosiñol, L.; Bataller, A.; Suárez-Lledó, M.; Cibeira, M.T.; de Larrea, C.F.; Garrote, M.; Jorge, S.; et al. Innovative Strategies Minimize Engraftment Syndrome in Multiple Myeloma Patients with Novel Induction Therapy Following Autologous Hematopoietic Stem Cell Transplantation. Bone Marrow Transpl. 2018, 53, 1541–1547. [Google Scholar] [CrossRef]
- Ziegler, M.; Han, J.H.; Landsburg, D.; Pegues, D.; Reesey, E.; Gilmar, C.; Gorman, T.; Bink, K.; Moore, A.; Kelly, B.J. Impact of Levofloxacin for the Prophylaxis of Bloodstream Infection on the Gut Microbiome in Patients With Hematologic Malignancy. Open Forum Infect. Dis. 2019, 6. [Google Scholar] [CrossRef] [PubMed]
- Kristinsson, S.Y.; Anderson, W.F.; Landgren, O. Improved Long-Term Survival in Multiple Myeloma up to the Age of 80 Years. Leukemia 2014, 28, 1346–1348. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.K.; Dispenzieri, A.; Lacy, M.Q.; Gertz, M.A.; Buadi, F.K.; Pandey, S.; Kapoor, P.; Dingli, D.; Hayman, S.R.; Leung, N.; et al. Continued Improvement in Survival in Multiple Myeloma: Changes in Early Mortality and Outcomes in Older Patients. Leukemia 2014, 28, 1122–1128. [Google Scholar] [CrossRef] [PubMed]
- Thorsteinsdottir, S.; Dickman, P.W.; Landgren, O.; Blimark, C.; Hultcrantz, M.; Turesson, I.; Björkholm, M.; Kristinsson, S.Y. Dramatically Improved Survival in Multiple Myeloma Patients in the Recent Decade: Results from a Swedish Population-Based Study. Haematologica 2018, 103, e412–e415. [Google Scholar] [CrossRef]
- June, C.H.; Sadelain, M. Chimeric Antigen Receptor Therapy. N. Engl. J. Med. 2018, 379, 64–73. [Google Scholar] [CrossRef]
- Maude, S.L.; Laetsch, T.W.; Buechner, J.; Rives, S.; Boyer, M.; Bittencourt, H.; Bader, P.; Verneris, M.R.; Stefanski, H.E.; Myers, G.D.; et al. Tisagenlecleucel in Children and Young Adults with B-Cell Lymphoblastic Leukemia. N. Engl. J. Med. 2018, 378, 439–448. [Google Scholar] [CrossRef]
- Schuster, S.J.; Svoboda, J.; Chong, E.A.; Nasta, S.D.; Mato, A.R.; Anak, Ö.; Brogdon, J.L.; Pruteanu-Malinici, I.; Bhoj, V.; Landsburg, D.; et al. Chimeric Antigen Receptor T Cells in Refractory B-Cell Lymphomas. N. Engl. J. Med. 2017, 377, 2545–2554. [Google Scholar] [CrossRef]
- Neelapu, S.S.; Locke, F.L.; Bartlett, N.L.; Lekakis, L.J.; Miklos, D.B.; Jacobson, C.A.; Braunschweig, I.; Oluwole, O.O.; Siddiqi, T.; Lin, Y.; et al. Axicabtagene Ciloleucel CAR T-Cell Therapy in Refractory Large B-Cell Lymphoma. N. Engl. J. Med. 2017, 377, 2531–2544. [Google Scholar] [CrossRef]
- Park, J.H.; Rivière, I.; Gonen, M.; Wang, X.; Sénéchal, B.; Curran, K.J.; Sauter, C.; Wang, Y.; Santomasso, B.; Mead, E.; et al. Long-Term Follow-up of CD19 CAR Therapy in Acute Lymphoblastic Leukemia. N. Engl. J. Med. 2018, 378, 449–459. [Google Scholar] [CrossRef]
- Rodríguez-Lobato, L.G.; Ganzetti, M.; Fernández de Larrea, C.; Hudecek, M.; Einsele, H.; Danhof, S. CAR T-Cells in Multiple Myeloma: State of the Art and Future Directions. Front. Oncol. 2020, 10, 1243. [Google Scholar] [CrossRef] [PubMed]
- Gagelmann, N.; Riecken, K.; Wolschke, C.; Berger, C.; Ayuk, F.A.; Fehse, B.; Kröger, N. Development of CAR-T Cell Therapies for Multiple Myeloma. Leukemia 2020, 34, 2317–2332. [Google Scholar] [CrossRef] [PubMed]
- Sonnenburg, J.L.; Bäckhed, F. Diet-Microbiota Interactions as Moderators of Human Metabolism. Nature 2016, 535, 56–64. [Google Scholar] [CrossRef] [PubMed]
- Thaiss, C.A.; Zeevi, D.; Levy, M.; Zilberman-Schapira, G.; Suez, J.; Tengeler, A.C.; Abramson, L.; Katz, M.N.; Korem, T.; Zmora, N.; et al. Transkingdom Control of Microbiota Diurnal Oscillations Promotes Metabolic Homeostasis. Cell 2014, 159, 514–529. [Google Scholar] [CrossRef]
- Barbaresko, J.; Koch, M.; Schulze, M.B.; Nöthlings, U. Dietary Pattern Analysis and Biomarkers of Low-Grade Inflammation: A Systematic Literature Review. Nutr. Rev. 2013, 71, 511–527. [Google Scholar] [CrossRef]
- Solans, M.; Castelló, A.; Benavente, Y.; Marcos-Gragera, R.; Amiano, P.; Gracia-Lavedan, E.; Costas, L.; Robles, C.; Gonzalez-Barca, E.; de la Banda, E.; et al. Adherence to the Western, Prudent, and Mediterranean Dietary Patterns and Chronic Lymphocytic Leukemia in the MCC-Spain Study. Haematologica 2018, 103, 1881–1888. [Google Scholar] [CrossRef]
- University of Michigan Rogel Cancer Center. Dietary Manipulation of the Microbiome-Metabolomic Axis for Mitigating GVHD in Allo HCT Patients. 2019. Available online: clinicaltrials.gov (accessed on 17 November 2020).
- Monteagudo-Mera, A.; Rastall, R.A.; Gibson, G.R.; Charalampopoulos, D.; Chatzifragkou, A. Adhesion Mechanisms Mediated by Probiotics and Prebiotics and Their Potential Impact on Human Health. Appl. Microbiol. Biotechnol. 2019, 103, 6463–6472. [Google Scholar] [CrossRef]
- Shoaf, K.; Mulvey, G.L.; Armstrong, G.D.; Hutkins, R.W. Prebiotic Galactooligosaccharides Reduce Adherence of Enteropathogenic Escherichia Coli to Tissue Culture Cells. Infect. Immun. 2006, 74, 6920–6928. [Google Scholar] [CrossRef]
- Fructooligosaccharides in Treating Patients With Blood Cancer Undergoing Donor Stem Cell Transplant-Full Text View-ClinicalTrials.Gov. Available online: https://clinicaltrials.gov/ct2/show/NCT02805075 (accessed on 17 November 2020).
- Duke University. A Phase I/II Trial of the Prebiotic Galacto-Oligosaccharide to Prevent Acute GVHD. 2020. Available online: clinicaltrials.gov (accessed on 17 November 2020).
- Maga, E.A.; Desai, P.T.; Weimer, B.C.; Dao, N.; Kültz, D.; Murray, J.D. Consumption of Lysozyme-Rich Milk Can Alter Microbial Fecal Populations. Appl. Environ. Microbiol. 2012, 78, 6153–6160. [Google Scholar] [CrossRef]
- Ko, K.Y.; Mendoncam, A.F.; Ismail, H.; Ahn, D.U. Ethylenediaminetetraacetate and Lysozyme Improves Antimicrobial Activities of Ovotransferrin against Escherichia Coli O157:H7. Poult. Sci. 2009, 88, 406–414. [Google Scholar] [CrossRef]
- Human Lysozyme Goat Milk for the Prevention of Graft Versus Host Disease in Patients With Blood Cancer Undergoing a Donor Stem Cell Transplant-Full Text View-ClinicalTrials.Gov. Available online: https://clinicaltrials.gov/ct2/show/NCT04177004 (accessed on 24 November 2020).
- Mills, J.P.; Rao, K.; Young, V.B. Probiotics for Prevention of Clostridium Difficile Infection. Curr. Opin. Gastroenterol. 2018, 34, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Klaenhammer, T.R.; Kleerebezem, M.; Kopp, M.V.; Rescigno, M. The Impact of Probiotics and Prebiotics on the Immune System. Nat. Rev. Immunol. 2012, 12, 728–734. [Google Scholar] [CrossRef] [PubMed]
- CBM588 in Improving Clinical Outcomes in Patients Who Have Undergone Donor Hematopoietic Stem Cell Transplant-Full Text View-ClinicalTrials.Gov. Available online: https://clinicaltrials.gov/ct2/show/NCT03922035 (accessed on 24 November 2020).
- Wang, Y.; Wiesnoski, D.H.; Helmink, B.A.; Gopalakrishnan, V.; Choi, K.; DuPont, H.L.; Jiang, Z.-D.; Abu-Sbeih, H.; Sanchez, C.A.; Chang, C.-C.; et al. Fecal Microbiota Transplantation for Refractory Immune Checkpoint Inhibitor-Associated Colitis. Nat. Med. 2018, 24, 1804. [Google Scholar] [CrossRef] [PubMed]
- Jonsson Comprehensive Cancer Center An Open-Label Phase 1 Pilot Study: Fecal Microbiota Transplantation (FMT) in Severe Acute Gut Graft-Versus-Host Disease Patients. 2020. Available online: https://clinicaltrials.gov/ct2/show/NCT04280471 (accessed on 24 November 2020).
- Medical University of Graz Fecal Microbiota Transplantation in Patients With Acute Gastrointestinal Graft-versus-Host-Disease After Allogeneic Stem Cell Transplantation. 2020. Available online: clinicaltrials.gov (accessed on 24 November 2020).
- Metheny, L. Fecal Microbiota Transplantation For The Treatment Of Gastro-Intestinal Acute Graft Versus Host Disease. 2020. Available online: clinicaltrials.gov (accessed on 24 November 2020).
- Moiseev, I.S. Pilot Study of Fecal Microbiota Transplantation in Combination With Ruxolitinib and Steroids for Severe Acute Intestinal Graft-versus-Host-Disease After Allogeneic Hematopoietic Stem Cell Transplantation. 2020. Available online: clinicaltrials.gov (accessed on 24 November 2020).
- Qing, C. Fecal Microbiota Transplantation for Treatment of Refractory Graft Versus Host Disease—A Pilot Study. 2019. Available online: clinicaltrials.gov (accessed on 24 November 2020).
- Wan, L. Fecal Microbiota Transplantation for the Treatment of Steroid Resistant/Dependent Acute Gastrointestinal Graft Versus Host Disease. 2020. Available online: clinicaltrials.gov (accessed on 24 November 2020).
- Liu, Q. Investigator-Initiated Trial to Explore the Efficacy and Safety of Fecal Microbiota Transplant (FMT) Capsule in the Treatment of Steroid-Refractory Gastrointestinal Acute Graft Versus Host Disease Patients. Available online: clinicaltrials.gov (accessed on 24 November 2020).
- FMT In High-Risk Acute GVHD After ALLO HCT-Full Text-ClinicalTrials.Gov. Available online: https://clinicaltrials.gov/ct2/show/NCT04139577 (accessed on 17 November 2020).
- FMT for Steroid Resistant Gut Acute GVHD-Full Text View-ClinicalTrials.Gov. Available online: https://clinicaltrials.gov/ct2/show/NCT04285424 (accessed on 17 November 2020).
- Masonic Cancer Center, University of Minnesota A Randomized Placebo-Controlled Clinical Trial of Fecal Microbiota Transplantation in Patients With Acute Myeloid Leukemia and Allogeneic Hematopoietic Cell Transplantation Recipients. 2020. Available online: clinicaltrials.gov (accessed on 24 November 2020).
- MaaT Pharma PreventiOn of DYSbioSis Complications With Autologous Fecal Microbiota Transplantation in AcutE MyEloid Leukemia Patients Undergoing Intensive Treatment: A Feasibility and Safety Study ODYSSEE STUDY. 2020. Available online: clinicaltrials.gov (accessed on 24 November 2020).
- Kao, D.; Roach, B.; Silva, M.; Beck, P.; Rioux, K.; Kaplan, G.G.; Chang, H.-J.; Coward, S.; Goodman, K.J.; Xu, H.; et al. Effect of Oral Capsule- vs Colonoscopy-Delivered Fecal Microbiota Transplantation on Recurrent Clostridium Difficile Infection: A Randomized Clinical Trial. JAMA 2017, 318, 1985–1993. [Google Scholar] [CrossRef] [PubMed]
- DeFilipp, Z.; Bloom, P.P.; Torres Soto, M.; Mansour, M.K.; Sater, M.R.A.; Huntley, M.H.; Turbett, S.; Chung, R.T.; Chen, Y.-B.; Hohmann, E.L. Drug-Resistant E. Coli Bacteremia Transmitted by Fecal Microbiota Transplant. N. Engl. J. Med. 2019, 381, 2043–2050. [Google Scholar] [CrossRef]
- FDA. Fecal Microbiota for Transplantation: Safety Alert-Risk of Serious Adverse Events Likely Due to Transmission of Pathogenic Organisms. 2020. Available online: https://www.fda.gov/safety/medical-product-safety-information/fecal-microbiota-transplantation-safety-alert-risk-serious-adverse-events-likely-due-transmission (accessed on 24 November 2020).
- Markey, K.A.; Schluter, J.; Gomes, A.L.C.; Littmann, E.R.; Pickard, A.J.; Taylor, B.P.; Giardina, P.A.; Weber, D.; Dai, A.; Docampo, M.D.; et al. The Microbe-Derived Short-Chain Fatty Acids Butyrate and Propionate Are Associated with Protection from Chronic GVHD. Blood 2020, 136, 130–136. [Google Scholar] [CrossRef]
- Wu, K.; Yuan, Y.; Yu, H.; Dai, X.; Wang, S.; Sun, Z.; Wang, F.; Fei, H.; Lin, Q.; Jiang, H.; et al. The Gut Microbial Metabolite Trimethylamine N-Oxide Aggravates GVHD by Inducing M1 Macrophage Polarization in Mice. Blood 2020, 136, 501–515. [Google Scholar] [CrossRef]
- Optimization of Antibiotic Treatment in Hematopoietic Stem Cell Receptors-Tabular View-ClinicalTrials.Gov. Available online: https://clinicaltrials.gov/ct2/show/record/NCT03727113 (accessed on 18 November 2020).

| Mechanism | Outcome | Involved Drug (s) |
|---|---|---|
| Enzymatic modification of the drugs | Decreased levels | Cladribine, gemcitabine, doxorubicin, Idarubicin, etoposide, mitoxantrone [99] |
| Increased activity | Fludarabine, 6-Mercaptopurine [99] | |
| Translocation of Gram-positive bacteria | Generation of Th17 and Th1 lymphocytes | Cyclophosphamide [74] |
| Modulation of genotoxicity | Reduction of DNA damage and apoptosis | Oxaliplatin, cisplatin [87] |
| Treatment | Microbiota Feature | Outcome |
|---|---|---|
| Allogeneic hematopoietic stem cell transplantation | Decontamination of gut anaerobes | Lower risk of GvHD [127,132] |
| Decreased duodenal Paneth cells | Higher GI GvHD and NRM [148] | |
| Low intestinal microbiota diversity | Higher TRM, lower OS and GvHD-related mortality [135,138] | |
| Enterococcus spp. domination | Increased GI GvHD severity [145] Increased GvHD-related and overall mortality [146,157] Associated with blood stream infections [110,158] | |
| Blautia abundance | Reduced GvHD-related mortality [136] | |
| Clostridia spp. depletion | Increased GvHD [150] | |
| Barnesiella spp. abundance | Protection against Enterococcus domination [159] | |
| Akkermansia muciniphila domination | Mucus degradation [128] | |
| Lactobacillales domination | Associated with GvHD development [140] | |
| Eubacterium limosum abundance | Lower risk of relapse or progression, higher OS [90] | |
| Picobirnivirus presence | Severe GI GvHD [160] | |
| Lower urinary 3-indoxyl sulfate | Higher risk of GvHD, higher TRM, lower OS, higher dysbiosis [145,155] | |
| Autologous stem cell transplantation | Reduction in diversity index and an increased dominance index | Development of mucositis [161] |
| Decreased in Firmicutes and Actinobacteria and increased in Proteobacteria | Development of GI mucositis [161] | |
| Chemotherapy | Baseline levels of Porphyromonadaceae | Predictor of infection during induction for acute myeloid leukemia [162] |
| Relative abundance of E hallii | Higher negative minimal residual disease rate in bone marrow for multiple myeloma [163] | |
| CAR T-cell therapy | Oscillospiraceae, Ruminococcacaeae and Lachnospiraceae enriched | Association with complete remission after CAR T cell therapy [164] |
| Higher abundance of Lachnospiraceae | Development of cytokine release syndrome and/or neurotoxicity [164] |
| Condition | Intervention | Administration | Primary Aim | Phase | Participants | Status | Identifier |
|---|---|---|---|---|---|---|---|
| aGvHD | FMT | Orally via capsule | Efficacy, safety, and tolerability | I | 10 | NYR | NCT04280471 |
| GI aGvHD | FMT | Colonoscopy or gastroscopy | Efficacy and safety | II | 30 | R | NCT03812705 |
| GI aGvHD | FMT | Nasojejunal tube | Efficacy and safety | I | 15 | NYR | NCT03549676 |
| GI aGvHD | FMT | Orally via capsule | Efficacy and safety | I/II | 20 | R | NCT04269850 |
| GI aGvHD | FMT | Orally via capsule | Efficacy and safety | II | 17 | NYR | NCT04059757 |
| GI aGvHD | FMT | Colonoscopy or gastroscopy | Efficacy | III | 15 | R | NCT03819803 |
| GI aGvHD | FMT | Orally via capsule | Efficacy and safety | N/A | 10 | R | NCT04622475 |
| aGvHD | FMT | Orally via capsule | Feasibility and efficacy | I | 11 | NYR | NCT04139577 |
| GI aGvHD | FMT | Colonoscopy or duodenal nutrition tube injection | Efficacy | I | 30 | R | NCT04285424 |
| Allo-HSCT, AML | FMT | Orally via capsule | Efficacy and incidence of infections | II | 120 | R | NCT03678493 |
| AML | FMT | Rectal enema | Evaluation in dysbiosis correction and in MDRB eradication | I/II | 20 | C | NCT02928523 |
| aGvHD | FMT | Not specified | Efficacy | II | 24 | C | NCT03359980 |
| Allo-HSCT | FMT | Orally via capsule | Feasibility and efficacy | I | 18 | C | NCT02733744 |
| Allo-HSCT | Potato-based dietary starch | Orally | Incidence of grade II-IV GVHD | II | 70 | R | NCT02763033 |
| Allo-HSCT | Fructo-oligosaccharides prebiotic | Orally | Safety and tolerability | I | 15 | C | NCT02805075 |
| Allo-HSCT | Galacto-oligosaccharide prebiotic | Orally | Tolerability and incidence of Grade II-IV aGVHD | I/II | 128 | NYR | NCT04373057 |
| Allo-HSCT | Human lysozyme goat milk prebiotic | Orally | Safety and tolerability | I | 36 | NYR | NCT04177004 |
| Allo-HSCT | Clostridium butyricum CBM 588 Probiotic Strain | Orally | Safety | I | 36 | R | NCT03922035 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Uribe-Herranz, M.; Klein-González, N.; Rodríguez-Lobato, L.G.; Juan, M.; Fernández de Larrea, C. Gut Microbiota Influence in Hematological Malignancies: From Genesis to Cure. Int. J. Mol. Sci. 2021, 22, 1026. https://doi.org/10.3390/ijms22031026
Uribe-Herranz M, Klein-González N, Rodríguez-Lobato LG, Juan M, Fernández de Larrea C. Gut Microbiota Influence in Hematological Malignancies: From Genesis to Cure. International Journal of Molecular Sciences. 2021; 22(3):1026. https://doi.org/10.3390/ijms22031026
Chicago/Turabian StyleUribe-Herranz, Mireia, Nela Klein-González, Luis Gerardo Rodríguez-Lobato, Manel Juan, and Carlos Fernández de Larrea. 2021. "Gut Microbiota Influence in Hematological Malignancies: From Genesis to Cure" International Journal of Molecular Sciences 22, no. 3: 1026. https://doi.org/10.3390/ijms22031026
APA StyleUribe-Herranz, M., Klein-González, N., Rodríguez-Lobato, L. G., Juan, M., & Fernández de Larrea, C. (2021). Gut Microbiota Influence in Hematological Malignancies: From Genesis to Cure. International Journal of Molecular Sciences, 22(3), 1026. https://doi.org/10.3390/ijms22031026






