NCs-Delivered Pesticides: A Promising Candidate in Smart Agriculture
Abstract
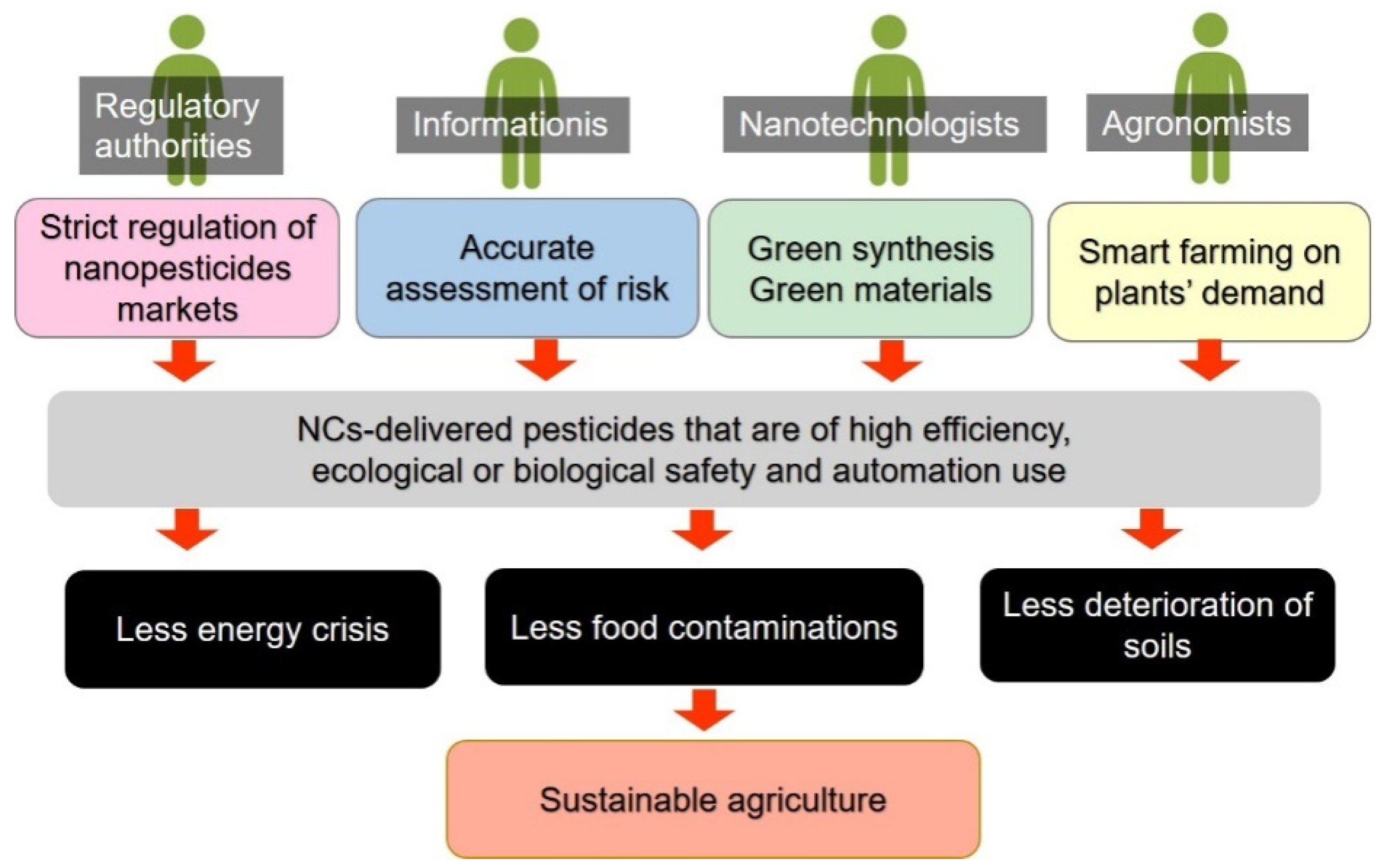
:1. Introduction
2. The Promising Applications of Nanocarriers in Pest Control
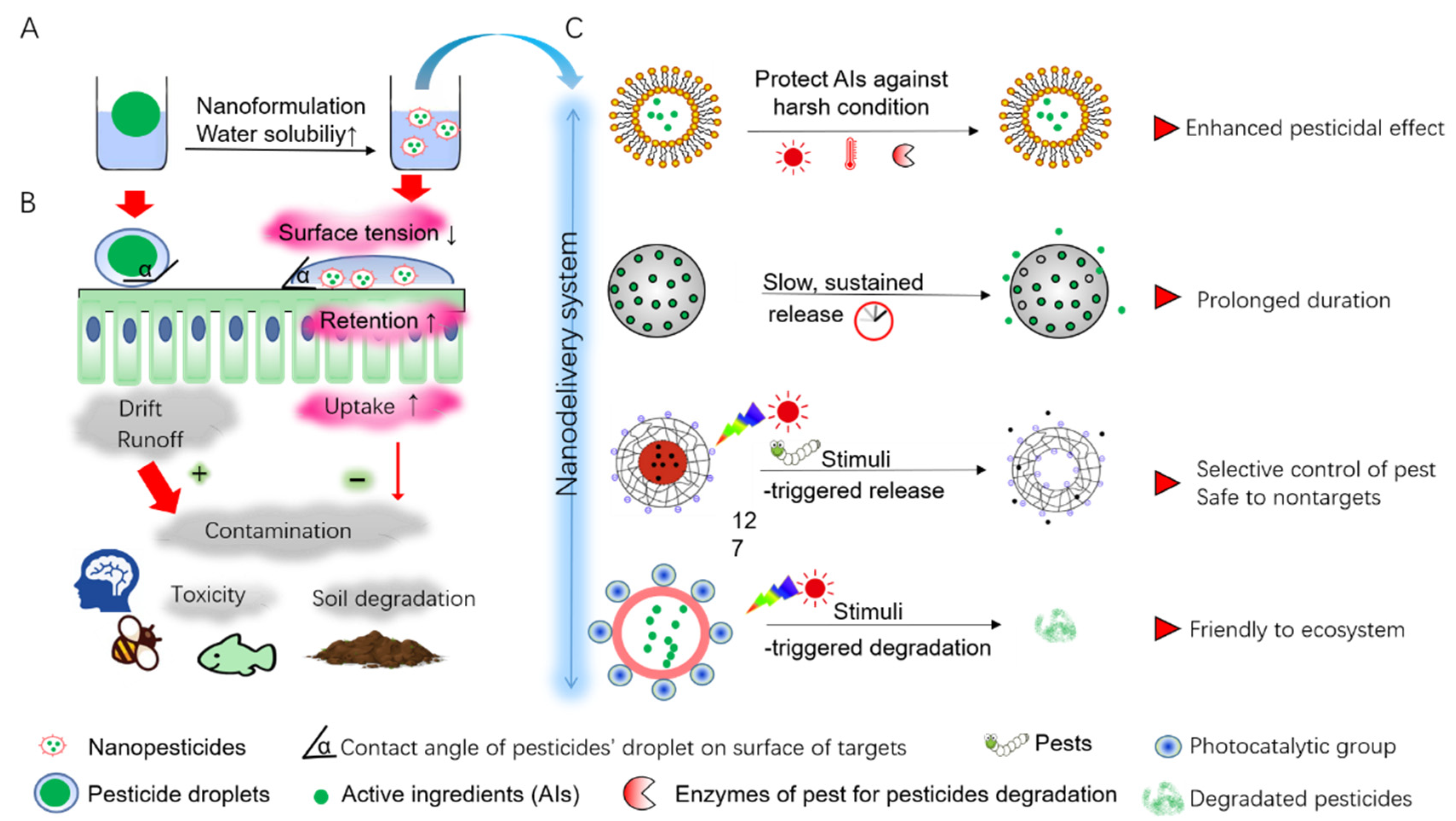
2.1. Nanoformulation for Increased Water-Solubility
2.2. NC-Based Delivery System to Protect Specific AIs against Harsh Condition
2.3. Sustained-Release Delivery Systems for Prolonged Duration of Pesticides
2.4. Stimuli-Responsive Systems for Controlled Release and Adjustable Degradation
3. Challenges and Perspective
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Conflicts of Interest
References
- Kah, M.; Johnston, L.J.; Kookana, R.S.; Bruce, W.; Haase, A.; Ritz, V.; Dinglasan, J.; Doak, S.; Garelick, H.; Gubala, V. Com-prehensive Framework for Human Health Risk Assessment of Nanopesticides. Nat. Nanotechnol. 2021, 16, 955–964. [Google Scholar] [CrossRef] [PubMed]
- Ecobichon, D.J. Pesticide use in developing countries. Toxicology 2001, 160, 27–33. [Google Scholar] [CrossRef]
- Gould, F.; Brown, Z.S.; Kuzma, J. Wicked evolution: Can we address the sociobiological dilemma of pesticide resistance? Science 2018, 360, 728–732. [Google Scholar] [CrossRef] [Green Version]
- Song, M.R.; Ju, J.; Luo, S.Q.; Han, Y.C.; Dong, Z.C.; Wang, Y.L.; Gu, Z.; Zhang, L.; Hao, R.R.; Jiang, L. Controlling Liquid Splash on Superhydrophobic Surfaces by a Vesicle Surfactant. Sci. Adv. 2017, 3, e1602188. [Google Scholar] [CrossRef] [Green Version]
- Zhao, X.; Cui, H.; Wang, Y.; Sun, C.; Cui, B.; Zeng, Z. Development Strategies and Prospects of Nano-based Smart Pesticide Formulation. J. Agric. Food Chem. 2018, 66, 6504–6512. [Google Scholar] [CrossRef]
- He, Y.; Zhao, B.; Yu, Y. Effect, Comparison and Analysis of Pesticide Electrostatic Spraying and Traditional Spraying. Bulg. Chem. Commun. 2016, 48, 340–344. [Google Scholar]
- Long, E.Y.; Krupke, C.H. Non-cultivated plants present a season-long route of pesticide exposure for honey bees. Nat. Commun. 2016, 7, 11629. [Google Scholar] [CrossRef]
- Kah, M.; Brown, C. Changes in pesticide adsorption with time at high soil to solution ratios. Chemosphere 2007, 68, 1335–1343. [Google Scholar] [CrossRef]
- Verger, P.J.P.; Boobis, A.R. Reevaluate Pesticides for Food Security and Safety. Science 2013, 341, 717–718. [Google Scholar] [CrossRef] [PubMed]
- Yamamuro, M.; Komuro, T.; Kamiya, H.; Kato, T.; Hasegawa, H.; Kameda, Y. Neonicotinoids disrupt aquatic food webs and decrease fishery yields. Science 2019, 366, 620–623. [Google Scholar] [CrossRef] [PubMed]
- Singh, B.K.; Walker, A. Microbial degradation of organophosphorus compounds. FEMS Microbiol. Rev. 2006, 30, 428–471. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Giraldo, J.P.; Wu, H.; Newkirk, G.M.; Kruss, S. Nanobiotechnology approaches for engineering smart plant sensors. Nat. Nanotechnol. 2019, 14, 541–553. [Google Scholar] [CrossRef]
- Kumar, S.; Nehra, M.; Dilbaghi, N.; Marrazza, G.; Hassan, A.A.; Kim, K.-H. Nano-based smart pesticide formulations: Emerging opportunities for agriculture. J. Control. Release 2019, 294, 131–153. [Google Scholar] [CrossRef]
- Ghormade, V.; Deshpande, M.V.; Paknikar, K. Perspectives for nano-biotechnology enabled protection and nutrition of plants. Biotechnol. Adv. 2011, 29, 792–803. [Google Scholar] [CrossRef]
- Demir, E. An in vivo study of nanorod, nanosphere, and nanowire forms of titanium dioxide using Drosophila melanogaster: Toxicity, cellular uptake, oxidative stress, and DNA damage. J. Toxicol. Environ. Health Part A 2020, 83, 456–469. [Google Scholar] [CrossRef] [PubMed]
- Ahmadi, Z.; Saber, M.; Bagheri, M.; Mahdavinia, G.R. Achillea millefolium essential oil and chitosan nanocapsules with enhanced activity against Tetranychusurticae. J. Pest Sci. 2018, 91, 837–848. [Google Scholar] [CrossRef]
- Cui, B.; Feng, L.; Wang, C.; Yang, D.; Yu, M.; Zeng, Z.; Wang, Y.; Sun, C.; Zhao, X.; Cui, H. Stability and Biological Activity Evaluation of Chlorantraniliprole Solid Nanodispersions Prepared by High Pressure Homogenization. PLoS ONE 2016, 11, e0160877. [Google Scholar] [CrossRef]
- Cao, L.; Zhang, H.; Cao, C.; Zhang, J.; Li, F.; Huang, Q. Quaternized Chitosan-Capped Mesoporous Silica Nanoparticles as Nanocarriers for Controlled Pesticide Release. Nanomaterials 2016, 6, 126. [Google Scholar] [CrossRef] [Green Version]
- Pérez Quiñones, J.; Brüggemann, O.; Kjems, J.; Shahavi, M.H.; PenicheCovas, C. Novel Brassinosteroid-Modified Polyeth-ylene Glycol Micelles for Controlled Release of Agrochemicals. J. Agric. Food Chem. 2018, 66, 1612–1619. [Google Scholar] [CrossRef]
- Liang, J.; Yu, M.; Guo, L.; Cui, B.; Zhao, X.; Sun, C.; Wang, Y.; Liu, G.; Cui, H.; Zeng, Z. Bioinspired Development of P(St–MAA)–Avermectin Nanoparticles with High Affinity for Foliage To Enhance Folia Retention. J. Agric. Food Chem. 2018, 66, 6578–6584. [Google Scholar] [CrossRef]
- Kah, M.; Kookana, R.S.; Gogos, A.; Bucheli, T.D. A critical evaluation of nanopesticides and nanofertilizers against their conventional analogues. Nat. Nanotechnol. 2018, 13, 677–684. [Google Scholar] [CrossRef] [PubMed]
- Pereira, A.E.; Grillo, R.; Mello, N.F.; Rosa, A.H.; Fraceto, L.F. Application of Poly(Epsilon-Caprolactone) Nanoparticles Con-taining Atrazine Herbicide as An Alternative Technique to Control Weeds and Reduce Damage to the Environment. J. HazardMater. 2014, 268, 207–215. [Google Scholar] [CrossRef] [PubMed]
- Balaji, A.; Sastry, T.P.; Manigandan, S.; Mukherjee, A.; Chandrasekaran, N. Environmental benignity of a pesticide in soft colloidal hydrodispersive nanometric form with improved toxic precision towards the target organisms than non-target organisms. Sci. Total. Environ. 2016, 579, 190–201. [Google Scholar] [CrossRef] [PubMed]
- Joga, M.R.; Zotti, M.J.; Smagghe, G.; Christiaens, O. RNAi Efficiency, Systemic Properties, and Novel Delivery Methods for Pest Insect Control: What We Know So Far. Front. Physiol. 2016, 7, 553. [Google Scholar] [CrossRef] [Green Version]
- Christiaens, O.; Whyard, S.; Vélez, A.M.; Smagghe, G. Double-Stranded RNA Technology to Control Insect Pests: Current Status and Challenges. Front. Plant Sci. 2020, 11, 451. [Google Scholar] [CrossRef]
- Baum, J.A.; Bogaert, T.; Clinton, W.; Heck, G.R.; Feldmann, P.; Ilagan, O.; Johnson, S.; Plaetinck, G.; Munyikwa, T.; Pleau, M.; et al. Control of coleopteran insect pests through RNA interference. Nat. Biotechnol. 2007, 25, 1322–1326. [Google Scholar] [CrossRef]
- Agrawal, N.; Dasaradhi, P.V.N.; Mohmmed, A.; Malhotra, P.; Bhatnagar, R.K.; Mukherjee, S.K. RNA Interference: Biology, Mechanism, and Applications. Microbiol. Mol. Biol. Rev. 2003, 67, 657–685. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhu, K.Y.; Palli, S.R. Mechanisms, Applications, and Challenges of Insect RNA Interference. Annu. Rev. Entomol. 2020, 65, 293–311. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yu, N.; Christiaens, O.; Liu, J.; Niu, J.; Cappelle, K.; Caccia, S.; Huvenne, H.; Smagghe, G. Delivery of dsRNA for RNAi in In-sects: An Overview and Future Directions. Insect Sci. 2013, 20, 4–14. [Google Scholar] [CrossRef]
- Yan, S.; Ren, B.Y.; Shen, J. Nanoparticle-Mediated Double-Stranded RNA Delivery System: A Promising Approach for Sustainable Pest Management. Insect Sci. 2021, 28, 21–34. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Zhang, J.; Zhu, K.Y. Chitosan/double-stranded RNA nanoparticle-mediated RNA interference to silence chitin synthase genes through larval feeding in the African malaria mosquito (Anopheles gambiae). Insect Mol. Biol. 2010, 19, 683–693. [Google Scholar] [CrossRef]
- Das, S.; Debnath, N.C.; Cui, Y.; Unrine, J.; Palli, S.R. Chitosan, Carbon Quantum Dot, and Silica Nanoparticle Mediated dsRNA Delivery for Gene Silencing in Aedes aegypti: A Comparative Analysis. ACS Appl. Mater. Interfaces 2015, 7, 19530–19535. [Google Scholar] [CrossRef]
- Zheng, Y.; Hu, Y.; Yan, S.; Zhou, H.; Songm, D.; Yin, M.; Shen, J. A polymer/detergent formulation improves dsRNA penetration through the body wall and RNAi-induced mortality in the soybean aphid Aphis glycines. Pest Manag. Sci. 2019, 75, 1993–1999. [Google Scholar] [CrossRef]
- Ali, A.; Ahmed, S. A review on chitosan and its nanocomposites in drug delivery. Int. J. Biol. Macromol. 2018, 109, 273–286. [Google Scholar] [CrossRef]
- Maluin, F.N.; Hussein, M.Z. Chitosan-Based Agronanochemicals as a Sustainable Alternative in Crop Protection. Molecules 2020, 25, 1611. [Google Scholar] [CrossRef] [Green Version]
- Safinya, C.R.; Ewert, K.K. Materials Chemistry: Liposomes Derived from Molecular Vases. Nature 2012, 489, 372–374. [Google Scholar] [CrossRef]
- Lin, Y.; Huang, J.-H.; Liu, Y.; Belles, X.; Lee, H.-J. Oral delivery of dsRNA lipoplexes to German cockroach protects dsRNA from degradation and induces RNAi response. Pest Manag. Sci. 2016, 73, 960–966. [Google Scholar] [CrossRef]
- Svenson, S.; Tomalia, D.A. Dendrimers in biomedical applications—reflections on the field. Adv. Drug Deliv. Rev. 2005, 57, 2106–2129. [Google Scholar] [CrossRef] [PubMed]
- Kesharwani, P.; Jain, K.; Jain, N.K. Dendrimer as nanocarrier for drug delivery. Prog. Polym. Sci. 2014, 39, 268–307. [Google Scholar] [CrossRef]
- Yu, G.S.; Bae, Y.M.; Choi, H.; Kong, B.; Choi, I.S.; Choi, J.S. Synthesis of PAMAM Dendrimer Derivatives with Enhanced Buffering Capacity and Remarkable Gene Transfection Efficiency. Bioconjugate Chem. 2011, 22, 1046–1055. [Google Scholar] [CrossRef]
- Lo, S.-T.; Kumar, A.; Hsieh, J.-T.; Sun, X. Dendrimer Nanoscaffolds for Potential Theranostics of Prostate Cancer with a Focus on Radiochemistry. Mol. Pharm. 2013, 10, 793–812. [Google Scholar] [CrossRef]
- Cheng, Y.; Zhao, L.; Li, Y.; Xu, T. Design of biocompatible dendrimers for cancer diagnosis and therapy: Current status and future perspectives. Chem. Soc. Rev. 2011, 40, 2673–2703. [Google Scholar] [CrossRef]
- He, B.; Chu, Y.; Yin, M.; Müllen, K.; An, C.; Shen, J. Fluorescent Nanoparticle Delivered dsRNA Toward Genetic Control of Insect Pests. Adv. Mater. 2013, 25, 4580–4584. [Google Scholar] [CrossRef] [PubMed]
- Shen, D.; Zhou, F.; Xu, Z.; He, B.; Li, M.; Shen, J.; Yin, M.; An, C. Systemically interfering with immune response by a fluorescent cationic dendrimer delivered gene suppression. J. Mater. Chem. B 2014, 2, 4653–4659. [Google Scholar] [CrossRef]
- Li, J.; Qian, J.; Xu, Y.; Yan, S.; Shen, J.; Yin, M. A Facile-Synthesized Star Polycation Constructed as a Highly Efficient Gene Vector in Pest Management. ACS Sustain. Chem. Eng. 2019, 7, 6316–6322. [Google Scholar] [CrossRef]
- Wang, K.; Peng, Y.; Chen, J.; Peng, Y.; Wang, X.; Shen, Z.; Han, Z. Comparison of Efficacy of RNAi Mediated by Various Na-noparticles in the Rice Striped Stem Borer (Chilosuppressalis). Pestic. Biochem. Physiol. 2020, 165, 104467. [Google Scholar] [CrossRef]
- Dhandapani, R.K.; Gurusamy, D.; Howell, J.L.; Palli, S.R. Development of CS-TPP-dsRNA Nanoparticles to Enhance RNAi Efficiency in the Yellow Fever Mosquito, Aedes aegypti. Sci. Rep. 2019, 9, 8775. [Google Scholar] [CrossRef]
- Thairu, M.W.; Skidmore, I.H.; Bansal, R.; Novakova, E.; Hansen, T.E.; Li-Byarlay, H.; Wickline, S.A.; Hansen, A. Efficacy of RNA interference knockdown using aerosolized short interfering RNAs bound to nanoparticles in three diverse aphid species. Insect Mol. Biol. 2017, 26, 356–368. [Google Scholar] [CrossRef]
- Liu, X.; He, B.; Xu, Z.; Yin, M.; Yang, W.; Zhang, H.; Cao, J.; Shen, J. A functionalized fluorescent dendrimer as a pesticide nanocarrier: Application in pest control. Nanoscale 2015, 7, 445–449. [Google Scholar] [CrossRef]
- Yan, S.; Qian, J.; Cai, C.; Ma, Z.Z.; Li, J.H.; Yin, M.Z.; Ren, B.Y.; Shen, J. Spray Method Application of Transdermal dsRNA De-livery System for Efficient Gene Silencing and Pest Control on Soybean Aphid Aphis glycines. J. Pest Sci. 2020, 93, 449–459. [Google Scholar] [CrossRef]
- Kim, J.H.; Moreau, J.A.; Zina, J.M.; Mazgaeen, L.; Yoon, K.S.; Pittendrigh, B.R.; Clark, J.M. Identification and interaction of multiple genes resulting in DDT resistance in the 91-R strain of Drosophila melanogaster by RNAi approaches. Pestic. Biochem. Physiol. 2018, 151, 90–99. [Google Scholar] [CrossRef]
- Bravo, A.; Gill, S.S.; Soberón, M. Mode of action of Bacillus thuringiensis Cry and Cyt toxins and their potential for insect control. Toxicon 2007, 49, 423–435. [Google Scholar] [CrossRef] [Green Version]
- Maghsoudi, S.; Jalali, E. Noble UV protective agent for Bacillus thuringiensis based on a combination of graphene oxide and olive oil. Sci. Rep. 2017, 7, 1–6. [Google Scholar] [CrossRef] [Green Version]
- Zheng, Y.; You, S.; Ji, C.; Yin, M.; Yang, W.; Shen, J. Development of an Amino Acid-Functionalized Fluorescent Nanocarrier to Deliver a Toxin to Kill Insect Pests. Adv. Mater. 2016, 28, 1375–1380. [Google Scholar] [CrossRef]
- Lin, L.; Cui, H.; Zhou, H.; Zhang, X.; Bortolini, C.; Chen, M.; Liu, L.; Dong, M. Nanoliposomes Containing Eucalyptus Citriodora as Antibiotic with Specific Antimicrobial Activity. Chem. Commun. 2015, 51, 2653–2655. [Google Scholar] [CrossRef]
- Nguyen, H.M.; Hwang, I.C.; Park, J.W.; Park, H.J. Enhanced Payload and Photo-Protection for Pesticides Using Nanostructured Lipid Carriers with Corn Oil as Liquid Lipid. J. Microencapsul. 2012, 29, 596–604. [Google Scholar] [CrossRef]
- Cantor, S.; Vargas, L.; Rojas, A.O.E.; Yarce, C.J.; Salamanca, C.H.; Oñate-Garzón, J. Evaluation of the Antimicrobial Activity of Cationic Peptides Loaded in Surface-Modified Nanoliposomes against Foodborne Bacteria. Int. J. Mol. Sci. 2019, 20, 680. [Google Scholar] [CrossRef] [Green Version]
- Yang, F.-L.; Li, X.-G.; Zhu, F.; Lei, C.-L. Structural Characterization of Nanoparticles Loaded with Garlic Essential Oil and Their Insecticidal Activity againstTriboliumcastaneum(Herbst) (Coleoptera: Tenebrionidae). J. Agric. Food Chem. 2009, 57, 10156–10162. [Google Scholar] [CrossRef]
- Cabrera, A.; Celis, R.; Hermosín, M.D.C. Imazamox-clay complexes with chitosan- and iron(III)-modified smectites and their use in nanoformulations. Pest Manag. Sci. 2015, 72, 1285–1294. [Google Scholar] [CrossRef] [Green Version]
- Li, Z.Z.; Chen, J.F.; Liu, F.; Liu, A.Q.; Wang, Q.; Sun, H.Y.; Wen, L.X. Study of UV-shielding Properties of Novel Porous Hollow Silica Nanoparticle Carriers for Avermectin. Pest Manag. Sci. 2007, 63, 241–246. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, A.; Wang, C.; Cui, B.; Sun, C.; Zhao, X.; Zeng, Z.; Shen, Y.; Gao, F.; Liu, G.; et al. Synthesis and Characterization of Emamectin-Benzoate Slow-Release Microspheres with Different Surfactants. Sci. Rep. 2017, 7, 12761. [Google Scholar] [CrossRef] [Green Version]
- Gabriel Paulraj, M.; Ignacimuthu, S.; Gandhi, M.R.; Shajahan, A.; Ganesan, P.; Packiam, S.M.; Al-Dhabi, N.A. Comparative Studies of Tripolyphosphate and Glutaraldehyde Cross-Linked Chitosan-Botanical Pesticide Nanoparticles and Their Agricultural Applications. Int. J. Biol. Macromol. 2017, 104, 1813–1819. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, H.M.; Hwang, I.-C.; Park, J.-W.; Park, H.-J. Photoprotection for deltamethrin using chitosan-coated beeswax solid lipid nanoparticles. Pest Manag. Sci. 2012, 68, 1062–1068. [Google Scholar] [CrossRef]
- Rani, P.U.; Madhusudhanamurthy, J.; Sreedhar, B. Dynamic adsorption of α-pinene and linalool on silica nanoparticles for enhanced antifeedant activity against agricultural pests. J. Pest Sci. 2014, 87, 191–200. [Google Scholar] [CrossRef]
- Mattos, B.; Rojas, O.; Magalhaes, W. Biogenic silica nanoparticles loaded with neem bark extract as green, slow-release biocide. J. Clean. Prod. 2017, 142, 4206–4213. [Google Scholar] [CrossRef]
- Flores-Céspedes, F.; Martínez-Domínguez, G.P.; Villafranca-Sánchez, M.; Fernández-Pérez, M. Preparation and Characteriza-tion of Azadirachtin Alginate-Biosorbent Based Formulations: Water Release Kinetics and Photodegradation Study. J. Agric. Food Chem. 2015, 63, 8391–8398. [Google Scholar] [CrossRef] [PubMed]
- Bakkali, F.; Averbeck, S.; Averbeck, D.; Idaomar, M. Biological Effects of Essential Oils—A Review. Food Chem. Toxicol. 2008, 46, 446–475. [Google Scholar] [CrossRef]
- Bai, X.; Lin, C.; Wang, Y.; Ma, J.; Wang, X.; Yao, X.; Tang, B. Preparation of Zn doped mesoporous silica nanoparticles (Zn-MSNs) for the improvement of mechanical and antibacterial properties of dental resin composites. Dent. Mater. 2020, 36, 794–807. [Google Scholar] [CrossRef] [PubMed]
- Lowry, G.V.; Avellan, A.; Gilbertson, L.M. Opportunities and challenges for nanotechnology in the Agri-Tech revolution. Nat. Nanotechnol. 2019, 14, 517–522. [Google Scholar] [CrossRef]
- Athanassiou, C.G.; Kavallieratos, N.G.; Benelli, G.; Losic, D.; Usha Rani, P.; Desneux, N. Nanoparticles for Pest Control: Cur-rent Status and Future Perspectives. J. Pest Sci. 2017, 91, 1–15. [Google Scholar] [CrossRef]
- Kaziem, A.E.; Gao, Y.; He, S.; Li, J. Synthesis and Insecticidal Activity of Enzyme-Triggered Functionalized Hollow Mesopo-rous Silica for Controlled Release. J. Agric. Food Chem. 2017, 65, 7854–7864. [Google Scholar] [CrossRef]
- Guo, M.; Zhang, W.; Ding, G.; Guo, D.; Zhu, J.; Wang, B.; Punyapitak, D.; Cao, Y. Preparation and Characterization of En-zyme-Responsive Emamectin Benzoate Microcapsules Based on a Copolymer Matrix of Silica–Epichlorohydrin–Carboxymethylcellulose. RSC Adv. 2015, 5, 93170–93179. [Google Scholar] [CrossRef]
- Peppas, N.A.; Sahlin, J.J. A simple equation for the description of solute release. III. Coupling of diffusion and relaxation. Int. J. Pharm. 1989, 57, 169–172. [Google Scholar] [CrossRef]
- Bystrzejewska-Piotrowska, G.; Golimowski, J.; Urban, P.L. Nanoparticles: Their potential toxicity, waste and environmental management. WasteManag. 2009, 29, 2587–2595. [Google Scholar] [CrossRef]
- Li, J.; Fan, T.; Xu, Y.; Wu, X. Ionic Liquids as Modulators of Physicochemical Properties and Nanostructures of Sodium Do-decyl Sulfate in Aqueous Solutions and Potential Application in Pesticide Microemulsions. Phys. Chem. Chem. Phys. 2016, 18, 29797–29807. [Google Scholar] [CrossRef]
- Zhao, Y. Rational design of light-controllable polymer micelles. Chem. Rec. 2007, 7, 286–294. [Google Scholar] [CrossRef]
- Edueng, K.; Mahlin, D.; Larsson, P.; Bergström, C.A.S. Mechanism-based Selection of Stabilization Strategy for Amorphous Formulations: Insights into Crystallization Pathways. J. Control. Release 2017, 256, 193–202. [Google Scholar] [CrossRef]
- Sharma, S.; Singh, S.; Ganguli, A.K.; Shanmugam, V. Anti-drift nano-stickers made of graphene oxide for targeted pesticide delivery and crop pest control. Carbon 2017, 115, 781–790. [Google Scholar] [CrossRef]
- Guan, H.N.; Chi, D.F.; Yu, J.; Zhang, S.Y. Novel Photodegradable Insecticide W/TiO2/Avermectin Nanocomposites Obtained by Polyelectrolytes Assembly. Colloids Surf. B 2011, 83, 148–154. [Google Scholar] [CrossRef]
- Atta, S.; Bera, M.; Chattopadhyay, T.; Paul, A.; Ikbal, M.; Maiti, M.K.; Singh, N. Nano-pesticide Formulation Based on Fluorescent Organic Photoresponsive Nanoparticles: For Controlled Release of 2,4-D and Real Time Monitoring of Morphological Changes Induced by 2,4-D in Plant Systems. RSC Adv. 2015, 5, 86990–86996. [Google Scholar] [CrossRef]
- Cao, L.; Zhou, Z.; Niu, S.; Cao, C.; Li, X.; Shan, Y.; Huang, Q. Positive-Charge Functionalized Mesoporous Silica Nanoparticles as Nanocarriers for Controlled 2,4-Dichlorophenoxy Acetic Acid Sodium Salt Release. J. Agric. Food Chem. 2018, 66, 6594–6603. [Google Scholar] [CrossRef]
- Liang, Y.; Guo, M.; Fan, C.; Dong, H.; Ding, G.; Zhang, W.; Tang, G.; Yang, J.; Kong, D.; Ca, O.Y. Development of Novel Urease-Responsive Pendimethalin Microcapsules Using Silica-Ipts-Pei as Controlled Release Carrier Materials. Acs. Sustain. Chem. Eng. 2017, 5, 4802–4810. [Google Scholar] [CrossRef]
- Chen, K.; Yu, G.; He, F.; Zhou, Q.; Xiao, D.; Li, J.; Feng, Y. A pH-Responsive Emulsion Stabilized by Alginate-Grafted Aniso-tropic Silica and Its Application in the Controlled Release of λ-Cyhalothrin. Carbohydr. Polym. 2017, 176, 203–213. [Google Scholar] [CrossRef]
- Kumar, S.; Chauhan, N.; Gopal, M.; Kumar, R.; Dilbaghi, N. Development and Evaluation of Alginate-Chitosan Nanocap-sules for Controlled Release of Acetamiprid. Int. J. Biol. Macromol. 2015, 81, 631–637. [Google Scholar] [CrossRef]
- Sarkar, D.J.; Singh, A. Base Triggered Release of Insecticide from Bentonite Reinforced Citric Acid Crosslinked Carboxyme-thyl Cellulose Hydrogel Composites. Carbohydr. Polym. 2017, 156, 303–311. [Google Scholar] [CrossRef]
- Ye, Z.; Guo, J.; Wu, D.; Tan, M.; Xiong, X.; Yin, Y.; He, G. Photo-responsive Shell Cross-Linked Micelles Based on Carboxymethyl Chitosan and Their Application in Controlled Release of Pesticide. Carbohydr. Polym. 2015, 132, 520–528. [Google Scholar] [CrossRef]
- Hardy, A.; Benford, D.; Halldorsson, T.; Jeger, M.J.; Knutsen, H.K.; More, S.; Naegeli, H.; Noteborn, H.; Ockleford, C.; Ricci, A.; et al. Guidance on risk assessment of the application of nanoscience and nanotechnologies in the food and feed chain: Part 1, human and animal health. EFSA J. 2011, 9, 2140. [Google Scholar] [CrossRef]
- Kookana, R.S.; Boxall, A.B.; Reeves, P.T.; Ashauer, R.; Beulke, S.; Chaudhry, Q.; Cornelis, G.; Fernandes, T.F.; Gan, J.; Kah, M.; et al. Nanopesticides: Guiding Principles for Regu-latory Evaluation of Environmental Risks. J. Agric. Food Chem. 2014, 62, 4227–4240. [Google Scholar] [CrossRef] [Green Version]
- Vladimir, G.; Johnston, L.J.; Liu, Z.W.; Harald, K.; Moore, C.J.; Ober, C.K.; Michael, S.; Michel, V. Engineered Nanomaterials and Human Health: Part 1. Preparation, Functionalization and Characterization (IUPAC Technical Report). Pure. Appl. Chem. 2018, 90, 1283–1324. [Google Scholar] [CrossRef] [Green Version]
- Mohd Firdaus, M.A.; Agatz, A.; Hodson, M.E.; Al-Khazrajy, O.S.A.; Boxall, A.B.A. Fate, Uptake, and Distribution of Nanoen-capsulated Pesticides in Soil-Earthworm Systems and Implications for Environmental Risk Assessment. Environ. Toxicol. Chem. 2018, 37, 1420–1429. [Google Scholar] [CrossRef] [Green Version]
- Glaser, J.A. Microplastics in the Environment. Clean Technol. Envir. 2015, 17, 1383–1391. [Google Scholar] [CrossRef]
- Wilpiszewska, K.; Spychaj, T.; Paździoch, W. Carboxymethyl starch/montmorillonite composite microparticles: Properties and controlled release of isoproturon. Carbohydr. Polym. 2016, 136, 101–106. [Google Scholar] [CrossRef] [PubMed]
- Oun, A.A.; Rhim, J.W. Isolation of Cellulose Nanocrystals from Grain Straws and Their Use for the Preparation of Carbox-ymethyl Cellulose-Based Nanocomposite Films. Carbohydr. Polym. 2016, 150, 187–200. [Google Scholar] [CrossRef] [PubMed]
- Giessen, T.W.; Silver, P.A. Encapsulation as a Strategy for the Design of Biological Compartmentalization. J. Mol. Biol. 2016, 428, 916–927. [Google Scholar] [CrossRef] [Green Version]
- Krishnaraj, C.; Jagan, E.G.; Rajasekar, S.; Selvakumar, P.; Kalaichelvan, P.T.; Mohan, N. Synthesis of Silver Nanoparticles Us-ingAcalyphaindica Leaf Extracts and Its Antibacterial Activity Against Water Borne Pathogens. Colloid. Surface. B 2010, 76, 50–56. [Google Scholar] [CrossRef]
- Shivaji, S.; Madhu, S.; Singh, S. Extracellular synthesis of antibacterial silver nanoparticles using psychrophilic bacteria. Process. Biochem. 2011, 46, 1800–1807. [Google Scholar] [CrossRef]
- Syed, A.; Saraswati, S.; Kundu, G.C.; Ahmad, A. Biological synthesis of silver nanoparticles using the fungus Humicola sp. and evaluation of their cytoxicity using normal and cancer cell lines. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2013, 114, 144–147. [Google Scholar] [CrossRef]
- Li, G.; He, D.; Qian, Y.; Guan, B.; Gao, S.; Cui, Y.; Yokoyama, K.; Wang, L. Fungus-Mediated Green Synthesis of Silver Nano-particles Using Aspergillus terreus. Int. J. Mol. Sci. 2012, 13, 466–476. [Google Scholar] [CrossRef] [Green Version]
- Mauricio, M.R.; Da Costa, P.G.; Haraguchi, S.K.; Guilherme, M.R.; Muniz, E.; Rubira, A. Synthesis of a microhydrogel composite from cellulose nanowhiskers and starch for drug delivery. Carbohydr. Polym. 2015, 115, 715–722. [Google Scholar] [CrossRef]
- Ribeiro, R.C.; Zanuncio, T.V.; Ramalho, F.D.S.; da Silva, C.A.D.; Serrão, J.E.; Zanuncio, J.C. Feeding and Oviposition of An-ticarsiagemmatalis (Lepidoptera: Noctuidae) with Sublethal Concentrations Of Ten Condiments Essential Oils. Ind. Crop Prod. 2015, 74, 139–143. [Google Scholar] [CrossRef]

| NCs | Pest | Improved Properties and Pest Control Effects | Reference |
|---|---|---|---|
| Chitosan | A. gambiae | Two key genes, AgCHS1 and AgCHS2, can be repressed by chitosan/AgCHS dsRNA-based nanoparticles through larval feeding | [31] |
| Chitosan | A. aegypti | Reduced expression level of targeted AaSNF7 (38%) and AaSRC (90%) through oral delivery of dsRNA | [32] |
| Chitosan | C. suppressalis | 57% reduction of glyceraldehyde 3 phosphate dehydrogenase gene in whole body and 92% mortality of larvae. | [46] |
| Chitosan | A. aegypti | Enhanced gene silencing effect (20–65%) depending on the targeted genes (IAP, SNF7, SSK, and OTK) and increased larvae mortality | [47] |
| liposomes | B. germanica | Slowing down the degradation of dsRNA in midgut, significantly inhibiting of α-tubulin expression and increased mortality of pests | [37] |
| Carbon quantum dot | A. glycines | Enhancing gene silencing effect by the delivery of aerosolized siRNA-NCs, increased reduction of aphid body mass. | [48] |
| Carbon quantum dot | A. aegypti | Reduced expression level of targeted AaSNF7 (60%) and AaSRC (29%) through feeding CQD/dsRNA; mortality of pests was 38% and 32% for dsAaSRC and dsAaSNF7 | [32] |
| Dendrimer | Heliothis armigera | Efficiently enter into various larval tissues and enhanced larvae mortality (83.3%) | [49] |
| Dendrimer | A. glycines | Efficiently knockdown of targeted genes (effect ranging from 86.86 to 58.87%) and high mortality up to 81.67% | [50] |
| Dendrimer | O. furnacalis | Efficient inhibition of targeted gene by oral delivery and severe defects in larval growth and molt leading to death | [43] |
| Dendrimer | D. melanogaster | Efficiently knock down of multiple genes involved in DDT resistance and enhanced sensitivity to DDT | [51] |
| NCs | AIs | Pest | Improved Capability and Pest Control Effects | Reference |
|---|---|---|---|---|
| Nanoliposomes | EOs from Eucalyptus citriodora | S. aureus | High efficiency and long-term availability of antibiotics | [55] |
| Nanolipid (corn oil, beeswax) | Insecticidal deltamethrin | - | Controlled release and increased photo-protection | [56] |
| Liposomes | Antimicrobial peptides | Foodborne pathogens | Enhanced antibacterial activity against Listeria monocytogenes and Escherichia coli | [57] |
| Polyethylene glycol | EOs from garlic | T. castaneum | Decreased volatilization of essential oils, retained 80% pest control efficacy over 5 months | [58] |
| Chitosan | EOs from Achillea millefolium L. | T. urticae Koch | A persistent release of AIs and prolonged acaricidal effect | [16] |
| Montmorillonite and chitosan | Herbicidal imazamox | Weeds | Only 27% release of herbicidal imazamox in the first 10 min, much lower than that of commercial formulations (86%) | [59] |
| Hollow silica NPs | Avermectin | - | remarkable UV-shielding for pesticides, a sustained release of pesticides over 30 days | [60] |
| Composite microspheres | Emamectin -benzoate | - | Excellent anti-photolysis and good leaf distribution, controlled release properties, | [61] |
| CSNs-TPP- PONNEEM 1 | Botanical PONNEEM (R) | H. armigera | 88.5% antifeedant activity and 90.2% larvicidal activity against pests | [62] |
| CH-BSLNs 2 | Insecticidal deltamethrin | - | 74.5% of deltamethrin remained after exposure to UV irradiation for 2 h | [63] |
| Alpha-pinene, linalool and silica | Insecticidal terpenes | S. litura. and A. janata L. | Enhanced the antifeedant against insects and longer shelf-life of pesticides | [64] |
| Biogenic silica | Theneem extract | A. crassispinus ant specie | Improving stability of pesticides to four-fold and sequential release profiles of pesticides | [65] |
| Copolymer (styrene and methacrylic acid) | Insecticidal avermectin | - | Excellent storage stability, improved resistance to ultraviolet light, sustained release and increased retention ratio on foliage | [20] |
| GO/olive oil | B. thuringiensis | E. kuehniella | Highest viability (50.62%) after exposure to for 96 h | [53] |
| Hybrid hydrogels (biosorbents and sodium alginate) | Botanical azadirachtin | - | Enhanced resistance against simulated sunlight | [66] |
| Nanocarriers | Pesticide | Stimuli | Improved Capability and Pest Control Effects | Reference |
|---|---|---|---|---|
| Hollow mesoporous silica | Chlorantraniliprole | Pest enzyme (α-amylase) | High larval mortality of P. xylostella | [74] |
| Copolymer matrix | Emamectin benzoate | Pest enzyme (cellulase) | Cellulase stimuli-responsive properties and sustained control of M. persicae | [75] |
| Silica-IPTS-PE 1 | Pendimethalin | Urease | Enhanced thermal and light stability of pendimethalin, increased duration and strengthened herbicidal activity | [65] |
| Natural alginate and chitosan | Insecticidal acetamiprid | Alkaline (pH of pest intestine) | Higher release of insecticides at alkaline pH condition than neutral pH | [76] |
| Clay hydrogels | Insecticidal thiamethoxam | Alkaline (pH of pest intestine) | Enhanced release of pesticides in alkaline pH condition | [19] |
| 2-nitrobenzyl carboxymethyl chitosan | Herbicidal diuron | Photo | High photo-controlled release rate of 96.8% while non-release without light exposure | [77] |
| PDA-PNIPAm 2 | Insecticidal imidacloprid | Photo | Good photothermal response capability, potential in selectively control of diurnal pests | [78] |
| GO NCs | Chlorpyrifos | Photo | Photothermal and photocatalytic performance, programmed pesticide residue degradation, resistance to drift, enhanced larval mortality (>35%) | [79] |
| TiO2 NCs | Abamectin | Photo | photocatalytic oxidation degradation of pesticides | [80] |
| Perylene-3-ylmethanol | Herbicidal 2,4-D 3 | Photo | Increased cell uptake of pesticides in plant, improved herbicidal activity, efficient photo-regulated release | [81] |
| Mesoporous silica | Herbicidal 2,4-D | temperature, pH and ionic strength | A controlled release pattern of pesticides, decreased soil leaching of 2, 4-D sodium salt | [82] |
| Salicylaldehyde modified mesoporous silica | Chlorpyrifos | pH | Decreased release of pesticides with the pH increasing | [83] |
| Alginate-grafted anisotropic silica | λ-cyhalothrin | pH | Decreased release of pesticides in the emulsion from 99.7% to 13.5% | [84] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hou, Q.; Zhang, H.; Bao, L.; Song, Z.; Liu, C.; Jiang, Z.; Zheng, Y. NCs-Delivered Pesticides: A Promising Candidate in Smart Agriculture. Int. J. Mol. Sci. 2021, 22, 13043. https://doi.org/10.3390/ijms222313043
Hou Q, Zhang H, Bao L, Song Z, Liu C, Jiang Z, Zheng Y. NCs-Delivered Pesticides: A Promising Candidate in Smart Agriculture. International Journal of Molecular Sciences. 2021; 22(23):13043. https://doi.org/10.3390/ijms222313043
Chicago/Turabian StyleHou, Qiuli, Hanqiao Zhang, Lixia Bao, Zeyu Song, Changpeng Liu, Zhenqi Jiang, and Yang Zheng. 2021. "NCs-Delivered Pesticides: A Promising Candidate in Smart Agriculture" International Journal of Molecular Sciences 22, no. 23: 13043. https://doi.org/10.3390/ijms222313043
APA StyleHou, Q., Zhang, H., Bao, L., Song, Z., Liu, C., Jiang, Z., & Zheng, Y. (2021). NCs-Delivered Pesticides: A Promising Candidate in Smart Agriculture. International Journal of Molecular Sciences, 22(23), 13043. https://doi.org/10.3390/ijms222313043






