Bacterial Cellulose: Production, Characterization, and Application as Antimicrobial Agent
Abstract
:1. Introduction
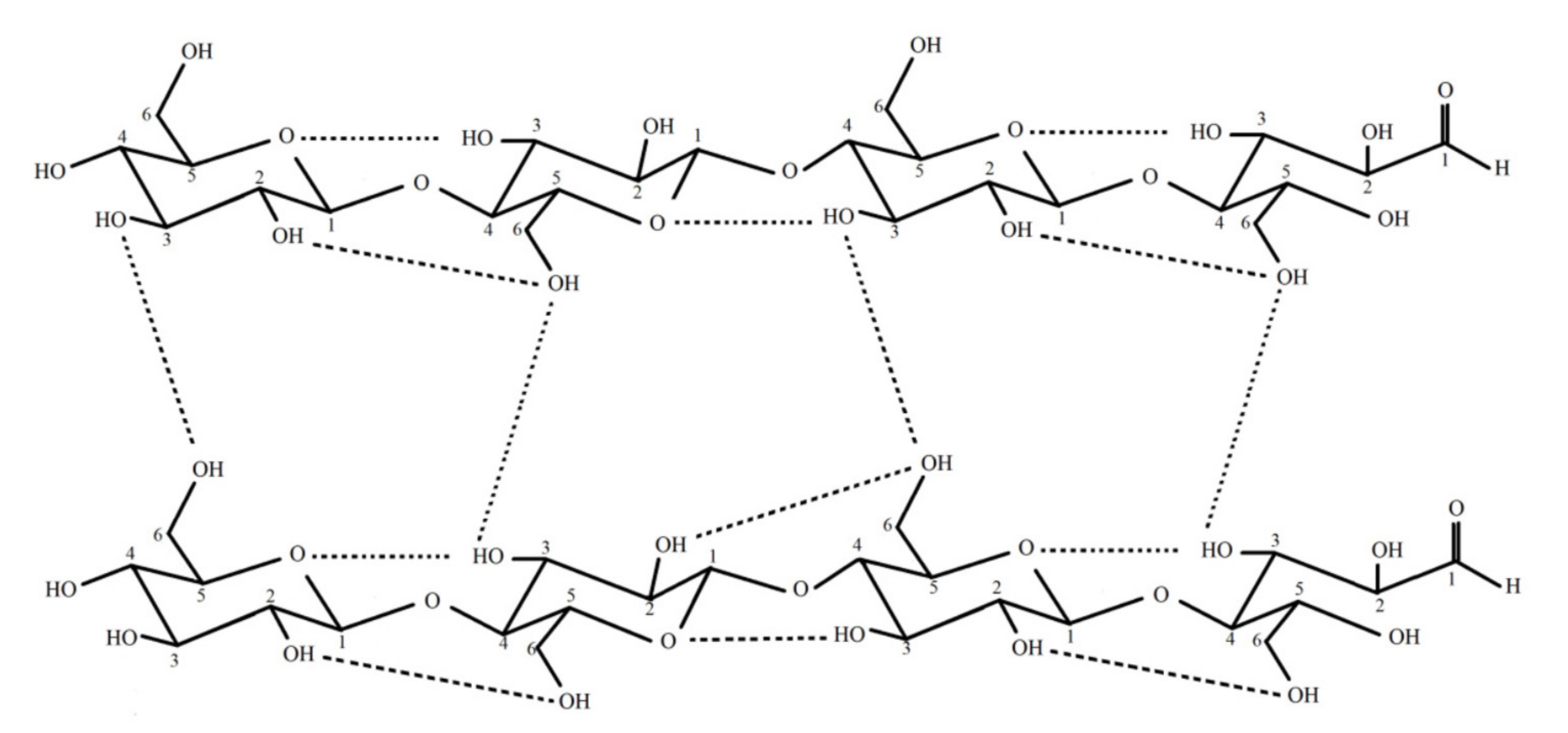
2. Properties of Bacterial Cellulose
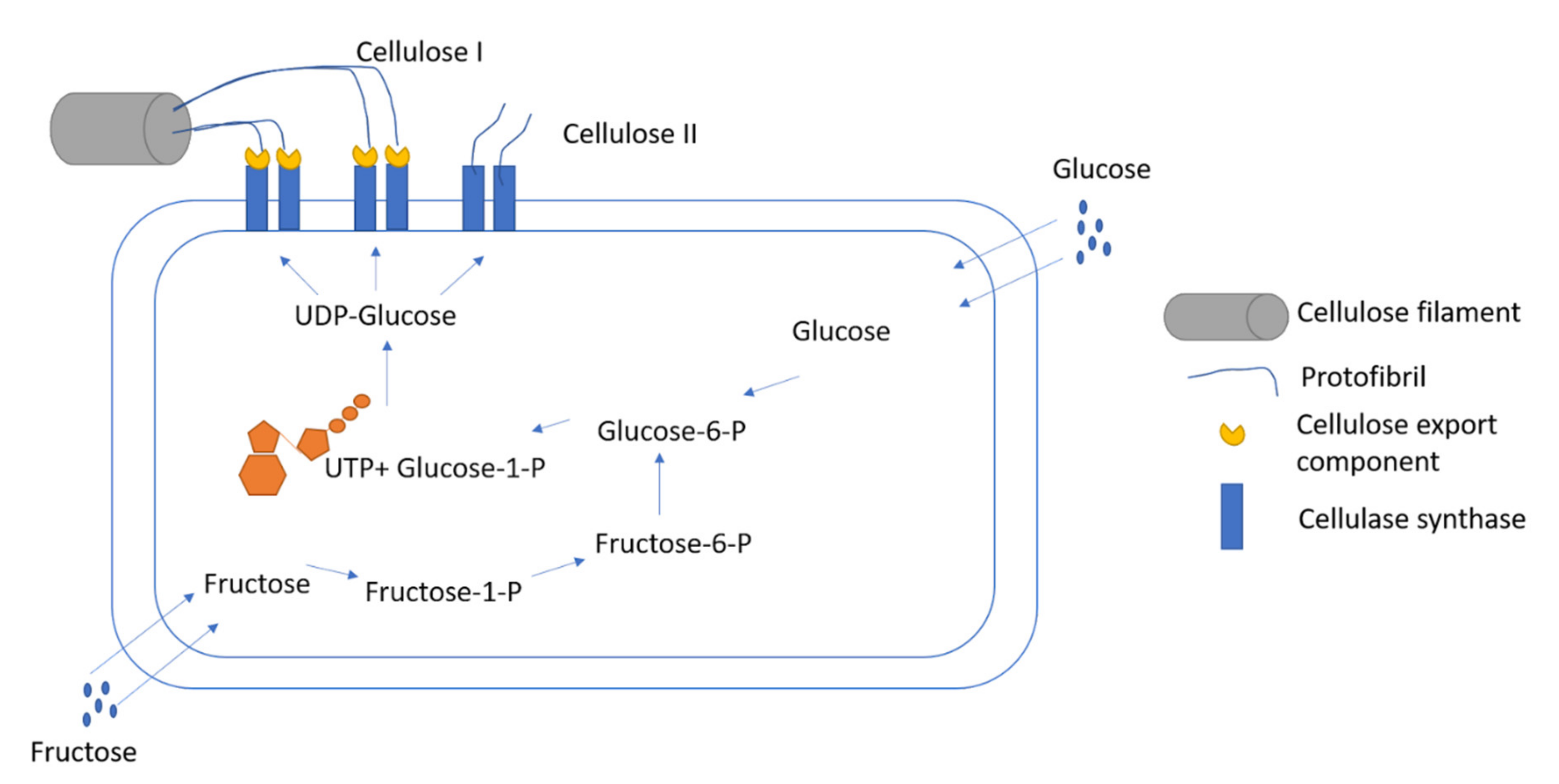
3. Bacterial Cellulose Producing Bacteria
4. Cultivation of Bacteria for the Production of Bacterial Cellulose
5. Parameters Controlling BC Production
5.1. Temperature
5.2. pH
5.3. Culture Media
5.4. Agitation Rate
5.5. Oxygen Level
5.6. Growth Curve
5.7. Yield of BC
6. Bacterial Cellulose Biocomposites and Their Characterization
7. Antimicrobial Activities of BC Composites
7.1. Deposition of BC on Electrospun PLA
7.2. BC-Reinforced Fabrics with Natural Fibers
7.3. BC-Reinforced Chitosan (BC-Ch) Composite
7.4. Montmorillonite (MMT)-Reinforced BC Composite
8. BC-CU NPs Nanocomposites
9. BC–Graphene-CuO Nanocomposites
10. BC–ZnO Nanocomposites
11. BC–Methylglyoxal Nanocomposites
12. Antimicrobial Activities of Bacterial Cellulose Augmented with Other Compounds
13. Antibiofilm Activities of BC Composites
13.1. Antibiofilm Effect of Bacterial Cellulose-Tannic Acid Composite
13.2. Antibiofilm Effect of Silver–BC Composite
13.3. Antibiofilm Activity of Chitosan–BC Composite
14. Dye- and Heavy-Metal-Removal Activities of BC Composites
15. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Rukmanikrishnan, B.; Ramalingam, S.; Rajasekharan, S.K.; Lee, J.; Lee, J. Binary and ternary sustainable composites of gellan gum, hydroxyethyl cellulose and lignin for food packaging applications: Biocompatibility, antioxidant activity, UV and water barrier properties. Int. J. Biol. Macromol. 2020, 153, 55–62. [Google Scholar] [CrossRef]
- Raj, V.; Raorane, C.J.; Lee, J.-H.; Lee, J. Appraisal of Chitosan-Gum Arabic-Coated Bipolymeric Nanocarriers for Efficient Dye Removal and Eradication of the Plant Pathogen Botrytis cinerea. ACS Appl. Mater. Interfaces 2021, 13, 47354–47370. [Google Scholar] [CrossRef] [PubMed]
- Abeer, M.M.; Mohd Amin, M.C.I.; Martin, C. A review of bacterial cellulose-based drug delivery systems: Their biochemistry, current approaches and future prospects. J. Pharm. Pharmacol. 2014, 66, 1047–1061. [Google Scholar] [CrossRef] [PubMed]
- Ross, P.; Mayer, R.; Benziman, M. Cellulose biosynthesis and function in bacteria. Microbiol. Rev. 1991, 55, 35–58. [Google Scholar] [CrossRef]
- Gallegos, A.M.A.; Carrera, S.H.; Parra, R.; Keshavarz, T.; Iqbal, H.M.N. Bacterial Cellulose: A Sustainable Source to Develop Value-Added Products—A Review. BioResources 2016, 11, 5641–5655. [Google Scholar] [CrossRef]
- Sarkar, T.; Salauddin, M.; Chakraborty, R. In-depth pharmacological and nutritional properties of bael (Aegle marmelos): A critical review. J. Agric. Food Res. 2020, 2, 100081. [Google Scholar] [CrossRef]
- Iqbal, H.M.N.; Kyazze, G.; Tron, T.; Keshavarz, T. Laccase-assisted grafting of poly(3-hydroxybutyrate) onto the bacterial cellulose as backbone polymer: Development and characterisation. Carbohydr. Polym. 2014, 113, 131–137. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, H.M.N.; Kyazze, G.; Tron, T.; Keshavarz, T. Laccase-Assisted Approach to Graft Multifunctional Materials of Interest: Keratin-EC Based Novel Composites and their Characterisation. Macromol. Mater. Eng. 2015, 300, 712–720. [Google Scholar] [CrossRef] [Green Version]
- Iqbal, H.M.N.; Kyazze, G.; Locke, I.C.; Tron, T.; Keshavarz, T. In Situ development of self-defensive antibacterial biomaterials: Phenol-g-keratin-EC based bio-composites with characteristics for biomedical applications. Green Chem. 2015, 17, 3858–3869. [Google Scholar] [CrossRef] [Green Version]
- Pandit, A.; Kumar, R. A Review on Production, Characterization and Application of Bacterial Cellulose and Its Biocomposites. J. Polym. Environ. 2021, 29, 2738–2755. [Google Scholar] [CrossRef]
- Wang, J.; Tavakoli, J.; Tang, Y. Bacterial cellulose production, properties and applications with different culture methods—A review. Carbohydr. Polym. 2019, 219, 63–76. [Google Scholar] [CrossRef] [Green Version]
- Sani, A.; Dahman, Y. Improvements in the production of bacterial synthesized biocellulose nanofibres using different culture methods. J. Chem. Technol. Biotechnol. 2010, 85, 151–164. [Google Scholar] [CrossRef]
- Figueiredo, A.R.P.; Figueiredo, A.G.P.R.; Silva, N.H.C.S.; Barros-Timmons, A.; Almeida, A.; Silvestre, A.J.D.; Freire, C.S.R. Antimicrobial bacterial cellulose nanocomposites prepared by in situ polymerization of 2-aminoethyl methacrylate. Carbohydr. Polym. 2015, 123, 443–453. [Google Scholar] [CrossRef] [PubMed]
- Förch, R.; Duque, L.; Lotz, A. 4.18—Antimicrobial Bioactive Polymer Coatings. In Comprehensive Materials Processing; Hashmi, S., Batalha, G.F., Van Tyne, C.J., Yilbas, B.B.T.-C.M.P., Eds.; Elsevier: Oxford, UK, 2014; pp. 449–461. ISBN 978-0-08-096533-8. [Google Scholar]
- Chau, C.-F.; Yang, P.; Yu, C.-M.; Yen, G.-C. Investigation on the lipid- and cholesterol-lowering abilities of biocellulose. J. Agric. Food Chem. 2008, 56, 2291–2295. [Google Scholar] [CrossRef]
- Czaja, W.K.; Young, D.J.; Kawecki, M.; Brown, R.M. The Future Prospects of Microbial Cellulose in Biomedical Applications. Biomacromolecules 2007, 8, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Petersen, N.; Gatenholm, P. Bacterial cellulose-based materials and medical devices: Current state and perspectives. Appl. Microbiol. Biotechnol. 2011, 91, 1277–1286. [Google Scholar] [CrossRef]
- Pogorelova, N.; Rogachev, E.; Digel, I.; Chernigova, S.; Nardin, D. Bacterial Cellulose Nanocomposites: Morphology and Mechanical Properties. Materials 2020, 13, 2849. [Google Scholar] [CrossRef]
- Chen, G.; Wu, G.; Alriksson, B.; Chen, L.; Wang, W.; Jönsson, L.J.; Hong, F.F. Scale-up of production of bacterial nanocellulose using submerged cultivation. J. Chem. Technol. Biotechnol. 2018, 93, 3418–3427. [Google Scholar] [CrossRef]
- Klemm, D.; Cranston, E.D.; Fischer, D.; Gama, M.; Kedzior, S.A.; Kralisch, D.; Kramer, F.; Kondo, T.; Lindström, T.; Nietzsche, S.; et al. Nanocellulose as a natural source for groundbreaking applications in materials science: Today’s state. Mater. Today 2018, 21, 720–748. [Google Scholar] [CrossRef] [Green Version]
- Esa, F.; Tasirin, S.M.; Rahman, N.A. Overview of Bacterial Cellulose Production and Application. Agric. Agric. Sci. Procedia 2014, 2, 113–119. [Google Scholar] [CrossRef] [Green Version]
- Zhong, C. Industrial-Scale Production and Applications of Bacterial Cellulose. Front. Bioeng. Biotechnol. 2020, 8, 1425. [Google Scholar] [CrossRef]
- Tanaka, M.; Murakami, S.; Shinke, R.; Aoki, K. Genetic characteristics of cellulose-forming acetic acid bacteria identified phenotypically as Gluconacetobacter xylinus. Biosci. Biotechnol. Biochem. 2000, 64, 757–760. [Google Scholar] [CrossRef]
- Keshk, S.M.A.S. Bacterial Cellulose Production and its Industrial Applications. J. Bioprocess. Biotech. 2014, 4, 1–10. [Google Scholar] [CrossRef]
- Jonas, R.; Farah, L.F. Production and application of microbial cellulose. Polym. Degrad. Stab. 1998, 59, 101–106. [Google Scholar] [CrossRef]
- Brown, R.M.J.; Montezinos, D. Cellulose microfibrils: Visualization of biosynthetic and orienting complexes in association with the plasma membrane. Proc. Natl. Acad. Sci. USA 1976, 73, 143–147. [Google Scholar] [CrossRef] [Green Version]
- Son, H.J.; Heo, M.S.; Kim, Y.G.; Lee, S.J. Optimization of fermentation conditions for the production of bacterial cellulose by a newly isolated Acetobacter sp. A9 in shaking cultures. Biotechnol. Appl. Biochem. 2001, 33, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Matthysse, A.G.; Thomas, D.L.; White, A.R. Mechanism of cellulose synthesis in Agrobacterium tumefaciens. J. Bacteriol. 1995, 177, 1076–1081. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, S.-S.; Han, Y.-H.; Chen, J.-L.; Zhang, D.-C.; Shi, X.-X.; Ye, Y.-X.; Chen, D.-L.; Li, M. Insights into Bacterial Cellulose Biosynthesis from Different Carbon Sources and the Associated Biochemical Transformation Pathways in Komagataeibacter sp. W1. Polymers 2018, 10, 963. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Benziman, M.; Burger-Rachamimov, H. Synthesis of cellulose from pyruvate by succinate-grown cells of Acetobacter xylinum. J. Bacteriol. 1962, 84, 625–630. [Google Scholar] [CrossRef]
- Schramm, M.; Gromet, Z.; Hestrin, S. Synthesis of cellulose by Acetobacter xylinum. 3. Substrates and inhibitors. Biochem. J. 1957, 67, 669–679. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pang, M.; Huang, Y.; Meng, F.; Zhuang, Y.; Liu, H.; Du, M.; Ma, Q.; Wang, Q.; Chen, Z.; Chen, L.; et al. Application of bacterial cellulose in skin and bone tissue engineering. Eur. Polym. J. 2020, 122, 109365. [Google Scholar] [CrossRef]
- Watanabe, A.; Morita, S.; Ozaki, Y. Temperature-Dependent Changes in Hydrogen Bonds in Cellulose Iα Studied by Infrared Spectroscopy in Combination with Perturbation-Correlation Moving-Window Two-Dimensional Correlation Spectroscopy: Comparison with Cellulose Iβ. Biomacromolecules 2007, 8, 2969–2975. [Google Scholar] [CrossRef]
- Watanabe, K.; Tabuchi, M.; Morinaga, Y.; Yoshinaga, F. Structural Features and Properties of Bacterial Cellulose Produced in Agitated Culture. Cellulose 1998, 5, 187–200. [Google Scholar] [CrossRef]
- Krystynowicz, A.; Czaja, W.K.; Wiktorowska-Jezierska, A.; Gonçalves-Miśkiewicz, M.; Turkiewicz, M.; Bielecki, S. Factors affecting the yield and properties of bacterial cellulose. J. Ind. Microbiol. Biotechnol. 2002, 29, 189–195. [Google Scholar] [CrossRef] [PubMed]
- Hestrin, S.; Schramm, M. Synthesis of cellulose by Acetobacter xylinum. II. Preparation of freeze-dried cells capable of polymerizing glucose to cellulose. Biochem. J. 1954, 58, 345–352. [Google Scholar] [CrossRef] [Green Version]
- Rivas, B.; Moldes, A.B.; Domínguez, J.M.; Parajó, J.C. Development of culture media containing spent yeast cells of Debaryomyces hansenii and corn steep liquor for lactic acid production with Lactobacillus rhamnosus. Int. J. Food Microbiol. 2004, 97, 93–98. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Jia, J.; Xing, J.; Chen, J.; Lu, S. Isolation and characteristics analysis of a novel high bacterial cellulose producing strain Gluconacetobacter intermedius CIs26. Carbohydr. Polym. 2013, 92, 2012–2017. [Google Scholar] [CrossRef] [PubMed]
- Buldum, G.; Bismarck, A.; Mantalaris, A. Recombinant biosynthesis of bacterial cellulose in genetically modified Escherichia coli. Bioprocess. Biosyst. Eng. 2018, 41, 265–279. [Google Scholar] [CrossRef] [Green Version]
- Velásquez-Riaño, M.; Bojacá, V. Production of bacterial cellulose from alternative low-cost substrates. Cellulose 2017, 24, 2677–2698. [Google Scholar] [CrossRef]
- Ullah, M.W.; Ul-Islam, M.; Khan, S.; Kim, Y.; Park, J.K. Innovative production of bio-cellulose using a cell-free system derived from a single cell line. Carbohydr. Polym. 2015, 132, 286–294. [Google Scholar] [CrossRef]
- Kim, Y.; Ullah, M.W.; Ul-Islam, M.; Khan, S.; Jang, J.H.; Park, J.K. Self-assembly of bio-cellulose nanofibrils through intermediate phase in a cell-free enzyme system. Biochem. Eng. J. 2019, 142, 135–144. [Google Scholar] [CrossRef]
- Brown, R.M.J.; Kudlicka, K.; Cousins, S.K.; Nagy, R. Gravity effects on cellulose assembly. Am. J. Bot. 1992, 79, 1247–1258. [Google Scholar] [CrossRef]
- De Wulf, P.; Joris, K.; Vandamme, E.J. Improved cellulose formation by an Acetobacter xylinum mutant limited in (keto)gluconate synthesis. J. Chem. Technol. Biotechnol. 1996, 67, 376–380. [Google Scholar] [CrossRef]
- Tokoh, C.; Takabe, K.; Fujita, M.; Saiki, H. Cellulose Synthesized by Acetobacter xylinum in the Presence of Acetyl Glucomannan. Cellulose 1998, 5, 249–261. [Google Scholar] [CrossRef]
- Ul-Islam, M.; Khan, S.; Ullah, M.W.; Park, J.K. Bacterial cellulose composites: Synthetic strategies and multiple applications in bio-medical and electro-conductive fields. Biotechnol. J. 2015, 10, 1847–1861. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.-Y.; Buldum, G.; Mantalaris, A.; Bismarck, A. More than meets the eye in bacterial cellulose: Biosynthesis, bioprocessing, and applications in advanced fiber composites. Macromol. Biosci. 2014, 14, 10–32. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yan, Z.; Chen, S.; Wang, H.; Wang, B.; Jiang, J. Biosynthesis of bacterial cellulose/multi-walled carbon nanotubes in agitated culture. Carbohydr. Polym. 2008, 74, 659–665. [Google Scholar] [CrossRef]
- Zahan, K.A.; Hedzir, M.S.A.; Mustapha, M. The potential use of papaya juice as fermentation medium for bacterial cellulose production by Acetobacter xylinum 0416. Pertanika J. Trop. Agric. Sci. 2017, 40, 343–350. [Google Scholar]
- Aswini, K.; Gopal, N.O.; Uthandi, S. Optimized culture conditions for bacterial cellulose production by Acetobacter senegalensis MA1. BMC Biotechnol. 2020, 20, 46. [Google Scholar] [CrossRef]
- Rangaswamy, B.E.; Vanitha, K.P.; Hungund, B.S. Microbial Cellulose Production from Bacteria Isolated from Rotten Fruit. Int. J. Polym. Sci. 2015, 2015, 280784. [Google Scholar] [CrossRef] [Green Version]
- Zakaria, J.; Nazeri, M.A. Optimization of Bacterial Cellulose Production from Pineapple Waste: Effect of Temperature, pH and Concentration. In Proceedings of the Engineering Towards Change—Empowering Green Solutions, Sarawak, Malaysia, 10–12 July 2012; pp. 1–7. [Google Scholar]
- Ramana, K.V.; Tomar, A.; Singh, L. Effect of various carbon and nitrogen sources on cellulose synthesis by Acetobacter xylinum. World J. Microbiol. Biotechnol. 2000, 16, 245–248. [Google Scholar] [CrossRef]
- Matsuoka, M.; Tsuchida, T.; Matsushita, K.; Adachi, O.; Yoshinaga, F. A Synthetic Medium for Bacterial Cellulose Production by Acetobacter xylinum subsp. sucrofermentans. Biosci. Biotechnol. Biochem. 1996, 60, 575–579. [Google Scholar] [CrossRef]
- Keshk, S.; Sameshima, K. The utilization of sugar cane molasses with/without the presence of lignosulfonate for the production of bacterial cellulose. Appl. Microbiol. Biotechnol. 2006, 72, 291–296. [Google Scholar] [CrossRef] [PubMed]
- Bae, S.; Shoda, M. Statistical optimization of culture conditions for bacterial cellulose production using Box-Behnken design. Biotechnol. Bioeng. 2005, 90, 20–28. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.-Y.; Kim, J.-N.; Wee, Y.-J.; Park, D.-H.; Ryu, H.-W. Production of bacterial cellulose by Gluconacetobacter sp. RKY5 isolated from persimmon vinegar. Appl. Biochem. Biotechnol. 2006, 131, 705–715. [Google Scholar] [CrossRef]
- Singhsa, P.; Narain, R.; Manuspiya, H. Physical structure variations of bacterial cellulose produced by different Komagataeibacter xylinus strains and carbon sources in static and agitated conditions. Cellulose 2018, 25, 1571–1581. [Google Scholar] [CrossRef]
- Jung, H.-I.; Jeong, J.-H.; Lee, O.-M.; Park, G.-T.; Kim, K.-K.; Park, H.-C.; Lee, S.-M.; Kim, Y.-G.; Son, H.-J. Influence of glycerol on production and structural-physical properties of cellulose from Acetobacter sp. V6 cultured in shake flasks. Bioresour. Technol. 2010, 101, 3602–3608. [Google Scholar] [CrossRef] [PubMed]
- Revin, V.V.; Liyas’kina, E.V.; Sapunova, N.B.; Bogatyreva, A.O. Isolation and Characterization of the Strains Producing Bacterial Cellulose. Microbiology 2020, 89, 86–95. [Google Scholar] [CrossRef]
- Hu, W.; Chen, S.; Yang, J.; Li, Z.; Wang, H. Functionalized bacterial cellulose derivatives and nanocomposites. Carbohydr. Polym. 2014, 101, 1043–1060. [Google Scholar] [CrossRef]
- Jung, J.Y.; Park, J.K.; Chang, H.N. Bacterial cellulose production by Gluconacetobacter hansenii in an agitated culture without living non-cellulose producing cells. Enzyme Microb. Technol. 2005, 37, 347–354. [Google Scholar] [CrossRef]
- Hodel, K.V.; Fonseca, L.M.; Santos, I.M.; Cerqueira, J.C.; Santos-Júnior, R.E.; Nunes, S.B.; Barbosa, J.D.; Machado, B.A. Evaluation of Different Methods for Cultivating Gluconacetobacter hansenii for Bacterial Cellulose and Montmorillonite Biocomposite Production: Wound-Dressing Applications. Polymers 2020, 12, 267. [Google Scholar] [CrossRef] [Green Version]
- Castro, C.; Zuluaga, R.; Putaux, J.-L.; Caro, G.; Mondragon, I.; Gañán, P. Structural characterization of bacterial cellulose produced by Gluconacetobacter swingsii sp. from Colombian agroindustrial wastes. Carbohydr. Polym. 2011, 84, 96–102. [Google Scholar] [CrossRef]
- De Medeiros, A.D.M.; Da Silva, C.J.G., Jr.; De Amorim, J.D.P.; Do Nascimento, H.A.; Converti, A.; De Santana Costa, A.F.; Sarubbo, L.A. Biocellulose for treatment of wastewaters generated by energy consuming industries: A review. Energies 2021, 14, 5066. [Google Scholar] [CrossRef]
- Hwang, J.W.; Yang, Y.K.; Hwang, J.K.; Pyun, Y.R.; Kim, Y.S. Effects of pH and dissolved oxygen on cellulose production by Acetobacter xylinum BRC5 in agitated culture. J. Biosci. Bioeng. 1999, 88, 183–188. [Google Scholar] [CrossRef]
- Liu, M.; Li, S.; Xie, Y.; Jia, S.; Hou, Y.; Zou, Y.; Zhong, C. Enhanced bacterial cellulose production by Gluconacetobacter xylinus via expression of Vitreoscilla hemoglobin and oxygen tension regulation. Appl. Microbiol. Biotechnol. 2018, 102, 1155–1165. [Google Scholar] [CrossRef]
- Molina-Ramírez, C.; Castro, M.; Osorio, M.; Torres-Taborda, M.; Gómez, B.; Zuluaga, R.; Gómez, C.; Gañán, P.; Rojas, O.J.; Castro, C. Effect of Different Carbon Sources on Bacterial Nanocellulose Production and Structure Using the Low pH Resistant Strain Komagataeibacter Medellinensis. Materials 2017, 10, 639. [Google Scholar] [CrossRef]
- Ashori, A.; Sheykhnazari, S.; Tabarsa, T.; Shakeri, A.; Golalipour, M. Bacterial cellulose/silica nanocomposites: Preparation and characterization. Carbohydr. Polym. 2012, 90, 413–418. [Google Scholar] [CrossRef] [PubMed]
- Sharip, N.S.; Ariffin, H. Cellulose nanofibrils for biomaterial applications. Mater. Today Proc. 2019, 16, 1959–1968. [Google Scholar] [CrossRef]
- Mohd Amin, M.C.I.; Ahmad, N.; Halib, N.; Ahmad, I. Synthesis and characterization of thermo- and pH-responsive bacterial cellulose/acrylic acid hydrogels for drug delivery. Carbohydr. Polym. 2012, 88, 465–473. [Google Scholar] [CrossRef]
- Dayal, M.S.; Goswami, N.; Sahai, A.; Jain, V.; Mathur, G.; Mathur, A. Effect of media components on cell growth and bacterial cellulose production from Acetobacter aceti MTCC 2623. Carbohydr. Polym. 2013, 94, 12–16. [Google Scholar] [CrossRef]
- Li, D.; Ao, K.; Wang, Q.; Lv, P.; Wei, Q. Preparation of Pd/Bacterial Cellulose Hybrid Nanofibers for Dopamine Detection. Moleucles 2016, 21, 618. [Google Scholar] [CrossRef] [PubMed]
- Vasconcelos, N.F.; Feitosa, J.P.A.; da Gama, F.M.P.; Morais, J.P.S.; Andrade, F.K.; de Souza, M.D.S.M.; de Freitas Rosa, M. Bacterial cellulose nanocrystals produced under different hydrolysis conditions: Properties and morphological features. Carbohydr. Polym. 2017, 155, 425–431. [Google Scholar] [CrossRef]
- Elazzouzi-Hafraoui, S.; Nishiyama, Y.; Putaux, J.-L.; Heux, L.; Dubreuil, F.; Rochas, C. The shape and size distribution of crystalline nanoparticles prepared by acid hydrolysis of native cellulose. Biomacromolecules 2008, 9, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Bi, J.-C.; Liu, S.-X.; Li, C.-F.; Li, J.; Liu, L.-X.; Deng, J.; Yang, Y.-C. Morphology and structure characterization of bacterial celluloses produced by different strains in agitated culture. J. Appl. Microbiol. 2014, 117, 1305–1311. [Google Scholar] [CrossRef]
- Feng, X.; Ullah, N.; Wang, X.; Sun, X.; Li, C.; Bai, Y.; Chen, L.; Li, Z. Characterization of Bacterial Cellulose by Gluconacetobacter hansenii CGMCC 3917. J. Food Sci. 2015, 80, E2217–E2227. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Ullah, N.; Sun, X.; Guo, Y.; Chen, L.; Li, Z.; Feng, X. Development and characterization of bacterial cellulose reinforced biocomposite films based on protein from buckwheat distiller’s dried grains. Int. J. Biol. Macromol. 2017, 96, 353–360. [Google Scholar] [CrossRef]
- Xiang, C.; Acevedo, N.C. In Situ Self-Assembled Nanocomposites from Bacterial Cellulose Reinforced with Eletrospun Poly(lactic acid)/Lipids Nanofibers. Polymers 2017, 9, 179. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Teixeira, M.A.; Paiva, M.C.; Amorim, M.T.P.; Felgueiras, H.P. Electrospun Nanocomposites Containing Cellulose and Its Derivatives Modified with Specialized Biomolecules for an Enhanced Wound Healing. Nanomaterials 2020, 10, 557. [Google Scholar] [CrossRef] [Green Version]
- Fan, T.; Daniels, R. Preparation and Characterization of Electrospun Polylactic Acid (PLA) Fiber Loaded with Birch Bark Triterpene Extract for Wound Dressing. AAPS PharmSciTech 2021, 22, 205. [Google Scholar] [CrossRef]
- Pommet, M.; Juntaro, J.; Heng, J.Y.Y.; Mantalaris, A.; Lee, A.F.; Wilson, K.; Kalinka, G.; Shaffer, M.S.P.; Bismarck, A. Surface Modification of Natural Fibers Using Bacteria: Depositing Bacterial Cellulose onto Natural Fibers To Create Hierarchical Fiber Reinforced Nanocomposites. Biomacromolecules 2008, 9, 1643–1651. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kalia, S.; Kaith, B.S.; Kaur, I. Cellulose Fibers: Bio- and Nano-Polymer Composites; Springer: Berlin/Heidelberg, Germany, 2011. [Google Scholar]
- Fernandes, M.; Souto, A.P.; Dourado, F.; Gama, M. Application of Bacterial Cellulose in the Textile and Shoe Industry: Development of Biocomposites. Polysaccharides 2021, 2, 566–581. [Google Scholar] [CrossRef]
- Yu, K.; Aubin-Tam, M.-E. Bacterially Grown Cellulose/Graphene Oxide Composites Infused with γ-Poly (Glutamic Acid) as Biodegradable Structural Materials with Enhanced Toughness. ACS Appl. Nano Mater. 2020, 3, 12055–12063. [Google Scholar] [CrossRef]
- Zhang, P.; Chen, L.; Zhang, Q.; Hong, F.F. Using In Situ Dynamic Cultures to Rapidly Biofabricate Fabric-Reinforced Composites of Chitosan/Bacterial Nanocellulose for Antibacterial Wound Dressings. Front. Microbiol. 2016, 7, 260. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kołodziejska, M.; Jankowska, K.; Klak, M.; Wszoła, M. Chitosan as an Underrated Polymer in Modern Tissue Engineering. Nanomaterials 2021, 11, 3019. [Google Scholar] [CrossRef]
- Savitskaya, I.; Kistaubayeva, A.; Digel, I.; Shokatayeva, D. Physicochemical and Antibacterial Properties of Composite Films Based on Bacterial Cellulose and Chitosan for Wound Dressing Materials. Eurasian Chem. J. 2017, 19. [Google Scholar] [CrossRef] [Green Version]
- Cabañas-Romero, L.V.; Valls, C.; Valenzuela, S.V.; Roncero, M.B.; Pastor, F.I.J.; Diaz, P.; Martínez, J. Bacterial Cellulose–Chitosan Paper with Antimicrobial and Antioxidant Activities. Biomacromolecules 2020, 21, 1568–1577. [Google Scholar] [CrossRef] [PubMed]
- Ostadhossein, F.; Mahmoudi, N.; Morales-Cid, G.; Tamjid, E.; Navas-Martos, F.J.; Soriano-Cuadrado, B.; Paniza, J.M.L.; Simchi, A. Development of Chitosan/Bacterial Cellulose Composite Films Containing Nanodiamonds as a Potential Flexible Platform for Wound Dressing. Materials 2015, 8, 6401–6418. [Google Scholar] [CrossRef] [PubMed]
- Abral, H.; Pratama, A.B.; Handayani, D.; Mahardika, M.; Aminah, I.; Sandrawati, N.; Sugiarti, E.; Muslimin, A.N.; Sapuan, S.M.; Ilyas, R.A. Antimicrobial Edible Film Prepared from Bacterial Cellulose Nanofibers/Starch/Chitosan for a Food Packaging Alternative. Int. J. Polym. Sci. 2021, 2021, 6641284. [Google Scholar] [CrossRef]
- Laromaine, A.; Tronser, T.; Pini, I.; Parets, S.; Levkin, P.A.; Roig, A. Free-standing three-dimensional hollow bacterial cellulose structures with controlled geometry via patterned superhydrophobic–hydrophilic surfaces. Soft Matter 2018, 14, 3955–3962. [Google Scholar] [CrossRef] [PubMed]
- Ul-Islam, M.; Khan, T.; Khattak, W.A.; Park, J.K. Bacterial cellulose-MMTs nanoreinforced composite films: Novel wound dressing material with antibacterial properties. Cellulose 2013, 20, 589–596. [Google Scholar] [CrossRef]
- Pinto, R.J.B.; Daina, S.; Sadocco, P.; Neto, C.P.; Trindade, T. Antibacterial Activity of Nanocomposites of Copper and Cellulose. Biomed. Res. Int. 2013, 2013, 280512. [Google Scholar] [CrossRef] [Green Version]
- Xie, Y.-Y.; Hu, X.-H.; Zhang, Y.-W.; Wahid, F.; Chu, L.-Q.; Jia, S.-R.; Zhong, C. Development and antibacterial activities of bacterial cellulose/graphene oxide-CuO nanocomposite films. Carbohydr. Polym. 2020, 229, 115456. [Google Scholar] [CrossRef] [PubMed]
- Wahid, F.; Duan, Y.-X.; Hu, X.-H.; Chu, L.-Q.; Jia, S.-R.; Cui, J.-D.; Zhong, C. A facile construction of bacterial cellulose/ZnO nanocomposite films and their photocatalytic and antibacterial properties. Int. J. Biol. Macromol. 2019, 132, 692–700. [Google Scholar] [CrossRef]
- Yang, M.; Ward, J.; Choy, K.-L. Nature-Inspired Bacterial Cellulose/Methylglyoxal (BC/MGO) Nanocomposite for Broad-Spectrum Antimicrobial Wound Dressing. Macromol. Biosci. 2020, 20, 2000070. [Google Scholar] [CrossRef]
- De Mattos, I.B.; Nischwitz, S.P.; Tuca, A.-C.; Groeber-Becker, F.; Funk, M.; Birngruber, T.; Mautner, S.I.; Kamolz, L.-P.; Holzer, J.C.J. Delivery of antiseptic solutions by a bacterial cellulose wound dressing: Uptake, release and antibacterial efficacy of octenidine and povidone-iodine. Burns 2020, 46, 918–927. [Google Scholar] [CrossRef]
- Yaşayan, G.; Karaca, G.; Akgüner, Z.P.; Öztürk, A.B. Chitosan/collagen composite films as wound dressings encapsulating allantoin and lidocaine hydrochloride. Int. J. Polym. Mater. Polym. Biomater. 2021, 70, 623–635. [Google Scholar] [CrossRef]
- Beekmann, U.; Schmölz, L.; Lorkowski, S.; Werz, O.; Thamm, J.; Fischer, D.; Kralisch, D. Process control and scale-up of modified bacterial cellulose production for tailor-made anti-inflammatory drug delivery systems. Carbohydr. Polym. 2020, 236, 116062. [Google Scholar] [CrossRef] [PubMed]
- Lahiri, D.; Dash, S.; Dutta, R.; Nag, M. Elucidating the effect of anti-biofilm activity of bioactive compounds extracted from plants. J. Biosci. 2019, 44, 52. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, S.; Lahiri, D.; Nag, M.; Dey, A.; Sarkar, T.; Pathak, S.K.; Edinur, H.A.; Pati, S.; Ray, R.R. Bacterial biopolymer: Its role in pathogenesis to effective biomaterials. Polymers 2021, 13, 1242. [Google Scholar] [CrossRef]
- Lahiri, D.; Nag, M.; Ghosh, A.; Dey, A.; Mukherjee, D.; Garai, S.; Ray, R.R. Biofilm and Antimicrobial Resistance. In Biofilm-Mediated Diseases: Causes and Controls; Ray, R.R., Nag, M., Lahiri, D., Eds.; Springer: Singapore, 2021; pp. 183–208. [Google Scholar]
- Clinton, A.; Carter, T. Chronic Wound Biofilms: Pathogenesis and Potential Therapies. Lab. Med. 2015, 46, 277–284. [Google Scholar] [CrossRef] [Green Version]
- Lee, J.-H.; Park, J.-H.; Cho, H.S.; Joo, S.W.; Cho, M.H.; Lee, J. Anti-biofilm activities of quercetin and tannic acid against Staphylococcus aureus. Biofouling 2013, 29, 491–499. [Google Scholar] [CrossRef]
- Siddiqui, M.F.; Oh, H.-S.; Rzechowicz, M.; Winters, H.; Chong, T.H.; Fane, A.G. Biofouling control potential of tannic acid, ellagic acid, and epigallocatechin against Pseudomonas aeruginosa and reverse osmosis membrane multispecies community. J. Ind. Eng. Chem. 2015, 30, 204–211. [Google Scholar] [CrossRef]
- Luo, J.; Lai, J.; Zhang, N.; Liu, Y.; Liu, R.; Liu, X. Tannic Acid Induced Self-Assembly of Three-Dimensional Graphene with Good Adsorption and Antibacterial Properties. ACS Sustain. Chem. Eng. 2016, 4, 1404–1413. [Google Scholar] [CrossRef]
- Barud, H.S.; Barrios, C.; Regiani, T.; Marques, R.F.C.; Verelst, M.; Dexpert-Ghys, J.; Messaddeq, Y.; Ribeiro, S.J.L. Self-supported silver nanoparticles containing bacterial cellulose membranes. Mater. Sci. Eng. C 2008, 28, 515–518. [Google Scholar] [CrossRef]
- Pinto, R.J.B.; Marques, P.A.A.P.; Neto, C.P.; Trindade, T.; Daina, S.; Sadocco, P. Antibacterial activity of nanocomposites of silver and bacterial or vegetable cellulosic fibers. Acta Biomater. 2009, 5, 2279–2289. [Google Scholar] [CrossRef]
- Morones, J.R.; Elechiguerra, J.L.; Camacho, A.; Holt, K.; Kouri, J.B.; Ramírez, J.T.; Yacaman, M.J. The bactericidal effect of silver nanoparticles. Nanotechnology 2005, 16, 2346–2353. [Google Scholar] [CrossRef] [Green Version]
- Nagy, A.; Harrison, A.; Sabbani, S.; Munson, R.S.J.; Dutta, P.K.; Waldman, W.J. Silver nanoparticles embedded in zeolite membranes: Release of silver ions and mechanism of antibacterial action. Int. J. Nanomed. 2011, 6, 1833–1852. [Google Scholar] [CrossRef] [Green Version]
- Divya, K.; Vijayan, S.; George, T.K.; Jisha, M.S. Antimicrobial properties of chitosan nanoparticles: Mode of action and factors affecting activity. Fibers Polym. 2017, 18, 221–230. [Google Scholar] [CrossRef]
- Pati, S.; Chatterji, A.; Dash, B.P.; Nelson, B.R.; Sarkar, T.; Shahimi, S.; Edinur, H.A.; Abd Manan, T.S.B.; Jena, P.; Mohanta, Y.K.; et al. Structural characterization and antioxidant potential of chitosan by γ-irradiation from the carapace of horseshoe crab. Polymers 2020, 12, 2361. [Google Scholar] [CrossRef]
- Pati, S.; Sarkar, T.; Sheikh, H.I.; Bharadwaj, K.K.; Mohapatra, P.K.; Chatterji, A.; Dash, B.P.; Edinur, H.A.; Nelson, B.R. γ-Irradiated Chitosan from Carcinoscorpius rotundicauda (Latreille, 1802) Improves the Shelf Life of Refrigerated Aquatic Products. Front. Mar. Sci. 2021, 8, 498. [Google Scholar] [CrossRef]
- Kong, M.; Chen, X.G.; Xing, K.; Park, H.J. Antimicrobial properties of chitosan and mode of action: A state of the art review. Int. J. Food Microbiol. 2010, 144, 51–63. [Google Scholar] [CrossRef] [PubMed]
- Jeon, Y.-J.; Park, P.-J.; Kim, S.-K. Antimicrobial effect of chitooligosaccharides produced by bioreactor. Carbohydr. Polym. 2001, 44, 71–76. [Google Scholar] [CrossRef]
- Darabpour, E.; Kashef, N.; Mashayekhan, S. Chitosan nanoparticles enhance the efficiency of methylene blue-mediated antimicrobial photodynamic inactivation of bacterial biofilms: An in vitro study. Photodiagn. Photodyn. Ther. 2016, 14, 211–217. [Google Scholar] [CrossRef]
- Chen, X.; Cui, J.; Xu, X.; Sun, B.; Zhang, L.; Dong, W.; Chen, C.; Sun, D. Bacterial cellulose/attapulgite magnetic composites as an efficient adsorbent for heavy metal ions and dye treatment. Carbohydr. Polym. 2020, 229, 115512. [Google Scholar] [CrossRef]
- Song, S.; Liu, Z.; Zhang, J.; Jiao, C.; Ding, L.; Yang, S. Synthesis and Adsorption Properties of Novel Bacterial Cellulose/Graphene Oxide/Attapulgite Materials for Cu and Pb Ions in Aqueous Solutions. Materials 2020, 13, 3703. [Google Scholar] [CrossRef] [PubMed]


| Medium | Bacterial Strain | Incubation Days | Yield of BC g/L | Reference |
|---|---|---|---|---|
| Glycerol | Gluconacetobacter sp. RKY5 | 6 | 4.59 a 5.63 b | [57] |
| Glucose yeast extract broth | Acetobacter xylinum K086 | 7 days | 0.14–0.39 a | [58] |
| Acetobacter xylinum K975 | 1.11–1.55 a | |||
| Acetobacter xylinum K428 | 0.09–0.22 a | |||
| Acetobacter xylinum K1011 | 0.57–1.46 a | |||
| Acetobacter xylinum KX | 1.14–1.84 a | |||
| Glycerol | Acetobacter sp. V6 | 7 days | 4.98 b | [59] |
| Molasse | Komagataeibacter sucrofernentans H110 Komagataeibacter hansenii C110 | 14 days | 8.2 ± 0.2 a, 8.1 ± 0.2 a | [60] |
| Stillage | 9.5 ± 0.1 a, 9.2 ± 0.1 a | |||
| Citrus waste solution | Gluconacetobacter intermedius CIs26 | 8 days | 7.2 a | [61] |
| HS media | 2.1 a | |||
| Citrus waste modified HS | 5.7 a | |||
| Glucose | Gluconacetobacter hansenii | 2 | 1.33 b | [62] |
| Glucose (modified HS Media) | 14 | 14.72 a | [63] | |
| Mannitol (modified HS media) | 20 | |||
| Pineapple peel juice | Gluconacetobacterswingsii | 13 | 2.8 a | [64] |
| Characterization | Properties | References | |
|---|---|---|---|
| Absorption Peak | Functional Groups | ||
| FTIR | ~1160; ~1361; ~2895; ~3338 | C–O–C antisymmetricbridge stretching of 1,4-β-d-glucoside; C–H bending; C–H stretching of CH2 and CH3; –OH stretching | [69] |
| ~1314; ~1426 | CH2 groups out-of-plane bending; O–H in-plane bending | [70] | |
| ~1108 | C–C bonds of the monomer units of polysaccharide | [71] | |
| ~1335 | C–H deformation/–OH in-plane bending | [64] | |
| ~900 | Antisymmetric out-of-phase ring stretching for β-glucosidic linkages | [72] | |
| ~1054 | Bending of the C–O–H bond of carbohydrate | [73] | |
| XRD | 14.5°, 16.4°, and 22.5° diffraction peak corresponds to crystallographic planes of 101 (amorphous) and 200 (crystalline) | [74] | |
| Due to the presence of preferential parallel orientation of the cellulose fibrils, 1 12 crystallographic planes are missed in BC diffraction pattern | [75] | ||
| intra and intermolecular H-bonding influences Crystallization | [76] | ||
| Tensile strength (TS) and Youngs’ modulus (YM) | 0.26 ± 0.02 MPa TS and 0.005 ± 0.0003 MPa YM for Wet BC | [77] | |
| 11.94 ± 1.15 MPa TS and 6.65 ± 0.16 MPa YM for lyophilized BC | |||
| Scannining Electron Microscopy (SEM) | Helps in providing the structural morphology of the BC | [78] | |
| Differential scanning calorimetry (DSC) | Helps in the determination of the stability of the BC | [78] | |
| Viscosity measurements of the film-forming solution (FFS) | Helps in the purpose of analyzying the viscosity of the BC | [78] | |
| Moisture content (MC) | Helps in the estimation of the water resistance capasity of the film | [78] | |
| Water vapor permeability (WVP) | [78] | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lahiri, D.; Nag, M.; Dutta, B.; Dey, A.; Sarkar, T.; Pati, S.; Edinur, H.A.; Abdul Kari, Z.; Mohd Noor, N.H.; Ray, R.R. Bacterial Cellulose: Production, Characterization, and Application as Antimicrobial Agent. Int. J. Mol. Sci. 2021, 22, 12984. https://doi.org/10.3390/ijms222312984
Lahiri D, Nag M, Dutta B, Dey A, Sarkar T, Pati S, Edinur HA, Abdul Kari Z, Mohd Noor NH, Ray RR. Bacterial Cellulose: Production, Characterization, and Application as Antimicrobial Agent. International Journal of Molecular Sciences. 2021; 22(23):12984. https://doi.org/10.3390/ijms222312984
Chicago/Turabian StyleLahiri, Dibyajit, Moupriya Nag, Bandita Dutta, Ankita Dey, Tanmay Sarkar, Siddhartha Pati, Hisham Atan Edinur, Zulhisyam Abdul Kari, Noor Haslina Mohd Noor, and Rina Rani Ray. 2021. "Bacterial Cellulose: Production, Characterization, and Application as Antimicrobial Agent" International Journal of Molecular Sciences 22, no. 23: 12984. https://doi.org/10.3390/ijms222312984
APA StyleLahiri, D., Nag, M., Dutta, B., Dey, A., Sarkar, T., Pati, S., Edinur, H. A., Abdul Kari, Z., Mohd Noor, N. H., & Ray, R. R. (2021). Bacterial Cellulose: Production, Characterization, and Application as Antimicrobial Agent. International Journal of Molecular Sciences, 22(23), 12984. https://doi.org/10.3390/ijms222312984








