Targeting Mitochondria by Plant Secondary Metabolites: A Promising Strategy in Combating Parkinson’s Disease
Abstract
:1. Introduction
2. Mitochondria in PD
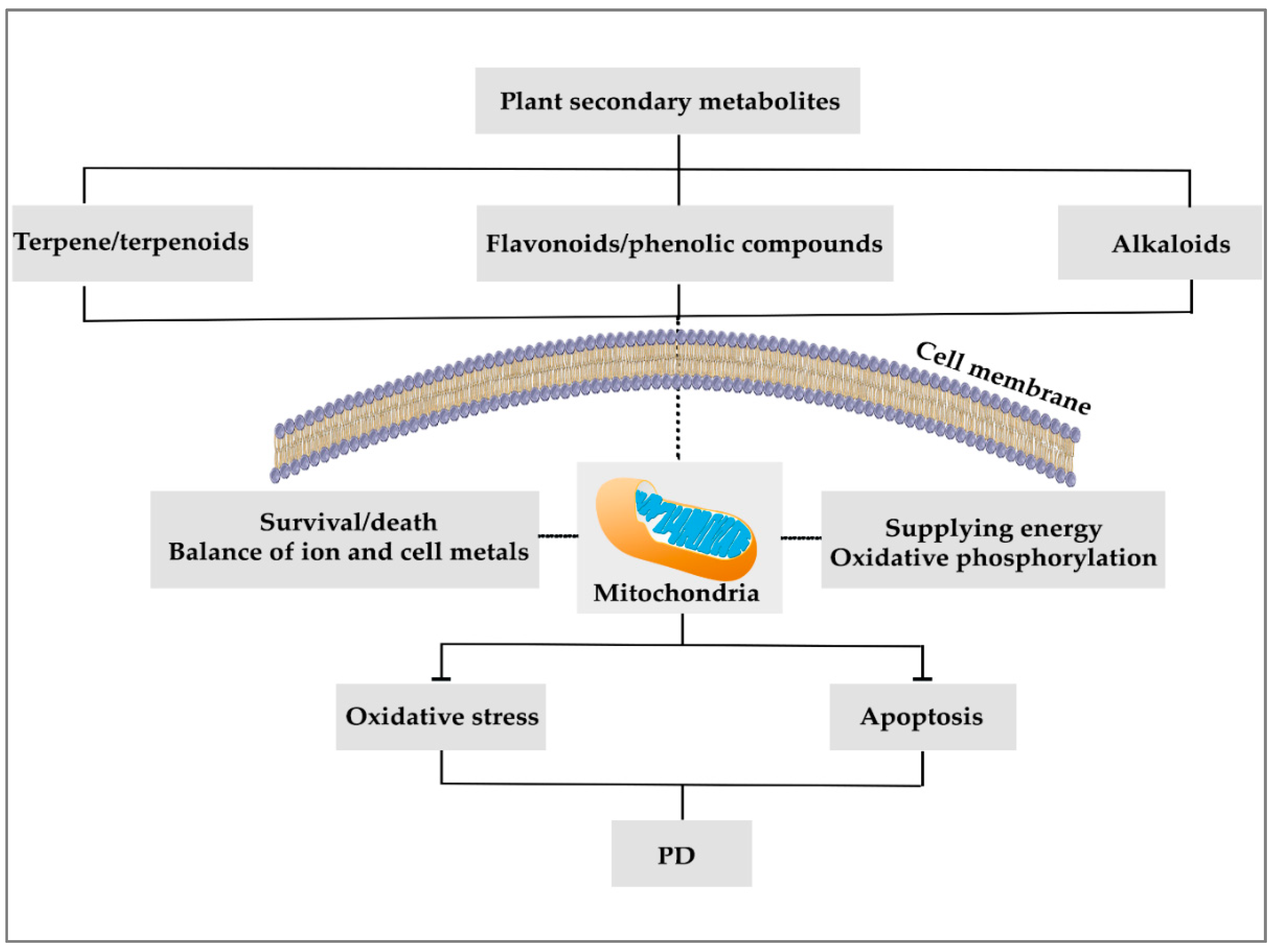
3. Plant Secondary Metabolites and Mitochondria
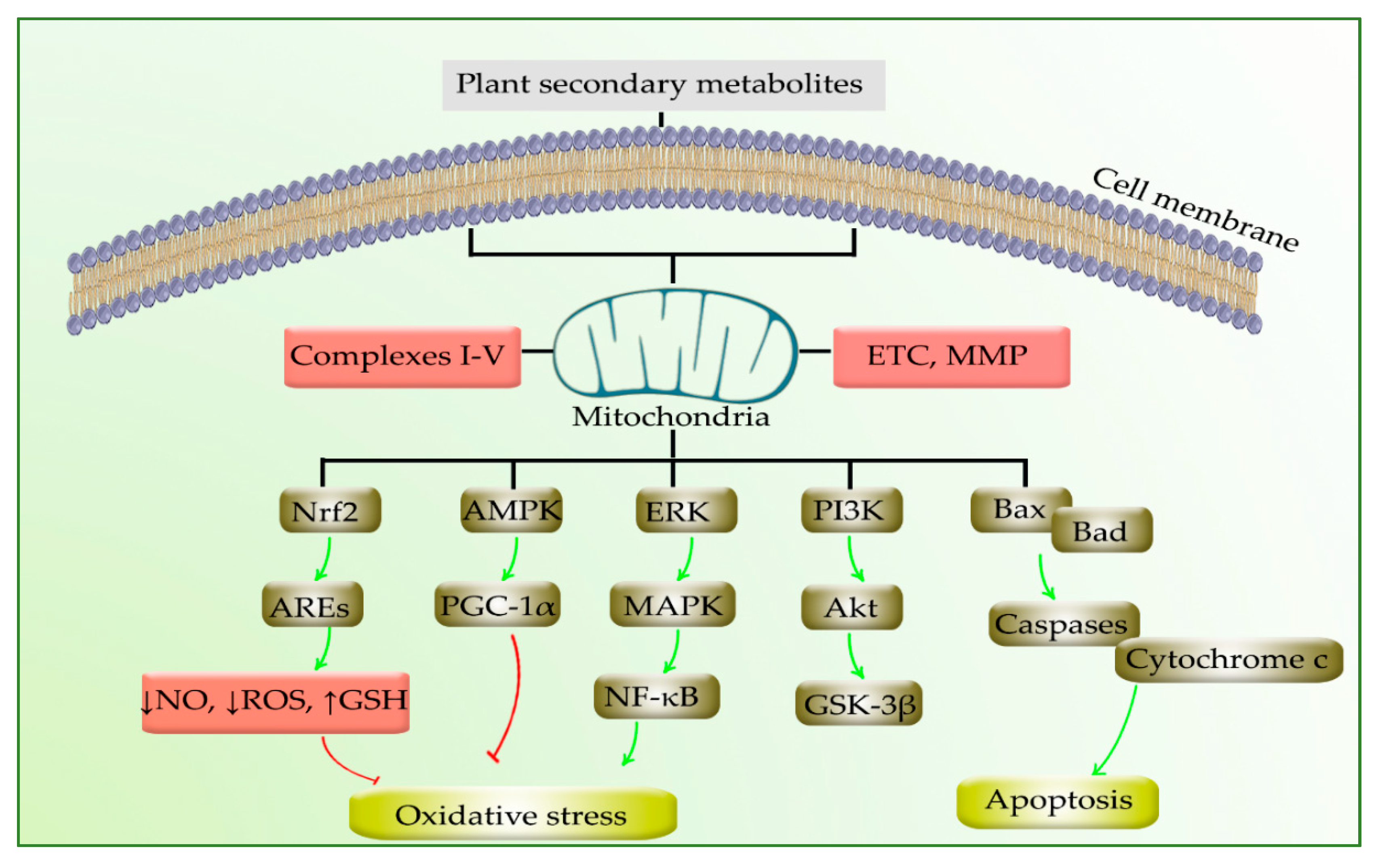
4. Modulation of PD by Phytochemicals through Targeting Mitochondria
4.1. Polyphenols Effects on Mitochondria in PD
4.2. Alkaloids and Mitochondria in PD
4.3. Terpenes and Mitochondria in PD
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Rekatsina, M.; Paladini, A.; Piroli, A.; Zis, P.; Pergolizzi, J.V.; Varrassi, G. Pathophysiology and Therapeutic Perspectives of Oxidative Stress and Neurodegenerative Diseases: A Narrative Review. Adv. Ther. 2019, 37, 113–139. [Google Scholar] [CrossRef] [Green Version]
- Barnham, K.J.; Masters, C.L.; Bush, A. Neurodegenerative diseases and oxidative stress. Nat. Rev. Drug Discov. 2004, 3, 205–214. [Google Scholar] [CrossRef]
- Dugger, B.N.; Dickson, D.W. Pathology of Neurodegenerative Diseases. Cold Spring Harb. Perspect. Biol. 2017, 9, a028035. [Google Scholar] [CrossRef]
- Palop, J.; Chin, J.; Mucke, L. A network dysfunction perspective on neurodegenerative diseases. Nature 2006, 443, 768–773. [Google Scholar] [CrossRef]
- Iborra, S.F.; Vila, M.; Perier, C. Mitochondrial Quality Control in Neurodegenerative Diseases: Focus on Parkinson’s Disease and Huntington’s Disease. Front. Neurosci. 2018, 12, 342. [Google Scholar] [CrossRef] [Green Version]
- Angeloni, C.; Malaguti, M.; Barbalace, M.C.; Hrelia, S.; Angeloni, C.; Malaguti, M.; Barbalace, M.C.; Hrelia, S. Bioactivity of Olive Oil Phenols in Neuroprotection. Int. J. Mol. Sci. 2017, 18, 2230. [Google Scholar] [CrossRef] [Green Version]
- Kramer, P.; Bressan, P. Our (Mother’s) Mitochondria and Our Mind. Perspect. Psychol. Sci. 2017, 13, 88–100. [Google Scholar] [CrossRef] [Green Version]
- Reddy, H.; Reddy, T.P. Mitochondria as a Therapeutic Target for Aging and Neurodegenerative Diseases. Curr. Alzheimer Res. 2011, 8, 393–409. [Google Scholar] [CrossRef]
- Sandoval, H.; Yao, C.-K.; Chen, K.; Jaiswal, M.; Donti, T.; Lin, Y.Q.; Bayat, V.; Xiong, B.; Zhang, K.; David, G.; et al. Mitochondrial fusion but not fission regulates larval growth and synaptic development through steroid hormone production. eLife 2014, 3. [Google Scholar] [CrossRef]
- Sun, N.; Youle, R.J.; Finkel, T. The Mitochondrial Basis of Aging. Mol. Cell 2016, 61, 654–666. [Google Scholar] [CrossRef] [Green Version]
- Melo, A.; Monteiro, L.; Lima, R.M.F.; de Oliveira, D.M.; De Cerqueira, M.D.; El-Bachá, R. Oxidative Stress in Neurodegenerative Diseases: Mechanisms and Therapeutic Perspectives. Oxidative Med. Cell. Longev. 2011, 2011, 1–14. [Google Scholar] [CrossRef]
- Kausar, S.; Wang, F.; Cui, H. The Role of Mitochondria in Reactive Oxygen Species Generation and Its Implications for Neurodegenerative Diseases. Cells 2018, 7, 274. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khan, H.; Ullah, H.; Aschner, M.; Cheang, W.S.; Akkol, E.K. Neuroprotective Effects of Quercetin in Alzheimer’s Disease. Biomolecules 2019, 10, 59. [Google Scholar] [CrossRef] [Green Version]
- Kalani, K.; Yan, S.F.; Yan, S.S. Mitochondrial permeability transition pore: A potential drug target for neurodegeneration. Drug Discov. Today 2018, 23, 1983–1989. [Google Scholar] [CrossRef] [PubMed]
- Guo, C.; Zhu, J.; Wang, J.; Duan, J.; Ma, S.; Yin, Y.; Quan, W.; Zhang, W.; Guan, Y.; Ding, Y.; et al. Neuroprotective effects of protocatechuic aldehyde through PLK2/p-GSK3β/Nrf2 signaling pathway in both in vivo and in vitro models of Parkinson’s disease. Aging 2019, 11, 9424–9441. [Google Scholar] [CrossRef]
- Mazzio, E.A.; Close, F.; Soliman, K.F. The Biochemical and Cellular Basis for Nutraceutical Strategies to Attenuate Neurodegeneration in Parkinson’s Disease. Int. J. Mol. Sci. 2011, 12, 506–569. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hajieva, P. The Effect of Polyphenols on Protein Degradation Pathways: Implications for Neuroprotection. Molecules 2017, 22, 159. [Google Scholar] [CrossRef] [Green Version]
- Seidl, S.E.; Santiago, J.A.; Bilyk, H.; Potashkin, J.A. The emerging role of nutrition in Parkinson’s disease. Front. Aging Neurosci. 2014, 6, 36. [Google Scholar] [CrossRef] [Green Version]
- Iriti, M.; Vitalini, S.; Fico, G.; Faoro, F. Neuroprotective Herbs and Foods from Different Traditional Medicines and Diets. Molecules 2010, 15, 3517–3555. [Google Scholar] [CrossRef] [Green Version]
- Caruana, M.; Cauchi, R.; Vassallo, N. Putative Role of Red Wine Polyphenols against Brain Pathology in Alzheimer’s and Parkinson’s Disease. Front. Nutr. 2016, 3, 31. [Google Scholar] [CrossRef] [Green Version]
- Boulos, C.; Yaghi, N.; El Hayeck, R.; Heraoui, G.N.; Fakhoury-Sayegh, N. Nutritional Risk Factors, Microbiota and Parkinson’s Disease: What Is the Current Evidence? Nutrients 2019, 11, 1896. [Google Scholar] [CrossRef] [Green Version]
- Kicinska, A.; Jarmuszkiewicz, W. Flavonoids and Mitochondria: Activation of Cytoprotective Pathways? Molecules 2020, 25, 3060. [Google Scholar] [CrossRef] [PubMed]
- Denzer, I.; Münch, G.; Friedland, K. Modulation of mitochondrial dysfunction in neurodegenerative diseases via activation of nuclear factor erythroid-2-related factor 2 by food-derived compounds. Pharmacol. Res. 2015, 103, 80–94. [Google Scholar] [CrossRef] [PubMed]
- Limboonreung, T.; Tuchinda, P.; Chongthammakun, S. Chrysoeriol mediates mitochondrial protection via PI3K/Akt pathway in MPP+ treated SH-SY5Y cells. Neurosci. Lett. 2019, 714, 134545. [Google Scholar] [CrossRef]
- Aryal, S.; Skinner, T.; Bridges, B.; Weber, J.T. The Pathology of Parkinson’s Disease and Potential Benefit of Dietary Polyphenols. Molecules 2020, 25, 4382. [Google Scholar] [CrossRef] [PubMed]
- Roshan, M.H.; Tambo, A.; Pace, N.P. Potential Role of Caffeine in the Treatment of Parkinson’s Disease. Open Neurol. J. 2016, 10, 42–58. [Google Scholar] [CrossRef] [PubMed]
- Kühlbrandt, W. Structure and function of mitochondrial membrane protein complexes. BMC Biol. 2015, 13, 1–11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marella, M.; Seo, B.B.; Yagi, T.; Matsuno-Yagi, A. Parkinson’s disease and mitochondrial complex I: A perspective on the Ndi1 therapy. J. Bioenerg. Biomembr. 2009, 41, 493–497. [Google Scholar] [CrossRef] [Green Version]
- Jaiswal, S.; Fontanillas, P.; Flannick, J.; Manning, A.; Grauman, P.V.; Mar, B.; Lindsley, C.; Mermel, C.; Burtt, N.; Chavez, A.; et al. Age-Related Clonal Hematopoiesis Associated with Adverse Outcomes. N. Engl. J. Med. 2014, 371, 2488–2498. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xiong, S.; Liu, W.; Zhou, Y.; Mo, Y.; Liu, Y.; Chen, X.; Pan, H.; Yuan, D.; Wang, Q.; Chen, T. Enhancement of oral bioavailability and anti-Parkinsonian efficacy of resveratrol through a nanocrystal formulation. Asian J. Pharm. Sci. 2019, 15, 518–528. [Google Scholar] [CrossRef]
- Fan, D.; Liu, L.; Wu, Z.; Cao, M. Combating Neurodegenerative Diseases with the Plant Alkaloid Berberine: Molecular Mechanisms and Therapeutic Potential. Curr. Neuropharmacol. 2019, 17, 563–579. [Google Scholar] [CrossRef]
- Cui, X.; Lin, Q.; Liang, Y. Plant-Derived Antioxidants Protect the Nervous System from Aging by Inhibiting Oxidative Stress. Front. Aging Neurosci. 2020, 12, 209. [Google Scholar] [CrossRef]
- Ganesan, P.; Ko, H.-M.; Kim, I.-S.; Choi, D.-K. Recent trends in the development of nanophytobioactive compounds and delivery systems for their possible role in reducing oxidative stress in Parkinson’s disease models. Int. J. Nanomed. 2015, 10, 6757–6772. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maiti, P.; Dunbar, G.L. Use of Curcumin, a Natural Polyphenol for Targeting Molecular Pathways in Treating Age-Related Neurodegenerative Diseases. Int. J. Mol. Sci. 2018, 19, 1637. [Google Scholar] [CrossRef] [Green Version]
- Vauzour, D.; Vafeiadou, K.; Rodriguez-Mateos, A.; Rendeiro, C.; Spencer, J.P.E. The neuroprotective potential of flavonoids: A multiplicity of effects. Genes Nutr. 2008, 3, 115–126. [Google Scholar] [CrossRef] [Green Version]
- Song, J.-X.; Sze, S.C.-W.; Ng, T.-B.; Lee, C.K.F.; Leung, G.P.H.; Shaw, P.-C.; Tong, Y.; Zhang, Y.-B. Anti-Parkinsonian drug discovery from herbal medicines: What have we got from neurotoxic models? J. Ethnopharmacol. 2011, 139, 698–711. [Google Scholar] [CrossRef]
- Li, Y.; Zhao, J.; Hölscher, C. Therapeutic Potential of Baicalein in Alzheimer’s Disease and Parkinson’s Disease. CNS Drugs 2017, 31, 639–652. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, X.-X.; He, G.-R.; Mu, X.; Xu, B.; Tian, S.; Yu, X.; Meng, F.-R.; Xuan, Z.-H.; Du, G.-H. Protective effects of baicalein against rotenone-induced neurotoxicity in PC12 cells and isolated rat brain mitochondria. Eur. J. Pharmacol. 2012, 674, 227–233. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Cheng, Y.; Yang, C.; Lau, S.; Lao, L.; Shuai, B.; Cai, J.; Rong, J. Botanical Drug Puerarin Attenuates 6-Hydroxydopamine (6-OHDA)-Induced Neurotoxicity via Upregulating Mitochondrial Enzyme Arginase-2. Mol. Neurobiol. 2015, 53, 2200–2211. [Google Scholar] [CrossRef]
- Cheng, Y.-F.; Zhu, G.-Q.; Wang, M.; Cheng, H.; Zhou, A.; Wang, N.; Fang, N.; Wang, X.-C.; Xiao, X.-Q.; Chen, Z.-W.; et al. Involvement of ubiquitin proteasome system in protective mechanisms of Puerarin to MPP+-elicited apoptosis. Neurosci. Res. 2009, 63, 52–58. [Google Scholar] [CrossRef]
- Lin, K.-L.; Wang, P.-W.; Chuang, J.-H.; Lin, H.-Y.; Chen, S.-D.; Chuang, Y.-C.; Huang, S.-T.; Tiao, M.-M.; Chen, J.-B.; Huang, P.-H.; et al. Resveratrol provides neuroprotective effects through modulation of mitochondrial dynamics and ERK1/2 regulated autophagy. Free. Radic. Res. 2018, 52, 1371–1386. [Google Scholar] [CrossRef]
- Uddin, S.; Al Mamun, A.; Kabir, T.; Ahmad, J.; Jeandet, P.; Sarwar, S.; Ashraf, G.M.; Aleya, L. Neuroprotective role of polyphenols against oxidative stress-mediated neurodegeneration. Eur. J. Pharmacol. 2020, 886, 173412. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.; Piao, Y.; Kang, Y.C.; Lim, S.; Pak, Y.K. Qi-activating quercetin alleviates mitochondrial dysfunction and neuroinflammation in vivo and in vitro. Arch. Pharmacal Res. 2020, 43, 553–566. [Google Scholar] [CrossRef]
- Wang, W.-W.; Han, R.; He, H.-J.; Li, J.; Chen, S.-Y.; Gu, Y.; Xie, C. Administration of quercetin improves mitochondria quality control and protects the neurons in 6-OHDA-lesioned Parkinson’s disease models. Aging 2021, 13, 11738–11751. [Google Scholar] [CrossRef]
- Karuppagounder, S.; Madathil, S.; Pandey, M.; Haobam, R.; Rajamma, U.; Mohanakumar, K. Quercetin up-regulates mitochondrial complex-I activity to protect against programmed cell death in rotenone model of Parkinson’s disease in rats. Neuroscience 2013, 236, 136–148. [Google Scholar] [CrossRef]
- Singh, A.; Tripathi, P.; Yadawa, A.K.; Singh, S. Promising Polyphenols in Parkinson’s Disease Therapeutics. Neurochem. Res. 2020, 45, 1731–1745. [Google Scholar] [CrossRef]
- Kujawska, M.; Jodynis-Liebert, J. Polyphenols in Parkinson’s Disease: A Systematic Review of In Vivo Studies. Nutrients 2018, 10, 642. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, X.; Liu, W.; Wang, C.; Chen, Y.; Liu, P.; Hayashi, T.; Mizuno, K.; Hattori, S.; Fujisaki, H.; Ikejima, T. Silibinin Attenuates Motor Dysfunction in a Mouse Model of Parkinson’s Disease by Suppression of Oxidative Stress and Neuroinflammation along with Promotion of Mitophagy. Physiol. Behav. 2021, 239, 113510. [Google Scholar] [CrossRef]
- Lee, Y.; Park, H.R.; Chun, H.J.; Lee, J. Silibinin prevents dopaminergic neuronal loss in a mouse model of Parkinson’s disease via mitochondrial stabilization. J. Neurosci. Res. 2015, 93, 755–765. [Google Scholar] [CrossRef] [PubMed]
- Fakhri, S.; Pesce, M.; Patruno, A.; Moradi, S.Z.; Iranpanah, A.; Farzaei, M.H.; Sobarzo-Sánchez, E. Attenuation of Nrf2/Keap1/ARE in Alzheimer’s Disease by Plant Secondary Metabolites: A Mechanistic Review. Molecules 2020, 25, 4926. [Google Scholar] [CrossRef]
- de Oliveira, M.R.; de Souza, I.C.C.; Fürstenau, C.R. Promotion of mitochondrial protection by naringenin in methylglyoxal-treated SH-SY5Y cells: Involvement of the Nrf2/GSH axis. Chem. Interact. 2019, 310, 108728. [Google Scholar] [CrossRef]
- Garabadu, D.; Agrawal, N. Naringin Exhibits Neuroprotection Against Rotenone-Induced Neurotoxicity in Experimental Rodents. NeuroMol. Med. 2020, 22, 314–330. [Google Scholar] [CrossRef] [PubMed]
- Ramalingam, M.; Kim, H.; Lee, Y.; Lee, Y.-I. Phytochemical and Pharmacological Role of Liquiritigenin and Isoliquiritigenin from Radix Glycyrrhizae in Human Health and Disease Models. Front. Aging Neurosci. 2018, 10, 348. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Balakrishnan, R.; Azam, S.; Cho, D.-Y.; Su-Kim, I.; Choi, D.-K. Natural Phytochemicals as Novel Therapeutic Strategies to Prevent and Treat Parkinson’s Disease: Current Knowledge and Future Perspectives. Oxidative Med. Cell. Longev. 2021, 2021, 1–32. [Google Scholar] [CrossRef]
- Fontanilla, C.; Ma, Z.; Wei, X.; Klotsche, J.; Zhao, L.; Wisniowski, P.; Dodel, R.; Farlow, M.; Oertel, W.; Du, Y. Caffeic acid phenethyl ester prevents 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-induced neurodegeneration. Neuroscience 2011, 188, 135–141. [Google Scholar] [CrossRef] [PubMed]
- Sun, W.; Wang, X.; Hou, C.; Yang, L.; Li, H.; Guo, J.; Huo, C.; Wang, M.; Miao, Y.; Liu, J.; et al. Oleuropein improves mitochondrial function to attenuate oxidative stress by activating the Nrf2 pathway in the hypothalamic paraventricular nucleus of spontaneously hypertensive rats. Neuropharmacology 2017, 113, 556–566. [Google Scholar] [CrossRef]
- Zhou, Z.D.; Xie, S.P.; Saw, W.T.; Ho, P.G.H.; Wang, H.; Lei, Z.; Yi, Z.; Tan, E.K. The Therapeutic Implications of Tea Polyphenols against Dopamine (DA) Neuron Degeneration in Parkinson’s Disease (PD). Cells 2019, 8, 911. [Google Scholar] [CrossRef] [Green Version]
- Kocot, J.; Kiełczykowska, M.; Luchowska-Kocot, D.; Kurzepa, J.; Musik, I. Antioxidant Potential of Propolis, Bee Pollen, and Royal Jelly: Possible Medical Application. Oxidative Med. Cell. Longev. 2018, 2018, 1–29. [Google Scholar] [CrossRef]
- Cole, G.M.; Teter, B.; Frautschy, S.A. Neuroprotective effects of curcumin. In The Molecular Targets and Therapeutic Uses of Curcumin in Health and Disease; Springer: Berlin/Heidelberg, Germany, 2007; Volume 595, pp. 197–212. [Google Scholar] [CrossRef] [Green Version]
- Zhao, G.; Yao-Yue, C.; Qin, G.-W.; Guo, L.-H. Luteolin from Purple Perilla mitigates ROS insult particularly in primary neurons. Neurobiol. Aging 2012, 33, 176–186. [Google Scholar] [CrossRef]
- Ding, Y.; Xin, C.; Zhang, C.-W.; Lim, K.-L.; Zhang, H.; Fu, Z.; Li, L.; Huang, W. Natural Molecules from Chinese Herbs Protecting Against Parkinson’s Disease via Anti-oxidative Stress. Front. Aging Neurosci. 2018, 10, 246. [Google Scholar] [CrossRef]
- Antunes, M.S.; Ladd, F.V.L.; Ladd, A.A.B.L.; Moreira, A.L.; Boeira, S.P.; Souza, L.C. Hesperidin protects against behavioral alterations and loss of dopaminergic neurons in 6-OHDA-lesioned mice: The role of mitochondrial dysfunction and apoptosis. Metab. Brain Dis. 2020, 36, 153–167. [Google Scholar] [CrossRef]
- Ahmad, I.; Fakhri, S.; Khan, H.; Jeandet, P.; Aschner, M.; Yu, Z.-L. Targeting cell cycle by β-carboline alkaloids in vitro: Novel therapeutic prospects for the treatment of cancer. Chem. Interact. 2020, 330, 109229. [Google Scholar] [CrossRef]
- Duan, W.-J.; Liang, L.; Pan, M.-H.; Lu, D.-H.; Wang, T.-M.; Li, S.-B.; Zhong, H.-B.; Yang, X.-J.; Cheng, Y.; Liu, B.; et al. Theacrine, a purine alkaloid from kucha, protects against Parkinson’s disease through SIRT3 activation. Phytomedicine 2020, 77, 153281. [Google Scholar] [CrossRef] [PubMed]
- Malińska, D.; Wieckowski, M.; Michalska, B.; Drabik, K.; Prill, M.; Patalas-Krawczyk, P.; Walczak, J.; Szymański, J.; Mathis, C.; van der Toorn, M.; et al. Mitochondria as a possible target for nicotine action. J. Bioenerg. Biomembr. 2019, 51, 259–276. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xie, Y.-X.; Bezard, E.; Zhao, B.-L. Investigating the Receptor-independent Neuroprotective Mechanisms of Nicotine in Mitochondria. J. Biol. Chem. 2005, 280, 32405–32412. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Godoy, J.A.; Valdivieso, A.G.; Inestrosa, N.C. Nicotine Modulates Mitochondrial Dynamics in Hippocampal Neurons. Mol. Neurobiol. 2018, 55, 8965–8977. [Google Scholar] [CrossRef]
- Cormier, A.; Morin, C.; Zini, R.; Tillement, J.-P.; Lagrue, G. Nicotine protects rat brain mitochondria against experimental injuries. Neuropharmacology 2003, 44, 642–652. [Google Scholar] [CrossRef]
- Liu, Y.; Zeng, X.; Hui, Y.; Zhu, C.; Wu, J.; Taylor, D.H.; Ji, J.; Fan, W.; Huang, Z.; Hu, J. Activation of α7 nicotinic acetylcholine receptors protects astrocytes against oxidative stress-induced apoptosis: Implications for Parkinson’s disease. Neuropharmacology 2014, 91, 87–96. [Google Scholar] [CrossRef] [PubMed]
- Schepici, G.; Silvestro, S.; Bramanti, P.; Mazzon, E. Caffeine: An Overview of Its Beneficial Effects in Experimental Models and Clinical Trials of Parkinson’s Disease. Int. J. Mol. Sci. 2020, 21, 4766. [Google Scholar] [CrossRef]
- Ahmed, T.; Gilani, A.; Abdollahi, M.; Daglia, M.; Nabavi, S.M. Berberine and neurodegeneration: A review of literature. Pharmacol. Rep. 2015, 67, 970–979. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Sheng, W.; Tan, Z.; Ren, Q.; Wang, R.; Stoika, R.; Liu, X.; Liu, K.; Shang, X.; Jin, M. Treatment of Parkinson’s disease in Zebrafish model with a berberine derivative capable of crossing blood brain barrier, targeting mitochondria, and convenient for bioimaging experiments. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2021, 249, 109151. [Google Scholar] [CrossRef] [PubMed]
- Fakhri, S.; Piri, S.; Majnooni, M.B.; Farzaei, M.H.; Echeverría, J. Targeting Neurological Manifestations of Coronaviruses by Candidate Phytochemicals: A Mechanistic Approach. Front. Pharmacol. 2021, 11, 621099. [Google Scholar] [CrossRef]
- Caruso, F.; Rossi, M.; Kaur, S.; Garcia-Villar, E.; Molasky, N.; Belli, S.; Sitek, J.D.; Gionfra, F.; Pedersen, J.Z.; Incerpi, S. Antioxidant Properties of Embelin in Cell Culture. Electrochemistry and Theoretical Mechanism of Scavenging. Potential Scavenging of Superoxide Radical through the Cell Membrane. Antioxidants 2020, 9, 382. [Google Scholar] [CrossRef] [PubMed]
- Rao, S.P.; Sharma, N.; Kalivendi, S.V. Embelin averts MPTP-induced dysfunction in mitochondrial bioenergetics and biogenesis via activation of SIRT1. Biochim. Biophys. Acta (BBA)-Bioenerg. 2020, 1861, 148157. [Google Scholar] [CrossRef]
- Lu, J.-H.; Tan, J.-Q.; Durairajan, S.S.K.; Liu, L.-F.; Zhang, Z.-H.; Ma, L.; Shen, H.-M.; Chan, H.E.; Li, M. Isorhynchophylline, a natural alkaloid, promotes the degradation of alpha-synuclein in neuronal cells via inducing autophagy. Autophagy 2012, 8, 98–108. [Google Scholar] [CrossRef] [Green Version]
- Li, X.-M.; Zhang, X.-J.; Dong, M.-X. Isorhynchophylline Attenuates MPP+-Induced Apoptosis Through Endoplasmic Reticulum Stress- and Mitochondria-Dependent Pathways in PC12 Cells: Involvement of Antioxidant Activity. NeuroMol. Med. 2017, 19, 480–492. [Google Scholar] [CrossRef]
- Zhu, Y.Z.; Wu, W.; Zhu, Q.; Liu, X. Discovery of Leonuri and therapeutical applications: From bench to bedside. Pharmacol. Ther. 2018, 188, 26–35. [Google Scholar] [CrossRef]
- Fakhri, S.; Moradi, S.Z.; Farzaei, M.H.; Bishayee, A. Modulation of dysregulated cancer metabolism by plant secondary metabolites: A mechanistic review. Semin. Cancer Biol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Park, H.-A.; Hayden, M.M.; Bannerman, S.; Jansen, J.; Crowe-White, K.M. Anti-Apoptotic Effects of Carotenoids in Neurodegeneration. Molecules 2020, 25, 3453. [Google Scholar] [CrossRef]
- Yi, F.; He, X.; Wang, D. Lycopene Protects Against MPP+-Induced Cytotoxicity by Maintaining Mitochondrial Function in SH-SY5Y Cells. Neurochem. Res. 2013, 38, 1747–1757. [Google Scholar] [CrossRef]
- Kaur, H.; Chauhan, S.; Sandhir, R. Protective Effect of Lycopene on Oxidative Stress and Cognitive Decline in Rotenone Induced Model of Parkinson’s Disease. Neurochem. Res. 2011, 36, 1435–1443. [Google Scholar] [CrossRef]
- Fakhri, S.; Aneva, I.Y.; Farzaei, M.H.; Sobarzo-Sánchez, E. The Neuroprotective Effects of Astaxanthin: Therapeutic Targets and Clinical Perspective. Molecules 2019, 24, 2640. [Google Scholar] [CrossRef] [Green Version]
- Wang, C.-C.; Shi, H.-H.; Xu, J.; Yanagita, T.; Xue, C.-H.; Zhang, T.-T.; Wang, Y.-M. Docosahexaenoic acid-acylated astaxanthin ester exhibits superior performance over non-esterified astaxanthin in preventing behav-ioral deficits coupled with apoptosis in MPTP-induced mice with Parkinson’s disease. Food Funct. 2020, 11, 8038–8050. [Google Scholar] [CrossRef]
- Meeran, M.F.N.; Goyal, S.N.; Suchal, K.; Sharma, C.; Patil, C.; Ojha, S.K. Pharmacological Properties, Molecular Mechanisms, and Pharmaceutical Development of Asiatic Acid: A Pentacyclic Triterpenoid of Therapeutic Promise. Front. Pharmacol. 2018, 9, 892. [Google Scholar] [CrossRef]
- Ding, H.; Xiong, Y.; Sun, J.; Chen, C.; Gao, J.; Xu, H. Asiatic Acid Prevents Oxidative Stress and Apoptosis by Inhibiting the Translocation of α-Synuclein into Mitochondria. Front. Neurosci. 2018, 12, 431. [Google Scholar] [CrossRef]
- Ahmed, S.; Kwatra, M.; Panda, S.R.; Murty, U.; Naidu, V. Andrographolide suppresses NLRP3 inflammasome activation in microglia through induction of parkin-mediated mitophagy in in-vitro and in-vivo models of Parkinson disease. Brain Behav. Immun. 2020, 91, 142–158. [Google Scholar] [CrossRef]
- Geng, J.; Liu, W.; Gao, J.; Jiang, C.; Fan, T.; Sun, Y.; Qin, Z.; Xu, Q.; Guo, W.; Gao, J. Andrographolide alleviates Parkinsonism in MPTP-PD mice via targeting mitochondrial fission mediated by dynamin-related protein. Br. J. Pharmacol. 2019, 176, 4574–4591. [Google Scholar] [CrossRef]
- Lin, C.-Y.; Chen, W.-J.; Fu, R.-H.; Tsai, C.-W. Upregulation of OPA1 by carnosic acid is mediated through induction of IKKγ ubiquitination by parkin and protects against neurotoxicity. Food Chem. Toxicol. 2019, 136, 110942. [Google Scholar] [CrossRef]
- De Oliveira, M.R.; Ferreira, G.C.; Schuck, P.F. Protective effect of carnosic acid against paraquat-induced redox impairment and mitochondrial dysfunction in SH-SY5Y cells: Role for PI3K/Akt/Nrf2 pathway. Toxicol. Vitr. 2016, 32, 41–54. [Google Scholar] [CrossRef]
- Lin, C.-Y.; Tsai, C.-W. PINK1/parkin-mediated mitophagy pathway is related to neuroprotection by carnosic acid in SH-SY5Y cells. Food Chem. Toxicol. 2019, 125, 430–437. [Google Scholar] [CrossRef]
- Yarmohammadi, F.; Hayes, A.W.; Najafi, N.; Karimi, G. The protective effect of natural compounds against rotenone-induced neurotoxicity. J. Biochem. Mol. Toxicol. 2020, 34. [Google Scholar] [CrossRef]
- Zhao, Q.; Ye, J.; Wei, N.; Fong, C.; Dong, X. Protection against MPP+-induced neurotoxicity in SH-SY5Y cells by tormentic acid via the activation of PI3-K/Akt/GSK3β pathway. Neurochem. Int. 2016, 97, 117–123. [Google Scholar] [CrossRef]
- Bhattamisra, S.K.; Yap, K.H.; Rao, V.; Choudhury, H. Multiple Biological Effects of an Iridoid Glucoside, Catalpol, and Its Underlying Molecular Mechanisms. Biomolecules 2019, 10, 32. [Google Scholar] [CrossRef] [Green Version]
- Dinda, B.; Dinda, M.; Kulsi, G.; Chakraborty, A.; Dinda, S. Therapeutic potentials of plant iridoids in Alzheimer’s and Parkinson’s diseases: A review. Eur. J. Med. Chem. 2019, 169, 185–199. [Google Scholar] [CrossRef]
- Song, L.; Xu, M.-B.; Zhou, X.-L.; Zhang, D.-P.; Zhang, S.-L.; Zheng, G.-Q. A Preclinical Systematic Review of Ginsenoside-Rg1 in Experimental Parkinson’s Disease. Oxidative Med. Cell. Longev. 2017, 2017, 1–14. [Google Scholar] [CrossRef]
- Wang, J.; Song, Y.; Gao, M.; Bai, X.; Chen, Z. Neuroprotective Effect of Several Phytochemicals and Its Potential Application in the Prevention of Neurodegenerative Diseases. Geriatrics 2016, 1, 29. [Google Scholar] [CrossRef]
- Abbaszadeh, F.; Fakhri, S.; Khan, H. Targeting apoptosis and autophagy following spinal cord injury: Therapeutic approaches to polyphenols and candidate phytochemicals. Pharmacol. Res. 2020, 160, 105069. [Google Scholar] [CrossRef]
- Zhou, T.; Zu, G.; Zhang, X.; Wang, X.; Li, S.; Gong, X.; Liang, Z.; Zhao, J. Neuroprotective effects of ginsenoside Rg1 through the Wnt/β-catenin signaling pathway in both in vivo and in vitro models of Parkinson’s disease. Neuropharmacology 2016, 101, 480–489. [Google Scholar] [CrossRef]
- González-Burgos, E.; Fernandez-Moriano, C.; Gómez-Serranillos, M.P. Potential Neuroprotective Activity of Ginseng in Parkinson’s Disease: A Review. J. Neuroimmune Pharmacol. 2014, 10, 14–29. [Google Scholar] [CrossRef]
- Hwang, C.K.; Chun, H.S. Isoliquiritigenin Isolated from LicoriceGlycyrrhiza uralensisPrevents 6-Hydroxydopamine-Induced Apoptosis in Dopaminergic Neurons. Biosci. Biotechnol. Biochem. 2012, 76, 536–543. [Google Scholar] [CrossRef] [Green Version]
- Javed, H.; Meeran, M.F.N.; Azimullah, S.; Adem, A.; Sadek, B.; Ojha, S.K. Plant Extracts and Phytochemicals Targeting α-Synuclein Aggregation in Parkinson’s Disease Models. Front. Pharmacol. 2019, 9, 1555. [Google Scholar] [CrossRef] [Green Version]
- Hatziagapiou, K.; Kakouri, E.; Lambrou, G.; Bethanis, K.; Tarantilis, P. Antioxidant Properties of Crocus Sativus L. and Its Constituents and Relevance to Neurodegenerative Diseases; Focus on Alzheimer’s and Parkinson’s Disease. Curr. Neuropharmacol. 2019, 17, 377–402. [Google Scholar] [CrossRef]
- Viswanathan, G.; Baby, S. Insights into the Molecular Aspects of Neuroprotective Bacoside A and Bacopaside I. Curr. Neuropharmacol. 2019, 17, 438–446. [Google Scholar] [CrossRef]
- Limanaqi, F.; Biagioni, F.; Busceti, C.L.; Ryskalin, L.; Polzella, M.; Frati, A.; Fornai, F. Phytochemicals Bridging Autophagy Induction and Alpha-Synuclein Degradation in Parkinsonism. Int. J. Mol. Sci. 2019, 20, 3274. [Google Scholar] [CrossRef] [Green Version]
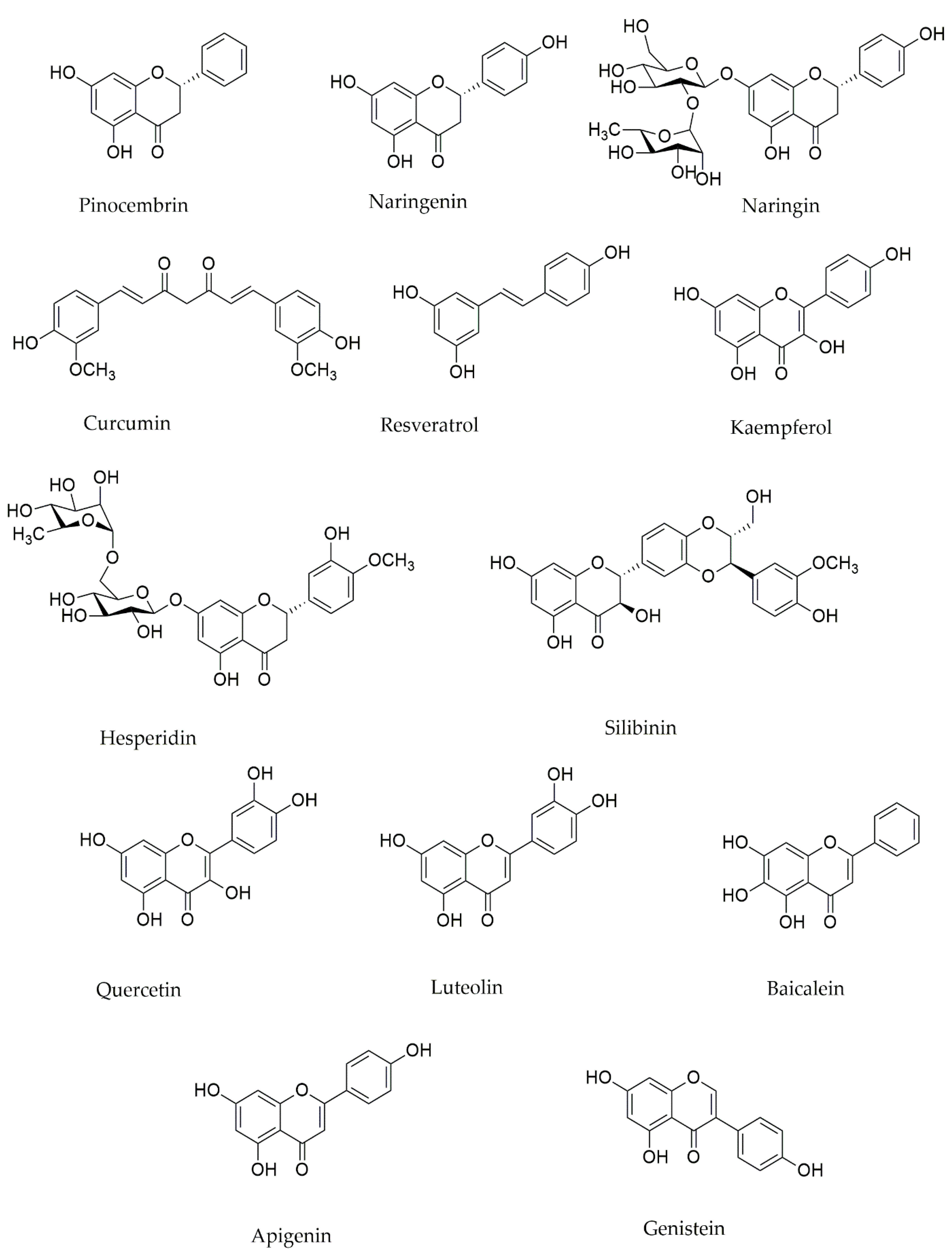
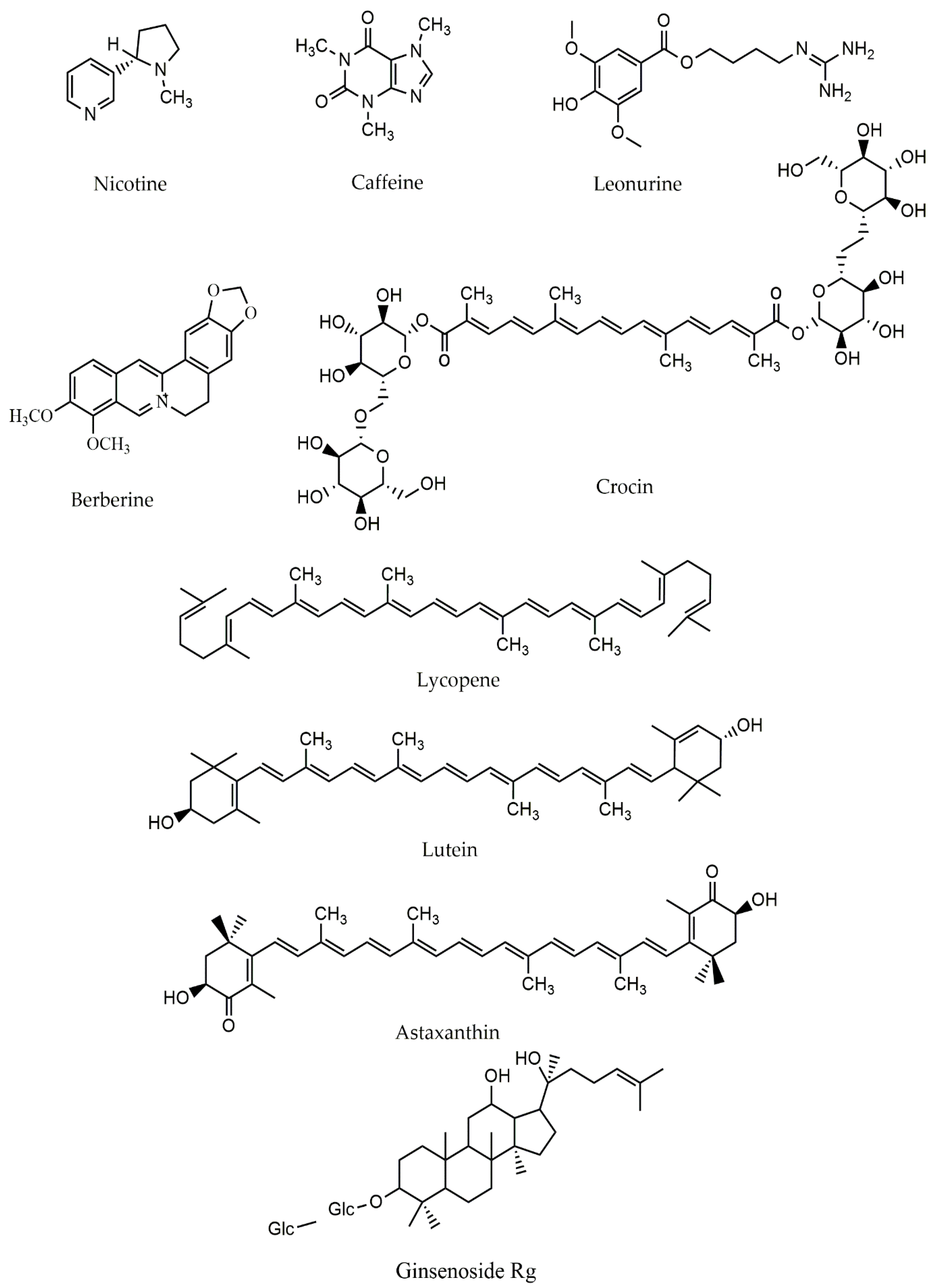
| Compound(s) | Class | In Vivo/In Vitro Models | Mechanisms and Outcomes | Reference |
|---|---|---|---|---|
| Pinocembrin | Flavonoid | In vitro: SH-SY5Y cells | Blocking ERK1/2 or silencing of Nrf2 | [58] |
| Naringin | In vivo: adult male Wistar albino rats | ↓Rotenone-induced dopaminergic toxicity, ↓mitochondrial function, stability, and bioenergetics in the animals’ SNpc via a Nrf2-mediated path | [54] | |
| Naringenin | In vivo/ In vitro: brains’s neurons of Sprague Dawley rats | ↑MMP, ↓ROS via affecting the Nrf2/ARE pathway | [50] | |
| In vitro: SH-SY5Y cells | ↓Mitochondria-related bioenergetics and redox dysfunctions via the Nrf2/GSH pathways | [53] | ||
| Hesperidin | In vivo: Mice | Attenuation of mitochondrial complex I, IV, V activity | [62] | |
| Isoliquiritigenin | In vitro: dopaminergic neuronal SN4741 cells | ↓Production of ROS as well as the dissipation of MMP and the presence of cytochrome c in the cytoplasm | [53] | |
| In vitro: dopaminergic neuronal SN4741 cells | ↓ROS, ↓NO, ↓JNK and p38 MAPK, ↑Bcl-2/Bax ratio, ↑MMP, ↑BDNF, ↓ cytochrome c, ↓caspase 3 and modulating PI3K/Akt | [101] | ||
| Luteolin | In vitro: SH-SY5Y cells | Preventive effect on ROS production and keep the activity of mitochondria in a normal way | [46,60] | |
| Baicalein | Flavone | In vivo/ in vitro: mice/ rat/ SH-SY5Y cells | ↓Mitochondrial malfunction in 6-OHDA-induced | [36] |
| In vitro: PC12 cells | Modulating ROS production, ↓rotenone-induced apoptotic in PC12 cells via ameliorating the mitochondrial dysfunction | [38] | ||
| Apigenin | In vivo: Rat | ↑Performance of mitochondrial enzyme | [102] | |
| Chrysoeriol | In vitro: SH-SY5Y cells | ↓Toxicity of MPP+—induced via PI3K/Akt | [22] | |
| Puerarin | Isoflavone | In vitro: PC12 neuronal cells | ↓Toxicity of MPP+—induced | [36] |
| In vitro: PC12 cells and primary rat midbrain neurons | Regulation of NO-mediated mitochondrial dysfunction | [39] | ||
| In vitro: SH-SY5Y cells | ↓Caspase-3 in MPP+-induced through modulating the activity of the ubiquitin proteasome system | [40] | ||
| Genistein | In vivo: ovariectomized rats | ↓Activity of ROS-induced NF-κB/ recovered the production of Bcl-2 mRNA | [18] | |
| Hyperoside | Flavonol glycoside | In vitro: PC12 neuronal cells | ↓Mitochondrial apoptotic pathway | [102] |
| In vitro: PC12 neuronal cells | ↓Mitochondrial apoptotic signaling | [48] | ||
| Schisandrin | Lignan | In vivo: Rat cortical cells | ↓Ca2+, ↓ROS, ↓cytochrome C | [25] |
| Ellagic acid | Phenolic acid | In vitro: Rat cortical neurons | Modulating ROS production, ↑Bcl-2/Bax | [32] |
| Ferulic acid | In vivo: Mice | Modulating ROS production by blocking p38MAPK,caspase-3, and COX-2, ↓Bax/Bcl2 | [32] | |
| Protocatechuic aldehyde | In vivo/ In vitro: Male C57BL/6 mice/ SH-SY5Y cells | ↓ROS, Modulating complex I’s activity in PLK2/p-GSK-3β/Nrf2 pathway | [15] | |
| Caffeic acid | In vitro: SH-SY5Y cells | ↓ROS production and keep the mitochondrial activity normal | [54] | |
| In vivo: male C57BL/6 mice | ↓Dopaminergic neurodegeneration and dopamine loss | [55] | ||
| Curcumin | Polyphenol | In vivo/ In vitro: mice / PC12 neuronal cell line | ↓MPP+ toxicity and ROS production | [36,42,59] |
| Tea polyphenols | In vitro: Human HEK293T and SH-SY5Y cells | Preserving DA neurons via inhibiting DA oxidation, conjugating with DA quinones (DAQ), scavenging ROS, and modulating Nrf2-Keap1 and PGC-1α | [57] | |
| Oleuropein | Secoiridoid | In vitro: PC12 neuronal cell line cells | ↓Superoxide anion, ↓complexes I, II, and IV activity, defensive function against MPP+ | [6] |
| Hydroxytyrosol | Phenolic compound | In vitro: dopaminergic SH-SY5Y cells | ↓Complexes I, II, and IV activity, defensive function against MPP+ | [6] |
| Quercetin | Flavonoid | In vivo/ In vitro: aged rats/ mice | ↓Apoptosis by downregulation of PI3K/Akt pathway, ↓mitochondrial fission | [42] |
| In vitro: SH-SY5Y cells | ↓Mitochondrial damage, ↑tyrosine hydroxylase, and mitochondrial controlling proteins | [43] | ||
| In vitro: PC12 cells | ↑Mitochondrial quality control, ↓ oxidative stress, ↑mitophagy markers | [44] | ||
| In vivo: adult Sprague-Dawley rats | Repair mitochondrial electron transport abnormalities | [45] | ||
| Silibinin | Flavonolig-nan | In vivo: mice | Modulating MMP and mitochondrial activity | [47] |
| In vivo: | ↓Mitochondrial damage, and strengthening of the oxidative defense system | [48] | ||
| In vivo: Male C57B/6 mice | ↓Motor impairment and dopaminergic neuronal degeneration, ↑stability of MMP | [49] | ||
| Resveratrol | Stilbene | In vivo/ In vitro: aged rats/ mice | ↓Apoptosis by downregulation of PI3K/Akt pathway | [20] |
| In vitro: SH-SY5Y cells | ↑MMP, ↑mitochondrial dynamics, elongated fragmented mitochondria | [41] | ||
| Vanillin | Phenolic aldehyde | In vitro: SH-SY5Y | ↓ROS production | [54] |
| α-Mangostin | Phenolic xanthone | In vitro: SH-SY5Y | Recovered mitochondrial membrane potential and cellular ATP | [102] |
| Nicotine | Alkaloid | In vivo/ In vitro: rats | ↓complex I activity, ↓ROS, Attenuating mitochondrial apoptosis pathway | [32,65] |
| In vitro: SH-SY5Y cells | ↓MPP+ and Ca2+-induced mitochondrial high amplitude swelling, ↓cytochrome c release from intact mitochondria | [66] | ||
| In vitro: cultured hippocampal neurons | Mediating mitochondrial dynamics, ↑ IP3 receptor clustering, modulating mitochondria-endoplasmic reticulum communications | [67] | ||
| In vivo/ In vitro: rats | Maintain mitochondrial function | [68] | ||
| In vivo/in vitro: C57BL/6 mouse | ↓H2O2-induced astrocyte apoptosis through the mitochondrial route and α7-nAChRs | [69] | ||
| Caffeine | In vivo/ In vitro: mice/ dopaminergic neurons | ↓ROS production, ↑mitochondrial formation by stimulating Nrf2-keap1 and PGC-1α pathway | [70] | |
| Leonurine | In vivo: mice | Modulating mitochondrial activity | [78] | |
| Berberine | In vitro: SH-SY5Y cells/ mice | ↓6-OHDA-induced cell death, ↓MPTP-induced PD-like behavior, ↓dopaminergic neuron loss | [72] | |
| Embelin | In vitro: Rat dopaminergic cell line, N27 | ↑pAMPK, ↑SIRT1, ↑PGC-1α, ↑mitochondrial biogenesis | [75] | |
| Isorhynchophylline | In vitro: PC12 cells | ↓MPP+-induced apoptotic cell death, ↓endoplasmic reticulum stress responses | [77] | |
| Lycopene | Carotenoid | In vivo: small mammals | Improving mitochondrial structural membrane ability, ↓Bax | [32,80] |
| In vitro: SH-SY5Y cells | ↓MPP+-induced mitochondrial ROS generation, ↓MPP+-induced opening of the mitochondrial permeability transition pore | [81] | ||
| In vivo: Adult male Wistar rats | ↓cytochrome c from mitochondria | [82] | ||
| Crocin | In vivo: an idiopathic Drosophila | ↓complex I_III activity | [103] | |
| Astaxanthin | In vivo/ In vitro: Rat/ SH-SY5Y | ↓Production of ROS | [83] | |
| Docosahexaenoic acid-acylated astaxanthin ester | Regulating dopaminergic neuron death in the brain through the mitochondria-mediated route and JNK and p38 MAPK pathways. | [84] | ||
| Asiatic acid | Pentacyclic triterpenoid | In vitro: SH-SY5Y cells/ mice | ↑Bcl-xL, ↓Bax, ↓H2O2 and rotenone adverse effects | [85] |
| In vitro: SH-SY5Y cells | ↓ROS, ↓cytochrome c, ↑MMP, preserving membrane integrity and ATP generation | [86] | ||
| Catalpol | Iridoid glucoside | In vivo: mouse | ↓Mitochondria malfunction via decreasing ATP | [94,95] |
| Ginsenosides-Rg1 | Triterpenoid | In vivo: mice | ↓Oxidative stress | [96,97] |
| Bacosides and Bacopasides | Triterpenoid saponin | in vivo/ in vitro: Drosophila /SH-SY5Y cells | ↓Mitochondrial malfunction and oxidative stress, Modulating complex I activity | [104,105] |
| Andrografolide | Diterpenoid lactone | In vivo/ In vitro: mice brain, N9 mouse microglia (RRID CVCL_0452) cell line | ↓ROS, balancing the level of ATP, ↑deletion of depolarized mitochondria via parkin dependent pathway | [87] |
| Alleviating behavioral impairments, ↓dopaminergic neuron loss and preventing excessive mitochondrial fission | [88] | |||
| Carnosic acid | Diterpene | In vitro: Human SH-SY5Y cells | ↑Fusion protein in mitochondria, ↓fission protein activity, ↑OPA1 protein production by parkin in IKK/NF-κB pathway | [89] |
| In vitro: SH-SY5Y cells | ↓ROS, ↓RNS, ↑Nrf2 via modulating the PI3K/Akt pathway, ↑GSH | [90] | ||
| In vitro: SH-SY5Y cells | ↑VDAC1, ↓cytosolic cytochrome c, ↑ PINK1/parkin-mediated mitophagy | [91] | ||
| Tormentic acid | Triterpene | In vitro: SH-SY5Y cells | ↓ROS, ↓Bax/Bcl-2 ratio through stimulation of the PI3K/Akt/GSK-3β signaling pathway | [93] |
| Boswellic acids | Pentacyclic triterpene | In vivo: Rats | ↓Rotenone-induced mitochondrial malfunction by inhibiting the IL-6/STAT3/NF-κB signaling pathways | [92] |
| Ginsenosides-Rg | Ginsenoside | In vivo: SN of MPTP-treated mice | Reducing iron-staining cells | [96] |
| Ginsenoside Rb1 | In vivo: mice primary cultured dopaminergic neuron | Recovering MMP and lessening Ca2+ over-influx into the mitochondria | [100] | |
| Ginsenoside Re | In vitro: PINK1-null dopaminergic cell line | Repair and offset particular mitochondrial complex IV deficiencies | ||
| Notoginsenoside R2 | In vitro: SH-SY5Y cells | ↓Mitochondrial death via the MEK1/2–ERK1/2 pathways | ||
| P-ginseng | In vitro: SH-SY5Y cells | ↓ROS excessive synthesis and inhibiting the mitochondria-dependent apoptotic pathway |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fakhri, S.; Abdian, S.; Zarneshan, S.N.; Akkol, E.K.; Farzaei, M.H.; Sobarzo-Sánchez, E. Targeting Mitochondria by Plant Secondary Metabolites: A Promising Strategy in Combating Parkinson’s Disease. Int. J. Mol. Sci. 2021, 22, 12570. https://doi.org/10.3390/ijms222212570
Fakhri S, Abdian S, Zarneshan SN, Akkol EK, Farzaei MH, Sobarzo-Sánchez E. Targeting Mitochondria by Plant Secondary Metabolites: A Promising Strategy in Combating Parkinson’s Disease. International Journal of Molecular Sciences. 2021; 22(22):12570. https://doi.org/10.3390/ijms222212570
Chicago/Turabian StyleFakhri, Sajad, Sadaf Abdian, Seyede Nazanin Zarneshan, Esra Küpeli Akkol, Mohammad Hosein Farzaei, and Eduardo Sobarzo-Sánchez. 2021. "Targeting Mitochondria by Plant Secondary Metabolites: A Promising Strategy in Combating Parkinson’s Disease" International Journal of Molecular Sciences 22, no. 22: 12570. https://doi.org/10.3390/ijms222212570
APA StyleFakhri, S., Abdian, S., Zarneshan, S. N., Akkol, E. K., Farzaei, M. H., & Sobarzo-Sánchez, E. (2021). Targeting Mitochondria by Plant Secondary Metabolites: A Promising Strategy in Combating Parkinson’s Disease. International Journal of Molecular Sciences, 22(22), 12570. https://doi.org/10.3390/ijms222212570








