Oxidative Stress and Respiratory Diseases in Preterm Newborns
Abstract
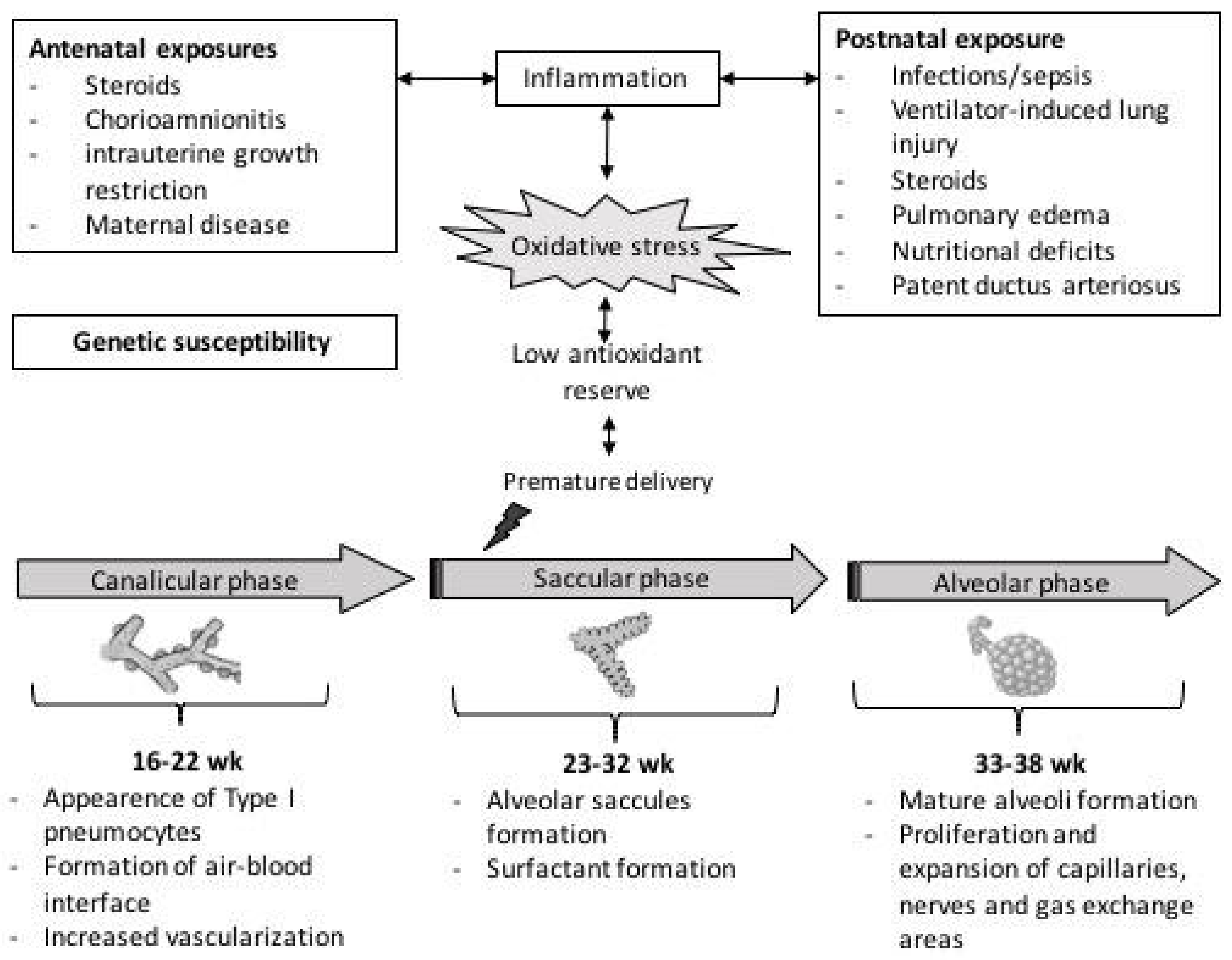
:1. Introduction
2. Oxidative Stress in Perinatal Period
3. Oxidative Stress and Respiratory Distress Syndrome
4. Oxidative Stress and Chronic Lung Disease
5. Oxidative Stress and Persistent Pulmonary Hypertension of the Newborn
6. Oxidative Stress and Ventilation-Induced Lung Injury
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Joshi, S.; Kotecha, S. Lung growth and development. Early Hum. Dev. 2007, 83, 789–794. [Google Scholar] [CrossRef] [PubMed]
- Gitto, E.; Reiter, R.J.; Karbownik, M.; Tan, D.X.; Gitto, P.; Barberi, S.; Barberi, I. Causes of oxidative stress in the pre- and perinatal period. Biol. Neonate 2002, 81, 146–157. [Google Scholar] [CrossRef] [PubMed]
- Check, J.; Gotteiner, N.; Liu, X.; Su, E.; Porta, N.; Steinhorn, R.; Mestan, K.K. Fetal growth restriction and pulmonary hypertension in premature infants with bronchopulmonary dysplasia. J. Perinatol. 2013, 33, 553–557. [Google Scholar] [CrossRef]
- Longini, M.; Perrone, S.; Kenanidis, A.; Vezzosi, P.; Marzocchi, B.; Petraglia, F.; Centini, G.; Buonocore, G. Isoprostanes in amniotic fluid: A predictive marker for fetal growth restriction in pregnancy. Free Radic. Biol. Med. 2005, 38, 1537–1541. [Google Scholar] [CrossRef] [PubMed]
- Rashid, C.S.; Bansal, A.; Simmons, R.A. Oxidative Stress, Intrauterine Growth Restriction, and Developmental Programming of Type 2 Diabetes. Physiology 2018, 33, 348–359. [Google Scholar] [CrossRef] [Green Version]
- Arguëlles, S.; Markado, M.J.; Ayala, A.; Machado, A.; Hervías, B. Correlation between circulating biomarkers of oxidative stress of maternal and umbilical cord blood at birth. Free Radic. Res. 2006, 40, 565–570. [Google Scholar] [CrossRef]
- Lee, J.W.; Davis, J.M. Future applications of antioxidants in premature infants. Curr. Opin. Pediatrics 2011, 23, 161–166. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Perrone, S.; Santacroce, A.; Picardi, A.; Buonocore, G. Fetal programming and early identification of newborns at high risk of free radical-mediated diseases. World J. Clin. Pediatrics 2016, 5, 172–181. [Google Scholar] [CrossRef] [PubMed]
- Aouache, R.; Biquard, L.; Vaiman, D.; Miralles, F. Oxidative Stress in Preeclampsia and Placental Diseases. Int. J. Mol. Sci. 2018, 19, 1496. [Google Scholar] [CrossRef] [Green Version]
- Perez, M.; Robbins, M.E.; Revhaug, C.; Saugstad, O.D. Oxygen radical disease in the newborn, revisited: Oxidative stress and disease in the newborn period. Free Radic. Biol. Med. 2019, 142, 61–72. [Google Scholar] [CrossRef] [PubMed]
- Negro, S.; Boutsikou, T.; Briana, D.D.; Tataranno, M.L.; Longini, M.; Proietti, F.; Bazzini, F.; Dani, C.; Malamitsi-Puchner, A.; Buonocore, G.; et al. Maternal obesity and perinatal oxidative stress: The strength of the association. J. Biol. Regul. Homeost. Agents 2017, 31, 221–227. [Google Scholar]
- Myatt, L.; Maloyan, A. Obesity and Placental Function. Semin. Reprod. Med. 2016, 34, 42–49. [Google Scholar] [PubMed] [Green Version]
- Adams Waldorf, K.M.; McAdams, R.M. Influence of infection during pregnancy on fetal development. Reproduction 2013, 146, R151–R162. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Saker, M.; Soulimane Mokhtari, N.; Merzouk, S.A.; Merzouk, H.; Belarbi, B.; Narce, M. Oxidant and antioxidant status in mothers and their newborns according to birthweight. Eur. J. Obstet. Gynecol. Reprod. Biol. 2008, 141, 95–99. [Google Scholar] [CrossRef] [PubMed]
- Matyas, M.; Hasmasanu, M.G.; Zaharie, G. Antioxidant Capacity of Preterm Neonates Assessed by Hydrogen Donor Value. Medicina 2019, 55, 720. [Google Scholar] [CrossRef] [PubMed]
- Laurie, S.; Mataz, Z.; Boaz, M.; Fux, A.; Golan, A.; Sadan, O. Different degrees of fetal oxidative stress in elective and emergent caesarean section. Neonatology 2007, 92, 111–115. [Google Scholar] [CrossRef] [PubMed]
- Shoji, H.; Koletzko, B. Oxidative stress and antioxidant protection in the perinatal period. Curr. Opin. Clin. Nutr. Metab. Care 2007, 10, 324–328. [Google Scholar] [CrossRef]
- Marseglia, L.; D’Angelo, G.; Granese, R.; Falsaperla, R.; Reiter, R.J.; Corsello, G.; Gitto, E. Role of oxidative stress in neonatal respiratory distress syndrome. Free Radic. Biol. Med. 2019, 142, 132–137. [Google Scholar] [CrossRef]
- Buonocore, G.; Perrone, S.; Tataranno, M.L. Oxygen toxicity: Chemistry and biology of reactive oxygen species. Semin. Fetal Neonatal Med. 2010, 15, 186–190. [Google Scholar] [CrossRef] [PubMed]
- Perrone, S.; Laschi, E.; Buonocore, G. Biomarkers of oxidative stress in the fetus and in the newborn. Free Radic. Biol. Med. 2019, 142, 23–31. [Google Scholar] [CrossRef]
- Dani, C.; Poggi, C. The role of genetic polymorphisms in antioxidant enzymes and potential antioxidant therapies in neonatal lung disease. Antioxid Redox Signal. 2014, 21, 1863–1880. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- D’Angelo, G.; Cannavò, L.; Reiter, R.J.; Gitto, E. Melatonin Administration from 2000 to 2020 to Human Newborns with Hypoxic-Ischemic Encephalopathy. Am. J. Perinatol. 2020. Epub ahead of print. [Google Scholar] [CrossRef]
- Aly, H.; Elmahdy, H.; El-Dib, M.; Rowisha, M.; Awny, M.; El-Gohary, T.; Elbatch, M.; Hamisa, M.; El-Mashad, A.R. Melatonin use for neuroprotection in perinatal asphyxia: A randomized controlled pilot study. J. Perinatol. 2015, 35, 186–191. [Google Scholar] [CrossRef]
- Hamid, E.; Ali, W.H.; Azmy, A.; Ahmed, H.H.; Sherif, L.S.; Saleh, M.T. Oxidative Stress and Anti-Oxidant Markers in Premature Infants with Respiratory Distress Syndrome. Open Access Maced. J. Med. Sci. 2019, 7, 2858–2863. [Google Scholar] [CrossRef] [Green Version]
- Kaltofen, T.; Haase, M.; Thome, U.H.; Laube, M. Male sex is associated with a reduced alveolar epithelial sodium transport. PLoS ONE 2015, 10, e0136178. [Google Scholar]
- Dani, C.; Corsini, L.; Longini, M.; Burchielli, S.; Dichiara, G.; Cantile Buonocore, G. Natural sufractant combined with superoxide dismutase and catalase decreases oxidative lung injury in the preterm lamb. Pediatr. Pulmonol. 2014, 49, 898–904. [Google Scholar] [CrossRef] [PubMed]
- Negi, R.; Pande, D.; Karki, K.; Kumar, A.; Khanna, R.S.; Khanna, H.D. A novel approach to study oxidative stress in neonatal respiratory distress syndrome. BBA Clin. 2014, 3, 65–69. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dizdar, E.A.; Uras, N.; Oguz, S.; Erdeve, O.; Sari, F.N.; Aydemir, C.; Dilmen, U. Total antioxidant capacity and total oxidant status after surfactant treatment in preterm infants with respiratory distress syndrome. Ann. Clin. Biochem. 2011, 48, 462–467. [Google Scholar] [CrossRef]
- Kellner, M.; Noonepalle, S.; Lu, Q.; Srivastava, A.; Zemskov, E.; Black, S.M. ROS Signaling in the Pathogenesis of Acute Lung Injury (ALI) and Acute Respiratory Distress Syndrome (ARDS). Adv. Exp. Med. Biol. 2017, 967, 105–137. [Google Scholar]
- Gitto, E.; Pellegrino, S.; D’Arrigo, S.; Barberi, I.; Reiter, R.J. Oxidative stress in resuscitation and in ventilation of newborns. Eur. Respir. J. 2009, 34, 1461–1469. [Google Scholar] [CrossRef]
- Marseglia, L.; D’Angelo, G.; Manti, S.; Arrigo, T.; Barberi, I.; Reiter, R.J.; Gitto, E. Oxidative stress-mediated aging during the fetal and perinatal periods. Oxid. Med. Cell. Longev. 2014, 104, 358375. [Google Scholar] [CrossRef] [Green Version]
- Nascimben, F.; Peri, F.M.; Impellizzeri, P.; Chimenz, R.; Cannavò, L.; Pellegrino, D.; Ceravolo, G.; Calabrò, M.P.; Gitto, E.; Romeo, C. Role of oxidative stress in the pathogenesis of congenital cardiopathies. J. Biol. Regul. Homeost. Agents 2020, 34, 85–90. [Google Scholar] [PubMed]
- Boda, D.; Nemeth, I.; Pinter, S. Surface tension, glutathione content and redox ratio of the tracheal aspirate fluid of premature infants with RDS. Biol. Neonate 1998, 74, 281–288. [Google Scholar] [CrossRef] [PubMed]
- Chang, L.Y.; Subramaniam, M.; Yoder, B.A.; Day, B.J.; Ellison, M.C.; Sunday, M.E.; Crapo, J.D. A catalytic antioxidant attenuates alveolar structural remodeling in bronchopulmonary dysplasia. Am. J. Respir. Crit Care Med. 2003, 167, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Fajardo, C.; Levin, D.; Garcia, M.; Abrams, D.; Adamson, I. Surfactant versus saline as a vehicle for corticosteroid delivery to the lungs of ventilated rabbits. Pediatr. Res. 1998, 43, 542–547. [Google Scholar] [CrossRef] [Green Version]
- Kothe, T.B.; Royse, E.; Kemp, M.W.; Schmidt, A.; Salomone, F.; Saito, M.; Usuda, H.; Watanabe, S.; Musk, G.C.; Jobe, A.H.; et al. Effects of budesonide and surfactant in preterm fetal sheep. Am. J. Physiol. Lung Cell. Mol. Physiol. 2018, 315, 193–202. [Google Scholar] [CrossRef]
- Yeh, T.F.; Lin, H.C.; Chang, C.H.; Wu, T.S.; Su, B.H.; Li, T.C.; Pyati, S.; Tsai, C.H. Early intratracheal instillation of budesonide using surfactant as a vehicle to prevent chronic lung disease in preterm infants: A pilot study. Pediatrics 2008, 121, 1310–1318. [Google Scholar] [CrossRef]
- Yeh, T.F.; Chen, C.M.; Wu, S.Y.; Husan, Z.; Li, T.C.; Hsieh, W.S.; Tsai, C.H.; Lin, H.C. Intratracheal Administration of Budesonide/Surfactant to Prevent Bronchopulmonary Dysplasia. Am. J. Respir. Crit. Care Med. 2016, 193, 86–95. [Google Scholar] [CrossRef] [PubMed]
- Schittny, J.C. Development of the lung. Cell Tissue Res. 2017, 367, 427–444. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dauger, S.; Ferkdadji, L.; Saumon, G.; Vardon, G.; Peuchmaur, M.; Gaultier, C.; Gallego, J. Neonatal exposure to 65% oxygen durably impairs lung architecture and breathing pattern in adult mice. Chest 2003, 123, 530–538. [Google Scholar] [CrossRef] [Green Version]
- Principi, N.; Di Pietro, G.M.; Esposito, S. Bronchopulmonary dysplasia: Clinical aspects and preventive and therapeutic strategies. J. Transl. Med. 2018, 16, 36. [Google Scholar] [CrossRef] [Green Version]
- Wang, J.; Dong, W. Oxidative stress and bronchopulmonary dysplasia. Gene 2018, 678, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Rehan, V.K.; Torday, J.S. The lung alveolar lipofibroblast: An evolutionary strategy against neonatal hyperoxic lung injury. Antioxid. Redox Signal. 2014, 21, 1893–1904. [Google Scholar] [CrossRef] [Green Version]
- West, J.B. Respiratory Physiology: The Essentials; Lippincott, Williams and Wilkins: Philadelphia, PA, USA, 2012. [Google Scholar]
- Torday, J.S.; Rehan, V.K. Cell-cell signaling drives the evolution of complex traits: Introduction-lung evo-devo. Integr. Comp. Biol. 2009, 49, 142–154. [Google Scholar] [CrossRef] [Green Version]
- Hu, E.; Tontonoz, P.; Spiegelman, B.M. Transdifferentiation of myoblasts by the adipogenic transcription factors PPAR gamma and C/EBP alpha. Proc. Natl. Acad. Sci. USA 1995, 92, 9856–9860. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rehan, V.K.; Torday, J.S. PPARγ Signaling Mediates the Evolution, Development, Homeostasis, and Repair of the Lung. PPAR Res. 2012, 71, 289867. [Google Scholar]
- Muramatsu, Y.; Ito, M.; Oshima, T.; Kojima, S.; Ohno, K. Hydrogen-rich water ameliorates bronchopulmonary dysplasia (BPD) in newborn rats. Pediatr. Pulmonol. 2016, 51, 928–935. [Google Scholar] [CrossRef] [PubMed]
- Mariduena, J.; Ramagopal, M.; Hiatt, M.; Chandra, S.; Laumbach, R.; Hegyi, T. Vascular endothelial growth factor levels and bronchopulmonary dysplasia in preterm infants. J. Matern. Fetal Neonatal Med. 2020, 4, 1–6. [Google Scholar] [CrossRef]
- Jiménez, J.; Lesage, F.; Richter, J.; Nagatomo, T.; Salaets, T.; Zia, S.; Mori Da Cunha, M.G.; Vanoirbeek, J.; Deprest, J.A.; Toelen, J. Up regulation of vascular endothelial growth factor in amniotic fluid stem cells enhances their potential to attenuate lung injury in a preterm rabbit model of bronchopulmonary dysplasia. Neonatology 2018, 113, 275–285. [Google Scholar] [CrossRef] [PubMed]
- Gilfillan, M.; Bhandari, A.; Bhandari, V. Diagnosis and management of bronchopulmonary dysplasia. BMJ 2021, 375, n1974. [Google Scholar] [CrossRef]
- Doyle, L.W.; Cheong, J.L.; Ehrenkranz, R.A.; Halliday, H.L. Early (<8 days) systemic postnatal corticosteroids for prevention of bronchopulmonary dysplasia in preterm infants. Cochrane Database Syst. Rev. 2017, 10, CD001146. [Google Scholar] [PubMed]
- Shaffer, M.L.; Baud, O.; Lacaze-Masmonteil, T.; Peltoniemi, O.M.; Bonsante, F.; Watterberg, K.L. Effect of prophylaxis for early adrenal insufficiency using low-dose hydrocortisone in very preterm infants: An individual patient data meta-analysis. J. Pediatr. 2019, 207, 136–142.e5. [Google Scholar] [CrossRef] [Green Version]
- Héneau, A.; Guimiot, F.; Mohamed, D.; Rideau, B.N.A.; Alberti, C.; Baud, O.; PREMILOC Trial Study Group. Placental findings and effect of prophylactic hydrocortisone in extremely preterm infants. Pediatrics 2018, 141, e20171788. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Perrone, S.; Tataranno, M.L.; Buonocore, G. Oxidative stress and bronchopulmonary dysplasia. J. Clin. Neonatol. 2012, 1, 109–114. [Google Scholar] [PubMed] [Green Version]
- Hsiao, C.; Chang, J.; Tsao, L.; Yang, R.; Chen, H.; Lee, C.; Lin, C.Y.; Tsa, Y.G. Correlates of elevated interleukin-6 and 8-hydroxy-2′-deoxyguanosine levels in tracheal aspirates from very low birth weight infants who develop bronchopulmonary dysplasia. Pediatr. Neonatol. 2017, 58, 63–69. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Joung, K.E.; Kim, H.; Lee, J.; Shim, G.H.; Choi, C.W.; Kim, E.K.; Kim, B.I.; Choi, J.H. Correlation of urinary inflammatory and oxidative stress markers in very low birth weight infants with subsequent development of bronchopulmonary dysplasia. Free Radic. Res. 2011, 45, 1024–1032. [Google Scholar] [CrossRef]
- Ferrante, G.; Carota, G.; Li Volti, G.; Giuffrè, M. Biomarkers of Oxidative Stress for Neonatal Lung Disease. Front. Pediatrics 2021, 18, 618867. [Google Scholar] [CrossRef] [PubMed]
- Piersigilli, F.; Bhandari, V.J. Biomarkers in neonatology: The new “omics” of bronchopulmonary dysplasia. Matern. Fetal Neonatal Med. 2016, 29, 1758–1764. [Google Scholar] [CrossRef]
- Piersigilli, F.; Bhandari, V. Metabolomics of bronchopulmonary dysplasia. Clin. Chim. Acta 2020, 500, 109–114. [Google Scholar] [CrossRef]
- Capasso, L.; Vento, G.; Loddo, C.; Tirone, C.; Iavarone, F.; Raimondi, F.; Dani, C.; Fanos, V. Oxidative Stress and Bronchopulmonary Dysplasia: Evidences From Microbiomics, Metabolomics, and Proteomics. Front. Pediatr. 2019, 7, 30. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Steurer, M.A.; Jelliffe-Pawlowski, L.L.; Baer, R.J.; Partridge, J.C.; Rogers, E.E.; Keller, R.L. Persistent Pulmonary Hypertension of the Newborn in Late Preterm and Term Infants in California. Pediatrics 2017, 81, 139. [Google Scholar] [CrossRef] [Green Version]
- Arjaans, S.; Zwart, E.A.H.; Ploegstra, M.J.; Bos, A.F.; Kooi, E.M.W.; Hillege, H.L.; Berger, R.M.F. Identification of gaps in the current knowledge on pulmonary hypertension in extremely preterm infants: A systematic review and meta-analysis. Paediatr. Perinat. Epidemiol. 2018, 32, 258–267. [Google Scholar] [CrossRef]
- Wedgwood, S.; Warford, C.; Agvateesiri, S.C.; Thai, P.; Berkelhamer, S.K.; Perez, M.; Underwood, M.A.; Steinhorn, R.H. Postnatal growth restriction augments oxygen-induced pulmonary hypertension in a neonatal rat model of bronchopulmonary dysplasia. Pediatr. Res. 2016, 80, 894–902. [Google Scholar] [CrossRef] [PubMed]
- Chimenz, R.; Cannavò, L.; Gasbarro, A.; Nascimben, F.; Sestito, S.; Rizzuti, L.; Ceravolo, G.; Ceravolo, M.D.; Calabrò, M.P.; Romeo, C.; et al. PPHN and oxidative stress: A review of literature. J. Biol. Regul. Homeost. Agents 2020, 34, 79–83. [Google Scholar] [PubMed]
- La Frano, M.R.; Fahrmann, J.F.; Grapov, D.; Fiehn, O.; Pedersen, T.L.; Newman, J.W.; Underwood, M.A.; Steinhorn, R.H.; Wedgwood, S. Metabolic perturbations of postnatal growth restriction and hyperoxia-induced pulmonary hypertension in a bronchopulmonary dysplasia model. Metabolomics 2017, 4, 12. [Google Scholar] [CrossRef]
- Farrow, K.; Wedgwood, S.; Lee, K.; Czech, L.; Gugino, S.; Lakshminrusimha, S.; Schumacker, P.; Steinhorn, R. Mitochondrial oxidant stress increases PDE5 activity in persistent pulmonary hypertension of the newborn. Respir. Physiol. Neurobiol. 2010, 174, 272–281. [Google Scholar] [CrossRef] [Green Version]
- Hilgendorff, A.; Apitz, C.; Bonnet, D.; Hansmann, G. Pulmonary hypertension associated with acute or chronic lung diseases in the preterm and term neonate and infant. The European Paediatric pulmonary vascular disease network, endorsed by ISHLT DGPK. Heart 2016, 102 (Suppl. 2), ii49–ii56. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carnesecchi, S.; Deffert, C.; Pagano, A.; Garrido-Urbani, S.; Metrailler-Ruchonnet, I.; Schappi, M.; Donati, Y.; Matthay, M.A.; Krause, K.M.; Barazzone Argiroffo, C. NADPH oxidase-1 plays a crucial role in hyperoxia-induced acute lung injury in mice. Am. J. Respir. Crit. Care Med. 2009, 180, 972–981. [Google Scholar] [CrossRef] [PubMed]
- Farrow, K.N.; Groh, B.S.; Schumacker, P.T.; Lakshminrusimha, S.; Czech, L.; Gugino, S.F.; Russell, J.A.; Steinhorn, R.H. Hyperoxia increases phosphodiesterase 5 expression and activity in ovine fetal pulmonary artery smooth muscle cells. Circ. Res. 2008, 102, 226–233. [Google Scholar] [CrossRef] [Green Version]
- Sharma, V.; Berkelhamer, S.; Lakshminrusimha, S. Persistent pulmonary hypertension of the newborn. Matern Heal. Neonatol Perinatol. 2015, 1, 14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, K.J.; Berkelhamer, S.K.; Kim, G.A.; Taylor, J.M.; O’Shea, K.M.; Steinhorn, R.H.; Farrow, K.N. Disrupted pulmonary artery cGMP signaling in mice with hyperoxia-induced pulmonary hypertension. Am. J. Respir Cell Mol. Biol. 2014, 50, 369–378. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gomez-Arroyo, J.; Mizuno, S.; Szczepanek, K.; Van Tassell, B.; Natarajan, R.; dos Remedios, C.G.; Drake, J.I.; Farkas, L.; Kraskauskas, D.; Wijesinghe, D.S.; et al. Metabolic gene remodeling and mitochondrial dysfunction in failing right ventricular hypertrophy secondary to pulmonary arterial hypertension. Circ. Hear. Fail. 2013, 6, 136–144. [Google Scholar] [CrossRef] [Green Version]
- Hanouni, M.; Bernal, G.; McBride, S.; Narvaez, V.R.F.; Ibe, B.O. Hypoxia and hyperoxia potentiate PAF receptor-mediated effects in newborn ovine pulmonary arterial smooth muscle cells: Significance in oxygen therapy of PPHN. Physiol Rep. 2016, 4, e12840. [Google Scholar] [CrossRef] [PubMed]
- Roberts, K.; Stepanovich, G.; Bhatt-Mehta, V.; Donn, S.M. New Pharmacologic Approaches to Bronchopulmonary Dysplasia. J. Exp. Pharmacol. 2021, 13, 377–396. [Google Scholar] [CrossRef]
- Farrow, K.N.; Lakshminrusimha, S.; Reda, W.J.; Wedgwood, S.; Czech, L.; Gugino, S.F.; Davis, J.M.; Russell, J.A.; Steinhorn, R.H. Superoxide dismutase restores eNOS expression and function in resistance pulmonary arteries from neonatal lambs with persistent pulmonary hypertension. Am. J. Phys. Lung Cell. Mol. Physiol. 2008, 295, 979–987. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Farrow, K.N.; Lakshminrusimha, S.; Czech, L.; Groh, B.S.; Gugino, S.; Davis, J.M.; Russell, J.A.; Steinhorn, R.H. SOD and inhaled nitric oxide normalize phosphodiesterase 5 expression and activity in neonatal lambs with persistent pulmonary hypertension. Am. J. Physiol. Lung Cell. Mol. Physiol. 2010, 299, 109–116. [Google Scholar] [CrossRef] [Green Version]
- Martinho, S.; Adão, R.; Leite-Moreira, A.F.; Brás-Silva, C. Persistent pulmonary hypertension of the newborn: Pathophysiological mechanisms and novel therapeutic approaches. Front. Pediatr. 2020, 24, 342. [Google Scholar] [CrossRef] [PubMed]
- Niermeyer, S.; Kattwinkel, J.; Van Reempts, P.; Nadkarni, V.; Phillips, B.; Zideman, D.; Azzopardi, D.; Berg, R.; Boyle, D.; Boyle, R.; et al. International Guidelines for Neonatal Resuscitation: An excerpt from the Guidelines 2000 for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care: International Consensus on Science. Contributors and Reviewers for the Neonatal Resuscitation Guidelines. Pediatrics 2000, 106, 29. [Google Scholar]
- Rook, D.; Schierbeek, H.; Vento, M.; Vlaardingerbroek, H.; van der Eijk, A.C.; Longini, M.; Buonocore, G.; Escobar, J.; van Goudoever, J.V.; Vermeulen, M.J. Resuscitation of preterm infants with different inspired oxygen fractions. J. Pediatr. 2014, 164, 1322–1326. [Google Scholar] [CrossRef]
- Vento, M.; Moro, M.; Escrig, R.; Arruza, L.; Villar, G.; Izquierdo, I.; Roberts, L.; Arduini, A.; Escobar, J.J.; Sastre, J.; et al. Preterm resuscitation with low oxygen causes less oxidative stress, inflammation, and chronic lung disease. Pediatrics 2009, 124, 439–449. [Google Scholar] [CrossRef]
- Raby, Y.; Rabi, D.; Yee, W. Room air resuscitation of the depressed newborn: A systematic review and meta-analysis. Resuscitation 2007, 72, 353–363. [Google Scholar] [CrossRef] [PubMed]
- Bhandari, V. Molecular mechanisms of hyperoxia-induced acute lung injury. Front. Biosci. 2008, 13, 6653–6661. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carvalho, C.G.; Procianoy, R.S.; Neto, E.C.; Silveira, R.C. Preterm Neonates with Respiratory Distress Syndrome: Ventilator-Induced Lung Injury and Oxidative Stress. J. Immunol. Res. 2018, 23, 6963754. [Google Scholar] [CrossRef] [Green Version]
- Vogel, E.R.; Britt, R.D., Jr.; Trinidadetal, M.C. Perinataloxygen in the developing lung. Can. J. Physiol. Pharmacol. 2015, 93, 119–127. [Google Scholar] [CrossRef] [Green Version]
- Sweet, D.G.; Carnielli, V.; Greisen, G.; Hallman, M.; Ozek, E.; Te Pas, A.; Plavka, R.; Roehr, C.C.; Saugstad, O.D.; Simeoni, U.; et al. European consensus guidelines on the Management of Respiratory Distress Syndrome 2019 update. Neonatology 2019, 115, 432–450. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bloom, R.; Yost, C.C. A consideration of neonatal resuscitation. Pediatr. Clin. N. Am. 2004, 51, 669–684. [Google Scholar] [CrossRef]
- Bohrer, B.; Silveira, R.C.; Neto, E.C.; Procianoy, R.S. Mechanical ventilation of newborns infant changes in plasma pro- and anti-inflammatory cytokines. J. Pediatr. 2010, 156, 16–19. [Google Scholar] [CrossRef]
- Beresfordand, M.W.; Shaw, N.J. Detectable IL-8 and IL-10 in bronchoalveolar lavage fluid from preterm infants ventilated for respiratory distress syndrome. Pediatr. Res. 2002, 52, 973–978. [Google Scholar] [CrossRef]
- Jónsson, B.; Li, Y.H.; Noack, G.; Brauner, A.; Tullus, K. Downregulatory cytokines in tracheobronchial aspirate fluid from infants with chronic lung disease of prematurity. Acta Paediatr. 2000, 89, 1375–1380. [Google Scholar] [CrossRef] [PubMed]
- Aversa, S.; Marseglia, L.; Manti, S.; D’Angelo, G.; Cuppari, C.; David, A.; Chirico, G.; Gitto, E. Ventilation strategies for preventing oxidative stress-induced injury in preterm infants with respiratory disease: An update. Paediatr. Respir. Rev. 2016, 17, 71–79. [Google Scholar] [CrossRef]
- Cannavò, L.; Rulli, I.; Falsaperla, R.; Corsello, G.; Gitto, E. Ventilation, oxidative stress and risk of brain injury in preterm newborn. Ital. J. Pediatr. 2020, 46, 100. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cannavò, L.; Perrone, S.; Viola, V.; Marseglia, L.; Di Rosa, G.; Gitto, E. Oxidative Stress and Respiratory Diseases in Preterm Newborns. Int. J. Mol. Sci. 2021, 22, 12504. https://doi.org/10.3390/ijms222212504
Cannavò L, Perrone S, Viola V, Marseglia L, Di Rosa G, Gitto E. Oxidative Stress and Respiratory Diseases in Preterm Newborns. International Journal of Molecular Sciences. 2021; 22(22):12504. https://doi.org/10.3390/ijms222212504
Chicago/Turabian StyleCannavò, Laura, Serafina Perrone, Valeria Viola, Lucia Marseglia, Gabriella Di Rosa, and Eloisa Gitto. 2021. "Oxidative Stress and Respiratory Diseases in Preterm Newborns" International Journal of Molecular Sciences 22, no. 22: 12504. https://doi.org/10.3390/ijms222212504





