Assessment of the Effects of Dietary Vitamin D Levels on Olanzapine-Induced Metabolic Side Effects: Focus on the Endocannabinoidome-Gut Microbiome Axis
Abstract
:1. Introduction
2. Results
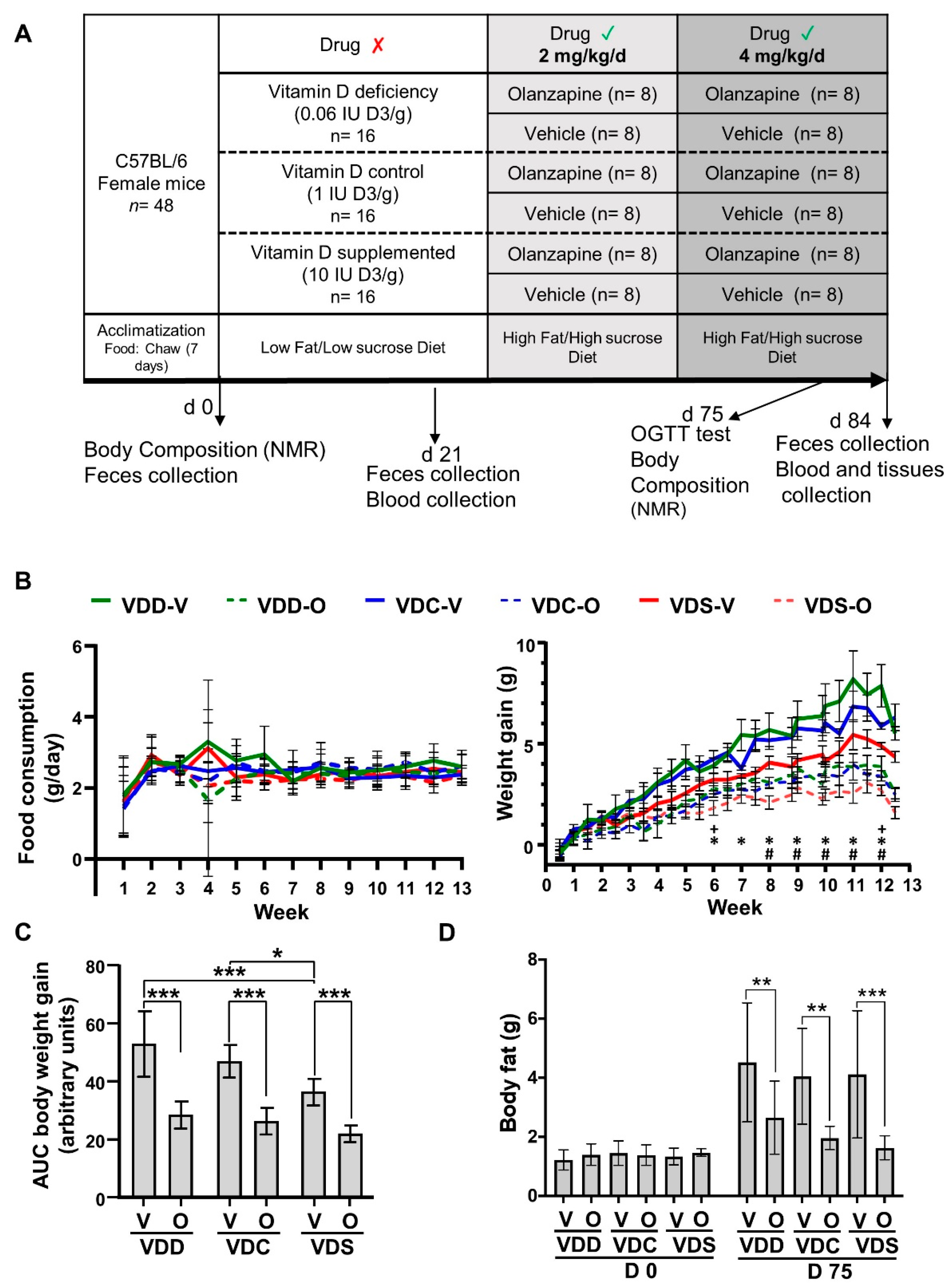
2.1. Olanzapine Reduces HFHS Diet-Mediated Weight Gain and Adiposity in Female Mice
2.2. Olanzapine Modifies Several Aspects of Adipose Tissue eCBome Signaling
2.3. Vitamin D Status and Olanzapine Effects on the Gut Microbiome of Mice on High-Fat, High-Sucrose Diets
3. Discussion
4. Materials and Methods
4.1. Animals and Housing
4.2. Drug Administration
4.3. Body Composition and Food Intake
4.4. Sample Collection
4.5. Oral Glucose Tolerance Test (OGTT)
4.6. RNA Isolation and qPCR-Based TaqMan Open Array
4.7. eCBome Lipid Mediator Analysis
4.8. 16S rRNA Gene Sequencing
4.9. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mackin, P.; Thomas, S.H.L. Atypical Antipsychotic Drugs. BMJ 2011, 342, d1126. [Google Scholar] [CrossRef] [PubMed]
- Seeman, P. Atypical Antipsychotics: Mechanism of Action. FOC 2004, 2, 48–58. [Google Scholar] [CrossRef] [Green Version]
- Tamminga, C.A. Treatment Mechanisms: Traditional and New Antipsychotic Drugs. Dialogues Clin. Neurosci. 2000, 2, 281–286. [Google Scholar] [PubMed]
- Nasrallah, H.A. Atypical Antipsychotic-Induced Metabolic Side Effects: Insights from Receptor-Binding Profiles. Mol. Psychiatry 2007, 13, 27. [Google Scholar] [CrossRef] [PubMed]
- Aucoin, M.; LaChance, L.; Cooley, K.; Kidd, S. Diet and Psychosis: A Scoping Review. Neuropsychobiology 2020, 79, 20–42. [Google Scholar] [CrossRef]
- Davey, K.J.; Cotter, P.D.; O’Sullivan, O.; Crispie, F.; Dinan, T.G.; Cryan, J.F.; O’Mahony, S.M. Antipsychotics and the Gut Microbiome: Olanzapine-Induced Metabolic Dysfunction Is Attenuated by Antibiotic Administration in the Rat. Transl. Psychiatry 2013, 3, e309. [Google Scholar] [CrossRef] [Green Version]
- Morgan, A.P.; Crowley, J.J.; Nonneman, R.J.; Quackenbush, C.R.; Miller, C.N.; Ryan, A.K.; Bogue, M.A.; Paredes, S.H.; Yourstone, S.; Carroll, I.M.; et al. The Antipsychotic Olanzapine Interacts with the Gut Microbiome to Cause Weight Gain in Mouse. PLoS ONE 2014, 9, e115225. [Google Scholar] [CrossRef] [PubMed]
- Dang, R.; Jiang, P.; Cai, H.; Li, H.; Guo, R.; Wu, Y.; Zhang, L.; Zhu, W.; He, X.; Liu, Y.; et al. Vitamin D Deficiency Exacerbates Atypical Antipsychotic-Induced Metabolic Side Effects in Rats: Involvement of the INSIG/SREBP Pathway. Eur. Neuropsychopharmacol. 2015, 25, 1239–1247. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.C.; Holick, M.F. Vitamin D Deficiency: A Worldwide Problem with Health Consequences. Am. J. Clin. Nutr. 2008, 87, 1080S–1086S. [Google Scholar] [CrossRef] [Green Version]
- Holick, M.F.; Binkley, N.C.; Bischoff-Ferrari, H.A.; Gordon, C.M.; Hanley, D.A.; Heaney, R.P.; Murad, M.H.; Weaver, C.M. Evaluation, Treatment, and Prevention of Vitamin D Deficiency: An Endocrine Society Clinical Practice Guideline. J. Clin. Endocrinol. Metab. 2011, 96, 1911–1930. [Google Scholar] [CrossRef] [Green Version]
- Vranić, L.; Mikolašević, I.; Milić, S. Vitamin D Deficiency: Consequence or Cause of Obesity? Medicina 2019, 55, 541. [Google Scholar] [CrossRef] [Green Version]
- Zakharova, I.; Klimov, L.; Kuryaninova, V.; Nikitina, I.; Malyavskaya, S.; Dolbnya, S.; Kasyanova, A.; Atanesyan, R.; Stoyan, M.; Todieva, A.; et al. Vitamin D Insufficiency in Overweight and Obese Children and Adolescents. Front. Endocrinol. 2019, 10, 103. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gracious, B.L.; Finucane, T.L.; Friedman-Campbell, M.; Messing, S.; Parkhurst, M.N. Vitamin D Deficiency and Psychotic Features in Mentally Ill Adolescents: A Cross-Sectional Study. BMC Psychiatry 2012, 12, 38. [Google Scholar] [CrossRef] [Green Version]
- Cuomo, A.; Maina, G.; Bolognesi, S.; Rosso, G.; Beccarini Crescenzi, B.; Zanobini, F.; Goracci, A.; Facchi, E.; Favaretto, E.; Baldini, I.; et al. Prevalence and Correlates of Vitamin D Deficiency in a Sample of 290 Inpatients With Mental Illness. Front. Psychiatry 2019, 10, 167. [Google Scholar] [CrossRef] [Green Version]
- Yüksel, R.N.; Altunsoy, N.; Tikir, B.; Cingi Külük, M.; Unal, K.; Goka, S.; Aydemir, C.; Goka, E. Correlation between Total Vitamin D Levels and Psychotic Psychopathology in Patients with Schizophrenia: Therapeutic Implications for Add-on Vitamin D Augmentation. Ther. Adv. Psychopharmacol. 2014, 4, 268–275. [Google Scholar] [CrossRef] [Green Version]
- Ghaderi, A.; Banafshe, H.R.; Mirhosseini, N.; Moradi, M.; Karimi, M.-A.; Mehrzad, F.; Bahmani, F.; Asemi, Z. Clinical and Metabolic Response to Vitamin D plus Probiotic in Schizophrenia Patients. BMC Psychiatry 2019, 19, 77. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Méndez-Salazar, E.O.; Ortiz-López, M.G.; Granados-Silvestre, M.d.l.Á.; Palacios-González, B.; Menjivar, M. Altered Gut Microbiota and Compositional Changes in Firmicutes and Proteobacteria in Mexican Undernourished and Obese Children. Front. Microbiol. 2018, 9, 2494. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Festi, D.; Schiumerini, R.; Eusebi, L.H.; Marasco, G.; Taddia, M.; Colecchia, A. Gut Microbiota and Metabolic Syndrome. World J. Gastroenterol. 2014, 20, 16079–16094. [Google Scholar] [CrossRef]
- Su, D.; Nie, Y.; Zhu, A.; Chen, Z.; Wu, P.; Zhang, L.; Luo, M.; Sun, Q.; Cai, L.; Lai, Y.; et al. Vitamin D Signaling through Induction of Paneth Cell Defensins Maintains Gut Microbiota and Improves Metabolic Disorders and Hepatic Steatosis in Animal Models. Front. Physiol. 2016, 7, 498. [Google Scholar] [CrossRef] [PubMed]
- Robles-Vera, I.; Callejo, M.; Ramos, R.; Duarte, J.; Perez-Vizcaino, F. Impact of Vitamin D Deficit on the Rat Gut Microbiome. Nutrients 2019, 11, 2564. [Google Scholar] [CrossRef] [Green Version]
- Bashir, M.; Prietl, B.; Tauschmann, M.; Mautner, S.I.; Kump, P.K.; Treiber, G.; Wurm, P.; Gorkiewicz, G.; Högenauer, C.; Pieber, T.R. Effects of High Doses of Vitamin D3 on Mucosa-Associated Gut Microbiome Vary between Regions of the Human Gastrointestinal Tract. Eur. J. Nutr. 2016, 55, 1479–1489. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- LeBlanc, J.G.; Milani, C.; de Giori, G.S.; Sesma, F.; van Sinderen, D.; Ventura, M. Bacteria as Vitamin Suppliers to Their Host: A Gut Microbiota Perspective. Curr. Opin. Biotechnol. 2013, 24, 160–168. [Google Scholar] [CrossRef] [PubMed]
- Allen, A.P.; Dinan, T.G.; Clarke, G.; Cryan, J.F. A Psychology of the Human Brain–Gut–Microbiome Axis. Soc. Personal. Psychol. Compass 2017, 11, e12309. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wilmes, L.; Collins, J.M.; O’Riordan, K.J.; O’Mahony, S.M.; Cryan, J.F.; Clarke, G. Of Bowels, Brain and Behavior: A Role for the Gut Microbiota in Psychiatric Comorbidities in Irritable Bowel Syndrome. Neurogastroenterol. Motil. 2021, 33, e14095. [Google Scholar] [CrossRef]
- Di Marzo, V. The Endocannabinoidome as a Substrate for Noneuphoric Phytocannabinoid Action and Gut Microbiome Dysfunction in Neuropsychiatric Disorders. Dialogues Clin. Neurosci. 2020, 22, 259–269. [Google Scholar] [CrossRef]
- Engeli, S. Dysregulation of the Endocannabinoid System in Obesity. J. Neuroendocrinol. 2008, 20, 110–115. [Google Scholar] [CrossRef]
- Cani, P.D.; Plovier, H.; Van Hul, M.; Geurts, L.; Delzenne, N.M.; Druart, C.; Everard, A. Endocannabinoids—At the Crossroads between the Gut Microbiota and Host Metabolism. Nat. Rev. Endocrinol. 2016, 12, 133. [Google Scholar] [CrossRef]
- Cristiano, C.; Pirozzi, C.; Coretti, L.; Cavaliere, G.; Lama, A.; Russo, R.; Lembo, F.; Mollica, M.P.; Meli, R.; Calignano, A.; et al. Palmitoylethanolamide Counteracts Autistic-like Behaviours in BTBR T+tf/J Mice: Contribution of Central and Peripheral Mechanisms. Brain Behav. Immun. 2018, 74, 166–175. [Google Scholar] [CrossRef]
- Guida, F.; Turco, F.; Iannotta, M.; Gregorio, D.D.; Palumbo, I.; Sarnelli, G.; Furiano, A.; Napolitano, F.; Boccella, S.; Luongo, L.; et al. Antibiotic-Induced Microbiota Perturbation Causes Gut Endocannabinoidome Changes, Hippocampal Neuroglial Reorganization and Depression in Mice. Brain Behav. Immun. 2018, 67, 230–245. [Google Scholar] [CrossRef] [PubMed]
- Dione, N.; Lacroix, S.; Taschler, U.; Deschênes, T.; Abolghasemi, A.; Leblanc, N.; Di Marzo, V.; Silvestri, C. Mgll Knockout Mouse Resistance to Diet-Induced Dysmetabolism Is Associated with Altered Gut Microbiota. Cells 2020, 9, 2705. [Google Scholar] [CrossRef]
- Iannotti, F.A.; Di Marzo, V. The Gut Microbiome, Endocannabinoids and Metabolic Disorders. J. Endocrinol. 2021, 248, R83–R97. [Google Scholar] [CrossRef] [PubMed]
- Manca, C.; Boubertakh, B.; Leblanc, N.; Deschênes, T.; Lacroix, S.; Martin, C.; Houde, A.; Veilleux, A.; Flamand, N.; Muccioli, G.G.; et al. Germ-Free Mice Exhibit Profound Gut Microbiota-Dependent Alterations of Intestinal Endocannabinoidome Signaling. J. Lipid Res. 2019, 61, P70–P85. [Google Scholar] [CrossRef]
- Muccioli, G.G.; Naslain, D.; Bäckhed, F.; Reigstad, C.S.; Lambert, D.M.; Delzenne, N.M.; Cani, P.D. The Endocannabinoid System Links Gut Microbiota to Adipogenesis. Mol. Syst. Biol. 2010, 6, 392. [Google Scholar] [CrossRef]
- Everard, A.; Belzer, C.; Geurts, L.; Ouwerkerk, J.P.; Druart, C.; Bindels, L.B.; Guiot, Y.; Derrien, M.; Muccioli, G.G.; Delzenne, N.M.; et al. Cross-Talk between Akkermansia Muciniphila and Intestinal Epithelium Controls Diet-Induced Obesity. Proc. Natl. Acad. Sci. USA 2013, 110, 9066. [Google Scholar] [CrossRef] [Green Version]
- Depommier, C.; Vitale, R.M.; Iannotti, F.A.; Silvestri, C.; Flamand, N.; Druart, C.; Everard, A.; Pelicaen, R.; Maiter, D.; Thissen, J.-P.; et al. Beneficial Effects of Akkermansia Muciniphila Are Not Associated with Major Changes in the Circulating Endocannabinoidome but Linked to Higher Mono-Palmitoyl-Glycerol Levels as New PPARα Agonists. Cells 2021, 10, 185. [Google Scholar] [CrossRef]
- Benetti, E.; Mastrocola, R.; Chiazza, F.; Nigro, D.; D’Antona, G.; Bordano, V.; Fantozzi, R.; Aragno, M.; Collino, M.; Minetto, M.A. Effects of Vitamin D on Insulin Resistance and Myosteatosis in Diet-Induced Obese Mice. PLoS ONE 2018, 13, e0189707. [Google Scholar] [CrossRef]
- Botella-Carretero, J.I.; Alvarez-Blasco, F.; Villafruela, J.J.; Balsa, J.A.; Vázquez, C.; Escobar-Morreale, H.F. Vitamin D Deficiency Is Associated with the Metabolic Syndrome in Morbid Obesity. Clin. Nutr. 2007, 26, 573–580. [Google Scholar] [CrossRef] [PubMed]
- Cieslak, K.; Feingold, J.; Antonius, D.; Walsh-Messinger, J.; Dracxler, R.; Rosedale, M.; Aujero, N.; Keefe, D.; Goetz, D.; Goetz, R.; et al. Low Vitamin D Levels Predict Clinical Features of Schizophrenia. Schizophr. Res. 2014, 159, 543–545. [Google Scholar] [CrossRef] [Green Version]
- Salehpour, A.; Hosseinpanah, F.; Shidfar, F.; Vafa, M.; Razaghi, M.; Dehghani, S.; Hoshiarrad, A.; Gohari, M. A 12-Week Double-Blind Randomized Clinical Trial of Vitamin D₃ Supplementation on Body Fat Mass in Healthy Overweight and Obese Women. Nutr. J. 2012, 11, 78. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kaviyani, S.; Bahadoram, M.; Houshmand, G.; Bahadoram, S. Therapeutic Impact of Cholecalciferol in Patients with Psychiatric Disorders Receiving Olanzapine. J. Parathyr. Dis. 2018, 6, 19–22. [Google Scholar] [CrossRef] [Green Version]
- Krivoy, A.; Onn, R.; Vilner, Y.; Hochman, E.; Weizman, S.; Paz, A.; Hess, S.; Sagy, R.; Kimhi-Nesher, S.; Kalter, E.; et al. Vitamin D Supplementation in Chronic Schizophrenia Patients Treated with Clozapine: A Randomized, Double-Blind, Placebo-Controlled Clinical Trial. EBioMedicine 2017, 26, 138–145. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fulton, B.; Goa, K.L. Olanzapine. Drugs 1997, 53, 281–298. [Google Scholar] [CrossRef]
- Boyda, H.N.; Tse, L.; Procyshyn, R.M.; Wong, D.; Wu, T.K.Y.; Pang, C.C.; Barr, A.M. A Parametric Study of the Acute Effects of Antipsychotic Drugs on Glucose Sensitivity in an Animal Model. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2010, 34, 945–954. [Google Scholar] [CrossRef]
- Boyda, H.N.; Procyshyn, R.M.; Tse, L.; Xu, J.; Jin, C.H.; Wong, D.; Pang, C.C.Y.; Honer, W.G.; Barr, A.M. Antipsychotic Polypharmacy Increases Metabolic Dysregulation in Female Rats. Exp. Clin. Psychopharmacol. 2013, 21, 164–171. [Google Scholar] [CrossRef]
- Auger, F.; Duriez, P.; Martin-Nizard, F.; Durieux, N.; Bordet, R.; Pétrault, O. Long-Term Risperidone Treatment Induces Visceral Adiposity Associated with Hepatic Steatosis in Mice: A Magnetic Resonance Approach. Schizophr. Res. Treat. 2014, 2014, 429291. [Google Scholar] [CrossRef]
- Hou, P.-H.; Chang, G.-R.; Chen, C.-P.; Lin, Y.-L.; Chao, I.-S.; Shen, T.-T.; Mao, F.C. Long-Term Administration of Olanzapine Induces Adiposity and Increases Hepatic Fatty Acid Desaturation Protein in Female C57BL/6J Mice. Iran J. Basic Med. Sci. 2018, 21, 495–501. [Google Scholar] [CrossRef]
- Davey, K.J.; O’Mahony, S.M.; Schellekens, H.; O’Sullivan, O.; Bienenstock, J.; Cotter, P.D.; Dinan, T.G.; Cryan, J.F. Gender-Dependent Consequences of Chronic Olanzapine in the Rat: Effects on Body Weight, Inflammatory, Metabolic and Microbiota Parameters. Psychopharmacology 2012, 221, 155–169. [Google Scholar] [CrossRef]
- Lazzari, P.; Serra, V.; Marcello, S.; Pira, M.; Mastinu, A. Metabolic Side Effects Induced by Olanzapine Treatment Are Neutralized by CB1 Receptor Antagonist Compounds Co-Administration in Female Rats. Eur. Neuropsychopharmacol. 2017, 27, 667–678. [Google Scholar] [CrossRef]
- Cussotto, S.; Walsh, J.; Golubeva, A.V.; Zhdanov, A.V.; Strain, C.R.; Fouhy, F.; Stanton, C.; Dinan, T.G.; Hyland, N.P.; Clarke, G.; et al. The Gut Microbiome Influences the Bioavailability of Olanzapine in Rats. EBioMedicine 2021, 66, 103307. [Google Scholar] [CrossRef] [PubMed]
- Templeman, N.M.; Skovsø, S.; Page, M.M.; Lim, G.E.; Johnson, J.D. A Causal Role for Hyperinsulinemia in Obesity. J. Endocrinol. 2017, 232, R173–R183. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Townsend, L.K.; Peppler, W.T.; Bush, N.D.; Wright, D.C. Obesity Exacerbates the Acute Metabolic Side Effects of Olanzapine. Psychoneuroendocrinology 2018, 88, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Coccurello, R.; Caprioli, A.; Ghirardi, O.; Conti, R.; Ciani, B.; Daniele, S.; Bartolomucci, A.; Moles, A. Chronic Administration of Olanzapine Induces Metabolic and Food Intake Alterations: A Mouse Model of the Atypical Antipsychotic-Associated Adverse Effects. Psychopharmacology 2006, 186, 561–571. [Google Scholar] [CrossRef]
- Shertzer, H.G.; Kendig, E.L.; Nasrallah, H.A.; Johansson, E.; Genter, M.B. Protection from Olanzapine-Induced Metabolic Toxicity in Mice by Acetaminophen and Tetrahydroindenoindole. Int. J. Obes. 2010, 34, 970–979. [Google Scholar] [CrossRef] [Green Version]
- Cope, M.B.; Jumbo-Lucioni, P.; Walton, R.G.; Kesterson, R.A.; Allison, D.B.; Nagy, T.R. No Effect of Dietary Fat on Short-Term Weight Gain in Mice Treated with Atypical Antipsychotic Drugs. Int. J. Obes. 2007, 31, 1014–1022. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Smith, G.C.; Vickers, M.H.; Shepherd, P.R. Olanzapine Effects on Body Composition, Food Preference, Glucose Metabolism and Insulin Sensitivity in the Rat. J. Metab. Dis. 2011, 117, 241–249. [Google Scholar] [CrossRef] [PubMed]
- Smith, R.C.; Lindenmayer, J.-P.; Davis, J.M.; Kelly, E.; Viviano, T.F.; Cornwell, J.; Hu, Q.; Khan, A.; Vaidhyanathaswamy, S. Effects of Olanzapine and Risperidone on Glucose Metabolism and Insulin Sensitivity in Chronic Schizophrenic Patients with Long-Term Antipsychotic Treatment: A Randomized 5-Month Study. J. Clin. Psychiatry 2009, 70, 1501–1513. [Google Scholar] [CrossRef]
- Fell, M.; Neill, J.; Anjum, N.; Peltola, L.; Marshall, K. Investigation into the Influence of a High Fat Diet on Antipsychotic-Induced Weight Gain in Female Rats. J. Psychopharmacol. 2008, 22, 182–186. [Google Scholar] [CrossRef] [PubMed]
- Di Marzo, V.; Silvestri, C. Lifestyle and Metabolic Syndrome: Contribution of the Endocannabinoidome. Nutrients 2019, 11, 1956. [Google Scholar] [CrossRef] [Green Version]
- Horn, H.; Böhme, B.; Dietrich, L.; Koch, M. Endocannabinoids in Body Weight Control. Pharmaceuticals 2018, 11, 55. [Google Scholar] [CrossRef] [Green Version]
- Di Marzo, V.; Goparaju, S.K.; Wang, L.; Liu, J.; Bátkai, S.; Járai, Z.; Fezza, F.; Miura, G.I.; Palmiter, R.D.; Sugiura, T.; et al. Leptin-Regulated Endocannabinoids Are Involved in Maintaining Food Intake. Nature 2001, 410, 822–825. [Google Scholar] [CrossRef] [PubMed]
- Pi-Sunyer, F.X.; Aronne, L.J.; Heshmati, H.M.; Devin, J.; Rosenstock, J. RIO-North America Study Group, for the Effect of Rimonabant, a Cannabinoid-1 Receptor Blocker, on Weight and Cardiometabolic Risk Factors in Overweight or Obese PatientsRIO-North America: A Randomized Controlled Trial. JAMA 2006, 295, 761–775. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Clapper, J.R.; Henry, C.L.; Niphakis, M.J.; Knize, A.M.; Coppola, A.R.; Simon, G.M.; Ngo, N.; Herbst, R.A.; Herbst, D.M.; Reed, A.W.; et al. Monoacylglycerol Lipase Inhibition in Human and Rodent Systems Supports Clinical Evaluation of Endocannabinoid Modulators. J. Pharmacol. Exp. Ther. 2018, 367, 494. [Google Scholar] [CrossRef] [Green Version]
- Hofer, P.; Taschler, U.; Schreiber, R.; Kotzbeck, P.; Schoiswohl, G. The Lipolysome—A Highly Complex and Dynamic Protein Network Orchestrating Cytoplasmic Triacylglycerol Degradation. Metabolites 2020, 10, 147. [Google Scholar] [CrossRef]
- Matias, I.; Bisogno, T.; Di Marzo, V. Endogenous Cannabinoids in the Brain and Peripheral Tissues: Regulation of Their Levels and Control of Food Intake. Int. J. Obes. 2006, 30, S7–S12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Delis, F.; Rosko, L.; Shroff, A.; Leonard, K.E.; Thanos, P.K. Oral Haloperidol or Olanzapine Intake Produces Distinct and Region-Specific Increase in Cannabinoid Receptor Levels That Is Prevented by High Fat Diet. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2017, 79, 268–280. [Google Scholar] [CrossRef]
- Iwasaki, Y.; Saito, O.; Tanabe, M.; Inayoshi, K.; Kobata, K.; Uno, S.; Morita, A.; Watanabe, T. Monoacylglycerols Activate Capsaicin Receptor, TRPV1. Lipids 2008, 43, 471–483. [Google Scholar] [CrossRef] [PubMed]
- Zygmunt, P.M.; Ermund, A.; Movahed, P.; Andersson, D.A.; Simonsen, C.; Jönsson, B.A.G.; Blomgren, A.; Birnir, B.; Bevan, S.; Eschalier, A.; et al. Monoacylglycerols Activate TRPV1–A Link between Phospholipase C and TRPV1. PLoS ONE 2013, 8, e81618. [Google Scholar] [CrossRef] [Green Version]
- Kang, J.-H.; Tsuyoshi, G.; Han, I.-S.; Kawada, T.; Kim, Y.M.; Yu, R. Dietary Capsaicin Reduces Obesity-Induced Insulin Resistance and Hepatic Steatosis in Obese Mice Fed a High-Fat Diet. Obesity 2010, 18, 780–787. [Google Scholar] [CrossRef]
- Iwasaki, Y.; Tamura, Y.; Inayoshi, K.; Narukawa, M.; Kobata, K.; Chiba, H.; Muraki, E.; Tsunoda, N.; Watanabe, T. TRPV1 Agonist Monoacylglycerol Increases UCP1 Content in Brown Adipose Tissue and Suppresses Accumulation of Visceral Fat in Mice Fed a High-Fat and High-Sucrose Diet. Biosci. Biotechnol. Biochem. 2011, 75, 904–909. [Google Scholar] [CrossRef] [Green Version]
- Motter, A.L.; Ahern, G.P. TRPV1-Null Mice Are Protected from Diet-Induced Obesity. FEBS Lett. 2008, 582, 2257–2262. [Google Scholar] [CrossRef] [Green Version]
- Lowin, T.; Apitz, M.; Anders, S.; Straub, R.H. Anti-Inflammatory Effects of N-Acylethanolamines in Rheumatoid Arthritis Synovial Cells Are Mediated by TRPV1 and TRPA1 in a COX-2 Dependent Manner. Arthritis Res. Ther. 2015, 17, 321. [Google Scholar] [CrossRef] [Green Version]
- Ambrosino, P.; Soldovieri, M.V.; Russo, C.; Taglialatela, M. Activation and Desensitization of TRPV1 Channels in Sensory Neurons by the PPARα Agonist Palmitoylethanolamide. Br. J. Pharmacol. 2013, 168, 1430–1444. [Google Scholar] [CrossRef] [Green Version]
- Geurts, L.; Everard, A.; Van Hul, M.; Essaghir, A.; Duparc, T.; Matamoros, S.; Plovier, H.; Castel, J.; Denis, R.G.P.; Bergiers, M.; et al. Adipose Tissue NAPE-PLD Controls Fat Mass Development by Altering the Browning Process and Gut Microbiota. Nat. Commun. 2015, 6, 6495. [Google Scholar] [CrossRef] [Green Version]
- Liu, X.; Nelson, A.; Wang, X.; Farid, M.; Gunji, Y.; Ikari, J.; Iwasawa, S.; Basma, H.; Feghali-Bostwick, C.; Rennard, S.I. Vitamin D Modulates Prostaglandin E2 Synthesis and Degradation in Human Lung Fibroblasts. Am. J. Respir. Cell Mol. Biol. 2013, 50, 40–50. [Google Scholar] [CrossRef]
- Schwartz, Z.; Swain, L.D.; Kelly, D.W.; Brooks, B.; Boyan, B.D. Regulation of Prostaglandin E2 Production by Vitamin D Metabolites in Growth Zone and Resting Zone Chondrocyte Cultures Is Dependent on Cell Maturation. Bone 1992, 13, 395–401. [Google Scholar] [CrossRef]
- Cheon, Y.; Park, J.-Y.; Modi, H.R.; Kim, H.-W.; Lee, H.-J.; Chang, L.; Rao, J.S.; Rapoport, S.I. Chronic Olanzapine Treatment Decreases Arachidonic Acid Turnover and Prostaglandin E2 Concentration in Rat Brain. J. Neurochem. 2011, 119, 364–376. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Faour-Nmarne, C.; Azab, A.N. Effects of Olanzapine on LPS-Induced Inflammation in Rat Primary Glia Cells. Innate Immun. 2015, 22, 40–50. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pisani, D.F.; Ghandour, R.A.; Beranger, G.E.; Le Faouder, P.; Chambard, J.-C.; Giroud, M.; Vegiopoulos, A.; Djedaini, M.; Bertrand-Michel, J.; Tauc, M.; et al. The Ω6-Fatty Acid, Arachidonic Acid, Regulates the Conversion of White to Brite Adipocyte through a Prostaglandin/Calcium Mediated Pathway. Mol. Metab. 2014, 3, 834–847. [Google Scholar] [CrossRef]
- García-Alonso, V.; López-Vicario, C.; Titos, E.; Morán-Salvador, E.; González-Périz, A.; Rius, B.; Párrizas, M.; Werz, O.; Arroyo, V.; Clària, J. Coordinate Functional Regulation between Microsomal Prostaglandin E Synthase-1 (MPGES-1) and Peroxisome Proliferator-Activated Receptor γ (PPARγ) in the Conversion of White-to-Brown Adipocytes. J. Biol. Chem. 2013, 288, 28230–28242. [Google Scholar] [CrossRef] [Green Version]
- García-Alonso, V.; Titos, E.; Alcaraz-Quiles, J.; Rius, B.; Lopategi, A.; López-Vicario, C.; Jakobsson, P.-J.; Delgado, S.; Lozano, J.; Clària, J. Prostaglandin E2 Exerts Multiple Regulatory Actions on Human Obese Adipose Tissue Remodeling, Inflammation, Adaptive Thermogenesis and Lipolysis. PLoS ONE 2016, 11, e0153751. [Google Scholar] [CrossRef] [Green Version]
- Hu, X.; Cifarelli, V.; Sun, S.; Kuda, O.; Abumrad, N.A.; Su, X. Major Role of Adipocyte Prostaglandin E2 in Lipolysis-Induced Macrophage Recruitment. J. Lipid Res. 2016, 57, 663–673. [Google Scholar] [CrossRef] [Green Version]
- Mazid, M.A.; Chowdhury, A.A.; Nagao, K.; Nishimura, K.; Jisaka, M.; Nagaya, T.; Yokota, K. Endogenous 15-Deoxy-Δ12,14-Prostaglandin J2 Synthesized by Adipocytes during Maturation Phase Contributes to Upregulation of Fat Storage. FEBS Lett. 2006, 580, 6885–6890. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mariat, D.; Firmesse, O.; Levenez, F.; Guimarăes, V.; Sokol, H.; Doré, J.; Corthier, G.; Furet, J.-P. The Firmicutes/Bacteroidetes Ratio of the Human Microbiota Changes with Age. BMC Microbiol. 2009, 9, 123. [Google Scholar] [CrossRef]
- Turnbaugh, P.J.; Hamady, M.; Yatsunenko, T.; Cantarel, B.L.; Duncan, A.; Ley, R.E.; Sogin, M.L.; Jones, W.J.; Roe, B.A.; Affourtit, J.P.; et al. A Core Gut Microbiome in Obese and Lean Twins. Nature 2009, 457, 480–484. [Google Scholar] [CrossRef] [Green Version]
- Turnbaugh, P.J.; Ley, R.E.; Mahowald, M.A.; Magrini, V.; Mardis, E.R.; Gordon, J.I. An Obesity-Associated Gut Microbiome with Increased Capacity for Energy Harvest. Nature 2006, 444, 1027–1031. [Google Scholar] [CrossRef]
- Luthold, R.V.; Fernandes, G.R.; Franco-de-Moraes, A.C.; Folchetti, L.G.D.; Ferreira, S.R.G. Gut Microbiota Interactions with the Immunomodulatory Role of Vitamin D in Normal Individuals. Metab. Clin. Exp. 2017, 69, 76–86. [Google Scholar] [CrossRef] [PubMed]
- Ooi, J.H.; Li, Y.; Rogers, C.J.; Cantorna, M.T. Vitamin D Regulates the Gut Microbiome and Protects Mice from Dextran Sodium Sulfate-Induced Colitis. J. Nutr. 2013, 143, 1679–1686. [Google Scholar] [CrossRef]
- Bora, S.A.; Kennett, M.J.; Smith, P.B.; Patterson, A.D.; Cantorna, M.T. The Gut Microbiota Regulates Endocrine Vitamin D Metabolism through Fibroblast Growth Factor 23. Front. Immunol. 2018, 9, 408. [Google Scholar] [CrossRef] [Green Version]
- Gong, S.; Ye, T.; Wang, M.; Wang, M.; Li, Y.; Ma, L.; Yang, Y.; Wang, Y.; Zhao, X.; Liu, L.; et al. Traditional Chinese Medicine Formula Kang Shuai Lao Pian Improves Obesity, Gut Dysbiosis, and Fecal Metabolic Disorders in High-Fat Diet-Fed Mice. Front. Pharmacol. 2020, 11, 297. [Google Scholar] [CrossRef] [PubMed]
- Gonai, M.; Shigehisa, A.; Kigawa, I.; Kurasaki, K.; Chonan, O.; Matsuki, T.; Yoshida, Y.; Aida, M.; Hamano, K.; Terauchi, Y. Galacto-Oligosaccharides Ameliorate Dysbiotic Bifidobacteriaceae Decline in Japanese Patients with Type 2 Diabetes. Benef. Microbes 2017, 8, 705–716. [Google Scholar] [CrossRef] [PubMed]
- McCabe, L.R.; Irwin, R.; Tekalur, A.; Evans, C.; Schepper, J.D.; Parameswaran, N.; Ciancio, M. Exercise Prevents High Fat Diet-Induced Bone Loss, Marrow Adiposity and Dysbiosis in Male Mice. Bone 2019, 118, 20–31. [Google Scholar] [CrossRef]
- Kulecka, M.; Paziewska, A.; Zeber-Lubecka, N.; Ambrozkiewicz, F.; Kopczynski, M.; Kuklinska, U.; Pysniak, K.; Gajewska, M.; Mikula, M.; Ostrowski, J. Prolonged Transfer of Feces from the Lean Mice Modulates Gut Microbiota in Obese Mice. Nutr. Metab. 2016, 13, 57. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Li, L.; Ye, C.; Yuan, J.; Qin, S. Alginate Oligosaccharide Improves Lipid Metabolism and Inflammation by Modulating Gut Microbiota in High-Fat Diet Fed Mice. Appl. Microbiol. Biotechnol. 2020, 104, 3541–3554. [Google Scholar] [CrossRef]
- Bligh, E.G.; Dyer, W.J. A Rapid Method of Total Lipid Extraction and Purification. Can. J. Biochem. Physiol. 1959, 37, 911–917. [Google Scholar] [CrossRef]
- Everard, A.; Plovier, H.; Rastelli, M.; Van Hul, M.; de Wouters d’Oplinter, A.; Geurts, L.; Druart, C.; Robine, S.; Delzenne, N.M.; Muccioli, G.G.; et al. Intestinal Epithelial N-Acylphosphatidylethanolamine Phospholipase D Links Dietary Fat to Metabolic Adaptations in Obesity and Steatosis. Nat. Commun. 2019, 10, 457. [Google Scholar] [CrossRef]
- Quast, C.; Pruesse, E.; Yilmaz, P.; Gerken, J.; Schweer, T.; Yarza, P.; Peplies, J.; Glöckner, F.O. The SILVA Ribosomal RNA Gene Database Project: Improved Data Processing and Web-Based Tools. Nucleic Acids Res. 2013, 41, D590–D596. [Google Scholar] [CrossRef]
- Paulson, J.N.; Stine, O.C.; Bravo, H.C.; Pop, M. Differential Abundance Analysis for Microbial Marker-Gene Surveys. Nat. Methods 2013, 10, 1200–1202. [Google Scholar] [CrossRef] [Green Version]
- McMurdie, P.J.; Holmes, S. Phyloseq: An R Package for Reproducible Interactive Analysis and Graphics of Microbiome Census Data. PLoS ONE 2013, 8, e61217. [Google Scholar] [CrossRef] [Green Version]




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abolghasemi, A.; Manca, C.; Iannotti, F.A.; Shen, M.; Leblanc, N.; Lacroix, S.; Martin, C.; Flamand, N.; Di Marzo, V.; Silvestri, C. Assessment of the Effects of Dietary Vitamin D Levels on Olanzapine-Induced Metabolic Side Effects: Focus on the Endocannabinoidome-Gut Microbiome Axis. Int. J. Mol. Sci. 2021, 22, 12361. https://doi.org/10.3390/ijms222212361
Abolghasemi A, Manca C, Iannotti FA, Shen M, Leblanc N, Lacroix S, Martin C, Flamand N, Di Marzo V, Silvestri C. Assessment of the Effects of Dietary Vitamin D Levels on Olanzapine-Induced Metabolic Side Effects: Focus on the Endocannabinoidome-Gut Microbiome Axis. International Journal of Molecular Sciences. 2021; 22(22):12361. https://doi.org/10.3390/ijms222212361
Chicago/Turabian StyleAbolghasemi, Armita, Claudia Manca, Fabio A. Iannotti, Melissa Shen, Nadine Leblanc, Sébastien Lacroix, Cyril Martin, Nicolas Flamand, Vincenzo Di Marzo, and Cristoforo Silvestri. 2021. "Assessment of the Effects of Dietary Vitamin D Levels on Olanzapine-Induced Metabolic Side Effects: Focus on the Endocannabinoidome-Gut Microbiome Axis" International Journal of Molecular Sciences 22, no. 22: 12361. https://doi.org/10.3390/ijms222212361






