L-dopa-Dependent Effects of GLP-1R Agonists on the Survival of Dopaminergic Cells Transplanted into a Rat Model of Parkinson Disease
Abstract
:1. Introduction
2. Results
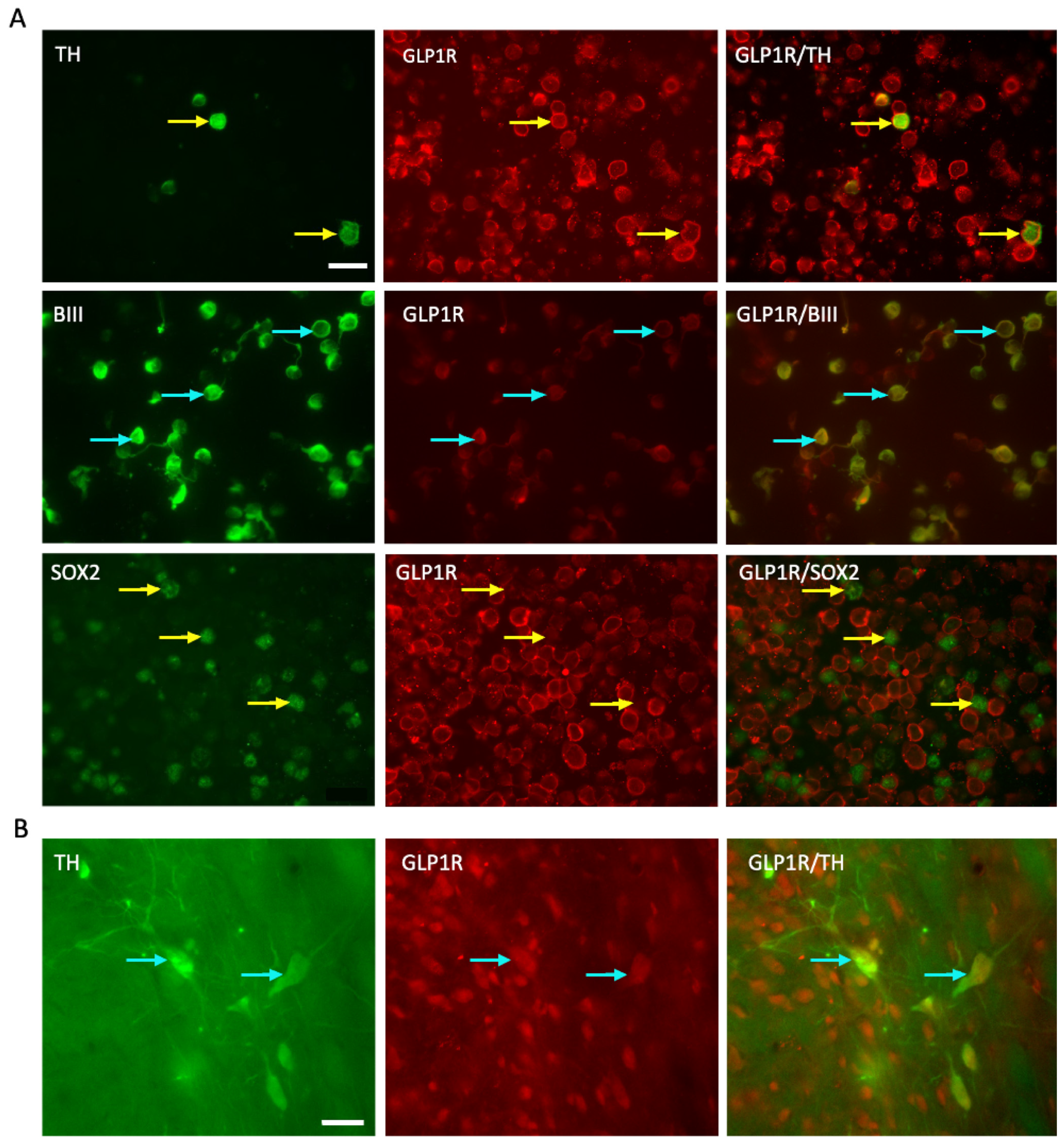
2.1. GLP-1R Receptor Expressed on the E14 VM Cells and Persists Post-Transplantation
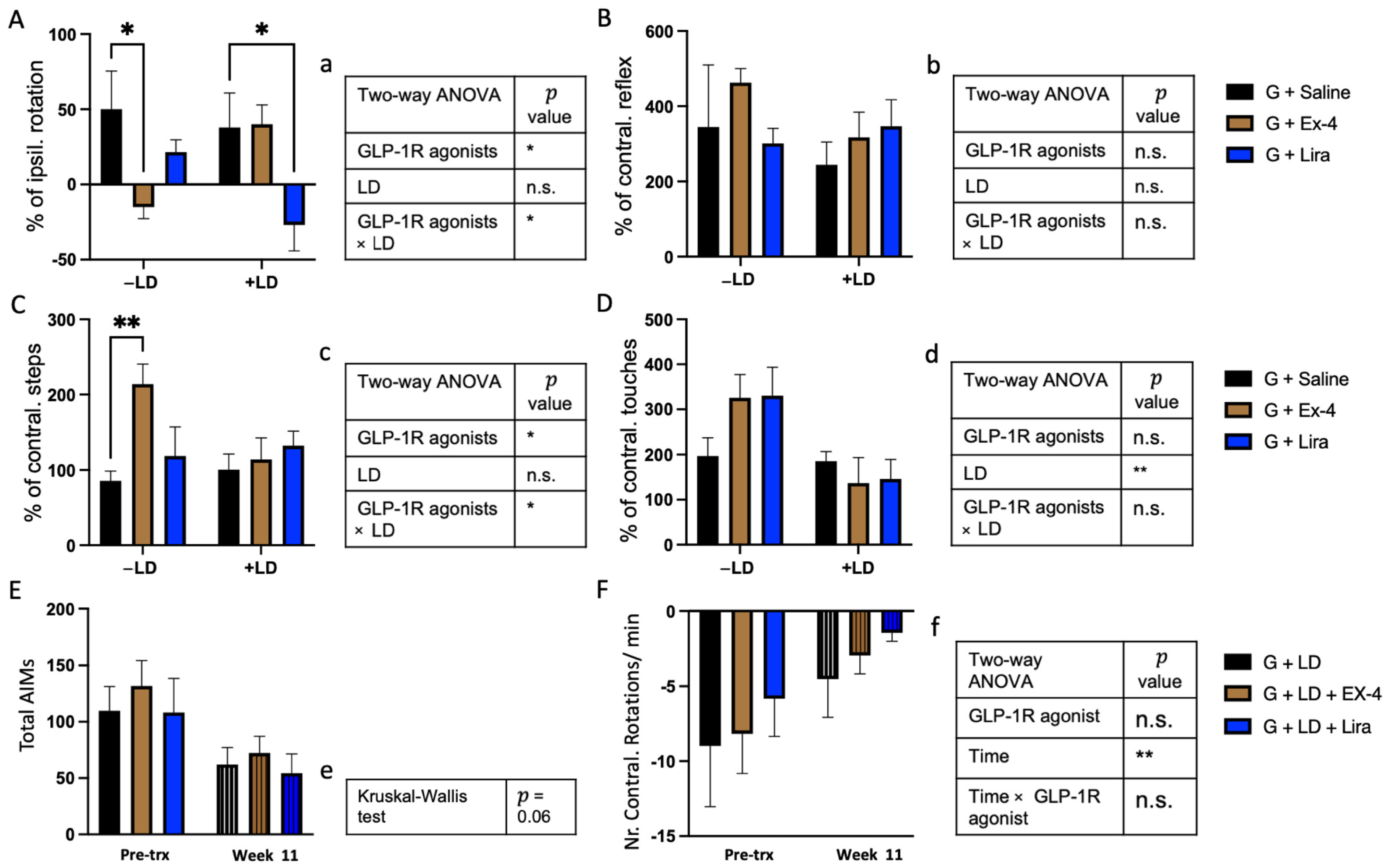
2.2. Differential Effects of Exendin-4 and Liraglutide on Graft Function in the Presence of L-dopa
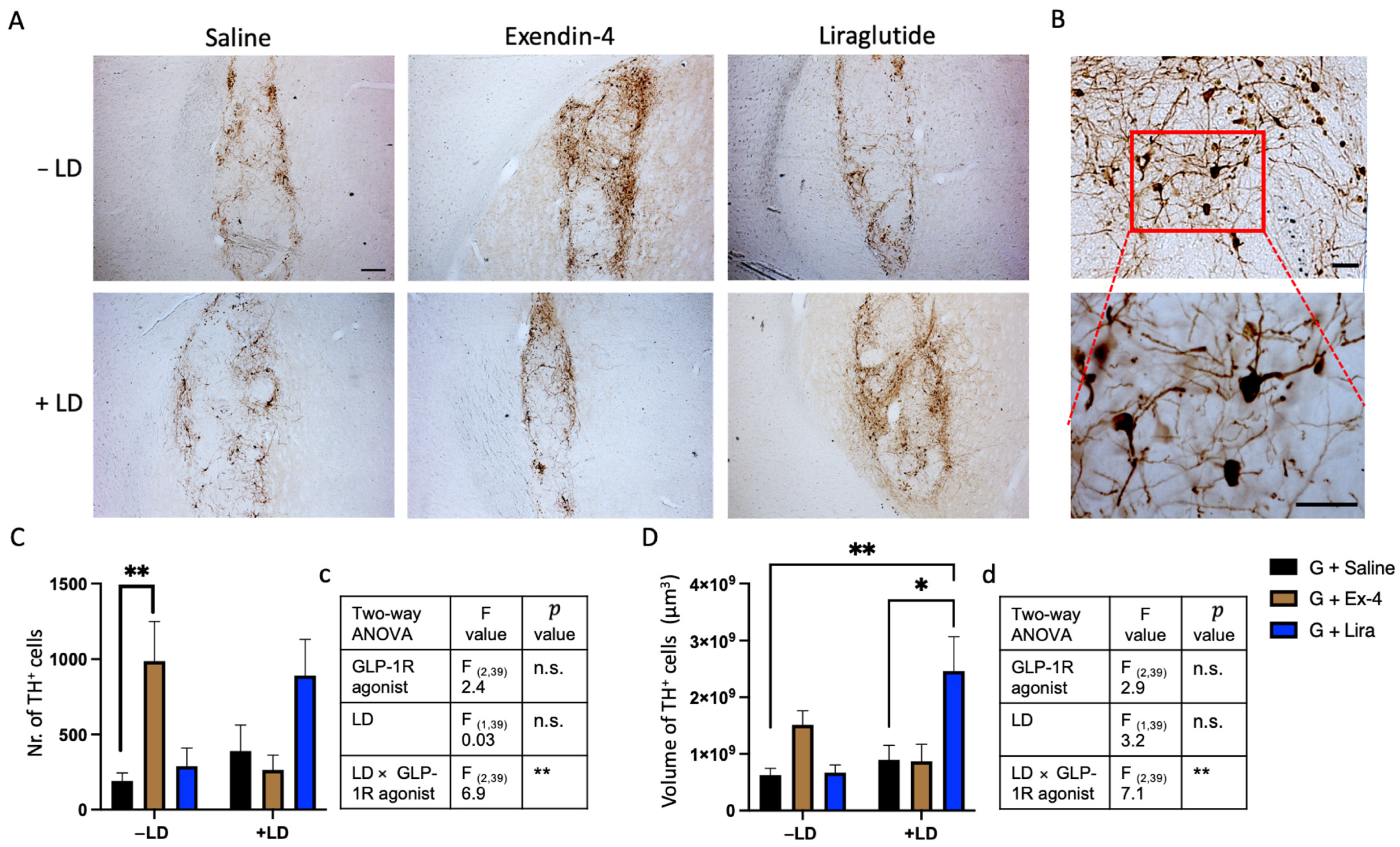
2.3. GLP-1R Agonists Induced Alterations in the Histological Characteristics of the Graft
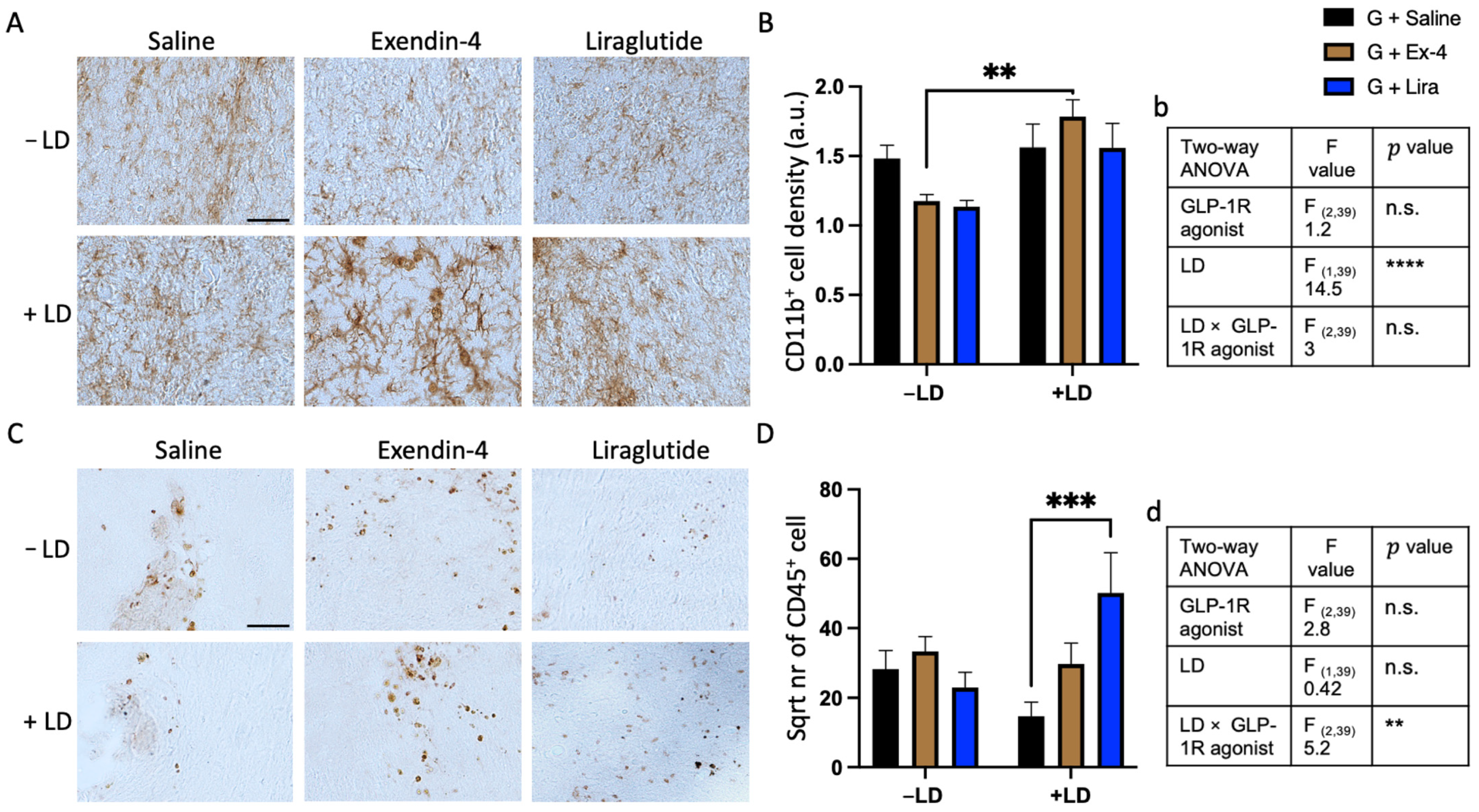
2.4. L-dopa and GLP-1R Agonists Altered the Inflammatory Response around the Graft
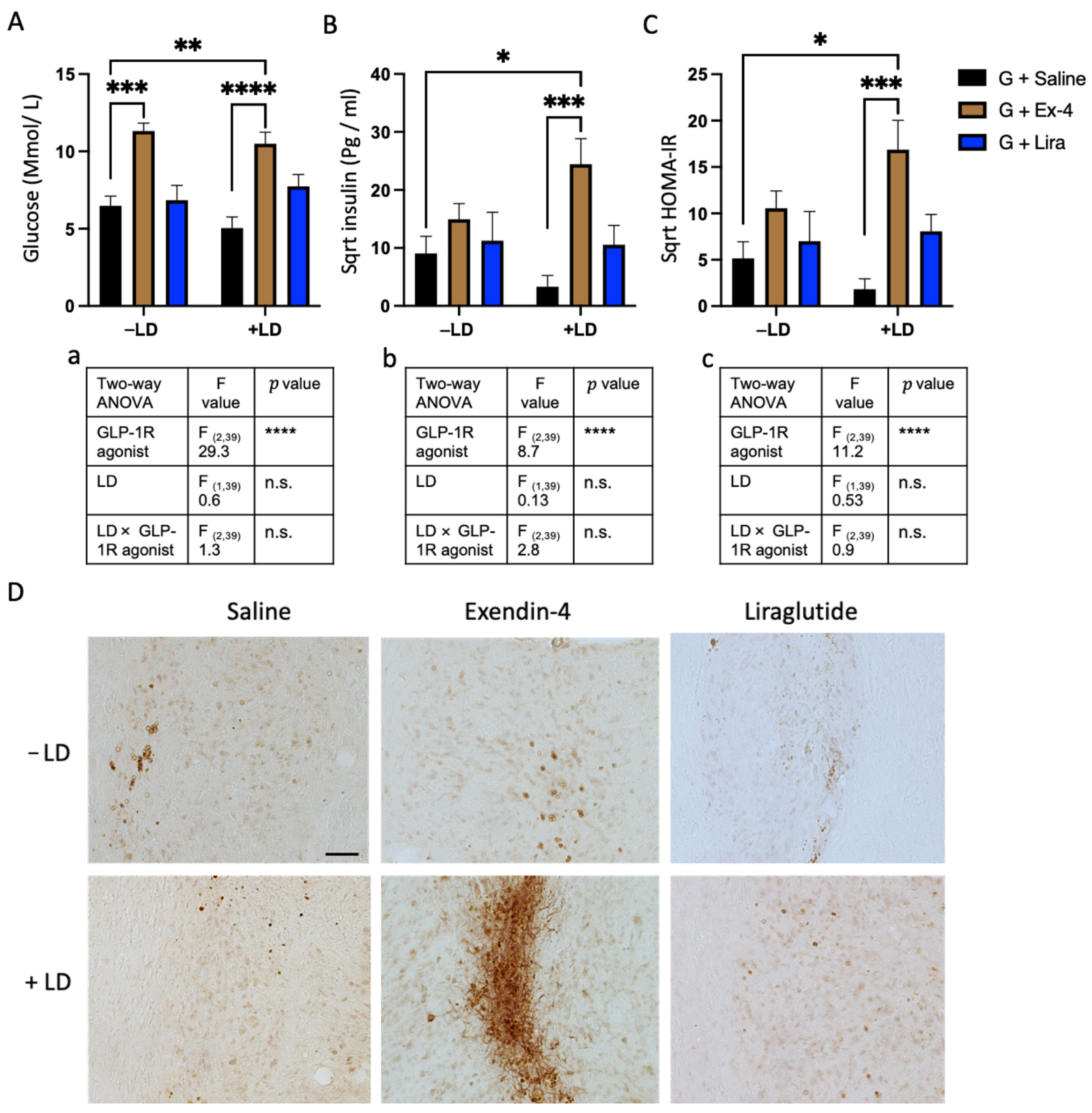
2.5. Exendin-4 Paradoxically Caused Insulin Resistance in the Grafted Rats Treated with L-dopa
3. Discussion
4. Material and Methods
4.1. Animals
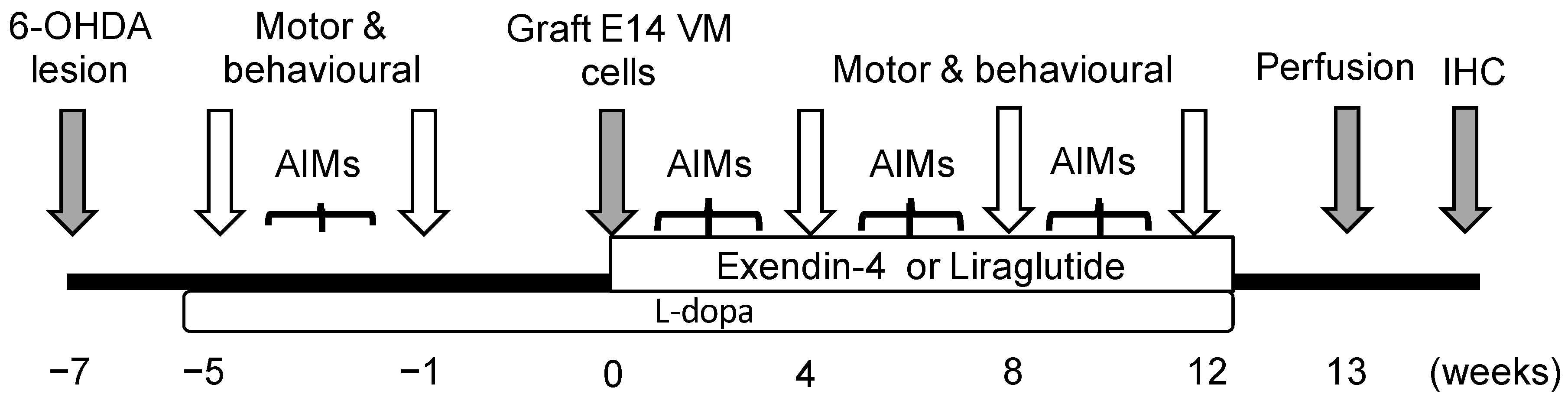
4.2. Experimental Design
4.3. Surgical Procedure
4.4. Treatments
4.5. Motor and L-dopa Induced Dyskinesia Tests
4.6. Blood Sampling, Perfusion and Fixation
4.7. Plasma Sample Analysis
4.8. Immunohistochemistry
4.9. Histological Analysis
4.10. Immunocytochemistry
4.11. Protein Lysis and Western Blot Analysis
4.12. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dorsey, E.R.; Bloem, B.R. The Parkinson Pandemic-A Call to Action. JAMA Neurol. 2018, 75, 9–10. [Google Scholar] [CrossRef]
- Fearnley, J.M.; Lees, A.J. Ageing and Parkinson’s disease: Substantia nigra regional selectivity. Brain 1991, 114, 2283–2301. [Google Scholar] [CrossRef]
- Kefalopoulou, Z.; Politis, M.; Piccini, P.; Mencacci, N.; Bhatia, K.; Jahanshahi, M.; Widner, H.; Rehncrona, S.; Brundin, P.; Bjorklund, A.; et al. Long-term clinical outcome of fetal cell transplantation for Parkinson disease: Two case reports. JAMA Neurol. 2014, 71, 83–87. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ma, Y.; Tang, C.; Chaly, T.; Greene, P.; Breeze, R.; Fahn, S.; Freed, C.; Dhawan, V.; Eidelberg, D. Dopamine cell implantation in Parkinson’s disease: Long-term clinical and (18)F-FDOPA PET outcomes. J. Nucl. Med. 2010, 51, 7–15. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hallett, P.J.; Cooper, O.; Sadi, D.; Robertson, H.; Mendez, I.; Isacson, O. Long-term health of dopaminergic neuron transplants in Parkinson’s disease patients. Cell Rep. 2014, 7, 1755–1761. [Google Scholar] [CrossRef] [Green Version]
- Freed, C.R.; Greene, P.E.; Breeze, R.E.; Tsai, W.Y.; DuMouchel, W.; Kao, R.; Dillon, S.; Winfield, H.; Culver, S.; Trojanowski, J.Q.; et al. Transplantation of embryonic dopamine neurons for severe Parkinson’s disease. N. Engl. J. Med. 2001, 344, 710–719. [Google Scholar] [CrossRef] [PubMed]
- Olanow, C.W.; Goetz, C.G.; Kordower, J.H.; Stoessl, A.J.; Sossi, V.; Brin, M.F.; Shannon, K.M.; Nauert, G.M.; Perl, D.P.; Godbold, J.; et al. A double-blind controlled trial of bilateral fetal nigral transplantation in Parkinson’s disease. Ann. Neurol. 2003, 54, 403–414. [Google Scholar] [CrossRef]
- TRANSEURO. Innovative Approach for the Treatment of Parkinson’s Disease. Available online: https://transeuro.org.uk (accessed on 20 September 2021).
- Kirkeby, A.; Parmar, M.; Barker, R.A. Strategies for bringing stem cell-derived dopamine neurons to the clinic: A European approach (STEM-PD). Prog. Brain Res. 2017, 230, 165–190. [Google Scholar] [CrossRef] [PubMed]
- Barker, R.A. Designing stem-cell-based dopamine cell replacement trials for Parkinson’s disease. Nat. Med. 2019, 25, 1045–1053. [Google Scholar] [CrossRef]
- Olanow, C.W.; Kordower, J.H.; Freeman, T.B. Fetal nigral transplantation as a therapy for Parkinson’s disease. Trends Neurosci. 1996, 19, 102–109. [Google Scholar] [CrossRef]
- Kordower, J.H.; Freeman, T.B.; Chen, E.Y.; Mufson, E.J.; Sanberg, P.R.; Hauser, R.A.; Snow, B.; Olanow, C.W. Fetal nigral grafts survive and mediate clinical benefit in a patient with Parkinson’s disease. Mov. Disord. 1998, 13, 383–393. [Google Scholar] [CrossRef]
- Brundin, P.; Karlsson, J.; Emgard, M.; Schierle, G.S.; Hansson, O.; Petersen, A.; Castilho, R.F. Improving the survival of grafted dopaminergic neurons: A review over current approaches. Cell Transpl. 2000, 9, 179–195. [Google Scholar] [CrossRef] [PubMed]
- Schierle, G.S.; Hansson, O.; Leist, M.; Nicotera, P.; Widner, H.; Brundin, P. Caspase inhibition reduces apoptosis and increases survival of nigral transplants. Nat. Med. 1999, 5, 97–100. [Google Scholar] [CrossRef] [PubMed]
- Cabre, S.; Alamilla, V.; Moriarty, N.; Pandit, A.; Dowd, E. Anti-inflammatory cytokine-eluting collagen hydrogel reduces the host immune response to dopaminergic cell transplants in a rat model of Parkinson’s disease. Neuronal Signal. 2021, 5, NS20210028. [Google Scholar] [CrossRef]
- Moriarty, N.; Pandit, A.; Dowd, E. Encapsulation of primary dopaminergic neurons in a GDNF-loaded collagen hydrogel increases their survival, re-innervation and function after intra-striatal transplantation. Sci. Rep. 2017, 7, 16033. [Google Scholar] [CrossRef] [PubMed]
- Grasbon-Frodl, E.M.; Nakao, N.; Brundin, P. The lazaroid U-83836E improves the survival of rat embryonic mesencephalic tissue stored at 4 degrees C and subsequently used for cultures or intracerebral transplantation. Brain Res. Bull. 1996, 39, 341–347. [Google Scholar] [CrossRef]
- Gutniak, M.; Orskov, C.; Holst, J.J.; Ahren, B.; Efendic, S. Antidiabetogenic effect of glucagon-like peptide-1 (7-36)amide in normal subjects and patients with diabetes mellitus. N. Engl. J. Med. 1992, 326, 1316–1322. [Google Scholar] [CrossRef] [PubMed]
- Gutniak, M.K.; Linde, B.; Holst, J.J.; Efendic, S. Subcutaneous injection of the incretin hormone glucagon-like peptide 1 abolishes postprandial glycemia in NIDDM. Diabetes Care 1994, 17, 1039–1044. [Google Scholar] [CrossRef]
- Augestad, I.L.; Dekens, D.; Karampatsi, D.; Elabi, O.; Zabala, A.; Pintana, H.; Larsson, M.; Nystrom, T.; Paul, G.; Darsalia, V.; et al. Normalisation of glucose metabolism by Exendin-4 in the chronic phase after stroke promotes functional recovery in male diabetic mice. Br. J. Pharmacol. 2021. [Google Scholar] [CrossRef]
- Zhang, J.; Yi, T.; Cheng, S.; Zhang, S. Glucagon-like peptide-1 receptor agonist Exendin-4 improves neurological outcomes by attenuating TBI- induced inflammatory responses and MAPK activation in rats. Int. Immunopharmacol. 2020, 86, 106715. [Google Scholar] [CrossRef]
- Ji, C.; Xue, G.F.; Lijun, C.; Feng, P.; Li, D.; Li, L.; Li, G.; Holscher, C. A novel dual GLP-1 and GIP receptor agonist is neuroprotective in the MPTP mouse model of Parkinson’s disease by increasing expression of BNDF. Brain Res. 2016, 1634, 1–11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aviles-Olmos, I.; Dickson, J.; Kefalopoulou, Z.; Djamshidian, A.; Kahan, J.; Ell, P.; Whitton, P.; Wyse, R.; Isaacs, T.; Lees, A.; et al. Motor and cognitive advantages persist 12 months after exenatide exposure in Parkinson’s disease. J. Parkinsons Dis. 2014, 4, 337–344. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Harkavyi, A.; Abuirmeileh, A.; Lever, R.; Kingsbury, A.E.; Biggs, C.S.; Whitton, P.S. Glucagon-like peptide 1 receptor stimulation reverses key deficits in distinct rodent models of Parkinson’s disease. J. Neuroinflamm. 2008, 5, 19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, S.; Moon, M.; Park, S. Exendin-4 protects dopaminergic neurons by inhibition of microglial activation and matrix metalloproteinase-3 expression in an animal model of Parkinson’s disease. J. Endocrinol. 2009, 202, 431–439. [Google Scholar] [CrossRef] [Green Version]
- Athauda, D.; Maclagan, K.; Skene, S.S.; Bajwa-Joseph, M.; Letchford, D.; Chowdhury, K.; Hibbert, S.; Budnik, N.; Zampedri, L.; Dickson, J.; et al. Exenatide once weekly versus placebo in Parkinson’s disease: A randomised, double-blind, placebo-controlled trial. Lancet 2017, 390, 1664–1675. [Google Scholar] [CrossRef]
- Foltynie, T.; Athauda, D. Repurposing anti-diabetic drugs for the treatment of Parkinson’s disease: Rationale and clinical experience. Prog. Brain Res. 2020, 252, 493–523. [Google Scholar] [CrossRef]
- Harkavyi, A.; Rampersaud, N.; Whitton, P.S. Neuroprotection by Exendin-4 Is GLP-1 Receptor Specific but DA D3 Receptor Dependent, Causing Altered BrdU Incorporation in Subventricular Zone and Substantia Nigra. J. Neurodegener. Dis. 2013, 2013, 407152. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, L.; Zhang, L.; Li, L.; Holscher, C. Semaglutide is Neuroprotective and Reduces alpha-Synuclein Levels in the Chronic MPTP Mouse Model of Parkinson’s Disease. J. Parkinsons Dis. 2019, 9, 157–171. [Google Scholar] [CrossRef]
- Li, Y.; Tweedie, D.; Mattson, M.P.; Holloway, H.W.; Greig, N.H. Enhancing the GLP-1 receptor signaling pathway leads to proliferation and neuroprotection in human neuroblastoma cells. J. Neurochem. 2010, 113, 1621–1631. [Google Scholar] [CrossRef] [Green Version]
- Lee, C.S.; Cenci, M.A.; Schulzer, M.; Bjorklund, A. Embryonic ventral mesencephalic grafts improve levodopa-induced dyskinesia in a rat model of Parkinson’s disease. Brain 2000, 123, 1365–1379. [Google Scholar] [CrossRef] [Green Version]
- Lane, E.L.; Winkler, C.; Brundin, P.; Cenci, M.A. The impact of graft size on the development of dyskinesia following intrastriatal grafting of embryonic dopamine neurons in the rat. Neurobiol. Dis. 2006, 22, 334–345. [Google Scholar] [CrossRef] [PubMed]
- Smith, G.A.; Breger, L.S.; Lane, E.L.; Dunnett, S.B. Pharmacological modulation of amphetamine-induced dyskinesia in transplanted hemi-parkinsonian rats. Neuropharmacology 2012, 63, 818–828. [Google Scholar] [CrossRef] [PubMed]
- Talbot, K.; Wang, H.Y.; Kazi, H.; Han, L.Y.; Bakshi, K.P.; Stucky, A.; Fuino, R.L.; Kawaguchi, K.R.; Samoyedny, A.J.; Wilson, R.S.; et al. Demonstrated brain insulin resistance in Alzheimer’s disease patients is associated with IGF-1 resistance, IRS-1 dysregulation, and cognitive decline. J. Clin. Investig. 2012, 122, 1316–1338. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Acuna-Goycolea, C.; van den Pol, A. Glucagon-like peptide 1 excites hypocretin/orexin neurons by direct and indirect mechanisms: Implications for viscera-mediated arousal. J. Neurosci. 2004, 24, 8141–8152. [Google Scholar] [CrossRef]
- Li, Y.; Perry, T.; Kindy, M.S.; Harvey, B.K.; Tweedie, D.; Holloway, H.W.; Powers, K.; Shen, H.; Egan, J.M.; Sambamurti, K.; et al. GLP-1 receptor stimulation preserves primary cortical and dopaminergic neurons in cellular and rodent models of stroke and Parkinsonism. Proc. Natl. Acad. Sci. USA 2009, 106, 1285–1290. [Google Scholar] [CrossRef] [Green Version]
- Hamilton, A.; Holscher, C. Receptors for the incretin glucagon-like peptide-1 are expressed on neurons in the central nervous system. NeuroReport 2009, 20, 1161–1166. [Google Scholar] [CrossRef]
- Rampersaud, N.; Harkavyi, A.; Giordano, G.; Lever, R.; Whitton, J.; Whitton, P. Exendin-4 reverts behavioural and neurochemical dysfunction in a pre-motor rodent model of Parkinson’s disease with noradrenergic deficit. Br. J. Pharmacol. 2012, 167, 1467–1479. [Google Scholar] [CrossRef] [Green Version]
- Athauda, D.; Maclagan, K.; Budnik, N.; Zampedri, L.; Hibbert, S.; Aviles-Olmos, I.; Chowdhury, K.; Skene, S.S.; Limousin, P.; Foltynie, T. Post hoc analysis of the Exenatide-PD trial-Factors that predict response. Eur. J. Neurosci. 2019, 49, 410–421. [Google Scholar] [CrossRef]
- Rosenblad, C.; Martinez-Serrano, A.; Bjorklund, A. Glial cell line-derived neurotrophic factor increases survival, growth and function of intrastriatal fetal nigral dopaminergic grafts. Neuroscience 1996, 75, 979–985. [Google Scholar] [CrossRef]
- Takayama, H.; Ray, J.; Raymon, H.K.; Baird, A.; Hogg, J.; Fisher, L.J.; Gage, F.H. Basic fibroblast growth factor increases dopaminergic graft survival and function in a rat model of Parkinson’s disease. Nat. Med. 1995, 1, 53–58. [Google Scholar] [CrossRef]
- Karlsson, J.; Love, R.M.; Clarke, D.J.; Brundin, P. Effects of anaesthetics and lazaroid U-83836E on survival of transplanted rat dopaminergic neurones. Brain Res. 1999, 821, 546–550. [Google Scholar] [CrossRef]
- Badawi, G.A.; El Fattah, A.M.A.; Zaki, H.F.; El Sayed, M.I. Sitagliptin and liraglutide reversed nigrostriatal degeneration of rodent brain in rotenone-induced Parkinson’s disease. Inflammopharmacology 2017, 25, 369–382. [Google Scholar] [CrossRef] [PubMed]
- Hansen, H.H.; Fabricius, K.; Barkholt, P.; Mikkelsen, J.D.; Jelsing, J.; Pyke, C.; Knudsen, L.B.; Vrang, N. Characterization of liraglutide, a glucagon-like peptide-1 (GLP-1) receptor agonist, in rat partial and full nigral 6-hydroxydopamine lesion models of Parkinson’s disease. Brain Res. 2016, 1646, 354–365. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Z.; Li, D.; Feng, P.; Xue, G.; Ji, C.; Li, G.; Holscher, C. A novel GLP-1/GIP dual agonist is more effective than liraglutide in reducing inflammation and enhancing GDNF release in the MPTP mouse model of Parkinson’s disease. Eur. J. Pharmacol. 2017, 812, 82–90. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hagell, P.; Brundin, P. Cell survival and clinical outcome following intrastriatal transplantation in Parkinson disease. J. Neuropathol. Exp. Neurol. 2001, 60, 741–752. [Google Scholar] [CrossRef] [Green Version]
- Falkenstein, G.; Rosenthal, C.; Reum, T.; Morgenstern, R.; Dobrossy, M.; Nikkhah, G. Pattern of long-term sensorimotor recovery following intrastriatal and--accumbens DA micrografts in a rat model of Parkinson’s disease. J. Comp. Neurol. 2009, 515, 41–55. [Google Scholar] [CrossRef]
- Breger, L.S.; Kienle, K.; Smith, G.A.; Dunnett, S.B.; Lane, E.L. Influence of chronic L-DOPA treatment on immune response following allogeneic and xenogeneic graft in a rat model of Parkinson’s disease. Brain Behav. Immun. 2017, 61, 155–164. [Google Scholar] [CrossRef]
- Del-Bel, E.; Bortolanza, M.; Dos-Santos-Pereira, M.; Bariotto, K.; Raisman-Vozari, R. l-DOPA-induced dyskinesia in Parkinson’s disease: Are neuroinflammation and astrocytes key elements? Synapse 2016, 70, 479–500. [Google Scholar] [CrossRef]
- Brabazon, F.; Bermudez, S.; Shaughness, M.; Khayrullina, G.; Byrnes, K.R. The effects of insulin on the inflammatory activity of BV2 microglia. PLoS ONE 2018, 13, e0201878. [Google Scholar] [CrossRef] [Green Version]
- Kappe, C.; Tracy, L.M.; Patrone, C.; Iverfeldt, K.; Sjoholm, A. GLP-1 secretion by microglial cells and decreased CNS expression in obesity. J. Neuroinflamm. 2012, 9, 276. [Google Scholar] [CrossRef] [Green Version]
- Elabi, O.F.; Cunha, J.; Gaceb, A.; Fex, M.; Paul, G. High-fat diet-induced diabetes leads to vascular alterations, pericyte reduction, and perivascular depletion of microglia in a 6-OHDA toxin model of Parkinson disease. J. Neuroinflamm. 2021, 18, 175. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Zhai, Y.Q.; Xu, L.L.; Qiao, C.; Sun, X.L.; Ding, J.H.; Lu, M.; Hu, G. Metabolic inflammation exacerbates dopaminergic neuronal degeneration in response to acute MPTP challenge in type 2 diabetes mice. Exp. Neurol. 2014, 251, 22–29. [Google Scholar] [CrossRef] [PubMed]
- Perez-Taboada, I.; Alberquilla, S.; Martin, E.D.; Anand, R.; Vietti-Michelina, S.; Tebeka, N.N.; Cantley, J.; Cragg, S.J.; Moratalla, R.; Vallejo, M. Diabetes Causes Dysfunctional Dopamine Neurotransmission Favoring Nigrostriatal Degeneration in Mice. Mov. Disord. 2020, 35, 1636–1648. [Google Scholar] [CrossRef] [PubMed]
- Pagano, G.; Polychronis, S.; Wilson, H.; Giordano, B.; Ferrara, N.; Niccolini, F.; Politis, M. Diabetes mellitus and Parkinson disease. Neurology 2018, 90, e1654–e1662. [Google Scholar] [CrossRef] [PubMed]
- Chohan, H.; Senkevich, K.; Patel, R.K.; Bestwick, J.P.; Jacobs, B.M.; Bandres Ciga, S.; Gan-Or, Z.; Noyce, A.J. Type 2 Diabetes as a Determinant of Parkinson’s Disease Risk and Progression. Mov. Disord. 2021, 36, 1420–1429. [Google Scholar] [CrossRef]
- Sergi, D.; Renaud, J.; Simola, N.; Martinoli, M.G. Diabetes, a Contemporary Risk for Parkinson’s Disease: Epidemiological and Cellular Evidences. Front. Aging Neurosci. 2019, 11, 302. [Google Scholar] [CrossRef] [Green Version]
- Shinoda, M.; Hudson, J.L.; Stromberg, I.; Hoffer, B.J.; Moorhead, J.W.; Olson, L. Allogeneic grafts of fetal dopamine neurons: Immunological reactions following active and adoptive immunizations. Brain Res. 1995, 680, 180–195. [Google Scholar] [CrossRef]
- Hudson, J.L.; Hoffman, A.; Stromberg, I.; Hoffer, B.J.; Moorhead, J.W. Allogeneic grafts of fetal dopamine neurons: Behavioral indices of immunological interactions. Neurosci. Lett. 1994, 171, 32–36. [Google Scholar] [CrossRef]
- De Felice, F.G.; Ferreira, S.T. Inflammation, defective insulin signaling, and mitochondrial dysfunction as common molecular denominators connecting type 2 diabetes to Alzheimer disease. Diabetes 2014, 63, 2262–2272. [Google Scholar] [CrossRef] [Green Version]
- Carr, L.; Tucker, A.; Fernandez-Botran, R. The enhancement of T cell proliferation by L-dopa is mediated peripherally and does not involve interleukin-2. J. Neuroimmunol. 2003, 142, 166–169. [Google Scholar] [CrossRef]
- Mori, T.; Kabashima, K.; Fukamachi, S.; Kuroda, E.; Sakabe, J.; Kobayashi, M.; Nakajima, S.; Nakano, K.; Tanaka, Y.; Matsushita, S.; et al. D1-like dopamine receptors antagonist inhibits cutaneous immune reactions mediated by Th2 and mast cells. J. Dermatol. Sci. 2013, 71, 37–44. [Google Scholar] [CrossRef]
- Shiraishi, D.; Fujiwara, Y.; Komohara, Y.; Mizuta, H.; Takeya, M. Glucagon-like peptide-1 (GLP-1) induces M2 polarization of human macrophages via STAT3 activation. Biochem. Biophys. Res. Commun. 2012, 425, 304–308. [Google Scholar] [CrossRef]
- Abuirmeileh, A.; Harkavyi, A.; Rampersaud, N.; Lever, R.; Tadross, J.A.; Bloom, S.R.; Whitton, P.S. Exendin-4 treatment enhances L-DOPA evoked release of striatal dopamine and decreases dyskinetic movements in the 6-hydoxydopamine lesioned rat. J. Pharm. Pharmacol. 2012, 64, 637–643. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.J.; Chen, S.; Yang, Y.Y.; Glotfelty, E.J.; Jung, J.; Kim, H.K.; Choi, H.I.; Choi, D.S.; Hoffer, B.J.; Greig, N.H.; et al. PT320, Sustained-Release Exendin-4, Mitigates L-DOPA-Induced Dyskinesia in a Rat 6-Hydroxydopamine Model of Parkinson’s Disease. Front. Neurosci. 2020, 14, 785. [Google Scholar] [CrossRef] [PubMed]
- Yun, S.P.; Kam, T.I.; Panicker, N.; Kim, S.; Oh, Y.; Park, J.S.; Kwon, S.H.; Park, Y.J.; Karuppagounder, S.S.; Park, H.; et al. Block of A1 astrocyte conversion by microglia is neuroprotective in models of Parkinson’s disease. Nat. Med. 2018, 24, 931–938. [Google Scholar] [CrossRef]
- Bu, L.L.; Liu, Y.Q.; Shen, Y.; Fan, Y.; Yu, W.B.; Jiang, D.L.; Tang, Y.L.; Yang, Y.J.; Wu, P.; Zuo, C.T.; et al. Neuroprotection of Exendin-4 by Enhanced Autophagy in a Parkinsonian Rat Model of alpha-Synucleinopathy. Neurotherapeutics 2021, 18, 962–978. [Google Scholar] [CrossRef] [PubMed]
- Bergkvist, L.; Johnson, M.E.; Mercado, G.; Steiner, J.A.; Meyerdirk, L.; Schulz, E.; Madaj, Z.; Ma, J.; Becker, K.; Li, Y.; et al. An extended release GLP-1 analogue increases alpha-synuclein accumulation in a mouse model of prodromal Parkinson’s disease. Exp. Neurol. 2021, 341, 113693. [Google Scholar] [CrossRef]
- Li, W.; Englund, E.; Widner, H.; Mattsson, B.; van Westen, D.; Latt, J.; Rehncrona, S.; Brundin, P.; Bjorklund, A.; Lindvall, O.; et al. Extensive graft-derived dopaminergic innervation is maintained 24 years after transplantation in the degenerating parkinsonian brain. Proc. Natl. Acad. Sci. USA 2016, 113, 6544–6549. [Google Scholar] [CrossRef] [Green Version]
- Kordower, J.H.; Goetz, C.G.; Chu, Y.; Halliday, G.M.; Nicholson, D.A.; Musial, T.F.; Marmion, D.J.; Stoessl, A.J.; Sossi, V.; Freeman, T.B.; et al. Robust graft survival and normalized dopaminergic innervation do not obligate recovery in a Parkinson disease patient. Ann. Neurol. 2017, 81, 46–57. [Google Scholar] [CrossRef]
- Torres, E.M.; Lane, E.L.; Heuer, A.; Smith, G.A.; Murphy, E.; Dunnett, S.B. Increased efficacy of the 6-hydroxydopamine lesion of the median forebrain bundle in small rats, by modification of the stereotaxic coordinates. J. Neurosci. Methods 2011, 200, 29–35. [Google Scholar] [CrossRef]
- Ungerstedt, U. 6-Hydroxy-dopamine induced degeneration of central monoamine neurons. Eur. J. Pharmacol. 1968, 5, 107–110. [Google Scholar] [CrossRef]
- Dunnett, S.B.; Björklund, A. Dissecting Embryonic Neural Tissues for Transplantation. In Neural Transplantation Methods; Dunnett, S.B., Boulton, A.A., Baker, G.B., Eds.; Humana Press: Totowa, NJ, USA, 2000; pp. 3–25. [Google Scholar]
- Bjorklund, A.; Stenevi, U.; Schmidt, R.H.; Dunnett, S.B.; Gage, F.H. Intracerebral grafting of neuronal cell suspensions. I. Introduction and general methods of preparation. Acta Physiol. Scand. Suppl. 1983, 522, 1–7. [Google Scholar]
- Torres, E.M.; Monville, C.; Gates, M.A.; Bagga, V.; Dunnett, S.B. Improved survival of young donor age dopamine grafts in a rat model of Parkinson’s disease. Neuroscience 2007, 146, 1606–1617. [Google Scholar] [CrossRef] [PubMed]
- McClean, P.L.; Gault, V.A.; Harriott, P.; Holscher, C. Glucagon-like peptide-1 analogues enhance synaptic plasticity in the brain: A link between diabetes and Alzheimer’s disease. Eur. J. Pharmacol. 2010, 630, 158–162. [Google Scholar] [CrossRef]
- Ungerstedt, U.; Arbuthnott, G.W. Quantitative recording of rotational behavior in rats after 6-hydroxy-dopamine lesions of the nigrostriatal dopamine system. Brain Res. 1970, 24, 485–493. [Google Scholar] [CrossRef]
- Kirik, D.; Rosenblad, C.; Bjorklund, A. Characterization of behavioral and neurodegenerative changes following partial lesions of the nigrostriatal dopamine system induced by intrastriatal 6-hydroxydopamine in the rat. Exp. Neurol. 1998, 152, 259–277. [Google Scholar] [CrossRef] [PubMed]
- Schallert, T.; Fleming, S.M.; Leasure, J.L.; Tillerson, J.L.; Bland, S.T. CNS plasticity and assessment of forelimb sensorimotor outcome in unilateral rat models of stroke, cortical ablation, parkinsonism and spinal cord injury. Neuropharmacology 2000, 39, 777–787. [Google Scholar] [CrossRef]
- Winkler, C.; Kirik, D.; Bjorklund, A.; Cenci, M.A. L-DOPA-induced dyskinesia in the intrastriatal 6-hydroxydopamine model of parkinson’s disease: Relation to motor and cellular parameters of nigrostriatal function. Neurobiol. Dis. 2002, 10, 165–186. [Google Scholar] [CrossRef]
- Breger, L.S.; Dunnett, S.B.; Lane, E.L. Comparison of rating scales used to evaluate L-DOPA-induced dyskinesia in the 6-OHDA lesioned rat. Neurobiol. Dis. 2013, 50, 142–150. [Google Scholar] [CrossRef]
- MILLIPLEX® MAP. Rat Metabolic Magnetic Bead Panel Kit 96 Well Plate Assay. Available online: https://filgen.jp/Product/Bioscience19-Bioplex/RMHMAG-84K.MPX.pdf (accessed on 20 September 2021).
- Matthews, D.R.; Hosker, J.P.; Rudenski, A.S.; Naylor, B.A.; Treacher, D.F.; Turner, R.C. Homeostasis model assessment: Insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985, 28, 412–419. [Google Scholar] [CrossRef] [Green Version]
- Hedreen, J.C. What was wrong with the Abercrombie and empirical cell counting methods? A review. Anat. Rec. 1998, 250, 373–380. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Elabi, O.F.; Davies, J.S.; Lane, E.L. L-dopa-Dependent Effects of GLP-1R Agonists on the Survival of Dopaminergic Cells Transplanted into a Rat Model of Parkinson Disease. Int. J. Mol. Sci. 2021, 22, 12346. https://doi.org/10.3390/ijms222212346
Elabi OF, Davies JS, Lane EL. L-dopa-Dependent Effects of GLP-1R Agonists on the Survival of Dopaminergic Cells Transplanted into a Rat Model of Parkinson Disease. International Journal of Molecular Sciences. 2021; 22(22):12346. https://doi.org/10.3390/ijms222212346
Chicago/Turabian StyleElabi, Osama F., Jeffrey S. Davies, and Emma L. Lane. 2021. "L-dopa-Dependent Effects of GLP-1R Agonists on the Survival of Dopaminergic Cells Transplanted into a Rat Model of Parkinson Disease" International Journal of Molecular Sciences 22, no. 22: 12346. https://doi.org/10.3390/ijms222212346






