Metal Sulfide Semiconductor Nanomaterials and Polymer Microgels for Biomedical Applications
Abstract
:1. Introduction
2. Synthesis of Nanomaterials
2.1. The Sol-Gel Method
2.2. Hydrothermal/Solvothermal Method
2.3. Vacuum Deposition Method
2.4. Ball Milling Method
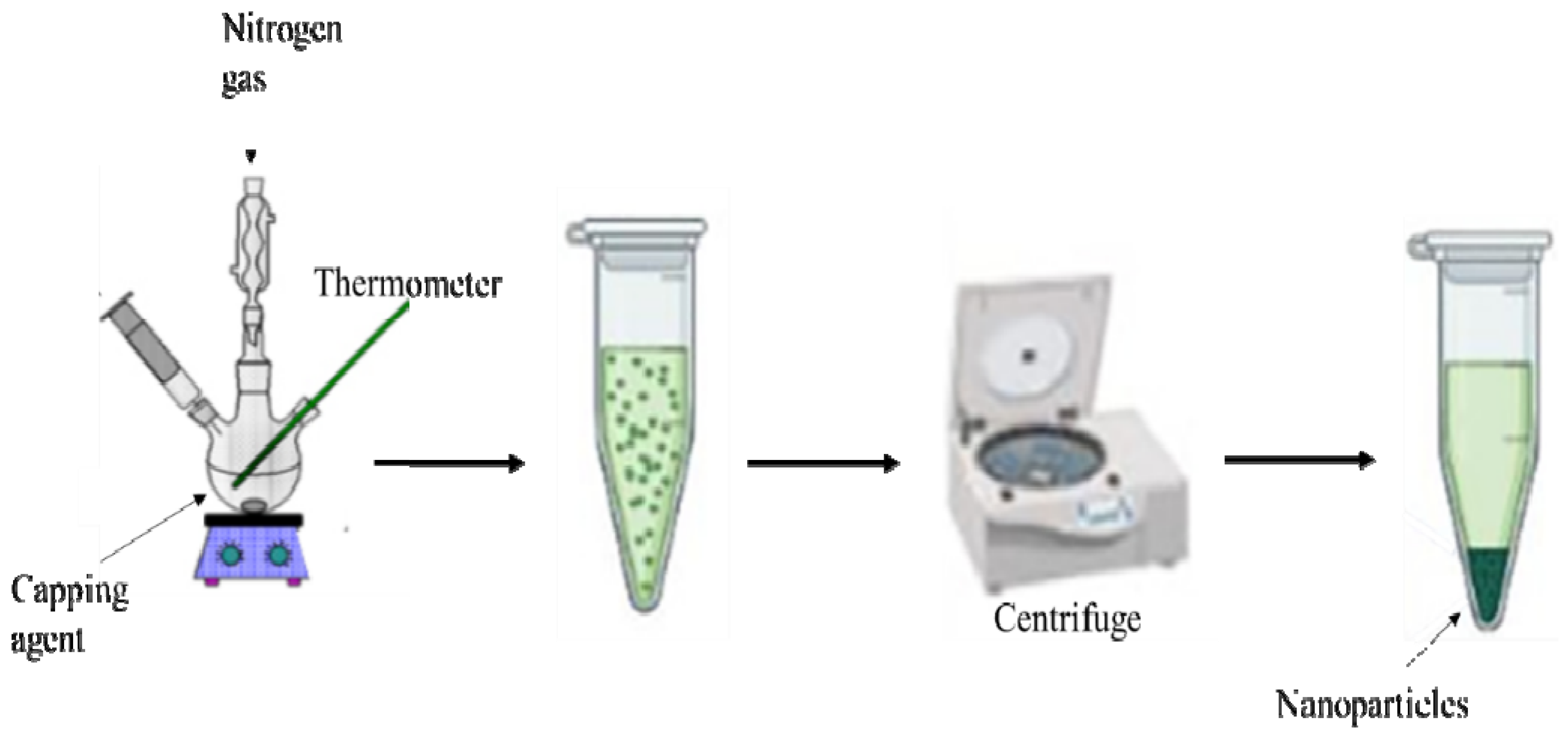
2.5. Thermolysis
3. Nanomaterials in Biomedicine
Metal Sulfide Nanomaterials in Cancer Therapy
4. Drug Delivery
4.1. Polymer Microgels as Drug Delivery Systems
4.2. Synthesis of Microgels
Precipitation Polymerization Method
4.3. Hybrid Metal Sulfide-Microgels
4.4. Polymer Nanofibers
4.4.1. Fabrication of Nanofibers
4.4.2. Formation of Nanofiber by Electrospinning
5. Conclusions
Author Contributions
Funding
Informed Consent Statement
Conflicts of Interest
References
- Boisseau, P.; Loubaton, B. Nanomedicine, nanotechnology in medicine. Comptes Rendus Physique 2011, 12, 620–636. [Google Scholar] [CrossRef] [Green Version]
- Khan, I.; Saeed, K.; Khan, I. Nanoparticles: Properties, applications and toxicities. Arab. J. Chem. 2019, 12, 908–931. [Google Scholar] [CrossRef]
- Gajanan, K.; Tijare, S. Applications of nanomaterials. Mater. Today Proc. 2018, 5, 1093–1096. [Google Scholar] [CrossRef]
- Pan, Y.; Bartneck, M.; Jahnen-Dechent, W. Cytotoxicity of gold nanoparticles. In Methods in Enzymology; Elsevier: Amsterdam, The Netherlands, 2012; Volume 509, pp. 225–242. [Google Scholar]
- Nayak, M.K.; Singh, J.; Singh, B.; Soni, S.; Pandey, V.S.; Tyagi, S. Metal Semiconductor Core-Shell Nanostructures for Energy and Environmental Applications; Elsevier: Amsterdam, The Netherlands, 2017; pp. 1–33. [Google Scholar]
- Bhatia, S. Natural Polymer Drug Delivery Systems; Springer: New York, NY, USA, 2016; pp. 1–33. [Google Scholar]
- Cartaxo, A.L.P. Nanoparticles types and properties–understanding these promising devices in the biomedical area. Int. J. Nanomed. 2018, 1–8. [Google Scholar]
- Reghunadhan, A.; Kalarikkal, N.; Thomas, S. Characterization of Nanomaterials; Elsevier: Amsterdam, The Netherlands, 2018; pp. 191–212. [Google Scholar]
- Gaiardo, A.; Fabbri, B.; Guidi, V.; Bellutti, P.; Giberti, A.; Gherardi, S.; Vanzetti, L.; Malagù, C.; Zonta, G. Metal sulfides as sensing materials for chemoresistive gas sensors. Sensors 2016, 16, 296. [Google Scholar] [CrossRef] [PubMed]
- Bobo, D.; Robinson, K.J.; Islam, J.; Thurecht, K.J.; Corrie, S.R. Nanoparticle-based medicines: A review of FDA-approved materials and clinical trials to date. Pharm. Res. 2016, 33, 2373–2387. [Google Scholar] [CrossRef] [PubMed]
- Rao, C.; Pisharody, K. Transition metal sulfides. Prog. Solid State Chem. 1976, 10, 207–270. [Google Scholar] [CrossRef]
- Gross, S.; Vittadini, A.; Dengo, N. Functionalisation of Colloidal Transition Metal Sulphides Nanocrystals: A Fascinating and Challenging Playground for the Chemist. Crystals 2017, 7, 110. [Google Scholar] [CrossRef] [Green Version]
- Lai, C.-H.; Lu, M.-Y.; Chen, L.-J. Metal sulfide nanostructures: Synthesis, properties and applications in energy conversion and storage. J. Mater. Chem. 2012, 22, 19–30. [Google Scholar] [CrossRef]
- Owens, G.J.; Singh, R.K.; Foroutan, F.; Alqaysi, M.; Han, C.-M.; Mahapatra, C.; Kim, H.-W.; Knowles, J.C. Sol-gel based materials for biomedical applications. Prog. Mater. Sci. 2016, 77, 1–79. [Google Scholar] [CrossRef] [Green Version]
- Lue, J.T. Physical properties of nanomaterials. Encycl. Nanosci. Nanotechnol. 2007, 10, 1–46. [Google Scholar]
- Khan, M.F.; Ansari, A.H.; Hameedullah, M.; Ahmad, E.; Husain, F.M.; Zia, Q.; Baig, U.; Zaheer, M.R.; Alam, M.M.; Khan, A.M. Sol-gel synthesis of thorn-like ZnO nanoparticles endorsing mechanical stirring effect and their antimicrobial activities: Potential role as nano-antibiotics. Sci. Rep. 2016, 6, 1–12. [Google Scholar] [CrossRef]
- Pudukudy, M.; Yaakob, Z.; Mazuki, M.Z.; Takriff, M.S.; Jahaya, S.S. One-pot sol-gel synthesis of MgO nanoparticles supported nickel and iron catalysts for undiluted methane decomposition into COx free hydrogen and nanocarbon. Appl. Catal. B 2017, 218, 298–316. [Google Scholar] [CrossRef]
- Thilagavathi, P.; Manikandan, A.; Sujatha, S.; Jaganathan, S.K.; Arul Antony, S. Sol-gel synthesis and characterization studies of NiMoO4 nanostructures for photocatalytic degradation of methylene blue dye. Nanosci. Nanotechnol. Lett. 2016, 8, 438–443. [Google Scholar] [CrossRef]
- Zorkipli, N.N.M.; Kaus, N.H.M.; Mohamad, A.A. Synthesis of NiO nanoparticles through sol-gel method. Procedia Chem. 2016, 19, 626–631. [Google Scholar] [CrossRef] [Green Version]
- Li, J.; Wu, Q.; Wu, J. Synthesis of Nanoparticles via Solvothermal and Hydrothermal Methods. In Handbook of Nanoparticles; Springer International Publishing: Cham, Switzerland, 2015; pp. 1–28. [Google Scholar]
- Camargo, P.H.C.; Satyanarayana, K.G.; Wypych, F. Nanocomposites: Synthesis, structure, properties and new application opportunities. Mat. Res. 2009, 12, 1–39. [Google Scholar] [CrossRef] [Green Version]
- Ajibade, P.A.; Botha, N.L. Synthesis and structural studies of copper sulfide nanocrystals. Res. Phys. 2016, 6, 581–589. [Google Scholar] [CrossRef] [Green Version]
- Paca, A.M.; Ajibade, P.A. Synthesis and structural studies of iron sulphide nanocomposites prepared from Fe(III) dithiocarbamates single source precursors. Mater. Chem. Phys. 2017, 202, 143–150. [Google Scholar] [CrossRef]
- Ajibade, P.A.; Oluwalana, A.E.; Andrew, F.P. Morphological studies, photocatalytic activity, and electrochemistry of platinum disulfide nanoparticles from bis(morpholinyl-4-carbodithioato)-platinum(II). ACS Omega 2020, 5, 27142–27153. [Google Scholar] [CrossRef]
- Paca, A.M.; Ajibade, P.A. Bis-(N-ethylphenyldithiocarbamato) palladium(II) as molecular precursor for palladium sulfide nanoparticles. J. Mol. Struct. 2021, 1243, 130777. [Google Scholar] [CrossRef]
- Paca, A.M.; Ajibade, P.A. Effect of temperature on structural and optical properties of iron sulfide nanocrystals prepared from tris(N-methylbenzyldithiocarbamato)iron(III) complex. Inorg. Nano-Met. Chem. 2021, 51, 322–331. [Google Scholar] [CrossRef]
- Angeloski, A.; Gentle, A.R.; Scott, J.A.; Cortie, M.B.; Hook, J.M.; Westerhausen, M.T.; Bhadbhade, M.; Baker, A.T.; McDonagh, A.M. From lead(II) dithiocarbamate precursors to a fast response PbS positive temperature coefficient thermistor. Inorg. Chem. 2018, 57, 2132–2140. [Google Scholar] [CrossRef] [PubMed]
- Ajibade, P.A.; Oluwalana, A.E.; Sikakane, B.M.; Singh, M. Structural, photocatalytic and anticancer studies of hexadecylamine capped ZnS nanoparticles. Chem. Phys. Lett. 2020, 755, 137813. [Google Scholar] [CrossRef]
- Boncher, W.L.; Regulacio, M.D.; Stoll, S.L. Thermolysis of lanthanide dithiocarbamate complexes. J. Solid State Chem. 2010, 183, 52–56. [Google Scholar] [CrossRef]
- Pradhan, N.; Efrima, S. Single-precursor, one-pot versatile synthesis under near ambient conditions of tunable, single and dual band fluorescing metal sulfide nanoparticles. J. Am. Chem. Soc. 2003, 125, 2050–2051. [Google Scholar] [CrossRef]
- Khan, A.; Hayat, F.; Butler, I.S.; Tahir, M.N. Mercury(II) dithiocarbamates: Structural aspects and their use as single source precursors for shape-controlled facile synthesis of HgS nanoparticles. Polyhedron 2020, 193, 1–33. [Google Scholar] [CrossRef]
- Arandhara, G.; Mostako, A.; Dutta, P.; Bora, J.; Saikia, P. Influence of thermolysis temperature on the morphology, structural and optical properties of nanocomposite ZnS-polyvinyl alcohol thin films: Fabrication and characterization of indium tin oxide/ZnS-polyvinyl alcohol/Al Schottky diode. Thin Solid Films 2020, 712, 138317. [Google Scholar] [CrossRef]
- Ul Ain, N.; Aamir, A.; Khan, Y.; Rehman, M.-u.; Lin, D.-J. Catalytic and photocatalytic efficacy of hexagonal CuS nanoplates derived from copper(II) dithiocarbamate. Mater. Chem. Phys. 2020, 242, 122408. [Google Scholar] [CrossRef]
- Cho, K.; Wang, X.; Nie, S.; Shin, D.M. Therapeutic nanoparticles for drug delivery in cancer. Clin. Cancer Res. 2008, 14, 1310–1316. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wong, I.Y.; Bhatia, S.N.; Toner, M. Nanotechnology: Emerging tools for biology and medicine. Genes Dev. 2013, 27, 2397–2408. [Google Scholar] [CrossRef] [Green Version]
- Banik, B.L.; Brown, J.L. Natural and Synthetic Biomedical Polymers; Elsevier: Amsterdam, The Netherlands, 2014; pp. 387–395. [Google Scholar]
- Argueta-Figueroa, L.; Martinez-Alvarez, O.; Santos-Cruz, J.; Garcia-Contreras, R.; Acosta-Torres, L.; De la Fuente-Hernandez, J.; Arenas-Arrocena, M. Nanomaterials made of non-toxic metallic sulfides: A systematic review of their potential biomedical applications. Mater. Sci. Eng. C 2017, 76, 1305–1315. [Google Scholar] [CrossRef] [PubMed]
- Kolahalam, L.A.; Viswanath, I.K.; Diwakar, B.S.; Govindh, B.; Reddy, V.; Murthy, Y. Review on nanomaterials: Synthesis and applications. Mater. Today 2019, 18, 2182–2190. [Google Scholar] [CrossRef]
- Li, C.; Wang, Y.; Jiang, H.; Wang, X. Biosensors Based on Advanced Sulfur-Containing Nanomaterials. Sensors 2020, 20, 3488. [Google Scholar] [CrossRef]
- Zhou, H.; Ge, J.; Miao, Q.; Zhu, R.; Wen, L.; Zeng, J.; Gao, M. Biodegradable Inorganic Nanoparticles for Cancer Theranostics: Insights into the Degradation Behavior. Bioconjugate Chem. 2019, 31, 315–331. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.; Wang, C.; Peng, Z.; Qi, K.; Guo, Z.; Zhang, Y.; Zhang, H. The chemistry of colloidal semiconductor nanocrystals: From metal-chalcogenides to emerging perovskite. Coord. Chem. Rev. 2020, 418, 1–20. [Google Scholar] [CrossRef]
- Yang, W.; Guo, W.; Chang, J.; Zhang, B. Protein/peptide-templated biomimetic synthesis of inorganic nanoparticles for biomedical applications. J. Mater. Chem. B 2017, 5, 401–417. [Google Scholar] [CrossRef] [PubMed]
- Yan, H.; Chen, J.; Li, Y.; Bai, Y.; Wu, Y.; Sheng, Z.; Song, L.; Liu, C.; Zhang, H. Ultrasmall hybrid protein–copper sulfide nanoparticles for targeted photoacoustic imaging of orthotopic hepatocellular carcinoma with a high signal-to-noise ratio. Biomater. Sci. 2019, 7, 92–103. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.; Chen, F.; He, S.; Shen, H.; Hu, Y.; Feng, N.; Wang, S.; Weng, L.; Luo, Z.; Wang, L. One-pot growth of triangular SnS nanopyramids for photoacoustic imaging and photothermal ablation of tumors. New J. Chem. 2019, 43, 13256–13262. [Google Scholar] [CrossRef]
- Wang, Y.; Wu, Y.; Liu, Y.; Shen, J.; Lv, L.; Li, L.; Yang, L.; Zeng, J.; Wang, Y.; Zhang, L.W. BSA-mediated synthesis of bismuth sulfide nanotheranostic agents for tumor multimodal imaging and thermoradiotherapy. Adv. Funct. Mater. 2016, 26, 5335–5344. [Google Scholar] [CrossRef]
- Lin, S.; Wang, Y.; Chen, Z.; Li, L.; Zeng, J.; Dong, Q.; Wang, Y.; Chai, Z. Biomineralized enzyme-like cobalt sulfide nanodots for synergetic phototherapy with tumor multimodal imaging navigation. ACS Sustain. Chem. Eng. 2018, 6, 12061–12069. [Google Scholar] [CrossRef]
- Li, Y.; Lu, W.; Huang, Q.; Li, C.; Chen, W. Copper sulfide nanoparticles for photothermal ablation of tumor cells. Nanomedicine 2010, 5, 1161–1171. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fan, J.-X.; Liu, M.-D.; Li, C.-X.; Hong, S.; Zheng, D.-W.; Liu, X.-H.; Chen, S.; Cheng, H.; Zhang, X.-Z. A metal–semiconductor nanocomposite as an efficient oxygen-independent photosensitizer for photodynamic tumor therapy. Nanoscale Horiz. 2017, 2, 349–355. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.; Yang, Y.; Jiang, T.; Zhang, X.; Zhao, Y.; Pang, G.; Feng, Y.; Zhang, S.; Wang, F.; Wang, Y. Effective Radiotherapy in Tumor Assisted by Ganoderma lucidum Polysaccharide-Conjugated Bismuth Sulfide Nanoparticles through Radiosensitization and Dendritic Cell Activation. ACS Appl. Mater. Inter. 2019, 11, 27536–27547. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Dong, H.; Li, M.; Cao, Y.; Yang, F.; Zhang, K.; Dai, W.; Wang, C.; Zhang, X. Erythrocyte–cancer hybrid membrane camouflaged hollow copper sulfide nanoparticles for prolonged circulation life and homotypic-targeting photothermal/chemotherapy of melanoma. ACS Nano 2018, 12, 5241–5252. [Google Scholar] [CrossRef]
- Sakthivel, R.; Kubendhiran, S.; Chen, S.-M.; Chen, T.-W.; Al-Zaqri, N.; Alsalme, A.; Alharthi, F.A.; Khanjer, M.M.A.; Tseng, T.-W.; Huang, C.-C. Exploring the promising potential of MoS2–RuS2 binary metal sulphide towards the electrocatalysis of antibiotic drug sulphadiazine. Anal. Chim. Acta 2019, 1086, 55–65. [Google Scholar] [CrossRef] [PubMed]
- Sangili, A.; Sakthivel, R.; Chen, S.M. Cost-effective single-step synthesis of flower-like cerium-ruthenium-sulfide for the determination of antipsychotic drug trifluoperazine in human urine samples. Anal. Chim. Acta 2020, 1131, 35–44. [Google Scholar] [CrossRef] [PubMed]
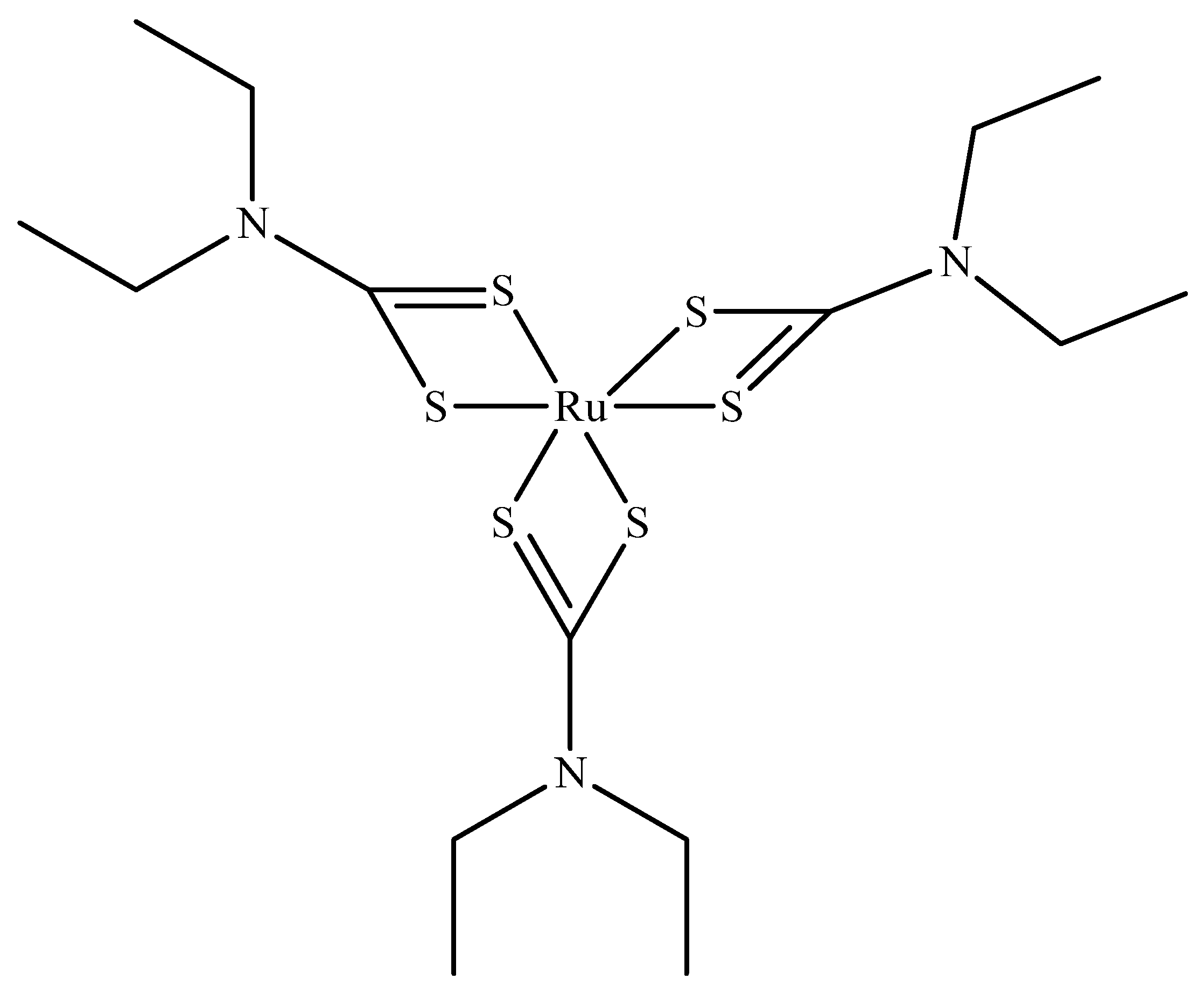
- Lu, Z.; Huang, F.-y.; Cao, R.; Zhang, L.; Tan, G.-h.; He, N.; Huang, J.; Wang, G.; Zhang, Z. Long blood residence and large tumor uptake of ruthenium sulfide nanoclusters for highly efficient cancer photothermal therapy. Sci. Rep. 2017, 7, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Xie, Z.; Wang, D.; Fan, T.; Xing, C.; Li, Z.; Tao, W.; Liu, L.; Bao, S.; Fan, D.; Zhang, H. Black phosphorus analogue tin sulfide nanosheets: Synthesis and application as near-infrared photothermal agents and drug delivery platforms for cancer therapy. J. Mater. Chem. B 2018, 6, 4747–4755. [Google Scholar] [CrossRef]
- Han, L.; Hao, Y.-N.; Wei, X.; Chen, X.-W.; Shu, Y.; Wang, J.-H. Hollow copper sulfide nanosphere–doxorubicin/graphene oxide core–shell nanocomposite for photothermo-chemotherapy. ACS Biomater. Sci. Eng. 2017, 3, 3230–3235. [Google Scholar] [CrossRef] [PubMed]
- Hou, L.; Shan, X.; Hao, L.; Feng, Q.; Zhang, Z. Copper sulfide nanoparticle-based localized drug delivery system as an effective cancer synergistic treatment and theranostic platform. Acta Biomater. 2017, 54, 307–320. [Google Scholar] [CrossRef] [PubMed]
- Lavik, E.B.; Kuppermann, B.D.; Humayun, M.S. Retina, 5th ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2012. [Google Scholar]
- Wu, W.; Zhou, T.; Berliner, A.; Banerjee, P.; Zhou, S. Smart core−shell hybrid nanogels with Ag nanoparticle core for cancer cell imaging and gel shell for pH-regulated drug delivery. Chem. Mater. 2010, 22, 1966–1976. [Google Scholar] [CrossRef]
- Kheirandish, S.; Ghaedi, M.; Dashtian, K.; Pourebrahim, F.; Jannesar, R.; Pezeshkpour, V. In vitro curcumin delivery and antibacterial activity of RuS2 and RuO2 nanoparticles loaded chitosan biopolymer. Appl. Organomet. Chem. 2018, 32, e4035. [Google Scholar] [CrossRef]
- Torchilin, V.P. Stimuli-Responsive Drug Delivery Systems; Royal Society of Chemistry: London, UK, 2018. [Google Scholar]
- Peer, D.; Karp, J.M.; Hong, S.; Farokhzad, O.C.; Margalit, R.; Langer, R. Nanocarriers as an emerging platform for cancer therapy. Nat. Nanotechnol. 2007, 2, 751. [Google Scholar] [CrossRef] [PubMed]
- Torchilin, V.P. Multifunctional nanocarriers. Adv. Drug Deliv. Rev. 2006, 58, 1532–1555. [Google Scholar] [CrossRef] [PubMed]
- Farokhzad, O.C.; Langer, R. Impact of nanotechnology on drug delivery. ACS Nano 2009, 3, 16–20. [Google Scholar] [CrossRef]
- Owens, D.E., III; Peppas, N.A. Opsonization, biodistribution, and pharmacokinetics of polymeric nanoparticles. Int. J. Pharm. Sci. Res. 2006, 307, 93–102. [Google Scholar] [CrossRef]
- De Jong, W.H.; Borm, P.J. Drug delivery and nanoparticles: Applications and hazards. Int. J. Nanomed. 2008, 3, 133–149. [Google Scholar] [CrossRef] [Green Version]
- Zhuang, J.; Gordon, M.R.; Ventura, J.; Li, L.; Thayumanavan, S. Multi-stimuli responsive macromolecules and their assemblies. Chem. Soc. Rev. 2013, 42, 7421–7435. [Google Scholar] [CrossRef] [PubMed]
- Abulateefeh, S.R.; Spain, S.G.; Aylott, J.W.; Chan, W.C.; Garnett, M.C.; Alexander, C. Thermoresponsive polymer colloids for drug delivery and cancer therapy. Macromol. Biosci. 2011, 11, 1722–1734. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Torchilin, V.P. Stimulus-responsive nanopreparations for tumor targeting. Integr. Biol. 2013, 5, 96–107. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, G.; Agrawal, R. Stimuli-Responsive Microgels and Microgel-Based Systems: Advances in the Exploitation of Microgel Colloidal Properties and Their Interfacial Activity. Polymers 2018, 10, 418. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Smeets, N.M.; Hoare, T. Designing responsive microgels for drug delivery applications. J. Polym. Sci. Part A Polym. Chem. 2013, 51, 3027–3043. [Google Scholar] [CrossRef]
- Vinogradov, S.V. Colloidal microgels in drug delivery applications. Curr. Pharm. Des. 2006, 12, 4703–4712. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oh, J.K.; Drumright, R.; Siegwart, D.J.; Matyjaszewski, K. The development of microgels/nanogels for drug delivery applications. Prog. Polym. Sci. 2008, 33, 448–477. [Google Scholar] [CrossRef]
- Bai, C.; Fang, Y.; Zhang, Y.; Chen, B. Synthesis of novel metal sulfide−polymer composite microspheres exhibiting patterned surface structures. Langmuir 2004, 20, 263–265. [Google Scholar] [CrossRef]
- Thomas, V.; Namdeo, M.; Murali Mohan, Y.; Bajpai, S.; Bajpai, M. Review on polymer, hydrogel and microgel metal nanocomposites: A facile nanotechnological approach. J. Macromol. Sci. Part A Pure Appl. Chem. 2007, 45, 107–119. [Google Scholar] [CrossRef]
- Nachev, P.; Van’t Zand, D.D.; Coger, V.; Wagener, P.; Reimers, K.; Vogt, P.M.; Barcikowski, S.; Pich, A. Synthesis of hybrid microgels by coupling of laser ablation and polymerization in aqueous medium. J. Laser Appl. 2012, 24, 1–7. [Google Scholar] [CrossRef]
- Zhang, J.; Xu, S.; Kumacheva, E. Polymer microgels: Reactors for semiconductor, metal, and magnetic nanoparticles. J. Am. Chem. Soc. 2004, 126, 7908–7914. [Google Scholar] [CrossRef] [PubMed]
- Gong, Y.; Gao, M.; Wang, D.; Möhwald, H. Incorporating fluorescent CdTe nanocrystals into a hydrogel via hydrogen bonding: Toward fluorescent microspheres with temperature-responsive properties. Chem. Mater. 2005, 17, 2648–2653. [Google Scholar] [CrossRef]
- Pich, A.; Zhang, F.; Shen, L.; Berger, S.; Ornatsky, O.; Baranov, V.; Winnik, M.A. Biocompatible hybrid nanogels. Small 2008, 4, 2171–2175. [Google Scholar] [CrossRef]
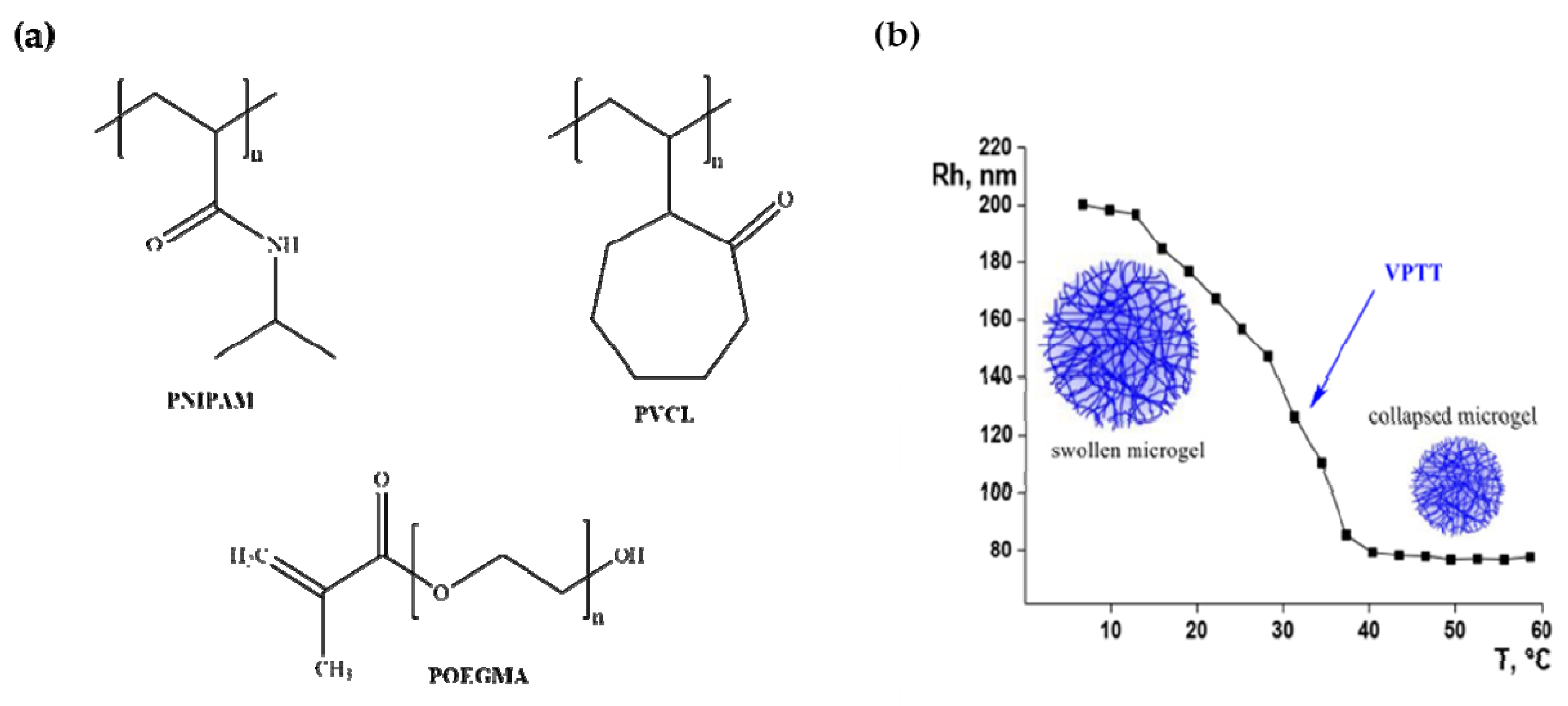
- Lutz, J.F. Thermo-switchable materials prepared using the OEGMA-platform. Adv. Mater. 2011, 23, 2237–2243. [Google Scholar] [CrossRef]
- Ramos, J.; Imaz, A.; Forcada, J. Temperature-sensitive nanogels: Poly(N-vinylcaprolactam) versus poly(N-isopropylacrylamide). Polym. Chem. 2012, 3, 852–856. [Google Scholar] [CrossRef]
- Bebis, K.; Jones, M.W.; Haddleton, D.M.; Gibson, M.I. Thermoresponsive behaviour of poly((oligo (ethyleneglycol methacrylate)) s and their protein conjugates: Importance of concentration and solvent system. Polym. Chem. 2011, 2, 975–982. [Google Scholar] [CrossRef]
- Lima, L.H.; Morales, Y.; Cabral, T. Ocular Biocompatibility of Poly-N-Isopropylacrylamide (pNIPAM). J. Ophthalmol. 2016, 2016. [Google Scholar] [CrossRef] [Green Version]
- Haq, M.A.; Su, Y.; Wang, D. Mechanical properties of PNIPAM based hydrogels: A review. Mater. Sci. Eng. C 2017, 70, 842–855. [Google Scholar] [CrossRef] [PubMed]
- Daly, E.; Saunders, B.R. A study of the effect of electrolyte on the swelling and stability of poly(N-isopropylacrylamide) microgel dispersions. Langmuir 2000, 16, 5546–5552. [Google Scholar] [CrossRef]
- Singh, R.; Deshmukh, S.A.; Kamath, G.; Sankaranarayanan, S.K.; Balasubramanian, G. Controlling the aqueous solubility of PNIPAM with hydrophobic molecular units. Comp. Mater. Sci. 2017, 126, 191–203. [Google Scholar] [CrossRef] [Green Version]
- Pelton, R. Temperature-sensitive aqueous microgels. Adv. Colloid Interface Sci. 2000, 85, 1–33. [Google Scholar] [CrossRef]
- Jung, J.; Lee, I.-H.; Lee, E.; Park, J.; Jon, S. pH-sensitive polymer nanospheres for use as a potential drug delivery vehicle. Biomacromolecules 2007, 8, 3401–3407. [Google Scholar] [CrossRef] [PubMed]
- Shi, L.; Khondee, S.; Linz, T.H.; Berkland, C. Poly(N-vinylformamide) nanogels capable of pH-sensitive protein release. Macromolecules 2008, 41, 6546–6554. [Google Scholar] [CrossRef]
- Hu, Y.; Atukorale, P.U.; Lu, J.J.; Moon, J.J.; Um, S.H.; Cho, E.C.; Wang, Y.; Chen, J.; Irvine, D.J. Cytosolic delivery mediated via electrostatic surface binding of protein, virus, or siRNA cargos to pH-responsive core−shell gel particles. Biomacromolecules 2009, 10, 756–765. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stubbs, M.; McSheehy, P.M.; Griffiths, J.R.; Bashford, C.L. Causes and consequences of tumour acidity and implications for treatment. Mol. Med. Today 2000, 6, 15–19. [Google Scholar] [CrossRef]
- Torchilin, V.P. Micellar nanocarriers: Pharmaceutical perspectives. Pharm. Res. 2007, 24, 1. [Google Scholar] [CrossRef] [PubMed]
- Coakley, R.D.; Grubb, B.R.; Paradiso, A.M.; Gatzy, J.T.; Johnson, L.G.; Kreda, S.M.; O’Neal, W.K.; Boucher, R.C. Abnormal surface liquid pH regulation by cultured cystic fibrosis bronchial epithelium. Proc. Natl. Acad. Sci. USA 2003, 100, 16083–16088. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Muratalin, M.; Luckham, P.F.; Esimova, A.; Aidarova, S.; Mutaliyeva, B.; Madybekova, G.; Sharipova, A.; Issayeva, A. Study of N-isopropylacrylamide-based microgel particles as a potential drug delivery agents. Colloids Surf. A 2017, 532, 8–17. [Google Scholar] [CrossRef]
- Khan, J.; Siddiq, M.; Akram, B.; Ashraf, M.A. In-situ synthesis of CuO nanoparticles in P (NIPAM-co-AAA) microgel, structural characterization, catalytic and biological applications. Arab. J. Chem. 2018, 11, 897–909. [Google Scholar] [CrossRef]
- Farooqi, Z.H.; Khan, S.R.; Begum, R.; Ijaz, A. Review on synthesis, properties, characterization, and applications of responsive microgels fabricated with gold nanostructures. Rev. Chem. Eng. 2016, 32, 49–69. [Google Scholar] [CrossRef]
- Pich, A.; Richtering, W. Chemical Design of Responsive Microgels; Springer: New York, NY, USA, 2010; pp. 1–37. [Google Scholar]
- Duracher, D.; Elaïssari, A.; Pichot, C. Preparation of poly(N-isopropylmethacrylamide) latexes kinetic studies and characterization. J. Polym. Sci. Part A Polym. Chem. 1999, 37, 1823–1837. [Google Scholar] [CrossRef]
- Huang, G.; Gao, J.; Hu, Z.; John, J.V.S.; Ponder, B.C.; Moro, D. Controlled drug release from hydrogel nanoparticle networks. J. Control. Release 2004, 94, 303–311. [Google Scholar] [CrossRef] [PubMed]
- Hoare, T.; Pelton, R. Functional group distributions in carboxylic acid containing poly(N-isopropylacrylamide) microgels. Langmuir 2004, 20, 2123–2133. [Google Scholar] [CrossRef] [PubMed]
- Leobandung, W.; Ichikawa, H.; Fukumori, Y.; Peppas, N.A. Monodisperse nanoparticles of poly (ethylene glycol) macromers and N-isopropyl acrylamide for biomedical applications. J. Appl. Polym. Sci. 2003, 87, 1678–1684. [Google Scholar] [CrossRef]
- Berndt, I.; Pedersen, J.S.; Lindner, P.; Richtering, W. Influence of Shell Thickness and Cross-Link Density on the Structure of Temperature-Sensitive Poly-N-Isopropylacrylamide−Poly-N-Isopropylmethacrylamide Core−Shell Microgels Investigated by Small-Angle Neutron Scattering. Langmuir 2006, 22, 459–468. [Google Scholar] [CrossRef] [PubMed]
- Boyko, V.; Pich, A.; Lu, Y.; Richter, S.; Arndt, K.-F.; Adler, H.-J.P. Thermo-sensitive poly(N-vinylcaprolactam-co-acetoacetoxyethyl methacrylate) microgels: 1—Synthesis and characterization. Polymer 2003, 44, 7821–7827. [Google Scholar] [CrossRef]
- Karg, M.; Pastoriza-Santos, I.; Pérez-Juste, J.; Hellweg, T.; Liz-Marzán, L.M. Nanorod-coated PNIPAM microgels: Thermoresponsive optical properties. Small 2007, 3, 1222–1229. [Google Scholar] [CrossRef]
- Kratz, K.; Hellweg, T.; Eimer, W. Structural changes in PNIPAM microgel particles as seen by SANS, DLS, and EM techniques. Polymer 2001, 42, 6631–6639. [Google Scholar] [CrossRef]
- Kratz, K.; Hellweg, T.; Eimer, W. Effect of connectivity and charge density on the swelling and local structural and dynamic properties of colloidal PNIPAM microgels. Ber. Bunsen Ges. Phys. Chem. 1998, 102, 1603–1608. [Google Scholar] [CrossRef]
- Das, M.; Sanson, N.; Fava, D.; Kumacheva, E. Microgels loaded with gold nanorods: Photothermally triggered volume transitions under physiological conditions. Langmuir 2007, 23, 196–201. [Google Scholar] [CrossRef]
- Gorelikov, I.; Field, L.M.; Kumacheva, E. Hybrid microgels photoresponsive in the near-infrared spectral range. J. Am. Chem. Soc. 2004, 126, 15938–15939. [Google Scholar] [CrossRef]
- Farooq, M.; Sagbas, S.; Yildiz, M.; Meral, K.; Siddiq, M.; Aktas, N.; Sahiner, N. Gum arabic microgels as template for in situ metal-sulfide based quantum dots preparation and their thermal, spectroscopic, optical, and magnetic characterization. J. Electron. Mater. 2017, 46, 4373–4383. [Google Scholar] [CrossRef]
- Sayed, A.; Asran, A.S. Electrospinning of Polymeric Nanofibers and Nanocomposite Materials: Fabrication, Physicochemical Characterization and Medical Applications. Ph.D. Thesis, Martin-Luther-Universität Halle-Wittenberg, Halle, Germany, 9 February 2011. [Google Scholar]
- Huang, Z.-M.; Zhang, Y.-Z.; Kotaki, M.; Ramakrishna, S. A review on polymer nanofibers by electrospinning and their applications in nanocomposites. Compos. Sci. Technol. 2003, 63, 2223–2253. [Google Scholar] [CrossRef]
- Kehren, D.; Pich, A. Fabrication and Characterisation of Microgel/Polymer Composite Microfibres. Macromol. Mater. Eng. 2013, 298, 1282–1293. [Google Scholar] [CrossRef]
- Koushik, O.; Rao, Y.; Kumar, P.; Karthikeyan, R. Nano Drug Delivery Systems to Overcome Cancer Drug Resistance-A Review. J. Nanomed. Nanotechnol. 2016, 7, 2. [Google Scholar]
- Yang, G.; Wang, J.; Wang, Y.; Li, L.; Guo, X.; Zhou, S. An implantable active-targeting micelle-in-nanofiber device for efficient and safe cancer therapy. ACS Nano 2015, 9, 1161–1174. [Google Scholar] [CrossRef] [PubMed]
- Shao, S.; Li, L.; Yang, G.; Li, J.; Luo, C.; Gong, T.; Zhou, S. Controlled green tea polyphenols release from electrospun PCL/MWCNTs composite nanofibers. Int. J. Pharm. Sci. Res. 2011, 421, 310–320. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Lim, C.T.; Ramakrishna, S.; Huang, Z.-M. Recent development of polymer nanofibers for biomedical and biotechnological applications. J. Mater. Sci. Mater. Med. 2005, 16, 933–946. [Google Scholar] [CrossRef] [PubMed]
- Xue, J.; Wu, T.; Dai, Y.; Xia, Y. Electrospinning and electrospun nanofibers: Methods, materials, and applications. Chem. Rev. 2019, 119, 5298–5415. [Google Scholar] [CrossRef] [PubMed]
- Islam, M.S.; Ang, B.C.; Andriyana, A.; Afifi, A.M. A review on fabrication of nanofibers via electrospinning and their applications. SN Appl. Sci. 2019, 1, 1–16. [Google Scholar] [CrossRef] [Green Version]
- Miyauchi, M.; Simmons, T.J.; Miao, J.; Gagner, J.E.; Shriver, Z.H.; Aich, U.; Dordick, J.S.; Linhardt, R.J. Electrospun polyvinylpyrrolidone fibers with high concentrations of ferromagnetic and superparamagnetic nanoparticles. ACS Appl. Mater. Interfaces 2011, 3, 1958–1964. [Google Scholar] [CrossRef]
- Mariello, M.; Qualtieri, A.; Mele, G.; De Vittorio, M. Metal-Free Multilayer Hybrid PENG Based on Soft Electrospun/-Sprayed Membranes with Cardanol Additive for Harvesting Energy from Surgical Face Masks. ACS Appl. Mater. Interfaces 2021, 13, 20606–20621. [Google Scholar] [CrossRef] [PubMed]
- Reneker, D.H.; Chun, I. Nanometre diameter fibres of polymer, produced by electrospinning. Nanotechnology 1996, 7, 216. [Google Scholar] [CrossRef] [Green Version]
- Jalili, R.; Hosseini, S.A.; Morshed, M. The effects of operating parameters on the morphology of electrospun polyacrilonitrile nanofibres. Iran. Polym. J. 2005, 14, 1074–1081. [Google Scholar]
- Gupta, P.; Asmatulu, R.; Claus, R.; Wilkes, G. Superparamagnetic flexible substrates based on submicron electrospun Estane® fibers containing MnZnFe—Ni nanoparticles. J. Appl. Polym. Sci. 2006, 100, 4935–4942. [Google Scholar] [CrossRef]
- Hu, X.; Liu, S.; Zhou, G.; Huang, Y.; Xie, Z.; Jing, X. Electrospinning of polymeric nanofibers for drug delivery applications. J. Control. Release 2014, 185, 12–21. [Google Scholar] [CrossRef]
- Deitzel, J.M.; Kleinmeyer, J.; Harris, D.; Tan, N.B. The effect of processing variables on the morphology of electrospun nanofibers and textiles. Polymer 2001, 42, 261–272. [Google Scholar] [CrossRef]
- Jin, H.-J.; Fridrikh, S.V.; Rutledge, G.C.; Kaplan, D.L. Electrospinning Bombyx mori silk with poly(ethylene oxide). Biomacromolecules 2002, 3, 1233–1239. [Google Scholar] [CrossRef]
- Huang, C.; Chen, S.; Lai, C.; Reneker, D.H.; Qiu, H.; Ye, Y.; Hou, H. Electrospun polymer nanofibres with small diameters. Nanotechnology 2006, 17, 1558–1563. [Google Scholar] [CrossRef] [PubMed]
- Yarin, A.L.; Koombhongse, S.; Reneker, D.H. Taylor cone and jetting from liquid droplets in electrospinning of nanofibers. J. Appl. Phys. 2001, 90, 4836–4846. [Google Scholar] [CrossRef] [Green Version]
- Cheng, J.; Jun, Y.; Qin, J.; Lee, S.-H. Electrospinning versus microfluidic spinning of functional fibers for biomedical applications. Biomaterials 2017, 114, 121–143. [Google Scholar] [CrossRef] [PubMed]
- Beachley, V.; Wen, X. Effect of electrospinning parameters on the nanofiber diameter and length. Mater. Sci. Eng. C 2009, 29, 663–668. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, C.; Yuan, X.; Wu, L.; Han, Y.; Sheng, J. Study on morphology of electrospun poly(vinyl alcohol) mats. Eur. Polym. J. 2005, 41, 423–432. [Google Scholar] [CrossRef]
- Son, W.K.; Youk, J.H.; Lee, T.S.; Park, W.H. Electrospinning of ultrafine cellulose acetate fibers: Studies of a new solvent system and deacetylation of ultrafine cellulose acetate fibers. J. Polym. Sci. Part B Polym. Phys. 2004, 42, 5–11. [Google Scholar] [CrossRef]
- Li, D.; Xia, Y. Electrospinning of nanofibers: Reinventing the wheel? Adv. Mater. 2004, 16, 1151–1170. [Google Scholar] [CrossRef]
- Casper, C.L.; Stephens, J.S.; Tassi, N.G.; Chase, D.B.; Rabolt, J.F. Controlling surface morphology of electrospun polystyrene fibers: Effect of humidity and molecular weight in the electrospinning process. Macromolecules 2004, 37, 573–578. [Google Scholar] [CrossRef]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Paca, A.M.; Ajibade, P.A. Metal Sulfide Semiconductor Nanomaterials and Polymer Microgels for Biomedical Applications. Int. J. Mol. Sci. 2021, 22, 12294. https://doi.org/10.3390/ijms222212294
Paca AM, Ajibade PA. Metal Sulfide Semiconductor Nanomaterials and Polymer Microgels for Biomedical Applications. International Journal of Molecular Sciences. 2021; 22(22):12294. https://doi.org/10.3390/ijms222212294
Chicago/Turabian StylePaca, Athandwe M., and Peter A. Ajibade. 2021. "Metal Sulfide Semiconductor Nanomaterials and Polymer Microgels for Biomedical Applications" International Journal of Molecular Sciences 22, no. 22: 12294. https://doi.org/10.3390/ijms222212294





