Memory Disorders Related to Hippocampal Function: The Interest of 5-HT4Rs Targeting
Abstract
1. Introduction
2. Episodic Memory Function and the Hippocampal Formation
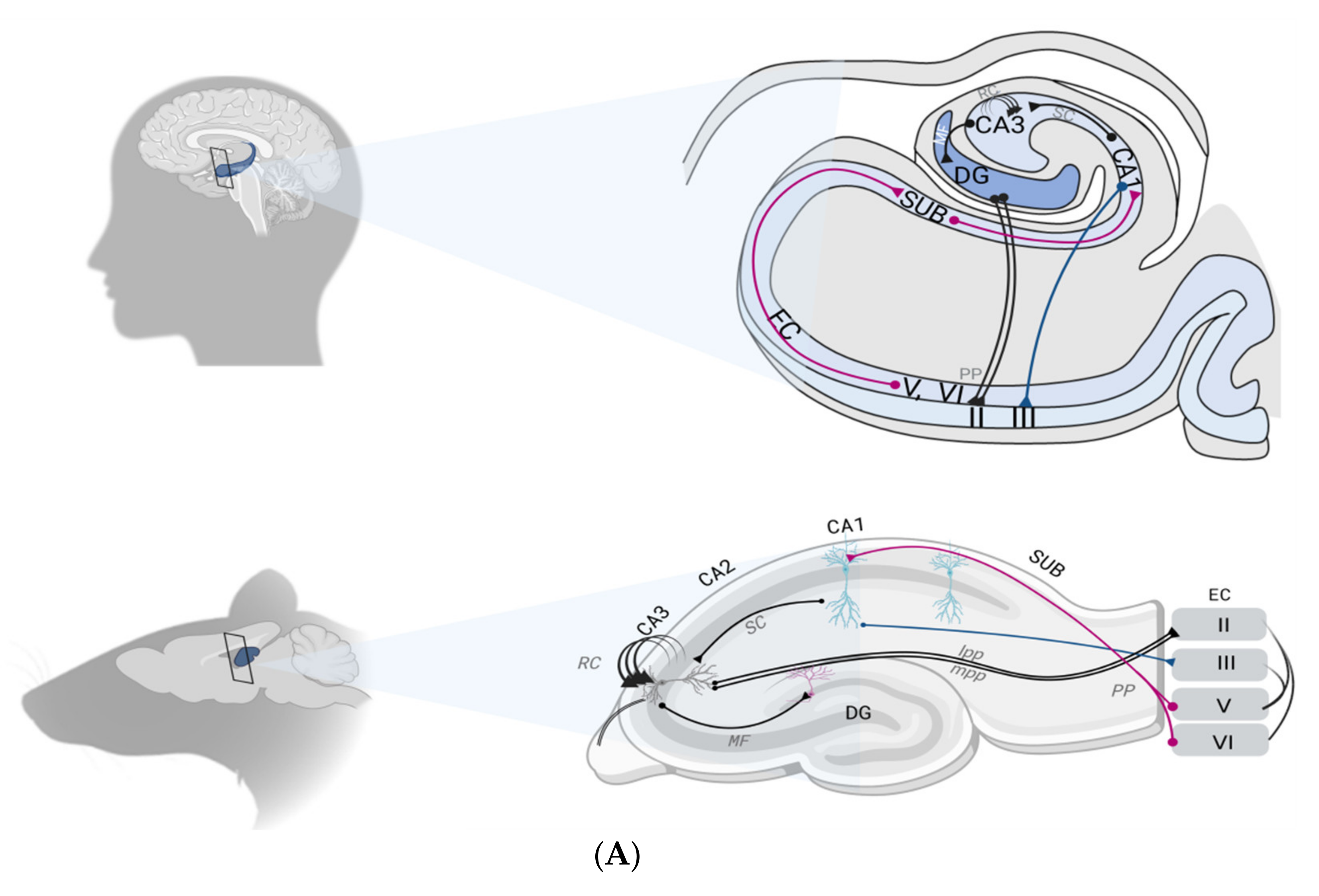
2.1. The Hippocampal Formation
2.2. The Hippocampal Formation Circuitry
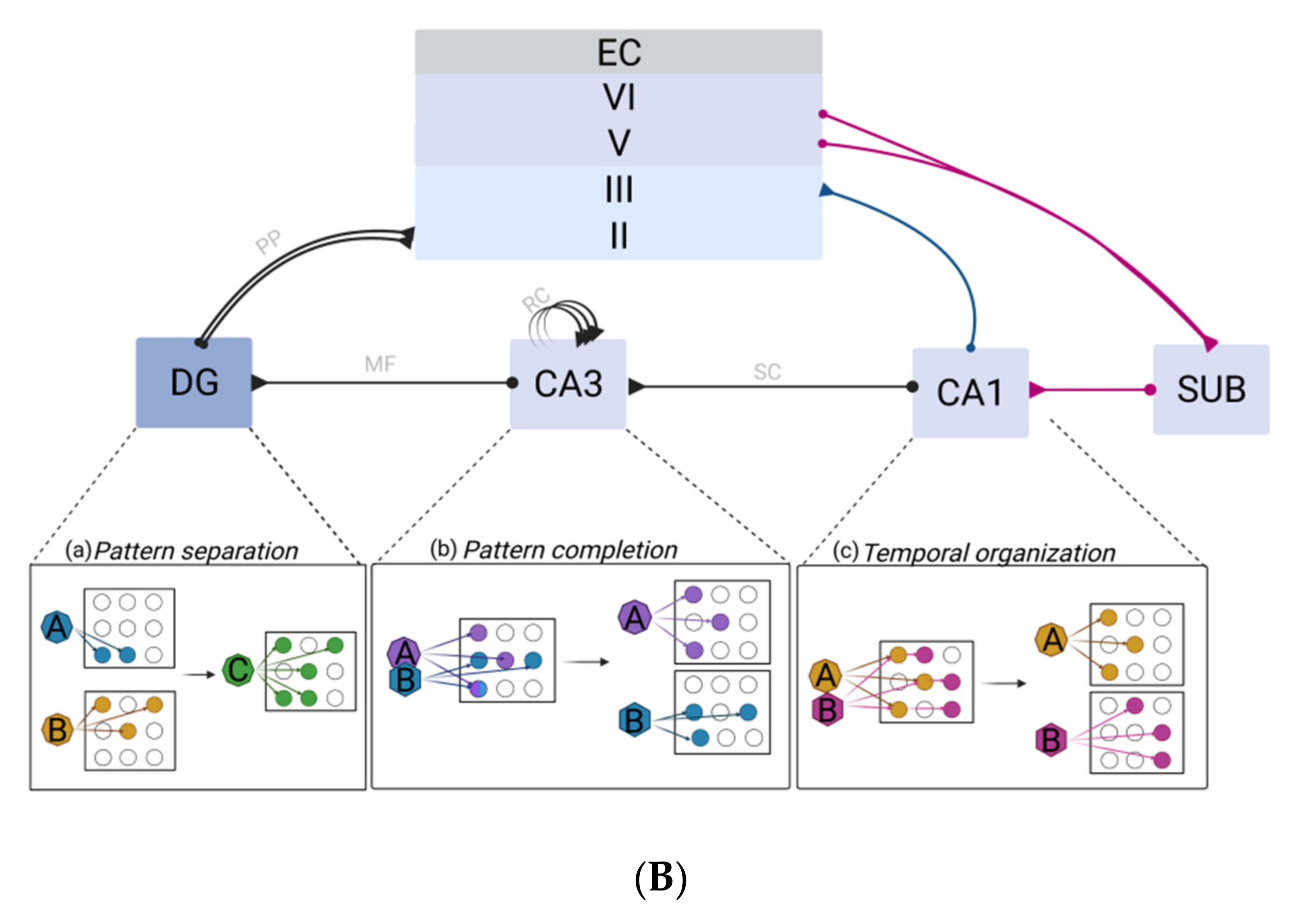
2.2.1. The Dentate Gyrus–CA3 Pathway: Pattern Separation and Completion
2.2.2. Multifaceted Roles of the CA1
2.3. Synaptic Plasticity as a Correlate of Hippocampal Memory
2.4. Neurotransmission Systems in the Hippocampus
2.4.1. The Glutamatergic System
2.4.2. The GABAergic System
2.4.3. The Cholinergic System
2.4.4. The Serotonergic System
3. Relevance of 5-HT4Rs Modulation in Memory Disorders
3.1. Insights from Animal Behavior Investigations
3.2. Distribution of 5-HT4Rs in CNS and Memory Disorders
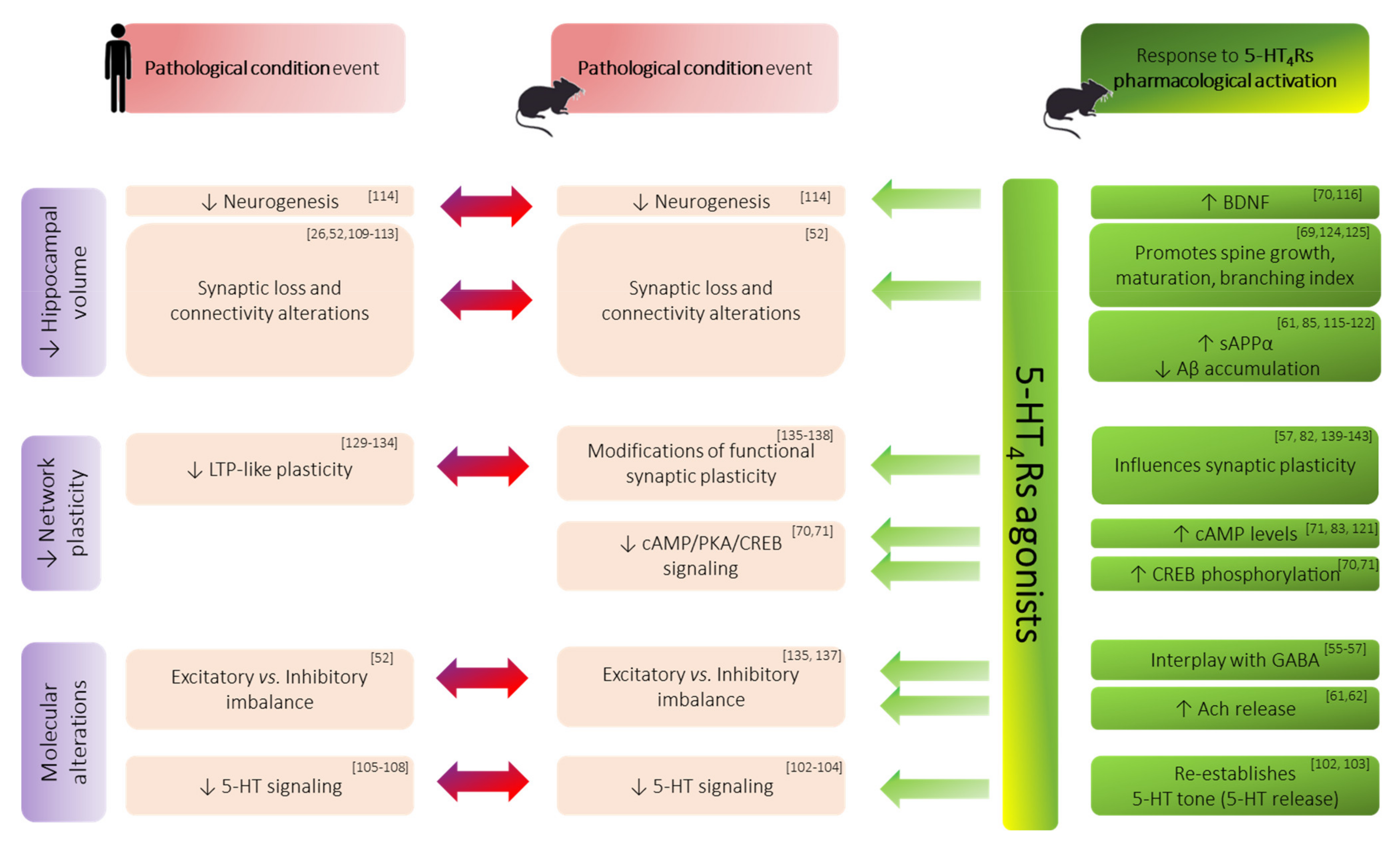
3.3. Morphological/Structural Alterations of Hippocampal Formation in Memory Disorders
3.4. Functional Synaptic Plasticity Impairments
3.5. 5-HT4Rs in Clinical Trials
4. Concluding Remarks and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- El Haj, M.; Roche, J.; Gallouj, K.; Gandolphe, M.-C. Autobiographical Memory Compromise in Alzheimer’s Disease: A Cognitive and Clinical Overview. Gériatrie Psychol. Neuropsychiatr. Viellissement 2017, 15, 443–451. [Google Scholar] [CrossRef]
- Das, T.; Hwang, J.J.; Poston, K.L. Episodic Recognition Memory and the Hippocampus in Parkinson’s Disease: A Review. Cortex 2019, 113, 191–209. [Google Scholar] [CrossRef] [PubMed]
- Darcet, F.; Gardier, A.; Gaillard, R.; David, D.; Guilloux, J.-P. Cognitive Dysfunction in Major Depressive Disorder. A Translational Review in Animal Models of the Disease. Pharmaceuticals 2016, 9, 9. [Google Scholar] [CrossRef]
- Guo, J.Y.; Ragland, J.D.; Carter, C.S. Memory and Cognition in Schizophrenia. Mol. Psychiatry 2019, 24, 633–642. [Google Scholar] [CrossRef] [PubMed]
- Klein, S.B.; Nichols, S. Memory and the Sense of Personal Identity. Mind 2012, 121, 677–702. [Google Scholar] [CrossRef]
- Schneider, L.S.; Mangialasche, F.; Andreasen, N.; Feldman, H.; Giacobini, E.; Jones, R.; Mantua, V.; Mecocci, P.; Pani, L.; Winblad, B.; et al. Clinical Trials and Late-Stage Drug Development for Alzheimer’s Disease: An Appraisal from 1984 to 2014. J. Intern. Med. 2014, 275, 251–283. [Google Scholar] [CrossRef] [PubMed]
- FDA. FDA Grants Accelerated Approval for Alzheimer’s Drug. 2021. Available online: https://www.fda.gov/news-events/press-announcements/fda-grants-accelerated-approval-alzheimers-drug (accessed on 13 July 2021).
- Lalut, J.; Karila, D.; Dallemagne, P.; Rochais, C. Modulating 5-HT4 and 5-HT6 Receptors in Alzheimer’s Disease Treatment. Future Med. Chem. 2017, 9, 781–795. [Google Scholar] [CrossRef]
- Lanthier, C.; Dallemagne, P.; Lecoutey, C.; Claeysen, S.; Rochais, C. Therapeutic Modulators of the Serotonin 5-HT4 Receptor: A Patent Review (2014–Present). Expert Opin. Ther. Pat. 2020, 30, 495–508. [Google Scholar] [CrossRef]
- Dumuis, A.; Bouhelal, R.; Sebben, M.; Cory, R.; Bockaert, J. A Nonclassical 5-Hydroxytryptamine Receptor Positively Coupled with Adenylate Cyclase in the Central Nervous System. Mol. Pharmacol. 1988, 34, 880–887. [Google Scholar]
- Squire, L.R. Memory Systems of the Brain: A Brief History and Current Perspective. Neurobiol. Learn. Mem. 2004, 82, 171–177. [Google Scholar] [CrossRef]
- Tulving, E.; Markowitsch, H.J. Episodic and Declarative Memory: Role of the Hippocampus. Hippocampus 1998, 8, 198–204. [Google Scholar] [CrossRef]
- Scoville, B.S.; Milner, B. Loss of Recent Memory after Bilateral Hippocampal Lesions. J. Neurol. Neurosurg. Psychiat. 1957, 20, 11. [Google Scholar] [CrossRef] [PubMed]
- Stein, D.G.; Kimble, D.P. Effects of Hippocampal Lesions and Posttrial Strychnine Administration on Maze Behavior in the Rat. J. Comp. Physiol. Psychol. 1966, 62, 243–249. [Google Scholar] [CrossRef]
- Thompson, R.; Langer, S.K.; Rich, I. Lesions of the limbic system and short-term memory in albino rats. Brain 1964, 87, 537–542. [Google Scholar] [CrossRef]
- Zola, S.M.; Squire, L.R.; Teng, E.; Stefanacci, L.; Buffalo, E.A.; Clark, R.E. Impaired Recognition Memory in Monkeys after Damage Limited to the Hippocampal Region. J. Neurosci. 2000, 20, 451–463. [Google Scholar] [CrossRef]
- Zola-Morgan, S.; Squirre, L.R.; Amaral, G. Human Amnesia and the Medial Temporal Region: Impairment Following a Bilateral Lesion Limited to Field CA1 of the Hippocampus. J. Neurosci. 1986, 6, 2950–2967. [Google Scholar] [CrossRef]
- Eichenbaum, H. Hippocampus. Neuron 2004, 44, 109–120. [Google Scholar] [CrossRef] [PubMed]
- O’Keefe, J.; Nadel, L. The Hippocampus as a Cognitive Map; Clarendon Press: Oxford, UK, 1978; ISBN 978-0-19-857206-0. [Google Scholar]
- Fanselow, M.S.; Dong, H.-W. Are the Dorsal and Ventral Hippocampus Functionally Distinct Structures? Neuron 2010, 65, 7–19. [Google Scholar] [CrossRef] [PubMed]
- Bannerman, D.M.; Rawlins, J.N.P.; McHugh, S.B.; Deacon, R.M.J.; Yee, B.K.; Bast, T.; Zhang, W.-N.; Pothuizen, H.H.J.; Feldon, J. Regional Dissociations within the Hippocampus—Memory and Anxiety. Neurosci. Biobehav. Rev. 2004, 28, 273–283. [Google Scholar] [CrossRef]
- Moser, M.B.; Moser, E.I. Functional Differentiation in the Hippocampus. Hippocampus 1998, 8, 608–619. [Google Scholar] [CrossRef]
- O’Keefe, J.; Recce, M.L. Phase Relationship between Hippocampal Place Units and the EEG Theta Rhythm. Hippocampus 1993, 3, 317–330. [Google Scholar] [CrossRef]
- Kesner, R.P.; Rolls, E.T. A Computational Theory of Hippocampal Function, and Tests of the Theory: New Developments. Neurosci. Biobehav. Rev. 2015, 48, 92–147. [Google Scholar] [CrossRef]
- Gilbert, P.E.; Kesner, R.P.; Lee, I. Dissociating Hippocampal Subregions: A Double Dissociation between Dentate Gyrus and CA1. Hippocampus 2001, 11, 626–636. [Google Scholar] [CrossRef]
- Yassa, M.A.; Mattfeld, A.T.; Stark, S.M.; Stark, C.E.L. Age-Related Memory Deficits Linked to Circuit-Specific Disruptions in the Hippocampus. Proc. Natl. Acad. Sci. USA 2011, 108, 8873–8878. [Google Scholar] [CrossRef] [PubMed]
- Grande, X.; Berron, D.; Horner, A.J.; Bisby, J.A.; Düzel, E.; Burgess, N. Holistic Recollection via Pattern Completion Involves Hippocampal Subfield CA3. J. Neurosci. 2019, 39, 8100–8111. [Google Scholar] [CrossRef] [PubMed]
- Rolls, E.T. A Quantitative Theory of the Functions of the Hippocampal CA3 Network in Memory. Front. Cell. Neurosci. 2013, 7, 98. [Google Scholar] [CrossRef]
- Barrientos, S.A.; Tiznado, V. Hippocampal CA1 Subregion as a Context Decoder. J. Neurosci. 2016, 36, 6602–6604. [Google Scholar] [CrossRef]
- Shimbo, A.; Izawa, E.-I.; Fujisawa, S. Scalable Representation of Time in the Hippocampus. Sci. Adv. 2021, 7, eabd7013. [Google Scholar] [CrossRef] [PubMed]
- Martin, S.J.; Grimwood, P.D.; Morris, R.G.M. Synaptic Plasticity and Memory: An Evaluation of the Hypothesis. Annu. Rev. Neurosci. 2000, 23, 649–711. [Google Scholar] [CrossRef]
- Hebb, D.O. The Organization of Behavior: A Neuropsychological Theory; Wiley: Oxford, UK, 1949. [Google Scholar]
- Bliss, T.V.P.; Collingridge, G.L. A Synaptic Model of Memory: Long-Term Potentiation in the Hippocampus. Nature 1993, 361, 31–39. [Google Scholar] [CrossRef]
- Bliss, T.V.P.; Lømo, T. Long-Lasting Potentiation of Synaptic Transmission in the Dentate Area of the Anaesthetized Rabbit Following Stimulation of the Perforant Path. J. Physiol. 1973, 232, 331–356. [Google Scholar] [CrossRef] [PubMed]
- Nicoll, R.A. A Brief History of Long-Term Potentiation. Neuron 2017, 93, 281–290. [Google Scholar] [CrossRef] [PubMed]
- Larson, J.; Munkácsy, E. Theta-Burst LTP. Brain Res. 2015, 1621, 38–50. [Google Scholar] [CrossRef]
- Bragin, A.; Jando, G.; Nadasdy, Z.; Hetke, J.; Wise, K.; Buzsaki, G. Gamma (40-100 Hz) Oscillation in the Hippocampus of the Behaving Rat. J. Neurosci. 1995, 15, 47–60. [Google Scholar] [CrossRef] [PubMed]
- Tort, A.B.L.; Kramer, M.A.; Thorn, C.; Gibson, D.J.; Kubota, Y.; Graybiel, A.M.; Kopell, N.J. Dynamic Cross-Frequency Couplings of Local Field Potential Oscillations in Rat Striatum and Hippocampus during Performance of a T-Maze Task. Proc. Natl. Acad. Sci. USA 2008, 105, 20517–20522. [Google Scholar] [CrossRef]
- Nuñez, A.; Buño, W. The Theta Rhythm of the Hippocampus: From Neuronal and Circuit Mechanisms to Behavior. Front. Cell. Neurosci. 2021, 15, 649262. [Google Scholar] [CrossRef] [PubMed]
- Morris, R.G.M.; Anderson, E.; Lynch, G.S.; Baudry, M. Selective Impairment of Learning and Blockade of Long-Term Potentiation by an N-Methyl-D-Aspartate Receptor Antagonist, AP5. Nature 1986, 319, 774–776. [Google Scholar] [CrossRef]
- Davis, S.; Butcher, S.P.; Morris, R.G. The NMDA Receptor Antagonist D-2-Amino-5-Phosphonopentanoate (D-AP5) Impairs Spatial Learning and LTP in Vivo at Intracerebral Concentrations Comparable to Those That Block LTP in Vitro. J. Neurosci. 1992, 12, 21–34. [Google Scholar] [CrossRef]
- Lynch, M.A. Long-Term Potentiation and Memory. Physiol. Rev. 2004, 84, 87–136. [Google Scholar] [CrossRef]
- Cooke, S.F. Plasticity in the Human Central Nervous System. Brain 2006, 129, 1659–1673. [Google Scholar] [CrossRef]
- Kemp, A.; Manahan-Vaughan, D. Hippocampal Long-Term Depression: Master or Minion in Declarative Memory Processes? Trends Neurosci. 2007, 30, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Bliss, T.V.P.; Cooke, S.F. Long-Term Potentiation and Long-Term Depression: A Clinical Perspective. Clinics 2011, 66, 3–17. [Google Scholar] [CrossRef]
- Myhrer, T. Neurotransmitter Systems Involved in Learning and Memory in the Rat: A Meta-Analysis Based on Studies of Four Behavioral Tasks. Brain Res. Rev. 2003, 41, 268–287. [Google Scholar] [CrossRef]
- Seyedabadi, M.; Fakhfouri, G.; Ramezani, V.; Mehr, S.E.; Rahimian, R. The Role of Serotonin in Memory: Interactions with Neurotransmitters and Downstream Signaling. Exp. Brain Res. 2014, 232, 723–738. [Google Scholar] [CrossRef]
- Monaghan, D.T.; Cotman, C.W. Identification and Properties of N-Methyl-D-Aspartate Receptors in Rat Brain Synaptic Plasma Membranes. Proc. Natl. Acad. Sci. USA 1986, 83, 7532–7536. [Google Scholar] [CrossRef]
- Folch, J.; Busquets, O.; Ettcheto, M.; Sánchez-López, E.; Castro-Torres, R.D.; Verdaguer, E.; Garcia, M.L.; Olloquequi, J.; Casadesús, G.; Beas-Zarate, C.; et al. Memantine for the Treatment of Dementia: A Review on Its Current and Future Applications. JAD 2018, 62, 1223–1240. [Google Scholar] [CrossRef] [PubMed]
- Cull-Candy, S.; Brickley, S.; Farrant, M. NMDA Receptor Subunits: Diversity, Development and Disease. Curr. Opin. Neurobiol. 2001, 11, 327–335. [Google Scholar] [CrossRef]
- King, M.; Marsden, C.; Fone, K. A Role for the 5-HT1A, 5-HT4 and 5-HT6 Receptors in Learning and Memory. Trends Pharmacol. Sci. 2008, 29, 482–492. [Google Scholar] [CrossRef]
- Pelkey, K.A.; Chittajallu, R.; Craig, M.T.; Tricoire, L.; Wester, J.C.; McBain, C.J. Hippocampal GABAergic Inhibitory Interneurons. Physiol. Rev. 2017, 97, 1619–1747. [Google Scholar] [CrossRef] [PubMed]
- Izquierdo, I.; da Cunha, C.; Rosat, R.; Jerusalinsky, D.; Ferreira, M.B.C.; Medina, J.H. Neurotransmitter Receptors Involved in Post-Training Memory Processing by the Amygdala, Medial Septum, and Hippocampus of the Rat. Behav. Neural Biol. 1992, 58, 16–26. [Google Scholar] [CrossRef]
- Grover, L.M.; Yan, C. Blockade of GABA A Receptors Facilitates Induction of NMDA Receptor-Independent Long-Term Potentiation. J. Neurophysiol. 1999, 81, 2814–2822. [Google Scholar] [CrossRef][Green Version]
- Bijak, M.; Misgeld, U. Effects of Serotonin through Serotonin1A and Serotonin4 Receptors on Inhibition in the Guinea-Pig Dentate Gyrus in Vitro. Neuroscience 1997, 78, 1017–1026. [Google Scholar] [CrossRef]
- Bianchi, C.; Rodi, D.; Marino, S.; Beani, L.; Siniscalchi, A. Dual Effects of 5-HT4 Receptor Activation on GABA Release from Guinea Pig Hippocampal Slices. Neuroreport 2002, 13, 2177–2180. [Google Scholar] [CrossRef] [PubMed]
- Lecouflet, P.; Roux, C.M.; Potier, B.; Leger, M.; Brunet, E.; Billard, J.-M.; Schumann-Bard, P.; Freret, T. Interplay between 5-HT4 Receptors and GABAergic System within CA1 Hippocampal Synaptic Plasticity. Cereb. Cortex 2021, 31, 694–701. [Google Scholar] [CrossRef] [PubMed]
- Teles-Grilo Ruivo, L.M.; Mellor, J.R. Cholinergic Modulation of Hippocampal Network Function. Front. Synaptic Neurosci. 2013, 5. [Google Scholar] [CrossRef]
- Miranda, M.I. Changes in neurotransmitter extracellular levels during memory formation. In Neural Plasticity and Memory: From Genes to Brain Imaging; Bermúdez-Rattoni, F., Ed.; CRC Press/Taylor & Francis: Boca Raton FL, USA, 2007; ISBN 978-0-8493-9070-8. [Google Scholar]
- Reis, H.; Guatimosim, C.; Paquet, M.; Santos, M.; Ribeiro, F.; Kummer, A.; Schenatto, G.; Salgado, J.; Vieira, L.; Teixeira, A.; et al. Neuro-Transmitters in the Central Nervous System & Their Implication in Learning and Memory Processes. CMC 2009, 16, 796–840. [Google Scholar] [CrossRef]
- Mohler, E.G.; Shacham, S.; Noiman, S.; Lezoualc’h, F.; Robert, S.; Gastineau, M.; Rutkowski, J.; Marantz, Y.; Dumuis, A.; Bockaert, J.; et al. VRX-03011, a Novel 5-HT4 Agonist, Enhances Memory and Hippocampal Acetylcholine Efflux. Neuropharmacology 2007, 53, 563–573. [Google Scholar] [CrossRef]
- Siniscalchi, A.; Badini, I.; Beani, L.; Bianchi, C. 5-HT4 Receptor Modulation of Acetylcholine Outflow in Guinea Pig Brain Slices. Neuroreport 1999, 10, 547–551. [Google Scholar] [CrossRef]
- Ihara, N.; Ueda, S.; Kawata, M.; Sano, Y. Immunohistochemical Demonstration of Serotonin-Containing Nerve Fibers in the Mammalian Hippocampal Formation. Acta Anat. 1988, 132, 335–346. [Google Scholar] [CrossRef]
- Oleskevich, S.; Descarries, L.; Watkins, K.C.; Se´gue´la, P.; Daszuta, A. Ultrastructural Features of the Serotonin Innervation in Adult Rat Hippocampus: An Immunocytochemical Description in Single and Serial Thin Sections. Neuroscience 1991, 42, 777–791. [Google Scholar] [CrossRef]
- Berumen, L.C.; Rodríguez, A.; Miledi, R.; García-Alcocer, G. Serotonin Receptors in Hippocampus. Sci. World J. 2012, 2012, 1–15. [Google Scholar] [CrossRef]
- Cowen, P.; Sherwood, A.C. The Role of Serotonin in Cognitive Function: Evidence from Recent Studies and Implications for Understanding Depression. J. Psychopharmacol. 2013, 27, 575–583. [Google Scholar] [CrossRef]
- Švob Štrac, D.; Pivac, N.; Mück-Šeler, D. The Serotonergic System and Cognitive Function. Transl. Neurosci. 2016, 7, 35–49. [Google Scholar] [CrossRef] [PubMed]
- Bockaert, J.; Claeysen, S.; Compan, V.; Dumuis, A. 5-HT4 Receptors: History, Molecular Pharmacology and Brain Functions. Neuropharmacology 2008, 55, 922–931. [Google Scholar] [CrossRef] [PubMed]
- Restivo, L.; Roman, F.; Dumuis, A.; Bockaert, J.; Marchetti, E.; Ammassari-Teule, M. The Promnesic Effect of G-Protein-Coupled 5-HT4 Receptors Activation Is Mediated by a Potentiation of Learning-Induced Spine Growth in the Mouse Hippocampus. Neuropsychopharmacology 2008, 33, 2427–2434. [Google Scholar] [CrossRef]
- Pascual-Brazo, J.; Castro, E.; Díaz, Á.; Valdizán, E.M.; Pilar-Cuéllar, F.; Vidal, R.; Treceño, B.; Pazos, Á. Modulation of Neuroplasticity Pathways and Antidepressant-like Behavioural Responses Following the Short-Term (3 and 7 Days) Administration of the 5-HT4 Receptor Agonist RS67333. Int. J. Neuropsychopharm. 2012, 15, 631–643. [Google Scholar] [CrossRef]
- Ishii, T.; Kinoshita, K.-i.; Muroi, Y. Serotonin 5-HT4 Receptor Agonists Improve Facilitation of Contextual Fear Extinction in An MPTP-Induced Mouse Model of Parkinson’s Disease. IJMS 2019, 20, 5340. [Google Scholar] [CrossRef] [PubMed]
- Bockaert, J.; Claeysen, S.; Compan, V.; Dumuis, A. 5-HT4 Receptors, a Place in the Sun: Act Two. Curr. Opin. Pharmacol. 2011, 11, 87–93. [Google Scholar] [CrossRef]
- Hagena, H.; Manahan-Vaughan, D. The Serotonergic 5-HT4 Receptor: A Unique Modulator of Hippocampal Synaptic Information Processing and Cognition. Neurobiol. Learn. Memory 2017, 138, 145–153. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, C.M.; Rosen, Z.B.; Suri, D.; Sun, Q.; Hersh, M.; Sargin, D.; Dincheva, I.; Morgan, A.A.; Spivack, S.; Krok, A.C.; et al. Hippocampal 5-HT Input Regulates Memory Formation and Schaffer Collateral Excitation. Neuron 2018, 98, 992–1004.e4. [Google Scholar] [CrossRef]
- Segu, L.; Lecomte, M.-J.; Wolff, M.; Santamaria, J.; Hen, R.; Dumuis, A.; Berrard, S.; Bockaert, J.; Buhot, M.-C.; Compan, V. Hyperfunction of Muscarinic Receptor Maintains Long-Term Memory in 5-HT4 Receptor Knock-Out Mice. PLoS ONE 2010, 5, e9529. [Google Scholar] [CrossRef]
- Lelong, V.; Dauphin, F.; Boulouard, M. RS 67333 and D-Cycloserine Accelerate Learning Acquisition in the Rat. Neuropharmacology 2001, 41, 517–522. [Google Scholar] [CrossRef]
- Lamirault, L.; Simon, H. Enhancement of Place and Object Recognition Memory in Young Adult and Old Rats by RS 67333, a Partial Agonist of 5-HT4 Receptors. Neuropharmacology 2001, 41, 844–853. [Google Scholar] [CrossRef]
- Freret, T.; Bouet, V.; Quiedeville, A.; Nee, G.; Dallemagne, P.; Rochais, C.; Boulouard, M. Synergistic Effect of Acetylcholinesterase Inhibition (Donepezil) and 5-HT4 Receptor Activation (RS67333) on Object Recognition in Mice. Behav. Brain Res. 2012, 230, 304–308. [Google Scholar] [CrossRef]
- Levallet, G.; Hotte, M.; Boulouard, M.; Dauphin, F. Increased Particulate Phosphodiesterase 4 in the Prefrontal Cortex Supports 5-HT4 Receptor-Induced Improvement of Object Recognition Memory in the Rat. Psychopharmacology 2009, 202, 125–139. [Google Scholar] [CrossRef]
- Marchetti-Gauthier, E.; Roman, F.S.; Dumuis, A.; Bockaert, J.; Soumireu-Mourat, B. BIMU1 Increases Associative Memory in Rats by Activating 5-HT4 Receptors. Neuropharmacology 1997, 36, 697–706. [Google Scholar] [CrossRef]
- Galeotti, N.; Ghelardini, C.; Bartolini, A. Role of 5-HT4 Receptors in the Mouse Passive Avoidance Test. J. Pharmacol. Exp. Ther. 1998, 286, 1115–1121. [Google Scholar]
- Matsumoto, M.; Togashi, H.; Mori, K.; Ueno, K.; Ohashi, S.; Kojima, T.; Yoshioka, M. Evidence for Involvement of Central 5-HT(4) Receptors in Cholinergic Function Associated with Cognitive Processes: Behavioral, Electrophysiological, and Neurochemical Studies. J. Pharmacol. Exp. Ther. 2001, 296, 676–682. [Google Scholar]
- Moser, P.C.; Bergis, O.E.; Jegham, S.; Lochead, A.; Duconseille, E.; Terranova, J.-P.; Caille, D.; Berque-Bestel, I.; Lezoualc’h, F.; Fischmeister, R.; et al. SL65.0155, a Novel 5-Hydroxytryptamine(4) Receptor Partial Agonist with Potent Cognition-Enhancing Properties. J. Pharmacol. Exp. Ther. 2002, 302, 731–741. [Google Scholar] [CrossRef] [PubMed]
- Baranger, K.; Giannoni, P.; Girard, S.D.; Girot, S.; Gaven, F.; Stephan, D.; Migliorati, M.; Khrestchatisky, M.; Bockaert, J.; Marchetti-Gauthier, E.; et al. Chronic Treatments with a 5-HT4 Receptor Agonist Decrease Amyloid Pathology in the Entorhinal Cortex and Learning and Memory Deficits in the 5xFAD Mouse Model of Alzheimer’s Disease. Neuropharmacology 2017, 126, 128–141. [Google Scholar] [CrossRef]
- Giannoni, P.; Gaven, F.; de Bundel, D.; Baranger, K.; Marchetti-Gauthier, E.; Roman, F.S.; Valjent, E.; Marin, P.; Bockaert, J.; Rivera, S.; et al. Early Administration of RS 67333, a Specific 5-HT4 Receptor Agonist, Prevents Amyloidogenesis and Behavioral Deficits in the 5XFAD Mouse Model of Alzheimer’s Disease. Front. Aging Neurosci. 2013, 5, 96. [Google Scholar] [CrossRef]
- Eydipour, Z.; Nasehi, M.; Vaseghi, S.; Jamaldini, S.H.; Zarrindast, M.-R. The Role of 5-HT4 Serotonin Receptors in the CA1 Hippocampal Region on Memory Acquisition Impairment Induced by Total (TSD) and REM Sleep Deprivation (RSD). Physiol. Behav. 2020, 215, 112788. [Google Scholar] [CrossRef]
- Murphy, S.E.; de Cates, A.N.; Gillespie, A.L.; Godlewska, B.R.; Scaife, J.C.; Wright, L.C.; Cowen, P.J.; Harmer, C.J. Translating the Promise of 5HT 4 Receptor Agonists for the Treatment of Depression. Psychol. Med. 2021, 51, 1111–1120. [Google Scholar] [CrossRef]
- Cachard-chastel, M.; Devers, S.; Sicsic, S.; Langlois, M.; Lezoualch, F.; Gardier, A.; Belzung, C. Prucalopride and Donepezil Act Synergistically to Reverse Scopolamine-Induced Memory Deficit in C57Bl/6j Mice. Behav. Brain Res. 2008, 187, 455–461. [Google Scholar] [CrossRef] [PubMed]
- Lecoutey, C.; Hedou, D.; Freret, T.; Giannoni, P.; Gaven, F.; Since, M.; Bouet, V.; Ballandonne, C.; Corvaisier, S.; Malzert Freon, A.; et al. Design of Donecopride, a Dual Serotonin Subtype 4 Receptor Agonist/Acetylcholinesterase Inhibitor with Potential Interest for Alzheimer’s Disease Treatment. Proc. Natl. Acad. Sci. USA 2014, 111, E3825–E3830. [Google Scholar] [CrossRef]
- Jansen, C.U.; Qvortrup, K.M. Small Molecule Drugs for Treatment of Alzheimer’s Diseases Developed on the Basis of Mechanistic Understanding of the Serotonin Receptors 4 and 6; IntechOpen: London, UK, 2021. [Google Scholar]
- Rebholz, H.; Friedman, E.; Castello, J. Alterations of Expression of the Serotonin 5-HT4 Receptor in Brain Disorders. IJMS 2018, 19, 3581. [Google Scholar] [CrossRef]
- Bonaventure, P.; Hall, H.; Gommeren, W.; Cras, P.; Langlois, X.; Jurzak, M.; Leysen, J.E. Mapping of Serotonin 5-HT(4) Receptor MRNA and Ligand Binding Sites in the Post-Mortem Human Brain. Synapse 2000, 36, 35–46. [Google Scholar] [CrossRef]
- Marner, L.; Gillings, N.; Madsen, K.; Erritzoe, D.; Baaré, W.F.C.; Svarer, C.; Hasselbalch, S.G.; Knudsen, G.M. Brain Imaging of Serotonin 4 Receptors in Humans with [11C]SB207145-PET. NeuroImage 2010, 50, 855–861. [Google Scholar] [CrossRef]
- Beliveau, V.; Ganz, M.; Feng, L.; Ozenne, B.; Højgaard, L.; Fisher, P.M.; Svarer, C.; Greve, D.N.; Knudsen, G.M. A High-Resolution In Vivo Atlas of the Human Brain’s Serotonin System. J. Neurosci. 2017, 37, 120–128. [Google Scholar] [CrossRef]
- Vilaró, M.T.; Cortés, R.; Mengod, G. Serotonin 5-HT4 Receptors and Their MRNAs in Rat and Guinea Pig Brain: Distribution and Effects of Neurotoxic Lesions: 5-HT4 Receptors in Rat and Guinea Pig Brain. J. Comp. Neurol. 2005, 484, 418–439. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, K.F.; Samuels, B.A.; Hen, R. Serotonin Receptor Expression along the Dorsal–Ventral Axis of Mouse Hippocampus. Phil. Trans. R. Soc. B 2012, 367, 2395–2401. [Google Scholar] [CrossRef]
- Haahr, M.E.; Fisher, P.; Holst, K.; Madsen, K.; Jensen, C.G.; Marner, L.; Lehel, S.; Baaré, W.; Knudsen, G.; Hasselbalch, S. The 5-HT4 Receptor Levels in Hippocampus Correlates Inversely with Memory Test Performance in Humans: The 5-HT4 Receptor and Memory Functions. Hum. Brain Mapp. 2013, 34, 3066–3074. [Google Scholar] [CrossRef] [PubMed]
- Madsen, K.; Neumann, W.-J.; Holst, K.; Marner, L.; Haahr, M.T.; Lehel, S.; Knudsen, G.M.; Hasselbalch, S.G. Cerebral Serotonin 4 Receptors and Amyloid-β in Early Alzheimer’s Disease. JAD 2011, 26, 457–466. [Google Scholar] [CrossRef]
- Meneses, A. Serotonin, Neural Markers, and Memory. Front. Pharmacol. 2015, 6, 143. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, G.P.; Mason, S.L.; Meldrum, A.; Keczer, S.; Parties, H.; Eglen, R.M.; Wong, E.H.F. 5-Hydroxytryptamine (5-HT)4 Receptors in Post Mortem Human Brain Tissue: Distribution, Pharmacology and Effects of Neurodegenerative Diseases. Br. J. Pharmacol. 1995, 114, 993–998. [Google Scholar] [CrossRef]
- Vidal, R.; Valdizán, E.M.; Mostany, R.; Pazos, A.; Castro, E. Long-Term Treatment with Fluoxetine Induces Desensitization of 5-HT4 Receptor-Dependent Signalling and Functionality in Rat Brain. J. Neurochem. 2009, 110, 1120–1127. [Google Scholar] [CrossRef] [PubMed]
- Licht, C.L.; Knudsen, G.M.; Sharp, T. Effects of the 5-HT4 Receptor Agonist RS67333 and Paroxetine on Hippocampal Extracellular 5-HT Levels. Neurosci. Lett. 2010, 476, 58–61. [Google Scholar] [CrossRef]
- Ge, J.; Barnes, N.M. 5-HT4 Receptor-Mediated Modulation of 5-HT Release in the Rat Hippocampus in Vivo. Br. J. Pharmacol. 1996, 117, 1475–1480. [Google Scholar] [CrossRef]
- Conductier, G.; Dusticier, N.; Lucas, G.; Côté, F.; Debonnel, G.; Daszuta, A.; Dumuis, A.; Nieoullon, A.; Hen, R.; Bockaert, J.; et al. Adaptive Changes in Serotonin Neurons of the Raphe Nuclei in 5-HT4 Receptor Knock-out Mouse. Eur. J. Neurosci. 2006, 24, 1053–1062. [Google Scholar] [CrossRef]
- Halliday, G.M.; McCann, H.L.; Pamphlett, R.; Brooks, W.S.; Creasey, H.; McCusker, E.; Cotton, R.G.H.; Broe, G.A.; Harper, C.G. Brain Stem Serotonin-Synthesizing Neurons in Alzheimer’s Disease: A Clinicopathological Correlation. Acta Neuropathol. 1992, 84, 638–650. [Google Scholar] [CrossRef]
- Ebinger, G.; Bruyland, M.; Martin, J.J.; Herregodts, P.; Cras, P.; Michotte, Y.; Gommé, L. Distribution of Biogenic Amines and Their Catabolites in Brains from Patients with Alzheimer’s Disease. J. Neurol. Sci. 1987, 77, 267–283. [Google Scholar] [CrossRef]
- Bowen, D.M.; Allen, S.J.; Benton, J.S.; Goodhardt, M.J.; Haan, E.A.; Palmer, A.M.; Sims, N.R.; Smith, C.C.T.; Spillane, J.A.; Esiri, M.M.; et al. Biochemical Assessment of Serotonergic and Cholinergic Dysfunction and Cerebral Atrophy in Alzheimer’s Disease. J. Neurochem. 1983, 41, 266–272. [Google Scholar] [CrossRef]
- Buhot, M.-C.; Martin, S.; Segu, L. Role of Serotonin in Memory Impairment. Ann. Med. 2000, 32, 210–221. [Google Scholar] [CrossRef] [PubMed]
- Small, S.A.; Schobel, S.A.; Buxton, R.B.; Witter, M.P.; Barnes, C.A. A Pathophysiological Framework of Hippocampal Dysfunction in Ageing and Disease. Nat. Rev. Neurosci. 2011, 12, 585–601. [Google Scholar] [CrossRef]
- Jack, C.R.; Petersen, R.C.; Xu, Y.; O’Brien, P.C.; Smith, G.E.; Ivnik, R.J.; Boeve, B.F.; Tangalos, E.G.; Kokmen, E. Rates of Hippocampal Atrophy Correlate with Change in Clinical Status in Aging and AD. Neurology 2000, 55, 484–489. [Google Scholar] [CrossRef]
- Sasabayashi, D.; Yoshimura, R.; Takahashi, T.; Takayanagi, Y.; Nishiyama, S.; Higuchi, Y.; Mizukami, Y.; Furuichi, A.; Kido, M.; Nakamura, M.; et al. Reduced Hippocampal Subfield Volume in Schizophrenia and Clinical High-Risk State for Psychosis. Front. Psychiatry 2021, 12, 642048. [Google Scholar] [CrossRef]
- Xu, R.; Hu, X.; Jiang, X.; Zhang, Y.; Wang, J.; Zeng, X. Longitudinal Volume Changes of Hippocampal Subfields and Cognitive Decline in Parkinson’s Disease. Quant. Imaging Med. Surg. 2020, 10, 220–232. [Google Scholar] [CrossRef]
- Santos, M.A.O.; Bezerra, L.S.; Carvalho, A.R.M.R.; Brainer-Lima, A.M. Global Hippocampal Atrophy in Major Depressive Disorder: A Meta-Analysis of Magnetic Resonance Imaging Studies. Trends Psychiatry Psychother. 2018, 40, 369–378. [Google Scholar] [CrossRef]
- Ruan, L.; Lau, B.W.-M.; Wang, J.; Huang, L.; ZhuGe, Q.; Wang, B.; Jin, K.; So, K.-F. Neurogenesis in Neurological and Psychiatric Diseases and Brain Injury: From Bench to Bedside. Progress Neurobiol. 2014, 115, 116–137. [Google Scholar] [CrossRef]
- Cachard-Chastel, M.; Lezoualc’h, F.; Dewachter, I.; Deloménie, C.; Croes, S.; Devijver, H.; Langlois, M.; Van Leuven, F.; Sicsic, S.; Gardier, A.M. 5-HT4 Receptor Agonists Increase SAPPα Levels in the Cortex and Hippocampus of Male C57BL/6j Mice: 5-HT4 Receptors and SAPPα in Vivo. Br. J. Pharmacol. 2007, 150, 883–892. [Google Scholar] [CrossRef]
- Lezoualc’h, F.; Robert, S.J. The Serotonin 5-HT4 Receptor and the Amyloid Precursor Protein Processing. Exp. Gerontol. 2003, 38, 159–166. [Google Scholar] [CrossRef]
- Robert, S.J.; Zugaza, J.L.; Fischmeister, R.; Gardier, A.M.; Lezoualc’h, F. The Human Serotonin 5-HT4 Receptor Regulates Secretion of Non-Amyloidogenic Precursor Protein. J. Biol. Chem. 2001, 276, 44881–44888. [Google Scholar] [CrossRef] [PubMed]
- Maillet, M.; Robert, S.J.; Cacquevel, M.; Gastineau, M.; Vivien, D.; Bertoglio, J.; Zugaza, J.L.; Fischmeister, R.; Lezoualc’h, F. Crosstalk between Rap1 and Rac Regulates Secretion of SAPPalpha. Nat. Cell Biol. 2003, 5, 633–639. [Google Scholar] [CrossRef] [PubMed]
- Cochet, M.; Donneger, R.; Cassier, E.; Gaven, F.; Lichtenthaler, S.F.; Marin, P.; Bockaert, J.; Dumuis, A.; Claeysen, S. 5-HT4 Receptors Constitutively Promote the Non-Amyloidogenic Pathway of APP Cleavage and Interact with ADAM10. ACS Chem. Neurosci. 2013, 4, 130–140. [Google Scholar] [CrossRef]
- Hashimoto, G.; Sakurai, M.; Teich, A.F.; Saeed, F.; Aziz, F.; Arancio, O. 5-HT₄ Receptor Stimulation Leads to Soluble AβPPα Production through MMP-9 Upregulation. J. Alzheimer’s Dis. 2012, 32, 437–445. [Google Scholar] [CrossRef]
- Tesseur, I.; Pimenova, A.A.; Lo, A.C.; Ciesielska, M.; Lichtenthaler, S.F.; De Maeyer, J.H.; Schuurkes, J.A.J.; D’Hooge, R.; De Strooper, B. Chronic 5-HT4 Receptor Activation Decreases Aβ Production and Deposition in HAPP/PS1 Mice. Neurobiol. Aging 2013, 34, 1779–1789. [Google Scholar] [CrossRef]
- Cho, S.; Hu, Y. Activation of 5-HT4 Receptors Inhibits Secretion of β-Amyloid Peptides and Increases Neuronal Survival. Exp. Neurol. 2007, 203, 274–278. [Google Scholar] [CrossRef]
- Koffie, R.M.; Hyman, B.T.; Spires-Jones, T.L. Alzheimer’s Disease: Synapses Gone Cold. Mol. Neurodegeneration 2011, 6, 63. [Google Scholar] [CrossRef]
- Schill, Y.; Bijata, M.; Kopach, O.; Cherkas, V.; Abdel-Galil, D.; Böhm, K.; Schwab, M.H.; Matsuda, M.; Compan, V.; Basu, S.; et al. Serotonin 5-HT4 Receptor Boosts Functional Maturation of Dendritic Spines via RhoA-Dependent Control of F-Actin. Commun. Biol. 2020, 3, 76. [Google Scholar] [CrossRef]
- Kozono, N.; Ohtani, A.; Shiga, T. Roles of the Serotonin 5-HT4 Receptor in Dendrite Formation of the Rat Hippocampal Neurons in Vitro. Brain Res. 2017, 1655, 114–121. [Google Scholar] [CrossRef]
- Preman, P.; Alfonso-Triguero, M.; Alberdi, E.; Verkhratsky, A.; Arranz, A.M. Astrocytes in Alzheimer’s Disease: Pathological Significance and Molecular Pathways. Cells 2021, 10, 540. [Google Scholar] [CrossRef]
- Saura, C.A.; Valero, J. The Role of CREB Signaling in Alzheimer’s Disease and Other Cognitive Disorders. Rev. Neurosci. 2011, 22, 153–169. [Google Scholar] [CrossRef]
- Clapp, W.C.; Hamm, J.P.; Kirk, I.J.; Teyler, T.J. Translating Long-Term Potentiation from Animals to Humans: A Novel Method for Noninvasive Assessment of Cortical Plasticity. Biol. Psychiatry 2012, 71, 496–502. [Google Scholar] [CrossRef] [PubMed]
- Freitas, C.; Perez, J.; Knobel, M.; Tormos, J.M.; Oberman, L.; Eldaief, M.; Bashir, S.; Vernet, M.; Peña-Gómez, C.; Pascual-Leone, A. Changes in Cortical Plasticity Across the Lifespan. Front. Aging Neurosci. 2011, 3, 5. [Google Scholar] [CrossRef]
- Kuhn, M.; Mainberger, F.; Feige, B.; Maier, J.G.; Mall, V.; Jung, N.H.; Reis, J.; Klöppel, S.; Normann, C.; Nissen, C. State-Dependent Partial Occlusion of Cortical LTP-Like Plasticity in Major Depression. Neuropsychopharmacology 2016, 41, 1521–1529. [Google Scholar] [CrossRef]
- Cantone, M.; Bramanti, A.; Lanza, G.; Pennisi, M.; Bramanti, P.; Pennisi, G.; Bella, R. Cortical Plasticity in Depression: A Neurochemical Perspective From Transcranial Magnetic Stimulation. ASN Neuro 2017, 9, 175909141771151. [Google Scholar] [CrossRef]
- Hasan, A.; Nitsche, M.A.; Rein, B.; Schneider-Axmann, T.; Guse, B.; Gruber, O.; Falkai, P.; Wobrock, T. Dysfunctional Long-Term Potentiation-like Plasticity in Schizophrenia Revealed by Transcranial Direct Current Stimulation. Behav. Brain Res. 2011, 224, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, H.K.; Roach, B.J.; Cavus, I.; Teyler, T.J.; Clapp, W.C.; Ford, J.M.; Tarakci, E.; Krystal, J.H.; Mathalon, D.H. Impaired Potentiation of Theta Oscillations During a Visual Cortical Plasticity Paradigm in Individuals With Schizophrenia. Front. Psychiatry 2020, 11, 590567. [Google Scholar] [CrossRef]
- Di Lorenzo, F.; Ponzo, V.; Bonnì, S.; Motta, C.; Negrão Serra, P.C.; Bozzali, M.; Caltagirone, C.; Martorana, A.; Koch, G. Long-Term Potentiation-like Cortical Plasticity Is Disrupted in Alzheimer’s Disease Patients Independently from Age of Onset: Cortical Plasticity in Alzheimer’s Disease. Ann. Neurol. 2016, 80, 202–210. [Google Scholar] [CrossRef] [PubMed]
- Lynch, G.; Rex, C.S.; Gall, C.M. Synaptic Plasticity in Early Aging. Ageing Res. Rev. 2006, 5, 255–280. [Google Scholar] [CrossRef]
- Marchetti, C.; Marie, H. Hippocampal Synaptic Plasticity in Alzheimer’s Disease: What Have We Learned so Far from Transgenic Models? Rev. Neurosci. 2011, 22, 373–402. [Google Scholar] [CrossRef] [PubMed]
- Percelay, S.; Billard, J.-M.; Freret, T.; Andrieux, A.; Boulouard, M.; Bouet, V. Functional Dysregulations in CA1 Hippocampal Networks of a 3-Hit Mouse Model of Schizophrenia. IJMS 2021, 22, 2644. [Google Scholar] [CrossRef]
- Yang, Y.; Ju, W.; Zhang, H.; Sun, L. Effect of Ketamine on LTP and NMDAR EPSC in Hippocampus of the Chronic Social Defeat Stress Mice Model of Depression. Front. Behav. Neurosci. 2018, 12, 229. [Google Scholar] [CrossRef]
- Wawra, M.; Fidzinski, P.; Heinemann, U.; Mody, I.; Behr, J. 5-HT4-Receptors Modulate Induction of Long-Term Depression but Not Potentiation at Hippocampal Output Synapses in Acute Rat Brain Slices. PLoS ONE 2014, 9, e88085. [Google Scholar] [CrossRef]
- Twarkowski, H.; Hagena, H.; Manahan-Vaughan, D. The 5-Hydroxytryptamine 4 Receptor Enables Differentiation of Informational Content and Encoding in the Hippocampus: Role of 5-HT4 Receptor in Hippocampal Plasticity. Hippocampus 2016, 26, 875–891. [Google Scholar] [CrossRef]
- Marchetti, E.; Chaillan, F.A.; Dumuis, A.; Bockaert, J.; Soumireu-Mourat, B.; Roman, F.S. Modulation of Memory Processes and Cellular Excitability in the Dentate Gyrus of Freely Moving Rats by a 5-HT4 Receptors Partial Agonist, and an Antagonist. Neuropharmacology 2004, 47, 1021–1035. [Google Scholar] [CrossRef] [PubMed]
- Kulla, A.; Manahan-Vaughan, D. Modulation by Serotonin 5-HT4 Receptors of Long-Term Potentiation and Depotentiation in the Dentate Gyrus of Freely Moving Rats. Cereb. Cortex 2002, 12, 150–162. [Google Scholar] [CrossRef] [PubMed]
- Kemp, A.; Manahan-Vaughan, D. The 5-Hydroxytryptamine4 Receptor Exhibits Frequency-Dependent Properties in Synaptic Plasticity and Behavioural Metaplasticity in the Hippocampal CA1 Region In Vivo. Cereb. Cortex 2005, 15, 1037–1043. [Google Scholar] [CrossRef]
- Perez, Y.; Chapman, C.A.; Woodhall, G.; Robitaille, R.; Lacaille, J.-C. Differential Induction of Long-Lasting Potentiation of Inhibitory Postsynaptic Potentials by Theta Patterned Stimulation versus 100-Hz Tetanization in Hippocampal Pyramidal Cells in Vitro. Neuroscience 1999, 90, 747–757. [Google Scholar] [CrossRef]
- Takeuchi, T.; Duszkiewicz, A.J.; Morris, R.G.M. The Synaptic Plasticity and Memory Hypothesis: Encoding, Storage and Persistence. Phil. Trans. R. Soc. B 2014, 369, 20130288. [Google Scholar] [CrossRef]
- Barnes, C.; Jung, M.; McNaughton, B.; Korol, D.; Andreasson, K.; Worley, P. LTP Saturation and Spatial Learning Disruption: Effects of Task Variables and Saturation Levels. J. Neurosci. 1994, 14, 5793–5806. [Google Scholar] [CrossRef]
- Lee, H.-K.; Kirkwood, A. Mechanisms of Homeostatic Synaptic Plasticity in Vivo. Front. Cell Neurosci. 2019, 13, 520. [Google Scholar] [CrossRef] [PubMed]
- Murphy, S.E.; Wright, L.C.; Browning, M.; Cowen, P.J.; Harmer, C.J. A Role for 5-HT4 Receptors in Human Learning and Memory. Psychol. Med. 2020, 50, 2722–2730. [Google Scholar] [CrossRef]
- Bakker, A.; Krauss, G.L.; Albert, M.S.; Speck, C.L.; Jones, L.R.; Stark, C.E.; Yassa, M.A.; Bassett, S.S.; Shelton, A.L.; Gallagher, M. Reduction of Hippocampal Hyperactivity Improves Cognition in Amnestic Mild Cognitive Impairment. Neuron 2012, 74, 467–474. [Google Scholar] [CrossRef] [PubMed]
- Mlinar, B.; Pugliese, A.M.; Corradetti, R. Selective Inhibition of Local Excitatory Synaptic Transmission by Serotonin through an Unconventional Receptor in the CA1 Region of Rat Hippocampus. J. Physiol. 2001, 534, 141–158. [Google Scholar] [CrossRef]
- Spencer, J.P.; Brown, J.T.; Richardson, J.C.; Medhurst, A.D.; Sehmi, S.S.; Calver, A.R.; Randall, A.D. Modulation of Hippocampal Excitability by 5-HT4 Receptor Agonists Persists in a Transgenic Model of Alzheimer’s Disease. Neuroscience 2004, 129, 49–54. [Google Scholar] [CrossRef] [PubMed]
- Karayol, R.; Medrihan, L.; Warner-Schmidt, J.L.; Fait, B.W.; Rao, M.N.; Holzner, E.B.; Greengard, P.; Heintz, N.; Schmidt, E.F. Serotonin Receptor 4 in the Hippocampus Modulates Mood and Anxiety. Mol. Psychiatry 2021, 26, 2334–2349. [Google Scholar] [CrossRef]
- Micale, V.; Marco Leggio, G.; Mazzola, C.; Drago, F. Cognitive Effects of SL65.0155, a Serotonin 5-HT4 Receptor Partial Agonist, in Animal Models of Amnesia. Brain Res. 2006, 1121, 207–215. [Google Scholar] [CrossRef]
- Shen, F.; Smith, J.A.M.; Chang, R.; Bourdet, D.L.; Tsuruda, P.R.; Obedencio, G.P.; Beattie, D.T. 5-HT4 Receptor Agonist Mediated Enhancement of Cognitive Function in Vivo and Amyloid Precursor Protein Processing in Vitro: A Pharmacodynamic and Pharmacokinetic Assessment. Neuropharmacology 2011, 61, 69–79. [Google Scholar] [CrossRef]
- De Cates, A.N.; Wright, L.C.; Martens, M.A.G.; Gibson, D.; Türkmen, C.; Filippini, N.; Cowen, P.J.; Harmer, C.J.; Murphy, S.E. Déjà-vu? Neural and Behavioural Effects of the 5-HT4 Receptor Agonist, Prucalopride, in a Hippocampal-Dependent Memory Task. Transl. Psychiatry 2021, 11, 497. [Google Scholar] [CrossRef]
- Frankland, P.W.; Bontempi, B. The Organization of Recent and Remote Memories. Nat. Rev. Neurosci. 2005, 6, 119–130. [Google Scholar] [CrossRef] [PubMed]
- Roux, C.M.; Lecouflet, P.; Billard, J.-M.; Corvaisier, S.; Schumann-Bard, P.; Leger, M.; Freret, T. Effects of 5-HT4Rs Activation on CA1 Hippocampal Synaptic Plasticity: An Add-on to the GABAergic Hypothesis. Neurofrance 2021. Unpublished Data. Abstract A-1225-0001-00696. [Google Scholar]
| Alteration | Contributing Factor | 5-HT4Rs Agonist | Dose/Con. | Treatment Duration | Preclinical Model | Target Brain Disease | Outcome of 5-HT4Rs Activation | References |
|---|---|---|---|---|---|---|---|---|
| Aβ-mediated cell death (Dysfunction in APP metabolism) | VRX-0311 | 0.1 nM–10 µM | 30 min | CHO cells stably expressing the human 5-HT4(e) receptor and APP695 | AD | Concentration-dependent ↑ sAPPα | Mohler et al. 2007 | |
| Prucalopride | 1 µM | 2 h | HEK-293 expressing SEAP-tagged APP and 5-HT4Rs | AD | ↑ sAPPα secretion (50%) through stimulation of α-secretase | Cochet et al. 2013 | ||
| Prucalopride | 1 µM | 30 min | CHO cell line expressing sAPPa and 5-HT4Rs | AD | ↑ sAPPα secretion | Lezoualc’h and Robert, 2003 Robert et al. 2001 | ||
| Hippocampal volume loss | SSP-002392 | 5 mg/kg | 26—37 days | APP/PS1 mice (4–5 and 12 months) | AD | ↓ soluble and insoluble hippocampal Aβ40 and Aβ42 ↓ total number of Aβ deposits in mice aged 4–5 months ↓ Bace-1, Adam17 (50%) and Nicastrin ↑ astrogliosis and microgliosis (Aβ degradation) | Tesseur et al. 2013 | |
| 10 nM | SH-SY5Y human neuroblastoma cell line | NA | ↑ sAPPα release | |||||
| RS67333 | 2 mg/kg | APP/PS1 mice (7–8 months) | AD | No change in Aβ | ||||
| RS67333 | 3 µM | 1 h, 2 h, 4 h, 8 h, 24 h and 48 h | H4/AβPP/5-HT4 cells | AD | ↑ sAPPα production (102%, 265%, 343% of control at 8 h, 24 h, and 48 h respectively) through MMP-9 (role in α-secretase activity) | Hashimoto et al. 2012 | ||
| RS67333 | 3 mg/kg | 10 days | Female Tg2576 transgenic mice (10–12 months) | AD | ↓ in Aβ load (30%) | |||
| RS67333 | - | 30 min | COS-7 cells transiently expressing 5-HT4Rs and SEAP-APP | AD | ↑ sAPPα release | Giannoni et al. 2013 | ||
| RS67333 | 1 mg/kg.d (twice a week) | 3 months | 5xFAD female mice | AD | ↑ hippocampal sAPPα (1.5 fold) ↓ in Aβ load (hippocampus: 48%, EC: 55%) ↓ Aβ42 levels in the insoluble and soluble factions (33% and 53 % respectively) | |||
| RS67333 | 1 mg/kg.d (twice a week) | 3 months | 5xFAD female mice | AD | ↓ in Aβ load in EC (31–33%) | Baranger et al. 2017 | ||
| RS67333 | 0.01µM-100µM | 2 days exposure | Cortical Primary culture from Tg2576 mice | AD | Dose-dependent ↓ of Aβ levels 90–95% depletion of both Aβ40 and Aβ42 at 30μM Protection from Aβ-mediated cell death (increase in neuronal survival) | Cho and Hu, 2007 | ||
| Neuro-inflammation | RS67333 | 1 mg/kg.d | 2 weeks | 5xFAD male mice (4 months) | NA | ↓ astroglial reactivity (61%) ↓ pro-inflammatory mediators IL-1β (25%) and MCP-1 30%) after 4 months’ treatment | Baranger et al. 2017 | |
| RS67333 | 1 mg/kg.d (twice a week | 3 months | 5xFAD male mice | AD | ↓ astrogliosis (49%) ↓ microgliosis (57%) | Giannoni et al. 2013 | ||
| Network plasticity impairments | Synaptic loss and connectivity alterations | SL65.0155 | 0.01 mg/kg | 4 days | Adult C57BL/6J mice | NA | Potentiates learning-induced spine growth (+6% relative to controls) | Restivo et al. 2008 |
| BIMU-8 | 10 µM | 10 min | N1E-155 Neuroblastoma cells | NA | Boosts phosphorylation of cofilin (regulator of neuronal morphology and spinogenesis) | Schill et al. 2020 | ||
| Hippocampal primary culture from C57BL/6J mice | NA | Prompts dendritic spine maturation (increasing the number of active axo-spinous excitatory synapses in dendritic branches of principal neurons) Boosts numbers of excitatory synapses | ||||||
| ↓ plasticity-related proteins | Prucalopride Velusetrag | 1.5- 3 mg/kg 3 mg/kg | Single dose | (MPTP)-induced PD model mice | PD | ↑ cAMP levels (with stronger effect of Velusetrag) ↑ pCREB positive cells in DG | Ishii et al. 2019 | |
| RS67333 | 1.5 mg/kd.d | 3–7 days | Adult male Sprague-Dawley rats | NA | ↑ pCREB/CREB ratio | Pascual-Brazo et al. 2011 | ||
| SSP-002392 Prucalopride | 0.0001 -1 mmol/L 0.01–1 mmol/L | SH-SY5Y human neuroblastoma cells | NA | ↑ cAMP production (with stronger effect of SSP-002392) | Tesseur et al. 2013 | |||
| Inhibitory vs. excitatory imbalance | BIMU-8 | 0.2–2 µM | 45 min | Guinea pig hippocampal slices | NA | Ach-dependent increase in electrically-evoked GABA release at low concentration (0.2–0.4 µM) Ach-dependent inhibition of electrically-evoked GABA release at higher concentration (0.7–2 µM) | Bianchi et al. 2002 | |
| Zacopride | 10 µM | 5 min | Guinea pig hippocampal slices | NA | ↑ IPSPs | Bijak and Misgeld, 1997 | ||
| VRX-03011 | 1–5 mg/kg | Single dose | Adult male Long Evans rats | NA | ↑ Ach outflow under mnemonic demand | Mohler et al. 2007 | ||
| Renzapride | 1 mg/kg (systemic) 100 µM (icv) | Single dose | Adult female Wistar rats | NA | Concentration-dependent ↑ hippocampal 5-HT levels (200%) | Ge and Barnes 1996 | ||
| RS67333 | 1.5 mg/kg.d | 3 days | Adult male Sprague-Dawley rats | NA | ↑ 5-HT levels (73%) ↓ 5-HIAA levels (27%) (no effect of 5-HT4Rs agonist in acute conditions) | Licht et al. 2010 | ||
| BIMU-8 | 0.2–4 µM | 5 min | Guinea pig hippocampal slices | NA | ↑ Ach outflow after electrical stimulation (but not at rest) | Siniscalchi et al. 1999 |
| Method | Hippocampal Area | Plasticity | Conditioning Stimulus | 5-HT4Rs Agonist | Effects of 5-HT4Rs Activation on Plasticity | Reference |
|---|---|---|---|---|---|---|
| In vivo | DG | LTP | HFS (200 Hz) | RS67333 | ↓ | Kulla and Manahan-Vaughan |
| LTP | HFS (200 Hz) | 5-Methoxytryptamine | = | |||
| LTP | HFS (10 × 400 Hz) | RS67333 | Transient ↑ and curtailed | Marchetti et al. 2004 | ||
| LTP | HFS (200 Hz) | RS67333 | Curtailed | Twarkowski et al. 2016 | ||
| DP | LFS (5 Hz) | RS67333 | Blocked | |||
| LTD | LFS (1 Hz) | RS67633 | ↓ | |||
| CA3 | LTP | HFS (4 × 100 Hz) | RS67333 | ↓ | Twarkowski et al. 2016 | |
| LTD | LFS (1 Hz) | RS67333 | ↓ | |||
| CA1 | LTP | HFS (5 × 400 Hz) | SC53116 | ↑ | Matsumoto et al. 2001 | |
| LTP | HFS (4 × 100 Hz) | RS67333 | = | Kemp and Manahan-Vaughan 2005 | ||
| LTD | LFS (1 Hz) | RS67333 | ↓ | |||
| Ex vivo | CA1 | LTP | HFS (1 × 100 Hz) | RS67333 | = | Lecouflet et al. 2020 |
| LTP | TBS (4 × 5 Hz) | RS67333 | ↓ | |||
| SUB | LTP | HFS (4 × 100 Hz) | RS67333 | = | ||
| LTD | LFS (1 Hz) | RS67333 | ↑ | Wawra et al. 2014 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Roux, C.M.; Leger, M.; Freret, T. Memory Disorders Related to Hippocampal Function: The Interest of 5-HT4Rs Targeting. Int. J. Mol. Sci. 2021, 22, 12082. https://doi.org/10.3390/ijms222112082
Roux CM, Leger M, Freret T. Memory Disorders Related to Hippocampal Function: The Interest of 5-HT4Rs Targeting. International Journal of Molecular Sciences. 2021; 22(21):12082. https://doi.org/10.3390/ijms222112082
Chicago/Turabian StyleRoux, Candice M., Marianne Leger, and Thomas Freret. 2021. "Memory Disorders Related to Hippocampal Function: The Interest of 5-HT4Rs Targeting" International Journal of Molecular Sciences 22, no. 21: 12082. https://doi.org/10.3390/ijms222112082
APA StyleRoux, C. M., Leger, M., & Freret, T. (2021). Memory Disorders Related to Hippocampal Function: The Interest of 5-HT4Rs Targeting. International Journal of Molecular Sciences, 22(21), 12082. https://doi.org/10.3390/ijms222112082






