Development of Nickel- and Magnetite-Promoted Carbonized Cellulose Bead-Supported Bimetallic Pd–Pt Catalysts for Hydrogenation of Chlorate Ions in Aqueous Solution
Abstract
:1. Introduction
2. Results and Discussion
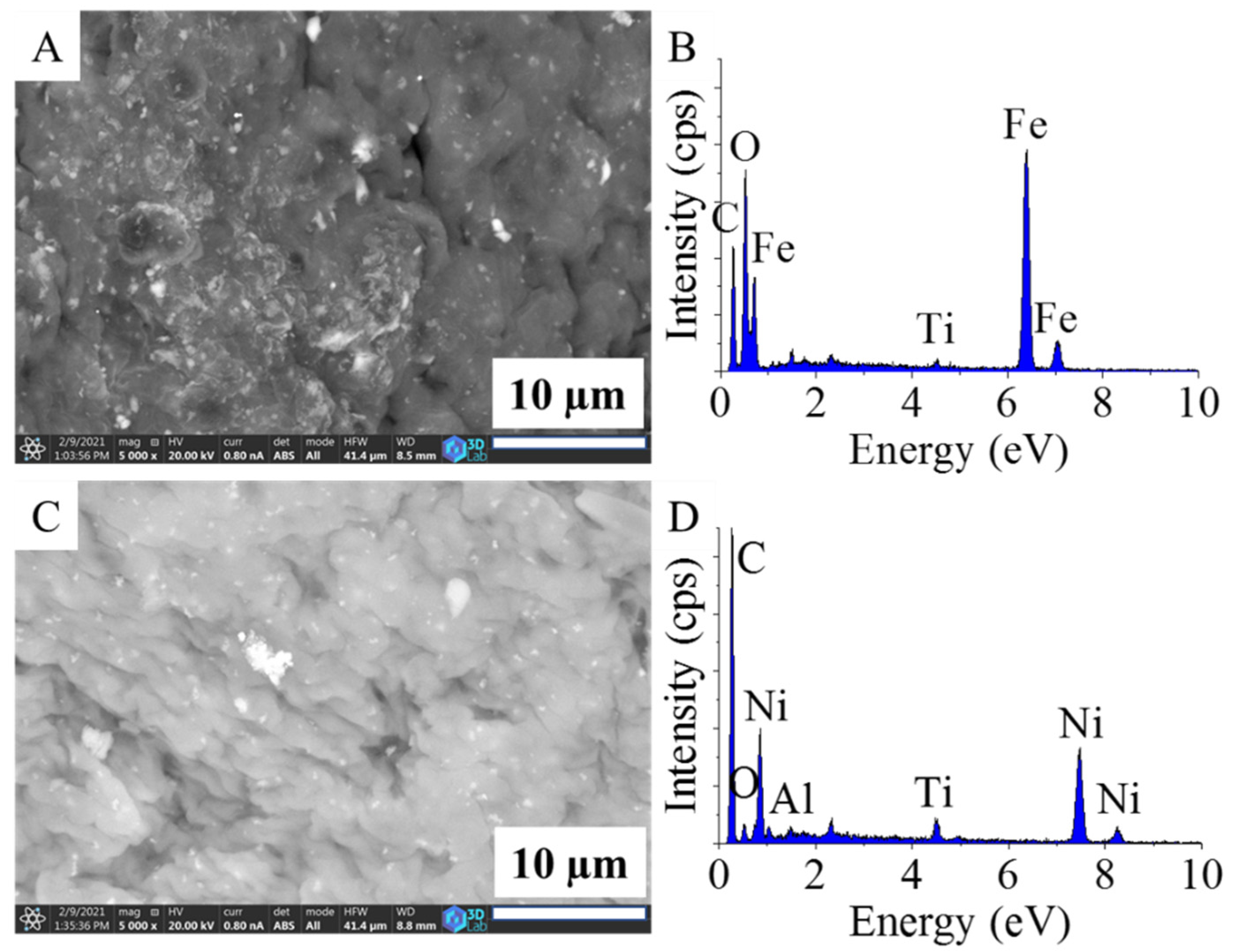
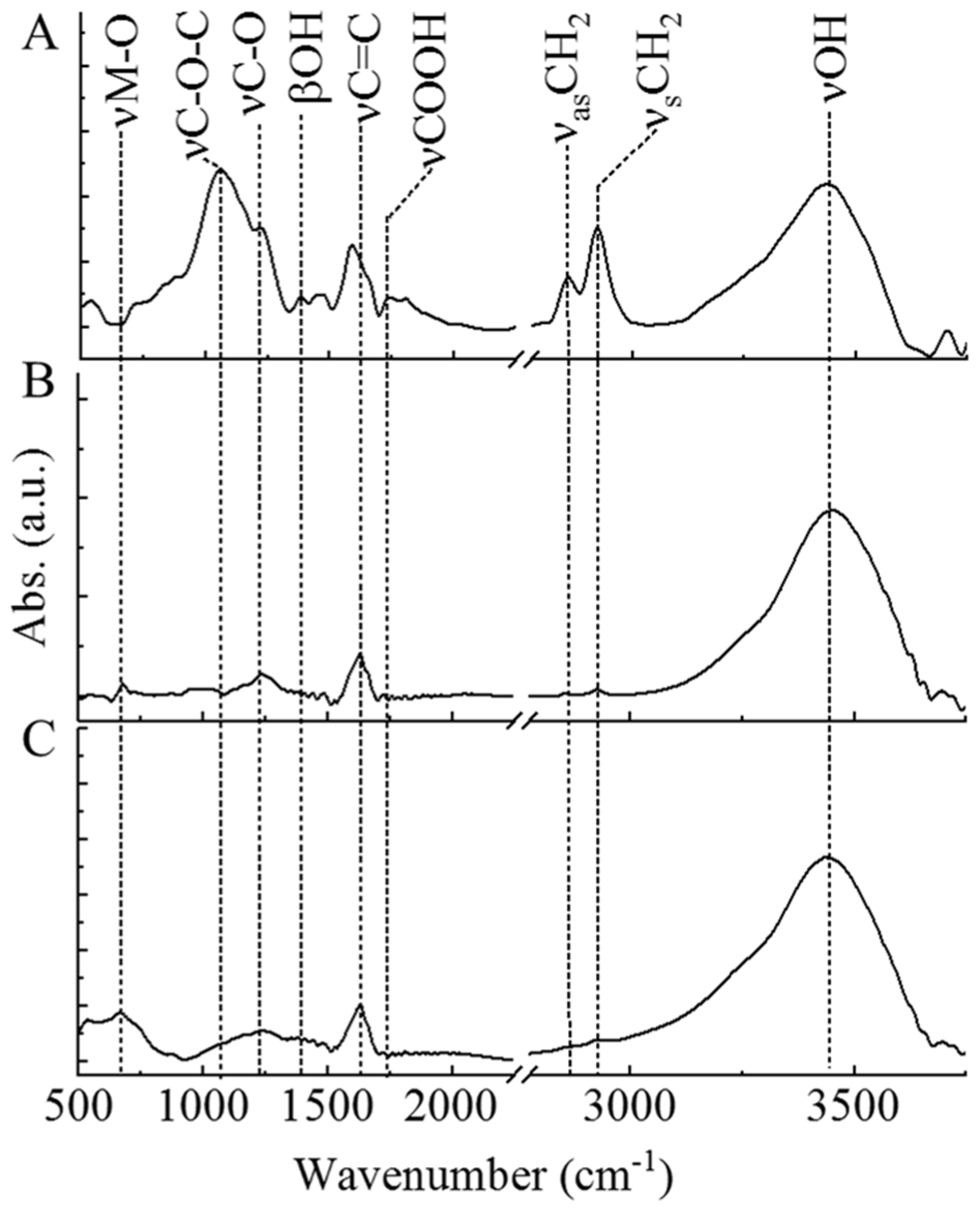
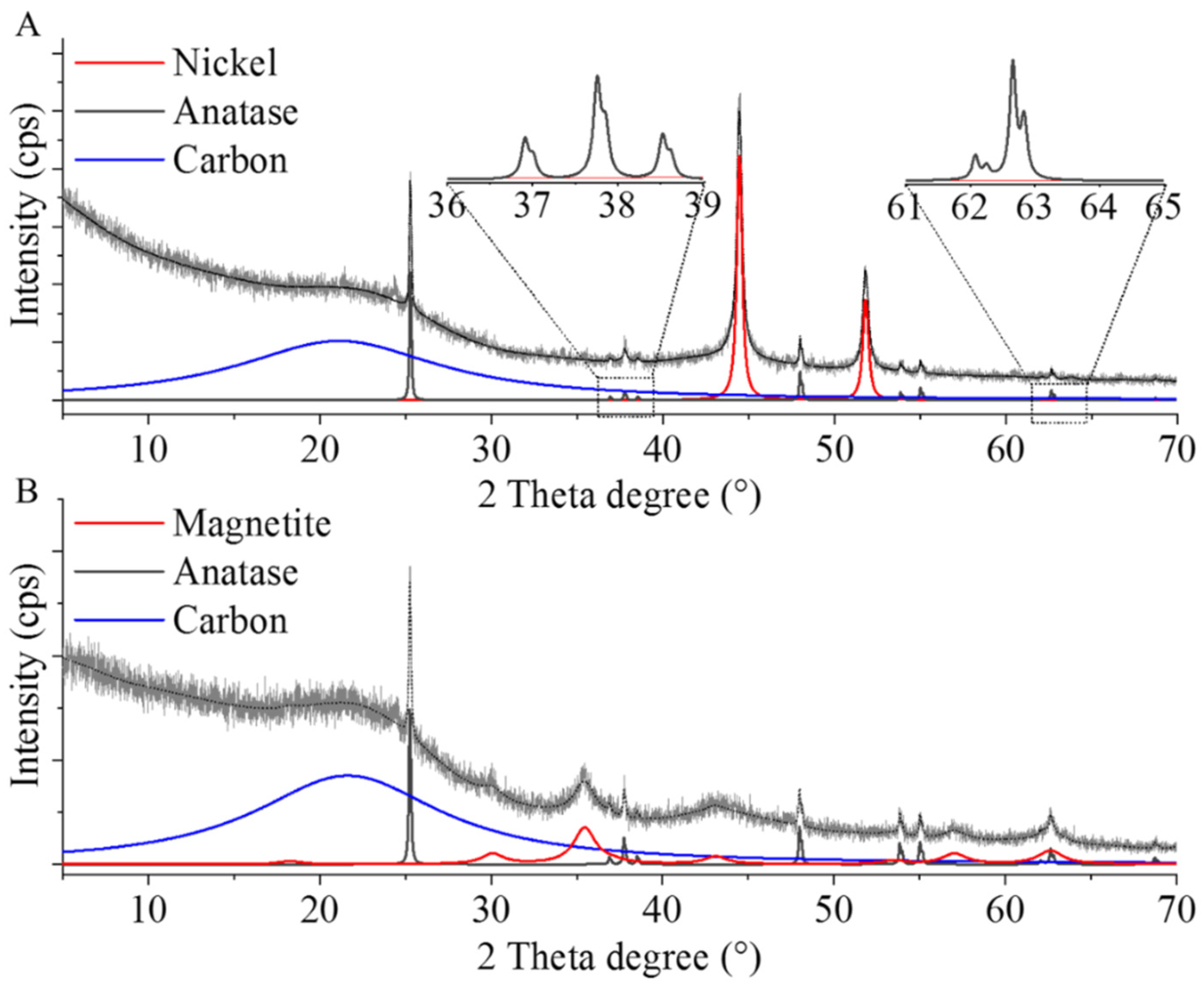
2.1. Characterization of the Catalyst Supports
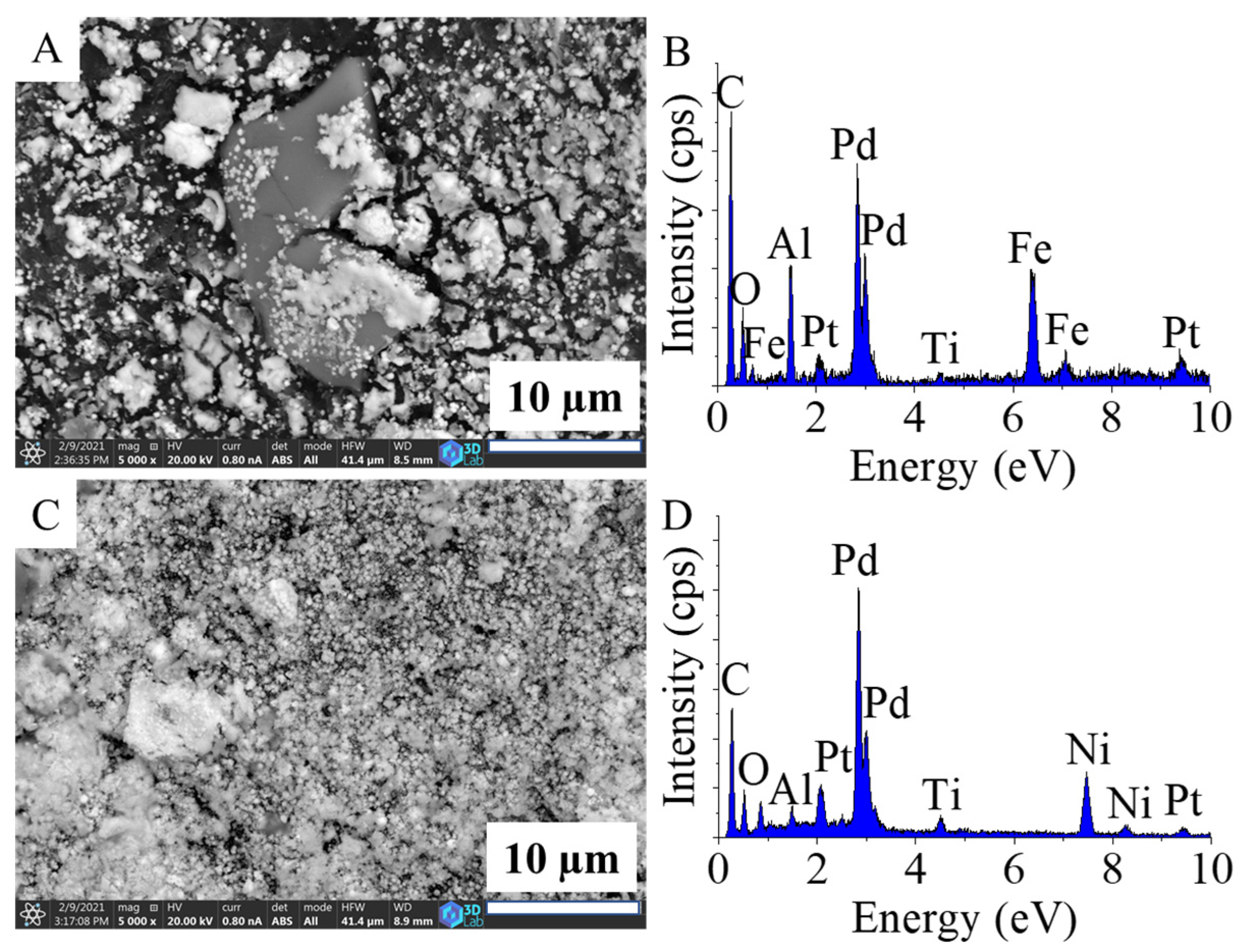
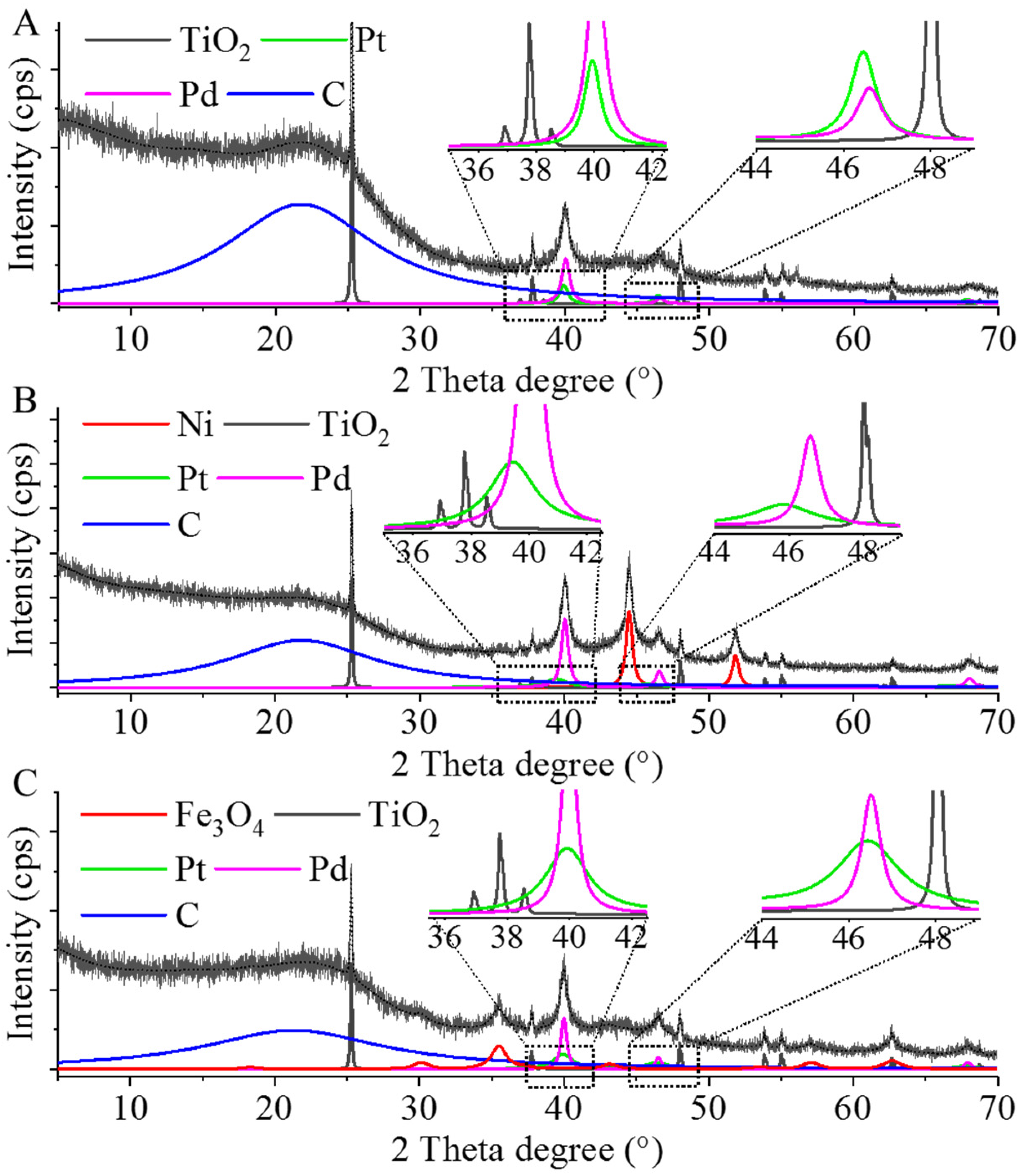
2.2. Characterization of the Pd–Pt/CCB, Pd–Pt/Ni–CCB, and Pd–Pt/Fe3O4–CCB Catalysts
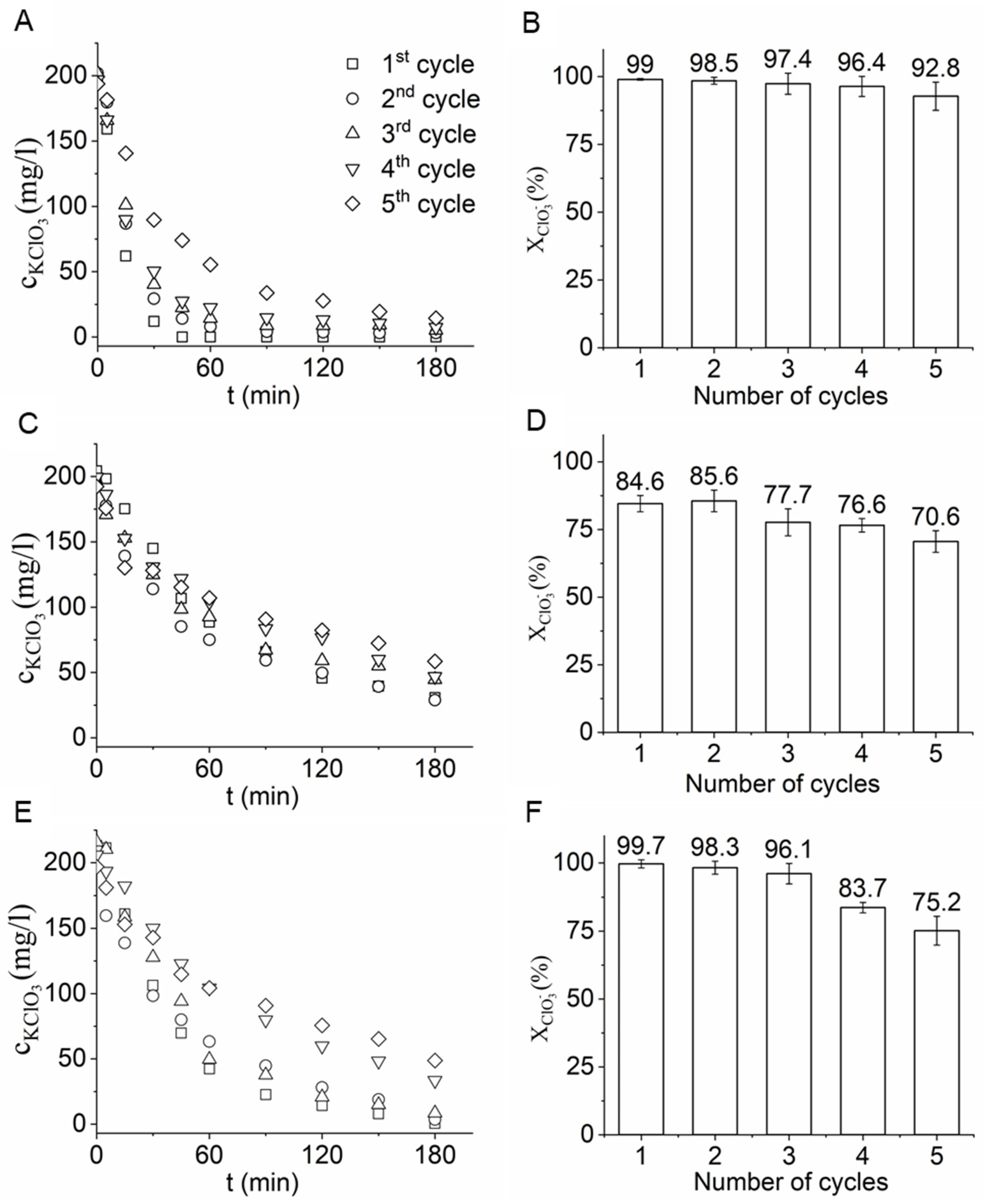
2.3. Catalytic Hydrogenation of Chlorate Ions in Aqueous Solution
3. Materials and Methods
3.1. Materials
3.2. Catalyst Preparation
3.3. Characterization Techniques
3.4. Catalytic Hydrogenation of Chlorate
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ren, C.; Yang, P.; Gao, J.; Huo, X.; Min, X.; Bi, E.Y.; Liu, Y.; Wang, Y.; Zhu, M.; Liu, J. Catalytic reduction of aqueous chlorate with MoOx immobilized on Pd/C. ACS Catal. 2020, 10, 8201–8211. [Google Scholar] [CrossRef]
- WHO. Chlorite and chlorate in drinking-water. In Guidelines for Drinking-Water Quality; World Health Organization (WHO): Geneva, Switzerland, 2005; p. 31. [Google Scholar]
- Brinkmann, T.; Giner Santonja, G.; Schorcht, F.; Roudier, S.; Delgado Sancho, L. Best Available Techniques Reference Document for the Production of Chlor-alkali; Jrc Science and Policy Reports: Luxembourg, 2014. [Google Scholar] [CrossRef]
- Jakab-Nácsa, A.; Stomp, D.; Farkas, L.; Kaptay, G. Large NaCl-effect on the decomposition rate of chlorate ions in HCl-containing brine solutions and its consequences for the chlor-alkali industry. Period. Polytech. Chem. Eng. 2020, 65, 238–242. [Google Scholar] [CrossRef]
- Rohonczy, J. Szervetlen Kémia, I. Az s-és p-Mező Elemeinek Kémiája Egyetemi Jegyzet; Eötvös Lóránd Tudományegyetem TTK: Budapest, Hungary, 2017; ISBN 9788632848532. [Google Scholar]
- Chlorine. Available online: http://www.essentialchemicalindustry.org/chemicals/chlorine.html (accessed on 26 November 2019).
- Maslamani, N.; Khan, S.B.; Danish, E.Y.; Bakhsh, E.M.; Zakeeruddin, S.M.; Asiri, A.M. Carboxymethyl cellulose nanocomposite beads as super-efficient catalyst for the catalytic reduction of organic and inorganic pollutants. Int. J. Biol. Macromol. 2020, 167, 101–116. [Google Scholar] [CrossRef]
- Bergner, D.; Hartmann, M. Chlorate and oxygen formation in alkali chloride membrane electrolysis. J. Appl. Electrochem. 1993, 23, 103–107. [Google Scholar] [CrossRef]
- Millet, P. Chlor-alkali technology: Fundamentals, processes and materials for diaphragms and membranes. In Handbook of Membrane Reactors; Woodhead Publishing: Sawston, UK, 2013; Volume 2, pp. 384–415. ISBN 9780857097347. [Google Scholar]
- Whiting, G.T.; Chung, S.H.; Stosic, D.; Chowdhury, A.D.; Van Der Wal, L.I.; Fu, D.; Zecevic, J.; Travert, A.; Houben, K.; Baldus, M.; et al. Multiscale mechanistic insights of shaped catalyst body formulations and their impact on catalytic properties. ACS Catal. 2019, 9, 4792–4803. [Google Scholar] [CrossRef]
- Moon, D.H.; Chung, W.J.; Chang, S.W.; Lee, S.M.; Kim, S.S.; Jeung, J.H.; Ro, Y.H.; Ahn, J.Y.; Guo, W.; Ngo, H.H.; et al. Fabrication and characterization of Ni-Ce-Zr ternary disk-shaped catalyst and its application for low-temperature CO2 methanation. Fuel 2020, 260, 116260. [Google Scholar] [CrossRef]
- Sevilla, M.; Fuertes, A.B. Graphitic carbon nanostructures from cellulose. Chem. Phys. Lett. 2010, 490, 63–68. [Google Scholar] [CrossRef] [Green Version]
- Habibi, Y.; Lucia, L.A.; Rojas, O.J. Cellulose nanocrystals: Chemistry, self-assembly, and applications. Chem. Rev. 2010, 110, 3479–3500. [Google Scholar] [CrossRef]
- Cheng, F.; Zhang, X.; Yang, X.; Li, R.; Wu, Y. Research on carbonization kinetic of cellulose-based materials and its application. J. Anal. Appl. Pyrolysis 2021, 158, 105232. [Google Scholar] [CrossRef]
- Zhou, W.; Jiang, C.; Duan, X.; Song, J.; Yuan, Y.; Chen, N. Fe3O4/carbonized cellulose micro-nano hybrid for high-performance microwave absorber. Carbohydr. Polym. 2020, 245, 116531. [Google Scholar] [CrossRef]
- Hwang, H.-C.; Woo, J.S.; Park, S.-Y. Flexible carbonized cellulose/single-walled carbon nanotube films with high conductivity. Carbohydr. Polym. 2018, 196, 168–175. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Lin, Z.; Zheng, M.; Wang, T.; Yang, J.; Yuan, F.; Lu, X.; Liu, L.; Sun, D. Amorphous Fe2O3 nanoshells coated on carbonized bacterial cellulose nanofibers as a flexible anode for high-performance lithium ion batteries. J. Power Sources 2016, 307, 649–656. [Google Scholar] [CrossRef]
- Prekob, A.; Hajdu, V.; Muránszky, G.; Fiser, B.; Sycheva, A.; Ferenczi, T.; Viskolcz, B.; Vanyorek, L. Application of carbonized cellulose-based catalyst in nitrobenzene hydrogenation. Mater. Today Chem. 2020, 17, 100337. [Google Scholar] [CrossRef]
- Chen, X.; Huo, X.; Liu, J.; Wang, Y.; Werth, C.J.; Strathmann, T.J. Exploring beyond palladium: Catalytic reduction of aqueous oxyanion pollutants with alternative platinum group metals and new mechanistic implications. Chem. Eng. J. 2017, 313, 745–752. [Google Scholar] [CrossRef] [Green Version]
- Kuznetsova, L.I.; Kuznetsova, N.I.; Koscheev, S.V.; Zaikovskii, V.I.; Lisitsyn, A.S.; Kaprielova, K.M.; Kirillova, N.V.; Twardowski, Z. Carbon-supported iridium catalyst for reduction of chlorate ions with hydrogen in concentrated solutions of sodium chloride. Appl. Catal. A Gen. 2012, 427–428, 8–15. [Google Scholar] [CrossRef]
- Rutger, V.S.; Klesing, A.; Neuenfeldt, G.; Ottmann, A. Method for Removing Chlorate Ions from Solutions. U.S. Patent 6,270,682, 7 August 2001. [Google Scholar]
- Kim, H.S.; Kim, T.W.; Koh, H.L.; Lee, S.H.; Min, B.R. Complete benzene oxidation over Pt-Pd bimental catalyst supported on γ-alumina: Influence of Pt-Pd ratio on the catalytic activity. Appl. Catal. A Gen. 2005, 280, 125–131. [Google Scholar] [CrossRef]
- Kim, G.J.; Shin, J.H.; Hong, S.C. Study on the role of Pt and Pd in Pt–Pd/TiO2 bimetallic catalyst for H2 oxidation at room temperature. Int. J. Hydrog. Energy 2020, 45, 17276–17286. [Google Scholar] [CrossRef]
- Saxena, R.; De, M. Enhanced performance of supported Pd-Pt bimetallic catalysts prepared by modified electroless deposition for butane dehydrogenation. Appl. Catal. A Gen. 2021, 610, 117933. [Google Scholar] [CrossRef]
- Persson, K.; Ersson, A.; Colussi, S.; Trovarelli, A.; Järås, S.G. Catalytic combustion of methane over bimetallic Pd-Pt catalysts: The influence of support materials. Appl. Catal. B Environ. 2006, 66, 175–185. [Google Scholar] [CrossRef]
- Niquille-Röthlisberger, A.; Prins, R. Hydrodesulfurization of 4,6-dimethyldibenzothiophene and dibenzothiophene over alumina-supported Pt, Pd, and Pt-Pd catalysts. J. Catal. 2006, 242, 207–216. [Google Scholar] [CrossRef]
- Martínez Guerrero, R.; Hernández-Gordillo, A.; Santes, V.; Vargas García, J.R.; Escobar, J.; Díaz-García, L.; Díaz Barriga Arceo, L.; Garibay Febles, V. Monometallic Pd and Pt and bimetallic Pd-Pt/Al2O3-TiO2 for the HDS of DBT: Effect of the Pd and Pt incorporation method. J. Chem. 2014, 1, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Matsubayashi, N.; Yasuda, H.; Imamura, M.; Yoshimura, Y. EXAFS study on Pd-Pt catalyst supported on USY zeolite. Catal. Today 1998, 45, 375–380. [Google Scholar] [CrossRef]
- Byun, S.; Song, Y.; Kim, B.M.; Kim, B.M. Heterogenized bimetallic Pd-Pt-Fe3O4 nanoflakes as extremely robust, magnetically recyclable catalysts for chemoselective nitroarene reduction. ACS Appl. Mater. Interfaces 2016, 8, 14637–14647. [Google Scholar] [CrossRef]
- Micheaud-Especel, C.; Bazin, D.; Guérin, M.; Marécot, P.; Barbier, J. Study of supported bimetallic Pd-Pt catalysts. Characterization and catalytic activity for toluene hydrogenation. React. Kinet. Catal. Lett. 2000, 69, 209–216. [Google Scholar] [CrossRef]
- Lei, Y.L.; Lin, D.Q.; Yao, S.J.; Zhu, Z.Q. Preparation and characterization of titanium oxide-densified cellulose beads for expanded bed adsorption. J. Appl. Polym. Sci. 2003, 90, 2848–2854. [Google Scholar] [CrossRef]
- Sikora, E.; Katona, K.K.; Muránszky, G.; Bánhidi, O.; Kristály, F.; Szabó, J.T.; Windisch, M.; Fiser, B.; Vanyorek, L. Cellulose-based catalyst design for efficient chlorate reduction. Arab. J. Chem. 2021, 14, 103202. [Google Scholar] [CrossRef]


| Ni (wt%) | Fe3O4 (wt%) | Pd (wt%) | Pt (wt%) | |
|---|---|---|---|---|
| Pd–Pt/Ni–CCB | 8.97 | - | 2.03 | 0.29 |
| Pd–Pt/Ni–CCB (5 × used) | 8.85 | - | 2.06 | 0.29 |
| Pd–Pt/Fe3O4–CCB | - | 6.27 | 1.71 | 0.34 |
| Pd–Pt/Fe3O4–CCB (5 × used) | - | 6.48 | 1.72 | 0.35 |
| Pd–Pt/CCB | - | - | 0.96 | 0.19 |
| Pd–Pt/CCB (5 × used) | - | - | 0.95 | 0.18 |
| k (s−1) | Pd–Pt/Ni–CCB | Pd–Pt/Fe3O4–CCB | Pd–Pt/CCB |
|---|---|---|---|
| 1st cycle | 1.6 × 10−3 ± 1.2 × 10−4 | 2.5 × 10−4 ± 1.3 × 10−5 | 4.2 × 10−4 ± 2.3 × 10−5 |
| 2nd cycle | 9.5 × 10−4 ± 5.5 × 10−5 | 2.7 × 10−4 ± 1.8 × 10−5 | 3.4 × 10−4 ± 1.9 × 10−5 |
| 3rd cycle | 7.7 × 10−4 ± 4.3 × 10−5 | 2.2 × 10−4 ± 1.7 × 10−5 | 3.1 × 10−4 ± 2.8 × 10−5 |
| 4th cycle | 6.3 × 10−4 ± 5.5 × 10−5 | 1.8 × 10−4 ± 1.7 × 10−5 | 1.7 × 10−4 ± 5.3 × 10−6 |
| 5th cycle | 3.7 × 10−4 ± 2.2 × 10−5 | 1.6 × 10−4 ± 1.8 × 10−5 | 1.3 × 10−4 ± 1.0 × 10−5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sikora, E.; Koncz-Horváth, D.; Muránszky, G.; Kristály, F.; Fiser, B.; Viskolcz, B.; Vanyorek, L. Development of Nickel- and Magnetite-Promoted Carbonized Cellulose Bead-Supported Bimetallic Pd–Pt Catalysts for Hydrogenation of Chlorate Ions in Aqueous Solution. Int. J. Mol. Sci. 2021, 22, 11846. https://doi.org/10.3390/ijms222111846
Sikora E, Koncz-Horváth D, Muránszky G, Kristály F, Fiser B, Viskolcz B, Vanyorek L. Development of Nickel- and Magnetite-Promoted Carbonized Cellulose Bead-Supported Bimetallic Pd–Pt Catalysts for Hydrogenation of Chlorate Ions in Aqueous Solution. International Journal of Molecular Sciences. 2021; 22(21):11846. https://doi.org/10.3390/ijms222111846
Chicago/Turabian StyleSikora, Emőke, Dániel Koncz-Horváth, Gábor Muránszky, Ferenc Kristály, Béla Fiser, Béla Viskolcz, and László Vanyorek. 2021. "Development of Nickel- and Magnetite-Promoted Carbonized Cellulose Bead-Supported Bimetallic Pd–Pt Catalysts for Hydrogenation of Chlorate Ions in Aqueous Solution" International Journal of Molecular Sciences 22, no. 21: 11846. https://doi.org/10.3390/ijms222111846
APA StyleSikora, E., Koncz-Horváth, D., Muránszky, G., Kristály, F., Fiser, B., Viskolcz, B., & Vanyorek, L. (2021). Development of Nickel- and Magnetite-Promoted Carbonized Cellulose Bead-Supported Bimetallic Pd–Pt Catalysts for Hydrogenation of Chlorate Ions in Aqueous Solution. International Journal of Molecular Sciences, 22(21), 11846. https://doi.org/10.3390/ijms222111846







