Stem Cells and Their Derivatives—Implications for Alveolar Bone Regeneration: A Comprehensive Review
Abstract
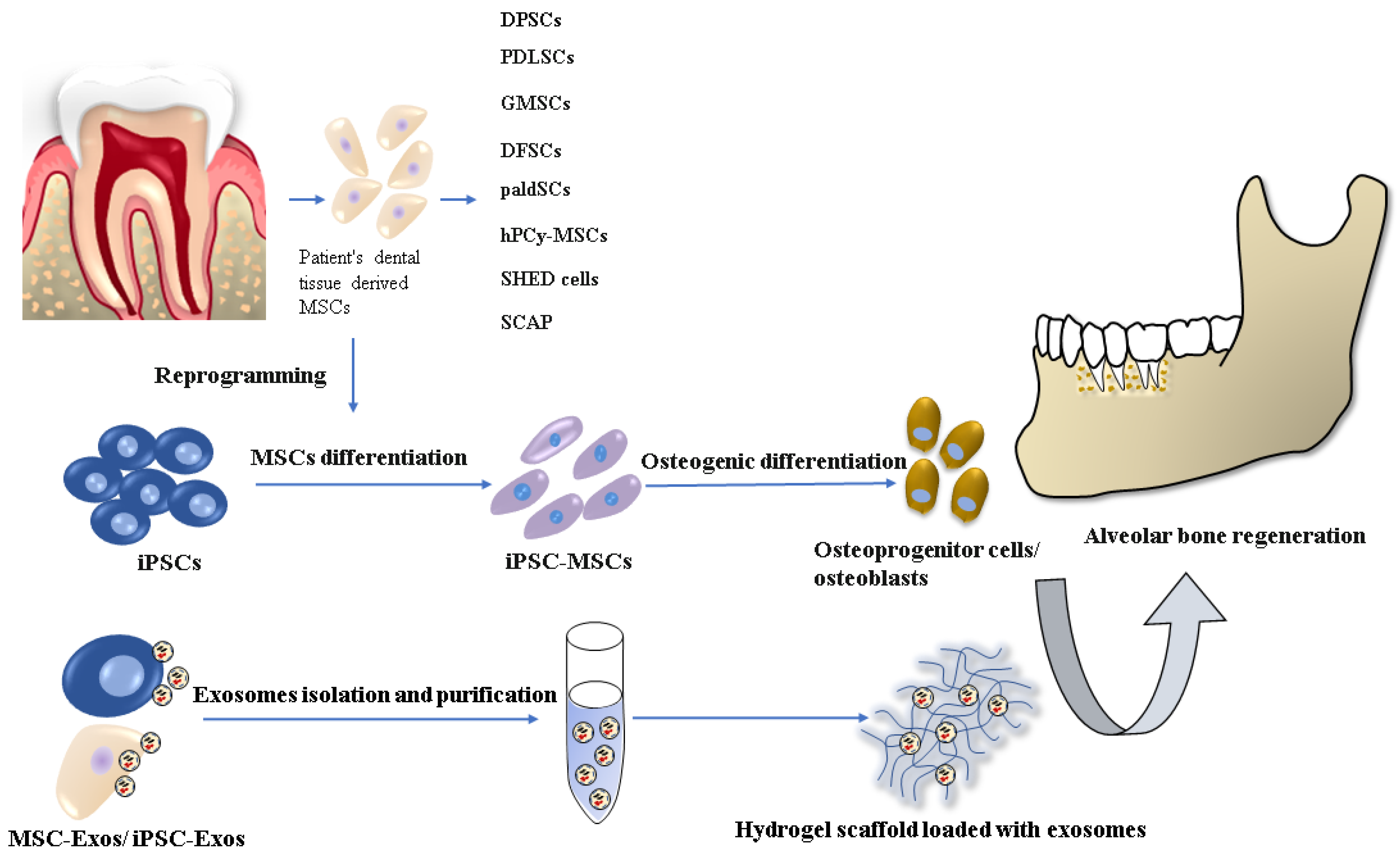
1. Introduction
2. Osteogenic Potential of Dental Tissue-Derived MSCs
| Type of Dental Tissue-Derived MSCs | In Vivo Models/Human Subjects | Site of Transplantation | Outcome | References |
|---|---|---|---|---|
| DPSCs | Immunocompromised mice | Dorsal surface | Generation of dentine/pulp-like structure | [27] |
| Immunocompromised rats | Subcutaneous site of dorsal surface | Generation of bone tissue with an integral blood supply | [29] | |
| Immunocompromised mice | Subcutaneous site of dorsal surface | Maintenance of MSC characteristics; higher stability compared with PDLSCs in vivo | [7] | |
| 6 patients aged 8 to 12 years old | Unilateral alveolar bone defect | Alveolar bone healing with no ectopic bone formation | [30] | |
| paldSCs | 30 patients | Alveolar bone defect | Improvement in vertical bone augmentation | [19] |
| DFSCs | Immunocompromised rats | Critical-sized calvarial defects | New bone formation | [35] |
| GMSCs | C57BL/6J mice | Second maxillary molar | Reduction in alveolar bone loss and new bone formation | [39] |
| Athymic rodent models | Maxillary alveolar bone defect | Enhanced bone regeneration | [40] | |
| PDLSCs | Immunocompromised rats | Calvarial critical-sized defect | Improvement of bone repair | [42] |
| 10 patients with chronic periodontitis | Root surface of defect site | Healing of deep periodontal defects | [43] | |
| SHED cells | Immunocompromised mice | Calvarial artificial bone defect | Formation of osteoid | [47] |
| SCAP | Minipig model of periodontitis | Local injection in the site of defects | Increased alveolar bone and periodontal tissue regeneration | [23] |
3. Osteogenic Potential of iPSCs in Dental Tissue Regeneration
4. Osteogenic Differentiation of iPSCs
5. Scaffolds Suitable for Alveolar Bone Regeneration
| Scaffold Material | Seeded Cells | Outcome | References |
|---|---|---|---|
| Apatite-coated silk scaffolds + EMD gel | iPSCs | Significant expression of Runx2; new bone tissue formation in vivo | [70] |
| RGD-CPC | iPSC-MSCs | Higher efficacy of osteogenic differentiation and bone matrix mineralization | [88] |
| RGD-CPC | iPSC-MSCs; + endothelial cells + pericytes | Increased scaffold pre-vascularization in vitro; new bone formation and vascularization in vivo | [73] |
| 3D-printed BG block/chitosan nanoparticles composites | BM-MSCs | New alveolar bone tissue formation in vivo | [93] |
| HA-Col | DPSCs | Supported attachment of DPSCs and formation of microenvironment for osteogenic differentiation in vitro | [94] |
| 3D collagen-based matrices + EDM + BMP-2 | MSCs | Significant expression of osteogenic markers; enhanced osteogenic differentiation in vitro | [95] |
| 3D BMP-6-hydrogel complex | iPSCs | new bone tissue formation in vivo | [72] |
| Graphene oxide-coated 3D-printed PCL scaffold | PDLSCs | Enhanced osteoinductivity and osteogenic differentiation in vitro | [97] |
6. Extracellular Vesicles—New Therapeutic Agents in Bone Regeneration
7. Conclusions and Future Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Tetè, G.; D’Orto, B.; Nagni, M.; Agostinacchio, M.; Polizzi, E.; Agliardi, E. Role of induced pluripotent stem cells (IPSCS) in bone tissue regeneration in dentistry: A narrative review. J. Biol. Regul. Homeost. Agents 2021, 34, 1–10. [Google Scholar]
- Paz, A.G.; Maghaireh, H.; Mangano, F.G. Stem Cells in Dentistry: Types of Intra- and Extraoral Tissue-Derived Stem Cells and Clinical Applications. Stem Cells Int. 2018, 2018, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Fernandez De Grado, G.; Keller, L.; Idoux-Gillet, Y.; Wagner, Q.; Musset, A.-M.; Benkirane-Jessel, N.; Bornert, F.; Offner, D. Bone substitutes: A review of their characteristics, clinical use, and perspectives for large bone defects management. J. Tissue Eng. 2018, 9, 2041731418776819. [Google Scholar] [CrossRef]
- Zhang, Y.; Wu, D.; Zhao, X.; Pakvasa, M.; Tucker, A.B.; Luo, H.; Qin, K.H.; Hu, D.A.; Wang, E.J.; Li, A.J.; et al. Stem Cell-Friendly Scaffold Biomaterials: Applications for Bone Tissue Engineering and Regenerative Medicine. Front. Bioeng. Biotechnol. 2020, 8, 598607. [Google Scholar] [CrossRef] [PubMed]
- Funda, G.; Taschieri, S.; Bruno, G.A.; Grecchi, E.; Paolo, S.; Girolamo, D.; Del Fabbro, M. Nanotechnology Scaffolds for Alveolar Bone Regeneration. Materials 2020, 13, 201. [Google Scholar] [CrossRef]
- Rodriguez, A.-M.; Elabd, C.; Amri, E.-Z.; Ailhaud, G.; Dani, C. The human adipose tissue is a source of multipotent stem cells. Biochimie 2005, 87, 125–128. [Google Scholar] [CrossRef] [PubMed]
- Lei, M.; Li, K.; Li, B.; Gao, L.-N.; Chen, F.-M.; Jin, Y. Mesenchymal stem cell characteristics of dental pulp and periodontal ligament stem cells after in vivo transplantation. Biomaterials 2014, 35, 6332–6343. [Google Scholar] [CrossRef]
- Gan, L.; Liu, Y.; Cui, D.; Pan, Y.; Zheng, L.; Wan, M. Dental Tissue—Derived Human Mesenchymal Stem Cells and Their Potential in Therapeutic Application. Stem Cells Int. 2020, 2020, 8864572. [Google Scholar] [CrossRef]
- Hynes, K.; Menichanin, D.; Bright, R.; Ivanovski, S.; Hutmacher, D.W.; Gronthos, S.; Bartold, P. Induced Pluripotent Stem Cells. J. Dent. Res. 2015, 94, 1508–1515. [Google Scholar] [CrossRef] [PubMed]
- Adamkov, M.; Halašová, E.; Rajcani, J.; Bencat, M.; Výbohová, D.; Rybárová, S.; Galbavy, S. Relation between expression pattern of p53 and survivin in cutaneous basal cell carcinomas. Med. Sci. Monit. 2011, 17, BR74–BR80. [Google Scholar] [CrossRef]
- Phillips, A.M. Overview of the fracture healing cascade. Injury 2005, 36, S5–S7. [Google Scholar] [CrossRef]
- Tsiridis, E.; Upadhyay, N.; Giannoudis, P. Molecular aspects of fracture healing:Which are the important molecules? Injury 2007, 38, S11–S25. [Google Scholar] [CrossRef] [PubMed]
- Huang, G.-J.; Gronthos, S.; Shi, S. Mesenchymal Stem Cells Derived from Dental Tissuesvs. Those from Other Sources: Their Biology and Role in Regenerative Medicine. J. Dent. Res. 2009, 88, 792–806. [Google Scholar] [CrossRef] [PubMed]
- Seo, B.-M.; Miura, M.; Gronthos, S.; Bartold, P.M.; Batouli, S.; Brahim, J.; Young, M.; Robey, P.G.; Wang, C.Y.; Shi, S. Investigation of multipotent postnatal stem cells from human periodontal ligament. Lancet 2004, 364, 149–155. [Google Scholar] [CrossRef]
- Tomokiyo, A.; Wada, N.; Maeda, H. Periodontal Ligament Stem Cells: Regenerative Potency in Periodontium. Stem Cells Dev. 2019, 28, 974–985. [Google Scholar] [CrossRef]
- Xu, Q.-C.; Wang, Z.-G.; Ji, Q.-X.; Yu, X.-B.; Xu, X.-Y.; Yuan, C.-Q.; Deng, J.; Yang, P.-S. Systemically transplanted human gingiva-derived mesenchymal stem cells contributing to bone tissue regeneration. Int. J. Clin. Exp. Pathol. 2014, 7, 4922–4929. [Google Scholar]
- Kim, D.; Lee, A.E.; Xu, Q.; Zhang, Q.; Le, A.D. Gingiva-Derived Mesenchymal Stem Cells: Potential Application in Tissue Engineering and Regenerative Medicine—A Comprehensive Review. Front. Immunol. 2021, 12, 667221. [Google Scholar] [CrossRef]
- Morsczeck, C. Effects of Cellular Senescence on Dental Follicle Cells. Pharmacology 2020, 106, 1–6. [Google Scholar] [CrossRef]
- Grimm, W.D.; Dannan, A.; Giesenhagen, B.; Schau, I.; Varga, G.; Vukovic, M.A.; Sirak, S.V. Translational Research: Palatal-derived Ecto-mesenchymal Stem Cells from Human Palate: A New Hope for Alveolar Bone and Cranio-Facial Bone Reconstruction. Int. J. Stem Cells 2014, 7, 23–29. [Google Scholar] [CrossRef]
- Naung, N.Y.; Duncan, W.; De Silva, R.; Coates, D. Localization and characterization of human palatal periosteum stem cells in serum-free, xeno-free medium for clinical use. Eur. J. Oral Sci. 2019, 127, 99–111. [Google Scholar] [CrossRef]
- Marrelli, M.; Paduano, F.; Tatullo, M. Human Periapical Cyst–Mesenchymal Stem Cells Differentiate Into Neuronal Cells. J. Dent. Res. 2015, 94, 843–852. [Google Scholar] [CrossRef]
- Ayoub, S.; Berbéri, A.; Fayyad-Kazan, M. An update on human periapical cyst-mesenchymal stem cells and their potential applications in regenerative medicine. Mol. Biol. Rep. 2020, 47, 2381–2389. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Han, N.; Zhang, X.; Yang, H.; Cao, Y.; Wang, S.; Fan, Z. Local Injection of Allogeneic Stem Cells from Apical Papilla Enhanced Periodontal Tissue Regeneration in Minipig Model of Periodontitis. BioMed Res. Int. 2018, 2018, 3960798. [Google Scholar] [CrossRef]
- Spagnuolo, G.; Codispoti, B.; Marrelli, M.; Rengo, C.; Rengo, S.; Tatullo, M. Commitment of Oral-Derived Stem Cells in Dental and Maxillofacial Applications. Dent. J. 2018, 6, 72. [Google Scholar] [CrossRef] [PubMed]
- Andrukhov, O.; Behm, C.; Blufstein, A.; Rausch-Fan, X. Immunomodulatory properties of dental tissue-derived mesenchymal stem cells: Implication in disease and tissue regeneration. World J. Stem Cells 2019, 11, 604–617. [Google Scholar] [CrossRef] [PubMed]
- Bajestan, M.N.; Rajan, A.; Edwards, S.P.; Aronovich, S.; Cevidanes, L.H.S.; Polymeri, A.; Travan, S.; Kaigler, D. Stem cell therapy for reconstruction of alveolar cleft and trauma defects in adults: A randomized controlled, clinical trial. Clin. Implant. Dent. Relat. Res. 2017, 19, 793–801. [Google Scholar] [CrossRef] [PubMed]
- Gronthos, S.; Mankani, M.; Brahim, J.; Robey, P.; Shi, S. Postnatal human dental pulp stem cells (DPSCs) in vitro and invivo. Proc. Natl. Acad. Sci. USA 2000, 97, 13625–13630. [Google Scholar] [CrossRef]
- Qu, G.; Li, Y.; Chen, L.; Chen, Q.; Zou, D.; Yang, C.; Zhou, Q. Comparison of Osteogenic Differentiation Potential of Human Dental-Derived Stem Cells Isolated from Dental Pulp, Periodontal Ligament, Dental Follicle, and Alveolar Bone. Stem Cells Int. 2021, 2021, 6631905. [Google Scholar] [CrossRef] [PubMed]
- D’Aquino, R.; Graziano, A.; Sampaolesi, M.; Laino, G.; Pirozzi, G.; De Rosa, A.; Papaccio, G. Human postnatal dental pulp cells co-differentiate into osteoblasts and endotheliocytes: A pivotal synergy leading to adult bone tissue formation. Cell Death Differ. 2007, 14, 1162–1171. [Google Scholar] [CrossRef]
- Tanikawa, D.Y.S.; Pinheiro, C.C.G.; Almeida, M.C.A.; Oliveira, C.R.G.C.M.; Coudry, R.D.A.; Rocha, D.L.; Bueno, D.F. Deciduous Dental Pulp Stem Cells for Maxillary Alveolar Reconstruction in Cleft Lip and Palate Patients. Stem Cells Int. 2020, 2020, 6234167. [Google Scholar] [CrossRef]
- Paduano, F.; Aiello, E.; Cooper, P.R.; Marrelli, B.; Makeeva, I.; Islam, M.; Spagnuolo, G.; Maged, D.; De Vito, D.; Tatullo, M. A Dedifferentiation Strategy to Enhance the Osteogenic Potential of Dental Derived Stem Cells. Front. Cell Dev. Biol. 2021, 9, 668558. [Google Scholar] [CrossRef]
- Zhang, J.; Ding, H.; Liu, X.; Sheng, Y.; Liu, X.; Jiang, C. Dental Follicle Stem Cells: Tissue Engineering and Immunomodulation. Stem Cells Dev. 2019, 28, 986–994. [Google Scholar] [CrossRef]
- Tsuchiya, S.; Ohshima, S.; Yamakoshi, Y.; Simmer, J.P.; Honda, M.J. Osteogenic Differentiation Capacity of Porcine Dental Follicle Progenitor Cells. Connect. Tissue Res. 2010, 51, 197–207. [Google Scholar] [CrossRef] [PubMed]
- Lucaciu, O.; Soritau, O.; Gheban, D.; Ciuca, D.R.; Virtic, O.; Vulpoi, A.; Dirzu, N.; Campian, R.; Băciuţ, G.; Popa, C.; et al. Dental follicle stem cells in bone regeneration on titanium implants. BMC Biotechnol. 2015, 15, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Bayat, H.; Shahabinejad, H.; Bayat, M.; Shirian, S.; Mohamadnia, A.; Alijani, M.; Godarzi, A.; Shojaei, P.; Shojaei, S.; Shevidi, A.; et al. Osteogenic differentiation of follicular stem cells on nano-Saghez scaffold containing BMP2. J. Orthop. Surg. Res. 2019, 14, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Honda, M.J.; Imaizumi, M.; Suzuki, H.; Ohshima, S.; Tsuchiya, S.; Satomura, K. Stem cells isolated from human dental follicles have osteogenic potential. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontol. 2011, 111, 700–708. [Google Scholar] [CrossRef]
- Gao, X. Gingiva-derived Mesenchymal Stem Cells and Their Potential Applications in Oral and Maxillofacial Diseases. Curr. Stem Cell Res. Ther. 2020, 15, 43–53. [Google Scholar] [CrossRef] [PubMed]
- Zhao, N.; Wu, Z.; Qin, L.; Guo, Z.; Li, D. Characteristics and Tissue Regeneration Properties of Gingiva-Derived Mesenchymal Stem Cells. Crit. Rev. Eukaryot. Gene Expr. 2015, 25, 135–144. [Google Scholar] [CrossRef]
- Al-Qadhi, G.; Al-Rai, S.; Hafed, L. The Therapeutic Potential of Inflamed Gingiva-Derived Mesenchymal Stem Cells in Preclinical Studies: A Scoping Review of a Unique Biomedical Waste. Stem Cells Int. 2021, 2021, 6619170. [Google Scholar] [CrossRef]
- Sun, Q.; Nakata, H.; Yamamoto, M.; Kasugai, S.; Kuroda, S. Comparison of gingiva-derived and bone marrow mesenchymal stem cells for osteogenesis. J. Cell. Mol. Med. 2019, 23, 7592–7601. [Google Scholar] [CrossRef]
- Kandalam, U.; Kawai, T.; Ravindran, G.; Brockman, R.; Romero, J.; Munro, M.; Ortiz, J.; Heidari, A.; Thomas, R.; Kuriakose, S.; et al. Predifferentiated Gingival Stem Cell-Induced Bone Regeneration in Rat Alveolar Bone Defect Model. Tissue Eng. Part A 2021, 27, 424–436. [Google Scholar] [CrossRef]
- Queiroz, A.; Albuquerque-Souza, E.; Gasparoni, L.M.; de França, B.N.; Pelissari, C.; Trierveiler, M.; Holzhausen, M. Therapeutic potential of periodontal ligament stem cells. World J. Stem Cells 2021, 13, 605–618. [Google Scholar] [CrossRef]
- Tour, G.; Wendel, M.; Tcacencu, I.; Moll, G. Bone Repair Using Periodontal Ligament Progenitor Cell-seeded Constructs. J. Dent. Res. 2012, 91, 789–794. [Google Scholar] [CrossRef]
- Iwata, T.; Yamato, M.; Washio, K.; Yoshida, T.; Tsumanuma, Y.; Yamada, A.; Onizuka, S.; Izumi, Y.; Ando, T.; Okano, T.; et al. Periodontal regeneration with autologous periodontal ligament-derived cell sheets—A safety and efficacy study in ten patients. Regen. Ther. 2018, 9, 38–44. [Google Scholar] [CrossRef]
- Marrelli, M.; Paduano, F.; Tatullo, M. Cells Isolated from Human Periapical Cysts Express Mesenchymal Stem Cell-like Properties. Int. J. Biol. Sci. 2013, 9, 1070–1078. [Google Scholar] [CrossRef] [PubMed]
- Tatullo, M.; Falisi, G.; Amantea, M.; Rastelli, C.; Paduano, F.; Marrelli, M. Dental pulp stem cells and human periap—Ical cyst mesenchymal stem cells in bone tissue regeneration: Comparison of basal and os—Teogenic differentiated gene expression of a newly discovered mesenchymal stem cell lineage. J. Biol. Regul. Homeost. Agents 2015, 29, 713–718. [Google Scholar]
- Tatullo, M.; Spagnuolo, G.; Codispoti, B.; Zamparini, F.; Zhang, A.; Degli Esposti, M.; Aparicio, C.; Rengo, C.; Nuzzolese, M.; Manzoli, L.; et al. PLA-Based Mineral—Doped Scaffolds Seeded with Human Periapical Cyst-Derived MSCs: A Promising Tool for Regenerative Healing in Dentistry. Materials 2019, 12, 597. [Google Scholar] [CrossRef] [PubMed]
- Nakajima, K.; Kunimatsu, R.; Ando, K.; Ando, T.; Hayashi, Y.; Kihara, T.; Hiraki, T.; Tsuka, Y.; Abe, T.; Kaku, M.; et al. Comparison of the bone regeneration ability between stem cells from human exfoliated deciduous teeth, human dental pulp stem cells and human bone marrow mesenchymal stem cells. Biochem. Biophys. Res. Commun. 2018, 497, 876–882. [Google Scholar] [CrossRef]
- Li, Q.; Yang, G.; Li, J.; Ding, M.; Zhou, N.; Dong, H.; Mou, Y. Stem cell therapies for periodontal tissue regeneration: A network meta-analysis of preclinical studies. Stem Cell Res. Ther. 2020, 11, 1–15. [Google Scholar] [CrossRef]
- Radwan, I.A.; Rady, D.; Abbass, M.; El Moshy, S.; Abubakr, N.; Dörfer, C.E.; El-Sayed, K.M.F. Induced Pluripotent Stem Cells in Dental and Nondental Tissue Regeneration: A Review of an Unexploited Potential. Stem Cells Int. 2020, 2020, 1941629. [Google Scholar] [CrossRef] [PubMed]
- Scesa, G.; Adami, R.; Bottai, D. iPSC Preparation and Epigenetic Memory: Does the Tissue Origin Matter? Cells 2021, 10, 1470. [Google Scholar] [CrossRef]
- Lee, J.-H.; Seo, S.-J. Biomedical Application of Dental Tissue-Derived Induced Pluripotent Stem Cells. Stem Cells Int. 2016, 2016, 9762465, Erratum in 2016, 2016, 2794510. [Google Scholar] [CrossRef]
- Yan, X.; Qin, H.; Qu, C.; Tuan, R.S.; Shi, S.; Huang, G.T.-J. iPS Cells Reprogrammed From Human Mesenchymal-Like Stem/Progenitor Cells of Dental Tissue Origin. Stem Cells Dev. 2010, 19, 469–480. [Google Scholar] [CrossRef]
- Saitoh, I.; Inada, E.; Iwase, Y.; Noguchi, H.; Murakami, T.; Soda, M.; Kubota, N.; Hasegawa, H.; Akasaka, E.; Matsumoto, Y.; et al. Choice of Feeders is Important When First Establishing iPSCs Derived from Primarily Cultured Human Deciduous Tooth Dental Pulp Cells. Cell Med. 2015, 8, 9–23. [Google Scholar] [CrossRef] [PubMed]
- Soda, M.; Saitoh, I.; Murakami, T.; Inada, E.; Iwase, Y.; Noguchi, H.; Shibasaki, S.; Kurosawa, M.; Sawami, T.; Terunuma, M.; et al. Repeated human deciduous tooth-derived dental pulp cell reprogramming factor transfection yields multipotent intermediate cells with enhanced iPS cell formation capability. Sci. Rep. 2019, 9, 1–13. [Google Scholar] [CrossRef]
- Oda, Y.; Yoshimura, Y.; Ohnishi, H.; Tadokoro, M.; Katsube, Y.; Sasao, M.; Kubo, Y.; Hattori, K.; Saito, S.; Horimoto, K.; et al. Induction of Pluripotent Stem Cells from Human Third Molar Mesenchymal Stromal Cells. J. Biol. Chem. 2010, 285, 29270–29278. [Google Scholar] [CrossRef]
- Miyoshi, K.; Tsuji, D.; Kudoh, K.; Satomura, K.; Muto, T.; Itoh, K.; Noma, T. Generation of human induced pluripotent stem cells from oral mucosa. J. Biosci. Bioeng. 2010, 110, 345–350. [Google Scholar] [CrossRef]
- Alvisi, G.; Trevisan, M.; Masi, G.; Canel, V.; Caenazzo, L.; Nespeca, P.; Barzon, L.; Di Iorio, E.; Barbaro, V.; Palù, G. Generation of a transgene-free human induced pluripotent stem cell line (UNIPDi001-A) from oral mucosa epithelial stem cells. Stem Cell Res. 2018, 28, 177–180. [Google Scholar] [CrossRef] [PubMed]
- Trevisan, M.; Alvisi, G.; Barbaro, V.; Barzon, L.; Raffa, P.; Migliorati, A.; Desole, G.; Ruzittu, S.; Masi, G.; Di Iorio, E.; et al. Oral Mucosa-Derived Induced Pluripotent Stem Cells from Patients with Ectrodactyly-Ectodermal Dysplasia-Clefting Syndrome. Cell. Reprogram. 2018, 20, 215–224. [Google Scholar] [CrossRef]
- Egusa, H.; Okita, K.; Kayashima, H.; Yu, G.; Fukuyasu, S.; Saeki, M.; Matsumoto, T.; Yamanaka, S.; Yatani, H. Gingival Fibroblasts as a Promising Source of Induced Pluripotent Stem Cells. PLoS ONE 2010, 5, e12743. [Google Scholar] [CrossRef]
- Wada, N.; Wang, B.; Lin, N.-H.; Laslett, A.; Gronthos, S.; Bartold, P.M. Induced pluripotent stem cell lines derived from human gingival fibroblasts and periodontal ligament fibroblasts. J. Periodontal Res. 2011, 46, 438–447. [Google Scholar] [CrossRef]
- Umezaki, Y.; Hashimoto, Y.; Nishishita, N.; Kawamata, S.; Baba, S. Human Gingival Integration-Free iPSCs; a Source for MSC-Like Cells. Int. J. Mol. Sci. 2015, 16, 13633–13648. [Google Scholar] [CrossRef]
- Yu, G.; Okawa, H.; Okita, K.; Kamano, Y.; Wang, F.; Saeki, M.; Yatani, H.; Egusa, H. Gingival Fibroblasts as Autologous Feeders for Induced Pluripotent Stem Cells. J. Dent. Res. 2015, 95, 110–118. [Google Scholar] [CrossRef]
- Limraksasin, P.; Okawa, H.; Zhang, M.; Kondo, T.; Osathanon, T.; Pavasant, P.; Egusa, H. Size—Optimized Microspace Culture Facilitates Differentiation of Mouse Induced Pluripotent Stem Cells into Osteoid-Rich Bone Constructs. Stem Cells Int. 2020, 2020, 1–14. [Google Scholar] [CrossRef]
- Sanchez-Duffhues, G.; Mikkers, H.; de Jong, D.; Szuhai, K.; de Vries, T.; Freund, C.; Bravenboer, N.; van Es, R.; Netelenbos, J.; Goumans, M.-J.; et al. Generation of Fibrodysplasia ossificans progressiva and control integration free iPSC lines from periodontal ligament fibroblasts. Stem Cell Res. 2019, 41, 101639. [Google Scholar] [CrossRef]
- Chang, Y.-C.; Li, W.-C.; Twu, N.-F.; Li, H.-Y.; Lo, W.-L.; Chang, Y.-L.; Lee, Y.-Y.; Lin, C.-F.; Shih, Y.-H.; Chen, M.-T. Induction of dental pulp-derived induced pluripotent stem cells in the absence of c-Myc for differentiation into neuron-like cells. J. Chin. Med. Assoc. 2014, 77, 618–625. [Google Scholar] [CrossRef][Green Version]
- Kawano, E.; Toriumi, T.; Iguchi, S.; Suzuki, D.; Sato, S.; Honda, M. Induction of neural crest cells from human dental pulp-derived induced pluripotent stem cells. Biomed. Res. 2017, 38, 135–147. [Google Scholar] [CrossRef]
- Pisal, R.V.; Suchanek, J.; Siller, R.; Soukup, T.; Hrebikova, H.; Bezrouk, A.; Kunke, D.; Micuda, S.; Filip, S.; Sullivan, G.; et al. Directed reprogramming of comprehensively characterized dental pulp stem cells extracted from natal tooth. Sci. Rep. 2018, 8, 6168. [Google Scholar] [CrossRef]
- Zhong, S.; He, X.; Li, Y.; Lou, X. Conditioned Medium Enhances Osteogenic Differentiation of Induced Pluripotent Stem Cell-Derived Mesenchymal Stem Cells. Tissue Eng. Regen. Med. 2019, 16, 141–150. [Google Scholar] [CrossRef]
- Duan, X.; Tu, Q.; Zhang, J.; Ye, J.; Sommer, C.; Mostoslavsky, G.; Kaplan, D.; Yang, P.; Chen, J. Application of induced pluripotent stem (iPS) cells in periodontal tissue regeneration. J. Cell. Physiol. 2011, 226, 150–157. [Google Scholar] [CrossRef] [PubMed]
- Hynes, K.; Menicanin, D.; Han, J.; Marino, V.; Mrozik, K.; Gronthos, S.; Bartold, P. Mesenchymal Stem Cells from iPS Cells Facilitate Periodontal Regeneration. J. Dent. Res. 2013, 92, 833–839. [Google Scholar] [CrossRef]
- Chien, K.-H.; Chang, Y.-L.; Wang, M.-L.; Chuang, J.-H.; Yang, Y.-C.; Tai, M.-C.; Wang, C.-Y.; Liu, Y.-Y.; Li, H.-Y.; Chen, J.-T.; et al. Promoting Induced Pluripotent Stem Cell-driven Biomineralization and Periodontal Regeneration in Rats with Maxillary-Molar Defects using Injectable BMP-6 Hydrogel. Sci. Rep. 2018, 8, 1–13. [Google Scholar] [CrossRef]
- Lin, Y.; Huang, S.; Zou, R.; Gao, X.; Ruan, J.; Weir, M.D.; Reynolds, M.A.; Qin, W.; Chang, X.; Fu, H.; et al. Calcium phosphate cement scaffold with stem cell co-culture and prevascularization for dental and craniofacial bone tissue engineering. Dent. Mater. 2019, 35, 1031–1041. [Google Scholar] [CrossRef]
- Csobonyeiova, M.; Štefan, P.; Zamborsky, R.; Danisovic, L. iPS cell technologies and their prospect for bone regeneration and disease modeling: A mini review. J. Adv. Res. 2017, 8, 321–327. [Google Scholar] [CrossRef]
- Kang, H.; Sung, J.; Jung, H.-M.; Woo, K.M.; Hong, S.-D.; Roh, S. Insulin-Like Growth Factor 2 Promotes Osteogenic Cell Differentiation in the Parthenogenetic Murine Embryonic Stem Cells. Tissue Eng. Part A 2012, 18, 331–341. [Google Scholar] [CrossRef]
- Ko, J.-Y.; Park, S.; Im, G.-I. Osteogenesis from Human Induced Pluripotent Stem Cells: An In Vitro and In Vivo Comparison with Mesenchymal Stem Cells. Stem Cells Dev. 2014, 23, 1788–1797. [Google Scholar] [CrossRef]
- Hayashi, T.; Misawa, H.; Nakahara, H.; Noguchi, H.; Yoshida, A.; Kobayashi, N.; Tanaka, M.; Ozaki, T. Transplantation of Osteogenically Differentiated Mouse iPS Cells for Bone Repair. Cell Transplant. 2012, 21, 591–600. [Google Scholar] [CrossRef]
- Ripamonti, U.; Crooks, J.; Petit, J.-C.; Rueger, D.C. Periodontal tissue regeneration by combined applications of recombinant human osteogenic protein-1 and bone morphogenetic protein-2. A pilot study in Chacma baboons (Papio ursinus). Eur. J. Oral Sci. 2001, 109, 241–248. [Google Scholar] [CrossRef]
- Mizrahi, O.; Sheyn, D.; Tawackoli, W.; Kallai, I.; Oh, A.; Su, S.; Da, X.; Zarrini, P.; Cook-Wiens, G.; Gazit, D.; et al. BMP-6 is more efficient in bone formation than BMP-2 when overexpressed in mesenchymal stem cells. Gene Ther. 2013, 20, 370–377. [Google Scholar] [CrossRef]
- Wu, Q.; Yang, B.; Cao, C.; Hu, K.; Wang, P.; Man, Y. Therapeutic antibody directed osteogenic differentiation of induced pluripotent stem cell derived MSCs. Acta Biomater. 2018, 74, 222–235. [Google Scholar] [CrossRef]
- Song, B.; Fu, H.; Liu, J.; Ren, K.; Weir, M.D.; Schneider, A.; Wang, P.; Song, Y.; Zhao, L.; Xu, H. Bioactive small molecules in calcium phosphate scaffold enhanced osteogenic differentiation of human induced pluripotent stem cells. Dent. Mater. J. 2021, 40, 615–624. [Google Scholar] [CrossRef] [PubMed]
- Zujur, D.; Kanke, K.; Onodera, S.; Tani, S.; Lai, J.; Azuma, T.; Xin, X.; Lichtler, A.C.; Rowe, D.W.; Saito, T.; et al. Stepwise strategy for generating osteoblasts from human pluripotent stem cells under fully defined xeno-free conditions with small-molecule inducers. Regen. Ther. 2020, 14, 19–31. [Google Scholar] [CrossRef]
- Li, J.; Lin, Q.; Lin, Y.; Lai, R.; Zhang, W. Effects of DLX3 on the osteogenic differentiation of induced pluripotent stem cell-derived mesenchymal stem cells. Mol. Med. Rep. 2021, 23, 1. [Google Scholar] [CrossRef]
- Akbulut, A.C.; Wasilewski, G.B.; Rapp, N.; Forin, F.; Singer, H.; Czogalla-Nitsche, K.J.; Schurgers, L.J. Menaquinone—7 Supplementation Improves Osteogenesis in Pluripotent Stem Cell Derived Mesenchymal Stem Cells. Front. Cell Dev. Biol. 2021, 8, 618760. [Google Scholar] [CrossRef]
- Sheikh, Z.; Hamdan, N.; Ikeda, Y.; Grynpas, M.; Ganss, B.; Glogauer, M. Natural graft tissues and synthetic biomaterials for periodontal and alveolar bone reconstructive applications: A review. Biomater. Res. 2017, 21, 9. [Google Scholar] [CrossRef]
- Iviglia, G.; Kargozar, S.; Baino, F. Biomaterials, Current Strategies, and Novel Nano-Technological Approaches for Periodontal Regeneration. J. Funct. Biomater. 2019, 10, 3. [Google Scholar] [CrossRef]
- Morrison, D.G.; Tomlinson, R.E. Leveraging advancements in tissue engineering for bioprinting dental tissues. Bioprinting 2021, 23, e00153. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Chen, W.; Zhao, Z.; Xu, H.H. Reprogramming of mesenchymal stem cells derived from iPSCs seeded on biofunctionalized calcium phosphate scaffold for bone engineering. Biomaterials 2013, 34, 7862–7872. [Google Scholar] [CrossRef]
- Liu, X.; Chen, W.; Zhang, C.; Thein-Han, W.; Hu, K.; Reynolds, M.A.; Bao, C.; Wang, P.; Zhao, L.; Xu, H.H. Co-Seeding Human Endothelial Cells with Human-Induced Pluripotent Stem Cell-Derived Mesenchymal Stem Cells on Calcium Phosphate Scaffold Enhances Osteogenesis and Vascularization in Rats. Tissue Eng. Part A 2017, 23, 546–555. [Google Scholar] [CrossRef]
- Miguez-Pacheco, V.; Hench, L.L.; Boccaccini, A.R. Bioactive glasses beyond bone and teeth: Emerging applications in contact with soft tissues. Acta Biomater. 2015, 13, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Kargozar, S.; Mozafari, M.; Ghodrat, S.; Fiume, E.; Baino, F. Copper-containing bioactive glasses and glass-ceramics: From tissue regeneration to cancer therapeutic strategies. Mater. Sci. Eng. C 2021, 121, 111741. [Google Scholar] [CrossRef]
- Wu, C.; Zhou, Y.; Xu, M.; Han, P.; Chen, L.; Chang, J.; Xiao, Y. Copper-containing mesoporous bioactive glass scaffolds with multifunctional properties of angiogenesis capacity, osteostimulation and antibacterial activity. Biomaterials 2013, 34, 422–433. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Chen, Y.; Xu, J.; Wang, J.; Li, C.; Wang, L. Tissue engineering using 3D printed nano-bioactive glass loaded with NELL1 gene for repairing alveolar bone defects. Regen. Biomater. 2018, 5, 213–220. [Google Scholar] [CrossRef] [PubMed]
- Trivedi, S.; Srivastava, K.; Saluja, T.; Shyam, H.; Kumar, S.; Singh, A.; Saxena, S.K.; Mehrotra, D.; Singh, S.K. Hydroxyapatite–collagen augments osteogenic differentiation of dental pulp stem cells. Odontology 2019, 108, 251–259. [Google Scholar] [CrossRef] [PubMed]
- Lin, Z.; Nica, C.; Sculean, A.; Asparuhova, M.B. Positive Effects of Three-Dimensional Collagen-Based Matrices on the Behavior of Osteoprogenitors. Front. Bioeng. Biotechnol. 2021, 9, 708830. [Google Scholar] [CrossRef] [PubMed]
- Guazzo, R.; Gardin, C.; Bellin, G.; Sbricoli, L.; Ferroni, L.; Ludovichetti, F.S.; Piattelli, A.; Antoniac, I.; Bressan, E.; Zavan, B. Graphene-Based Nanomaterials for Tissue Engineering in the Dental Field. Nanomaterials 2018, 8, 349. [Google Scholar] [CrossRef]
- Park, J.; Park, S.; Kim, J.; Jang, K.-J.; Seonwoo, H.; Chung, J. Enhanced Osteogenic Differentiation of Periodontal Ligament Stem Cells Using a Graphene Oxide-Coated Poly(ε-caprolactone) Scaffold. Polymers 2021, 13, 797. [Google Scholar] [CrossRef]
- Joo, H.S.; Suh, J.H.; Lee, H.J.; Bang, E.S. Current Knowledge and Future Perspectives on Mesenchymal Stem Cell-Derived Exosomes as a New Therapeutic Agent. Int. J. Mol. Sci. 2020, 21, 727. [Google Scholar] [CrossRef]
- Zhao, T.; Sun, F.; Liu, J.; Ding, T.; She, J.; Mao, F.; Xu, W.; Qian, H.; Yan, Y. Emerging Role of Mesenchymal Stem Cell-derived Exosomes in Regenerative Medicine. Curr. Stem Cell Res. Ther. 2019, 14, 482–494. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Li, L.; Rong, Y.; Qian, D.; Chen, J.; Zhou, Z.; Luo, Y.; Jiang, D.; Cheng, L.; Zhao, S.; et al. Hypoxic mesenchymal stem cell-derived exosomes promote bone fracture healing by the transfer of miR-126. Acta Biomater. 2020, 103, 196–212. [Google Scholar] [CrossRef]
- Zhang, Y.; Xie, Y.; Hao, Z.; Zhou, P.; Wang, P.; Fang, S.; Li, L.; Xu, S.; Xia, Y. Umbilical Mesenchymal Stem Cell-Derived Exosome-Encapsulated Hydrogels Accelerate Bone Repair by Enhancing Angiogenesis. ACS Appl. Mater. Interfaces 2021, 13, 18472–18487. [Google Scholar] [CrossRef] [PubMed]
- Livshits, M.A.; Khomyakova, E.; Evtushenko, E.; Lazarev, V.N.; Kulemin, N.; Semina, S.E.; Generozov, E.; Govorun, V.M. Isolation of exosomes by differential centrifugation: Theoretical analysis of a commonly used protocol. Sci. Rep. 2015, 5, 17319, Erratum in 2016, 6, 21447. [Google Scholar] [CrossRef] [PubMed]
- Cooper, L.F.; Ravindran, S.; Huang, C.-C.; Kang, M. A Role for Exosomes in Craniofacial Tissue Engineering and Regeneration. Front. Physiol. 2020, 10, 1569. [Google Scholar] [CrossRef]
- Macías, I.; Alcorta-Sevillano, N.; Infante, A.; Rodríguez, C. Cutting Edge Endogenous Promoting and Exogenous Driven Strategies for Bone Regeneration. Int. J. Mol. Sci. 2021, 22, 7724. [Google Scholar] [CrossRef] [PubMed]
- De Cássia Noronha, N.; Mizukami, A.; Caliári-Oliveira, C.; Cominal, J.G.; Rocha, J.L.M.; Covas, D.T.; Swiech, K.; Malmegrim, K.C.R. Priming approaches to improve the efficacy of mesenchymal stromal cell-based therapies. Stem Cell Res. Ther. 2019, 10, 131, Erratum in 2019, 10, 132. [Google Scholar] [CrossRef]
- Furuta, T.; Miyaki, S.; Ishitobi, H.; Ogura, T.; Kato, Y.; Kamei, N.; Miyado, K.; Higashi, Y.; Ochi, M. Mesenchymal Stem Cell-Derived Exosomes Promote Fracture Healing in a Mouse Model. Stem Cells Transl. Med. 2016, 5, 1620–1630. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Liu, Y.; Zhang, P.; Tang, Y.; Zhou, M.; Jiang, W.; Zhang, X.; Wu, G.; Zhou, Y. Tissue-Engineered Bone Immobilized with Human Adipose Stem Cells-Derived Exosomes Promotes Bone Regeneration. ACS Appl. Mater. Interfaces 2018, 10, 5240–5254. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Jiao, G.; Ren, S.; Zhang, X.; Li, C.; Wu, W.; Wang, H.; Liu, H.; Zhou, H.; Chen, Y. Exosomes from bone marrow mesenchymal stem cells enhance fracture healing through the promotion of osteogenesis and angiogenesis in a rat model of nonunion. Stem Cell Res. Ther. 2020, 11, 1–15. [Google Scholar] [CrossRef]
- Swanson, W.B.; Zhang, Z.; Xiu, K.; Gong, T.; Eberle, M.; Wang, Z.; Ma, P.X. Scaffolds with controlled release of pro-mineralization exosomes to promote craniofacial bone healing without cell transplantation. Acta Biomater. 2020, 118, 215–232. [Google Scholar] [CrossRef]
- Behera, J.; Kumar, A.; Voor, M.J.; Tyagi, N. Exosomal lncRNA-H19 promotes osteogenesis and angiogenesis through mediating Angpt1/Tie2-NO signaling in CBS-heterozygous mice. Theranostics 2021, 11, 7715–7734. [Google Scholar] [CrossRef]
- Pishavar, E.; Luo, H.; Naserifar, M.; Hashemi, M.; Toosi, S.; Atala, A.; Ramakrishna, S.; Behravan, J. Advanced Hydrogels as Exosome Delivery Systems for Osteogenic Differentiation of MSCs: Application in Bone Regeneration. Int. J. Mol. Sci. 2021, 22, 6203. [Google Scholar] [CrossRef] [PubMed]
- Qi, X.; Zhang, J.; Yuan, H.; Xu, Z.; Li, Q.; Niu, X.; Hu, B.; Wang, Y.; Li, X. Exosomes Secreted by Human-Induced Pluripotent Stem Cell-Derived Mesenchymal Stem Cells Repair Critical-Sized Bone Defects through Enhanced Angiogenesis and Osteogenesis in Osteoporotic Rats. Int. J. Biol. Sci. 2016, 12, 836–849. [Google Scholar] [CrossRef]
- Zhang, J.; Liu, X.; Li, H.; Chen, C.; Hu, B.; Niu, X.; Li, Q.; Zhao, B.; Xie, Z.; Wang, Y. Exosomes/tricalcium phosphate combination scaffolds can enhance bone regeneration by activating the PI3K/Akt signaling pathway. Stem Cell Res. Ther. 2016, 7, 1–14. [Google Scholar] [CrossRef]
- Liu, X.; Li, Q.; Niu, X.; Hu, B.; Chen, S.; Song, W.; Ding, J.; Zhang, C.; Wang, Y. Exosomes Secreted from Human-Induced Pluripotent Stem Cell-Derived Mesenchymal Stem Cells Prevent Osteonecrosis of the Femoral Head by Promoting Angiogenesis. Int. J. Biol. Sci. 2017, 13, 232–244. [Google Scholar] [CrossRef] [PubMed]
- Khayambashi, P.; Iyer, J.; Pillai, S.; Upadhyay, A.; Zhang, Y.; Tran, S.D. Hydrogel Encapsulation of Mesenchymal Stem Cells and Their Derived Exosomes for Tissue Engineering. Int. J. Mol. Sci. 2021, 22, 684. [Google Scholar] [CrossRef] [PubMed]
- Kargozar, S.; Mozafari, M.; Hamzehlou, S.; Milan, P.B.; Kim, H.-W.; Baino, F. Bone Tissue Engineering Using Human Cells: A Comprehensive Review on Recent Trends, Current Prospects, and Recommendations. Appl. Sci. 2019, 9, 174. [Google Scholar] [CrossRef]
| Exosome Source | Isolation Method | Outcome | References |
|---|---|---|---|
| BM-MSCs | Ultracentrifugation of BM-MSC-conditioned media | MSC-Exos facilitated femur fracture healing in mice | [105] |
| iPSC-MSCs | Ultracentrifugation of iPSC-MSC-conditioned media | iPSC-MSC-Exos efficaciously stimulated bone regeneration and angiogenesis in critical-sized calvarial defects in rats | [111] |
| iPSC-MSCs | Ultracentrifugation of iPSC-MSC-conditioned media | iPSC-MSC-Exos significantly prevented osteonecrosis and increased microvessel density in femoral head | [113] |
| ADSCs | Ultracentrifugation of ADSC-conditioned media | ADSC-Exos increased bone formation in critical-sized mice calvarial defects | [107] |
| Umbilical MSCs treated under hypoxic condition | Ultracentrifugation of media with sucrose/D2O cushion conjunction | Hypo-exosomes promoted femoral fracture healing by transferring miR-126 in mice | [99] |
| BM-MSCs | Ultracentrifugation of BM-MSC-conditioned media | Osteogenesis, angiogenesis, and bone healing in a fracture model of rat femoral nonunion | [107] |
| hDPSCs | Ultracentrifugation of hDPSC-conditioned media | hDPSC-Exos facilitated osteogenic differentiation of BM-MSCs; mice calvarial defect repair by hDPSC-Exo loaded constructs | [108] |
| BM-MSCs | Ultracentrifugation of BM-MSC-conditioned media | MSC-Exos promoted angiogenesis and osteogenesis in vitro; restoration of bone formation and mechanical quality in vivo | [109] |
| Umbilical MSCs | Ultracentrifugation of umbilical MSC-conditioned media | MSC-Exos seeded on 3D hydrogel scaffold promoted the repair of cranial defects in rats | [100] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hollý, D.; Klein, M.; Mazreku, M.; Zamborský, R.; Polák, Š.; Danišovič, Ľ.; Csöbönyeiová, M. Stem Cells and Their Derivatives—Implications for Alveolar Bone Regeneration: A Comprehensive Review. Int. J. Mol. Sci. 2021, 22, 11746. https://doi.org/10.3390/ijms222111746
Hollý D, Klein M, Mazreku M, Zamborský R, Polák Š, Danišovič Ľ, Csöbönyeiová M. Stem Cells and Their Derivatives—Implications for Alveolar Bone Regeneration: A Comprehensive Review. International Journal of Molecular Sciences. 2021; 22(21):11746. https://doi.org/10.3390/ijms222111746
Chicago/Turabian StyleHollý, Dušan, Martin Klein, Merita Mazreku, Radoslav Zamborský, Štefan Polák, Ľuboš Danišovič, and Mária Csöbönyeiová. 2021. "Stem Cells and Their Derivatives—Implications for Alveolar Bone Regeneration: A Comprehensive Review" International Journal of Molecular Sciences 22, no. 21: 11746. https://doi.org/10.3390/ijms222111746
APA StyleHollý, D., Klein, M., Mazreku, M., Zamborský, R., Polák, Š., Danišovič, Ľ., & Csöbönyeiová, M. (2021). Stem Cells and Their Derivatives—Implications for Alveolar Bone Regeneration: A Comprehensive Review. International Journal of Molecular Sciences, 22(21), 11746. https://doi.org/10.3390/ijms222111746








