Hyaluronic Acid as a Carrier Supports the Effects of Glucocorticoids and Diminishes the Cytotoxic Effects of Local Anesthetics in Human Articular Chondrocytes In Vitro
Abstract
1. Introduction
2. Results
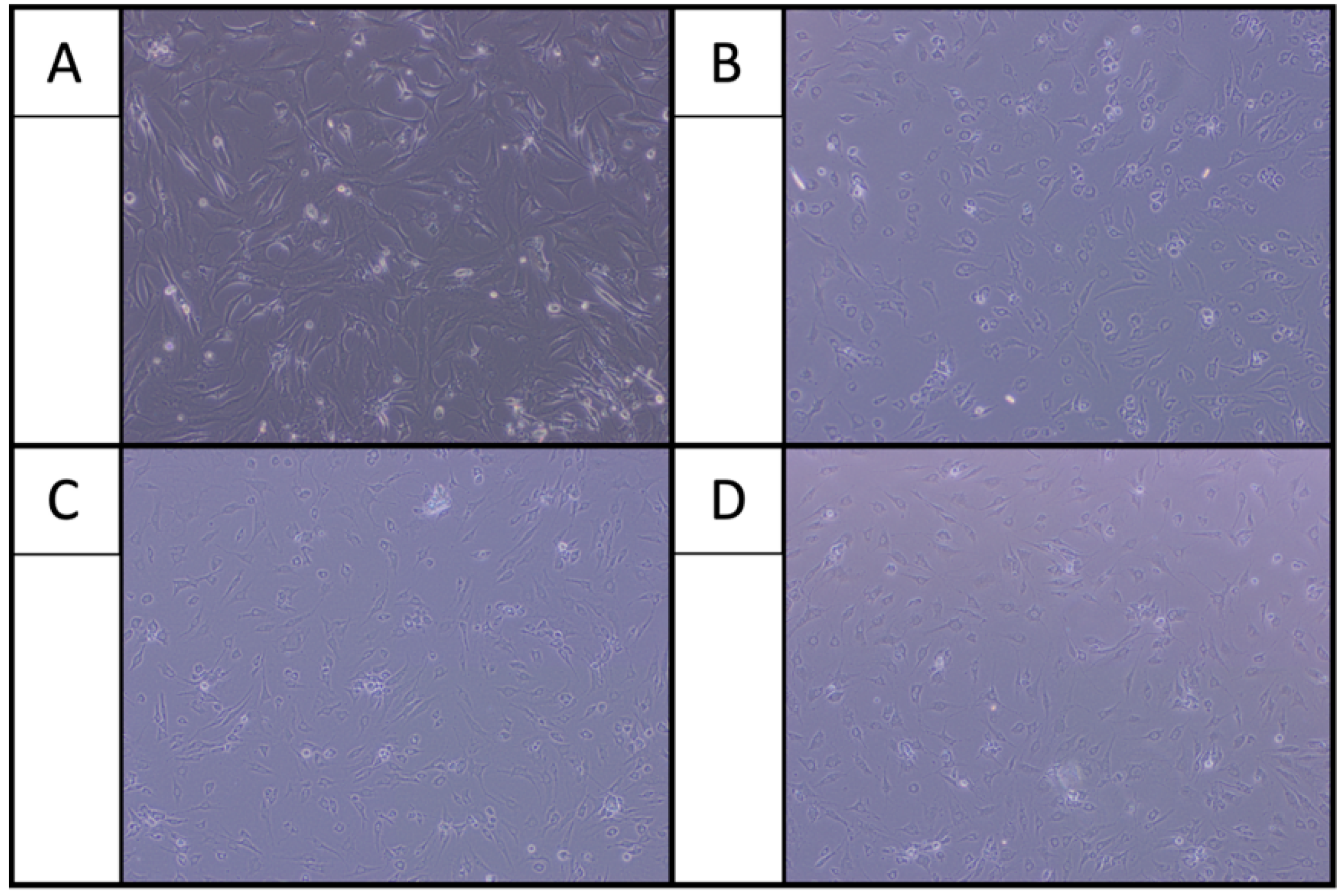
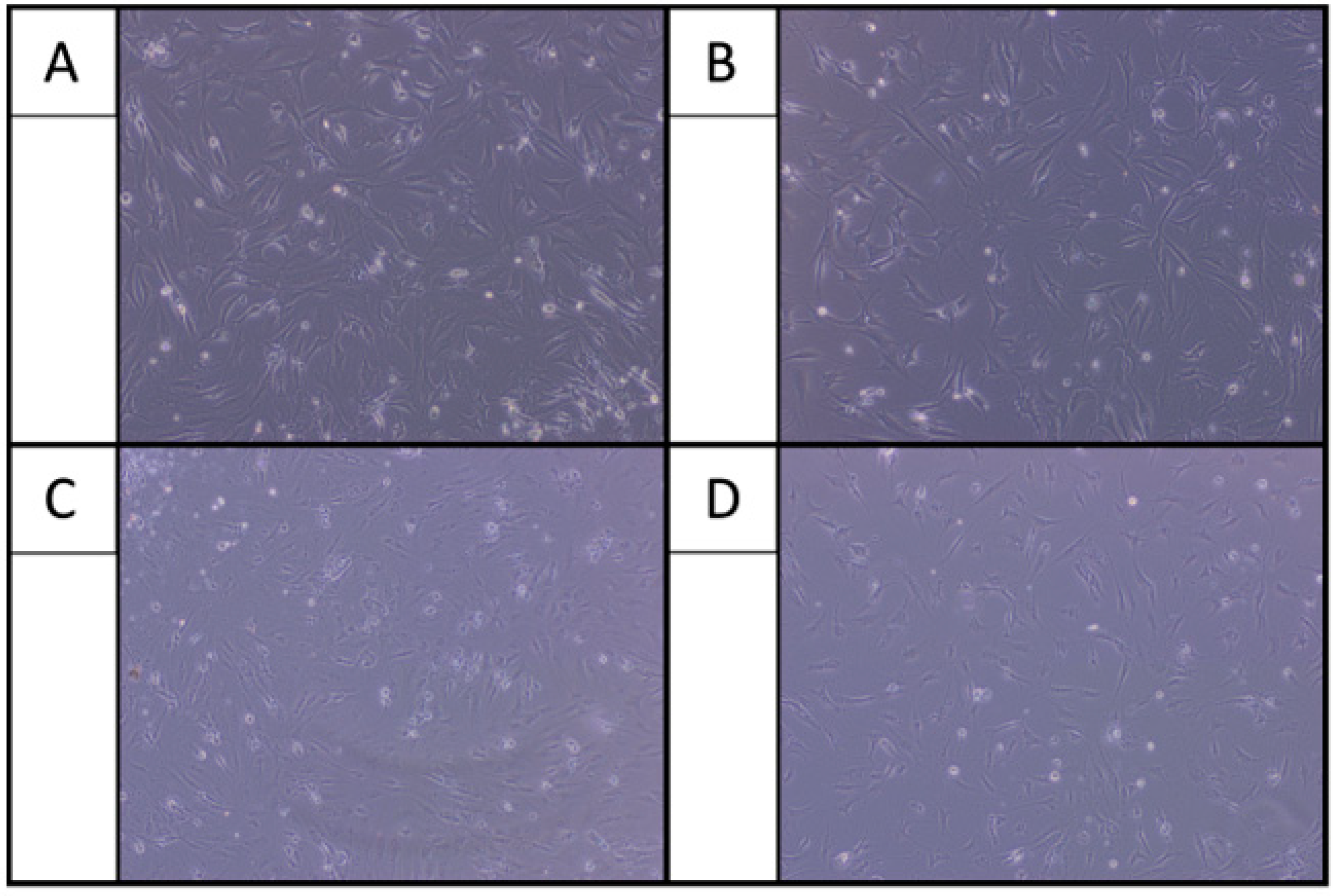
2.1. Morphological Examination
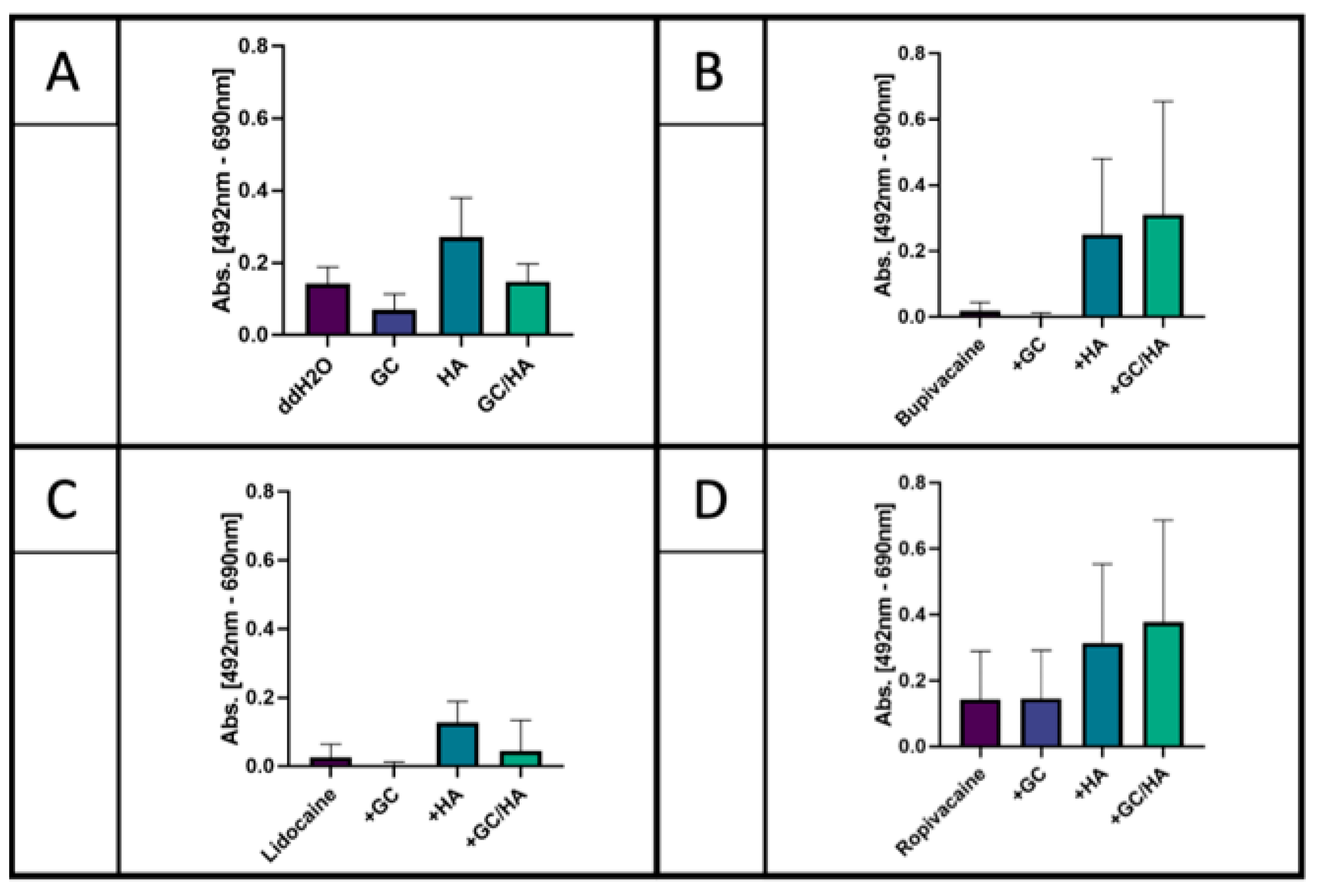
2.2. XTT Assay
2.3. Flow Cytometry
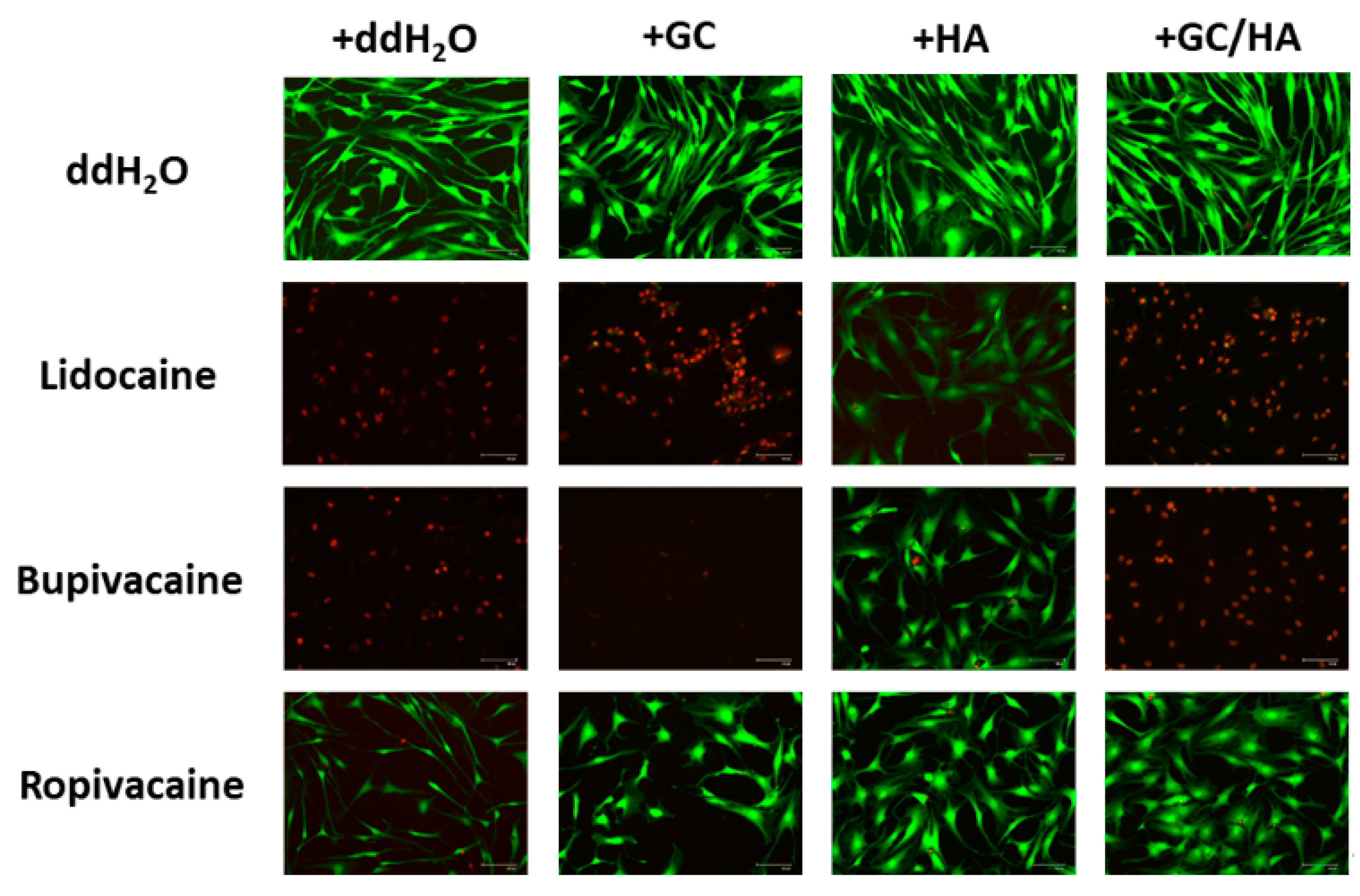
2.4. Live/Dead Assay
3. Discussion
4. Materials and Methods
4.1. Isolation and Cultivation of Human Chondrocytes
4.2. Treatment of the Cells
4.3. Morphological Examination
4.4. Metabolic Activity
4.5. Flow Cytometry
4.6. Live/Dead Assay
4.7. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Szwedowski, D.; Szczepanek, J.; Paczesny, Ł.; Pękała, P.; Zabrzyński, J.; Kruczyński, J. Genetics in Cartilage Lesions: Basic Science and Therapy Approaches. Int. J. Mol. Sci. 2020, 21, 5430. [Google Scholar] [CrossRef]
- Sophia Fox, A.J.; Bedi, A.; Rodeo, S.A. The Basic Science of Articular Cartilage. Sports Health 2009, 1, 461–468. [Google Scholar] [CrossRef] [PubMed]
- Zheng, L.; Zhang, Z.; Sheng, P.; Mobasheri, A. The Role of Metabolism in Chondrocyte Dysfunction and the Progression of Osteoarthritis. Ageing Res. Rev. 2021, 66, 101249. [Google Scholar] [CrossRef]
- Kreuz, P.C.; Steinwachs, M.; Angele, P. Single-Dose Local Anesthetics Exhibit a Type-, Dose-, and Time-Dependent Chondrotoxic Effect on Chondrocytes and Cartilage: A Systematic Review of the Current Literature. Knee Surg. Sports Traumatol. Arthrosc. 2018, 26, 819–830. [Google Scholar] [CrossRef] [PubMed]
- Breu, A.; Rosenmeier, K.; Kujat, R.; Angele, P.; Zink, W. The Cytotoxicity of Bupivacaine, Ropivacaine, and Mepivacaine on Human Chondrocytes and Cartilage. Anesth. Analg. 2013, 117, 514–522. [Google Scholar] [CrossRef]
- Gomoll, A.H.; Kang, R.W.; Williams, J.M.; Bach, B.R.; Cole, B.J. Chondrolysis after Continuous Intra-Articular Bupivacaine Infusion: An Experimental Model Investigating Chondrotoxicity in the Rabbit Shoulder. Arthroscopy 2006, 22, 813–819. [Google Scholar] [CrossRef]
- Hansen, B.P.; Beck, C.L.; Beck, E.P.; Townsley, R.W. Postarthroscopic Glenohumeral Chondrolysis. Am. J. Sports Med. 2007, 35, 1628–1634. [Google Scholar] [CrossRef] [PubMed]
- Grishko, V.; Xu, M.; Wilson, G.; Pearsall, A.W. Apoptosis and Mitochondrial Dysfunction in Human Chondrocytes Following Exposure to Lidocaine, Bupivacaine, and Ropivacaine. J. Bone Joint Surg. Am. 2010, 92, 609–618. [Google Scholar] [CrossRef]
- Wernecke, C.; Braun, H.J.; Dragoo, J.L. The Effect of Intra-Articular Corticosteroids on Articular Cartilage: A Systematic Review. Orthop. J. Sports Med. 2015, 3, 2325967115581163. [Google Scholar] [CrossRef] [PubMed]
- Balazs, E.A.; Denlinger, J.L. Viscosupplementation: A New Concept in the Treatment of Osteoarthritis. J. Rheumatol. Suppl. 1993, 39, 3–9. [Google Scholar] [PubMed]
- Akmal, M.; Singh, A.; Anand, A.; Kesani, A.; Aslam, N.; Goodship, A.; Bentley, G. The Effects of Hyaluronic Acid on Articular Chondrocytes. J. Bone Joint Surg. Br. 2005, 87, 1143–1149. [Google Scholar] [CrossRef]
- Hangody, L.; Szody, R.; Lukasik, P.; Zgadzaj, W.; Lénárt, E.; Dokoupilova, E.; Bichovsk, D.; Berta, A.; Vasarhelyi, G.; Ficzere, A.; et al. Intraarticular Injection of a Cross-Linked Sodium Hyaluronate Combined with Triamcinolone Hexacetonide (Cingal) to Provide Symptomatic Relief of Osteoarthritis of the Knee: A Randomized, Double-Blind, Placebo-Controlled Multicenter Clinical Trial. Cartilage 2018, 9, 276–283. [Google Scholar] [CrossRef] [PubMed]
- Onur, T.S.; Sitron, C.S.; Dang, A. Co-Administration of Hyaluronic Acid with Local Anaesthetics Shows Lower Cytotoxicity than Local Anaesthetic Treatment Alone in Bovine Articular Chondrocytes. Bone Jt. Res. 2013, 2, 270–275. [Google Scholar] [CrossRef][Green Version]
- Piper, S.L.; Kramer, J.D.; Kim, H.T.; Feeley, B.T. Effects of Local Anesthetics on Articular Cartilage. Am. J. Sports Med. 2011, 39, 2245–2253. [Google Scholar] [CrossRef]
- Jayaram, P.; Kennedy, D.J.; Yeh, P.; Dragoo, J. Chondrotoxic Effects of Local Anesthetics on Human Knee Articular Cartilage: A Systematic Review. PM&R 2019, 11, 379–400. [Google Scholar] [CrossRef]
- Jacob, B.; Zippelius, T.; Kloss, N.; Benad, K.; Schwerdt, C.; Hoff, P.; Matziolis, G.; Röhner, E. Local Anesthetics’ Toxicity toward Human Cultured Chondrocytes: A Comparative Study between Lidocaine, Bupivacaine, and Ropivacaine. Cartilage 2019, 10, 364–369. [Google Scholar] [CrossRef]
- Braun, H.J.; Wilcox-Fogel, N.; Kim, H.J.; Pouliot, M.A.; Harris, A.H.S.; Dragoo, J.L. The Effect of Local Anesthetic and Corticosteroid Combinations on Chondrocyte Viability. Knee Surg. Sports Traumatol. Arthrosc. 2012, 20, 1689–1695. [Google Scholar] [CrossRef]
- Farkas, B.; Kvell, K.; Czömpöly, T.; Illés, T.; Bárdos, T. Increased Chondrocyte Death after Steroid and Local Anesthetic Combination. Clin. Orthop. Relat. Res. 2010, 468, 3112–3120. [Google Scholar] [CrossRef]
- Klaus, G.; Jux, C.; Fernandez, P.; Rodriguez, J.; Himmele, R.; Mehls, O. Suppression of Growth Plate Chondrocyte Proliferation by Corticosteroids. Pediatr. Nephrol. 2000, 14, 612–615. [Google Scholar] [CrossRef] [PubMed]
- Verschure, P.J.; Van Noorden, C.J.; Van Marle, J.; Van den Berg, W.B. Articular Cartilage Destruction in Experimental Inflammatory Arthritis: Insulin-like Growth Factor-1 Regulation of Proteoglycan Metabolism in Chondrocytes. Histochem. J. 1996, 28, 835–857. [Google Scholar] [CrossRef]
- Patti, A.M.; Gabriele, A.; Vulcano, A.; Ramieri, M.T.; Della Rocca, C. Effect of Hyaluronic Acid on Human Chondrocyte Cell Lines from Articular Cartilage. Tissue Cell 2001, 33, 294–300. [Google Scholar] [CrossRef]
- Ishida, O.; Tanaka, Y.; Morimoto, I.; Takigawa, M.; Eto, S. Chondrocytes Are Regulated by Cellular Adhesion through CD44 and Hyaluronic Acid Pathway. J. Bone Miner. Res. 1997, 12, 1657–1663. [Google Scholar] [CrossRef]
- Altman, R.; Manjoo, A.; Fierlinger, A.; Niazi, F.; Nicholls, M. The Mechanism of Action for Hyaluronic Acid Treatment in the Osteoarthritic Knee: A Systematic Review. BMC Musculoskelet. Disord. 2015, 16, 321. [Google Scholar] [CrossRef]
- Moreland, L.W. Intra-Articular Hyaluronan (Hyaluronic Acid) and Hylans for the Treatment of Osteoarthritis: Mechanisms of Action. Arthritis Res. Ther. 2003, 5, 54–67. [Google Scholar] [CrossRef]
- Gupta, R.C.; Lall, R.; Srivastava, A.; Sinha, A. Hyaluronic Acid: Molecular Mechanisms and Therapeutic Trajectory. Front. Vet. Sci. 2019, 6, 192. [Google Scholar] [CrossRef] [PubMed]
- Jones, I.A.; Togashi, R.; Wilson, M.L.; Heckmann, N.; Vangsness, C.T. Intra-Articular Treatment Options for Knee Osteoarthritis. Nat. Rev. Rheumatol. 2019, 15, 77–90. [Google Scholar] [CrossRef] [PubMed]
- Jevsevar, D.S.; Brown, G.A.; Jones, D.L.; Matzkin, E.G.; Manner, P.A.; Mooar, P.; Schousboe, J.T.; Stovitz, S.; Sanders, J.O.; Bozic, K.J.; et al. The American Academy of Orthopaedic Surgeons Evidence-Based Guideline on: Treatment of Osteoarthritis of the Knee, 2nd Edition. J. Bone Jt. Surg. Am. 2013, 95, 1885–1886. [Google Scholar] [CrossRef]
- Hochberg, M.C.; Altman, R.D.; April, K.T.; Benkhalti, M.; Guyatt, G.; McGowan, J.; Towheed, T.; Welch, V.; Wells, G.; Tugwell, P.; et al. American College of Rheumatology 2012 Recommendations for the Use of Nonpharmacologic and Pharmacologic Therapies in Osteoarthritis of the Hand, Hip, and Knee. Arthritis Care Res. 2012, 64, 465–474. [Google Scholar] [CrossRef]
- McAlindon, T.E.; LaValley, M.P.; Harvey, W.F.; Price, L.L.; Driban, J.B.; Zhang, M.; Ward, R.J. Effect of Intra-Articular Triamcinolone vs Saline on Knee Cartilage Volume and Pain in Patients With Knee Osteoarthritis: A Randomized Clinical Trial. JAMA 2017, 317, 1967–1975. [Google Scholar] [CrossRef]
- National Clinical Guideline Centre (UK). Osteoarthritis: Care and Management in Adults. In National Institute for Health and Clinical Excellence: Guidance; National Institute for Health and Care Excellence (UK): London, UK, 2014. [Google Scholar]
- Bedard, N.A.; Pugely, A.J.; Elkins, J.M.; Duchman, K.R.; Westermann, R.W.; Liu, S.S.; Gao, Y.; Callaghan, J.J.; John, N. Insall Award: Do Intraarticular Injections Increase the Risk of Infection After TKA? Clin. Orthop. Relat. Res. 2017, 475, 45–52. [Google Scholar] [CrossRef] [PubMed]
- McAlindon, T.E.; Bannuru, R.R.; Sullivan, M.C.; Arden, N.K.; Berenbaum, F.; Bierma-Zeinstra, S.M.; Hawker, G.A.; Henrotin, Y.; Hunter, D.J.; Kawaguchi, H.; et al. OARSI Guidelines for the Non-Surgical Management of Knee Osteoarthritis. Osteoarthr. Cartil. 2014, 22, 363–388. [Google Scholar] [CrossRef] [PubMed]
- Bannuru, R.R.; Natov, N.S.; Obadan, I.E.; Price, L.L.; Schmid, C.H.; McAlindon, T.E. Therapeutic Trajectory of Hyaluronic Acid versus Corticosteroids in the Treatment of Knee Osteoarthritis: A Systematic Review and Meta-Analysis. Arthritis Rheum. 2009, 61, 1704–1711. [Google Scholar] [CrossRef]
- Bellamy, N.; Campbell, J.; Robinson, V.; Gee, T.; Bourne, R.; Wells, G. Intraarticular Corticosteroid for Treatment of Osteoarthritis of the Knee. Cochrane Database Syst. Rev. 2006, CD005328. [Google Scholar] [CrossRef]
- Rutjes, A.W.S.; Jüni, P.; da Costa, B.R.; Trelle, S.; Nüesch, E.; Reichenbach, S. Viscosupplementation for Osteoarthritis of the Knee: A Systematic Review and Meta-Analysis. Ann. Intern. Med. 2012, 157, 180–191. [Google Scholar] [CrossRef]
- Szwedowski, D.; Szczepanek, J.; Paczesny, Ł.; Zabrzyński, J.; Gagat, M.; Mobasheri, A.; Jeka, S. The Effect of Platelet-Rich Plasma on the Intra-Articular Microenvironment in Knee Osteoarthritis. Int. J. Mol. Sci. 2021, 22, 5492. [Google Scholar] [CrossRef]
- Andia, I.; Maffulli, N. Platelet-Rich Plasma for Managing Pain and Inflammation in Osteoarthritis. Nat. Rev. Rheumatol. 2013, 9, 721–730. [Google Scholar] [CrossRef] [PubMed]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moser, L.B.; Bauer, C.; Jeyakumar, V.; Niculescu-Morzsa, E.-P.; Nehrer, S. Hyaluronic Acid as a Carrier Supports the Effects of Glucocorticoids and Diminishes the Cytotoxic Effects of Local Anesthetics in Human Articular Chondrocytes In Vitro. Int. J. Mol. Sci. 2021, 22, 11503. https://doi.org/10.3390/ijms222111503
Moser LB, Bauer C, Jeyakumar V, Niculescu-Morzsa E-P, Nehrer S. Hyaluronic Acid as a Carrier Supports the Effects of Glucocorticoids and Diminishes the Cytotoxic Effects of Local Anesthetics in Human Articular Chondrocytes In Vitro. International Journal of Molecular Sciences. 2021; 22(21):11503. https://doi.org/10.3390/ijms222111503
Chicago/Turabian StyleMoser, Lukas B., Christoph Bauer, Vivek Jeyakumar, Eugenia-Paulina Niculescu-Morzsa, and Stefan Nehrer. 2021. "Hyaluronic Acid as a Carrier Supports the Effects of Glucocorticoids and Diminishes the Cytotoxic Effects of Local Anesthetics in Human Articular Chondrocytes In Vitro" International Journal of Molecular Sciences 22, no. 21: 11503. https://doi.org/10.3390/ijms222111503
APA StyleMoser, L. B., Bauer, C., Jeyakumar, V., Niculescu-Morzsa, E.-P., & Nehrer, S. (2021). Hyaluronic Acid as a Carrier Supports the Effects of Glucocorticoids and Diminishes the Cytotoxic Effects of Local Anesthetics in Human Articular Chondrocytes In Vitro. International Journal of Molecular Sciences, 22(21), 11503. https://doi.org/10.3390/ijms222111503





