Modification of Magnetite Nanoparticles with Triazine-Based Dendrons and Their Application as Drug-Transporting Systems
Abstract
:1. Introduction
2. Results and Discussion
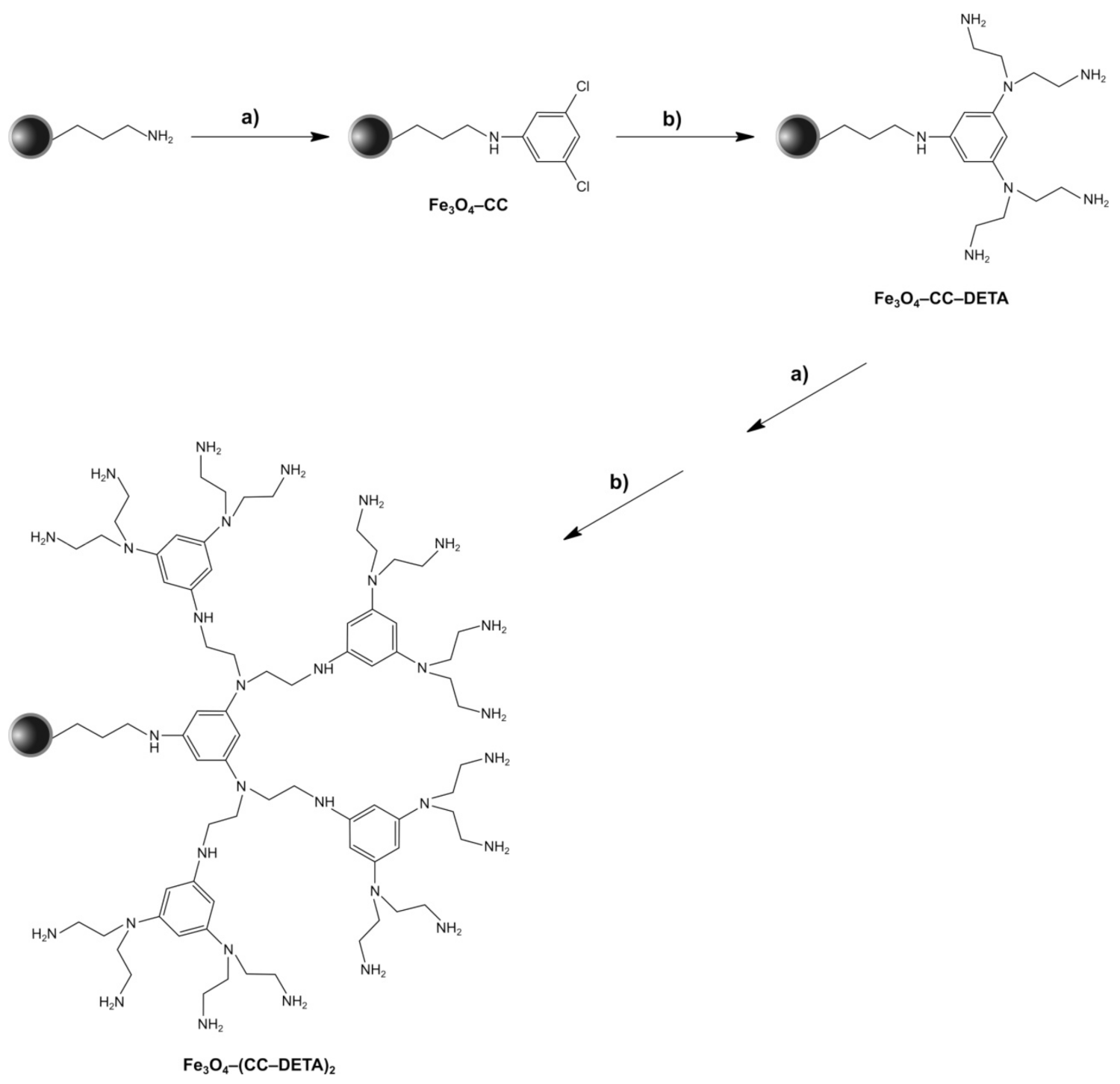
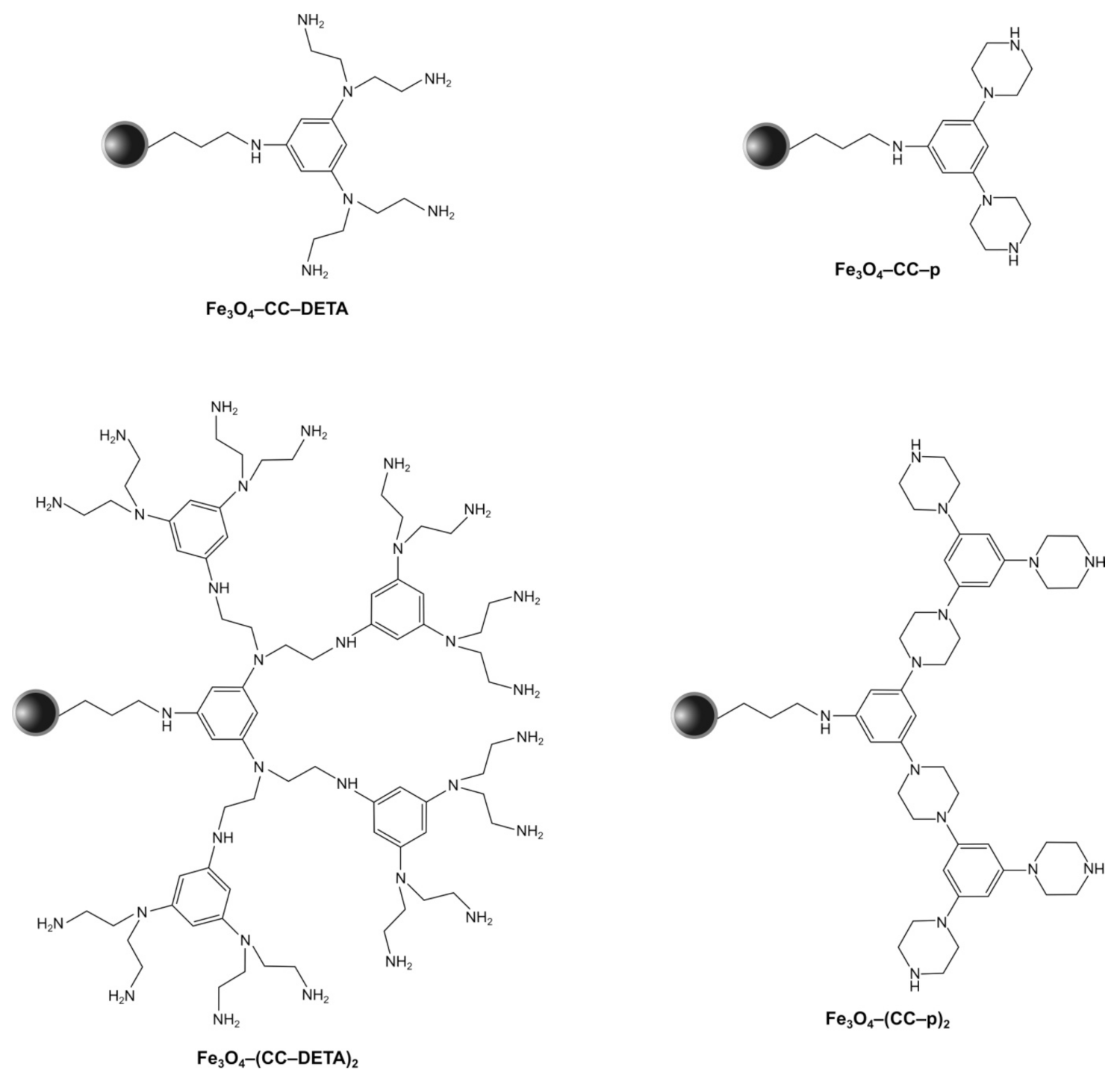
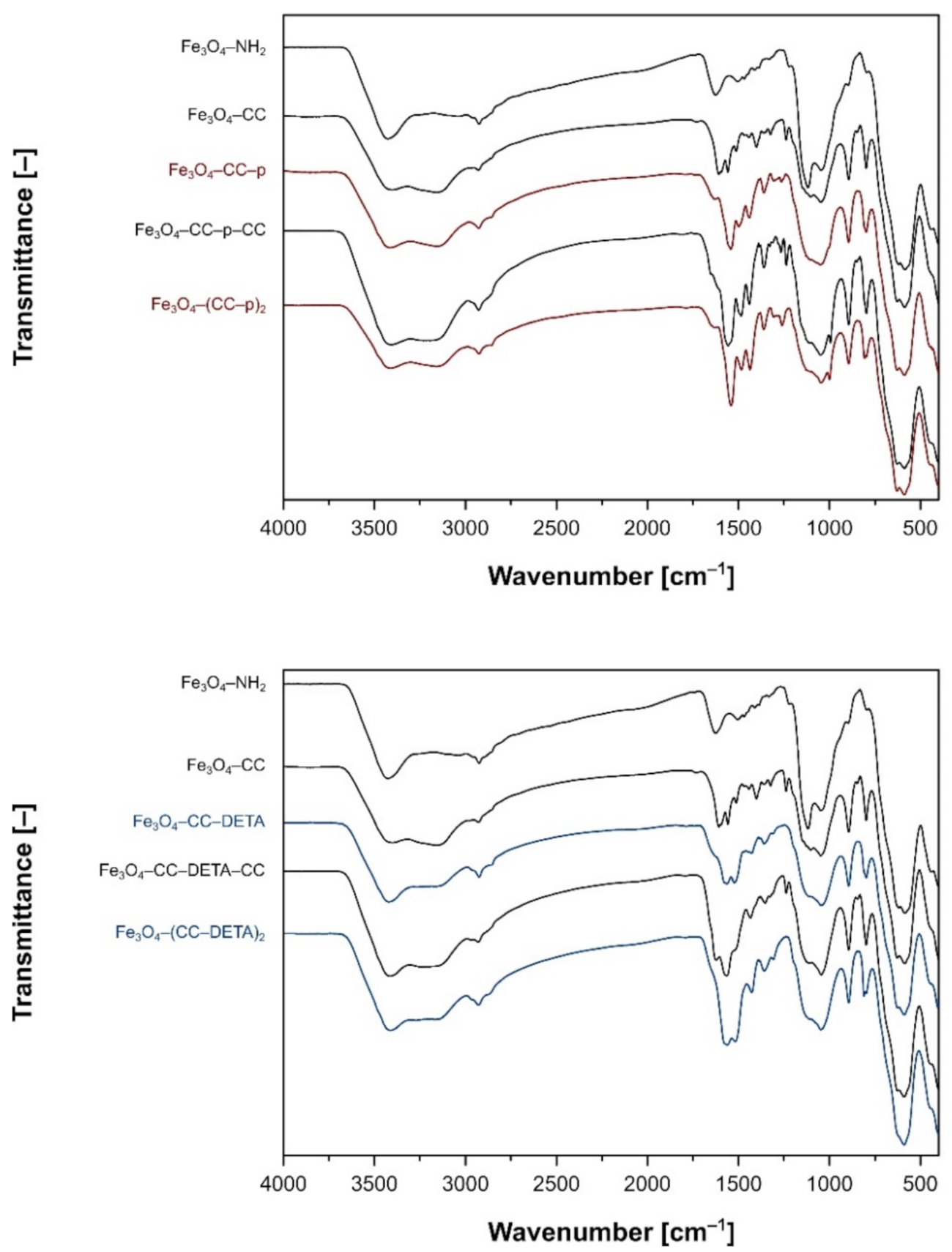
2.1. Synthesis and Characterization of Triazine Dendron-Functionalized Magnetite Nanoparticles
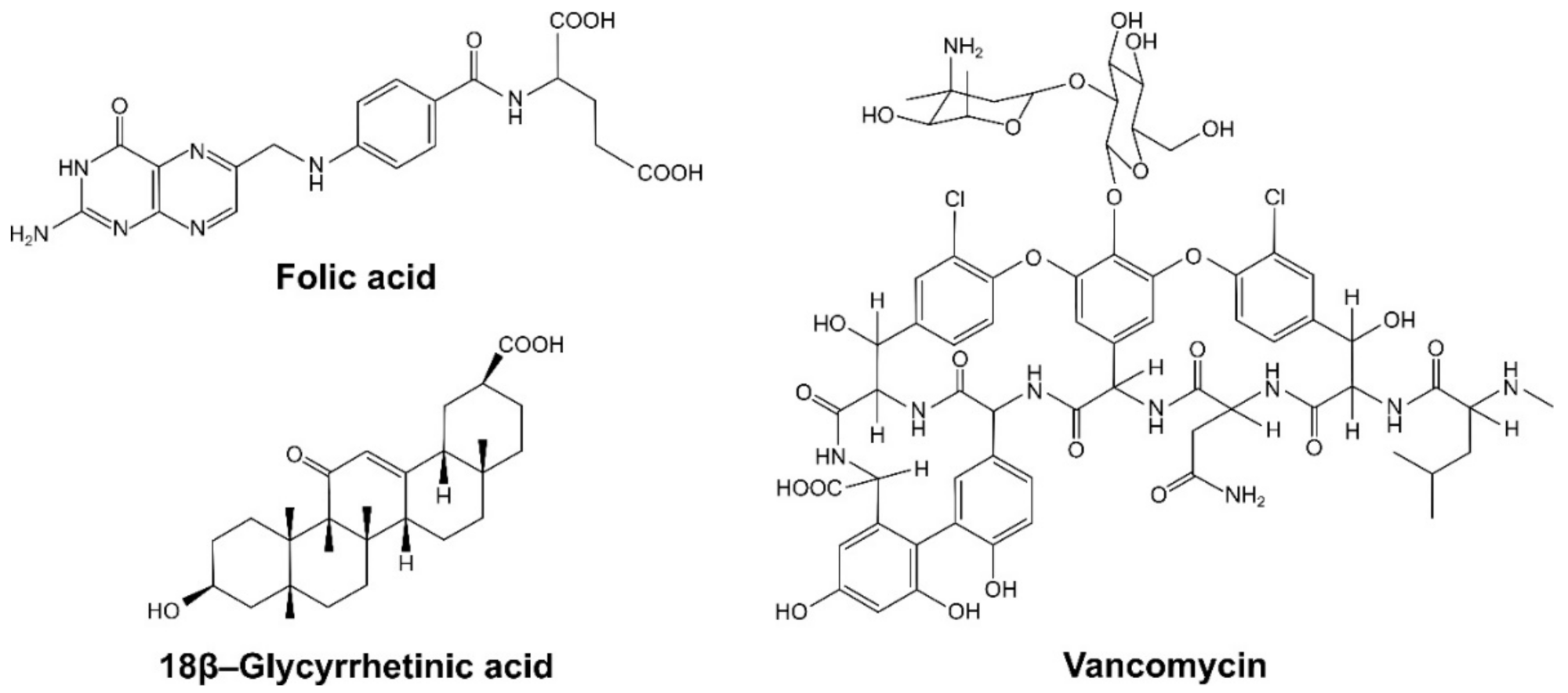
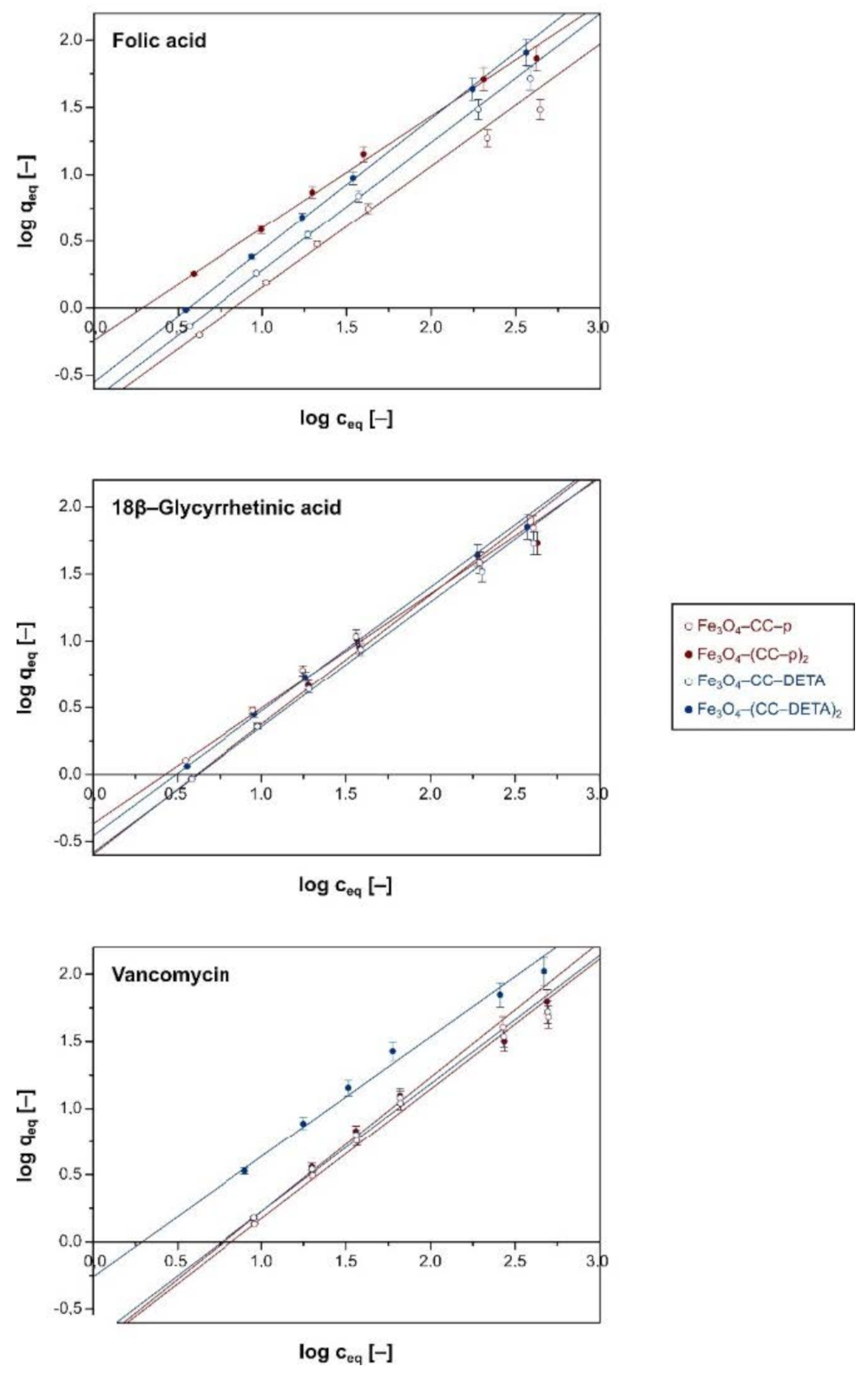
2.2. Investigation of Adsorptive Properties of the Dendron-Functionalized Magnetic Materials towards Bioactive Compounds
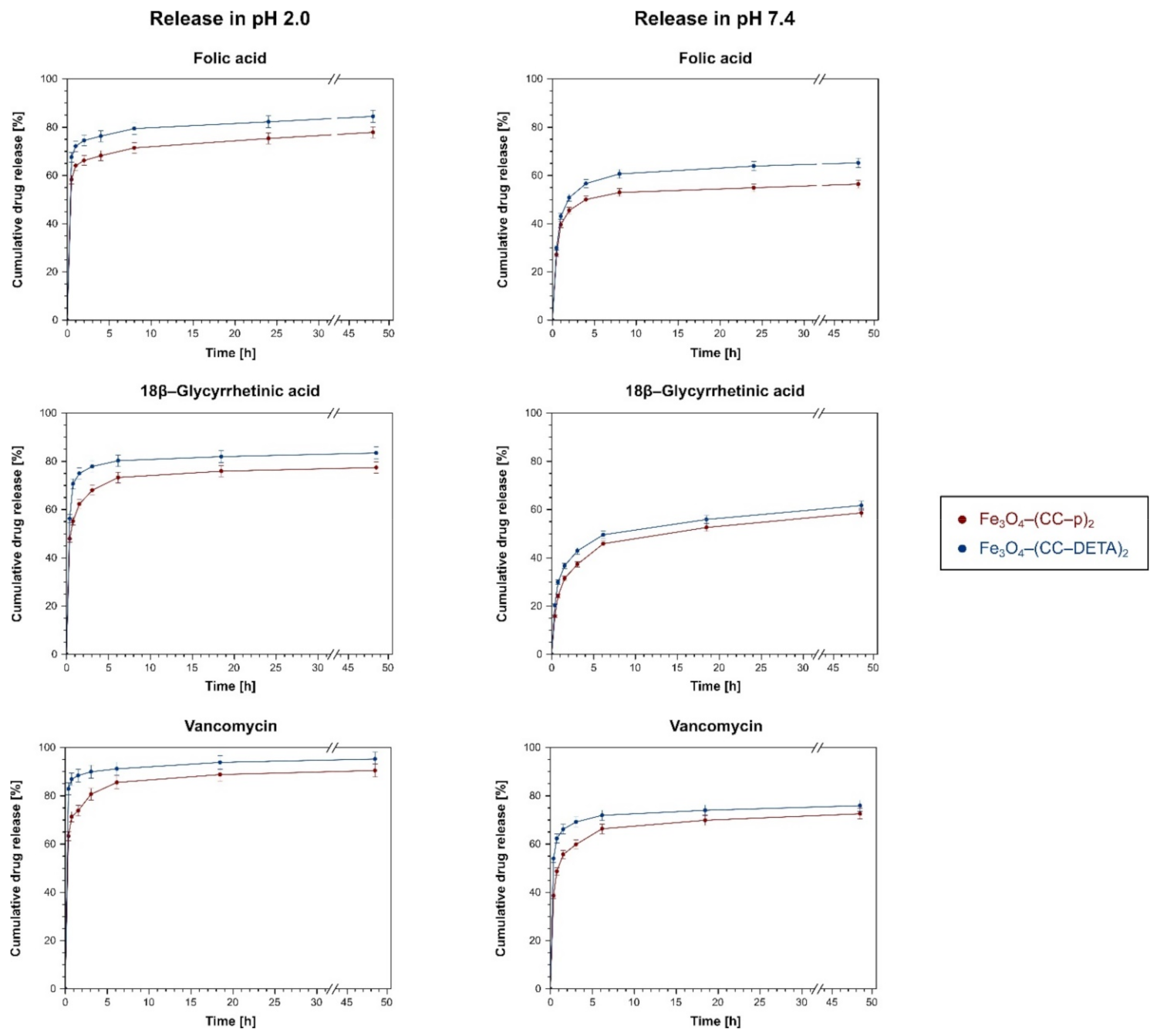
2.3. Investigation of the Materials’ Drug-Delivery Potential
3. Materials and Methods
3.1. Chemicals
3.2. Instruments
3.3. Synthesis of Magnetite Nanoparticles Functionalized with Triazine-Based Dendrons
3.3.1. Synthesis of Amino-Functionalized Magnetic Particles
3.3.2. Quantification of the Amino Groups Loading
3.3.3. Synthesis of Fe3O4–CC Precursor
3.3.4. Synthesis of Triazine-Dendrons Containing Piperazine
3.3.5. Synthesis of Triazine-Dendrons Containing Diethylenetriamine (DETA)
3.3.6. Quantification of the Dendrons Loading on the Materials’ Surface
3.4. Sorption Experiments
3.4.1. Adsorption of the Chosen Acidic Bioactive Compounds
3.4.2. Biocompounds Release Experiments
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ramachandran, R.; Jung, D.; Bernier, N.A.; Logan, J.K.; Waddington, M.A.; Spokoyny, A.M. Sonochemical synthesis of small boron oxide nanoparticles. Inorg. Chem. 2018, 57, 8037–8041. [Google Scholar] [CrossRef]
- Oleshkevich, E.; Teixidor, F.; Rosell, A.; Vinas, C. Merging icosahedral boron clusters and magnetic nanoparticles: Aiming toward multifunctional nanohybrid materials. Inorg. Chem. 2018, 57, 462–470. [Google Scholar] [CrossRef] [PubMed]
- Xiao, D.; Lu, T.; Zeng, R.; Bi, Y. Preparation and highlighted applications of magnetic microparticles and nanoparticles: A review on recent advances. Microchim. Acta 2016, 183, 2655–2675. [Google Scholar] [CrossRef]
- Ghazanfari, M.R.; Kashefi, M.; Shams, S.F.; Jaafari, M.R. Perspective of Fe3O4 Nanoparticles Role in Biomedical Applications. Biochem. Res. Int. 2016, 2016, 7840161. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mieloch, A.A.; Kręcisz, M.; Rybka, J.D.; Strugała, A.; Krupiński, M.; Urbanowicz, A.; Kozak, M.; Skalski, B.; Figlerowicz, M.; Giersig, M. The influence of ligand charge and length on the assembly of Brome mosaic virus derived virus–like particles with magnetic core. AIP Adv. 2018, 8, 35005–35013. [Google Scholar] [CrossRef] [Green Version]
- Su, C. Environmental implications and applications of engineered nanoscale magnetite and its hybrid nanocomposites: A review of recent literature. J. Hazard. Mater. 2017, 332, 48–84. [Google Scholar] [CrossRef]
- Guć, M.; Schroeder, G. Application of Molecularly Imprinted Polymers (MIP) and Magnetic Molecularly Imprinted Polymers (mag-MIP) to Selective Analysis of Quercetin in Flowing Atmospheric-Pressure Afterglow Mass Spectrometry (FAPA-MS) and in Electrospray Ionization Mass Spectrometry (ESI-MS). Molecules 2019, 24, 2364. [Google Scholar]
- Mintzer, M.A.; Grinstaff, M.W. Biomedical applications of dendrimers: A tutorial. Chem. Soc. Rev. 2011, 40, 173–190. [Google Scholar] [CrossRef]
- Ambekar, R.S.; Choudhary, M.; Kandasubramanian, B. Recent advances in dendrimer–based nanoplatform for cancer treatment: A review. Eur. Polym. J. 2020, 126, 109546. [Google Scholar] [CrossRef]
- Viltres, H.; Lopez, Y.C.; Leyva, C.; Gupta, N.K.; Naranjo, A.G.; Acevedo–Pena, P.; Sanchez–Diaz, A.; Bae, J.; Kim, K.S. Polyamidoamine dendrimer–based materials for environmental applications: A review. J. Mol. Liq. 2021, 334, 116017. [Google Scholar] [CrossRef]
- Zheng, W.; Simanek, E.E. Dendrimers Based on Melamine. Divergent and Orthogonal, Convergent Syntheses of a G3 Dendrimer. Org. Lett. 2000, 2, 843–845. [Google Scholar] [CrossRef] [PubMed]
- Patel, R.M.; Patel, H.N.; Gajjar, D.; Patel, P.M. Enhanced Solubility of Non–Steroidal Anti–Inflammatory Drugs by Hydroxyl Terminated S–Triazine Based Dendrimers. Asian J. Pharm. Clin. Res. 2014, 7, 156–161. [Google Scholar]
- Gajjar, D.; Patel, R.M.; Patel, V.A.; Patel, P.M. Novel hydroxyl terminated dendrimers as potential drug carriers: Sustained release, hemolysis and cytotoxicity study. Int. J. Appl. Pharm. 2015, 7, 5–9. [Google Scholar]
- Vembu, S.; Pazhamalai, S.; Gopalakrishnan, M. Potential antibacterial activity of triazine dendrimer: Synthesis and controllable drug release properties. Bioorg. Med. Chem. 2015, 23, 4561–4566. [Google Scholar] [CrossRef] [PubMed]
- Vembu, S.; Pazhamalai, S.; Gopalakrishnan, M. Synthesis, spectral characterization, and effective antifungal evaluation of 1H–tetrazole containing 1,3,5–triazine dendrimers. Med. Chem. Res. 2016, 25, 1916–1924. [Google Scholar] [CrossRef]
- Lim, J.; Lo, S.T.; Hill, S.; Pavan, G.M.; Sun, X.; Simanek, E.E. Antitumor Activity and Molecular Dynamics Simulations of Paclitaxel–Laden Triazine Dendrimers. Mol. Pharm. 2012, 9, 404–412. [Google Scholar] [CrossRef]
- Merkel, O.M.; Mintzer, M.A.; Librizzi, D.; Samsonova, O.; Dicke, T.; Sproat, B.; Garn, H.; Barth, P.J.; Simanek, E.E.; Kissel, T. Triazine Dendrimers as Nonviral Vectors for in Vitro and in Vivo RNAi: The Effects of Peripheral Groups and Core Structure on Biological Activity. Mol. Pharm. 2010, 7, 969–983. [Google Scholar] [CrossRef] [Green Version]
- Golikand, A.N.; Didehban, K.; Irannejad, L. Synthesis and Characterization of Triazine–Based Dendrimers and Their Application in Metal Ion Adsorption. J. Appl. Polym. Sci. 2012, 123, 1245–1251. [Google Scholar] [CrossRef]
- Gajjar, D.; Patel, R.M.; Patel, H.N.; Patel, P.M. Removal of heavy metal ions from water by Hydroxyl terminated Triazine–based Dendrimer. Desalin. Water Treat. 2015, 55, 1209–1219. [Google Scholar] [CrossRef]
- Wu, Q.; You, R.; Lv, Q.; Xu, Y.; You, W.; Yu, Y. Efficient simultaneous removal of Cu(II) and Cr2O72– from aqueous solution by a renewable amphoteric functionalized mesoporous silica. Chem. Eng. J. 2015, 281, 491–501. [Google Scholar] [CrossRef]
- Anbia, M.; Haqshenas, M. Adsorption studies of Pb(II) and Cu(II) ions on mesoporous carbon nitride functionalized with melamine–based dendrimer amine. Int. J. Environ. Sci. Technol. 2015, 12, 2649–2664. [Google Scholar] [CrossRef]
- Arshadi, M.; Faraji, A.R.; Amiri, M.J. Modification of aluminum–silicate nanoparticles by melamine–based dendrimer L–cysteine methyl esters for adsorptive characteristic of Hg(II) ions from the synthetic and Persian Gulf water. Chem. Eng. J. 2015, 266, 345–355. [Google Scholar] [CrossRef]
- Beyki, M.H.; Feizi, F.; Shemirani, F. Melamine–based dendronized magnetic polymer in the adsorption of Pb(ΙΙ) and preconcentration of rhodamine B. React. Funt. Polym. 2016, 103, 81–91. [Google Scholar] [CrossRef]
- Anbia, M.; Khoshobooei, S. Functionalized magnetic MCM–48 nanoporous silica by cyanuric chloride for removal of chlorophenol and bromophenol from aqueous media. J. Nanostruct. Chem. 2015, 5, 139–146. [Google Scholar] [CrossRef] [Green Version]
- Fardjahromi, M.A.; Moghadam, M.; Tangestaninejad, S.; Mirkhani, V.; Mohammadpoor–Baltork, I. Manganese(III) Salophen Supported on Silica Containing Triazine Dendrimer: An Efficient Catalyst for Epoxidation of Alkenes with Sodium Periodate. RSC Adv. 2016, 6, 20128–20134. [Google Scholar] [CrossRef]
- Asadi, B.; Mohammadpoor–Baltork, I.; Tangestaninejad, S.; Moghadam, M.; Mirkhani, V.; Nasr–Esfahani, M. Synthesis and characterization of Bi(III) immobilized on triazine dendrimer–stabilized magnetic nanoparticles: A reusable catalyst for synthesis of aminonaphthoquinones and bisaminonaphthoquinones. New J. Chem. 2016, 40, 6171–6184. [Google Scholar] [CrossRef]
- Pan, S.; Yao, S.; Osako, T.; Uozumi, Y. Controlled Aerobic Oxidation of Primary Benzylic Alcohols to Aldehydes Catalyzed by Polymer–Supported Triazine–Based Dendrimer–Copper Composites. Synlett 2018, 29, 1152–1156. [Google Scholar] [CrossRef]
- Eskandari, A.; Jafarpour, M.; Rezaeifard, A.; Salimi, M. A dendritic TiO2–Co(II) nanocomposite based on the melamine catalyzed one–pot aerobic photocatalytic synthesis of benzimidazoles. New J. Chem. 2018, 42, 6449–6456. [Google Scholar] [CrossRef]
- Pan, S.; Yao, S.; Osako, T.; Uozumi, Y. Batch and Continuous–Flow Huisgen 1,3–Dipolar Cycloadditions with an Amphiphilic Resin–Supported Triazine–Based Polyethyleneamine Dendrimer Copper Catalyst. ACS Sustain. Chem. Eng. 2017, 5, 10722–10734. [Google Scholar] [CrossRef]
- Young, K.L.; Xu, C.; Xie, J.; Sun, S. Conjugating Methotrexate to magnetite (Fe3O4) nanoparticles via trichloro–s–triazine. J. Mater. Chem. 2009, 19, 6400–6409. [Google Scholar] [CrossRef]
- Isfahani, A.L.; Moghadam, M.; Mohammadi, S.; Royvaran, M.; Moshtael–Arani, N.; Rezaei, S.; Tangestaninejad, S.; Mirkhani, V.; Mohammadpoor–Baltork, I. An Elegant pH–Responsive Nanovehicle for Drug Delivery Based on Triazine Dendrimer Modified Magnetic Nanoparticles. Langmuir 2017, 34, 8503–8515. [Google Scholar] [CrossRef] [PubMed]
- Fu, R.; Jin, X.; Liang, J.; Zheng, W.; Zhuang, J.; Yang, W. Preparation of nearly monodispersed Fe3O4/SiO2 composite particles from aggregates of Fe3O4 nanoparticles. J. Mater. Chem. 2011, 21, 15352. [Google Scholar] [CrossRef]
- Moreno, K.X.; Simanek, E.E. Identification of diamine linkers with differing reactivity and their application in the synthesis of melamine dendrimers. Tetrahedron Lett. 2008, 49, 1152–1154. [Google Scholar] [CrossRef] [Green Version]
- Orta, M.D.M.; Martin, J.; Medina–Carrasco, S.; Santos, J.L.; Aparicio, I.; Alonso, E. Adsorption of propranolol onto montmorillonite: Kinetic, isotherm and pH studies. Appl. Clay Sci. 2019, 173, 107–114. [Google Scholar] [CrossRef]
- Kim, Y.S.; Kim, J.H. Isotherm, kinetic and thermodynamic studies on the adsorption of paclitaxel onto Sylopute. J. Chem. Thermodyn. 2019, 130, 104–113. [Google Scholar] [CrossRef]
- Wua, I.Y.; Balaa, S.; Skalko–Basneta, N.; Pio di Cagno, M. Interpreting non–linear drug diffusion data: Utilizing Korsmeyer–Peppas model to study drug release from liposomes. Eur. J. Pharm. Sci. 2019, 138, 105026. [Google Scholar] [CrossRef]
- Guć, M.; Reszke, E.; Cegłowski, M.; Schroeder, G. Construction of Plasma Ion Sources to be Applied in Analysis of Small Organic Compounds Using Mass Spectrometry. Plasma Chem. Plasma Process. 2020, 40, 235–260. [Google Scholar] [CrossRef]
- Pawlaczyk, M.; Pasieczna-Patkowska, S.; Schroeder, G. Photoacoustic Spectroscopy of Surface–Functionalized Fe3O4–SiO2 Nanoparticles. Appl. Spectr. 2020, 74, 712–719. [Google Scholar] [CrossRef] [PubMed]
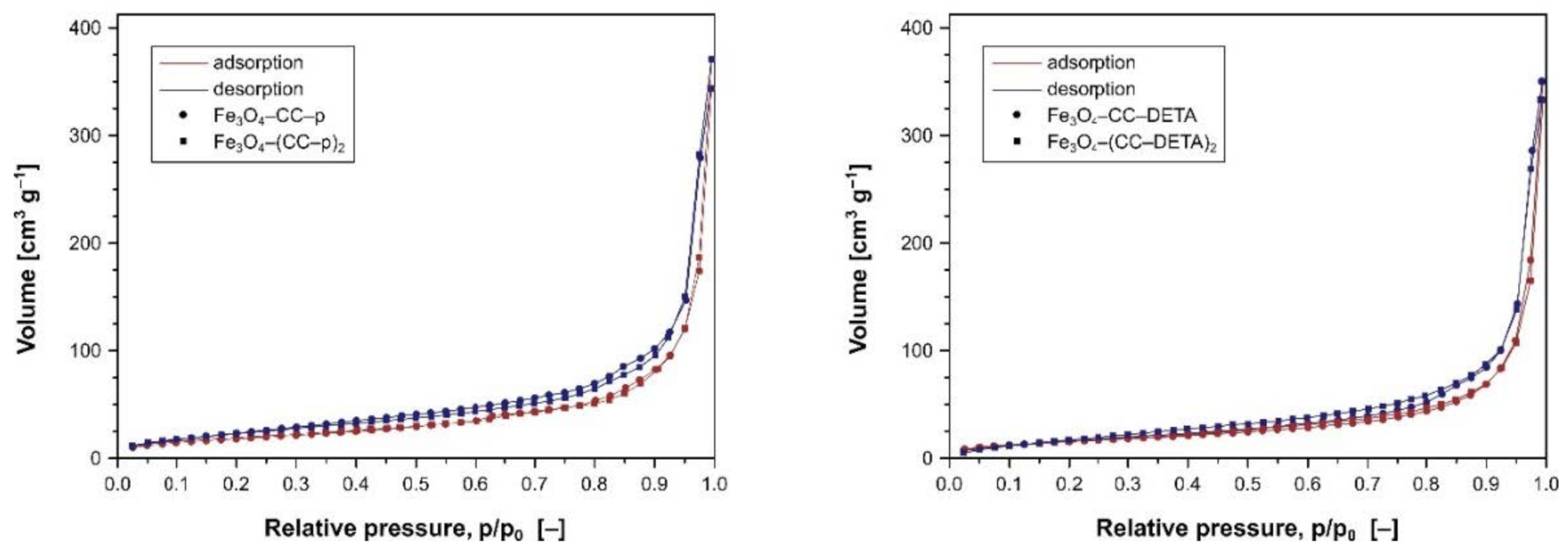

| Material | Loading (mmol g−1) | N2 Adsorption/Desorption Analysis | |||
|---|---|---|---|---|---|
| La | Ld | Surface Area (m2 g−1) | Pore Size (nm) | Pore Volume (cm3 g−1) | |
| Fe3O4–CC–p | 0.390 | 0.195 | 68.914 | 30.9 | 0.533 |
| Fe3O4–(CC–p)2 | 0.719 | 0.180 | 71.672 | 32.1 | 0.575 |
| Fe3O4–CC–DETA | 0.467 | 0.117 | 58.259 | 86.0 | 0.542 |
| Fe3O4–(CC–DETA)2 | 1.104 | 0.069 | 62.063 | 65.9 | 0.515 |
| Material | Langmuir Isotherm | Freundlich Isotherm | ||||
|---|---|---|---|---|---|---|
| qm (mg g−1) | R2 | χ2 | 1/n (–) | R2 | χ2 | |
| Folic Acid | ||||||
| Fe3O4–CC–p | 53.33 ± 4.44 | 0.9794 | 0.072 | 0.91 ± 0.02 | 0.9930 | 0.029 |
| Fe3O4–(CC–p)2 | 123.92 ± 9.54 | 0.9476 | 0.044 | 0.84 ± 0.03 | 0.9971 | 0.008 |
| Fe3O4–CC–DETA | 170.07 ± 18.32 | 0.9880 | 0.047 | 0.96 ± 0.02 | 0.9991 | 0.005 |
| Fe3O4–(CC–DETA)2 | 401.61 ± 10.56 | 0.9960 | 0.005 | 0.99 ± 0.02 | 0.9995 | 0.003 |
| 18β–Glycyrrhetinic Acid | ||||||
| Fe3O4–CC–p | 75.82 ± 4.90 | 0.9658 | 0.102 | 0.86 ± 0.01 | 0.9902 | 0.034 |
| Fe3O4–(CC–p)2 | 176.99 ± 20.68 | 0.9801 | 0.009 | 0.97 ± 0.02 | 0.9987 | 0.007 |
| Fe3O4–CC–DETA | 114.16 ± 11.75 | 0.9894 | 0.019 | 0.94 ± 0.02 | 0.9969 | 0.015 |
| Fe3O4–(CC–DETA)2 | 223.71 ± 21.68 | 0.9723 | 0.013 | 0.93 ± 0.02 | 0.9988 | 0.005 |
| Vancomycin | ||||||
| Fe3O4–CC–p | 68.17 ± 8.38 | 0.9698 | 0.274 | 0.83 ± 0.02 | 0.9932 | 0.022 |
| Fe3O4–(CC–p)2 | 97.66 ± 9.07 | 0.9747 | 0.149 | 0.86 ± 0.02 | 0.9971 | 0.017 |
| Fe3O4–CC–DETA | 73.10 ± 6.62 | 0.9639 | 0.307 | 0.82 ± 0.02 | 0.9991 | 0.019 |
| Fe3O4–(CC–DETA)2 | 132.45 ± 6.70 | 0.9682 | 0.172 | 0.77 ± 0.03 | 0.9995 | 0.019 |
| Drug Release Model | Linear Representation | Parameters |
|---|---|---|
| Zero-order model | F0 and Ft—the initial and the cumulative amount of the drug released at time t, respectively (mg) k0—the zero-order release constant (mg h−1) kH–C—the Hixson-Crowell release constant (mg1/3 h−1) | |
| Hixson–Crowell model | ||
| First-order model | Qt—the cumulative percentage of the drug releases at time t (%) k1—the first-order release constant (% h−1) kH—the Higuchi release constant (% h−1/2) n—the Korsmeyer–Peppas exponent of release (–) kK–P—the Korsmeyer–Peppas release constant (% h−1) | |
| Higuchi model | ||
| Korsmeyer–Peppas model |
| Adsorbent | Higuchi Model | Korsemeyer-Peppas Model | ||||
|---|---|---|---|---|---|---|
| kH (% h−1/2) | R2 | n | kK–P (% h−1) | R2 | ||
| Folic Acid | ||||||
| pH 2.0 | Fe3O4–(CC–p)2 | 2.7 ± 0.5 | 0.8561 | 0.06 | 62.7 ± 0.6 | 0.9676 |
| Fe3O4–(CC–DETA)2 | 2.4 ± 0.4 | 0.8435 | 0.05 | 71.4 ± 0.5 | 0.9717 | |
| pH 7.4 | Fe3O4–(CC–p)2 | 3.5 ± 1.3 | 0.5944 | 0.14 | 37.1 ± 2.7 | 0.7633 |
| Fe3O4–(CC–DETA)2 | 4.4 ± 1.5 | 0.6341 | 0.15 | 41.0 ± 2.9 | 0.7994 | |
| 18β–Glycyrrhetinic Acid | ||||||
| pH 2.0 | Fe3O4–(CC–p)2 | 4.1 ± 1.2 | 0.7087 | 0.10 | 55.6 ± 1.8 | 0.8967 |
| Fe3O4–(CC–DETA)2 | 3.0 ± 1.2 | 0.5353 | 0.07 | 67.0 ± 2.7 | 0.7295 | |
| pH 7.4 | Fe3O4–(CC–p)2 | 6.2 ± 1.1 | 0.8603 | 0.27 | 23.3 ± 1.7 | 0.9273 |
| Fe3O4–(CC–DETA)2 | 5.8 ± 1.1 | 0.8388 | 0.22 | 28.5 ± 1.8 | 0.9192 | |
| Vancomycin | ||||||
| pH 2.0 | Fe3O4–(CC–p)2 | 3.8 ± 0.9 | 0.7603 | 0.08 | 70.0 ± 1.4 | 0.9302 |
| Fe3O4–(CC–DETA)2 | 1.7 ± 0.3 | 0.8205 | 0.03 | 86.0 ± 0.5 | 0.9581 | |
| pH 7.4 | Fe3O4–(CC–p)2 | 4.6 ± 1.2 | 0.7395 | 0.13 | 47.5 ± 2.0 | 0.8922 |
| Fe3O4–(CC–DETA)2 | 2.7 ± 0.8 | 0.6832 | 0.07 | 60.8 ± 1.5 | 0.8664 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pawlaczyk, M.; Schroeder, G. Modification of Magnetite Nanoparticles with Triazine-Based Dendrons and Their Application as Drug-Transporting Systems. Int. J. Mol. Sci. 2021, 22, 11353. https://doi.org/10.3390/ijms222111353
Pawlaczyk M, Schroeder G. Modification of Magnetite Nanoparticles with Triazine-Based Dendrons and Their Application as Drug-Transporting Systems. International Journal of Molecular Sciences. 2021; 22(21):11353. https://doi.org/10.3390/ijms222111353
Chicago/Turabian StylePawlaczyk, Mateusz, and Grzegorz Schroeder. 2021. "Modification of Magnetite Nanoparticles with Triazine-Based Dendrons and Their Application as Drug-Transporting Systems" International Journal of Molecular Sciences 22, no. 21: 11353. https://doi.org/10.3390/ijms222111353
APA StylePawlaczyk, M., & Schroeder, G. (2021). Modification of Magnetite Nanoparticles with Triazine-Based Dendrons and Their Application as Drug-Transporting Systems. International Journal of Molecular Sciences, 22(21), 11353. https://doi.org/10.3390/ijms222111353






