Mesenchymal Stem Cells and Extracellular Vesicles in Osteosarcoma Pathogenesis and Therapy
Abstract
1. Introduction
2. Mesenchymal Stem Cells’ Role in Osteosarcoma
2.1. Cellular Origin of OS
2.2. MSCs in OS Microenvironment
2.3. Recruitment of MSCs to the Tumor Site
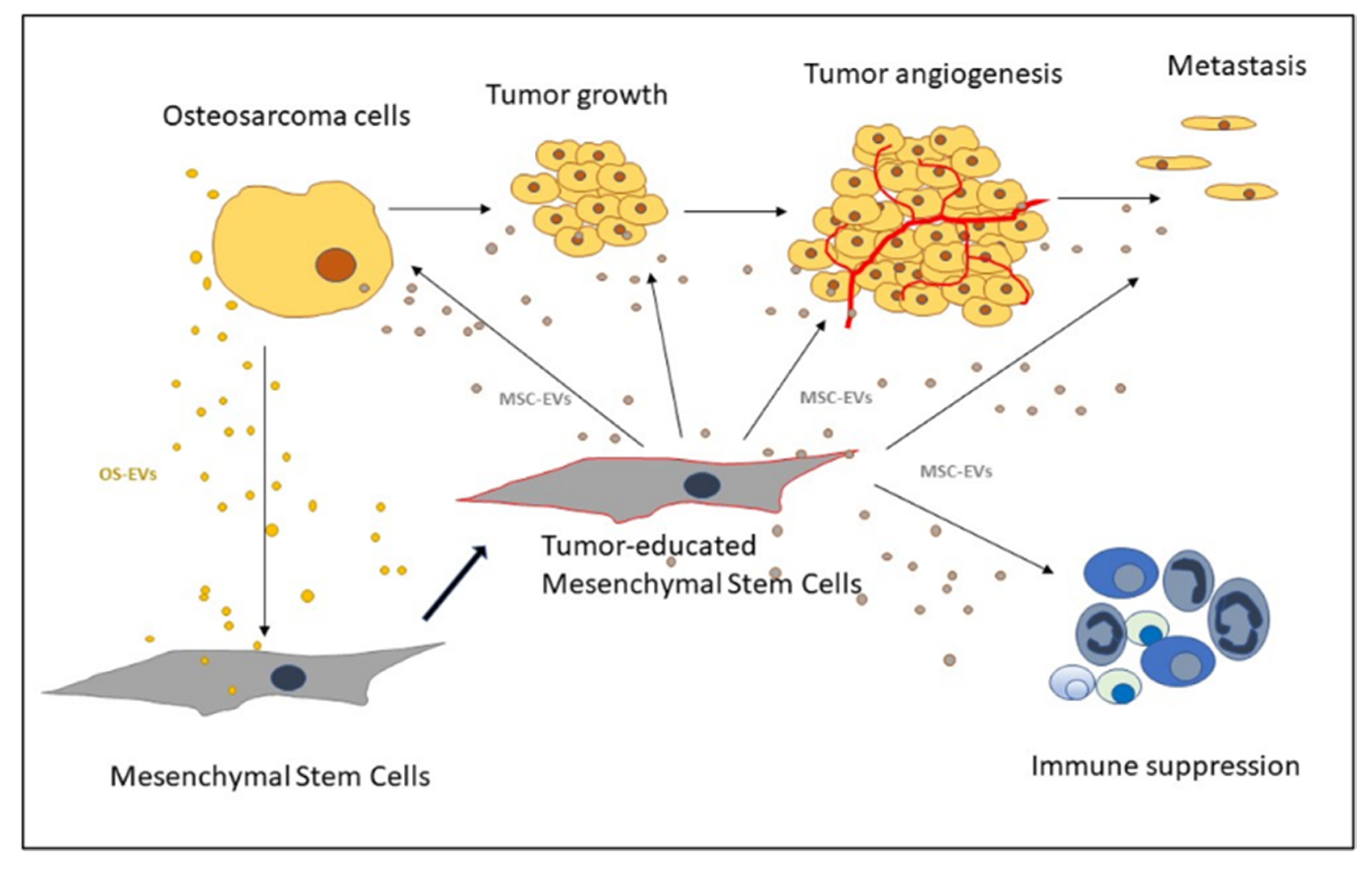
2.4. OS Microenvironment-Induced Changes in MSCs
2.5. Transformation of MSCs into Carcinoma-Associated Fibroblasts
2.6. MSCs’ Role in Promoting OS Growth and Progression
2.7. MSCs’ Role in Angiogenesis
2.8. MSCs’ Role in Metastasis
2.9. MSCs’ Role in Drug Resistance
2.10. MSCs as Suppressors of OS Growth
3. Extracellular Vesicles as Mediators of MSCs and Osteosarcoma Crosstalk
3.1. OS-EVs
3.2. MSC-EVs
3.3. MSC-EVs in Angiogenesis
3.4. MSC-EVs in OS Metastasis
3.5. MSC-EVs in Immune Response
4. MSCs in OS Therapy
MSC-EVs in OS Therapy
5. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Mirabello, L.; Troisi, R.J.; Savage, S.A. Osteosarcoma Incidence and Survival Rates from 1973 to 2004: Data from the Surveillance, Epidemiology, and End Results Program. Cancer 2009, 115, 1531–1543. [Google Scholar] [CrossRef]
- ESMO European Sarcoma Network Working Group. Bone Sarcomas: ESMO Clinical Practice Guidelines for Diagnosis, Treatment and Follow-Up. Ann. Oncol. 2012, 23, vii100-9. [Google Scholar] [CrossRef]
- Broadhead, M.L.; Clark, J.C.; Myers, D.E.; Dass, C.R.; Choong, P.F. The Molecular Pathogenesis of Osteosarcoma: A Review. Sarcoma 2011, 2011, 959248. [Google Scholar] [CrossRef]
- Kempf-Bielack, B.; Bielack, S.S.; Jurgens, H.; Branscheid, D.; Berdel, W.E.; Exner, G.U.; Gobel, U.; Helmke, K.; Jundt, G.; Kabisch, H.; et al. Osteosarcoma Relapse After Combined Modality Therapy: An Analysis of Unselected Patients in the Cooperative Osteosarcoma Study Group (COSS). J. Clin. Oncol. 2005, 23, 559–568. [Google Scholar] [CrossRef] [PubMed]
- Bhaskar, B.; Mekala, N.K.; Baadhe, R.R.; Rao, P.S. Role of Signaling Pathways in Mesenchymal Stem Cell Differentiation. Curr. Stem Cell. Res. Ther. 2014, 9, 508–512. [Google Scholar] [CrossRef]
- Main, H.; Munsie, M.; O’Connor, M.D. Managing the Potential and Pitfalls during Clinical Translation of Emerging Stem Cell Therapies. Clin. Transl. Med. 2014, 3, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Yang, R.; Roth, M.; Piperdi, S.; Zhang, W.; Dorfman, H.; Rao, P.; Park, A.; Tripathi, S.; Freeman, C.; et al. Genetically Transforming Human Osteoblasts to Sarcoma: Development of an Osteosarcoma Model. Genes Cancer 2017, 8, 484–494. [Google Scholar] [CrossRef] [PubMed]
- Rubio, R.; Gutierrez-Aranda, I.; Saez-Castillo, A.I.; Labarga, A.; Rosu-Myles, M.; Gonzalez-Garcia, S.; Toribio, M.L.; Menendez, P.; Rodriguez, R. The Differentiation Stage of p53-Rb-Deficient Bone Marrow Mesenchymal Stem Cells Imposes the Phenotype of in vivo Sarcoma Development. Oncogene 2013, 32, 4970–4980. [Google Scholar] [CrossRef]
- Mutsaers, A.J.; Walkley, C.R. Cells of Origin in Osteosarcoma: Mesenchymal Stem Cells Or Osteoblast Committed Cells? Bone 2014, 62, 56–63. [Google Scholar] [CrossRef]
- Uccelli, A.; Moretta, L.; Pistoia, V. Mesenchymal Stem Cells in Health and Disease. Nat. Rev. Immunol. 2008, 8, 726–736. [Google Scholar] [CrossRef]
- Jo, V.Y.; Fletcher, C.D. WHO Classification of Soft Tissue Tumours: An Update Based on the 2013 (4th) Edition. Pathology 2014, 46, 95–104. [Google Scholar] [CrossRef]
- Mohseny, A.B.; Szuhai, K.; Romeo, S.; Buddingh, E.P.; Briaire-de Bruijn, I.; de Jong, D.; van Pel, M.; Cleton-Jansen, A.M.; Hogendoorn, P.C. Osteosarcoma Originates from Mesenchymal Stem Cells in Consequence of Aneuploidization and Genomic Loss of Cdkn2. J. Pathol. 2009, 219, 294–305. [Google Scholar] [CrossRef]
- Wang, J.Y.; Wu, P.K.; Chen, P.C.; Lee, C.W.; Chen, W.M.; Hung, S.C. Generation of Osteosarcomas from a Combination of Rb Silencing and C-Myc Overexpression in Human Mesenchymal Stem Cells. Stem Cells Transl. Med. 2017, 6, 512–526. [Google Scholar] [CrossRef] [PubMed]
- Walkley, C.R.; Qudsi, R.; Sankaran, V.G.; Perry, J.A.; Gostissa, M.; Roth, S.I.; Rodda, S.J.; Snay, E.; Dunning, P.; Fahey, F.H.; et al. Conditional Mouse Osteosarcoma, Dependent on p53 Loss and Potentiated by Loss of Rb, Mimics the Human Disease. Genes Dev. 2008, 22, 1662–1676. [Google Scholar] [CrossRef] [PubMed]
- Dani, N.; Olivero, M.; Mareschi, K.; van Duist, M.M.; Miretti, S.; Cuvertino, S.; Patane, S.; Calogero, R.; Ferracini, R.; Scotlandi, K.; et al. The MET Oncogene Transforms Human Primary Bone-Derived Cells into Osteosarcomas by Targeting Committed Osteo-Progenitors. J. Bone Miner. Res. 2012, 27, 1322–1334. [Google Scholar] [CrossRef] [PubMed]
- Chang, X.; Ma, Z.; Zhu, G.; Lu, Y.; Yang, J. New Perspective into Mesenchymal Stem Cells: Molecular Mechanisms Regulating Osteosarcoma. J. Bone Oncol. 2021, 29, 100372. [Google Scholar] [CrossRef] [PubMed]
- Abarrategi, A.; Tornin, J.; Martinez-Cruzado, L.; Hamilton, A.; Martinez-Campos, E.; Rodrigo, J.P.; Gonzalez, M.V.; Baldini, N.; Garcia-Castro, J.; Rodriguez, R. Osteosarcoma: Cells-of-Origin, Cancer Stem Cells, and Targeted Therapies. Stem Cells Int. 2016, 2016, 3631764. [Google Scholar] [CrossRef] [PubMed]
- Corre, I.; Verrecchia, F.; Crenn, V.; Redini, F.; Trichet, V. The Osteosarcoma Microenvironment: A Complex but Targetable Ecosystem. Cells 2020, 9, 976. [Google Scholar] [CrossRef]
- Sun, Z.; Wang, S.; Zhao, R.C. The Roles of Mesenchymal Stem Cells in Tumor Inflammatory Microenvironment. J. Hematol. Oncol. 2014, 7, 14. [Google Scholar] [CrossRef] [PubMed]
- Pietrovito, L.; Leo, A.; Gori, V.; Lulli, M.; Parri, M.; Becherucci, V.; Piccini, L.; Bambi, F.; Taddei, M.L.; Chiarugi, P. Bone Marrow-Derived Mesenchymal Stem Cells Promote Invasiveness and Transendothelial Migration of Osteosarcoma Cells Via a Mesenchymal to Amoeboid Transition. Mol. Oncol. 2018, 12, 659–676. [Google Scholar] [CrossRef]
- Gutova, M.; Najbauer, J.; Frank, R.T.; Kendall, S.E.; Gevorgyan, A.; Metz, M.Z.; Guevorkian, M.; Edmiston, M.; Zhao, D.; Glackin, C.A.; et al. Urokinase Plasminogen Activator and Urokinase Plasminogen Activator Receptor Mediate Human Stem Cell Tropism to Malignant Solid Tumors. Stem Cells 2008, 26, 1406–1413. [Google Scholar] [CrossRef] [PubMed]
- Ho, I.A.; Yulyana, Y.; Sia, K.C.; Newman, J.P.; Guo, C.M.; Hui, K.M.; Lam, P.Y. Matrix metalloproteinase-1-mediated mesenchymal stem cell tumor tropism is dependent on crosstalk with stromal derived growth factor 1/C-X-C chemokine receptor 4 axis. FASEB J. 2014, 28, 4359–4368. [Google Scholar] [CrossRef] [PubMed]
- Baglio, S.R.; Lagerweij, T.; Perez-Lanzon, M.; Ho, X.D.; Leveille, N.; Melo, S.A.; Cleton-Jansen, A.M.; Jordanova, E.S.; Roncuzzi, L.; Greco, M.; et al. Blocking Tumor-Educated MSC Paracrine Activity Halts Osteosarcoma Progression. Clin. Cancer Res. 2017, 23, 3721–3733. [Google Scholar] [CrossRef] [PubMed]
- Mannerstrom, B.; Kornilov, R.; Abu-Shahba, A.G.; Chowdhury, I.M.; Sinha, S.; Seppanen-Kaijansinkko, R.; Kaur, S. Epigenetic Alterations in Mesenchymal Stem Cells by Osteosarcoma-Derived Extracellular Vesicles. Epigenetics 2019, 14, 352–364. [Google Scholar] [CrossRef] [PubMed]
- Tu, B.; Zhu, J.; Liu, S.; Wang, L.; Fan, Q.; Hao, Y.; Fan, C.; Tang, T.T. Mesenchymal Stem Cells Promote Osteosarcoma Cell Survival and Drug Resistance through Activation of STAT3. Oncotarget 2016, 7, 48296–48308. [Google Scholar] [CrossRef]
- Cortini, M.; Massa, A.; Avnet, S.; Bonuccelli, G.; Baldini, N. Tumor-Activated Mesenchymal Stromal Cells Promote Osteosarcoma Stemness and Migratory Potential Via IL-6 Secretion. PLoS ONE 2016, 11, e0166500. [Google Scholar] [CrossRef]
- Avnet, S.; Di Pompo, G.; Chano, T.; Errani, C.; Ibrahim-Hashim, A.; Gillies, R.J.; Donati, D.M.; Baldini, N. Cancer-Associated Mesenchymal Stroma Fosters the Stemness of Osteosarcoma Cells in Response to Intratumoral Acidosis Via NF-kappaB Activation. Int. J. Cancer 2017, 140, 1331–1345. [Google Scholar] [CrossRef]
- Wang, Y.; Chu, Y.; Li, K.; Zhang, G.; Guo, Z.; Wu, X.; Qiu, C.; Li, Y.; Wan, X.; Sui, J.; et al. Exosomes Secreted by Adipose-Derived Mesenchymal Stem Cells Foster Metastasis and Osteosarcoma Proliferation by Increasing COLGALT2 Expression. Front. Cell. Dev. Biol. 2020, 8, 353. [Google Scholar] [CrossRef]
- Wang, Y.; Chu, Y.; Yue, B.; Ma, X.; Zhang, G.; Xiang, H.; Liu, Y.; Wang, T.; Wu, X.; Chen, B. Adipose-Derived Mesenchymal Stem Cells Promote Osteosarcoma Proliferation and Metastasis by Activating the STAT3 Pathway. Oncotarget 2017, 8, 23803–23816. [Google Scholar] [CrossRef]
- Kawano, M.; Tanaka, K.; Itonaga, I.; Iwasaki, T.; Tsumura, H. Interaction between Human Osteosarcoma and Mesenchymal Stem Cells Via an Interleukin-8 Signaling Loop in the Tumor Microenvironment. Cell. Commun. Signal. 2018, 16, 13. [Google Scholar] [CrossRef]
- Lu, M.; Xie, K.; Lu, X.; Lu, L.; Shi, Y.; Tang, Y. Notoginsenoside R1 Counteracts Mesenchymal Stem Cell-Evoked Oncogenesis and Doxorubicin Resistance in Osteosarcoma Cells by Blocking IL-6 Secretion-Induced JAK2/STAT3 Signaling. Invest. New Drugs 2021, 39, 416–425. [Google Scholar] [CrossRef]
- Vallabhaneni, K.C.; Hassler, M.Y.; Abraham, A.; Whitt, J.; Mo, Y.Y.; Atfi, A.; Pochampally, R. Mesenchymal Stem/Stromal Cells Under Stress Increase Osteosarcoma Migration and Apoptosis Resistance Via Extracellular Vesicle Mediated Communication. PLoS ONE 2016, 11, e0166027. [Google Scholar] [CrossRef] [PubMed]
- Tu, B.; Du, L.; Fan, Q.M.; Tang, Z.; Tang, T.T. STAT3 Activation by IL-6 from Mesenchymal Stem Cells Promotes the Proliferation and Metastasis of Osteosarcoma. Cancer Lett. 2012, 325, 80–88. [Google Scholar] [CrossRef] [PubMed]
- Tu, B.; Peng, Z.X.; Fan, Q.M.; Du, L.; Yan, W.; Tang, T.T. Osteosarcoma Cells Promote the Production of Pro-Tumor Cytokines in Mesenchymal Stem Cells by Inhibiting their Osteogenic Differentiation through the TGF-Beta/Smad2/3 Pathway. Exp. Cell Res. 2014, 320, 164–173. [Google Scholar] [CrossRef] [PubMed]
- Du, L.; Han, X.G.; Tu, B.; Wang, M.Q.; Qiao, H.; Zhang, S.H.; Fan, Q.M.; Tang, T.T. CXCR1/Akt Signaling Activation Induced by Mesenchymal Stem Cell-Derived IL-8 Promotes Osteosarcoma Cell Anoikis Resistance and Pulmonary Metastasis. Cell. Death Dis. 2018, 9, 714. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.; Zhu, B.; Huang, G.; Zeng, Q.; Wang, C. Microvesicles Derived from Human Bone Marrow Mesenchymal Stem Cells Promote U2OS Cell Growth Under Hypoxia: The Role of PI3K/AKT and HIF-1alpha. Hum. Cell 2019, 32, 64–74. [Google Scholar] [CrossRef] [PubMed]
- Qi, J.; Zhou, Y.; Jiao, Z.; Wang, X.; Zhao, Y.; Li, Y.; Chen, H.; Yang, L.; Zhu, H.; Li, Y. Exosomes Derived from Human Bone Marrow Mesenchymal Stem Cells Promote Tumor Growth through Hedgehog Signaling Pathway. Cell. Physiol. Biochem. 2017, 42, 2242–2254. [Google Scholar] [CrossRef]
- Liu, T.; Zhou, L.; Li, D.; Andl, T.; Zhang, Y. Cancer-Associated Fibroblasts Build and Secure the Tumor Microenvironment. Front. Cell. Dev. Biol. 2019, 7, 60. [Google Scholar]
- Zhu, H.; Guo, S.; Zhang, Y.; Yin, J.; Yin, W.; Tao, S.; Wang, Y.; Zhang, C. Proton-Sensing GPCR-YAP Signalling Promotes Cancer-Associated Fibroblast Activation of Mesenchymal Stem Cells. Int. J. Biol. Sci. 2016, 12, 389–396. [Google Scholar] [CrossRef]
- Lin, L.; Huang, K.; Guo, W.; Zhou, C.; Wang, G.; Zhao, Q. Conditioned Medium of the Osteosarcoma Cell Line U2OS Induces hBMSCs to Exhibit Characteristics of Carcinoma-Associated Fibroblasts Via Activation of IL-6/STAT3 Signalling. J. Biochem. 2020, 168, 265–271. [Google Scholar] [CrossRef]
- Wang, Y.M.; Wang, W.; Qiu, E.D. Osteosarcoma Cells Induce Differentiation of Mesenchymal Stem Cells into Cancer Associated Fibroblasts through Notch and Akt Signaling Pathway. Int. J. Clin. Exp. Pathol. 2017, 10, 8479–8486. [Google Scholar]
- Maurizi, G.; Verma, N.; Gadi, A.; Mansukhani, A.; Basilico, C. Sox2 is Required for Tumor Development and Cancer Cell Proliferation in Osteosarcoma. Oncogene 2018, 37, 4626–4632. [Google Scholar] [CrossRef]
- Xu, W.T.; Bian, Z.Y.; Fan, Q.M.; Li, G.; Tang, T.T. Human Mesenchymal Stem Cells (hMSCs) Target Osteosarcoma and Promote its Growth and Pulmonary Metastasis. Cancer Lett. 2009, 281, 32–41. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S. TGF-Beta Regulates Beta-Catenin Signaling and Osteoblast Differentiation in Human Mesenchymal Stem Cells. J. Cell. Biochem. 2011, 112, 1651–1660. [Google Scholar] [CrossRef] [PubMed]
- Li, G.C.; Zhang, H.W.; Zhao, Q.C.; Sun, L.I.; Yang, J.J.; Hong, L.; Feng, F.; Cai, L. Mesenchymal stem cells promote tumor angiogenesis via the action of transforming growth factor β1. Oncol. Lett. 2016, 11, 1089–1094. [Google Scholar] [CrossRef] [PubMed]
- Batlle, R.; Andrés, E.; Gonzalez, L.; Llonch, E.; Igea, A.; Gutierrez-Prat, N.; Berenguer-Llergo, A.; Nebreda, A.R. Regulation of tumor angiogenesis and mesenchymal-endothelial transition by p38α through TGF-β and JNK signaling. Nat. Commun. 2019, 10, 3071. [Google Scholar] [CrossRef]
- Chaturvedi, P.; Gilkes, D.M.; Wong, C.C.; Kshitiz, J.; Luo, W.; Zhang, H.; Wei, H.; Takano, N.; Schito, L.; Levchenko, A.; et al. Hypoxia-Inducible Factor-Dependent Breast Cancer-Mesenchymal Stem Cell Bidirectional Signaling Promotes Metastasis. J. Clin. Invest. 2013, 123, 189–205. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Dong, L.; Yan, K.; Long, H.; Yang, T.T.; Dong, M.Q.; Zhou, Y.; Fan, Q.Y.; Ma, B.A. CXCR4-Mediated Osteosarcoma Growth and Pulmonary Metastasis is Promoted by Mesenchymal Stem Cells through VEGF. Oncol. Rep. 2013, 30, 1753–1761. [Google Scholar] [CrossRef]
- Pelagalli, A.; Nardelli, A.; Fontanella, R.; Zannetti, A. Inhibition of AQP1 Hampers Osteosarcoma and Hepatocellular Carcinoma Progression Mediated by Bone Marrow-Derived Mesenchymal Stem Cells. Int. J. Mol. Sci. 2016, 17, 1102. [Google Scholar] [CrossRef]
- Birru, B.; Durthi, C.P.; Kacham, S.; Pola, M.; Rajulapati, S.B.; Parcha, S.R.; Kamal, M.A. Stem Cells in Tumour Microenvironment Aid in Prolonged Survival Rate of Cancer Cells and Developed Drug Resistance: Major Challenge in Osteosarcoma Treatment. Curr. Drug Metab. 2020, 21, 44–52. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Wang, C.; Zuo, D.; Zhang, T.; Yin, F.; Zhou, Z.; Wang, H.; Xu, J.; Mao, M.; Wang, G.; et al. Attenuation of STAT3 Phosphorylation Promotes Apoptosis and Chemosensitivity in Human Osteosarcoma Induced by Raddeanin A. Int. J. Biol. Sci. 2019, 15, 668–679. [Google Scholar] [CrossRef]
- Bharti, R.; Dey, G.; Mandal, M. Cancer Development, Chemoresistance, Epithelial to Mesenchymal Transition and Stem Cells: A Snapshot of IL-6 Mediated Involvement. Cancer Lett. 2016, 375, 51–61. [Google Scholar] [CrossRef]
- Roodhart, J.M.; Daenen, L.G.; Stigter, E.C.; Prins, H.J.; Gerrits, J.; Houthuijzen, J.M.; Gerritsen, M.G.; Schipper, H.S.; Backer, M.J.; van Amersfoort, M.; et al. Mesenchymal stem cells induce resistance to chemotherapy through the release of platinum-induced fatty acids. Cancer Cell 2011, 20, 370–383. [Google Scholar] [CrossRef]
- van der Velden, D.L.; Cirkel, G.A.; Houthuijzen, J.M.; van Werkhoven, E.; Roodhart, J.M.L.; Daenen, L.G.M.; Kaing, S.; Gerrits, J.; Verhoeven-Duif, N.M.; Grootscholten, C.; et al. Phase I study of combined indomethacin and platinum-based chemotherapy to reduce platinum-induced fatty acids. Cancer Chemother Pharmacol. 2018, 81, 911–921. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Wang, J.; Ren, T.; Huang, Y.; Liang, X.; Yu, Y.; Wang, W.; Niu, J.; Guo, W. Bone Marrow Mesenchymal Stem Cell-Derived Exosomal miR-206 Inhibits Osteosarcoma Progression by Targeting TRA2B. Cancer Lett. 2020, 490, 54–65. [Google Scholar] [CrossRef] [PubMed]
- Eiro, N.; Fraile, M.; Fernandez-Francos, S.; Sanchez, R.; Costa, L.A.; Vizoso, F.J. Importance of the Origin of Mesenchymal (Stem) Stromal Cells in Cancer Biology: “Alliance” or “War” in Intercellular Signals. Cell Biosci. 2021, 11, 109. [Google Scholar] [CrossRef] [PubMed]
- Aanstoos, M.E.; Regan, D.P.; Rose, R.J.; Chubb, L.S.; Ehrhart, N.P. Do Mesenchymal Stromal Cells Influence Microscopic Residual Or Metastatic Osteosarcoma in a Murine Model? Clin. Orthop. Relat. Res. 2016, 474, 707–715. [Google Scholar] [CrossRef]
- Lee, S.W.; Jeon, T.J.; Biswal, S. Effect of Local Treatment with Adipose Tissue-Derived Mesenchymal Stem Cells in the Early Tumorigenesis of Osteosarcoma. Oncol. Rep. 2015, 33, 1381–1387. [Google Scholar] [CrossRef][Green Version]
- Ghannam, S.; Bouffi, C.; Djouad, F.; Jorgensen, C.; Noel, D. Immunosuppression by Mesenchymal Stem Cells: Mechanisms and Clinical Applications. Stem Cell. Res. Ther. 2010, 1, 2. [Google Scholar] [CrossRef]
- Paino, F.; La Noce, M.; Di Nucci, D.; Nicoletti, G.F.; Salzillo, R.; De Rosa, A.; Ferraro, G.A.; Papaccio, G.; Desiderio, V.; Tirino, V. Human Adipose Stem Cell Differentiation is Highly Affected by Cancer Cells both in vitro and in vivo: Implication for Autologous Fat Grafting. Cell. Death Dis. 2017, 8, e2568. [Google Scholar] [CrossRef]
- Gauthaman, K.; Yee, F.C.; Cheyyatraivendran, S.; Biswas, A.; Choolani, M.; Bongso, A. Human Umbilical Cord Wharton’s Jelly Stem Cell (hWJSC) Extracts Inhibit Cancer Cell Growth in vitro. J. Cell. Biochem. 2012, 113, 2027–2039. [Google Scholar] [CrossRef] [PubMed]
- Shen, W.C.; Lai, Y.C.; Li, L.H.; Liao, K.; Lai, H.C.; Kao, S.Y.; Wang, J.; Chuong, C.M.; Hung, S.C. Methylation and PTEN Activation in Dental Pulp Mesenchymal Stem Cells Promotes Osteogenesis and Reduces Oncogenesis. Nat. Commun. 2019, 10, 2226. [Google Scholar] [CrossRef] [PubMed]
- Kalluri, R.; LeBleu, V.S. The Biology, Function, and Biomedical Applications of Exosomes. Science 2020, 367, 977. [Google Scholar] [CrossRef] [PubMed]
- Raimondi, L.; De Luca, A.; Gallo, A.; Costa, V.; Russelli, G.; Cuscino, N.; Manno, M.; Raccosta, S.; Carina, V.; Bellavia, D.; et al. Osteosarcoma Cell-Derived Exosomes Affect Tumor Microenvironment by Specific Packaging of microRNAs. Carcinogenesis 2020, 41, 666–677. [Google Scholar] [CrossRef]
- Wortzel, I.; Dror, S.; Kenific, C.M.; Lyden, D. Exosome-Mediated Metastasis: Communication from a Distance. Dev. Cell 2019, 49, 347–360. [Google Scholar] [CrossRef]
- Lobb, R.J.; Lima, L.G.; Moller, A. Exosomes: Key Mediators of Metastasis and Pre-Metastatic Niche Formation. Semin. Cell Dev. Biol. 2017, 67, 3–10. [Google Scholar] [CrossRef]
- Zhuang, G.; Wu, X.; Jiang, Z.; Kasman, I.; Yao, J.; Guan, Y.; Oeh, J.; Modrusan, Z.; Bais, C.; Sampath, D.; et al. Tumour-Secreted miR-9 Promotes Endothelial Cell Migration and Angiogenesis by Activating the JAK-STAT Pathway. EMBO J. 2012, 31, 3513–3523. [Google Scholar] [CrossRef]
- Perut, F.; Roncuzzi, L.; Zini, N.; Massa, A.; Baldini, N. Extracellular Nanovesicles Secreted by Human Osteosarcoma Cells Promote Angiogenesis. Cancers 2019, 11, 779. [Google Scholar] [CrossRef]
- Li, Y.; Lin, S.; Xie, X.; Zhu, H.; Fan, T.; Wang, S. Highly Enriched Exosomal lncRNA OIP5-AS1 Regulates Osteosarcoma Tumor Angiogenesis and Autophagy through miR-153 and ATG5. Am. J. Transl. Res. 2021, 13, 4211–4223. [Google Scholar]
- Huang, Y.; Liu, W.; He, B.; Wang, L.; Zhang, F.; Shu, H.; Sun, L. Exosomes Derived from Bone Marrow Mesenchymal Stem Cells Promote Osteosarcoma Development by Activating Oncogenic Autophagy. J. Bone Oncol. 2020, 21, 100280. [Google Scholar] [CrossRef]
- Li, F.; Chen, X.; Shang, C.; Ying, Q.; Zhou, X.; Zhu, R.; Lu, H.; Hao, X.; Dong, Q.; Jiang, Z. Bone Marrow Mesenchymal Stem Cells-Derived Extracellular Vesicles Promote Proliferation, Invasion and Migration of Osteosarcoma Cells via the lncRNA MALAT1/miR-143/NRSN2/Wnt/Beta-Catenin Axis. Onco Targets Ther. 2021, 14, 737–749. [Google Scholar] [CrossRef]
- Zhao, W.; Qin, P.; Zhang, D.; Cui, X.; Gao, J.; Yu, Z.; Chai, Y.; Wang, J.; Li, J. Long Non-Coding RNA PVT1 Encapsulated in Bone Marrow Mesenchymal Stem Cell-Derived Exosomes Promotes Osteosarcoma Growth and Metastasis by Stabilizing ERG and Sponging miR-183-5p. Aging 2019, 11, 9581–9596. [Google Scholar] [CrossRef]
- Qin, F.; Tang, H.; Zhang, Y.; Zhang, Z.; Huang, P.; Zhu, J. Bone Marrow-Derived Mesenchymal Stem Cell-Derived Exosomal microRNA-208a Promotes Osteosarcoma Cell Proliferation, Migration, and Invasion. J. Cell. Physiol. 2020, 235, 4734–4745. [Google Scholar] [CrossRef]
- Macklin, R.; Wang, H.; Loo, D.; Martin, S.; Cumming, A.; Cai, N.; Lane, R.; Ponce, N.S.; Topkas, E.; Inder, K.; et al. Extracellular Vesicles Secreted by Highly Metastatic Clonal Variants of Osteosarcoma Preferentially Localize to the Lungs and Induce Metastatic Behaviour in Poorly Metastatic Clones. Oncotarget 2016, 7, 43570–43587. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Dong, C.; Chen, M.; Yang, T.; Wang, X.; Gao, Y.; Wang, L.; Wen, Y.; Chen, G.; Wang, X.; et al. Extracellular Vesicle-Mediated Delivery of miR-101 Inhibits Lung Metastasis in Osteosarcoma. Theranostics 2020, 10, 411–425. [Google Scholar] [CrossRef] [PubMed]
- Shimbo, K.; Miyaki, S.; Ishitobi, H.; Kato, Y.; Kubo, T.; Shimose, S.; Ochi, M. Exosome-Formed Synthetic microRNA-143 is Transferred to Osteosarcoma Cells and Inhibits their Migration. Biochem. Biophys. Res. Commun. 2014, 445, 381–387. [Google Scholar]
- Kobayashi, E.; Hornicek, F.J.; Duan, Z. MicroRNA Involvement in Osteosarcoma. Sarcoma 2012, 2012, 359739. [Google Scholar] [CrossRef]
- Alcayaga-Miranda, F.; Varas-Godoy, M.; Khoury, M. Harnessing the Angiogenic Potential of Stem Cell-Derived Exosomes for Vascular Regeneration. Stem Cells Int. 2016, 2016, 3409169. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-King, H.; Garcia, N.A.; Ontoria-Oviedo, I.; Ciria, M.; Montero, J.A.; Sepulveda, P. Hypoxia Inducible Factor-1alpha Potentiates Jagged 1-Mediated Angiogenesis by Mesenchymal Stem Cell-Derived Exosomes. Stem Cells 2017, 35, 1747–1759. [Google Scholar] [CrossRef] [PubMed]
- Gong, M.; Yu, B.; Wang, J.; Wang, Y.; Liu, M.; Paul, C.; Millard, R.W.; Xiao, D.S.; Ashraf, M.; Xu, M. Mesenchymal Stem Cells Release Exosomes that Transfer miRNAs to Endothelial Cells and Promote Angiogenesis. Oncotarget 2017, 8, 45200–45212. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.K.; Park, S.R.; Jung, B.K.; Jeon, Y.K.; Lee, Y.S.; Kim, M.K.; Kim, Y.G.; Jang, J.Y.; Kim, C.W. Exosomes Derived from Mesenchymal Stem Cells Suppress Angiogenesis by Down-Regulating VEGF Expression in Breast Cancer Cells. PLoS ONE 2013, 8, e84256. [Google Scholar]
- Zhu, W.; Huang, L.; Li, Y.; Zhang, X.; Gu, J.; Yan, Y.; Xu, X.; Wang, M.; Qian, H.; Xu, W. Exosomes Derived from Human Bone Marrow Mesenchymal Stem Cells Promote Tumor Growth in vivo. Cancer Lett. 2012, 315, 28–37. [Google Scholar] [CrossRef]
- Bian, Z.Y.; Fan, Q.M.; Li, G.; Xu, W.T.; Tang, T.T. Human Mesenchymal Stem Cells Promote Growth of Osteosarcoma: Involvement of Interleukin-6 in the Interaction between Human Mesenchymal Stem Cells and Saos-2. Cancer Sci. 2010, 101, 2554–2560. [Google Scholar] [CrossRef]
- Le Blanc, K.; Ringden, O. Immunobiology of Human Mesenchymal Stem Cells and Future use in Hematopoietic Stem Cell Transplantation. Biol. Blood Marrow Transplant. 2005, 11, 321–334. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Yin, Y.; Lai, R.C.; Tan, S.S.; Choo, A.B.; Lim, S.K. Mesenchymal Stem Cells Secrete Immunologically Active Exosomes. Stem Cells Dev. 2014, 23, 1233–1244. [Google Scholar] [CrossRef]
- Mardpour, S.; Hamidieh, A.A.; Taleahmad, S.; Sharifzad, F.; Taghikhani, A.; Baharvand, H. Interaction between Mesenchymal Stromal Cell-Derived Extracellular Vesicles and Immune Cells by Distinct Protein Content. J. Cell. Physiol. 2019, 234, 8249–8258. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Fu, L.; Liang, Y.; Guo, Z.; Wang, L.; Ma, C.; Wang, H. Exosomes Originating from MSCs Stimulated with TGF-Beta and IFN-Gamma Promote Treg Differentiation. J. Cell. Physiol. 2018, 233, 6832–6840. [Google Scholar] [CrossRef] [PubMed]
- Khare, D.; Or, R.; Resnick, I.; Barkatz, C.; Almogi-Hazan, O.; Avni, B. Mesenchymal Stromal Cell-Derived Exosomes Affect mRNA Expression and Function of B-Lymphocytes. Front. Immunol. 2018, 9, 3053. [Google Scholar] [CrossRef] [PubMed]
- Luo, Z.W.; Liu, P.P.; Wang, Z.X.; Chen, C.Y.; Xie, H. Macrophages in Osteosarcoma Immune Microenvironment: Implications for Immunotherapy. Front. Oncol. 2020, 10, 586580. [Google Scholar] [CrossRef]
- Cersosimo, F.; Lonardi, S.; Bernardini, G.; Telfer, B.; Mandelli, G.E.; Santucci, A.; Vermi, W.; Giurisato, E. Tumor-Associated Macrophages in Osteosarcoma: From Mechanisms to Therapy. Int. J. Mol. Sci. 2020, 21, 5207. [Google Scholar] [CrossRef] [PubMed]
- Showalter, M.R.; Wancewicz, B.; Fiehn, O.; Archard, J.A.; Clayton, S.; Wagner, J.; Deng, P.; Halmai, J.; Fink, K.D.; Bauer, G.; et al. Primed Mesenchymal Stem Cells Package Exosomes with Metabolites Associated with Immunomodulation. Biochem. Biophys. Res. Commun. 2019, 512, 729–735. [Google Scholar] [CrossRef] [PubMed]
- Jia, X.H.; Feng, G.W.; Wang, Z.L.; Du, Y.; Shen, C.; Hui, H.; Peng, D.; Li, Z.J.; Kong, D.L.; Tian, J. Activation of mesenchymal stem cells by macrophages promotes tumor progression through immune suppressive effects. Oncotarget 2016, 12, 20934–20944. [Google Scholar] [CrossRef]
- Shao, X.J.; Xiang, S.F.; Chen, Y.Q.; Zhang, N.; Cao, J.; Zhu, H.; Yang, B.; Zhou, Q.; Ying, M.D.; He, Q.J. Inhibition of M2-like macrophages by all-trans retinoic acid prevents cancer initiation and stemness in osteosarcoma cells. Acta. Pharmacol. Sin. 2019, 40, 1343–1350. [Google Scholar] [CrossRef] [PubMed]
- Gunassekaran, G.R.; Poongkavithai Vadevoo, S.M.; Baek, M.C.; Lee, B. M1 macrophage exosomes engineered to foster M1 polarization and target the IL-4 receptor inhibit tumor growth by reprogramming tumor-associated macrophages into M1-like macrophages. Biomaterials 2021, 278, 121137. [Google Scholar] [CrossRef] [PubMed]
- Grisendi, G.; Spano, C.; D’souza, N.; Rasini, V.; Veronesi, E.; Prapa, M.; Petrachi, T.; Piccinno, S.; Rossignoli, F.; Burns, J.S.; et al. Mesenchymal Progenitors Expressing TRAIL Induce Apoptosis in Sarcomas. Stem Cells 2015, 33, 859–869. [Google Scholar] [CrossRef]
- Gamie, Z.; Kapriniotis, K.; Papanikolaou, D.; Haagensen, E.; Da Conceicao Ribeiro, R.; Dalgarno, K.; Krippner-Heidenreich, A.; Gerrand, C.; Tsiridis, E.; Rankin, K.S. TNF-Related Apoptosis-Inducing Ligand (TRAIL) for Bone Sarcoma Treatment: Pre-Clinical and Clinical Data. Cancer Lett. 2017, 409, 66–80. [Google Scholar] [CrossRef]
- Qiao, B.; Shui, W.; Cai, L.; Guo, S.; Jiang, D. Human Mesenchymal Stem Cells as Delivery of Osteoprotegerin Gene: Homing and Therapeutic Effect for Osteosarcoma. Drug Des. Devel. Ther. 2015, 9, 969–976. [Google Scholar] [CrossRef] [PubMed]
- Duan, X.; Guan, H.; Cao, Y.; Kleinerman, E.S. Murine Bone Marrow-Derived Mesenchymal Stem Cells as Vehicles for Interleukin-12 Gene Delivery into Ewing Sarcoma Tumors. Cancer 2009, 115, 13–22. [Google Scholar] [CrossRef]
- Duchi, S.; Sotgiu, G.; Lucarelli, E.; Ballestri, M.; Dozza, B.; Santi, S.; Guerrini, A.; Dambruoso, P.; Giannini, S.; Donati, D.; et al. Mesenchymal Stem Cells as Delivery Vehicle of Porphyrin Loaded Nanoparticles: Effective Photoinduced in vitro Killing of Osteosarcoma. J. Control. Release 2013, 168, 225–237. [Google Scholar] [CrossRef]
- Lenna, S.; Bellotti, C.; Duchi, S.; Martella, E.; Columbaro, M.; Dozza, B.; Ballestri, M.; Guerrini, A.; Sotgiu, G.; Frisoni, T.; et al. Mesenchymal stromal cells mediated delivery of photoactive nanoparticles inhibits osteosarcoma growth in vitro and in a murine in vivo ectopic model. J. Exp. Clin. Cancer Res. 2020, 39, 40. [Google Scholar] [CrossRef]
- Ruano, D.; López-Martín, J.A.; Moreno, L.; Lassaletta, Á.; Bautista, F.; Andión, M.; Hernández, C.; González-Murillo, Á.; Melen, G.; Alemany, R.; et al. First-in-Human, First-in-Child Trial of Autologous MSCs Carrying the Oncolytic Virus Icovir-5 in Patients with Advanced Tumors. Mol. Ther. 2020, 28, 1033–1042. [Google Scholar] [CrossRef] [PubMed]
- Morales-Molina, A.; Gambera, S.; Leo, A.; García-Castro, J. Combination immunotherapy using G-CSF and oncolytic virotherapy reduces tumor growth in osteosarcoma. J. Immunother. Cancer 2021, 9, e001703. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Zhou, X.; Wu, J.; Cui, X.; Wang, M.; Wang, X.; Gao, Z. Mesenchymal stem cell-derived exosomes carrying microRNA-150 suppresses the proliferation and migration of osteosarcoma cells via targeting IGF2BP1. Transl. Cancer Res. 2020, 9, 5323–5335. [Google Scholar] [CrossRef]
- Xu, Z.; Zeng, S.; Gong, Z.; Yan, Y. Exosome-based immunotherapy: A promising approach for cancer treatment. Mol. Cancer 2020, 19, 160. [Google Scholar] [CrossRef]

| OS Signal | Effect on MSCs | MSC Signal | Effect on OS | Reference |
|---|---|---|---|---|
| Exosomes | Increase in COLGALT2 and proliferation. | Wang et al. 2020 [28] | ||
| CM 1/co-culture | Increase in MMP2/9; STAT3 activation. Increased proliferation, invasion, and metastasis. | Wang et al. 2017 [29] | ||
| IL-8 (co-culture) | Increased IL-8. | IL-8 (co-culture) | Increased IL-8. Increased metastatic potential. | Kawano et al. 2018 [30] |
| IL-6 CM/co-culture | Increase in MMP2/9; JAK2/STAT3 activation. Increased proliferation, migration, and doxorubicin resistance. | Lu et al. 2021 [31] | ||
| MCP-1, GRO-α, and TGFβ | Mesenchymal-to-amoeboid transition. Increase in MCP-1, GRO-α, IL-6, and IL-8 in the tumor environment. | Increased migration, invasion, and trans-endothelial migration. | Pietrovito et al. 2018 [20] | |
| OS-EVs | LINE-1 hypomethylation increased VEGF-A. | Mannerström et al. 2019 [24] | ||
| CM | STAT3 activation. Promote survival and drug resistance. | Tu et al. 2016 [25] | ||
| MSC-EVs (under stress) | Increased migration. Apoptosis resistance. | Vallabhaneni et al. 2016 [32] | ||
| Co-culture | Increased TGFβ. | Co-culture IL-6 | Increased OS proliferation, stemness & migration. | Cortini et al. 2016 [26] |
| IL-6 | STAT3 activation. Increased proliferation and metastasis. | Tu et al. 2012 [33] | ||
| CM/TGFβ | Increased IL-6, VEGF. Inhibit osteogenic differentiation. | Tu et al. 2014 [34] | ||
| IL-8 | CXCR1/Akt activation. Promotes metastasis. | Du et al. 2018 [35] | ||
| EVs/TGFβ | Increased IL-6. | Tumor-educated MSC | Activation of STAT3 signaling. | Baglio et al. 2017 [23] |
| EVs | Cell growth under hypoxia. Activation of PI3K/AKT & HIF-1α. | Lin et al. 2019 [36] | ||
| EVs | Activation of Hedgehog signaling. Tumor growth. | Qi et al. 2017 [37] |
| Source of EVs | miRNAs/other RNAs in EVs | Proteins in EVs | Function | Reference |
|---|---|---|---|---|
| OS cells | miR-146a-5p, miR-10b-5p, miR-143-3p, miR-382-5p, miR-150-5p, miR-125b-5p, miR-27a-3p, miR-145-5p, miR-26a-5p, miR-93-5p, miR-21-5p, miR-92a-3p, and miR-106a-5p | serpin-E1, serpin-F1, TIMP-1, thrombospondin-1, urokinase-type plasminogen activator (uPA), VEGF, pentraxin-3, PDGF-AA, angiopoietin-2, coagulation factor-III, CD26, CD105, endostatin, endothelin-1, and HB-EGF | Angiogenesis. | Perut et al. 2019 [68] |
| OS cells & tissue | lncRNA OIP5-AS1 | Angiogenesis. | Li et al. 2021 [69] | |
| OS cells | miR-148a-3p and miR-21-5p | TME remodeling. | Raimondi et al. 2020 [64] | |
| OS cells | TGFβ | Increase IL6 in AD-MSCs, tumor growth, STAT3 activation, and lung metastasis. Autophagy. | Baglio et al. 2017, Tu et al. 2012, Huang et al. 2020 [23,33,70] | |
| BM-MSC | lncRNA MALAT1 | Proliferation, invasion, and migration of OS cells via lncRNA MALAT1/miR-143/NRSN2/Wnt/β-Catenin Axis. | Li et al. 2021 [71] | |
| BM-MSC | non-coding RNA PVT1 | OS migration by upregulating ERG and sponging miR-183-5p in OS cells. | Zhao et al. 2019 [72] | |
| BM-MSC | miR-206 | Tumor suppression and apoptosis. | Zhang et al. 2020 [55] | |
| BM-MSC | microRNA-208a LCP1 | OS cell migration & invasion. OS proliferation and metastasis via the JAK2/STAT3 pathway. | Qin et al. 2020 [73] | |
| Highly metastatic OS cells | NPM1, CCT2, CCT4, CCT6A, CCT8, VIM, CLTC, COL6A2, HNRNPC, PKM, ACTN4, MYH10, PAICS, VCP, ANXA1, ACLY | Metastasis. | Macklin et al. 2016 [74] | |
| Engineered AD-MSC | miR-101; miR-150 synthetic miR-143 | OS Therapy. Suppress OS growth. | Zhang et al. 2020; Shimbo et al. 2014; Xu et al. 2020; [75,76] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sarhadi, V.K.; Daddali, R.; Seppänen-Kaijansinkko, R. Mesenchymal Stem Cells and Extracellular Vesicles in Osteosarcoma Pathogenesis and Therapy. Int. J. Mol. Sci. 2021, 22, 11035. https://doi.org/10.3390/ijms222011035
Sarhadi VK, Daddali R, Seppänen-Kaijansinkko R. Mesenchymal Stem Cells and Extracellular Vesicles in Osteosarcoma Pathogenesis and Therapy. International Journal of Molecular Sciences. 2021; 22(20):11035. https://doi.org/10.3390/ijms222011035
Chicago/Turabian StyleSarhadi, Virinder Kaur, Ravindra Daddali, and Riitta Seppänen-Kaijansinkko. 2021. "Mesenchymal Stem Cells and Extracellular Vesicles in Osteosarcoma Pathogenesis and Therapy" International Journal of Molecular Sciences 22, no. 20: 11035. https://doi.org/10.3390/ijms222011035
APA StyleSarhadi, V. K., Daddali, R., & Seppänen-Kaijansinkko, R. (2021). Mesenchymal Stem Cells and Extracellular Vesicles in Osteosarcoma Pathogenesis and Therapy. International Journal of Molecular Sciences, 22(20), 11035. https://doi.org/10.3390/ijms222011035






