Neuroprotective Effect of Vascular Endothelial Growth Factor on Motoneurons of the Oculomotor System
Abstract
1. Vascular Endothelial Growth Factor (VEGF)
1.1. History
1.2. VEGF Family
1.3. Functions of VEGF
1.4. VEGF Expression
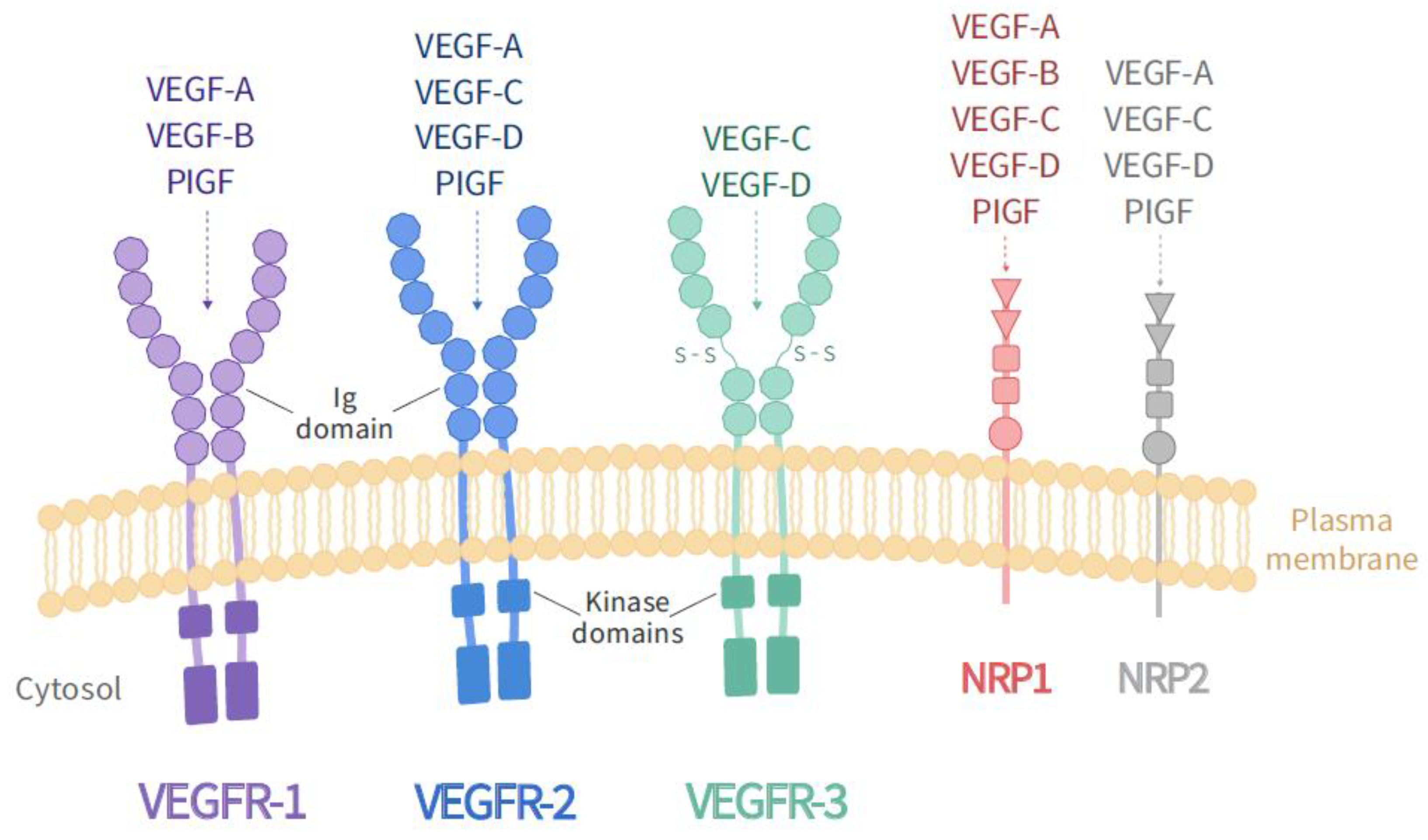
2. VEGF Receptors
3. Effects of Low Levels of VEGF
VEGF and ALS
4. Neuroprotective Effect of VEGF
4.1. Anti-Apoptotic Effects of VEGF
4.2. Role of Excitotoxicity in Neurodegeneration and VEGF Protection
5. Selective Vulnerability of Motoneurons to Neurodegeneration
6. Properties of Ocular Motoneurons
6.1. VEGF and FLK-1 Expression in the Oculomotor System
6.2. VEGF Sources to Ocular Motoneurons
6.2.1. Via Autocrine
6.2.2. Via Paracrine
6.2.3. Retrograde Via
6.3. Characteristics of the Ocular Motor System after Axotomy
6.3.1. Regulation of Trophic Factors
6.3.2. Administration of Trophic Factors
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Senger, D.R.; Galli, S.J.; Dvorak, A.M.; Perruzzi, C.A.; Harvey, S.V.; Dvorak, H.F. Tumor cells secrete a vascular permeability factor that promotes accumulation of ascites fluid. Science 1983, 219, 983–985. [Google Scholar] [CrossRef]
- Ferrara, N.; Henzel, W.J. Pituitary follicular cells secrete a novel heparin-binding growth factor specific for vascular endothelial cells. Biochem. Biophys. Res. Commun. 1989, 161, 851–858. [Google Scholar] [CrossRef]
- Keck, P.J.; Hauser, S.D.; Krivi, G.; Sanzo, K.; Warren, T.; Feder, J.; Connolly, D.T. Vascular permeability factor, an endothelial cell mitogen related to PDGF. Science 1989, 246, 1309–1312. [Google Scholar] [CrossRef]
- Ferrara, N.; Gerber, H.-P.; LeCouter, J. The biology of VEGF and its receptors. Nat. Med. 2003, 9, 669–676. [Google Scholar] [CrossRef]
- Oosthuyse, B.; Moons, L.; Storkebaum, E.; Beck, H.; Nuyens, D.; Brusselmans, K.; Van Dorpe, J.; Hellings, P.; Gorselink, M.; Heymans, S.; et al. Deletion of the hypoxia-response element in the vascular endothelial growth factor promoter causes motor neuron degeneration. Nat. Genet. 2001, 28, 131–138. [Google Scholar] [CrossRef]
- Grimmond, S.; Lagercrantz, J.; Drinkwater, C.; Silins, G.; Townson, S.; Pollock, P.; Gotley, D.; Carson, E.; Rakar, S.; Nordenskjöld, M.; et al. Cloning and characterization of a novel human gene related to vascular endothelial growth factor. Genome Res. 1996, 6, 124–131. [Google Scholar] [CrossRef]
- Olofsson, B.; Pajusola, K.; Kaipainen, A.; Von Euler, G.; Joukov, V.; Saksela, O.; Orpana, A.; Pettersson, R.F.; Alitalo, K.; Eriksson, U. Vascular endothelial growth factor B, a novel growth factor for endothelial cells. Proc. Natl. Acad. Sci. USA 1996, 93, 2576–2581. [Google Scholar] [CrossRef]
- Joukov, V.; Pajusola, K.; Kaipainen, A.; Chilov, D.; Lahtinen, I.; Kukk, E.; Saksela, O.; Kalkkinen, N.; Alitalo, K. A novel vascular endothelial growth factor, VEGF-C, is a ligand for the Flt4 (VEGFR-3) and KDR (VEGFR-2) receptor tyrosine kinases. EMBO J. 1996, 15, 290–298. [Google Scholar] [CrossRef]
- Lee, J.; Gray, A.; Yuan, J.; Luoh, S.M.; Avraham, H.; Wood, W.I. Vascular endothelial growth factor-related protein: A ligand and specific activator of the tyrosine kinase receptor Flt4. Proc. Natl. Acad. Sci. USA 1996, 93, 1988–1992. [Google Scholar] [CrossRef]
- Orlandini, M.; Marconcini, L.; Ferruzzi, R.; Oliviero, S. Identification of a c-fos-induced gene that is related to the platelet-derived growth factor/vascular endothelial growth factor family. Proc. Natl. Acad. Sci. USA 1996, 93, 11675–11680. [Google Scholar] [CrossRef]
- Yamada, Y.; Nezu, J.I.; Shimane, M.; Hirata, Y. Molecular cloning of a novel vascular endothelial growth factor, VEGF-D. Genomics 1997, 42, 483–488. [Google Scholar] [CrossRef]
- Ogawa, S.; Oku, A.; Sawano, A.; Yamaguchi, S.; Yazaki, Y.; Shibuya, M. A novel type of vascular endothelial growth factor, VEGF-E (NZ-7 VEGF), preferentially utilizes KDR/Flk-1 receptor and carries a potent mitotic activity without heparin-binding domain. J. Biol. Chem. 1998, 273, 31273–31282. [Google Scholar] [CrossRef] [PubMed]
- Yamazaki, Y.; Takani, K.; Atoda, H.; Morita, T. Snake venom vascular endothelial growth factors (VEGFs) exhibit potent activity through their specific recognition of KDR (VEGF Receptor 2). J. Biol. Chem. 2003, 278, 51985–51988. [Google Scholar] [CrossRef] [PubMed]
- Maglione, D.; Guerriero, V.; Viglietto, G.; Delli-Bovi, P.; Persico, M.G. Isolation of a human placenta cDNA coding for a protein related to the vascular permeability factor. Proc. Natl. Acad. Sci. USA 1991, 88, 9267–9271. [Google Scholar] [CrossRef] [PubMed]
- Sathasivam, S. VEGF and ALS. Neurosci. Res. 2008, 62, 71–77. [Google Scholar] [CrossRef] [PubMed]
- Jin, K.; Zhu, Y.; Sun, Y.; Mao, X.O.; Xie, L.; Greenberg, D.A. Vascular endothelial growth factor (VEGF) stimulates neurogenesis in vitro and in vivo. Proc. Natl. Acad. Sci. USA 2002, 99, 11946–11950. [Google Scholar] [CrossRef]
- Lambrechts, D.; Storkebaum, E.; Morimoto, M.; Del-Favero, J.; Desmet, F.; Marklund, S.L.; Wyns, S.; Thijs, V.; Andersson, J.; Van Marion, I.; et al. VEGF is a modifier of amyotrophic lateral sclerosis in mice and humans and protects motoneurons against ischemic death. Nat. Genet. 2003, 34, 383–394. [Google Scholar] [CrossRef]
- Li, X.; Tjwa, M.; Van Hove, I.; Enholm, B.; Neven, E.; Paavonen, K.; Juan, T.D.; Sievers, R.E.; Chorianopoulos, E.; Wada, H.; et al. Reevaluation of the role of VEGF-B suggests a restricted role in the revascularization of the ischemic myocardium. Arter. Thromb. Vasc. Biol. 2008, 28, 1614–1620. [Google Scholar] [CrossRef]
- Poesen, K.; Lambrechts, D.; Van Damme, P.; Dhondt, J.; Bender, F.; Frank, N.; Bogaert, E.; Claes, B.; Heylen, L.; Verheyen, A.; et al. Novel role for vascular endothelial growth factor (VEGF) receptor-1 and its ligand VEGF-B in motor neuron degeneration. J. Neurosci. 2008, 28, 10451–10459. [Google Scholar] [CrossRef]
- Carmeliet, P.; Ruiz de Almodovar, C. VEGF ligands and receptors: Implications in neurodevelopment and neurodegeneration. Cell. Mol. Life Sci. 2013, 70, 1763–1778. [Google Scholar] [CrossRef]
- Shibuya, M. Vascular endothelial growth factor (VEGF) and its receptor (VEGFR) signaling in angiogenesis: A crucial target for anti- and pro-angiogenic therapies. Genes Cancer 2011, 2, 1097–1105. [Google Scholar] [CrossRef] [PubMed]
- Ogunshola, O.O.; Antic, A.; Donoghue, M.J.; Fan, S.Y.; Kim, H.; Stewart, W.B.; Madri, J.A.; Ment, L.R. Paracrine and autocrine functions of neuronal vascular endothelial growth factor (VEGF) in the central nervous system. J. Biol. Chem. 2002, 277, 11410–11415. [Google Scholar] [CrossRef] [PubMed]
- Van Den Bosch, L.; Storkebaum, E.; Vleminckx, V.; Moons, L.; Vanopdenbosch, L.; Scheveneels, W.; Carmeliet, P.; Robberecht, W. Effects of vascular endothelial growth factor (VEGF) on motor neuron degeneration. Neurobiol. Dis. 2004, 17, 21–28. [Google Scholar] [CrossRef]
- Ijichi, A.; Sakuma, S.; Tofilon, P.J. Hypoxia-induced vascular endothelial growth factor expression in normal rat astrocyte cultures. Glia 1995, 14, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Bartholdi, D.; Rubin, B.P.; Schwab, M.E. VEGF mRNA induction correlates with changes in the vascular architecture upon spinal cord damage in the rat. Eur. J. Neurosci. 1997, 9, 2549–2560. [Google Scholar] [CrossRef] [PubMed]
- Silverman, W.F.; Krum, J.M.; Mani, N.; Rosenstein, J.M. Vascular, glial and neuronal effects of vascular endothelial growth factor in mesencephalic explant cultures. Neuroscience 1999, 90, 1529–1541. [Google Scholar] [CrossRef]
- Sondell, M.; Lundborg, G.; Kanje, M. Vascular endothelial growth factor stimulates Schwann cell invasion and neovascularization of acellular nerve grafts. Brain Res. 1999, 846, 219–228. [Google Scholar] [CrossRef]
- Sondell, M.; Lundborg, G.; Kanje, M. Vascular endothelial growth factor has neurotrophic activity and stimulates axonal outgrowth, enhancing cell survival and Schwann cell proliferation in the peripheral nervous system. J. Neurosci. 1999, 19, 5731–5740. [Google Scholar] [CrossRef]
- Sondell, M.; Sundler, F.; Kanje, M. Vascular endothelial growth factor is a neurotrophic factor which stimulates axonal outgrowth through the flk-1 receptor. Eur. J. Neurosci. 2000, 12, 4243–4254. [Google Scholar] [CrossRef]
- Jin, K.L.; Mao, X.O.; Greenberg, D.A. Vascular endothelial growth factor: Direct neuroprotective effect in in vitro ischemia. Proc. Natl. Acad. Sci. USA 2000, 97, 10242–10247. [Google Scholar] [CrossRef]
- Svensson, B.; Peters, M.; König, H.G.; Poppe, M.; Levkau, B.; Rothermundt, M.; Arolt, V.; Kögel, D.; Prehn, J.H.M. Vascular endothelial growth factor protects cultured rat hippocampal neurons against hypoxic injury via an antiexcitotoxic, caspase-independent mechanism. J. Cereb. Blood Flow Metab. 2002, 22, 1170–1175. [Google Scholar] [CrossRef]
- Wick, A.; Wick, W.; Waltenberger, J.; Weller, M.; Dichgans, J.; Schulz, J.B. Neuroprotection by hypoxic preconditioning requires sequential activation of vascular endothelial growth factor receptor and Akt. J. Neurosci. 2002, 22, 6401–6407. [Google Scholar] [CrossRef]
- Licht, T.; Goshen, I.; Avital, A.; Kreisel, T.; Zubedat, S.; Eavri, R.; Segal, M.; Yirmiya, R.; Keshet, E. Reversible modulations of neuronal plasticity by VEGF. Proc. Natl. Acad. Sci. USA 2011, 108, 5081–5086. [Google Scholar] [CrossRef]
- Ruiz de Almodovar, C.; Lambrechts, D.; Mazzone, M.; Carmeliet, P. Role and therapeutic potential of VEGF in the nervous system. Physiol. Rev. 2009, 89, 607–648. [Google Scholar] [CrossRef]
- Li, B.; Xu, W.; Luo, C.; Gozal, D.; Liu, R. VEGF-induced activation of the PI3-K/Akt pathway reduces mutant SOD1-mediated motor neuron cell death. Mol. Brain Res. 2003, 111, 155–164. [Google Scholar] [CrossRef]
- Tovar-y-Romo, L.B.; Zepeda, A.; Tapia, R. Vascular endothelial growth factor prevents paralysis and motoneuron death in a rat model of excitotoxic spinal cord neurodegeneration. J. Neuropathol. Exp. Neurol. 2007, 66, 913–922. [Google Scholar] [CrossRef]
- Tolosa, L.; Mir, M.; Asensio, V.J.; Olmos, G.; Lladó, J. Vascular endothelial growth factor protects spinal cord motoneurons against glutamate-induced excitotoxicity via phosphatidylinositol 3-kinase. J. Neurochem. 2008, 105, 1080–1090. [Google Scholar] [CrossRef]
- Tovar-y-Romo, L.B.; Tapia, R. VEGF protects spinal motor neurons against chronic excitotoxic degeneration in vivo by activation of PI3-K pathway and inhibition of p38MAPK. J. Neurochem. 2010, 115, 1090–1101. [Google Scholar] [CrossRef]
- Ruiz de Almodovar, C.; Fabre, P.J.; Knevels, E.; Coulon, C.; Segura, I.; Haddick, P.C.G.; Aerts, L.; Delattin, N.; Strasser, G.; Oh, W.-J.; et al. VEGF mediates commissural axon chemoattraction through its receptor Flk1. Neuron 2011, 70, 966–978. [Google Scholar] [CrossRef]
- Brockington, A.; Wharton, S.B.; Fernando, M.; Gelsthorpe, C.H.; Baxter, L.; Ince, P.G.; Lewis, C.E.; Shaw, P.J. Expression of vascular endothelial growth factor and its receptors in the central nervous system in amyotrophic lateral sclerosis. J. Neuropathol. Exp. Neurol. 2006, 65, 26–36. [Google Scholar] [CrossRef]
- Folkman, J.; Shing, Y. Angiogenesis. J. Biol. Chem. 1992, 267, 10931–10934. [Google Scholar] [CrossRef]
- Pertovaara, L.; Kaipainen, A.; Mustonen, T.; Orpana, A.; Ferrara, N.; Saksela, O.; Alitalo, K. Vascular endothelial growth factor is induced in response to transforming growth factor-β in fibroblastic and epithelial cells. J. Biol. Chem. 1994, 269, 6271–6274. [Google Scholar] [CrossRef]
- Cohen, T.; Nahari, D.; Cerem, L.W.; Neufeld, G.; Levin, B.Z. Interleukin 6 induces the expression of vascular endothelial growth factor. J. Biol. Chem. 1996, 271, 736–741. [Google Scholar] [CrossRef] [PubMed]
- Ryuto, M.; Ono, M.; Izumi, H.; Yoshida, S.; Weich, H.A.; Kohno, K.; Kuwano, M. Induction of vascular endothelial growth factor by tumor necrosis factor α in human glioma cells: Possible roles of SP-1. J. Biol. Chem. 1996, 271, 28220–28228. [Google Scholar] [CrossRef]
- Deroanne, C.F.; Hajitou, A.; Calberg-Bacq, C.M.; Nusgens, B.V.; Lapière, C.M. Angiogenesis by fibroblast growth factor 4 is mediated through an autocrine up-regulation of vascular endothelial growth factor expression. Cancer Res. 1997, 57, 5590–5597. [Google Scholar]
- Kroll, J.; Waltenberger, J. A novel function of VEGF receptor-2 (KDR): Rapid release of nitric oxide in response to VEGF-A stimulation in endothelial cells. Biochem. Biophys. Res. Commun. 1999, 265, 636–639. [Google Scholar] [CrossRef]
- Shen, B.Q.; Lee, D.Y.; Zioncheck, T.F. Vascular endothelial growth factor governs endothelial nitric-oxide synthase expression via a KDR/Flk-1 receptor and a protein kinase C signaling pathway. J. Biol. Chem. 1999, 274, 33057–33063. [Google Scholar] [CrossRef]
- Richard, D.E.; Berra, E.; Pouysségur, J. Angiogenesis: How a tumor adapts to hypoxia. Biochem. Biophys. Res. Commun. 1999, 266, 718–722. [Google Scholar] [CrossRef]
- Stein, I.; Neeman, M.; Shweiki, D.; Itin, A.; Keshet, E. Stabilization of vascular endothelial growth factor mRNA by hypoxia and hypoglycemia and coregulation with other ischemia-induced genes. Mol. Cell. Biol. 1995, 15, 5363–5368. [Google Scholar] [CrossRef]
- Ikeda, E.; Achen, M.G.; Breier, G.; Risau, W. Hypoxia-induced transcriptional activation and increased mRNA stability of vascular endothelial growth factor in C6 glioma cells. J. Biol. Chem. 1995, 270, 19761–19766. [Google Scholar] [CrossRef]
- Forsythe, J.A.; Jiang, B.H.; Iyer, N.V.; Agani, F.; Leung, S.W.; Koos, R.D.; Semenza, G.L. Activation of vascular endothelial growth factor gene transcription by hypoxia-inducible factor 1. Mol. Cell. Biol. 1996, 16, 4604–4613. [Google Scholar] [CrossRef]
- Semenza, G.L. Hypoxia-inducible factor 1: Master regulator of O2 homeostasis. Curr. Opin. Genet. Dev. 1998, 8, 588–594. [Google Scholar] [CrossRef]
- Kaelin, W.G. HIF2 inhibitor joins the kidney cancer armamentarium. J. Clin. Oncol. 2018, 36, 908–910. [Google Scholar] [CrossRef]
- Levy, A.P.; Levy, N.S.; Wegner, S.; Goldberg, M.A. Transcriptional regulation of the rat vascular endothelial growth factor gene by hypoxia. J. Biol. Chem. 1995, 270, 13333–13340. [Google Scholar] [CrossRef]
- Liu, Y.; Cox, S.R.; Morita, T.; Kourembanas, S. Hypoxia regulates vascular endothelial growth factor gene expression in endothelial cells: Identification of a 5’ enhancer. Circ. Res. 1995, 77, 638–643. [Google Scholar] [CrossRef]
- Semenza, G.L.; Prabhakar, N.R. The role of hypoxia-inducible factors in carotid body (patho) physiology. J. Physiol. 2018, 596, 2977–2983. [Google Scholar] [CrossRef]
- Ratcliffe, P.J.; Ebert, B.L.; Ferguson, D.J.P.; Firth, J.D.; Gleadle, J.M.; Maxwell, P.H.; Pugh, C.W. State of the art lecture: Regulation of the erythropoietin gene. Nephrol. Dial. Transplant. 1995, 10, 18–27. [Google Scholar] [CrossRef]
- Semenza, G.L. Expression of hypoxia-inducible factor 1: Mechanisms and consequences. Biochem. Pharmacol. 2000, 59, 47–53. [Google Scholar] [CrossRef]
- Wenger, R.H. Mammalian oxygen sensing, signalling and gene regulation. J. Exp. Biol. 2000, 203, 1253–1263. [Google Scholar]
- Kolodkin, A.L.; Levengood, D.V.; Rowe, E.G.; Tai, Y.T.; Giger, R.J.; Ginty, D.D. Neuropilin is a semaphorin III receptor. Cell 1997, 90, 753–762. [Google Scholar] [CrossRef]
- Giger, R.J.; Urquhart, E.R.; Gillespie, S.K.H.; Levengood, D.V.; Ginty, D.D.; Kolodkin, A.L. Neuropilin-2 is a receptor for semaphorin IV: Insight into the structural basis of receptor function and specificity. Neuron 1998, 21, 1079–1092. [Google Scholar] [CrossRef]
- Whitaker, G.B.; Limberg, B.J.; Rosenbaum, J.S. Vascular endothelial growth factor receptor-2 and neuropilin-1 form a receptor complex that is responsible for the differential signaling potency of VEGF165 and VEGF121. J. Biol. Chem. 2001, 276, 25520–25531. [Google Scholar] [CrossRef] [PubMed]
- Geretti, E.; Shimizu, A.; Klagsbrun, M. Neuropilin structure governs VEGF and semaphorin binding and regulates angiogenesis. Angiogenesis 2008, 11, 31–39. [Google Scholar] [CrossRef] [PubMed]
- Lladó, J.; Tolosa, L.; Olmos, G. Cellular and molecular mechanisms involved in the neuroprotective effects of VEGF on motoneurons. Front. Cell. Neurosci. 2013, 7, 181. [Google Scholar] [CrossRef] [PubMed]
- Autiero, M.; Waltenberger, J.; Communi, D.; Kranz, A.; Moons, L.; Lambrechts, D.; Kroll, J.; Plaisance, S.; De Mol, M.; Bono, F.; et al. Role of PlGF in the intra- and intermolecular cross talk between the VEGF receptors Flt1 and Flk1. Nat. Med. 2003, 9, 936–943. [Google Scholar] [CrossRef]
- Zachary, I. Neuroprotective role of vascular endothelial growth factor: Signalling mechanisms, biological function, and therapeutic potential. NeuroSignals 2005, 14, 207–221. [Google Scholar] [CrossRef]
- Storkebaum, E.; Lambrechts, D.; Dewerchin, M.; Moreno-Murciano, M.P.; Appelmans, S.; Oh, H.; Van Damme, P.; Rutten, B.; Man, W.Y.; De Mol, M.; et al. Treatment of motoneuron degeneration by intracerebroventricular delivery of VEGF in a rat model of ALS. Nat. Neurosci. 2005, 8, 85–92. [Google Scholar] [CrossRef]
- Bogaert, E.; Van Damme, P.; Van Den Bosch, L.; Robberecht, W. Vascular endothelial growth factor in amyotrophic lateral sclerosis and other neurodegenerative diseases. Muscle Nerve 2006, 34, 391–405. [Google Scholar] [CrossRef]
- Foster, F.M.; Traer, C.J.; Abraham, S.M.; Fry, M.J. The phosphoinositide (PI) 3-kinase family. J. Cell Sci. 2003, 116, 3037–3040. [Google Scholar] [CrossRef]
- Tolosa, L.; Mir, M.; Olmos, G.; Lladó, J. Vascular endothelial growth factor protects motoneurons from serum deprivation-induced cell death through phosphatidylinositol 3-kinase-mediated p38 mitogen-activated protein kinase inhibition. Neuroscience 2009, 158, 1348–1355. [Google Scholar] [CrossRef]
- Shiote, M.; Nagano, I.; Ilieva, H.; Murakami, T.; Narai, H.; Ohta, Y.; Nagata, T.; Shoji, M.; Abe, K. Reduction of a vascular endothelial growth factor receptor, fetal liver kinase-1, by antisense oligonucleotides induces motor neuron death in rat spinal cord exposed to hypoxia. Neuroscience 2005, 132, 175–182. [Google Scholar] [CrossRef] [PubMed]
- Boillée, S.; Vande Velde, C.; Cleveland, D.W. ALS: A disease of motor neurons and their nonneuronal neighbors. Neuron 2006, 52, 39–59. [Google Scholar] [CrossRef] [PubMed]
- Boillée, S.; Yamanaka, K.; Lobsiger, C.S.; Copeland, N.G.; Jenkins, N.A.; Kassiotis, G.; Kollias, G.; Cleveland, D.W. Onset and progression in inherited ALS determined by motor neurons and microglia. Science 2006, 312, 1389–1392. [Google Scholar] [CrossRef] [PubMed]
- Andersen, P.M.; Sims, K.B.; Xin, W.W.; Kiely, R.; O’Neill, G.; Ravits, J.; Pioro, E.; Harati, Y.; Brower, R.D.; Levine, J.S.; et al. Sixteen novel mutations in the Cu/Zn superoxide dismutase gene in amyotrophic lateral sclerosis: A decade of discoveries, defects and disputes. Amyotroph. Lateral Scler. Other Mot. Neuron Disord. 2003, 4, 62–73. [Google Scholar] [CrossRef] [PubMed]
- Gurney, M.E.; Pu, H.; Chiu, A.Y.; Dal Canto, M.C.; Polchow, C.Y.; Alexander, D.D.; Caliendo, J.; Hentati, A.; Kwon, Y.W.; Deng, H.X. Motor neuron degeneration in mice that express a human Cu,Zn superoxide dismutase mutation. Science 1994, 264, 1772–1775. [Google Scholar] [CrossRef] [PubMed]
- Lu, L.; Zheng, L.; Viera, L.; Suswam, E.; Li, Y.; Li, X.; Estévez, A.G.; King, P.H. Mutant Cu/Zn-superoxide dismutase associated with amyotrophic lateral sclerosis destabilizes vascular endothelial growth factor mRNA and downregulates its expression. J. Neurosci. 2007, 27, 7929–7938. [Google Scholar] [CrossRef]
- Dupuis, L.; Gonzalez de Aguilar, J.L.; Echaniz-Laguna, A.; Eschbach, J.; Rene, F.; Oudart, H.; Halter, B.; Huze, C.; Schaeffer, L.; Bouillaud, F.; et al. Muscle mitochondrial uncoupling dismantles neuromuscular junction and triggers distal degeneration of motor neurons. PLoS ONE 2009, 4, e5390. [Google Scholar] [CrossRef]
- Dadon-Nachum, M.; Melamed, E.; Offen, D. The “dying-back” phenomenon of motor neurons in ALS. J. Mol. Neurosci. 2011, 43, 470–477. [Google Scholar] [CrossRef]
- Rocha, M.C.; Pousinha, P.A.; Correia, A.M.; Sebastião, A.M.; Ribeiro, J.A. Early changes of neuromuscular transmission in the SOD1(G93A) mice model of ALS start long before motor symptoms onset. PLoS ONE 2013, 8, e73846. [Google Scholar] [CrossRef]
- Storkebaum, E.; Carmeliet, P. VEGF: A critical player in neurodegeneration. J. Clin. Investig. 2004, 113, 14–18. [Google Scholar] [CrossRef]
- Storkebaum, E.; Lambrechts, D.; Carmeliet, P. VEGF: Once regarded as a specific angiogenic factor, now implicated in neuroprotection. Bioessays 2004, 26, 943–954. [Google Scholar] [CrossRef] [PubMed]
- Azzouz, M.; Ralph, G.S.; Storkebaum, E.; Walmsley, L.E.; Mitrophanous, K.A.; Kingsman, S.M.; Carmeliet, P.; Mazarakis, N.D. VEGF delivery with retrogradely transported lentivector prolongs survival in a mouse ALS model. Nature 2004, 429, 413–417. [Google Scholar] [CrossRef] [PubMed]
- Hwang, D.H.; Lee, H.J.; Park, I.H.; Seok, J.I.; Kim, B.G.; Joo, I.S.; Kim, S.U. Intrathecal transplantation of human neural stem cells overexpressing VEGF provide behavioral improvement, disease onset delay and survival extension in transgenic ALS mice. Gene Ther. 2009, 16, 1234–1244. [Google Scholar] [CrossRef] [PubMed]
- Gould, T.W.; Oppenheim, R.W. Motor neuron trophic factors: Therapeutic use in ALS? Brain Res. Rev. 2011, 67, 1–39. [Google Scholar] [CrossRef]
- Rosenstein, J.M.; Mani, N.; Khaibullina, A.; Krum, J.M. Neurotrophic effects of vascular endothelial growth factor on organotypic cortical explants and primary cortical neurons. J. Neurosci. 2003, 23, 11036–11044. [Google Scholar] [CrossRef]
- Vande Velde, C.; Cleveland, D.W. VEGF: Multitasking in ALS. Nat. Neurosci. 2005, 8, 5–7. [Google Scholar] [CrossRef]
- Widenfalk, J.; Lipson, A.; Jubran, M.; Hofstetter, C.; Ebendal, T.; Cao, Y.; Olson, L. Vascular endothelial growth factor improves functional outcome and decreases secondary degeneration in experimental spinal cord contusion injury. Neuroscience 2003, 120, 951–960. [Google Scholar] [CrossRef]
- Lu, Y.; Tang, C.; Zhu, L.; Li, J.; Liang, H.; Zhang, J.; Xu, R. The overexpression of TDP-43 protein in the neuron and oligodendrocyte cells causes the progressive motor neuron degeneration in the SOD1 G93A transgenic mouse model of amyotrophic lateral sclerosis. Int. J. Biol. Sci. 2016, 12, 1140–1149. [Google Scholar] [CrossRef]
- Gerber, H.P.; Dixit, V.; Ferrara, N. Vascular endothelial growth factor induces expression of the antiapoptotic proteins Bcl-2 and A1 in vascular endothelial cells. J. Biol. Chem. 1998, 273, 13313–13316. [Google Scholar] [CrossRef]
- Corona, J.C.; Tapia, R. Ca2+-permeable AMPA receptors and intracellular Ca2+ determine motoneuron vulnerability in rat spinal cord in vivo. Neuropharmacology 2007, 52, 1219–1228. [Google Scholar] [CrossRef]
- Tapia, R.; Medina-Ceja, L.; Peña, F. On the relationship between extracellular glutamate, hyperexcitation and neurodegeneration, in vivo. Neurochem. Int. 1999, 34, 23–31. [Google Scholar] [CrossRef]
- Meldrum, B.S. Glutamate and glutamine in the brain glutamate as a neurotransmitter in the brain: Review of physiology and pathology. J. Nutr. 2018, 130, 1007S–1015S. [Google Scholar] [CrossRef] [PubMed]
- Lipton, S.A.; Rosenberg, P.A. Excitatory amino acids as a final common pathway for neurologic disorders. N. Engl. J. Med. 1994, 330, 613–622. [Google Scholar] [PubMed]
- Bettler, B.; Mulle, C. AMPA and kainate receptors. Neuropharmacology 1995, 34, 123–139. [Google Scholar] [CrossRef]
- Heath, P.R.; Shaw, P.J. Update on the glutamatergic neurotransmitter system and the role of excitotoxicity in amyotrophic lateral sclerosis. Muscle Nerve 2002, 26, 438–458. [Google Scholar] [CrossRef]
- Siesjo, B.K. Calcium-mediated processes in neuronal degeneration. Ann. N. Y. Acad. Sci. 1994, 747, 140–161. [Google Scholar] [CrossRef] [PubMed]
- Van Den Bosch, L.; Vandenberghe, W.; Klaassen, H.; Van Houtte, E.; Robberecht, W. Ca2+-permeable AMPA receptors and selective vulnerability of motor neurons. J. Neurol. Sci. 2000, 180, 29–34. [Google Scholar] [CrossRef]
- Van Damme, P.; Van Den Bosch, L.; Van Houtte, E.; Callewaert, G.; Robberecht, W. GluR2-dependent properties of AMPA receptors determine the selective vulnerability of motor neurons to excitotoxicity. J. Neurophysiol. 2002, 88, 1279–1287. [Google Scholar] [CrossRef]
- Van Damme, P.; Braeken, D.; Callewaert, G.; Robberecht, W.; Van Den Bosch, L. GluR2 deficiency accelerates motor neuron degeneration in a mouse model of amyotrophic lateral sclerosis. J. Neuropathol. Exp. Neurol. 2005, 64, 605–612. [Google Scholar] [CrossRef]
- Carriedo, S.G.; Sensi, S.L.; Yin, H.Z.; Weiss, J.H. AMPA exposures induce mitochondrial Ca(2+) overload and ROS generation in spinal motor neurons in vitro. J. Neurosci. 2000, 20, 240–250. [Google Scholar] [CrossRef]
- Rothstein, J.D.; Dykes-Hoberg, M.; Pardo, C.A.; Bristol, L.A.; Jin, L.; Kuncl, R.W.; Kanai, Y.; Hediger, M.A.; Wang, Y.; Schielke, J.P.; et al. Knockout of glutamate transporters reveals a major role for astroglial transport in excitotoxicity and clearance of glutamate. Neuron 1996, 16, 675–686. [Google Scholar] [CrossRef]
- Matsuzaki, H.; Tamatani, M.; Yamaguchi, A.; Namikawa, K.; Kiyama, H.; Vitek, M.P.; Mitsuda, N.; Tohyama, M. Vascular endothelial growth factor rescues hippocampal neurons from glutamate-induced toxicity: Signal transduction cascades. FASEB J. 2001, 15, 1218–1220. [Google Scholar] [CrossRef]
- Bogaert, E.; Van Damme, P.; Poesen, K.; Dhondt, J.; Hersmus, N.; Kiraly, D.; Scheveneels, W.; Robberecht, W.; Van Den Bosch, L. VEGF protects motor neurons against excitotoxicity by upregulation of GluR2. Neurobiol. Aging 2010, 31, 2185–2191. [Google Scholar] [CrossRef] [PubMed]
- Nimchinsky, E.A.; Young, W.G.; Yeung, G.; Shah, R.A.; Gordon, J.W.; Bloom, F.E.; Morrison, J.H.; Hof, P.R. Differential vulnerability of oculomotor, facial, and hypoglossal nuclei in G86R superoxide dismutase transgenic mice. J. Comp. Neurol. 2000, 416, 112–125. [Google Scholar] [CrossRef]
- Haenggeli, C.; Kato, A.C. Differential vulnerability of cranial motoneurons in mouse models with motor neuron degeneration. Neurosci. Lett. 2002, 335, 39–43. [Google Scholar] [CrossRef]
- DePaul, R.; Abbs, J.H.; Caligiuri, M.; Gracco, V.L.; Brooks, B.R. Hypoglossal, trigeminal, and facial motoneuron involvement in amyotrophic lateral sclerosis. Neurology 1988, 38, 281–283. [Google Scholar] [CrossRef] [PubMed]
- Brockington, A.; Ning, K.; Heath, P.R.; Wood, E.; Kirby, J.; Fusi, N.; Lawrence, N.; Wharton, S.B.; Ince, P.G.; Shaw, P.J. Unravelling the enigma of selective vulnerability in neurodegeneration: Motor neurons resistant to degeneration in ALS show distinct gene expression characteristics and decreased susceptibility to excitotoxicity. Acta Neuropathol. 2013, 125, 95–109. [Google Scholar] [CrossRef] [PubMed]
- Comley, L.; Allodi, I.; Nichterwitz, S.; Nizzardo, M.; Simone, C.; Corti, S.; Hedlund, E. Motor neurons with differential vulnerability to degeneration show distinct protein signatures in health and ALS. Neuroscience 2015, 291, 216–229. [Google Scholar] [CrossRef]
- Reiner, A.; Medina, L.; Figueredo-Cardenas, G.; Anfinson, S. Brainstem motoneuron pools that are selectively resistant in amyotrophic lateral sclerosis are preferentially enriched in parvalbumin: Evidence from monkey brainstem for a calcium-mediated mechanism in sporadic ALS. Exp. Neurol. 1995, 131, 239–250. [Google Scholar] [CrossRef]
- Tjust, A.E.; Brannstrom, T.; Pedrosa Domellof, F. Unaffected motor endplate occupancy in eye muscles of ALS G93A mouse model. Front. Biosci. 2012, 1547–1555. [Google Scholar]
- Liu, J.X.; Brännström, T.; Andersen, P.M.; Pedrosa-Domellöf, F. Different impact of ALS on laminin isoforms in human extraocular muscles versus limb muscles. Investig. Ophthalmol. Vis. Sci. 2011, 52, 4842–4852. [Google Scholar] [CrossRef]
- Alexianu, M.E.; Ho, B.; Mohamed, A.H.; La Bella, V.; Smith, R.G.; Appel, S.H. The role of calcium-binding proteins in selective motoneuron vulnerability in amyotrophic lateral sclerosis. Ann. Neurol. 1994, 36, 846–858. [Google Scholar] [CrossRef] [PubMed]
- Vanselow, B.K.; Keller, B.U. Calcium dynamics and buffering in oculomotor neurones from mouse that are particularly resistant during amyotrophic lateral sclerosis (ALS)-related motoneurone disease. J. Physiol. 2000, 525 Pt 2, 433–445. [Google Scholar] [CrossRef]
- Laslo, P.; Lipski, J.; Nicholson, L.F.; Miles, G.B.; Funk, G.D. Calcium binding proteins in motoneurons at low and high risk for degeneration in ALS. Neuroreport 2000, 11, 3305–3308. [Google Scholar] [CrossRef] [PubMed]
- Ince, P.; Stout, N.; Shaw, P.; Slade, J.; Hunziker, W.; Heizmann, C.W.; Baimbridge, K.G. Parvalbumin and calbindin D-28k in the human motor system and in motor neuron disease. Neuropathol. Appl. Neurobiol. 1993, 19, 291–299. [Google Scholar] [CrossRef] [PubMed]
- de la Cruz, R.R.; Pastor, A.M.; Martínez-Guijarro, F.J.; López-García, C.; Delgado-García, J.M. Localization of parvalbumin, calretinin, and calbindin D-28k in identified extraocular motoneurons and internuclear neurons of the cat. J. Comp. Neurol. 1998, 390, 377–391. [Google Scholar] [CrossRef]
- Beers, D.R.; Ho, B.K.; Siklós, L.; Alexianu, M.E.; Mosier, D.R.; Habib Mohamed, A.; Otsuka, Y.; Kozovska, M.E.; McAlhany, R.E.; Glenn Smith, R.; et al. Parvalbumin overexpression alters immune-mediated increases in intracellular calcium, and delays disease onset in a transgenic model of familial amyotrophic lateral sclerosis. J. Neurochem. 2001, 79, 499–509. [Google Scholar] [CrossRef] [PubMed]
- Van Den Bosch, L.; Schwaller, B.; Vleminckx, V.; Meijers, B.; Stork, S.; Ruehlicke, T.; Van Houtte, E.; Klaassen, H.; Celio, M.R.; Missiaen, L.; et al. Protective effect of parvalbumin on excitotoxic motor neuron death. Exp. Neurol. 2002, 174, 150–161. [Google Scholar] [CrossRef] [PubMed]
- Fukuoka, T.; Tokunaga, A.; Kondo, E.; Miki, K.; Tachibana, T.; Noguchi, K. Differential regulation of alpha- and beta-CGRP mRNAs within oculomotor, trochlear, abducens, and trigeminal motoneurons in response to axotomy. Mol. Brain Res. 1999, 63, 304–315. [Google Scholar] [CrossRef]
- Morcuende, S.; Matarredona, E.R.; Benítez-Temiño, B.; Muñoz-Hernández, R.; Pastor, A.M.; de la Cruz, R.R. Differential regulation of the expression of neurotrophin receptors in rat extraocular motoneurons after lesion. J. Comp. Neurol. 2011, 519, 2335–2352. [Google Scholar] [CrossRef]
- González-Forero, D.; de la Cruz, R.R.; Delgado-García, J.M.; Álvarez, F.J.; Pastor, A.M. Correlation between CGRP immunoreactivity and firing activity in cat abducens motoneurons. J. Comp. Neurol. 2002, 451, 201–212. [Google Scholar] [CrossRef] [PubMed]
- Allodi, I.; Comley, L.; Nichterwitz, S.; Nizzardo, M.; Simone, C.; Aguila Benitez, J.; Cao, M.; Corti, S.; Hedlund, E. Differential neuronal vulnerability identifies IGF-2 as a protective factor in ALS and SMA. Sci. Rep. 2016, 6, 25960. [Google Scholar] [CrossRef] [PubMed]
- Hedlund, E.; Karlsson, M.; Osborn, T.; Ludwig, W.; Isacson, O. Global gene expression profiling of somatic motor neuron populations with different vulnerability identify molecules and pathways of degeneration and protection. Brain 2010, 133, 2313–2330. [Google Scholar] [CrossRef] [PubMed]
- Hafezparast, M.; Klocke, R.; Ruhrberg, C.; Marquardt, A.; Ahmad-Annuar, A.; Bowen, S.; Lalli, G.; Witherden, A.S.; Hummerich, H.; Nicholson, S.; et al. Mutations in dynein link motor neuron degeneration to defects in retrograde transport. Science 2003, 300, 808–812. [Google Scholar] [CrossRef]
- Millecamps, S.; Robertson, J.; Lariviere, R.; Mallet, J.; Julien, J.P. Defective axonal transport of neurofilament proteins in neurons overexpressing peripherin. J. Neurochem. 2006, 98, 926–938. [Google Scholar] [CrossRef]
- Jiang, Y.M.; Yamamoto, M.; Tanaka, F.; Ishigaki, S.; Katsuno, M.; Adachi, H.; Niwa, J.I.; Doyu, M.; Yoshida, M.; Hashizume, Y.; et al. Gene expressions specifically detected in motor neurons (dynactin 1, early growth response 3, acetyl-CoA transporter, death receptor 5, and cyclin C) differentially correlate to pathologic markers in sporadic amyotrophic lateral sclerosis. J. Neuropathol. Exp. Neurol. 2007, 66, 617–627. [Google Scholar] [CrossRef]
- Collard, J.F.; Côté, F.; Julien, J.P. Defective axonal transport in a transgenic mouse model of amyotrophic lateral sclerosis. Nature 1995, 375, 61–64. [Google Scholar] [CrossRef]
- Oppenheim, R.; Haverkamp, L.; Prevette, D.; McManaman, J.; Appel, S. Reduction of naturally occurring motoneuron death in vivo by a target-derived neurotrophic factor. Science 1988, 240, 919–922. [Google Scholar] [CrossRef]
- Grieshammer, U.; Lewandoski, M.; Prevette, D.; Oppenheim, R.W.; Martin, G.R. Muscle-specific cell ablation conditional upon Cre-mediated DNA recombination in transgenic mice leads to massive spinal and cranial motoneuron loss. Dev. Biol. 1998, 197, 234–247. [Google Scholar] [CrossRef][Green Version]
- Gould, T.W.; Yonemura, S.; Oppenheim, R.W.; Ohmori, S.; Enomoto, H. The neurotrophic effects of glial cell line-derived neurotrophic factor on spinal motoneurons are restricted to fusimotor subtypes. J. Neurosci. 2008, 28, 2131–2146. [Google Scholar] [CrossRef]
- DiStefano, P.S.; Friedman, B.; Radziejewski, C.; Alexander, C.; Boland, P.; Schick, C.M.; Lindsay, R.M.; Wiegand, S.J. The neurotrophins BDNF, NT-3, and NGF display distinct patterns of retrograde axonal transport in peripheral and central neurons. Neuron 1992, 8, 983–993. [Google Scholar] [CrossRef]
- Koliatsos, V.E.; Crawford, T.O.; Price, D.L. Axotomy induces nerve growth factor receptor immunoreactivity in spinal motor neurons. Brain Res. 1991, 549, 297–304. [Google Scholar] [CrossRef]
- Connor, B.; Young, D.; Lawlor, P.; Gai, W.; Waldvogel, H.; Faull, R.L.M.; Dragunow, M. Trk receptor alterations in Alzheimer’s disease. Mol. Brain Res. 1996, 42, 1–17. [Google Scholar] [CrossRef]
- Canals, J.M.; Checa, N.; Marco, S.; Michels, A.; Pérez-Navarro, E.; Alberch, J. The neurotrophin receptors trkA, trkB and trkC are differentially regulated after excitotoxic lesion in rat striatum. Mol. Brain Res. 1999, 69, 242–248. [Google Scholar] [CrossRef]
- Duprey-Díaz, M.V.; Soto, I.; Blagburn, J.M.; Blanco, R.E. Changes in brain-derived neurotrophic factor and trkB receptor in the adult Rana pipiens retina and optic tectum after optic nerve injury. J. Comp. Neurol. 2002, 454, 456–469. [Google Scholar] [CrossRef]
- Koliatsos, V.E.; Clatterbuck, R.E.; Winslow, J.W.; Cayouette, M.H.; Prices, D.L. Evidence that brain-derived neurotrophic factor is a trophic factor for motor neurons in vivo. Neuron 1993, 10, 359–367. [Google Scholar] [CrossRef]
- Merlio, J.P.; Ernfors, P.; Jaber, M.; Persson, H. Molecular cloning of rat trkC and distribution of cells expressing messenger RNAs for members of the trk family in the rat central nervous system. Neuroscience 1992, 51, 513–532. [Google Scholar] [CrossRef]
- Henderson, C.E.; Camu, W.; Mettling, C.; Gouin, A.; Poulsen, K.; Karihaloo, M.; Ruilamas, J.; Evans, T.; McMahon, S.B.; Armanini, M.P.; et al. Neurotrophins promote motor neuron survival and are present in embryonic limb bud. Nature 1993, 363, 266–270. [Google Scholar] [CrossRef]
- Piehl, F.; Frisén, J.; Risling, M.; Hökfelt, T.; Cullheim, S. Increased trkB mRNA expression by axotomized motoneurones. Neuroreport 1994, 5, 697–700. [Google Scholar] [CrossRef]
- Davis-López de Carrizosa, M.A.; Morado-Díaz, C.J.; Morcuende, S.; de la Cruz, R.R.; Pastor, A.M. Nerve growth factor regulates the firing patterns and synaptic composition of motoneurons. J. Neurosci. 2010, 30, 8308–8319. [Google Scholar] [CrossRef]
- Benítez-Temiño, B.; Morcuende, S.; Mentis, G.Z.; de la Cruz, R.R.; Pastor, A.M. Expression of Trk receptors in the oculomotor system of the adult cat. J. Comp. Neurol. 2004, 473, 538–552. [Google Scholar] [CrossRef]
- Morcuende, S.; Muñoz-Hernández, R.; Benítez-Temiño, B.; Pastor, A.M.; de la Cruz, R.R. Neuroprotective effects of NGF, BDNF, NT-3 and GDNF on axotomized extraocular motoneurons in neonatal rats. Neuroscience 2013, 250, 31–48. [Google Scholar] [CrossRef] [PubMed]
- Silva-Hucha, S.; Hernández, R.G.; Benítez-Temiño, B.; Pastor, A.M.; de la Cruz, R.R.; Morcuende, S. Extraocular motoneurons of the adult rat show higher levels of vascular endothelial growth factor and its receptor Flk-1 than other cranial motoneurons. PLoS ONE 2017, 12, e0178616. [Google Scholar] [CrossRef] [PubMed]
- McCloskey, D.P.; Hintz, T.M.; Scharfman, H.E. Modulation of vascular endothelial growth factor (VEGF) expression in motor neurons and its electrophysiological effects. Brain Res. Bull. 2008, 76, 36–44. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Millauer, B.; Wizigmann-Voos, S.; Schnürch, H.; Martinez, R.; Møller, N.P.H.; Risau, W.; Ullrich, A. High affinity VEGF binding and developmental expression suggest Flk-1 as a major regulator of vasculogenesis and angiogenesis. Cell 1993, 72, 835–846. [Google Scholar] [CrossRef]
- Wang, Y.; Duan, W.; Wang, W.; Wen, D.; Liu, Y.; Liu, Y.; Li, Z.; Hu, H.; Lin, H.; Cui, C.; et al. scAAV9-VEGF prolongs the survival of transgenic ALS mice by promoting activation of M2 microglia and the PI3K/Akt pathway. Brain Res. 2016, 1648, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Pettersson, A.; Nagy, J.A.; Brown, L.F.; Sundberg, C.; Morgan, E.; Jungles, S.; Carter, R.; Krieger, J.E.; Manseau, E.J.; Harvey, V.S.; et al. Heterogeneity of the angiogenic response induced in different normal adult tissues by vascular permeability factor/vascular endothelial growth factor. Lab. Investig. 2000, 80, 99–115. [Google Scholar] [CrossRef]
- Hernández, R.G.; Calvo, P.M.; Blumer, R.; de la Cruz, R.R.; Pastor, A.M. Functional diversity of motoneurons in the oculomotor system. Proc. Natl. Acad. Sci. USA 2019, 116, 3837–3846. [Google Scholar] [CrossRef]
- Benítez-Temiño, B.; Davis-López de Carrizosa, M.A.; Morcuende, S.; Matarredona, E.R.; de la Cruz, R.R.; Pastor, A.M. Functional diversity of neurotrophin actions on the oculomotor system. Int. J. Mol. Sci. 2016, 17, 2016. [Google Scholar] [CrossRef]
- Ernfors, P.; Henschen, A.; Olson, L.; Persson, H. Expression of Nerve Growth Factor Receptor mRNA Is Developmentally Regulated and Increased after Axotomy in Rat Spinal Cord Motoneurons. Neuron 1989, 2, 1605–1613. [Google Scholar] [CrossRef]
- Silva-Hucha, S.; Carrero-Rojas, G.; de Sevilla, M.E.F.; Benítez-Temiño, B.; de Carrizosa, M.A.D.L.; Pastor, A.M.; Morcuende, S. Sources and lesion-induced changes of VEGF expression in brainstem motoneurons. Brain Struct. Funct. 2020, 225, 1033–1053. [Google Scholar] [CrossRef]
- Vijayalakshmi, K.; Ostwal, P.; Sumitha, R.; Shruthi, S.; Varghese, A.M.; Mishra, P.; Manohari, S.G.; Sagar, B.C.; Sathyaprabha, T.N.; Nalini, A.; et al. Role of VEGF and VEGFR2 receptor in reversal of ALS-CSF induced degeneration of NSC-34 motor neuron cell line. Mol. Neurobiol. 2015, 51, 995–1007. [Google Scholar] [CrossRef] [PubMed]
- Falkowska, A.; Gutowska, I.; Goschorska, M.; Nowacki, P.; Chlubek, D.; Baranowska-Bosiacka, I. Energy metabolism of the brain, including the cooperation between astrocytes and neurons, especially in the context of glycogen metabolism. Int. J. Mol. Sci. 2015, 16, 25959–25981. [Google Scholar] [CrossRef]
- Chung, W.S.; Clarke, L.E.; Wang, G.X.; Stafford, B.K.; Sher, A.; Chakraborty, C.; Joung, J.; Foo, L.C.; Thompson, A.; Chen, C.; et al. Astrocytes mediate synapse elimination through MEGF10 and MERTK pathways. Nature 2013, 504, 394–400. [Google Scholar] [CrossRef] [PubMed]
- Taylor, J.P.; Brown, R.H.; Cleveland, D.W. Decoding ALS: From genes to mechanism. Nature 2016, 539, 197–206. [Google Scholar] [CrossRef] [PubMed]
- Barbeito, L.H.; Pehar, M.; Cassina, P.; Vargas, M.R.; Peluffo, H.; Viera, L.; Estévez, A.G.; Beckman, J.S. A role for astrocytes in motor neuron loss in amyotrophic lateral sclerosis. Brain Res. Rev. 2004, 47, 263–274. [Google Scholar] [CrossRef]
- Saijo, K.; Glass, C.K. Microglial cell origin and phenotypes in health and disease. Nat. Rev. Immunol. 2011, 11, 775–787. [Google Scholar] [CrossRef]
- Schafer, D.P.; Lehrman, E.K.; Kautzman, A.G.; Koyama, R.; Mardinly, A.R.; Yamasaki, R.; Ransohoff, R.M.; Greenberg, M.E.; Barres, B.A.; Stevens, B. Microglia sculpt postnatal neural circuits in an activity and complement-dependent manner. Neuron 2012, 74, 691–705. [Google Scholar] [CrossRef]
- Perry, V.H.; Nicoll, J.A.R.; Holmes, C. Microglia in neurodegenerative disease. Nat. Rev. Neurol. 2010, 6, 193–201. [Google Scholar] [CrossRef]
- Kreutzberg, G.W. Microglia: A sensor for pathological events in the CNS. Trends Neurosci. 1996, 19, 312–318. [Google Scholar] [CrossRef]
- Krum, J.M.; Rosenstein, J.M. VEGF mRNA and its receptor flt-1 are expressed in reactive astrocytes following neural grafting and tumor cell implantation in the adult CNS. Exp. Neurol. 1998, 154, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Krum, J.M.; Mani, N.; Rosenstein, J.M. Roles of the endogenous VEGF receptors flt-1 and flk-1 in astroglial and vascular remodeling after brain injury. Exp. Neurol. 2008, 212, 108–117. [Google Scholar] [CrossRef] [PubMed]
- Lennmyr, F.; Ata, K.A.; Funa, K.; Olsoon, Y.; Terént, A. Expression of vascular endothelial growth factor (VEGF) and its receptors (Flt-1 and Flk-1) following permanent and transient occlusion of the middle cerebral artery in the rat. J. Neuropathol. Exp. Neurol. 1998, 57, 874–882. [Google Scholar] [CrossRef] [PubMed]
- Acosta, L.; Morcuende, S.; Silva-Hucha, S.; Pastor, A.M.; de la Cruz, R.R. Vascular endothelial growth factor (VEGF) prevents the downregulation of the cholinergic phenotype in axotomized motoneurons of the adult rat. Front. Mol. Neurosci. 2018, 11, 241. [Google Scholar] [CrossRef] [PubMed]
- Büttner, U.; Büttner-Ennever, J.A. Present concepts of oculomotor organization. Prog. Brain Res. 2006, 151, 1–42. [Google Scholar]
- Nijssen, J.; Comley, L.H.; Hedlund, E. Motor neuron vulnerability and resistance in amyotrophic lateral sclerosis. Acta Neuropathol. 2017, 133, 863–885. [Google Scholar] [CrossRef]
- Zimmermann, L.; Morado-Díaz, C.J.; Carrizosa, M.A.D.L.; de la Cruz, R.R.; May, P.J.; Streicher, J.; Pastor, A.M.; Blumer, R. Axons giving rise to the palisade endings of feline extraocular muscles display motor features. J. Neurosci. 2013, 33, 2784–2793. [Google Scholar] [CrossRef]
- Jacoby, J.; Ko, K.; Weiss, C.; Rushbrook, J.I. Systematic variation in myosin expression along extraocular muscle fibres of the adult rat. J. Muscle Res. Cell Motil. 1990, 11, 25–40. [Google Scholar] [CrossRef]
- Valdez, G.; Tapia, J.C.; Lichtman, J.W.; Fox, M.A.; Sanes, J.R. Shared resistance to aging and als in neuromuscular junctions of specific muscles. PLoS ONE 2012, 7, e34640. [Google Scholar] [CrossRef]
- Tjust, A.E.; Danielsson, A.; Andersen, P.M.; Brännström, T.; Domellöf, F.P. Impact of amyotrophic lateral sclerosis on slow tonic myofiber composition in human extraocular muscles. Investig. Ophthalmol. Vis. Sci. 2017, 58, 3708–3715. [Google Scholar] [CrossRef]
- Heerssen, H.M.; Segal, R.A. Location, location, location: A spatial view of neurotrophin signal transduction. Trends Neurosci. 2002, 25, 160–165. [Google Scholar] [CrossRef]
- Hernández, R.G.; Silva-Hucha, S.; Morcuende, S.; de la Cruz, R.R.; Pastor, A.M.; Benítez-Temiño, B. Extraocular motor system exhibits a higher expression of neurotrophins when compared with other brainstem motor systems. Front. Neurosci. 2017, 11, 399. [Google Scholar] [CrossRef] [PubMed]
- Calvo, P.M.; de la Cruz, R.R.; Pastor, A.M. Synaptic loss and firing alterations in Axotomized Motoneurons are restored by vascular endothelial growth factor (VEGF) and VEGF-B. Exp. Neurol. 2018, 304, 67–81. [Google Scholar] [CrossRef] [PubMed]
- Gao, L.; Zhou, S.; Cai, H.; Gong, Z.; Zang, D. VEGF levels in CSF and serum in mild ALS patients. J. Neurol. Sci. 2014, 346, 216–220. [Google Scholar] [CrossRef] [PubMed]
- Nicoletti, J.N.; Shah, S.K.; McCloskey, D.P.; Goodman, J.H.; Elkady, A.; Atassi, H.; Hylton, D.; Rudge, J.S.; Scharfman, H.E.; Croll, S.D. Vascular endothelial growth factor is up-regulated after status epilepticus and protects against seizure-induced neuronal loss in hippocampus. Neuroscience 2008, 151, 232–241. [Google Scholar] [CrossRef][Green Version]
- Sun, Y.; Jin, K.; Xie, L.; Childs, J.; Mao, X.O.; Logvinova, A.; Greenberg, D.A. VEGF-induced neuroprotection, neurogenesis, and angiogenesis after focal cerebral ischemia. J. Clin. Investig. 2003, 111, 1843–1851. [Google Scholar] [CrossRef]
- Zheng, C.; Nennesmo, I.; Fadeel, B.; Henter, J.I. Vascular endothelial growth factor prolongs survival in a transgenic mouse model of ALS. Ann. Neurol. 2004, 56, 564–567. [Google Scholar] [CrossRef]
- Krakora, D.; Mulcrone, P.; Meyer, M.; Lewis, C.; Bernau, K.; Gowing, G.; Zimprich, C.; Aebischer, P.; Svendsen, C.N.; Suzuki, M. Synergistic effects of GDNF and VEGF on lifespan and disease progression in a familial ALS rat model. Mol. Ther. 2013, 21, 1602–1610. [Google Scholar] [CrossRef]
- Kobayashi, N.R.; Bedard, A.M.; Hincke, M.T.; Tetzlaff, W. Increased expression of BDNF and trkB mRNA in rat facial motoneurons after axotomy. Eur. J. Neurosci. 1996, 8, 1018–1029. [Google Scholar] [CrossRef]
- Nagata, Y.; Okuya, M.; Watanabe, R.; Honda, M. Regional distribution of cholinergic neurons in human spinal cord transections in the patients with and without motor neuron disease. Brain Res. 1982, 244, 223–229. [Google Scholar] [CrossRef]
- Virgo, L.; de Belleroche, J.; Rossi, M.; Steiner, T.J. Characterisation of the distribution of choline acetyltransferase messenger RNA in human spinal cord and its depletion in motor neurone disease. J. Neurol. Sci. 1992, 112, 126–132. [Google Scholar] [CrossRef]
- Oda, Y.; Imai, S.; Nakanishi, I.; Ichikawa, T.; Deguchi, T. Immunohistochemical study on choline acetyltransferase in the spinal cord of patients with amyotrophic lateral sclerosis. Pathol. Int. 1995, 45, 933–939. [Google Scholar] [CrossRef] [PubMed]
- Kato, T. Choline acetyltransferase activities in single spinal motor neurons from patients with amyotrophic lateral sclerosis. J. Neurochem. 1989, 52, 636–640. [Google Scholar] [CrossRef]
- Oda, Y. Choline acetyltransferase: The structure, distribution and pathologic changes in the central nervous system. Pathol. Int. 1999, 49, 921–937. [Google Scholar] [CrossRef] [PubMed]
- Morcuende, S.; Benítez-Temiño, B.; Pecero, M.L.; Pastor, A.M.; de la Cruz, R.R. Abducens internuclear neurons depend on their target motoneurons for survival during early postnatal development. Exp. Neurol. 2005, 195, 244–256. [Google Scholar] [CrossRef]
- Davis-López de Carrizosa, M.A.; Morado-Díaz, C.J.; Tena, J.J.; Benítez-Temiño, B.; Pecero, M.L.; Morcuende, S.; de la Cruz, R.R.; Pastor, A.M. Complementary actions of BDNF and neurotrophin-3 on the firing patterns and synaptic composition of motoneurons. J. Neurosci. 2009, 29, 575–587. [Google Scholar] [CrossRef]
- de la Cruz, R.R.; Delgado-García, J.M.; Pastor, A.M. Discharge characteristics of axotomized abducens internuclear neurons in the adult cat. J. Comp. Neurol. 2000, 427, 391–404. [Google Scholar] [CrossRef]
- Pastor, A.M.; Delgado-García, J.M.; Martínez-Guijarro, F.J.; López-García, C.; de la Cruz, R.R. Response of abducens internuclear neurons to axotomy in the adult cat. J. Comp. Neurol. 2000, 427, 370–390. [Google Scholar] [CrossRef]
- de la Cruz, R.R.; Benítez-Temiño, B.; Pastor, A.M. Intrinsic determinants of synaptic phenotype: An experimental study of abducens internuclear neurons connecting with anomalous targets. Neuroscience 2002, 112, 759–771. [Google Scholar] [CrossRef]
- Lams, B.E.E.; Isacson, O.; Sofroniew, M.V. Loss of transmitter-associated enzyme staining following axotomy does not indicate death of brainstem cholinergic neurons. Brain Res. 1988, 475, 401–406. [Google Scholar] [CrossRef]
- Matsuura, J.; Ajiki, K.; Ichikawa, T.; Misawa, H. Changes of expression levels of choline acetyltransferase and vesicular acetylcholine transporter mRNAs after transection of the hypoglossal nerve in adult rats. Neurosci. Lett. 1997, 236, 95–98. [Google Scholar] [CrossRef]
- Wang, W.; Salvaterra, P.M.; Loera, S.; Chiu, A.Y. Brain-derived neurotrophic factor spares choline acetyltransferase mRNA following axotomy of motor neurons in vivo. J. Neurosci. Res. 1997, 47, 134–143. [Google Scholar] [CrossRef]
- Morado-Díaz, C.J.; Matarredona, E.R.; Morcuende, S.; Talaverón, R.; Davis-López de Carrizosa, M.A.; de la Cruz, R.R.; Pastor, A.M. Neural progenitor cell implants in the lesioned medial longitudinal fascicle of adult cats regulate synaptic composition and firing properties of abducens internuclear neurons. J. Neurosci. 2014, 34, 7007–7017. [Google Scholar] [CrossRef]
- Lambrechts, D.; Carmeliet, P. VEGF at the neurovascular interface: Therapeutic implications for motor neuron disease. Biochim. Biophys. Acta 2006, 1762, 1109–1121. [Google Scholar] [CrossRef] [PubMed]
- Tovar-y-Romo, L.B.; Ramírez-Jarquín, U.N.; Lazo-Gómez, R.; Tapia, R. Trophic factors as modulators of motor neuron physiology and survival: Implications for ALS therapy. Front. Cell. Neurosci. 2014, 8, 61. [Google Scholar] [CrossRef] [PubMed]
- Dodge, J.C.; Treleaven, C.M.; Fidler, J.A.; Hester, M.; Haidet, A.; Handy, C.; Rao, M.; Eagle, A.; Matthews, J.C.; Taksir, T.V.; et al. AAV4-mediated expression of IGF-1 and VEGF within cellular components of the ventricular system improves survival outcome in familial ALS mice. Mol. Ther. 2010, 18, 2075–2084. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Silva-Hucha, S.; Pastor, A.M.; Morcuende, S. Neuroprotective Effect of Vascular Endothelial Growth Factor on Motoneurons of the Oculomotor System. Int. J. Mol. Sci. 2021, 22, 814. https://doi.org/10.3390/ijms22020814
Silva-Hucha S, Pastor AM, Morcuende S. Neuroprotective Effect of Vascular Endothelial Growth Factor on Motoneurons of the Oculomotor System. International Journal of Molecular Sciences. 2021; 22(2):814. https://doi.org/10.3390/ijms22020814
Chicago/Turabian StyleSilva-Hucha, Silvia, Angel M. Pastor, and Sara Morcuende. 2021. "Neuroprotective Effect of Vascular Endothelial Growth Factor on Motoneurons of the Oculomotor System" International Journal of Molecular Sciences 22, no. 2: 814. https://doi.org/10.3390/ijms22020814
APA StyleSilva-Hucha, S., Pastor, A. M., & Morcuende, S. (2021). Neuroprotective Effect of Vascular Endothelial Growth Factor on Motoneurons of the Oculomotor System. International Journal of Molecular Sciences, 22(2), 814. https://doi.org/10.3390/ijms22020814




