Comparative Time-Course Physiological Responses and Proteomic Analysis of Melatonin Priming on Promoting Germination in Aged Oat (Avena sativa L.) Seeds
Abstract
1. Introduction
2. Results
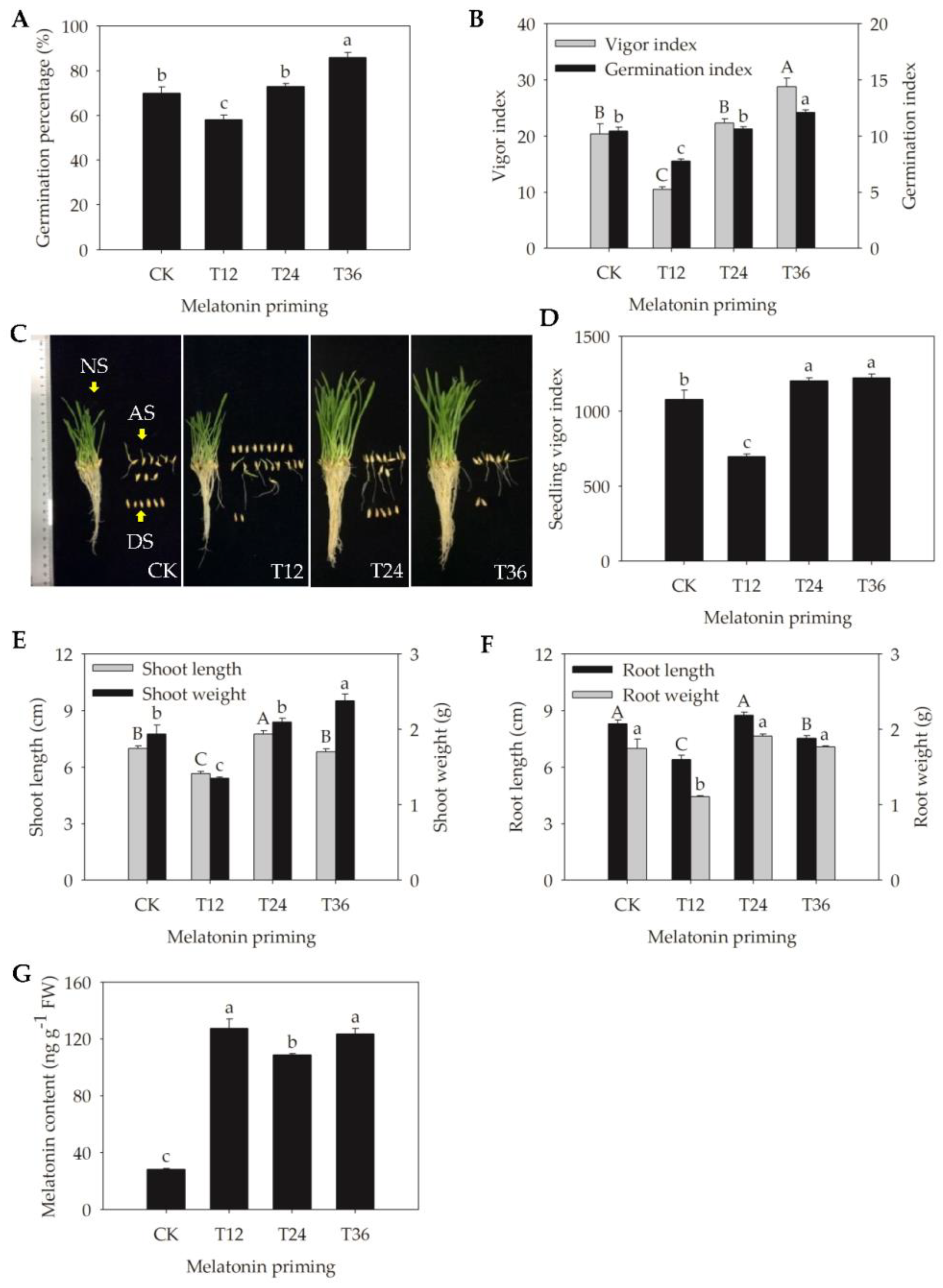
2.1. Germinability and Seedling Performance of Aged Oat Seeds Following Melatonin Priming
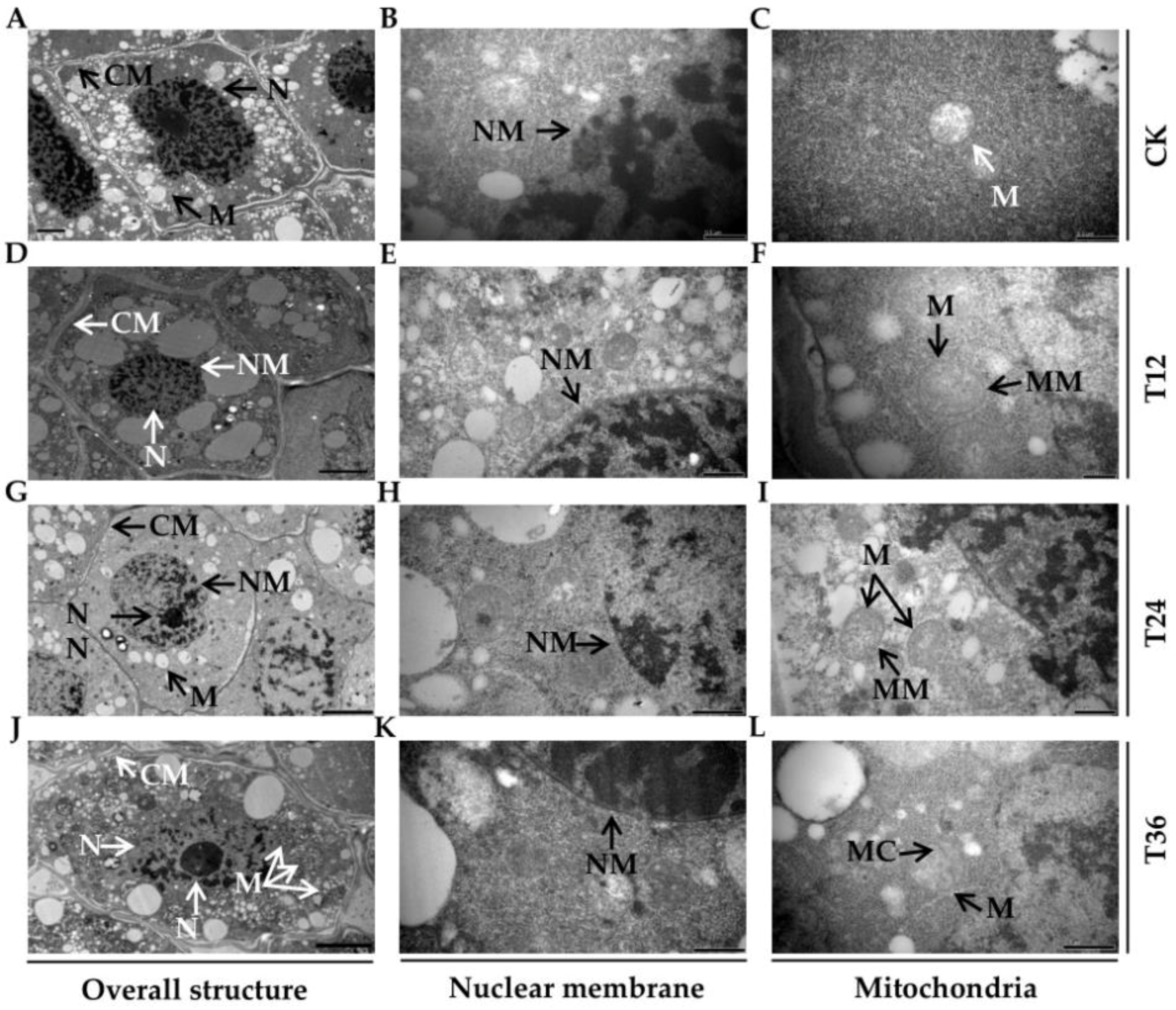
2.2. Ultrastructural Charicteristics of Embryos from Aged Oat Seeds Following Melatonin Priming
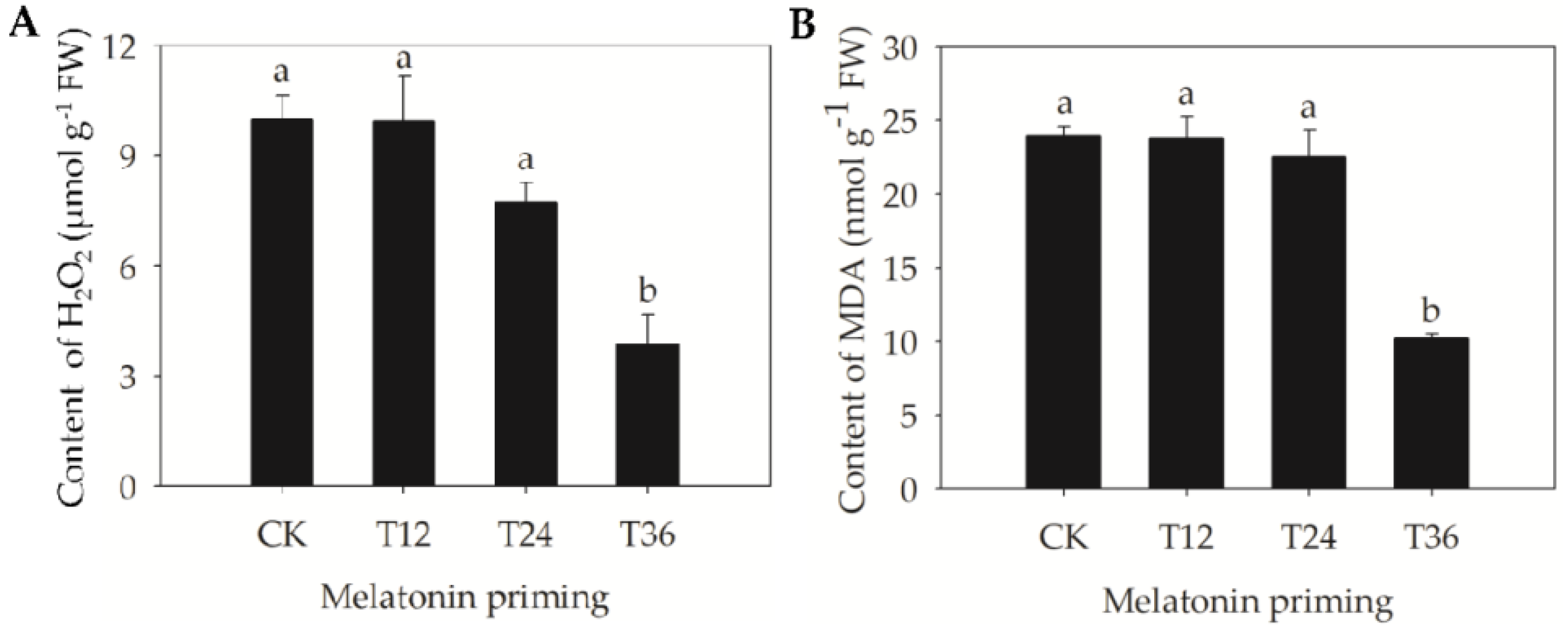
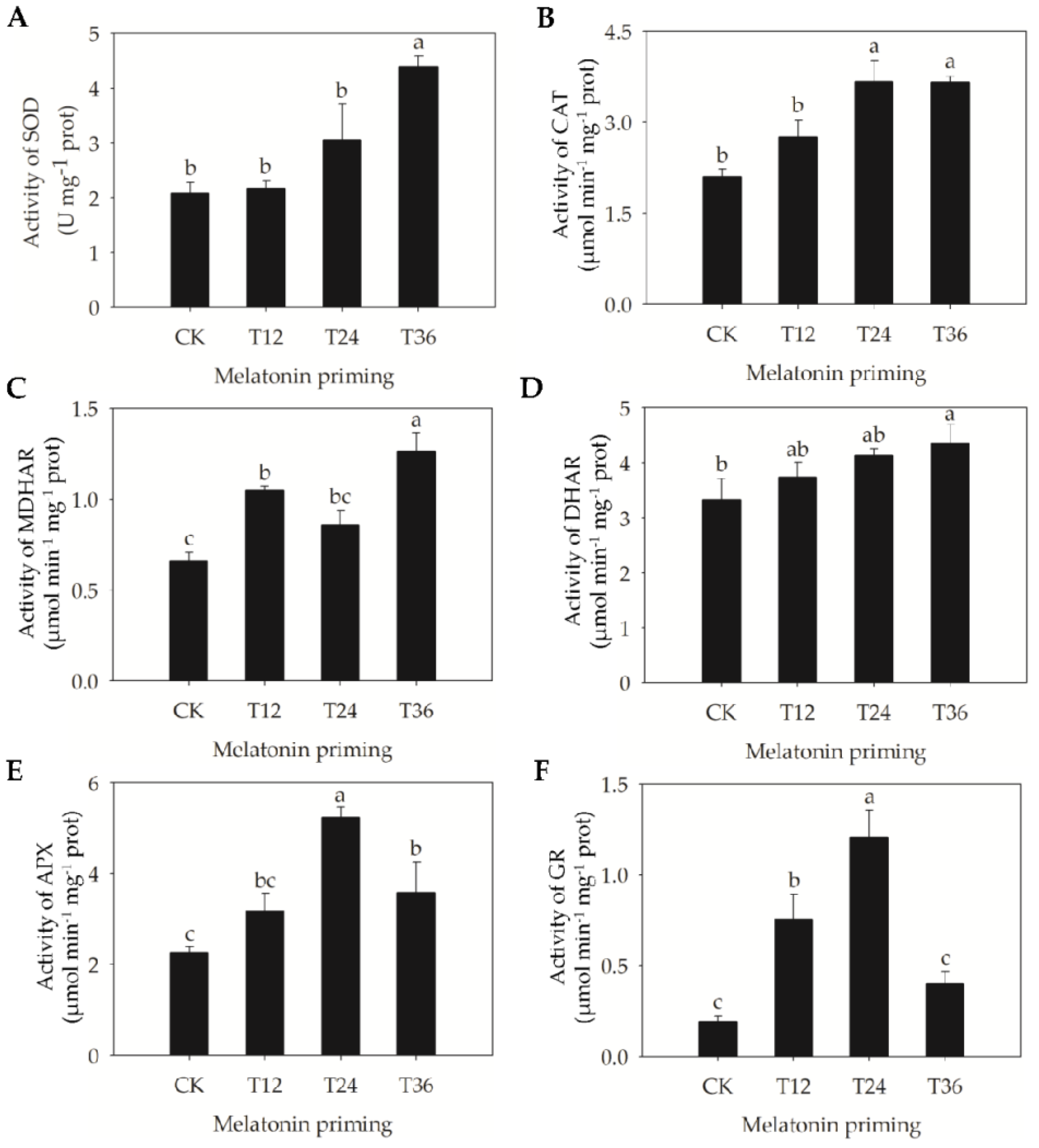
2.3. Changes of H2O2 Accumulation, Lipid Peroxidation, and Antioxidant Capacity in Aged Oat Seeds Following Melatonin Priming
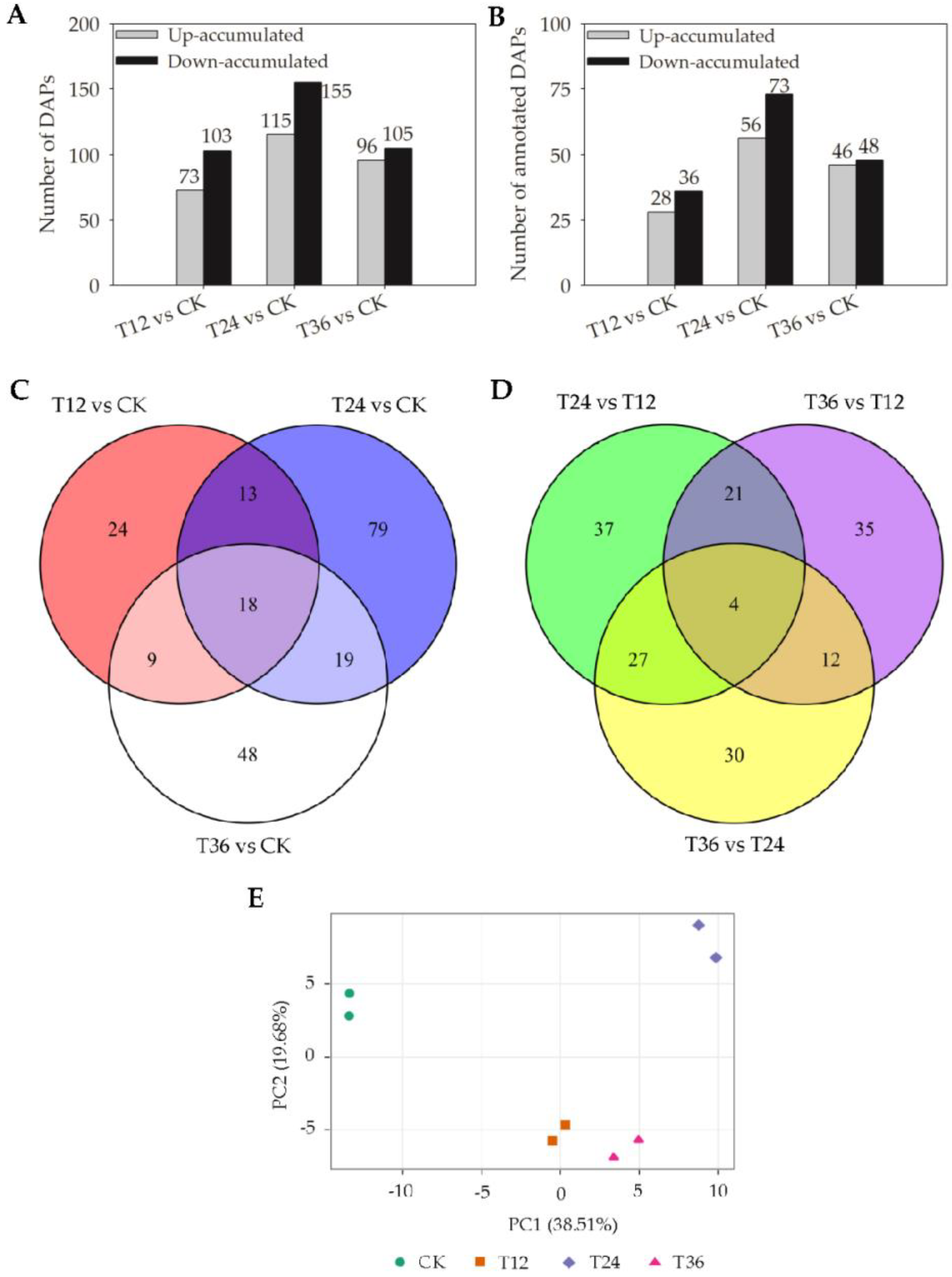
2.4. Quantitative Proteomic Analysis Profiles of Oat Seed Embryos Following Melatonin Priming
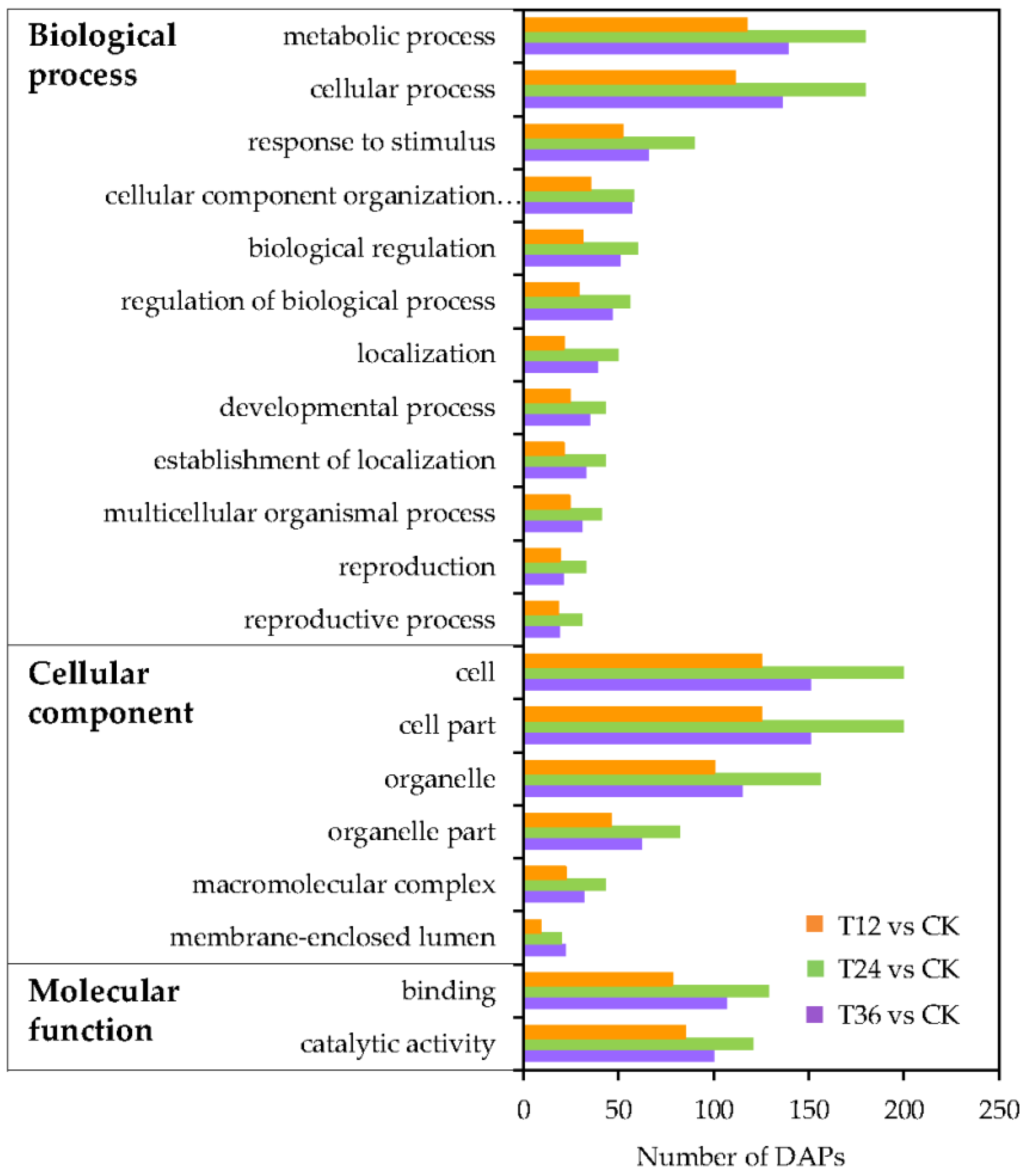
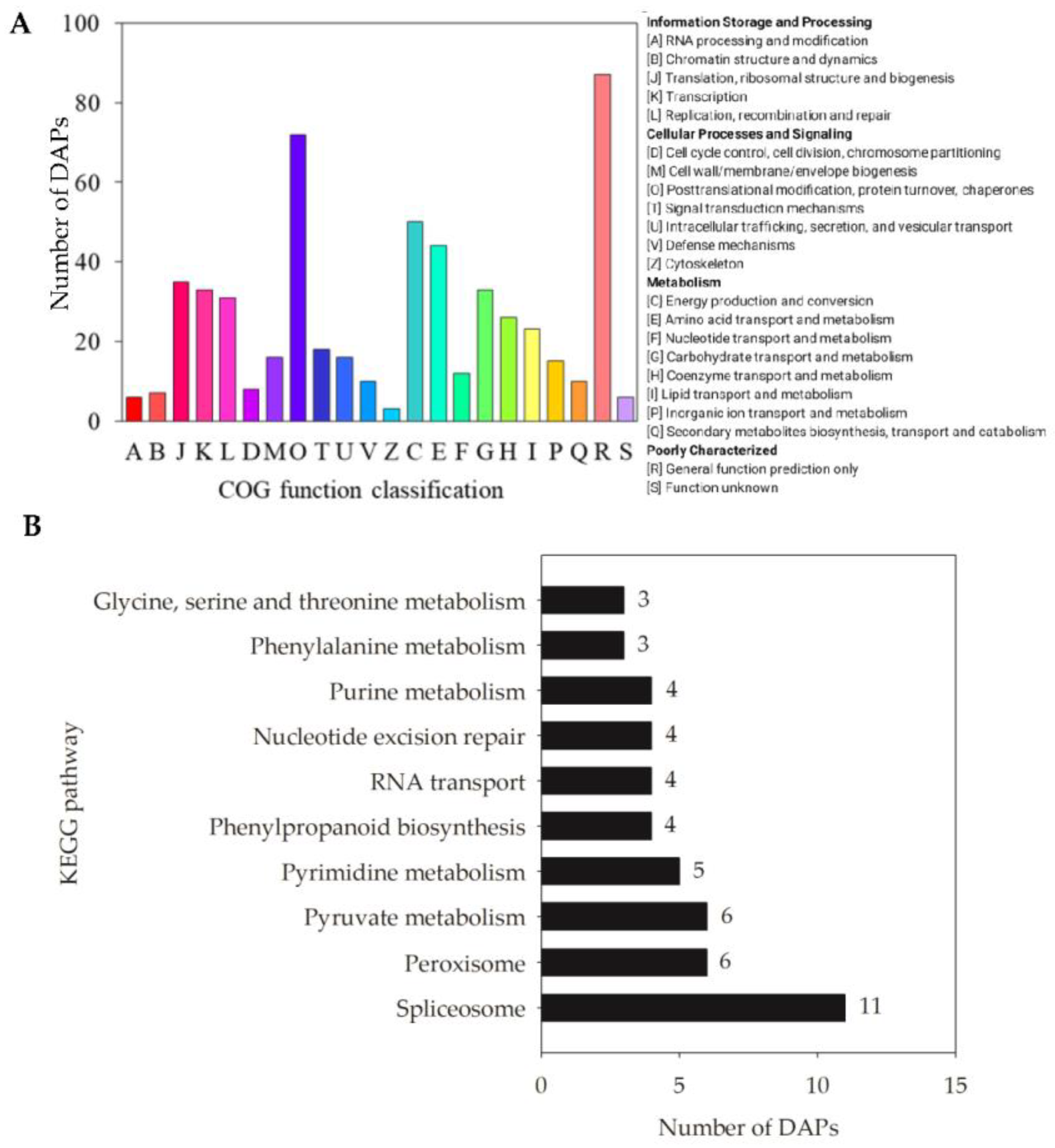
2.5. Functional Classification Analysis of DAPs in Oat Seed Embryos Following Melatonin Priming
2.6. Confirmation of DAPs by qRT-PCR at the Transcriptional Level
3. Discussion
3.1. DAPs Involved in Carbon Metabolism and Energy Production
3.2. DAPs Involved in Amino Acid Metabolism and Phenylpropanoid Biosynthesis
3.3. DAPs Involved in Phosphatidylinositol Signaling
3.4. DAPs Involved in Alpha-Linolenic Acid Metabolism
4. Materials and Methods
4.1. Seed Materials
4.2. Seed Aging and Melatonin Priming Treatments
4.3. Quantification of Endogenous Melatonin
4.4. Germination Test and Seedling Growth Assay
4.5. Ultrastructural Observation of Radicle Cells
4.6. Determination of H2O2 and MDA Contents
4.7. Assay of Antioxidant Enzymes
4.8. Extraction and Quantification of Embryo Proteins
4.9. Protein Reduction, Digestion, and iTRAQ Labeling
4.10. NanoLC-MS/MS Analysis
4.11. Database Search, Bioinformatic Analysis, Protein Annotation, and Functional Analysis
4.12. RNA Extraction and qRT-PCR
4.13. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ACO | Aconitate hydratase |
| APX | Ascorbate peroxidase |
| AsA–GSH | Ascorbate–glutathione |
| ATP-PFK | ATP-dependent 6-phosphofructokinase |
| CAT | Catalase |
| DHAR | Dehydroascorbate reductase |
| DLD | Dihydrolipoyl dehydrogenase |
| DLDH | D-lactate dehydrogenase |
| GAPDH | Glyceraldehyde-3-phosphate dehydrogenase |
| H2O2 | Hydrogen peroxide |
| HSDH | Homoserine dehydrogenase |
| iTRAQ | Isobaric tags for relative and absolute quantification |
| MDA | Malondialdehyde |
| MDHAR | Monodehydroascorbate reductase |
| OPR | 12-oxo-phytodienoic acid reductase |
| PAL | Phenylalanine ammonia-lyase |
| P5CS | δ-1-pyrroline-5-carboxylate synthase |
| PDC1 | Pyruvate decarboxylase 1 |
| PDDA | Phospho-2-dehydro-3-deoxyheptonate aldolase |
| PI-PLC | Phosphoinositide phospholipase C |
| PK | Pyruvate kinase |
| POD | Peroxidase |
| ROS | Reactive oxygen species |
| SHMT | Serine hydroxymethyltransferase |
| SOD | Superoxide dismutase |
| SuSy | Sucrose synthase |
| UDP-GlcDH | UDP-glucose 6-dehydrogenase |
References
- Xu, D.; Ren, G.Y.; Liu, L.L.; Zhu, W.X.; Liu, Y.H. The influences of drying process on crude protein content of naked oat cut herbage (Avena nuda L.). Dry Technol. 2014, 32, 321–332. [Google Scholar]
- Klose, C.; Arendt, E.K. Proteins in oats; their synthesis and changes during germination: A review. Crit. Rev. Food Sci. 2012, 52, 629–639. [Google Scholar] [CrossRef]
- Kumar, S.P.J.; Prasad, S.R.; Banerjee, R.; Thammineni, C. Seed birth to death: Dual functions of reactive oxygen species in seed physiology. Ann. Bot. 2015, 116, 663–668. [Google Scholar] [CrossRef]
- Wang, W.Q.; Liu, S.J.; Song, S.Q.; Møller, I.M. Proteomics of seed development, desiccation tolerance, germination and vigor. Plant Physiol. Biochem. 2015, 86, 1–15. [Google Scholar] [CrossRef]
- Wojtyla, L.; Lechowska, K.; Kubala, S.; Garnczarska, M. Different modes of hydrogen peroxide action during seed germination. Front. Plant Sci. 2016, 7, 66. [Google Scholar] [CrossRef]
- Xia, F.; Chen, L.; Sun, Y.; Mao, P. Relationships between ultrastructure of embryo cells and biochemical variations during ageing of oat (Avena sativa L.) seeds with different moisture content. Acta Physiol. Plant. 2015, 37, 89. [Google Scholar] [CrossRef]
- Mao, C.; Zhu, Y.; Cheng, H.; Yan, H.; Zhao, L.; Tang, J.; Ma, X.; Mao, P. Nitric oxide regulates seedling growth and mitochondrial responses in aged oat seeds. Int. J. Mol. Sci. 2018, 19, 1052. [Google Scholar] [CrossRef]
- Chen, L.; Chen, Q.; Kong, L.; Xia, F.; Yan, H.; Zhu, Y.; Mao, P. Proteomic and physiological analysis of the response of oat (Avena sativa) seeds to heat stress under different moisture conditions. Front. Plant Sci. 2016, 7, 896. [Google Scholar] [CrossRef]
- Yan, H.; Jia, S.; Mao, P. Melatonin priming alleviates aging-induced germination inhibition by regulating β-oxidation, protein translation and antioxidant metabolism in oat (Avena sativa L.) seeds. Int. J. Mol. Sci. 2020, 21, 1898. [Google Scholar] [CrossRef]
- Sheteiwy, M.S.; Guan, Y.; Cao, D.; Li, J.; Nawaz, A.; Hu, Q.; Hu, W.; Ning, M.; Hu, J. Seed priming with polyethylene glycol regulating the physiological and molecular mechanism in rice (Oryza sativa L.) under nano-ZnO stress. Sci. Rep. 2015, 5, 14278. [Google Scholar]
- Yan, H.F.; Mao, C.L.; Zhu, Y.Q.; Cheng, H.; Mao, P.S. Exogenous glutathione pre-treatment improves germination and resistance of Elymus sibiricus seeds subjected to different ageing conditions. Seed Sci. Technol. 2017, 45, 607–621. [Google Scholar] [CrossRef]
- Paparella, S.; Araújo, S.S.; Rossi, G.; Wijayasinghe, M.; Carbonera, D.; Balestrazzi, A. Seed priming: State of the art and new perspectives. Plant Cell Rep. 2015, 34, 1281–1293. [Google Scholar] [CrossRef]
- Xia, F.; Cheng, H.; Chen, L.; Zhu, H.; Mao, P.; Wang, M. Influence of exogenous ascorbic acid and glutathione priming on mitochondrial structural and functional systems to alleviate aging damage in oat seeds. BMC Plant Biol. 2020, 20, 104. [Google Scholar] [CrossRef]
- Nazari, R.; Parsa, S.; Afshari, R.T.; Mahmoodi, S.; Seyyedi, S.M. Salicylic acid priming before and after accelerated aging process increases seedling vigor in aged soybean seed. J. Crop Improv. 2020, 34, 218–237. [Google Scholar] [CrossRef]
- Siadat, S.A.; Moosavi, A.; Zadeh, M.S. Effects of seed priming on antioxidant activity and germination characteristics of maize seeds under different ageing treatment. Res. J. Seed Sci. 2012, 5, 51–62. [Google Scholar] [CrossRef]
- Krainart, C.; Siri, B.; Vichitphan, K. Effects of accelerated aging and subsequent priming on seed quality and biochemical change of hybrid cucumber (Cucumis sativa Linn.) seeds. Int. J. Agric. Technol. 2015, 11, 165–179. [Google Scholar]
- Kołodziejczyk, I.; Dzitko, K.; Szewczyk, R.; Posmyk, M.M. Exogenous melatonin improves corn (Zea mays L.) embryo proteome in seeds subjected to chilling stress. J. Plant Physiol. 2016, 193, 47–56. [Google Scholar] [CrossRef]
- Xiao, S.; Liu, L.; Wang, H.; Li, D.; Bai, Z.; Zhang, Y.; Sun, H.; Zhang, K.; Li, C. Exogenous melatonin accelerates seed germination in cotton (Gossypium hirsutum L.). PLoS ONE 2019, 14, e0216575. [Google Scholar] [CrossRef]
- Wang, P.; Sun, X.; Li, C.; Wei, Z.; Liang, D.; Ma, F. Long-term exogenous application of melatonin delays drought-induced leaf senescence in apple. J. Pineal Res. 2013, 54, 292–302. [Google Scholar] [CrossRef]
- Cao, Q.; Li, G.; Cui, Z.; Yang, F.; Jiang, X.; Diallo, L.; Kong, F. Seed priming with melatonin improves the seed germination of waxy maize under chilling stress via promoting the antioxidant system and starch metabolism. Sci. Rep. 2019, 9, 15044. [Google Scholar] [CrossRef]
- Li, J.; Zeng, L.; Cheng, Y.; Lu, G.; Fu, G.; Ma, H.; Liu, Q.; Zhang, X.; Zou, X.; Li, C. Exogenous melatonin alleviates damage from drought stress in Brassica napus L. (rapeseed) seedlings. Acta Physiol. Plant. 2018, 40, 43. [Google Scholar] [CrossRef]
- Cen, H.; Wang, T.; Liu, H.; Tian, D.; Zhang, Y. Melatonin application improves salt tolerance of alfalfa (Medicago sativa L.) by enhancing antioxidant capacity. Plants 2020, 9, 220. [Google Scholar] [CrossRef] [PubMed]
- Manchester, L.C.; Coto-Montes, A.; Boga, J.A.; Andersen, L.P.H.; Zhou, Z.; Galano, A.; Vriend, J.; Tan, D.X.; Reiter, R.J. Melatonin: An ancient molecule that makes oxygen metabolically tolerable. J. Pineal Res. 2015, 59, 403–419. [Google Scholar] [CrossRef] [PubMed]
- Arora, D.; Bhatla, S.C. Melatonin and nitric oxide regulate sunflower seedling growth under salt stress accompanying differential expression of Cu/Zn SOD and Mn SOD. Free Radic. Biol. Med. 2017, 106, 315–328. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Feng, C.; Zheng, X.; Guo, Y.; Zhou, F.; Shan, D.; Liu, X.; Kong, J. Plant mitochondria synthesize melatonin and enhance the tolerance of plants to drought stress. J. Pineal Res. 2017, 63, e12429. [Google Scholar] [CrossRef]
- Su, X.; Xin, L.; Li, Z.; Zheng, H.; Mao, J.; Yang, Q. Physiology and transcriptome analyses reveal a protective effect of the radical scavenger melatonin in aging maize seeds. Free Radical Res. 2018, 52, 1094–1109. [Google Scholar] [CrossRef]
- Liu, X.; Chen, Z.; Gao, Y.; Liu, Q.; Zhou, W.; Zhao, T.; Jiang, W.; Cui, X.; Cui, J.; Wang, Q. Combinative effects of Azospirillum brasilense inoculation and chemical priming on germination behavior and seedling growth in aged grass seeds. PLoS ONE 2019, 14, e0210453. [Google Scholar] [CrossRef]
- Bahcesular, B.; Yildirim, E.D.; Karacocuk, M.; Kulak, M.; Karaman, S. Seed priming with melatonin effects on growth, essential oil compounds and antioxidant activity of basil (Ocimum basilicum L.) under salinity stress. Ind. Crop. Prod. 2020, 146, 112165. [Google Scholar] [CrossRef]
- Cui, G.; Sun, F.; Gao, X.; Xie, K.; Zhang, C.; Liu, S.; Xi, Y. Proteomic analysis of melatonin-mediated osmotic tolerance by improving energy metabolism and autophagy in wheat (Triticum aestivum L.). Planta 2018, 248, 69–87. [Google Scholar] [CrossRef]
- Szafrańska, K.; Glińska, S.; Janas, K.M. Ameliorative effect of melatonin on meristematic cells of chilled and re-warmed Vigna radiata roots. Biol. Plantarum 2013, 57, 91–96. [Google Scholar] [CrossRef]
- Bailly, C. Active oxygen species and antioxidants in seed biology. Seed Sci. Res. 2004, 14, 93–107. [Google Scholar] [CrossRef]
- Sharma, S.N.; Maheshwari, A.; Sharma, C.; Shukla, N. Gene expression patterns regulating the seed metabolism in relation to deterioration/ageing of primed mung bean (Vigna radiata L.) seeds. Plant Physiol. Biochem. 2018, 124, 40–49. [Google Scholar] [CrossRef] [PubMed]
- Lv, Y.; Zhang, S.; Wang, J.; Hu, Y. Quantitative proteomic analysis of wheat seeds during artificial ageing and priming using the isobaric tandem mass tag labeling. PLoS ONE 2016, 11, e0162851. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.J.; Xu, H.H.; Wang, W.Q.; Li, N.; Wang, W.P.; Møller, I.M.; Song, S.Q. A proteomic analysis of rice seed germination as affected by high temperature and ABA treatment. Physiol. Plantarum 2015, 154, 142–161. [Google Scholar] [CrossRef]
- Yang, M.; Geng, M.; Shen, P.; Chen, X.; Li, Y.; Wen, X. Effect of post-silking drought stress on the expression profiles of genes involved incarbon and nitrogen metabolism during leaf senescence in maize (Zea mays L.). Plant Physiol. Biochem. 2019, 135, 304–309. [Google Scholar] [CrossRef]
- Kärkönen, A.; Murigneux, A.; Martinant, J.P.; Pepey, E.; Tatout, C.; Dudley, B.J.; Fry, S.C. UDP-glucose dehydrogenases of maize: A role in cell wall pentose biosynthesis. Biochem. J. 2005, 391, 409–415. [Google Scholar] [CrossRef]
- Wang, L.; Ma, H.; Song, L.; Shu, Y.; Gu, W. Comparative proteomics analysis reveals the mechanism of pre-harvest seed deterioration of soybean under high temperature and humidity stress. J. Proteomics 2012, 75, 2109–2127. [Google Scholar] [CrossRef]
- Dong, K.; Zhen, S.; Cheng, Z.; Cao, H.; Ge, P.; Yan, Y. Proteomic analysis reveals key proteins and phosphoproteins upon seed germination of wheat (Triticum aestivum L.). Front. Plant Sci. 2015, 6, 1017. [Google Scholar] [CrossRef]
- Kondoh, H.; Lleonart, M.E.; Gil, J.; Wang, J.; Degan, P.; Peters, G.; Martinez, D.; Carnero, A.; Beach, D. Glycolytic enzymes can modulate cellular life span. Cancer Res. 2005, 65, 177–185. [Google Scholar]
- Chen, X.; Yin, G.; Börner, A.; Xin, X.; He, J.; Nagel, M.; Liu, X.; Lu, X. Comparative physiology and proteomics of two wheat genotypes differing in seed storage tolerance. Plant Physiol. Biochem. 2018, 130, 455–463. [Google Scholar] [CrossRef]
- Zeng, L.; Deng, R.; Guo, Z.; Yang, S.; Deng, X. Genome-wide identification and characterization of Glyceraldehyde-3-phosphate dehydrogenase genes family in wheat (Triticum aestivum). BMC Genom. 2016, 17, 240. [Google Scholar] [CrossRef] [PubMed]
- Zhang, N.; Zhang, H.J.; Sun, Q.Q.; Cao, Y.Y.; Li, X.; Zhao, B.; Wu, P.; Guo, Y.D. Proteomic analysis reveals a role of melatonin in promoting cucumber seed germination under high salinity by regulating energy production. Sci. Rep. 2017, 7, 503. [Google Scholar] [CrossRef] [PubMed]
- Izard, T.; Aevarsson, A.; Allen, M.D.; Westphal, A.H.; Perham, R.N.; de Kok, A.; Hol, W.G.J. Principles of quasi-equivalence and Euclidean geometry govern the assembly of cubic and dodecahedral cores of pyruvate dehydrogenase complexes. Proc. Natl. Acad. Sci. USA 1999, 96, 1240–1245. [Google Scholar] [CrossRef]
- Zhang, Y.X.; Xu, H.H.; Liu, S.J.; Li, N.; Wang, W.Q.; Møller, I.M.; Song, S.Q. Proteomic analysis reveals different involvement of embryo and endosperm proteins during aging of Yliangyou 2 hybrid rice seeds. Front. Plant Sci. 2016, 7, 1394. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Huang, S.; Millar, A.H. Succinate dehydrogenase: The complex roles of a simple enzyme. Curr. Opin. Plant Biol. 2013, 16, 344–349. [Google Scholar] [CrossRef]
- Renwick, S.B.; Snell, K.; Baumann, U. The crystal structure of human cytosolic serine hydroxymethyltransferase: A target for cancer chemotherapy. Structure 1998, 6, 1105–1116. [Google Scholar] [CrossRef]
- Engel, N.; Ewald, R.; Gupta, K.J.; Zrenner, R.; Hagemann, M.; Bauwe, H. The presequence of Arabidopsis serine hydroxymethyltransferase SHM2 selectively prevents import into mesophyll mitochondria. Plant Physiol. 2011, 157, 1711–1720. [Google Scholar] [CrossRef][Green Version]
- Muehlbauer, G.J.; Somers, D.A.; Matthews, B.F.; Gengenbach, B.G. Molecular genetics of the maize (Zea mays L.) aspartate kinase-homoserine dehydrogenase gene family. Plant Physiol. 1994, 106, 1303–1312. [Google Scholar] [CrossRef]
- Yang, X.; Cui, X.; Zhao, L.; Guo, D.; Feng, L.; Wei, S.; Zhao, C.; Huang, D. Exogenous glycine nitrogen enhances accumulation of glycosylated flavonoids and antioxidant activity in lettuce (Lactuca sativa L.). Front. Plant Sci. 2017, 8, 2098. [Google Scholar] [CrossRef]
- Ábrahám, E.; Rigó, G.; Székely, G.; Nagy, R.; Koncz, C.; Szabados, L. Light-dependent induction of proline biosynthesis by abscisic acid and salt stress is inhibited by brassinosteroid in Arabidopsis. Plant Mol. Biol. 2003, 51, 363–372. [Google Scholar] [CrossRef]
- Light, S.H.; Anderson, W.F. The diversity of allosteric controls at the gateway to aromatic amino acid biosynthesis. Protein Sci. 2013, 22, 395–404. [Google Scholar] [CrossRef] [PubMed]
- Yoo, H.; Widhalm, J.R.; Qian, Y.; Maeda, H.; Cooper, B.R.; Jannasch, A.S.; Gonda, I.; Lewinsohn, E.; Rhodes, D.; Dudareva, N. An alternative pathway contributes to phenylalanine biosynthesis in plants via a cytosolic tyrosine: Phenylpyruvate aminotransferase. Nat. Commun. 2013, 4, 2833. [Google Scholar] [CrossRef] [PubMed]
- Andrea, G.B.; Raymond, R.; Dane, W. Phenylalanine ammonia lyase activity, antioxidant properties, fatty acid profile, mineral content and physiochemical analyses of Cissus sicyoides berries. J. Berry Res. 2017, 7, 117–127. [Google Scholar]
- Brunetti, C.; Guidi, L.; Sebastiani, F.; Tattini, M. Isoprenoids and phenylpropanoids are key components of the antioxidant defense system of plants facing severe excess light stress. Environ. Exp. Bot. 2015, 119, 54–62. [Google Scholar] [CrossRef]
- Lindbäck, T.; Granum, P.E. The Comprehensive Sourcebook of Bacterial Protein Toxins, 4th ed.; Bacillus cereus phospholipases, enterotoxins, and other hemolysins; Elsevier Press: Amsterdam, The Netherlands, 2015; pp. 839–857. ISBN 978-0-12-800188-2. [Google Scholar]
- Shi, H.; Jiang, C.; Ye, T.; Tan, D.; Reiter, R.J.; Zhang, H.; Liu, R.; Chan, Z. Comparative physiological, metabolomic, and transcriptomic analyses reveal mechanisms of improved abiotic stress resistance in bermudagrass [Cynodon dactylon (L). Pers.] by exogenous melatonin. J. Exp. Bot. 2015, 66, 681–694. [Google Scholar] [CrossRef]
- Chen, Y.; Li, B.; Cen, K.; Lu, Y.; Zhang, S.; Wang, C. Diverse effect of phosphatidylcholine biosynthetic genes on phospholipid homeostasis, cell autophagy and fungal developments in Metarhizium robertsii. Environ. Microbiol. 2018, 20, 293–304. [Google Scholar] [CrossRef]
- Sun, M.; Peng, F.; Xiao, Y.; Yu, W.; Zhang, Y.; Gao, H. Exogenous phosphatidylcholine treatment alleviates drought stress and maintains the integrity of root cell membranes in peach. Sci. Hortic. 2020, 259, 108821. [Google Scholar] [CrossRef]
- Sharma, R.; Moffatt, R.; Singh, R.B.; Katz, J. Omega-3 fatty acids in prevention of cardiovascular disease in humans. In Bioactive Food as Dietary Interventions for Cardiovascular Disease; Watson, R.R., Preedy, V., Eds.; Morgan Kaufmann Publishers: San Francisco, CA, USA, 2013; pp. 465–489. [Google Scholar]
- Xin, Z.; Zhang, J.; Ge, L.; Lei, S.; Han, J.; Zhang, X.; Li, X.; Sun, X. A putative 12-oxophytodienoate reductase gene CsOPR3 from Camellia sinensis, is involved in wound and herbivore infestation responses. Gene 2017, 615, 18–24. [Google Scholar] [CrossRef]
- Raviv, Z.; Cohen, S.; Reischer-Pelech, D. The anti-cancer activities of jasmonates. Cancer Chemother. Pharmacol. 2013, 71, 275–285. [Google Scholar] [CrossRef]
- Arnao, M.B.; Hernández-Ruiz, J. Melatonin in flowering, fruit set and fruit ripening. Plant Reprod. 2020, 33, 77–87. [Google Scholar] [CrossRef]
- Ahmad, S.; Su, W.; Kamran, M.; Ahmad, I.; Meng, X.; Wu, X.; Javed, T.; Han, Q. Foliar application of melatonin delay leaf senescence in maize by improving the antioxidant defense system and enhancing photosynthetic capacity under semi-arid regions. Protoplasma 2020, 25, 1079–1092. [Google Scholar] [CrossRef] [PubMed]
- Posmyk, M.M.; Bałabusta, M.; Wieczorek, M.; Sliwinska, E.; Janas, K.M. Melatonin applied to cucumber (Cucumis sativus L.) seeds improves germination during chilling stress. J. Pineal Res. 2009, 46, 214–223. [Google Scholar] [CrossRef] [PubMed]
- Arnao, M.B.; Hernández-Ruiz, J. Growth activity, rooting capacity, and tropism: Three auxinic precepts fulfilled by melatonin. Acta Physiol. Plant. 2017, 39, 127. [Google Scholar] [CrossRef]
- Moustafa-Farag, M.; Almoneafy, A.; Mahmoud, A.; Elkelish, A.; Arnao, M.B.; Li, L.; Ai, S. Melatonin and its protective role against biotic stress impacts on plants. Biomolecules 2020, 10, 54. [Google Scholar] [CrossRef]
- Reiter, R.J.; Tan, D.X.; Galano, A. Melatonin reduces lipid peroxidation and membrane viscosity. Front. Physiol. 2014, 5, 377. [Google Scholar] [CrossRef] [PubMed]
- Liang, D.; Shen, Y.; Ni, Z.; Wang, Q.; Lei, Z.; Xu, N.; Deng, Q.; Lin, L.; Wang, J.; Lv, X.; et al. Exogenous melatonin application delays senescence of kiwifruit leaves by regulating the antioxidant capacity and biosynthesis of flavonoids. Front. Plant Sci. 2018, 9, 426. [Google Scholar] [CrossRef]
- Pothinuch, P.; Tongchitpakdee, S. Melatonin contents in mulberry (Morus spp.) leaves: Effects of sample preparation, cultivar, leaf age and tea processing. Food Chem. 2011, 128, 415–419. [Google Scholar] [CrossRef]
- ISTA. International Rules for Seed Testing; International Seed Testing Association: Bassersdorf, Switzerland, 2015. [Google Scholar]
- Abdul-Baki, A.A.; Anderson, J.D. Vigour determination in soybean seed multiple criteria. Crop Sci. 1973, 13, 630–633. [Google Scholar] [CrossRef]
- Bailly, C.; Benamar, A.; Corbineau, F.; Côme, D. Changes in malondialdehyde content and superoxide dismutase, catalase and glutathione reductase activities in sunflower seeds as related to deterioration during accelerated aging. Physiol. Plantarum 1996, 97, 104–111. [Google Scholar] [CrossRef]
- Beauchamp, C.; Fridovich, I. Superoxide dismutase: Improved assays and an assay applicable to acrylamide gels. Anal. Biochem. 1971, 44, 276–287. [Google Scholar] [CrossRef]
- Dhindsa, R.S.; Matowe, W. Drought tolerance in two mosses: Correlated with enzymatic defense against lipid peroxidation. J. Exp. Bot. 1981, 32, 79–91. [Google Scholar] [CrossRef]
- Cakmak, I.; Marschner, H. Magnesium deficiency and high light intensity enhance activities of superoxide dismutase, ascorbate peroxidase, and glutathione reductase in bean leaves. Plant Physiol. 1992, 98, 1222–1227. [Google Scholar] [CrossRef] [PubMed]
- Arrigoni, O.; Dipierro, S.; Borraccino, G. Ascorbate free radical reductase: A key enzyme of the ascorbic acid system. FEBS Lett. 1981, 125, 242–244. [Google Scholar] [CrossRef]
- Dalton, D.A.; Baird, L.M.; Langeberg, L.; Taugher, C.Y.; Anyan, W.R.; Vance, C.P.; Sarath, G. Subcellular localization of oxygen defense enzymes in soybean (Glycine max [L.] Merr.) root nodules. Plant Physiol. 1993, 102, 481–489. [Google Scholar] [CrossRef] [PubMed]
- Nakano, Y.; Asada, K. Hydrogen peroxide scavenged by ascorbate-specific peroxidase in spinach chloroplast. Plant Cell Physiol. 1981, 22, 867–880. [Google Scholar]
- Madamanchi, N.R.; Alscher, R.G. Metabolic bases for differences in sensitivity of two pea cultivars to sulfur dioxide. Plant Physiol. 1991, 97, 88–93. [Google Scholar] [CrossRef]


| Accession | Description | T12 vs. CK | T24 vs. CK | T36 vs. CK | |||
|---|---|---|---|---|---|---|---|
| FC | p-Value | FC | p-Value | FC | p-Value | ||
| Carbon Metabolism and Energy Production | |||||||
| Starch and Sucrose Metabolism | |||||||
| A0A0Q3GVZ0 | Sucrose synthase | 1.200 | 0.0094 | 1.509 | 0.0001 | 1.309 | 0.0074 |
| W5DP16 | UDP-glucose 6-dehydrogenase | 1.182 | 0.2406 | 0.902 | 0.4743 | 1.523 | 0.0294 |
| Glycolysis/Gluconeogenesis | |||||||
| W4ZRX8 | ATP-dependent 6-phosphofructokinase | 1.307 | 0.2254 | 2.459 | 0.0112 | 1.682 | 0.0598 |
| Q6Z9G0 | Glyceraldehyde-3-phosphate dehydrogenase | 1.160 | 0.7688 | 1.683 | 0.0828 | 2.091 | 0.0355 |
| A0A1J7GV79 | Pyruvate kinase | 1.367 | 0.0435 | 1.398 | 0.0200 | 1.722 | 0.0055 |
| A0A1J3JHF1 | S-(hydroxymethyl)glutathione dehydrogenase (Fragment) | 2.110 | 0.0164 | 1.998 | 0.0101 | 1.837 | 0.0157 |
| Pyruvate metabolism | |||||||
| A0A0D3EN96 | Dihydrolipoyl dehydrogenase | 1.288 | 0.1383 | 1.551 | 0.0333 | 1.475 | 0.0628 |
| Q9FVE1 | Pyruvate decarboxylase 1 (Fragment) | 0.733 | 0.0275 | 0.659 | 0.0010 | 0.682 | 0.0053 |
| K3Z7G1 | Lactoylglutathione lyase | 1.224 | 0.0194 | 1.297 | 0.0029 | 1.653 | 0.0041 |
| A0A1J3DH40 | D-lactate dehydrogenase [cytochrome], mitochondrial (Fragment) | 0.234 | 0.0023 | 0.535 | 0.0096 | 0.618 | 0.0294 |
| Citrate cycle (TCA cycle) | |||||||
| Q10S34 | Aconitate hydratase | 1.680 | 0.0076 | 1.206 | 0.1089 | 1.602 | 0.0193 |
| F2E611 | Succinate dehydrogenase [ubiquinone] flavoprotein subunit, mitochondrial | 0.520 | 0.0072 | 0.818 | 0.0199 | 1.004 | 0.9945 |
| Amino acid metabolism | |||||||
| Phenylalanine, tyrosine and tryptophan biosynthesis | |||||||
| A0A1D6RN38 | Phospho-2-dehydro-3-deoxyheptonate aldolase | 0.863 | 0.1420 | 2.406 | 0.0033 | 1.656 | 0.0617 |
| B6TMW7 | Transaminase/ transferase isoform 1 | 1.716 | 0.0775 | 2.051 | 0.0366 | 2.062 | 0.0479 |
| Phenylalanine metabolism | |||||||
| A0A1D6B9G2 | Phenylalanine ammonia-lyase | 0.628 | 0.1998 | 1.336 | 0.0155 | 2.739 | 0.0393 |
| Arginine and proline metabolism | |||||||
| Q43559 | Delta-1-pyrroline-5-carboxylate synthase | 1.455 | 0.2157 | 3.981 | 0.0004 | 1.970 | 0.1216 |
| Q53UC8 | Delta-1-pyrroline-5-carboxylate synthase | 1.405 | 0.0044 | 1.334 | 0.0282 | 1.608 | 0.0024 |
| Glycine, serine and threonine metabolism | |||||||
| A0A0C4BJE5 | Serine hydroxymethyltransferase | 1.574 | 0.1904 | 1.473 | 0.2255 | 2.576 | 0.0203 |
| A0A0D3HHP5 | Homoserine dehydrogenase | 1.177 | 0.8198 | 1.722 | 0.1573 | 2.398 | 0.0495 |
| Phenylpropanoid biosynthesis | |||||||
| W5AX51 | Peroxidase | 1.457 | NA | 1.362 | 0.0445 | 1.696 | 0.0441 |
| A0A0D3A374 | Peroxidase | 1.399 | 0.0117 | 1.358 | 0.0206 | 1.604 | 0.0006 |
| A0A1D6B9G2 | Phenylalanine ammonia-lyase | 0.628 | 0.1998 | 1.336 | 0.0155 | 2.739 | 0.0393 |
| Phosphatidylinositol signaling | |||||||
| I6YMA7 | Phosphoinositide phospholipase C | 1.208 | 0.0467 | 1.704 | 0.0002 | 1.548 | 0.0023 |
| alpha-linolenic acid metabolism | |||||||
| H9CWE9 | 12-oxo-phytodienoic acid reductase | 3.499 | 0.0330 | 4.034 | 0.0355 | 4.771 | 0.0185 |
| Accession | Primer F (5′-3′) | Primer R (5′-3′) |
|---|---|---|
| H9CWE9 | GACCACGGCATCCTCTACC | CTTGGGCAGGTCTGGGTT |
| A0A078HA44 | CAGCGTCATTGGTACATCCAG | TTTCATCAAGCTCACGGCAC |
| A0A1J3DH40 | CTTGTGGGCTTGCTTTGC | TTCGCTGCTCTTCGTTGC |
| Q2R2B4 | ATGATGAACTGGTGCCTGGT | GCCATTGCCATTGCTCTT |
| ACTIN2 | GATGATGCGCCAAGAGCTG | GCCTCATCACCTACGTAGGCAT |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yan, H.; Mao, P. Comparative Time-Course Physiological Responses and Proteomic Analysis of Melatonin Priming on Promoting Germination in Aged Oat (Avena sativa L.) Seeds. Int. J. Mol. Sci. 2021, 22, 811. https://doi.org/10.3390/ijms22020811
Yan H, Mao P. Comparative Time-Course Physiological Responses and Proteomic Analysis of Melatonin Priming on Promoting Germination in Aged Oat (Avena sativa L.) Seeds. International Journal of Molecular Sciences. 2021; 22(2):811. https://doi.org/10.3390/ijms22020811
Chicago/Turabian StyleYan, Huifang, and Peisheng Mao. 2021. "Comparative Time-Course Physiological Responses and Proteomic Analysis of Melatonin Priming on Promoting Germination in Aged Oat (Avena sativa L.) Seeds" International Journal of Molecular Sciences 22, no. 2: 811. https://doi.org/10.3390/ijms22020811
APA StyleYan, H., & Mao, P. (2021). Comparative Time-Course Physiological Responses and Proteomic Analysis of Melatonin Priming on Promoting Germination in Aged Oat (Avena sativa L.) Seeds. International Journal of Molecular Sciences, 22(2), 811. https://doi.org/10.3390/ijms22020811




