Feeding Rhythm-Induced Hypothalamic Agouti-Related Protein Elevation via Glucocorticoids Leads to Insulin Resistance in Skeletal Muscle
Abstract
1. Introduction
2. Results
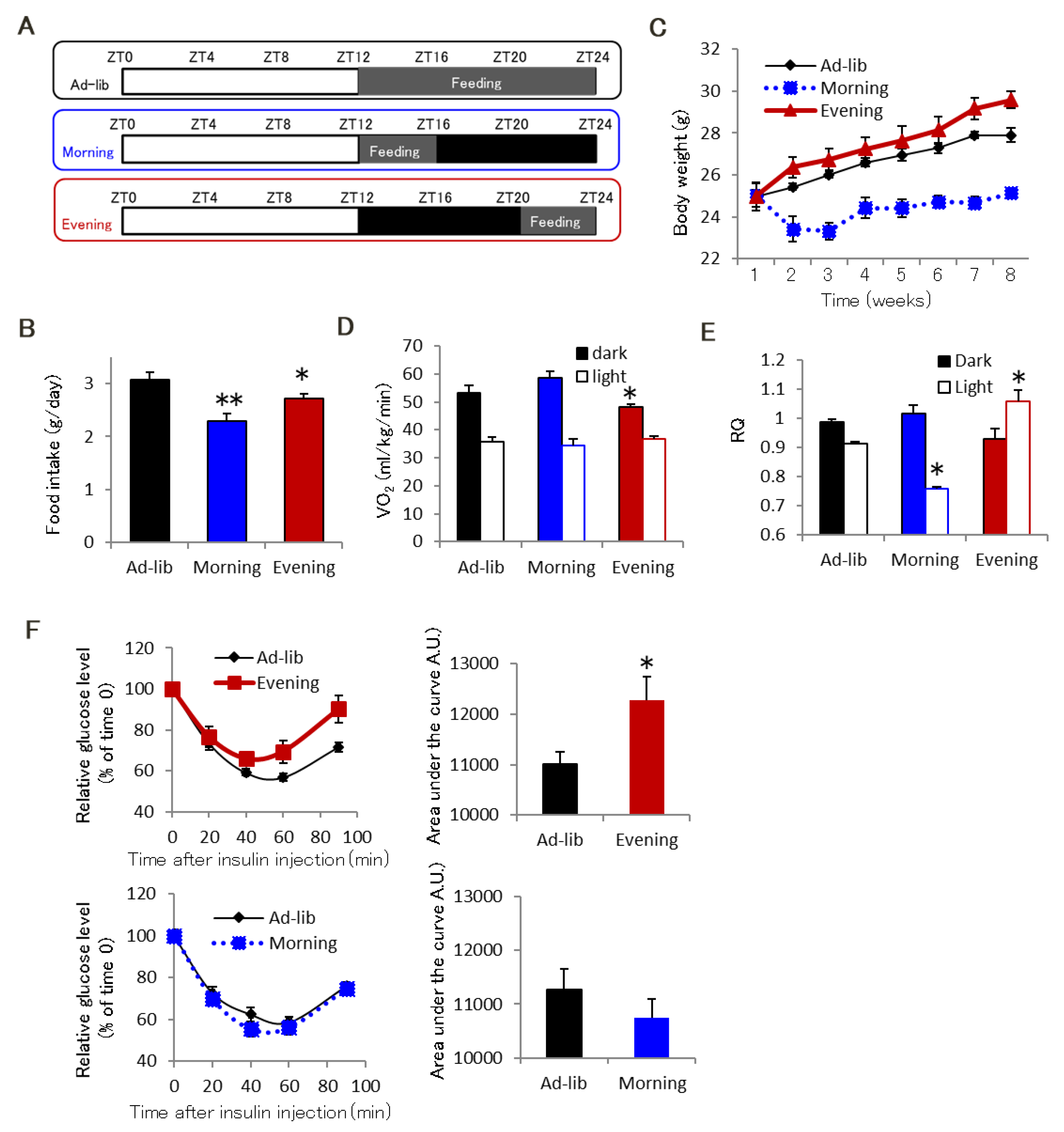
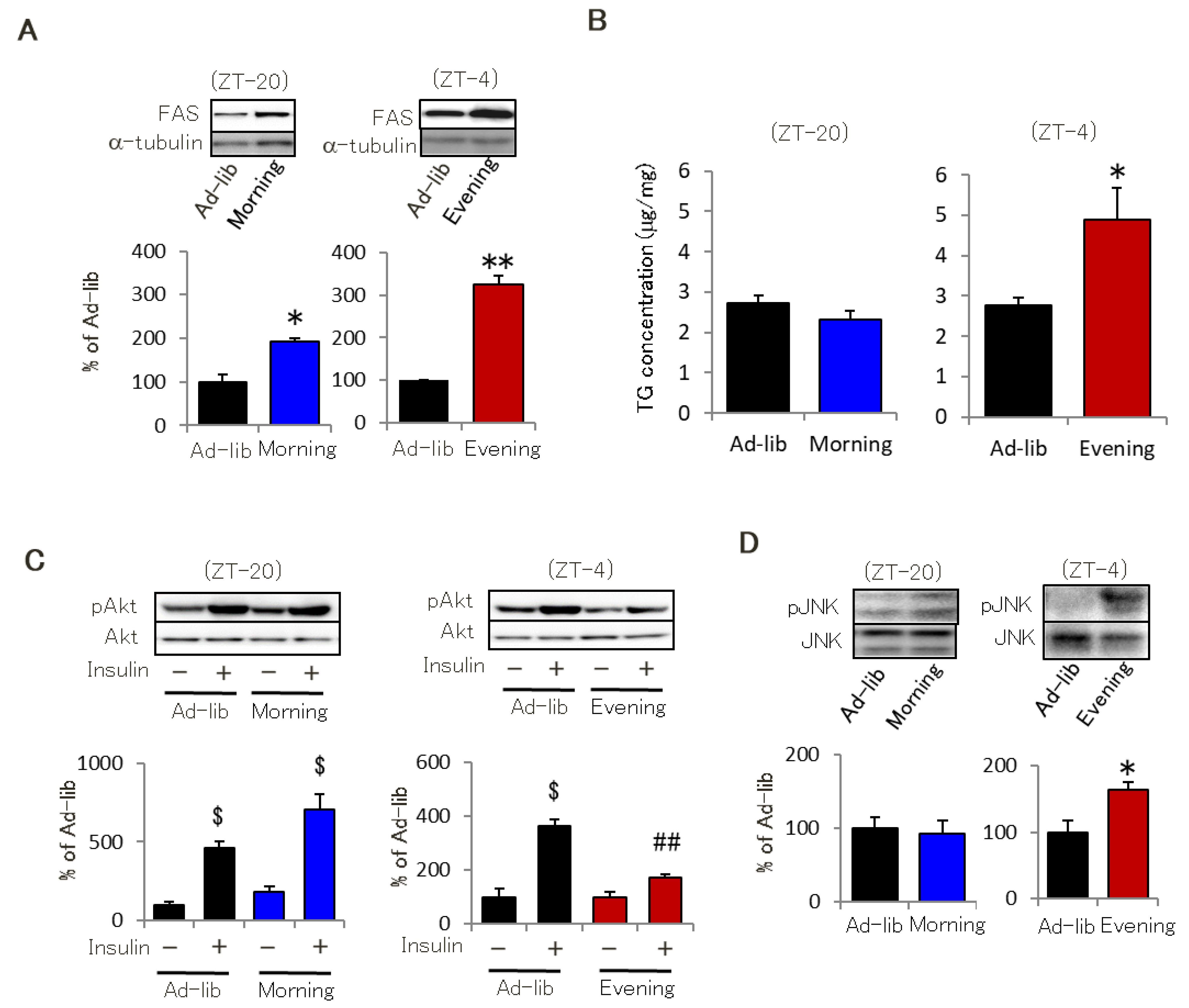
2.1. Evening-Type Feeding Rhythm Leads to Skeletal Muscle Insulin Resistance
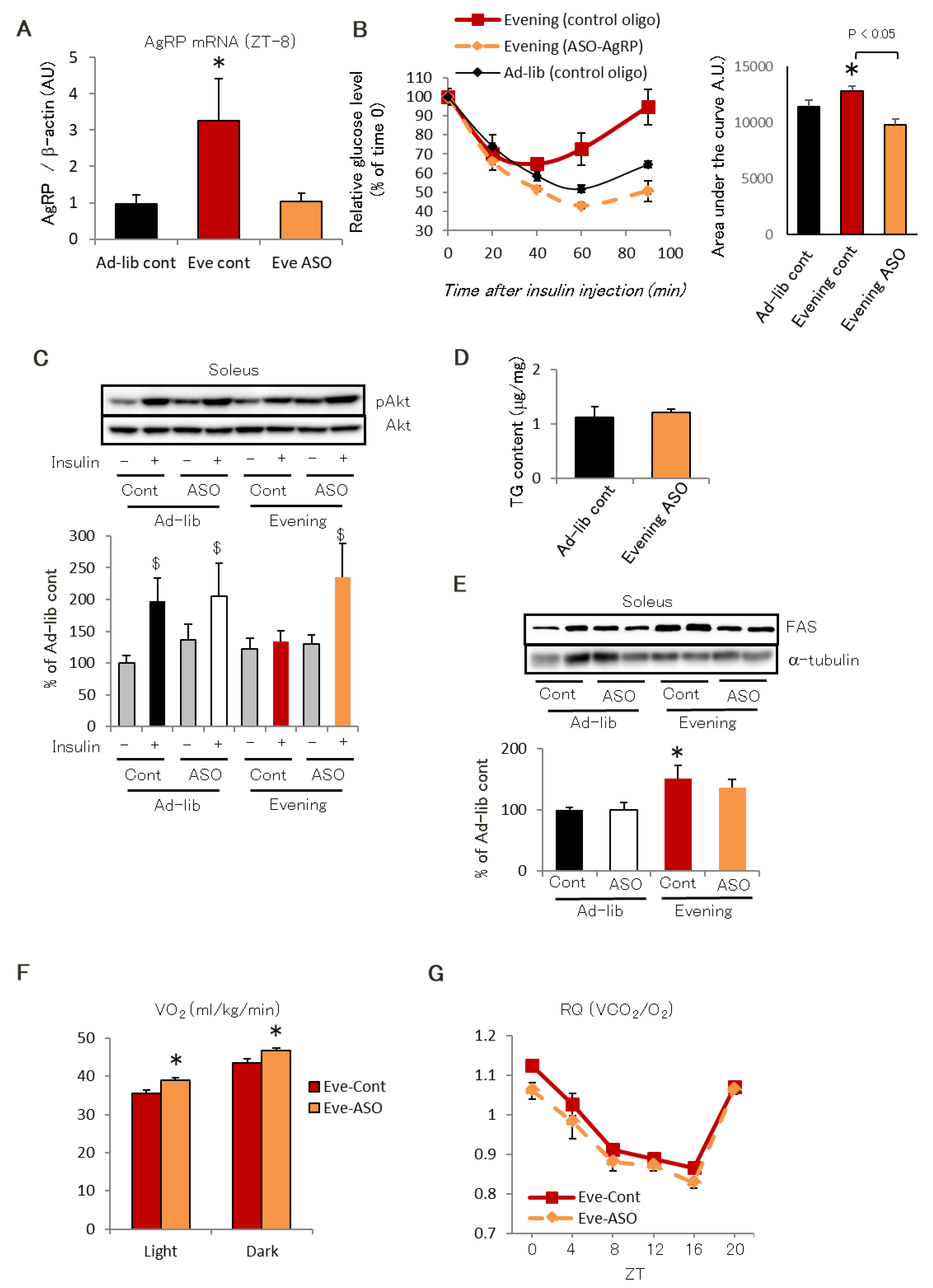
2.2. AgRP Is Responsible for Evening-Type Feeding Rhythm-Induced Insulin Resistance
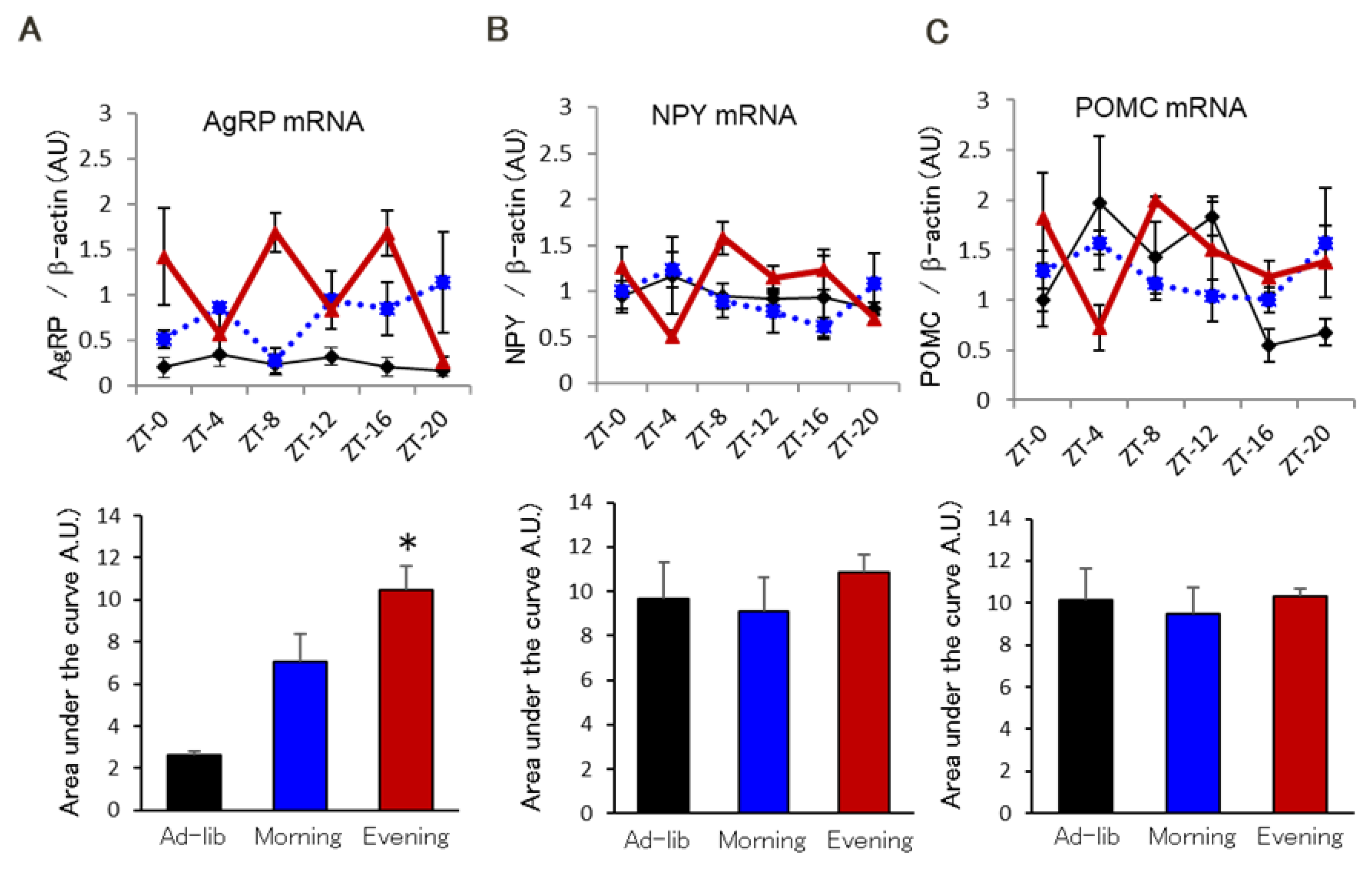
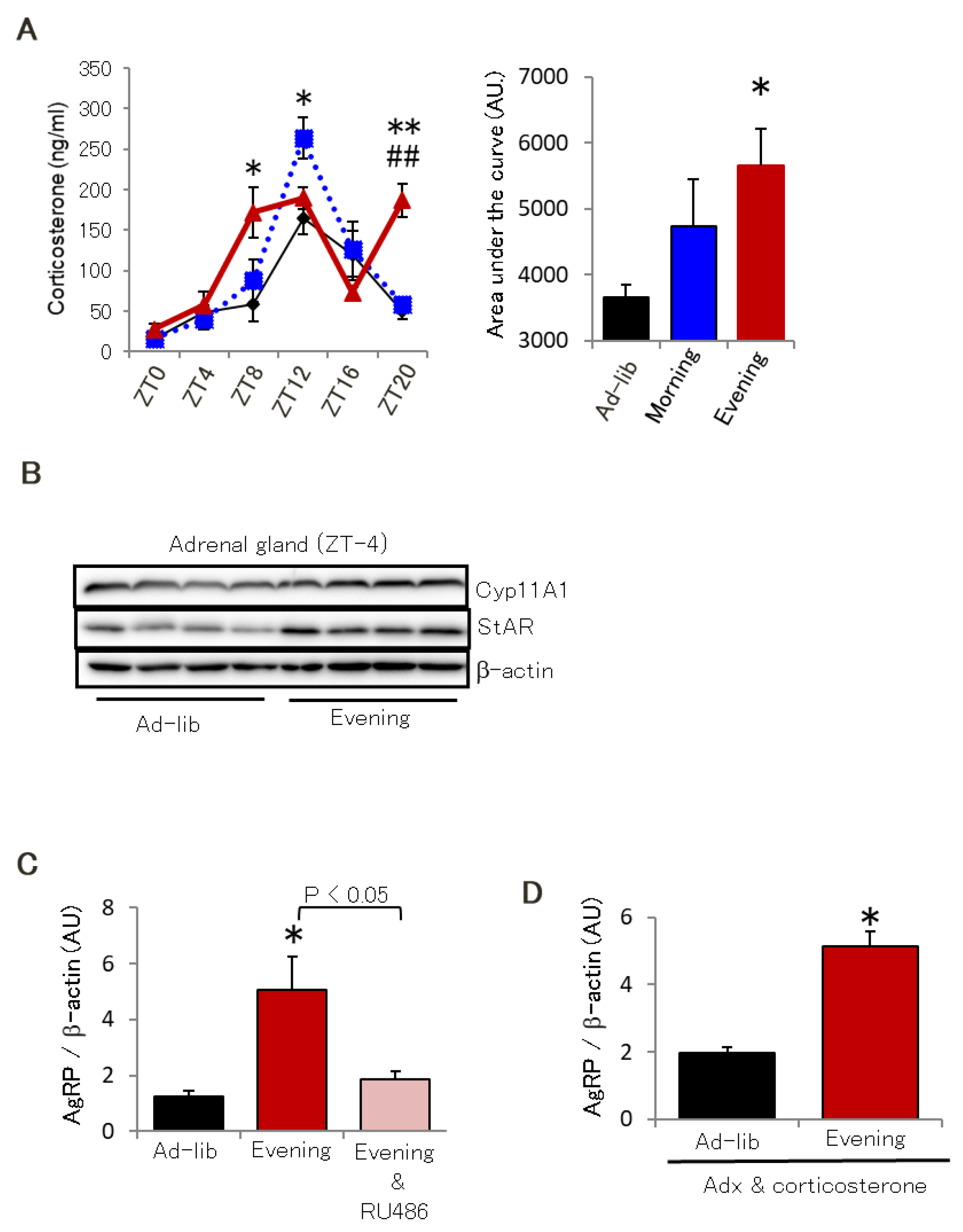
2.3. Glucocorticoid Sensitivity in the Hypothalamus Is Critical for AgRP Expression Due to the Evening-Type Feeding Rhythm
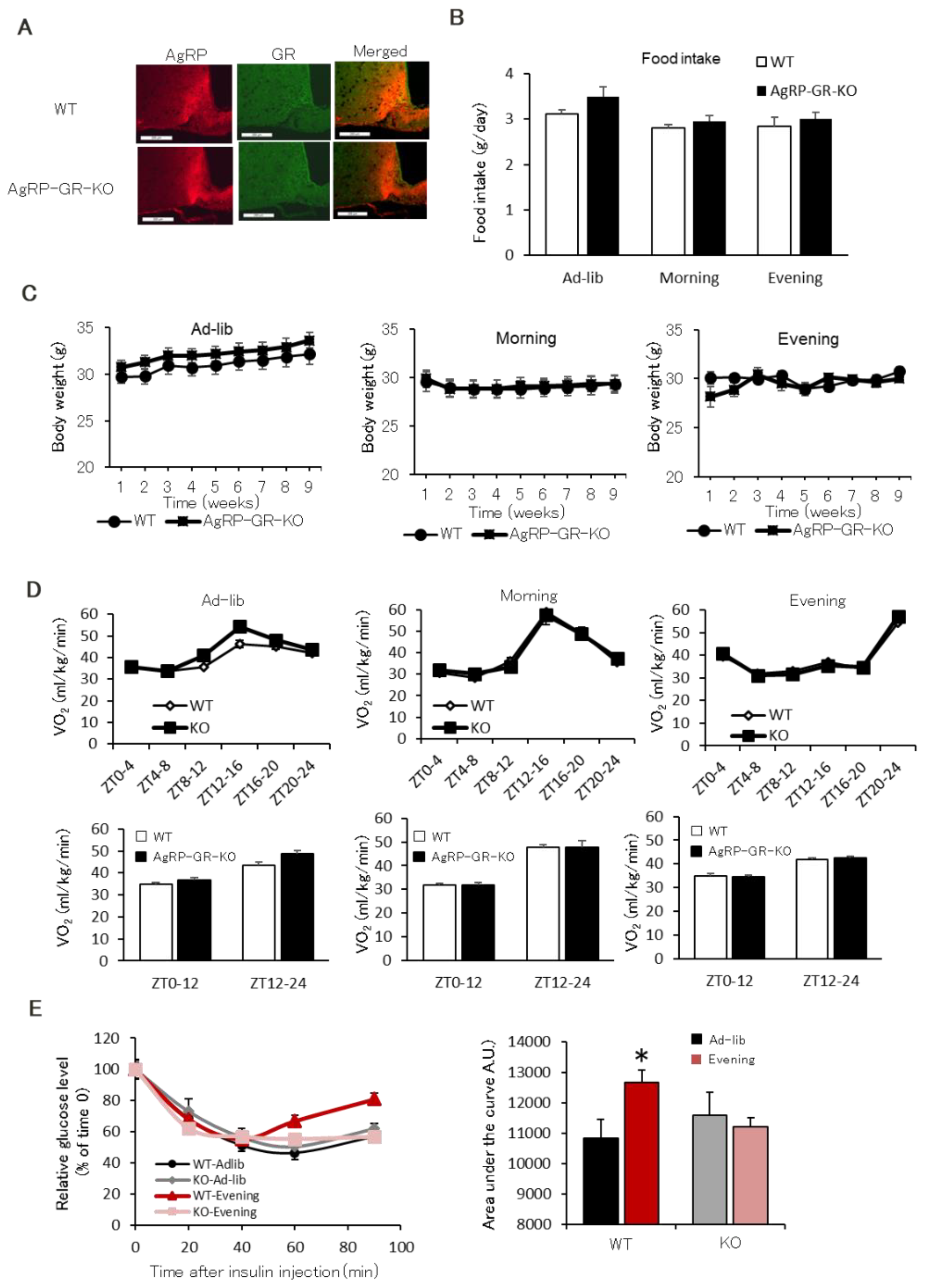
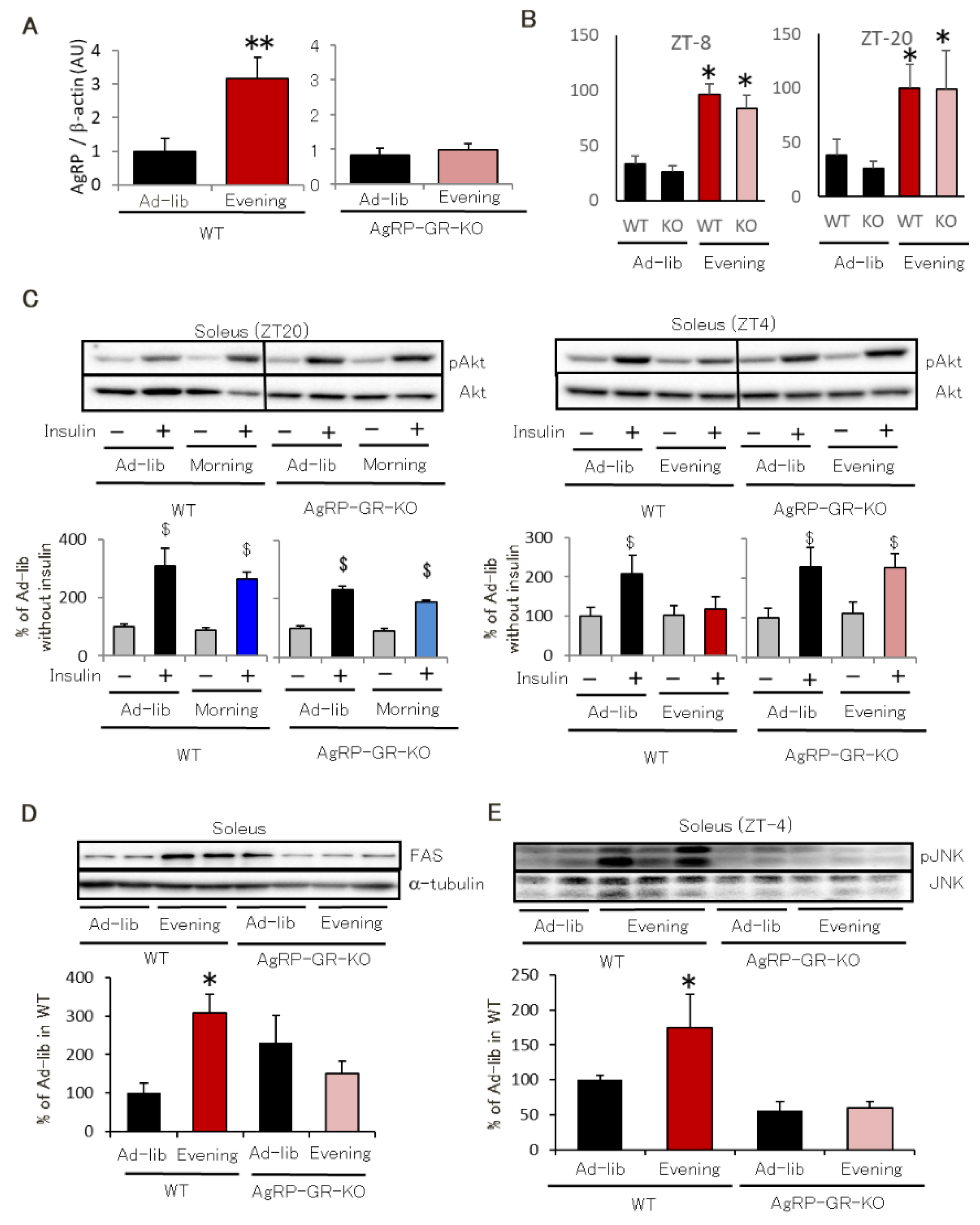
2.4. AgRP Neuron-Specific GR Knockout Mice Have Normal Insulin Sensitivity Even in the Evening-Type Feeding Rhythm
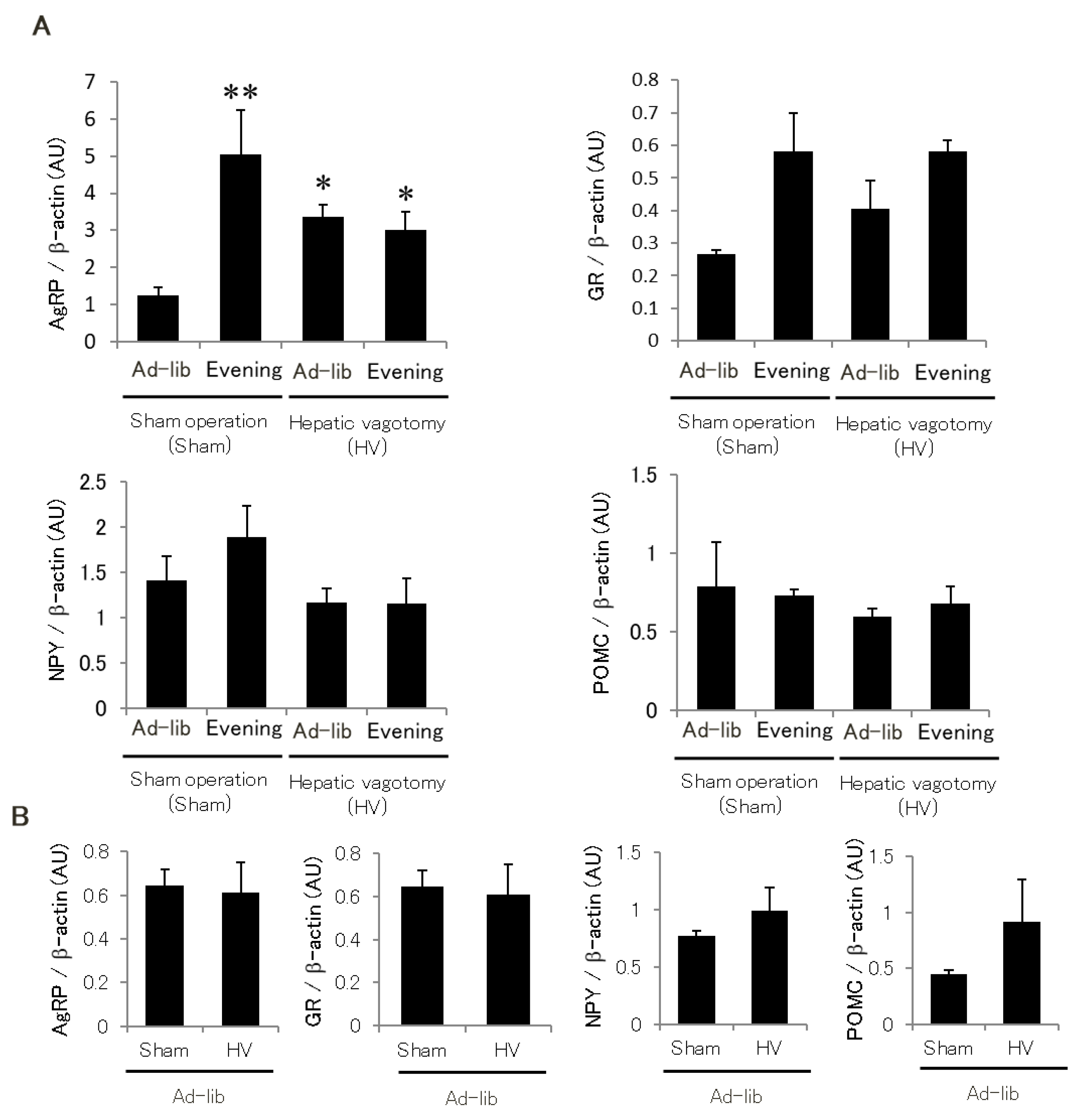
2.5. Hypothalamic Glucocorticoid Sensitivity Is Enhanced by Hepatic Afferent Nerve in the Evening-Type Feeding Rhythm
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Cre-LoxP System
4.3. Western Blotting
4.4. Immunohistochemistry
4.5. Quantitative Real-Time PCR
4.6. ITTs
4.7. GTT
4.8. Brain Cannula Implantation
4.9. Antisense Oligonucleotides
4.10. Adrenalectomy
4.11. Microarray
4.12. TG Content
4.13. NE Turnover
4.14. VO2 and RQ
4.15. Plasma Concentrations
4.16. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pot, G.K. Sleep and dietary habits in the urban environment: The role of chrono-nutrition. Proc. Nutr. Soc. 2018, 77, 189–198. [Google Scholar] [CrossRef]
- Sagris, M.; Kokkinidis, D.G.; Lempesis, I.G.; Giannopoulos, S.; Rallidis, L.; Mena-Hurtado, C.; Bakoyiannis, C. Nutrition, dietary habits, and weight management to prevent and treat patients with peripheral artery disease. Rev. Cardiovasc. Med. 2020, 21, 565–575. [Google Scholar] [PubMed]
- Panda, S. Circadian physiology of metabolism. Science 2016, 354, 1008–1015. [Google Scholar] [CrossRef] [PubMed]
- Stenvers, D.J.; Jonkers, C.F.; Fliers, E.; Bisschop, P.H.; Kalsbeek, A. Nutrition and the circadian timing system. Prog. Brain Res. 2012, 199, 359–376. [Google Scholar]
- Mohawk, J.A.; Green, C.B.; Takahashi, J.S. Central and peripheral circadian clocks in mammals. Ann. Rev. Neurosci. 2012, 35, 445–462. [Google Scholar] [CrossRef]
- Bass, J.; Takahashi, J.S. Circadian integration of metabolism and energetics. Science 2010, 330, 1349–1354. [Google Scholar] [CrossRef]
- Hatori, M.; Vollmers, C.; Zarrinpar, A.; DiTacchio, L.; Bushong, E.A.; Gill, S.; Leblanc, M.; Chaix, A.; Joens, M.; Fitzpatrick, J.A.; et al. Time-restricted feeding without reducing caloric intake prevents metabolic diseases in mice fed a high-fat diet. Cell Metab. 2012, 15, 848–860. [Google Scholar] [CrossRef] [PubMed]
- Kuroda, H.; Tahara, Y.; Saito, K.; Ohnishi, N.; Kubo, Y.; Seo, Y.; Otsuka, M.; Fuse, Y.; Ohura, Y.; Hirao, A.; et al. Meal frequency patterns determine the phase of mouse peripheral circadian clocks. Sci. Rep. 2012, 2, 711. [Google Scholar] [CrossRef]
- Tahara, Y.; Shibata, S. Chronobiology and nutrition. Neuroscience 2013, 253, 78–88. [Google Scholar] [CrossRef]
- Asher, G.; Sassone-Corsi, P. Time for food: The intimate interplay between nutrition, metabolism, and the circadian clock. Cell 2015, 161, 84–92. [Google Scholar] [CrossRef]
- Oishi, K.; Hashimoto, C. Short-term time-restricted feeding during the resting phase is sufficient to induce leptin resistance that contributes to development of obesity and metabolic disorders in mice. Chronobiol. Int. 2018, 35, 1576–1594. [Google Scholar] [CrossRef] [PubMed]
- Okauchi, H.; Hashimoto, C.; Nakao, R.; Oishi, K. Timing of food intake is more potent than habitual voluntary exercise to prevent diet-induced obesity in mice. Chronobiol. Int. 2019, 36, 57–74. [Google Scholar] [CrossRef]
- Minokoshi, Y.; Haque, M.S.; Shimazu, T. Microinjection of leptin into the ventromedial hypothalamus increases glucose uptake in peripheral tissues in rats. Diabetes 1999, 48, 287–291. [Google Scholar] [CrossRef]
- Haque, M.S.; Minokoshi, Y.; Hamai, M.; Iwai, M.; Horiuchi, M.; Shimazu, T. Role of the sympathetic nervous system and insulin in enhancing glucose uptake in peripheral tissues after intrahypothalamic injection of leptin in rats. Diabetes 1999, 48, 1706–1712. [Google Scholar] [CrossRef]
- Minokoshi, Y.; Kim, Y.B.; Peroni, O.D.; Fryer, L.G.; Müller, C.; Carling, D.; Kahn, B.B. Leptin stimulates fatty-acid oxidation by activating AMP-activated protein kinase. Nature 2002, 415, 339–343. [Google Scholar] [CrossRef] [PubMed]
- Inoue, H.; Ogawa, W.; Asakawa, A.; Okamoto, Y.; Nishizawa, A.; Matsumoto, M.; Teshigawara, K.; Matsuki, Y.; Watanabe, E.; Hiramatsu, R.; et al. Role of hepatic STAT3 in brain-insulin action on hepatic glucose production. Cell Metab. 2006, 3, 267–275. [Google Scholar] [CrossRef]
- Tanaka, T.; Masuzaki, H.; Yasue, S.; Ebihara, K.; Shiuchi, T.; Ishii, T.; Arai, N.; Hirata, M.; Yamamoto, H.; Hayashi, T.; et al. Central melanocortin signaling restores skeletal muscle AMP-activated protein kinase phosphorylation in mice fed a high-fat diet. Cell Metab. 2007, 5, 395–402. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Shiuchi, T.; Haque, M.S.; Okamoto, S.; Inoue, T.; Kageyama, H.; Lee, S.; Toda, C.; Suzuki, A.; Bachman, E.S.; Kim, Y.B.; et al. Hypothalamic orexin stimulates feeding-associated glucose utilization in skeletal muscle via sympathetic nervous system. Cell Metab. 2009, 10, 466–480. [Google Scholar] [CrossRef]
- Toda, C.; Shiuchi, T.; Lee, S.; Yamato-Esaki, M.; Fujino, Y.; Suzuki, A.; Okamoto, S.; Minokoshi, Y. Distinct effects of leptin and a melanocortin receptor agonist injected into medial hypothalamic nuclei on glucose uptake in peripheral tissues. Diabetes 2009, 58, 2757–2765. [Google Scholar] [CrossRef]
- Toda, C.; Shiuchi, T.; Kageyama, H.; Okamoto, S.; Coutinho, E.A.; Sato, T.; Okamatsu-Ogura, Y.; Yokota, S.; Takagi, K.; Tang, L.; et al. Extracellular signal-regulated kinase in the ventromedial hypothalamus mediates leptin-induced glucose uptake in red-type skeletal muscle. Diabetes 2013, 62, 2295–2307. [Google Scholar] [CrossRef][Green Version]
- Kimura, K.; Nakamura, Y.; Inaba, Y.; Matsumoto, M.; Kido, Y.; Asahara, S.; Matsuda, T.; Watanabe, H.; Maeda, A.; Inagaki, F.; et al. Inoue H Histidine augments the suppression of hepatic glucose production by central insulin action. Diabetes 2013, 62, 2266–2277. [Google Scholar] [CrossRef]
- Shiuchi, T.; Toda, C.; Okamoto, S.; Coutinho, E.A.; Saito, K.; Miura, S.; Ezaki, O.; Minokoshi, Y. Induction of glucose uptake in skeletal muscle by central leptin is mediated by muscle β 2-adrenergic receptor but not by AMPK. Sci. Rep. 2017, 7, 15141. [Google Scholar] [CrossRef] [PubMed]
- Kalsbeek, A.; Bruinstroop, E.; Yi, C.X.; Klieverik, L.P.; La Fleur, S.E.; Fliers, E. Hypothalamic control of energy metabolism via the autonomic nervous system. Ann. N. Y. Acad. Sci. 2010, 1212, 114–129. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, M.W.; Woods, S.C.; Porte, D., Jr.; Seeley, R.J.; Baskin, D.G. Central nervous system control of food intake. Nature 2000, 404, 661–671. [Google Scholar] [CrossRef] [PubMed]
- Yamada, T.; Oka, Y.; Katagiri, H. Inter-organ metabolic communication involved in energy homeostasis: Potential therapeutic targets for obesity and metabolic syndrome. Pharmacol. Ther. 2008, 117, 188–198. [Google Scholar] [CrossRef]
- Kimura, K.; Tanida, M.; Nagata, N.; Inaba, Y.; Watanabe, H.; Nagashimada, M.; Ota, T.; Asahara, S.; Kido, Y.; Matsumoto, M.; et al. Central Insulin Action Activates Kupffer Cells by Suppressing Hepatic Vagal Activation via the Nicotinic Alpha 7 Acetylcholine Receptor. Cell Rep. 2016, 14, 2362–2374. [Google Scholar] [CrossRef]
- Nakayama, S.; Nishiyama, M.; Iwasaki, Y.; Shinahara, M.; Okada, Y.; Tsuda, M.; Okazaki, M.; Tsugita, M.; Taguchi, T.; Makino, S.; et al. Corticotropin-releasing hormone (CRH) transgenic mice display hyperphagia with increased Agouti-related protein mRNA in the hypothalamic arcuate nucleus. Endocr. J. 2011, 58, 279–286. [Google Scholar] [CrossRef]
- Lee, B.; Kim, S.-G.; Kim, J.; Choi, K.Y.; Lee, S.; Lee, S.-K.; Lee, J.W. Brain-specific homeobox factor as a target selector for glucocorticoid receptor in energy balance. Mol. Cell. Biol. 2013, 33, 2650–2658. [Google Scholar] [CrossRef]
- Lu, X.Y.; Kim, S.G.; Kim, J.; Choi, K.Y.; Lee, S.; Lee, S.K.; Lee, J.W. Diurnal rhythm of agouti-related protein and its relation to corticosterone and food intake. Endocrinology 2002, 33, 2650–2658. [Google Scholar] [CrossRef]
- Stunkard, A.J.; Allison, K.C.; Lundgren, J.D.; O’Reardon, J.P. A biobehavioural model of the night eating syndrome. Obes. Rev. 2009, 10, 69–77. [Google Scholar] [CrossRef]
- Vander Wal, J.S. Night eating syndrome: A critical review of the literature. Clin. Psychol. Rev. 2012, 32, 49–59. [Google Scholar] [CrossRef]
- Adamovich, Y.; Rousso-Noori, L.; Zwighaft, Z.; Neufeld-Cohen, A.; Golik, M.; Kraut-Cohen, J.; Wang, M.; Han, X.; Asher, G. Circadian clocks and feeding time regulate the oscillations and levels of hepatic triglycerides. Cell Metab. 2014, 19, 319–330. [Google Scholar] [CrossRef]
- Chaix, A.; Zarrinpar, A.; Miu, P.; Panda, S. Time-restricted feeding is a preventative and therapeutic intervention against diverse nutritional challenges. Cell Metab. 2014, 20, 991–1005. [Google Scholar] [CrossRef]
- Coen, P.M.; Dubé, J.J.; Amati, F.; Stefanovic-Racic, M.; Ferrell, R.E.; Toledo, F.G.; Goodpaster, B.H. Insulin resistance is associated with higher intramyocellular triglycerides in type I but not type II myocytes concomitant with higher ceramide content. Diabetes 2010, 59, 80–88. [Google Scholar] [CrossRef]
- Liu, L.; Zhang, Y.; Chen, N.; Shi, X.; Tsang, B.; Yu, Y.H. Upregulation of myocellular DGAT1 augments triglyceride synthesis in skeletal muscle and protects against fat-induced insulin resistance. J. Clin. Investig. 2007, 117, 1679–1689. [Google Scholar] [CrossRef] [PubMed]
- Henstridge, D.C.; Bruce, C.R.; Pang, C.P.; Lancaster, G.I.; Allen, T.L.; Estevez, E.; Gardner, T.; Weir, J.M.; Meikle, P.J.; Lam, K.S.L.; et al. Skeletal muscle-specific overproduction of constitutively activated c-Jun N-terminal kinase (JNK) induces insulin resistance in mice. Diabetologia 2012, 55, 2769–2778. [Google Scholar] [CrossRef] [PubMed]
- Corander, M.P.; Rimmington, D.; Challis, B.G.; O’Rahilly, S.; Coll, A.P. Loss of agouti-related peptide does not significantly impact the phenotype of murine POMC deficiency. Endocrinology 2011, 152, 1819–1828. [Google Scholar] [CrossRef]
- Wortley, K.E.; Anderson, K.D.; Yasenchak, J.; Murphy, A.; Valenzuela, D.; Diano, S.; Yancopoulos, G.D.; Wiegand, S.J.; Sleeman, M.W. Agouti-related protein-deficient mice display an age-related lean phenotype. Cell Metab. 2005, 2, 421–427. [Google Scholar] [CrossRef] [PubMed]
- Joly-Amado, A.; Denis, R.G.; Castel, J.; Lacombe, A.; Cansell, C.; Rouch, C.; Kassis, N.; Dairou, J.; Cani, P.D.; Ventura-Clapier, R.; et al. Hypothalamic AgRP-neurons control peripheral substrate utilization and nutrient partitioning. EMBO J. 2012, 31, 4276–4288. [Google Scholar] [CrossRef]
- Kim, S.G.; Lee, B.; Kim, D.H.; Kim, J.; Lee, S.; Lee, S.K.; Lee, J.W. Control of energy balance by hypothalamic gene circuitry involving two nuclear receptors, neuron-derived orphan receptor 1 and glucocorticoid receptor. Mol. Cell. Biol. 2013, 33, 3826–3834. [Google Scholar] [CrossRef]
- Hagimoto, S.; Arima, H.; Adachi, K.; Ito, Y.; Suga, H.; Sugimura, Y.; Goto, M.; Banno, R.; Oiso, Y. Expression of neuropeptide Y and agouti-related protein mRNA stimulated by glucocorticoids is attenuated via NF-κB p65 under ER stress in mouse hypothalamic cultures. Neurosci. Lett. 2013, 553, 165–169. [Google Scholar] [CrossRef]
- Shibata, M.; Banno, R.; Sugiyama, M.; Tominaga, T.; Onoue, T.; Tsunekawa, T.; Azuma, Y.; Hagiwara, D.; Lu, W.; Ito, Y.; et al. AgRP neuron-specific deletion of glucocorticoid receptor leads to increased energy expenditure and decreased body weight in female mice on a high-fat diet. Endocrinology 2016, 157, 1457–1466. [Google Scholar] [CrossRef]
- Izumida, Y.; Yahagi, N.; Takeuchi, Y.; Nishi, M.; Shikama, A.; Takarada, A.; Masuda, Y.; Kubota, M.; Matsuzaka, T.; Nakagawa, Y.; et al. Glycogen shortage during fasting triggers liver-brain-adipose neurocircuitry to facilitate fat utilization. Nat. Commun. 2013, 4, 2316. [Google Scholar] [CrossRef]
- Gyengesi, E.; Liu, Z.W.; D’Agostino, G.; Gan, G.; Horvath, T.L.; Gao, X.B.; Diano, S. Corticosterone regulates synaptic input organization of POMC and NPY/AgRP neurons in adult mice. Endocrinology 2010, 151, 5395–5402. [Google Scholar] [CrossRef] [PubMed]
- Watt, M.J. Storing up trouble: Does accumulation of intramyocellular triglyceride protect skeletal muscle from insulin resistance? Clin. Exp. Pharmacol. Physiol. 2009, 36, 5–11. [Google Scholar] [CrossRef] [PubMed]
- Watt, M.J.; Hevener, A.; Lancaster, G.I.; Febbraio, M.A. Ciliary neurotrophic factor prevents acute lipid-induced insulin resistance by attenuating ceramide accumulation and phosphorylation of c-Jun N-terminal kinase in peripheral tissues. Endocrinology 2006, 147, 2077–2085. [Google Scholar] [CrossRef]
- Padilla, S.L.; Qiu, J.; Soden, M.E.; Sanz, E.; Nestor, C.C.; Barker, F.D.; Quintana, A.; Zweifel, L.S.; Rønnekleiv, O.K.; Kelly, M.J.; et al. Agouti-related peptide neural circuits mediate adaptive behaviors in the starved state. Nat. Neurosci. 2016, 19, 734–741. [Google Scholar] [CrossRef]
- Burnett, C.J.; Li, C.; Webber, E.; Tsaousidou, E.; Xue, S.Y.; Brüning, J.C.; Krashes, M.J. Hunger-driven motivational state competition. Neuron 2016, 92, 187–201. [Google Scholar] [CrossRef] [PubMed]
- Jikomes, N.; Ramesh, R.N.; Mandelblat-Cerf, Y.; Andermann, M.L. Preemptive stimulation of AgRP neurons in fed mice enables conditioned food seeking under threat. Curr. Biol. 2016, 26, 2500–2507. [Google Scholar] [CrossRef]
- Dietrich, M.O.; Zimmer, M.R.; Bober, J.; Horvath, T.L. Hypothalamic AgRP neurons drive stereotypic behaviors beyond feeding. Cell 2015, 160, 1222–1232. [Google Scholar] [CrossRef]
- Paxinos, G.; Franklin, K. Paxinos and Franklin’s the Mouse Brain in Stereotaxic Coordinates, 4th ed.; Academic Press: Cambridge, MA, USA, 2012. [Google Scholar]
- Reissner, K.J.; Sartor, G.C.; Vazey, E.M.; Dunn, T.E.; Aston-Jones, G.; Kalivas, P.W. Use of vivo-morpholinos for control of protein expression in the adult rat brain. J. Neurosci. Methods 2012, 203, 354–360. [Google Scholar] [CrossRef][Green Version]
- Séi, H.; Oishi, K.; Chikahisa, S.; Kitaoka, K.; Takeda, E.; Ishida, N. Diurnal amplitudes of arterial pressure and heart rate are dampened in Clock mutant mice and adrenalectomized mice. Endocrinology 2008, 149, 3576–3580. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Mueller, A.D.; Parfyonov, M.; Pavlovski, I.; Marchant, E.G.; Mistlberger, R.E. The inhibitory effect of sleep deprivation on cell proliferation in the hippocampus of adult mice is eliminated by corticosterone clamp combined with interleukin-1 receptor 1 knockout. Brain Behav. Immun. 2014, 35, 182–188. [Google Scholar] [CrossRef] [PubMed]
- Chikahisa, S.; Tominaga, K.; Kawai, T.; Kitaoka, K.; Oishi, K.; Ishida, N.; Rokutan, K.; Séi, H. Bezafibrate, a peroxisome proliferator-activated receptors agonist, decreases body temperature and enhances electroencephalogram delta-oscillation during sleep in mice. Endocrinology 2008, 149, 5262–5271. [Google Scholar] [CrossRef] [PubMed]
- Saito, M.; Minokoshi, Y.; Shimazu, T. Accelerated norepinephrine turnover in peripheral tissues after ventromedial hypothalamic stimulation in rats. Brain Res. 1989, 481, 298–303. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shiuchi, T.; Otsuka, A.; Shimizu, N.; Chikahisa, S.; Séi, H. Feeding Rhythm-Induced Hypothalamic Agouti-Related Protein Elevation via Glucocorticoids Leads to Insulin Resistance in Skeletal Muscle. Int. J. Mol. Sci. 2021, 22, 10831. https://doi.org/10.3390/ijms221910831
Shiuchi T, Otsuka A, Shimizu N, Chikahisa S, Séi H. Feeding Rhythm-Induced Hypothalamic Agouti-Related Protein Elevation via Glucocorticoids Leads to Insulin Resistance in Skeletal Muscle. International Journal of Molecular Sciences. 2021; 22(19):10831. https://doi.org/10.3390/ijms221910831
Chicago/Turabian StyleShiuchi, Tetsuya, Airi Otsuka, Noriyuki Shimizu, Sachiko Chikahisa, and Hiroyoshi Séi. 2021. "Feeding Rhythm-Induced Hypothalamic Agouti-Related Protein Elevation via Glucocorticoids Leads to Insulin Resistance in Skeletal Muscle" International Journal of Molecular Sciences 22, no. 19: 10831. https://doi.org/10.3390/ijms221910831
APA StyleShiuchi, T., Otsuka, A., Shimizu, N., Chikahisa, S., & Séi, H. (2021). Feeding Rhythm-Induced Hypothalamic Agouti-Related Protein Elevation via Glucocorticoids Leads to Insulin Resistance in Skeletal Muscle. International Journal of Molecular Sciences, 22(19), 10831. https://doi.org/10.3390/ijms221910831





