Transcriptomic Profiling of Adult-Onset Asthma Related to Damp and Moldy Buildings and Idiopathic Environmental Intolerance
Abstract
:1. Introduction
2. Results
2.1. Demographic and Clinical Data
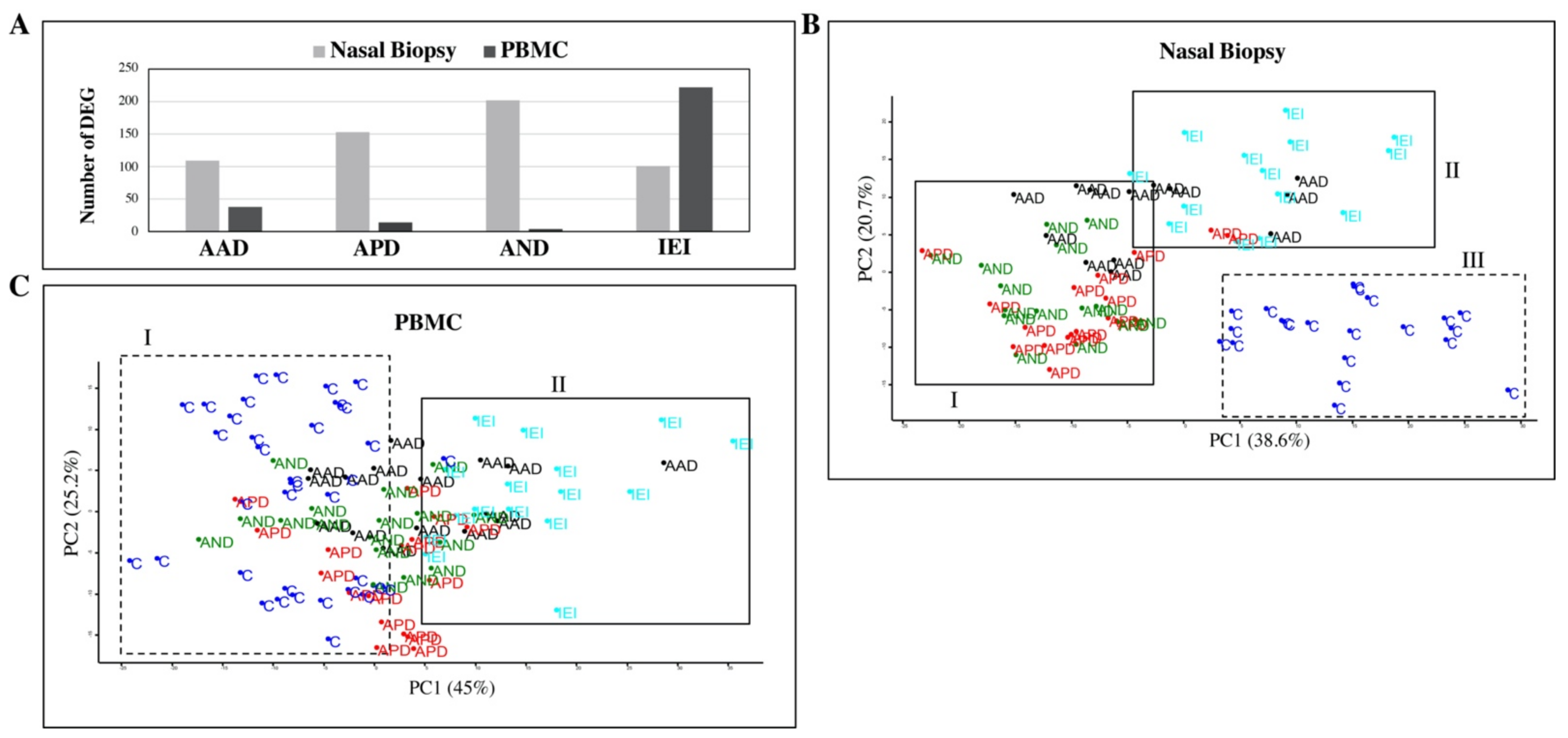
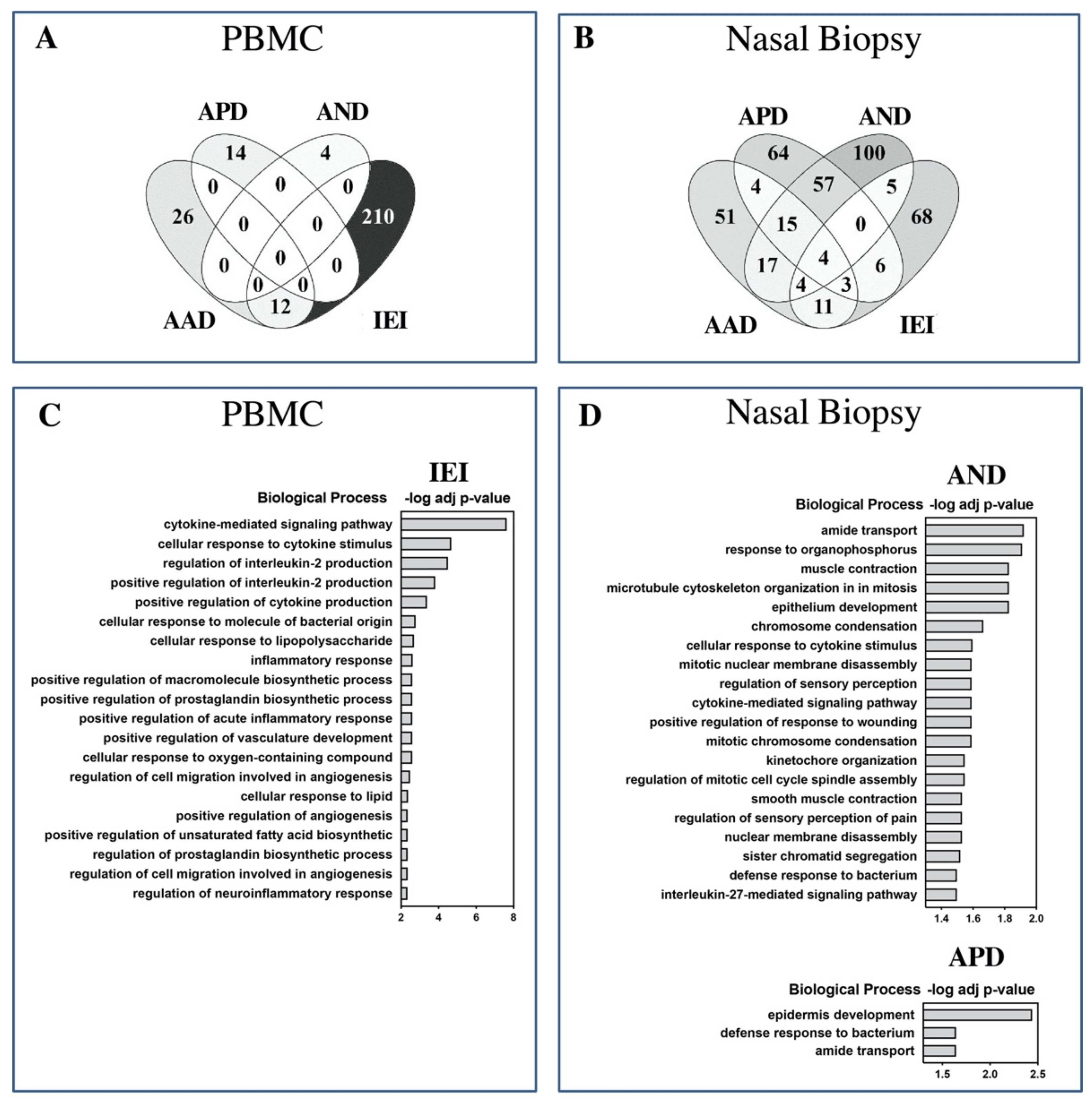
2.2. Inflammatory Cytokine and Gene Expression Profile of Patients and Controls
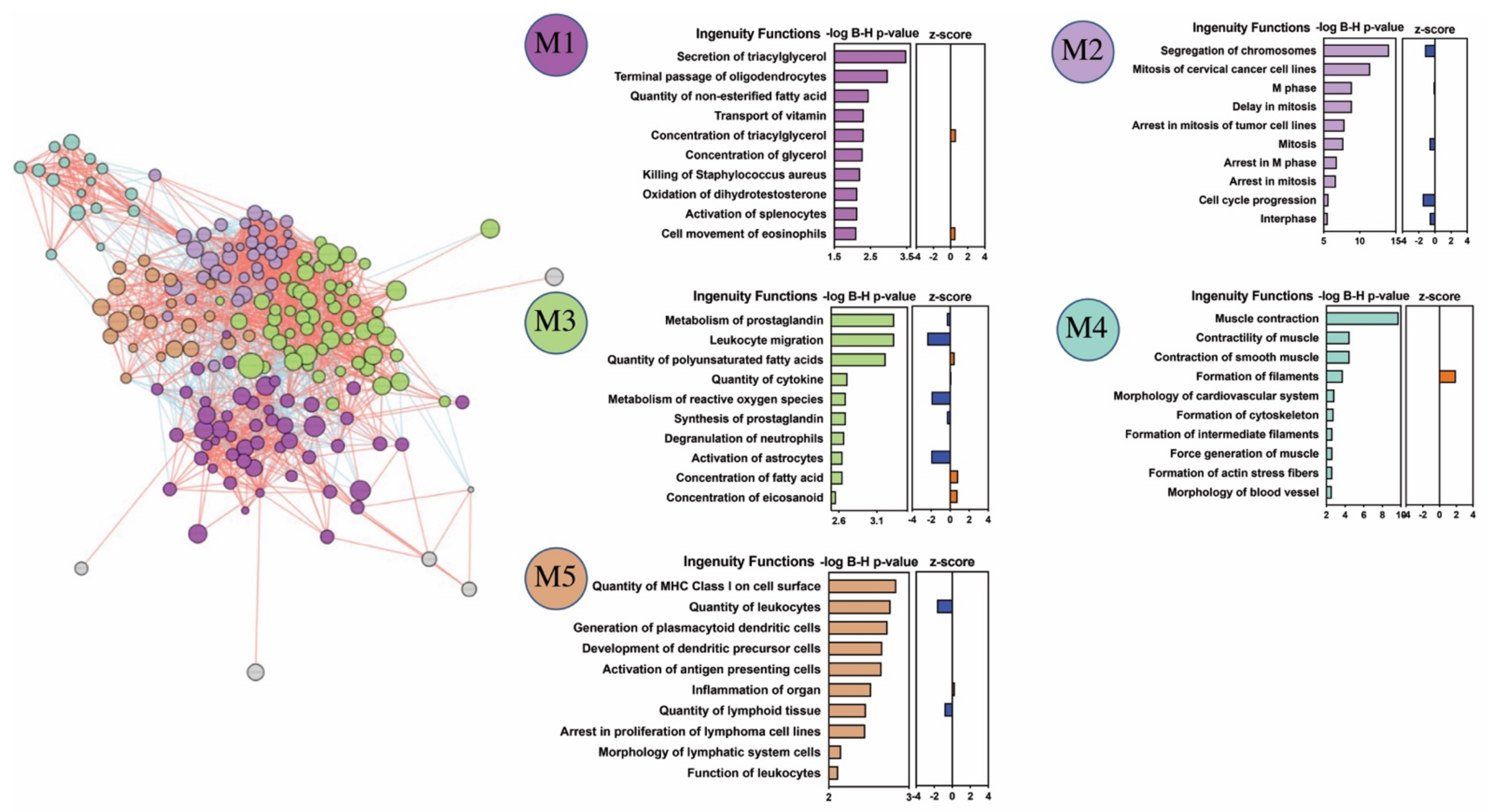
2.3. Biological Significance of Differentially Expressed Genes
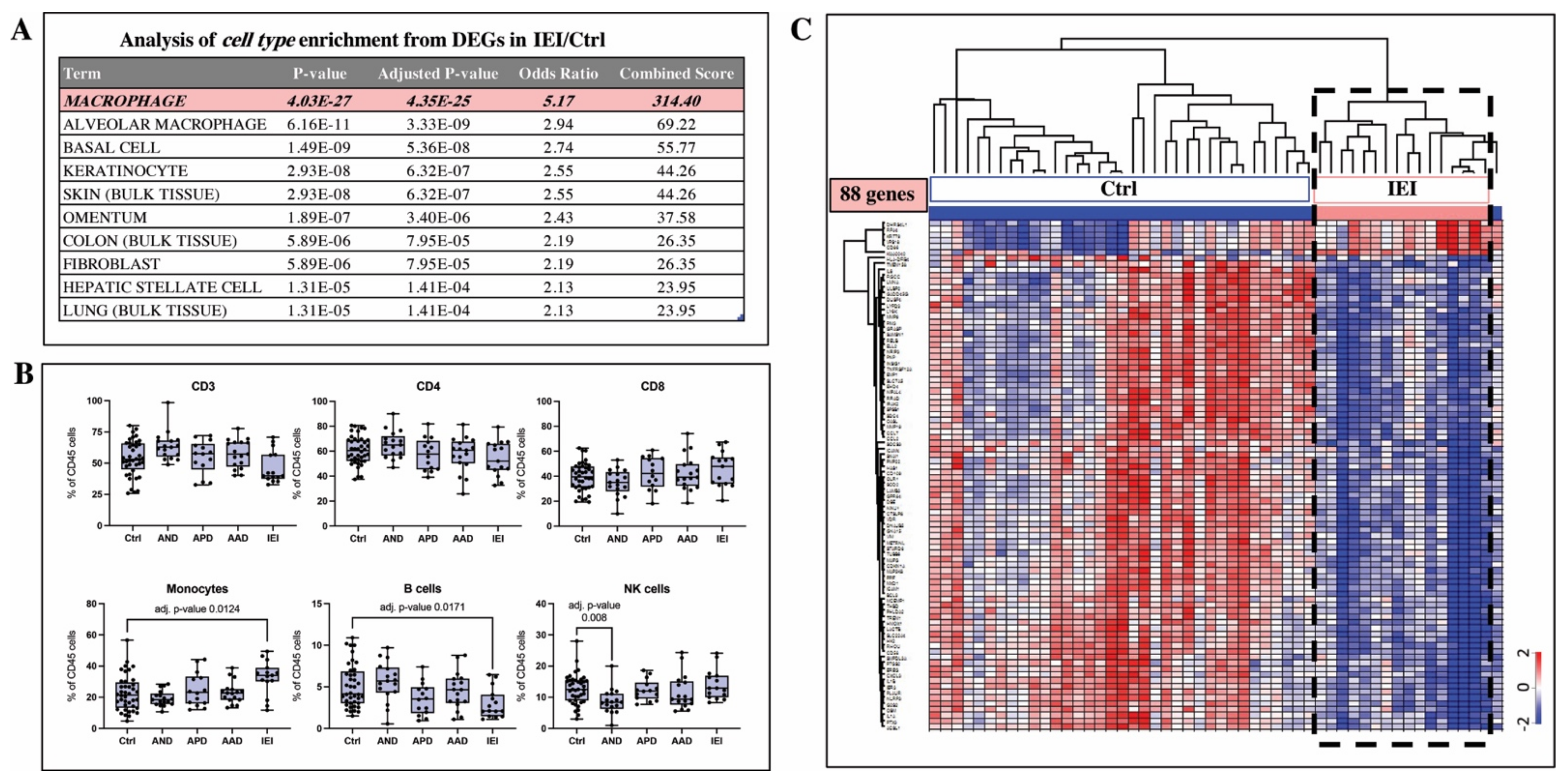
2.4. Cell Type Analysis Based on Gene Expression Signature and Flow Cytometry Indicate Macrophages Are Key Mechanistic Players in IEI Pathobiology
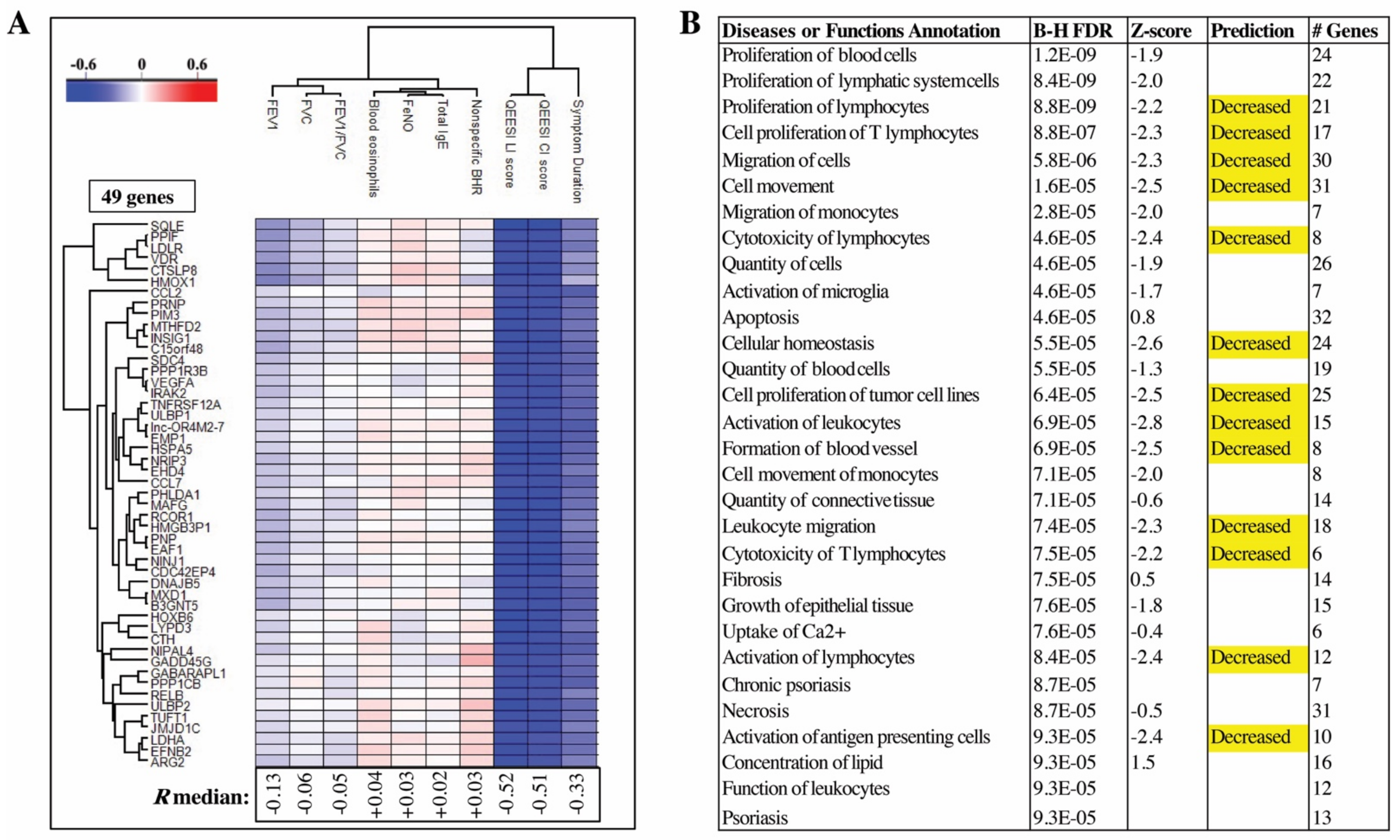
2.5. QEESI Score and Disease Duration Have the Strongest Correlations to DEGs Identified in Blood Cells
3. Discussion
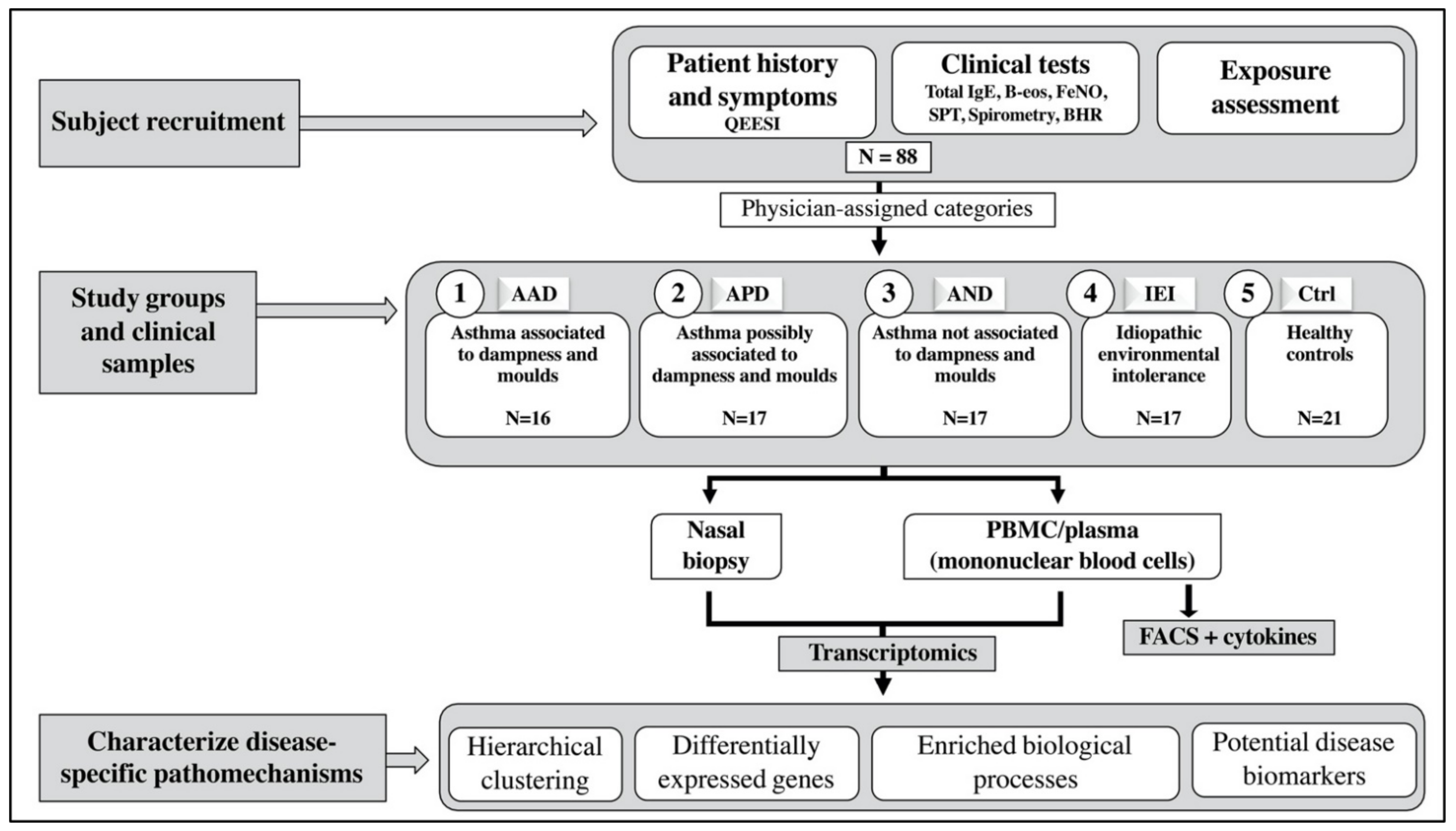
4. Materials and Methods
4.1. Exposure Assessment
4.2. Study Population and Groups
- Asthma associated with dampness and molds (AAD) had ‘substantial exposure to dampness and molds’—during the asthma onset their symptoms deteriorated at the damaged building.
- Asthma possibly associated with dampness and molds (APD) had ‘possible exposure to dampness and molds’—during the asthma onset their symptoms deteriorated at the workplace or at home.
- Asthma not associated with dampness and molds (AND) had ‘no exposure to dampness and molds’ or deterioration of asthma symptoms at home or at the workplace during asthma onset.
- Idiopathic environmental intolerance (IEI) group inclusion criteria were modified from WHO’s IEI criteria [16] and that of Lacour et al. [44]: (1) respiratory symptoms, (2) symptoms associated environmental factors in low exposure levels which do not cause symptoms to the majority of people, (3) symptoms from several organ systems, (4) recurrent symptoms related to certain environments that diminish or end when the factor related to symptoms is removed, (5) the condition had lasted at least 6 months and causes major living restrictions in ability of function, (6) symptoms cannot be explained by any known somatic or psychiatric disease, (7) the person experienced that the symptoms are caused by environmental factors. Asthma patients were not excluded.
- Healthy controls (Ctrl) had no allergic symptoms related to common environmental allergens, asthma, or chronic cough or dyspnea. They had no known long-term working or living in a building with dampness or mold damage. They did not fulfill the criteria of IEI.
4.3. Inhaled Steroid Course for Healthy Controls
4.4. Sampling
4.5. PBMC Separation and Flow Cytometry
4.6. Cytokine Profiling by Luminex Assay
4.7. Transcriptomics
4.8. Data Analysis
4.9. Pathway Enrichment Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kaur, R.; Chupp, G. Phenotypes and endotypes of adult asthma: Moving toward precision medicine. J. Allergy Clin. Immunol. 2019, 144, 1–12. [Google Scholar] [CrossRef]
- De Nijs, S.B.; Venekamp, L.N.; Bel, E.H. Adult-onset asthma: Is it really different? Eur. Respir. Rev. 2013, 22, 44–52. [Google Scholar] [CrossRef]
- Tuomisto, L.E.; Ilmarinen, P.; Kankaanranta, H. Prognosis of new-onset asthma diagnosed at adult age. Respir. Med. 2015, 109, 944–954. [Google Scholar] [CrossRef]
- Wang, J.; Pindus, M.; Janson, C.; Sigsgaard, T.; Kim, J.-L.; Holm, M.; Sommar, J.; Orru, H.; Gislason, T.; Johannessen, A.; et al. Dampness, mould, onset and remission of adult respiratory symptoms, asthma and rhinitis. Eur. Respir. J. 2019, 53, 1801921. [Google Scholar] [CrossRef] [PubMed]
- Karvala, K.; Toskala, E.; Luukkonen, R.; Lappalainen, S.; Uitti, J.; Nordman, H. New-onset adult asthma in relation to damp and moldy workplaces. Int. Arch. Occup. Environ. Health 2010, 83, 855–865. [Google Scholar] [CrossRef] [PubMed]
- Karvala, K.; Uitti, J.; Luukkonen, R.; Nordman, H. Quality of life of patients with asthma related to damp and moldy work environments. Scand. J. Work Environ. Health 2012, 39, 96–105. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lessof, M. Report of Multiple Chemical Sensitivities (MCS) Workshop, Berlin, Germany, 21–23 February 1996. PCS/96.29 IPCS, Geneva, Switzerland. Hum. Exp. Toxicol. 1997, 16, 233–234. [Google Scholar] [CrossRef] [PubMed]
- Hetherington, L.H.; Battershill, J.M. Review of evidence for a toxicological mechanism of idiopathic environmental intolerance. Hum. Exp. Toxicol. 2012, 32, 3–17. [Google Scholar] [CrossRef]
- Nordin, S. Mechanisms underlying nontoxic indoor air health problems: A review. Int. J. Hyg. Environ. Health 2020, 226, 113489. [Google Scholar] [CrossRef]
- Karvala, K.; Sainio, M.; Palmquist, E.; Nyback, M.H.; Nordin, S. Prevalence of various environmental intolerances in a Swedish and Finnish general population. Environ. Res. 2018, 161, 220–228. [Google Scholar] [CrossRef]
- Karvala, K.; Sainio, M.; Palmquist, E.; Claeson, A.S.; Nyback, M.H.; Nordin, S. Building-Related Environmental Intolerance and Associated Health in the General Population. Int. J. Environ. Res. Public Health 2018, 15, 2047. [Google Scholar] [CrossRef] [Green Version]
- Lind, N.; Söderholm, A.; Palmquist, E.; Andersson, L.; Millqvist, E.; Nordin, S. Comorbidity and Multimorbidity of Asthma and Allergy and Intolerance to Chemicals and Certain Buildings. J. Occup. Environ. Med. 2017, 59, 80–84. [Google Scholar] [CrossRef]
- Chung, K.F.; Wenzel, S.E.; Brozek, J.L.; Bush, A.; Castro, M.; Sterk, P.J.; Adcock, I.M.; Bateman, E.D.; Bel, E.H.; Bleecker, E.R.; et al. International ERS/ATS guidelines on definition, evaluation and treatment of severe asthma. Eur. Respir. J. 2014, 43, 343–373. [Google Scholar] [CrossRef] [Green Version]
- Lachmann, A.; Torre, D.; Keenan, A.B.; Jagodnik, K.M.; Lee, H.J.; Wang, L.; Silverstein, M.C.; Ma’ayan, A. Massive mining of publicly available RNA-seq data from human and mouse. Nat. Commun. 2018, 9, 1366. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sze, E.; Bhalla, A.; Nair, P. Mechanisms and therapeutic strategies for non-T2 asthma. Allergy 2020, 75, 311–325. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- IPCS/WHO. Conclusions and recommendations of a workshop on multiple chemical sensitivities (MCS). Regul. Toxicol. Pharm. 1996, 24, 188–189. [Google Scholar] [CrossRef]
- Ilmarinen, P.; Tuomisto, L.E.; Niemela, O.; Tommola, M.; Haanpaa, J.; Kankaanranta, H. Cluster Analysis on Longitudinal Data of Patients with Adult-Onset Asthma. J. Allergy Clin. Immunol. Pract. 2017, 5, 967–978.e963. [Google Scholar] [CrossRef] [PubMed]
- Kuruvilla, M.E.; Lee, F.E.; Lee, G.B. Understanding Asthma Phenotypes, Endotypes, and Mechanisms of Disease. Clin. Rev. Allergy Immunol. 2019, 56, 219–233. [Google Scholar] [CrossRef] [PubMed]
- Skovbjerg, S.; Berg, N.D.; Elberling, J.; Christensen, K.B. Evaluation of the quick environmental exposure and sensitivity inventory in a Danish population. J. Environ. Public Health 2012, 2012, 304314. [Google Scholar] [CrossRef] [PubMed]
- Nordin, S.; Andersson, L. Evaluation of a Swedish version of the Quick Environmental Exposure and Sensitivity Inventory. Int. Arch. Occup. Environ. Health 2010, 83, 95–104. [Google Scholar] [CrossRef]
- Drizik, E.; Corbett, S.; Zheng, Y.; Vermeulen, R.; Dai, Y.; Hu, W.; Ren, D.; Duan, H.; Niu, Y.; Xu, J.; et al. Transcriptomic changes in the nasal epithelium associated with diesel engine exhaust exposure. Environ. Int. 2020, 137, 105506. [Google Scholar] [CrossRef]
- Matsuoka, H.; Niimi, A.; Matsumoto, H.; Ueda, T.; Takemura, M.; Yamaguchi, M.; Jinnai, M.; Chang, L.; Otsuka, K.; Oguma, T.; et al. Patients’ characteristics associated with unsuccessful sputum induction in asthma. J. Allergy Clin. Immunol. 2008, 121, 774–776. [Google Scholar] [CrossRef]
- Laan, M.; Lotvall, J.; Chung, K.F.; Linden, A. IL-17-induced cytokine release in human bronchial epithelial cells in vitro: Role of mitogen-activated protein (MAP) kinases. Br. J. Pharm. 2001, 133, 200–206. [Google Scholar] [CrossRef] [Green Version]
- Yokoyama, A.; Kohno, N.; Fujino, S.; Hamada, H.; Inoue, Y.; Fujioka, S.; Ishida, S.; Hiwada, K. Circulating interleukin-6 levels in patients with bronchial asthma. Am. J. Respir. Crit. Care Med. 1995, 151, 1354–1358. [Google Scholar] [CrossRef]
- Virchow, J.C., Jr.; Kroegel, C.; Walker, C.; Matthys, H. Inflammatory determinants of asthma severity: Mediator and cellular changes in bronchoalveolar lavage fluid of patients with severe asthma. J. Allergy Clin. Immunol. 1996, 98, S27–S33, discussion S33–S40. [Google Scholar] [CrossRef]
- Mitchell, C.S.; Donnay, A.; Hoover, D.R.; Margolick, J.B. Immunologic parameters of multiple chemical sensitivity. Occup. Med. 2000, 15, 647–665. [Google Scholar] [PubMed]
- De Luca, C.; Scordo, M.G.; Cesareo, E.; Pastore, S.; Mariani, S.; Maiani, G.; Stancato, A.; Loreti, B.; Valacchi, G.; Lubrano, C.; et al. Biological definition of multiple chemical sensitivity from redox state and cytokine profiling and not from polymorphisms of xenobiotic-metabolizing enzymes. Toxicol. Appl. Pharm. 2010, 248, 285–292. [Google Scholar] [CrossRef] [PubMed]
- Dantoft, T.M.; Elberling, J.; Brix, S.; Szecsi, P.B.; Vesterhauge, S.; Skovbjerg, S. An elevated pro-inflammatory cytokine profile in multiple chemical sensitivity. Psychoneuroendocrinology 2014, 40, 140–150. [Google Scholar] [CrossRef] [PubMed]
- Bigler, J.; Boedigheimer, M.; Schofield, J.P.R.; Skipp, P.J.; Corfield, J.; Rowe, A.; Sousa, A.R.; Timour, M.; Twehues, L.; Hu, X.; et al. A Severe Asthma Disease Signature from Gene Expression Profiling of Peripheral Blood from U-BIOPRED Cohorts. Am. J. Respir. Crit. Care Med. 2017, 195, 1311–1320. [Google Scholar] [CrossRef] [PubMed]
- Doeing, D.C.; Solway, J. Airway smooth muscle in the pathophysiology and treatment of asthma. J. Appl. Physiol. 2013, 114, 834–843. [Google Scholar] [CrossRef] [Green Version]
- Zuyderduyn, S.; Sukkar, M.B.; Fust, A.; Dhaliwal, S.; Burgess, J.K. Treating asthma means treating airway smooth muscle cells. Eur. Respir. J. 2008, 32, 265–274. [Google Scholar] [CrossRef]
- Alcala, S.E.; Benton, A.S.; Watson, A.M.; Kureshi, S.; Reeves, E.M.; Damsker, J.; Wang, Z.; Nagaraju, K.; Anderson, J.; Williams, A.M.; et al. Mitotic asynchrony induces transforming growth factor-beta1 secretion from airway epithelium. Am. J. Respir. Cell Mol. Biol. 2014, 51, 363–369. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Freishtat, R.J.; Watson, A.M.; Benton, A.S.; Iqbal, S.F.; Pillai, D.K.; Rose, M.C.; Hoffman, E.P. Asthmatic airway epithelium is intrinsically inflammatory and mitotically dyssynchronous. Am. J. Respir. Cell Mol. Biol. 2011, 44, 863–869. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.N.; Su, R.N.; Yang, B.Y.; Yang, K.X.; Yang, L.F.; Yan, Y.; Chen, Z.G. Potential Role of Cellular Senescence in Asthma. Front. Cell Dev. Biol. 2020, 8, 59. [Google Scholar] [CrossRef]
- Ndika, J.; Suojalehto, H.; Taubel, M.; Lehto, M.; Karvala, K.; Pallasaho, P.; Sund, J.; Auvinen, P.; Jarvi, K.; Pekkanen, J.; et al. Nasal mucosa and blood cell transcriptome profiles do not reflect respiratory symptoms associated with moisture damage. Indoor Air 2018, 28, 721–731. [Google Scholar] [CrossRef]
- Van den Bergh, O.; Witthoft, M.; Petersen, S.; Brown, R.J. Symptoms and the body: Taking the inferential leap. Neurosci. Biobehav. Rev. 2017, 74, 185–203. [Google Scholar] [CrossRef] [Green Version]
- Segerstrom, S.C.; Miller, G.E. Psychological stress and the human immune system: A meta-analytic study of 30 years of inquiry. Psychol. Bull. 2004, 130, 601–630. [Google Scholar] [CrossRef] [Green Version]
- Tian, R.; Hou, G.; Li, D.; Yuan, T.F. A possible change process of inflammatory cytokines in the prolonged chronic stress and its ultimate implications for health. Sci. World J. 2014, 2014, 780616. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.; Woo, J.K.; Jang, Y.-S.; Kang, J.-H.; Hwang, J.-I.; Seong, J.K.; Yoon, Y.S.; Oh, S.H. Ninjurin1 Plays a Crucial Role in Pulmonary Fibrosis by Promoting Interaction between Macrophages and Alveolar Epithelial Cells. Sci. Rep. 2018, 8, 17542. [Google Scholar] [CrossRef]
- Ifergan, I.; Kebir, H.; Terouz, S.; Alvarez, J.I.; Lécuyer, M.-A.; Gendron, S.; Bourbonnière, L.; Dunay, I.R.; Bouthillier, A.; Moumdjian, R.; et al. Role of ninjurin-1 in the migration of myeloid cells to central nervous system inflammatory lesions. Ann. Neurol. 2011, 70, 751–763. [Google Scholar] [CrossRef] [PubMed]
- Jennewein, C.; Sowa, R.; Faber, A.C.; Dildey, M.; von Knethen, A.; Meybohm, P.; Scheller, B.; Dröse, S.; Zacharowski, K. Contribution of Ninjurin1 to Toll-Like Receptor 4 Signaling and Systemic Inflammation. Am. J. Respir. Cell Mol. Biol. 2015, 53, 656–663. [Google Scholar] [CrossRef]
- Padyana, A.K.; Gross, S.; Jin, L.; Cianchetta, G.; Narayanaswamy, R.; Wang, F.; Wang, R.; Fang, C.; Lv, X.; Biller, S.A.; et al. Structure and inhibition mechanism of the catalytic domain of human squalene epoxidase. Nat. Commun. 2019, 10, 97. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tahtinen, K.; Lappalainen, S.; Karvala, K.; Remes, J.; Salonen, H. Association between Four-Level Categorisation of Indoor Exposure and Perceived Indoor Air Quality. Int. J. Environ. Res. Public Health 2018, 15, 679. [Google Scholar] [CrossRef] [Green Version]
- Lacour, M.; Zunder, T.; Schmidtke, K.; Vaith, P.; Scheidt, C. Multiple chemical sensitivity syndrome (MCS)--suggestions for an extension of the U.S. MCS-case definition. Int. J. Hyg. Environ. Health 2005, 208, 141–151. [Google Scholar] [CrossRef]
- Miller, C.S.; Prihoda, T.J. The Environmental Exposure and Sensitivity Inventory (EESI): A standardized approach for measuring chemical intolerances for research and clinical applications. Toxicol. Ind. Health 1999, 15, 370–385. [Google Scholar] [CrossRef] [PubMed]
- Marwah, V.S.; Scala, G.; Kinaret, P.A.S.; Serra, A.; Alenius, H.; Fortino, V.; Greco, D. eUTOPIA: SolUTion for Omics data PreprocessIng and Analysis. Source Code Biol. Med. 2019, 14, 1. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ritchie, M.E.; Phipson, B.; Wu, D.; Hu, Y.; Law, C.W.; Shi, W.; Smyth, G.K. limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res. 2015, 43, e47. [Google Scholar] [CrossRef]
- Kallio, M.A.; Tuimala, J.T.; Hupponen, T.; Klemelä, P.; Gentile, M.; Scheinin, I.; Koski, M.; Käki, J.; Korpelainen, E.I. Chipster: User-friendly analysis software for microarray and other high-throughput data. BMC Genom. 2011, 12, 507. [Google Scholar] [CrossRef] [Green Version]
- Tyanova, S.; Temu, T.; Sinitcyn, P.; Carlson, A.; Hein, M.Y.; Geiger, T.; Mann, M.; Cox, J. The Perseus computational platform for comprehensive analysis of (prote)omics data. Nat. Methods 2016, 13, 731–740. [Google Scholar] [CrossRef]
- Marwah, V.; Kinaret, P.; Serra, A.; Scala, G.; Lauerma, A.; Fortino, V.; Greco, D. INfORM: Inference of NetwOrk Response Modules. Bioinformatic 2018, 34, 2136–2138. [Google Scholar] [CrossRef] [Green Version]

| Asthma Associated with Dampness and Molds (AAD) n = 16 | Asthma Possibly Associated with Dampness and Molds (APD) n = 17 | Asthma Not Associated with Dampness and Molds (AND) n = 17 | Idiopathic Environmental Intolerance (IEI) a n = 17 | Healthy Controls (Ctrl) n = 21 | p-Value | |
|---|---|---|---|---|---|---|
| Age, years, md (IQR) | 47.3 (40.9–53.4) | 50.5 (43.7–55.7) | 51.8 (38.5–57.4) | 50.3 (44.4–55.1) | 41.2 (32.8–47.0) | 0.021 |
| Body mass index, kg/m2, md (IQR) | 24.9 (24.1–30.4) | 27.3 (24.0–32.9) | 29.0 (26.0–34.5) | 25.1 (23.7–27.3) | 25.5 (21.1–28.7) | 0.060 |
| Ex-smoker, n (%) | 3 (19) | 6 (35) | 4 (24) | 3 (18) | 1 (10) | 0.399 |
| Atopy b, n (%) | 3 (19) | 4 (24) | 1 (6) | 6 (35) | 7 (33) | 0.238 |
| Duration of respiratory symptoms, years, md (IQR) | 3.0 (2.0–5.0) | 5.0 (1.5–12.5) | 8.0 (2.0–16.5) | 5.0 (2.5–10.5) | NA | 0.122 |
| Use of inhaled steroid within one month, n (%) | 16 (100) | 16 (94) | 17 (100) | 10 (59) | 0 (0) | <0.001 |
| Inhaled steroid dose, n (%) c | 0.786 | |||||
| Low | 5 (31) | 2 (13) | 2 (12) | 2 (20) | NA | |
| Medium | 6 (37) | 6 (38) | 7 (41) | 3 (30) | NA | |
| High | 5 (31) | 8 (50) | 8 (47) | 5 (50) | NA | |
| Daily use of short-acting β2-agonist, n (%) | 0 (0) | 1 (6) | 2 (12) | 1 (17) | NA | 0.696 |
| Severe asthma d, n (%) | 3 (19) | 4 (24) | 5 (29) | 1 (17) | NA | 0.875 |
| Chronic rhinitis, n (%) | 11 (69) | 11 (65) | 10 (59) | 10 (59) | 3 (14) | 0.004 |
| QEESI chemical intolerance score, md (IQR), n = 87 | 39 (24–53) | 34 (19–55) | 29 (9–71) | 76 (39–85) | 5 (3–15) | <0.001 |
| QEESI symptom score, md (IQR) | 42 (16–61) | 35 (15–44) | 29 (17–47) | 51 (32–78) | 6 (3–13) | <0.001 |
| QEESI life impact score, md (IQR) | 23 (4–52) | 22 (9–40) | 15 (15–38) | 64 (43–88) | 2 (0–7) | <0.001 |
| Total IgE kU/L, md (IQR) | 27 (15–78) | 14 (7–41) | 26 (13–69) | 20 (12–52) | 22 (16–35) | 0.690 |
| Blood eosinophils /µL, md (IQR) | 175 (93–215) | 130 (95–220) | 180 (120–250) | 120 (120–175) | 160 (110–210) | 0.918 |
| Fractional exhaled nitric oxide, md (IQR) | 13.5 (11.5–22.1) | 17.5 (12.3–23.5) | 14.5 (9.0–30.5) | 11.5 (9.1–20.6) | 12.0 (9.3–16.3) | 0.445 |
| FEV1 % pred, md (IQR) | 91 (86–97) | 84 (78–100) | 85 (70–95) | 95 (88–103) | 89 (83–98) | 0.093 |
| FVC % pred, md (IQR) | 93 (86–101) | 92 (79–102) | 86 (78–81) | 96 (94–103) | 95 (86–105) | 0.033 |
| FEV1/FVC, md (IQR) | 78 (75–84) | 78 (73–83) | 80 (75–81) | 81 (76–84) | 79 (76–82) | 0.671 |
| Nonspecific bronchial hyperresponsiveness, n (%), n = 84 | 0.029 | |||||
| Moderate | 4 (29) | 3 (18) | 1 (7) | 1 (6) | 0 (0) | |
| Mild | 2 (14) | 5 (29) | 3 (20) | 4 (24) | 0 (0) | |
| No | 8 (57) | 9 (53) | 11 (73) | 12 (71) | 21 (100) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Suojalehto, H.; Ndika, J.; Lindström, I.; Airaksinen, L.; Karvala, K.; Kauppi, P.; Lauerma, A.; Toppila-Salmi, S.; Karisola, P.; Alenius, H. Transcriptomic Profiling of Adult-Onset Asthma Related to Damp and Moldy Buildings and Idiopathic Environmental Intolerance. Int. J. Mol. Sci. 2021, 22, 10679. https://doi.org/10.3390/ijms221910679
Suojalehto H, Ndika J, Lindström I, Airaksinen L, Karvala K, Kauppi P, Lauerma A, Toppila-Salmi S, Karisola P, Alenius H. Transcriptomic Profiling of Adult-Onset Asthma Related to Damp and Moldy Buildings and Idiopathic Environmental Intolerance. International Journal of Molecular Sciences. 2021; 22(19):10679. https://doi.org/10.3390/ijms221910679
Chicago/Turabian StyleSuojalehto, Hille, Joseph Ndika, Irmeli Lindström, Liisa Airaksinen, Kirsi Karvala, Paula Kauppi, Antti Lauerma, Sanna Toppila-Salmi, Piia Karisola, and Harri Alenius. 2021. "Transcriptomic Profiling of Adult-Onset Asthma Related to Damp and Moldy Buildings and Idiopathic Environmental Intolerance" International Journal of Molecular Sciences 22, no. 19: 10679. https://doi.org/10.3390/ijms221910679
APA StyleSuojalehto, H., Ndika, J., Lindström, I., Airaksinen, L., Karvala, K., Kauppi, P., Lauerma, A., Toppila-Salmi, S., Karisola, P., & Alenius, H. (2021). Transcriptomic Profiling of Adult-Onset Asthma Related to Damp and Moldy Buildings and Idiopathic Environmental Intolerance. International Journal of Molecular Sciences, 22(19), 10679. https://doi.org/10.3390/ijms221910679






