Comparative Transcriptome Analysis of Chemoreception Organs of Laodelphax striatellus in Response to Rice Stripe Virus Infection
Abstract
:1. Introduction
2. Results
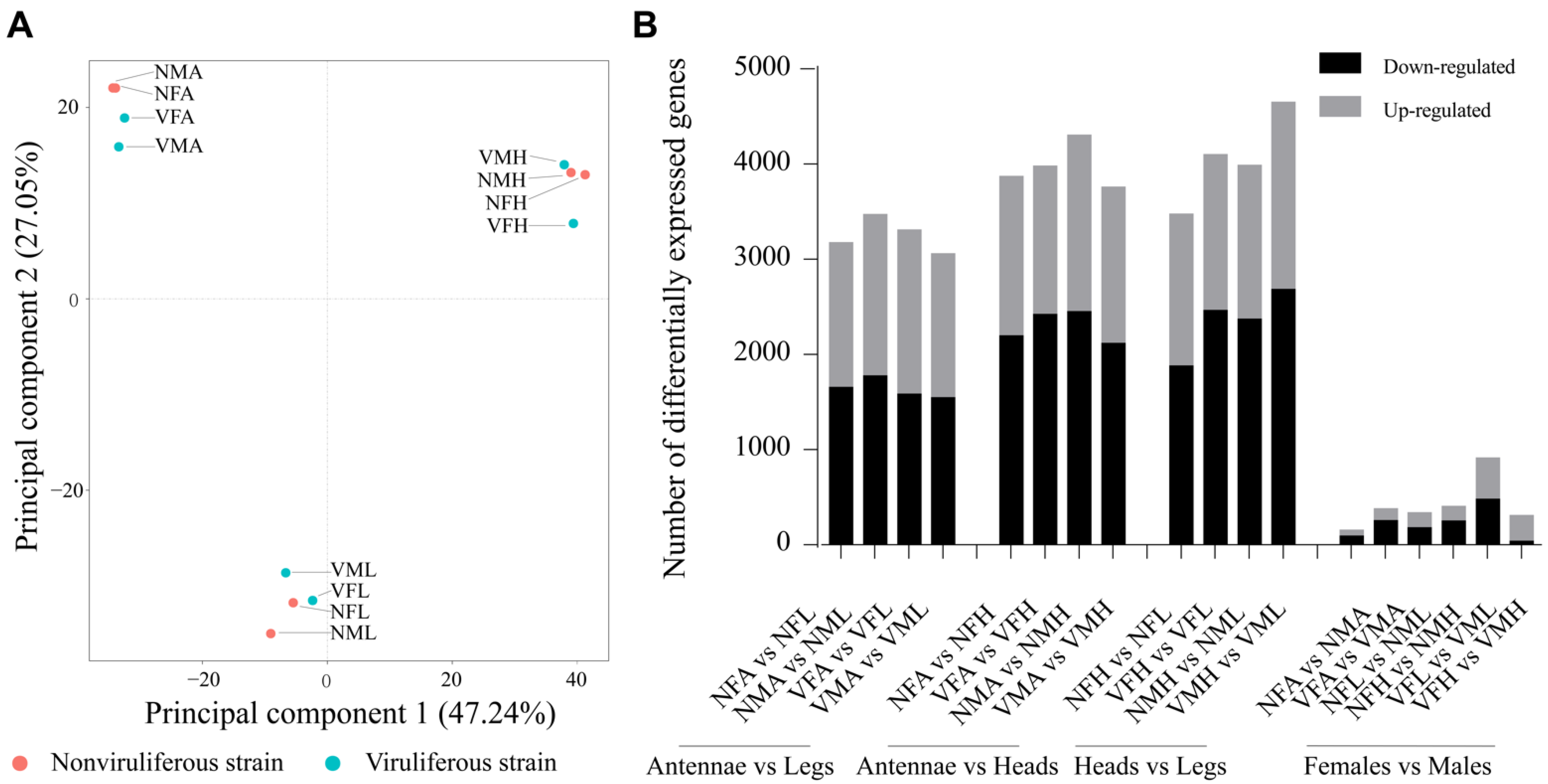
2.1. Illumina Sequencing and Assembly of the Chemoreception Organs of Female and Male Laodelphax striatellus
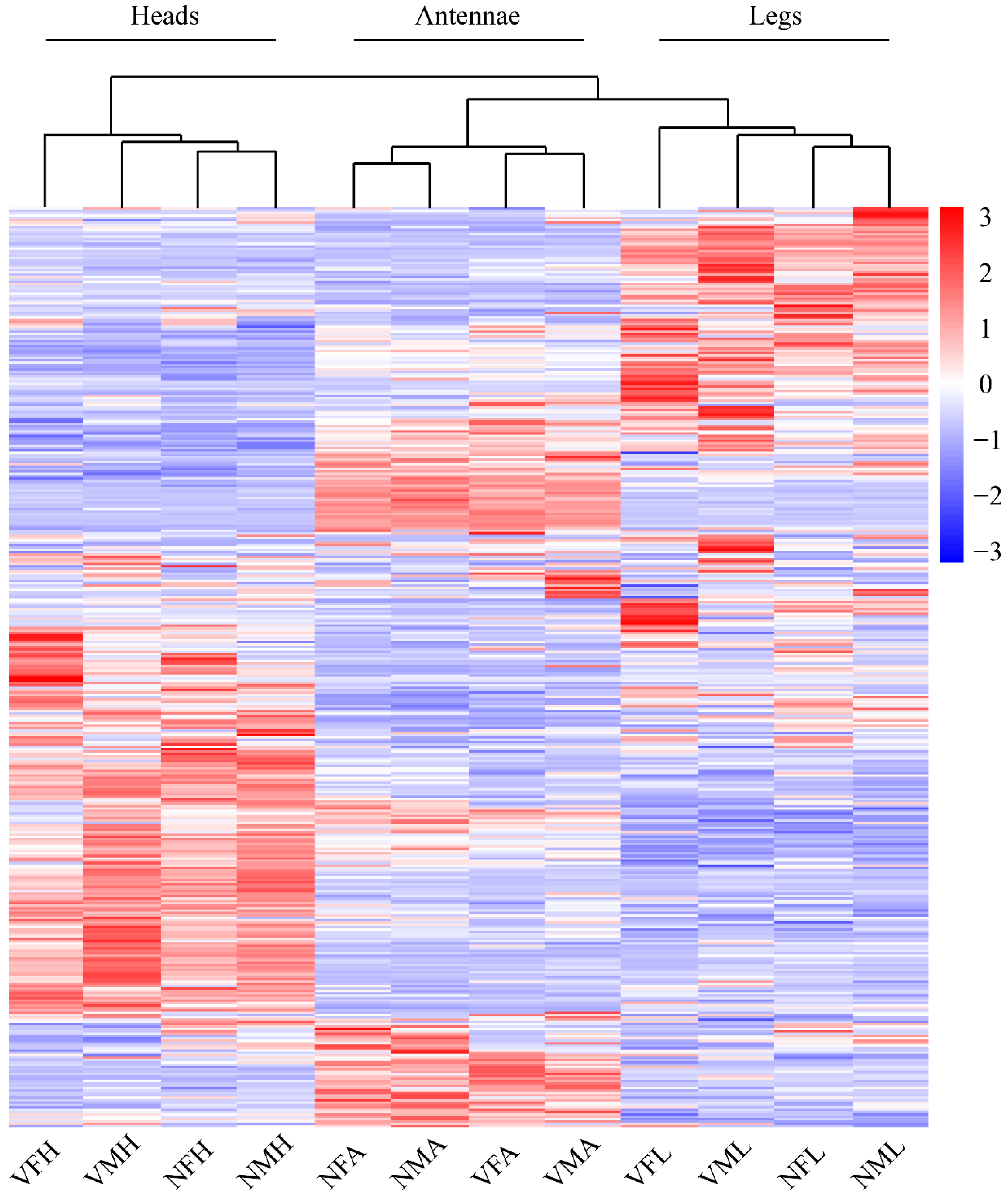
2.2. Gene Ontology Classification of Differentially Expressed Genes among the Chemoreception Organs of Females and Males
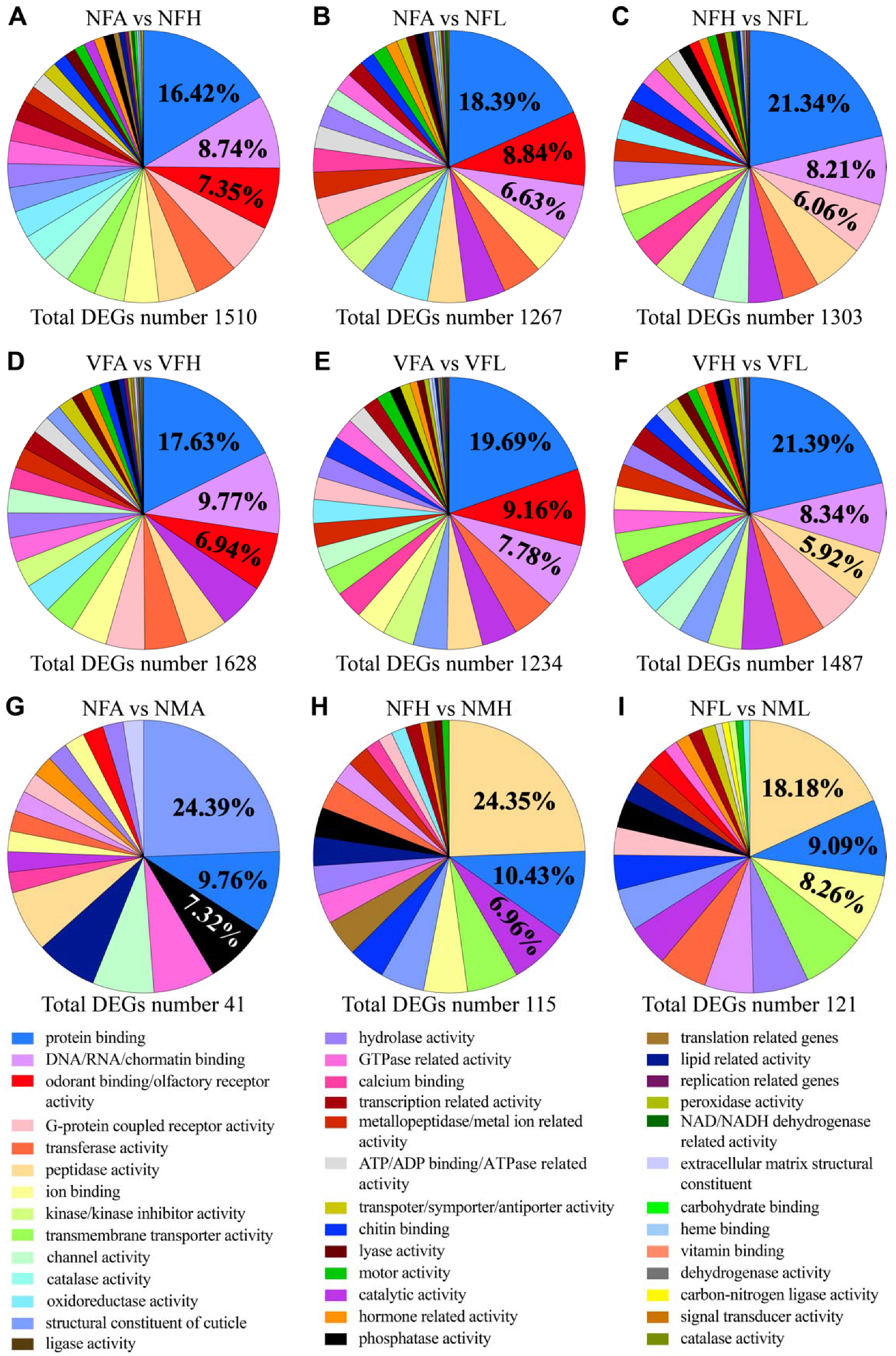
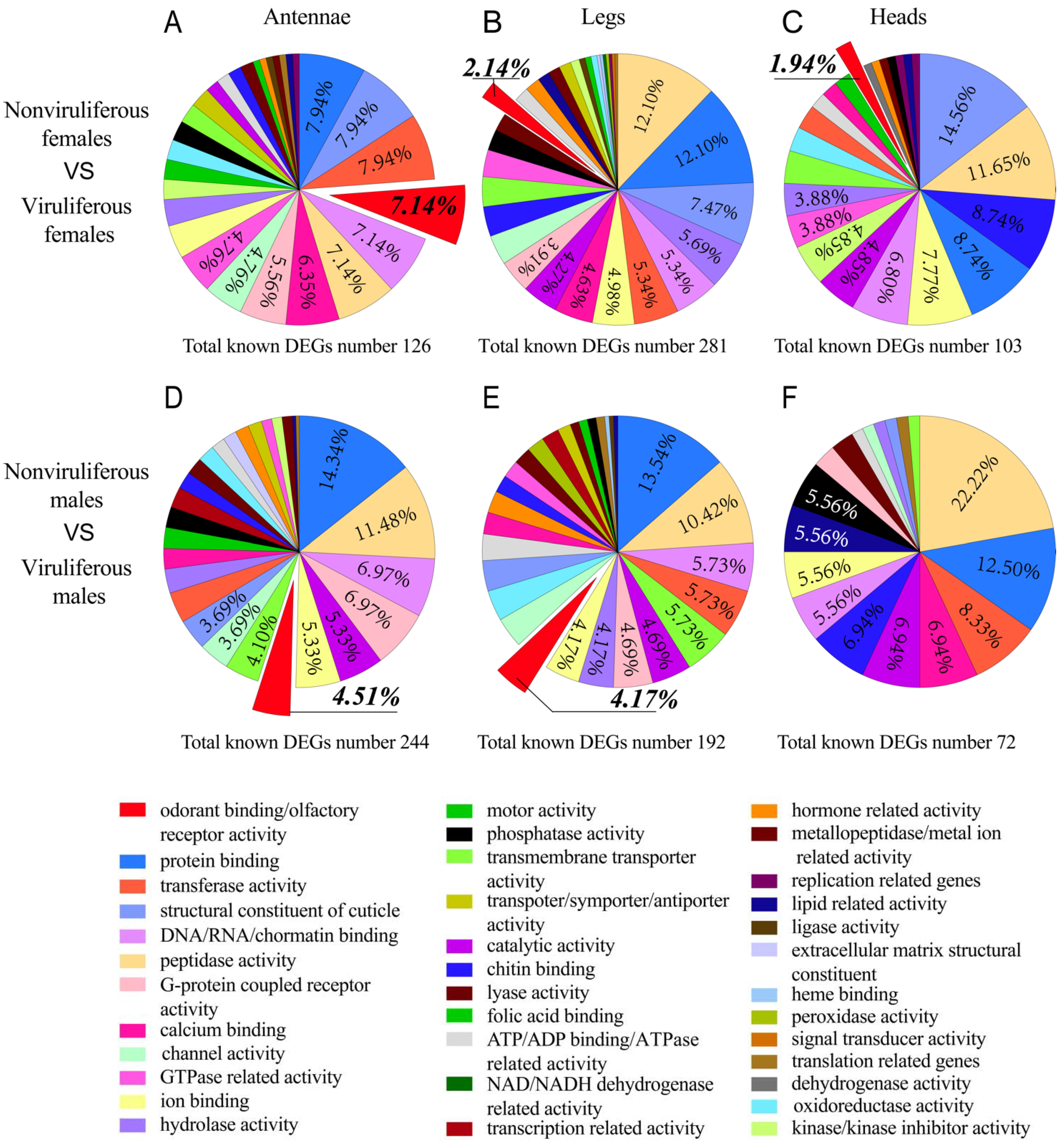
2.3. Differentially Expressed Genes Induced by RSV Infection
2.4. Gene Ontology Classification of Differentially Expressed Genes Induced by RSV Infection
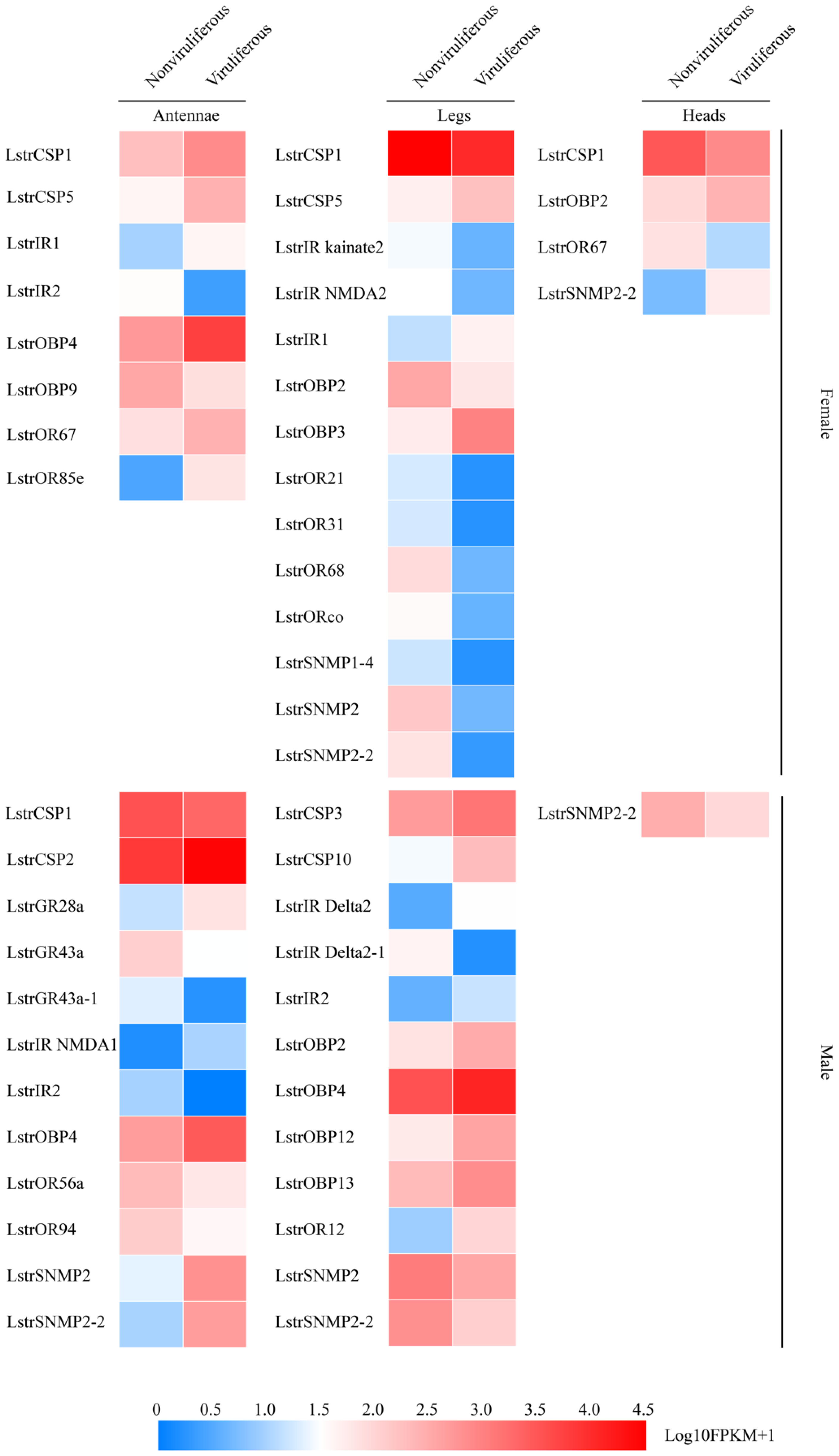
2.5. Changes in the Expression of Olfactory-Related Genes in the Chemoreception Organs Infected by RSV
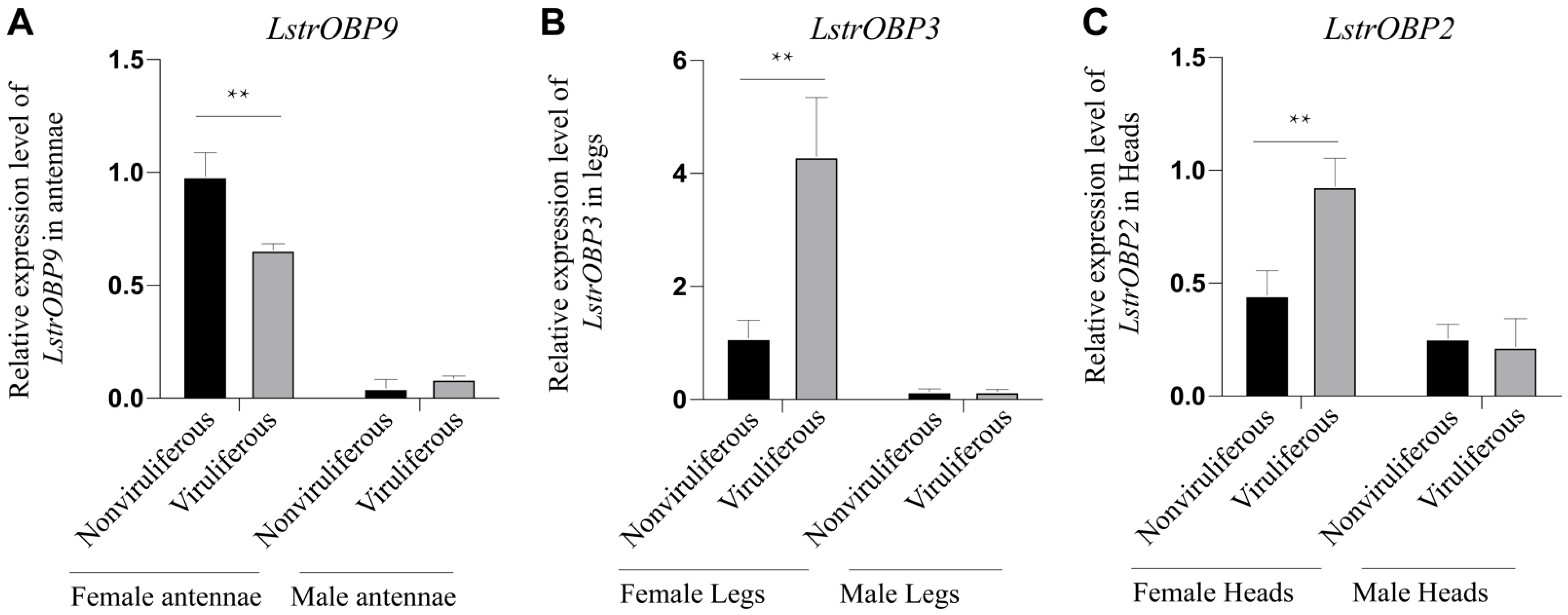
2.6. Validation of the OBPs Expressions in RSV-Infected Chemoreception Organs by Quantitative Real-Time PCR
3. Discussion
4. Materials and Methods
4.1. Nonviruliferous and Viruliferous L. striatellus Rearing
4.2. Samples Preparation and Transcriptomic Sequencing
4.3. Transcriptomic Assembly and GO Annotation
4.4. Analysis of Differentially Expressed Genes
4.5. Identification and Comparative Expression Profiles of Olfactory Related Genes
4.6. Quantitative Real-Time PCR Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| DEGs | Differentially expressed genes |
| RSV | Rice stripe virus |
| CMV | Cucumber mosaic virus |
| PLV | Potato leafroll virus |
| TYLCV | Tomato yellow leaf curl virus |
| SRBDV | Southern rice black streaked dwarf virus |
| TCV | Tomato chlorosis virus |
| OBPs | Odorant-binding proteins |
| CSPs | Chemosensory proteins |
| IRs | Ionotropic receptors |
| GRs | Gustatory receptors |
| ORs | Odorant receptors |
| ORco | Olfactory co-receptor receptor |
| SNMPs | Sensory neuron membrane proteins |
| VFA | Viruliferous female antennae |
| VMA | Viruliferous male antennae |
| VFL | Viruliferous female legs |
| VML | Viruliferous male legs |
| VFH | Viruliferous female heads |
| VMH | Viruliferous male heads |
| NFA | Nonviruliferous female antennae |
| NMA | Nonviruliferous male antennae |
| NFL | Nonviruliferous female legs |
| NML | Nonviruliferous male legs |
| NFH | Nonviruliferous female heads |
| NMH | Nonviruliferous male heads |
| FPKM | Fragments per Kilobase of transcript per Million mapped reads |
| PCA | Principal component analysis |
| GO | Gene ontology |
| qPCR | Real-Time Quantitative PCR |
| VOC | volatile organic compounds |
| GC–EAD | gas chromatography–electroantennographic detection |
References
- Ng, J.C.K.; Falk, B.W. Virus-Vector Interactions Mediating Nonpersistent and Semipersistent Transmission of Plant Viruses. Annu. Rev. Phytopathol. 2006, 44, 183–212. [Google Scholar] [CrossRef]
- Hogenhout, S.A.; Ammar, E.-D.; Whitfield, A.E.; Redinbaugh, M.G. Insect vector interactions with persistently transmitted viruses. Annu. Rev. Phytopathol. 2008, 46, 327–359. [Google Scholar] [CrossRef] [PubMed]
- Mauck, K.E.; De Moraes, C.M.; Mescher, M.C. Deceptive chemical signals induced by a plant virus attract insect vectors to inferior hosts. Proc. Natl. Acad. Sci. USA 2010, 107, 3600–3605. [Google Scholar] [CrossRef] [PubMed]
- Keesey, I.W.; Koerte, S.; Khallaf, M.A.; Retzke, T.; Guillou, A.; Grosse-Wilde, E.; Buchon, N.; Knaden, M.; Hansson, B.S. Pathogenic bacteria enhance dispersal through alteration of Drosophila social communication. Nat. Commun. 2017, 8, 265. [Google Scholar] [CrossRef]
- Ngumbi, E.; Eigenbrode, S.D.; Bosque-Pérez, N.A.; Ding, H.; Rodriguez, A. Myzus persicae is arrested more by blends than by individual compounds elevated in headspace of plrv-infected potato. J. Chem. Ecol. 2007, 33, 1733–1747. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Guo, H.; Ge, F.; Sun, Y. Apoptotic neurodegeneration in whitefly promotes the spread of tylcv. Elife 2020, 9, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Hu, K.; Yang, H.; Liu, S.; He, H.; Li, Y. Odorant-Binding Protein 2 is Involved in the Preference of Sogatella furcifera (Hemiptera: Delphacidae) for Rice Plants Infected with the Southern Rice Black-Streaked Dwarf Virus. Florida Entomol. 2019, 102, 353. [Google Scholar] [CrossRef]
- Shi, X.B.; Wang, X.Z.; Zhang, D.Y.; Zhang, Z.H.; Zhang, Z.; Cheng, J.; Zheng, L.M.; Zhou, X.G.; Tan, X.Q.; Liu, Y. Silencing of odorant-binding protein gene OBP3 using RNA interference reduced virus transmission of tomato chlorosis virus. Int. J. Mol. Sci. 2019, 20, 4969. [Google Scholar] [CrossRef]
- Renou, M.; Guerrero, A. Insect Parapheromones in Olfaction Research and Semiochemical-Based Pest Control Strategies. Annu. Rev. Entomol. 2000, 45, 605–630. [Google Scholar] [CrossRef]
- Morita, H. Primary processes of insect chemoreception. Adv. Biophys. 1972, 3, 161. [Google Scholar]
- Benton, R.; Sachse, S.; Michnick, S.W.; Vosshall, L.B. Atypical membrane topology and heteromeric function of Drosophila odorant receptors in vivo. PLoS Biol. 2006, 4, e20. [Google Scholar] [CrossRef] [PubMed]
- Benton, R.; Vannice, K.S.; Vosshall, L.B. An essential role for a CD36-related receptor in pheromone detection in Drosophila. Nature 2007, 450, 289. [Google Scholar] [CrossRef] [PubMed]
- Clyne, P.J.; Warr, C.G.; Carlson, J.R. Candidate taste receptors in Drosophila. Science 2000, 287, 1830–1834. [Google Scholar] [CrossRef]
- Leal, S.W. Odorant Reception in Insects: Roles of Receptors, Binding Proteins, and Degrading Enzymes. Annu. Rev. Entomol. 2013, 58, 373–391. [Google Scholar] [CrossRef] [PubMed]
- Pelosi, P.; Iovinella, I.; Zhu, J.; Wang, G.; Dani, F.R. Beyond chemoreception: Diverse tasks of soluble olfactory proteins in insects. Biol. Rev. 2018, 93, 184–200. [Google Scholar] [CrossRef]
- Pelosi, P.; Zhou, J.-J.; Ban, L.P.; Calvello, M. Soluble proteins in insect chemical communication. Cell. Mol. Life Sci. C 2006, 63, 1658–1676. [Google Scholar] [CrossRef]
- Pelosi, P.; Iovinella, I.; Felicioli, A.; Dani, F.R. Soluble proteins of chemical communication: An overview across arthropods. Front. Physiol. 2014, 5, 320. [Google Scholar] [CrossRef]
- Toriyama, S. Rice stripe virus: Prototype of a new group of viruses that replicate in plants and insects. Microbiol. Sci. 1986, 3, 347–351. [Google Scholar]
- Falk, B.W.; Tsai, J.H. Biology and molecular biology of viruses in the genus Tenuivirus. Annu. Rev. Phytopathol. 1998, 36, 139–163. [Google Scholar] [CrossRef]
- Hibino, H. Biology and Epidemiology of Rice Viruses. Annu. Rev. Phytopathol. 1996, 34, 249–274. [Google Scholar] [CrossRef]
- Heong, K.L.; Cheng, J.; Escalada, M.M. Rice Planthoppers—Ecology, Management, Socio Economics and Policy; Springer: Dordrecht, The Netherlands, 2015; ISBN 9789401795340. [Google Scholar]
- Li, Y.; Chen, D.; Hu, J.; Zhang, K.; Liu, F. The α-tubulin of Laodelphax striatellus mediates the passage of rice stripe virus (RSV) and enhances horizontal transmission. PLoS Pathog. 2020, 16, e1008710. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.; Guo, H.; Zheng, H.; Zhou, T.; Zhou, Y.; Wang, S.; Fang, R.; Qian, W.; Chen, X. Massively parallel pyrosequencing-based transcriptome analyses of small brown planthopper (Laodelphax striatellus), a vector insect transmitting rice stripe virus (RSV). BMC Genom. 2010, 11, 303. [Google Scholar] [CrossRef]
- Yang, M.; Xu, Z.; Zhao, W.; Liu, Q.; Li, Q.; Lu, L.; Liu, R.; Zhang, X.; Cui, F. Rice stripe virus-derived siRNAs play different regulatory roles in rice and in the insect vector Laodelphax striatellus. BMC Plant Biol. 2018, 18, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Choi, J.Y.; Tao, X.Y.; Kim, J.S.; Kim, W.; Je, Y.H. Transcriptome analysis of the small brown planthopper, Laodelphax striatellus carrying Rice stripe virus. Plant Pathol. J. 2013, 29, 330–337. [Google Scholar] [CrossRef]
- Zhao, W.; Lu, L.; Yang, P.; Cui, N.; Kang, L.; Cui, F. Organ-specific transcriptome response of the small brown planthopper toward rice stripe virus. Insect Biochem. Mol. Biol. 2016, 70, 60–72. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Hu, J.; Xiang, Y.; Zhang, Y.; Liu, F. Identification and comparative expression profiles of chemosensory genes in major chemoreception organs of a notorious pests, Laodelphax striatellus. Comp. Biochem. Physiol. Part D Genom. Proteom. 2019, 33, 100646. [Google Scholar] [CrossRef]
- Li, S.; Zhou, C.; Zhou, Y. Olfactory co-receptor Orco stimulated by Rice stripe virus is essential for host seeking behavior in small brown planthopper. Pest Manag. Sci. 2019, 75, 187–194. [Google Scholar] [CrossRef]
- Xu, Y.; Fu, S.; Tao, X.; Zhou, X. Rice stripe virus: Exploring Molecular Weapons in the Arsenal of a Negative-Sense RNA Virus. Annu. Rev. Phytopathol. 2021, 59, 351–371. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Jiang, F.; Wang, X.; Yang, P.; Bao, Y.; Zhao, W.; Wang, W.; Lu, H.; Wang, Q.; Cui, N. Genome sequence of the small brown planthopper, Laodelphax Striatellus. Gigascience 2017, 6, gix109. [Google Scholar] [CrossRef] [PubMed]
- Naters, W.; Carlson, J.R. Insects as chemosensors of humans and crops. Nature 2006, 444, 302–307. [Google Scholar] [CrossRef]
- Sim, S.; Ramirez, J.L.; Dimopoulos, G. Dengue virus infection of the aedes aegypti salivary gland and chemosensory apparatus induces genes that modulate infection and blood-feeding behavior. PLoS Pathog. 2012, 8, e1002631. [Google Scholar] [CrossRef]
- Wang, G.Z.; Zhou, Y.J.; Chen, Z.X.; University, Z. Hangzhou Production of monoclonal antibodies to Rice stripe virus and application in virus detection. Acta Phytopathol. Sin. 2004, 34, 302–306. [Google Scholar]
- Pertea, M.; Kim, D.; Pertea, G.M.; Leek, J.T.; Salzberg, S.L. Transcript-level expression analysis of RNA-seq experiments with HISAT, StringTie and Ballgown. Nat. Protoc. 2016, 11, 1650–1667. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Smyth, G.K.; Wei, S. FeatureCounts: An efficient general purpose program for assigning sequence reads to genomic features. Bioinformatics 2014, 30, 923–930. [Google Scholar]
- Deng, W.; Wang, Y.; Liu, Z.; Cheng, H.; Xue, Y. HemI: A Toolkit for Illustrating Heatmaps. PLoS ONE 2014, 9, e111988. [Google Scholar] [CrossRef]
- Robinson, M.D.; McCarthy, D.J.; Smyth, G.K. EdgeR: A Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 2010, 26, 139–140. [Google Scholar] [CrossRef]
- Haynes, W. Benjamini—Hochberg Method BT. In Encyclopedia of Systems Biology; Dubitzky, W., Wolkenhauer, O., Cho, K.H., Yokota, H., Eds.; Springer: New York, NY, USA, 2013; p. 78. ISBN 978-1-4419-9863-7. [Google Scholar]
- Rao, X.; Huang, X.; Zhou, Z.; Lin, X. An improvement of the 2−ΔΔCT method for quantitative real-time polymerase chain reaction data analysis. Biostat. Bioinform. Biomath. 2013, 3, 71–85. [Google Scholar] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, Y.; Zhang, Y.; Xiang, Y.; Chen, D.; Hu, J.; Liu, F. Comparative Transcriptome Analysis of Chemoreception Organs of Laodelphax striatellus in Response to Rice Stripe Virus Infection. Int. J. Mol. Sci. 2021, 22, 10299. https://doi.org/10.3390/ijms221910299
Li Y, Zhang Y, Xiang Y, Chen D, Hu J, Liu F. Comparative Transcriptome Analysis of Chemoreception Organs of Laodelphax striatellus in Response to Rice Stripe Virus Infection. International Journal of Molecular Sciences. 2021; 22(19):10299. https://doi.org/10.3390/ijms221910299
Chicago/Turabian StyleLi, Yao, Yunye Zhang, Yin Xiang, Danyu Chen, Jia Hu, and Fang Liu. 2021. "Comparative Transcriptome Analysis of Chemoreception Organs of Laodelphax striatellus in Response to Rice Stripe Virus Infection" International Journal of Molecular Sciences 22, no. 19: 10299. https://doi.org/10.3390/ijms221910299
APA StyleLi, Y., Zhang, Y., Xiang, Y., Chen, D., Hu, J., & Liu, F. (2021). Comparative Transcriptome Analysis of Chemoreception Organs of Laodelphax striatellus in Response to Rice Stripe Virus Infection. International Journal of Molecular Sciences, 22(19), 10299. https://doi.org/10.3390/ijms221910299




