A Review of the Use of Microparticles for Cartilage Tissue Engineering
Abstract
1. Introduction
1.1. Tissue Engineering (TE)
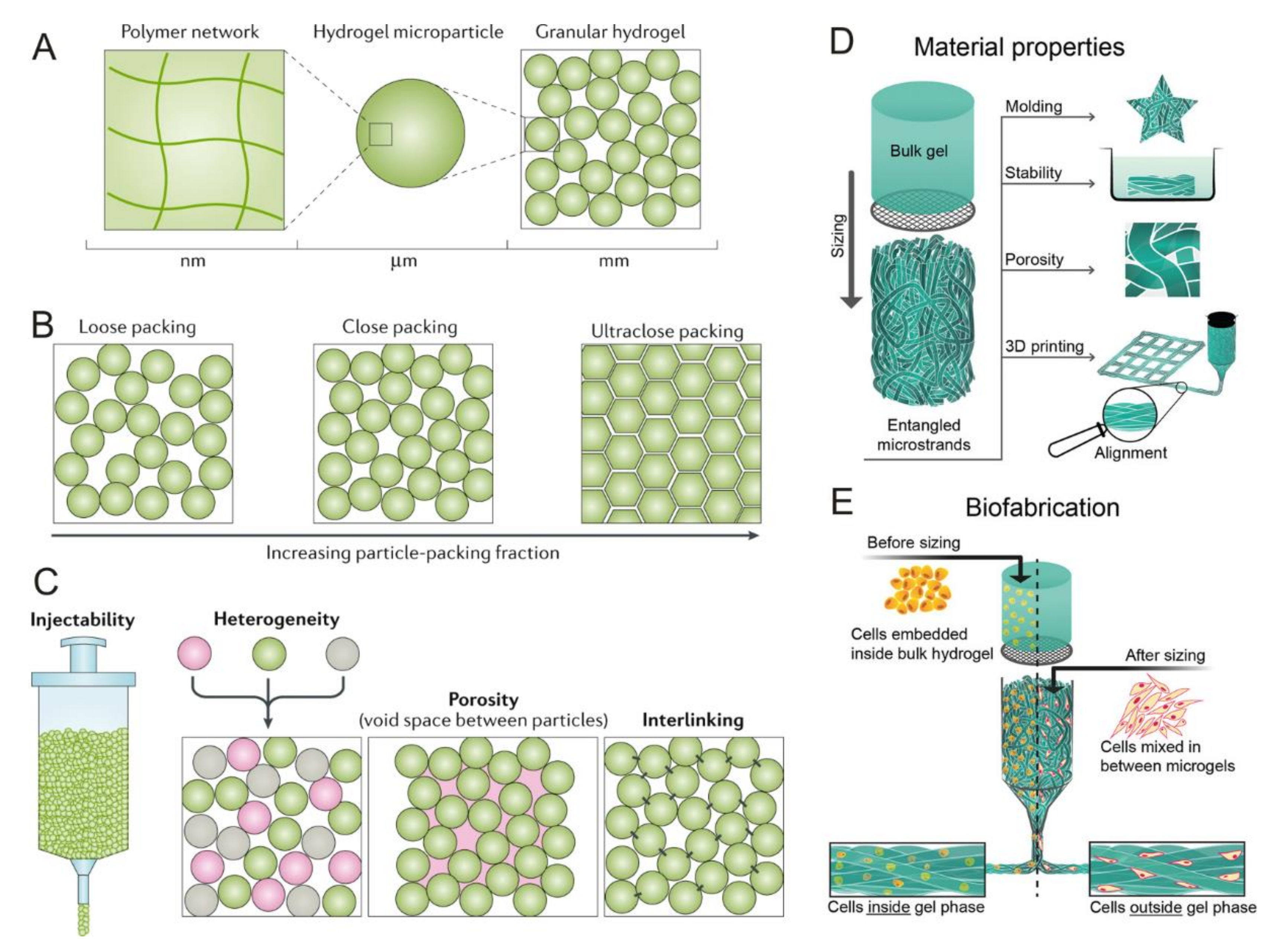
1.2. Background on Microspheres, Microcarriers, and Granular Hydrogels
1.3. Overview of the Review
2. Cartilage Regeneration and Tissue Engineering (TE)
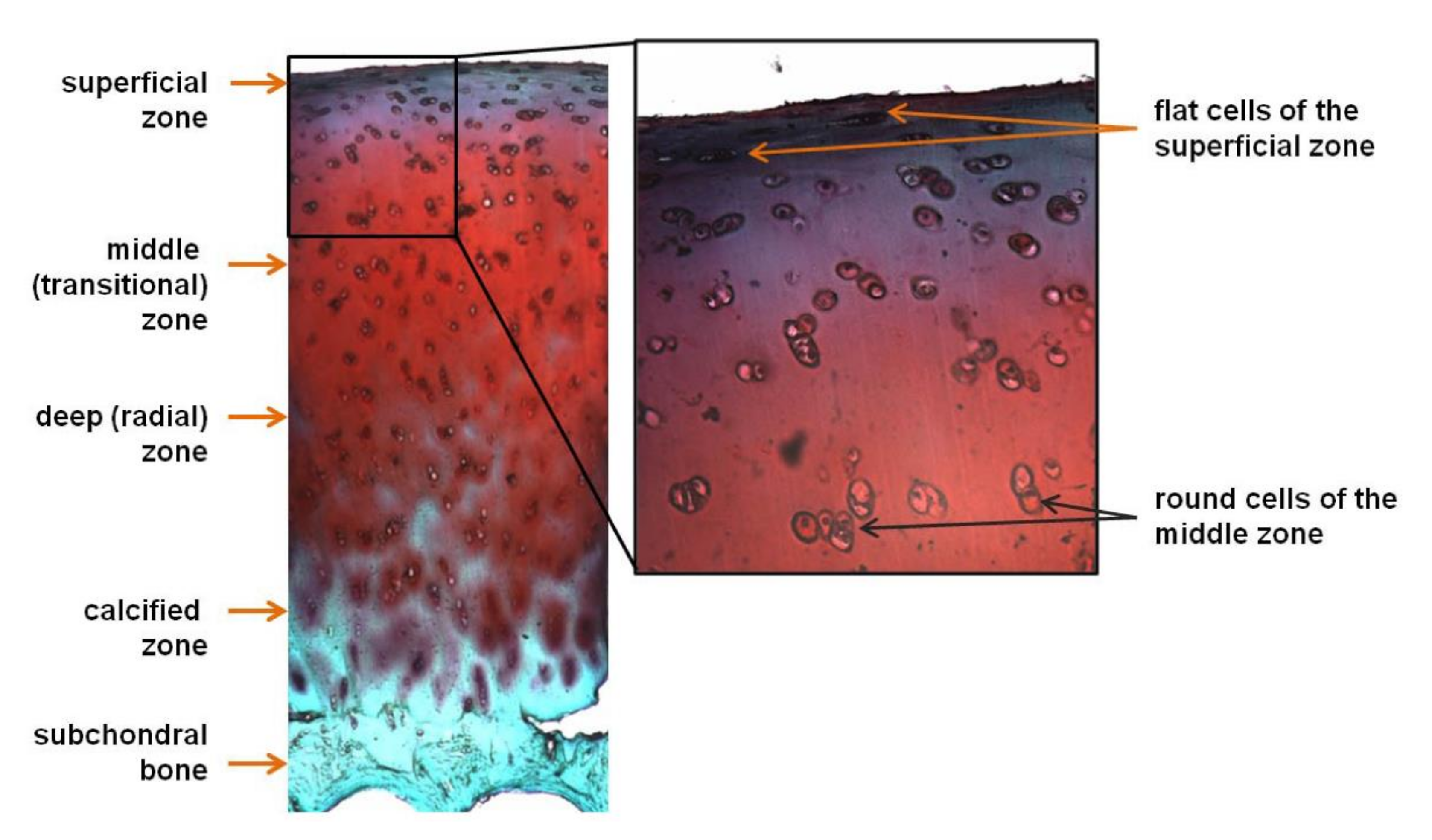
2.1. Cartilage Tissue and Disease and Current Treatment Options
2.2. Cartilage TE Specifics
3. Microspheres as Delivery Vehicles for Growth Factors and Drugs
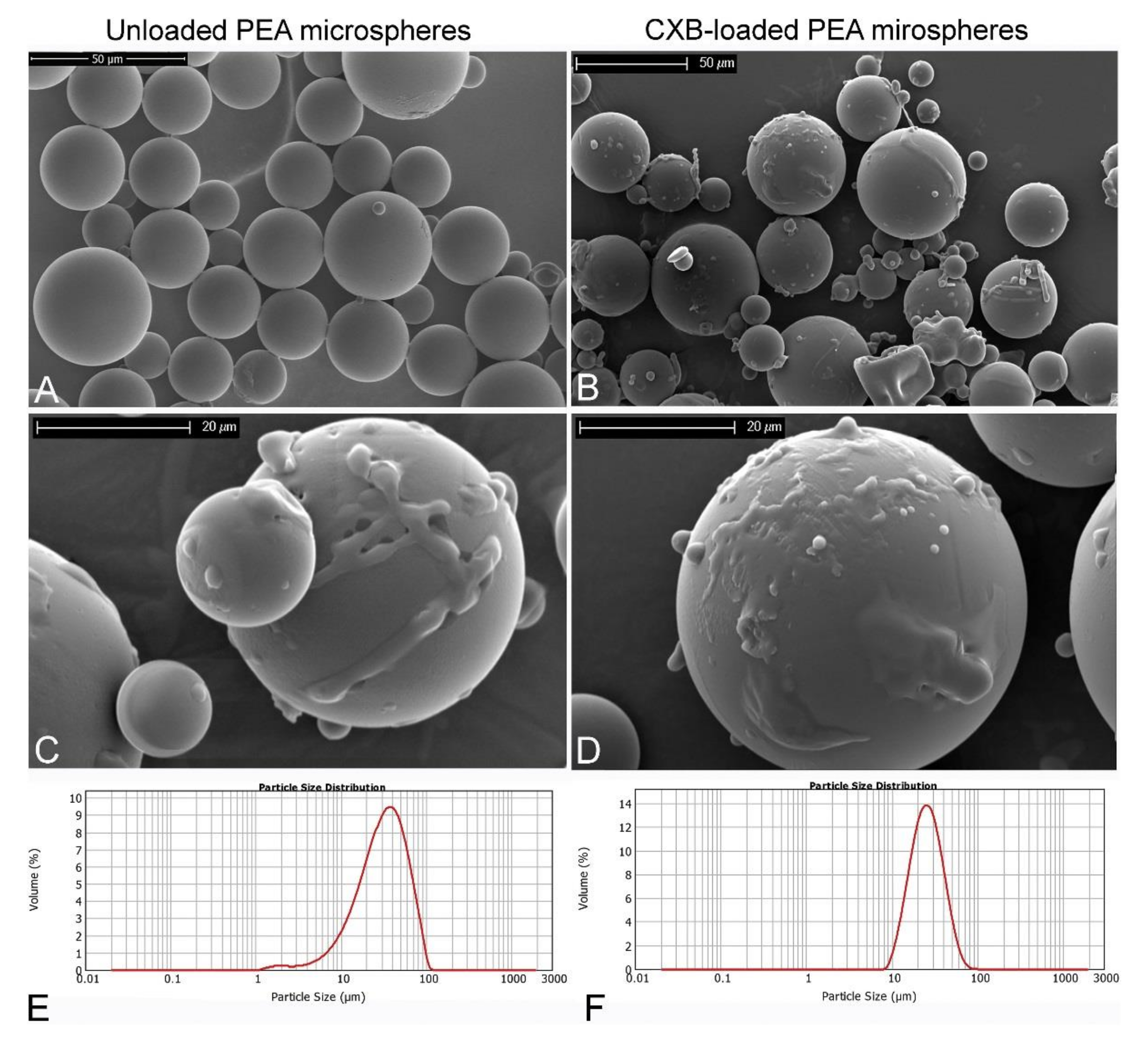
3.1. Corticosteroids
3.2. Anti-Inflammatories
3.3. Melatonin
3.4. Gene Therapy
3.5. Decellularized Cartilage Matrix (dCM)
3.6. Growth Factors
4. Microcarriers for Cell Expansion
4.1. Microcarrier Fundamentals
4.2. Expansion in Bioreactors
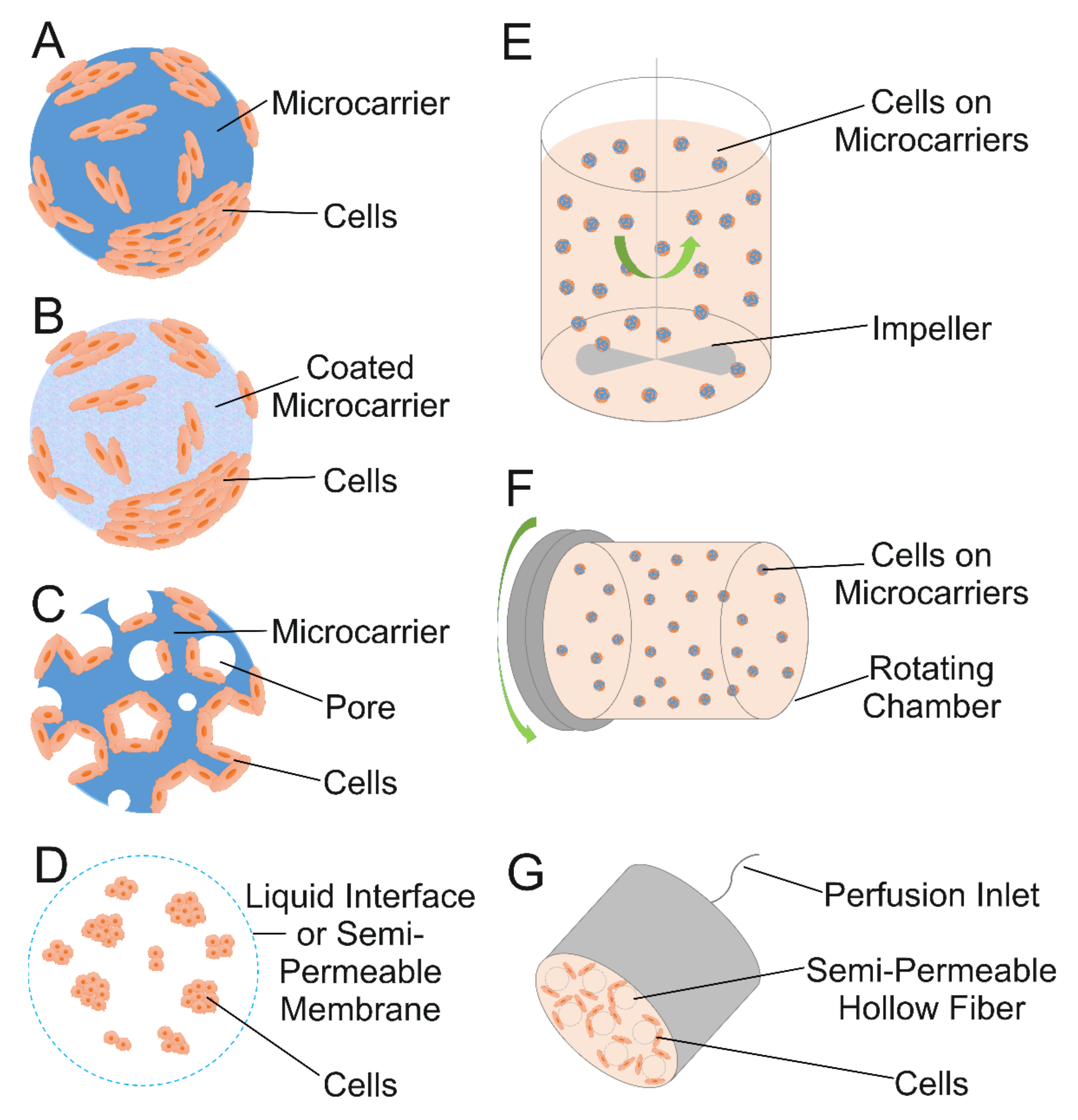
4.3. Cell Expansion on Microcarriers Relevant to Cartilage TE
5. Microspheres Forming Granular Hydrogels as 3D Tissue Engineering (TE) Constructs
5.1. Granular Hydrogels for Bioprinting Applications
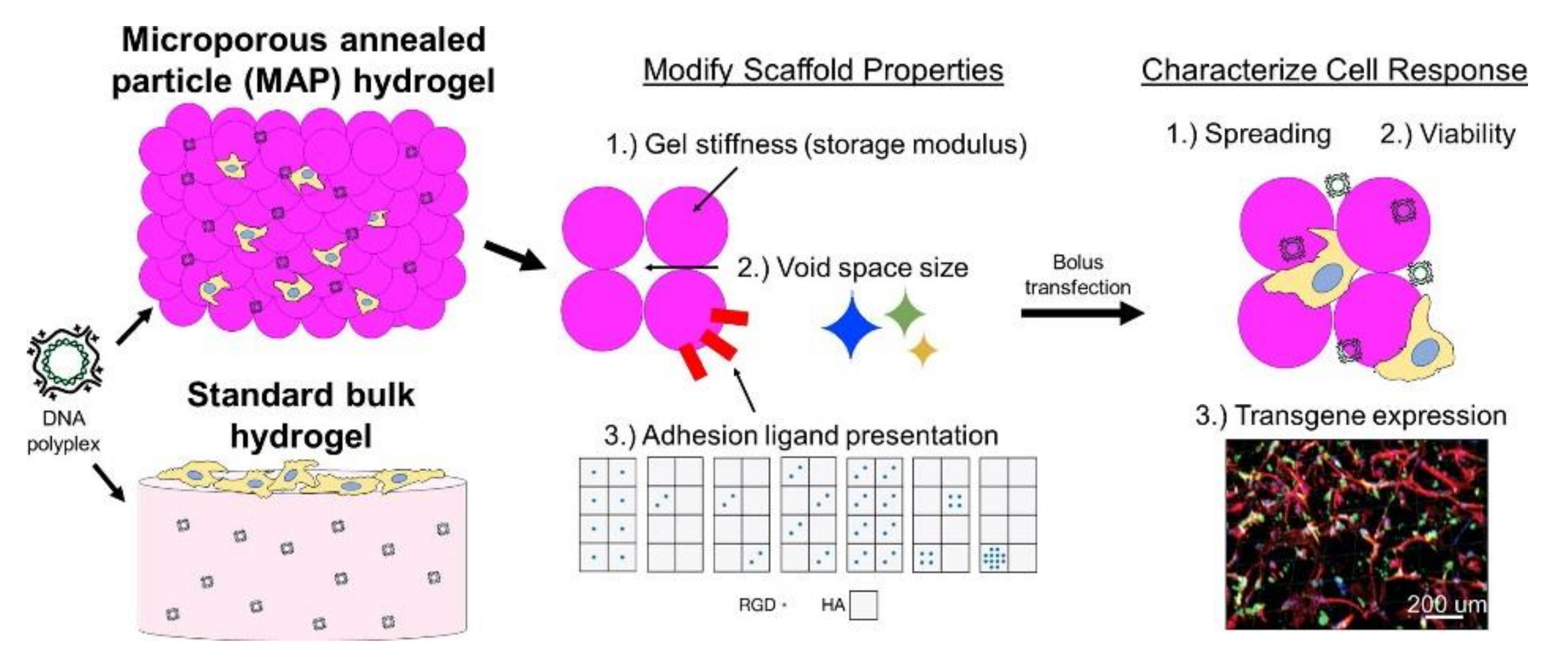
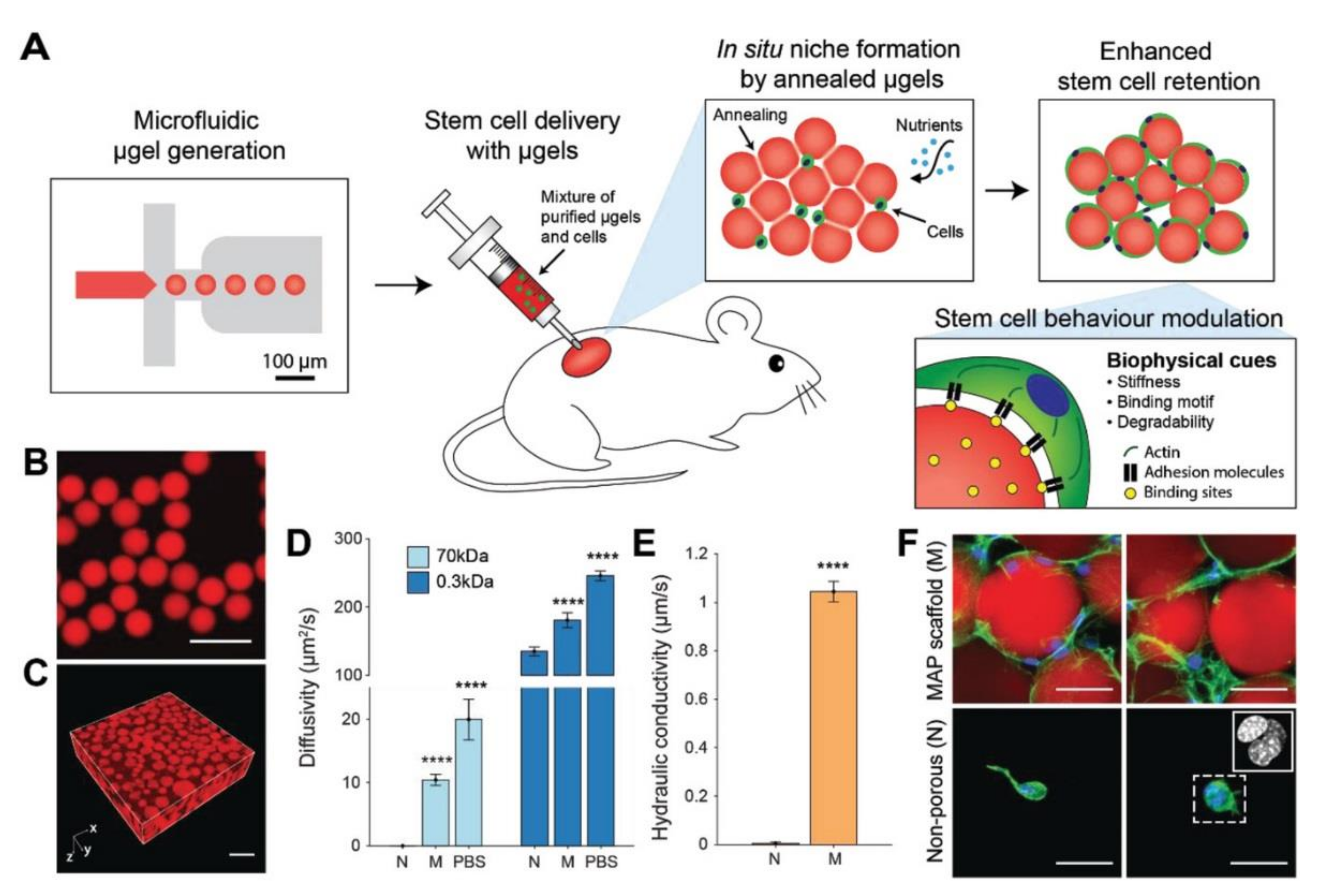
5.2. Microporous Annealed Particle (MAP) Hydrogels
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ACI | Autologous chondrocyte implantation |
| BMP | Bone morphogenetic protein |
| BMSC | Bone marrow-derived mesenchymal stem cell |
| COX-2 | Cyclo-oxygenase-2 |
| CXB | Celecoxib |
| dCM | Decellularized cartilage matrix |
| DLMS | Dexamethasone-loaded microspheres |
| ECM | Extracellular matrix |
| hADSC | Human adipose-derived stem cell |
| hESC | Human embryonic stem cell |
| HMP | Hydrogel microparticle |
| hMSC | Human mesenchymal stem cell |
| hPDC | Human periosteum-derived cell |
| hPSC | Human pluripotent stem cell |
| IL | Interleukin |
| INJ | Injected group |
| IVD | Intervertebral disc |
| MAP | Microporous annealed particle |
| MEF | Mouse embryonic fibroblast |
| MSC | Mesenchymal stem cell |
| NP | Nucleus pulposus |
| NSAID | Non-steroidal anti-inflammatory drug |
| OA | Osteoarthritis |
| OARSI | Osteoarthritis Research Society International |
| pDNA | Plasmid deoxyribonucleic acid |
| PEA | Polyesteramide |
| PLA | Polylactic acid |
| PLGA | Polylactic-co-glycolic acid |
| RGD | Arginine-glycine-aspartic acid |
| TAA | Triamcinolone acetonide |
| TE | Tissue engineering |
| TGF | Transforming growth factor |
| US | United States |
| 3D | Three-dimensional |
| µCT | Microcomputed tomography |
References
- Bentley, T.S.; Ortner, N.J. 2020 U.S. Organ and Tissue Transplants: Cost Estimates, Discussion, and Emerging Issues; Milliman: Seattle, WA, USA, 2020. [Google Scholar]
- Langer, R.; Vacanti, J.P. Tissue Engineering. Science 1993, 260, 920–926. [Google Scholar] [CrossRef] [PubMed]
- Lanza, R.P.; Langer, R.; Vacanti, J.P.; Atala, A. (Eds.) Principles of Tissue Engineering, 5th ed.; Academic Press, an imprint of Elsevier: London, UK, 2020; ISBN 978-0-12-821401-5. [Google Scholar]
- Griffith, L.G. Tissue Engineering--Current Challenges and Expanding Opportunities. Science 2002, 295, 1009–1014. [Google Scholar] [CrossRef]
- Chapekar, M.S. Tissue Engineering: Challenges and Opportunities. J. Biomed. Mater. Res. 2000, 53, 617–620. [Google Scholar] [CrossRef]
- Organ Donation Statistics | Organdonor.Gov. Available online: https://www.organdonor.gov/learn/organ-donation-statistics#:%7E:text=Learn%20More%20About%20Organ%20Transplantation%20by%20the%20Numbers&text=Who%20is%20on%20the%20transplant,the%20national%20transplant%20waiting%20list (accessed on 23 July 2021).
- Jagur-Grodzinski, J. Polymers for Tissue Engineering, Medical Devices, and Regenerative Medicine. Concise General Review of Recent Studies. Polym. Adv. Technol. 2006, 17, 395–418. [Google Scholar] [CrossRef]
- Howard, D.; Buttery, L.D.; Shakesheff, K.M.; Roberts, S.J. Tissue Engineering: Strategies, Stem Cells and Scaffolds. J. Anat. 2008, 213, 66–72. [Google Scholar] [CrossRef]
- Tuli, R.; Li, W.-J.; Tuan, R.S. Current State of Cartilage Tissue Engineering. Arthritis Res. Ther. 2003, 5, 235. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, A.S. Hydrogels for Biomedical Applications. Adv. Drug Deliv. Rev. 2012, 64, 18–23. [Google Scholar] [CrossRef]
- Koh, J.; Griffin, D.R.; Archang, M.M.; Feng, A.; Horn, T.; Margolis, M.; Zalazar, D.; Segura, T.; Scumpia, P.O.; Carlo, D. Enhanced In Vivo Delivery of Stem Cells Using Microporous Annealed Particle Scaffolds. Small 2019, 15, 1903147. [Google Scholar] [CrossRef]
- Sahil, K.; Akanksha, M.; Premjeet, S.; Bilandi, A.; Kapoor, B. Microsphere: A Review. Int. J. Res. Pharm. Chem. 2011, 1, 1184–1198. [Google Scholar]
- Virmani, T.; Gupta, J. Pharmaceutical Application of Microspheres: An Approach for Treatment of Various Diseases. Int. J. Pharm. Sci. Res. 2017, 8, 3252–3260. [Google Scholar]
- Tavassoli, H.; Alhosseini, S.N.; Tay, A.; Chan, P.P.Y.; Weng Oh, S.K.; Warkiani, M.E. Large-Scale Production of Stem Cells Utilizing Microcarriers: A Biomaterials Engineering Perspective from Academic Research to Commercialized Products. Biomaterials 2018, 181, 333–346. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.-Y.; Chen, J.-Y.; Tong, X.-M.; Mei, J.-G.; Chen, Y.-F.; Mou, X.-Z. Recent Advances in the Use of Microcarriers for Cell Cultures and Their Ex Vivo and in Vivo Applications. Biotechnol. Lett. 2020, 42, 1–10. [Google Scholar] [CrossRef]
- Koh, B.; Sulaiman, N.; Fauzi, M.B.; Law, J.X.; Ng, M.H.; Idrus, R.B.H.; Yazid, M.D. Three Dimensional Microcarrier System in Mesenchymal Stem Cell Culture: A Systematic Review. Cell Biosci. 2020, 10, 75. [Google Scholar] [CrossRef] [PubMed]
- Chen, A.K.-L.; Reuveny, S.; Oh, S.K.W. Application of Human Mesenchymal and Pluripotent Stem Cell Microcarrier Cultures in Cellular Therapy: Achievements and Future Direction. Biotechnol. Adv. 2013, 31, 1032–1046. [Google Scholar] [CrossRef]
- Tellegen, A.R.; Rudnik-Jansen, I.; Beukers, M.; Miranda-Bedate, A.; Bach, F.C.; de Jong, W.; Woike, N.; Mihov, G.; Thies, J.C.; Meij, B.P.; et al. Intradiscal Delivery of Celecoxib-Loaded Microspheres Restores Intervertebral Disc Integrity in a Preclinical Canine Model. J. Controlled Release 2018, 286, 439–450. [Google Scholar] [CrossRef]
- Agnihotri, N.; Soni, G.C.; Chanchal, D.K.; Khan, A.; Tiwari, S. A Review on Microspheres a Novel Drug Delivery System for Multiparticulate Drug Release. Int. J. Life Sci. Rev. 2019, 5, 6–15. [Google Scholar]
- Cheng, W.; Zhang, J.; Liu, J.; Yu, Z. Granular Hydrogels for 3D Bioprinting Applications. View 2020, 1, 20200060. [Google Scholar] [CrossRef]
- Riley, L.; Schirmer, L.; Segura, T. Granular Hydrogels: Emergent Properties of Jammed Hydrogel Microparticles and Their Applications in Tissue Repair and Regeneration. Curr. Opin. Biotechnol. 2019, 60, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, E.M. Hydrogel: Preparation, Characterization, and Applications: A Review. J. Adv. Res. 2015, 6, 105–121. [Google Scholar] [CrossRef]
- Kessel, B.; Lee, M.; Bonato, A.; Tinguely, Y.; Tosoratti, E.; Zenobi-Wong, M. 3D Bioprinting of Architected Hydrogels from Entangled Microstrands. Adv. Sci. 2020, 7. [Google Scholar] [CrossRef] [PubMed]
- Walker, M.; Luo, J.; Pringle, E.W.; Cantini, M. ChondroGELesis: Hydrogels to Harness the Chondrogenic Potential of Stem Cells. Mater. Sci. Eng. C 2021, 121, 111822. [Google Scholar] [CrossRef]
- Stampoultzis, T.; Karami, P.; Pioletti, D.P. Thoughts on Cartilage Tissue Engineering: A 21st Century Perspective. Curr. Res. Transl. Med. 2021, 69, 103299. [Google Scholar] [CrossRef]
- Ngadimin, K.D.; Stokes, A.; Gentile, P.; Ferreira, A.M. Biomimetic Hydrogels Designed for Cartilage Tissue Engineering. Biomater. Sci. 2021, 9, 4246–4259. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Dai, B.; Guo, J.; Zheng, L.; Guo, Q.; Peng, J.; Xu, J.; Qin, L. Nanoparticle–Cartilage Interaction: Pathology-Based Intra-Articular Drug Delivery for Osteoarthritis Therapy. Nano-Micro Lett. 2021, 13, 149. [Google Scholar] [CrossRef] [PubMed]
- Younas, A.; Gu, H.; Zhao, Y.; Zhang, N. Novel Approaches of the Nanotechnology-Based Drug Delivery Systems for Knee Joint Injuries: A Review. Int. J. Pharm. 2021, 608, 121051. [Google Scholar] [CrossRef]
- Akkiraju, H.; Nohe, A. Role of Chondrocytes in Cartilage Formation, Progression of Osteoarthritis and Cartilage Regeneration. J. Dev. Biol. 2015, 3, 177–192. [Google Scholar] [CrossRef] [PubMed]
- Morgese, G.; Benetti, E.M.; Zenobi-Wong, M. Molecularly Engineered Biolubricants for Articular Cartilage. Adv. Healthc. Mater. 2018, 7, 1701463. [Google Scholar] [CrossRef]
- Lin, W.; Klein, J. Recent Progress in Cartilage Lubrication. Adv. Mater. 2021, 33, 2005513. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, T.A.; Gastelum, N.S.; Nguyen, Q.T.; Schumacher, B.L.; Sah, R.L. Boundary Lubrication of Articular Cartilage: Role of Synovial Fluid Constituents. Arthritis Rheum. 2007, 56, 882–891. [Google Scholar] [CrossRef] [PubMed]
- Tuan, R.S.; Chen, A.F.; Klatt, B.A. Cartilage Regeneration. J. Am. Acad. Orthop. Surg. 2013, 21, 303–311. [Google Scholar] [CrossRef] [PubMed]
- Kuo, C.K.; Li, W.-J.; Mauck, R.L.; Tuan, R.S. Cartilage Tissue Engineering: Its Potential and Uses. Curr. Opin. Rheumatol. 2006, 18, 64–73. [Google Scholar] [CrossRef] [PubMed]
- Urban, J.P.G.; Roberts, S. Degeneration of the Intervertebral Disc. Arthritis Res. Ther. 2003, 5, 120–130. [Google Scholar] [CrossRef]
- Secretariat, M.A. Artificial Discs for Lumbar and Cervical Degenerative Disc Disease-Update: An Evidence-Based Analysis. Ont. Health Technol. Assess. Ser. 2006, 6, 1–98. [Google Scholar]
- Yalcinkaya, E.; Celik, M.; Bugan, B. Extracellular Matrix Turnover: A Balance between MMPs and Their Inhibitors. Arq. Bras. Cardiol. 2014. [Google Scholar] [CrossRef]
- Steinwachs, M.R.; Guggi, T.; Kreuz, P.C. Marrow Stimulation Techniques. Injury 2008, 39, 26–31. [Google Scholar] [CrossRef] [PubMed]
- Medvedeva, E.; Grebenik, E.; Gornostaeva, S.; Telpuhov, V.; Lychagin, A.; Timashev, P.; Chagin, A. Repair of Damaged Articular Cartilage: Current Approaches and Future Directions. Int. J. Mol. Sci. 2018, 19, 2366. [Google Scholar] [CrossRef]
- Ikada, Y. Challenges in Tissue Engineering. J. R. Soc. Interface 2006, 3, 589–601. [Google Scholar] [CrossRef]
- Heath, C.A. Cells for Tissue Engineering. Trends Biotechnol. 2000, 18, 17–19. [Google Scholar] [CrossRef]
- Ude, C.C.; Miskon, A.; Idrus, R.B.H.; Abu Bakar, M.B. Application of Stem Cells in Tissue Engineering for Defense Medicine. Mil. Med. Res. 2018, 5, 7. [Google Scholar] [CrossRef]
- Ferretti, C. Periosteum Derived Stem Cells for Regenerative Medicine Proposals: Boosting Current Knowledge. World J. Stem Cells 2014, 6, 266. [Google Scholar] [CrossRef] [PubMed]
- Hassan, M.N.F.B.; Yazid, M.D.; Yunus, M.H.M.; Chowdhury, S.R.; Lokanathan, Y.; Idrus, R.B.H.; Ng, A.M.H.; Law, J.X. Large-Scale Expansion of Human Mesenchymal Stem Cells. Stem Cells Int. 2020, 2020, 1–17. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, C.; Laslett, A.L. Suspended in Culture — Human Pluripotent Cells for Scalable Technologies. Stem Cell Res. 2012, 9, 167–170. [Google Scholar] [CrossRef] [PubMed]
- Kumbar, S.; Laurencin, C.; Deng, M. (Eds.) Natural and Synthetic Biomedical Polymers, 1st ed.; Elsevier: Burlington, MA, USA, 2014; ISBN 978-0-12-396983-5. [Google Scholar]
- Ducheyne, P.; Grainger, D.W.; Healy, K.E.; Hutmacher, D.W.; Kirkpatrick, C.J. Comprehensive Biomaterials II; Elsevier: Amsterdam, The Netherlands, 2017; ISBN 978-0-08-100692-4. [Google Scholar]
- Mas-Moruno, C. Surface functionalization of biomaterials for bone tissue regeneration and repair. In Peptides and Proteins as Biomaterials for Tissue Regeneration and Repair; Barbosa, M.A., Martins, M.C.L., Eds.; Woodhead Publishing Series in Biomaterials; Woodhead Publishing: Duxford, UK, 2018; ISBN 978-0-08-100803-4. [Google Scholar]
- Das, M.K.; Ahmed, A.B.; Saha, D. MICROSPHERE A DRUG DELIVERY SYSTEM–A REVIEW. Int. J. Curr. Pharm. Res. 2019, 34–41. [Google Scholar] [CrossRef]
- Berkland, C.; King, M.; Cox, A.; Kim, K.; Pack, D.W. Precise Control of PLG Microsphere Size Provides Enhanced Control of Drug Release Rate. J. Controlled Release 2002, 82, 137–147. [Google Scholar] [CrossRef]
- Singh, U.V.; Bisht, K.; Rao, S.; Devi, U.; Udupa, N. Plumbagin-Loaded PLGA Microspheres with Reduced Toxicity and Enhanced Antitumour Efficacy in Mice. Pharm. Pharmacol. Commun. 1996, 2, 407–409. [Google Scholar] [CrossRef]
- Gurung, B.D.; Kakar, S. An Overview on Microspheres. Int. J. Health Clin. Res. 2020, 3, 11–24. [Google Scholar]
- Lengyel, M.; Kállai-Szabó, N.; Antal, V.; Laki, A.J.; Antal, I. Microparticles, Microspheres, and Microcapsules for Advanced Drug Delivery. Sci. Pharm. 2019, 87, 20. [Google Scholar] [CrossRef]
- Sulaiman, S.B.; Idrus, R.B.H.; Hwei, N.M. Gelatin Microsphere for Cartilage Tissue Engineering: Current and Future Strategies. Polymers 2020, 12, 2404. [Google Scholar] [CrossRef]
- Mercier, N.R.; Costantino, H.R.; Tracy, M.A.; Bonassar, L.J. A Novel Injectable Approach for Cartilage Formation in Vivo Using PLG Microspheres. Ann. Biomed. Eng. 2004, 32, 418–429. [Google Scholar] [CrossRef]
- Martín, A.R.; Patel, J.M.; Zlotnick, H.M.; Carey, J.L.; Mauck, R.L. Emerging Therapies for Cartilage Regeneration in Currently Excluded ‘Red Knee’ Populations. npj Regen. Med. 2019, 4, 12. [Google Scholar] [CrossRef] [PubMed]
- Wernecke, C.; Braun, H.J.; Dragoo, J.L. The Effect of Intra-Articular Corticosteroids on Articular Cartilage: A Systematic Review. Orthop. J. Sports Med. 2015, 3, 232596711558116. [Google Scholar] [CrossRef]
- Paik, J.; Duggan, S.T.; Keam, S.J. Triamcinolone Acetonide Extended-Release: A Review in Osteoarthritis Pain of the Knee. Drugs 2019, 79, 455–462. [Google Scholar] [CrossRef] [PubMed]
- Wehling, P.; Evans, C.; Wehling, J.; Maixner, W. Effectiveness of Intra-Articular Therapies in Osteoarthritis: A Literature Review. Ther. Adv. Musculoskelet. Dis. 2017, 9, 183–196. [Google Scholar] [CrossRef]
- Rudnik-Jansen, I.; Tellegen, A.; Beukers, M.; Öner, F.; Woike, N.; Mihov, G.; Thies, J.; Meij, B.; Tryfonidou, M.; Creemers, L. Safety of Intradiscal Delivery of Triamcinolone Acetonide by a Poly(Esteramide) Microsphere Platform in a Large Animal Model of Intervertebral Disc Degeneration. Spine J. 2019, 19, 905–919. [Google Scholar] [CrossRef] [PubMed]
- Kraus, V.B.; Conaghan, P.G.; Aazami, H.A.; Mehra, P.; Kivitz, A.J.; Lufkin, J.; Hauben, J.; Johnson, J.R.; Bodick, N. Synovial and Systemic Pharmacokinetics (PK) of Triamcinolone Acetonide (TA) Following Intra-Articular (IA) Injection of an Extended-Release Microsphere-Based Formulation (FX006) or Standard Crystalline Suspension in Patients with Knee Osteoarthritis (OA). Osteoarthritis Cartilage 2018, 26, 34–42. [Google Scholar] [CrossRef] [PubMed]
- Flexion Therapeutics, Inc. Treatment Utilizing Extended-Release Microsphere Technology. Available online: https://zilrettapro.com/how-zilretta-works/extended-release-microspheres/ (accessed on 2 August 2021).
- Flexion Therapeutics Announces New Drug Application for ZilrettaTM (FX006) Accepted by U.S. Food and Drug Administration | Flexion Therapeutics, Inc. Available online: https://ir.flexiontherapeutics.com/news-releases/news-release-details/flexion-therapeutics-announces-new-drug-application-zilrettatm (accessed on 2 August 2021).
- Bodick, N.; Lufkin, J.; Willwerth, C.; Kumar, A.; Bolognese, J.; Schoonmaker, C.; Ballal, R.; Hunter, D.; Clayman, M. An Intra-Articular, Extended-Release Formulation of Triamcinolone Acetonide Prolongs and Amplifies Analgesic Effect in Patients with Osteoarthritis of the Knee: A Randomized Clinical Trial. J. Bone Jt. Surg. 2015, 97, 877–888. [Google Scholar] [CrossRef]
- Huebner, K.D.; Shrive, N.G.; Frank, C.B. Dexamethasone Inhibits Inflammation and Cartilage Damage in a New Model of Post-Traumatic Osteoarthritis: DEXAMETHASONE PREVENTS POST-TRAUMATIC ARTHRITIS IN A RABBIT. J. Orthop. Res. 2014, 32, 566–572. [Google Scholar] [CrossRef]
- Zolnik, B.S.; Burgess, D.J. Evaluation of in Vivo–in Vitro Release of Dexamethasone from PLGA Microspheres. J. Controlled Release 2008, 127, 137–145. [Google Scholar] [CrossRef]
- Stefani, R.M.; Lee, A.J.; Tan, A.R.; Halder, S.S.; Hu, Y.; Guo, X.E.; Stoker, A.M.; Ateshian, G.A.; Marra, K.G.; Cook, J.L.; et al. Sustained Low-Dose Dexamethasone Delivery via a PLGA Microsphere-Embedded Agarose Implant for Enhanced Osteochondral Repair. Acta Biomater. 2020, 102, 326–340. [Google Scholar] [CrossRef]
- Zhang, Y.; Pizzute, T.; Pei, M. Anti-Inflammatory Strategies in Cartilage Repair. Tissue Eng. Part B Rev. 2014, 20, 655–668. [Google Scholar] [CrossRef]
- Janssen, M.; Timur, U.T.; Woike, N.; Welting, T.J.M.; Draaisma, G.; Gijbels, M.; van Rhijn, L.W.; Mihov, G.; Thies, J.; Emans, P.J. Celecoxib-Loaded PEA Microspheres as an Auto Regulatory Drug-Delivery System after Intra-Articular Injection. J. Controlled Release 2016, 244, 30–40. [Google Scholar] [CrossRef]
- Park, E.; Hart, M.L.; Rolauffs, B.; Stegemann, J.P.; Annamalai, T.R. Bioresponsive Microspheres for On-demand Delivery of Anti-inflammatory Cytokines for Articular Cartilage Repair. J. Biomed. Mater. Res. A 2020, 108, 722–733. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.; Wang, L.; Li, H.; Ke, Y.; Yao, Y. Function of Sustained Released Resveratrol on IL-1 β -Induced HBMSC MMP13 Secretion Inhibition and Chondrogenic Differentiation Promotion. J. Biomater. Appl. 2016, 30, 930–939. [Google Scholar] [CrossRef] [PubMed]
- Goto, N.; Okazaki, K.; Akasaki, Y.; Ishihara, K.; Murakami, K.; Koyano, K.; Ayukawa, Y.; Yasunami, N.; Masuzaki, T.; Nakashima, Y. Single Intra-Articular Injection of Fluvastatin-PLGA Microspheres Reduces Cartilage Degradation in Rabbits with Experimental Osteoarthritis: FLUVASTATIN-PLGA MICROSPHERES REDUCE OSTEOARTHRITIS. J. Orthop. Res. 2017, 35, 2465–2475. [Google Scholar] [CrossRef]
- Gao, W.; Lin, M.; Liang, A.; Zhang, L.; Chen, C.; Liang, G.; Xu, C.; Peng, Y.; Chen, C.; Huang, D.; et al. Melatonin Enhances Chondrogenic Differentiation of Human Mesenchymal Stem Cells. J. Pineal Res. 2014, 56, 62–70. [Google Scholar] [CrossRef] [PubMed]
- Atoufi, Z.; Kamrava, S.K.; Davachi, S.M.; Hassanabadi, M.; Saeedi Garakani, S.; Alizadeh, R.; Farhadi, M.; Tavakol, S.; Bagher, Z.; Hashemi Motlagh, G. Injectable PNIPAM/Hyaluronic Acid Hydrogels Containing Multipurpose Modified Particles for Cartilage Tissue Engineering: Synthesis, Characterization, Drug Release and Cell Culture Study. Int. J. Biol. Macromol. 2019, 139, 1168–1181. [Google Scholar] [CrossRef]
- Naghizadeh, Z.; Karkhaneh, A.; Khojasteh, A. Simultaneous Release of Melatonin and Methylprednisolone from an Injectable in Situ Self-Crosslinked Hydrogel/Microparticle System for Cartilage Tissue Engineering: Hydrogel/Microparticle System for Cartilage Tissue Engineering. J. Biomed. Mater. Res. A 2018, 106, 1932–1940. [Google Scholar] [CrossRef]
- Song, W.; Ma, Z.; Wang, C.; Li, H.; He, Y. Pro-Chondrogenic and Immunomodulatory Melatonin-Loaded Electrospun Membranes for Tendon-to-Bone Healing. J. Mater. Chem. B 2019, 7, 6564–6575. [Google Scholar] [CrossRef]
- Kouhi, M.; Varshosaz, J.; Hashemibeni, B.; Sarmadi, A. Injectable Gellan Gum/Lignocellulose Nanofibrils Hydrogels Enriched with Melatonin Loaded Forsterite Nanoparticles for Cartilage Tissue Engineering: Fabrication, Characterization and Cell Culture Studies. Mater. Sci. Eng. C 2020, 115, 111114. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Llamas, F.; Hernández-Ruiz, J.; Cuesta, A.; Zamora, S.; Arnao, M.B. Development of a Phytomelatonin-Rich Extract from Cultured Plants with Excellent Biochemical and Functional Properties as an Alternative to Synthetic Melatonin. Antioxidants 2020, 9, 158. [Google Scholar] [CrossRef]
- Grol, M.W.; Lee, B.H. Gene Therapy for Repair and Regeneration of Bone and Cartilage. Curr. Opin. Pharmacol. 2018, 40, 59–66. [Google Scholar] [CrossRef]
- Bellavia, D.; Veronesi, F.; Carina, V.; Costa, V.; Raimondi, L.; De Luca, A.; Alessandro, R.; Fini, M.; Giavaresi, G. Gene Therapy for Chondral and Osteochondral Regeneration: Is the Future Now? Cell. Mol. Life Sci. 2018, 75, 649–667. [Google Scholar] [CrossRef]
- Feng, G.; Zhang, Z.; Dang, M.; Zhang, X.; Doleyres, Y.; Song, Y.; Chen, D.; Ma, P.X. Injectable Nanofibrous Spongy Microspheres for NR4A1 Plasmid DNA Transfection to Reverse Fibrotic Degeneration and Support Disc Regeneration. Biomaterials 2017, 131, 86–97. [Google Scholar] [CrossRef]
- Gilpin, A.; Yang, Y. Decellularization Strategies for Regenerative Medicine: From Processing Techniques to Applications. BioMed Res. Int. 2017, 2017, 1–13. [Google Scholar] [CrossRef]
- Ghosh, P.; Gruber, S.M.S.; Lin, C.-Y.; Whitlock, P.W. Microspheres Containing Decellularized Cartilage Induce Chondrogenesis in Vitro and Remain Functional after Incorporation within a Poly(Caprolactone) Filament Useful for Fabricating a 3D Scaffold. Biofabrication 2018, 10, 025007. [Google Scholar] [CrossRef]
- Fortier, L.A.; Barker, J.U.; Strauss, E.J.; McCarrel, T.M.; Cole, B.J. The Role of Growth Factors in Cartilage Repair. Clin. Orthop. 2011, 469, 2706–2715. [Google Scholar] [CrossRef]
- Kudva, A.K.; Dikina, A.D.; Luyten, F.P.; Alsberg, E.; Patterson, J. Gelatin Microspheres Releasing Transforming Growth Factor Drive in Vitro Chondrogenesis of Human Periosteum Derived Cells in Micromass Culture. Acta Biomater. 2019, 90, 287–299. [Google Scholar] [CrossRef]
- Solorio, L.D.; Vieregge, E.L.; Dhami, C.D.; Dang, P.N.; Alsberg, E. Engineered Cartilage via Self-Assembled HMSC Sheets with Incorporated Biodegradable Gelatin Microspheres Releasing Transforming Growth Factor-Β1. J. Controlled Release 2012, 158, 224–232. [Google Scholar] [CrossRef]
- Sun, Q.; Zhang, L.; Xu, T.; Ying, J.; Xia, B.; Jing, H.; Tong, P. Combined Use of Adipose Derived Stem Cells and TGF-Β3 Microspheres Promotes Articular Cartilage Regeneration in Vivo. Biotech. Histochem. 2018, 93, 168–176. [Google Scholar] [CrossRef]
- Morille, M.; Toupet, K.; Montero-Menei, C.N.; Jorgensen, C.; Noël, D. PLGA-Based Microcarriers Induce Mesenchymal Stem Cell Chondrogenesis and Stimulate Cartilage Repair in Osteoarthritis. Biomaterials 2016, 88, 60–69. [Google Scholar] [CrossRef]
- Vayas, R.; Reyes, R.; Rodríguez-Évora, M.; del Rosario, C.; Delgado, A.; Évora, C. Evaluation of the Effectiveness of a BMSC and BMP-2 Polymeric Trilayer System in Cartilage Repair. Biomed. Mater. 2017, 12, 045001. [Google Scholar] [CrossRef]
- Gavenis, K.; Schneider, U.; Groll, J.; Schmidt-Rohlfing, B. BMP-7-Loaded PGLA Microspheres as a New Delivery System for the Cultivation of Human Chondrocytes in a Collagen Type I Gel: The Common Nude Mouse Model. Int. J. Artif. Organs 2010, 33, 45–53. [Google Scholar] [CrossRef]
- Serra, M.; Brito, C.; Correia, C.; Alves, P.M. Process Engineering of Human Pluripotent Stem Cells for Clinical Application. Trends Biotechnol. 2012, 30, 350–359. [Google Scholar] [CrossRef] [PubMed]
- Tseng, P.-C.; Young, T.-H.; Wang, T.-M.; Peng, H.-W.; Hou, S.-M.; Yen, M.-L. Spontaneous Osteogenesis of MSCs Cultured on 3D Microcarriers through Alteration of Cytoskeletal Tension. Biomaterials 2012, 33, 556–564. [Google Scholar] [CrossRef] [PubMed]
- Simaria, A.S.; Hassan, S.; Varadaraju, H.; Rowley, J.; Warren, K.; Vanek, P.; Farid, S.S. Allogeneic Cell Therapy Bioprocess Economics and Optimization: Single-use Cell Expansion Technologies. Biotechnol. Bioeng. 2014, 111, 69–83. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Cui, Z.; Field, R.; Moloney, M.G.; Rimmer, S.; Ye, H. Thermo-Responsive Microcarriers Based on Poly(N-Isopropylacrylamide). Eur. Polym. J. 2015, 67, 346–364. [Google Scholar] [CrossRef]
- Murasiewicz, H.; Nienow, A.W.; Hanga, M.P.; Coopman, K.; Hewitt, C.J.; Pacek, A.W. Engineering Considerations on the Use of Liquid/Liquid Two-Phase Systems as a Cell Culture Platform: Engineering Considerations on the Use of Liquid/Liquid Two-Phase Systems. J. Chem. Technol. Biotechnol. 2017, 92, 1690–1698. [Google Scholar] [CrossRef]
- Stephenson, M.; Grayson, W. Recent Advances in Bioreactors for Cell-Based Therapies. F1000Research 2018, 7, 517. [Google Scholar] [CrossRef] [PubMed]
- Hundt, B.; Best, C.; Schlawin, N.; Kaßner, H.; Genzel, Y.; Reichl, U. Establishment of a Mink Enteritis Vaccine Production Process in Stirred-Tank Reactor and Wave® Bioreactor Microcarrier Culture in 1–10L Scale. Vaccine 2007, 25, 3987–3995. [Google Scholar] [CrossRef]
- Kiesslich, S.; Vila-Chã Losa, J.P.; Gélinas, J.-F.; Kamen, A.A. Serum-Free Production of RVSV-ZEBOV in Vero Cells: Microcarrier Bioreactor versus Scale-XTM Hydro Fixed-Bed. J. Biotechnol. 2020, 310, 32–39. [Google Scholar] [CrossRef]
- Valle, M.A.; Kaufman, J.; Bentley, W.E.; Shiloach, J. Evaluation of porous microcarriers in fluidized bed reactor for protein production by Heik 293 cells. In New Developments and New Applications in Animal Cell Technology; Merten, O.-W., Perrin, P., Griffiths, B., Eds.; Springer Netherlands: Dordrecht, The Netherlands, 2002; pp. 381–384. ISBN 978-0-7923-5016-3. [Google Scholar]
- Warnock, J.N.; Al-Rubeai, M. Bioreactor Systems for the Production of Biopharmaceuticals from Animal Cells. Biotechnol. Appl. Biochem. 2006, 45, 1. [Google Scholar] [CrossRef]
- Hewitt, C.J.; Lee, K.; Nienow, A.W.; Thomas, R.J.; Smith, M.; Thomas, C.R. Expansion of Human Mesenchymal Stem Cells on Microcarriers. Biotechnol. Lett. 2011, 33, 2325–2335. [Google Scholar] [CrossRef]
- Rafiq, Q.A.; Brosnan, K.M.; Coopman, K.; Nienow, A.W.; Hewitt, C.J. Culture of Human Mesenchymal Stem Cells on Microcarriers in a 5 l Stirred-Tank Bioreactor. Biotechnol. Lett. 2013, 35, 1233–1245. [Google Scholar] [CrossRef] [PubMed]
- Berry, J.D.; Liovic, P.; Šutalo, I.D.; Stewart, R.L.; Glattauer, V.; Meagher, L. Characterisation of Stresses on Microcarriers in a Stirred Bioreactor. Appl. Math. Model. 2016, 40, 6787–6804. [Google Scholar] [CrossRef]
- Torizal, F.G.; Horiguchi, I.; Sakai, Y. Physiological Microenvironmental Conditions in Different Scalable Culture Systems for Pluripotent Stem Cell Expansion and Differentiation. Open Biomed. Eng. J. 2019, 13, 41–54. [Google Scholar] [CrossRef]
- Schrobback, K.; Klein, T.J.; Schuetz, M.; Upton, Z.; Leavesley, D.I.; Malda, J. Adult Human Articular Chondrocytes in a Microcarrier-Based Culture System: Expansion and Redifferentiation: MICROCARRIER-EXPANSION OF HUMAN ARTICULAR CHONDROCYTES. J. Orthop. Res. 2011, 29, 539–546. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.; Hsiung, M.; Cheng, C.; Tzanakakis, E.S. Facile Engineering of Xeno-Free Microcarriers for the Scalable Cultivation of Human Pluripotent Stem Cells in Stirred Suspension. Tissue Eng. Part A 2014, 20, 588–599. [Google Scholar] [CrossRef]
- Zhou, Z.; Wu, W.; Fang, J.; Yin, J. Polymer-Based Porous Microcarriers as Cell Delivery Systems for Applications in Bone and Cartilage Tissue Engineering. Int. Mater. Rev. 2021, 66, 77–113. [Google Scholar] [CrossRef]
- Georgi, N.; van Blitterswijk, C.; Karperien, M. Mesenchymal Stromal/Stem Cell–or Chondrocyte-Seeded Microcarriers as Building Blocks for Cartilage Tissue Engineering. Tissue Eng. Part A 2014, 20, 2513–2523. [Google Scholar] [CrossRef] [PubMed]
- Phillips, B.W.; Horne, R.; Lay, T.S.; Rust, W.L.; Teck, T.T.; Crook, J.M. Attachment and Growth of Human Embryonic Stem Cells on Microcarriers. J. Biotechnol. 2008, 138, 24–32. [Google Scholar] [CrossRef]
- Nie, Y.; Bergendahl, V.; Hei, D.J.; Jones, J.M.; Palecek, S.P. Scalable Culture and Cryopreservation of Human Embryonic Stem Cells on Microcarriers. Biotechnol. Prog. 2009, 25, 20–31. [Google Scholar] [CrossRef]
- Muoio, F.; Panella, S.; Jossen, V.; Lindner, M.; Harder, Y.; Müller, M.; Eibl, R.; Tallone, T. Human Adipose Stem Cells (HASCs) Grown on Biodegradable Microcarriers in Serum- and Xeno-Free Medium Preserve Their Undifferentiated Status. J. Funct. Biomater. 2021, 12, 25. [Google Scholar] [CrossRef] [PubMed]
- Saei Arezoumand, K.; Alizadeh, E.; Pilehvar-Soltanahmadi, Y.; Esmaeillou, M.; Zarghami, N. An Overview on Different Strategies for the Stemness Maintenance of MSCs. Artif. Cells Nanomed. Biotechnol. 2017, 45, 1255–1271. [Google Scholar] [CrossRef]
- Gupta, P.; Geris, L.; Luyten, F.P.; Papantoniou, I. An Integrated Bioprocess for the Expansion and Chondrogenic Priming of Human Periosteum-Derived Progenitor Cells in Suspension Bioreactors. Biotechnol. J. 2018, 13, 1700087. [Google Scholar] [CrossRef]
- Murphy, S.V.; Atala, A. 3D Bioprinting of Tissues and Organs. Nat. Biotechnol. 2014, 32, 773–785. [Google Scholar] [CrossRef]
- Daly, A.C.; Riley, L.; Segura, T.; Burdick, J.A. Hydrogel Microparticles for Biomedical Applications. Nat. Rev. Mater. 2020, 5, 20–43. [Google Scholar] [CrossRef]
- Zhang, Y.; Ellison, S.T.; Duraivel, S.; Morley, C.D.; Taylor, C.R.; Angelini, T.E. 3D Printed Collagen Structures at Low Concentrations Supported by Jammed Microgels. Bioprinting 2021, 21, e00121. [Google Scholar] [CrossRef]
- Jeon, O.; Lee, Y.B.; Hinton, T.J.; Feinberg, A.W.; Alsberg, E. Cryopreserved Cell-Laden Alginate Microgel Bioink for 3D Bioprinting of Living Tissues. Mater. Today Chem. 2019, 12, 61–70. [Google Scholar] [CrossRef]
- Truong, N.F.; Kurt, E.; Tahmizyan, N.; Lesher-Pérez, S.C.; Chen, M.; Darling, N.J.; Xi, W.; Segura, T. Microporous Annealed Particle Hydrogel Stiffness, Void Space Size, and Adhesion Properties Impact Cell Proliferation, Cell Spreading, and Gene Transfer. Acta Biomater. 2019, 94, 160–172. [Google Scholar] [CrossRef]
- Caldwell, A.S.; Campbell, G.T.; Shekiro, K.M.T.; Anseth, K.S. Clickable Microgel Scaffolds as Platforms for 3D Cell Encapsulation. Adv. Healthc. Mater. 2017, 6, 1700254. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kulchar, R.J.; Denzer, B.R.; Chavre, B.M.; Takegami, M.; Patterson, J. A Review of the Use of Microparticles for Cartilage Tissue Engineering. Int. J. Mol. Sci. 2021, 22, 10292. https://doi.org/10.3390/ijms221910292
Kulchar RJ, Denzer BR, Chavre BM, Takegami M, Patterson J. A Review of the Use of Microparticles for Cartilage Tissue Engineering. International Journal of Molecular Sciences. 2021; 22(19):10292. https://doi.org/10.3390/ijms221910292
Chicago/Turabian StyleKulchar, Rachel J., Bridget R. Denzer, Bharvi M. Chavre, Mina Takegami, and Jennifer Patterson. 2021. "A Review of the Use of Microparticles for Cartilage Tissue Engineering" International Journal of Molecular Sciences 22, no. 19: 10292. https://doi.org/10.3390/ijms221910292
APA StyleKulchar, R. J., Denzer, B. R., Chavre, B. M., Takegami, M., & Patterson, J. (2021). A Review of the Use of Microparticles for Cartilage Tissue Engineering. International Journal of Molecular Sciences, 22(19), 10292. https://doi.org/10.3390/ijms221910292





