Fractionated Irradiation of Right Thorax Induces Abscopal Damage on Bone Marrow Cells via TNF-α and SAA
Abstract
:1. Introduction
2. Results
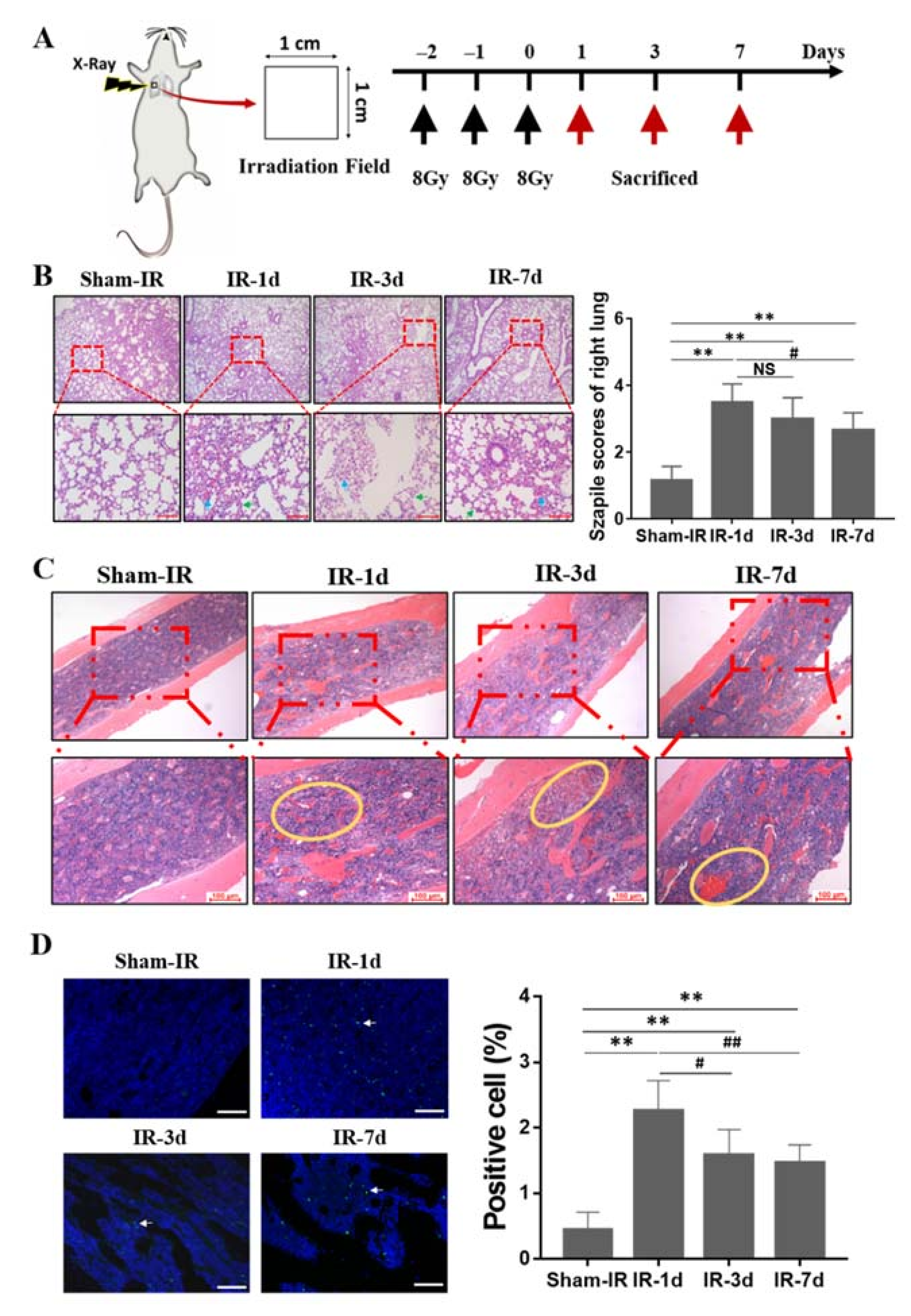
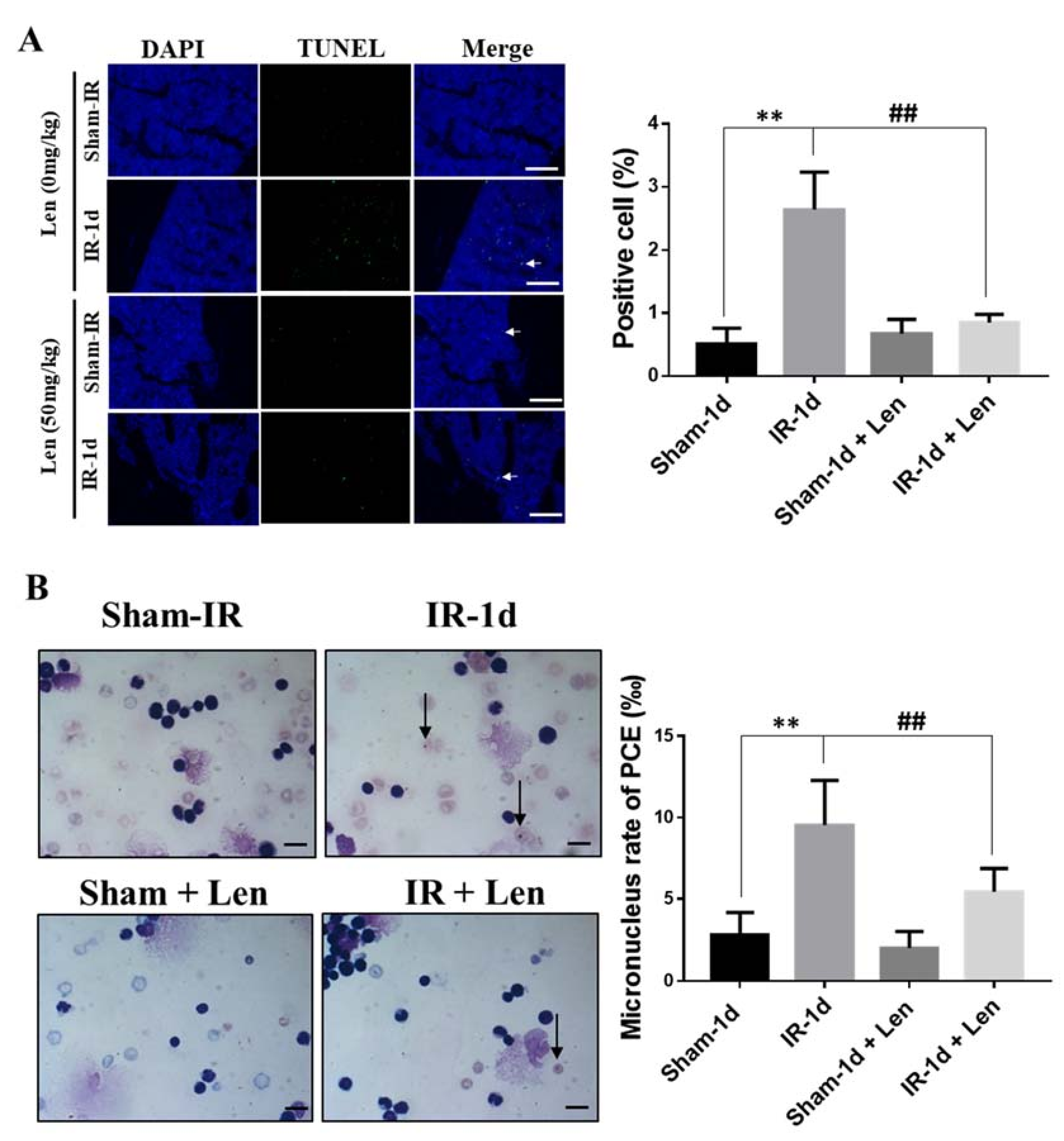
2.1. Thoracic Irradiation Induces Tissue Injury in Bone Marrow Cells
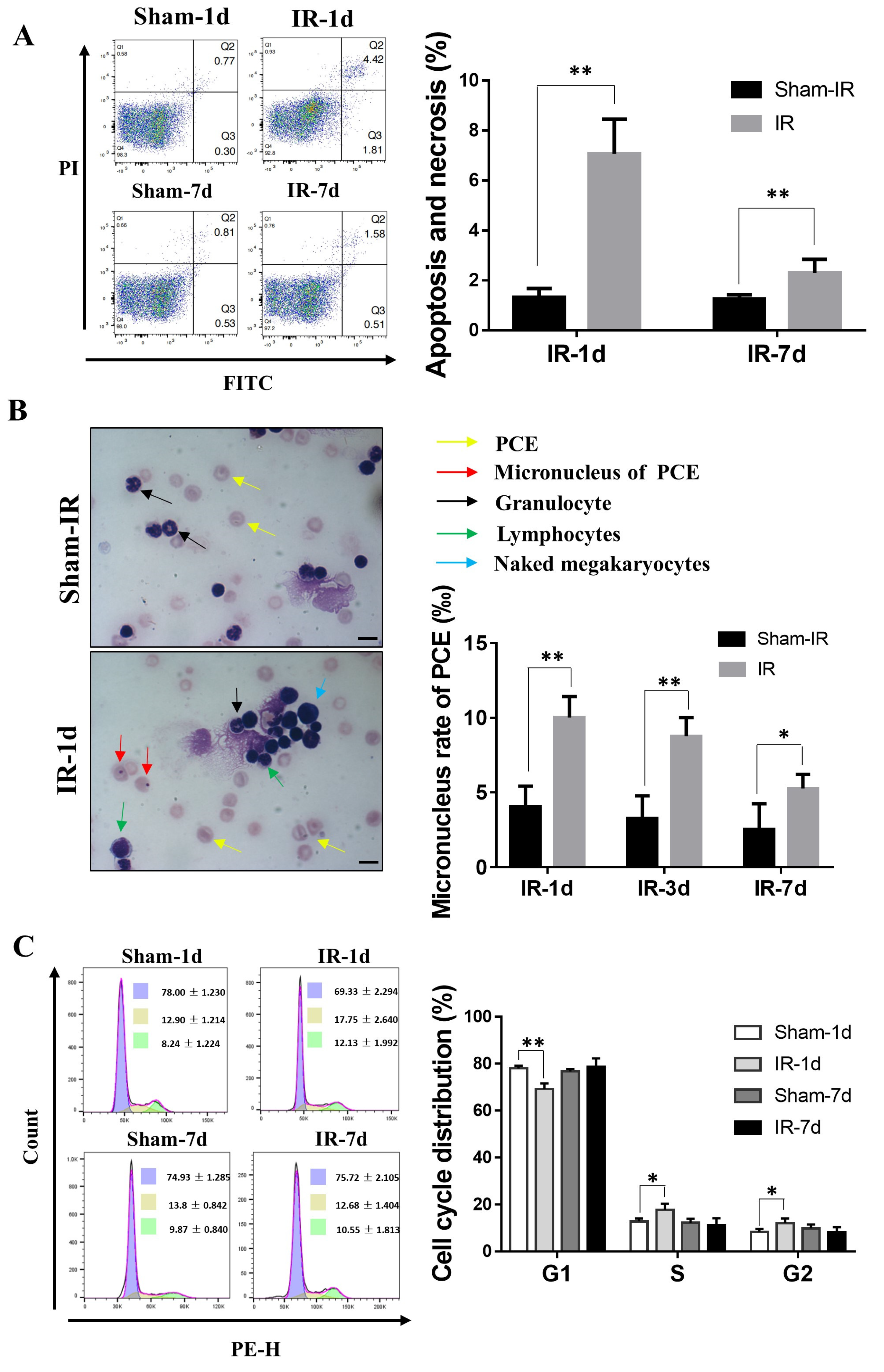
2.2. Th-IR Induces Bone Marrow Cell Damage and Cell Cycle Alteration
2.3. Proteomic Analysis of Differentially Expressed Proteins in the Serum of Mice after Th-IR
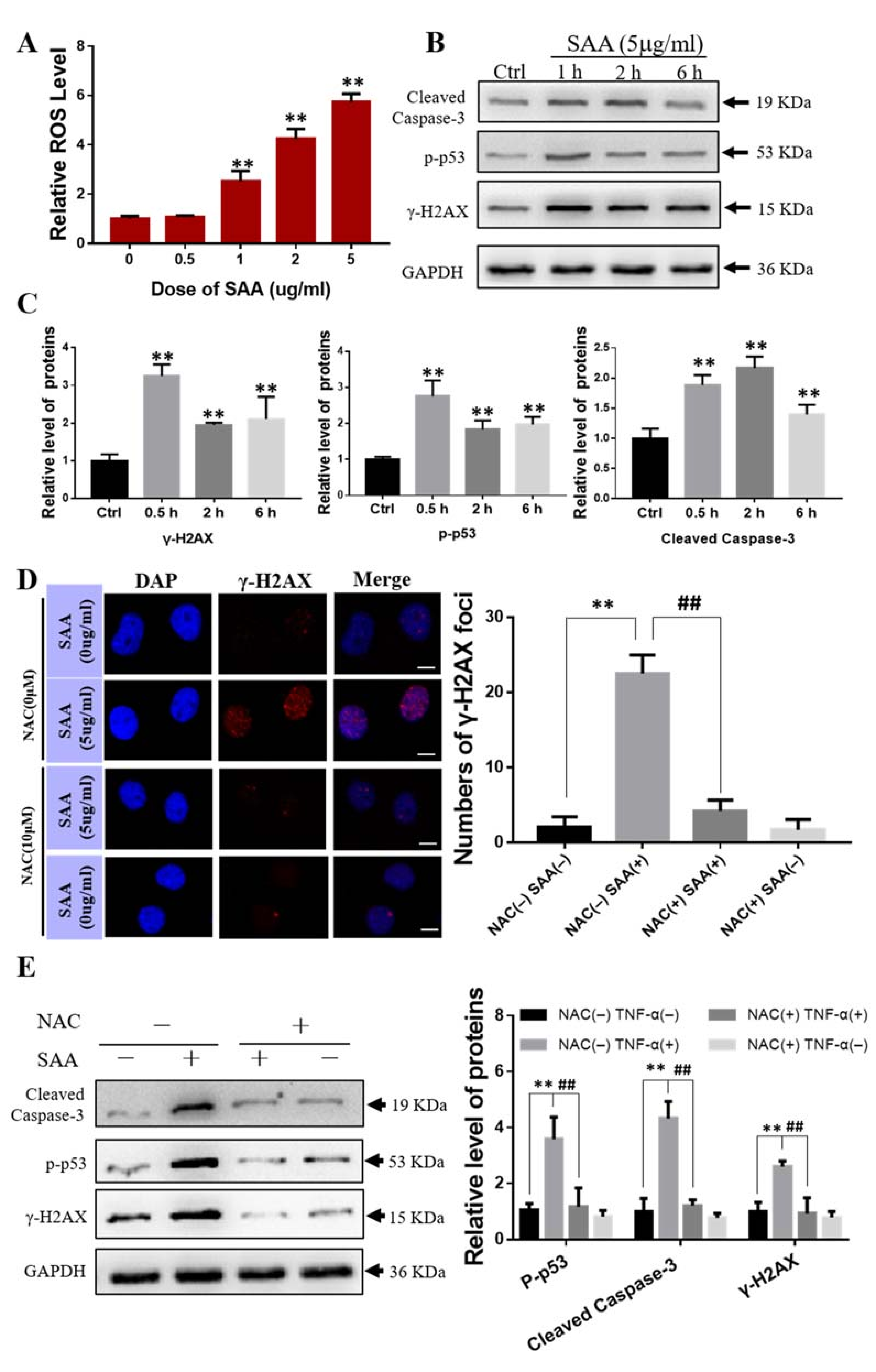
2.4. Effect of Exogenous SAA on Bone Marrow Mesenchymal Stem Cells
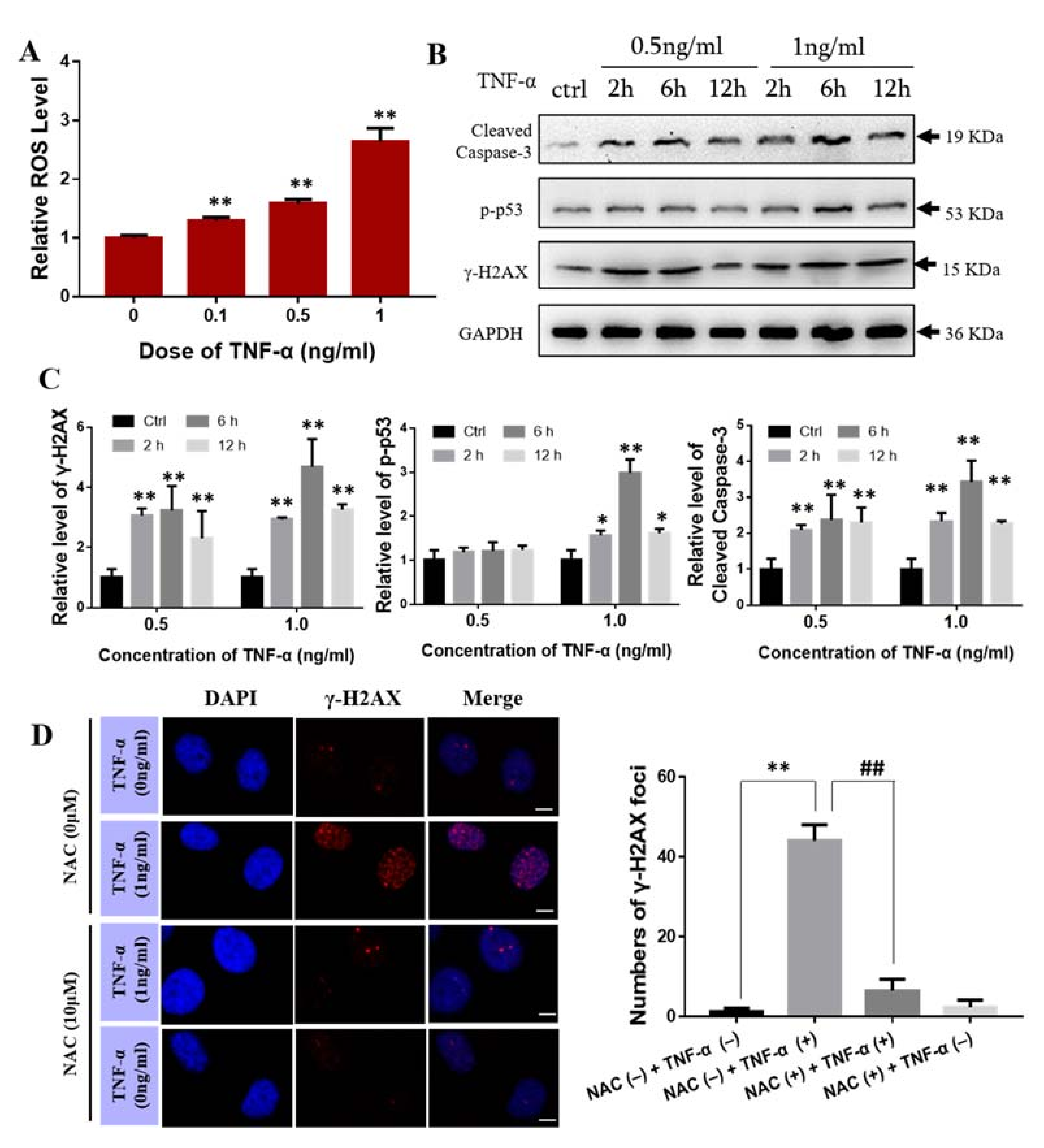
2.5. Effect of TNF-α on Bone Marrow Mesenchymal Stem Cells
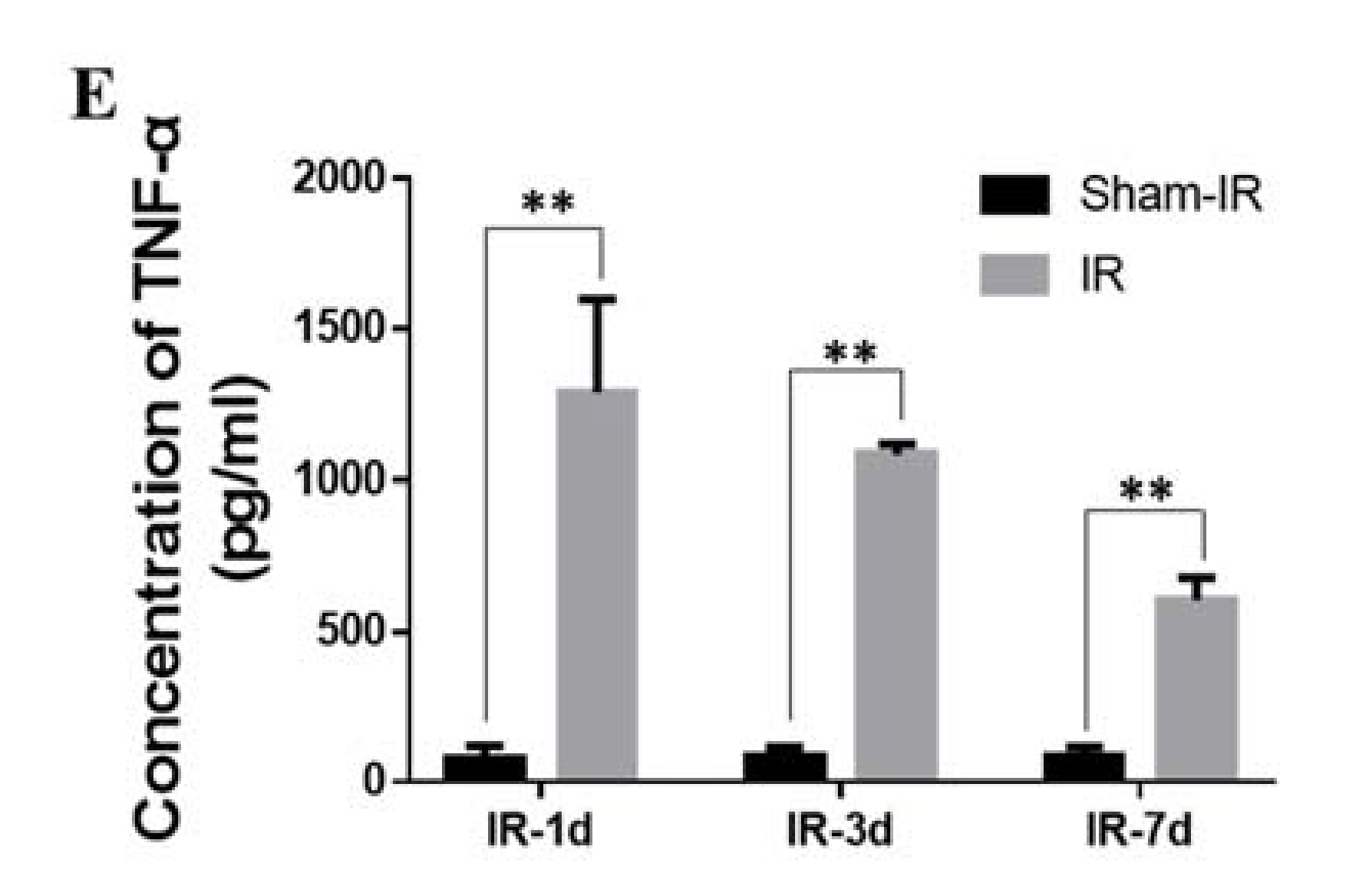
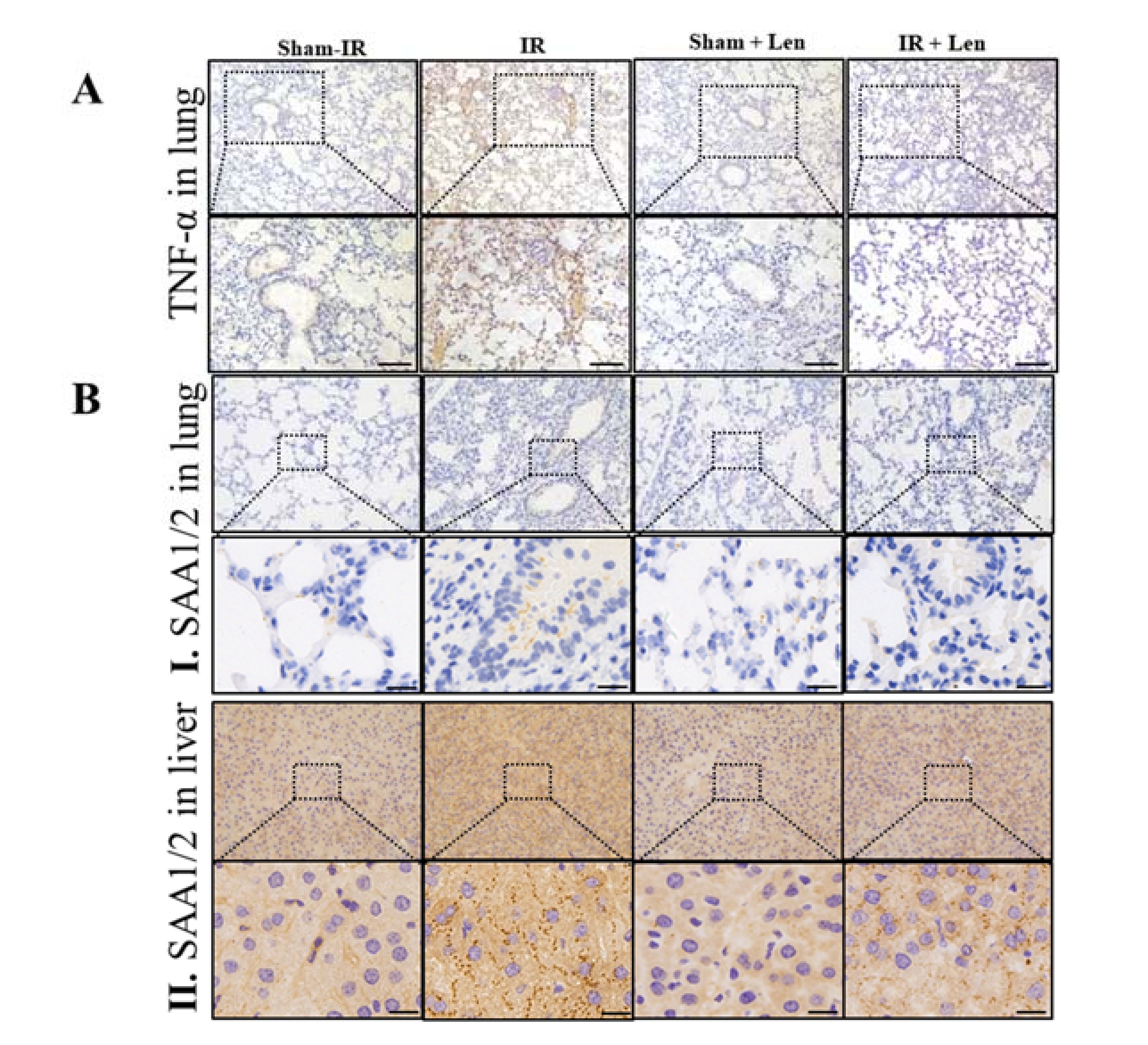
2.6. TNF-α Induces the Expression of SAA In Vivo and In Vitro
2.7. TNF-α Contributes to the Abscopal Bone Marrow Damage in the Th-IR Mice
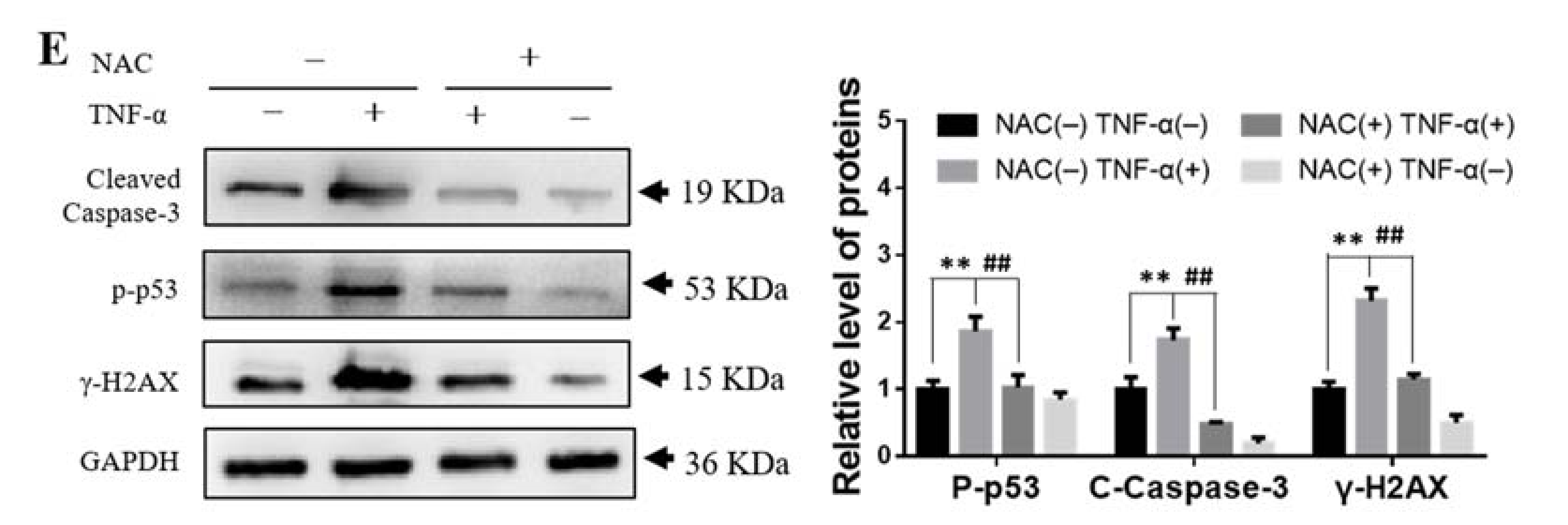
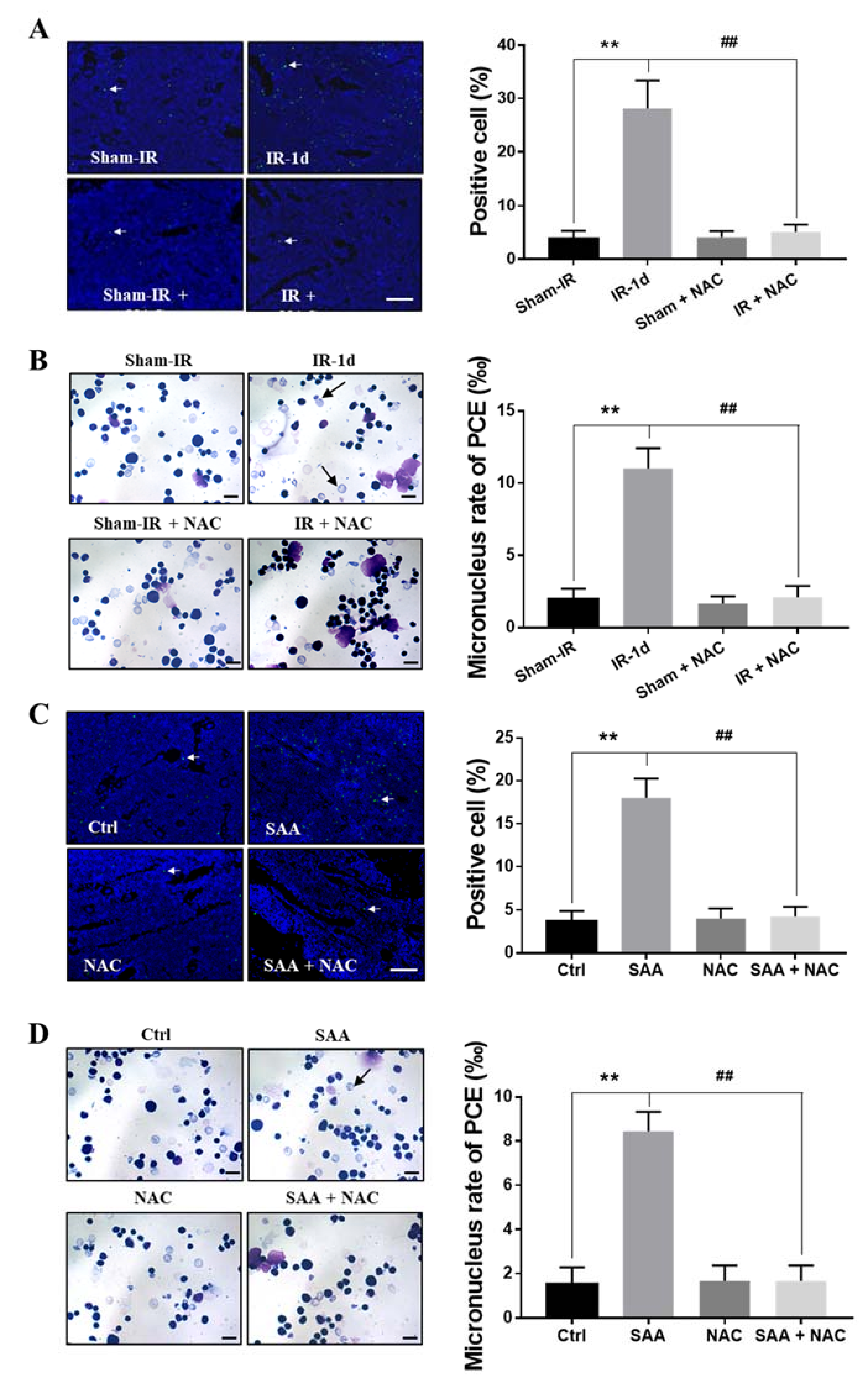
2.8. ROS Scavenger NAC Eliminated Th-IR/SAA-Induced Bone Marrow Damage in Mice
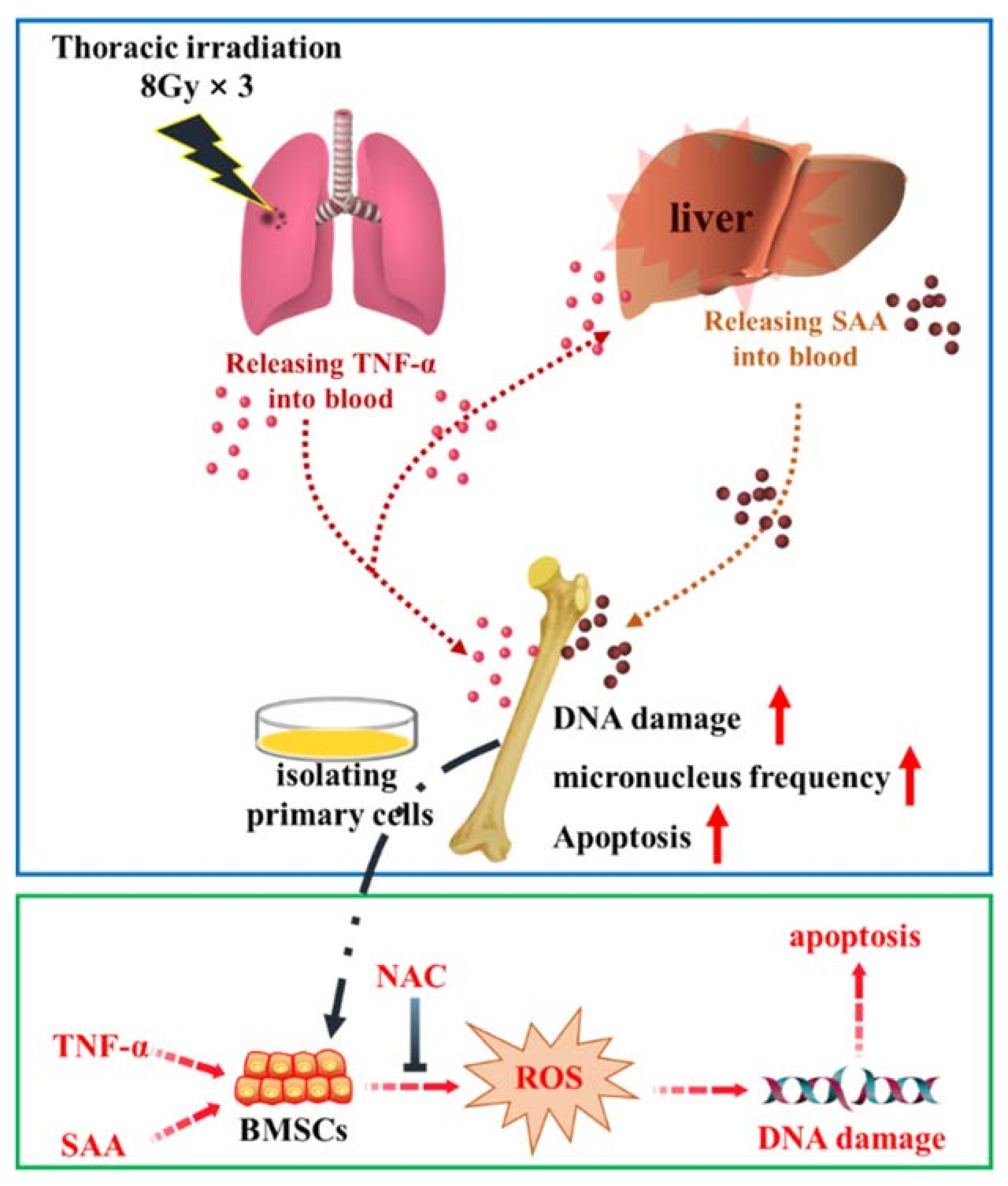
3. Discussion
4. Materials and Methods
4.1. Animal and Treatment
4.2. Detection of Bone Marrow Cell Apoptosis
4.3. Cell Cycle Analysis of Bone Marrow Cells
4.4. Measurement of Micronucleus in Polychromatic Erythrocytes (PCEs)
4.5. Bone Marrow Stem Cells (BMSCs) Isolation, Culture and Identification
4.6. Western Blot Assay
4.7. Histological Analysis
4.8. TUNEL Assay
4.9. Tandem Mass Tagging (TMT) Proteomics Analysis
4.10. Detection of Inflammatory Factors
4.11. ROS Measurement
4.12. Cell Immunofluorescence Assay
4.13. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Das, M. Individualised adaptive radiotherapy in liver cancer. Lancet Oncol. 2017, 18, e520. [Google Scholar] [CrossRef]
- Chen, Y.P.; Chan, A.T.C.; Le, Q.T.; Blanchard, P.; Sun, Y.; Ma, J. Nasopharyngeal carcinoma. Lancet 2019, 394, 64–80. [Google Scholar] [CrossRef]
- Palma, D.A.; Olson, R.; Harrow, S.; Gaede, S.; Louie, A.V.; Haasbeek, C.; Mulroy, L.; Lock, M.; Rodrigues, G.B.; Yaremko, B.P.; et al. Stereotactic Ablative Radiotherapy for the Comprehensive Treatment of Oligometastatic Cancers: Long-Term Results of the SABR-COMET Phase II Randomized Trial. J. Clin. Oncol. 2020, 38, 2830–2838. [Google Scholar] [CrossRef]
- Farias, V.A.; Tovar, I.; Del Moral, R.; O’Valle, F.; Exposito, J.; Oliver, F.J.; Ruiz de Almodovar, J.M. Enhancing the Bystander and Abscopal Effects to Improve Radiotherapy Outcomes. Front. Oncol. 2019, 9, 1381. [Google Scholar] [CrossRef] [Green Version]
- Mohye El-Din, A.A.; Abdelrazzak, A.B.; Ahmed, M.T.; El-Missiry, M.A. Radiation induced bystander effects in the spleen of cranially-irradiated rats. Br. J. Radiol. 2017, 90, 20170278. [Google Scholar] [CrossRef] [PubMed]
- Tamminga, J.; Koturbash, I.; Baker, M.; Kutanzi, K.; Kathiria, P.; Pogribny, I.P.; Sutherland, R.J.; Kovalchuk, O. Paternal cranial irradiation induces distant bystander DNA damage in the germline and leads to epigenetic alterations in the offspring. Cell Cycle 2008, 7, 1238–1245. [Google Scholar] [CrossRef] [Green Version]
- Ngwa, W.; Irabor, O.C.; Schoenfeld, J.D.; Hesser, J.; Demaria, S.; Formenti, S.C. Using immunotherapy to boost the abscopal effect. Nat. Rev. Cancer 2018, 18, 313–322. [Google Scholar] [CrossRef] [PubMed]
- Koturbash, I.; Rugo, R.E.; Hendricks, C.A.; Loree, J.; Thibault, B.; Kutanzi, K.; Pogribny, I.; Yanch, J.C.; Engelward, B.P.; Kovalchuk, O. Irradiation induces DNA damage and modulates epigenetic effectors in distant bystander tissue in vivo. Oncogene 2006, 25, 4267–4275. [Google Scholar] [CrossRef] [Green Version]
- Suzuki, K.; Yamashita, S. Radiation-Induced Bystander Response: Mechanism and Clinical Implications. Adv. Wound Care New Rochelle 2014, 3, 16–24. [Google Scholar] [CrossRef] [Green Version]
- Mancuso, M.; Pasquali, E.; Leonardi, S.; Tanori, M.; Rebessi, S.; Di Majo, V.; Pazzaglia, S.; Toni, M.P.; Pimpinella, M.; Covelli, V.; et al. Oncogenic bystander radiation effects in Patched heterozygous mouse cerebellum. Proc. Natl. Acad. Sci. USA 2008, 105, 12445–12450. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hekim, N.; Cetin, Z.; Nikitaki, Z.; Cort, A.; Saygili, E.I. Radiation triggering immune response and inflammation. Cancer Lett. 2015, 368, 156–163. [Google Scholar] [CrossRef]
- Diegeler, S.; Hellweg, C.E. Intercellular Communication of Tumor Cells and Immune Cells after Exposure to Different Ionizing Radiation Qualities. Front. Immunol. 2017, 8, 664. [Google Scholar] [CrossRef] [Green Version]
- Vacchelli, E.; Aranda, F.; Bloy, N.; Buqué, A.; Cremer, I.; Eggermont, A.; Fridman, W.H.; Fucikova, J.; Galon, J.; Spisek, R.; et al. Trial Watch-Immunostimulation with cytokines in cancer therapy. Oncoimmunology 2016, 5, e1115942. [Google Scholar] [CrossRef] [Green Version]
- Hu, L.; Chen, H.; Zhang, X.; Feng, Z.; Zhang, H.; Meng, Q. Rosiglitazone ameliorates radiation-induced intestinal inflammation in rats by inhibiting NLRP3 inflammasome and TNF-α production. J. Radiat. Res. 2020, 61, 842–850. [Google Scholar] [CrossRef]
- Ala, M.; Mohammad Jafari, R.; Ala, M.; Agbele, A.T.; Hejazi, S.M.; Tavangar, S.M.; Mahdavi, S.R.M.; Dehpour, A.R. Sumatriptan alleviates radiation-induced oral mucositis in rats by inhibition of NF-kB and ERK activation, prevention of TNF-α and ROS release. Arch. Oral. Biol. 2020, 119, 104919. [Google Scholar] [CrossRef] [PubMed]
- Malik, G.; Wilting, J.; Hess, C.F.; Ramadori, G.; Malik, I.A. PECAM-1 modulates liver damage induced by synergistic effects of TNF-α and irradiation. J. Cell. Mol. Med. 2019, 23, 3336–3344. [Google Scholar] [CrossRef] [PubMed]
- Cachaco, A.S.; Carvalho, T.; Santos, A.C.; Igreja, C.; Fragoso, R.; Osorio, C.; Ferreira, M.; Serpa, J.; Correia, S.; Pinto-do, O.P.; et al. TNF-alpha regulates the effects of irradiation in the mouse bone marrow microenvironment. PLoS ONE 2010, 5, e8980. [Google Scholar] [CrossRef] [Green Version]
- Zhang, J.; Yao, D.; Song, Y.; Pan, Y.; Zhu, L.; Bai, Y.; Xu, Y.; Zhang, J.; Shao, C. Fractionated irradiation of right thorax induces abscopal damage on testes leading to decline in fertility. Sci. Rep. 2019, 9, 15221. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.; Zhu, L.; Song, Y.; Zhao, X.; Chen, Q.; Pan, Y.; Zhang, J.; Bai, Y.; Zhang, H.; Shao, C. Radiation-induced abscopal reproductive effect is driven by TNF-α/p38 MAPK/Rac1 axis in Sertoli cells. Theranostics 2021, 11, 5742–5758. [Google Scholar] [CrossRef]
- Huang, S.; Xu, L.; Sun, Y.; Wu, T.; Wang, K.; Li, G. An improved protocol for isolation and culture of mesenchymal stem cells from mouse bone marrow. J. Orthop. Transl. 2015, 3, 26–33. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fardid, R.; Najafi, M.; Salajegheh, A.; Kazemi, E.; Rezaeyan, A. Radiation-induced non-targeted effect in vivo: Evaluation of cyclooygenase-2 and endothelin-1 gene expression in rat heart tissues. J. Cancer Res. Ther. 2017, 13, 51–55. [Google Scholar] [CrossRef]
- Zhang, J.; Zhou, S.; Zhou, Y.; Feng, F.; Wang, Q.; Zhu, X.; Zhao, J.; Fu, H.; Lv, M.; Ai, H.; et al. Adipose-Derived Mesenchymal Stem Cells (ADSCs) with the Potential to Ameliorate Platelet Recovery, Enhance Megakaryopoiesis, and Inhibit Apoptosis of Bone Marrow Cells in a Mouse Model of Radiation-Induced Thrombocytopenia. Cell Transplant. 2016, 25, 261–273. [Google Scholar] [CrossRef] [PubMed]
- Selig, C.; Kreja, L.; Müller, H.; Seifried, E.; Nothdurft, W. Hematologic effects of recombinant human interleukin-6 in dogs exposed to a total-body radiation dose of 2.4 Gy. Exp. Hematol. 1994, 22, 551–558. [Google Scholar] [PubMed]
- Rastogi, S.; Coates, P.J.; Lorimore, S.A.; Wright, E.G. Bystander-type effects mediated by long-lived inflammatory signaling in irradiated bone marrow. Radiat. Res. 2012, 177, 244–250. [Google Scholar] [CrossRef]
- Szatmári, T.; Kis, D.; Bogdándi, E.N.; Benedek, A.; Bright, S.; Bowler, D.; Persa, E.; Kis, E.; Balogh, A.; Naszályi, L.N.; et al. Extracellular Vesicles Mediate Radiation-Induced Systemic Bystander Signals in the Bone Marrow and Spleen. Front. Immunol. 2017, 8, 347. [Google Scholar] [CrossRef] [Green Version]
- Watson, G.E.; Lorimore, S.A.; Macdonald, D.A.; Wright, E.G. Chromosomal instability in unirradiated cells induced in vivo by a bystander effect of ionizing radiation. Cancer Res. 2000, 60, 5608–5611. [Google Scholar]
- Lorimore, S.A.; McIlrath, J.M.; Coates, P.J.; Wright, E.G. Chromosomal instability in unirradiated hemopoietic cells resulting from a delayed in vivo bystander effect of gamma radiation. Cancer Res. 2005, 65, 5668–5673. [Google Scholar] [CrossRef] [Green Version]
- Bannister, L.A.; Mantha, R.R.; Devantier, Y.; Petoukhov, E.S.; Brideau, C.L.; Serran, M.L.; Klokov, D.Y. Dose and Radioadaptive Response Analysis of Micronucleus Induction in Mouse Bone Marrow. Int. J. Mol. Sci. 2016, 17, 1548. [Google Scholar] [CrossRef] [Green Version]
- Ye, R.D.; Sun, L. Emerging functions of serum amyloid A in inflammation. J. Leukoc. Biol. 2015, 98, 923–929. [Google Scholar] [CrossRef] [PubMed]
- Urieli-Shoval, S.; Cohen, P.; Eisenberg, S.; Matzner, Y. Widespread expression of serum amyloid A in histologically normal human tissues. Predominant localization to the epithelium. J. Histochem. Cytochem. 1998, 46, 1377–1384. [Google Scholar] [CrossRef] [Green Version]
- De Buck, M.; Gouwy, M.; Wang, J.M.; Van Snick, J.; Opdenakker, G.; Struyf, S.; Van Damme, J. Structure and Expression of Different Serum Amyloid A (SAA) Variants and their Concentration-Dependent Functions during Host Insults. Curr. Med. Chem. 2016, 23, 1725–1755.29. [Google Scholar] [CrossRef] [Green Version]
- Ossetrova, N.I.; Sandgren, D.J.; Blakely, W.F. Protein biomarkers for enhancement of radiation dose and injury assessment in nonhuman primate total-body irradiation model. Radiat. Prot. Dosim. 2014, 159, 61–76. [Google Scholar] [CrossRef] [PubMed]
- Morrow, J.F.; Stearman, R.S.; Peltzman, C.G.; Potter, D.A. Induction of hepatic synthesis of serum amyloid A protein and actin. Proc. Natl. Acad. Sci. USA 1981, 78, 4718–4722. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sproull, M.; Kramp, T.; Tandle, A.; Shankavaram, U.; Camphausen, K. Multivariate Analysis of Radiation Responsive Proteins to Predict Radiation Exposure in Total-Body Irradiation and Partial-Body Irradiation Models. Radiat. Res. 2017, 187, 251–258. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sproull, M.; Kramp, T.; Tandle, A.; Shankavaram, U.; Camphausen, K. Serum Amyloid A as a Biomarker for Radiation Exposure. Radiat. Res. 2015, 184, 14–23. [Google Scholar] [CrossRef] [Green Version]
- Di Meo, S.; Reed, T.T.; Venditti, P.; Victor, V.M. Role of ROS and RNS Sources in Physiological and Pathological Conditions. Oxid. Med. Cell. Longev. 2016, 2016, 1245049. [Google Scholar] [CrossRef]
- Lee, S.; Lee, J.Y.; Lee, E.W.; Park, S.; Kang, D.H.; Min, C.; Lee, D.J.; Kang, D.; Song, J.; Kwon, J.; et al. Absence of Cytosolic 2-Cys Prx Subtypes I and II Exacerbates TNF-α-Induced Apoptosis via Different Routes. Cell Rep. 2019, 26, 2194–2211. [Google Scholar] [CrossRef] [Green Version]
- Otano, I.; Alvarez, M.; Minute, L.; Ochoa, M.C.; Migueliz, I.; Molina, C.; Azpilikueta, A.; de Andrea, C.E.; Etxeberria, I.; Sanmamed, M.F.; et al. Human CD8 T cells are susceptible to TNF-mediated activation-induced cell death. Theranostics 2020, 10, 4481–4489. [Google Scholar] [CrossRef]
- Lee, A.S.; Kim, J.S.; Lee, Y.J.; Kang, D.G.; Lee, H.S. Anti-TNF-α activity of Portulaca oleracea in vascular endothelial cells. Int. J. Mol. Sci. 2012, 13, 5628–5644. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhao, W.; Feng, H.; Guo, S.; Han, Y.; Chen, X. Danshenol A inhibits TNF-α-induced expression of intercellular adhesion molecule-1 (ICAM-1) mediated by NOX4 in endothelial cells. Sci. Rep. 2017, 7, 12953. [Google Scholar] [CrossRef] [Green Version]
- Mann, J.; Yang, N.; Montpetit, R.; Kirschenman, R.; Lemieux, H.; Goping, I.S. BAD sensitizes breast cancer cells to docetaxel with increased mitotic arrest and necroptosis. Sci. Rep. 2020, 10, 355. [Google Scholar] [CrossRef] [PubMed]
- De Buck, M.; Gouwy, M.; Wang, J.M.; Van Snick, J.; Proost, P.; Struyf, S.; Van Damme, J. The cytokine-serum amyloid A-chemokine network. Cytokine Growth Factor Rev. 2016, 30, 55–69. [Google Scholar] [CrossRef]
- Thorn, C.F.; Lu, Z.Y.; Whitehead, A.S. Regulation of the human acute phase serum amyloid A genes by tumour necrosis factor-alpha, interleukin-6 and glucocorticoids in hepatic and epithelial cell lines. Scand. J. Immunol. 2004, 59, 152–158. [Google Scholar] [CrossRef] [PubMed]
- Fong, Y.; Tracey, K.J.; Moldawer, L.L.; Hesse, D.G.; Manogue, K.B.; Kenney, J.S.; Lee, A.T.; Kuo, G.C.; Allison, A.C.; Lowry, S.F.; et al. Antibodies to cachectin/tumor necrosis factor reduce interleukin 1 beta and interleukin 6 appearance during lethal bacteremia. J. Exp. Med. 1989, 170, 1627–1633. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van Deventer, S.J.; Büller, H.R.; ten Cate, J.W.; Aarden, L.A.; Hack, C.E.; Sturk, A. Experimental endotoxemia in humans: Analysis of cytokine release and coagulation, fibrinolytic, and complement pathways. Blood 1990, 76, 2520–2526. [Google Scholar] [CrossRef] [Green Version]
- Kumar, A.; Anand, T.; Bhattacharyya, J.; Sharma, A.; Jaganathan, B.G. K562 chronic myeloid leukemia cells modify osteogenic differentiation and gene expression of bone marrow stromal cells. J. Cell Commun. Signal. 2018, 12, 441–450. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Song, Y.; Hu, S.; Zhang, J.; Zhu, L.; Zhao, X.; Chen, Q.; Zhang, J.; Bai, Y.; Pan, Y.; Shao, C. Fractionated Irradiation of Right Thorax Induces Abscopal Damage on Bone Marrow Cells via TNF-α and SAA. Int. J. Mol. Sci. 2021, 22, 9964. https://doi.org/10.3390/ijms22189964
Song Y, Hu S, Zhang J, Zhu L, Zhao X, Chen Q, Zhang J, Bai Y, Pan Y, Shao C. Fractionated Irradiation of Right Thorax Induces Abscopal Damage on Bone Marrow Cells via TNF-α and SAA. International Journal of Molecular Sciences. 2021; 22(18):9964. https://doi.org/10.3390/ijms22189964
Chicago/Turabian StyleSong, Yimeng, Songling Hu, Junling Zhang, Lin Zhu, Xinrui Zhao, Qianping Chen, Jianghong Zhang, Yang Bai, Yan Pan, and Chunlin Shao. 2021. "Fractionated Irradiation of Right Thorax Induces Abscopal Damage on Bone Marrow Cells via TNF-α and SAA" International Journal of Molecular Sciences 22, no. 18: 9964. https://doi.org/10.3390/ijms22189964
APA StyleSong, Y., Hu, S., Zhang, J., Zhu, L., Zhao, X., Chen, Q., Zhang, J., Bai, Y., Pan, Y., & Shao, C. (2021). Fractionated Irradiation of Right Thorax Induces Abscopal Damage on Bone Marrow Cells via TNF-α and SAA. International Journal of Molecular Sciences, 22(18), 9964. https://doi.org/10.3390/ijms22189964






