Formulation, Characterization and Cytotoxicity Effects of Novel Thymoquinone-PLGA-PF68 Nanoparticles
Abstract
:1. Introduction
2. Results
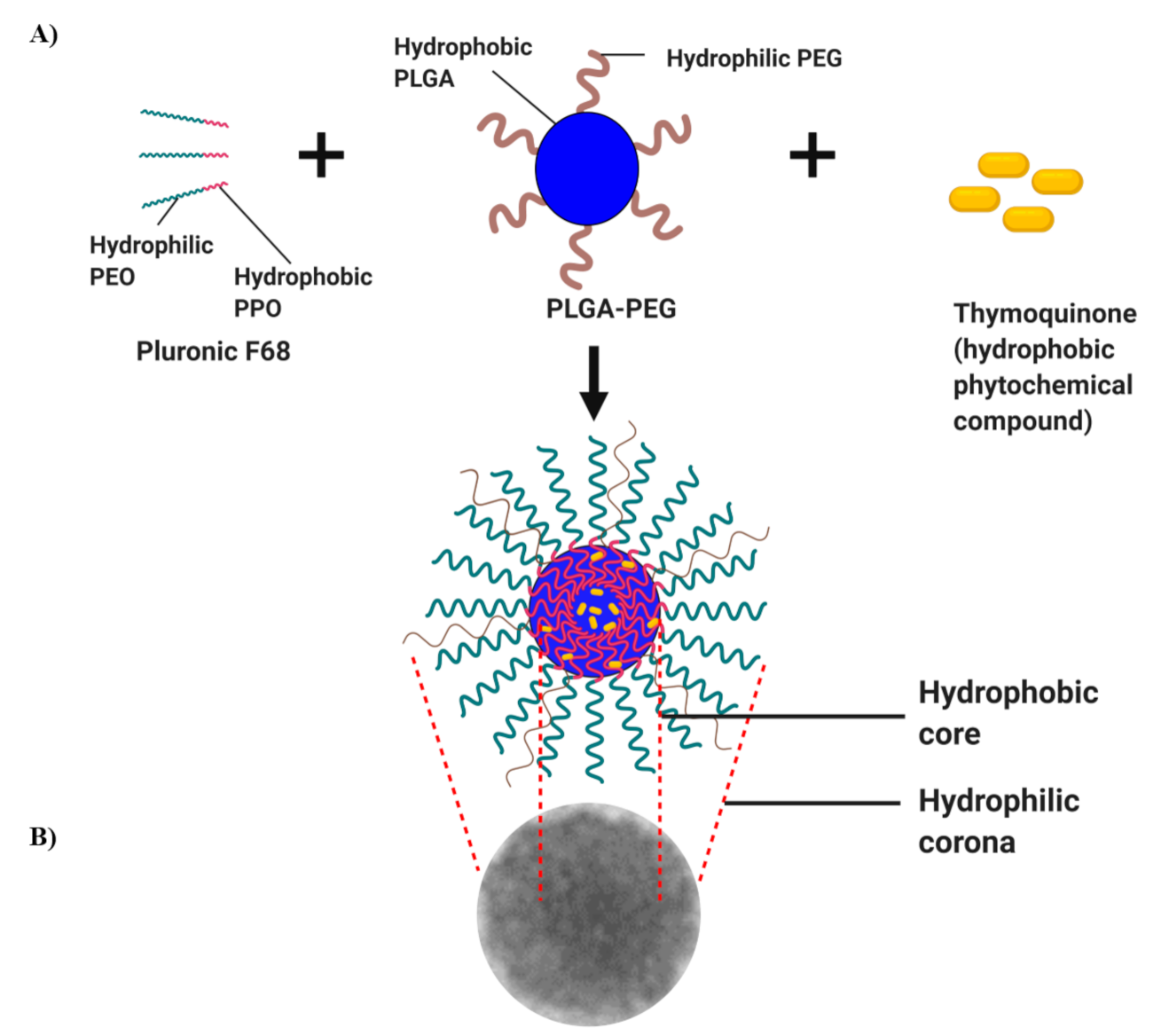
2.1. Characterization of TQ-PLGA-PF68 Nanoparticles
2.1.1. FTIR Spectroscopic Analysis
2.1.2. Polydispersity Index (PDI) and Zeta Potential
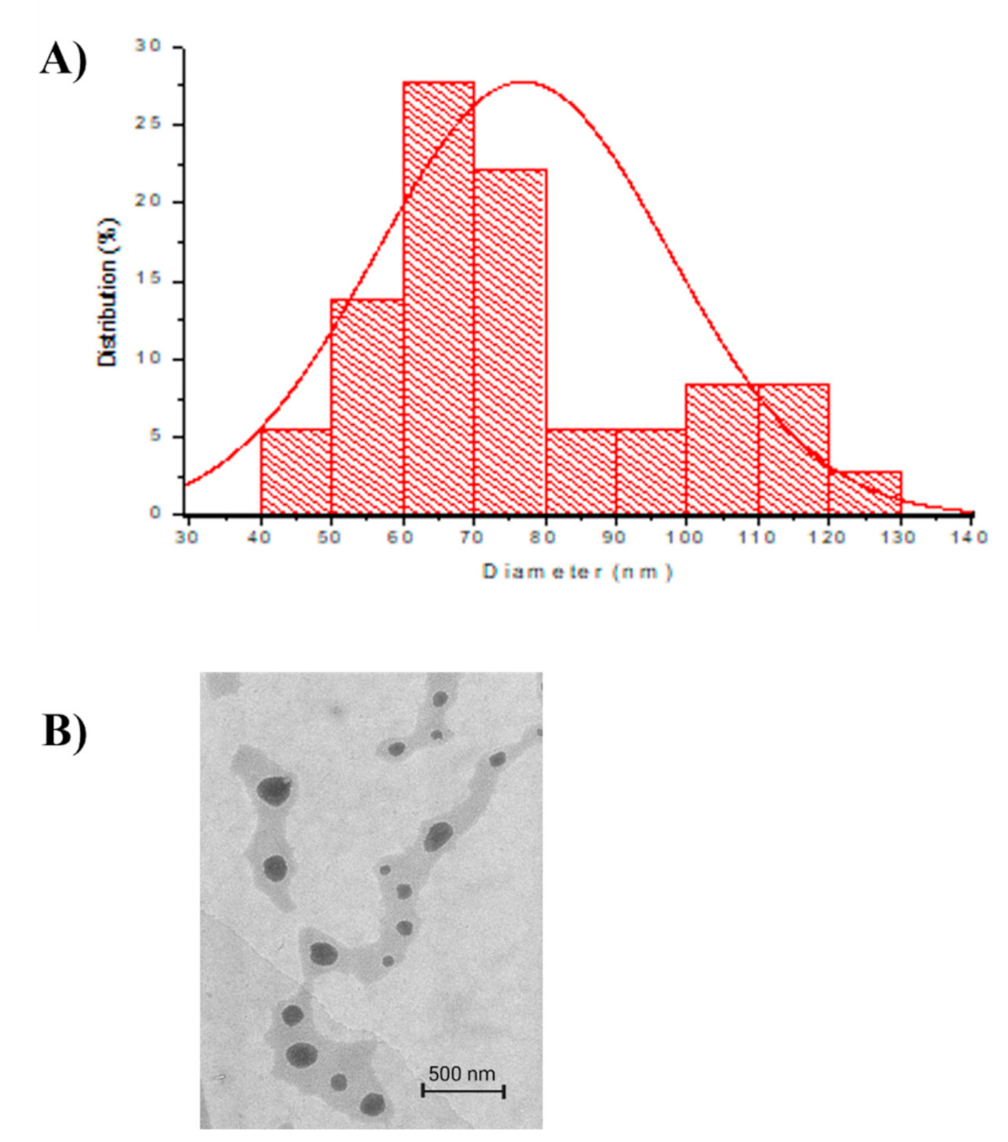
2.1.3. Transmission Electron Microscope (TEM)
2.2. Encapsulation Efficiency (EE) of TQ-PLGA-PF68 Nanoparticles
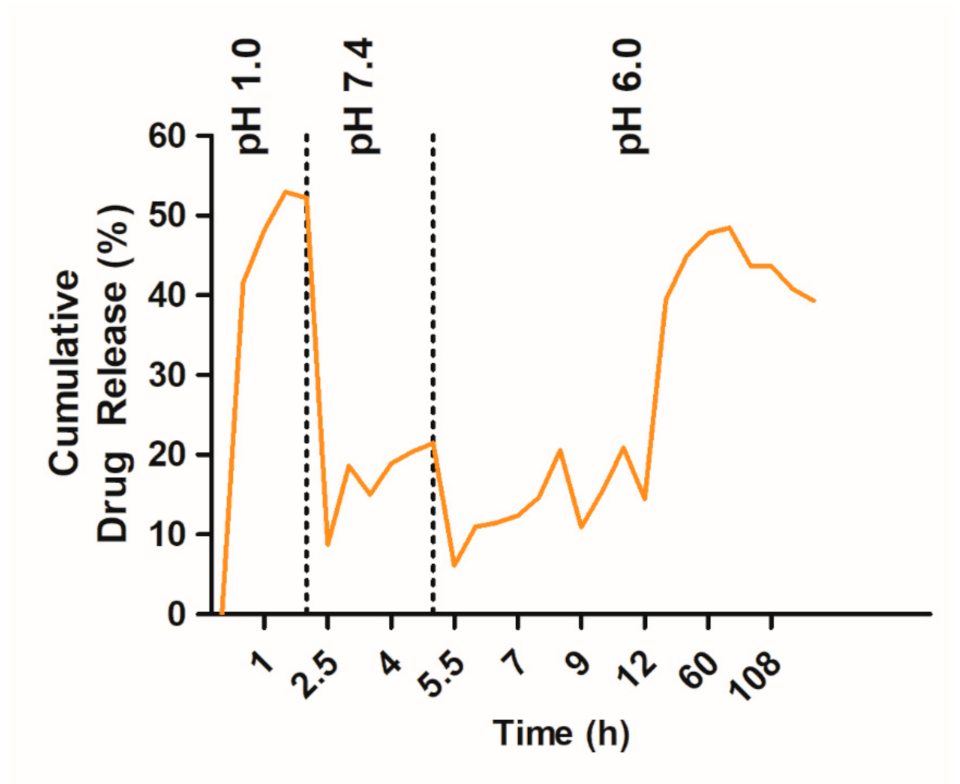
2.3. Effect of pH of the Release Medium on In Vitro TQ Release
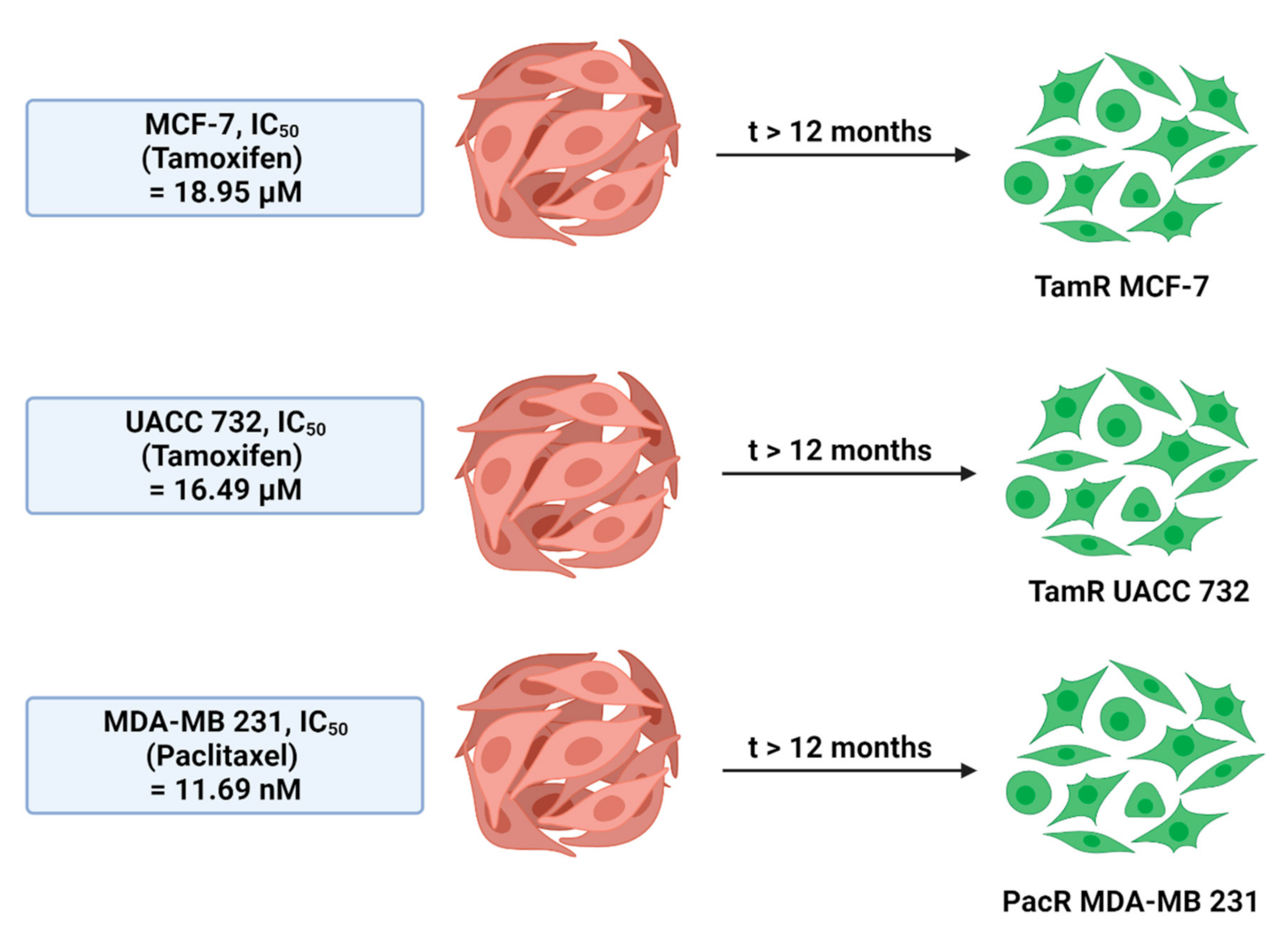
2.4. Titration of Drug Sensitivity on Drug-Resistant Cell Lines
2.5. In Vitro Cytotoxicity Study
3. Discussion
4. Materials and Methods
4.1. Synthesis of TQ-PLGA-PF68 Nanoparticles
4.2. Characterization of TQ-PLGA-PF68 Nanoparticles
4.2.1. FTIR Spectroscopic Analysis
4.2.2. Polydispersity Index (PDI) and Zeta Potential
4.2.3. Transmission Electron Microscope (TEM)
4.3. Encapsulation Efficiency (EE) of TQ-PLGA-PF68 Nanoparticles
4.4. In Vitro Release of TQ from of TQ-PLGA-PF68
4.5. Cell Culture and Culture Conditions
4.6. Cell Viability Assay
4.7. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kamarudin, S.N.; Tripathy, M.; Iezhitsa, I.; Froemming, G.R.A.F.; Alyautdin, R.; Ismail, N.M. Optimization of poloxamer 188 concentration as media for nanoparticle dispersion: Effect of concentration, nanoparticle size and in vitro penetration through blood brain barrier. Int. J. Pharma Bio Sci. 2017, 8, 68–76. [Google Scholar] [CrossRef]
- Anselmo, A.C.; Mitragotri, S. Nanoparticles in the clinic: An update. Bioeng. Transl. Med. 2019, 4, e10143. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hortobagyi, G.N. Treatment of breast cancer. N. Engl. J. Med. 1998, 339, 974–984. [Google Scholar] [CrossRef] [PubMed]
- Vilar, G.; Tulla-Puche, J.; Albericio, F. Polymers and drug delivery systems. Curr. Drug Deliv. 2012, 9, 367–394. [Google Scholar] [CrossRef] [PubMed]
- Chan, J.M.; Valencia, P.M.; Zhang, L.; Langer, R.; Farokhzad, O.C. Polymeric Nanoparticles for Drug Delivery. Methods Mol. Biol. 2010, 624, 163–175. [Google Scholar]
- Manavitehrani, I.; Fathi, A.; Badr, H.; Daly, S.; Shirazi, A.N.; Dehghani, F. Biomedical applications of biodegradable polyesters. Polymers 2016, 8, 20. [Google Scholar] [CrossRef] [Green Version]
- Makadia, H.K.; Siegel, S.J. Poly lactic-co-glycolic acid (PLGA) As biodegradable controlled drug delivery carrier. Polymers 2011, 3, 1377–1397. [Google Scholar] [CrossRef]
- Sharma, N.; Madan, P.; Lin, S. Effect of process and formulation variables on the preparation of parenteral paclitaxel-loaded biodegradable polymeric nanoparticles: A co-surfactant study. Asian J. Pharm. Sci. 2016, 11, 404–416. [Google Scholar] [CrossRef] [Green Version]
- Patel, H.R.; Patel, R.P.; Patel, M.M. Poloxamers: A pharmaceutical excipients with therapeutic behaviors. Int. J. PharmTech. Res. 2009, 1, 299–303. [Google Scholar]
- Norton, K.-A.; Popel, A.; Pandey, N.B. Heterogeneity of chemokine cell-surface receptor expression in triple-negative breast cancer. Am. J. Cancer Res. 2015, 5, 1295–1307. [Google Scholar]
- Batrakova, E.V.; Bronich, T.K.; Vetro, J.A.; Kabanov, A.V. Polymer Micelles as Drug Carriers. Nanopart. Drug Carr. 2006, 57–93. [Google Scholar]
- Trivedi, R.; Kompella, U.B. Nanomicellar formulations for sustained drug delivery: Strategies and underlying principles. Nanomedicine 2010, 5, 485–505. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, F.; Bronich, T.K.; Kabanov, A.V.; Rauh, R.D.; Roovers, J. Synthesis and evaluation of a star amphiphilic block copolymer from poly(epsilon-caprolactone) and poly(ethylene glycol) as a potential drug delivery carrier. Bioconjug. Chem. 2005, 16, 397–405. [Google Scholar] [CrossRef] [PubMed]
- Russo, E.; Villa, C. Poloxamer Hydrogels for Biomedical Applications. Pharmaceutics 2019, 11, 671. [Google Scholar] [CrossRef] [Green Version]
- Oh, K.S.; Song, J.Y.; Cho, S.H.; Lee, B.S.; Kim, S.Y.; Kim, K.; Jeon, H.; Kwon, I.C.; Yuk, S.H. Paclitaxel-loaded Pluronic nanoparticles formed by a temperature-induced phase transition for cancer therapy. J. Control Release 2010, 148, 344–350. [Google Scholar] [CrossRef]
- Mei, L.; Zhang, Y.; Zheng, Y.; Tian, G.; Song, C.; Yangqing, Z.; Chen, H.; Sun, H.; Tian, Y.; Liu, K.; et al. A Novel Docetaxel-Loaded Poly (ε-Caprolactone)/Pluronic F68 Nanoparticle Overcoming Multidrug Resistance for Breast Cancer Treatment. Nanoscale Res. Lett. 2009, 4, 1530–1539. [Google Scholar] [CrossRef] [Green Version]
- Pereverzeva, E.; Treschalin, I.; Treschalin, M.; Arantseva, D.; Ermolenko, Y.; Kumskova, N.; Maksimenko, O.; Balabanyan, V.; Kreuter, J.; Gelperina, S. Toxicological study of doxorubicin-loaded PLGA nanoparticles for the treatment of glioblastoma. Int. J. Pharm. 2019, 554, 161–178. [Google Scholar] [CrossRef]
- Ansary, R.H.; Awang, M.B.; Rahman, M.M. Biodegradable Poly(D,L-lactic-co-glycolic acid)-Based Micro/Nanoparticles for Sustained Release of Protein Drugs—A Review. Trop. J. Pharm. Res. 2014, 13, 1179. [Google Scholar] [CrossRef] [Green Version]
- Chen, Y.-C.; Hsieh, W.-Y.; Lee, W.-F.; Zeng, D.-T. Effects of surface modification of PLGA-PEG-PLGA nanoparticles on loperamide delivery efficiency across the blood–brain barrier. J. Biomater. Appl. 2011, 27, 909–922. [Google Scholar] [CrossRef]
- Khan, A.; Chen, H.-C.; Tania, M.; Zhang, D.-Z. Anticancer Activities of Nigella sativa (Black Cumin). Afr. J. Tradit. Complement. Altern. Med. 2011, 8, 226–232. [Google Scholar] [CrossRef]
- Majdalawieh, A.F.; Fayyad, M.W.; Nasrallah, G. Anti-cancer properties and mechanisms of action of thymoquinone, the major active ingredient ofNigella sativa. Crit. Rev. Food Sci. Nutr. 2017, 57, 3911–3928. [Google Scholar] [CrossRef] [PubMed]
- Ballout, F.; Habli, Z.; Rahal, O.N.; Fatfat, M.; Gali-Muhtasib, H. Thymoquinone-based nanotechnology for cancer therapy: Promises and challenges. Drug Discov. Today 2018, 23, 1089–1098. [Google Scholar] [CrossRef] [PubMed]
- Ganea, G.M.; Fakayode, S.O.; Losso, J.N.; Van Nostrum, C.F.; Sabliov, C.M.; Warner, I.M. Delivery of phytochemical thymoquinone using molecular micelle modified poly(D, L lactide-co-glycolide) (PLGA) nanoparticles. Nanotechnology 2010, 21, 285104. [Google Scholar] [CrossRef] [PubMed]
- Nallamuthu, I.; Parthasarathi, A.; Khanum, F. Thymoquinone-loaded PLGA nanoparticles: Antioxidant and anti-microbial properties. Int. Curr. Pharm. J. 2013, 2, 202–207. [Google Scholar] [CrossRef]
- Gache, S.A.M.; Angelini, A.A.R.; Sabeckis, M.L.; Flecha, F.L.G. Improving the stability of the malachite green method for the determination of phosphate using Pluronic F68. Anal. Biochem. 2020, 597, 113681. [Google Scholar]
- De Assis, D.N.; Mosqueira, V.C.F.; Vilela, J.M.C.; Andrade, M.S.; Cardoso, V.N. Release profiles and morphological characterization by atomic force microscopy and photon correlation spectroscopy of 99mTechnetium-fluconazole nanocapsules. Int. J. Pharm. 2008, 349, 152–160. [Google Scholar] [CrossRef]
- Acharya, S.; Dilnawaz, F.; Sahoo, S.K. Targeted epidermal growth factor receptor nanoparticle bioconjugates for breast cancer therapy. Biomaterials 2009, 30, 5737–5750. [Google Scholar] [CrossRef]
- Guo, C.; Liu, F.; Qi, J.; Ma, J.; Lin, S.; Zhang, C.; Zhang, Q.; Zhang, H.; Lu, R.; Li, X. A Novel Synthetic Dihydroindeno[1,2-b] Indole Derivative (LS-2-3j) Reverses ABCB1- and ABCG2-Mediated Multidrug Resistance in Cancer Cells. Molecules 2018, 23, 3264. [Google Scholar] [CrossRef] [Green Version]
- McDermott, M.; Eustace, A.J.; Busschots, S.; Breen, L.; Crown, J.; Clynes, M.; O’Donovan, N.; Stordal, B. In vitro Development of Chemotherapy and Targeted Therapy Drug-Resistant Cancer Cell Lines: A Practical Guide with Case Studies. Front. Oncol. 2014, 4, 40. [Google Scholar] [CrossRef] [Green Version]
- Harker, W.G.; Slade, D.L.; Dalton, W.S.; Meltzer, P.S.; Trent, J.M. Multidrug resistance in mitoxantrone-selected HL-60 leukemia cells in the absence of P-glycoprotein overexpression. Cancer Res. 1989, 49, 4542–4549. [Google Scholar] [PubMed]
- Gliszczyńska, A.; Niezgoda, N.; Gładkowski, W.; Czarnecka, M.; Świtalska, M.; Wietrzyk, J. Synthesis and Biological Evaluation of Novel Phosphatidylcholine Analogues Containing Monoterpene Acids as Potent Antiproliferative Agents. PLoS ONE 2016, 11, e0157278. [Google Scholar] [CrossRef]
- Agrawal, Y.K.; Patel, V.R. Nanosuspension: An approach to enhance solubility of drugs. J. Adv. Pharm. Technol. Res. 2011, 2, 81–87. [Google Scholar] [CrossRef]
- Lagreca, E.; Onesto, V.; Di Natale, C.; La Manna, S.; Netti, P.A.; Vecchione, R. Recent advances in the formulation of PLGA microparticles for controlled drug delivery. Prog. Biomater. 2020, 9, 153–174. [Google Scholar] [CrossRef]
- Baysal, I.; Ucar, G.; Gultekinoglu, M.; Ulubayram, K.; Yabanoglu-Ciftci, S. Donepezil loaded PLGA-b-PEG nanoparticles: Their ability to induce destabilization of amyloid fibrils and to cross blood brain barrier in vitro. J. Neural Transm. 2017, 124, 33–45. [Google Scholar] [CrossRef]
- Danaei, M.; Dehghankhold, M.; Ataei, S.; Hasanzadeh Davarani, F.; Javanmard, R.; Dokhani, A.; Khorasani, S.; Mozafari, M.R. Impact of Particle Size and Polydispersity Index on the Clinical Applications of Lipidic Nanocarrier Systems. Pharmaceutics 2018, 10, 57. [Google Scholar] [CrossRef] [Green Version]
- Aw-Yong, P.Y.; Sasmita, A.O.; Gan, P.H.; Mak, S.T.; Ling, A.P.K. Nanoparticles as Carriers of Phytochemicals: Recent Applications Against Lung Cancer. Int. J. Res. Biomed. Biotechnol. 2018, 7, 1–11. [Google Scholar]
- Park, J.H.; Oh, N. Endocytosis and exocytosis of nanoparticles in mammalian cells. Int. J. Nanomed. 2014, 1, 51. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Clarke, S.P. Development of Hierarchical Magnetic Nanocomposite Materials for Biomedical Applications.PhD thesis submitted for the Degree of Doctor of Philosophy, Dublin City University, Dublin Ireland, 13. Ph.D. Thesis submitted for the Degree of Doctor of Philosophy, Dublin City University, Dublin, Ireland, January 2013. [Google Scholar]
- Albanese, A.; Tang, P.S.; Chan, W.C.W. The Effect of Nanoparticle Size, Shape, and Surface Chemistry on Biological Systems. Annu. Rev. Biomed. Eng. 2012, 14, 1–16. [Google Scholar] [CrossRef] [Green Version]
- Venkataraman, S.; Hedrick, J.L.; Ong, Z.Y.; Yang, C.; Ee, P.L.R.; Hammond, P.T.; Yang, Y.Y. The effects of polymeric nanostructure shape on drug delivery. Adv. Drug Deliv. Rev. 2011, 63, 1228–1246. [Google Scholar] [CrossRef]
- Leary, J.F. The Importance of Zeta Potential for Drug/Gene Delivery in Nanomedicine Zeta Potential—Electrostatics in Fluids. In In Proceedings of the Malvern Instruments Workshop, Purdue University, West Lafayette, IN, USA, 21 September 2011; Available online: https://nanohub.org/resources/13793/download/2011.09.20-Leary-Malvern.pdf (accessed on 7 September 2018).
- Hoshyar, N.; Gray, S.; Han, H.; Bao, G. The effect of nanoparticle size on in vivo pharmacokinetics and cellular interaction. Nanomedicine 2016, 11, 673–692. [Google Scholar] [CrossRef] [Green Version]
- Gomes, C.; Moreira, R.G.; Castell-Perez, E. Poly (DL-lactide-co-glycolide) (PLGA) Nanoparticles with Entrapped trans-Cinnamaldehyde and Eugenol for Antimicrobial Delivery Applications. J. Food Sci. 2011, 76, N16–N24. [Google Scholar] [CrossRef]
- Simon, L.C.; Stout, R.W.; Sabliov, C. Bioavailability of Orally Delivered Alpha-Tocopherol by Poly (Lactic-Co-Glycolic) Acid (PLGA) Nanoparticles and Chitosan Covered PLGA Nanoparticles in F344 Rats. Nanobiomedicine 2016, 3, 8. [Google Scholar] [CrossRef] [PubMed]
- Mirakabad, F.S.T.; Akbarzadeh, A.; Milani, M.; Zarghami, N.; Taheri-Anganeh, M.; Zeighamian, V.; Badrzadeh, F.; Rahmati-Yamchi, M. A Comparison between the cytotoxic effects of pure curcumin and curcumin-loaded PLGA-PEG nanoparticles on the MCF-7 human breast cancer cell line. Artif. Cells Nanomed. Biotechnol. 2014, 44, 1–8. [Google Scholar] [CrossRef]
- Fang, X.B.; Zhang, J.M.; Xie, X.; Liu, D.; He, C.W.; Wan, J.B.; Chen, M.W. PH-sensitive micelles based on acid-labile pluronic F68-curcumin conjugates for improved tumor intracellular drug delivery. Int. J. Pharm. 2016, 502, 28–37. [Google Scholar] [CrossRef]
- Arafa, E.-S.A.; Zhu, Q.; Shah, Z.I.; Wani, G.; Barakat, B.M.; Racoma, I.; El-Mahdy, M.A.; Wani, A.A. Thymoquinone up-regulates PTEN expression and induces apoptosis in doxorubicin-resistant human breast cancer cells. Mutat. Res. Mol. Mech. Mutagen. 2011, 706, 28–35. [Google Scholar] [CrossRef] [Green Version]
- Ahmad, A.; Mishra, R.K.; Vyawahare, A.; Kumar, A.; Rehman, M.U.; Qamar, W.; Khan, R. Thymoquinone (2-Isoprpyl-5-methyl-1, 4-benzoquinone) as a chemopreventive / anticancer agent: Chemistry and Biological effects. Saudi Pharm. J. 2019, 27, 1113–1126. [Google Scholar] [CrossRef]
- Ahmad, R.; Kaus, N.H.M.; Hamid, S. Synthesis and Characterization of PLGA-PEG Thymoquinone Nanoparticles and Its Cytotoxicity Effects in Tamoxifen-Resistant Breast Cancer Cells. Stud. Biomark. New Targets Aging Res. Iran 2018, 1292, 65–82. [Google Scholar]
- Grever, M.R.; Schepartz, S.A.; Chabner, B.A. The National Cancer Institute: Cancer drug discovery and development program. Semin. Oncol. 1992, 19, 622–638. [Google Scholar]
- Al Khateb, K.; Ozhmukhametova, E.K.; Mussin, M.N.; Seilkhanov, S.K.; Rakhypbekov, T.K.; Lau, W.M.; Khutoryanskiy, V.V. In situ gelling systems based on Pluronic F127/Pluronic F68 formulations for ocular drug delivery. Int. J. Pharm. 2016, 502, 70–79. [Google Scholar] [CrossRef]
- Almeida, H.; Lobao, P.; Frigerio, C.; Fonseca, J.; Silva, R.; Lobo, J.M.S.; Amaral, M.H. Preparation, characterization and biocompatibility studies of thermoresponsive eyedrops based on the combination of nanostructured lipid carriers (NLC) and the polymer Pluronic F-127 for controlled delivery of ibuprofen. Pharm. Dev. Technol. 2015, 22, 336–349. [Google Scholar] [CrossRef]
- Schmolka, I.R. Physical Basis for Poloxamer Interactions. Ann. N. Y. Acad. Sci. 1994, 720, 92–97. [Google Scholar] [CrossRef]
- Park, N.; Kang, D.; Ahn, M.C.; Kang, S.; Lee, S.M.; Ahn, T.K.; Jaung, J.Y.; Shin, H.; Son, S.U. Electronic Supplementary Information for Hollow and Sulfonated Microporous Organic Polymers: Versatile Platforms for Non-Covalent Fixation of Molecular Photocatalysts. RSC Adv. 2015, 5, 47270–47274. [Google Scholar] [CrossRef]
- Clogston, J.D.; Patri, A.K. Zeta Potential Measurement. In Characterization of Nanoparticles Intended for Drug Delivery; McNeil, S.E., Ed.; Humana Press: Totowa, NJ, USA, 2011; pp. 63–70. [Google Scholar]
- Li, C.; Li, X.; Li, S.; Weng, Y.; Wang, K.; Zhang, T.; Chen, S.; Lu, X.; Jiang, Y.; Liang, J.X.A.X. Development and Validation of a Method for Determination of Encapsulation Efficiency of CPT-11/DSPE-mPEG2000 Nanoparticles. Med. Chem. 2016, 6, 345–348. [Google Scholar] [CrossRef] [Green Version]
- Coley, H.M. Development of drug-resistant models. Methods Mol. Med. 2004, 88, 267–273. [Google Scholar] [PubMed]
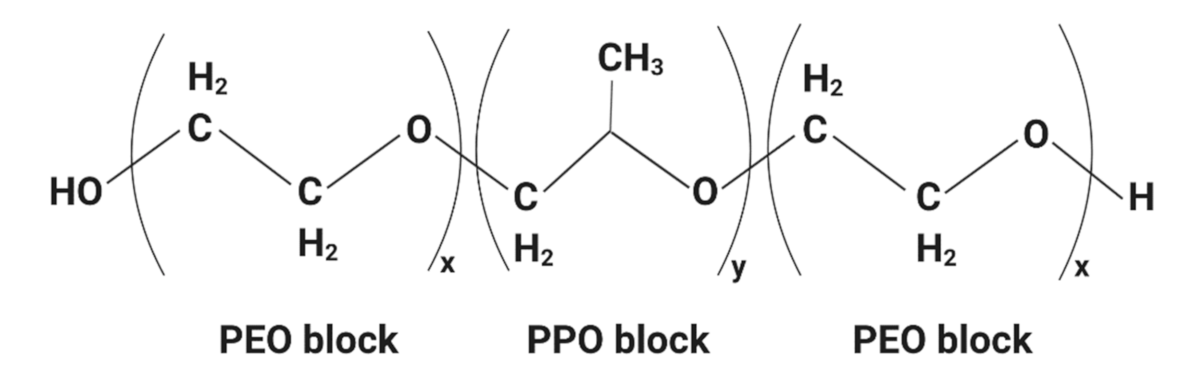

| Functional Group | Type of Compounds | |||
|---|---|---|---|---|
| TQ | Pluronics F68 | PLGA-PEG | TQ-PLGA-PF68 | |
| sp2 CH | 3257 (w) | - | - | - |
| sp3 CH | 2967 (m) | 2882 (m) | - | 2882 (m) |
| N-H | - | - | 2954 (w) | 3345 (m) |
| C=C | 1645 (s) | - | - | - |
| C=O | - | - | 1748 (s) | 1751 (m) |
| C=CH | 3039 (w) | - | - | - |
| C-O-C | - | 1100 (s); 1242 (m) | - | 1103 (s) |
| C-O | - | - | 1085 (s); 1127(s); 1164 (s) | - |
| Resistant Cell Line | Drug | RI | * p-Value | RI Classification |
|---|---|---|---|---|
| TamR MCF-7 | Tamoxifen | 2.32 | <0.05 | Moderately drug- resistant |
| PacR MDA-MB- 231 | Paclitaxel | 1.59 | <0.05 | Drug-resistant |
| TamR UACC 732 | Tamoxifen | 1.32 | <0.05 | Drug-sensitive |
| A Subtype of Breast Cancer Cells | IC50 Value (Mean ± SE) | ||
|---|---|---|---|
| Parental Cell Line | Resistant Cell Line | * p-Value | |
| MCF-7 | 90.46 ± 22.63 | 96.34 ± 1.70 | > 0.05 |
| UACC 732 | 118.5 ± 23.49 | 14.81 ± 3.2 | <0.05 |
| MDA-MB 231 | 34.28 ± 11.81 | 28.89 ± 5.12 | <0.05 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Noor, N.S.; Kaus, N.H.M.; Szewczuk, M.R.; Hamid, S.B.S. Formulation, Characterization and Cytotoxicity Effects of Novel Thymoquinone-PLGA-PF68 Nanoparticles. Int. J. Mol. Sci. 2021, 22, 9420. https://doi.org/10.3390/ijms22179420
Noor NS, Kaus NHM, Szewczuk MR, Hamid SBS. Formulation, Characterization and Cytotoxicity Effects of Novel Thymoquinone-PLGA-PF68 Nanoparticles. International Journal of Molecular Sciences. 2021; 22(17):9420. https://doi.org/10.3390/ijms22179420
Chicago/Turabian StyleNoor, Nurul Shahfiza, Noor Haida Mohd Kaus, Myron R. Szewczuk, and Shahrul Bariyah Sahul Hamid. 2021. "Formulation, Characterization and Cytotoxicity Effects of Novel Thymoquinone-PLGA-PF68 Nanoparticles" International Journal of Molecular Sciences 22, no. 17: 9420. https://doi.org/10.3390/ijms22179420
APA StyleNoor, N. S., Kaus, N. H. M., Szewczuk, M. R., & Hamid, S. B. S. (2021). Formulation, Characterization and Cytotoxicity Effects of Novel Thymoquinone-PLGA-PF68 Nanoparticles. International Journal of Molecular Sciences, 22(17), 9420. https://doi.org/10.3390/ijms22179420








