Abstract
Tissues and organs are not composed of solely cellular components; instead, they converge with an extracellular matrix (ECM). The composition and function of the ECM differ depending on tissue types. The ECM provides a microenvironment that is essential for cellular functionality and regulation. However, during aging, the ECM undergoes significant changes along with the cellular components. The ECM constituents are over- or down-expressed, degraded, and deformed in senescence cells. ECM aging contributes to tissue dysfunction and failure of stem cell maintenance. Aging is the primary risk factor for prevalent diseases, and ECM aging is directly or indirectly correlated to it. Hence, rejuvenation strategies are necessitated to treat various age-associated symptoms. Recent rejuvenation strategies focus on the ECM as the basic biomaterial for regenerative therapies, such as tissue engineering. Modified and decellularized ECMs can be used to substitute aged ECMs and cell niches for culturing engineered tissues. Various tissue engineering approaches, including three-dimensional bioprinting, enable cell delivery and the fabrication of transplantable engineered tissues by employing ECM-based biomaterials.
1. Introduction
The extracellular matrix (ECM) is an essential non-cellular component of the body that is present in all tissues and organs [1]. The ECM organizes large fibrillar three-dimensional (3D) networks composed of glycosaminoglycans (GAGs) and various types of proteins such as collagen, fibronectin, elastin, and laminin. Among the numerous constituents of the ECM, fibrous proteins (i.e., collagen and elastin), adhesive glycoproteins (fibronectin and laminin), and glycosaminoglycans (i.e., hyaluronic acid) are recognized as the main components [2]. Although the compositions differ based on the type of tissue, type I and II collagen are the primary constituents and are associated with other ECM constituents [3]. The ECM offers biophysical, biochemical, and biomechanical cues for cellular components. Furthermore, it provides structural support, functions as an adhesive substrate, presents growth factors to its receptors, sequesters, stores growth factors, senses, and transduces mechanical signals [4,5]. Furthermore, the ECM is involved in regulating several cellular functions, such as survival, adhesion, migration, proliferation, differentiation, and supporting cells for binding [4,6].
The ECM undergoes changes in the aging cell. The ECM is degraded and deformed, whereas the expression of ECM components is up or downregulated [7]. As the ECM provides a microenvironment for cells, alterations in the ECM may affect cellular functionalities. Abnormal changes in the ECM contribute to alterations in pathological conditions, and stem or progenitor cells fail to proceed with normal regeneration [8]. Hence, regeneration strategies must be applied to overcome age-associated dysfunctions and disorders. Currently, ECM-based biomaterials are prominently utilized in regenerative therapies owing to their high biocompatibility and biomimetic properties. ECM-based biomaterials can be employed in relatively simple applications (i.e., injectable fillers) to complicate biofabrication methods (i.e., bioink). Owing to the injectability and biomimetic characteristics of ECM-based materials, more detailed biomimicking engineering is now feasible. Among the latest biofabrication technologies, 3D bioprinting technology that adopts biomaterials as bioinks has been highlighted [9].
Herein, we describe the aging and rejuvenation within the framework of ECM. The functions of the ECM components, changes in the ECM with aging, age-related disorders, and rejuvenating strategies are introduced in the following sections. The ECM is mentioned in every section of this article to emphasize its importance in both understanding the aging process and developing regeneration strategies. We discuss current rejuvenating approaches using ECM-based biomaterials and biofabrication methods used in the past five years to facilitate the further development of ECM-based materials such that issues in the field of tissue engineering can be solved.
2. Changes in ECM Components with Aging
Tissues and organs undergo physical and physiological changes during aging, and the aging process varies depending on the tissue and its components. Not only cellular components but also non-cellular components such as ECM [8,10] undergo aging (Table 1). The ECM undergoes constant regulation through deposition, modification, and degradation, primarily via matrix metalloproteinases (MMPs) in normal tissues. However, at the senescence stage, changes occur in both the ECM components and enzymes. The roles and alterations of some abundant and important ECM components, including collagen, are partially known; however, not every ECM component has been investigated in terms of their specific functions and aging process [7].

Table 1.
Features of senescence-related extracellular matrix (ECM) components.
2.1. Collagen
Collagen is one of the most abundant proteins in the human body. More than 28 different types of collagen exist, and the ratio varies for different tissues. Collagen primarily provides structural properties and resilience to tissues. Typically, collagen constitutes a significant portion of the ECM, and in the dermis, collagen is a major component, constituting up to 75% of the total composition [11]. However, aging cells fail to regulate collagen synthesis, resulting in changes in the amount and ratio of collagen. Aging modulates the expression of collagen differently, and several types of collagen in various tissues are down- or upregulated depending on the circumstances [12,13,14]. In some cases, collagen is overexpressed with aging, inducing fibrosis [15]. It has been reported the expression of collagen IV increased in age-related neural disorders [16]. Meanwhile, the downregulation of collagen due to degradation has been observed in the senescence path [17]. Additionally, collagens undergo modifications (i.e., mineralization and accumulation of advanced glycation end-products), and these modifications affect the susceptibility of collagens to MMP-mediated degradation [18].
2.2. Fibronectin
Fibronectin is an abundant glycoprotein that is essential for ECM assembly. Fibronectin can bind various ECM molecules such as collagen I and III, gelatin, thrombospondin, and heparin [19]. Fibronectin typically functions through interactions with the ECM network. Fibronectin mediates cellular activities by binding to integrin or syndecan receptors on the cell surface [20]. The gene expression of fibronectin is downregulated with collagen in senescent hepatic stellate cells [21]. Meanwhile, fibronectin expression increases in aortic endothelial cells in vivo with the age of donors [22]. Furthermore, the expression of fibronectin increases in patients with Alzheimer’s disease [16]. Increased fibronectin mRNA accumulation has been observed in in vitro cellular senescence [23]. Both the downregulation and upregulation of fibronectin were indicated in senescent cells.
2.3. Elastin
Elastin is a fibrous self-assembling protein that contributes significantly to the mechanical strength, elasticity, firmness, and suppleness of tissues. Elastin facilitates the appropriate functioning of organs and is present in almost all connective tissues [24]. The primary age-dependent change in elastin is degradation. The expression levels of elastin-degrading enzymes (i.e., cathepsins and elastase) increase with aging [25,26]. Age-related destruction of the elastin fiber structure has been investigated in the skin and artery [25,27]. It was reported that aging affects the structural and mechanical properties of elastic fibers, resulting in the fragmentation and structural reorganization of elastin [27,28].
2.4. Laminin
Laminin is a large heterotrimeric glycoprotein that is integral to the basement membrane. Laminin is a major component of the basal lamina; it is one of the layers of the basement membrane and contributes significantly to the lamina’s structure. Laminins aid mediating processes such as cell differentiation, proliferation, adhesion, migration, and tissue maintenance based on receptor- and matrix-binding properties [29]. Some laminins have been reported to be upregulated in some aging tissues, particularly in diabetic basement membranes [30]. A recent study demonstrated that decreased laminin production is an early indicator of an aging lung [31]. Another recent study reported that the loss of laminin-α4 results in non-progressive impaired neurotransmissions and premature morphological alterations typically associated with an aging neuromuscular junction [32].
2.5. Hyaluronic Acid
Hyaluronic acid (HA) is a substantial GAG component that comprises glucuronic acid and hyaluronan. HA exhibits the unique characteristic of water binding, thereby allowing it to preserve a large amount of moisture, which is important for tissue hydration [33,34]. HA contributes significantly to the structure and organization of the ECM. However, during aging, the elasticity, turgidity, and mechanical strength of HA deteriorate [35]. Age-induced deterioration, which is relevant to symptoms of aged skin such as wrinkles, has been observed in the skin [34]. The decline in both the total mass and polymer size was investigated in aging skin [36]. Furthermore, the water-binding capacity decreased as senescence progressed [37]. The level of HA decreased in the lamina propria with age [38].
3. Influence of Age-Related Changes of ECM on Tissues
Changes in the ECM during aging not only result in cellular senescence but also affect tissue dysfunction. Aging is characterized by reduced tissue function and regenerative capacity [39]. During cellular senescence, the ECM and ECM-related components undergo abnormal regulation and remodeling. The mechanisms underlying these abnormalities and cell behavior have not yet been elucidated at the molecular level; however, it is certain that such defect changes in the ECM dynamics affect cell and tissue functions [40]. Age-related changes in the ECM mediate cell proliferation, differentiation, inflammation, and apoptosis. Furthermore, it affects the recruitment, differentiation, and functional integration of stem- and tissue-specific progenitor cells [8]. As the strict regulation of ECM synthesis and remodeling is directly associated with healthy tissue conditions, alterations in ECM remodeling can affect the course and progression of several other pathological conditions, including fibrosis, disorders, and cancers [2]. A recent study revealed that the ECM compositions of young, adult, and aged tissues are different and that the ECM age significantly affects the proliferative ability, maturity, and stress response of cells. Aging cues, which accelerate the aging process, are presented from aged ECM on an in vitro test platform [41]. The major alterations in tissues related to the ECM during aging, which are described in detail in the subsections below, are mass loss, moisture loss, fibrosis, and dysfunction.
3.1. Mass Loss
The regenerative function of most tissues declines with age, and mass loss occurs in various tissues. Although the correlation between ECM alteration and tissue loss is yet to be elucidated, it is certain that the ECM comprises tissues and may likely contribute to deviations. As the ECM promotes cell proliferation and adhesion, structural and biochemical changes in the ECM may affect cell maintenance [42]. The number of proliferating cells and the number of newly synthesized ECMs typically decrease during aging, resulting in mass loss. In particular, certain muscle tissues indicate a considerable decline associated with aging, resulting in interference with physical activities. Additionally, brain tissues decline gradually with aging, and this tendency is consistent with the decline in cognitive performance among the elderly [43]. ECM alterations induce neural cell death, and the decline of neurons may result in ECM degradation caused by the proteolytic activity of MMPs [44]. Dermal collagen fibrils undergo gradual loss and fragmentation. Alterations and reduced production of collagen at the dermal site impair the structural integrity and induce thin skin, rendering the skin more vulnerable with increased age [45,46].
3.2. Moisture Loss
Connective tissues, such as ligaments and tendons, are innately stiffened with aging. This is the result of age-related water content loss and ECM turnover. During aging, the mechanical properties and solubility of collagen diminish, thereby increasing mechanical stiffness; consequently, joint motion decreases and pain increases [47]. Proteoglycans, one of the major components of the ECM, maintain the hydration of tissues and the viscoelasticity of the disk to support mechanical loads. Nucleus pulposus, the core of the disk, receives water molecules through the effect of chondroitin sulfate proteoglycans. However, as the disk ages, the composition of the ECM shifts from proteoglycan-rich to fibrotic collagen-rich. Consequently, the water-binding ability degenerates, and the disc becomes stiffer [48]. Nevertheless, distinct from water content matter, the ECM constitutes 90% of the dry weight of the cartilage; hence, pathological changes in the ECM are regarded as key for understanding cartilage disorders [49]. Additionally, skin aging is associated with moisture loss. HA, which is abundant in the skin, loses its capacity to retain water with aging; eventually, the skin becomes dry and lusterless [37].
3.3. Fibrosis
Fibrosis is the excess of fibrous tissues, and sclerosis is the stiffening of tissues. Both pathological states are typically caused by the deposition of connective tissues with abundant collagen and glycosaminoglycans. They are indicators of aging and are related to ECM alterations. Aging promotes the structural remodeling of the ECM, resulting in fibrosis and tissue function deterioration. Fibrosis is primarily caused by increased concentrations of collagen fibers, which results in the development of fibrotic conditions. Collagen deposition in the heart contributes to cardiac fibrosis, which eventually decreases cardiac function [50]. Additionally, vascular stiffening is correlated with fibrosis. Arterial stiffening is primarily caused by the alteration of ECM components during aging [51]. As we age, elastin fibers become weaker, and stiff collagen fibers are burdensome. Unlike elastin, the concentration of collagen increases with age; this causes fibrosis, which increases arterial stiffness significantly and induces cardiovascular diseases [52]. Hepatic fibrosis is recognized as ECM dyshomeostasis, which is caused by ECM aging. Fibrillar ECMs such as collagens accumulate in the liver and can progress to cirrhosis or worse [53]. Idiopathic pulmonary fibrosis, an age-related lung disorder, occurs primarily in the elderly population. Lung fibrosis occurring in the elderly is characterized by increased type I and III collagen, elastin, and fibronectin [54].
3.4. Tissue Dysfunctions
Many disorders and tissue dysfunctions are assumed to be associated with aging. They occur because of epigenetic changes, protein deformation, and many other factors. Apart from mass loss and fibrosis, several disorders, including diabetes and Alzheimer’s disease, are associated with aging [55]. A recent study showed that elastin in the brain might induce pathological changes in Alzheimer’s disease, and elastin is known to be fragmented and released with aging [56]. Significant ECM changes, including increased expression of collagen IV and fibronectin, are presumed to affect disease progression in patients with Alzheimer’s disease [16]. The glycemic condition caused by diabetes and aging can affect the ECM constituents and result in complications [57]. Although not many studies of age-related diseases investigated the direct relationship with ECM aging, some studies have reported that ECM aging and age-related tissue dysfunctions are indirectly linked. The pathogenesis of diabetic kidney disease is characterized by increased ECM accumulation, which causes glomerular thickening and tubulointerstitial fibrosis. Furthermore, the increased levels of MMPs are correlated with the development and progression of diabetic nephropathy [58]. The glycation of proteins that leads to the formation of advanced glycation end-products (AGEs) is also known to be mainly related to aging [59]. The modification of proteins (e.g., collagen and fibronectin) by AGEs may contribute to an alteration in their structure and function, indicating the consequent progression of several diseases, such as diabetic nephropathy, retinopathy, atherosclerosis, arthritis, cardiovascular diseases, and neurodegenerative diseases [60].
4. Utilization of ECM for Regeneration Strategies
The senescence of the ECM affects tissue dysfunction and tissue regeneration failure. To rejuvenate aged tissues and restore their functions, regenerative strategies are indispensable. Tissue engineering, which focuses on restoring, maintaining, and improving the damaged tissues, is a possible treatment. Tissue engineering is an interdisciplinary field in which cells and matrices are applied. The cell source can be varied from primary lineages to stem cells (SCs), and synthetic polymers or the ECM can be employed as a matrix [61]. Synthetic polymers such as polycaprolactone (PCL), polylactic acid (PLA), and polyvinyl alcohol (PVA) have been typically used in early studies. However, owing to the hydrophobicity and non-natural residues of most synthetic polymers, naturally occurring biomaterials such as ECM-based materials and some hybrid biomaterials have been extensively investigated for biomedical applications [62]. Furthermore, numerous recent 3D tissue engineering strategies have been actively researched to treat age-related tissue dysfunctions by employing ECM-based materials [63,64,65]. In this section, the types of ECM-based materials and their applications as tissue engineering strategies are introduced for the regeneration of age-dependent tissue dysfunctions.
4.1. Types of ECM-Based Materials for Biomedical Applications
As aged ECMs adversely affect cellular regulations and appear in fewer numbers, replacing them with ECM-based materials is an option. ECM is a typical biomaterial that can be used for in situ tissue regeneration. This biomaterial can interact with the microenvironment and induce in situ tissue regeneration [66]. Additionally, ECM-based materials are used as an injectable soft tissue filler for filling up the volume lost from ECM degradation during aging. Many commercial filler products based on ECM components can be used in clinical applications [67]. At the dermal site, in particular, the number of ECM components decreases on the outside. Aging features such as wrinkles and loss of skin elasticity can be rejuvenated through antiaging approaches, including dermal filling [68]. Soft tissue fillers based on ECM constituents have been reported to affect tissue regeneration [69,70]. ECM-based materials can be processed from native tissues to products through isolation, decellularization, and purification (Figure 1) [71]. Native collagen, fibronectin, elastin, and the like are commercially available. They are typically isolated from specific ECM-enriched tissues, such as collagen from the dermis and fibronectin from the plasma. Purified ECM components are used for various applications, from biomedical to cosmetics, and have demonstrated potential for therapeutic use [72,73,74,75,76].
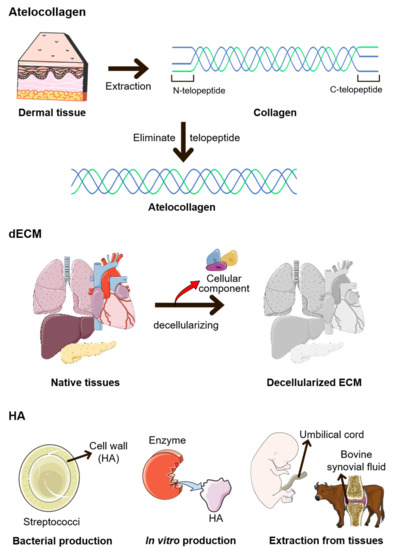
Figure 1.
Methods for manufacturing modified ECMs. Atelocollagen is manufactured by removing telopeptides from collagen. Decellularized ECM (dECM) is manufactured by eliminating cellular components from native tissues. Finally, hyaluronic acid (HA) is produced using several methods. Bacteria such as Streptococci are representative bacterium that produce HA. Additionally, HA can be manufactured using enzymes, the umbilical cord, or bovine synovial fluid [83].
Collagen is frequently used as a basic biomaterial because it is present in high amounts in the ECM. In some cases, the ECM is modified to enhance its unique characteristics or to eliminate its harmful effects, and atelocollagen is one of the most popular modified ECMs [76]. By removing telopeptide from collagen to form atelocollagen, the chance of an immune reaction is reduced. Protocols to produce atelocollagen with high purity at a faster speed have been developed in some studies [77]. In other studies, atelocollagen was used to treat tendons and periodontal tissues [78,79]. Additionally, some examples of improvement in collagen functions have been reported, including the enhancement in the ability of collagen to promote cell proliferation and anti-thrombogenic activity via conjugation with chondroitin sulfate. Although collagen is known to induce thrombosis, chondroitin-sulfate-modified collagen may weaken the induction [80]. Apart from modification, a novel scaffold can be achieved by obtaining an anisotropic structure or inducing the orientation of collagen fibers. The aligned structure of collagen provides insights into neural and vascular endothelial cells [81,82].
HA is an injectable hydrogel that is vital to hydration and the space-filling capacity [37]. However, HA exhibits relatively weak mechanical properties, and it dissolves and degrades rapidly. Hence, HA has been modified using numerous methods for biomedical applications, such as controlling the molecular size or viscoelasticity [37,84]. Recently, novel methods for modifying HA-based hydrogels to achieve enhanced stiffness and control degradability while sustaining injectability have been reported. The combination of dynamic covalent crosslinking with thermoresponsive engineered proteins enhanced mechanical stiffness, and the crosslinking of monoaldehyde-modified HA with carbohydrazide-modified gelatin decelerated the degradation [85,86]. Skin rejuvenation and joint treatment are representative applications of HA that involve the injectability of HA [87,88]. HA is introduced in joints via either an injectable hydrogel or solid scaffold form to relieve inflammation and repair the cartilage [89]. Enhancing the water-binding capability of HA is another example of a modification that may aid tissue hydration. A modified interpenetrating polymer network that entraps HA and water, thereby aiding moisturization, has been reported [90].
To apply a multicomponent matrix for the microenvironment, the decellularized ECM (dECM) is typically employed for biomedical applications. The dECM is created by eliminating cellular components from the native tissue. Tissue-specific ECMs are important for the development and functioning of normal tissues, and the dECM is a representative example [91]. Through decellularization, only bioactive ECM components that do not harm the immune system remain and have a similar ratio of ECM components as the native tissues [92]. A decellularizing protocol was developed to maintain the delicate structure and composition of the native tissue. Hence, the dECM was compared to the native tissue using proteomics and protein staining imaging [93]. Furthermore, the dECM can be classified based on the source of the original tissues. Using only specific tissues as the source of the dECM, tissue-specific compositions can be provided [91,94,95]. The dECM-based hydrogel can be applied as a bioink for 3D bioprinting, injectable hydrogels, cell delivery, and translational medicine [96]. However, dECM lacks mechanical strength and stability. Post-decellularization processes (e.g., hybridization of dECM with a crosslinking agent) have been developed to enhance the mechanical properties of dECM [97]. A recent study introduced the light-activated dECM, which can be polymerized via a dityrosine-based crosslinking system, to improve the inferior mechanical properties of the dECM [98]. In the case of cartilage tissue, resistance to compressive forces is required. Synthetic polymers are often hybridized with dECM using crosslinking agents to satisfy the physical properties of cartilage tissues. The hybridization of dECM with cell-encapsulated microparticles or platelet-rich plasma (PRP) is regarded as a post-decellularization process [97].
4.2. ECM-Based Materials as Cell Delivery Carriers
Because the surrounding of an aged ECM is damaged, replacing only the ECM might not be sufficient to regenerate the aged tissues. Hence, cell delivery may be required for successful tissue regeneration. However, there are limitations to cell delivery; thus, tissue engineering techniques are being studied to supplement the insufficient capacity of past cell delivery systems [99]. Tissue engineering is a complex process that involves recellularizing the ECM matrix. Recellularization requires a specific environment that provides biochemical and biomechanical conditions similar to those of a specific organ [97]. Similarly, niches that provide a microenvironment for the delivered cells are essential to design effective cell carriers [64]. For instance, type V collagen has been shown to induce the formation of human islet organoids and the generation of endocrine cells by profiling extracellular proteins in the pancreatic ECM. Type V collagen is an indispensable factor in the recapitulation of niches for pancreatic cells [100]. Essential factors regulating cell functions, cell survival, and cell retention must be considered when deciding the materials for cell delivery [101]. Many studies have revealed that ECM-based biomaterials showed improved results in cell delivery and successfully regenerated defective tissues. Therapeutic cell delivery restored the functions of organs such as the heart, nerve, joint, thyroid, and the like using cells and ECM-based biomaterials [102,103,104,105]. The cell-delivering material replenishes the volume first; subsequently, the delivered cells secrete matrix components, thereby restoring the regeneration capacity [106]. Generally, progenitor cells, such as embryonic SCs, induced pluripotent SCs, and mesenchymal stem cells (MSCs), are combined with ECM-based biomaterials for cell therapeutics [107]. MSCs showed improved potency when expanded on the dECM [108]. Moreover, ECM-based hydrogels may enable human organoids to be delivered in vivo for clinical purposes [109]. Although current clinical trials based on SCs are still at an early stage, several studies have revealed that SC-based approaches are potential treatments for degenerative diseases, such as Parkinson’s disease and osteoarthritis [110,111].
Cardiovascular diseases are the leading cause of death and are of high risk in the elderly. To prevent fatal diseases, cell delivery for the cardiac disease has been actively investigated to restore tissue function and cardiac regeneration [112]. The application of ECM-based biomaterials has resulted in successful cardio cell delivery [113]. A recent study validated that the combination of dual SCs and ECM-enhanced cell retention, engraftment, and maturation. The result of cell delivery showed significantly higher gene expression, indicating better integration with the host myocardium [114]. MSCs were delivered in patch form to improve cell adhesion and effectively enhance cardiac remodeling [115]. The development of a heart decellularization method for retaining ECM-derived biochemical cues and promoting angiogenesis has been attempted [116].
Understanding neural-specific ECMs is crucial for the treatment of neurodegenerative diseases because the neural extracellular matrix is known to differ from the normal ECM in other organs. The neural ECM has a low fibrous protein content and high glycoprotein and carbohydrate contents. One of the key carbohydrate components in the brain ECM is chondroitin sulfate proteoglycans. It is found that chondroitin sulfate GAG matrices promote neural stem cell maintenance and efficacy [117]. Proteomic analysis showed sufficient chondroitin sulfate proteoglycans in decellularized nerve tissues, indicating its suitability for cell therapy applications. Moreover, a recent study established the genipin and EDC crosslinking of dECM to enhance stability [118]. Therefore, some studies have optimized the neural microenvironment using the dECM. dECM made from brain tissue accelerated the neural network formation in vitro [119]. The use of laminin enriched ECM hydrogel enhanced the retention rate of the delivered cells. Because of the importance of laminin in the neural ECM, laminin-derived peptides have been applied to promote neural cell adhesion in neural progenitor cell delivery research [120].
Osteoarthritis is another age-related joint disease in which cell delivery therapy has progressed. The cartilage is composed of specialized cells known as chondrocytes, which produce a large number of collagenous extracellular matrices, and osteoarthritis is a degenerative cartilage disorder [121]. Adipose-derived SCs and MSCs delivered with the ECM were investigated to secrete the newly synthesized ECM, which can restore the ECM-producing function of cartilage [104,122]. The differentiation of MSCs was promoted by dECM, and the ECM derived from human umbilical vein endothelial cells (HUVECs): MSC co-culture improved the osteogenic and angiogenic potential [123]. For further applications, ECM-polymer hybrid or cross-linkable injectable hydrogels are often used as functional biomaterials because the cartilage must endure high pressures [124]. PVA is typically used in combination with ECM materials to achieve both favorable mechanical properties and biocompatibility [125]. Recently, PLGA (poly(lactic-co-glycolic acid)) has also been used with collagen to prepare synergetic hybrid biomaterials [126]. Moreover, several MSC-based clinical trials have been reported to have an effect on cartilage renewal, and trials are ongoing to improve long-term osteoarthritis [111].
4.3. 3D Bioprinting Using ECM-Based Bioinks
Aged ECMs around the delivery site may cause delivered cells to fail initially. To overcome the limitation of cell delivery by mimicking the original tissues, transplantable engineered tissues are being developed to produce substitutes [99]. Tissue development is not achieved via cell culture alone. In fact, the surrounding environment is important, and 3D cell deposition is necessitated. Tissue engineering technology enables the fabrication of various types of engineered organs such as the pancreas, liver, kidney, ovary, bladder, cornea, muscle, skin, vessel, esophagus, and the like using SCs and biomaterials (Figure 2) [127,128,129,130,131,132,133,134,135,136,137]. Although numerous hurdles (i.e., low cell survival, immune response, and insufficient functions) remain to be overcome, many studies have revealed the effectiveness of engineered tissue transplantation as a novel regeneration method [138,139]. Some simple tissues (i.e., artificial epidermis) that are relatively easier to engineer, as well as some vital organs that are in high demand for transplantation, have been developed and are progressing favorably [66,127]. Furthermore, to achieve highly precise engineered tissues, novel biofabrication methods such as 3D bioprinting are being developed actively [140,141,142]. Conventional tissue engineering approaches, such as scaffolding and molding, are limited in their capacity to produce precise tissue constructs. Unlike the former fabrication methods, 3D bioprinting is a rapid and efficient method for constructing elaborate engineered tissues [143]. Three-dimensional bioprinting is a type of additive manufacturing system that allows controlled deposition, i.e., several cells can be deposited at the desired locations to bio-mimic natural tissues using bioinks [144]. A 3D bioprinting system can facilitate complex construction using multiple materials and multiple cells by utilizing multiple nozzles [145]. Tissue-specific geometric structures (i.e., convoluted tubules as well as chamber- and lobule-like structures) constructed via 3D bioprinting demonstrated enhanced function and structural maturity [146,147]. The 3D bioprinting system uses bioinks and biomaterial inks to print the product, and ECM-based materials for bioinks have become the popular choice recently [148].
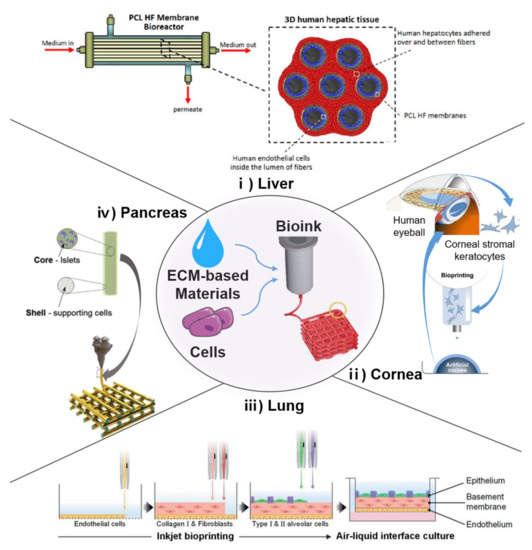
Figure 2.
Applications of tissue engineering based on 3D bioprinting. (i) Poly(ε-caprolactone) (PCL) hollow fiber membrane bioreactor and scheme of 3D human hepatic tissue realized by culturing human hepatocytes over and between PCL HF membranes [129]. (ii) Bioprinting of functional and biomimetic 3D corneal model using hydrogels and cultivated human corneal stromal keratocytes [168]. (iii) Fabrication of alveolar barrier model with all cells inkjet-printed in a layer-by-layer manner [169]. (iv) coaxial printing approach for establishing vascularized bioartificial pancreatic constructs with pancreatic insulin-secreting cells housed in core component [170]. (clockwise).
Numerous stem cell therapy companies are attempting to engineer pancreatic tissues [149]. The pancreas is an organ of both the endocrine and exocrine systems, typically known for its role in controlling blood glucose levels. The pancreas undergoes pathological alterations during aging, including fibrosis, and these changes may result in diabetes mellitus [150]. The specific microenvironment of the ECM is vital to the function of metabolic tissues such as pancreatic islets. Pancreatic cells are expected to function as intended in a specific microenvironment (i.e., collagen type I, IV, and VI abundant), where ECM–cell interactions exist [151]. In recent studies, a microenvironment was created using porcine dECM and endothelial cells via novel biofabrication methods. Three-dimensional printing and a dECM bioink enabled the fabrication of 3D pancreatic tissue constructs with enhanced functions [128]. Stem-cell-based insulin-producing cells for forming islet-like structures have been fabricated as biomimicking pancreatic islets [152]. Emerging evidence suggests that clustering insulin-producing cells inspired by pancreatic islet cells improve cell maturity and function [153,154]. To enable engineered pancreatic tissues to be transplanted, encapsulation strategies involving porous membranes or ECM-like hydrogels are typically adopted [155]. The cell encapsulation device can protect insulin-producing cells from the immune response, promote cell viability, and further advance successful transplantations [156,157].
Moderate fibrosis of the liver is a histological indicator of aging; however, some studies suggest that altered components in the ECM may initiate pathological progression as well [15]. Hepatic fibrosis, i.e., the accumulation of fibrillar ECMs, is a type of dyshomeostasis that results in end-stage liver disease [53]. Hepatic transplantation is an established treatment; however, to fill the gap between donor shortage and high demand, applied hepatocyte transplantation has been applied as an alternative solution in many cases [158]. As the microenvironment of the liver ECM is a critical cue for hepatocyte behaviors and functions, the liver dECM has been used in several studies [95,159,160,161,162]. The potential cell sources, as well as the MSCs and their derivatives of engineered hepatic tissues, were promoted during cell differentiation and hepatic-specific functions in the liver dECM hydrogel [161,162]. MSCs can differentiate into hepatocyte-like cells with therapeutic potential [163]. Biomimetic architecture is a necessary factor for the development of complex artificial tissues. The hollow fiber membrane, which mimics the layer of the actual liver, is a representative example [164]. A recent study used 3D bioprinting to construct the liver lobule architecture, thereby expanding the long-term function of hepatocytes and provided a physiologically relevant mechanical environment [159]. Another 3D bioprinting-based liver tissue engineering study verified that providing suitable biochemical and biomechanical surroundings enhanced the functions of HepG2 cells [95]. Furthermore, physiologically relevant hepatic models are being developed from advanced liver tissue engineering studies by building complex structures and applying co-culture systems [165]. A co-culture of hepatocytes, primary human sinusoidal endothelial cells, and stellate cells on a hollow fiber construct demonstrated self-reorganization and presented a tube-like structure that resembled a real liver tissue [164].
Vascular dysfunctions, which exhibit a higher risk with aging, can result in the decreased activity and deterioration of other organs. Regenerative strategies, including stem cell therapy, have been discussed [166]. Fabricating tubular-shaped tissues is challenging because maintaining a hollow structure with only cells and ECM materials is difficult. However, state-of-the-art biofabrication techniques have enabled the production of functional tubular tissues [167]. Diverse human blood vessel structures have been fabricated with multilevel and multibranch structures. The hollow channel was formed by removing a sacrificial material after the printing process [145]. Newly established coaxial bioprinting enabled the fabrication of hollow blood vessels, which can replace impaired blood vessels. A vessel dECM was mixed with alginate as a bioink and printed through a coaxial nozzle with a controllable, broad range of dimensions [136].
Retinal disorders, including macular degeneration and glaucoma, are age-related diseases. Additionally, cornea changes with age, including the thickening of both epithelial and endothelial basement membranes. As the cornea is known to be organized in a lattice pattern of collagen fibrils, which affords the transparency of the cornea, collagen rich biomaterials are used for inducing collagen fibril orientation. The corneal 3D models could be fabricated via drop-on-demand bioprinting using collagen-based bioink [168]. A recent study introduced a transparent, bioengineered corneal structure for transplantation. The engineered cornea was fabricated by inducing shear stress on a corneal stroma-derived decellularized extracellular matrix bioink based on a 3D cell-printing technique. The aligned collagen fibrils of dECM resulted in a highly mature and transparent corneal stroma analog. Epithelial and stromal cells were used for generating full-thickness corneas, and cell alignment was also investigated [133].
5. Future Perspectives and Conclusions
The ECM is vital to the human body. In addition to its structural role as a physical scaffold for cellular constituents, the ECM participates in numerous biological functions. Although the ECM is composed of non-cellular molecules, it undergoes significant changes during aging, including morphological changes that occur at the tissue level. Consequentially, tissues lose mass, dehydrate, undergo fibrosis, fail to regenerate, and cause dysfunctions. Age-related changes in the ECM are investigated in various forms. ECM-based biomaterials and tissue engineering approaches have been introduced to rejuvenate and regenerate aged tissues. ECM-based biomaterials, including the dECM, closely resemble the actual ECM and have tissue-specific compositions. ECM injection may aid tissue maintenance and compensate for volume losses caused by aging. Moreover, the ECM can be used as a carrier for cell delivery. The ECM provides a tissue-specific microenvironment for cells in vitro and in vivo.
Additionally, ECM-based materials can be used as cell delivery carriers and bioinks. SCs and their derivations can be encapsulated in ECM-based biomaterials and delivered to tissues with age-related dysfunction. The ECM supports the delivered cells, and the delivered cells successfully reconstruct the ECM, thereby validating its effect on restoring tissue function and regeneration. Furthermore, the ECM has been applied in tissue engineering. Engineered tissue can be fabricated using the ECM and cells using precise manufacturing methods such as 3D bioprinting. Fabricated organs, which are suitable for transplantation, can be substituted for donated organs. Biofabrication methods are currently being developed; with the progression of tissue engineering technology, every organ dysfunction associated with aging will soon be treatable.
The ECM demonstrated its versatility in various tissue engineering applications, and 3D bioprinting systems employ ECM-based materials as bioinks and biomaterial inks. The development of 3D bioprinting techniques has enabled the fabrication of complex engineered tissues. The potency of bioprinted tissues for regeneration strategies was verified to be efficient. Bioprinted tissues are used not only for rejuvenating aged tissues but also for treating injuries and chronic diseases. As biomimetics resemble real tissues, bioprinted tissues can be utilized as an in vitro test platform as well. Additionally, owing to the development of tissue engineering technology using ECM-based materials, transplanting without donors and performing in vivo tests without live animals can soon be realized. However, the properties of bioinks must be enhanced, and the accuracy of the manufacturing methods should be improved to fabricate more realistic tissues. Although ECM-based materials offer many advantages, some of their disadvantages must be addressed. They lack mechanical strength, are difficult to deposit precisely, and require a certain amount of time to crosslink. Therefore, further studies are required to improve their mechanical properties and address their weaknesses while maintaining their cell-friendly environment.
Author Contributions
Conceptualization, Y.J. and S.H.H.; investigation, Y.J.; writing—original draft preparation, Y.J.; writing—review and editing, Y.J.; visualization, S.H.H.; supervision, J.J. All authors have read and agreed to the published version of the manuscript.
Funding
This work was funded by a National Research Foundation of South Korea (NRF) grant funded by the Ministry of Science and ICT (No. 2021R1A2C2004981) and the Research Leader Program of the National Research Foundation of Korea (NRF) funded by the Korean government (MSIT) (No. 2020R1A3B2079741).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
| ECM | Extracellular matrix |
| 3D dECM | Tree-dimensional Decellularized extracellular matrix |
| GAGs | Glycosaminoglycans |
| HA | Hyaluronic acid |
| MMPs | Matrix metalloproteinase |
| AGEs | Advanced glycation end products |
| SC | Stem cell |
| MSC | Mesenchymal stem cell |
| ESC | Embryonic stem cell |
| iPSC PRP | Induced pluripotent stem cell Platelet-rich plasma |
| PCL PLA PVC PLGA | Polycaprolactone Polylactic acid Polyvinyl alcohol Poly(lactic-co-glycolic acid) |
References
- Frantz, C.; Stewart, K.M.; Weaver, V.M. The extracellular matrix at a glance. J. Cell Sci. 2010, 123, 4195–4200. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arseni, L.; Lombardi, A.; Orioli, D. From structure to phenotype: Impact of collagen alterations on human health. Int. J. Mol. Sci. 2018, 19, 1407. [Google Scholar] [CrossRef] [Green Version]
- Theocharis, A.D.; Skandalis, S.S.; Gialeli, C.; Karamanos, N.K. Extracellular matrix structure. Adv. Drug Deliv. Rev. 2016, 97, 4–27. [Google Scholar] [CrossRef]
- Trapani, V.; Bonaldo, P.; Corallo, D.; Teti, A. Role of the ECM in notochord formation, function and disease. J. Cell Sci. 1992, 2, 3203–3211. [Google Scholar] [CrossRef] [Green Version]
- Taipale, J.; Keski-Oja, J. Growth factors in the extraceilular. FASEB J. 1997, 11, 51–59. [Google Scholar] [CrossRef] [PubMed]
- Teti, A. Regulation of cellular functions by extracellular matrix. J. Am. Soc. Nephrol. 1992, 2, S83. [Google Scholar] [CrossRef] [PubMed]
- Levi, N.; Papismadov, N.; Solomonov, I.; Sagi, I.; Krizhanovsky, V. The ECM path of senescence in aging: Components and modifiers. FEBS J. 2020, 287, 2636–2646. [Google Scholar] [CrossRef]
- Kurtz, A.; Oh, S.J. Age related changes of the extracellular matrix and stem cell maintenance. Prev. Med. 2012, 54, S50–S56. [Google Scholar] [CrossRef]
- Nicolas, J.; Magli, S.; Rabbachin, L.; Sampaolesi, S.; Nicotra, F.; Russo, L. 3D Extracellular Matrix Mimics: Fundamental Concepts and Role of Materials Chemistry to Influence Stem Cell Fate. Biomacromolecules 2020, 21, 1968–1994. [Google Scholar] [CrossRef]
- Kostrominova, T.Y.; Brooks, S.V. Age-related changes in structure and extracellular matrix protein expression levels in rat tendons. Age 2013, 35, 2203–2214. [Google Scholar] [CrossRef] [Green Version]
- León-López, A.; Morales-Peñaloza, A.; Martínez-Juárez, V.M.; Vargas-Torres, A.; Zeugolis, D.I.; Aguirre-Álvarez, G. Hydrolyzed collagen-sources and applications. Molecules 2019, 24, 4031. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khorramizadeh, M.; Tredget, E.; Telasky, C.; Shen, Q.; Ghahary, A. Aging differentially modulates the expression of collagen and collagenase in dermal fibroblasts. Mol. Cell Biochem. 1999, 194, 99–108. [Google Scholar] [CrossRef]
- Calabresi, C.; Arosio, B.; Galimberti, L.; Scanziani, E.; Bergottini, R.; Annoni, G.; Vergani, C. Natural aging, expression of fibrosis-related genes and collagen deposition in rat lung. Exp. Gerontol. 2007, 42, 1003–1011. [Google Scholar] [CrossRef]
- Shelton, D.N.; Chang, E.; Whittier, P.S.; Choi, D.; Funk, W.D. Microarray analysis of replicative senescence. Curr. Biol. 1999, 9, 939–945. [Google Scholar] [CrossRef] [Green Version]
- Gagliano, N.; Arosio, B.; Grizzi, F.; Masson, S.; Tagliabue, J.; Dioguardi, N.; Vergani, C.; Annoni, G. Reduced collagenolytic activity of matrix metalloproteinases and development of liver fibrosis in the aging rat. Mech. Ageing Dev. 2002, 123, 413–425. [Google Scholar] [CrossRef] [Green Version]
- Lepelletier, F.X.; Mann, D.M.A.; Robinson, A.C.; Pinteaux, E.; Boutin, H. Early changes in extracellular matrix in Alzheimer’s disease. Neuropathol. Appl. Neurobiol. 2017, 43, 167–182. [Google Scholar] [CrossRef]
- Eckes, B.; Mauch, C.; Hüppe, G.; Krieg, T. Downregulation of collagen synthesis in fibroblasts within three-dimensional collagen lattices involves transcriptional and posttranscriptional mechanisms. FEBS Lett. 1993, 318, 129–133. [Google Scholar] [CrossRef] [Green Version]
- Panwar, P.; Butler, G.S.; Jamroz, A.; Azizi, P.; Overall, C.M.; Brömme, D. Aging-associated modifications of collagen affect its degradation by matrix metalloproteinases. Matrix Biol. 2018, 65, 30–44. [Google Scholar] [CrossRef]
- Singh, P.; Carraher, C.; Schwarzbauer, J.E. Assembly of fibronectin extracellular matrix. Annu. Rev. Cell Dev. Biol. 2010, 26, 397–419. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Takahashi, S.; Leiss, M.; Moser, M.; Ohashi, T.; Kitao, T.; Heckmann, D.; Pfeifer, A.; Kessler, H.; Takagi, J.; Erickson, H.P.; et al. The RGD motif in fibronectin is essential for development but dispensable for fibril assembly. J. Cell Biol. 2007, 178, 167–178. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Krizhanovsky, V.; Yon, M.; Dickins, R.A.; Hearn, S.; Simon, J.; Miething, C.; Yee, H.; Zender, L.; Lowe, S.W. Senescence of Activated Stellate Cells Limits Liver Fibrosis. Cell 2008, 134, 657–667. [Google Scholar] [CrossRef] [Green Version]
- Kumazaki, T.; Kobayashi, M.; Mitsui, Y. Enhanced expression of fibronectin during in vivo cellular aging of human vascular endothelial cells and skin fibroblasts. Exp. Cell Res. 1993, 205, 396–402. [Google Scholar] [CrossRef]
- Kumazaki, T.; Robetorye, R.S.; Robetorye, S.C.; Smith, J.R. Fibronectin expression increases during in vitro cellular senescence: Correlation with increased cell area. Exp. Cell Res. 1991, 195, 13–19. [Google Scholar] [CrossRef]
- Yeo, G.C.; Mithieux, S.M.; Weiss, A.S. The elastin matrix in tissue engineering and regeneration. Curr. Opin. Biomed. Eng. 2018, 6, 27–32. [Google Scholar] [CrossRef]
- Panwar, P.; Hedtke, T.; Heinz, A.; Andrault, P.M.; Hoehenwarter, W.; Granville, D.J.; Schmelzer, C.E.H.; Brömme, D. Expression of elastolytic cathepsins in human skin and their involvement in age-dependent elastin degradation. Biochim. Biophys. Acta Gen. Subj. 2020, 1864, 129544. [Google Scholar] [CrossRef] [PubMed]
- Antonicelli, F.; Bellon, G.; Debelle, L.; Hornebeck, W. Elastin-Elastases and Inflamm-Aging. Curr. Top. Dev. Biol. 2007, 79, 99–155. [Google Scholar] [CrossRef] [PubMed]
- Åstrand, H.; Stålhand, J.; Karlsson, J.; Karlsson, M.; Sonesson, B.; Länne, T.; Fonck, E.; Feigl, G.G.; Fasel, J.; Sage, D.; et al. In vivo estimation of the contribution of elastin and collagen to the mechanical properties in the human abdominal aorta: Effect of age and sex. Curr. Top. Dev. Biol. 2011, 40, 2552–2556. [Google Scholar] [CrossRef] [Green Version]
- Fonck, E.; Feigl, G.G.; Fasel, J.; Sage, D.; Unser, M.; Rüfenacht, D.A.; Stergiopulos, N. Effect of aging on elastin functionality in human cerebral arteries. Stroke 2009, 40, 2552–2556. [Google Scholar] [CrossRef]
- Colognato, H.; Yurchenco, P.D. Form and function: The laminin family of heterotrimers. Dev. Dyn. 2000, 218, 213–234. [Google Scholar] [CrossRef]
- Labat-Robert, J. Age-dependent remodeling of connective tissue: Role of fibronectin and laminin. Pathol. Biol. 2003, 51, 563–568. [Google Scholar] [CrossRef] [PubMed]
- Godin, L.M.; Sandri, B.J.; Wagner, D.E.; Meyer, C.M.; Price, A.P.; Akinnola, I.; Weiss, D.J.; Panoskaltsis-Mortari, A.P.M. Decreased laminin expression by human lung epithelial cells and fibroblasts cultured in acellular lung scaffolds from aged mice. PLoS ONE 2016, 11, 1–17. [Google Scholar] [CrossRef] [Green Version]
- Lee, K.M.; Chand, K.K.; Hammond, L.A.; Lavidis, N.A.; Noakes, P.G. Functional decline at the aging neuromuscular junction is associated with altered laminin-α4 expression. Aging 2017, 9, 880–899. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stocks, D.; Sundaram, H.; Michaels, J.; Durrani, M.J.; Wortzman, M.S.; Nelson, D.B. Rheological Evaluation of the Physical Properties. J. Drugs Dermatol. 2011, 10, 974–980. [Google Scholar] [PubMed]
- Zhang, W.; Mu, H.; Zhang, A.; Cui, G.; Chen, H.; Duan, J.; Wang, S. A decrease in moisture absorption-retention capacity of N-deacetylation of hyaluronic acid. Glycoconj. J. 2013, 30, 577–583. [Google Scholar] [CrossRef]
- Ghersetich, I.; Lotti, T.; Campanile, G.; Grappone, C.; Dini, G. Hyaluronic Acid in Cutaneous Intrinsic Aging. Int. J. Dermatol. 1994, 33, 119–122. [Google Scholar] [CrossRef]
- Longas, M.O.; Burden, J.D.; Lesniak, J.; Booth, R.M.; McPencow, J.A.; Park, J.I. Hyaluronic acid N-deacetylase assay in whole skin. Biomacromolecules 2003, 4, 189–192. [Google Scholar] [CrossRef]
- Papakonstantinou, E.; Roth, M.; Karakiulakis, G. Hyaluronic acid: A key molecule in skin aging. Dermato-Endocrinology 2012, 4, 253–258. [Google Scholar] [CrossRef] [Green Version]
- Branco, A.; Rodrigues, S.A.; Fabro, A.T.; Fonseca-Alves, C.E.; Martins, R.H.G. Hyaluronic acid behavior in the lamina propria of the larynx with advancing age. Otolaryngol. Head Neck Surg. 2014, 151, 652–656. [Google Scholar] [CrossRef] [PubMed]
- López-Otín, C.; Blasco, M.A.; Partridge, L.; Serrano, M.; Kroemer, G. The hallmarks of aging. Cell 2013, 153, 1194. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lu, P.; Takai, K.; Weaver, V.M.; Werb, Z. Extracellular Matrix degradation and remodeling in development and disease. Cold Spring Harb. Perspect. Biol. 2011, 3, a005058. [Google Scholar] [CrossRef] [PubMed]
- Ozcebe, S.G.; Bahcecioglu, G.; Yue, X.S.; Zorlutuna, P. Effect of cellular and ECM aging on human iPSC-derived cardiomyocyte performance, maturity and senescence. Biomaterials 2021, 268, 120554. [Google Scholar] [CrossRef] [PubMed]
- Kragstrup, T.W.; Kjaer, M.; Mackey, A.L. Structural, biochemical, cellular, and functional changes in skeletal muscle extracellular matrix with aging. Scand. J. Med. Sci. Sport. 2011, 21, 749–757. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Colcombe, S.J.; Erickson, K.I.; Raz, N.; Webb, A.G.; Cohen, N.J.; McAuley, E.; Kramer, A.F. Aerobic fitness reduces brain tissue loss in aging humans. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2003, 58, 176–180. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bonneh-Barkay, D.; Wiley, C.A. Brain extracellular matrix in neurodegeneration. Brain Pathol. 2009, 19, 573–585. [Google Scholar] [CrossRef] [PubMed]
- Quan, T.; Fisher, G.J. Role of age-associated alterations of the dermal extracellular matrix microenvironment in human skin aging: A mini-review. Gerontology 2015, 61, 427–434. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tu, Y.; Quan, T. Oxidative stress and human skin connective tissue aging. Cosmetics 2016, 3, 28. [Google Scholar] [CrossRef]
- Kannus, P.; Paavola, M.; Józsa, L. Aging and degeneration of tendons. In Tendon Injuries; Springer: London, UK, 2005; pp. 25–31. [Google Scholar] [CrossRef]
- Ohnishi, T.; Novais, E.J.; Risbud, M.V. Alterations in ECM signature underscore multiple sub-phenotypes of intervertebral disc degeneration. Matrix Biol. Plus 2020, 6–7, 100036. [Google Scholar] [CrossRef] [PubMed]
- Peng, Z.; Sun, H.; Bunpetch, V.; Koh, Y.; Wen, Y.; Wu, D.; Ouyang, H. The regulation of cartilage extracellular matrix homeostasis in joint cartilage degeneration and regeneration. Biomaterials 2021, 268, 120555. [Google Scholar] [CrossRef] [PubMed]
- Murtha, L.A.; Morten, M.; Schuliga, M.J.; Mabotuwana, N.S.; Hardy, S.A.; Waters, D.W.; Burgess, J.K.; Ngo, D.T.M.; Sverdlov, A.L.; Knight, D.A.; et al. The role of pathological aging in cardiac and pulmonary fibrosis. Aging Dis. 2019, 10, 419–428. [Google Scholar] [CrossRef] [Green Version]
- Díez, J. Arterial stiffness and extracellular matrix. Adv. Cardiol. 2007, 44, 76–95. [Google Scholar] [CrossRef] [PubMed]
- Kohn, J.C.; Lampi, M.C.; Reinhart-King, C.A. Age-related vascular stiffening: Causes and consequences. Front. Genet. 2015, 6, 1–17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arteel, G.E.; Naba, A. The liver matrisome—Looking beyond collagens. JHEP Rep. 2020, 2, 100115. [Google Scholar] [CrossRef]
- Gulati, S.; Thannickal, V.J.; Murtha, L.A.; Morten, M.; Schuliga, M.J.; Mabotuwana, N.S.; Hardy, S.A.; Waters, D.W.; Burgess, J.K.; Ngo, D.T.M.; et al. The Aging Lung and Idiopathic Pulmonary Fibrosis. Am. J. Med. Sci. 2019, 10, 384–389. [Google Scholar] [CrossRef] [PubMed]
- Brunet, A.; Berger, S.L. Epigenetics of aging and aging-related disease. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2014, 69, 17–20. [Google Scholar] [CrossRef] [Green Version]
- Li, J.; Sun, Y.; Liang, Y.; Ma, J.; Li, B.; Ma, C.; Tanzi, R.E.; Zhang, H.; Liu, K.; Zhang, C. Extracellular Elastin Molecule Modulates Alzheimer’s Aβ Dynamics In Vitro and In Vivo by Affecting Microglial Activities. CCS Chem. 2020, 3, 1830–1837. [Google Scholar] [CrossRef]
- Francis-Sedlak, M.E.; Uriel, S.; Larson, J.C.; Greisler, H.P.; Venerus, D.C.; Brey, E.M. Characterization of type I collagen gels modified by glycation. Biomaterials 2020, 30, 472. [Google Scholar] [CrossRef]
- Garcia-Fernandez; Jacobs-Cachá; Mora-Gutiérrez; Vergara; Orbe; Soler Matrix Metalloproteinases in Diabetic Kidney Disease. J. Clin. Med. 2020, 9, 472. [CrossRef] [Green Version]
- Fournet, M.; Bonté, F.; Desmoulière, A. Glycation damage: A possible hub for major pathophysiological disorders and aging. Aging Dis. 2018, 9, 880–900. [Google Scholar] [CrossRef] [Green Version]
- Bansode, S.B.; Gacche, R.N. Glycation-induced modification of tissue-specific ECM proteins: A pathophysiological mechanism in degenerative diseases. Biochim. Biophys. Acta Gen. Subj. 2019, 1863, 129411. [Google Scholar] [CrossRef]
- Saska, S.; Pilatti, L.; Blay, A.; Shibli, J.A. Bioresorbable polymers: Advanced materials and 4D printing for tissue engineering. Polymers 2021, 13, 563. [Google Scholar] [CrossRef] [PubMed]
- Unal, A.Z.; West, J.L. Synthetic ECM: Bioactive Synthetic Hydrogels for 3D Tissue Engineering. Bioconjug. Chem. 2020, 31, 2253–2271. [Google Scholar] [CrossRef] [PubMed]
- Das, D.; Noh, I. Biomimetic Medical Materials: From Nanotechnology to 3D Bioprinting; Springer: Singapore, 2018; Vol. 1064, ISBN 9789811304446. [Google Scholar]
- Hinderer, S.; Layland, S.L.; Schenke-Layland, K. ECM and ECM-like materials—Biomaterials for applications in regenerative medicine and cancer therapy. Adv. Drug Deliv. Rev. 2016, 97, 260–269. [Google Scholar] [CrossRef] [PubMed]
- Xing, H.; Lee, H.; Luo, L.; Kyriakides, T.R. Extracellular matrix-derived biomaterials in engineering cell function. Biotechnol. Adv. 2020, 42, 107421. [Google Scholar] [CrossRef]
- Gaharwar, A.K.; Singh, I.; Khademhosseini, A. Engineered biomaterials for in situ tissue regeneration. Nat. Rev. Mater. 2020, 5, 686–705. [Google Scholar] [CrossRef]
- Dayan, S.H.; Bassichis, B.A. Facial Dermal Fillers: Selection of Appropriate Products and Techniques. Aesthet. Surg. J. 2008, 28, 335–347. [Google Scholar] [CrossRef] [Green Version]
- Shin, J.W.; Kwon, S.H.; Choi, J.Y.; Na, J.I.; Huh, C.H.; Choi, H.R.; Park, K.C. Molecular mechanisms of dermal aging and antiaging approaches. Int. J. Mol. Sci. 2019, 20, 2126. [Google Scholar] [CrossRef] [Green Version]
- Fallacara, A.; Manfredini, S.; Durini, E.; Vertuani, S. Hyaluronic Acid Fillers in Soft Tissue Regeneration. Facial Plast. Surg. 2017, 33, 87–96. [Google Scholar] [CrossRef] [PubMed]
- Puls, T.J.; Fisher, C.S.; Cox, A.; Plantenga, J.M.; McBride, E.L.; Anderson, J.L.; Goergen, C.J.; Bible, M.; Moller, T.; Voytik-Harbin, S.L. Regenerative tissue filler for breast conserving surgery and other soft tissue restoration and reconstruction needs. Sci. Rep. 2021, 11, 1–17. [Google Scholar] [CrossRef]
- Xing, Q.; Qian, Z.; Jia, W.; Ghosh, A.; Tahtinen, M.; Zhao, F. Natural Extracellular Matrix for Cellular and Tissue Biomanufacturing. ACS Biomater. Sci. Eng. 2017, 3, 1462–1476. [Google Scholar] [CrossRef]
- Rittié, L. Type I collagen purification from rat tail tendons. Methods Mol. Biol. 2017, 1627, 287–308. [Google Scholar] [CrossRef]
- Avila Rodríguez, M.I.; Rodríguez Barroso, L.G.; Sánchez, M.L. Collagen: A review on its sources and potential cosmetic applications. J. Cosmet. Dermatol. 2018, 17, 20–26. [Google Scholar] [CrossRef]
- Kiyozumi, D.; Nakano, I.; Sato-Nishiuchi, R.; Tanaka, S.; Sekiguchi, K. Laminin is the ECM niche for trophoblast stem cells. Life Sci. Alliance 2020, 3, 1–9. [Google Scholar] [CrossRef]
- Patten, J.; Wang, K. Fibronectin in development and wound healing. Adv. Drug Deliv. Rev. 2021, 170, 353–368. [Google Scholar] [CrossRef]
- Bazrafshan, Z.; Stylios, G.K. Spinnability of collagen as a biomimetic material: A review. Int. J. Biol. Macromol. 2019, 129, 693–705. [Google Scholar] [CrossRef]
- Hsieh, D.J.; Srinivasan, P. Protocols for accelerated production and purification of collagen scaffold and atelocollagen from animal tissues. Biotechniques 2020, 69, 221–225. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Kim, D.J.; Lee, H.J.; Kim, B.K.; Kim, Y.S. Atelocollagen Injection Improves Tendon Integrity in Partial-Thickness Rotator Cuff Tears: A Prospective Comparative Study. Orthop. J. Sport. Med. 2020, 8, 1–7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wyganowska-Swiatkowska, M.; Duda-Sobczak, A.; Corbo, A.; Matthews-Brzozowska, T. Atelocollagen application in human periodontal tissue treatment—A pilot study. Life 2020, 10, 114. [Google Scholar] [CrossRef]
- Jia, W.; Li, M.; Liu, L.; Zhou, H.; Liu, X.; Gu, G.; Xiao, M.; Chen, Z. Fabrication and assessment of chondroitin sulfate-modified collagen nanofibers for small-diameter vascular tissue engineering applications. Carbohydr. Polym. 2021, 257, 117573. [Google Scholar] [CrossRef] [PubMed]
- Wakuda, Y.; Nishimoto, S.; Suye, S.I.; Fujita, S. Native collagen hydrogel nanofibres with anisotropic structure using core-shell electrospinning. Sci. Rep. 2018, 8, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Antman-Passig, M.; Levy, S.; Gartenberg, C.; Schori, H.; Shefi, O. Mechanically Oriented 3D Collagen Hydrogel for Directing Neurite Growth. Tissue Eng. Part A 2017, 23, 403–414. [Google Scholar] [CrossRef]
- Boeriu, C.G.; Springer, J.; Kooy, F.K.; van den Broek, L.A.M.; Eggink, G. Production Methods for Hyaluronan. Int. J. Carbohydr. Chem. 2013, 2013, 1–14. [Google Scholar] [CrossRef] [Green Version]
- Tiwari, S.; Bahadur, P. Modified hyaluronic acid based materials for biomedical applications. Int. J. Biol. Macromol. 2019, 121, 556–571. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Zhu, D.; Paul, A.; Cai, L.; Enejder, A.; Yang, F.; Heilshorn, S.C. Covalently Adaptable Elastin-Like Protein–Hyaluronic Acid (ELP–HA) Hybrid Hydrogels with Secondary Thermoresponsive Crosslinking for Injectable Stem Cell Delivery. Adv. Funct. Mater. 2017, 27. [Google Scholar] [CrossRef]
- Hozumi, T.; Kageyama, T.; Ohta, S.; Fukuda, J.; Ito, T. Injectable Hydrogel with Slow Degradability Composed of Gelatin and Hyaluronic Acid Cross-Linked by Schiff’s Base Formation. Biomacromolecules 2018, 19, 288–297. [Google Scholar] [CrossRef]
- Chen, L.H.; Xue, J.F.; Zheng, Z.Y.; Shuhaidi, M.; Thu, H.E.; Hussain, Z. Hyaluronic acid, an efficient biomacromolecule for treatment of inflammatory skin and joint diseases: A review of recent developments and critical appraisal of preclinical and clinical investigations. Int. J. Biol. Macromol. 2018, 116, 572–584. [Google Scholar] [CrossRef] [PubMed]
- Gupta, R.C.; Lall, R.; Srivastava, A.; Sinha, A. Hyaluronic acid: Molecular mechanisms and therapeutic trajectory. Front. Vet. Sci. 2019, 6, 1–24. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jin, Y.; Koh, R.H.; Kim, S.H.; Kim, K.M.; Park, G.K.; Hwang, N.S. Injectable anti-inflammatory hyaluronic acid hydrogel for osteoarthritic cartilage repair. Mater. Sci. Eng. C 2020, 115, 111096. [Google Scholar] [CrossRef]
- Majewski, G.P.; Rodan, K.; Fields, K.; Falla, T.J. Characterization of bound water in skin hydrators prepared with and without a 3D3P interpenetrating polymer network. Ski. Res. Technol. 2019, 25, 150–157. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.S.; Das, S.; Jang, J.; Cho, D.W. Decellularized Extracellular Matrix-based Bioinks for Engineering Tissue- And Organ-specific Microenvironments. Chem. Rev. 2020, 120, 10608–10661. [Google Scholar] [CrossRef]
- Dziki, J.L.; Huleihel, L.; Scarritt, M.E.; Badylak, S.F. Extracellular Matrix Bioscaffolds as Immunomodulatory Biomaterials. Tissue Eng. Part A 2017, 23, 1152–1159. [Google Scholar] [CrossRef]
- Mayorca-Guiliani, A.E.; Madsen, C.D.; Cox, T.R.; Horton, E.R.; Venning, F.A.; Erler, J.T. ISDoT: In situ decellularization of tissues for high-resolution imaging and proteomic analysis of native extracellular matrix. Nat. Med. 2017, 23, 890–898. [Google Scholar] [CrossRef]
- Das, S.; Jang, J. 3D bioprinting and decellularized ECM-based biomaterials for in vitro CV tissue engineering. J. 3D Print. Med. 2018, 2, 69–87. [Google Scholar] [CrossRef]
- Lee, H.; Han, W.; Kim, H.; Ha, D.H.; Jang, J.; Kim, B.S.; Cho, D.W. Development of Liver Decellularized Extracellular Matrix Bioink for Three-Dimensional Cell Printing-Based Liver Tissue Engineering. Biomacromolecules 2017, 18, 1229–1237. [Google Scholar] [CrossRef] [PubMed]
- Yao, Q.; Zheng, Y.W.; Lan, Q.H.; Kou, L.; Xu, H.L.; Zhao, Y.Z. Recent development and biomedical applications of decellularized extracellular matrix biomaterials. Mater. Sci. Eng. C 2019, 104, 109942. [Google Scholar] [CrossRef] [PubMed]
- Nouri Barkestani, M.; Naserian, S.; Uzan, G.; Shamdani, S. Post-decellularization techniques ameliorate cartilage decellularization process for tissue engineering applications. J. Tissue Eng. 2021, 12, 12–14. [Google Scholar] [CrossRef]
- Kim, H.; Kang, B.; Cui, X.; Lee, S.H.; Lee, K.; Cho, D.W.; Hwang, W.; Woodfield, T.B.F.; Lim, K.S.; Jang, J. Light-Activated Decellularized Extracellular Matrix-Based Bioinks for Volumetric Tissue Analogs at the Centimeter Scale. Adv. Funct. Mater. 2021, 31, 2011252. [Google Scholar]
- Kwon, S.G.; Kwon, Y.W.; Lee, T.W.; Park, G.T.; Kim, J.H. Recent advances in stem cell therapeutics and tissue engineering strategies. Biomater. Res. 2018, 22, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Bi, H.; Ye, K.; Jin, S. Proteomic analysis of decellularized pancreatic matrix identifies collagen V as a critical regulator for islet organogenesis from human pluripotent stem cells. Biomaterials 2020, 233, 119673. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Rivera-Bolanos, N.; Jiang, B.; Ameer, G.A. Advanced Functional Biomaterials for Stem Cell Delivery in Regenerative Engineering and Medicine. Adv. Funct. Mater. 2019, 29, 1809009. [Google Scholar] [CrossRef]
- Bejleri, D.; Streeter, B.W.; Nachlas, A.L.Y.; Brown, M.E.; Gaetani, R.; Christman, K.L.; Davis, M.E. A Bioprinted Cardiac Patch Composed of Cardiac-Specific Extracellular Matrix and Progenitor Cells for Heart Repair. Adv. Healthc. Mater. 2018, 7, 1800672. [Google Scholar] [CrossRef]
- Wollenberg, A.L.; O’Shea, T.M.; Kim, J.H.; Czechanski, A.; Reinholdt, L.G.; Sofroniew, M.V.; Deming, T.J. Injectable polypeptide hydrogels via methionine modification for neural stem cell delivery. Biomaterials 2018, 178, 527–545. [Google Scholar] [CrossRef] [PubMed]
- Yuan, X.; Wei, Y.; Villasante, A.; Ng, J.J.D.; Arkonac, D.E.; Chao, P.; Hsiu, G.; Vunjak-Novakovic, G. Stem cell delivery in tissue-specific hydrogel enabled meniscal repair in an orthotopic rat model. Biomaterials 2017, 132, 59–71. [Google Scholar] [CrossRef]
- Pan, J.; Li, H.; Fang, Y.; Shen, Y.-B.; Zhou, X.Y.; Zhu, F.; Zhu, L.X.; Du, Y.H.; Yu, X.F.; Wang, Y.; et al. Regeneration of a bioengineered thyroid using decellularized thyroid matrix. Thyroid 2019, 29, 142–152. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Kong, W.; Li, B.; Ni, Y.; Yuan, T.; Guo, L.; Lin, H.; Fan, H.; Fan, Y.; Zhang, X. Fabrication and characterization of collagen-based injectable and self-crosslinkable hydrogels for cell encapsulation. Colloids Surf. B Biointerfaces 2018, 167, 448–456. [Google Scholar] [CrossRef] [PubMed]
- Bai, R.; Tian, L.; Li, Y.; Zhang, J.; Wei, Y.; Jin, Z.; Liu, Z.; Liu, H. Combining ECM Hydrogels of Cardiac Bioactivity with Stem Cells of High Cardiomyogenic Potential for Myocardial Repair. Stem Cells Int. 2019, 2019, 6708435. [Google Scholar] [CrossRef]
- Shakouri-Motlagh, A.; O’Connor, A.J.; Brennecke, S.P.; Kalionis, B.; Heath, D.E. Native and solubilized decellularized extracellular matrix: A critical assessment of their potential for improving the expansion of mesenchymal stem cells. Acta Biomater. 2017, 55, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Giobbe, G.G.; Crowley, C.; Luni, C.; Campinoti, S.; Khedr, M.; Kretzschmar, K.; De Santis, M.M.; Zambaiti, E.; Michielin, F.; Meran, L.; et al. Extracellular matrix hydrogel derived from decellularized tissues enables endodermal organoid culture. Nat. Commun. 2019, 10, 5658. [Google Scholar] [CrossRef] [Green Version]
- Doi, D.; Magotani, H.; Kikuchi, T.; Ikeda, M.; Hiramatsu, S.; Yoshida, K.; Amano, N.; Nomura, M.; Umekage, M.; Morizane, A.; et al. Pre-clinical study of induced pluripotent stem cell-derived dopaminergic progenitor cells for Parkinson’s disease. Nat. Commun. 2020, 11, 3369. [Google Scholar] [CrossRef]
- Lee, W.Y.; Wang, B. Cartilage repair by mesenchymal stem cells: Clinical trial update and perspectives. J. Orthop. Transl. 2017, 9, 76–88. [Google Scholar] [CrossRef]
- Hashimoto, H.; Olson, E.N.; Bassel-Duby, R. Therapeutic approaches for cardiac regeneration and repair. Nat. Rev. Cardiol. 2018, 15, 585–600. [Google Scholar] [CrossRef] [PubMed]
- Yong, U.; Lee, S.; Jung, S.; Jang, J. Interdisciplinary approaches to advanced cardiovascular tissue engineering: ECM-based biomaterials, 3D bioprinting, and its assessment. Prog. Biomed. Eng. 2020, 2, 042003. [Google Scholar] [CrossRef]
- Park, S.J.; Kim, R.Y.; Park, B.W.; Lee, S.; Choi, S.W.; Park, J.H.; Choi, J.J.; Kim, S.W.; Jang, J.; Cho, D.W.; et al. Dual stem cell therapy synergistically improves cardiac function and vascular regeneration following myocardial infarction. Nat. Commun. 2019, 10, 1–12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, I.G.; Hwang, M.P.; Park, J.S.; Kim, S.H.; Kim, J.H.; Kang, H.J.; Subbiah, R.; Ko, U.H.; Shin, J.H.; Kim, C.H.; et al. Stretchable ECM Patch Enhances Stem Cell Delivery for Post-MI Cardiovascular Repair. Adv. Healthc. Mater. 2019, 8, 1900593. [Google Scholar] [CrossRef] [PubMed]
- Seo, Y.; Jung, Y.; Kim, S.H. Decellularized heart ECM hydrogel using supercritical carbon dioxide for improved angiogenesis. Acta Biomater. 2018, 67, 270–281. [Google Scholar] [CrossRef] [PubMed]
- Betancur, M.I.; Mason, H.D.; Alvarado-Velez, M.; Holmes, P.V.; Bellamkonda, R.V.; Karumbaiah, L. Chondroitin Sulfate Glycosaminoglycan Matrices Promote Neural Stem Cell Maintenance and Neuroprotection Post-Traumatic Brain Injury. ACS Biomater. Sci. Eng. 2017, 3, 420–430. [Google Scholar] [CrossRef] [Green Version]
- Výborný, K.; Vallová, J.; Kočí, Z.; Kekulová, K.; Jiráková, K.; Jendelová, P.; Hodan, J.; Kubinová, Š. Genipin and EDC crosslinking of extracellular matrix hydrogel derived from human umbilical cord for neural tissue repair. Sci. Rep. 2019, 9, 1–15. [Google Scholar] [CrossRef] [Green Version]
- Lam, D.; Enright, H.A.; Cadena, J.; Peters, S.K.G.; Sales, A.P.; Osburn, J.J.; Soscia, D.A.; Kulp, K.S.; Wheeler, E.K.; Fischer, N.O. Tissue-specific extracellular matrix accelerates the formation of neural networks and communities in a neuron-glia co-culture on a multi-electrode array. Sci. Rep. 2019, 9, 1–15. [Google Scholar] [CrossRef]
- Zhao, T.; Sellers, D.L.; Cheng, Y.; Horner, P.J.; Pun, S.H. Tunable, Injectable Hydrogels Based on Peptide-Cross-Linked, Cyclized Polymer Nanoparticles for Neural Progenitor Cell Delivery. Biomacromolecules 2017, 18, 2723–2731. [Google Scholar] [CrossRef]
- Nasiri, N.; Hosseini, S.; Alini, M.; Khademhosseini, A.; Baghaban Eslaminejad, M. Targeted cell delivery for articular cartilage regeneration and osteoarthritis treatment. Drug Discov. Today 2019, 24, 2212–2224. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Wang, J.; Huang, X.; Fang, W.; Tao, Y.; Zhao, T.; Liang, C.; Hua, J.; Chen, Q.; Li, F. Injectable decellularized nucleus pulposus-based cell delivery system for differentiation of adipose-derived stem cells and nucleus pulposus regeneration. Acta Biomater. 2018, 81, 115–128. [Google Scholar] [CrossRef]
- Carvalho, M.S.; Silva, J.C.; Cabral, J.M.S.; da Silva, C.L.; Vashishth, D. Cultured cell-derived extracellular matrices to enhance the osteogenic differentiation and angiogenic properties of human mesenchymal stem/stromal cells. J. Tissue Eng. Regen. Med. 2019, 13, 1544–1558. [Google Scholar] [CrossRef]
- Koh, R.H.; Jin, Y.; Kim, J.; Hwang, N.S. Inflammation-Modulating Hydrogels for Osteoarthritis Cartilage Tissue Engineering. Cells 2020, 9, 419. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barbon, S.; Contran, M.; Stocco, E.; Todros, S.; Macchi, V.; De Caro, R.; Porzionato, A. Enhanced biomechanical properties of polyvinyl alcohol-based hybrid scaffolds for cartilage tissue engineering. Processes 2021, 9, 730. [Google Scholar] [CrossRef]
- Chen, Y.; Lee, K.; Yang, Y.; Kawazoe, N.; Chen, G. PLGA-collagen-ECM hybrid meshes mimicking stepwise osteogenesis and their influence on the osteogenic differentiation of hMSCs. Biofabrication 2020, 12, 25027. [Google Scholar] [CrossRef] [PubMed]
- Sohn, S.; Van Buskirk, M.; Buckenmeyer, M.J.; Londono, R.; Faulk, D. Whole Organ Engineering: Approaches, Challenges, and Future Directions. Appl. Sci. 2020, 10, 4277. [Google Scholar] [CrossRef]
- Kim, J.; Shim, I.K.; Hwang, D.G.; Lee, Y.N.; Kim, M.; Kim, H.; Kim, S.-W.; Lee, S.; Kim, S.C.; Cho, D.-W.; et al. 3D cell printing of islet-laden pancreatic tissue-derived extracellular matrix bioink constructs for enhancing pancreatic functions. J. Mater. Chem. B 2019, 7, 1773–1781. [Google Scholar] [CrossRef] [PubMed]
- Salerno, S.; Tasselli, F.; Drioli, E.; De Bartolo, L. Poly(ε-caprolactone) hollow fiber membranes for the biofabrication of a vascularized human liver tissue. Membranes 2020, 10, 112. [Google Scholar] [CrossRef]
- Singh, N.K.; Han, W.; Nam, S.A.; Kim, J.W.; Kim, J.Y.; Kim, Y.K.; Cho, D.W. Three-dimensional cell-printing of advanced renal tubular tissue analogue. Biomaterials 2020, 232, 119734. [Google Scholar] [CrossRef]
- Yoon, H.J.; Lee, Y.J.; Baek, S.; Chung, Y.S.; Kim, D.H.; Lee, J.H.; Shin, Y.C.; Shin, Y.M.; Ryu, C.; Kim, H.S.; et al. Hormone autocrination by vascularized hydrogel delivery of ovary spheroids to rescue ovarian dysfunctions. Sci. Adv. 2021, 7, eabe8873. [Google Scholar] [CrossRef]
- Kim, E.; Choi, S.; Kang, B.; Kong, J.H.; Kim, Y.; Yoon, W.H.; Lee, H.R.; Kim, S.E.; Kim, H.M.; Lee, H.S.; et al. Creation of bladder assembloids mimicking tissue regeneration and cancer. Nature 2020, 588, 664–669. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Jang, J.; Park, J.; Lee, K.P.; Lee, S.; Lee, D.M.; Kim, K.H.; Kim, H.K.; Cho, D.W. Shear-induced alignment of collagen fibrils using 3D cell printing for corneal stroma tissue engineering. Biofabrication 2019, 11, 35017. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.J.; Jun, Y.J.; Kim, D.Y.; Yi, H.G.; Chae, S.H.; Kang, J.; Lee, J.; Gao, G.; Kong, J.S.; Jang, J.; et al. A 3D cell printed muscle construct with tissue-derived bioink for the treatment of volumetric muscle loss. Biomaterials 2019, 206, 160–169. [Google Scholar] [CrossRef]
- Kim, B.S.; Kwon, Y.W.; Kong, J.S.; Park, G.T.; Gao, G.; Han, W.; Kim, M.B.; Lee, H.; Kim, J.H.; Cho, D.W. 3D cell printing of in vitro stabilized skin model and in vivo pre-vascularized skin patch using tissue-specific extracellular matrix bioink: A step towards advanced skin tissue engineering. Biomaterials 2018, 168, 38–53. [Google Scholar] [CrossRef]
- Gao, G.; Lee, J.H.; Jang, J.; Lee, D.H.; Kong, J.S.; Kim, B.S.; Choi, Y.J.; Jang, W.B.; Hong, Y.J.; Kwon, S.M.; et al. Tissue Engineered Bio-Blood-Vessels Constructed Using a Tissue-Specific Bioink and 3D Coaxial Cell Printing Technique: A Novel Therapy for Ischemic Disease. Adv. Funct. Mater. 2017, 27, 1700798. [Google Scholar] [CrossRef]
- Nam, H.; Jeong, H.J.; Jo, Y.; Lee, J.Y.; Ha, D.H.; Kim, J.H.; Chung, J.H.; Cho, Y.S.; Cho, D.W.; Lee, S.J.; et al. Multi-layered Free-form 3D Cell-printed Tubular Construct with Decellularized Inner and Outer Esophageal Tissue-derived Bioinks. Sci. Rep. 2020, 10, 1–14. [Google Scholar] [CrossRef]
- Desai, T.A.; Tang, Q. Islet encapsulation therapy—Racing towards the finish line? Nat. Rev. Endocrinol. 2018, 14, 630–632. [Google Scholar] [CrossRef] [PubMed]
- Song, H.H.G.; Rumma, R.T.; Ozaki, C.K.; Edelman, E.R.; Chen, C.S. Vascular Tissue Engineering: Progress, Challenges, and Clinical Promise. Cell Stem Cell 2018, 22, 340–354. [Google Scholar] [CrossRef] [Green Version]
- Pedde, R.D.; Mirani, B.; Navaei, A.; Styan, T.; Wong, S.; Mehrali, M.; Thakur, A.; Mohtaram, N.K.; Bayati, A.; Dolatshahi-Pirouz, A.; et al. Emerging Biofabrication Strategies for Engineering Complex Tissue Constructs. Adv. Mater. 2017, 29, 1606061. [Google Scholar] [CrossRef]
- Gao, G.; Kim, B.S.; Jang, J.; Cho, D.W. Recent Strategies in Extrusion-Based Three-Dimensional Cell Printing toward Organ Biofabrication. ACS Biomater. Sci. Eng. 2019, 5, 1150–1169. [Google Scholar] [CrossRef]
- Sun, W.; Starly, B.; Daly, A.C.; Burdick, J.A.; Groll, J.; Skeldon, G.; Shu, W.; Sakai, Y.; Shinohara, M.; Nishikawa, M.; et al. The bioprinting roadmap. Biofabrication 2020, 12, 22002. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.S.; Yue, K.; Aleman, J.; Mollazadeh-Moghaddam, K.; Bakht, S.M.; Yang, J.; Jia, W.; Dell’Erba, V.; Assawes, P.; Shin, S.R.; et al. 3D Bioprinting for Tissue and Organ Fabrication. Ann. Biomed. Eng. 2017, 45, 148–163. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Murphy, S.V.; De Coppi, P.; Atala, A. Opportunities and challenges of translational 3D bioprinting. Nat. Biomed. Eng. 2020, 4, 370–380. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Hu, Y.; Liu, C.; Yao, H.; Liu, B.; Mi, S. A novel strategy for creating tissue-engineered biomimetic blood vessels using 3D bioprinting technology. Materials 2018, 11, 1581. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jang, J.; Park, J.Y.; Gao, G.; Cho, D.W. Biomaterials-based 3D cell printing for next-generation therapeutics and diagnostics. Biomaterials 2018, 156, 88–106. [Google Scholar] [CrossRef]
- Hwang, D.G.; Choi, Y.M.; Jang, J. 3D Bioprinting-Based Vascularized Tissue Models Mimicking Tissue-Specific Architecture and Pathophysiology for in vitro Studies. Front. Bioeng. Biotechnol. 2021, 9, 685507. [Google Scholar] [CrossRef]
- Groll, J.; Burdick, J.A.; Cho, D.W.; Derby, B.; Gelinsky, M.; Heilshorn, S.C.; Jüngst, T.; Malda, J.; Mironov, V.A.; Nakayama, K.; et al. A definition of bioinks and their distinction from biomaterial inks. Biofabrication 2019, 11, 13001. [Google Scholar] [CrossRef]
- Lewis, P.L.; Wells, J.M. Engineering-inspired approaches to study β-cell function and diabetes. Stem Cells 2021, 39, 522–535. [Google Scholar] [CrossRef]
- Matsuda, Y. Age related morphological alterations of the pancreas. Front. Biosci. 2018, 10, 137–142. [Google Scholar] [CrossRef] [Green Version]
- Huang, G.; Greenspan, D.S. ECM roles in the function of metabolic tissues. Trends Endocrinol. Metab. 2012, 23, 16–22. [Google Scholar] [CrossRef] [Green Version]
- Kumar, S.A.; Delgado, M.; Mendez, V.E.; Joddar, B. Applications of stem cells and bioprinting for potential treatment of diabetes. World J. Stem Cells 2019, 11, 13–32. [Google Scholar] [CrossRef]
- Tran, R.; Moraes, C.; Hoesli, C.A. Controlled clustering enhances PDX1 and NKX6.1 expression in pancreatic endoderm cells derived from pluripotent stem cells. Sci. Rep. 2020, 10, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Fukuda, Y.; Akagi, T.; Asaoka, T.; Eguchi, H.; Sasaki, K.; Iwagami, Y.; Yamada, D.; Noda, T.; Kawamoto, K.; Gotoh, K.; et al. Layer-by-layer cell coating technique using extracellular matrix facilitates rapid fabrication and function of pancreatic β-cell spheroids. Biomaterials 2018, 160, 82–91. [Google Scholar] [CrossRef] [PubMed]
- Desai, T.; Shea, L.D. Advances in islet encapsulation technologies. Nat. Rev. Drug Discov. 2017, 16, 338–350. [Google Scholar] [CrossRef] [PubMed]
- Nyitray, C.E.; Chang, R.; Faleo, G.; Lance, K.D.; Bernards, D.A.; Tang, Q.; Desai, T.A. Polycaprolactone Thin-Film Micro- and Nanoporous Cell-Encapsulation Devices. ACS Nano 2015, 9, 5675–5682. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- An, D.; Wang, L.H.; Ernst, A.U.; Chiu, A.; Lu, Y.C.; Flanders, J.A.; Datta, A.K.; Ma, M. An Atmosphere-Breathing Refillable Biphasic Device for Cell Replacement Therapy. Adv. Mater. 2019, 31, 1905135. [Google Scholar] [CrossRef] [PubMed]
- Ye, S.; Boeter, J.W.B.; Penning, L.C.; Spee, B.; Schneeberger, K. Hydrogels for liver tissue engineering. Bioengineering 2019, 6, 59. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ma, X.; Yu, C.; Wang, P.; Xu, W.; Wan, X.; Lai, C.S.E.; Liu, J.; Koroleva-Maharajh, A.; Chen, S. Rapid 3D bioprinting of decellularized extracellular matrix with regionally varied mechanical properties and biomimetic microarchitecture. Biomaterials 2018, 185, 310–321. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Chae, S.; Kim, J.Y.; Han, W.; Kim, J.; Choi, Y.; Cho, D.W. Cell-printed 3D liver-on-a-chip possessing a liver microenvironment and biliary system. Biofabrication 2019, 11, 25001. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Li, W.; Dean, D.; Mishra, M.K.; Wekesa, K.S.; Saheli, M.; Sepantafar, M.; Pournasr, B.; Farzaneh, Z.; Vosough, M.; et al. Three-dimensional liver-derived extracellular matrix hydrogel promotes liver organoids function. J. Biomed. Mater. Res. Part A 2018, 119, 4320–4333. [Google Scholar]
- Wang, B.; Li, W.; Dean, D.; Mishra, M.K.; Wekesa, K.S. Enhanced hepatogenic differentiation of bone marrow derived mesenchymal stem cells on liver ECM hydrogel. J. Biomed. Mater. Res. Part A 2018, 106, 829–838. [Google Scholar] [CrossRef] [PubMed]
- Afshari, A.; Shamdani, S.; Uzan, G.; Naserian, S.; Azarpira, N. Different approaches for transformation of mesenchymal stem cells into hepatocyte-like cells. Stem Cell Res. Ther. 2020, 11, 1–14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ahmed, H.M.M.; Salerno, S.; Morelli, S.; Giorno, L.; De Bartolo, L. 3D liver membrane system by co-culturing human hepatocytes, sinusoidal endothelial and stellate cells. Biofabrication 2017, 9, 25022. [Google Scholar] [CrossRef] [PubMed]
- Baze, A.; Parmentier, C.; Hendriks, D.F.G.; Hurrell, T.; Heyd, B.; Bachellier, P.; Schuster, C.; Ingelman-Sundberg, M.; Richert, L. Three-Dimensional Spheroid Primary Human Hepatocytes in Monoculture and Coculture with Nonparenchymal Cells. Tissue Eng. Part C Methods 2018, 24, 534–545. [Google Scholar] [CrossRef] [PubMed]
- Mistriotis, P.; Andreadis, S.T. Vascular aging: Molecular mechanisms and potential treatments for vascular rejuvenation. Ageing Res. Rev. 2017, 37, 94–116. [Google Scholar] [CrossRef]
- Jeong, H.J.; Nam, H.; Jang, J.; Lee, S.J. 3D bioprinting strategies for the regeneration of functional tubular tissues and organs. Bioengineering 2020, 7, 32. [Google Scholar] [CrossRef] [Green Version]
- Campos, D.F.D.; Rohde, M.; Ross, M.; Anvari, P.; Blaeser, A.; Vogt, M.; Panfil, C.; Yam, G.H.; Mehta, J.S.; Fischer, H.; et al. Corneal bioprinting utilizing collagen-based bioinks and primary human keratocytes. J. Biomed. Mater. Res. 2019, 107A, 1945–1953. [Google Scholar] [CrossRef]
- Kang, D.; Park, J.A.; Kim, W.; Kim, S.; Lee, H.R.; Kim, W.J.; Yoo, J.Y.; Jung, S. All-Inkjet-Printed 3D Alveolar Barrier Model with Physiologically Relevant Microarchitecture. Adv. Sci. 2021, 8, 1–11. [Google Scholar] [CrossRef]
- Liu, X.; Carter, S.S.D.; Renes, M.J.; Kim, J.; Rojas-Canales, D.M.; Penko, D.; Angus, C.; Beirne, S.; Drogemuller, C.J.; Yue, Z.; et al. Development of a Coaxial 3D Printing Platform for Biofabrication of Implantable Islet-Containing Constructs. Adv. Healthc. Mater. 2019, 8, 1–12. [Google Scholar] [CrossRef] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).