Molecular Basis of Endometriosis and Endometrial Cancer: Current Knowledge and Future Perspectives
Abstract
1. Morphological Features of the Human Endometrium
2. Endometriosis
2.1. Definition, Epidemiology and Classification
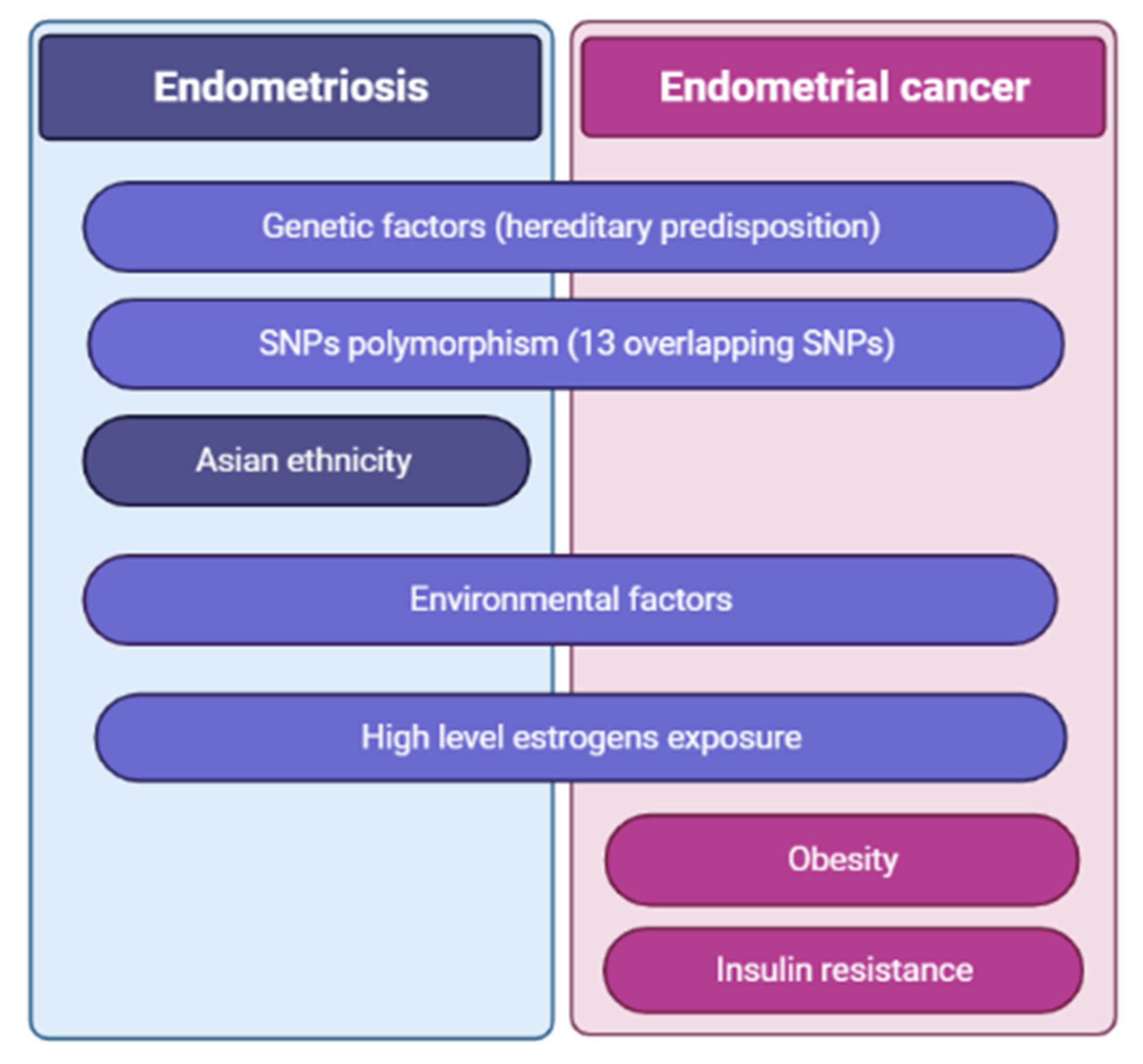
2.2. Risk Factors of Endometriosis
2.3. Pathophysiology of Endometriosis
2.4. Clinical Presentation and Diagnostic Tools
3. Endometrial Cancer
3.1. Definition, Epidemiology and Classification
3.2. Risk and Protective Factors of Endometrial Cancer
3.3. Pathophysiology of Endometrial Cancer
- Cowden syndrome: Cowden syndrome is an autosomal dominant syndrome characterized by PTEN mutations. It is associated with a 19% to 28% risk of EC by age 70 [90].
3.4. Clinical Presentation and Diagnostic Tools
4. Molecular Basis of Endometriosis and Endometrial Pathology
4.1. Genetic and Epigenetic Changes in Endometriosis
4.1.1. Genetic Association and Meta-Analyses Studies
4.1.2. Genome Mapping Studies and Targeted Gene Sequencing
4.1.3. Genome-Wide Sequencing Studies
4.2. Endometrial Stem Cells in Pathogenesis of Endometrial Pathology
4.3. Endometriosis as a Risk Factor for Endometrial Cancer
5. Clinical Applications of Current Knowledge and Directions for Future Research
5.1. Molecular Basis for a Specific Therapeutic Approach
5.2. Prognostic Biomarkers for Endometrial Cancer
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Garry, R.; Hart, R.; Karthigasu, K.A.; Burke, C. A re-appraisal of the morphological changes within the endometrium during menstruation: A hysteroscopic, histological and scanning electron microscopic study. Hum. Reprod. 2009, 24, 1393–1401. [Google Scholar] [CrossRef]
- Emera, D.; Wagner, G. Transposable element recruitments in the mammalian placenta: Impacts and mechanisms. Brief. Funct. Genom. 2012, 11, 267–276. [Google Scholar] [CrossRef]
- Amant, F.; Moerman, P.; Neven, P.; Timmerman, D.; Van Limbergen, E.; Vergote, I. Endometrial cancer. Lancet 2005, 366, 491–505. [Google Scholar] [CrossRef]
- Yamaguchi, M.; Yoshihara, K.; Suda, K.; Nakaoka, H.; Yachida, N.; Ueda, H.; Sugino, K.; Mori, Y.; Yamawaki, K.; Tamura, R.; et al. Three-dimensional understanding of the morphological complexity of the human uterine endometrium. iScience 2021, 24, 102258. [Google Scholar] [CrossRef]
- Cooke, P.S.; Spencer, T.; Bartol, F.F.; Hayashi, K. Uterine glands: Development, function and experimental model systems. Mol. Hum. Reprod. 2013, 19, 547–558. [Google Scholar] [CrossRef]
- Gray, C.A.; Bartol, F.F.; Tarleton, B.J.; Wiley, A.A.; Johnson, G.A.; Bazer, F.W.; Spencer, T.E. Developmental Biology of Uterine Glands1. Biol. Reprod. 2001, 65, 1311–1323. [Google Scholar] [CrossRef] [PubMed]
- Kyo, S.; Maida, Y.; Inoue, M. Stem cells in endometrium and endometrial cancer: Accumulating evidence and unresolved questions. Cancer Lett. 2011, 308, 123–133. [Google Scholar] [CrossRef]
- Miyazaki, K.; Maruyama, T.; Masuda, H.; Yamasaki, A.; Uchida, S.; Oda, H.; Uchida, H.; Yoshimura, Y. Stem Cell-Like Differentiation Potentials of Endometrial Side Population Cells as Revealed by a Newly Developed In Vivo Endometrial Stem Cell Assay. PLoS ONE 2012, 7, e50749. [Google Scholar] [CrossRef]
- Prianishnikov, V.A. A functional model of the structure of the epithelium of normal, hyperplastic, and malignant human endometrium: A review. Gynecol. Oncol. 1978, 6, 420–428. [Google Scholar] [CrossRef]
- Garry, R.; Hart, R.; Karthigasu, K.; Burke, C. Structural changes in endometrial basal glands during menstruation. BJOG Int. J. Obstet. Gynaecol. 2010, 117, 1175–1185. [Google Scholar] [CrossRef]
- Gellersen, B.; Brosens, J. Cyclic Decidualization of the Human Endometrium in Reproductive Health and Failure. Endocr. Rev. 2014, 35, 851–905. [Google Scholar] [CrossRef]
- Santamaria, X.; Mas, A.; Cervelló, I.; Taylor, H.; Simón, C. Uterine stem cells: From basic research to advanced cell therapies. Hum. Reprod. Updat. 2018, 24, 673–693. [Google Scholar] [CrossRef]
- E McLennan, C.; Rydell, A.H. Extent of endometrial shedding during normal menstruation. Obstet. Gynecol. 1965, 26, 605–621. [Google Scholar]
- Filho, J.H.; Cedenho, A.P.; De Freitas, V. Correlation between endometrial dating of luteal phase days 6 and 10 of the same menstrual cycle. Sao Paulo Med. J. 1998, 116, 1734–1737. [Google Scholar] [CrossRef][Green Version]
- Johannisson, E.; Parker, R.A.; Landgren, B.-M.; Diczfalusy, E. Morphometric analysis of the human endometrium in relation to peripheral hormone levels. Fertil. Steril. 1982, 38, 564–571. [Google Scholar] [CrossRef]
- García-Solares, J.; Donnez, J.; Donnez, O.; Dolmans, M.-M. Pathogenesis of uterine adenomyosis: Invagination or metaplasia? Fertil. Steril. 2018, 109, 371–379. [Google Scholar] [CrossRef] [PubMed]
- Koninckx, P.R.; Ussia, A.; Adamyan, L.; Wattiez, A.; Gomel, V.; Martin, D.C. Pathogenesis of endometriosis: The genetic/epigenetic theory. Fertil. Steril. 2019, 111, 327–340. [Google Scholar] [CrossRef] [PubMed]
- Morice, P.; Leary, A.; Creutzberg, C.; Abu-Rustum, N.; Darai, E. Endometrial cancer. Lancet 2016, 387, 1094–1108. [Google Scholar] [CrossRef]
- Matalliotaki, C.; Matalliotakis, M.; Zervou, M.I.; Patelarou, A.; Koliarakis, I.; Spandidos, D.A.; Arici, A.; Matalliotakis, I.; Goulielmos, G.N. Epidemiological aspects of the outcomes from the treatment of endometriosis: Experience from two different geographical areas. Exp. Ther. Med. 2019, 19, 1079–1083. [Google Scholar] [CrossRef]
- Johnatty, S.E.; Tan, Y.; Buchanan, D.D.; Bowman, M.; Walters, R.J.; Obermair, A.; Quinn, M.A.; Blomfield, P.B.; Brand, A.; Leung, Y.; et al. Family history of cancer predicts endometrial cancer risk independently of Lynch Syndrome: Implications for genetic counselling. Gynecol. Oncol. 2017, 147, 381–387. [Google Scholar] [CrossRef]
- Laganà, A.S.; Vitale, S.G.; Salmeri, F.M.; Triolo, O.; Frangež, H.B.; Vrtačnik-Bokal, E.; Stojanovska, L.; Apostolopoulos, V.; Granese, R.; Sofo, V. Unus pro omnibus, omnes pro uno: A novel, evidence-based, unifying theory for the pathogenesis of endometriosis. Med. Hypotheses 2017, 103, 10–20. [Google Scholar] [CrossRef] [PubMed]
- Raffaelli, R.; Garzon, S.; Baggio, S.; Genna, M.; Pomini, P.; Laganà, A.S.; Ghezzi, F.; Franchi, M. Mesenteric vascular and nerve sparing surgery in laparoscopic segmental intestinal resection for deep infiltrating endometriosis. Eur. J. Obstet. Gynecol. Reprod. Biol. 2018, 231, 214–219. [Google Scholar] [CrossRef]
- Bulun, S.E. Endometriosis. N. Engl. J. Med. 2009, 360, 268–279. [Google Scholar] [CrossRef]
- E Bulun, S.; Yilmaz, B.D.; Sison, C.; Miyazaki, K.; Bernardi, L.; Liu, S.; Kohlmeier, A.; Yin, P.; Milad, M.; Wei, J. Endometriosis. Endocr. Rev. 2019, 40, 1048–1079. [Google Scholar] [CrossRef] [PubMed]
- Falcone, T.; Flyckt, R. Clinical Management of Endometriosis. Obstet. Gynecol. 2018, 131, 557–571. [Google Scholar] [CrossRef] [PubMed]
- Terzic, M.; Aimagambetova, G.; Garzon, S.; Bapayeva, G.; Ukybassova, T.; Terzic, S.; Norton, M.; Laganà, A.S. Ovulation induction in infertile women with endometriotic ovarian cysts: Current evidence and potential pitfalls. Minerva Med. 2020, 111, 50–61. [Google Scholar] [CrossRef]
- Hemmert, R.; Schliep, K.C.; Willis, S.; Peterson, C.M.; Louis, G.B.; Allen-Brady, K.; Simonsen, S.E.; Stanford, J.B.; Byun, J.; Smith, K.R. Modifiable life style factors and risk for incident endometriosis. Paediatr. Périnat. Epidemiol. 2019, 33, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Shafrir, A.; Farland, L.; Shah, D.; Harris, H.; Kvaskoff, M.; Zondervan, K.; Missmer, S. Risk for and consequences of endometriosis: A critical epidemiologic review. Best Pr. Res. Clin. Obstet. Gynaecol. 2018, 51, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Adam, A.M.; Altalhi, T.A.; El-Megharbel, S.M.; Saad, H.A.; Refat, M.S. Using a Modified Polyamidoamine Fluorescent Dendrimer for Capturing Environment Polluting Metal Ions Zn2+, Cd2+, and Hg2+: Synthesis and Characterizations. Crystals 2021, 11, 92. [Google Scholar] [CrossRef]
- Capezzuoli, T.; Clemenza, S.; Sorbi, F.; Campana, D.; Vannuccini, S.; Chapron, C.; Petraglia, F. Classification/staging systems for endometriosis: The state of the art. Gynecol. Reprod. Endocrinol. Metab. 2020, 1, 14–22. [Google Scholar]
- Canis, M.; Donnez, J.G.; Guzick, D.S.; Halme, J.K.; Rock, J.A.; Schenken, R.S.; Vernon, M.W. Revised American Society for Reproductive Medicine classification of endometriosis: 1996. Fertil. Steril. 1997, 67, 817–821. [Google Scholar] [CrossRef]
- Vercellini, P.; Viganò, P.; Somigliana, E.; Fedele, L. Endometriosis: Pathogenesis and treatment. Nat. Rev. Endocrinol. 2014, 10, 261–275. [Google Scholar] [CrossRef] [PubMed]
- Laganà, A.S.; Vitale, S.G.; Trovato, M.A.; Palmara, V.I.; Rapisarda, A.M.C.; Granese, R.; Sturlese, E.; De Dominici, R.; Alecci, S.; Padula, F.; et al. Full-Thickness Excision versus Shaving by Laparoscopy for Intestinal Deep Infiltrating Endometriosis: Rationale and Potential Treatment Options. BioMed Res. Int. 2016, 2016, 3617179. [Google Scholar] [CrossRef] [PubMed]
- Mehedintu, C.; Plotogea, M.N.; Ionescu, S.; Antonovici, M. Endometriosis still a challenge. J. Med. Life 2014, 7, 349–357. [Google Scholar] [PubMed]
- Maruyama, S.; Imanaka, S.; Nagayasu, M.; Kimura, M.; Kobayashi, H. Relationship between adenomyosis and endometriosis; Different phenotypes of a single disease? Eur. J. Obstet. Gynecol. Reprod. Biol. 2020, 253, 191–197. [Google Scholar] [CrossRef]
- Tosti, C.; Pinzauti, S.; Santulli, P.; Chapron, C.; Petraglia, F. Pathogenetic Mechanisms of Deep Infiltrating Endometriosis. Reprod. Sci. 2015, 22, 1053–1059. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.-W. The Pathogenesis of Adenomyosis vis-à-vis Endometriosis. J. Clin. Med. 2020, 9, 485. [Google Scholar] [CrossRef]
- Chapron, C.; Tosti, C.; Marcellin, L.; Bourdon, M.; Lafay-Pillet, M.-C.; Millischer, A.-E.; Streuli, I.; Borghese, B.; Petraglia, F.; Santulli, P. Relationship between the magnetic resonance imaging appearance of adenomyosis and endometriosis phenotypes. Hum. Reprod. 2017, 32, 1393–1401. [Google Scholar] [CrossRef]
- Rolla, E. Endometriosis: Advances and controversies in classification, pathogenesis, diagnosis, and treatment. F1000Research 2019, 8, 529. [Google Scholar] [CrossRef]
- Vitale, S.G.; La Rosa, V.L.; Rapisarda, A.M.C.; Laganà, A.S. Impact of endometriosis on quality of life and psychological well-being. J. Psychosom. Obstet. Gynecol. 2016, 38, 317–319. [Google Scholar] [CrossRef]
- Brasil, D.L.; Montagna, E.; Trevisan, C.M.; La Rosa, V.L.; Laganà, A.S.; Barbosa, C.P.; Bianco, B.; Zaia, V. Psychological stress levels in women with endometriosis: Systematic review and meta-analysis of observational studies. Minerva Med. 2020, 111, 90–102. [Google Scholar] [CrossRef]
- Abrao, M.S.; Miller, C.E. An endometriosis classification, designed to be validated. NewsScope 2012, 25, 6. [Google Scholar]
- Dai, Y.; Li, X.; Shi, J.; Leng, J. A review of the risk factors, genetics and treatment of endometriosis in Chinese women: A comparative update. Reprod. Health 2018, 15, 82. [Google Scholar] [CrossRef] [PubMed]
- Baranov, V.S.; Ivaschenko, T.E.; Liehr, T.; Yarmolinskaya, M.I. Systems genetics view of endometriosis: A common complex disorder. Eur. J. Obstet. Gynecol. Reprod. Biol. 2015, 185, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Laganà, A.S.; Garzon, S.; Götte, M.; Viganò, P.; Franchi, M.; Ghezzi, F.; Martin, D.C. The Pathogenesis of Endometriosis: Molecular and Cell Biology Insights. Int. J. Mol. Sci. 2019, 20, 5615. [Google Scholar] [CrossRef]
- Williams, C.; Long, A.J.; Noga, H.; Allaire, C.; Bedaiwy, M.A.; Lisonkova, S.; Yong, P.J. East and South East Asian Ethnicity and Moderate-to-Severe Endometriosis. J. Minim. Invasive Gynecol. 2018, 26, 507–515. [Google Scholar] [CrossRef] [PubMed]
- Missmer, S.A.; Hankinson, S.E.; Spiegelman, D.; Barbieri, R.L.; Marshall, L.M.; Hunter, D.J. Incidence of Laparoscopically Confirmed Endometriosis by Demographic, Anthropometric, and Lifestyle Factors. Am. J. Epidemiol. 2004, 160, 784–796. [Google Scholar] [CrossRef] [PubMed]
- McLeod, B.S.; Retzloff, M.G. Epidemiology of endometriosis: An assessment of risk factors. Clin. Obstet. Gynecol. 2010, 53, 389–396. [Google Scholar] [CrossRef] [PubMed]
- Schliep, K.C.; Schisterman, E.; Mumford, S.; Pollack, A.; Zhang, C.; Ye, A.; Stanford, J.B.; O Hammoud, A.; Porucznik, C.; Wactawski-Wende, J. Caffeinated beverage intake and reproductive hormones among premenopausal women in the BioCycle Study. Am. J. Clin. Nutr. 2012, 95, 488–497. [Google Scholar] [CrossRef]
- Fiuza-Luces, C.; Garatachea, N.; Berger, N.A.; Lucia, A. Exercise is the Real Polypill. Physiology 2013, 28, 330–358. [Google Scholar] [CrossRef]
- Sofo, V.; Götte, M.; Laganà, A.S.; Salmeri, F.M.; Triolo, O.; Sturlese, E.; Retto, G.; Alfa, M.; Granese, R.; Abrão, M.S. Correlation between dioxin and endometriosis: An epigenetic route to unravel the pathogenesis of the disease. Arch. Gynecol. Obstet. 2015, 292, 973–986. [Google Scholar] [CrossRef] [PubMed]
- Painter, J.N.; O’Mara, T.A.; Morris, A.P.; Cheng, T.H.T.; Gorman, M.; Martin, L.; Hodson, S.; Jones, A.; Martin, N.G.; Gordon, S.; et al. Genetic overlap between endometriosis and endometrial cancer: Evidence from cross-disease genetic correlation and GWAS meta-analyses. Cancer Med. 2018, 7, 1978–1987. [Google Scholar] [CrossRef] [PubMed]
- Burney, R.O.; Giudice, L.C. Pathogenesis and pathophysiology of endometriosis. Fertil. Steril. 2012, 98, 511–519. [Google Scholar] [CrossRef]
- Coxon, L.; Horne, A.W.; Vincent, K. Pathophysiology of endometriosis-associated pain: A review of pelvic and central nervous system mechanisms. Best Pr. Res. Clin. Obstet. Gynaecol. 2018, 51, 53–67. [Google Scholar] [CrossRef]
- Sasson, I.E.; Taylor, H.S. Stem Cells and the Pathogenesis of Endometriosis. Ann. N. Y. Acad. Sci. 2008, 1127, 106–115. [Google Scholar] [CrossRef]
- Poli-Neto, O.B.; Meola, J.; Rosa-E-Silva, J.C.; Tiezzi, D. Transcriptome meta-analysis reveals differences of immune profile between eutopic endometrium from stage I-II and III-IV endometriosis independently of hormonal milieu. Sci. Rep. 2020, 10, 313. [Google Scholar] [CrossRef] [PubMed]
- Cousins, F.L.; Dorien, F.O.; Gargett, C.E. Endometrial stem/progenitor cells and their role in the pathogenesis of endometriosis. Best Pract. Res. Clin. Obstet. Gynaecol. 2018, 50, 27–38. [Google Scholar] [CrossRef] [PubMed]
- Vetvicka, V.; Laganà, A.S.; Salmeri, F.M.; Triolo, O.; Palmara, V.I.; Vitale, S.G.; Sofo, V.; Kralickova, M. Regulation of apoptotic pathways during endometriosis: From the molecular basis to the future perspectives. Arch. Gynecol. Obstet. 2016, 294, 897–904. [Google Scholar] [CrossRef] [PubMed]
- Salmeri, F.M.; Laganà, A.S.; Sofo, V.; Triolo, O.; Sturlese, E.; Retto, G.; Pizzo, A.; D’Ascola, A.; Campo, S. Behavior of Tumor Necrosis Factor-α and Tumor Necrosis Factor Receptor 1/Tumor Necrosis Factor Receptor 2 System in Mononuclear Cells Recovered From Peritoneal Fluid of Women With Endometriosis at Different Stages. Reprod. Sci. 2014, 22, 165–172. [Google Scholar] [CrossRef] [PubMed]
- Sturlese, E.; Salmeri, F.M.; Retto, G.; Pizzo, A.; De Dominici, R.; Ardita, F.V.; Borrielli, I.; Licata, N.; Laganà, A.S.; Sofo, V. Dysregulation of the Fas/FasL system in mononuclear cells recovered from peritoneal fluid of women with endometriosis. J. Reprod. Immunol. 2011, 92, 74–81. [Google Scholar] [CrossRef]
- Patel, B.G.; Lenk, E.E.; Lebovic, D.I.; Shu, Y.; Yu, J.; Taylor, R.N. Pathogenesis of endometriosis: Interaction between Endocrine and inflammatory pathways. Best Pract. Res. Clin. Obstet. Gynaecol. 2018, 50, 50–60. [Google Scholar] [CrossRef] [PubMed]
- Chapron, C.; Marcellin, L.; Borghese, B.; Santulli, P. Rethinking mechanisms, diagnosis and management of endometriosis. Nat. Rev. Endocrinol. 2019, 15, 666–682. [Google Scholar] [CrossRef] [PubMed]
- Terzic, M.; Aimagambetova, G.; Norton, M.; Della Corte, L.; Marín-Buck, A.; Lisón, J.F.; Amer-Cuenca, J.J.; Zito, G.; Garzon, S.; Caruso, S.; et al. Scoring systems for the evaluation of adnexal masses nature: Current knowledge and clinical applications. J. Obstet. Gynaecol. 2021, 41, 340–347. [Google Scholar] [CrossRef] [PubMed]
- Terzic, M.; Rapisarda, A.M.C.; Della Corte, L.; Manchanda, R.; Aimagambetova, G.; Norton, M.; Garzon, S.; Riemma, G.; King, C.R.; Chiofalo, B.; et al. Diagnostic work-up in paediatric and adolescent patients with adnexal masses: An evidence-based approach. J. Obstet. Gynaecol. 2021, 41, 503–515. [Google Scholar] [CrossRef]
- Giudice, L.C. Clinical practice. Endometriosis. N. Engl. J. Med. 2010, 362, 2389–2398. [Google Scholar] [CrossRef]
- DiVasta, A.D.; Vitonis, A.F.; Laufer, M.R.; Missmer, S.A. Spectrum of symptoms in women diagnosed with endometriosis during adolescence vs adulthood. Am. J. Obstet. Gynecol. 2018, 218, 324.e1–324.e11. [Google Scholar] [CrossRef]
- D’Alterio, M.N.; Saponara, S.; D’Ancona, G.; Russo, M.; Laganà, A.S.; Sorrentino, F.; Nappi, L.; Angioni, S. Role of surgical treatment in endometriosis. Minerva Obstet. Gynecol. 2021, 73, 317–332. [Google Scholar] [CrossRef]
- Chapron, C.; Santulli, P.; De Ziegler, D.; Noel, J.-C.; Anaf, V.; Streuli, I.; Foulot, H.; Souza, C.; Borghese, B. Ovarian endometrioma: Severe pelvic pain is associated with deeply infiltrating endometriosis. Hum. Reprod. 2012, 27, 702–711. [Google Scholar] [CrossRef]
- Tanbo, T.; Fedorcsak, P. Endometriosis-associated infertility: Aspects of pathophysiological mechanisms and treatment options. Acta Obstet. Gynecol. Scand. 2017, 96, 659–667. [Google Scholar] [CrossRef] [PubMed]
- Terzic, M.; Dotlic, J.; Brndusic, N.; Likic, I.; Andrijasevic, S.; Arsenovic, N.; Ladjevic, N.; Maričić, S. Histopathological diagnoses of adnexal masses: Which parameters are relevant in preoperative assessment? Ginekol. Polska 2013, 84, 700–708. [Google Scholar] [CrossRef]
- Terzic, M.; Dotlic, J.; Likic, I.; Nikolic, B.; Brndusic, N.; Pilic, I.; Bila, J.; Maricic, S.; Arsenovic, N. Diagnostic value of serum tumor markers evaluation for adnexal masses. Open Med. 2014, 9, 210–216. [Google Scholar] [CrossRef]
- Bazot, M.; Daraï, E. Diagnosis of deep endometriosis: Clinical examination, ultrasonography, magnetic resonance imaging, and other techniques. Fertil. Steril. 2017, 108, 886–894. [Google Scholar] [CrossRef]
- Scioscia, M.; Virgilio, B.A.; Laganà, A.S.; Bernardini, T.; Fattizzi, N.; Neri, M.; Guerriero, S. Differential Diagnosis of Endometriosis by Ultrasound: A Rising Challenge. Diagnostics 2020, 10, 848. [Google Scholar] [CrossRef]
- Djokovic, D.; Pinto, A.P.; van Herendael, B.J.; Laganà, A.S.; Thomas, V.; Keckstein, J. Structured report for dynamic ultrasonography in patients with suspected or known endometriosis: Recommendations of the International Society for Gynecologic Endoscopy (ISGE). Eur. J. Obstet. Gynecol. Reprod. Biol. 2021, 263, 252–260. [Google Scholar] [CrossRef] [PubMed]
- Barra, F.; Biscaldi, E.; Scala, C.; Laganà, A.S.; Vellone, V.G.; Stabilini, C.; Ghezzi, F.; Ferrero, S. A Prospective Study Comparing Three-Dimensional Rectal Water Contrast Transvaginal Ultrasonography and Computed Tomographic Colonography in the Diagnosis of Rectosigmoid Endometriosis. Diagnostics 2020, 10, 252. [Google Scholar] [CrossRef] [PubMed]
- Noventa, M.; Scioscia, M.; Schincariol, M.; Cavallin, F.; Pontrelli, G.; Virgilio, B.; Vitale, S.G.; Laganà, A.S.; Dessole, F.; Cosmi, E.; et al. Imaging Modalities for Diagnosis of Deep Pelvic Endometriosis: Comparison between Trans-Vaginal Sonography, Rectal Endoscopy Sonography and Magnetic Resonance Imaging. A Head-to-Head Meta-Analysis. Diagnostics 2019, 9, 225. [Google Scholar] [CrossRef]
- Nisenblat, V.; Bossuyt, P.M.M.; Farquhar, C.; Johnson, N.; Hull, M.L. Imaging modalities for the non-invasive diagnosis of endometriosis. Cochrane Database Syst. Rev. 2016, 2, CD009591. [Google Scholar] [CrossRef] [PubMed]
- Raglan, O.; Kalliala, I.; Markozannes, G.; Cividini, S.; Gunter, M.J.; Nautiyal, J.; Gabra, H.; Paraskevaidis, E.; Martin-Hirsch, P.; Tsilidis, K.K.; et al. Risk factors for endometrial cancer: An umbrella review of the literature. Int. J. Cancer 2019, 145, 1719–1730. [Google Scholar] [CrossRef]
- Terzic, M.; Norton, M.; Terzic, S.; Bapayeva, G.; Aimagambetova, G. Fertility preservation in endometrial cancer patients: Options, challenges and perspectives. Ecancermedicalscience 2020, 14, 1030. [Google Scholar] [CrossRef]
- Lortet-Tieulent, J.; Ferlay, J.; Bray, F.; Jemal, A. International Patterns and Trends in Endometrial Cancer Incidence, 1978–2013. J. Natl. Cancer Inst. 2017, 110, 354–361. [Google Scholar] [CrossRef]
- Moore, K.; Brewer, M.A. Endometrial Cancer: Is This a New Disease? Am. Soc. Clin. Oncol. Educ. Book 2017, 37, 435–442. [Google Scholar] [CrossRef]
- American Cancer Society. Facts & Figures 2021; American Cancer Society: Atlanta, GA, USA, 2021; Available online: https://www.cancer.org/cancer/endometrial-cancer.html (accessed on 2 July 2021).
- Urick, M.E.; Bell, D.W. Clinical actionability of molecular targets in endometrial cancer. Nat. Rev. Cancer 2019, 19, 510–521. [Google Scholar] [CrossRef]
- Terzic, M.; Aimagambetova, G.; Terzic, S.; Norton, M.; Bapayeva, G.; Garzon, S. Current role of Pipelle endometrial sampling in early diagnosis of endometrial cancer. Transl. Cancer Res. 2020, 9, 7716–7724. [Google Scholar] [CrossRef]
- Braun, M.M.; Overbeek-Wager, E.A.; Grumbo, R.J. Diagnosis and Management of Endometrial Cancer. Am. Fam. Phys. 2016, 93, 468–474. [Google Scholar]
- Vitale, S.G.; Capriglione, S.; Zito, G.; Lopez, S.; Gulino, F.A.; Di Guardo, F.; Vitagliano, A.; Noventa, M.; La Rosa, V.L.; Sapia, F.; et al. Management of endometrial, ovarian and cervical cancer in the elderly: Current approach to a challenging condition. Arch. Gynecol. Obstet. 2018, 299, 299–315. [Google Scholar] [CrossRef]
- Howlader, N.N.A.K.M.; Noone, A.M.; Krapcho, M.; Garshell, J.; Miller, D.; Altekruse, S.F.; Kosary, C.L.; Yu, M.; Ruhl, J.; Tatalovich, Z.; et al. SEER Cancer Statistics Review, 1975–2015; National Cancer Institute: Bethesda, MD, USA, 2015. Available online: https://seer.cancer.gov/csr/1975_2015/ (accessed on 2 July 2021).
- Lu, K.H.; Broaddus, R.R. Endometrial Cancer. New Engl. J. Med. 2020, 383, 2053–2064. [Google Scholar] [CrossRef] [PubMed]
- Benati, M.; Montagnana, M.; Danese, E.; Mazzon, M.; Paviati, E.; Garzon, S.; Laganà, A.S.; Casarin, J.; Giudici, S.; Raffaelli, R.; et al. Aberrant Telomere Length in Circulating Cell-Free DNA as Possible Blood Biomarker with High Diagnostic Performance in Endometrial Cancer. Pathol. Oncol. Res. 2020, 26, 2281–2289. [Google Scholar] [CrossRef]
- Passarello, K.; Kurian, S.; Villanueva, V. Endometrial Cancer: An Overview of Pathophysiology, Management, and Care. Semin. Oncol. Nurs. 2019, 35, 157–165. [Google Scholar] [CrossRef]
- Chiofalo, B.; Mazzon, I.; Antonio, S.D.A.; Amadore, D.; Vizza, E.; Laganà, A.S.; Vocaturo, G.; Calagna, G.; Favilli, A.; Palmara, V.; et al. Hysteroscopic Evaluation of Endometrial Changes in Breast Cancer Women with or without Hormone Therapies: Results from a Large Multicenter Cohort Study. J. Minim. Invasive Gynecol. 2020, 27, 832–839. [Google Scholar] [CrossRef] [PubMed]
- Lindemann, K.; Vatten, L.J.; Ellstrøm-Engh, M.; Eskild, A. Body mass, diabetes and smoking, and endometrial cancer risk: A follow-up study. Br. J. Cancer 2008, 98, 1582–1585. [Google Scholar] [CrossRef] [PubMed]
- Njoku, K.; Campbell, A.; Geary, B.; MacKintosh, M.; Derbyshire, A.; Kitson, S.; Sivalingam, V.; Pierce, A.; Whetton, A.; Crosbie, E. Metabolomic Biomarkers for the Detection of Obesity-Driven Endometrial Cancer. Cancers 2021, 13, 718. [Google Scholar] [CrossRef]
- Renehan, A.G.; Tyson, M.; Egger, M.; Heller, R.F.; Zwahlen, M. Body-mass index and incidence of cancer: A systematic review and meta-analysis of prospective observational studies. Lancet 2008, 371, 569–578. [Google Scholar] [CrossRef]
- Kalliala, I.; Markozannes, G.; Gunter, M.J.; Paraskevaidis, E.; Gabra, H.; Mitra, A.; Terzidou, V.; Bennett, P.; Martin-Hirsch, P.; Tsilidis, K.K.; et al. Obesity and gynaecological and obstetric conditions: Umbrella review of the literature. BMJ 2017, 359, j4511. [Google Scholar] [CrossRef] [PubMed]
- Kyrgiou, M.; Kalliala, I.; Markozannes, G.; Gunter, M.J.; Paraskevaidis, E.; Gabra, H.; Martin-Hirsch, P.; Tsilidis, K.K. Adiposity and cancer at major anatomical sites: Umbrella review of the literature. BMJ 2017, 356, j477. [Google Scholar] [CrossRef]
- Tsilidis, K.K.; Kasimis, J.C.; Lopez, D.S.; E Ntzani, E.; Ioannidis, J.P.A. Type 2 diabetes and cancer: Umbrella review of meta-analyses of observational studies. BMJ 2015, 350, g7607. [Google Scholar] [CrossRef] [PubMed]
- Nead, K.T.; Sharp, S.J.; Thompson, D.J.; Painter, J.N.; Savage, D.B.; Semple, R.; Barker, A.; Perry, J.R.B.; Attia, J.; Dunning, A.M.; et al. Evidence of a Causal Association Between Insulinemia and Endometrial Cancer: A Mendelian Randomization Analysis. J. Natl. Cancer Inst. 2015, 107, 178. [Google Scholar] [CrossRef] [PubMed]
- Collaborative Group on Epidemiological Studies on Endometrial Cancer. Endometrial cancer and oral contraceptives: An in-dividual participant meta-analysis of 27,276 women with endometrial cancer from 36 epidemiological studies. Lancet Oncol. 2015, 16, 1061–1070. [Google Scholar] [CrossRef]
- Wang, A.; Wang, S.; Zhu, C.; Huang, H.; Wu, L.; Wan, X.; Yang, X.; Zhang, H.; Miao, R.; He, L.; et al. Coffee and cancer risk: A meta-analysis of prospective observational studies. Sci. Rep. 2016, 6, 33711. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Q.; Luo, M.-L.; Li, H.; Li, M.; Zhou, J.-G. Coffee consumption and risk of endometrial cancer: A dose-response meta-analysis of prospective cohort studies. Sci. Rep. 2015, 5, 13410. [Google Scholar] [CrossRef]
- Al-Zoughool, M.; Dossus, L.; Kaaks, R.; Clavel-Chapelon, F.; Tjønneland, A.; Olsen, A.; Overvad, K.; Boutron-Ruault, M.-C.; Gauthier, E.; Linseisen, J.; et al. Risk of endometrial cancer in relationship to cigarette smoking: Results from the EPIC study. Int. J. Cancer 2007, 121, 2741–2747. [Google Scholar] [CrossRef] [PubMed]
- Setiawan, V.W.; Pike, M.C.; Kolonel, L.N.; Nomura, A.M.; Goodman, M.T.; Henderson, B.E. Racial/Ethnic Differences in Endometrial Cancer Risk: The Multiethnic Cohort Study. Am. J. Epidemiol. 2006, 165, 262–270. [Google Scholar] [CrossRef]
- Arend, R.C.; Jones, B.A.; Martinez, A.; Goodfellow, P. Endometrial cancer: Molecular markers and management of advanced stage disease. Gynecol. Oncol. 2018, 150, 569–580. [Google Scholar] [CrossRef] [PubMed]
- Hutt, S.; Tailor, A.; Ellis, P.; Michael, A.; Butler-Manuel, S.; Chatterjee, J. The role of biomarkers in endometrial cancer and hyperplasia: A literature review. Acta Oncol. 2019, 58, 342–352. [Google Scholar] [CrossRef] [PubMed]
- Aimagambetova, G.; Kaiyrlykyzy, A.; Bapayeva, G.; Ukybassova, T.; Kenbayeva, K.; Ibrayimov, B.; Lyasova, A.; Bonaldo, G.; Buzzaccarini, G.; Noventa, M.; et al. Validation of Pipelle endometrial biopsy in patients with abnormal uterine bleeding in Kazakhstani healthcare setting. Clin. Exp. Obstet. Gynecol. 2021, 48, 670–675. [Google Scholar] [CrossRef]
- Terzic, M.M.; Aimagambetova, G.; Terzic, S.; Norton, M.; Bapayeva, G.; Garzon, S. Pipelle endometrial sampling success rates in Kazakhstani settings: Results from a prospective cohort analysis. J. Obstet. Gyn. 2021. [Google Scholar] [CrossRef]
- Scioscia, M.; Noventa, M.; Laganà, A.S. Abnormal uterine bleeding and the risk of endometrial cancer: Can subendometrial vascular ultrasound be of help to discriminate cancer from adenomyosis? Am. J. Obstet. Gynecol. 2020, 223, 605–606. [Google Scholar] [CrossRef]
- Laganà, A.S.; Scioscia, M. Endometrial Cancer in Women with Adenomyosis: An Underestimated Risk? Int. J. Fertil. Steril. 2020, 14, 260–261. [Google Scholar]
- Stachowicz, N.; Smoleń, A.; Ciebiera, M.; Łoziński, T.; Poziemski, P.; Borowski, D.; Czekierdowski, A. Risk Assessment of Endometrial Hyperplasia or Endometrial Cancer with Simplified Ultrasound-Based Scoring Systems. Diagnostics 2021, 11, 442. [Google Scholar] [CrossRef]
- Will, A.J.; Sanchack, K.E. Endometrial Biopsy. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2021. [Google Scholar]
- Costas, T.; Belda, R.; Alcazar, J.L. Transvaginal three-dimensional ultrasound for preoperative assessment of myometrial invasion in patients with endometrial cancer: A systematic review and meta-analysis. Med. Ultrason. 2021. [Google Scholar] [CrossRef]
- Capozzi, V.A.; Rosati, A.; Rumolo, V.; Ferrari, F.; Gullo, G.; Karaman, E.; Karaaslan, O.; Hacioğlu, L. Novelties of ultrasound imaging for endometrial cancer preoperative workup. Minerva Med. 2021, 112, 3–11. [Google Scholar] [CrossRef] [PubMed]
- Zondervan, K.T.; Becker, C.M.; Koga, K.; Missmer, S.A.; Taylor, R.N.; Viganò, P. Endometriosis. Nat. Rev. Dis. Primers 2018, 4, 9. [Google Scholar] [CrossRef] [PubMed]
- Zondervan, K.T.; Becker, C.M.; Missmer, S.A. Endometriosis. New Engl. J. Med. 2020, 382, 1244–1256. [Google Scholar] [CrossRef] [PubMed]
- Gardner, G.H.; Greene, R.R.; Ranney, B. The histogenesis of endometriosis; recent contributions. Obstet. Gynecol. 1953, 1, 615–637. [Google Scholar]
- Simpson, J.L.; Elias, S.; Malinak, L.; Buttram, V.C. Heritable aspects of endometriosis. Am. J. Obstet. Gynecol. 1980, 137, 327–331. [Google Scholar] [CrossRef]
- Parasar, P.; Ozcan, P.; Terry, K.L. Endometriosis: Epidemiology, Diagnosis and Clinical Management. Curr. Obstet. Gynecol. Rep. 2017, 6, 34–41. [Google Scholar] [CrossRef]
- Frey, G. The Familial Occurrence of Endometriosis. Am. J. Obstet. Gynecol. 1957, 73, 418–421. [Google Scholar] [CrossRef]
- Kennedy, S.; Hadfield, R.; Mardon, H.; Barlow, D. Age of onset of pain symptoms in non-twin sisters concordant for endometriosis. Hum. Reprod. 1996, 11, 403–405. [Google Scholar] [CrossRef] [PubMed][Green Version]
- A Treloar, S.; O’Connor, D.T.; O’Connor, V.M.; Martin, N. Genetic influences on endometriosis in an Australian twin sample. Fertil. Steril. 1999, 71, 701–710. [Google Scholar] [CrossRef]
- Stefansson, H.; Geirsson, R.; Steinthorsdottir, V.; Jonsson, H.; Manolescu, A.; Kong, A.; Ingadottir, G.; Gulcher, J. Genetic factors contribute to the risk of developing endometriosis. Hum. Reprod. 2002, 17, 555–559. [Google Scholar] [CrossRef] [PubMed]
- Treloar, S.A.; Wicks, J.; Nyholt, D.; Montgomery, G.; Bahlo, M.; Smith, V.; Dawson, G.; Mackay, I.J.; Weeks, D.; Bennett, S.T.; et al. Genomewide Linkage Study in 1,176 Affected Sister Pair Families Identifies a Significant Susceptibility Locus for Endometriosis on Chromosome 10q26. Am. J. Hum. Genet. 2005, 77, 365–376. [Google Scholar] [CrossRef] [PubMed]
- Zondervan, K.T.; Treloar, S.A.; Lin, J.; Weeks, D.; Nyholt, D.; Mangion, J.; Mackay, I.J.; Cardon, L.R.; Martin, N.; Kennedy, S.H.; et al. Significant evidence of one or more susceptibility loci for endometriosis with near-Mendelian inheritance on chromosome 7p13–15. Hum. Reprod. 2007, 22, 717–728. [Google Scholar] [CrossRef]
- Daftary, G.S.; Taylor, H.S. EMX2 Gene Expression in the Female Reproductive Tract and Aberrant Expression in the Endometrium of Patients with Endometriosis. J. Clin. Endocrinol. Metab. 2004, 89, 2390–2396. [Google Scholar] [CrossRef][Green Version]
- Du, H.; Taylor, H.S. Molecular Regulation of Müllerian Development by Hox Genes. Ann. N. Y. Acad. Sci. 2004, 1034, 152–165. [Google Scholar] [CrossRef] [PubMed]
- Dinulescu, D.M.; A Ince, T.; Quade, B.J.; A Shafer, S.; Crowley, D.; Jacks, T. Role of K-ras and Pten in the development of mouse models of endometriosis and endometrioid ovarian cancer. Nat. Med. 2005, 11, 63–70. [Google Scholar] [CrossRef]
- Mutter, G.L.; Lin, M.-C.; Fitzgerald, J.T.; Kum, J.B.; Baak, J.P.A.; Lees, J.A.; Weng, L.-P.; Eng, C. Altered PTEN Expression as a Diagnostic Marker for the Earliest Endometrial Precancers. J. Natl. Cancer Inst. 2000, 92, 924–930. [Google Scholar] [CrossRef] [PubMed]
- Maxwell, G.L.; I Risinger, J.; Gumbs, C.; Shaw, H.; Bentley, R.C.; Barrett, J.C.; Berchuck, A.; A Futreal, P. Mutation of the PTEN tumor suppressor gene in endometrial hyperplasias. Cancer Res. 1998, 58, 2500–2503. [Google Scholar]
- Martini, M.; Ciccarone, M.; Garganese, G.; Maggiore, C.; Evangelista, A.; Rahimi, S.; Zannoni, G.; Vittori, G.; Larocca, L.M. Possible involvement ofhMLH1, p16INK4a andPTEN in the malignant transformation of endometriosis. Int. J. Cancer 2002, 102, 398–406. [Google Scholar] [CrossRef]
- Treloar, S.A.; Zhao, Z.Z.; Le, L.; Zondervan, K.; Martin, N.; Kennedy, S.; Nyholt, D.; Montgomery, G. Variants in EMX2 and PTEN do not contribute to risk of endometriosis. Mol. Hum. Reprod. 2007, 13, 587–594. [Google Scholar] [CrossRef] [PubMed]
- Painter, J.N.; Nyholt, D.; Morris, A.; Zhao, Z.Z.; Henders, A.; Lambert, A.; Wallace, L.; Martin, N.; Kennedy, S.H.; Treloar, S.A.; et al. High-density fine-mapping of a chromosome 10q26 linkage peak suggests association between endometriosis and variants close to CYP2C19. Fertil. Steril. 2011, 95, 2236–2240. [Google Scholar] [CrossRef]
- Painter, J.N.; Nyholt, D.R.; Krause, L.; Zhao, Z.Z.; Chapman, B.; Zhang, C.; Medland, S.; Martin, N.G.; Kennedy, S.; Treloar, S.; et al. Common variants in the CYP2C19 gene are associated with susceptibility to endometriosis. Fertil. Steril. 2014, 102, 496–502. [Google Scholar] [CrossRef]
- Zanger, U.M.; Schwab, M. Cytochrome P450 enzymes in drug metabolism: Regulation of gene expression, enzyme activities, and impact of genetic variation. Pharmacol. Ther. 2013, 138, 103–141. [Google Scholar] [CrossRef]
- Lee, A.J.; Cai, M.X.; Thomas, P.E.; Conney, A.H.; Zhu, B.T. Characterization of the Oxidative Metabolites of 17β-Estradiol and Estrone Formed by 15 Selectively Expressed Human Cytochrome P450 Isoforms. Endocrinology 2003, 144, 3382–3398. [Google Scholar] [CrossRef]
- Painter, J.N.; Anderson, C.A.; Nyholt, D.; MacGregor, S.; Lin, J.; Lee, S.H.; Lambert, A.; Zhao, Z.Z.; Roseman, F.; Guo, Q.; et al. Genome-wide association study identifies a locus at 7p15.2 associated with endometriosis. Nat. Genet. 2010, 43, 51–54. [Google Scholar] [CrossRef] [PubMed]
- Nyholt, D.R.; Low, S.-K.; Anderson, C.A.; Painter, J.N.; Uno, S.; Morris, A.P.; MacGregor, S.; Gordon, S.D.; Henders, A.; Martin, N.; et al. Genome-wide association meta-analysis identifies new endometriosis risk loci. Nat. Genet. 2012, 44, 1355–1359. [Google Scholar] [CrossRef] [PubMed]
- Uno, S.; Zembutsu, H.; Hirasawa, A.; Takahashi, A.; Kubo, M.; Akahane, T.; Aoki, D.; Kamatani, N.; Hirata, K.; Nakamura, Y. A genome-wide association study identifies genetic variants in the CDKN2BAS locus associated with endometriosis in Japanese. Nat. Genet. 2010, 42, 707–710. [Google Scholar] [CrossRef] [PubMed]
- Adachi, S.; Tajima, A.; Quan, J.; Haino, K.; Yoshihara, K.; Masuzaki, H.; Katabuchi, H.; Ikuma, K.; Suginami, H.; Nishida, N.; et al. Meta-analysis of genome-wide association scans for genetic susceptibility to endometriosis in Japanese population. J. Hum. Genet. 2010, 55, 816–821. [Google Scholar] [CrossRef] [PubMed]
- Albertsen, H.M.; Chettier, R.; Farrington, P.; Ward, K. Genome-Wide Association Study Link Novel Loci to Endometriosis. PLoS ONE 2013, 8, e58257. [Google Scholar] [CrossRef]
- Uimari, O.; Rahmioglu, N.; Nyholt, D.; Vincent, K.; Missmer, S.A.; Becker, C.; Morris, A.P.; Montgomery, G.; Zondervan, K.T. Genome-wide genetic analyses highlight mitogen-activated protein kinase (MAPK) signaling in the pathogenesis of endometriosis. Hum. Reprod. 2017, 32, 780–793. [Google Scholar] [CrossRef] [PubMed]
- Sapkota, Y.; De Vivo, I.; Steinthorsdottir, V.; Fassbender, A.; Bowdler, L.; Buring, J.E.; Edwards, T.L.; Jones, S.; Dorien, O.; Peterse, D.; et al. Analysis of potential protein-modifying variants in 9000 endometriosis patients and 150,000 controls of European ancestry. Sci. Rep. 2017, 7, 11380. [Google Scholar] [CrossRef] [PubMed]
- Sapkota, Y.; Fassbender, A.; Bowdler, L.; Fung, J.N.T.; Peterse, D.O.D.; Montgomery, G.; Nyholt, D.; D’Hooghe, T.M. Independent Replication and Meta-Analysis for Endometriosis Risk Loci. Twin Res. Hum. Genet. 2015, 18, 518–525. [Google Scholar] [CrossRef]
- Sapkota, Y.; Steinthorsdottir, V.; Morris, A.P.; Fassbender, A.; Rahmioglu, N.; De Vivo, I.; Buring, J.E.; Zhang, F.; Edwards, T.L.; Jones, S.; et al. Meta-analysis identifies five novel loci associated with endometriosis highlighting key genes involved in hormone metabolism. Nat. Commun. 2017, 8, 15539. [Google Scholar] [CrossRef] [PubMed]
- Rahmioglu, N.; Missmer, S.A.; Montgomery, G.W.; Zondervan, K.T. Insights into Assessing the Genetics of Endometriosis. Curr. Obstet. Gynecol. Rep. 2012, 1, 124–137. [Google Scholar] [CrossRef] [PubMed]
- Rahmioglu, N.; Montgomery, G.; Zondervan, K. Genetics of Endometriosis. Women’s Health 2015, 11, 577–586. [Google Scholar] [CrossRef] [PubMed]
- Neto, J.S.; Kho, R.M.; Siufi, D.F.D.S.; Baracat, E.C.; Anderson, K.S.; Abrão, M.S. Cellular, Histologic, and Molecular Changes Associated with Endometriosis and Ovarian Cancer. J. Minim. Invasive Gynecol. 2014, 21, 55–63. [Google Scholar] [CrossRef]
- Wiegand, K.C.; Shah, S.P.; Al-Agha, O.M.; Zhao, Y.; Tse, K.; Zeng, T.; Senz, J.; McConechy, M.K.; Anglesio, M.S.; Kalloger, S.E.; et al. ARID1AMutations in Endometriosis-Associated Ovarian Carcinomas. N. Engl. J. Med. 2010, 363, 1532–1543. [Google Scholar] [CrossRef]
- Melin, A.; Sparén, P.; Persson, I.; Bergqvist, A. Endometriosis and the risk of cancer with special emphasis on ovarian cancer. Hum. Reprod. 2006, 21, 1237–1242. [Google Scholar] [CrossRef] [PubMed]
- Anglesio, M.S.; Papadopoulos, N.; Ayhan, A.; Nazeran, T.M.; Noë, M.; Horlings, H.M.; Lum, A.; Jones, S.; Senz, J.; Seckin, T.; et al. Cancer-Associated Mutations in Endometriosis without Cancer. N. Engl. J. Med. 2017, 376, 1835–1848. [Google Scholar] [CrossRef]
- Suda, K.; Nakaoka, H.; Yoshihara, K.; Ishiguro, T.; Tamura, R.; Mori, Y.; Yamawaki, K.; Adachi, S.; Takahashi, T.; Kase, H.; et al. Clonal Expansion and Diversification of Cancer-Associated Mutations in Endometriosis and Normal Endometrium. Cell Rep. 2018, 24, 1777–1789. [Google Scholar] [CrossRef] [PubMed]
- Noë, M.; Ayhan, A.; Wang, T.-L.; Shih, I.-M. Independent development of endometrial epithelium and stroma within the same endometriosis. J. Pathol. 2018, 245, 265–269. [Google Scholar] [CrossRef]
- Rathore, N.; Kriplani, A.; Yadav, R.K.; Jaiswal, U.; Netam, R. Distinct peritoneal fluid ghrelin and leptin in infertile women with endometriosis and their correlation with interleukin-6 and vascular endothelial growth factor. Gynecol. Endocrinol. 2014, 30, 671–675. [Google Scholar] [CrossRef]
- Martincorena, I.; Campbell, P.J. Somatic mutation in cancer and normal cells. Science 2015, 349, 1483–1489. [Google Scholar] [CrossRef] [PubMed]
- Moore, L.; Leongamornlert, D.; Coorens, T.H.H.; Sanders, M.A.; Ellis, P.; Dentro, S.C.; Dawson, K.J.; Butler, T.; Rahbari, R.; Mitchell, T.J.; et al. The mutational landscape of normal human endometrial epithelium. Nat. Cell Biol. 2020, 580, 640–646. [Google Scholar] [CrossRef] [PubMed]
- Pon, J.R.; Marra, M.A. Driver and Passenger Mutations in Cancer. Annu. Rev. Pathol. Mech. Dis. 2015, 10, 25–50. [Google Scholar] [CrossRef] [PubMed]
- Leyendecker, G.; Herbertz, M.; Kunz, G.; Mall, G. Endometriosis results from the dislocation of basal endometrium. Hum. Reprod. 2002, 17, 2725–2736. [Google Scholar] [CrossRef] [PubMed]
- Králíčková, M.; Laganà, A.S.; Ghezzi, F.; Vetvicka, V. Endometriosis and risk of ovarian cancer: What do we know? Arch. Gynecol. Obstet. 2019, 301, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Alderman, M.H.; Yoder, N.; Taylor, H.S. The Systemic Effects of Endometriosis. Semin. Reprod. Med. 2017, 35, 263–270. [Google Scholar] [CrossRef]
- Kajiyama, H.; Suzuki, S.; Yoshihara, M.; Tamauchi, S.; Yoshikawa, N.; Niimi, K.; Shibata, K.; Kikkawa, F. Endometriosis and cancer. Free. Radic. Biol. Med. 2019, 133, 186–192. [Google Scholar] [CrossRef]
- Vargas-Hernández, V.M. La endometriosis como factor de riesgo para cáncer de ovario. Cirugía Cirujanos 2013, 81, 163–168. (in Spanish). [Google Scholar]
- Saavalainen, L.; Lassus, H.; But, A.; Tiitinen, A.; Härkki, P.; Gissler, M.; Pukkala, E.; Heikinheimo, O. Risk of Gynecologic Cancer According to the Type of Endometriosis. Obstet. Gynecol. 2018, 131, 1095–1102. [Google Scholar] [CrossRef]
- Poole, E.M.; Lin, W.T.; Kvaskoff, M.; De Vivo, I.; Terry, K.L.; Missmer, S.A. Endometriosis and risk of ovarian and endometrial cancers in a large prospective cohort of U.S. nurses. Cancer Causes Control. 2017, 28, 437–445. [Google Scholar] [CrossRef]
- Rowlands, I.J.; Nagle, C.M.; Spurdle, A.B.; Webb, P.M. Gynecological conditions and the risk of endometrial cancer. Gynecol. Oncol. 2011, 123, 537–541. [Google Scholar] [CrossRef]
- Kalaitzopoulos, D.R.; Mitsopoulou, A.; Iliopoulou, S.M.; Daniilidis, A.; Samartzis, E.P.; Economopoulos, K.P. Association between endometriosis and gynecological cancers: A critical review of the literature. Arch. Gynecol. Obstet. 2020, 301, 355–367. [Google Scholar] [CrossRef]
- Chen, J.-J.; Xiao, Z.-J.; Meng, X.; Wang, Y.; Yu, M.K.; Huang, W.Q.; Sun, X.; Chen, H.; Duan, Y.-G.; Jiang, C.; et al. MRP4 sustains Wnt/β-catenin signaling for pregnancy, endometriosis and endometrial cancer. Theranostics 2019, 9, 5049–5064. [Google Scholar] [CrossRef]
- Johnatty, S.E.; Stewart, C.J.R.; Smith, D.; Nguyen, A.; Dwyer, J.O.; O’Mara, T.A.; Webb, P.M.; Spurdle, A.B. Co-existence of leiomyomas, adenomyosis and endometriosis in women with endometrial cancer. Sci. Rep. 2020, 10, 3621. [Google Scholar] [CrossRef] [PubMed]
- Mogensen, J.B.; Kjaer, S.K.; Mellemkjaer, L.; Jensen, A. Endometriosis and risks for ovarian, endometrial and breast cancers: A nationwide cohort study. Gynecol. Oncol. 2016, 143, 87–92. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.-C.; Lin, C.-Y.; Chang, W.-C.; Shen, B.-J.; Chang, W.-P.; Chuang, C.-M. Increased association between endo-metriosis and endometrial cancer: A nationwide population-based retrospective cohort study. Int. J. Gynecol. Cancer 2015, 25, 447–452. [Google Scholar] [CrossRef]
- Franchi, M.; Garzon, S.; Zorzato, P.C.; Laganà, A.S.; Casarin, J.; Locantore, L.; Raffaelli, R.; Ghezzi, F. PET-CT scan in the preoperative workup of early stage intermediate- and high-risk endometrial cancer. Minim. Invasive Ther. Allied Technol. 2019, 29, 232–239. [Google Scholar] [CrossRef] [PubMed]
- Cignini, P.; Vitale, S.G.; Laganà, A.S.; Biondi, A.; La Rosa, V.L.; Cutillo, G. Preoperative work-up for definition of lymph node risk involvement in early stage endometrial cancer: 5-year follow-up. Updates Surg. 2017, 69, 75–82. [Google Scholar] [CrossRef]
- Vitale, S.G.; Rossetti, D.; Tropea, A.; Biondi, A.; Laganà, A.S. Fertility sparing surgery for stage IA type I and G2 endometrial cancer in reproductive-aged patients: Evidence-based approach and future perspectives. Updates Surg. 2017, 69, 29–34. [Google Scholar] [CrossRef]
- Casarin, J.; Bogani, G.; Piovano, E.; Falcone, F.; Ferrari, F.; Odicino, F.; Puppo, A.; Bonfiglio, F.; Donadello, N.; Pinelli, C.; et al. Survival implication of lymphadenectomy in patients surgically treated for apparent early-stage uterine serous carcinoma. J. Gynecol. Oncol. 2020, 31, e64. [Google Scholar] [CrossRef]
- Freytag, D.; Pape, J.; Dhanawat, J.; Günther, V.; Maass, N.; Gitas, G.; Laganà, A.S.; Allahqoli, L.; Meinhold-Heerlein, I.; Moawad, G.N.; et al. Challenges Posed by Embryonic and Anatomical Factors in Systematic Lymphadenectomy for Endometrial Cancer. J. Clin. Med. 2020, 9, 4107. [Google Scholar] [CrossRef]
- Lee, S.-Y. Tailored Therapy Based on Molecular Characteristics in Endometrial Cancer. BioMed Res. Int. 2021, 2021, 2068023. [Google Scholar] [CrossRef]
- Casarin, J.; Bogani, G.; Serati, M.; Pinelli, C.; Laganà, A.S.; Garzon, S.; Raspagliesi, F.; Ghezzi, F. Presence of Glandular Cells at the Preoperative Cervical Cytology and Local Recurrence in Endometrial Cancer. Int. J. Gynecol. Pathol. 2020, 39, 522–528. [Google Scholar] [CrossRef] [PubMed]
- Winterhoff, B.; Thomaier, L.; Mullany, S.; Powell, M.A. Molecular characterization of endometrial cancer and therapeutic implications. Curr. Opin. Obstet. Gynecol. 2020, 32, 76–83. [Google Scholar] [CrossRef]
- Cancer Genome Atlas Research Network, Kandoth C, Schultz N, et al. Integrated genomic characterization of endometrial carcinoma. Nature 2013, 497, 67–73. [Google Scholar] [CrossRef]
- Huvila, J.; Pors, J.; Thompson, E.F.; Gilks, C.B. Endometrial carcinoma: Molecular subtypes, precursors and the role of pathology in early diagnosis. J. Pathol. 2021, 253, 355–365. [Google Scholar] [CrossRef] [PubMed]
- León-Castillo, A.; De Boer, S.M.; Powell, M.E.; Mileshkin, L.R.; Mackay, H.J.; Leary, A.; Nijman, H.W.; Singh, N.; Pollock, P.M.; Bessette, P.; et al. Molecular Classification of the PORTEC-3 Trial for High-Risk Endometrial Cancer: Impact on Prognosis and Benefit from Adjuvant Therapy. J. Clin. Oncol. 2020, 38, 3388–3397. [Google Scholar] [CrossRef] [PubMed]
- McAlpine, J.; Leon-Castillo, A.; Bosse, T. The rise of a novel classification system for endometrial carcinoma; integration of mo-lecular subclasses. J. Pathol. 2018, 244, 538–549. [Google Scholar] [CrossRef]
- Carlson, J.; McCluggage, W.G. Reclassifying endometrial carcinomas with a combined morphological and molecular approach. Curr. Opin. Oncol. 2019, 31, 411–419. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, M.; Obata, T.; Daikoku, T.; Fujiwara, H. The Association and Significance of p53 in Gynecologic Cancers: The Potential of Targeted Therapy. Int. J. Mol. Sci. 2019, 20, 5482. [Google Scholar] [CrossRef] [PubMed]
- Salvesen, H.B.; Haldorsen, I.S.; Trovik, J. Markers for individualised therapy in endometrial carcinoma. Lancet Oncol. 2012, 13, e353–e361. [Google Scholar] [CrossRef]
- Alexa, M.; Hasenburg, A.; Battista, M. The TCGA Molecular Classification of Endometrial Cancer and Its Possible Impact on Adjuvant Treatment Decisions. Cancers 2021, 13, 1478. [Google Scholar] [CrossRef] [PubMed]
- Menderes, G.; Lopez, S.; Han, C.; Altwerger, G.; Gysler, S.; Varughese, J.; E Schwartz, P.; Santin, A.D. Mechanisms of resistance to HER2-targeted therapies in HER2-amplified uterine serous carcinoma, and strategies to overcome it. Discov. Med. 2018, 26, 39–50. [Google Scholar] [PubMed]
- Erickson, B.K.; Zeybek, B.; Santin, A.D.; Fader, A.N. Targeting human epidermal growth factor receptor 2 (HER2) in gynecologic malignancies. Curr. Opin. Obstet. Gynecol. 2020, 32, 57–64. [Google Scholar] [CrossRef]
- Ferriss, J.S.; Erickson, B.K.; Shih, I.-M.; Fader, A.N. Uterine serous carcinoma: Key advances and novel treatment approaches. Int. J. Gynecol. Cancer 2021, 31, 1165–1174. [Google Scholar] [CrossRef] [PubMed]
- Stelloo, E.; Nout, R.A.; Osse, E.M.; Juergenliemk-Schulz, I.J.; Jobsen, J.J.; Lutgens, L.C.; Van Der Steen-Banasik, E.M.; Nijman, H.W.; Putter, H.; Bosse, T.; et al. Improved Risk Assessment by Integrating Molecular and Clinicopathological Factors in Early-stage Endometrial Cancer—Combined Analysis of the PORTEC Cohorts. Clin. Cancer Res. 2016, 22, 4215–4224. [Google Scholar] [CrossRef] [PubMed]
- Talhouk, A.; DeRocher, H.; Schmidt, P.; Leung, S.; Milne, K.; Gilks, C.B.; Anglesio, M.S.; Nelson, B.H.; McAlpine, J.N. Molecular Subtype Not Immune Response Drives Outcomes in Endometrial Carcinoma. Clin. Cancer Res. 2019, 25, 2537–2548. [Google Scholar] [CrossRef]
- Reijnen, C.; Küsters-Vandevelde, H.V.; Prinsen, C.F.; Massuger, L.F.; Snijders, M.P.; Kommoss, S.; Brucker, S.Y.; Kwon, J.S.; McAlpine, J.N.; Pijnenborg, J.M. Mismatch repair deficiency as a predictive marker for response to adjuvant radiotherapy in endometrial cancer. Gynecol. Oncol. 2019, 154, 124–130. [Google Scholar] [CrossRef]
- Mackay, H.J.; Freixinos, V.R.; Fleming, G.F. Therapeutic Targets and Opportunities in Endometrial Cancer: Update on Endocrine Therapy and Nonimmunotherapy Targeted Options. Am. Soc. Clin. Oncol. Educ. Book 2020, 40, 245–255. [Google Scholar] [CrossRef]
- Gómez-Raposo, C.; Salvador, M.M.; Zamora, C.A.; de Santiago, B.G.; Sáenz, E.C. Immune checkpoint inhibitors in endometrial cancer. Crit. Rev. Oncol. 2021, 161, 103306. [Google Scholar] [CrossRef]
- Jamieson, A.; Thompson, E.F.; Huvila, J.; Gilks, C.B.; McAlpine, J.N. p53abn Endometrial Cancer: Understanding the most aggressive endometrial cancers in the era of molecular classification. Int. J. Gynecol. Cancer 2021, 31, 907–913. [Google Scholar] [CrossRef] [PubMed]
- Nm, A.E.M.; El-Gelany, S. Differential Expression Patterns of PTEN in Cyclic, Hyperplastic and Malignant Endome-trium: Its Relation with ER, PR and Clinicopathological Parameters. J. Egypt. Natl. Cancer Inst. 2009, 21, 323–331. [Google Scholar]
- Giono, L.E.; Manfredi, J.J. The p53 tumor suppressor participates in multiple cell cycle checkpoints. J. Cell. Physiol. 2006, 209, 13–20. [Google Scholar] [CrossRef] [PubMed]
- D’Andrilli, G.; Bovicelli, A.; Paggi, M.G.; Giordano, A. New insights in endometrial carcinogenesis. J. Cell. Physiol. 2012, 227, 2842–2846. [Google Scholar] [CrossRef]
- Angioli, R.; Miranda, A.; Aloisi, A.; Montera, R.; Capriglione, S.; Nardone, C.D.C.; Terranova, C.; Plotti, F. A critical review on HE4 performance in endometrial cancer: Where are we now? Tumor Biol. 2014, 35, 881–887. [Google Scholar] [CrossRef] [PubMed]
- Kemik, P.; Saatli, B.; Yıldırım, N.; Kemik, V.D.; Deveci, B.; Terek, M.C.; Koçtürk, S.; Koyuncuoğlu, M.; Saygılı, U. Diagnostic and prognostic values of preoperative serum levels ofYKL-40, HE-4 and DKK-3 in endometrial cancer. Gynecol. Oncol. 2016, 140, 64–69. [Google Scholar] [CrossRef]
| Classification | ||||||
|---|---|---|---|---|---|---|
| American Society for Reproductive Medicine (ASRM) | ||||||
| Staging | Points | |||||
| Stage 1—Minimal Endometriosis | 1–5 | |||||
| Stage 2—Mild Endometriosis | 6–15 | |||||
| Stage 3—Moderate Endometriosis | 16–40 | |||||
| Stage 4—Severe Endometriosis | >40 | |||||
| ENZIAN (supplement to ASRM) | ||||||
| Compartments | ||||||
| Compartment A: vagina, recto-vaginal septum; | ||||||
| Compartment B: uterosacral ligaments to the pelvic wall (BB: bilateral involvement); | ||||||
| Compartment C: rectum and sigmoid colon. | ||||||
| Disease severity | Invasion | |||||
| Grade 1: | <1 cm | |||||
| Grade 2: | 1–3 cm | |||||
| Grade 3: | >3 cm | |||||
| Deep endometriosis invasion beyond the pelvis | ||||||
| FA: adenomyosis | ||||||
| FB: bladder invasion | ||||||
| FU: intrinsic ureteral endometriosis; | ||||||
| FI: bowel disease cranial to the sigmoid colon | ||||||
| F0: other locations | ||||||
| The Endometriosis Fertility Index (EFI) | ||||||
| Historical factor | Years | Points | ||||
| Patient age | ≤35 | 2 | ||||
| 36–39 | 1 | |||||
| ≥40 | 0 | |||||
| Duration of infertility | ≤3 | 1 | ||||
| >3 | 0 | |||||
| Prior pregnancy | Yes | 1 | ||||
| No | 0 | |||||
| Score | Description | |||||
| 4 | Normal | |||||
| 3 | Mild | |||||
| 2 | Moderate | |||||
| 1 | Severe | |||||
| 0 | Absent or nonfunctional | |||||
| Endometrial Cancer Type | Molecular Factors/Genes | Changes in Function Leading to Endometrial Cancer |
|---|---|---|
| Type 1 | ||
| CTNNB1 | Mutation | |
| POLE | Mutation | |
| KRAS | Mutation | |
| PTEN | Loss | |
| AKT | Up-regulation | |
| PI3KSA | Up-regulation | |
| G1/S cell cycle phase | Progression | |
| Bcl-2 | Loss of down-regulation | |
| MLH1/MSH6 | Instability | |
| DNA | Mismatch | |
| Type 2 | ||
| TP53 | Mutation | |
| ERBB-2 (HER2/neu) | Overexpression | |
| p-16 | Inactivation | |
| E-cadherin | Reduction | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Terzic, M.; Aimagambetova, G.; Kunz, J.; Bapayeva, G.; Aitbayeva, B.; Terzic, S.; Laganà, A.S. Molecular Basis of Endometriosis and Endometrial Cancer: Current Knowledge and Future Perspectives. Int. J. Mol. Sci. 2021, 22, 9274. https://doi.org/10.3390/ijms22179274
Terzic M, Aimagambetova G, Kunz J, Bapayeva G, Aitbayeva B, Terzic S, Laganà AS. Molecular Basis of Endometriosis and Endometrial Cancer: Current Knowledge and Future Perspectives. International Journal of Molecular Sciences. 2021; 22(17):9274. https://doi.org/10.3390/ijms22179274
Chicago/Turabian StyleTerzic, Milan, Gulzhanat Aimagambetova, Jeannette Kunz, Gauri Bapayeva, Botagoz Aitbayeva, Sanja Terzic, and Antonio Simone Laganà. 2021. "Molecular Basis of Endometriosis and Endometrial Cancer: Current Knowledge and Future Perspectives" International Journal of Molecular Sciences 22, no. 17: 9274. https://doi.org/10.3390/ijms22179274
APA StyleTerzic, M., Aimagambetova, G., Kunz, J., Bapayeva, G., Aitbayeva, B., Terzic, S., & Laganà, A. S. (2021). Molecular Basis of Endometriosis and Endometrial Cancer: Current Knowledge and Future Perspectives. International Journal of Molecular Sciences, 22(17), 9274. https://doi.org/10.3390/ijms22179274









