Experimental Evidence of 2,3,7,8-Tetrachlordibenzo-p-Dioxin (TCDD) Transgenerational Effects on Reproductive Health
Abstract
1. Introduction
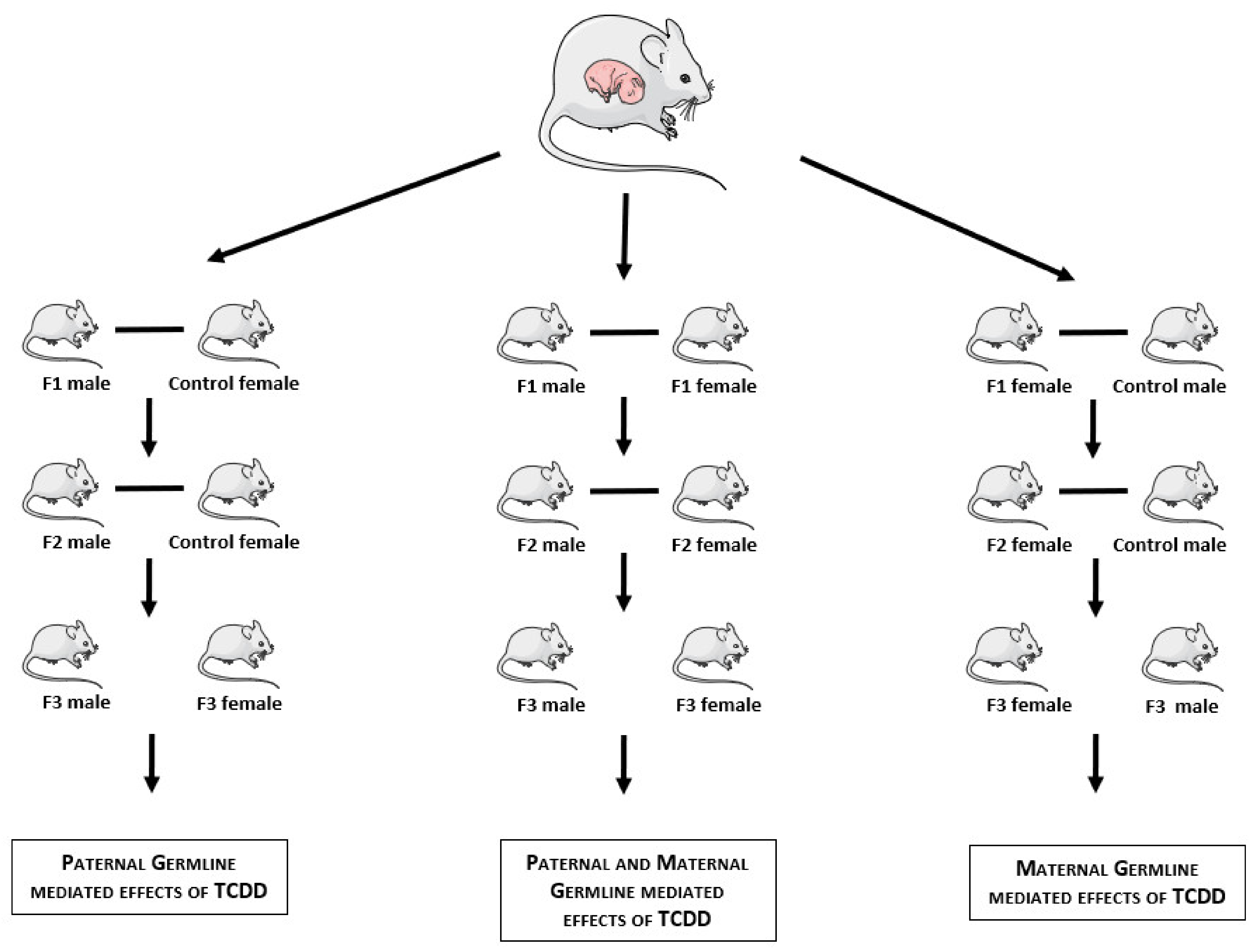
2. Materials and Methods
3. Results
3.1. Articles Selection
3.2. TCDD Transgenerational Effects on Female Reproductive Health
3.3. TCDD Transgenerational Effects on Male Reproduction
3.4. Transgenerational Epigenetic Mechanisms
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Barker, D.J. The developmental origins of adult disease. Eur. J. Epidemiol. 2003, 18, 733–736. [Google Scholar] [CrossRef]
- Welsh, M.; Saunders, P.T.; Fisken, M.; Scott, H.M.; Hutchison, G.R.; Smith, L.B.; Sharpe, R.M. Identification in rats of a programming window for reproductive tract masculinization, disruption of which leads to hypospadias and cryptorchidism. J. Clin. Investig. 2008, 118, 1479–1490. [Google Scholar] [CrossRef] [PubMed]
- Buser, M.C.; Abadin, H.G.; Irwin, J.L.; Pohl, H.R. Windows of sensitivity to toxic chemicals in the development of reproductive effects: An analysis of ATSDR’s toxicological profile database. Int. J. Environ. Health Res. 2018, 28, 553–578. [Google Scholar] [CrossRef] [PubMed]
- DeRosa, C.; Richter, P.; Pohl, H.; Jones, D.E. Environmental exposures that affect the endocrine system: Public health implications. J. Toxicol. Environ. Health Part B 1998, 1, 3–26. [Google Scholar] [CrossRef] [PubMed]
- Rakyan, V.; Whitelaw, E. Transgenerational epigenetic inheritance. Curr. Biol. 2003, 13, R6. [Google Scholar] [CrossRef]
- Anway, M.D.; Cupp, A.S.; Uzumcu, M.; Skinner, M.K. Epigenetic Transgenerational Actions of Endocrine Disruptors and Male Fertility. Science 2005, 308, 1466–1469. [Google Scholar] [CrossRef]
- Anway, M.D.; Skinner, M.K. Epigenetic transgenerational actions of endocrine disruptors. Endocrinology 2006, 147, S43–S49. [Google Scholar] [CrossRef]
- Skinner, M.K.; Anway, M.D. Seminiferous Cord Formation and Germ-Cell Programming: Epigenetic Transgenerational Actions of Endocrine Disruptors. Ann. N. Y. Acad. Sci. 2005, 1061, 18–32. [Google Scholar] [CrossRef]
- Skinner, M.K.; Manikkam, M.; Guerrero-Bosagna, C. Epigenetic transgenerational actions of environmental factors in disease etiology. Trends Endocrinol. Metab. 2010, 21, 214–222. [Google Scholar] [CrossRef]
- Ben Maamar, M.; Nilsson, E.; Thorson, J.L.; Beck, D.; Skinner, M.K. Transgenerational disease specific epigenetic sperm biomarkers after ancestral exposure to dioxin. Environ. Res. 2021, 192, 110279. [Google Scholar] [CrossRef]
- Kimbel, H.; Nilsson, E.; Skinner, M. Environmentally Induced Epigenetic Transgenerational Inheritance of Ovarian Disease. Ovary 2018, 4, 149–154. [Google Scholar] [CrossRef]
- Morán, F.; Conley, A.; Corbin, C.; Enan, E.; Vandevoort, C.; Overstreet, J.; Lasley, B. 2,3,7,8-Tetrachlorodibenzo-p-Dioxin Decreases Estradiol Production Without Altering the Enzyme Activity of Cytochrome P450 Aromatase of Human Luteinized Granulosa Cells in Vitro. Biol. Reprod. 2000, 62, 1102–1108. [Google Scholar] [CrossRef]
- Orlowska, K.; Swigonska, S.; Sadowska, A.; Ruszkowska, M.; Nynca, A.; Molcan, T.; Zmijewska, A.; Ciereszko, R.E. Proteomic changes of aryl hydrocarbon receptor (AhR)-silenced porcine granulosa cells exposed to 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD). PLoS ONE 2019, 14, e0223420. [Google Scholar] [CrossRef]
- Ho, H.; Ohshima, K.-I.; Watanabe, G.; Taya, K.; Strawn, E.; Hutz, R.J. TCDD Increases Inhibin A Production by Human Luteinized Granulosa Cells In Vitro. J. Reprod. Dev. 2006, 52, 523–528. [Google Scholar] [CrossRef] [PubMed]
- Tai, P.T.; Nishijo, M.; Kido, T.; Nakagawa, H.; Maruzeni, S.; Naganuma, R.; Anh, N.T.; Morikawa, Y.; Luong, H.V.; Anh, T.H.; et al. Dioxin concentrations in breast milk of Vietnamese nursing mothers: A survey four decades after the herbicide spraying. Environ. Sci. Technol. 2011, 45, 6625–6632. [Google Scholar] [CrossRef]
- Weiss, J.; Päpke, O.; Bignert, A.; Jensen, S.; Greyerz, E.; Agostoni, C.; Besana, R.; Riva, E.; Giovannini, M.; Zetterström, R. Concentrations of dioxins and other organochlorines (PCBs, DDTs, HCHs) in human milk from Seveso, Milan and a Lombardian rural area in Italy: A study performed 25 years after the heavy dioxin exposure in Seveso. Acta Paediatr. 2007, 92, 467–472. [Google Scholar] [CrossRef] [PubMed]
- Michalek, J.E.; Pirkle, J.L.; Needham, L.L.; Patterson, D.G., Jr.; Caudill, S.P.; Tripathi, R.C.; Mocarelli, P. Pharmacokinetics of 2,3,7,8-tetrachlorodibenzo-p-dioxin in Seveso adults and veterans of operation Ranch Hand. J. Expo. Anal. Environ. Epidemiol. 2002, 12, 44–53. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Brulport, A.; Le Corre, L.; Chagnon, M.-C. Chronic exposure of 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) induces an obesogenic effect in C57BL/6J mice fed a high fat diet. Toxicology 2017, 390, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Egeland, G.M.; Sweeney, M.H.; Fingerhut, M.A.; Wille, K.K.; Schnorr, T.M.; Halperin, W.E. Total Serum Testosterone and Gonadotropins in Workers Exposed to Dioxin. Am. J. Epidemiol. 1994, 139, 272–281. [Google Scholar] [CrossRef] [PubMed]
- Bois, F.Y.; Eskenazi, B. Possible risk of endometriosis for Seveso, Italy, residents: An assessment of exposure to dioxin. Environ. Health Perspect 1994, 102, 476–477. [Google Scholar] [CrossRef][Green Version]
- Kido, T.; Van Dao, T.; Ho, M.D.; Dang, N.D.; Pham, N.T.; Okamoto, R.; Pham, T.T.; Maruzeni, S.; Nishijo, M.; Nakagawa, H.; et al. High cortisol and cortisone levels are associated with breast milk dioxin concentrations in Vietnamese women. Eur. J. Endocrinol. 2014, 170, 131–139. [Google Scholar] [CrossRef] [PubMed]
- Eskenazi, B.; Ames, J.; Rauch, S.; Signorini, S.; Brambilla, P.; Mocarelli, P.; Siracusa, C.; Holland, N.; Warner, M. Dioxin exposure associated with fecundability and infertility in mothers and daughters of Seveso, Italy. Hum. Reprod. 2021, 36, 794–807. [Google Scholar] [CrossRef] [PubMed]
- Eskenazi, B.; Mocarelli, P.; Warner, M.; Chee, W.Y.; Gerthoux, P.M.; Samuels, S.; Needham, L.L.; Patterson, D.G., Jr. Maternal serum dioxin levels and birth outcomes in women of Seveso, Italy. Environ. Health Perspect. 2003, 111, 947–953. [Google Scholar] [CrossRef] [PubMed]
- Eskenazi, B.; Mocarelli, P.; Warner, M.; Samuels, S.; Vercellini, P.P.; Olive, D.; Needham, L.; Patterson, D.; Brambilla, P. Seveso Women’s Health Study: A study of the effects of 2,3,7,8-tetrachlorodibenzo- p -dioxin on reproductive health. Chemosphere 2000, 40, 1247–1253. [Google Scholar] [CrossRef]
- Eskenazi, B.; Mocarelli, P.; Warner, M.; Samuels, S.; Vercellini, P.; Olive, D.; Needham, L.L.; Patterson, D.G.; Brambilla, P.; Gavoni, N.; et al. Serum dioxin concentrations and endometriosis: A cohort study in Seveso, Italy. Environ. Health Perspect. 2002, 110, 629–634. [Google Scholar] [CrossRef] [PubMed]
- Eskenazi, B.; Warner, M.; Brambilla, P.; Signorini, S.; Ames, J.; Mocarelli, P. The Seveso accident: A look at 40 years of health research and beyond. Environ. Int. 2018, 121, 71–84. [Google Scholar] [CrossRef]
- Eskenazi, B.; Warner, M.; Marks, A.R.; Samuels, S.; Gerthoux, P.A.; Vercellini, P.; Olive, D.L.; Needham, L.; Patterson, D.G.; Mocarelli, P. Serum Dioxin Concentrations and Age at Menopause-0. Environ. Health Perspect 2014, 29, 858–862. [Google Scholar] [CrossRef]
- Eskenazi, B.; Warner, M.; Marks, A.; Samuels, S.; Needham, L.; Brambilla, P.; Mocarelli, P. Serum Dioxin Concentrations and Time to Pregnancy. Epidemiology 2010, 21, 224–231. [Google Scholar] [CrossRef]
- Eskenazi, B.; Warner, M.; Mocarelli, P.; Samuels, S.; Needham, L.L.; Patterson, N.G.; Lippman, S.; Vercellini, P.; Gerthoux, P.M.; Brambilla, P.; et al. Serum dioxin concentrations and menstrual cycle characteristics. Am. J. Epidemiol. 2002, 156, 383–392. [Google Scholar] [CrossRef]
- Eskenazi, B.; Warner, M.; Samuels, S.; Young, J.; Gerthoux, P.M.; Needham, L.; Patterson, D.; Olive, D.; Gavoni, N.; Vercellini, P.; et al. Serum dioxin concentrations and risk of uterine leiomyoma in the Seveso Women’s Health Study. Am. J. Epidemiol. 2007, 166, 79–87. [Google Scholar] [CrossRef]
- Mocarelli, P.; Brambilla, P.; Gerthoux, P.M.; Peterson, D.G., Jr.; Needham, L.L. Change in sex ratio with exposure to dioxin. Lancet 1996, 348, 409. [Google Scholar] [CrossRef]
- Mocarelli, P.; Gerthoux, P.M.; Ferrari, E.; Patterson, D.G.; Kieszak, S.M.; Brambilla, P.; Vincoli, N.; Signorini, S.; Tramacere, P.; Carreri, V.; et al. Paternal concentrations of dioxin and sex ratio of offspring. Lancet 2000, 355, 1858–1863. [Google Scholar] [CrossRef]
- Mocarelli, P.; Gerthoux, P.M.; Needham, L.L.; Patterson, D.G.; Limonta, G.; Falbo, R.; Signorini, S.; Bertona, M.; Crespi, C.; Sarto, C.; et al. Perinatal Exposure to Low Doses of Dioxin Can Permanently Impair Human Semen Quality. Environ. Health Perspect. 2011, 119, 713–718. [Google Scholar] [CrossRef]
- Mocarelli, P.; Gerthoux, P.M.; Patterson, D.G.; Milani, S.; Limonta, G.; Bertona, M.; Signorini, S.; Tramacere, P.; Colombo, L.; Crespi, C.; et al. Dioxin Exposure, from Infancy through Puberty, Produces Endocrine Disruption and Affects Human Semen Quality. Environ. Health Perspect. 2008, 116, 70–77. [Google Scholar] [CrossRef] [PubMed]
- Mocarelli, P.; Marocchi, A.; Brambilla, P.; Gerthoux, P.; Young, D.S.; Mantel, N. Clinical laboratory manifestations of exposure to dioxin in children. A six-year study of the effects of an environmental disaster near Seveso, Italy. JAMA 1986, 256, 2687–2695. [Google Scholar] [CrossRef] [PubMed]
- Mocarelli, P.; Needham, L.L.; Marocchi, A.; Patterson, N.G.; Brambilla, P.; Gerthoux, P.M.; Meazza, L.; Carreri, V. Serum concentrations of 2,3,7,8-tetrachlorodibenzo-p-dioxin and test results from selected residents of Seveso, Italy. J. Toxicol. Environ. Heal. Part A 1991, 32, 357–366. [Google Scholar] [CrossRef] [PubMed]
- Warner, M.; Eskenazi, B.; Olive, D.L.; Samuels, S.; Quick-Miles, S.; Vercellini, P.; Gerthoux, P.M.; Needham, L.; Patterson, N.G.; Mocarelli, P. Serum Dioxin Concentrations and Quality of Ovarian Function in Women of Seveso. Environ. Health Perspect. 2007, 115, 336–340. [Google Scholar] [CrossRef]
- Field, B.; Kerr, C. Reproductive behaviour and consistent patterns of abnormality in offspring of Vietnam veterans. J. Med. Genet. 1988, 25, 819–826. [Google Scholar] [CrossRef]
- Warner, M.; Rauch, S.; Ames, J.; Mocarelli, P.; Brambilla, P.; Eskenazi, B. Age at menarche in Seveso daughters exposed in utero to 2,3,7,8-tetrachlorodibenzo-p-dioxin. Environ. Epidemiol. 2020, 4, e111. [Google Scholar] [CrossRef]
- Ryan, J.J.; Amirova, Z.; Carrier, G. Sex ratios of children of Russian pesticide producers exposed to dioxin. Environ. Health Perspect. 2002, 110, A699–A701. [Google Scholar] [CrossRef]
- Mannetje, A.T.; Eng, A.; Walls, C.; Dryson, E.; Kogevinas, M.; Brooks, C.; McLean, D.; Cheng, S.; Smith, A.H.; Pearce, N. Sex ratio of the offspring of New Zealand phenoxy herbicide producers exposed to 2,3,7,8-tetrachlorodibenzo-p-dioxin. Occup. Environ. Med. 2016, 74, 24–29. [Google Scholar] [CrossRef]
- Mínguez-Alarcón, L.; Sergeyev, O.; Burns, J.S.; Williams, P.L.; Lee, M.M.; Korrick, S.A.; Smigulina, L.; Revich, B.; Hauser, R. A Longitudinal Study of Peripubertal Serum Organochlorine Concentrations and Semen Parameters in Young Men: The Russian Children’s Study. Environ. Health Perspect. 2017, 125, 460–466. [Google Scholar] [CrossRef]
- Ruegg, J.; Swedenborg, E.; Wahlstrom, D.; Escande, A.; Balaguer, P.; Pettersson, K.; Pongratz, I. The transcription factor aryl hydrocarbon receptor nuclear translocator functions as an estrogen receptor beta-selective coactivator, and its recruitment to alternative pathways mediates antiestrogenic effects of dioxin. Mol. Endocrinol. 2008, 22, 304–316. [Google Scholar] [CrossRef] [PubMed]
- Swedenborg, E.; Kotka, M.; Seifert, M.; Kanno, J.; Pongratz, I.; Rüegg, J. The aryl hydrocarbon receptor ligands 2,3,7,8-tetrachlorodibenzo-p-dioxin and 3-methylcholanthrene regulate distinct genetic networks. Mol. Cell. Endocrinol. 2012, 362, 39–47. [Google Scholar] [CrossRef]
- Klip, H.; Verloop, J.; van Gool, J.D.; Koster, M.E.; Burger, C.W.; van Leeuwen, F.E.; Group, O.P. Hypospadias in sons of women exposed to diethylstilbestrol in utero: A cohort study. Lancet 2002, 359, 1102–1107. [Google Scholar] [CrossRef]
- Pons, J.C.; Papiernik, E.; Billon, A.; Hessabi, M.; Duyme, M. Hypospadias in sons of women exposed to diethylstilbestrol in utero. Prenat. Diagn. 2005, 25, 418–419. [Google Scholar] [CrossRef] [PubMed]
- Brouwers, M.M.; Feitz, W.F.J.; Roelofs, L.A.J.; Kiemeney, L.; De Gier, R.P.E.; Roeleveld, N. Hypospadias: A transgenerational effect of diethylstilbestrol? Hum. Reprod. 2006, 21, 666–669. [Google Scholar] [CrossRef] [PubMed]
- Kalfa, N.; Paris, F.; Soyer-Gobillard, M.-O.; Daures, J.-P.; Sultan, C. Prevalence of hypospadias in grandsons of women exposed to diethylstilbestrol during pregnancy: A multigenerational national cohort study. Fertil. Steril. 2011, 95, 2574–2577. [Google Scholar] [CrossRef] [PubMed]
- Gaspari, L.; Paris, F.; Soyer-Gobillard, M.-O.; Hamamah, S.; Kalfa, N.; Sultan, C. “Idiopathic” partial androgen insensitivity syndrome in 11 grandsons of women treated by diethylstilbestrol during gestation: A multi-generational impact of endocrine disruptor contamination? J. Endocrinol. Investig. 2021, 44, 379–381. [Google Scholar] [CrossRef] [PubMed]
- Titus-Ernstoff, L.; Troisi, R.; Hatch, E.; Wise, L.; Palmer, J.; Hyer, M.; Kaufman, R.; Adam, E.; Strohsnitter, W.; Noller, K.; et al. Menstrual and reproductive characteristics of women whose mothers were exposed in utero to diethylstilbestrol (DES). Int. J. Epidemiol. 2006, 35, 862–868. [Google Scholar] [CrossRef]
- Titus, L.; Hatch, E.E.; Drake, K.M.; Parker, S.E.; Hyer, M.; Palmer, J.R.; Strohsnitter, W.C.; Adam, E.; Herbst, A.L.; Huo, D.; et al. Reproductive and hormone-related outcomes in women whose mothers were exposed in utero to diethylstilbestrol (DES): A report from the US National Cancer Institute DES Third Generation Study. Reprod. Toxicol. 2019, 84, 32–38. [Google Scholar] [CrossRef]
- Titus-Ernstoff, L.; Troisi, R.; Hatch, E.; Palmer, J.; Hyer, M.; Kaufman, R.; Adam, E.; Noller, K.; Hoover, R.N. Birth defects in the sons and daughters of women who were exposed in utero to diethylstilbestrol (DES). Int. J. Androl. 2010, 33, 377–384. [Google Scholar] [CrossRef]
- Gaspari, L.; Paris, F.; Cassel-Knipping, N.; Villeret, J.; Verschuur, A.; Soyer-Gobillard, M.-O.; Carcopino-Tusoli, X.; Hamamah, S.; Kalfa, N.; Sultan, C. Diethylstilbestrol exposure during pregnancy with primary clear cell carcinoma of the cervix in an 8-year-old granddaughter: A multigenerational effect of endocrine disruptors? Hum. Reprod. 2020, 36, 82–86. [Google Scholar] [CrossRef] [PubMed]
- Manikkam, M.; Tracey, R.; Guerrero-Bosagna, C.; Skinner, M.K. Dioxin (TCDD) Induces Epigenetic Transgenerational Inheritance of Adult Onset Disease and Sperm Epimutations. PLoS ONE 2012, 7, e46249. [Google Scholar] [CrossRef]
- Nilsson, E.; Larsen, G.; Manikkam, M.; Guerrero-Bosagna, C.; Savenkova, M.I.; Skinner, M.K. Environmentally Induced Epigenetic Transgenerational Inheritance of Ovarian Disease. PLoS ONE 2012, 7, e36129. [Google Scholar] [CrossRef] [PubMed]
- Manikkam, M.; Guerrero-Bosagna, C.; Tracey, R.; Haque, M.; Skinner, M.K. Transgenerational Actions of Environmental Compounds on Reproductive Disease and Identification of Epigenetic Biomarkers of Ancestral Exposures. PLoS ONE 2012, 7, e31901. [Google Scholar] [CrossRef] [PubMed]
- Bruner-Tran, K.L.; Osteen, K.G. Developmental exposure to TCDD reduces fertility and negatively affects pregnancy outcomes across multiple generations. Reprod. Toxicol. 2011, 31, 344–350. [Google Scholar] [CrossRef]
- Yu, K.; Zhang, X.; Tan, X.; Ji, M.; Chen, Y.; Tao, Y.; Liu, M.; Yu, Z. Transgenerational impairment of ovarian induced by 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) associated with Igf2 and H19 in adult female rat. Toxicology 2019, 428, 152311. [Google Scholar] [CrossRef]
- Yu, K.; Zhang, X.; Tan, X.; Ji, M.; Chen, Y.; Wan, Z.; Yu, Z. Multigenerational and transgenerational effects of 2,3,7,8-tetrachlorodibenzo-p-dioxin exposure on ovarian reserve and follicular development through AMH/AMHR2 pathway in adult female rats. Food Chem. Toxicol. 2020, 140, 111309. [Google Scholar] [CrossRef] [PubMed]
- Bruner-Tran, K.L.; Duleba, A.J.; Taylor, H.S.; Osteen, K.G. Developmental Toxicant Exposure Is Associated with Transgenerational Adenomyosis in a Murine Model. Biol. Reprod. 2016, 95, 73. [Google Scholar] [CrossRef] [PubMed]
- Bruner-Tran, K.L.; Ding, T.; Yeoman, K.B.; Archibong, A.; Arosh, J.A.; Osteen, K.G. Developmental Exposure of Mice to Dioxin Promotes Transgenerational Testicular Inflammation and an Increased Risk of Preterm Birth in Unexposed Mating Partners. PLoS ONE 2014, 9, e105084. [Google Scholar] [CrossRef]
- Sanabria, M.; Cucielo, M.S.; Guerra, M.T.; Borges, C.D.S.; Banzato, T.P.; Perobelli, J.E.; Leite, G.A.A.; Anselmo-Franci, J.A.; Kempinas, W.D.G. Sperm quality and fertility in rats after prenatal exposure to low doses of TCDD: A three-generation study. Reprod. Toxicol. 2016, 65, 29–38. [Google Scholar] [CrossRef]
- Murray, F.; Smith, F.; Nitschke, K.; Humiston, C.; Kociba, R.; Schwetz, B. Three-generation reproduction study of rats given 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) in the diet. Toxicol. Appl. Pharmacol. 1979, 50, 241–252. [Google Scholar] [CrossRef]
- Ding, T.; Mokshagundam, S.; Rinaudo, P.F.; Osteen, K.G.; Bruner-Tran, K.L. Paternal developmental toxicant exposure is associated with epigenetic modulation of sperm and placental Pgr and Igf2 in a mouse model. Biol. Reprod. 2018, 99, 864–876. [Google Scholar] [CrossRef]
- Rowlands, J.; Budinsky, R.; Aylward, L.; Faqi, A.; Carney, E. Sex ratio of the offspring of Sprague–Dawley rats exposed to 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) in utero and lactationally in a three-generation study. Toxicol. Appl. Pharmacol. 2006, 216, 29–33. [Google Scholar] [CrossRef]
- Ding, T.; Lambert, L.A.; Aronoff, D.M.; Osteen, K.G.; Bruner-Tran, K.L. Sex-Dependent Influence of Developmental Toxicant Exposure on Group B Streptococcus-Mediated Preterm Birth in a Murine Model. Reprod. Sci. 2018, 25, 662–673. [Google Scholar] [CrossRef]
- Prokopec, S.D.; Viluksela, M.; Miettinen, H.M.; Boutros, P.C.; Pohjanvirta, R. Transgenerational epigenetic and transcriptomic effects of 2,3,7,8-tetrachlorodibenzo-p-dioxin exposure in rat. Arch. Toxicol. 2020, 94, 1613–1624. [Google Scholar] [CrossRef] [PubMed]
- Garavaglia, E.; Sala, C.; Taccagni, G.; Traglia, M.; Barbieri, C.; Ferrari, S.; Candiani, M.; Panina-Bordignon, P.; Toniolo, D. Fertility Preservation in Endometriosis Patients: Anti-Müllerian Hormone Is a Reliable Marker of the Ovarian Follicle Density. Front. Surg. 2017, 4, 40. [Google Scholar] [CrossRef] [PubMed]
- Pan, M.-L.; Chen, L.-R.; Tsao, H.-M.; Chen, K.-H. Polycystic ovarian syndrome and the risk of subsequent primary ovarian insufficiency: A nationwide population-based study. Menopause 2017, 24, 803–809. [Google Scholar] [CrossRef] [PubMed]
- Horton, J.; Sterrenburg, M.; Lane, S.; Maheshwari, A.; Li, T.C.; Cheong, Y. Reproductive, obstetric, and perinatal outcomes of women with adenomyosis and endometriosis: A systematic review and meta-analysis. Hum. Reprod. Updat. 2019, 25, 593–633. [Google Scholar] [CrossRef]
- Rier, S.E.; Martin, D.; Bowman, R.E.; Dmowski, W.P.; Becker, J.L. Endometriosis in Rhesus Monkeys (Macaca mulatta) Following Chronic Exposure to 2,3,7,8-Tetrachlorodibenzo-p-dioxin. Toxicol. Sci. 1993, 21, 433–441. [Google Scholar] [CrossRef]
- Nayyar, T.; Bruner-Tran, K.L.; Piestrzeniewicz-Ulanska, D.; Osteen, K.G. Developmental exposure of mice to TCDD elicits a similar uterine phenotype in adult animals as observed in women with endometriosis. Reprod. Toxicol. 2007, 23, 326–336. [Google Scholar] [CrossRef] [PubMed]
- Bruner-Tran, K.L.; Resuehr, D.; Ding, T.; Lucas, J.A.; Osteen, K.G. The Role of Endocrine Disruptors in the Epigenetics of Reproductive Disease and Dysfunction: Potential Relevance to Humans. Curr. Obstet. Gynecol. Rep. 2012, 1, 116–123. [Google Scholar] [CrossRef] [PubMed]
- Igarashi, T.M.; Bruner-Tran, K.L.; Yeaman, G.R.; Lessey, B.A.; Edwards, D.P.; Eisenberg, E.; Osteen, K.G. Reduced expression of progesterone receptor-B in the endometrium of women with endometriosis and in cocultures of endometrial cells exposed to 2,3,7,8-tetrachlorodibenzo-p-dioxin. Fertil. Steril. 2005, 84, 67–74. [Google Scholar] [CrossRef]
- Bruner-Tran, K.L.; Ding, T.; Osteen, K.G. Dioxin and Endometrial Progesterone Resistance. Semin. Reprod. Med. 2010, 28, 059–068. [Google Scholar] [CrossRef]
- The Rotterdam ESHRE/ASRM-Sponsored PCOS Consensus Workshop Group. Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome (PCOS). Hum. Reprod. 2004, 19, 41–47. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, K.L.; Kryger-Baggesen, N.; Byskov, A.G.; Andersen, C.Y. Anti-Mullerian hormone initiates growth of human primordial follicles in vitro. Mol. Cell. Endocrinol. 2005, 234, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Jablonska, O.; Shi, Z.; Valdez, K.E.; Ting, A.Y.; Petroff, B.K. Temporal and anatomical sensitivities to the aryl hydrocarbon receptor agonist 2,3,7,8-tetrachlorodibenzo-p-dioxin leading to premature acyclicity with age in rats. Int. J. Androl. 2010, 33, 405–412. [Google Scholar] [CrossRef]
- Ikeda, M.; Tamura, M.; Yamashita, J.; Suzuki, C.; Tomita, T. Repeated in utero and lactational 2,3,7,8-tetrachlorodibenzo-p-dioxin exposure affects male gonads in offspring, leading to sex ratio changes in F2 progeny. Toxicol. Appl. Pharmacol. 2005, 206, 351–355. [Google Scholar] [CrossRef]
- Roberts, S.A.; Kaiser, U.B. Genetics in Endocrinology: Genetic etiologies of central precocious puberty and the role of imprinted genes. Eur. J. Endocrinol. 2020, 183, R107–R117. [Google Scholar] [CrossRef]
- Méar, L.; Herr, M.; Fauconnier, A.; Pineau, C.; Vialard, F. Polymorphisms and endometriosis: A systematic review and meta-analyses. Hum. Reprod. Update 2019, 26, 73–102. [Google Scholar] [CrossRef] [PubMed]
- Ajmal, N.; Khan, S.Z.; Shaikh, R. Polycystic ovary syndrome (PCOS) and genetic predisposition: A review article. Eur. J. Obstet. Gynecol. Reprod. Biol. X 2019, 3, 100060. [Google Scholar] [CrossRef] [PubMed]
- La Marca, A.; Mastellari, E. Fertility preservation for genetic diseases leading to premature ovarian insufficiency (POI). J. Assist. Reprod. Genet. 2021, 38, 759–777. [Google Scholar] [CrossRef] [PubMed]
- Bjerke, D.L.; Peterson, R.E. Reproductive toxicity of 2,3,7,8-tetrachlorodibenzo-p-dioxin in male rats: Different effects of in utero versus lactational exposure. Toxicol. Appl. Pharmacol. 1994, 127, 241–249. [Google Scholar] [CrossRef] [PubMed]
- Straub, R.H. Interaction of the endocrine system with inflammation: A function of energy and volume regulation. Arthritis Res. Ther. 2014, 16, 203. [Google Scholar] [CrossRef] [PubMed]
- Mai, X.; Dong, Y.; Xiang, L.; Er, Z. Maternal exposure to 2,3,7,8-tetrachlorodibenzo-p-dioxin suppresses male reproductive functions in their adulthood. Hum. Exp. Toxicol. 2020, 39, 890–905. [Google Scholar] [CrossRef]
- Zhang, X.; Ji, M.; Tan, X.; Yu, K.; Liu, X.; Li, N.; Yu, Z. Impairment of ovaries by 2,3,7,8-tetrachlorobenzo-p-dioxin (TCDD) exposure in utero associated with BMP15 and GDF9 in the female offspring rat. Toxicology 2018, 410, 16–25. [Google Scholar] [CrossRef]
- Johnson, K.; Passage, J.; Lin, H.; Sriram, S.; Budinsky, R.A. Dioxin male rat reproductive toxicity mode of action and relative potency of 2,3,7,8-tetrachlorodibenzo-p-dioxin and 2,3,7,8-tetrachlorodibenzofuran characterized by fetal pituitary and testis transcriptome profiling. Reprod. Toxicol. 2020, 93, 146–162. [Google Scholar] [CrossRef]
- Melekoglu, R.; Ciftci, O.; Cetin, A.; Basak, N.; Celik, E. The beneficial effects of Montelukast against 2,3,7,8-tetrachlorodibenzo-p-dioxin toxicity in female reproductive system in rats. Acta Cir. Bras. 2016, 31, 557–563. [Google Scholar] [CrossRef]
- Latchoumycandane, C.; Chitra, C.; Mathur, P. Induction of oxidative stress in rat epididymal sperm after exposure to 2,3,7,8-tetrachlorodibenzo-p-dioxin. Arch. Toxicol. 2002, 76, 113–118. [Google Scholar] [CrossRef]
- Latchoumycandane, C.; Chitra, K.; Mathur, P. The effect of 2,3,7,8-tetrachlorodibenzo-p-dioxin on the antioxidant system in mitochondrial and microsomal fractions of rat testis. Toxicology 2002, 171, 127–135. [Google Scholar] [CrossRef]
- Lettieri, G.; D’Agostino, G.; Mele, E.; Cardito, C.; Esposito, R.; Cimmino, A.; Giarra, A.; Trifuoggi, M.; Raimondo, S.; Notari, T.; et al. Discovery of the Involvement in DNA Oxidative Damage of Human Sperm Nuclear Basic Proteins of Healthy Young Men Living in Polluted Areas. Int. J. Mol. Sci. 2020, 21, 4198. [Google Scholar] [CrossRef]
- Lettieri, G.; Marra, F.; Moriello, C.; Prisco, M.; Notari, T.; Trifuoggi, M.; Giarra, A.; Bosco, L.; Montano, L.; Piscopo, M. Molecular Alterations in Spermatozoa of a Family Case Living in the Land of Fires. A First Look at Possible Transgenerational Effects of Pollutants. Int. J. Mol. Sci. 2020, 21, 6710. [Google Scholar] [CrossRef]
- Zhang, X.; Ji, M.; Tan, X.; Yu, K.; Xu, L.; Chen, G.; Yu, Z. Role of epigenetic regulation of Igf2 and H19 in 2,3,7,8-Tetrachlorobenzo-p-dioxin (TCDD)-induced ovarian toxicity in offspring rats. Toxicol. Lett. 2019, 311, 98–104. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.; Ohsako, S.; Ishimura, R.; Suzuki, J.S.; Tohyama, C. Exposure of Mouse Preimplantation Embryos to 2,3,7,8-Tetrachlorodibenzo-p-dioxin (TCDD) Alters the Methylation Status of Imprinted Genes H19 and Igf21. Biol. Reprod. 2004, 70, 1790–1797. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Chen, X.; Liu, Y.; Xie, Q.; Sun, Y.; Chen, J.; Leng, L.; Yan, H.; Zhao, B.; Tang, N. Ancestral TCDD exposure promotes epigenetic transgenerational inheritance of imprinted gene Igf2: Methylation status and DNMTs. Toxicol. Appl. Pharmacol. 2015, 289, 193–202. [Google Scholar] [CrossRef] [PubMed]
- Devillers, M.M.; Petit, F.; Giton, F.; Francois, C.M.; Juricek, L.; Coumoul, X.; Magre, S.; Cohen-Tannoudji, J.; Guigon, C.J. Age-dependent vulnerability of the ovary to AhR-mediated TCDD action before puberty: Evidence from mouse models. Chemosphere 2020, 258, 127361. [Google Scholar] [CrossRef] [PubMed]
- Reik, W.; Dean, W.; Walter, J. Epigenetic reprogramming in mammalian development. Science 2001, 293, 1089–1093. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, H.; Matsui, Y. Epigenetic events in mammalian germ-cell development: Reprogramming and beyond. Nat. Rev. Genet. 2008, 9, 129–140. [Google Scholar] [CrossRef] [PubMed]
- Guerrero-Bosagna, C.; Settles, M.; Lucker, B.; Skinner, M.K. Epigenetic Transgenerational Actions of Vinclozolin on Promoter Regions of the Sperm Epigenome. PLoS ONE 2010, 5, e13100. [Google Scholar] [CrossRef]
- Patrizi, B.; Siciliani de Cumis, M. TCDD Toxicity Mediated by Epigenetic Mechanisms. Int. J. Mol. Sci. 2018, 19, 12. [Google Scholar] [CrossRef]
- Lensu, S.; Tuomisto, J.T.; Tuomisto, J.; Viluksela, M.; Niittynen, M.; Pohjanvirta, R. Immediate and highly sensitive aversion response to a novel food item linked to AH receptor stimulation. Toxicol. Lett. 2011, 203, 252–257. [Google Scholar] [CrossRef]
- Pohjanvirta, R.; Mahiout, S. Aryl hydrocarbon receptor is indispensable for beta-naphthoflavone-induced novel food avoidance and may be involved in LiCl-triggered conditioned taste aversion in rats. Physiol. Behav. 2019, 204, 58–64. [Google Scholar] [CrossRef]
- Dixon, G.; Liao, Y.; Bay, L.K.; Matz, M.V. Role of gene body methylation in acclimatization and adaptation in a basal metazoan. Proc. Natl. Acad. Sci. USA 2018, 115, 13342–13346. [Google Scholar] [CrossRef] [PubMed]
- Arechederra, M.; Daian, F.; Yim, A.; Bazai, S.K.; Richelme, S.; Dono, R.; Saurin, A.J.; Habermann, B.H.; Maina, F. Hypermethylation of gene body CpG islands predicts high dosage of functional oncogenes in liver cancer. Nat. Commun. 2018, 9, 3164. [Google Scholar] [CrossRef] [PubMed]
- Azzi, S.; Abi Habib, W.; Netchine, I. Beckwith-Wiedemann and Russell-Silver Syndromes: From new molecular insights to the comprehension of imprinting regulation. Curr. Opin. Endocrinol. Diabetes Obes. 2014, 21, 30–38. [Google Scholar] [CrossRef] [PubMed]
- Peluso, C.; Fonseca, F.L.; Gastaldo, G.G.; Christofolini, D.M.; Cordts, E.B.; Barbosa, C.P.; Bianco, B. AMH and AMHR2 polymorphisms and AMH serum level can predict assisted reproduction outcomes: A cross-sectional study. Cell Physiol. Biochem. 2015, 35, 1401–1412. [Google Scholar] [CrossRef] [PubMed]
- Zhou, R.; Yu, S.M.Y.; Ge, W. Expression and functional characterization of intrafollicular GH–IGF system in the zebrafish ovary. Gen. Comp. Endocrinol. 2016, 232, 32–42. [Google Scholar] [CrossRef]
| F3 Females | Paternal Germline | Paternal and Maternal Germline | Maternal Germline |
|---|---|---|---|
| Fertility | ↓ [57] 2 | ||
| Ovarian weight | N [54] 1 | ↓ [58] 3 | |
| Ovarian primordial follicles | ↓ [55] 1 | ↓ [58] 3 | |
| Granulosa cell apoptosis around growing follicles | ↑ [59] 3 | ||
| AMH concentration | N [59] 3 | ||
| Earlier pubertal maturation | ↑ [54,56] 1 | ↑ [58] 3 | |
| Estrous cycle abnormalities | ↑ [58] 3 | ||
| Polycystic ovaries | ↑ [54] 1 | ||
| Adenomyosis | ↑ [60] 2 | ||
| F3 Males | Paternal Germline | Paternal and Maternal Germline | Maternal Germline |
| Fertility | ↓ [61] 2 | ||
| Testosterone concentration | ↓ (ns) [61] 2 ↓ (ns) [62] 4 | ↓ [56] 1 | |
| LH concentration | N [61] 2 N [62] 4 | N [56] 1 | |
| Sperm number | N [61] 2 | ||
| Normal sperm morphology | ↓ [61] 2 N [62] 4 | ||
| Epididymal sperm concentration and motility | N [62] 4 | N [56] 1 | |
| Epididymis and prostate weight | N [54] 1 | ||
| Testis and prostate diseases/abnormalities | N [54] 1 | ||
| Pubertal development | N [54] 1 | ||
| F3 Pregnancies | Paternal Germline | Paternal and Maternal Germline | Maternal Germline |
| Premature birth | ↑ [61] 2 | ↑ [57] 2 | |
| Litter size at birth | ↓ [63] 6 N [54] 1 | ||
| Number of implanted embryos and number of implanted embryos per corpus luteum | ↓ [62] 4 | ||
| Placental weight | ↓ [64] 5 | ||
| Pup weight | ↓ [64] 5 | ||
| Gestation survival index | ↓ [63] 6 | ||
| Male/female sex ratio | N [65] 6 N [54] 1 | ||
| Ascending GBS infection during pregnancy | Maternal health and gestation length N, neonatal survival ↓ [66] 5 | 100% maternal and fetal mortality [66] 5 | |
| Infertility and premature birth after latent MPV | ↑ [57] 2 |
| F3 Females | Paternal Germline | Paternal and Maternal Germline | Maternal Germline |
|---|---|---|---|
| AMH mRNA ↓ and AMHR2 mRNA ↑ in the ovary [58] 1 | |||
| Igf2 mRNA ↓ and H19 mRNA ↑ in the ovary [66] 1 | |||
| F3 Males | Paternal Germline | Paternal and Maternal Germline | Maternal Germline |
| DMR epimutations in testes [67] 2 | DMR epimutations in sperm [54] 4 | ||
| DMR epimutations in sperm [64] 3 | Disease-specific DMR epimutations in sperm [10] 4 | ||
| DMR epimutations in male-derived placentae [64] 3 | |||
| Pgr, Igf2 and H19 mRNA ↓ in male-derived placentae [64] 3 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gaspari, L.; Paris, F.; Kalfa, N.; Soyer-Gobillard, M.-O.; Sultan, C.; Hamamah, S. Experimental Evidence of 2,3,7,8-Tetrachlordibenzo-p-Dioxin (TCDD) Transgenerational Effects on Reproductive Health. Int. J. Mol. Sci. 2021, 22, 9091. https://doi.org/10.3390/ijms22169091
Gaspari L, Paris F, Kalfa N, Soyer-Gobillard M-O, Sultan C, Hamamah S. Experimental Evidence of 2,3,7,8-Tetrachlordibenzo-p-Dioxin (TCDD) Transgenerational Effects on Reproductive Health. International Journal of Molecular Sciences. 2021; 22(16):9091. https://doi.org/10.3390/ijms22169091
Chicago/Turabian StyleGaspari, Laura, Françoise Paris, Nicolas Kalfa, Marie-Odile Soyer-Gobillard, Charles Sultan, and Samir Hamamah. 2021. "Experimental Evidence of 2,3,7,8-Tetrachlordibenzo-p-Dioxin (TCDD) Transgenerational Effects on Reproductive Health" International Journal of Molecular Sciences 22, no. 16: 9091. https://doi.org/10.3390/ijms22169091
APA StyleGaspari, L., Paris, F., Kalfa, N., Soyer-Gobillard, M.-O., Sultan, C., & Hamamah, S. (2021). Experimental Evidence of 2,3,7,8-Tetrachlordibenzo-p-Dioxin (TCDD) Transgenerational Effects on Reproductive Health. International Journal of Molecular Sciences, 22(16), 9091. https://doi.org/10.3390/ijms22169091





