Stress Adaptation and the Brainstem with Focus on Corticotropin-Releasing Hormone
Abstract
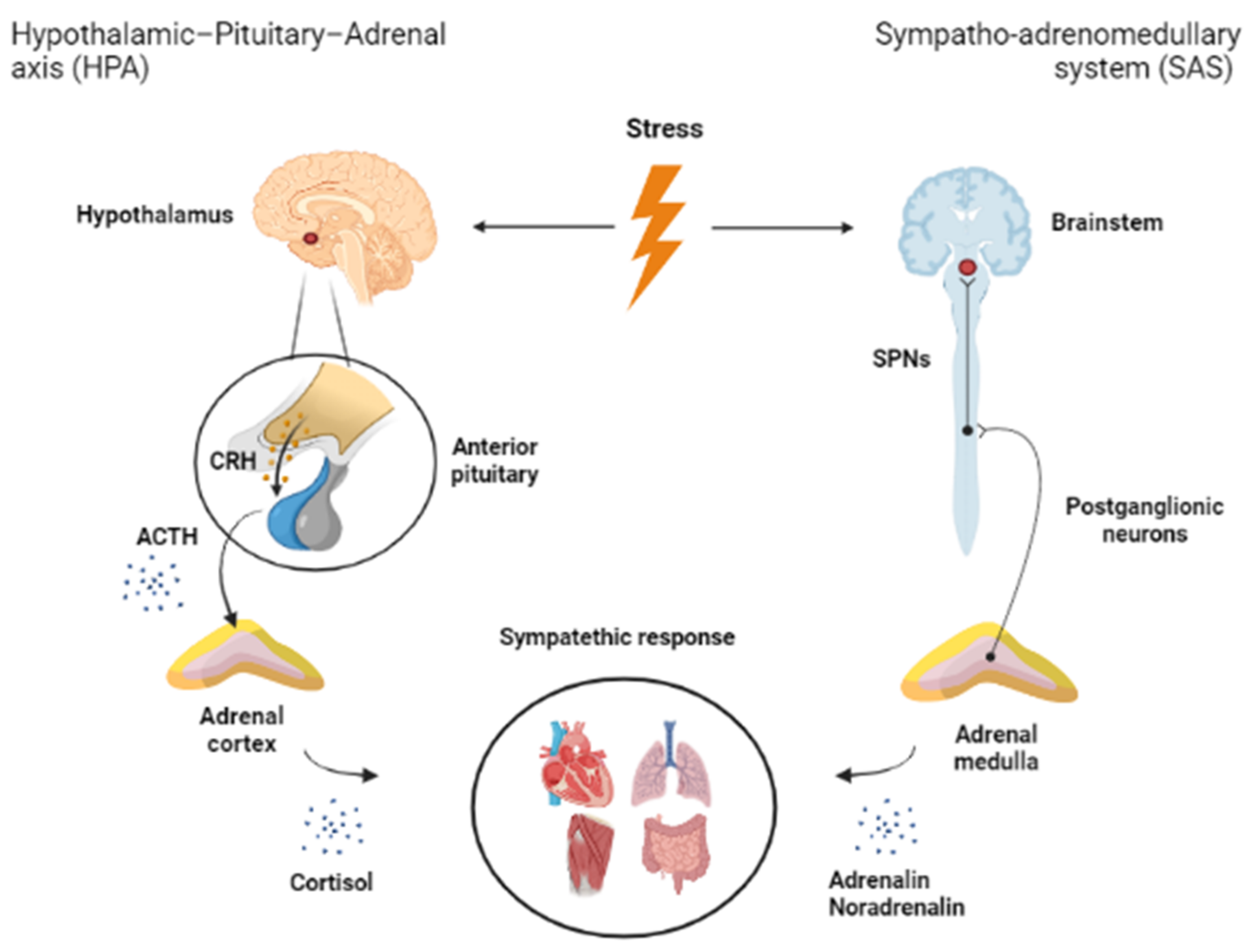
1. Introduction
2. Acute or Chronic Stress
3. The PVN-CRH Neurons and Stress Adaptation
4. Function of Brainstem in Stress
4.1. Some Anatomical Considerations
4.2. Neurotransmitters
4.2.1. Inhibition: GABA
4.2.2. Excitation: Glutamate
4.2.3. Monoamines
Serotonin
Catecholamines
Catecholamines: Noradrenaline (NA) and Locus Coeruleus (LC)
Catecholamines: Dopamine (DA)
5. CRH Cells in the Brainstem
6. Assumed Function of Each CRH-Containing Brainstem Nuclei in Stress
7. Conclusions
Funding
Conflicts of Interest
References
- Anisman, H.; Merali, Z. Understanding stress: Characteristics and caveats. Alcohol. Res. Health 1999, 23, 241–249. [Google Scholar] [PubMed]
- Hayley, S.; Borowski, T.; Merali, Z.; Anisman, H. Central monoamine activity in genetically distinct strains of mice following a psychogenic stressor: Effects of predator exposure. Brain Res. 2001, 892, 293–300. [Google Scholar] [CrossRef]
- Myers, B.; Scheimann, J.R.; Franco-Villanueva, A.; Herman, J.P. Ascending mechanisms of stress integration: Implications for brainstem regulation of neuroendocrine and behavioral stress responses. Neurosci. Biobehav. Rev. 2017, 74, 366–375. [Google Scholar] [CrossRef] [PubMed]
- Buller, K.M. Neuroimmune stress responses: Reciprocal connections between the hypothalamus and the brainstem. Stress 2003, 6, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Aguilera, G.; Liu, Y. The molecular physiology of CRH neurons. Front. Neuroendocrinol. 2012, 33, 67–84. [Google Scholar] [CrossRef] [PubMed]
- Herman, J.P.; McKlveen, J.M.; Ghosal, S.; Kopp, B.; Wulsin, A.; Makinson, R.; Scheimann, J.; Myers, B. Regulation of the Hypothalamic-Pituitary-Adrenocortical Stress Response. Compr. Physiol. 2016, 6, 603–621. [Google Scholar] [CrossRef]
- Goncharova, N.D. Stress responsiveness of the hypothalamic-pituitary-adrenal axis: Age-related features of the vasopressinergic regulation. Front. Endocrinol. 2013, 4, 26. [Google Scholar] [CrossRef]
- Gjerstad, J.K.; Lightman, S.L.; Spiga, F. Role of glucocorticoid negative feedback in the regulation of HPA axis pulsatility. Stress 2018, 21, 403–416. [Google Scholar] [CrossRef]
- Tasker, J.G.; Herman, J.P. Mechanisms of rapid glucocorticoid feedback inhibition of the hypothalamic-pituitary-adrenal axis. Stress 2011, 14, 398–406. [Google Scholar] [CrossRef]
- Henry, J.P. Biological basis of the stress response. Integr. Physiol. Behav. Sci. 1992, 27, 66–83. [Google Scholar] [CrossRef]
- Jansen, A.S.; Nguyen, X.V.; Karpitskiy, V.; Mettenleiter, T.C.; Loewy, A.D. Central command neurons of the sympathetic nervous system: Basis of the fight-or-flight response. Science 1995, 270, 644–646. [Google Scholar] [CrossRef]
- Deuchars, S.A.; Lall, V.K. Sympathetic preganglionic neurons: Properties and inputs. Compr. Physiol. 2015, 5, 829–869. [Google Scholar] [CrossRef] [PubMed]
- Tucker, D.C.; Saper, C.B. Specificity of spinal projections from hypothalamic and brainstem areas which innervate sympathetic preganglionic neurons. Brain Res. 1985, 360, 159–164. [Google Scholar] [CrossRef]
- Fuzesi, T.; Daviu, N.; Wamsteeker Cusulin, J.I.; Bonin, R.P.; Bains, J.S. Hypothalamic CRH neurons orchestrate complex behaviours after stress. Nat. Commun. 2016, 7, 11937. [Google Scholar] [CrossRef] [PubMed]
- McCarty, R. Learning about stress: Neural, endocrine and behavioral adaptations. Stress 2016, 19, 449–475. [Google Scholar] [CrossRef] [PubMed]
- Agorastos, A.; Chrousos, G.P. The neuroendocrinology of stress: The stress-related continuum of chronic disease development. Mol. Psychiatry 2021. [Google Scholar] [CrossRef]
- Musazzi, L.; Tornese, P.; Sala, N.; Popoli, M. Acute or Chronic? A Stressful Question. Trends Neurosci. 2017, 40, 525–535. [Google Scholar] [CrossRef]
- Aguilera, G. Regulation of pituitary ACTH secretion during chronic stress. Front. Neuroendocrinol. 1994, 15, 321–350. [Google Scholar] [CrossRef]
- Chen, J.; Young, S.; Subburaju, S.; Sheppard, J.; Kiss, A.; Atkinson, H.; Wood, S.; Lightman, S.; Serradeil-Le Gal, C.; Aguilera, G. Vasopressin does not mediate hypersensitivity of the hypothalamic pituitary adrenal axis during chronic stress. Ann. N. Y. Acad. Sci. 2008, 1148, 349–359. [Google Scholar] [CrossRef]
- Makara, G.B.; Mergl, Z.; Zelena, D. The role of vasopressin in hypothalamo-pituitary-adrenal axis activation during stress: An assessment of the evidence. Ann. N. Y. Acad Sci. 2004, 1018, 151–161. [Google Scholar] [CrossRef]
- Zelena, D.; Pinter, O.; Balazsfi, D.G.; Langnaese, K.; Richter, K.; Landgraf, R.; Makara, G.B.; Engelmann, M. Vasopressin signaling at brain level controls stress hormone release: The vasopressin-deficient Brattleboro rat as a model. Amino Acids 2015, 47, 2245–2253. [Google Scholar] [CrossRef]
- Zelena, D.; Filaretova, L. Stress Adaptation with Special Emphasis on Gastric Erosion: Is Stress a Bad Guy or a Good Guy? In Chronic Stress and Health; Nova Science Publisher: Hauppauge, NY, USA, 2017; Volume 4, pp. 65–88. [Google Scholar]
- Rohleder, N. Stress and inflammation—The need to address the gap in the transition between acute and chronic stress effects. Psychoneuroendocrinology 2019, 105, 164–171. [Google Scholar] [CrossRef]
- Swanson, L.W.; Sawchenko, P.E.; Rivier, J.; Vale, W.W. Organization of ovine corticotropin-releasing factor immunoreactive cells and fibers in the rat brain: An immunohistochemical study. Neuroendocrinology 1983, 36, 165–186. [Google Scholar] [CrossRef]
- Carlin, K.M.; Vale, W.W.; Bale, T.L. Vital functions of corticotropin-releasing factor (CRF) pathways in maintenance and regulation of energy homeostasis. Proc. Natl. Acad. Sci. USA 2006, 103, 3462–3467. [Google Scholar] [CrossRef]
- Wang, X.D.; Chen, Y.; Wolf, M.; Wagner, K.V.; Liebl, C.; Scharf, S.H.; Harbich, D.; Mayer, B.; Wurst, W.; Holsboer, F.; et al. Forebrain CRHR1 deficiency attenuates chronic stress-induced cognitive deficits and dendritic remodeling. Neurobiol. Dis. 2011, 42, 300–310. [Google Scholar] [CrossRef]
- Ma, X.M.; Levy, A.; Lightman, S.L. Rapid changes in heteronuclear RNA for corticotrophin-releasing hormone and arginine vasopressin in response to acute stress. J. Endocrinol. 1997, 152, 81–89. [Google Scholar] [CrossRef]
- Aguilera, G. Corticotropin releasing hormone, receptor regulation and the stress response. Trends Endocrinol. Metab. 1998, 9, 329–336. [Google Scholar] [CrossRef]
- Ma, X.M.; Levy, A.; Lightman, S.L. Emergence of an isolated arginine vasopressin (AVP) response to stress after repeated restraint: A study of both AVP and corticotropin-releasing hormone messenger ribonucleic acid (RNA) and heteronuclear RNA. Endocrinology 1997, 138, 4351–4357. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Shanks, N.; Windle, R.J.; Perks, P.; Wood, S.; Ingram, C.D.; Lightman, S.L. The hypothalamic-pituitary-adrenal axis response to endotoxin is attenuated during lactation. J. Neuroendocrinol. 1999, 11, 857–865. [Google Scholar] [CrossRef] [PubMed]
- Chung, S.; Son, G.H.; Kim, K. Circadian rhythm of adrenal glucocorticoid: Its regulation and clinical implications. Biochim. Biophys Acta 2011, 1812, 581–591. [Google Scholar] [CrossRef] [PubMed]
- Watts, A.G.; Tanimura, S.; Sanchez-Watts, G. Corticotropin-releasing hormone and arginine vasopressin gene transcription in the hypothalamic paraventricular nucleus of unstressed rats: Daily rhythms and their interactions with corticosterone. Endocrinology 2004, 145, 529–540. [Google Scholar] [CrossRef]
- Row, B.W.; Dohanich, G.P. Post-training administration of corticotropin-releasing hormone (CRH) enhances retention of a spatial memory through a noradrenergic mechanism in male rats. Neurobiol. Learn. Mem 2008, 89, 370–378. [Google Scholar] [CrossRef] [PubMed]
- Radulovic, J.; Ruhmann, A.; Liepold, T.; Spiess, J. Modulation of learning and anxiety by corticotropin-releasing factor (CRF) and stress: Differential roles of CRF receptors 1 and 2. J. Neurosci. 1999, 19, 5016–5025. [Google Scholar] [CrossRef]
- Blank, T.; Nijholt, I.; Eckart, K.; Spiess, J. Priming of long-term potentiation in mouse hippocampus by corticotropin-releasing factor and acute stress: Implications for hippocampus-dependent learning. J. Neurosci. 2002, 22, 3788–3794. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Dube, C.M.; Rice, C.J.; Baram, T.Z. Rapid loss of dendritic spines after stress involves derangement of spine dynamics by corticotropin-releasing hormone. J. Neurosci. 2008, 28, 2903–2911. [Google Scholar] [CrossRef]
- Conrad, C.D.; McLaughlin, K.J.; Harman, J.S.; Foltz, C.; Wieczorek, L.; Lightner, E.; Wright, R.L. Chronic glucocorticoids increase hippocampal vulnerability to neurotoxicity under conditions that produce CA3 dendritic retraction but fail to impair spatial recognition memory. J. Neurosci. 2007, 27, 8278–8285. [Google Scholar] [CrossRef] [PubMed]
- Conrad, C.D. What is the functional significance of chronic stress-induced CA3 dendritic retraction within the hippocampus? Behav. Cogn Neurosci. Rev. 2006, 5, 41–60. [Google Scholar] [CrossRef]
- Maniam, J.; Morris, M.J. The link between stress and feeding behaviour. Neuropharmacology 2012, 63, 97–110. [Google Scholar] [CrossRef]
- Ulrich-Lai, Y.M.; Ryan, K.K. Neuroendocrine circuits governing energy balance and stress regulation: Functional overlap and therapeutic implications. Cell Metab. 2014, 19, 910–925. [Google Scholar] [CrossRef]
- Ulrich-Lai, Y.M.; Herman, J.P. Neural regulation of endocrine and autonomic stress responses. Nat. Rev. Neurosci. 2009, 10, 397–409. [Google Scholar] [CrossRef]
- Melnick, I.; Krishtal, O.A.; Colmers, W.F. Integration of energy homeostasis and stress by parvocellular neurons in rat hypothalamic paraventricular nucleus. J. Physiol. 2020, 598, 1073–1092. [Google Scholar] [CrossRef]
- Yuan, Y.; Wu, W.; Chen, M.; Cai, F.; Fan, C.; Shen, W.; Sun, W.; Hu, J. Reward Inhibits Paraventricular CRH Neurons to Relieve Stress. Curr. Biol. 2019, 29, 1243–1251.e4. [Google Scholar] [CrossRef]
- Foster, M.T.; Warne, J.P.; Ginsberg, A.B.; Horneman, H.F.; Pecoraro, N.C.; Akana, S.F.; Dallman, M.F. Palatable foods, stress, and energy stores sculpt corticotropin-releasing factor, adrenocorticotropin, and corticosterone concentrations after restraint. Endocrinology 2009, 150, 2325–2333. [Google Scholar] [CrossRef]
- Laugero, K.D.; Bell, M.E.; Bhatnagar, S.; Soriano, L.; Dallman, M.F. Sucrose ingestion normalizes central expression of corticotropin-releasing-factor messenger ribonucleic acid and energy balance in adrenalectomized rats: A glucocorticoid-metabolic-brain axis? Endocrinology 2001, 142, 2796–2804. [Google Scholar] [CrossRef]
- Nemeroff, C.B.; Widerlov, E.; Bissette, G.; Walleus, H.; Karlsson, I.; Eklund, K.; Kilts, C.D.; Loosen, P.T.; Vale, W. Elevated concentrations of CSF corticotropin-releasing factor-like immunoreactivity in depressed patients. Science 1984, 226, 1342–1344. [Google Scholar] [CrossRef] [PubMed]
- Jokinen, J.; Ouda, J.; Nordstrom, P. Noradrenergic function and HPA axis dysregulation in suicidal behaviour. Psychoneuroendocrinology 2010, 35, 1536–1542. [Google Scholar] [CrossRef] [PubMed]
- Basinger, H.; Hogg, J.P. Neuroanatomy, Brainstem; StatPearls: Treasure Island, FL, USA, 2021. [Google Scholar]
- Littlejohn, E.L.; Fedorchak, S.; Boychuk, C.R. Sex-steroid-dependent plasticity of brain-stem autonomic circuits. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2020, 319, R60–R68. [Google Scholar] [CrossRef] [PubMed]
- Suudhof, T.C. Neurotransmitter release. Handb Exp. Pharmacol. 2008, 184, 1–21. [Google Scholar] [CrossRef]
- Jahn, R.; Hell, J.; Maycox, P.R. Synaptic vesicles: Key organelles involved in neurotransmission. J. Physiol. 1990, 84, 128–133. [Google Scholar]
- Bowery, N.G.; Smart, T.G. GABA and glycine as neurotransmitters: A brief history. Br. J. Pharmacol. 2006, 147 (Suppl. S1), S109–S119. [Google Scholar] [CrossRef]
- Ross, J.A.; Van Bockstaele, E.J. The Locus Coeruleus- Norepinephrine System in Stress and Arousal: Unraveling Historical, Current, and Future Perspectives. Front. Psychiatry 2020, 11, 601519. [Google Scholar] [CrossRef]
- Vizi, E.S.; Labos, E. Non-synaptic interactions at presynaptic level. Prog. Neurobiol. 1991, 37, 145–163. [Google Scholar] [CrossRef]
- Leng, G.; Ludwig, M. Neurotransmitters and peptides: Whispered secrets and public announcements. J. Physiol. 2008, 586, 5625–5632. [Google Scholar] [CrossRef]
- McBain, C.J.; Fisahn, A. Interneurons unbound. Nat. Rev. Neurosci. 2001, 2, 11–23. [Google Scholar] [CrossRef] [PubMed]
- Scalettar, B.A. How neurosecretory vesicles release their cargo. Neuroscientist 2006, 12, 164–176. [Google Scholar] [CrossRef] [PubMed]
- Hrabovszky, E.; Wittmann, G.; Turi, G.F.; Liposits, Z.; Fekete, C. Hypophysiotropic thyrotropin-releasing hormone and corticotropin-releasing hormone neurons of the rat contain vesicular glutamate transporter-2. Endocrinology 2005, 146, 341–347. [Google Scholar] [CrossRef] [PubMed]
- Partridge, J.G.; Forcelli, P.A.; Luo, R.; Cashdan, J.M.; Schulkin, J.; Valentino, R.J.; Vicini, S. Stress increases GABAergic neurotransmission in CRF neurons of the central amygdala and bed nucleus stria terminalis. Neuropharmacology 2016, 107, 239–250. [Google Scholar] [CrossRef] [PubMed]
- Obata, K. Synaptic inhibition and gamma-aminobutyric acid in the mammalian central nervous system. Proc. Jpn Acad. Ser. B Phys. Biol. Sci. 2013, 89, 139–156. [Google Scholar] [CrossRef]
- Nuss, P. Anxiety disorders and GABA neurotransmission: A disturbance of modulation. Neuropsychiatr Dis. Treat. 2015, 11, 165–175. [Google Scholar] [CrossRef] [PubMed]
- Riemann, D.; Nissen, C.; Palagini, L.; Otte, A.; Perlis, M.L.; Spiegelhalder, K. The neurobiology, investigation, and treatment of chronic insomnia. Lancet Neurol. 2015, 14, 547–558. [Google Scholar] [CrossRef]
- Schwartz, R.D. The GABAA receptor-gated ion channel: Biochemical and pharmacological studies of structure and function. Biochem. Pharmacol. 1988, 37, 3369–3375. [Google Scholar] [CrossRef]
- Dedic, N.; Kuhne, C.; Jakovcevski, M.; Hartmann, J.; Genewsky, A.J.; Gomes, K.S.; Anderzhanova, E.; Pohlmann, M.L.; Chang, S.; Kolarz, A.; et al. Chronic CRH depletion from GABAergic, long-range projection neurons in the extended amygdala reduces dopamine release and increases anxiety. Nat. Neurosci. 2018, 21, 803–807. [Google Scholar] [CrossRef] [PubMed]
- Somogyi, J. Functional significance of co-localization of GABA and Glu in nerve terminals: A hypothesis. Curr. Top. Med. Chem. 2006, 6, 969–973. [Google Scholar] [CrossRef]
- Wagner, S.; Castel, M.; Gainer, H.; Yarom, Y. GABA in the mammalian suprachiasmatic nucleus and its role in diurnal rhythmicity. Nature 1997, 387, 598–603. [Google Scholar] [CrossRef]
- Cullinan, W.E.; Ziegler, D.R.; Herman, J.P. Functional role of local GABAergic influences on the HPA axis. Brain Struct. Funct. 2008, 213, 63–72. [Google Scholar] [CrossRef]
- Fogaca, M.V.; Duman, R.S. Cortical GABAergic Dysfunction in Stress and Depression: New Insights for Therapeutic Interventions. Front. Cell Neurosci. 2019, 13, 87. [Google Scholar] [CrossRef]
- Marques de Souza, L.; Franci, C.R. GABAergic mediation of stress-induced secretion of corticosterone and oxytocin, but not prolactin, by the hypothalamic paraventricular nucleus. Life Sci. 2008, 83, 686–692. [Google Scholar] [CrossRef]
- Sarkar, J.; Wakefield, S.; MacKenzie, G.; Moss, S.J.; Maguire, J. Neurosteroidogenesis is required for the physiological response to stress: Role of neurosteroid-sensitive GABAA receptors. J. Neurosci. 2011, 31, 18198–18210. [Google Scholar] [CrossRef]
- Gottesmann, C. GABA mechanisms and sleep. Neuroscience 2002, 111, 231–239. [Google Scholar] [CrossRef]
- Nemeroff, C.B. The role of GABA in the pathophysiology and treatment of anxiety disorders. Psychopharmacol Bull. 2003, 37, 133–146. [Google Scholar] [PubMed]
- Petty, F. GABA and mood disorders: A brief review and hypothesis. J. Affect. Disord 1995, 34, 275–281. [Google Scholar] [CrossRef]
- Zhu, H.; Karolewicz, B.; Nail, E.; Stockmeier, C.A.; Szebeni, K.; Ordway, G.A. Normal [3H]flunitrazepam binding to GABAA receptors in the locus coeruleus in major depression and suicide. Brain Res. 2006, 1125, 138–146. [Google Scholar] [CrossRef] [PubMed]
- Biggio, G.; Corda, M.G.; Concas, A.; Demontis, G.; Rossetti, Z.; Gessa, G.L. Rapid changes in GABA binding induced by stress in different areas of the rat brain. Brain Res. 1981, 229, 363–369. [Google Scholar] [CrossRef]
- Elekes, I.; Patthy, A.; Lang, T.; Palkovits, M. Concentrations of GABA and glycine in discrete brain nuclei. Stress-induced changes in the levels of inhibitory amino acids. Neuropharmacology 1986, 25, 703–709. [Google Scholar] [CrossRef]
- DiMicco, J.A.; Sarkar, S.; Zaretskaia, M.V.; Zaretsky, D.V. Stress-induced cardiac stimulation and fever: Common hypothalamic origins and brainstem mechanisms. Auton. Neurosci. 2006, 126–127, 106–119. [Google Scholar] [CrossRef] [PubMed]
- Kunos, G.; Varga, K. The tachycardia associated with the defense reaction involves activation of both GABAA and GABAB receptors in the nucleus tractus solitarii. Clin. Exp. Hypertens 1995, 17, 91–100. [Google Scholar] [CrossRef]
- Horvath, H.R.; Fazekas, C.L.; Balazsfi, D.; Jain, S.K.; Haller, J.; Zelena, D. Contribution of Vesicular Glutamate Transporters to Stress Response and Related Psychopathologies: Studies in VGluT3 Knockout Mice. Cell Mol. Neurobiol. 2018, 38, 37–52. [Google Scholar] [CrossRef] [PubMed]
- Zelena, D.; Mergl, Z.; Makara, G.B. Glutamate agonists activate the hypothalamic-pituitary-adrenal axis through hypothalamic paraventricular nucleus but not through vasopressinerg neurons. Brain Res. 2005, 1031, 185–193. [Google Scholar] [CrossRef]
- Zelena, D.; Makara, G.B.; Jezova, D. Simultaneous blockade of two glutamate receptor subtypes (NMDA and AMPA) results in stressor-specific inhibition of prolactin and corticotropin release. Neuroendocrinology 1999, 69, 316–323. [Google Scholar] [CrossRef]
- Kusek, M.; Tokarski, K.; Hess, G. Repeated restraint stress enhances glutamatergic transmission in the paraventricular nucleus of the rat hypothalamus. J. Physiol. Pharmacol. 2013, 64, 565–570. [Google Scholar]
- Ziegler, D.R.; Edwards, M.R.; Ulrich-Lai, Y.M.; Herman, J.P.; Cullinan, W.E. Brainstem origins of glutamatergic innervation of the rat hypothalamic paraventricular nucleus. J. Comp. Neurol. 2012, 520, 2369–2394. [Google Scholar] [CrossRef]
- Zheng, H.; Stornetta, R.L.; Agassandian, K.; Rinaman, L. Glutamatergic phenotype of glucagon-like peptide 1 neurons in the caudal nucleus of the solitary tract in rats. Brain Struct. Funct. 2015, 220, 3011–3022. [Google Scholar] [CrossRef]
- Kinzig, K.P.; D’Alessio, D.A.; Herman, J.P.; Sakai, R.R.; Vahl, T.P.; Figueiredo, H.F.; Murphy, E.K.; Seeley, R.J. CNS glucagon-like peptide-1 receptors mediate endocrine and anxiety responses to interoceptive and psychogenic stressors. J. Neurosci. 2003, 23, 6163–6170. [Google Scholar] [CrossRef] [PubMed]
- Larsen, P.J.; Tang-Christensen, M.; Jessop, D.S. Central administration of glucagon-like peptide-1 activates hypothalamic neuroendocrine neurons in the rat. Endocrinology 1997, 138, 4445–4455. [Google Scholar] [CrossRef]
- Nakade, Y.; Tsukamoto, K.; Pappas, T.N.; Takahashi, T. Central glucagon like peptide-1 delays solid gastric emptying via central CRF and peripheral sympathetic pathway in rats. Brain Res. 2006, 1111, 117–121. [Google Scholar] [CrossRef]
- Moller, C.; Sommer, W.; Thorsell, A.; Rimondini, R.; Heilig, M. Anxiogenic-like action of centrally administered glucagon-like peptide-1 in a punished drinking test. Prog. Neuropsychopharmacol. Biol. Psychiatry 2002, 26, 119–122. [Google Scholar] [CrossRef]
- Venzala, E.; Garcia-Garcia, A.L.; Elizalde, N.; Tordera, R.M. Social vs. environmental stress models of depression from a behavioural and neurochemical approach. Eur. Neuropsychopharmacol. 2013, 23, 697–708. [Google Scholar] [CrossRef] [PubMed]
- Fodor, A.; Kovacs, K.B.; Balazsfi, D.; Klausz, B.; Pinter, O.; Demeter, K.; Daviu, N.; Rabasa, C.; Rotllant, D.; Nadal, R.; et al. Depressive- and anxiety-like behaviors and stress-related neuronal activation in vasopressin-deficient female Brattleboro rats. Physiol. Behav. 2016, 158, 100–111. [Google Scholar] [CrossRef]
- Lkhagvasuren, B.; Nakamura, Y.; Oka, T.; Sudo, N.; Nakamura, K. Social defeat stress induces hyperthermia through activation of thermoregulatory sympathetic premotor neurons in the medullary raphe region. Eur. J. Neurosci. 2011, 34, 1442–1452. [Google Scholar] [CrossRef]
- Kataoka, N.; Hioki, H.; Kaneko, T.; Nakamura, K. Psychological stress activates a dorsomedial hypothalamus-medullary raphe circuit driving brown adipose tissue thermogenesis and hyperthermia. Cell Metab. 2014, 20, 346–358. [Google Scholar] [CrossRef]
- Machado, N.L.S.; Abbott, S.B.G.; Resch, J.M.; Zhu, L.; Arrigoni, E.; Lowell, B.B.; Fuller, P.M.; Fontes, M.A.P.; Saper, C.B. A Glutamatergic Hypothalamomedullary Circuit Mediates Thermogenesis, but Not Heat Conservation, during Stress-Induced Hyperthermia. Curr. Biol. 2018, 28, 2291–2301.e5. [Google Scholar] [CrossRef] [PubMed]
- Dygalo, N.N.; Kalinina, T.S.; Shishkina, G.T. Stress-induced expression pattern of glutamate signaling genes associated with anhedonia. Stress 2020, 23, 700–707. [Google Scholar] [CrossRef] [PubMed]
- Stelly, C.E.; Pomrenze, M.B.; Cook, J.B.; Morikawa, H. Repeated social defeat stress enhances glutamatergic synaptic plasticity in the VTA and cocaine place conditioning. Elife 2016, 5, e15448. [Google Scholar] [CrossRef] [PubMed]
- Shinohara, F.; Asaoka, Y.; Kamii, H.; Minami, M.; Kaneda, K. Stress augments the rewarding memory of cocaine via the activation of brainstem-reward circuitry. Addict. Biol. 2019, 24, 509–521. [Google Scholar] [CrossRef]
- Daftary, S.S.; Panksepp, J.; Dong, Y.; Saal, D.B. Stress-induced, glucocorticoid-dependent strengthening of glutamatergic synaptic transmission in midbrain dopamine neurons. Neurosci. Lett. 2009, 452, 273–276. [Google Scholar] [CrossRef] [PubMed]
- Hornung, J.P. The human raphe nuclei and the serotonergic system. J. Chem. Neuroanat. 2003, 26, 331–343. [Google Scholar] [CrossRef]
- Felten, D.L.; Sladek, J.R., Jr. Monoamine distribution in primate brain V. Monoaminergic nuclei: Anatomy, pathways and local organization. Brain Res. Bull. 1983, 10, 171–284. [Google Scholar] [CrossRef]
- Larsen, P.J.; Hay-Schmidt, A.; Vrang, N.; Mikkelsen, J.D. Origin of projections from the midbrain raphe nuclei to the hypothalamic paraventricular nucleus in the rat: A combined retrograde and anterograde tracing study. Neuroscience 1996, 70, 963–988. [Google Scholar] [CrossRef]
- Hermann, D.M.; Luppi, P.H.; Peyron, C.; Hinckel, P.; Jouvet, M. Afferent projections to the rat nuclei raphe magnus, raphe pallidus and reticularis gigantocellularis pars alpha demonstrated by iontophoretic application of choleratoxin (subunit b). J. Chem. Neuroanat 1997, 13, 1–21. [Google Scholar] [CrossRef]
- Vertes, R.P.; Fortin, W.J.; Crane, A.M. Projections of the median raphe nucleus in the rat. J. Comp. Neurol. 1999, 407, 555–582. [Google Scholar] [CrossRef]
- Vertes, R.P. A PHA-L analysis of ascending projections of the dorsal raphe nucleus in the rat. J. Comp. Neurol. 1991, 313, 643–668. [Google Scholar] [CrossRef]
- Zhang, S.; Lv, F.; Yuan, Y.; Fan, C.; Li, J.; Sun, W.; Hu, J. Whole-Brain Mapping of Monosynaptic Afferent Inputs to Cortical CRH Neurons. Front. Neurosci. 2019, 13, 565. [Google Scholar] [CrossRef]
- Linthorst, A.C.; Penalva, R.G.; Flachskamm, C.; Holsboer, F.; Reul, J.M. Forced swim stress activates rat hippocampal serotonergic neurotransmission involving a corticotropin-releasing hormone receptor-dependent mechanism. Eur. J. Neurosci. 2002, 16, 2441–2452. [Google Scholar] [CrossRef]
- Hammack, S.E.; Pepin, J.L.; DesMarteau, J.S.; Watkins, L.R.; Maier, S.F. Low doses of corticotropin-releasing hormone injected into the dorsal raphe nucleus block the behavioral consequences of uncontrollable stress. Behav. Brain Res. 2003, 147, 55–64. [Google Scholar] [CrossRef]
- Nishitani, N.; Nagayasu, K.; Asaoka, N.; Yamashiro, M.; Andoh, C.; Nagai, Y.; Kinoshita, H.; Kawai, H.; Shibui, N.; Liu, B.; et al. Manipulation of dorsal raphe serotonergic neurons modulates active coping to inescapable stress and anxiety-related behaviors in mice and rats. Neuropsychopharmacology 2019, 44, 721–732. [Google Scholar] [CrossRef] [PubMed]
- Hammack, S.E.; Schmid, M.J.; LoPresti, M.L.; Der-Avakian, A.; Pellymounter, M.A.; Foster, A.C.; Watkins, L.R.; Maier, S.F. Corticotropin releasing hormone type 2 receptors in the dorsal raphe nucleus mediate the behavioral consequences of uncontrollable stress. J. Neurosci. 2003, 23, 1019–1025. [Google Scholar] [CrossRef]
- Summers, C.H.; Kampshoff, J.L.; Ronan, P.J.; Lowry, C.A.; Prestbo, A.A.; Korzan, W.J.; Renner, K.J. Monoaminergic activity in subregions of raphe nuclei elicited by prior stress and the neuropeptide corticotropin-releasing factor. J. Neuroendocrinol. 2003, 15, 1122–1133. [Google Scholar] [CrossRef] [PubMed]
- Kelly, K.J.; Donner, N.C.; Hale, M.W.; Lowry, C.A. Swim stress activates serotonergic and nonserotonergic neurons in specific subdivisions of the rat dorsal raphe nucleus in a temperature-dependent manner. Neuroscience 2011, 197, 251–268. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Goebel, M.; Stengel, A.; Wang, L.; Tache, Y. Restraint stress activates nesfatin-1-immunoreactive brain nuclei in rats. Brain Res. 2009, 1300, 114–124. [Google Scholar] [CrossRef] [PubMed]
- Pham-Le, N.M.; Cockburn, C.; Nowell, K.; Brown, J. Activation of GABAA or 5HT1A receptors in the raphe pallidus abolish the cardiovascular responses to exogenous stress in conscious rats. Brain Res. Bull. 2011, 86, 360–366. [Google Scholar] [CrossRef]
- Said, N.; Lakehayli, S.; El Khachibi, M.; El Ouahli, M.; Nadifi, S.; Hakkou, F.; Tazi, A. Effect of prenatal stress on memory, nicotine withdrawal and 5HT1A expression in raphe nuclei of adult rats. Int. J. Dev. Neurosci. 2015, 43, 92–98. [Google Scholar] [CrossRef] [PubMed]
- Nyhuis, T.J.; Masini, C.V.; Taufer, K.L.; Day, H.E.; Campeau, S. Reversible inactivation of rostral nucleus raphe pallidus attenuates acute autonomic responses but not their habituation to repeated audiogenic stress in rats. Stress 2016, 19, 248–259. [Google Scholar] [CrossRef] [PubMed]
- Cao, W.H.; Morrison, S.F. Disinhibition of rostral raphe pallidus neurons increases cardiac sympathetic nerve activity and heart rate. Brain Res. 2003, 980, 1–10. [Google Scholar] [CrossRef]
- Ishida, Y.; Hashiguchi, H.; Takeda, R.; Ishizuka, Y.; Mitsuyama, Y.; Kannan, H.; Nishimori, T.; Nakahara, D. Conditioned-fear stress increases Fos expression in monoaminergic and GABAergic neurons of the locus coeruleus and dorsal raphe nuclei. Synapse 2002, 45, 46–51. [Google Scholar] [CrossRef]
- Da Silva, G.S.; Giusti, H.; Castro, O.W.; Garcia-Cairasco, N.; Gargaglioni, L.H.; Branco, L.G.; Glass, M.L. Serotonergic neurons in the nucleus raphe obscurus are not involved in the ventilatory and thermoregulatory responses to hypoxia in adult rats. Respir. Physiol. Neurobiol. 2013, 187, 139–148. [Google Scholar] [CrossRef]
- Krukoff, T.L.; Khalili, P. Stress-induced activation of nitric oxide-producing neurons in the rat brain. J. Comp. Neurol. 1997, 377, 509–519. [Google Scholar] [CrossRef]
- Howerton, A.R.; Roland, A.V.; Fluharty, J.M.; Marshall, A.; Chen, A.; Daniels, D.; Beck, S.G.; Bale, T.L. Sex differences in corticotropin-releasing factor receptor-1 action within the dorsal raphe nucleus in stress responsivity. Biol. Psychiatry 2014, 75, 873–883. [Google Scholar] [CrossRef]
- Kijima, T.; Muroi, Y.; Ishii, T. Regulation of maternal care by corticotropin-releasing factor receptors in the dorsal raphe nucleus in mice. Behav. Neurosci. 2021, 135, 359–368. [Google Scholar] [CrossRef]
- Chamas, F.M.; Underwood, M.D.; Arango, V.; Serova, L.; Kassir, S.A.; Mann, J.J.; Sabban, E.L. Immobilization stress elevates tryptophan hydroxylase mRNA and protein in the rat raphe nuclei. Biol. Psychiatry 2004, 55, 278–283. [Google Scholar] [CrossRef]
- Boyarskikh, U.A.; Bondar, N.P.; Filipenko, M.L.; Kudryavtseva, N.N. Downregulation of serotonergic gene expression in the Raphe nuclei of the midbrain under chronic social defeat stress in male mice. Mol. Neurobiol. 2013, 48, 13–21. [Google Scholar] [CrossRef]
- Kovacs, G.L.; Kishonti, J.; Lissak, K.; Telegdy, G. Inhibitory action of midbrain raphe stimulation on stress-induced elevation of plasma corticosterone level in rats. Neurosci. Lett. 1976, 3, 305–310. [Google Scholar] [CrossRef]
- Lazarini-Lopes, W.; Corsi-Zuelli, F.; Padovan, C.M. Attenuation of stress-induced behavioral changes by activation of serotonin type 7 receptors in the median raphe nucleus of rats. J. Psychopharmacol. 2020, 34, 901–913. [Google Scholar] [CrossRef] [PubMed]
- Morrissette, D.A.; Stahl, S.M. Modulating the serotonin system in the treatment of major depressive disorder. CNS Spectr. 2014, 19 (Suppl. S1), 54–57, 68. [Google Scholar] [CrossRef]
- Mikics, E.; Guirado, R.; Umemori, J.; Toth, M.; Biro, L.; Miskolczi, C.; Balazsfi, D.; Zelena, D.; Castren, E.; Haller, J.; et al. Social Learning Requires Plasticity Enhanced by Fluoxetine Through Prefrontal Bdnf-TrkB Signaling to Limit Aggression Induced by Post-Weaning Social Isolation. Neuropsychopharmacology 2018, 43, 235–245. [Google Scholar] [CrossRef]
- Bucci, D.; Busceti, C.L.; Calierno, M.T.; Di Pietro, P.; Madonna, M.; Biagioni, F.; Ryskalin, L.; Limanaqi, F.; Nicoletti, F.; Fornai, F. Systematic Morphometry of Catecholamine Nuclei in the Brainstem. Front. Neuroanat. 2017, 11, 98. [Google Scholar] [CrossRef]
- Gaillet, S.; Alonso, G.; Le Borgne, R.; Barbanel, G.; Malaval, F.; Assenmacher, I.; Szafarczyk, A. Effects of discrete lesions in the ventral noradrenergic ascending bundle on the corticotropic stress response depend on the site of the lesion and on the plasma levels of adrenal steroids. Neuroendocrinology 1993, 58, 408–419. [Google Scholar] [CrossRef] [PubMed]
- Dayas, C.V.; Buller, K.M.; Crane, J.W.; Xu, Y.; Day, T.A. Stressor categorization: Acute physical and psychological stressors elicit distinctive recruitment patterns in the amygdala and in medullary noradrenergic cell groups. Eur. J. Neurosci. 2001, 14, 1143–1152. [Google Scholar] [CrossRef] [PubMed]
- Itoi, K.; Sugimoto, N. The brainstem noradrenergic systems in stress, anxiety and depression. J. Neuroendocrinol. 2010, 22, 355–361. [Google Scholar] [CrossRef]
- Valentino, R.J.; Van Bockstaele, E. Convergent regulation of locus coeruleus activity as an adaptive response to stress. Eur. J. Pharmacol. 2008, 583, 194–203. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Hunt, S.; Sah, P. Norepinephrine and Corticotropin-Releasing Hormone: Partners in the Neural Circuits that Underpin Stress and Anxiety. Neuron 2015, 87, 468–470. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Pruessner, J.C.; Champagne, F.; Meaney, M.J.; Dagher, A. Dopamine release in response to a psychological stress in humans and its relationship to early life maternal care: A positron emission tomography study using [11C]raclopride. J. Neurosci 2004, 24, 2825–2831. [Google Scholar] [CrossRef]
- Bloomfield, M.A.; McCutcheon, R.A.; Kempton, M.; Freeman, T.P.; Howes, O. The effects of psychosocial stress on dopaminergic function and the acute stress response. Elife 2019, 8, e46797. [Google Scholar] [CrossRef] [PubMed]
- Baik, J.H. Stress and the dopaminergic reward system. Exp. Mol. Med. 2020, 52, 1879–1890. [Google Scholar] [CrossRef] [PubMed]
- Missale, C.; Nash, S.R.; Robinson, S.W.; Jaber, M.; Caron, M.G. Dopamine receptors: From structure to function. Physiol. Rev. 1998, 78, 189–225. [Google Scholar] [CrossRef]
- Belujon, P.; Grace, A.A. Regulation of dopamine system responsivity and its adaptive and pathological response to stress. Proc. Biol. Sci. 2015, 282, 20142516. [Google Scholar] [CrossRef] [PubMed]
- Hamner, M.B.; Diamond, B.I. Elevated plasma dopamine in posttraumatic stress disorder: A preliminary report. Biol. Psychiatry 1993, 33, 304–306. [Google Scholar] [CrossRef]
- Hong, S. Dopamine system: Manager of neural pathways. Front. Hum. Neurosci. 2013, 7, 854. [Google Scholar] [CrossRef]
- Barbeau, A. The pathogenesis of Parkinson’s disease: A new hypothesis. Can. Med. Assoc. J. 1962, 87, 802–807. [Google Scholar] [PubMed]
- Narayanan, N.S.; Guarnieri, D.J.; DiLeone, R.J. Metabolic hormones, dopamine circuits, and feeding. Front. Neuroendocrinol. 2010, 31, 104–112. [Google Scholar] [CrossRef]
- Helbing, C.; Brocka, M.; Scherf, T.; Lippert, M.T.; Angenstein, F. The role of the mesolimbic dopamine system in the formation of blood-oxygen-level dependent responses in the medial prefrontal/anterior cingulate cortex during high-frequency stimulation of the rat perforant pathway. J. Cereb. Blood Flow Metab. 2016, 36, 2177–2193. [Google Scholar] [CrossRef]
- Massaly, N.; Moron, J.A.; Al-Hasani, R. A Trigger for Opioid Misuse: Chronic Pain and Stress Dysregulate the Mesolimbic Pathway and Kappa Opioid System. Front. Neurosci. 2016, 10, 480. [Google Scholar] [CrossRef] [PubMed]
- Sogabe, S.; Yagasaki, Y.; Onozawa, K.; Kawakami, Y. Mesocortical dopamine system modulates mechanical nociceptive responses recorded in the rat prefrontal cortex. BMC Neurosci. 2013, 14, 65. [Google Scholar] [CrossRef][Green Version]
- Oresland, T.; Fasth, S.; Akervall, S.; Nordgren, S.; Hulten, L. Manovolumetric and sensory characteristics of the ileoanal J pouch compared with healthy rectum. Br. J. Surg. 1990, 77, 803–806. [Google Scholar] [CrossRef]
- Briffaud, V.; Williams, P.; Courty, J.; Broberger, C. Excitation of tuberoinfundibular dopamine neurons by oxytocin: Crosstalk in the control of lactation. J. Neurosci. 2015, 35, 4229–4237. [Google Scholar] [CrossRef]
- Lyons, D.J.; Hellysaz, A.; Broberger, C. Prolactin regulates tuberoinfundibular dopamine neuron discharge pattern: Novel feedback control mechanisms in the lactotrophic axis. J. Neurosci. 2012, 32, 8074–8083. [Google Scholar] [CrossRef]
- Gudelsky, G.A. Tuberoinfundibular dopamine neurons and the regulation of prolactin secretion. Psychoneuroendocrinology 1981, 6, 3–16. [Google Scholar] [CrossRef]
- Vaessen, T.; Hernaus, D.; Myin-Germeys, I.; van Amelsvoort, T. The dopaminergic response to acute stress in health and psychopathology: A systematic review. Neurosci. Biobehav. Rev. 2015, 56, 241–251. [Google Scholar] [CrossRef]
- Berridge, K.C.; Robinson, T.E. What is the role of dopamine in reward: Hedonic impact, reward learning, or incentive salience? Brain Res. Brain Res. Rev. 1998, 28, 309–369. [Google Scholar] [CrossRef]
- Salamone, J.D.; Cousins, M.S.; Snyder, B.J. Behavioral functions of nucleus accumbens dopamine: Empirical and conceptual problems with the anhedonia hypothesis. Neurosci. Biobehav. Rev. 1997, 21, 341–359. [Google Scholar] [CrossRef]
- Piazza, P.V.; Le Moal, M. The role of stress in drug self-administration. Trends Pharmacol. Sci. 1998, 19, 67–74. [Google Scholar] [CrossRef]
- Levine, S.; Muneyyirci-Delale, O. Stress-Induced Hyperprolactinemia: Pathophysiology and Clinical Approach. Obstet Gynecol. Int. 2018, 2018, 9253083. [Google Scholar] [CrossRef] [PubMed]
- Ochoa-Amaya, J.E.; Malucelli, B.E.; Cruz-Casallas, P.E.; Nasello, A.G.; Felicio, L.F.; Carvalho-Freitas, M.I. Acute and chronic stress and the inflammatory response in hyperprolactinemic rats. Neuroimmunomodulation 2010, 17, 386–395. [Google Scholar] [CrossRef]
- Ruiz-Herrera, X.; de Los Rios, E.A.; Diaz, J.M.; Lerma-Alvarado, R.M.; Martinez de la Escalera, L.; Lopez-Barrera, F.; Lemini, M.; Arnold, E.; Martinez de la Escalera, G.; Clapp, C.; et al. Prolactin Promotes Adipose Tissue Fitness and Insulin Sensitivity in Obese Males. Endocrinology 2017, 158, 56–68. [Google Scholar] [CrossRef] [PubMed]
- Jaroenporn, S.; Nagaoka, K.; Kasahara, C.; Ohta, R.; Watanabe, G.; Taya, K. Physiological roles of prolactin in the adrenocortical response to acute restraint stress. Endocr. J. 2007, 54, 703–711. [Google Scholar] [CrossRef] [PubMed]
- Bánrévi, K.; Chaves, T.; Correia, P.; Fazekas, C.L.; Szabó, A.; Török, B.; Zelena, D. Brain corticotropin releasing hormone and stress reactivity. Integrativnaja Physiologija 2021, 2, 6–14. [Google Scholar] [CrossRef]
- Keegan, C.E.; Herman, J.P.; Karolyi, I.J.; O’Shea, K.S.; Camper, S.A.; Seasholtz, A.F. Differential expression of corticotropin-releasing hormone in developing mouse embryos and adult brain. Endocrinology 1994, 134, 2547–2555. [Google Scholar] [CrossRef]
- Bloom, F.E.; Battenberg, E.L.; Rivier, J.; Vale, W. Corticotropin releasing factor (CRF): Immunoreactive neurones and fibers in rat hypothalamus. Regul. Pept. 1982, 4, 43–48. [Google Scholar] [CrossRef]
- Wang, L.; Goebel-Stengel, M.; Stengel, A.; Wu, S.V.; Ohning, G.; Tache, Y. Comparison of CRF-immunoreactive neurons distribution in mouse and rat brains and selective induction of Fos in rat hypothalamic CRF neurons by abdominal surgery. Brain Res. 2011, 1415, 34–46. [Google Scholar] [CrossRef]
- Palkovits, M.; Kovacs, K.; Young, W.S.; Makara, G.B. Corticotropin-releasing hormone expression in supraoptic neurons after bilateral lesioning of the paraventricular nucleus in rats. Neurobiology 1997, 5, 423–430. [Google Scholar] [PubMed]
- Kovacs, K.J.; Sawchenko, P.E. Mediation of osmoregulatory influences on neuroendocrine corticotropin-releasing factor expression by the ventral lamina terminalis. Proc. Natl. Acad. Sci. USA 1993, 90, 7681–7685. [Google Scholar] [CrossRef]
- Wamsteeker Cusulin, J.I.; Fuzesi, T.; Watts, A.G.; Bains, J.S. Characterization of corticotropin-releasing hormone neurons in the paraventricular nucleus of the hypothalamus of Crh-IRES-Cre mutant mice. PLoS ONE 2013, 8, e64943. [Google Scholar] [CrossRef]
- Alon, T.; Zhou, L.; Perez, C.A.; Garfield, A.S.; Friedman, J.M.; Heisler, L.K. Transgenic mice expressing green fluorescent protein under the control of the corticotropin-releasing hormone promoter. Endocrinology 2009, 150, 5626–5632. [Google Scholar] [CrossRef] [PubMed]
- Kono, J.; Konno, K.; Talukder, A.H.; Fuse, T.; Abe, M.; Uchida, K.; Horio, S.; Sakimura, K.; Watanabe, M.; Itoi, K. Distribution of corticotropin-releasing factor neurons in the mouse brain: A study using corticotropin-releasing factor-modified yellow fluorescent protein knock-in mouse. Brain Struct. Funct. 2017, 222, 1705–1732. [Google Scholar] [CrossRef] [PubMed]
- Peng, J.; Long, B.; Yuan, J.; Peng, X.; Ni, H.; Li, X.; Gong, H.; Luo, Q.; Li, A. A Quantitative Analysis of the Distribution of CRH Neurons in Whole Mouse Brain. Front. Neuroanat. 2017, 11, 63. [Google Scholar] [CrossRef] [PubMed]
- Yao, M.; Westphal, N.J.; Denver, R.J. Distribution and acute stressor-induced activation of corticotrophin-releasing hormone neurones in the central nervous system of Xenopus laevis. J. Neuroendocrinol. 2004, 16, 880–893. [Google Scholar] [CrossRef]
- Austin, M.C.; Janosky, J.E.; Murphy, H.A. Increased corticotropin-releasing hormone immunoreactivity in monoamine-containing pontine nuclei of depressed suicide men. Mol. Psychiatry 2003, 8, 324–332. [Google Scholar] [CrossRef]
- Kawatani, M.; Itoi, K.; Talukder, A.H.; Uchida, K.; Sakimura, K.; Kawatani, M. Cholinergic modulation of CRH and non-CRH neurons in Barrington’s nucleus of the mouse. J. Neurophysiol. 2020, 124, 443–457. [Google Scholar] [CrossRef]
- Sved, A.F.; Cano, G.; Passerin, A.M.; Rabin, B.S. The locus coeruleus, Barrington’s nucleus, and neural circuits of stress. Physiol. Behav. 2002, 77, 737–742. [Google Scholar] [CrossRef]
- Holschneider, D.P.; Wang, Z.; Guo, Y.; Sanford, M.T.; Yeh, J.; Mao, J.J.; Zhang, R.; Rodriguez, L.V. Exercise modulates neuronal activation in the micturition circuit of chronically stressed rats: A multidisciplinary approach to the study of urologic chronic pelvic pain syndrome (MAPP) research network study. Physiol. Behav. 2020, 215, 112796. [Google Scholar] [CrossRef] [PubMed]
- Butler, S.; Luz, S.; McFadden, K.; Fesi, J.; Long, C.; Spruce, L.; Seeholzer, S.; Canning, D.; Valentino, R.; Zderic, S. Murine social stress results in long lasting voiding dysfunction. Physiol. Behav. 2018, 183, 10–17. [Google Scholar] [CrossRef] [PubMed]
- Wood, S.K.; McFadden, K.; Griffin, T.; Wolfe, J.H.; Zderic, S.; Valentino, R.J. A corticotropin-releasing factor receptor antagonist improves urodynamic dysfunction produced by social stress or partial bladder outlet obstruction in male rats. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2013, 304, R940–R950. [Google Scholar] [CrossRef][Green Version]
- Schweighofer, N.; Lang, E.J.; Kawato, M. Role of the olivo-cerebellar complex in motor learning and control. Front. Neural. Circuits 2013, 7, 94. [Google Scholar] [CrossRef]
- Gutstein, H.B.; Thome, J.L.; Fine, J.L.; Watson, S.J.; Akil, H. Pattern of c-fos mRNA induction in rat brain by acute morphine. Can. J. Physiol. Pharmacol. 1998, 76, 294–303. [Google Scholar] [CrossRef]
- Rub, U.; Burk, K.; Schols, L.; Brunt, E.R.; de Vos, R.A.; Diaz, G.O.; Gierga, K.; Ghebremedhin, E.; Schultz, C.; Del Turco, D.; et al. Damage to the reticulotegmental nucleus of the pons in spinocerebellar ataxia type 1, 2, and 3. Neurology 2004, 63, 1258–1263. [Google Scholar] [CrossRef]
- May, P.J.; Billig, I.; Gamlin, P.D.; Quinet, J. Central mesencephalic reticular formation control of the near response: Lens accommodation circuits. J. Neurophysiol. 2019, 121, 1692–1703. [Google Scholar] [CrossRef]
- Palmiter, R.D. The Parabrachial Nucleus: CGRP Neurons Function as a General Alarm. Trends Neurosci. 2018, 41, 280–293. [Google Scholar] [CrossRef]
- Tiriac, A.; Blumberg, M.S. Gating of reafference in the external cuneate nucleus during self-generated movements in wake but not sleep. eLife 2016, 5, e18749. [Google Scholar] [CrossRef]
- Schwarz, C.; Thier, P. Binding of signals relevant for action: Towards a hypothesis of the functional role of the pontine nuclei. Trends Neurosci. 1999, 22, 443–451. [Google Scholar] [CrossRef]
- Martins, A.P.; Marras, R.A.; Guimaraes, F.S. Anxiolytic effect of a CRH receptor antagonist in the dorsal periaqueductal gray. Depress. Anxiety 2000, 12, 99–101. [Google Scholar] [CrossRef]
- Martins, A.P.; Marras, R.A.; Guimaraes, F.S. Anxiogenic effect of corticotropin-releasing hormone in the dorsal periaqueductal grey. Neuroreport 1997, 8, 3601–3604. [Google Scholar] [CrossRef] [PubMed]
- Sos, K.E.; Mayer, M.I.; Cserep, C.; Takacs, F.S.; Szonyi, A.; Freund, T.F.; Nyiri, G. Cellular architecture and transmitter phenotypes of neurons of the mouse median raphe region. Brain Struct. Funct. 2017, 222, 287–299. [Google Scholar] [CrossRef] [PubMed]
- Ohmura, Y.; Izumi, T.; Yamaguchi, T.; Tsutsui-Kimura, I.; Yoshida, T.; Yoshioka, M. The serotonergic projection from the median raphe nucleus to the ventral hippocampus is involved in the retrieval of fear memory through the corticotropin-releasing factor type 2 receptor. Neuropsychopharmacology 2010, 35, 1271–1278. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hammack, S.E.; Richey, K.J.; Schmid, M.J.; LoPresti, M.L.; Watkins, L.R.; Maier, S.F. The role of corticotropin-releasing hormone in the dorsal raphe nucleus in mediating the behavioral consequences of uncontrollable stress. J. Neurosci. 2002, 22, 1020–1026. [Google Scholar] [CrossRef] [PubMed]
- Szonyi, A.; Sos, K.E.; Nyilas, R.; Schlingloff, D.; Domonkos, A.; Takacs, V.T.; Posfai, B.; Hegedus, P.; Priestley, J.B.; Gundlach, A.L.; et al. Brainstem nucleus incertus controls contextual memory formation. Science 2019, 364, eaaw0445. [Google Scholar] [CrossRef]
- Ryan, P.J.; Ma, S.; Olucha-Bordonau, F.E.; Gundlach, A.L. Nucleus incertus—An emerging modulatory role in arousal, stress and memory. Neurosci. Biobehav. Rev. 2011, 35, 1326–1341. [Google Scholar] [CrossRef]
- Bittencourt, J.C.; Sawchenko, P.E. Do centrally administered neuropeptides access cognate receptors?: An analysis in the central corticotropin-releasing factor system. J. Neurosci. 2000, 20, 1142–1156. [Google Scholar] [CrossRef]
- Gliddon, C.M.; Darlington, C.L.; Smith, P.F. Activation of the hypothalamic-pituitary-adrenal axis following vestibular deafferentation in pigmented guinea pig. Brain Res. 2003, 964, 306–310. [Google Scholar] [CrossRef]
- Markia, B.; Kovacs, Z.I.; Palkovits, M. Projections from the vestibular nuclei to the hypothalamic paraventricular nucleus: Morphological evidence for the existence of a vestibular stress pathway in the rat brain. Brain Struct. Funct. 2008, 213, 239–245. [Google Scholar] [CrossRef]
- Sherafat, Y.; Bautista, M.; Fowler, J.P.; Chen, E.; Ahmed, A.; Fowler, C.D. The Interpeduncular-Ventral Hippocampus Pathway Mediates Active Stress Coping and Natural Reward. eNeuro 2020, 7. [Google Scholar] [CrossRef]
- Herman, J.P. Regulation of Hypothalamo-Pituitary-Adrenocortical Responses to Stressors by the Nucleus of the Solitary Tract/Dorsal Vagal Complex. Cell Mol. Neurobiol. 2018, 38, 25–35. [Google Scholar] [CrossRef]

| Density of CRH + Neurons. | |||||
|---|---|---|---|---|---|
| Wang et al. 2011 | Alon et al. 2009 | Kono et al. 2016 | Peng et al. 2017 | Recent Results | |
| Method | Colchicin | CRH-GFP | CRH-Venus | CRH-EYFP | CRH-Cre × DIO-dtTomato |
| Inferior olivary complex (IO) | ++ | ++++ | ++++ | +++++ | +++ |
| Barrington’s nucleus (B) | ++ | ++++ | ++++ | ++++ | ++ |
| Tegmental reticular nucleus (TRN) | - | ++ | ++ | +++ | ++ |
| Parabrachial nuclei (PB) | ++ | ++ | + | + | + |
| Periaqueductal gray (PAG) | - | ++ | + | ++ | + |
| External cunate nucleus (ECN) | - | + | +++ | + | |
| Raphe nucleus | - | + | ++ (MRN) | + (DRN) | + |
| Nucleus incertus | + | + | + | + | |
| Mesencephalic reticular formation | - | + | + | + | + |
| Interpeduncular nucleus | + | + | + | ||
| Pontine gray | +++ | + | |||
| Medial vestibular nucleus | - | + | ++ | + | |
| A11 | + | ||||
| Vagus nuclei (NTS, motor) | ++ | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chaves, T.; Fazekas, C.L.; Horváth, K.; Correia, P.; Szabó, A.; Török, B.; Bánrévi, K.; Zelena, D. Stress Adaptation and the Brainstem with Focus on Corticotropin-Releasing Hormone. Int. J. Mol. Sci. 2021, 22, 9090. https://doi.org/10.3390/ijms22169090
Chaves T, Fazekas CL, Horváth K, Correia P, Szabó A, Török B, Bánrévi K, Zelena D. Stress Adaptation and the Brainstem with Focus on Corticotropin-Releasing Hormone. International Journal of Molecular Sciences. 2021; 22(16):9090. https://doi.org/10.3390/ijms22169090
Chicago/Turabian StyleChaves, Tiago, Csilla Lea Fazekas, Krisztina Horváth, Pedro Correia, Adrienn Szabó, Bibiána Török, Krisztina Bánrévi, and Dóra Zelena. 2021. "Stress Adaptation and the Brainstem with Focus on Corticotropin-Releasing Hormone" International Journal of Molecular Sciences 22, no. 16: 9090. https://doi.org/10.3390/ijms22169090
APA StyleChaves, T., Fazekas, C. L., Horváth, K., Correia, P., Szabó, A., Török, B., Bánrévi, K., & Zelena, D. (2021). Stress Adaptation and the Brainstem with Focus on Corticotropin-Releasing Hormone. International Journal of Molecular Sciences, 22(16), 9090. https://doi.org/10.3390/ijms22169090





