Diabetic Kinome Inhibitors—A New Opportunity for β-Cells Restoration
Abstract
:1. Introduction
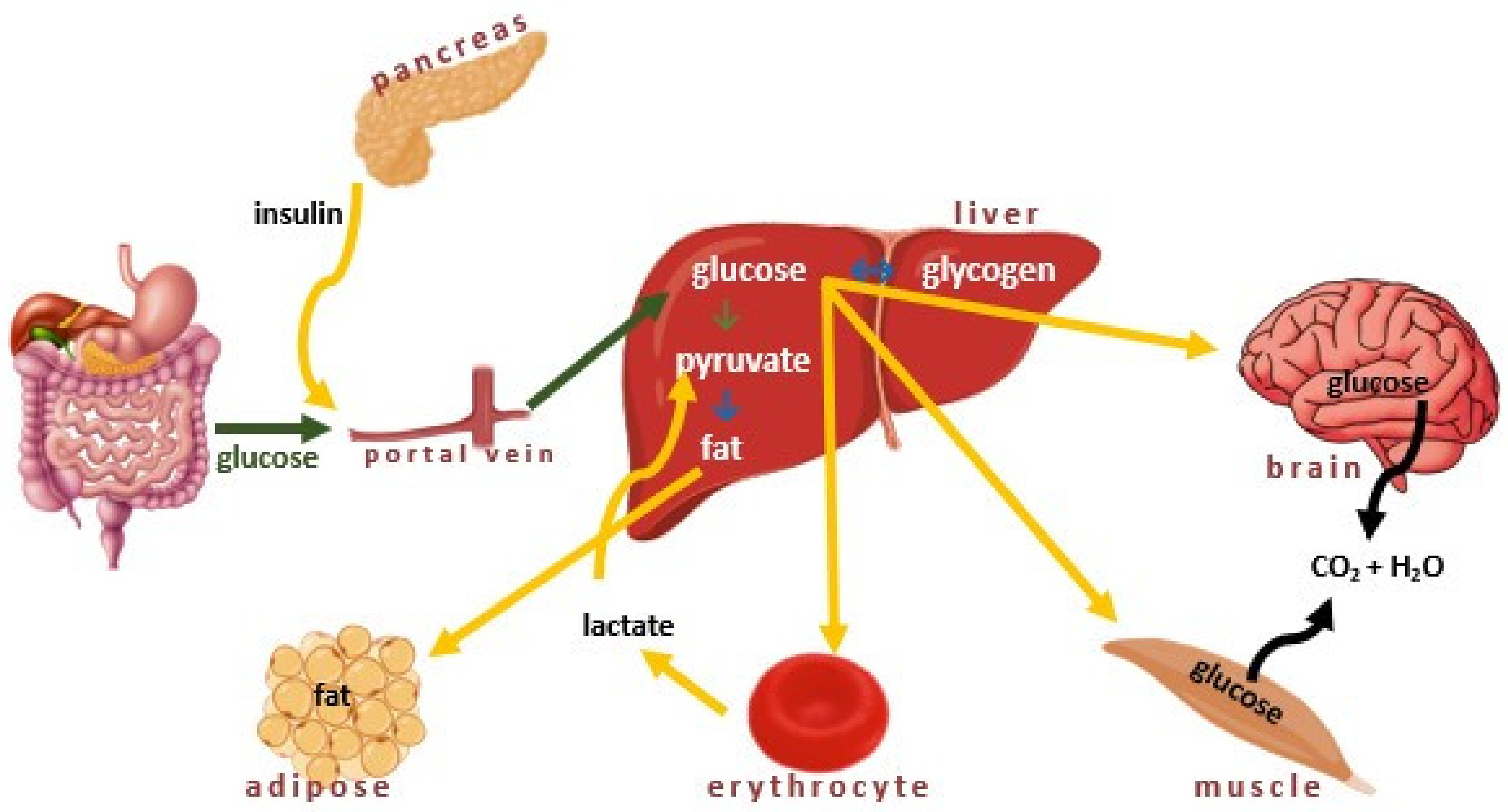
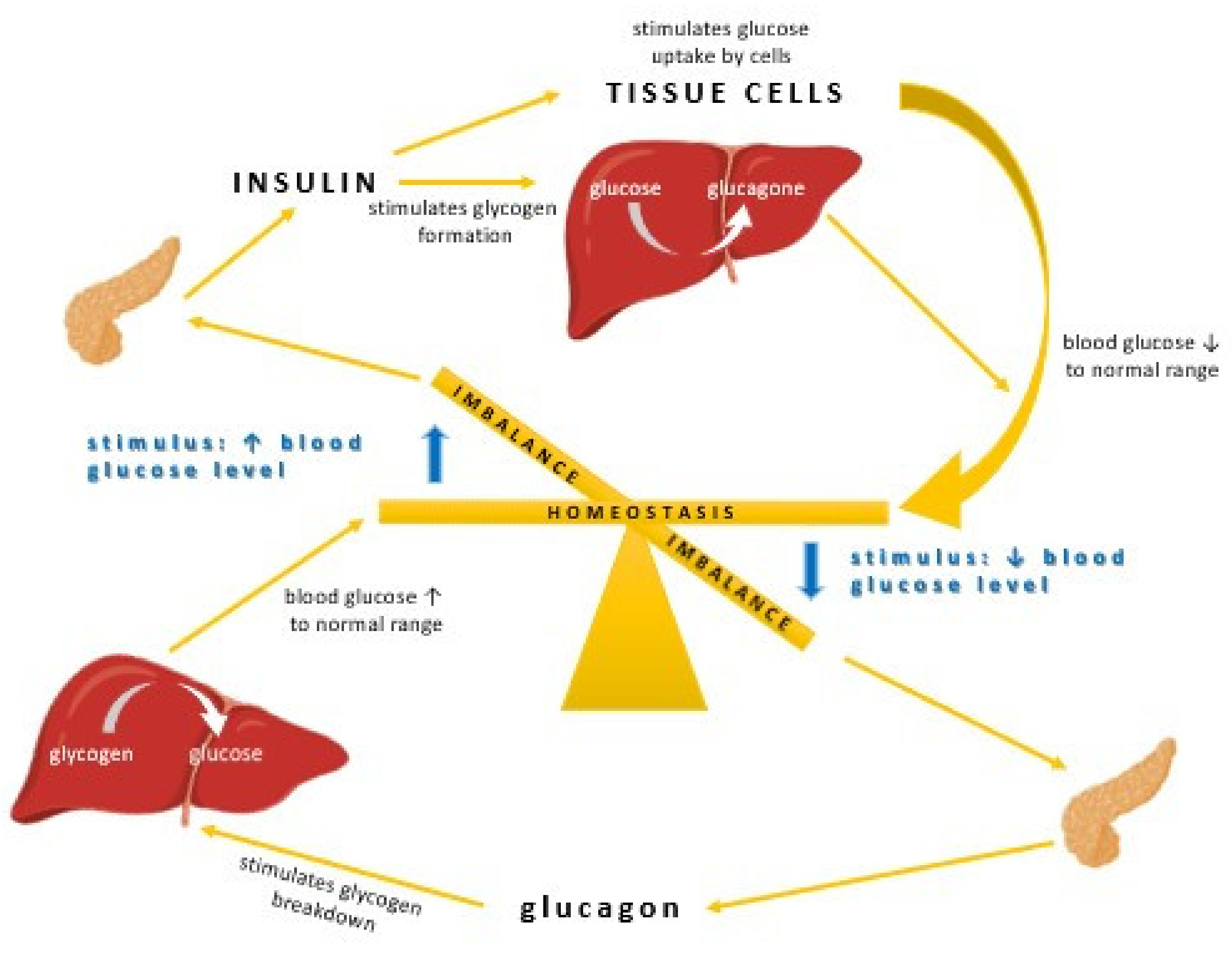
2. Insulin Homeostasis and Diabetes
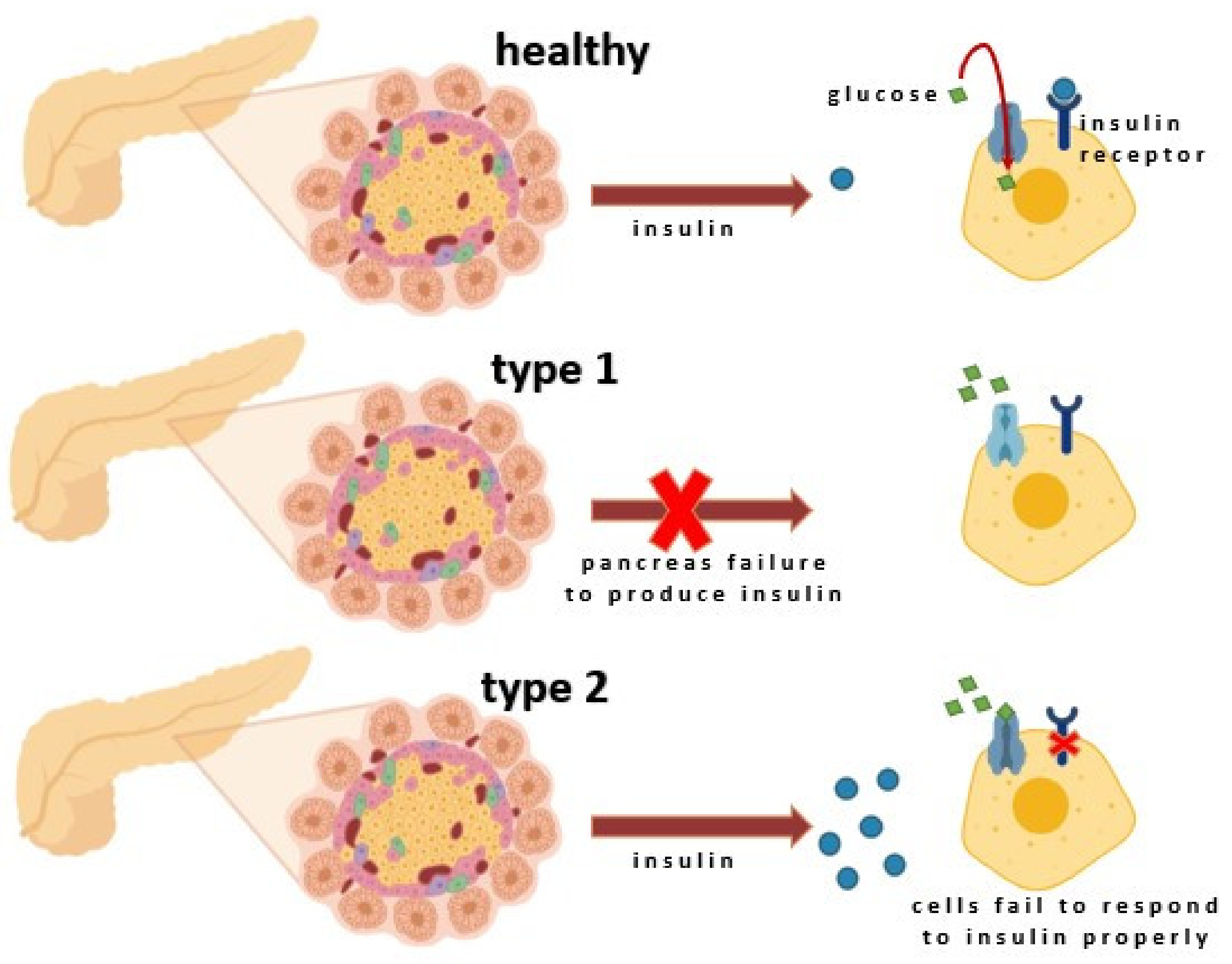
2.1. Type I Diabetes (T1D)
2.2. Type 2 Diabetes (T2D)
2.3. Maturity-Onset Diabetes of the Young (MODY)
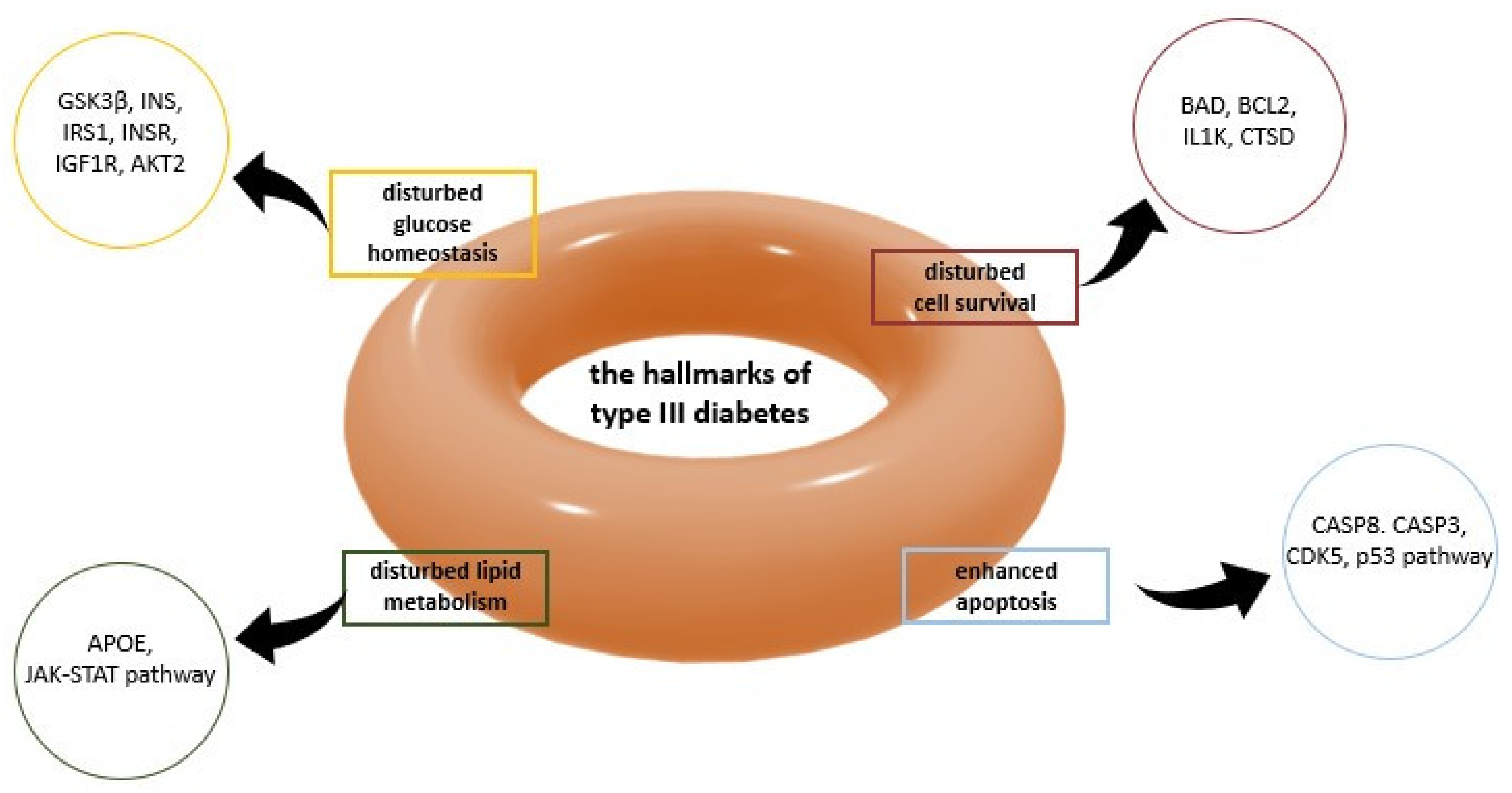
2.4. Alzheimer’s Disease—Diabetes Mellitus (AD-DM) Relation and Type 3 Diabetes (T3D)
3. Molecular Basis of Diabetes
3.1. Diabetic Kinome
Inflammation, Diabetes, and Kinase Inhibition
4. DYRK Family of Protein Kinases
4.1. DYRKs Activity and Regulation
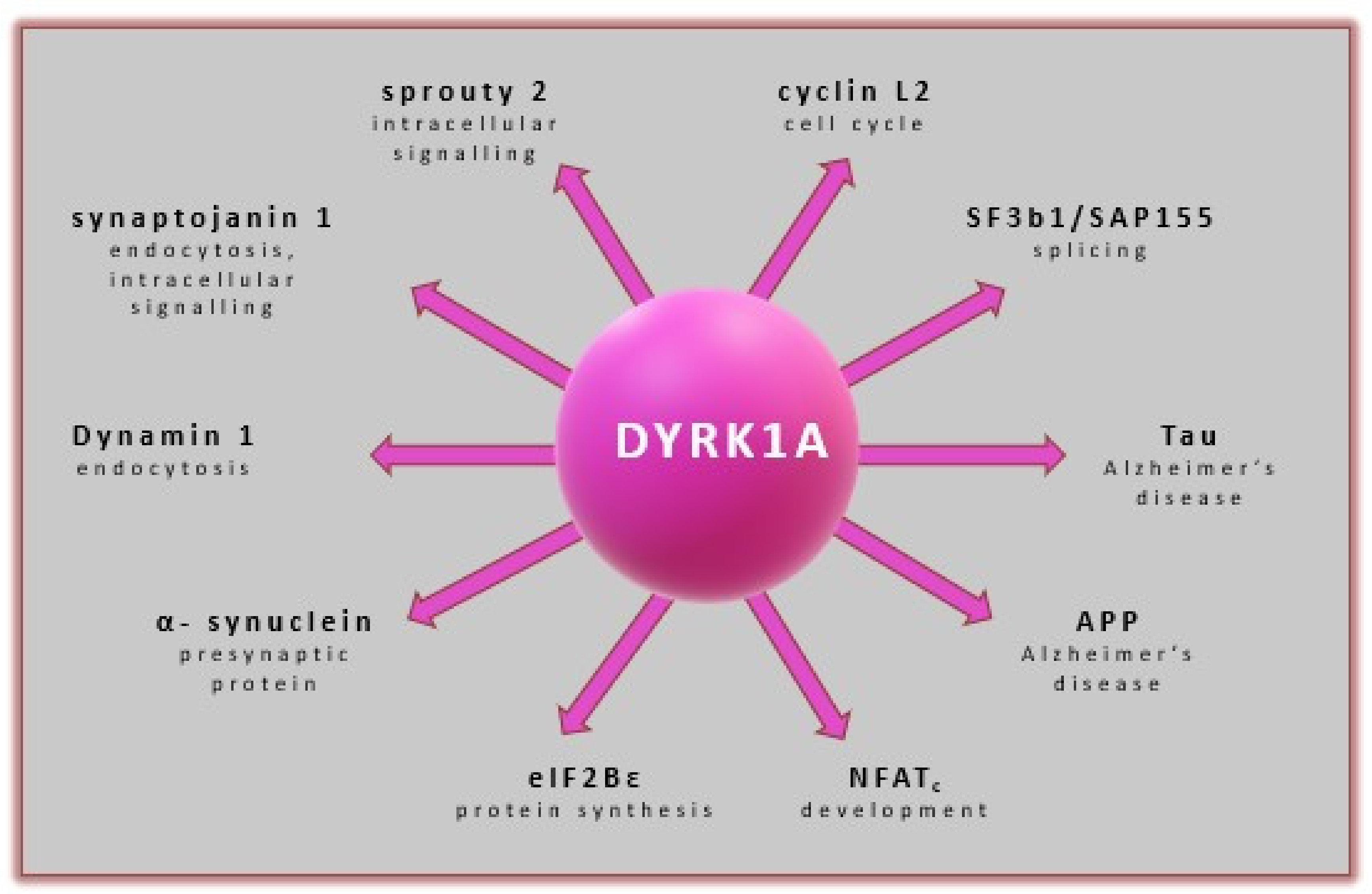
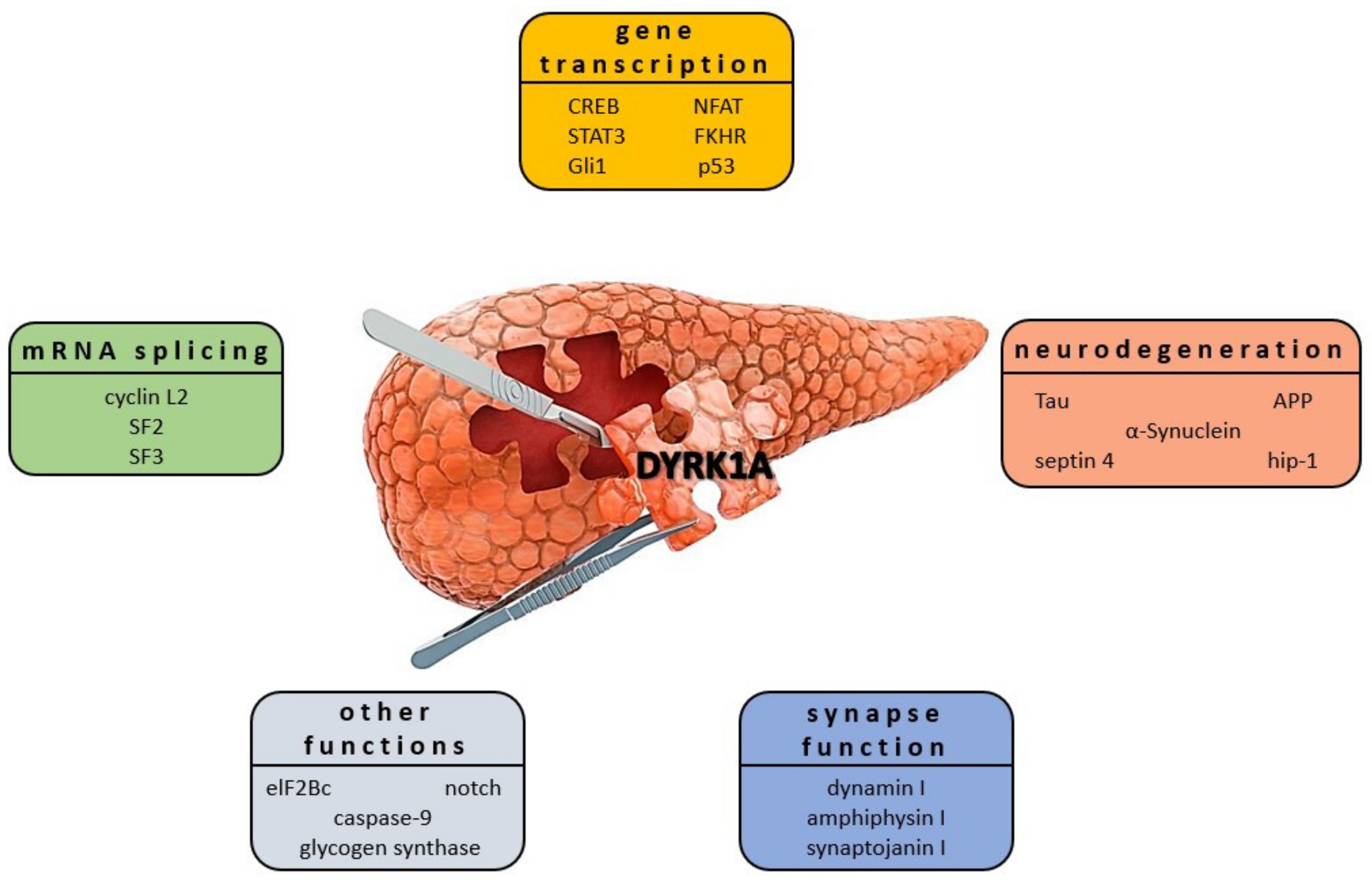
4.2. DYRK1A Expression and Its Role in Neurological Diseases
4.3. DYRK1A Expression Affects Mechanisms of Diabetes
5. Current Treatments of Diabetes
5.1. DYRK1A Inhibitors for β-Cell Function Restoration
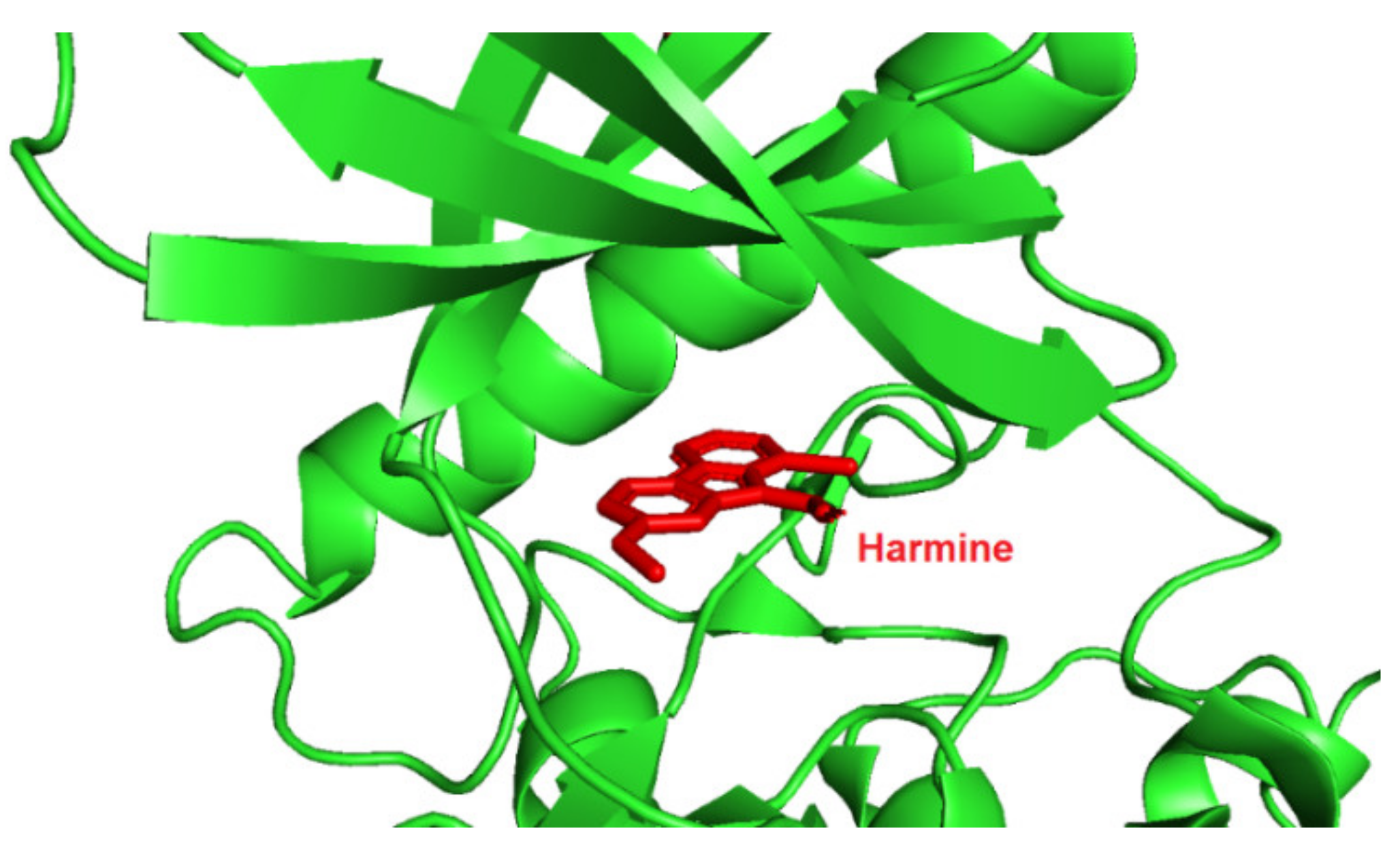
5.1.1. Harmine and Its Analogues—SAR Approach
5.1.2. Perha Pharmaceutics Inhibitors
5.1.3. Azaindoles
5.1.4. Aminopyrazines
5.1.5. AC Inhibitors
5.1.6. Miscellaneous Scaffolds and Drug Combinations
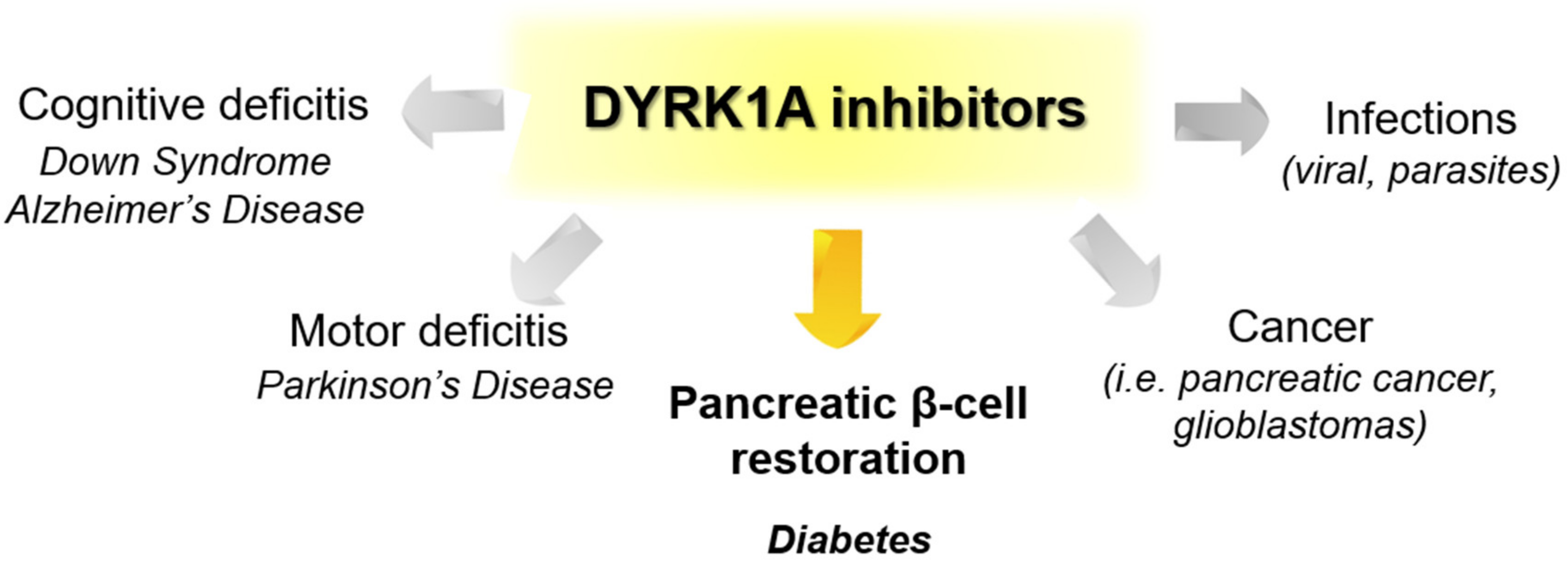
6. Biological Effects of DYRK1A Inhibitors
6.1. Diabetes
6.2. Other Diseases
6.2.1. Neurological Disorders
6.2.2. Cancer
7. Summary
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| AD | Alzheimer’s Disease |
| Akt | Protein kinase B |
| AMPK | 5′AMP-activated protein kinase |
| BBB | Blood-brain barrier |
| β-FGF | Fibroblast growth factor |
| cAMP | 3′,5′-cyclic adenosine monophosphate |
| CDKLs | CDK-like kinases |
| CDKN | Cyclin-dependent kinase inhibitor |
| CDKs | Cyclin-dependent kinases |
| CK2 | Casein kinase 2 |
| CLKs | CDC-like kinases |
| CMGC | Kinase family is named after the initials of its subfamily members, including cyclin-dependent kinase (CDK), mitogen-activated protein kinase (MAPK), glycogen synthase kinase (GSK) and CDC-like kinase (CLK) |
| c-Myc | Family of regulator genes and proto-oncogenes that code for transcription factors |
| CNS | Central nervous system |
| CREB | cAMP response element-binding protein |
| DM | Diabetes mellitus |
| DN | Diabetic Neuropathy |
| DPP-4 | Dipeptidyl peptidase-4 |
| DREAM | Dimerization partner, RB-like, E2F, and multi-vulval class B |
| DS | Down Syndrome |
| DSCR1 gene | Down Syndrome critical region gene 1 |
| DYRK1A | Dual-specificity tyrosine phosphorylation-regulated kinase A |
| EGCG | Epigallocatechin gallate |
| ERK/MAPK | A chain of proteins in the cell that communicates a signal from a receptor on the cell’s surface to the DNA in the nucleus. The pathway includes many proteins, including MAPK (mitogen-activated protein kinases, originally-called ERK, extracellular signal-regulated kinases) |
| FDA | Food and Drug Administration |
| FKHR | Forkhead protein |
| FOX | Forkhead box |
| FOXM1 | Forkhead Box M1 |
| GAD65 | Glutamic acid decarboxylase |
| GADA | Glutamic acid decarboxylase antibodies |
| GDM | Gestational diabetes mellitus |
| GLP-1 | Glucagon-like peptide-1 |
| GLUT4 | Glucose transporter type 4 |
| GPAIS | Glucose potentiates arginine-induced insulin secretion |
| GSK3 | Glycogen synthase kinase 3 |
| GSKs | Glycogen synthase kinases |
| HbA1c | Glycated hemoglobin |
| HK | Hexokinase |
| HLA | Human leukocyte antigen |
| HNF-1α | Hepatic transcription nuclear factor |
| HTS | High-throughput screening |
| IDE | Insulin-degrading Enzyme |
| IGF-1R | Insulin-like growth factor 1 receptor |
| IKKβ | I kappa beta kinase |
| IL-1β | Interleukin 1 beta |
| IL-12 | Interleukin 12 |
| IR | Insulin receptor |
| IRS1 | Insulin receptor substrate 1 |
| IRS2 | Insulin receptor substrate-2 |
| JNK | Jun N-terminal kinase |
| MAO | Monoamine oxidase |
| MAPK | Mitogen-activated protein kinase |
| MODY | Maturity-onset diabetes of the young |
| mTOR | Mammalian target of rapamycin kinase |
| MYBL2 | Myb-related protein B |
| NFAT | Nuclear factor of activated T-cell |
| NKX6.1 | Protein that in humans is encoded by the NKX6-1 gene |
| NME1/2 | Nucleoside diphosphate kinase 1 and 2 |
| p53 | tumor protein P53 |
| PI3K | Phosphatidylinositol 3-kinase |
| PI3K-AKT/PKB | Signal transduction pathway that promotes survival and growth in response to extracellular signals. Key proteins involved are PI3K (phosphatidylinositol 3-kinase) and Akt (protein kinase B) |
| PKC | Protein kinase C |
| PKR | RNA-activated protein kinase |
| PPAR | Peroxisome proliferator-activated receptors |
| PPX | Partial pancreatectomy model |
| PTHRP | Parathyroid hormone-related protein |
| RNA | Ribonucleic acid |
| ROCK | Rho-associated coiled-coil containing protein kinase |
| S6K1 | S6 kinase-1 |
| SAR | Structure-activity relationship |
| SGLT-2 | Sodium-glucose co-transporter-2 |
| SMAD | Family of structurally similar proteins that are the primary signal transducers for receptors of the transforming growth factor-beta |
| SPRED2 | Sprouty-related protein with an EVH1 domain |
| Sprouty2 | Sprouty homolog 2 |
| STAT3 | Signal transducer and activator of transcription 3 |
| TGF-β | Transforming growth factor β |
| TGFβSF | Transforming growth factor-beta superfamily |
| TNF-α | Tumor necrosis factor α |
| VGF | Nerve growth factor |
References
- Frayling, T.M.; Evans, J.C.; Bulman, M.P.; Pearson, E.; Allen, L.; Owen, K.; Bingham, C.; Hannemann, M.; Shepherd, M.; Ellard, S. beta-cell genes and diabetes: Molecular and clinical characterization of mutations in transcription factors. Diabetes 2001, 50 (Suppl. S1). [Google Scholar]
- Eisenbarth, G.S. Type 1 Diabetes: Molecular, Cellular and Clinical Immunology; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2004; Volume 552. [Google Scholar]
- Inadera, H. Developmental origins of obesity and type 2 diabetes: Molecular aspects and role of chemicals. Environ. Health Prev. Med. 2013, 18, 185–197. [Google Scholar]
- Pal, M.; Febbraio, M.A.; Lancaster, G.I. The roles of c-Jun NH2-terminal kinases (JNKs) in obesity and insulin resistance. J. Physiol. 2016, 594, 267–279. [Google Scholar]
- Lu, Z.; Hunter, T. Metabolic kinases moonlighting as protein kinases. Trends Biochem. Sci. 2018, 43, 301–310. [Google Scholar]
- Rondinone, C.M. Serine kinases as new drug targets for the treatment of type 2 diabetes. Curr. Med. Chem. Immunol. Endocr. Metab. Agents 2005, 5, 529–536. [Google Scholar]
- Roskoski, R., Jr. Properties of FDA-approved small molecule protein kinase inhibitors: A 2020 update. Pharmacol. Res. 2020, 152, 104609. [Google Scholar]
- Wu, P.; Nielsen, T.E.; Clausen, M.H. FDA-approved small-molecule kinase inhibitors. Trends Pharmacol. Sci. 2015, 36, 422–439. [Google Scholar]
- Li, Y.H.; Wang, P.P.; Li, X.X.; Yu, C.Y.; Yang, H.; Zhou, J.; Xue, W.W.; Tan, J.; Zhu, F. The human kinome targeted by FDA approved multi-target drugs and combination products: A comparative study from the drug-target interaction network perspective. PLoS ONE 2016, 11, e0165737. [Google Scholar]
- Available online: https://clinicaltrials.gov/ct2/show/NCT01781975 (accessed on 3 January 2021).
- Dirice, E.; Walpita, D.; Vetere, A.; Meier, B.C.; Kahraman, S.; Hu, J.; Dančík, V.; Burns, S.M.; Gilbert, T.J.; Olson, D.E. Inhibition of DYRK1A stimulates human β-cell proliferation. Diabetes 2016, 65, 1660–1671. [Google Scholar]
- Nguyen, T.L.; Fruit, C.; Hérault, Y.; Meijer, L.; Besson, T. Dual-specificity tyrosine phosphorylation-regulated kinase 1A (DYRK1A) inhibitors: A survey of recent patent literature. Expert Opin. Ther. Pat. 2017, 27, 1183–1199. [Google Scholar]
- Patel, K.; Gadewar, M.; Tripathi, R.; Prasad, S.; Patel, D.K. A review on medicinal importance, pharmacological activity and bioanalytical aspects of beta-carboline alkaloid “Harmine”. Asian Pac. J. Trop. Biomed. 2012, 2, 660–664. [Google Scholar]
- Cuny, G.D.; Ulyanova, N.P.; Patnaik, D.; Liu, J.-F.; Lin, X.; Auerbach, K.; Ray, S.S.; Xian, J.; Glicksman, M.A.; Stein, R.L. Structure–activity relationship study of beta-carboline derivatives as haspin kinase inhibitors. Bioorg. Med. Chem. Lett. 2012, 22, 2015–2019. [Google Scholar]
- Kumar, K.; Wang, P.; Sanchez, R.; Swartz, E.A.; Stewart, A.F.; DeVita, R.J. Development of kinase-selective, harmine-based DYRK1A inhibitors that induce pancreatic human β-cell proliferation. J. Med. Chem. 2018, 61, 7687–7699. [Google Scholar]
- Wang, P.; Alvarez-Perez, J.-C.; Felsenfeld, D.P.; Liu, H.; Sivendran, S.; Bender, A.; Kumar, A.; Sanchez, R.; Scott, D.K.; Garcia-Ocaña, A. A high-throughput chemical screen reveals that harmine-mediated inhibition of DYRK1A increases human pancreatic beta cell replication. Nat. Med. 2015, 21, 383–388. [Google Scholar]
- Dakic, V.; de Moraes Maciel, R.; Drummond, H.; Nascimento, J.M.; Trindade, P.; Rehen, S.K. Harmine stimulates proliferation of human neural progenitors. PeerJ 2016, 4, e2727. [Google Scholar]
- Wu, L.-W.; Zhang, J.-K.; Rao, M.; Zhang, Z.-Y.; Zhu, H.-J.; Zhang, C. Harmine suppresses the proliferation of pancreatic cancer cells and sensitizes pancreatic cancer to gemcitabine treatment. OncoTargets Ther. 2019, 12, 4585. [Google Scholar]
- Delehouzé, C.; Godl, K.; Loaëc, N.; Bruyère, C.; Desban, N.; Oumata, N.; Galons, H.; Roumeliotis, T.I.; Giannopoulou, E.G.; Grenet, J. CDK/CK1 inhibitors roscovitine and CR8 downregulate amplified MYCN in neuroblastoma cells. Oncogene 2014, 33, 5675–5687. [Google Scholar]
- Jarhad, D.B.; Mashelkar, K.K.; Kim, H.-R.; Noh, M.; Jeong, L.S. Dual-specificity tyrosine phosphorylation-regulated kinase 1A (DYRK1A) inhibitors as potential therapeutics. J. Med. Chem. 2018, 61, 9791–9810. [Google Scholar]
- Rothweiler, U.; Eriksson, J.; Stensen, W.; Leeson, F.; Engh, R.A.; Svendsen, J.S. Luciferin and derivatives as a DYRK selective scaffold for the design of protein kinase inhibitors. Eur. J. Med. Chem. 2015, 94, 140–148. [Google Scholar]
- Stotani, S.; Giordanetto, F.; Medda, F. DYRK1A inhibition as potential treatment for Alzheimer’s disease. Future Med. Chem. 2016, 8, 681–696. [Google Scholar]
- Smith, B.; Medda, F.; Gokhale, V.; Dunckley, T.; Hulme, C. Recent advances in the design, synthesis, and biological evaluation of selective DYRK1A inhibitors: A new avenue for a disease modifying treatment of Alzheimer’s? ACS Chem. Neurosci. 2012, 3, 857–872. [Google Scholar]
- Saeedi, P.; Petersohn, I.; Salpea, P.; Malanda, B.; Karuranga, S.; Unwin, N.; Colagiuri, S.; Guariguata, L.; Motala, A.A.; Ogurtsova, K. Global and regional diabetes prevalence estimates for 2019 and projections for 2030 and 2045: Results from the International Diabetes Federation Diabetes Atlas. Diabetes Res. Clin. Pract. 2019, 157, 107843. [Google Scholar]
- American Diabetes Association. Diagnosis and classification of diabetes mellitus. Diabetes Care 2010, 33 (Suppl. S1), S62–S69. [Google Scholar]
- Dronavalli, S.; Duka, I.; Bakris, G.L. The pathogenesis of diabetic nephropathy. Nat. Clin. Pract. Endocrinol. Metab. 2008, 4, 444–452. [Google Scholar]
- Said, G. Diabetic neuropathy—A review. Nat. Clin. Pract. Neurol. 2007, 3, 331–340. [Google Scholar]
- Kalaria, R.N. Diabetes, microvascular pathology and Alzheimer disease. Nat. Rev. Neurol. 2009, 5, 305–306. [Google Scholar]
- Redondo, M.J.; Hagopian, W.A.; Oram, R.; Steck, A.K.; Vehik, K.; Weedon, M.; Balasubramanyam, A.; Dabelea, D. The clinical consequences of heterogeneity within and between different diabetes types. Diabetologia 2020, 63, 2040–2048. [Google Scholar]
- Shen, W.; Taylor, B.; Jin, Q.; Nguyen-Tran, V.; Meeusen, S.; Zhang, Y.-Q.; Kamireddy, A.; Swafford, A.; Powers, A.F.; Walker, J. Inhibition of DYRK1A and GSK3B induces human β-cell proliferation. Nat. Commun. 2015, 6, 1–11. [Google Scholar]
- Butler, A.E.; Janson, J.; Bonner-Weir, S.; Ritzel, R.; Rizza, R.A.; Butler, P.C. β-cell deficit and increased β-cell apoptosis in humans with type 2 diabetes. Diabetes 2003, 52, 102–110. [Google Scholar]
- Schäfer, S.A.; Machicao, F.; Fritsche, A.; Häring, H.-U.; Kantartzis, K. New type 2 diabetes risk genes provide new insights in insulin secretion mechanisms. Diabetes Res. Clin. Pract. 2011, 93, S9–S24. [Google Scholar]
- American Diabetes Association. Diagnosis and classification of diabetes mellitus. Diabetes Care 2014, 37 (Suppl. S1), S81–S90. [Google Scholar]
- American Diabetes Association. Diagnosis and classification of diabetes mellitus. Standards of medical care in diabetes. Diabetes Care 2019, 41 (Suppl. S1), S13–S27. [Google Scholar]
- Qaid, M.M.; Abdelrahman, M.M. Role of insulin and other related hormones in energy metabolism—A review. Cogent Food Agric. 2016, 2, 1267691. [Google Scholar]
- Singh, S.P.; Ninov, N. The triumvirate of beta-cell regeneration: Solutions and bottlenecks to curing diabetes. Int. J. Dev. Biol. 2018, 62, 453–464. [Google Scholar]
- Grote, C.W.; Wright, D.E. A role for insulin in diabetic neuropathy. Front. Neurosci. 2016, 10, 581. [Google Scholar]
- Modi, P. Diabetes beyond insulin: Review of new drugs for treatment of diabetes mellitus. Curr. Drug Discov. Technol. 2007, 4, 39–47. [Google Scholar]
- Holst, J.J.; Gromada, J. Role of incretin hormones in the regulation of insulin secretion in diabetic and nondiabetic humans. Am. J. Physiol. Endocrinol. Metab. 2004, 287, E199–E206. [Google Scholar]
- Tokarz, V.L.; MacDonald, P.E.; Klip, A. The cell biology of systemic insulin function. J. Cell Biol. 2018, 217, 2273–2289. [Google Scholar]
- Röder, P.V.; Wu, B.; Liu, Y.; Han, W. Pancreatic regulation of glucose homeostasis. Exp. Mol. Med. 2016, 48, e219. [Google Scholar]
- Roep, B.O.; Wheeler, D.C.; Peakman, M. Antigen-based immune modulation therapy for type 1 diabetes: The era of precision medicine. Lancet Diabetes Endocrinol. 2019, 7, 65–74. [Google Scholar]
- Pathak, V.; Pathak, N.M.; O’Neill, C.L.; Guduric-Fuchs, J.; Medina, R.J. Therapies for type 1 diabetes: Current scenario and future perspectives. Clin. Med. Insights Endocrinol. Diabetes 2019, 12, 1179551419844521. [Google Scholar]
- Norris, J.M.; Johnson, R.K.; Stene, L.C. Type 1 diabetes—Early life origins and changing epidemiology. Lancet Diabetes Endocrinol. 2020, 8, 226–238. [Google Scholar]
- Vantyghem, M.-C.; de Koning, E.J.; Pattou, F.; Rickels, M.R. Advances in β-cell replacement therapy for the treatment of type 1 diabetes. Lancet 2019, 394, 1274–1285. [Google Scholar]
- Stumvoll, M.; Goldstein, B.J.; van Haeften, T.W. Type 2 diabetes: Pathogenesis and treatment. Lancet 2008, 371, 2153–2156. [Google Scholar]
- Chao, E.C.; Henry, R.R. SGLT2 inhibition—A novel strategy for diabetes treatment. Nat. Rev. Drug Discov. 2010, 9, 551–559. [Google Scholar]
- Ferrannini, E.; Solini, A. SGLT2 inhibition in diabetes mellitus: Rationale and clinical prospects. Nat. Rev. Endocrinol. 2012, 8, 495. [Google Scholar]
- Rosenstock, J.; Ferrannini, E. Euglycemic diabetic ketoacidosis: A predictable, detectable, and preventable safety concern with SGLT2 inhibitors. Diabetes Care 2015, 38, 1638–1642. [Google Scholar]
- Ackeifi, C.; Wang, P.; Karakose, E.; Fox, J.E.M.; González, B.J.; Liu, H.; Wilson, J.; Swartz, E.; Berrouet, C.; Li, Y. GLP-1 receptor agonists synergize with DYRK1A inhibitors to potentiate functional human β cell regeneration. Sci. Transl. Med. 2020, 12, eaaw9996. [Google Scholar]
- Meier, J.J. GLP-1 receptor agonists for individualized treatment of type 2 diabetes mellitus. Nat. Rev. Endocrinol. 2012, 8, 728–742. [Google Scholar]
- Nauck, M.; Vardarli, I.; Deacon, C.; Holst, J.J.; Meier, J. Secretion of glucagon-like peptide-1 (GLP-1) in type 2 diabetes: What is up, what is down? Diabetologia 2011, 54, 10–18. [Google Scholar]
- Shields, B.; Hicks, S.; Shepherd, M.; Colclough, K.; Hattersley, A.T.; Ellard, S. Maturity-onset diabetes of the young (MODY): How many cases are we missing? Diabetologia 2010, 53, 2504–2508. [Google Scholar]
- Gupta, R.K.; Kaestner, K.H. HNF-4α: From MODY to late-onset type 2 diabetes. Trends Mol. Med. 2004, 10, 521–524. [Google Scholar]
- Thomas, H.; Jaschkowitz, K.; Bulman, M.; Frayling, T.M.; Mitchell, S.M.; Roosen, S.; Lingott-Frieg, A.; Tack, C.J.; Ellard, S.; Ryffel, G.U. A distant upstream promoter of the HNF-4 α gene connects the transcription factors involved in maturity-onset diabetes of the young. Hum. Mol. Genet. 2001, 10, 2089–2097. [Google Scholar]
- Ellard, S. Hepatocyte nuclear factor 1 alpha (HNF-1α) mutations in maturity-onset diabetes of the young. Hum. Mutat. 2000, 16, 377–385. [Google Scholar]
- McDonald, T.J.; McEneny, J.; Pearson, E.R.; Thanabalasingham, G.; Szopa, M.; Shields, B.M.; Ellard, S.; Owen, K.R.; Malecki, M.T.; Hattersley, A.T. Lipoprotein composition in HNF1A-MODY: Differentiating between HNF1A-MODY and type 2 diabetes. Clin. Chim. Acta 2012, 413, 927–932. [Google Scholar]
- Matschinsky, F.; Liang, Y.; Kesavan, P.; Wang, L.; Froguel, P.; Velho, G.; Cohen, D.; Permutt, M.; Tanizawa, Y.; Jetton, T. Glucokinase as pancreatic beta cell glucose sensor and diabetes gene. J. Clin. Investig. 1993, 92, 2092–2098. [Google Scholar]
- Galán, M.; Vincent, O.; Roncero, I.; Azriel, S.; Boix-Pallares, P.; Delgado-Alvarez, E.; Díaz-Cadórniga, F.; Blázquez, E.; Navas, M.-A. Effects of novel maturity-onset diabetes of the young (MODY)-associated mutations on glucokinase activity and protein stability. Biochem. J. 2006, 393, 389–396. [Google Scholar]
- Hattersley, A.; Turner, R.; Patel, P.; O’Rahilly, S.; Wainscoat, J.; Permutt, M.; Tanazawa, Y.; Chin, K.; Watkins, P. Linkage of type 2 diabetes to the glucokinase gene. Lancet 1992, 339, 1307–1310. [Google Scholar]
- Molven, A.; Ringdal, M.; Nordbø, A.M.; Ræder, H.; Støy, J.; Lipkind, G.M.; Steiner, D.F.; Philipson, L.H.; Bergmann, I.; Aarskog, D. Mutations in the insulin gene can cause MODY and autoantibody-negative type 1 diabetes. Diabetes 2008, 57, 1131–1135. [Google Scholar]
- Carlsson, A.; Shepherd, M.; Ellard, S.; Weedon, M.; Lernmark, Å.; Forsander, G.; Colclough, K.; Brahimi, Q.; Valtonen-Andre, C.; Ivarsson, S.A. Absence of islet autoantibodies and modestly raised glucose values at diabetes diagnosis should lead to testing for MODY: Lessons from a 5-year pediatric swedish national cohort study. Diabetes Care 2020, 43, 82–89. [Google Scholar]
- Fajans, S.S.; Bell, G.I. MODY: History, genetics, pathophysiology, and clinical decision making. Diabetes Care 2011, 34, 1878–1884. [Google Scholar]
- Sims-Robinson, C.; Kim, B.; Rosko, A.; Feldman, E.L. How does diabetes accelerate Alzheimer disease pathology? Nat. Rev. Neurol. 2010, 6, 551. [Google Scholar]
- Li, L.; Hölscher, C. Common pathological processes in Alzheimer disease and type 2 diabetes: A review. Brain Res. Rev. 2007, 56, 384–402. [Google Scholar]
- Arnold, S.E.; Arvanitakis, Z.; Macauley-Rambach, S.L.; Koenig, A.M.; Wang, H.-Y.; Ahima, R.S.; Craft, S.; Gandy, S.; Buettner, C.; Stoeckel, L.E. Brain insulin resistance in type 2 diabetes and Alzheimer disease: Concepts and conundrums. Nat. Rev. Neurol. 2018, 14, 168–181. [Google Scholar]
- Suzanne, M. Type 3 diabetes is sporadic Alzheimer’s disease: Mini-review. Eur. Neuropsychopharmacol. 2014, 24, 1954–1960. [Google Scholar]
- Freude, S.; Schilbach, K.; Schubert, M. The role of IGF-1 receptor and insulin receptor signaling for the pathogenesis of Alzheimer’s disease: From model organisms to human disease. Curr. Alzheimer Res. 2009, 6, 213–223. [Google Scholar]
- Moloney, A.M.; Griffin, R.J.; Timmons, S.; O’Connor, R.; Ravid, R.; O’Neill, C. Defects in IGF-1 receptor, insulin receptor and IRS-1/2 in Alzheimer’s disease indicate possible resistance to IGF-1 and insulin signalling. Neurobiol. Aging 2010, 31, 224–243. [Google Scholar]
- Talbot, K.; Wang, H.-Y.; Kazi, H.; Han, L.-Y.; Bakshi, K.P.; Stucky, A.; Fuino, R.L.; Kawaguchi, K.R.; Samoyedny, A.J.; Wilson, R.S. Demonstrated brain insulin resistance in Alzheimer’s disease patients is associated with IGF-1 resistance, IRS-1 dysregulation, and cognitive decline. J. Clin. Investig. 2012, 122, 1316–1338. [Google Scholar]
- Kariharan, T.; Nanayakkara, G.; Parameshwaran, K.; Bagasrawala, I.; Ahuja, M.; Abdel-Rahman, E.; Amin, A.T.; Dhanasekaran, M.; Suppiramaniam, V.; Amin, R.H. Central activation of PPAR-gamma ameliorates diabetes induced cognitive dysfunction and improves BDNF expression. Neurobiol. Aging 2015, 36, 1451–1461. [Google Scholar]
- Qiu, W.Q.; Folstein, M.F. Insulin, insulin-degrading enzyme and amyloid-β peptide in Alzheimer’s disease: Review and hypothesis. Neurobiol. Aging 2006, 27, 190–198. [Google Scholar]
- Li, H.; Wu, J.; Zhu, L.; Sha, L.; Yang, S.; Wei, J.; Ji, L.; Tang, X.; Mao, K.; Cao, L. Insulin degrading enzyme contributes to the pathology in a mixed model of Type 2 diabetes and Alzheimer’s disease: Possible mechanisms of IDE in T2D and AD. Biosci. Rep. 2018, 38, BSR20170862. [Google Scholar]
- Rad, S.K.; Arya, A.; Karimian, H.; Madhavan, P.; Rizwan, F.; Koshy, S.; Prabhu, G. Mechanism involved in insulin resistance via accumulation of β-amyloid and neurofibrillary tangles: Link between type 2 diabetes and Alzheimer’s disease. Drug Des. Dev. Ther. 2018, 12, 3999. [Google Scholar]
- Wang, P.; Alvarez-Perez, J.-C.; Felsenfeld, D.P.; Liu, H.; Sivendran, S.; Bender, A.; Kumar, A.; Sanchez, R.; Scott, D.K.; Garcia-Ocaña, A. Induction of human pancreatic beta cell replication by inhibitors of dual specificity tyrosine regulated kinase. Nat. Med. 2015, 21, 383. [Google Scholar]
- Chen, C.; Cohrs, C.M.; Stertmann, J.; Bozsak, R.; Speier, S. Human beta cell mass and function in diabetes: Recent advances in knowledge and technologies to understand disease pathogenesis. Mol. Metab. 2017, 6, 943–957. [Google Scholar]
- Rachdi, L.; Kariyawasam, D.; Guez, F.; Aïello, V.; Arbonés, M.L.; Janel, N.; Delabar, J.-M.; Polak, M.; Scharfmann, R. Dyrk1a haploinsufficiency induces diabetes in mice through decreased pancreatic beta cell mass. Diabetologia 2014, 57, 960–969. [Google Scholar]
- Janel, N.; Sarazin, M.; Corlier, F.; Corne, H.; De Souza, L.C.; Hamelin, L.; Aka, A.; Lagarde, J.; Blehaut, H.; Hindié, V. Plasma DYRK1A as a novel risk factor for Alzheimer’s disease. Transl. Psychiatry 2014, 4, e425. [Google Scholar]
- Kargbo, R.B. Selective DYRK1A Inhibitor for the Treatment of Neurodegenerative Diseases: Alzheimer, Parkinson, Huntington, and Down Syndrome; ACS Publications: Washington, DC, USA, 2020. [Google Scholar]
- Mittal, K.; Mani, R.J.; Katare, D.P. Type 3 diabetes: Cross talk between differentially regulated proteins of type 2 diabetes mellitus and Alzheimer’s disease. Sci. Rep. 2016, 6, 25589. [Google Scholar]
- Cohen, P. The role of protein phosphorylation in human health and disease. FEBS J. 2001, 268, 5001–5010. [Google Scholar]
- Melnikova, I.; Golden, J. Targeting protein kinases. Nat. Rev. Drug Discov. 2004, 3, 993–994. [Google Scholar]
- Saini, V. Molecular mechanisms of insulin resistance in type 2 diabetes mellitus. World J. Diabetes 2010, 1, 68. [Google Scholar]
- Tanti, J.-F.; Jager, J. Cellular mechanisms of insulin resistance: Role of stress-regulated serine kinases and insulin receptor substrates (IRS) serine phosphorylation. Curr. Opin. Pharmacol. 2009, 9, 753–762. [Google Scholar]
- Zick, Y. Role of Ser/Thr kinases in the uncoupling of insulin signaling. Int. J. Obes. 2003, 27, S56–S60. [Google Scholar]
- Gao, Z.; Zhang, X.; Zuberi, A.; Hwang, D.; Quon, M.J.; Lefevre, M.; Ye, J. Inhibition of insulin sensitivity by free fatty acids requires activation of multiple serine kinases in 3T3-L1 adipocytes. Mol. Endocrinol. 2004, 18, 2024–2034. [Google Scholar]
- Farese, R.V.; Sajan, M.P.; Standaert, M.L. Insulin-sensitive protein kinases (atypical protein kinase C and protein kinase B/Akt): Actions and defects in obesity and type II diabetes. Exp. Biol. Med. 2005, 230, 593–605. [Google Scholar]
- Schultze, S.M.; Hemmings, B.A.; Niessen, M.; Tschopp, O. PI3K/AKT, MAPK and AMPK signalling: Protein kinases in glucose homeostasis. Expert Rev. Mol. Med. 2012, 14, e1. [Google Scholar]
- Choi, K.; Kim, Y.-B. Molecular mechanism of insulin resistance in obesity and type 2 diabetes. Korean J. Intern. Med. 2010, 25, 119. [Google Scholar]
- Liu, L.; Tan, L.; Lai, J.; Li, S.; Wang, D.W. Enhanced Rho-kinase activity: Pathophysiological relevance in type 2 diabetes. Clin. Chim. Acta 2016, 462, 107–110. [Google Scholar]
- Hotamisligil, G.S.; Peraldi, P.; Budavari, A.; Ellis, R.; White, M.F.; Spiegelman, B.M. IRS-1-mediated inhibition of insulin receptor tyrosine kinase activity in TNF-α-and obesity-induced insulin resistance. Science 1996, 271, 665–670. [Google Scholar]
- Chopra, I.; Li, H.; Wang, H.; Webster, K.A. Phosphorylation of the insulin receptor by AMP-activated protein kinase (AMPK) promotes ligand-independent activation of the insulin signalling pathway in rodent muscle. Diabetologia 2012, 55, 783–794. [Google Scholar]
- Donath, M.Y.; Shoelson, S.E. Type 2 diabetes as an inflammatory disease. Nat. Rev. Immunol. 2011, 11, 98–107. [Google Scholar]
- Lontchi-Yimagou, E.; Sobngwi, E.; Matsha, T.E.; Kengne, A.P. Diabetes mellitus and inflammation. Curr. Diabetes Rep. 2013, 13, 435–444. [Google Scholar]
- Rehman, K.; Akash, M.S.H. Mechanisms of inflammatory responses and development of insulin resistance: How are they interlinked? J. Biomed. Sci. 2016, 23, 1–18. [Google Scholar]
- Park, S.-H.; Liu, Z.; Sui, Y.; Helsley, R.N.; Zhu, B.; Powell, D.K.; Kern, P.A.; Zhou, C. IKKβ is essential for adipocyte survival and adaptive adipose remodeling in obesity. Diabetes 2016, 65, 1616–1629. [Google Scholar]
- Xu, J.; Jia, Y.-F.; Tapadar, S.; Weaver, J.D.; Raji, I.O.; Pithadia, D.J.; Javeed, N.; García, A.J.; Choi, D.-S.; Matveyenko, A.V. Inhibition of TBK1/IKKε promotes regeneration of pancreatic β-cells. Sci. Rep. 2018, 8, 15587. [Google Scholar]
- Becker, W.; Joost, H.-G. Structural and functional characteristics of Dyrk, a novel subfamily of protein kinases with dual specificity. In Progress in Nucleic Acid Research and Molecular Biology; Elsevier: Amsterdam, The Netherlands, 1998; Volume 62, pp. 1–17. [Google Scholar]
- Kentrup, H.; Becker, W.; Heukelbach, J.; Wilmes, A.; Schürmann, A.; Huppertz, C.; Kainulainen, H.; Joost, H.-G. Dyrk, a dual specificity protein kinase with unique structural features whose activity is dependent on tyrosine residues between subdomains VII and VIII. J. Biol. Chem. 1996, 271, 3488–3495. [Google Scholar]
- Becker, W.; Weber, Y.; Wetzel, K.; Eirmbter, K.; Tejedor, F.J.; Joost, H.-G. Sequence characteristics, subcellular localization, and substrate specificity of DYRK-related kinases, a novel family of dual specificity protein kinases. J. Biol. Chem. 1998, 273, 25893–25902. [Google Scholar]
- Nolen, B.; Taylor, S.; Ghosh, G. Regulation of protein kinases: Controlling activity through activation segment conformation. Mol. Cell 2004, 15, 661–675. [Google Scholar]
- Becker, W.; Sippl, W. Activation, regulation, and inhibition of DYRK1A. FEBS J. 2011, 278, 246–256. [Google Scholar]
- Wiechmann, S.; Czajkowska, H.; de Graaf, K.; Grötzinger, J.; Joost, H.-G.; Becker, W. Unusual function of the activation loop in the protein kinase DYRK1A. Biochem. Biophys. Res. Commun. 2003, 302, 403–408. [Google Scholar]
- Soundararajan, M.; Roos, A.K.; Savitsky, P.; Filippakopoulos, P.; Kettenbach, A.N.; Olsen, J.V.; Gerber, S.A.; Eswaran, J.; Knapp, S.; Elkins, J.M. Structures of Down syndrome kinases, DYRKs, reveal mechanisms of kinase activation and substrate recognition. Structure 2013, 21, 986–996. [Google Scholar]
- Himpel, S.; Panzer, P.; Eirmbter, K.; Czajkowska, H.; Sayed, M.; Packman, L.C.; Blundell, T.; Kentrup, H.; Grötzinger, J.; Joost, H.-G. Identification of the autophosphorylation sites and characterization of their effects in the protein kinase DYRK1A. Biochem. J. 2001, 359, 497–505. [Google Scholar]
- Walte, A.; Rüben, K.; Birner-Gruenberger, R.; Preisinger, C.; Bamberg-Lemper, S.; Hilz, N.; Bracher, F.; Becker, W. Mechanism of dual specificity kinase activity of DYRK 1 A. FEBS J. 2013, 280, 4495–4511. [Google Scholar]
- Lochhead, P.A.; Sibbet, G.; Morrice, N.; Cleghon, V. Activation-loop autophosphorylation is mediated by a novel transitional intermediate form of DYRKs. Cell 2005, 121, 925–936. [Google Scholar]
- Kii, I.; Sumida, Y.; Goto, T.; Sonamoto, R.; Okuno, Y.; Yoshida, S.; Kato-Sumida, T.; Koike, Y.; Abe, M.; Nonaka, Y. Selective inhibition of the kinase DYRK1A by targeting its folding process. Nat. Commun. 2016, 7, 1–14. [Google Scholar]
- Duchon, A.; Herault, Y. DYRK1A, a dosage-sensitive gene involved in neurodevelopmental disorders, is a target for drug development in Down syndrome. Front. Behav. Neurosci. 2016, 10, 104. [Google Scholar]
- Soppa, U.; Becker, W. DYRK protein kinases. Curr. Biol. 2015, 25, R488–R489. [Google Scholar]
- Kim, M.-Y.; Jeong, B.C.; Lee, J.H.; Kee, H.J.; Kook, H.; Kim, N.S.; Kim, Y.H.; Kim, J.-K.; Ahn, K.Y.; Kim, K.K. A repressor complex, AP4 transcription factor and geminin, negatively regulates expression of target genes in nonneuronal cells. Proc. Natl. Acad. Sci. USA 2006, 103, 13074–13079. [Google Scholar]
- Yadav, R.R.; Sharma, S.; Joshi, P.; Wani, A.; Vishwakarma, R.A.; Kumar, A.; Bharate, S.B. Meridianin derivatives as potent Dyrk1A inhibitors and neuroprotective agents. Bioorg. Med. Chem. Lett. 2015, 25, 2948–2952. [Google Scholar]
- Maenz, B.; Hekerman, P.; Vela, E.M.; Galceran, J.; Becker, W. Characterization of the human DYRK1A promoter and its regulation by the transcription factor E2F1. BMC Mol. Biol. 2008, 9, 30. [Google Scholar]
- Aranda, S.; Laguna, A.; Luna, S.D.L. DYRK family of protein kinases: Evolutionary relationships, biochemical properties, and functional roles. FASEB J. 2011, 25, 449–462. [Google Scholar]
- Lee, Y.; Ha, J.; Kim, H.J.; Kim, Y.-S.; Chang, E.-J.; Song, W.-J.; Kim, H.-H. Negative feedback Inhibition of NFATc1 by DYRK1A regulates bone homeostasis. J. Biol. Chem. 2009, 284, 33343–33351. [Google Scholar]
- Minematsu, H.; Shin, M.J.; Aydemir, A.B.C.; Kim, K.-O.; Nizami, S.A.; Chung, G.-J.; Lee, F.Y.-I. Nuclear presence of nuclear factor of activated T cells (NFAT) c3 and c4 is required for Toll-like receptor-activated innate inflammatory response of monocytes/macrophages. Cell. Signal. 2011, 23, 1785–1793. [Google Scholar]
- Arron, J.R.; Winslow, M.M.; Polleri, A.; Chang, C.-P.; Wu, H.; Gao, X.; Neilson, J.R.; Chen, L.; Heit, J.J.; Kim, S.K. NFAT dysregulation by increased dosage of DSCR1 and DYRK1A on chromosome 21. Nature 2006, 441, 595–600. [Google Scholar]
- Liao, J.-M.; Zhou, X.; Zhang, Y.; Lu, H. MiR-1246: A new link of the p53 family with cancer and Down syndrome. Cell Cycle 2012, 11, 2624–2630. [Google Scholar]
- Fernandez-Martinez, P.; Zahonero, C.; Sanchez-Gomez, P. DYRK1A: The double-edged kinase as a protagonist in cell growth and tumorigenesis. Mol. Cell. Oncol. 2015, 2, e970048. [Google Scholar]
- Miyata, Y.; Shibata, T.; Aoshima, M.; Tsubata, T.; Nishida, E. The molecular chaperone TRiC/CCT binds to the Trp-Asp 40 (WD40) repeat protein WDR68 and promotes its folding, protein kinase DYRK1A binding, and nuclear accumulation. J. Biol. Chem. 2014, 289, 33320–33332. [Google Scholar]
- Yousefelahiyeh, M.; Xu, J.; Alvarado, E.; Yu, Y.; Salven, D.; Nissen, R.M. DCAF7/WDR68 is required for normal levels of DYRK1A and DYRK1B. PLoS ONE 2018, 13, e0207779. [Google Scholar]
- Miyata, Y.; Nishida, E. DYRK1A binds to an evolutionarily conserved WD40-repeat protein WDR68 and induces its nuclear translocation. Biochim. Biophys. Acta Mol. Cell Res. 2011, 1813, 1728–1739. [Google Scholar]
- Kurabayashi, N.; Hirota, T.; Sakai, M.; Sanada, K.; Fukada, Y. DYRK1A and glycogen synthase kinase 3β, a dual-kinase mechanism directing proteasomal degradation of CRY2 for circadian timekeeping. Mol. Cell. Biol. 2010, 30, 1757–1768. [Google Scholar]
- Li, D.; Jackson, R.A.; Yusoff, P.; Guy, G.R. Direct association of Sprouty-related protein with an EVH1 domain (SPRED) 1 or SPRED2 with DYRK1A modifies substrate/kinase interactions. J. Biol. Chem. 2010, 285, 35374–35385. [Google Scholar]
- Woods, Y.L.; Cohen, P.; Becker, W.; Jakes, R.; Goedert, M.; Wang, X.; Proud, C.G. The kinase DYRK phosphorylates protein-synthesis initiation factor eIF2Bε at Ser539 and the microtubule-associated protein tau at Thr212: Potential role for DYRK as a glycogen synthase kinase 3-priming kinase. Biochem. J. 2001, 355, 609–615. [Google Scholar]
- Jung, M.-S.; Park, J.-H.; Ryu, Y.S.; Choi, S.-H.; Yoon, S.-H.; Kwen, M.-Y.; Oh, J.Y.; Song, W.-J.; Chung, S.-H. Regulation of RCAN1 protein activity by Dyrk1A protein-mediated phosphorylation. J. Biol. Chem. 2011, 286, 40401–40412. [Google Scholar]
- Wiseman, F.K.; Alford, K.A.; Tybulewicz, V.L.; Fisher, E.M. Down syndrome—Recent progress and future prospects. Hum. Mol. Genet. 2009, 18, R75–R83. [Google Scholar]
- Di Vona, C.; Bezdan, D.; Islam, A.B.; Salichs, E.; López-Bigas, N.; Ossowski, S.; de la Luna, S. Chromatin-wide profiling of DYRK1A reveals a role as a gene-specific RNA polymerase II CTD kinase. Mol. Cell 2015, 57, 506–520. [Google Scholar]
- Aranda, S.; Alvarez, M.; Turró, S.; Laguna, A.; de la Luna, S. Sprouty2-mediated inhibition of fibroblast growth factor signaling is modulated by the protein kinase DYRK1A. Mol. Cell. Biol. 2008, 28, 5899–5911. [Google Scholar]
- Litovchick, L.; Florens, L.A.; Swanson, S.K.; Washburn, M.P.; DeCaprio, J.A. DYRK1A protein kinase promotes quiescence and senescence through DREAM complex assembly. Genes Dev. 2011, 25, 801–813. [Google Scholar]
- Yang, E.J.; Ahn, Y.S.; Chung, K.C. Protein kinase Dyrk1 activates cAMP response element-binding protein during neuronal differentiation in hippocampal progenitor cells. J. Biol. Chem. 2001, 276, 39819–39824. [Google Scholar]
- Rena, G.; Woods, Y.L.; Prescott, A.R.; Peggie, M.; Unterman, T.G.; Williams, M.R.; Cohen, P. Two novel phosphorylation sites on FKHR that are critical for its nuclear exclusion. EMBO J. 2002, 21, 2263–2271. [Google Scholar]
- Seifert, A.; Allan, L.A.; Clarke, P.R. DYRK1A phosphorylates caspase 9 at an inhibitory site and is potently inhibited in human cells by harmine. FEBS J. 2008, 275, 6268–6280. [Google Scholar]
- Seifert, A.; Clarke, P.R. p38α-and DYRK1A-dependent phosphorylation of caspase-9 at an inhibitory site in response to hyperosmotic stress. Cell. Signal. 2009, 21, 1626–1633. [Google Scholar]
- Fernandez-Martinez, J.; Vela, E.M.; Tora-Ponsioen, M.; Ocaña, O.H.; Nieto, M.A.; Galceran, J. Attenuation of Notch signalling by the Down-syndrome-associated kinase DYRK1A. J. Cell Sci. 2009, 122, 1574–1583. [Google Scholar]
- Skurat, A.V.; Dietrich, A.D. Phosphorylation of Ser640 in muscle glycogen synthase by DYRK family protein kinases. J. Biol. Chem. 2004, 279, 2490–2498. [Google Scholar]
- Chen, J.-Y.; Lin, J.-R.; Tsai, F.-C.; Meyer, T. Dosage of Dyrk1a shifts cells within a p21-cyclin D1 signaling map to control the decision to enter the cell cycle. Mol. Cell 2013, 52, 87–100. [Google Scholar]
- Kumar, K.; Suebsuwong, C.; Wang, P.; Garcia-Ocana, A.; Stewart, A.F.; DeVita, R.J. DYRK1A inhibitors as potential therapeutics for β-cell regeneration for diabetes. J. Med. Chem. 2021, 64, 2901–2922. [Google Scholar]
- Park, J.; Song, W.-J.; Chung, K.C. Function and regulation of Dyrk1A: Towards understanding Down syndrome. Cell. Mol. Life Sci. 2009, 66, 3235–3240. [Google Scholar]
- American Diabetes Association. 9. Pharmacologic approaches to glycemic treatment: Standards of medical care in diabetes—2019. Diabetes Care 2019, 42 (Suppl. S1), S90–S102. [Google Scholar]
- Saunders, D.; Powers, A.C. Replicative capacity of β-cells and type 1 diabetes. J. Autoimmun. 2016, 71, 59–68. [Google Scholar]
- Rathwa, N.; Patel, R.; Palit, S.P.; Parmar, N.; Rana, S.; Ansari, M.I.; Ramachandran, A.; Begum, R. β-cell replenishment: Possible curative approaches for diabetes mellitus. Nutr. Metab. Cardiovasc. Dis. 2020, 30, 1870–1881. [Google Scholar]
- Bain, J.; Plater, L.; Elliott, M.; Shpiro, N.; Hastie, C.J.; Mclauchlan, H.; Klevernic, I.; Arthur, J.S.C.; Alessi, D.R.; Cohen, P. The selectivity of protein kinase inhibitors: A further update. Biochem. J. 2007, 408, 297–315. [Google Scholar]
- Göckler, N.; Jofre, G.; Papadopoulos, C.; Soppa, U.; Tejedor, F.J.; Becker, W. Harmine specifically inhibits protein kinase DYRK1A and interferes with neurite formation. FEBS J. 2009, 276, 6324–6337. [Google Scholar]
- Ogawa, Y.; Nonaka, Y.; Goto, T.; Ohnishi, E.; Hiramatsu, T.; Kii, I.; Yoshida, M.; Ikura, T.; Onogi, H.; Shibuya, H. Development of a novel selective inhibitor of the Down syndrome-related kinase Dyrk1A. Nat. Commun. 2010, 1, 1–9. [Google Scholar]
- Wurzlbauer, A.; Rüben, K.; Gürdal, E.; Chaikuad, A.; Knapp, S.; Sippl, W.; Becker, W.; Bracher, F. How to separate kinase inhibition from undesired monoamine oxidase a inhibition—The development of the DYRK1A inhibitor AnnH75 from the Alkaloid Harmine. Molecules 2020, 25, 5962. [Google Scholar]
- Kumar, K.; Wang, P.; Swartz, E.A.; Khamrui, S.; Secor, C.; BLazarus, M.; Sanchez, R.; Stewart, A.F.; DeVita, R.J. Structure–activity relationships and biological evaluation of 7-substituted harmine analogs for human β-cell proliferation. Molecules 2020, 25, 1983. [Google Scholar]
- Kumar, K.; Wang, P.; Wilson, J.; Zlatanic, V.; Berrouet, C.; Khamrui, S.; Secor, C.; Swartz, E.A.; Lazarus, M.; Sanchez, R. Synthesis and biological validation of a harmine-based, central nervous system (CNS)-Avoidant, selective, human β-cell regenerative dual-specificity tyrosine phosphorylation-regulated kinase a (DYRK1A) inhibitor. J. Med. Chem. 2020, 63, 2986–3003. [Google Scholar]
- Ackeifi, C.; Swartz, E.; Kumar, K.; Liu, H.; Chalada, S.; Karakose, E.; Scott, D.K.; Garcia-Ocaña, A.; Sanchez, R.; DeVita, R.J. Pharmacologic and genetic approaches define human pancreatic β cell mitogenic targets of DYRK1A inhibitors. JCI Insight 2020, 5, e132594. [Google Scholar]
- Tahtouh, T.; Elkins, J.M.; Filippakopoulos, P.; Soundararajan, M.; Burgy, G.; Durieu, E.; Cochet, C.; Schmid, R.S.; Lo, D.C.; Delhommel, F. Selectivity, cocrystal structures, and neuroprotective properties of leucettines, a family of protein kinase inhibitors derived from the marine sponge alkaloid leucettamine B. J. Med. Chem. 2012, 55, 9312–9330. [Google Scholar]
- Fant, X.; Durieu, E.; Chicanne, G.; Payrastre, B.; Sbrissa, D.; Shisheva, A.; Limanton, E.; Carreaux, F.; Bazureau, J.-P.; Meijer, L. CLK/DYRK kinases inhibitor Leucettine L41 induces mTOR-dependent autophagy. Implication for Alzheimers’ disease. Mol. Pharmacol. 2013, 85, 837. [Google Scholar]
- Souchet, B.; Audrain, M.; Billard, J.M.; Dairou, J.; Fol, R.; Orefice, N.S.; Tada, S.; Gu, Y.; Dufayet-Chaffaud, G.; Limanton, E. Inhibition of DYRK1A proteolysis modifies its kinase specificity and rescues Alzheimer phenotype in APP/PS1 mice. Acta Neuropathol. Commun. 2019, 7, 46. [Google Scholar]
- Grabher, P.; Durieu, E.; Kouloura, E.; Halabalaki, M.; Skaltsounis, L.A.; Meijer, L.; Hamburger, M.; Potterat, O. Library-based discovery of DYRK1A/CLK1 inhibitors from natural product extracts. Planta Med. 2012, 78, 951–956. [Google Scholar]
- Neagoie, C.; Vedrenne, E.; Buron, F.; Mérour, J.-Y.; Rosca, S.; Bourg, S.; Lozach, O.; Meijer, L.; Baldeyrou, B.; Lansiaux, A. Synthesis of chromeno [3, 4-b] indoles as Lamellarin D analogues: A novel DYRK1A inhibitor class. Eur. J. Med. Chem. 2012, 49, 379–396. [Google Scholar]
- Falke, H.; Chaikuad, A.; Becker, A.; Loaëc, N.; Lozach, O.; Abu Jhaisha, S.; Becker, W.; Jones, P.G.; Preu, L.; Baumann, K. 10-Iodo-11 H-indolo [3, 2-c] quinoline-6-carboxylic acids are selective inhibitors of DYRK1A. J. Med. Chem. 2015, 58, 3131–3143. [Google Scholar]
- Foucourt, A.; Hédou, D.; Dubouilh-Benard, C.; Désiré, L.; Casagrande, A.-S.; Leblond, B.; Loäec, N.; Meijer, L.; Besson, T. Design and synthesis of thiazolo [5, 4-f] quinazolines as DYRK1A inhibitors, Part I. Molecules 2014, 19, 15546–15571. [Google Scholar]
- Foucourt, A.; Hédou, D.; Dubouilh-Benard, C.; Girard, A.; Taverne, T.; Casagrande, A.-S.; Désiré, L.; Leblond, B.; Besson, T. Design and synthesis of thiazolo [5, 4-f] quinazolines as DYRK1A inhibitors, part II. Molecules 2014, 19, 15411–15439. [Google Scholar]
- Fruit, C.; Couly, F.; Bhansali, R.; Rammohan, M.; Lindberg, M.F.; Crispino, J.D.; Meijer, L.; Besson, T. Biological characterization of 8-cyclopropyl-2-(pyridin-3-yl) thiazolo [5, 4-f] quinazolin-9 (8h)-one, a promising inhibitor of DYRK1A. Pharmaceuticals 2019, 12, 185. [Google Scholar]
- Lechner, C.; Flaßhoff, M.; Falke, H.; Preu, L.; Loaëc, N.; Meijer, L.; Knapp, S.; Chaikuad, A.; Kunick, C. [b]-annulated halogen-substituted indoles as potential DYRK1A inhibitors. Molecules 2019, 24, 4090. [Google Scholar]
- Meine, R.; Becker, W.; Falke, H.; Preu, L.; Loaëc, N.; Meijer, L.; Kunick, C. Indole-3-carbonitriles as DYRK1A inhibitors by fragment-based drug design. Molecules 2018, 23, 64. [Google Scholar]
- Kassis, P.; Brzeszcz, J.; Bénéteau, V.; Lozach, O.; Meijer, L.; Le Guével, R.; Guillouzo, C.; Lewiński, K.; Bourg, S.; Colliandre, L. Synthesis and biological evaluation of new 3-(6-hydroxyindol-2-yl)-5-(Phenyl) pyridine or pyrazine V-Shaped molecules as kinase inhibitors and cytotoxic agents. Eur. J. Med. Chem. 2011, 46, 5416–5434. [Google Scholar]
- Loidreau, Y.; Marchand, P.; Dubouilh-Benard, C.; Nourrisson, M.-R.; Duflos, M.; Loaëc, N.; Meijer, L.; Besson, T. Synthesis and biological evaluation of N-aryl-7-methoxybenzo [b] furo [3, 2-d] pyrimidin-4-amines and their N-arylbenzo [b] thieno [3, 2-d] pyrimidin-4-amine analogues as dual inhibitors of CLK1 and DYRK1A kinases. Eur. J. Med. Chem. 2013, 59, 283–295. [Google Scholar]
- Dehbi, O.; Tikad, A.; Bourg, S.; Bonnet, P.; Lozach, O.; Meijer, L.; Aadil, M.; Akssira, M.; Guillaumet, G.; Routier, S. Synthesis and optimization of an original V-shaped collection of 4-7-disubstituted Pyrido [3, 2-d] pyrimidines as CDK5 and DYRK1A inhibitors. Eur. J. Med. Chem. 2014, 80, 352–363. [Google Scholar]
- Ferris, J.K.; Inglis, J.T.; Madden, K.M.; Boyd, L.A. Brain and body: A review of central nervous system contributions to movement impairments in diabetes. Diabetes 2020, 69, 3–11. [Google Scholar]
- Bruel, A.; Bénéteau, R.; Chabanne, M.; Lozach, O.; Le Guevel, R.; Ravache, M.; Bénédetti, H.; Meijer, L.; Logé, C.; Robert, J.-M. Synthesis of new pyridazino [4, 5-b] indol-4-ones and pyridazin-3 (2H)-one analogs as DYRK1A inhibitors. Bioorg. Med. Chem. Lett. 2014, 24, 5037–5040. [Google Scholar]
- Marti, G.; Eparvier, V.; Morleo, B.; Ven, J.L.; Apel, C.; Bodo, B.; Amand, S.; Dumontet, V.; Lozach, O.; Meijer, L. Natural aristolactams and aporphine alkaloids as inhibitors of CDK1/Cyclin B and DYRK1A. Molecules 2013, 18, 3018–3027. [Google Scholar]
- Motati, D.R.; Amaradhi, R.; Ganesh, T. Azaindole therapeutic agents. Bioorg. Med. Chem. 2020, 28, 115830. [Google Scholar]
- Shirakawa, J.; Kulkarni, R.N. Novel factors modulating human β-cell proliferation. Diabetes Obes. Metab. 2016, 18, 71–77. [Google Scholar]
- Mérour, J.-Y.; Buron, F.; Plé, K.; Bonnet, P.; Routier, S. The azaindole framework in the design of kinase inhibitors. Molecules 2014, 19, 19935–19979. [Google Scholar]
- Huang, Z.; Tremblay, M.S.; Wu, T.Y.-H.; Ding, Q.; Hao, X.; Baaten, J.; Hill, R.; Liu, B.; Meeusen, S.; Zou, Y. Discovery of 5-(3, 4-Difluorophenyl)-3-(pyrazol-4-yl)-7-azaindole (GNF3809) for β-cell survival in type 1 diabetes. ACS Omega 2019, 4, 3571–3581. [Google Scholar]
- Liu, Y.A.; Jin, Q.; Ding, Q.; Hao, X.; Mo, T.; Yan, S.; Zou, Y.; Huang, Z.; Zhang, X.; Gao, W. A dual inhibitor of DYRK1A and GSK3β for β-cell proliferation: Aminopyrazine derivative GNF4877. ChemMedChem 2020, 15, 1562–1570. [Google Scholar]
- Liu, Y.A.; Jin, Q.; Zou, Y.; Ding, Q.; Yan, S.; Wang, Z.; Hao, X.; Nguyen, B.; Zhang, X.; Pan, J. Selective DYRK1A inhibitor for the treatment of Type 1 Diabetes: Discovery of 6-azaindole derivative GNF2133. J. Med. Chem. 2020, 63, 2958–2973. [Google Scholar]
- Hohmeier, H.E.; Zhang, L.; Taylor, B.; Stephens, S.; Lu, D.; McNamara, P.; Laffitte, B.; Newgard, C.B. Identification of a small molecule that stimulates human β-cell proliferation and insulin secretion, and protects against cytotoxic stress in rat insulinoma cells. PLoS ONE 2020, 15, e0224344. [Google Scholar]
- Schisler, J.C.; Jensen, P.B.; Taylor, D.G.; Becker, T.C.; Knop, F.K.; Takekawa, S.; German, M.; Weir, G.C.; Lu, D.; Mirmira, R.G. The Nkx6. 1 homeodomain transcription factor suppresses glucagon expression and regulates glucose-stimulated insulin secretion in islet beta cells. Proc. Natl. Acad. Sci. USA 2005, 102, 7297–7302. [Google Scholar]
- Schisler, J.C.; Fueger, P.T.; Babu, D.A.; Hohmeier, H.E.; Tessem, J.S.; Lu, D.; Becker, T.C.; Naziruddin, B.; Levy, M.; Mirmira, R.G. Stimulation of human and rat islet β-cell proliferation with retention of function by the homeodomain transcription factor Nkx6. 1. Mol. Cell. Biol. 2008, 28, 3465–3476. [Google Scholar]
- Eurofins DiscoverX Products. KINOMEscan Technology Platform. Available online: https://www.discoverx.com/technologies-platforms/competitive-binding-technology/kinomescan-technology-platform (accessed on 5 May 2021).
- Czarna, A.; Wang, J.; Zelencova, D.; Liu, Y.; Deng, X.; Choi, H.G.; Zhang, T.; Zhou, W.; Chang, J.W.; Kildalsen, H. Novel scaffolds for Dual specificity tyrosine-phosphorylation-regulated kinase (DYRK1A) inhibitors. J. Med. Chem. 2018, 61, 7560–7572. [Google Scholar]
- Lee, H.-J.; Woo, H.; Lee, H.-E.; Jeon, H.; Ryu, K.-Y.; Han Nam, J.; Jeon, S.G.; Park, H.; Lee, J.-S.; Han, K.-M. The novel DYRK1A inhibitor KVN93 regulates cognitive function, amyloid-beta pathology, and neuroinflammation. Free Radic. Biol. Med. 2020, 160, 575–595. [Google Scholar]
- Allegretti, P.A.; Horton, T.M.; Abdolazimi, Y.; Moeller, H.P.; Yeh, B.; Caffet, M.; Michel, G.; Smith, M.; Annes, J.P. Generation of highly potent DYRK1A-dependent inducers of human β-Cell replication via Multi-Dimensional compound optimization. Bioorg. Med. Chem. 2020, 28, 115193. [Google Scholar]
- Abdolazimi, Y.; Zhao, Z.; Lee, S.; Xu, H.; Allegretti, P.; Horton, T.M.; Yeh, B.; Moeller, H.P.; Nichols, R.J.; McCutcheon, D. CC-401 promotes β-cell replication via pleiotropic consequences of DYRK1A/B inhibition. Endocrinology 2018, 159, 3143–3157. [Google Scholar]
- Wang, P.; Karakose, E.; Liu, H.; Swartz, E.; Ackeifi, C.; Zlatanic, V.; Wilson, J.; González, B.J.; Bender, A.; Takane, K.K. Combined inhibition of DYRK1A, SMAD, and trithorax pathways synergizes to induce robust replication in adult human beta cells. Cell Metab. 2019, 29, 638–652. [Google Scholar]
- Lu, M.; Ma, L.; Shan, P.; Liu, A.; Yu, X.; Jiang, W.; Wang, X.; Zhao, X.; Ye, X.; Wang, T. DYRK1A aggravates β cell dysfunction and apoptosis by promoting the phosphorylation and degradation of IRS2. Exp. Gerontol. 2019, 125, 110659. [Google Scholar]
- Scavuzzo, M.A.; Borowiak, M. Two drugs converged in a pancreatic β cell. Sci. Transl. Med. 2020, 12, eaba7359. [Google Scholar]
- Dowjat, W.K.; Adayev, T.; Kuchna, I.; Nowicki, K.; Palminiello, S.; Hwang, Y.W.; Wegiel, J. Trisomy-driven overexpression of DYRK1A kinase in the brain of subjects with Down syndrome. Neurosci. Lett. 2007, 413, 77–81. [Google Scholar]
- Neumann, F.; Gourdain, S.; Albac, C.; Dekker, A.D.; Bui, L.C.; Dairou, J.; Schmitz-Afonso, I.; Hue, N.; Rodrigues-Lima, F.; Delabar, J.M. DYRK1A inhibition and cognitive rescue in a Down syndrome mouse model are induced by new fluoro-DANDY derivatives. Sci. Rep. 2018, 8, 2859. [Google Scholar]
- Park, J.; Chung, K.C. New perspectives of Dyrk1A role in neurogenesis and neuropathologic features of Down syndrome. Exp. Neurobiol. 2013, 22, 244. [Google Scholar]
- Wegiel, J.; Gong, C.X.; Hwang, Y.W. The role of DYRK1A in neurodegenerative diseases. FEBS J. 2011, 278, 236–245. [Google Scholar]
- De la Torre, R.; De Sola, S.; Pons, M.; Duchon, A.; de Lagran, M.M.; Farré, M.; Fitó, M.; Benejam, B.; Langohr, K.; Rodriguez, J. Epigallocatechin-3-gallate, a DYRK1A inhibitor, rescues cognitive deficits in D own syndrome mouse models and in humans. Mol. Nutr. Food Res. 2014, 58, 278–288. [Google Scholar]
- García-Cerro, S.; Rueda, N.; Vidal, V.; Lantigua, S.; Martínez-Cué, C. Normalizing the gene dosage of Dyrk1A in a mouse model of Down syndrome rescues several Alzheimer’s disease phenotypes. Neurobiol. Dis. 2017, 106, 76–88. [Google Scholar]
- Birger, Y.; Izraeli, S. DYRK1A in Down syndrome: An oncogene or tumor suppressor? J. Clin. Investig. 2012, 122, 807–810. [Google Scholar]
- Xu, X.; Liu, Q.; Zhang, C.; Ren, S.; Xu, L.; Zhao, Z.; Dou, H.; Li, P.; Zhang, X.; Gong, Y. Inhibition of DYRK1A-EGFR axis by p53-MDM2 cascade mediates the induction of cellular senescence. Cell Death Dis. 2019, 10, 282. [Google Scholar]
- Popowicz, G.M.; Czarna, A.; Wolf, S.; Wang, K.; Wang, W.; Dömling, A.; Holak, T.A. Structures of low molecular weight inhibitors bound to MDMX and MDM2 reveal new approaches for p53-MDMX/MDM2 antagonist drug discovery. Cell Cycle 2010, 9, 1104–1111. [Google Scholar]
- Kim, J.-H.; Li, L.; Resar, L.M. Doubling up on function: Dual-specificity tyrosine-regulated kinase 1A (DYRK1A) in B cell acute lymphoblastic leukemia. J. Clin. Investig. 2021, 131, e142627. [Google Scholar]
- Hurtz, C.; Carroll, M.P.; Tasian, S.K.; Wertheim, G.; Bhansali, R.S.; Lee, S.J.; Kumar, A.; Lehman, A.; Loftus, J.P.; Jeschke, G. DYRK1A Is Required to Alleviate Replication Stress in KMT2A-Rearranged Acute Lymphoblastic Leukemia. Blood 2020, 136, 39–40. [Google Scholar]
- Zhao, C.; Wang, D.; Gao, Z.; Kan, H.; Qiu, F.; Chen, L.; Li, H. Licocoumarone induces BxPC-3 pancreatic adenocarcinoma cell death by inhibiting DYRK1A. Chem. Biol. Interact. 2020, 316, 108913. [Google Scholar]
- Luna, J.; Boni, J.; Cuatrecasas, M.; Bofill-De Ros, X.; Núñez-Manchón, E.; Gironella, M.; Vaquero, E.C.; Arbones, M.L.; de la Luna, S.; Fillat, C. DYRK1A modulates c-MET in pancreatic ductal adenocarcinoma to drive tumour growth. Gut 2019, 68, 1465–1476. [Google Scholar]
- Bai, Z.; Du, Y.; Cong, L.; Cheng, Y. The USP22 promotes the growth of cancer cells through the DYRK1A in pancreatic ductal adenocarcinoma. Gene 2020, 758, 144960. [Google Scholar]
- Tarpley, M.; Oladapo, H.O.; Strepay, D.; Caligan, T.B.; Chdid, L.; Shehata, H.; Roques, J.R.; Thomas, R.; Laudeman, C.P.; Onyenwoke, R.U. Identification of harmine and β-carboline analogs from a high-throughput screen of an approved drug collection; profiling as differential inhibitors of DYRK1A and monoamine oxidase A and for in vitro and in vivo anti-cancer studies. Eur. J. Pharm. Sci. 2021, 162, 105821. [Google Scholar]
- Recasens, A.; Humphrey, S.J.; Ellis, M.; Hoque, M.; Abbassi, R.H.; Chen, B.; Longworth, M.; Needham, E.J.; James, D.E.; Johns, T.G. Global phosphoproteomics reveals DYRK1A regulates CDK1 activity in glioblastoma cells. Cell Death Discov. 2021, 7, 81. [Google Scholar]
- Li, Y.L.; Ding, K.; Hu, X.; Wu, L.W.; Zhou, D.M.; Rao, M.J.; Lin, N.M.; Zhang, C. DYRK1A inhibition suppresses STAT3/EGFR/Met signalling and sensitizes EGFR wild-type NSCLC cells to AZD9291. J. Cell. Mol. Med. 2019, 23, 7427–7437. [Google Scholar]
- Pozo, N.; Zahonero, C.; Fernández, P.; Liñares, J.M.; Ayuso, A.; Hagiwara, M.; Pérez, A.; Ricoy, J.R.; Hernández-Laín, A.; Sepúlveda, J.M. Inhibition of DYRK1A destabilizes EGFR and reduces EGFR-dependent glioblastoma growth. J. Clin. Investig. 2013, 123, 2475–2487. [Google Scholar]
- Kim, H.; Lee, K.-S.; Kim, A.-K.; Choi, M.; Choi, K.; Kang, M.; Chi, S.-W.; Lee, M.-S.; Lee, J.-S.; Lee, S.-Y. A chemical with proven clinical safety rescues Down-syndrome-related phenotypes in through DYRK1A inhibition. Dis. Models Mech. 2016, 9, 839–848. [Google Scholar]
- Müller, T.; Clemmensen, C.; Finan, B.; DiMarchi, R.; Tschöp, M. Anti-obesity therapy: From rainbow pills to polyagonists. Pharmacol. Rev. 2018, 70, 712–746. [Google Scholar]

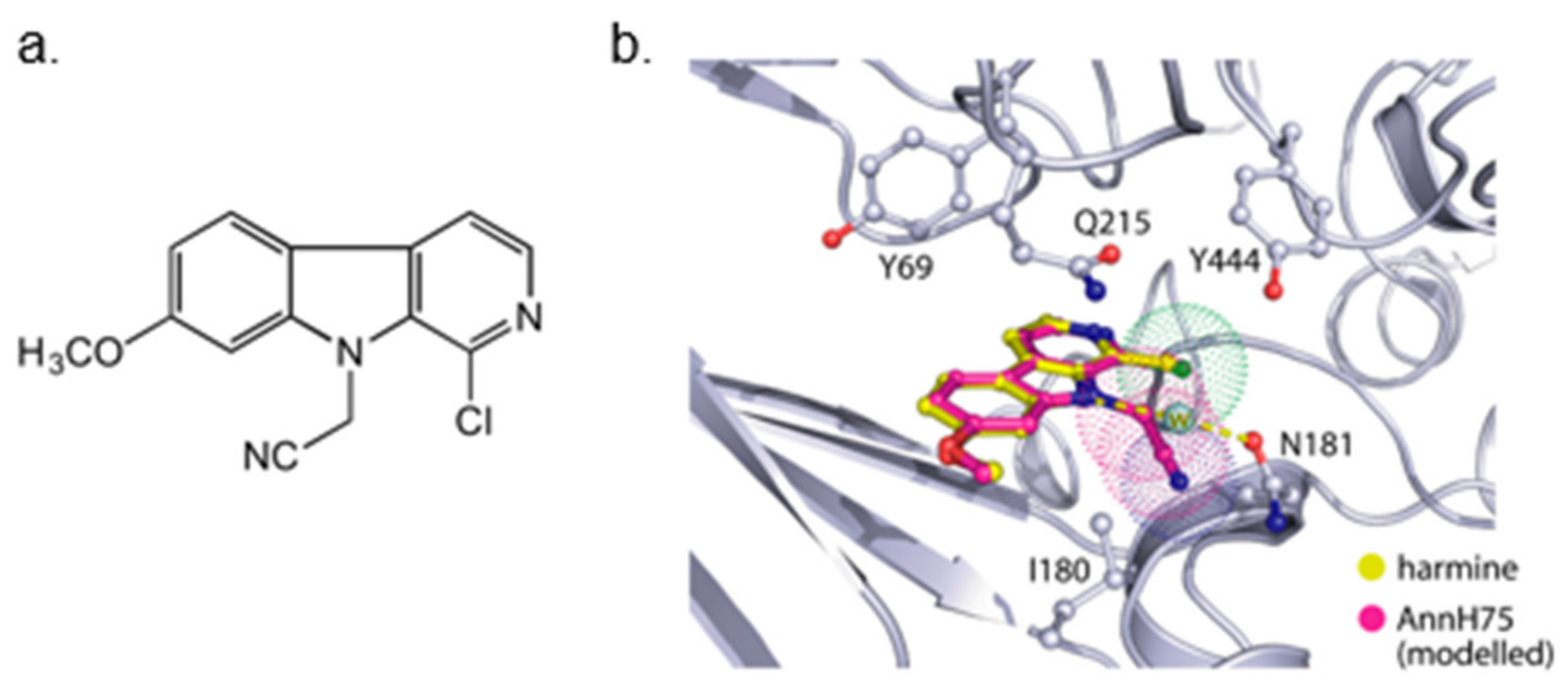
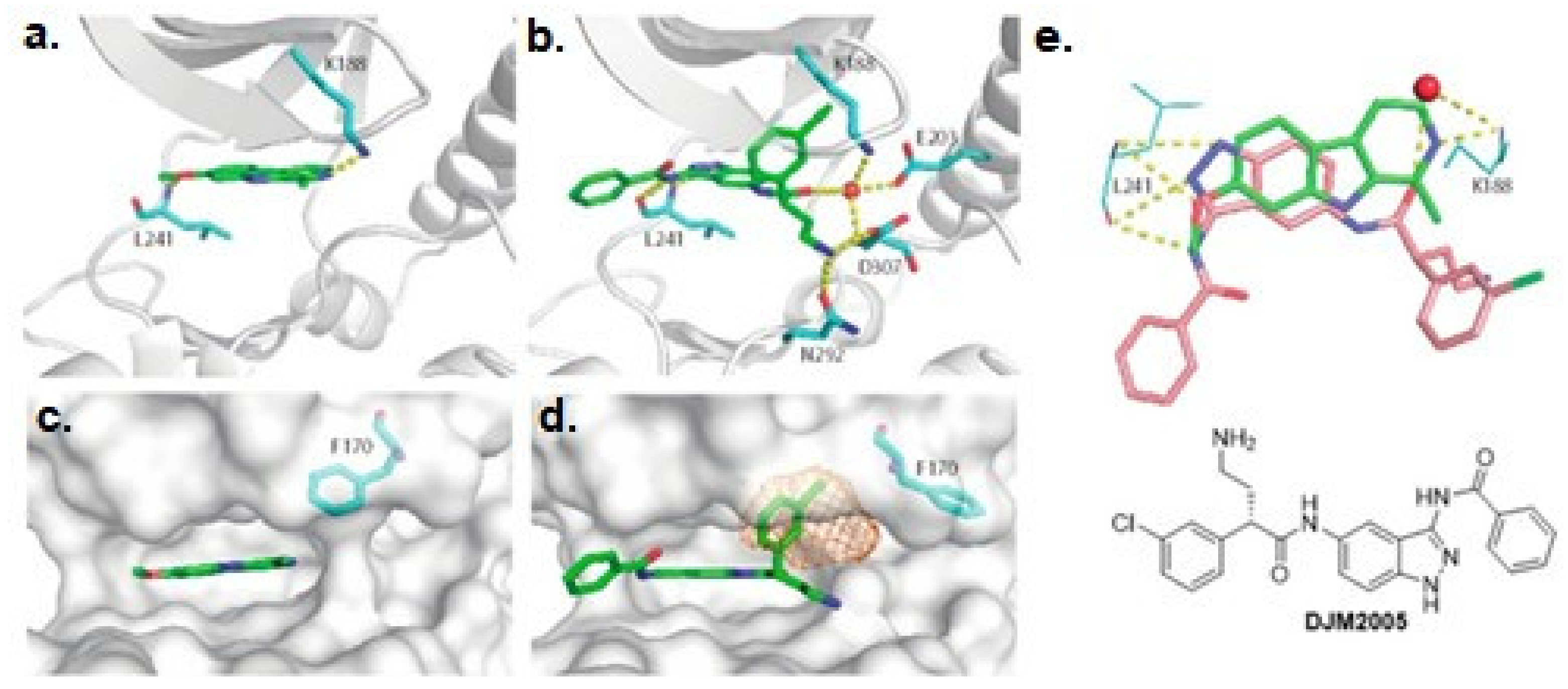

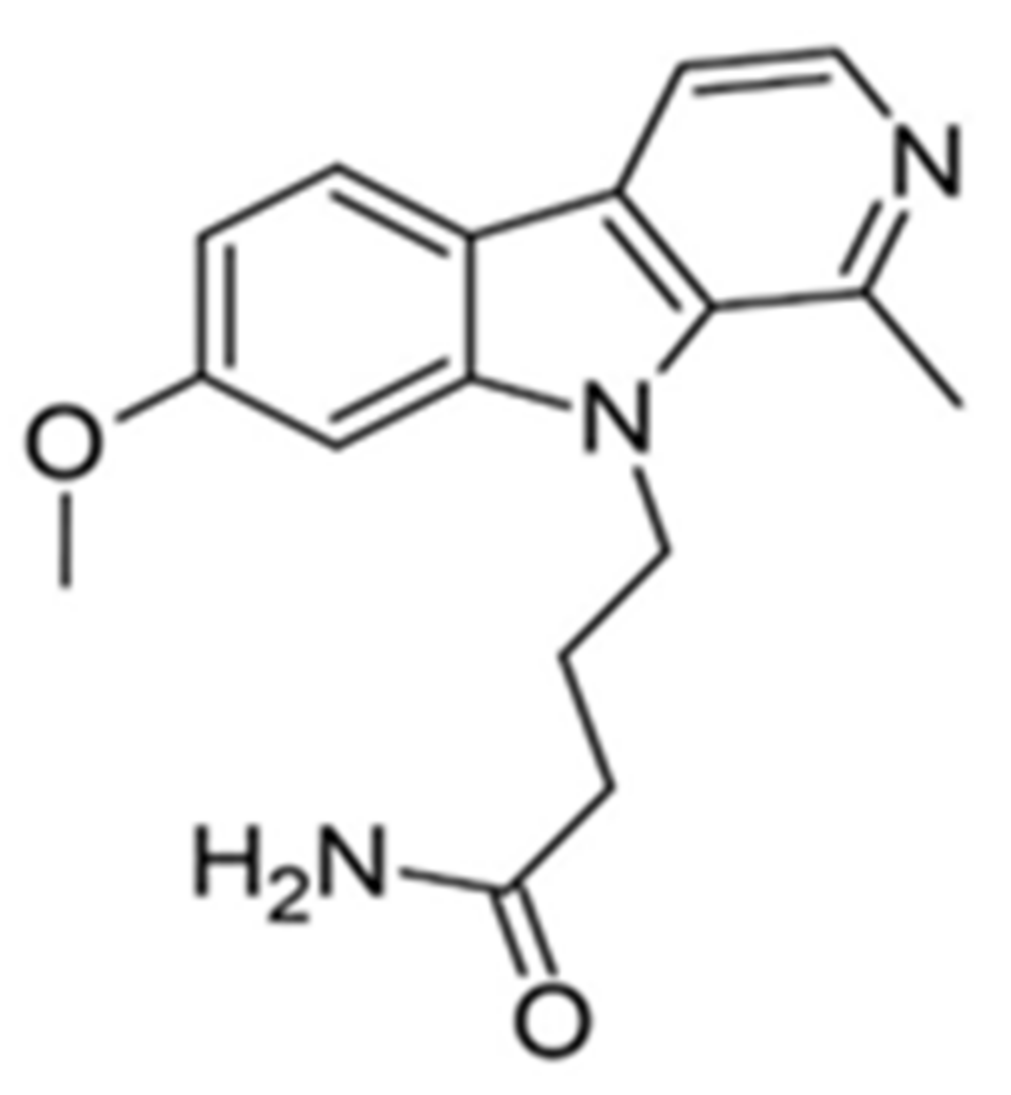
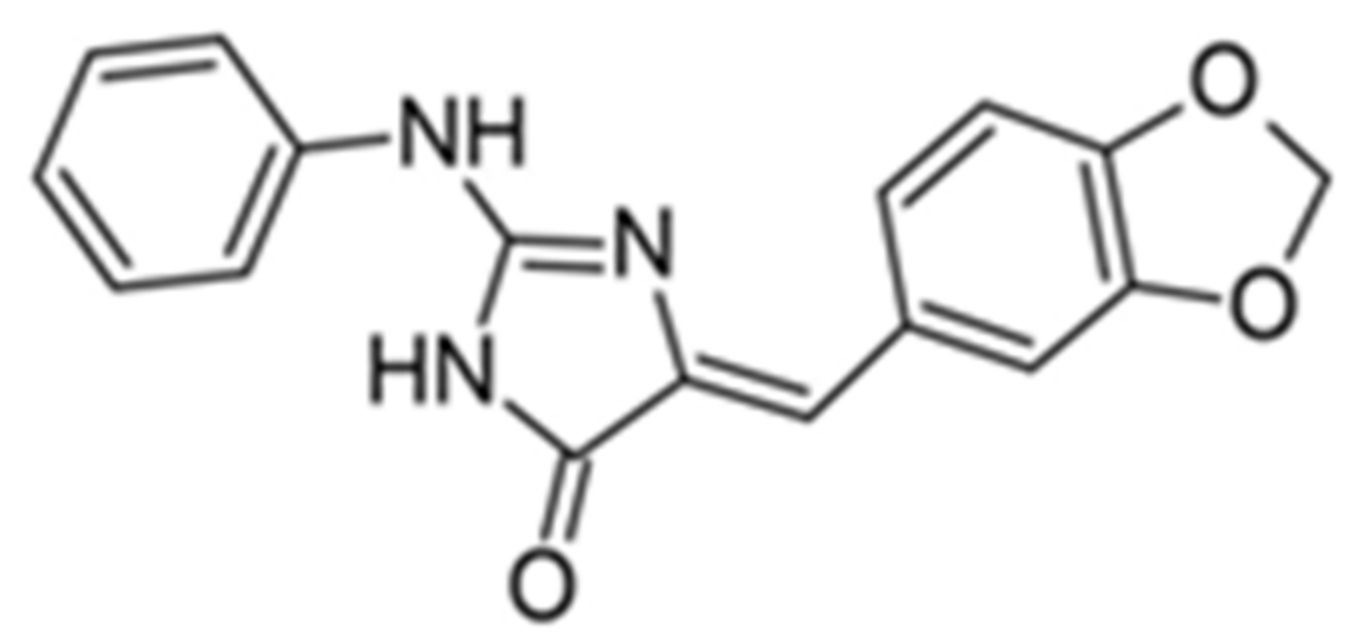
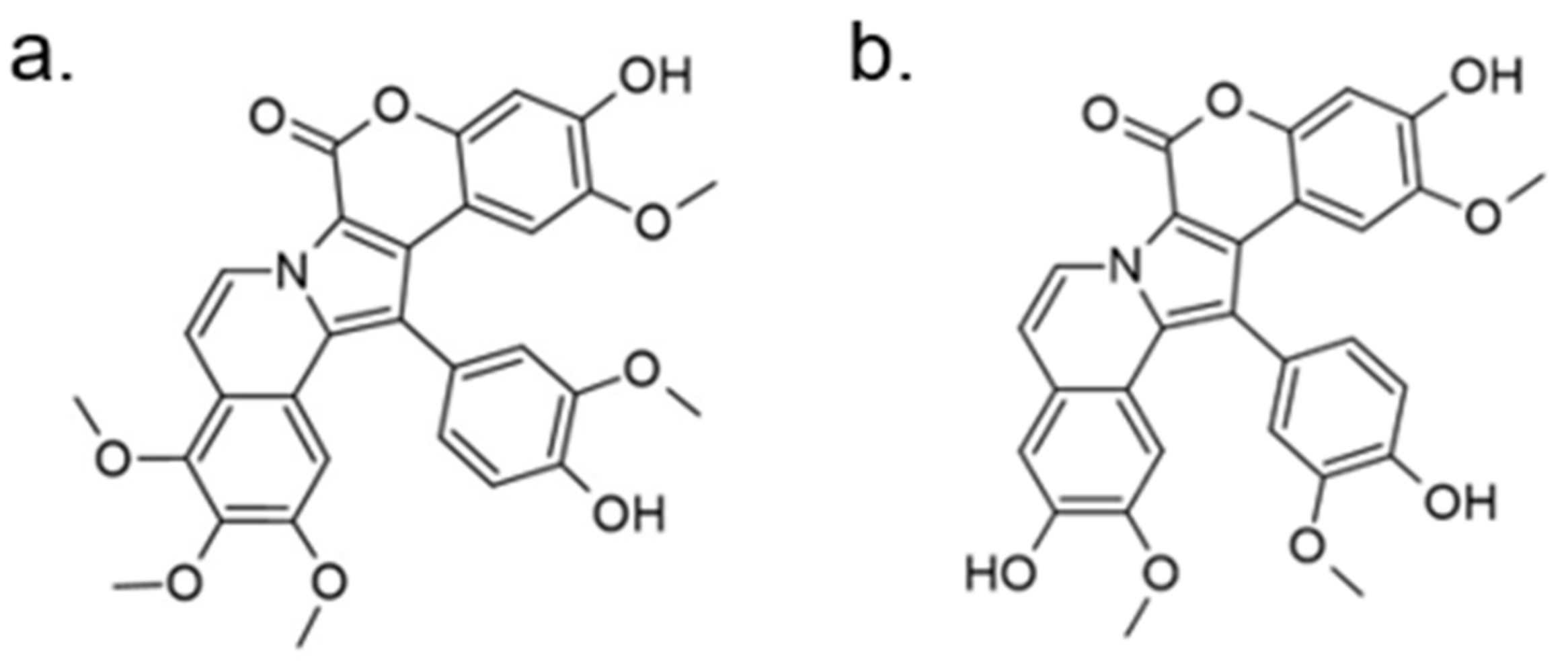
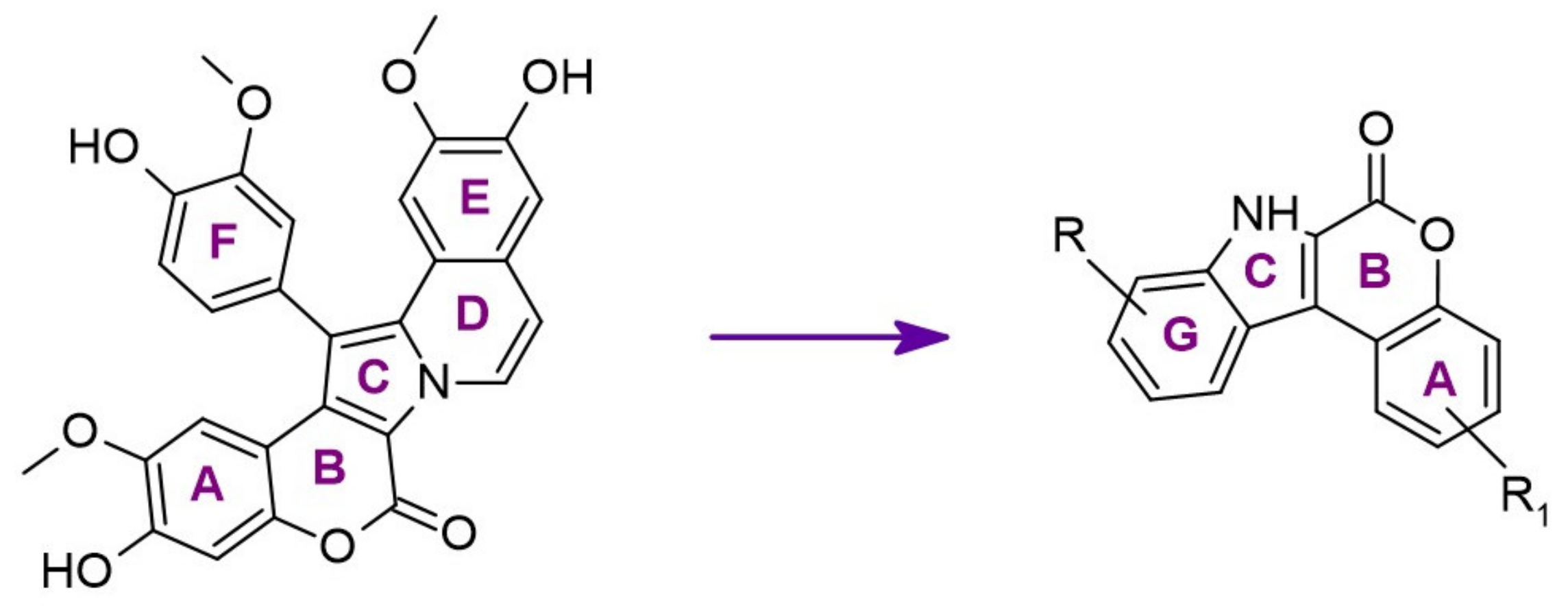
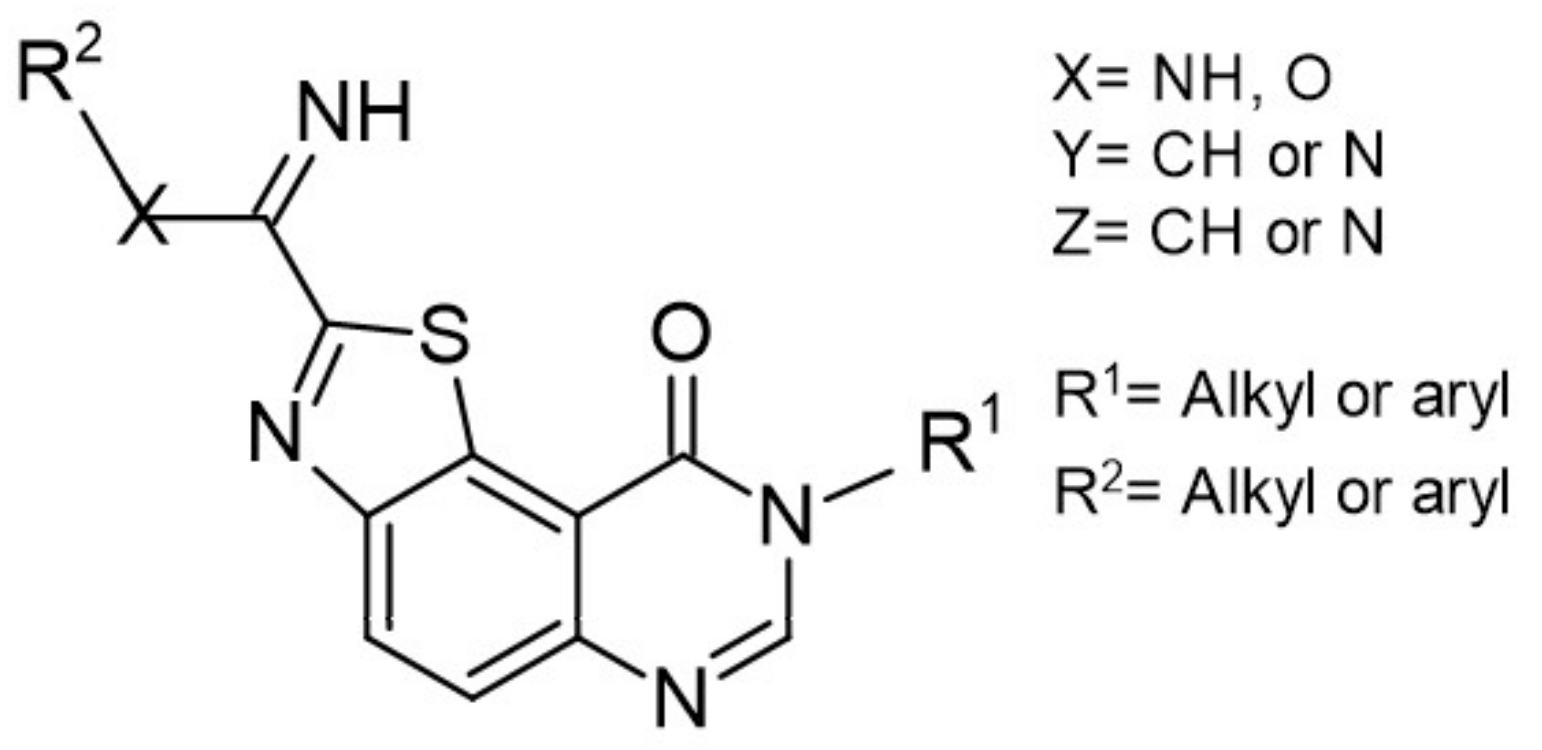
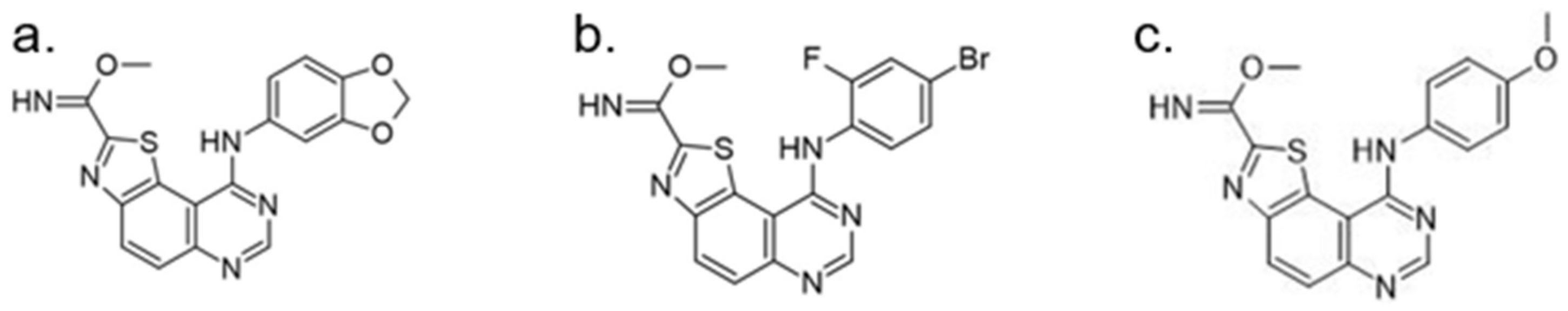
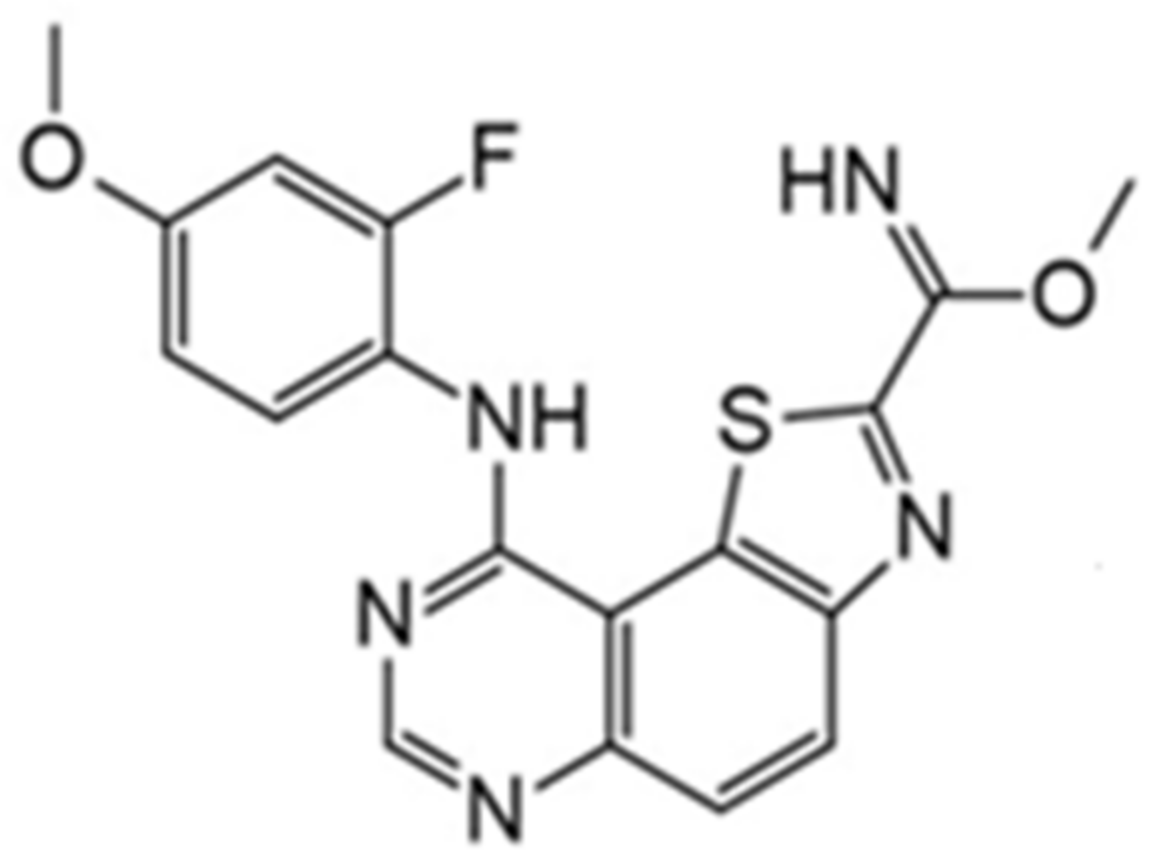
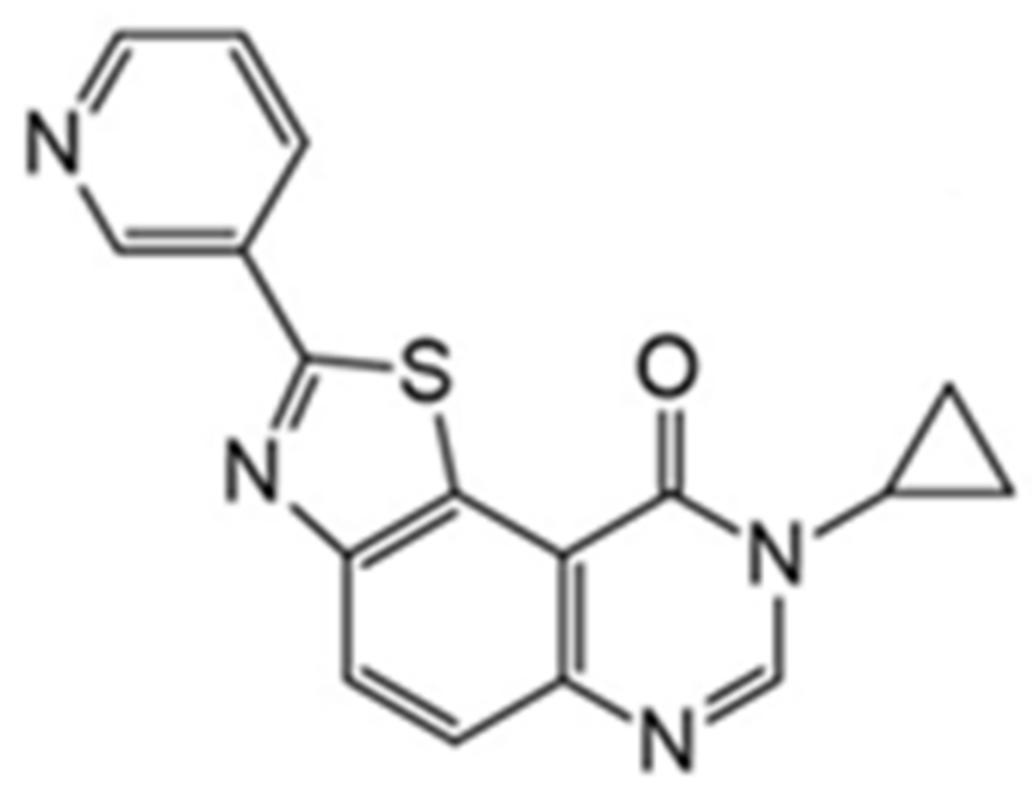
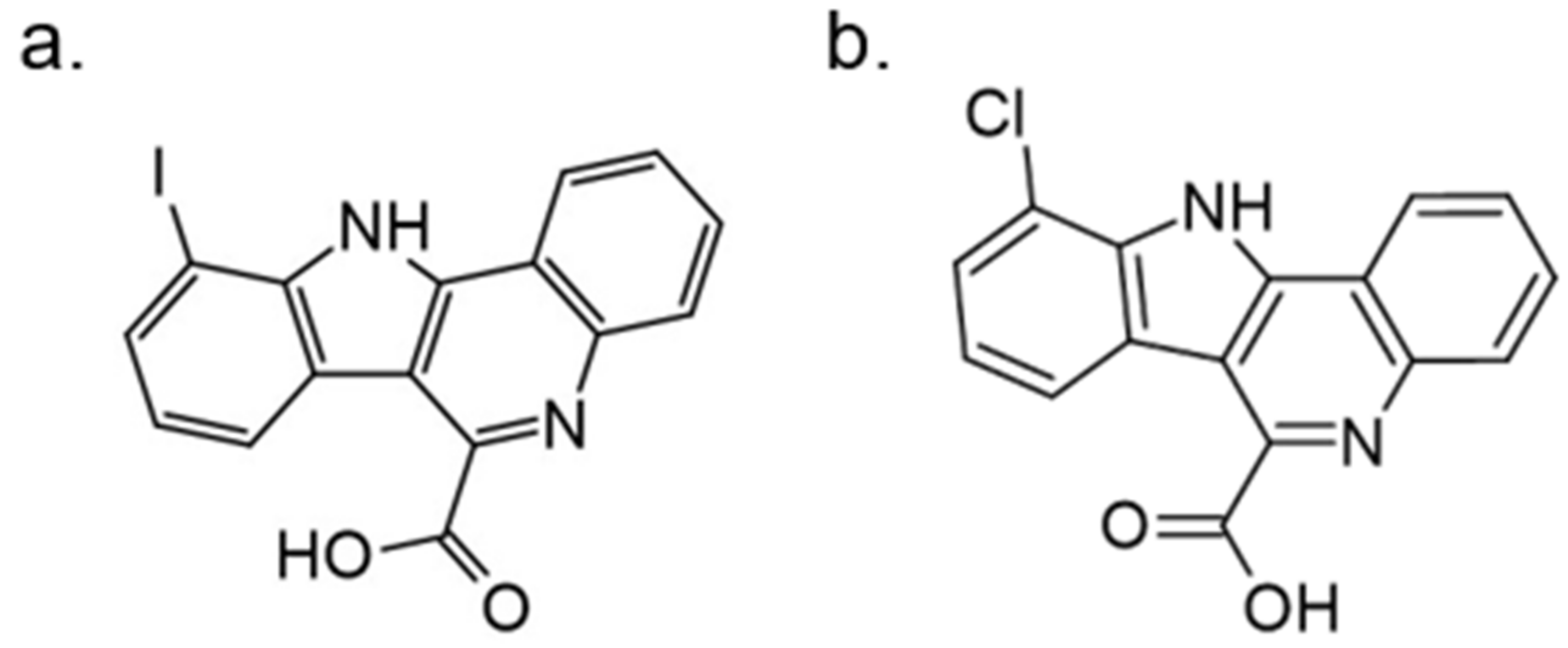
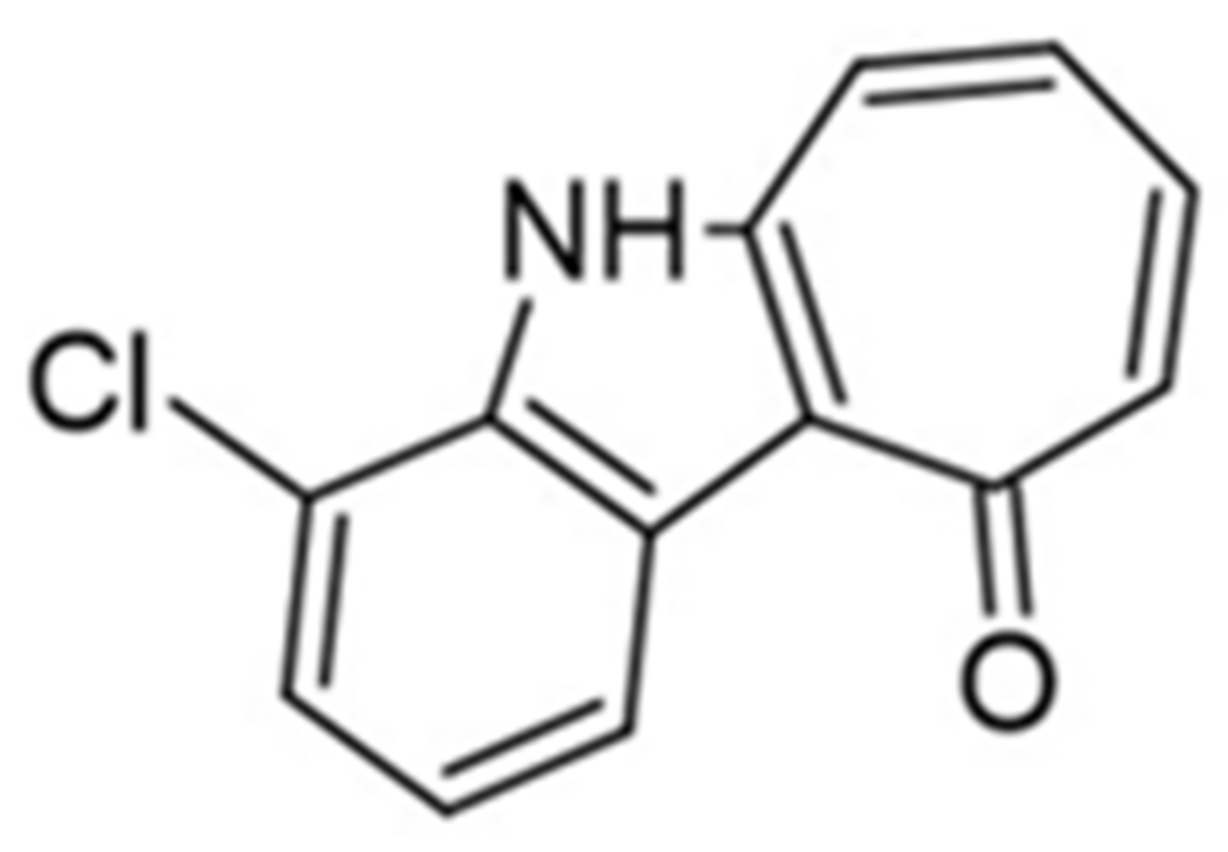
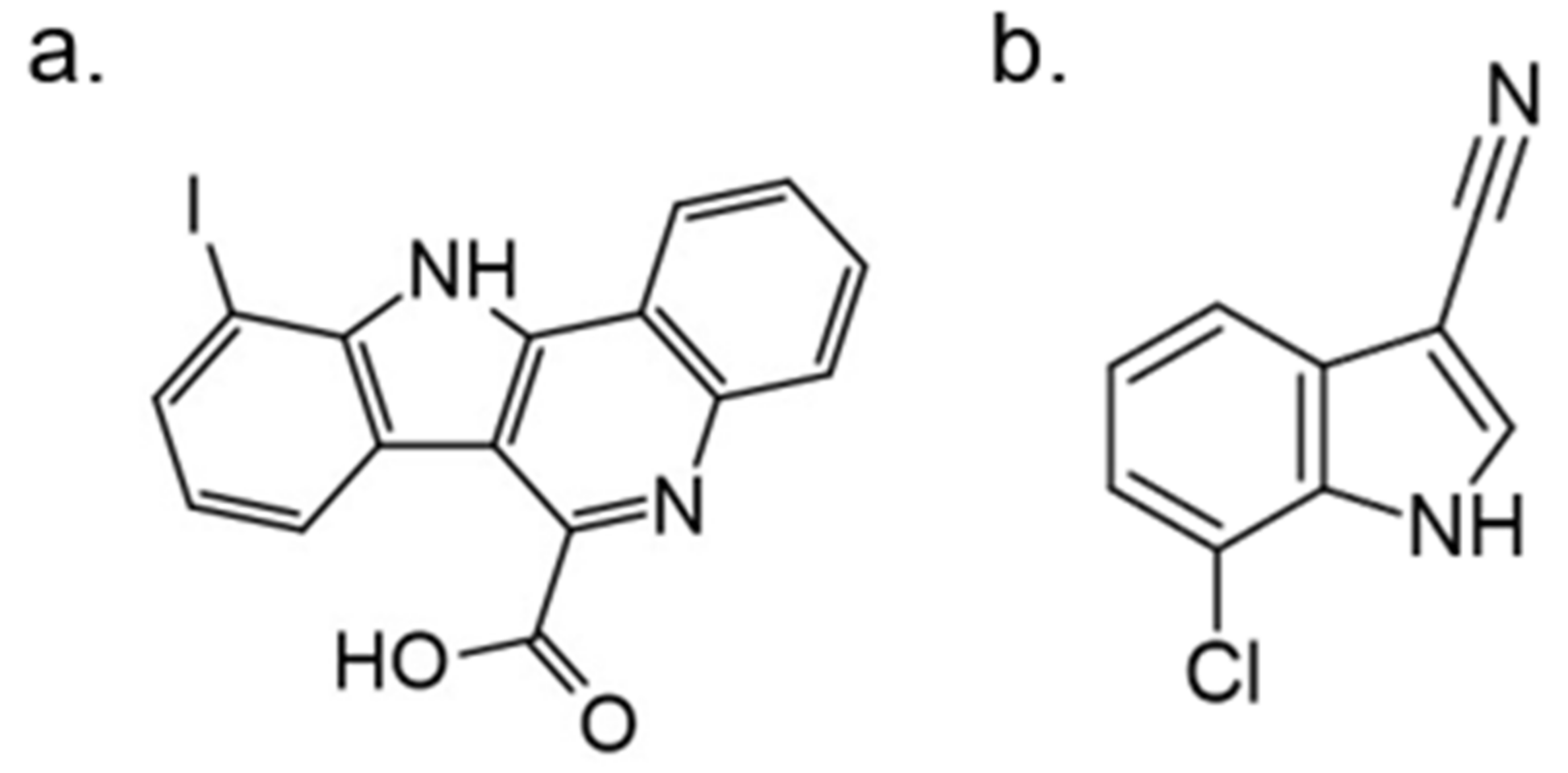
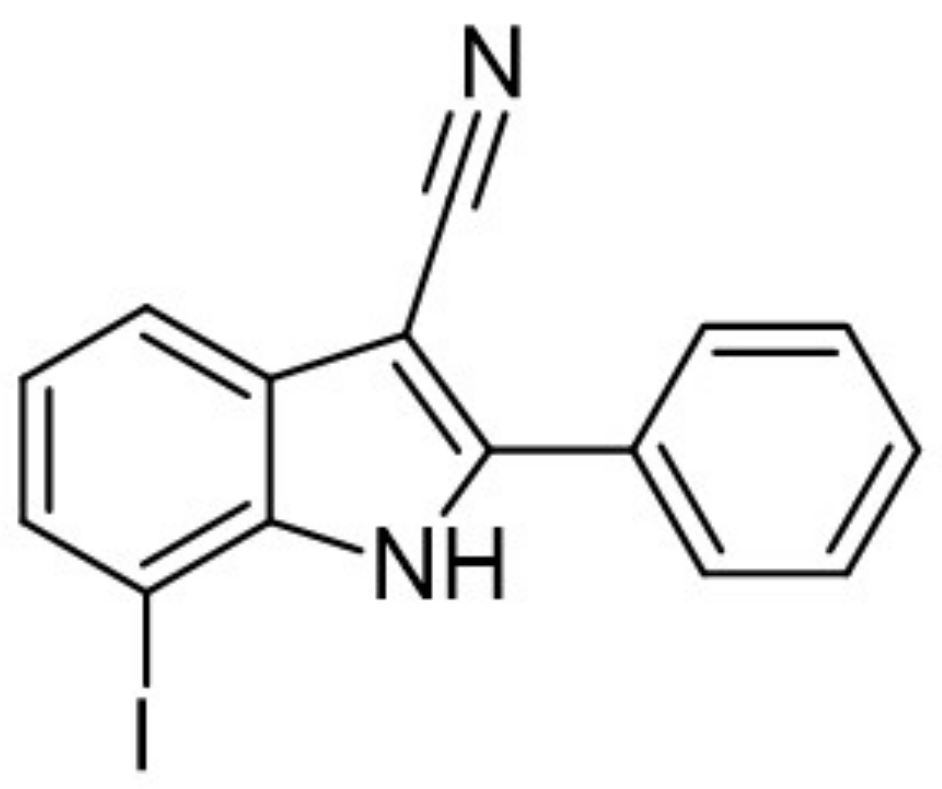
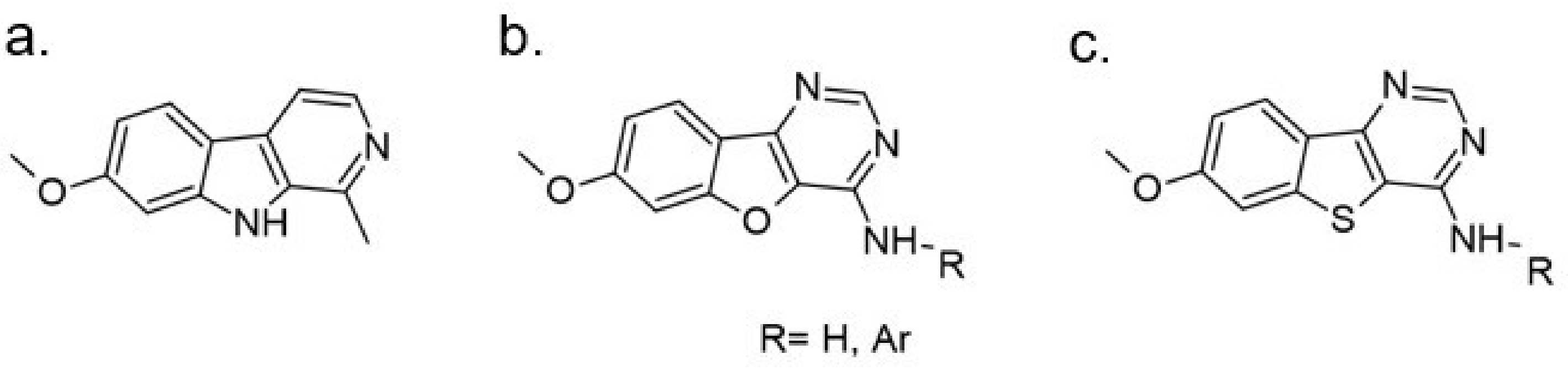
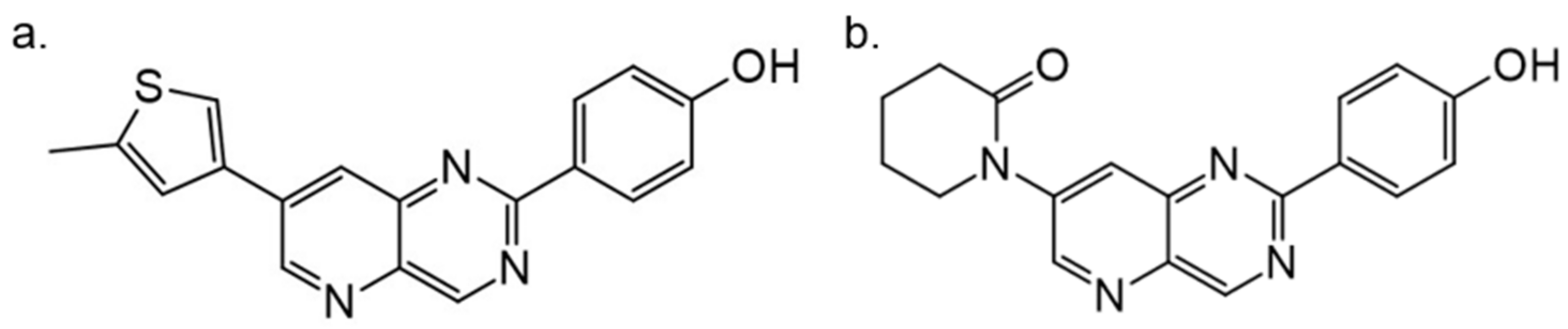

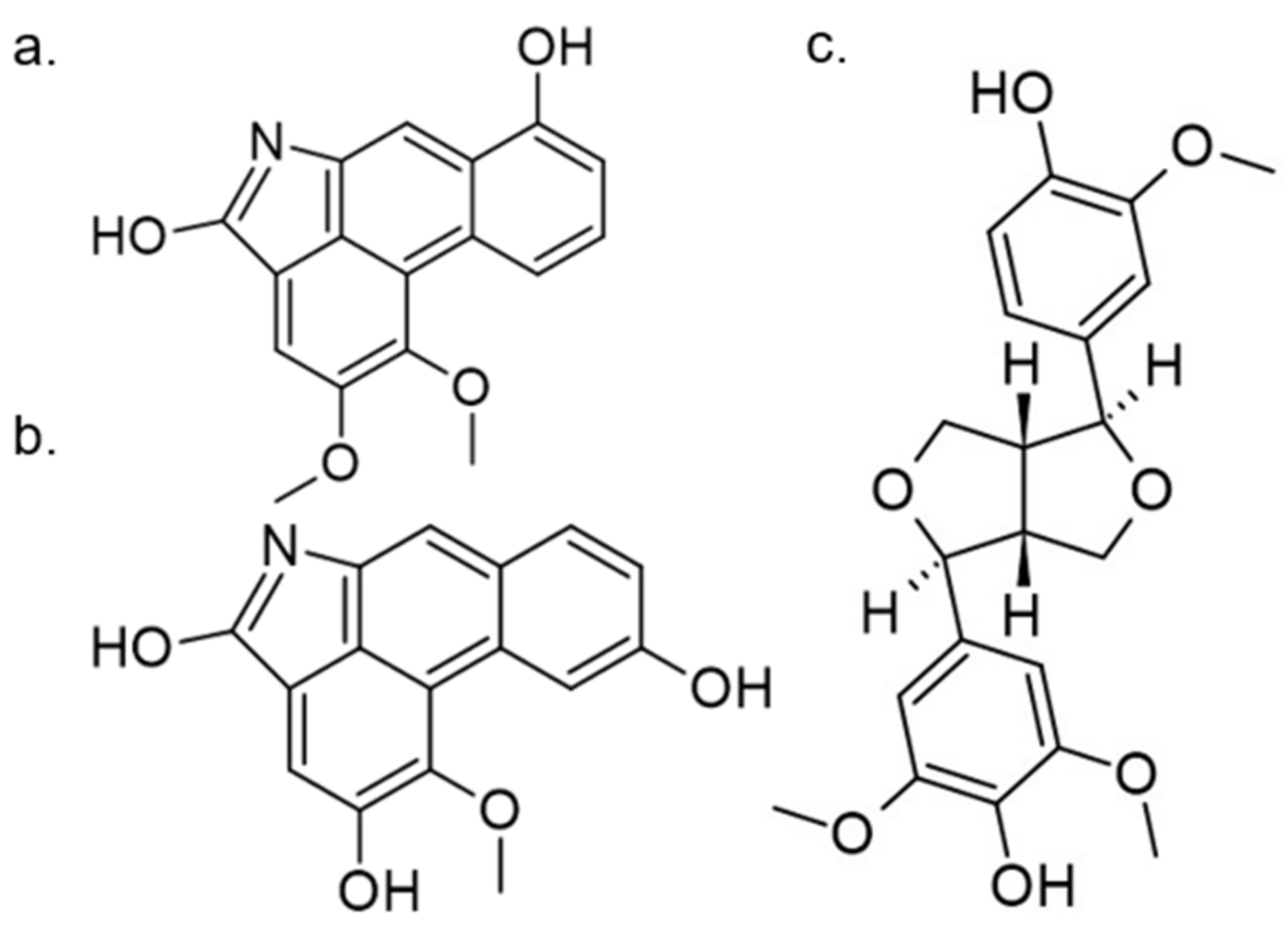

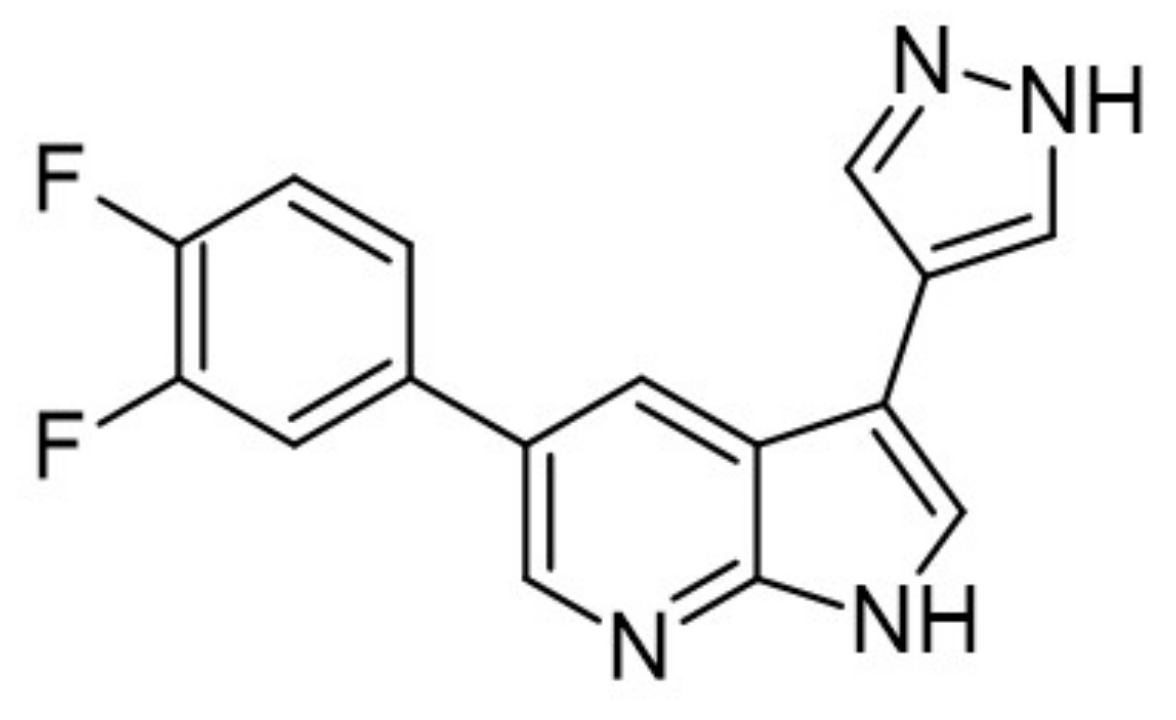
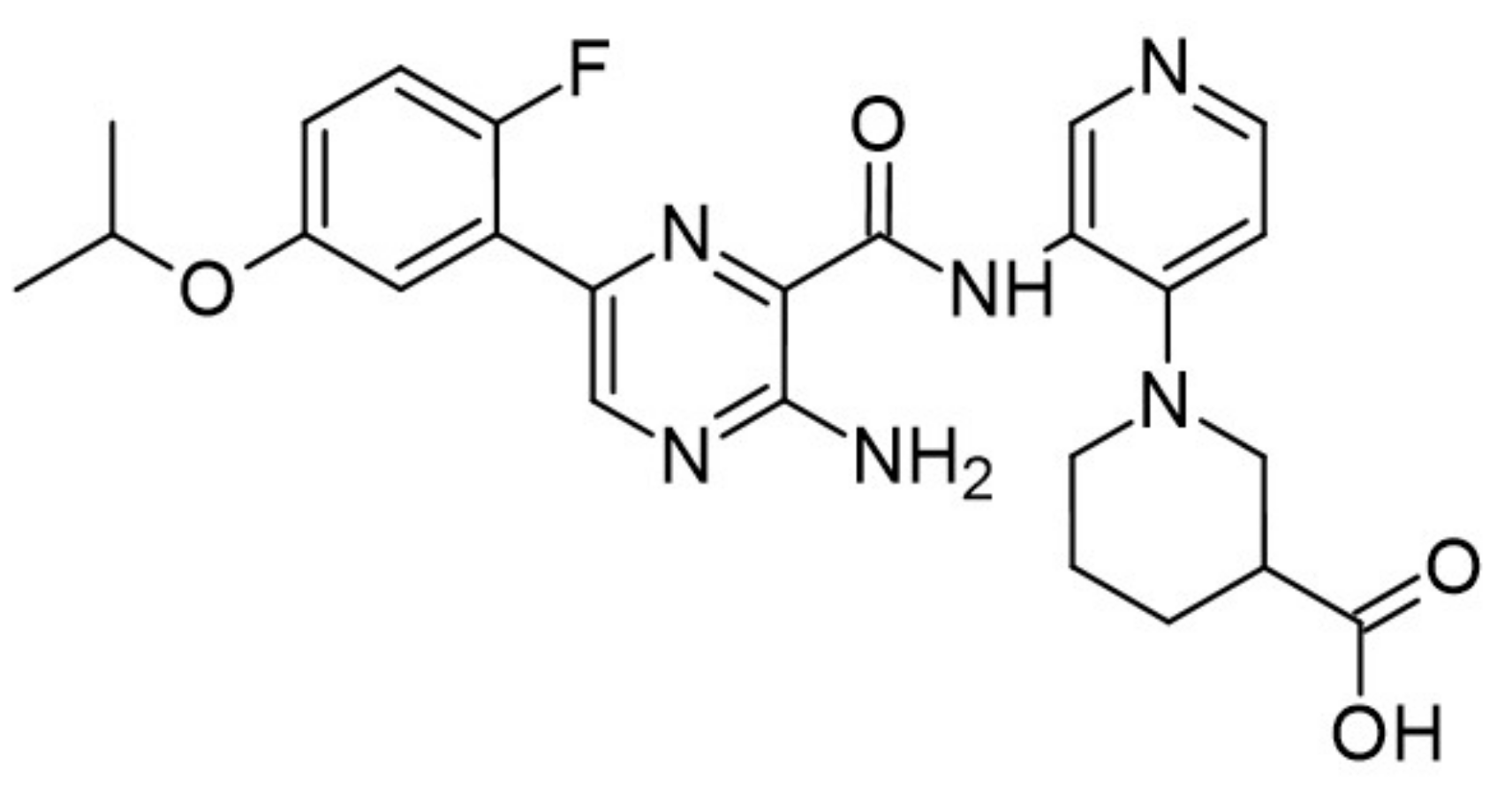
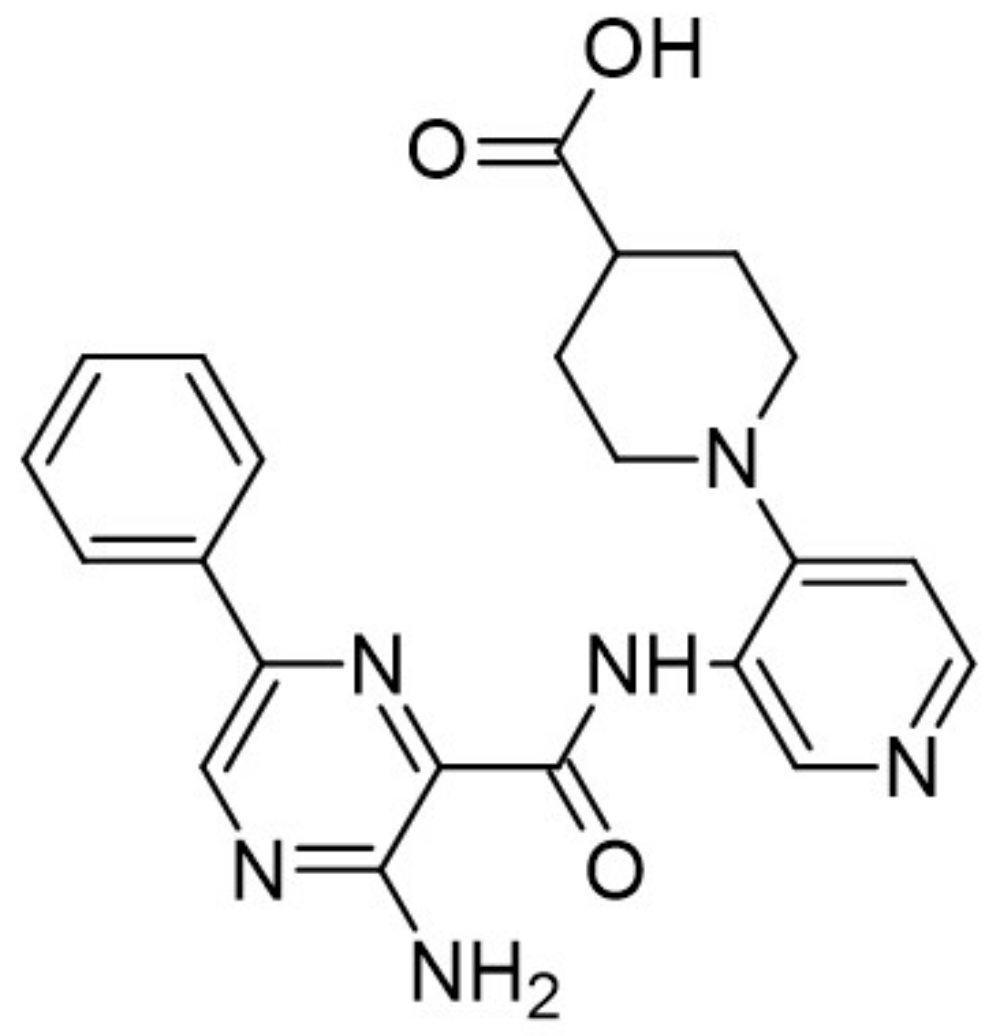




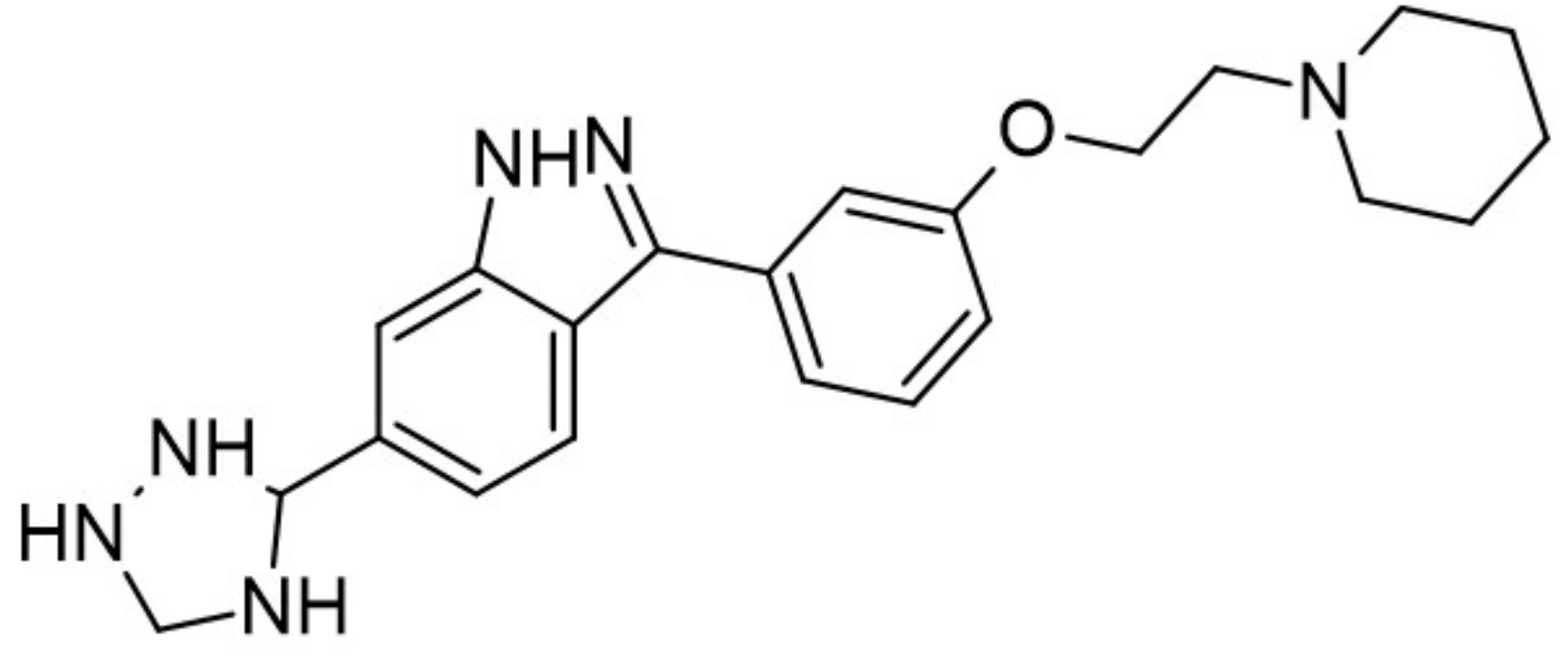
| Type | Etiology |
|---|---|
| type I diabetes | Immunologically-mediated Idiopathic |
| type II diabetes | Genetic predisposition, Insulin resistance (aging, physical inactivity, and overweight) Relative insulin deficiency and decreased β-cell function |
| gestational diabetes | β-cell dysfunction on a background of chronic insulin resistance during pregnancy |
| other types of diabetes | Genetic defects of β-cell function: • MODY 3, MODY 2, MODY 1 and others • transient and permanent neonatal diabetes • mitochondrial DNA and others Genetic defects in insulin action: • type A insulin resistance • leprechaunism, Rabson-Mendenhall syndrome • Lipoathropic diabetes and others The disease of the exocrine pancreas: • pancreatitis, trauma/pancreatectomy, neoplasia, cystic fibrosis, hemochromatosis, fibrocalculous pancreatopathy, and others Endocrinopathies: • acromegaly, Cushing’s syndrome, glucagonoma, pheochromocytoma, hyperthyroidism, somatostatinoma, aldosteronoma Drug or chemical-induced: • e.g., vacor, pentamidine, nicotinic acid, glucocorticoids, thyroid hormone, diazoxide, β-adrenergic agonists, thiazides, Dilantin, γ-IFN Infections: • congenital rubella • cytomegalovirus and others Uncommon forms of immune-mediated diabetes: • Stiff-man syndrome • anti-insulin receptor antibodies and others Other genetic syndromes sometimes associated with diabetes • Down syndrome, Klinefelter syndrome, Wolfram syndrome, Friedreich ataxia, Huntington chorea, Laurence-Mood-Biedl syndrome, myotonic dystrophy, porphyria, Prader-Willi syndrome and others |
| No. | Compound | Name | IC50 DYRK1A[nM] | Other Targets | In Vitro Results | Future Directions |
|---|---|---|---|---|---|---|
| 1. | 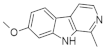 | harmine | 33 | MAO-A CK1 PIM3 | ↑↑↑ | increasing the selectivity of the compound |
| 2. | 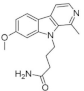 | 4-(7-methoxy-1-methyl-β-carbolin-9-yl)butanamide | 25 | MAO-A | ↑↑↑ | the research direction is definitely worth pursuing, large-scale clinical trials are needed |
| 3. | 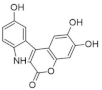 | 4-hydroxychromeno[3,4-b]indol-6(7H)-one | 0.074 | CDK5 GSK3 | not available | results will be use to the chromeno[3,4-b]indole as a pharmacophore |
| 4. | 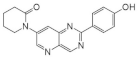 | 1-[2-(4-hydroxyphenyl)pyrido[3,2-d]pyrimidin-7-yl]piperidin-2-one | 60 | CDK5 GSK3 | not available | promising scaffolds for targetingprotein kinases involved in the central nervous system |
| 5. | 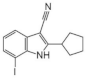 | 2-cyclopentyl-7-iodo-1H-indole-3-carbonitrile | 70 | CLK1 CLK2 CLK4 GSK3 | minimal cytotoxicity, more data not available | further modifications are underway, aiming at the development of potent, highly selective and water-soluble DYRK1A inhibitors |
| 6. | 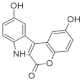 | 3-hydroxychromeno[3,4-b]indol-6(7H)-one) | 500 | CDK5 GSK3 | not available | results will be used to the chromeno[3,4-b]indole as a pharmacophore |
| 7. | 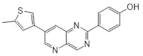 | 4-[7-(5-methyl-thiophen-3-yl)-pyrido[3,2-d]pyrimidin-2-yl]-phenol | 24 | CDK5 GSK3 | not available | promising scaffolds for targetingprotein kinases involved in the central nervous system |
| 8. | 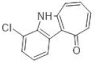 | 4-chlorocyclohepta[b]indol-10(5H)-one | 200 | CLK1 | not available | biological data are needed |
| 9. | 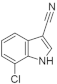 | 7-chloro-1H-indole-3-carbonitrile | 3300 | CLK1 CLK2 CLK3 CLK4 GSK3 | not available | use for the development of new DYRK1A inhibitors |
| 10. | 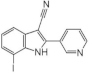 | 7-iodo-2-(pyridin-3-yl)-1H-indole-3-carbonitrile | 80 | CLK1 CLK2 CLK4 | not available | biological data are needed |
| 11. | 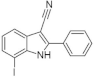 | 7-iodo-2-phenyl-1H-indole-3-carbonitrile | 10 | CLK1 CLK2 CLK3 CLK4 GSK3 | minimal cytotoxicity, more data not available | biological data are needed |
| 12. | 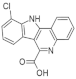 | 10-chloro-11H-indolo[3,2-c]quinoline-6-carboxylic acid | 23 | CLK1 CLK2 CLK3 CLK4 | not available | biological data are needed |
| 13. | 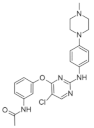 | AC2 | >16000 | not available | not available | new scaffolds offer novel opportunities to design DYRK1A inhibitors |
| 14. | 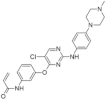 | AC8 | >8000 | not available | not available | new scaffolds offer novel opportunities to design DYRK1A inhibitors |
| 15. | 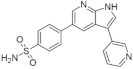 | AC12 | 216 | pan kinase | not available | new scaffolds offer novel opportunities to design DYRK1A inhibitors |
| 16. | 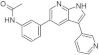 | AC13 | 350 | not available | not available | biological research in progress |
| 17. |  | AC14 | 221 | not available | not available | new scaffolds offer novel opportunities to design DYRK1A inhibitors |
| 18. | 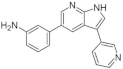 | AC15 | 329 | pan kinase | not available | new scaffolds offer novel opportunities to design DYRK1A inhibitors |
| 19. |  | AC16 | >6000 | not available | not available | new scaffolds offer novel opportunities to design DYRK1A inhibitors |
| 20. | 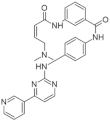 | AC18 | >4800 | not available | not available | new scaffolds offer novel opportunities to design DYRK1A inhibitors |
| 21. | 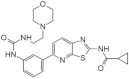 | AC20 | 3500 | CLK2 ABL PDGFR | not available | new scaffolds offer novel opportunities to design DYRK1A inhibitors |
| 22. | 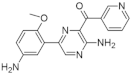 | AC22 | >6000 | GSK3β CLK2 HIPK1/2 CDK7 | not available | biological research in progress |
| 23. | 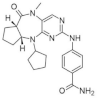 | AC23 | 4200 | DRAK1/2 ERK5 a | not available | biological research in progress |
| 24. |  | AC24 | 800 | CLK2 | not available | new scaffolds offer novel opportunities to design DYRK1A inhibitors |
| 25. | 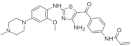 | AC25 | 1200 | CLK2 | not available | biological research in progress |
| 26. |  | AC27 | 532 | GSK3β PIK3CG PIK4CB | not available | biological research in progress |
| 27. | 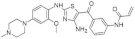 | AC28 | - | CLK2 | not available | biological research in progress |
| 28. | 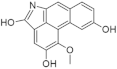 | Aristolactam AIIIA | 80000 | CDK1 CDK2 CDK4 | not available | possible use in viral infections, cancer and neurodegenerative pathologies (e.g., Alzheimer’s and Parkinson’s diseases) |
| 29. | 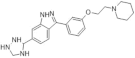 | CC-401 | - | c-Jun-N-terminal kinase PRKD2 PRKD3 CSNK1G3 MAPK9 | ↑↑ | key issues are the development of strategies to target regenerative compounds selectively to the β cell |
| 30. | 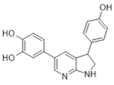 | 3,5-di-(4-hydroxyphenyl)-1H-pyrrolo[2,3-b]pyridine | 3 | Akt CDK2 CDK5 CK1 CLK1 ERK2 GSK3 JAK3 TRKA pim 1 kinase | Noncytotoxic, more data not available | studies on appropriate animal models |
| 31. | 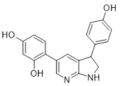 | 3-(4-hydroxyphenyl)-5-(2,4-dihydroxyphenyl)-1H-pyrrolo[2,3-b]- pyridine | 11.7 | Akt CDK2 CDK5 CK1 CLK1 ERK2 GSK3 JAK3 TRKA pim 1 kinase | Noncytotoxic, more data not available | studies on appropriate animal models |
| 32. |  | 3,5-di-(3,4-dihydroxyphenyl)-1H-pyrrolo[2,3-b]pyridine | 12.4 | Akt CDK2 CDK5 CK1 CLK1 ERK2 GSK3 JAK3 TRKA pim 1 kinase | Noncytotoxic, more data not available | studies on appropriate animal models |
| 33. | 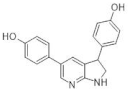 | 3-(4-hydroxyphenyl)-5-(3,4-dihydroxyphenyl)-1H-pyrrolo[2,3-b]- pyridine | 23.1 | Akt CDK2 CDK5 CK1 CLK1 ERK2 GSK3 JAK3 TRKA pim 1 kinase | Noncytotoxic, more data not available | studies on appropriate animal models |
| 34. | 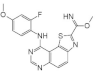 | EHT5372 | 0.22 | CLK1 CLK2 CLK4 GSK3 | not available | high-potential therapy for AD and other tauopathies |
| 35. | 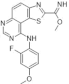 | EHT1610 | 0.36 | CLK1 CLK2 CLK4 GSK3 | not available | high-potential therapy for AD and other tauopathies |
| 36. | 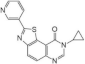 | FC162 | 11 | CLK1 GSK3 | FC162 modified Tau phosphorylation and could alter cell cycle progression of pre-B cells | development as a DYRK1A kinase inhibitor |
| 37. | 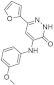 | furan-2-yl-substituted | >10000 | CDK5/p25 GSK3α/β PI3Kα | cytotoxic effects was determined against six human cancer cell lines | candidate for further evaluations |
| 38. | 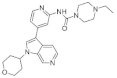 | GNF2133 | 6 | GSK3 | ↑↑ | requires strategies to mitigate the observedhypertrophic effects in nonpancreatic tissues |
| 39. | 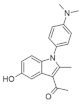 | GNF1346 | - | undetermined | ↑ | for now, untested |
| 40. | 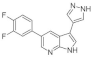 | GNF3809 | - | CDK CLK MAP4K4 GSK3 FLT HIPK JAK3 | ↑↑ | further optimization and elucidation of its molecular mechanism of action needed |
| 41. | 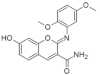 | GNF4088 | - | undetermined | ↑ | for now, untested |
| 42. |  | GNF4877 | 6 | GSK3 | ↑↑ | GNF4877 was notprogressed beyond preclinical research |
| 43. | 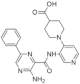 | GNF7156 | 100 | GSK3 | ↑↑ | may provide a path forward to develop new drugs to treat diabetes |
| 44. | 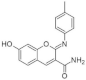 | GNF9228 | - | undetermined | ↑↑↑ | development of chemically modified versions of GNF9228 with enhanced bioavailability to allow in vivo testing |
| 45. | 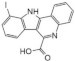 | Kufal194 | 6 | CLK-1 | data available for zebrafish | promising starting point for the development of therapeutics in DS |
| 46. | 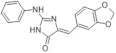 | L41 | 170 | CLKs mTOR/PI3K | not available | biological research in progress, promising compound forthe development of novel AD therapeutic agents |
| 47. | 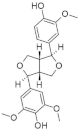 | medioresinol | 100 | CDK1 | not available | for now, untested |
| 48. |  | methyl 9-(4-bromo-2-fluorophenylamino)thiazolo[5,4-f]quinazoline-2-carbimidate | 50 | CK1 CDK5 GSK3 | not available | a promising source for the synthesis of novel bioactive molecules |
| 49. | 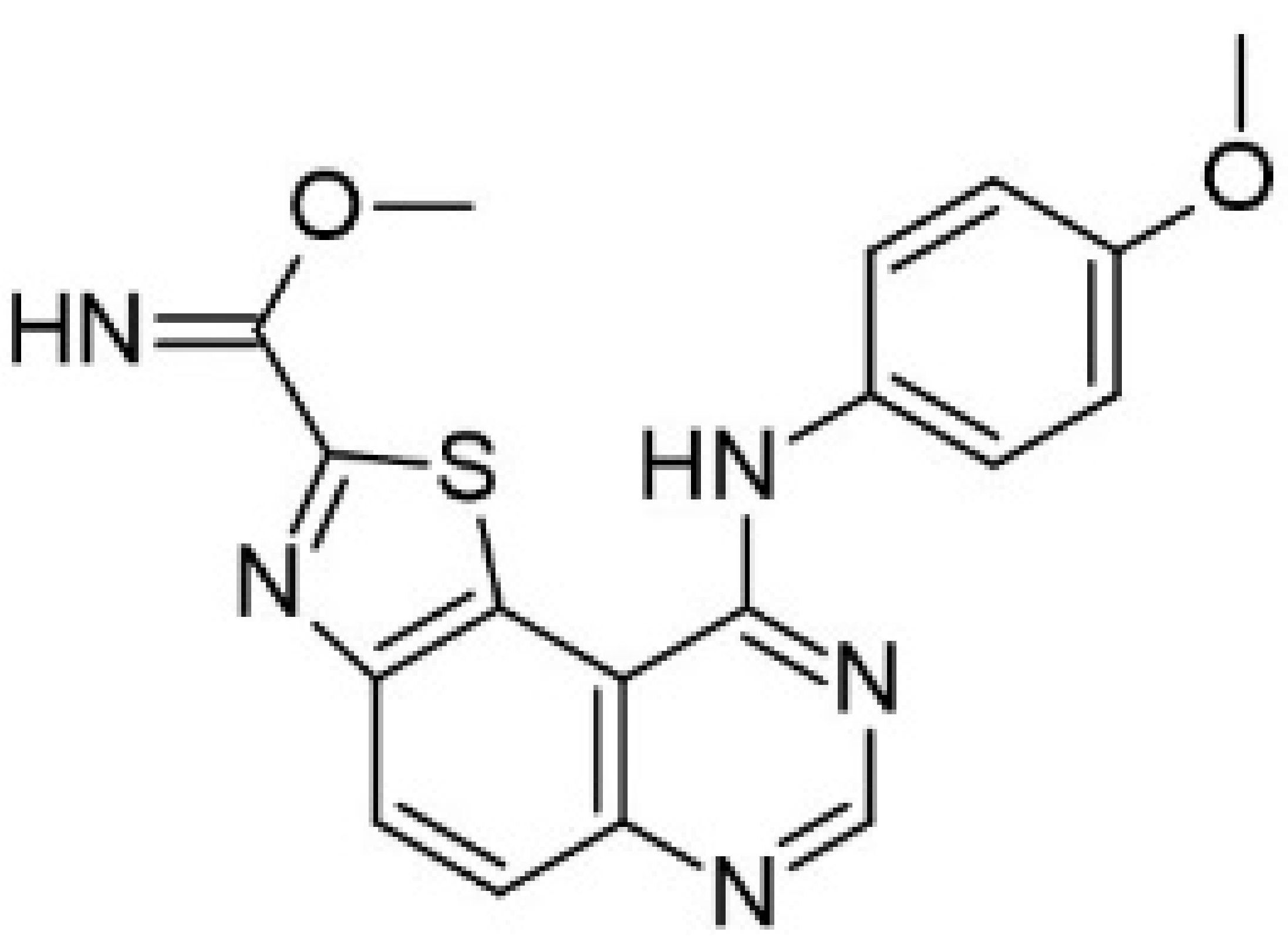 | methyl 9-(4-methoxyphenylamino)thiazolo[5,4-f]quinazoline-2-carbimidate | 40 | CK1 CDK5 GSK3 | not available | a promising source for the synthesis of novel bioactive molecules |
| 50. | 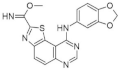 | methyl 9-(benzo[d][1,3]dioxol-5-ylamino)thiazolo[5,4-f]quinazoline-2-carbimidate | 47 | CK1 CDK5 GSK3 | not available | a promising source for the synthesis of novel bioactive molecules |
| 51. |  | N-(2,3-dihydrobenzo[b][1,4]dioxin-6-yl)-7-methoxybenzothieno[3,2-d]pyrimidin-4-amine | 0.5 | TRPV1 CLK1 | not available | starting point of a larger program |
| 52. | 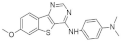 | N1-(7-methoxybenzothieno[3,2-d]pyrimidin-4-yl)-N4,N4-dimethylbenzene-1,3-diamine | 0.68 | CLK1 | not available | starting point of a larger program |
| 53. | 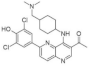 | OTS167 | - | undetermined | cytotoxic | a model compound for the design of less toxic compounds |
| 54. |  | velutinam | 600 | CDK1 | not available | for now, untested |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pucelik, B.; Barzowska, A.; Dąbrowski, J.M.; Czarna, A. Diabetic Kinome Inhibitors—A New Opportunity for β-Cells Restoration. Int. J. Mol. Sci. 2021, 22, 9083. https://doi.org/10.3390/ijms22169083
Pucelik B, Barzowska A, Dąbrowski JM, Czarna A. Diabetic Kinome Inhibitors—A New Opportunity for β-Cells Restoration. International Journal of Molecular Sciences. 2021; 22(16):9083. https://doi.org/10.3390/ijms22169083
Chicago/Turabian StylePucelik, Barbara, Agata Barzowska, Janusz M. Dąbrowski, and Anna Czarna. 2021. "Diabetic Kinome Inhibitors—A New Opportunity for β-Cells Restoration" International Journal of Molecular Sciences 22, no. 16: 9083. https://doi.org/10.3390/ijms22169083
APA StylePucelik, B., Barzowska, A., Dąbrowski, J. M., & Czarna, A. (2021). Diabetic Kinome Inhibitors—A New Opportunity for β-Cells Restoration. International Journal of Molecular Sciences, 22(16), 9083. https://doi.org/10.3390/ijms22169083







