Chronic and Intermittent Hyperglycemia Modulates Expression of Key Molecules of PI3K/AKT Pathway in Differentiating Human Visceral Adipocytes
Abstract
1. Introduction
2. Results
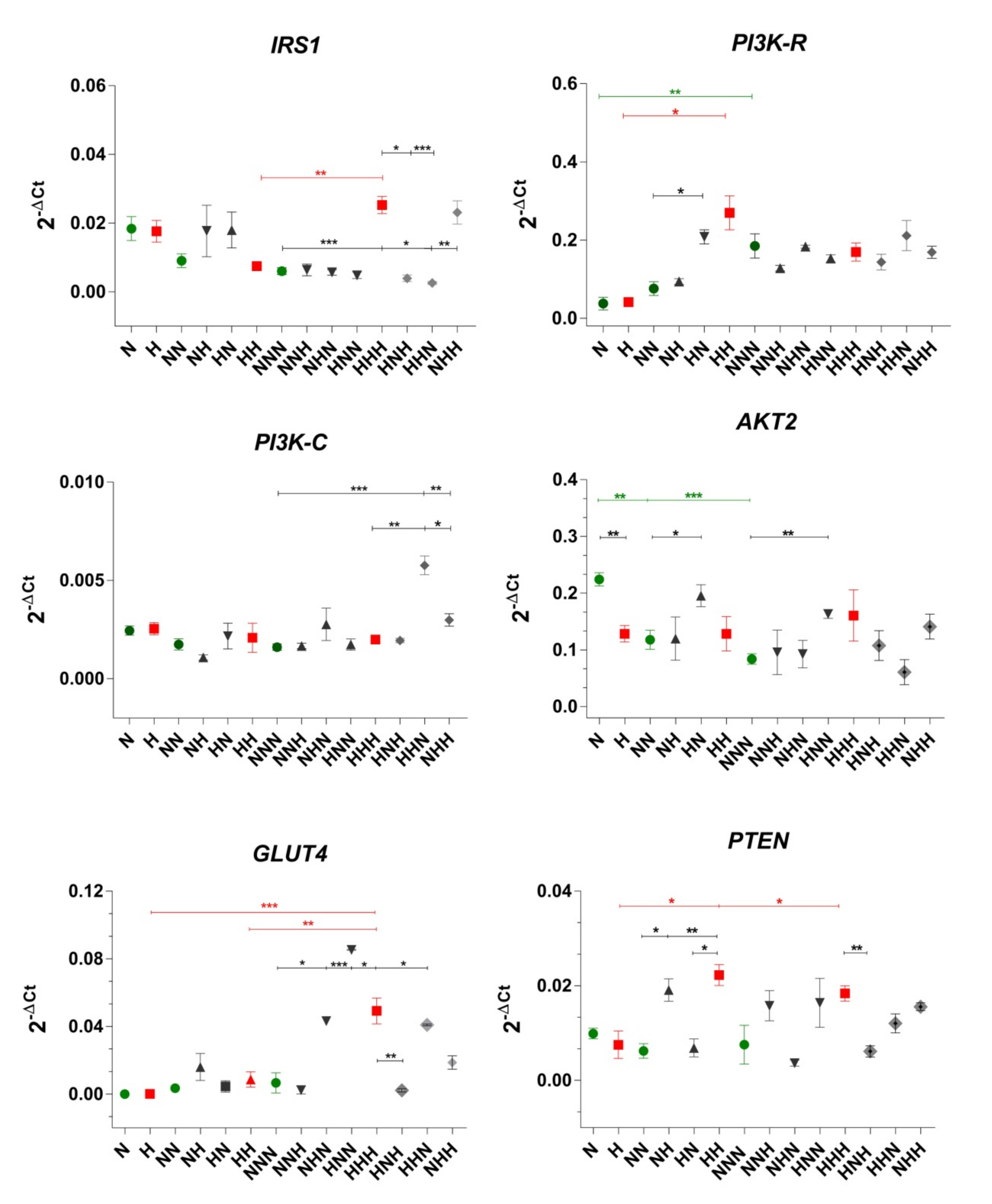
2.1. Evaluation of the Gene Expression Profile of Insulin Signaling Pathway Molecules under CHG and IHG
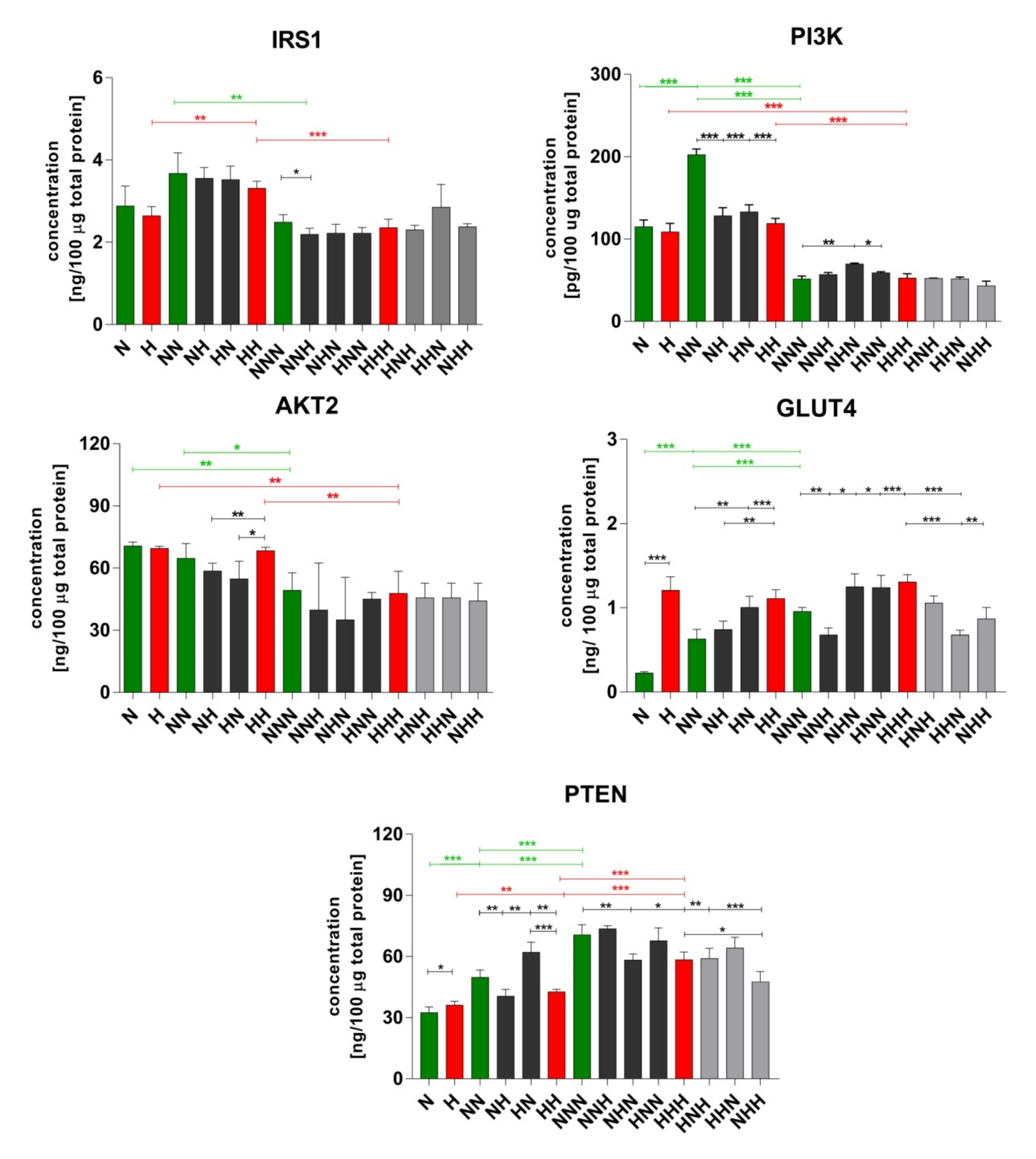
2.2. Evaluation of Insulin Signaling Proteins Expression during ADG in CHG and IHG
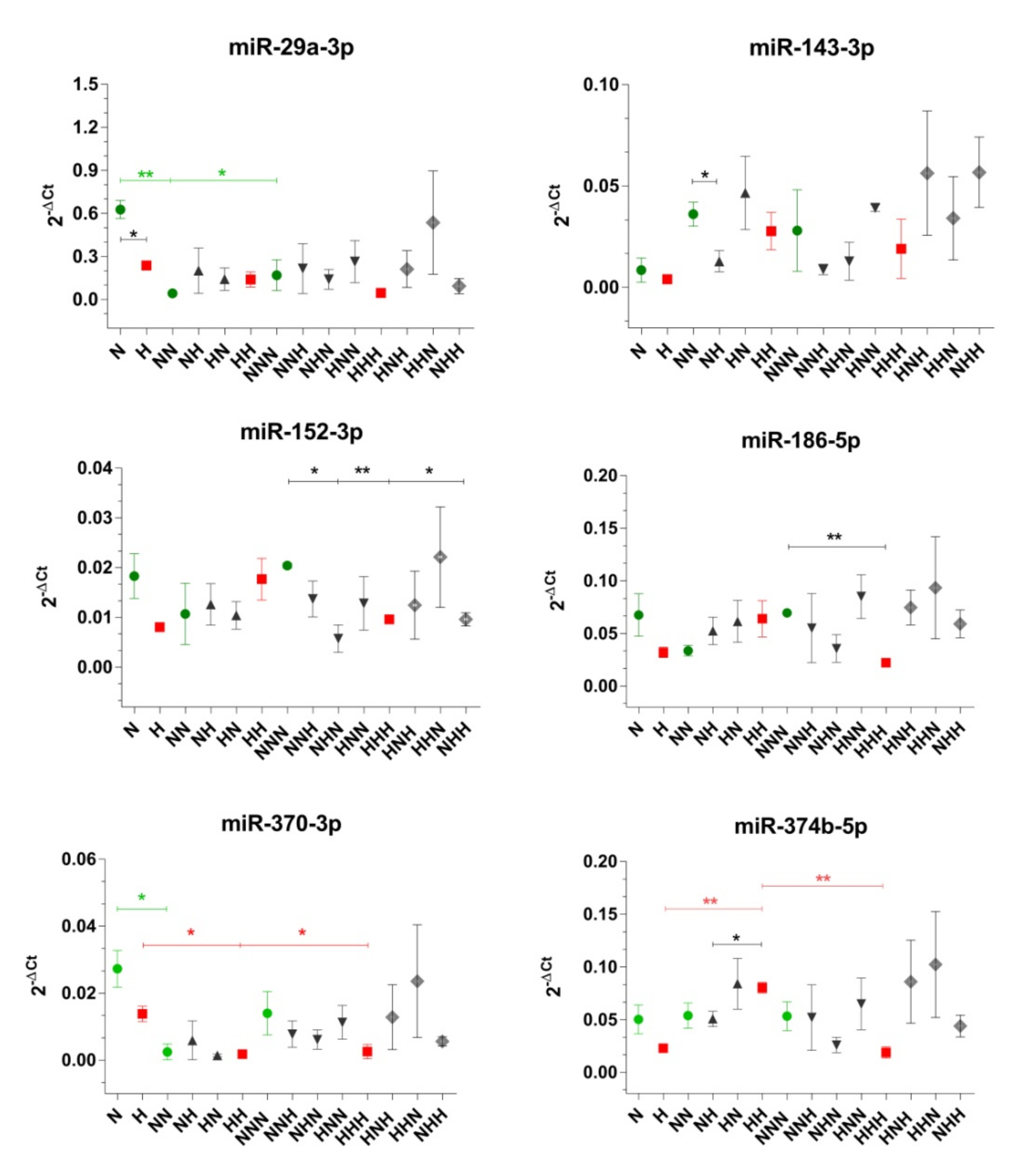
2.3. Expression of Selected miRs That Target Insulin Signaling Pathway Molecules during ADG in CHG and IHG
3. Discussion
3.1. ADG in CHG
3.2. ADG in IHG
3.3. Study Limitations
3.4. Overall Conclusions and Clinical Output
4. Materials and Methods
4.1. Cell Culture
4.2. RNA Isolation and mRNA Expression Profiling
4.3. Protein Isolation and Expression Profiling
4.4. miRs Isolation and Expression Profiling
4.5. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ADG | adipogenesis |
| Ads | adipocytes |
| AKT | protein kinase B |
| AT | adipose tissue |
| CHG | chronic hyperglycemia |
| C/EBPs | CCAAT enhancer-binding proteins |
| FOXO1 | forkhead box O1 |
| GLUT4 | glucose transporter 4 |
| HG | hyperglycemia |
| IHG | intermittent hyperglycemia |
| mTOR2 | mammalian target of rapamycin 2 |
| NG | normoglycemia |
| pAds | preadipocytes |
| PDK1 | phosphoinositide-dependent kinase 1 |
| PIP2 | phophatidylinositol-4,5-biphospate |
| PIP3 | phophatidylinositol-3,4,5-triphospate |
| PI3K | phosphatidylinositol-4,5-biphosphate 3-kinase |
| PI3K-C | phosphatidylinositol-4,5-biphosphate 3-kinase catalytic subunit |
| PI3K-R | phosphatidylinositol-4,5-biphosphate 3-kinase regulatory subunit |
| PPARG | peroxisome proliferator-activated receptor gamma |
| PTEN | phosphatase and tensin homolog |
| T2DM | type 2 diabetes mellitus |
| VAT | visceral adipose tissue |
References
- Gastaldelli, A.; Gaggini, M.; DeFronzo, R.A. Role of Adipose Tissue Insulin Resistance in the Natural History of Type 2 Diabetes: Results from the San Antonio Metabolism Study. Diabetes 2017, 66, 815–822. [Google Scholar] [CrossRef] [PubMed]
- Grundy, S.M. Adipose tissue and metabolic syndrome: Too much, too little or neither. Eur. J. Clin. Investig. 2015, 45, 1209–1217. [Google Scholar] [CrossRef] [PubMed]
- Wróblewski, A.; Strycharz, J.; Świderska, E.; Balcerczyk, A.; Szemraj, J.; Drzewoski, J.; Śliwińska, A. Chronic and Transient Hyperglycemia Induces Changes in the Expression Patterns of IL6 and ADIPOQ Genes and Their Associated Epigenetic Modifications in Differentiating Human Visceral Adipocytes. Int. J. Mol. Sci. 2021, 22, 6964. [Google Scholar] [CrossRef]
- Deng, T.; Lyon, C.J.; Bergin, S.; Caligiuri, M.A.; Hsueh, W.A. Obesity, Inflammation, and Cancer. Annu. Rev. Pathol. 2016, 11, 421–449. [Google Scholar] [CrossRef]
- Swiderska, E.; Podolska, M.; Strycharz, J.; Szwed, M.; Abramczyk, H.; Brozek-Pluska, B.; Wroblewski, A.; Szemraj, J.; Majsterek, I.; Drzewoski, J.; et al. Hyperglycemia Changes Expression of Key Adipogenesis Markers (C/EBPα and PPARγ) and Morphology of Differentiating Human Visceral Adipocytes. Nutrients 2019, 11, 1835. [Google Scholar] [CrossRef]
- Ishaq, A.; Dufour, D.; Cameron, K.; von Zglinicki, T.; Saretzki, G. Metabolic memory of dietary restriction ameliorates DNA damage and adipocyte size in mouse visceral adipose tissue. Exp. Gerontol. 2018, 113, 228–236. [Google Scholar] [CrossRef]
- Strycharz, J.; Swiderska, E.; Wroblewski, A.; Podolska, M.; Czarny, P.; Szemraj, J.; Balcerczyk, A.; Drzewoski, J.; Kasznicki, J.; Sliwinska, A. Hyperglycemia Affects miRNAs Expression Pattern during Adipogenesis of Human Visceral Adipocytes-Is Memorization Involved? Nutrients 2018, 10, 1774. [Google Scholar] [CrossRef] [PubMed]
- Vanhaesebroeck, B.; Stephens, L.; Hawkins, P. PI3K signalling: The path to discovery and understanding. Nat. Rev. Mol. Cell Biol. 2012, 13, 195–203. [Google Scholar] [CrossRef]
- Świderska, E.; Strycharz, J.; Wróblewski, A.; Szemraj, J.; Drzewoski, J.; Śliwińska, A. Role of PI3K/AKT pathway in insulin-mediated glucose uptake. In Blood Glucose Levels; IntechOpen: London, UK, 2018. [Google Scholar]
- Li, A.; Qiu, M.; Zhou, H.; Wang, T.; Guo, W. PTEN, Insulin Resistance and Cancer. Curr. Pharm. Des. 2017, 23, 3667–3676. [Google Scholar] [CrossRef]
- Gao, Y.; Moten, A.; Lin, H.-K. Akt: A new activation mechanism. Cell Res. 2014, 24, 785–786. [Google Scholar] [CrossRef] [PubMed]
- Fischer-Posovszky, P.; Tews, D.; Horenburg, S.; Debatin, K.M.; Wabitsch, M. Differential function of Akt1 and Akt2 in human adipocytes. Mol. Cell Endocrinol. 2012, 358, 135–143. [Google Scholar] [CrossRef] [PubMed]
- Baudry, A.; Yang, Z.Z.; Hemmings, B.A. PKBalpha is required for adipose differentiation of mouse embryonic fibroblasts. J. Cell Sci. 2006, 119, 889–897. [Google Scholar] [CrossRef]
- Cai, H.; Dong, L.Q.; Liu, F. Recent Advances in Adipose mTOR Signaling and Function: Therapeutic Prospects. Trends Pharmacol. Sci. 2016, 37, 303–317. [Google Scholar] [CrossRef]
- Nakae, J.; Kitamura, T.; Kitamura, Y.; Biggs, W.H.; Arden, K.C.; Accili, D. The Forkhead Transcription Factor Foxo1 Regulates Adipocyte Differentiation. Dev. Cell 2003, 4, 119–129. [Google Scholar] [CrossRef]
- Andersen, E.; Ingerslev, L.R.; Fabre, O.; Donkin, I.; Altintas, A.; Versteyhe, S.; Bisgaard, T.; Kristiansen, V.B.; Simar, D.; Barres, R. Preadipocytes from obese humans with type 2 diabetes are epigenetically reprogrammed at genes controlling adipose tissue function. Int. J. Obes. 2019, 43, 306–318. [Google Scholar] [CrossRef] [PubMed]
- Ramzan, F.; D’Souza, R.F.; Durainayagam, B.R.; Milan, A.M.; Markworth, J.F.; Miranda-Soberanis, V.; Sequeira, I.R.; Roy, N.C.; Poppitt, S.D.; Mitchell, C.J.; et al. Circulatory miRNA biomarkers of metabolic syndrome. Acta Diabetol. 2020, 57, 203–214. [Google Scholar] [CrossRef]
- Herrera, B.M.; Lockstone, H.E.; Taylor, J.M.; Ria, M.; Barrett, A.; Collins, S.; Kaisaki, P.; Argoud, K.; Fernandez, C.; Travers, M.E.; et al. Global microRNA expression profiles in insulin target tissues in a spontaneous rat model of type 2 diabetes. Diabetologia 2010, 53, 1099–1109. [Google Scholar] [CrossRef]
- Landrier, J.F.; Derghal, A.; Mounien, L. MicroRNAs in Obesity and Related Metabolic Disorders. Cells 2019, 8, 859. [Google Scholar] [CrossRef]
- Yang, W.M.; Jeong, H.J.; Park, S.Y.; Lee, W. Induction of miR-29a by saturated fatty acids impairs insulin signaling and glucose uptake through translational repression of IRS-1 in myocytes. FEBS Lett. 2014, 588, 2170–2176. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Sun, Y.C.; Cheng, P.; Shao, H.G. Adipose tissue macrophage-derived exosomal miR-29a regulates obesity-associated insulin resistance. Biochem. Biophys. Res. Commun. 2019, 515, 352–358. [Google Scholar] [CrossRef] [PubMed]
- Esteves, J.V.; Enguita, F.J.; Machado, U.F. MicroRNAs-Mediated Regulation of Skeletal Muscle GLUT4 Expression and Translocation in Insulin Resistance. J. Diabetes Res. 2017, 2017, 7267910. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Song, J.; Cui, J.; Hou, J.; Zheng, X.; Li, C.; Liu, L. microRNAs regulate adipocyte differentiation. Cell Biol. Int. 2013, 37, 533–546. [Google Scholar] [CrossRef]
- He, A.; Zhu, L.; Gupta, N.; Chang, Y.; Fang, F. Overexpression of micro ribonucleic acid 29, highly up-regulated in diabetic rats, leads to insulin resistance in 3T3-L1 adipocytes. Mol. Endocrinol. 2007, 21, 2785–2794. [Google Scholar] [CrossRef]
- Boudreau, R.L.; Jiang, P.; Gilmore, B.L.; Spengler, R.M.; Tirabassi, R.; Nelson, J.A.; Ross, C.A.; Xing, Y.; Davidson, B.L. Transcriptome-wide discovery of microRNA binding sites in human brain. Neuron 2014, 81, 294–305. [Google Scholar] [CrossRef] [PubMed]
- Grosswendt, S.; Filipchyk, A.; Manzano, M.; Klironomos, F.; Schilling, M.; Herzog, M.; Gottwein, E.; Rajewsky, N. Unambiguous identification of miRNA:target site interactions by different types of ligation reactions. Mol. Cell 2014, 54, 1042–1054. [Google Scholar] [CrossRef]
- Kameswaran, V.; Bramswig, N.C.; McKenna, L.B.; Penn, M.; Schug, J.; Hand, N.J.; Chen, Y.; Choi, I.; Vourekas, A.; Won, K.J.; et al. Epigenetic regulation of the DLK1-MEG3 microRNA cluster in human type 2 diabetic islets. Cell Metab. 2014, 19, 135–145. [Google Scholar] [CrossRef] [PubMed]
- Kong, G.; Zhang, J.; Zhang, S.; Shan, C.; Ye, L.; Zhang, X. Upregulated microRNA-29a by hepatitis B virus X protein enhances hepatoma cell migration by targeting PTEN in cell culture model. PLoS ONE 2011, 6, e19518. [Google Scholar] [CrossRef]
- Wang, S.; Wang, L.; Dou, L.; Guo, J.; Fang, W.; Li, M.; Meng, X.; Man, Y.; Shen, T.; Huang, X.; et al. MicroRNA 152 regulates hepatic glycogenesis by targeting PTEN. FEBS J. 2016, 283, 1935–1946. [Google Scholar] [CrossRef]
- Xu, Q.; Jiang, Y.; Yin, Y.; Li, Q.; He, J.; Jing, Y.; Qi, Y.-T.; Xu, Q.; Li, W.; Lu, B.; et al. A regulatory circuit of miR-148a/152 and DNMT1 in modulating cell transformation and tumor angiogenesis through IGF-IR and IRS1. J. Mol. Cell Biol. 2013, 5, 3–13. [Google Scholar] [CrossRef]
- Xiang, Y.; Chen, Y.-J.; Yan, Y.-B.; Liu, Y.; Qiu, J.; Tan, R.-Q.; Tian, Q.; Guan, L.; Niu, S.-S.; Xin, H.-W. MiR-186 bidirectionally regulates cisplatin sensitivity of ovarian cancer cells via suppressing targets PIK3R3 and PTEN and upregulating APAF1 expression. J. Cancer 2020, 11, 3446–3453. [Google Scholar] [CrossRef]
- Feng, H.; Zhang, Z.; Qing, X.; French, S.W.; Liu, D. miR-186-5p promotes cell growth, migration and invasion of lung adenocarcinoma by targeting PTEN. Exp. Mol. Pathol. 2019, 108, 105–113. [Google Scholar] [CrossRef]
- Hou, W.Z.; Chen, X.L.; Wu, W.; Hang, C.H. MicroRNA-370-3p inhibits human vascular smooth muscle cell proliferation via targeting KDR/AKT signaling pathway in cerebral aneurysm. Eur. Rev. Med. Pharmacol. Sci. 2017, 21, 1080–1087. [Google Scholar]
- Xue, Y.; Ouyang, K.; Huang, J.; Zhou, Y.; Ouyang, H.; Li, H.; Wang, G.; Wu, Q.; Wei, C.; Bi, Y.; et al. Direct conversion of fibroblasts to neurons by reprogramming PTB-regulated microRNA circuits. Cell 2013, 152, 82–96. [Google Scholar] [CrossRef]
- Skalsky, R.L.; Corcoran, D.L.; Gottwein, E.; Frank, C.L.; Kang, D.; Hafner, M.; Nusbaum, J.D.; Feederle, R.; Delecluse, H.J.; Luftig, M.A.; et al. The viral and cellular microRNA targetome in lymphoblastoid cell lines. PLoS Pathog. 2012, 8, e1002484. [Google Scholar] [CrossRef]
- Kishore, S.; Jaskiewicz, L.; Burger, L.; Hausser, J.; Khorshid, M.; Zavolan, M. A quantitative analysis of CLIP methods for identifying binding sites of RNA-binding proteins. Nat. Methods 2011, 8, 559–564. [Google Scholar] [CrossRef]
- Damania, B.; Haecker, I.; Gay, L.A.; Yang, Y.; Hu, J.; Morse, A.M.; McIntyre, L.M.; Renne, R. Ago HITS-CLIP Expands Understanding of Kaposi’s Sarcoma-associated Herpesvirus miRNA Function in Primary Effusion Lymphomas. PLoS Pathog. 2012, 8, e1002884. [Google Scholar] [CrossRef]
- Leonardini, A.; Laviola, L.; Perrini, S.; Natalicchio, A.; Giorgino, F. Cross-Talk between PPARgamma and Insulin Signaling and Modulation of Insulin Sensitivity. PPAR Res. 2009, 2009, 818945. [Google Scholar] [CrossRef] [PubMed]
- Guo, S. Insulin signaling, resistance, and the metabolic syndrome: Insights from mouse models into disease mechanisms. J. Endocrinol. 2014, 220, T1–T23. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Liu, G.; Guo, J.; Su, Z. The PI3K/AKT pathway in obesity and type 2 diabetes. Int. J. Biol. Sci. 2018, 14, 1483–1496. [Google Scholar] [CrossRef]
- Varma, S.; Lal, B.K.; Zheng, R.; Breslin, J.W.; Saito, S.; Pappas, P.J.; Hobson, R.W., 2nd; Duran, W.N. Hyperglycemia alters PI3k and Akt signaling and leads to endothelial cell proliferative dysfunction. Am. J. Physiol. Heart Circ. Physiol. 2005, 289, H1744–H1751. [Google Scholar] [CrossRef] [PubMed]
- Hernandez, R.; Teruel, T.; Lorenzo, M. Akt mediates insulin induction of glucose uptake and up-regulation of GLUT4 gene expression in brown adipocytes. FEBS Lett. 2001, 494, 225–231. [Google Scholar] [CrossRef]
- Tremblay, F.; Lavigne, C.; Jacques, H.; Marette, A. Defective Insulin-Induced GLUT4 Translocation in Skeletal Muscle of High Fat–Fed Rats Is Associated with Alterations in both Akt/Protein Kinase B and Atypical Protein Kinase C (ζ/λ) Activities. Diabetes 2001, 50, 1901–1910. [Google Scholar] [CrossRef] [PubMed]
- Xi, G.; Shen, X.; Wai, C.; White, M.F.; Clemmons, D.R. Hyperglycemia induces vascular smooth muscle cell dedifferentiation by suppressing insulin receptor substrate-1-mediated p53/KLF4 complex stabilization. J. Biol. Chem. 2019, 294, 2407–2421. [Google Scholar] [CrossRef]
- Wang, Y.; He, G.; Wang, F.; Zhang, C.; Ge, Z.; Zheng, X.; Deng, H.; Yuan, C.; Zhou, B.; Tao, X.; et al. Aspirin inhibits adipogenesis of tendon stem cells and lipids accumulation in rat injury tendon through regulating PTEN/PI3K/AKT signalling. J. Cell Mol. Med. 2019, 23, 7535–7544. [Google Scholar] [CrossRef] [PubMed]
- Meugnier, E.; Faraj, M.; Rome, S.; Beauregard, G.; Michaut, A.; Pelloux, V.; Chiasson, J.L.; Laville, M.; Clement, K.; Vidal, H.; et al. Acute hyperglycemia induces a global downregulation of gene expression in adipose tissue and skeletal muscle of healthy subjects. Diabetes 2007, 56, 992–999. [Google Scholar] [CrossRef] [PubMed]
- Xi, G.; Wai, C.; White, M.F.; Clemmons, D.R. Down-regulation of Insulin Receptor Substrate 1 during Hyperglycemia Induces Vascular Smooth Muscle Cell Dedifferentiation. J. Biol. Chem. 2017, 292, 2009–2020. [Google Scholar] [CrossRef]
- Nelson, B.A.; Robinson, K.A.; Buse, M.G. High glucose and glucosamine induce insulin resistance via different mechanisms in 3T3-L1 adipocytes. Diabetes 2000, 49, 981–991. [Google Scholar] [CrossRef]
- Buren, J.; Liu, H.; Lauritz, J.; Eriksson, J. High glucose and insulin in combination cause insulin receptor substrate-1 and -2 depletion and protein kinase B desensitisation in primary cultured rat adipocytes: Possible implications for insulin resistance in type 2 diabetes. Eur. J. Endocrinol. 2003, 148, 157. [Google Scholar] [CrossRef]
- Back, K.; Islam, R.; Johansson, G.S.; Chisalita, S.I.; Arnqvist, H.J. Insulin and IGF1 receptors in human cardiac microvascular endothelial cells: Metabolic, mitogenic and anti-inflammatory effects. J. Endocrinol. 2012, 215, 89–96. [Google Scholar] [CrossRef][Green Version]
- Laybutt, D.R.; Chisholm, D.J.; Kraegen, E.W. Specific adaptations in muscle and adipose tissue in response to chronic systemic glucose oversupply in rats. Am. J. Physiol. Endocrinol. Metab. 1997, 273, E1–E9. [Google Scholar] [CrossRef]
- Chouchani, E.T.; Kajimura, S. Metabolic adaptation and maladaptation in adipose tissue. Nat. Metab. 2019, 1, 189–200. [Google Scholar] [CrossRef]
- Chuang, C.C.; Yang, R.S.; Tsai, K.S.; Ho, F.M.; Liu, S.H. Hyperglycemia enhances adipogenic induction of lipid accumulation: Involvement of extracellular signal-regulated protein kinase 1/2, phosphoinositide 3-kinase/Akt, and peroxisome proliferator-activated receptor gamma signaling. Endocrinology 2007, 148, 4267–4275. [Google Scholar] [CrossRef] [PubMed]
- Masi, L.N.; Lotufo, P.A.; Ferreira, F.M.; Rodrigues, A.C.; Serdan, T.D.A.; Souza-Siqueira, T.; Braga, A.A.; Saldarriaga, M.E.G.; Alba-Loureiro, T.C.; Borges, F.T.; et al. Profiling plasma-extracellular vesicle proteins and microRNAs in diabetes onset in middle-aged male participants in the ELSA-Brasil study. Physiol. Rep. 2021, 9, e14731. [Google Scholar] [CrossRef]
- Nusca, A.; Tuccinardi, D.; Albano, M.; Cavallaro, C.; Ricottini, E.; Manfrini, S.; Pozzilli, P.; Di Sciascio, G. Glycemic variability in the development of cardiovascular complications in diabetes. Diabetes Metab. Res. Rev. 2018, 34, e3047. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.-Y.; Miao, L.-F.; Qian, L.-L.; Wang, N.; Qi, M.-M.; Zhang, Y.-M.; Dang, S.-P.; Wu, Y.; Wang, R.-X. Molecular Mechanisms of Glucose Fluctuations on Diabetic Complications. Front. Endocrinol. 2019, 10. [Google Scholar] [CrossRef] [PubMed]
- Shi, X.L.; Ren, Y.Z.; Wu, J. Intermittent high glucose enhances apoptosis in INS-1 cells. Exp. Diabetes Res. 2011, 2011, 754673. [Google Scholar] [CrossRef][Green Version]
- Ceriello, A.; Kilpatrick, E.S. Glycemic variability: Both sides of the story. Diabetes Care 2013, 36 (Suppl. 2), S272–S275. [Google Scholar] [CrossRef]
- Testa, R.; Bonfigli, A.R.; Prattichizzo, F.; La Sala, L.; De Nigris, V.; Ceriello, A. The “Metabolic Memory” Theory and the Early Treatment of Hyperglycemia in Prevention of Diabetic Complications. Nutrients 2017, 9, 437. [Google Scholar] [CrossRef]
- Strycharz, J.; Wroblewski, A.; Zieleniak, A.; Swiderska, E.; Matyjas, T.; Rucinska, M.; Pomorski, L.; Czarny, P.; Szemraj, J.; Drzewoski, J.; et al. Visceral Adipose Tissue of Prediabetic and Diabetic Females Shares a Set of Similarly Upregulated microRNAs Functionally Annotated to Inflammation, Oxidative Stress and Insulin Signaling. Antioxidants 2021, 10, 101. [Google Scholar] [CrossRef]
- Ying, C.; Liu, T.; Ling, H.; Cheng, M.; Zhou, X.; Wang, S.; Mao, Y.; Chen, L.; Zhang, R.; Li, W. Glucose variability aggravates cardiac fibrosis by altering AKT signalling path. Diabetes Vasc. Dis. Res. 2017, 14, 327–335. [Google Scholar] [CrossRef]
- Manning, B.D.; Cantley, L.C. AKT/PKB signaling: Navigating downstream. Cell 2007, 129, 1261–1274. [Google Scholar] [CrossRef]
- Aamir, K.; Khan, H.U.; Sethi, G.; Hossain, M.A.; Arya, A. Wnt signaling mediates TLR pathway and promote unrestrained adipogenesis and metaflammation: Therapeutic targets for obesity and type 2 diabetes. Pharmacol. Res. 2020, 152, 104602. [Google Scholar] [CrossRef] [PubMed]
- Wu, K.K.; Cheung, S.W.; Cheng, K.K. NLRP3 Inflammasome Activation in Adipose Tissues and Its Implications on Metabolic Diseases. Int. J. Mol. Sci. 2020, 21, 4148. [Google Scholar] [CrossRef]
- Vecchie, A.; Dallegri, F.; Carbone, F.; Bonaventura, A.; Liberale, L.; Portincasa, P.; Fruhbeck, G.; Montecucco, F. Obesity phenotypes and their paradoxical association with cardiovascular diseases. Eur. J. Intern. Med. 2018, 48, 6–17. [Google Scholar] [CrossRef] [PubMed]
- Xie, F.; Xiao, P.; Chen, D.; Xu, L.; Zhang, B. miRDeepFinder: A miRNA analysis tool for deep sequencing of plant small RNAs. Plant Mol. Biol. 2012, 80, 75–84. [Google Scholar] [CrossRef] [PubMed]
| Mir | Target Genes | References |
|---|---|---|
| 29a-3p | IRS, AKT, PI3KR, PTEN, GLUT4 | [18,20,21,22,23,24,25,26,27,28] |
| 143-3p | AKT, GLUT4 | [18] |
| 152-3p | IRS, PTEN | [29,30] |
| 186-5p | PI3KR, PTEN | [31,32] |
| 370-3p | IRS | [33] |
| 374b-5p | IRS, PI3KC, PTEN | [34,35,36,37] |
| Proliferation | Differentiation | Maturation |
|---|---|---|
| N | - | - |
| H | - | - |
| N | N | - |
| N | H | - |
| H | N | - |
| H | H | - |
| N | N | N |
| N | N | H |
| N | H | H |
| N | H | N |
| H | H | H |
| H | H | N |
| H | N | H |
| H | N | N |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Świderska, E.; Strycharz, J.; Wróblewski, A.; Czarny, P.; Szemraj, J.; Drzewoski, J.; Śliwińska, A. Chronic and Intermittent Hyperglycemia Modulates Expression of Key Molecules of PI3K/AKT Pathway in Differentiating Human Visceral Adipocytes. Int. J. Mol. Sci. 2021, 22, 7712. https://doi.org/10.3390/ijms22147712
Świderska E, Strycharz J, Wróblewski A, Czarny P, Szemraj J, Drzewoski J, Śliwińska A. Chronic and Intermittent Hyperglycemia Modulates Expression of Key Molecules of PI3K/AKT Pathway in Differentiating Human Visceral Adipocytes. International Journal of Molecular Sciences. 2021; 22(14):7712. https://doi.org/10.3390/ijms22147712
Chicago/Turabian StyleŚwiderska, Ewa, Justyna Strycharz, Adam Wróblewski, Piotr Czarny, Janusz Szemraj, Józef Drzewoski, and Agnieszka Śliwińska. 2021. "Chronic and Intermittent Hyperglycemia Modulates Expression of Key Molecules of PI3K/AKT Pathway in Differentiating Human Visceral Adipocytes" International Journal of Molecular Sciences 22, no. 14: 7712. https://doi.org/10.3390/ijms22147712
APA StyleŚwiderska, E., Strycharz, J., Wróblewski, A., Czarny, P., Szemraj, J., Drzewoski, J., & Śliwińska, A. (2021). Chronic and Intermittent Hyperglycemia Modulates Expression of Key Molecules of PI3K/AKT Pathway in Differentiating Human Visceral Adipocytes. International Journal of Molecular Sciences, 22(14), 7712. https://doi.org/10.3390/ijms22147712






