Screening Anti-Inflammatory Effects of Flavanones Solutions
Abstract
1. Introduction
2. Results
2.1. In Silico Analyses
2.2. Model of Mice Ear Inflammation Induced with TPA
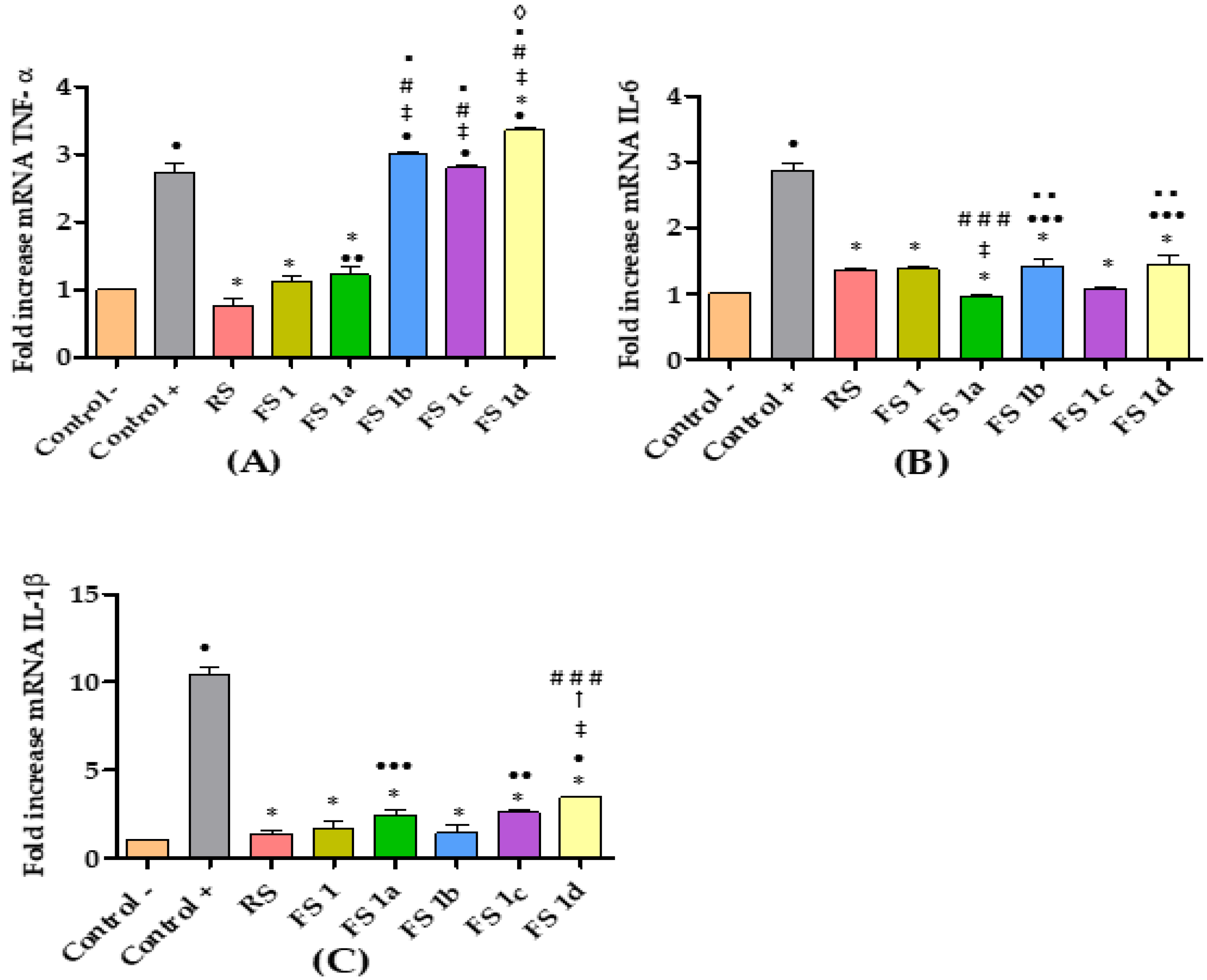
2.3. Model of Rat Ear Inflammation Induced with AA and Anti-Inflammatory Response after Flavanone Solution Treatment
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Plant Extraction and Derivatization of Flavanones
4.3. In Silico Analysis
4.4. Flavanone Solutions (FS)
4.5. In Vivo Anti-Inflammatory Testing
4.5.1. The TPA-Induced Rat Ear Inflammation Model
4.5.2. Arachidonic Acid (AA)-Induced Rat Ear Inflammation Model
4.5.3. Gene Expression Analysis by RT-qPCR
4.5.4. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Abdel-Mottaleb, M.M.; Try, C.; Pellequer, Y.; Lamprecht, A. Nanomedicine strategies for targeting skin inflammation. Nanomedicine 2014, 9, 1727–1743. [Google Scholar] [CrossRef]
- Maleki, S.J.; Crespo, J.F.; Cabanillas, B. Anti-inflammatory effects of flavonoids. Food Chem. 2019, 299, 125124. [Google Scholar] [CrossRef]
- Paoletti, T.; Fallarini, S.; Gugliesi, F.; Minassi, A.; Appendino, G.B.; Lombardi, G. Anti-inflammatory and vascular protective properties of 8-prenylapigenin. Eur. J. Pharmacol. 2009, 620, 120–130. [Google Scholar] [CrossRef] [PubMed]
- Park, K.E.; Qin, Y.; Bavry, A.A. Nonsteroidal anti-inflammatory drugs and their effects in the elderly. Aging Health 2012, 8, 167–177. [Google Scholar] [CrossRef]
- Li, Y.-S.; Zhang, J.; Tian, G.-H.; Shang, H.-C.; Tang, H.-B. Kirenol, darutoside and hesperidin contribute to the anti-inflammatory and analgesic activities of Siegesbeckia pubescens makino by inhibiting COX-2 expression and inflammatory cell infiltration. J. Ethnopharmacol. 2021, 268, 113547. [Google Scholar] [CrossRef] [PubMed]
- Keseroglu, H.O.; Gonul, M. Traditional topical herbal therapies in psoriasis. Tang Humanit. Med. 2014, 4, 23.1–23.8. [Google Scholar] [CrossRef]
- Dhingra, A.K.; Chopra, B.; Bonthagarala, B. Natural Anti-Inflammatory Agents: Recent Progress and Future Perspectives. Ann. Pharmacol. Pharm. 2018, 3, 1158. [Google Scholar]
- Singh, M.R.; Nag, M.K.; Patel, S.; Daharwal, S.J. Novel Approaches for Dermal and Transdermal Delivery of Herbal Drugs. J. Pharmacogn. Phytochem. 2013, 5, 271–279. [Google Scholar]
- Scheau, C.; Badarau, I.A.; Mihai, L.-G.; Scheau, A.-E.; Costache, D.O.; Constantin, C.; Calina, D.; Caruntu, C.; Costache, R.S.; Caruntu, A. Cannabinoids in the Pathophysiology of Skin Inflammation. Molecules 2020, 25, 652. [Google Scholar] [CrossRef]
- Kim, H.P.; Son, K.H.; Chang, H.W.; Kang, S.S. Anti-inflammatory Plant Polyphenolics and Cellular Action Mechanisms. Curr. Bioact. Compd. 2020, 16, 809–817. [Google Scholar] [CrossRef]
- Chen, J.; Li, W.; Yao, H.; Xu, J. Insights into drug discovery from natural products through structural modification. Fitoterapia 2015, 103, 231–241. [Google Scholar] [CrossRef]
- Deenonpoe, R.; Prayong, P.; Thippamom, N.; Meephansan, J.; Na-Bangchang, K. Anti-inflammatory effect of naringin and sericin combination on human peripheral blood mononuclear cells (hPBMCs) from patient with psoriasis. BMC Complement. Altern. Med. 2019, 19, 1–11. [Google Scholar] [CrossRef]
- Al-Roujayee, A.S. Naringenin improves the healing process of thermally-induced skin damage in rats. J. Int. Med. Res. 2017, 45, 570–582. [Google Scholar] [CrossRef]
- Chi, Y.S.; Lim, H.; Park, H.; Kim, H.P. Effects of wogonin, a plant flavone from Scutellaria radix, on skin inflammation: In vivo regulation of inflammation-associated gene expression. Biochem. Pharmacol. 2003, 66, 1271–1278. [Google Scholar] [CrossRef]
- Alalaiwe, A.; Lin, C.-F.; Hsiao, C.-Y.; Chen, E.-L.; Lin, C.-Y.; Lien, W.-C.; Fang, J.-Y. Development of flavanone and its derivatives as topical agents against psoriasis: The prediction of therapeutic efficiency through skin permeation evaluation and cell-based assay. Int. J. Pharm. 2020, 581, 119256. [Google Scholar] [CrossRef] [PubMed]
- Barreca, D.; Gattuso, G.; Bellocco, E.S.; Calderaro, A.; Trombetta, D.; Smeriglio, A.; Laganà, G.; Daglia, M.; Meneghini, S.; Nabavi, S.M. Flavanones: Citrus phytochemical with health-promoting properties. BioFactors 2017, 43, 495–506. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Campoy, A.; Garcia, E.; Muñiz-Ramirez, A. Phytochemical and Pharmacological Study of the Eysenhardtia Genus. Plants 2020, 9, 1124. [Google Scholar] [CrossRef] [PubMed]
- Narváez-Mastache, J.M.; Soto, C.; Delgado, G. Antioxidant Evaluation of Eysenhardtia Species (Fabaceae): Relay Synthesis of 3-O-Acetyl-11α, 12α-epoxy-oleanan-28, 13β-olide Isolated from E. platycarpa and Its Protective Effect in Experimental Diabetes. Biol. Pharm. Bull. 2007, 30, 1503–1510. [Google Scholar] [CrossRef]
- Wächter, G.A.; Hoffmann, J.J.; Furbacher, T.; Blake, M.E.; Timmermann, B.N. Antibacterial and antifungal flavanones from Eysenhardtia texana. Phytochemistry 1999, 52, 1469–1471. [Google Scholar] [CrossRef]
- Pérez-Guitierrez, R.M.; Vargas-solís, R.; García-Dueñas, L.M.; Dávila-Badillo, L. Efecto de isoflavonas aisladas de la corteza de Eysenhardtia polystachya sobre el crecimiento de cristales de oxalato y fosfato de calcio urinario. Boletín del Col. Mex. Urol. 2002, 17, 134–139. [Google Scholar]
- Alvarez, L.; Rios, M.Y.; Esquivel, C.; Chávez, M.I.; Delgado, G.; Aguilar, M.I.; Villarreal, M.L.; Navarro, V. Cytotoxic Isoflavans from Eysenhardtia polystachya. J. Nat. Prod. 1998, 61, 767–770. [Google Scholar] [CrossRef] [PubMed]
- Domínguez-Villegas, V.; Domínguez-Villegas, V.; García, M.L.; Calpena, A.C.; Clares-Naveros, B.; Garduño-Ramirez, M.L. Anti-inflammatory, Antioxidant and Cytotoxicity Activities of Methanolic Extract and Prenylated Flavanones Isolated from Leaves of Eysehardtia platycarpa. Nat. Prod. Commun. 2013, 8, 177–180. [Google Scholar] [CrossRef]
- Shi, L.; Feng, X.E.; Cui, J.R.; Fang, L.H.; Du, G.H.; Li, Q.S. Synthesis and biological activity of flavanone derivatives. Bioorg. Med. Chem. Lett. 2010, 20, 5466–5468. [Google Scholar] [CrossRef]
- Gordaliza, M. Natural products as leads to anticancer drugs. Clin. Transl. Oncol. 2007, 9, 767–776. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Xiong, Q.; Lai, X.; Li, X.; Wan, M.; Zhang, J.; Yan, Y.; Cao, M.; Lu, L.; Guan, J.; et al. Molecular Modification of Polysaccharides and Resulting Bioactivities. Compr. Rev. Food Sci. Food Saf. 2015, 15, 237–250. [Google Scholar] [CrossRef]
- Filimonov, D.; Lagunin, A.A.; Gloriozova, T.A.; Rudik, A.; Druzhilovskii, D.S.; Pogodin, P.V.; Poroikov, V.V. Prediction of the Biological Activity Spectra of Organic Compounds Using the Pass Online Web Resource. Chem. Heterocycl. Compd. 2014, 50, 444–457. [Google Scholar] [CrossRef]
- Nadeem, S.; Sirajuddin, M.; Ahmad, S.; Tirmizi, S.A.; Ali, M.I.; Hameed, A. Synthesis, spectral characterization and in vitro antibacterial evaluation and Petra/Osiris/Molinspiration analyses of new Palladium(II) iodide complexes with thioamides. Alex. J. Med. 2016, 52, 279–288. [Google Scholar] [CrossRef]
- Farias, I.V.; Faqueti, L.G.; Noldin, V.F.; Junior, G.F.; Nowil, A.E.; Schuquel, I.T.; Monache, F.D.; García, P.A.; López-Pérez, J.L.; Feliciano, A.S.; et al. Cytotoxic phloroglucinol meroterpenoid from Eugenia umbelliflora fruits. Phytochem. Lett. 2018, 27, 187–192. [Google Scholar] [CrossRef]
- Jarrahpour, A.; Fathi, J.; Mimouni, M.; Hadda, T.B.; Sheikh, J.; Chohan, Z.; Parvez, A. Petra, Osiris and Molinspiration (POM) together as a successful support in drug design: Antibacterial activity and biopharmaceutical characterization of some azo Schiff bases. Med. Chem. Res. 2011, 21, 1984–1990. [Google Scholar] [CrossRef]
- Baskar, V.; Jayalakshmi, C.; Pavithra, N.; Veronica Grite, S. Validating therapeutically active phytochemical compounds for anti-ageing by in silico pharmacokinetic approach. J. Biol. Inf. Sci. 2014, 3, 1–7. [Google Scholar]
- Yi, Y.-S. Flavonoids: Nutraceuticals for Rheumatic Diseases via Targeting of Inflammasome Activation. Int. J. Mol. Sci. 2021, 22, 488. [Google Scholar] [CrossRef] [PubMed]
- Chibli, L.A.; Rodrigues, K.C.; Gasparetto, C.M.; Pinto, N.C.; Fabri, R.L.; Scio, E.; Alves, M.S.; Del-Vechio-Vieira, G.; Sousa, O.V. Anti-inflammatory effects of Bryophyllum pinnatum (Lam.) Oken ethanol extract in acute and chronic cutaneous inflammation. J. Ethnopharmacol. 2014, 154, 330–338. [Google Scholar] [CrossRef]
- Siddiqui, F.; Naqvi, S.; Abidi, L.; Faizi, S.; Avesi, L.; Mirza, T.; Farooq, A.D. Opuntia dillenii cladode: Opuntiol and opuntioside attenuated cytokines and eicosanoids mediated inflammation. J. Ethnopharmacol. 2016, 182, 221–234. [Google Scholar] [CrossRef]
- Bustos-Salgado, P.; Andrade-Carrera, B.; Domínguez-Villegas, V.; Rodríguez-Lagunas, M.J.; Boix-Montañes, A.; Calpena-Campmany, A.; Garduño-Ramírez, M.L. Biopharmaceutic study and in vivo efficacy of natural and derivatives flavanones formulations. Nanomedicine 2021, 16, 205–220. [Google Scholar] [CrossRef]
- Gomes, A.; Fernandes, E.; Lima, J.; Mira, L.; Corvo, M.L. Molecular Mechanisms of Anti-Inflammatory Activity Mediated by Flavonoids. Curr. Med. Chem. 2008, 15, 1586–1605. [Google Scholar] [CrossRef]
- Gautam, R.; Jachak, S.M. Recent developments in anti-inflammatory natural products. Med. Res. Rev. 2009, 29, 767–820. [Google Scholar] [CrossRef]
- Otuki, M.F.; Vieira-Lima, F.; Malheiros, Â.; Yunes, R.A.; Calixto, J.B. Topical anti-inflammatory effects of the ether extract from Protium kleinii and α-amyrin pentacyclic triterpene. Eur. J. Pharmacol. 2005, 507, 253–259. [Google Scholar] [CrossRef]
- Ren, X.; Zhang, M.; Chen, L.; Zhang, W.; Huang, Y.; Luo, H.; Li, L.; He, H. The anti-inflammatory effects of Yunnan Baiyao are involved in regulation of the phospholipase A2/arachidonic acid metabolites pathways in acute inflammation rat model. Mol. Med. Rep. 2017, 16, 4045–4053. [Google Scholar] [CrossRef]
- Sanaki, T.; Kasai-Yamamoto, E.; Yoshioka, T.; Sakai, S.; Yuyama, K.; Fujiwara, T.; Numata, Y.; Igarashi, Y. Direct Involvement of Arachidonic Acid in the Development of Ear Edema via TRPV3. J. Oleo Sci. 2017, 66, 591–599. [Google Scholar] [CrossRef][Green Version]
- Rodrigues, K.C.M.; Chibli, L.A.; Santos, B.C.S.; Temponi, V.S.; Pinto, N.C.C.; Scio, E.; Del-Vechio-Vieira, G.; Alves, M.S.; Sousa, O.V. Evidence of Bioactive Compounds from Vernonia polyanthes Leaves with Topical Anti-Inflammatory Potential. Int. J. Mol. Sci. 2016, 17, 1929. [Google Scholar] [CrossRef]
- Griswold, D.E.; Martin, L.D.; Badger, A.M.; Breton, J.; Chabot-Fletcher, M. Evaluation of the cutaneous anti-inflammatory activity of azaspiranes. Inflamm. Res. 1998, 47, 56–61. [Google Scholar] [CrossRef]
- Passos, G.F.; Medeiros, R.; Marcon, R.; Nascimento, A.F.; Calixto, J.B.; Pianowski, L.F. The role of PKC/ERK1/2 signaling in the anti-inflammatory effect of tetracyclic triterpene euphol on TPA-induced skin inflammation in mice. Eur. J. Pharmacol. 2013, 698, 413–420. [Google Scholar] [CrossRef]
- Gábor, MThe mouse ear as a model for cutaneous irritation. J. Toxicol. Cutan. Ocul. Toxicol. 2002, 21, 191–202. [CrossRef]
- Saraiva, R.A.; Araruna, M.K.; Oliveira, R.C.; Menezes, K.D.; Leite, G.O.; Kerntopf, M.R.; Costa, J.G.; Rocha, J.B.; Tomé, A.R.; Campos, A.R.; et al. Topical anti-inflammatory effect of Caryocar coriaceum Wittm. (Caryocaraceae) fruit pulp fixed oil on mice ear edema induced by different irritant agents. J. Ethnopharmacol. 2011, 136, 504–510. [Google Scholar] [CrossRef]
- Peinnequin, A.; Mouret, C.; Birot, O.; Alonso, A.; Mathieu, J.; Clarençon, D.; Agay, D.; Chancerelle, Y.; Multon, E. Rat pro-inflammatory cytokine and cytokine related mRNA quantification by real-time polymerase chain reaction using SYBR green. BMC Immunol. 2004, 5, 3. [Google Scholar] [CrossRef]
- Spies, M.; Nesic, O.; Barrow, R.E.; Perez-Polo, J.R.; Herndon, D.N. Liposomal IGF-1 gene transfer modulates pro- and anti-inflammatory cytokine mRNA expression in the burn wound. Gene Ther. 2001, 8, 1409–1415. [Google Scholar] [CrossRef][Green Version]
- Xiao, S.; Yu, H.; Xie, Y.; Guo, Y.; Fan, J.; Yao, W. The anti-inflammatory potential of Cinnamomum camphora (L.) J.Presl essential oil in vitro and in vivo. J. Ethnopharmacol. 2021, 267, 113516. [Google Scholar] [CrossRef] [PubMed]
- Giongo, J.L.; Vaucher, R.D.A.; Sagrillo, M.R.; Santos, R.C.V.; Duarte, M.M.; Rech, V.C.; Lopes, L.Q.S.; da Cruz, I.; Tatsch, E.; Moresco, R.N.; et al. Anti-inflammatory effect of geranium nanoemulsion macrophages induced with soluble protein of Candida albicans. Microb. Pathog. 2017, 110, 694–702. [Google Scholar] [CrossRef]
- Wang, H.; Peters, T.; Kess, D.; Sindrilaru, A.; Oreshkova, T.; Van Rooijen, N.; Stratis, A.; Renkl, A.C.; Sunderkötter, C.; Wlaschek, M.; et al. Activated macrophages are essential in a murine model for T cell-mediated chronic psoriasiform skin inflammation. J. Clin. Investig. 2006, 116, 2105–2114. [Google Scholar] [CrossRef]
- Yao, F.; Xue, Q.; Li, K.; Cao, X.; Sun, L.; Liu, Y. Phenolic Compounds and Ginsenosides in Ginseng Shoots and Their Antioxidant and Anti-Inflammatory Capacities in LPS-Induced RAW264.7 Mouse Macrophages. Int. J. Mol. Sci. 2019, 20, 2951. [Google Scholar] [CrossRef]
- Owona, B.A.; Abia, W.A.; Moundipa, P.F. Natural compounds flavonoids as modulators of inflammasomes in chronic diseases. Int. Immunopharmacol. 2020, 84, 106498. [Google Scholar] [CrossRef]
- Denaro, M.; Smeriglio, A.; Trombetta, D. Antioxidant and anti-inflammatory activity of citrus flavanones mix and its stability after in vitro simulated digestion. Antioxidants 2021, 10, 140. [Google Scholar] [CrossRef]
- Tejada, S.; Pinya, S.; Martorell, M.; Capó, X.; Tur, J.A.; Pons, A.; Sureda, A. Potential Anti-inflammatory Effects of Hesperidin from the Genus Citrus. Curr. Med. Chem. 2019, 25, 4929–4945. [Google Scholar] [CrossRef]
- Pahl, H.L. Activators and target genes of Rel/NF-kappaB transcription factors. Oncogene 1999, 18, 6853–6866. [Google Scholar] [CrossRef]
- Sarango-Granda, P.; Silva-Abreu, M.; Calpena, A.; Halbaut, L.; Fábrega, M.-J.; Rodríguez-Lagunas, M.; Díaz-Garrido, N.; Badia, J.; Espinoza, L. Apremilast Microemulsion as Topical Therapy for Local Inflammation: Design, Characterization and Efficacy Evaluation. Pharmaceuticals 2020, 13, 484. [Google Scholar] [CrossRef]
- Xian, Y.-F.; Hu, Z.; Ip, S.-P.; Chen, J.-N.; Su, Z.-R.; Lai, X.-P.; Lin, Z.-X. Comparison of the anti-inflammatory effects of Sinapis alba and Brassica juncea in mouse models of inflammation. Phytomedicine 2018, 50, 196–204. [Google Scholar] [CrossRef]
- Andrade-Carrera, B.; Clares, B.; Noé, V.; Mallandrich, M.; Calpena, A.C.; García, M.L.; Garduño-Ramírez, M.L. Cytotoxic Evaluation of (2S)-5,7-Dihydroxy-6-prenylflavanone Derivatives Loaded PLGA Nanoparticles against MiaPaCa-2 Cells. Molecules 2017, 22, 1553. [Google Scholar] [CrossRef]
- Tariq, M.; Sirajuddin, M.; Ali, S.; Khalid, N.; Tahir, M.N.; Khan, H.; Ansari, T.M. Pharmacological investigations and Petra/Osiris/Molinspiration (POM) analyses of newly synthesized potentially bioactive organotin(IV) carboxylates. J. Photochem. Photobiol. B Biol. 2016, 158, 174–183. [Google Scholar] [CrossRef]
- Imran, M.; Aziz, M.; Kumar, N.; Kousar, Z.; Shabnam, S.; Nohri, F. Synthesis, spectroscopic characterization and Petra Osiris Molinspiration (POM) analyses of dicarboxylic acid amides. Int. J. Pharm. Sci. Res. 2016, 7, 1915–1927. [Google Scholar] [CrossRef]
- Bustos-Salgado, P.; Andrade-Carrera, B.; Garduño-Ramírez, M.L.; Alvarado, H.; Calpena-Campmany, A. Quantification of one Prenylated Flavanone from Eysenhardtia platycarpa and four derivatives in Ex Vivo Human Skin Permeation Samples Applying a Validated HPLC Method. Biomolecules 2020, 10, 889. [Google Scholar] [CrossRef]
- Rincón, M.; Calpena, A.C.; Clares, B.; Espina, M.; Garduño-Ramírez, M.L.; Rodríguez-Lagunas, M.J.; García, M.L.; Abrego, G. Skin-controlled release lipid nanosystems of pranoprofen for the treatment of local inflammation and pain. Nanomedicine 2018, 13, 2397–2413. [Google Scholar] [CrossRef]
- Espinoza, L.C.; Silva-Abreu, M.; Calpena, A.C.; Rodríguez-Lagunas, M.J.; Fábrega, M.-J.; Garduño-Ramírez, M.L.; Clares, B. Nanoemulsion strategy of pioglitazone for the treatment of skin inflammatory diseases. Nanomed. Nanotechnol. Biol. Med. 2019, 19, 115–125. [Google Scholar] [CrossRef]
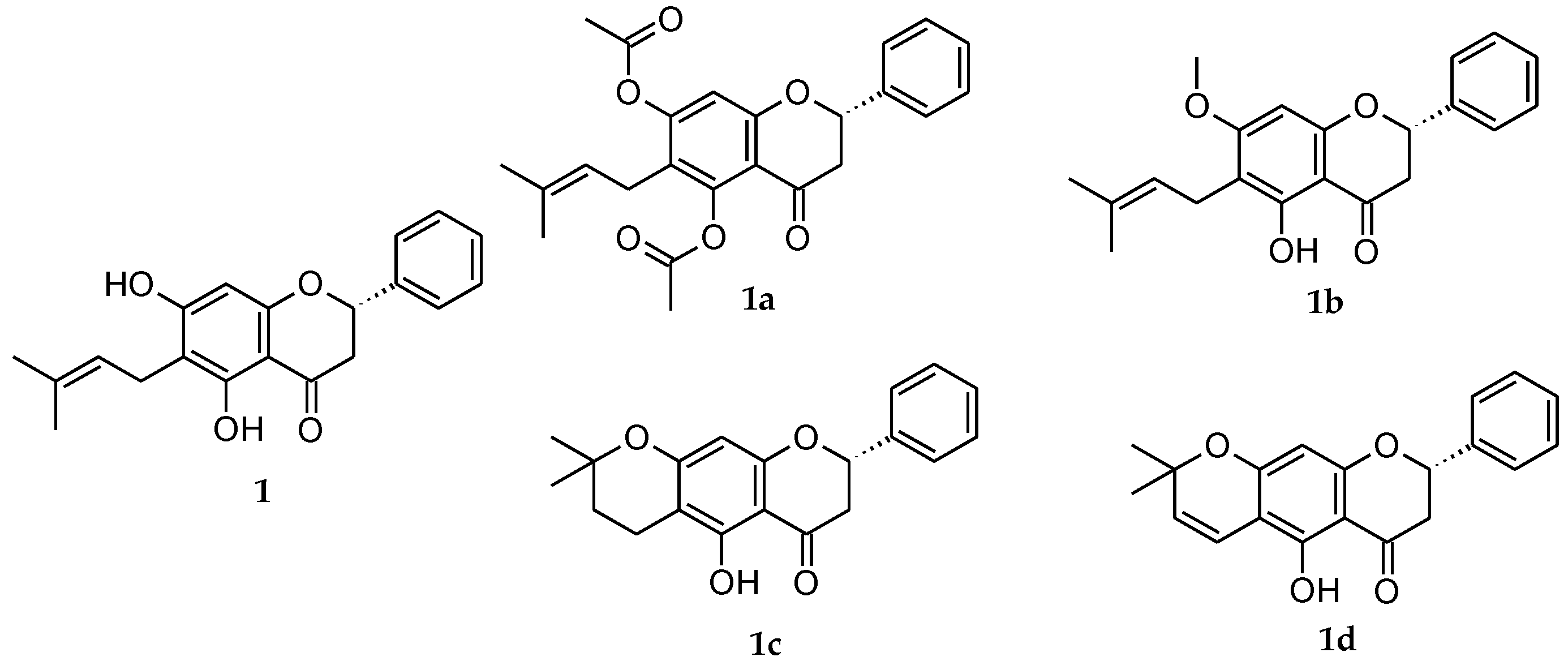
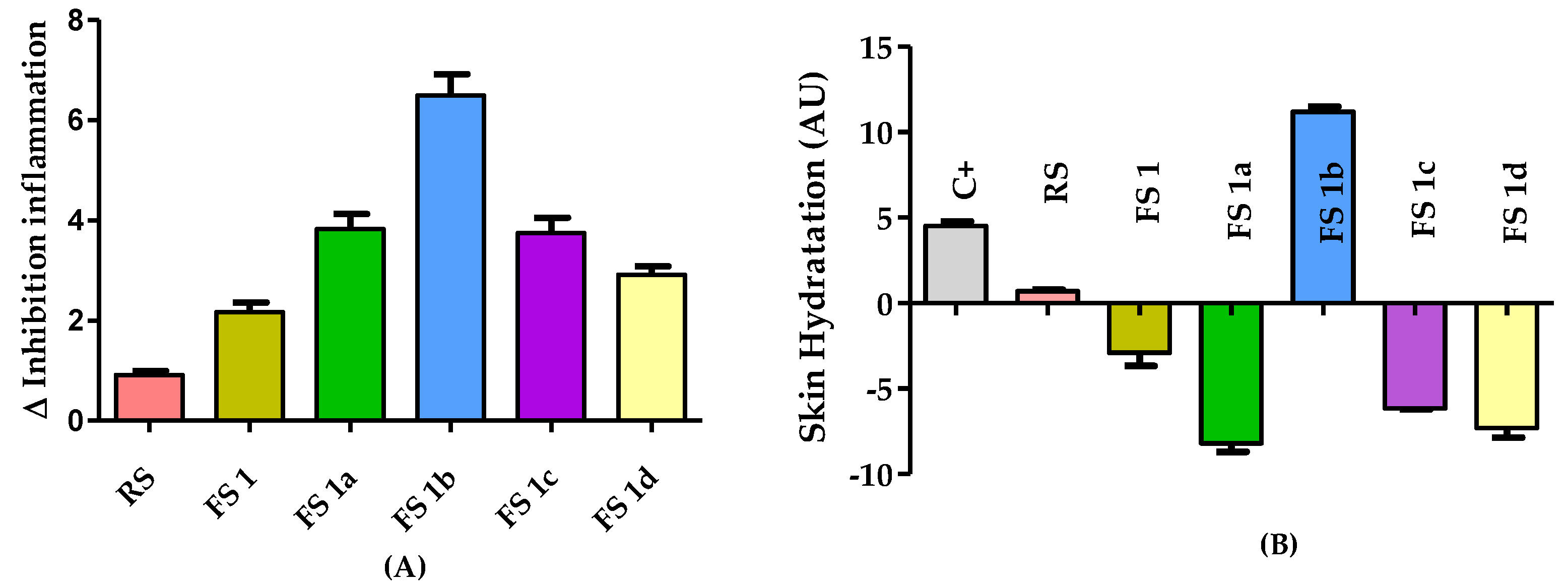
 auricular cartilage,
auricular cartilage,  dermis,
dermis,  epidermis,
epidermis,  stratum corneum. Arrows indicate presence of edema. Scale bar = 200 µm.
stratum corneum. Arrows indicate presence of edema. Scale bar = 200 µm.
 auricular cartilage,
auricular cartilage,  dermis,
dermis,  epidermis,
epidermis,  stratum corneum. Arrows indicate presence of edema. Scale bar = 200 µm.
stratum corneum. Arrows indicate presence of edema. Scale bar = 200 µm.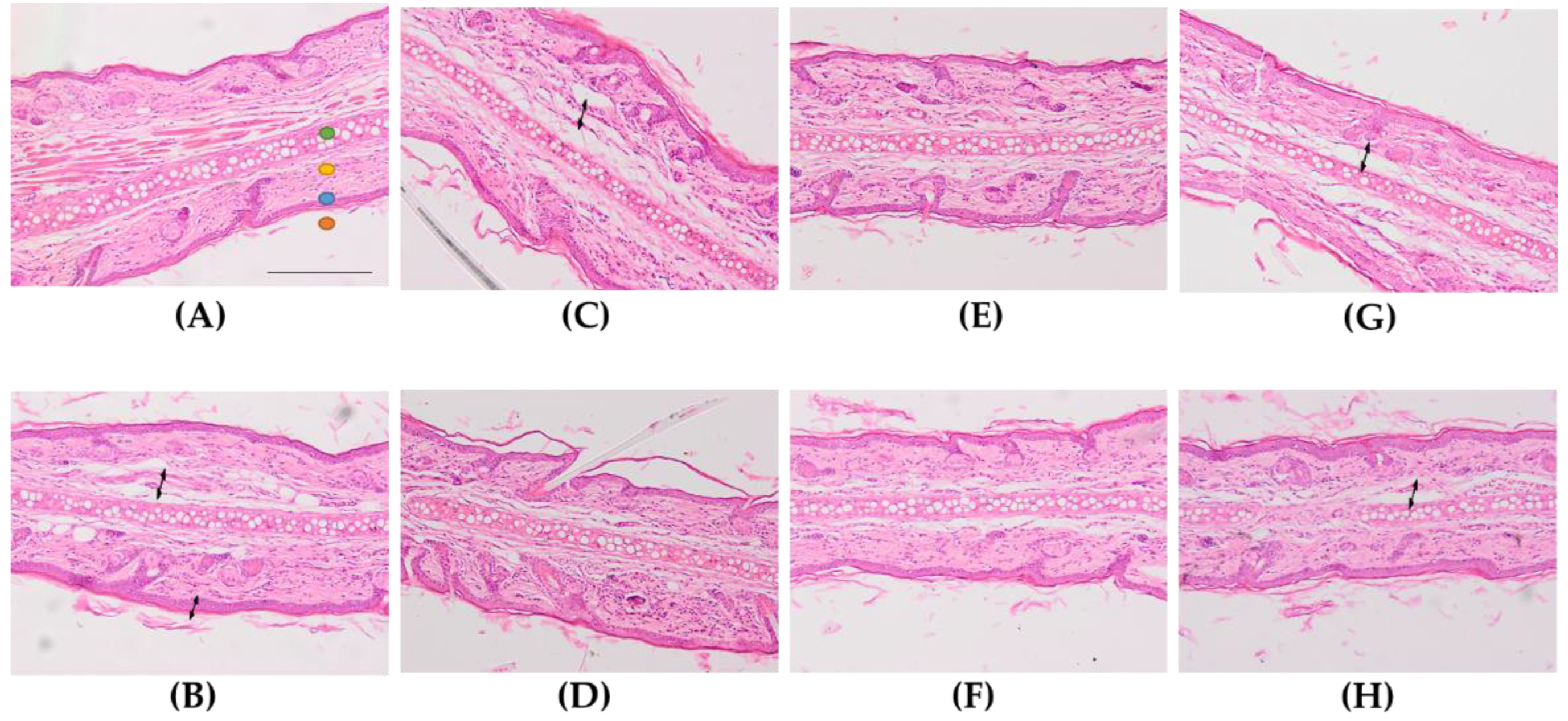
| Data | Flavanone | Diclofenac | Indomethacin | ||||
|---|---|---|---|---|---|---|---|
| 1 | 1a | 1b | 1c | 1d | |||
| Anti-inflammatory (Pa) | 0.66 | 0.72 | 0.61 | 0.74 | 0.62 | 0.79 | 0.71 |
| miLogP | 4.49 | 3.82 | 4.56 | 4.22 | 4.07 | 4.57 | 3.99 |
| TPSA | 66.76 | 78.92 | 55.77 | 55.77 | 55.77 | 49.33 | 68.54 |
| natoms | 23 | 29 | 24 | 24 | 24 | 19 | 25 |
| MW | 310.35 | 394.42 | 324.38 | 324.38 | 322.36 | 296.15 | 357.79 |
| Volume | 282.78 | 355.81 | 300.31 | 295.6 | 289.41 | 238.73 | 303.24 |
| Solutions | FS1 | FS1a | FS1b | FS1c | FS1d | Indomethacin |
|---|---|---|---|---|---|---|
| % Inhibition | 66.67 ± 1.55 | 10.27 ± 0.21 | 25.69 ± 0.52 | 40.61 ± 0.81 | 96.27 ± 1.93 | 91.35 ± 0.47 |
| Gene (Rat) | Primer Sequences (5′-3′) | Gene Accession Number |
|---|---|---|
| IL-6 | FW: AGAAAAGAGTTGTGCAATGGCA RV: GGCAAATTTCCTGGTTATATCC | NM_012589.2 |
| TNF-α | FW: AAATGGGCTCCCTCTCATCAGTTC RV: TCTGCTTGGTGGTTTGCTACGAC | NM_012675.3 |
| IL-1β | FW: CACCTCTCAAGCAGAGCACAG RV: GGGTTCCATGGTGAAGTCAAC | NM_031512.2 |
| β-actin | FW: AAGTCCCTCACCCTCCCAAAA RV: AAGCAATGCTGTCACCTTCCC | V01217.1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bustos-Salgado, P.; Andrade-Carrera, B.; Domínguez-Villegas, V.; Díaz-Garrido, N.; Rodríguez-Lagunas, M.J.; Badía, J.; Baldomà, L.; Mallandrich, M.; Calpena-Campmany, A.; Garduño-Ramírez, M.L. Screening Anti-Inflammatory Effects of Flavanones Solutions. Int. J. Mol. Sci. 2021, 22, 8878. https://doi.org/10.3390/ijms22168878
Bustos-Salgado P, Andrade-Carrera B, Domínguez-Villegas V, Díaz-Garrido N, Rodríguez-Lagunas MJ, Badía J, Baldomà L, Mallandrich M, Calpena-Campmany A, Garduño-Ramírez ML. Screening Anti-Inflammatory Effects of Flavanones Solutions. International Journal of Molecular Sciences. 2021; 22(16):8878. https://doi.org/10.3390/ijms22168878
Chicago/Turabian StyleBustos-Salgado, Paola, Berenice Andrade-Carrera, Valeri Domínguez-Villegas, Natalia Díaz-Garrido, María J. Rodríguez-Lagunas, Josefa Badía, Laura Baldomà, Mireia Mallandrich, Ana Calpena-Campmany, and María Luisa Garduño-Ramírez. 2021. "Screening Anti-Inflammatory Effects of Flavanones Solutions" International Journal of Molecular Sciences 22, no. 16: 8878. https://doi.org/10.3390/ijms22168878
APA StyleBustos-Salgado, P., Andrade-Carrera, B., Domínguez-Villegas, V., Díaz-Garrido, N., Rodríguez-Lagunas, M. J., Badía, J., Baldomà, L., Mallandrich, M., Calpena-Campmany, A., & Garduño-Ramírez, M. L. (2021). Screening Anti-Inflammatory Effects of Flavanones Solutions. International Journal of Molecular Sciences, 22(16), 8878. https://doi.org/10.3390/ijms22168878









