Exploring the Biological Activity and Mechanism of Xenoestrogens and Phytoestrogens in Cancers: Emerging Methods and Concepts
Abstract
1. Introduction
2. Xenoestrogens and Phytoestrogens: Definitions and Origins
2.1. Xenoestrogens: Synthetic Industrial Chemicals
2.2. Phytoestrogens: Plant-Derived Chemicals
3. Advanced Methodology in Studying the Biological Effects of Xenoestrogens and Phytoestrogens
3.1. In Vitro Models with Cultured Cells
3.2. In Vivo Models with Rodents
3.3. Single-Cell RNA-Sequencing (scRNA-seq)
4. Biological Activities and Mechanisms of Xenoestrogens and Phytoestrogens in Cancers
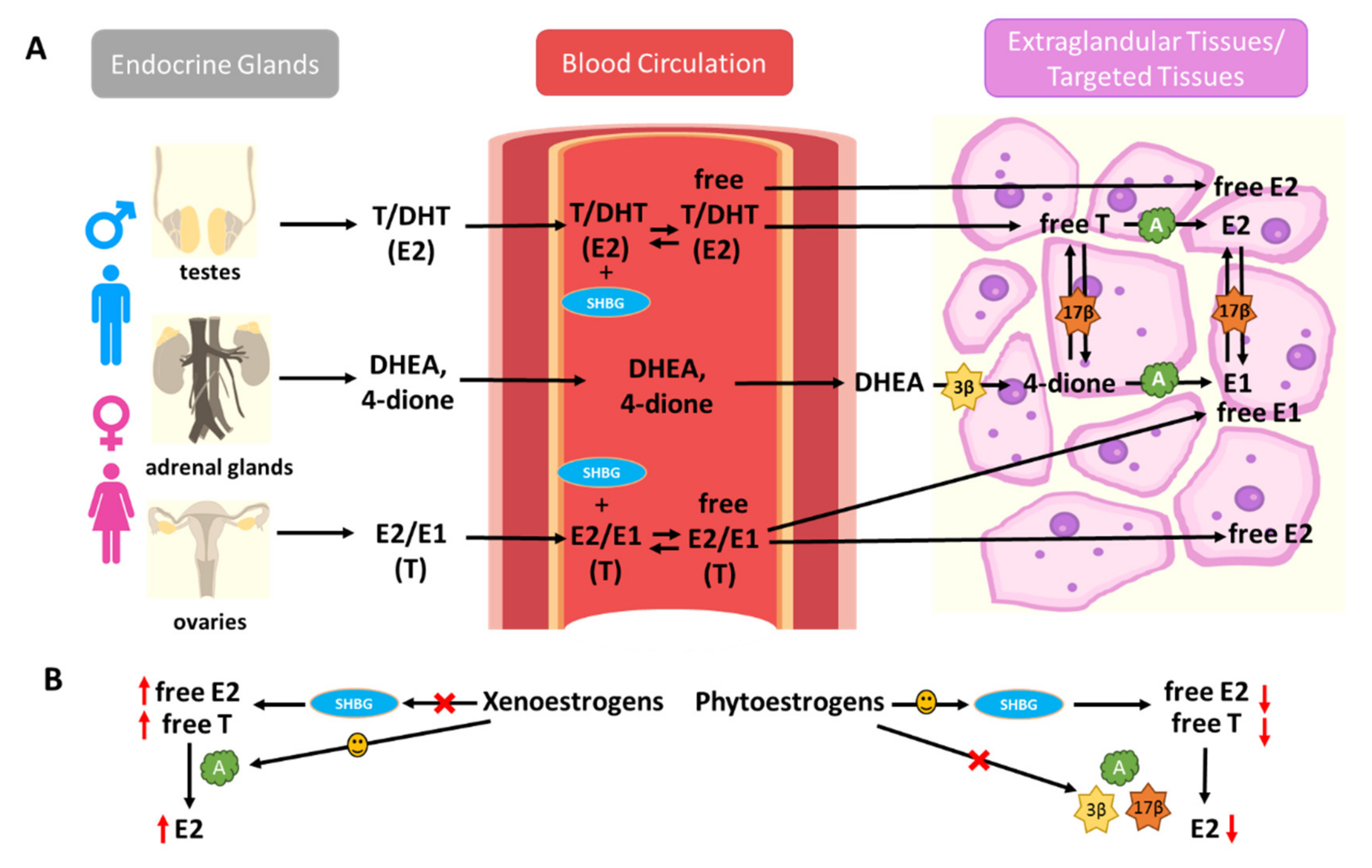
4.1. Effects of Xenoestrogens and Phytoestrogens on the Bioavailability and Formation of Endogenous Estrogens
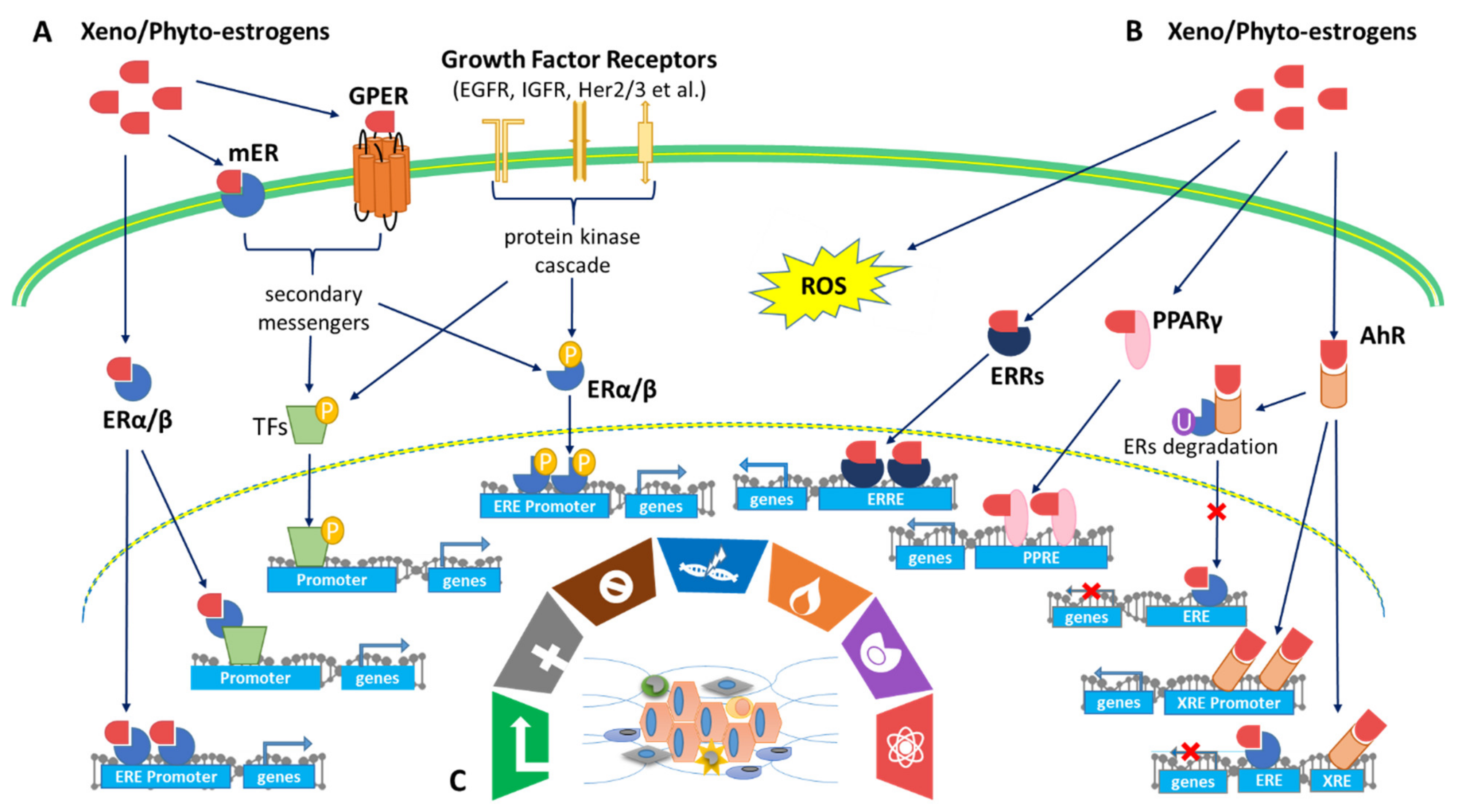
4.2. Effects of Xenoestrogens and Phytoestrogens on Estrogen Receptor Activation and Signaling
4.3. Effect of Xenoestrogens/Phytoestrogens on Activation of AhR/PPARγ/ROS Pathways
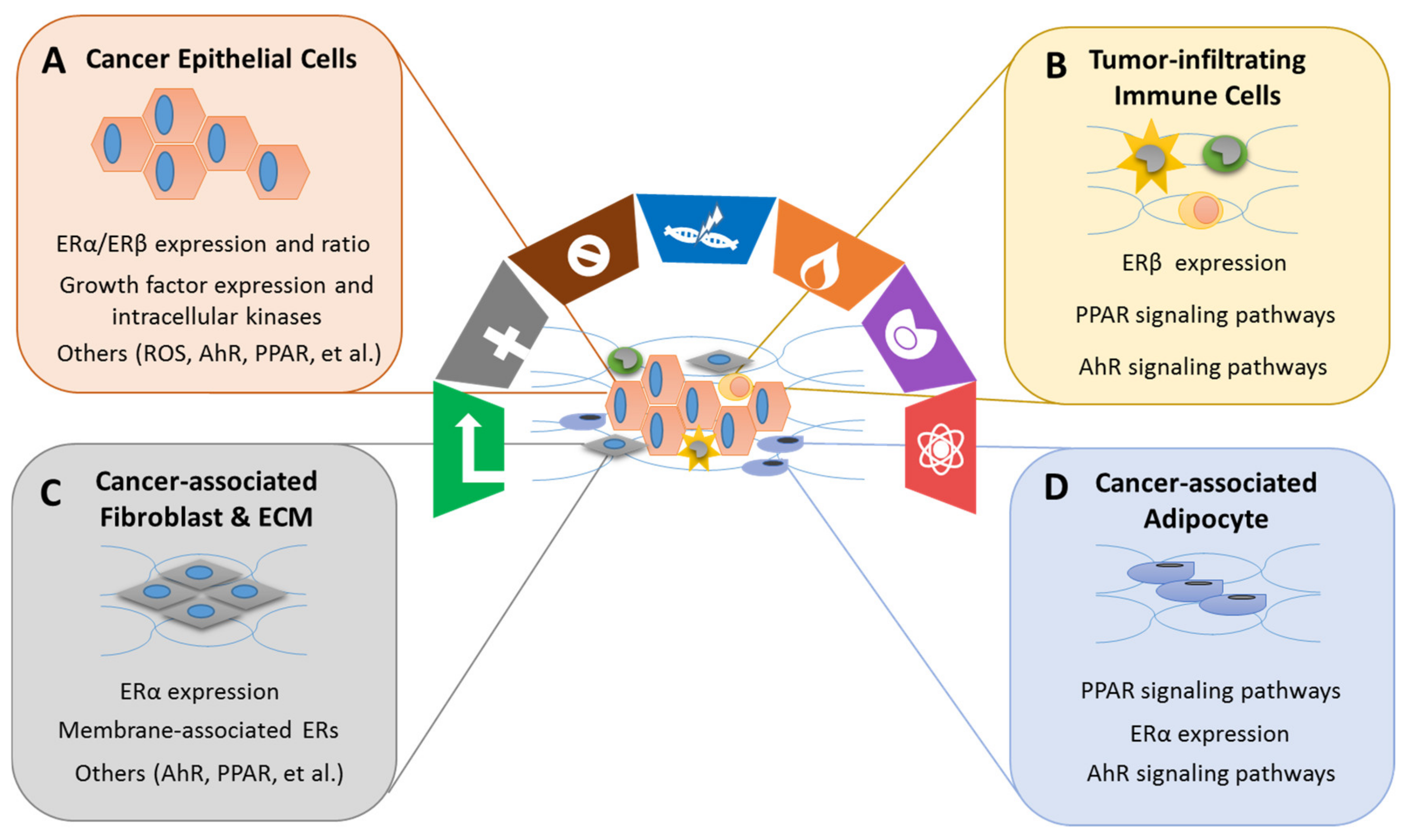
4.4. Effects of Xenoestrogens/Phytoestrogens on Modulating the Hallmarks of Cancer
4.5. Effects of Xenoestrogens/Phytoestrogens Determines on Critical Timing of Exposure
5. Application of Phytoestrogens in the Prevention or Treatment of Cancers: Evidence from Clinical Trials
6. Future Directions and Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Tazuke, S.; Khaw, K.-T.; Barrett-Connor, E. Exogenous estrogen and endogenous sex hormones. Medicine 1992, 71, 44–51. [Google Scholar] [CrossRef]
- Wendy, Y.; Chen, M.D. Exogenous and endogenous hormones and breast cancer. Best Pract. Res. Clin. Endocrinol. Metab. 2008, 22, 573–585. [Google Scholar]
- Hu, W.Y.; Shi, G.B.; Hu, D.P.; Nelles, J.L.; Prins, G.S. Actions of estrogens and endocrine disrupting chemicals on human prostate stem/progenitor cells and prostate cancer risk. Mol. Cell Endocrinol. 2012, 354, 63–73. [Google Scholar] [CrossRef]
- Hsu, L.H.; Chu, N.M.; Kao, S.H. Estrogen, Estrogen Receptor and Lung Cancer. Int. J. Mol. Sci. 2017, 18, 1713. [Google Scholar] [CrossRef]
- Barzi, A.; Lenz, A.M.; LaBonte, M.J.; Lenz, H.J. Molecular pathways: Estrogen pathway in colorectal cancer. Clin. Cancer Res. 2013, 19, 5842–5848. [Google Scholar] [CrossRef]
- Ur Rahman, M.S.; Cao, J. Estrogen receptors in gastric cancer: Advances and perspectives. World J. Gastroenterol. 2016, 22, 2475–2482. [Google Scholar] [CrossRef]
- Miller, W.L.; Auchus, R.J. The molecular biology, biochemistry, and physiology of human steroidogenesis and its disorders. Endocr. Rev. 2011, 32, 81–151. [Google Scholar] [CrossRef]
- Barakat, R.; Oakley, O.; Kim, H.; Jin, J.; Ko, C.J. Extra-gonadal sites of estrogen biosynthesis and function. BMB Rep. 2016, 49, 488–496. [Google Scholar] [CrossRef] [PubMed]
- Ingle, J.N. Estrogen as therapy for breast cancer. Breast Cancer Res. 2002, 4, 133–136. [Google Scholar] [CrossRef][Green Version]
- Kerdivel, G.; Habauzit, D.; Pakdel, F. Assessment and molecular actions of endocrine-disrupting chemicals that interfere with estrogen receptor pathways. Int. J. Endocrinol. 2013, 2013, 501851. [Google Scholar] [CrossRef] [PubMed]
- Fucic, A.; Gamulin, M.; Ferencic, Z.; Katic, J.; von Kauss, M.K.; Bartonova, A.; Merlo, D.F. Environmental exposure to xenoestrogens and oestrogen related cancers: Reproductive system, breast, lung, kidney, pancreas, and brain. Environ. Health 2012, 11, S8. [Google Scholar] [CrossRef] [PubMed]
- Paterni, I.; Granchi, C.; Minutolo, F. Risks and benefits related to alimentary exposure to xenoestrogens. Crit. Rev. Food Sci. Nutr. 2017, 57, 3384–3404. [Google Scholar] [CrossRef]
- Patisaul, H.B.; Jefferson, W. The pros and cons of phytoestrogens. Front. Neuroendocrinol. 2010, 31, 400–419. [Google Scholar] [CrossRef]
- Virk-Baker, M.K.; Nagy, T.R.; Barnes, S. Role of phytoestrogens in cancer therapy. Planta Med. 2010, 76, 1132–1142. [Google Scholar] [CrossRef]
- Torrens-Mas, M.; Roca, P. Phytoestrogens for Cancer Prevention and Treatment. Biology 2020, 9, 427. [Google Scholar] [CrossRef]
- Fuentes, N.; Silveyra, P. Estrogen receptor signaling mechanisms. Adv. Protein Chem. Struct. Biol. 2019, 116, 135–170. [Google Scholar] [PubMed]
- Yaşar, P.; Ayaz, G.; User, S.D.; Güpür, G.; Muyan, M. Molecular mechanism of estrogen-estrogen receptor signaling. Reprod. Med. Biol. 2016, 16, 4–20. [Google Scholar] [CrossRef]
- Márquez, D.C.; Lee, J.; Lin, T.; Pietras, R.J. Epidermal growth factor receptor and tyrosine phosphorylation of estrogen receptor. Endocrine 2001, 16, 73–81. [Google Scholar] [CrossRef]
- Gong, P.; Madak-Erdogan, Z.; Flaws, J.A.; Shapiro, D.J.; Katzenellenbogen, J.A.; Katzenellenbogen, B.S. Estrogen receptor-α and aryl hydrocarbon receptor involvement in the actions of botanical estrogens in target cells. Mol. Cell Endocrinol. 2016, 437, 190–200. [Google Scholar] [CrossRef]
- Huang, Q.; Chen, Q. Mediating Roles of PPARs in the Effects of Environmental Chemicals on Sex Steroids. PPAR Res. 2017, 2017, 3203161. [Google Scholar] [CrossRef]
- Kumari, K.; Adhya, A.K.; Rath, A.K.; Reddy, P.B.; Mishra, S.K. Estrogen-related receptors alpha, beta and gamma expression and function is associated with transcriptional repressor EZH2 in breast carcinoma. BMC Cancer 2018, 18, 690. [Google Scholar] [CrossRef]
- Rothenberger, N.J.; Somasundaram, A.; Stabile, L.P. The Role of the Estrogen Pathway in the Tumor Microenvironment. Int. J. Mol. Sci. 2018, 19, 611. [Google Scholar] [CrossRef]
- Terry, M.B.; Michels, K.B.; Brody, J.G.; Byrne, C.; Chen, S.; Jerry, D.J.; Malecki, K.M.C.; Martin, M.B.; Miller, R.L.; Neuhausen, S.L.; et al. Environmental exposures during windows of susceptibility for breast cancer: A framework for prevention research. Breast Cancer Res. 2019, 21, 96. [Google Scholar] [CrossRef] [PubMed]
- Breast Cancer and the Environment Research Program. Endocrine-Disrupting Chemicals. Available online: https://bcerp.org/health-professionals/endocrine-disrupting-chemicals/ (accessed on 16 May 2021).
- Kuiper, G.G.; Lemmen, J.G.; Carlsson, B.; Safe, S.H.; van der Saag, P.T.; van der Burg, B.; Gustafsson, J.A. Interaction of Estrogenic Chemicals and Phytoestrogens with Estrogen Receptor Beta. Endocrinology 1998, 139, 4252–4263. [Google Scholar] [CrossRef]
- Wetherill, Y.B.; Akingbemi, B.T.; Kanno, J.; McLachlan, J.A.; Nadal, A.; Sonnenschein, C.; Watson, C.S.; Zoeller, R.T.; Belcher, S.M. In Vitro Molecular Mechanisms of Bisphenol a Action. Reprod. Toxicol. 2007, 24, 178–198. [Google Scholar] [CrossRef]
- Lee, H.J.; Chattopadhyay, S.; Gong, E.; Ahn, R.S.; Lee, K. Antiandrogenic Effects of Bisphenol A and Nonylphenol on the Function of Androgen Receptor. Toxicol. Sci. 2003, 75, 40–46. [Google Scholar] [CrossRef] [PubMed]
- Seachrist, D.D.; Bonk, K.W.; Ho, S.; Prins, G.S.; Soto, A.M.; Keri, R.A. A Review of the Carcinogenic Potential of Bisphenol A. Reprod. Toxicol. 2016, 59, 167–182. [Google Scholar] [CrossRef] [PubMed]
- La Merrill, M.A.; Vandenberg, L.N.; Smith, M.T.; Goodson, W.; Browne, P.; Patisual, H.B.; Guyton, K.Z.; Kortenkamp, A.; Cogliano, V.J.; Woodruff, T.J.; et al. Consensus on the Key Characteristics of Endocrine-Disrupting Chemicals as a Basis for Hazard Identification. Nat. Rev. Endocrinol. 2020, 16, 45–57. [Google Scholar] [CrossRef]
- Nicolopoulou-Stamati, P.; Maipas, S.; Kotampasi, C.; Stamatis, P.; Hens, L. Chemical Pesticides and Human Health: The Urgent Need for a New Concept in Agriculture. Front. Public Health 2016, 4, 148. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Zhao, M.; Zhuang, S.; Yang, Y.; Yang, Y.; Liu, W. Low Concentrations of o,p’-DDT Inhibit Gene Expression and Prostaglandin Synthesis by Estrogen Receptor-Independent Mechanism in Rat Ovarian Cells. PLoS ONE 2012, 11, e49916. [Google Scholar] [CrossRef]
- Cohn, B.A.; Cirillo, P.M.; Terry, M.B. DDT and Breast Cancer: Prospective Study of Induction Time and Susceptibility Windows. J. Natl. Cancer Inst. 2019, 111, 803–810. [Google Scholar] [CrossRef] [PubMed]
- Maness, S.C.; McDonnell, D.P.; Gaido, K.W. Inhibition of Androgen Receptor-Dependent Transcriptional Activity by DDT isomers and Methoxychlor in HepG2 Human Heptaoma Cells. Toxicol. Appl. Pharmacol. 1998, 151, 135–142. [Google Scholar] [CrossRef]
- Cariati, F.; D’Uonno, N.; Borrillo, F.; Iervolino, S.; Galdiero, G.; Tomaiuolo, R. Bisphenol a: An emerging threat to male fertility. Reprod. Biol. Endocrinol. 2019, 17, 6. [Google Scholar] [CrossRef]
- Perry, M.J.; Young, H.A.; Grandjean, P.; Halling, J.; Petersen, M.S.; Martenies, S.E.; Karimi, P.; Weihe, P. Sperm Aneuploidy in Faroese Men with Lifetime Exposure to Dichlorodiphenyldichloroethylene (p,p’-DDE) and Polychlorinated Biphenyl (PCB) Pollutants. Environ. Health Perspect. 2016, 124, 951–956. [Google Scholar] [CrossRef]
- Schrader, T.J.; Cooke, G.M. Effects of Aroclors and Individual PCB Congeners on Activation of the Human Androgen Receptor In Vitro. Reprod. Toxicol. 2003, 17, 15–23. [Google Scholar] [CrossRef]
- Connor, K.; Ramamoorthy, K.; Moore, M.; Mustain, M.; Chen, I.; Safe, S.; Zacharewski, T.; Gillesby, B.; Joyeux, A.; Balaguer, P. Hydroxylated Polychlorinated Biphenyls (PCBs) as Estrogens and Antiestrogens: Structure-Activity Relationships. Toxicol. Appl. Pharmacol. 1997, 145, 111–123. [Google Scholar] [CrossRef]
- Breast Cancer Prevention Partners. Polychlorinated Biphenyls (PCBs). Available online: https://www.bcpp.org/resource/polychlorinated-biphenyls/ (accessed on 20 May 2021).
- Hurley, S.; Goldberg, D.; Park, J.; Petreas, M.; Bernstein, L.; Anton-Culver, H.; Neuhausen, S.L.; Nelson, D.O.; Reynolds, P. A Breast Cancer Case-Control Study of Polybrominated Diphenyl Ether (PBDE) Serum Levels among California Women. Environ. Int. 2019, 127, 412–419. [Google Scholar] [CrossRef]
- Cao, L.; Ren, X.; Yang, Y.; Wan, B.; Guo, L.; Chen, D.; Fan, Y. Hydroxylated Polybrominated Biphenyl Ethers Exert Estrogenic Effects via Non-genomic G Protein-Coupled Estrogen Receptor Mediated Pathways. Environ. Health Perspect. 2018, 126, 057005. [Google Scholar] [CrossRef]
- Luthe, G.; Jacobus, J.A.; Robertson, L.W. Receptor Interactions by Polybrominated Diphenyl Ethers Versus Polychlorinated Biphenyls: A Theoretical Structure-Activity Assessment. Environ. Toxicol. Pharmacol. 2008, 25, 202–210. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Liu, X.; Wang, N.; Chen, J.; Chen, Y.; Huang, J.; Su, C.; Xie, F.; Yu, B.; Chen, D. Effects of Decabrominated Diphenyl Ether (PBDE-209) in Regulation of Growth and Apoptosis of Breast, Ovarian, and Cervical Cancer Cells. Environ. Health Perspect. 2012, 120, 541–546. [Google Scholar] [CrossRef] [PubMed]
- Mancini, F.R.; Cano-Sancho, G.; Mohamed, O.; Cervenka, I.; Omichessan, H.; Marchand, P.; Boutron-Ruault, M.; Arveux, P.; Severi, G.; Antignac, P.; et al. Plasma Concentration of Brominated Flame Retardants and Postmenopausal Breast Cancer Risk: A Nested Case-Control Study in the French E3N Cohort. Environ. Health 2020, 19, 54. [Google Scholar] [CrossRef]
- Hoover, R.N.; Hyer, M.; Pfeiffer, R.M.; Adam, E.; Bond, B.; Cheville, A.L.; Colton, T.; Hartge, P.; Hatch, E.E.; Herbst, A.L.; et al. Adverse health outcomes in women exposed in utero to diethylstilbestrol. N. Engl. J. Med. 2011, 365, 1304–1314. [Google Scholar] [CrossRef] [PubMed]
- Adam, A.H.B.; de Haan, L.H.J.; Estruch, I.M.; Hooiveld, G.J.E.J.; Louisse, J.; Rietjens, I.M.C.M. Estrogen receptor alpha (ERα)-mediated coregulator binding and gene expression discriminates the toxic ERα agonist diethylstilbestrol (DES) from the endogenous ERα agonist 17β-estradiol (E2). Cell Biol. Toxicol. 2020, 36, 417–435. [Google Scholar] [CrossRef]
- Tang, Z.; Liu, Z.H.; Wang, H.; Dang, Z.; Liu, Y. A review of 17α-ethynylestradiol (EE2) in surface water across 32 countries: Sources, concentrations, and potential estrogenic effects. J. Environ. Manag. 2021, 292, 112804. [Google Scholar] [CrossRef] [PubMed]
- Ahern, T.P.; Broe, A.; Lash, T.L.; Cronin-Fenton, D.P.; Ulrichsen, S.P.; Christiansen, P.M.; Cole, B.F.; Tamimi, R.M.; Sorensen, H.T.; Damkier, P. Phthalate Exposure and Breast Cancer Incidence: A Danish Nationwide Cohort Study. J. Clin. Oncol. 2019, 37, 1800–1809. [Google Scholar] [CrossRef] [PubMed]
- López-Carrillo, L.; Hernández-Ramírez, R.U.; Calafat, A.M.; Torres-Sánchez, L.; Galván-Portillo, M.; Needham, L.L.; Ruiz-Ramos, R.; Cebrián, M.E. Exposure to Phthalates and Breast Cancer Risk in Northern Mexico. Environ. Health Perspect. 2010, 118, 539–544. [Google Scholar] [CrossRef]
- Crobeddu, B.; Ferraris, E.; Kolasa, E.; Plante, I. Di(2-ethylhexyl) Phthalate (DEHP) Increases Proliferation of Epithelial Breast Cancer Cells Through Progesterone Receptor Dysregulation. Environ. Res. 2019, 173, 165–173. [Google Scholar] [CrossRef]
- Gan, W.; Zhou, M.; Xiang, Z.; Han, X.; Li, D. Combined Effects of Nonylphenol and Bisphenol A on the Human Prostate Epithelial Cell Line RWPE-1. Int. J. Environ. Res. Public Health 2015, 12, 4141–4155. [Google Scholar] [CrossRef] [PubMed]
- Forte, M.; Di Lorenzo, M.; Carrizzo, A.; Valiante, S.; Vecchione, C.; Laforgia, V.; De Falco, M. Nonylphenol Effects on Human Prostate non Tumorigenic Cells. Toxicology 2016, 357–358, 21–32. [Google Scholar] [CrossRef]
- Golden, R.; Gandy, J.; Vollmer, G. A Review of the Endocrine Activity of Parabens and Implications for Potential Risks to Human Health. Crit. Rev. Toxicol. 2005, 35, 435–458. [Google Scholar] [CrossRef] [PubMed]
- Varinska, L.; Gal, P.; Mojzisova, G.; Mirossay, L.; Majzis, J.; Srivastava, S.K. Soy and Breast Cancer: Focus on Angiogenesis. Int. J. Mol. Sci. 2015, 16, 11728–11749. [Google Scholar] [CrossRef]
- Miodini, P.; Fioravanti, L.; Di Fronzo, G.; Cappelletti, V. The Two Phyto-oestrogens Genistein and Quercetin Exert Different Effects on Oestrogen Receptor Function. Br. J. Cancer 1999, 80, 1150–1155. [Google Scholar] [CrossRef] [PubMed]
- Obiorah, I.E.; Fan, P.; Jordan, V.C. Breast Cancer Cell Apoptosis with Phytoestrogens is Dependent on an Estrogen-deprived State. Cancer Prev. Res. (Phila) 2014, 7, 939–949. [Google Scholar] [CrossRef]
- Dandawate, P.R.; Subramaniam, D.; Jensen, R.A.; Anant, S. Targeting Cancer Stem Cells and Signaling Pathways by Phytochemicals: Novel Approach for Breast Cancer Therapy. Semin. Cancer Biol. 2016, 40–41, 192–208. [Google Scholar] [CrossRef]
- Liu, R.; Yu, X.; Chen, X.; Zhong, H.; Liang, C.; Xu, X.; Xu, W.; Cheng, Y.; Wang, W.; Yu, L.; et al. Individual Factors Define the Overall Effects of Dietary Genistein Exposure on Breast Cancer Patients. Nutr. Res. 2019, 67, 1–16. [Google Scholar] [CrossRef]
- Chan, K.K.L.; Siu, M.K.Y.; Jiang, Y.; Wang, J.; Leung, T.H.Y.; Ngan, H.Y.S. Estrogen Receptor Modulators Genistein, Daidzein and ERB-041 Inhibit Cell Migration, Invasion, Proliferation and Sphere Formation via Modulation of FAK and PI3K/AKT Signaling in Ovarian Cancer. Cancer Cell Int. 2018, 18, 65. [Google Scholar] [CrossRef]
- Yu, M.; Qi, H.; Gao, X. Daidzein Promotes Milk Synthesis and Proliferation of Mammary Epithelial Cells via the Estrogen Receptor-dependent NF-B1 Activation. Anim. Biotechnol. 2020, 1–10. [Google Scholar]
- Staar, S.; Richter, D.; Makovitzky, J.; Briese, V.; Bergemann, C. Stimulation of Endometrial Glandular Cells with Genistein and Daidzein and Their Effects on ERα- and ERβ-mRNA and Protein Expresion. Anticancer Res. 2005, 25, 1713–1718. [Google Scholar] [PubMed]
- Pendleton, J.M.; Tan, W.W.; Anai, S.; Chang, M.; Hou, W.; Shiverick, K.T.; Rosser, C.J. Phase II Trial of Isoflavone in Prostate-Specific Antigen Recurrent Prostate Cancer after Previous Local Therapy. BMC Cancer 2008, 8, 1–10. [Google Scholar] [CrossRef]
- Couture, R.; Mora, N.; Al Bittar, S.; Najih, M.; Touaibia, M.; Martin, L.J. Luteolin Modulates Gene Expression Related to Steroidogenesis, Apoptosis, and Stress Response in Rat LC540 Tumor Leydig Cells. Cell Biol. Toxicol. 2020, 36, 31–49. [Google Scholar] [CrossRef] [PubMed]
- Nordeen, S.K.; Bona, B.J.; Jones, D.N.; Lambert, J.R.; Jackson, T.A. Endocrine Disrupting Activities of the Flavonoid Nutraceuticals Luteolin and Quercetin. Horm. Cancer 2013, 4, 293–300. [Google Scholar] [CrossRef] [PubMed]
- Maggiolini, M.; Bonofiglio, D.; Marsico, S.; Panno, M.L.; Cenni, B.; Picard, D.; Andó, S. Estrogen Receptor Alpha Mediates the Proliferation but not the Cytotoxic Dose-dependent Effects of Major Phytoestrogens on Human Breast Cancer Cells. Mol. Pharmacol. 2001, 60, 595–602. [Google Scholar]
- Wei, L.; Liu, T.; Wang, H.; Hong, H.; Yu, A.L.; Feng, H.; Chang, W. Hsp27 Participates in the Maintenance of Breast Cancer Stem Cells Through Regulation of Epithelial-mesenchymal Transition and Nuclear Factor-B. Breast Cancer Res. 2011, 13, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Selvakumar, P.; Badgeley, A.; Murphy, P.; Anwar, H.; Sharma, U.; Lawrence, K.; Lakshmikuttyamma, A. Flavonoids and Other Polyphenols Act as Epigenetic Modifiers in Breast Cancer. Nutrients 2020, 12, 761. [Google Scholar] [CrossRef]
- Xu, Z.; Zhao, D.; Zheng, X.; Huang, B.; Xia, X.; Pan, X. Quercetin Exerts Bidirectional Regulation Effects on the Efficacy of Tamoxifen in Estrogen Receptor-positive Breast Cancer Therapy: An In Vitro Study. Environ. Toxicol. 2020, 35, 1179–1193. [Google Scholar] [CrossRef] [PubMed]
- Hashemzaei, M.; Far, A.D.; Yari, A.; Heravi, R.E.; Tabrizian, K.; Taghdisi, S.M.; Sadegh, S.E.; Tsarouhas, K.; Kouretas, D.; Tzanakakis, G.; et al. Anticancer and Apoptosis-inducing Effects of Quercetin In Vitro and In Vivo. Oncol. Rep. 2017, 38, 819–828. [Google Scholar] [CrossRef]
- Shankar, E.; Goel, A.; Gupta, K.; Gupta, S. Plant Flavone Apigenin: An Emerging Anticancer Agent. Curr. Pharmacol. Rep. 2017, 3, 423–446. [Google Scholar] [CrossRef]
- Dean, M.; Murphy, B.T.; Burdette, J.E. Phytosteroids Beyond Estrogens: Regulators of Reproductive and Endocrine Function in Natural Products. Mol. Cell Endocrinol. 2017, 442, 98–105. [Google Scholar] [CrossRef] [PubMed]
- Shukla, S.; Gupta, S. Apigenin: A Promising Molecule for Cancer Prevention. Pharm. Res. 2010, 27, 962–978. [Google Scholar] [CrossRef]
- Long, X.; Fan, M.; Bigsby, R.M.; Nephew, K.P. Apigenin Inhibits Antiestrogen-resisitent Breast Cancer Cell Growth through Estrogen Receptor-α-dependent and -independent Mechanisms. Mol. Cancer Ther. 2008, 7, 2096–2108. [Google Scholar] [CrossRef] [PubMed]
- Sirotkin, A.; Záhoranska, Z.; Tarko, A.; Fabova, Z.; Alwasel, S.; Harrath, A.H. Plant Polyphenols Can Directly Affect Ovarian Cell Functions and Modify Toluene Effects. J. Anim. Physiol. Anim. Nutr. 2020, 105, 80–89. [Google Scholar] [CrossRef] [PubMed]
- Barakat, R.; Park, C.J.; Perez, P.A.; Chiu, K.; Ko, C. Female Antiestrogens. Encycl. Reprod. 2018, 2, 740–747. [Google Scholar]
- Bowers, J.L.; Tyulmenkov, V.V.; Jernigan, S.C.; Klinge, C.M. Resveratrol Acts as a Mixed Agonist/Antagonist for Estrogen Receptors. Endocrinology 2000, 141, 3657–3667. [Google Scholar] [CrossRef] [PubMed]
- Carter, L.G.; D’Orazio, J.A.; Pearson, K.J. Resveratrol and Cancer: Focus on In Vivo Evidence. Endocr. Relat. Cancer 2014, 21, R209–R225. [Google Scholar] [CrossRef]
- Harada, N.; Murata, Y.; Yamaji, R.; Miura, T.; Inui, H.; Nakano, Y. Resvertrol Down-regulates the Androgen Receptor at the Post-translational Level in Prostate Cancer Cells. J. Nutr. Sci. Vitaminol. (Tokyo) 2007, 53, 556–560. [Google Scholar] [CrossRef]
- Wang, Y.; Romigh, T.; He, X.; Orloff, M.S.; Silverman, R.H.; Heston, W.D.; Eng, C. Resveratrol Regulates the PTEN/AKT Pathway Through Androgen Receptor-dependent and -independent Mechanisms in Prostate Cancer Cell Lines. Hum. Mol. Genet. 2010, 19, 4319–4329. [Google Scholar] [CrossRef] [PubMed]
- Waite, K.A.; Sinden, M.R.; Eng, C. Phytoestrogen Exposure Elevates PTEN Levels. Hum. Mol. Genet. 2005, 14, 1457–1463. [Google Scholar] [CrossRef] [PubMed]
- Ci, Y.; Zhang, Y.; Liu, Y.; Lu, S.; Cao, J.; Li, H.; Zhang, J.; Huang, Z.; Zhu, X.; Gao, J.; et al. Myricetin Suppresses Breast Cancer Metastasis Through Down-regulating the Activity of Matrix Metalloproteinase (MMP)-2/9. Phytother. Res. 2018, 32, 1373–1381. [Google Scholar] [CrossRef] [PubMed]
- McCarty, M.F.; Assanga, S.I.; Lujan, L.L. Flavones and Flavonols May Have Clinical Potential as CK2 Inhibitors in Cancer Therapy. Med. Hypotheses 2020, 141, 109723. [Google Scholar] [CrossRef]
- Maggiolini, M.; Recchia, A.G.; Bonofiglio, D.; Catalano, S.; Vivacqua, A.; Carpino, A.; Rago, V.; Rossi, R.; Andò, A. The Red Wine Phenolics Piceatannol and Myricetin Act as Agonists for Estrogen Receptor Alpha in Human Breast Cancer Cells. J. Mol. Endocrinol. 2005, 35, 269–281. [Google Scholar] [CrossRef]
- Xu, R.; Zhang, Y.; Ye, X.; Xue, S.; Shi, J.; Pan, J.; Chen, Q. Inhibition Effects and Induction of Apoptosis of Flavonoids on the Prostate Cancer Cell Line PC-3 In Vitro. Food Chem. 2013, 138, 48–53. [Google Scholar] [CrossRef] [PubMed]
- Semwal, D.K.; Semwal, R.B.; Combrinck, S.; Viljoen, A. Myricetin: A Dietary Molecule with Diverse Biological Activities. Nutrients 2016, 8, 90. [Google Scholar] [CrossRef] [PubMed]
- Sajedi, N.; Homayoun, M.; Mohammadi, F.; Soleimani, M. Myricetin Exerts its Apoptotic Effects on MCF-7 Breast Cancer Cells through Evoing the BRCA1-GADD45 Pathway. Asian Pac. J. Cancer Prev. 2020, 21, 3461–3468. [Google Scholar] [CrossRef] [PubMed]
- Chen, A.Y.; Chen, Y.C. A Review of the Dietary Flavnoid, Kaempferol on Human Health and Cancer Chemoprevention. Food Chem. 2013, 138, 2099–2107. [Google Scholar] [CrossRef]
- Imran, M.; Salehi, B.; Sharifi-Rad, J.; Gondal, T.A.; Saeed, F.; Imran, A.; Shahbaz, M.; Fokou, P.V.T.; Arshad, M.U.; Khan, H.; et al. Kaempferol: A Key Emphasis to its Anticancer Potential. Molecules 2019, 24, 2277. [Google Scholar] [CrossRef]
- Resende, F.A.; de Oliveira, A.P.S.; de Camargo, M.S.; Vilegas, W.; Varanda, E.A. Evaulation of Estrogenic Potential of Flavonoids Using a Recombinant Yeast Strain an dMCF7/BUS Cell Proliferation Assay. PLoS ONE 2013, 8, e74881. [Google Scholar] [CrossRef] [PubMed]
- Zeng, J.; Xu, H.; Fan, P.; Xie, J.; He, J.; Yu, J.; Gu, X.; Zhang, C. Kaempferol Blocks Neutrophil Extracellular Traps Formation and Reduces Tumour Metastasis by Inhibiting ROS-PAD4 Pathway. J. Cell Mol. Med. 2020, 24, 7590–7599. [Google Scholar] [CrossRef]
- Wang, X.; Yang, Y.; An, Y.; Fang, G. The Mechanism of Anticancer Action and Potential Clinical Use of Kaempferol in the Treatment of Breast Cancer. Biomed. Pharmacother. 2019, 117, 109086. [Google Scholar] [CrossRef]
- Pham, H.N.T.; Sakoff, J.A.; Vuong, Q.V.; Bowyer, M.C.; Scarlett, C.J. Comparative Cytotoxic Activity Between Kaempferol and Gallic Acid Against Various Cancer Cell Lines. Data Brief 2018, 21, 1033–1036. [Google Scholar] [CrossRef]
- Seelinger, G.; Merfort, I.; Wolfle, U.; Schempp, C.M. Anti-carcinogenic Effects of the Flavonoid Luteolin. Molecules 2008, 13, 2628–2651. [Google Scholar] [CrossRef] [PubMed]
- Bachmeier, B.E.; Mirisola, V.; Romeo, F.; Generoso, L.; Esposito, A.; Dell’eva, R.; Blengio, F.; Killian, P.H.; Albini, A.; Ufeffer, U. Reference Profile Correlation Reveals Estrogen-like Transcriptional Activity of Curcumin. Cell Physiol. Biochem. 2010, 26, 471–482. [Google Scholar] [CrossRef]
- Hallman, K.; Aleck, K.; Dwyer, B.; Lloyd, V.; Quigley, M.; Sitto, N.; Siebert, A.E.; Dinda, S. The Effects of Tumeric (Curcumin) on Tumor Suppressor Protein (p53) and Estrogen Receptor in Breast Cancer Cells. Breast Cancer 2017, 9, 153–161. [Google Scholar] [PubMed]
- Mohajeri, M.; Vianconi, V.; Ávila-Rodriguez, M.F.; Barreto, G.E.; Jamialahmadi, T.; Pirro, M.; Sahebkar, A. Curcumin: A Phytochemical Modulator of Estrogens and Androgens in Tumors of the Reproductive System. Pharmacol. Res. 2020, 156, 104765. [Google Scholar] [CrossRef]
- Ávila-Gálvez, M.A.; González-Sarrias, A.; Martínez-Díaz, F.; Abellán, B.; Martínez-Torrano, A.J.; Fernández-López, A.J.; Giménez-Bastida, J.A.; Espín, J.C. Disposition of Dietary Polypenols in Breast Cancer Patients’ Tumors, and Their Associated Anticancer Activity: The Particular Case of Curcumin. Mol. Nutr. Food Res. 2021, 65, 2100163. [Google Scholar] [CrossRef] [PubMed]
- Dorai, T.; Dutcher, J.P.; Dempster, D.W.; Wiernik, P.H. Therapeutic Potential of Curcumin in Prostate Cancer-V: Interference with the Osteomimetic Properties of Hormone Refractory C4–2B Prostate Cancer Cells. Prostate 2004, 60, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Zafar, A.; Singh, S.; Satija, Y.K.; Saluja, D.; Naseem, I. Deciphering the Molecular Mechanism Underlying Anticancer Activity of Coumestrol in Triple-negative Breast Cancer Cells. Toxicol. Vitr. 2018, 46, 19–28. [Google Scholar] [CrossRef] [PubMed]
- Zafar, A.; Singh, S.; Satija, Y.K.; Naseem, I. Cytotoxic Activity of Soy Phytoestrogen Coumetrol Against Human Breast Cancer MCF-7 Cells: Insights into the Molecular Mechanism. Food Chem. Toxicol. 2017, 99, 149–161. [Google Scholar] [CrossRef] [PubMed]
- Lim, W.; Jeong, M.; Bazar, F.W.; Song, G. Coumestrol Inhibits Proliferation and Migration of Prostate Cancer Cells by Regulating AKT, ERK1/2, and JNK MAPK Cell Signaling Cascades. J. Cell Physiol. 2017, 232, 862–871. [Google Scholar] [CrossRef]
- Hu, Y.; Wu, C.; Lai, J.; Tsai, Y. Detection of a Negative Correlation Between Prescription of Chinese Herbal Products Containing Coumestrol, Genistein or Daidzein and Risk of Subsequent Endometrial Cancer Among Tamoxifen-treated Female Breast Cancer Survivors in Taiwan between 1998 and 2008: A Population-based Study. J. Ethnopharmacol. 2015, 169, 356–362. [Google Scholar] [PubMed]
- Gea, M.; Toso, A.; Schilirò, T. Estrogenic activity of biological samples as a biomarker. Sci. Total Environ. 2020, 740, 140050. [Google Scholar] [CrossRef]
- Wang, L.H.; Chen, L.R.; Chen, K.H. In vitro and vivo identification, metabolism and action of xenoestrogens: An overview. Int. J. Mol. Sci. 2021, 22, 4013. [Google Scholar] [CrossRef]
- Lin, H.; Li, H.; Lu, G.; Chen, Z.; Sun, W.; Shi, Y.; Fu, Z.; Huang, B.; Zhu, X.; Lu, W.; et al. Low dose of bisphenol a modulates ovarian cancer gene expression profile and promotes epithelial to mesenchymal transition via canonical wnt pathway. Toxicol. Sci. 2018, 164, 527–538. [Google Scholar]
- Chen, Z.J.; Zhang, K.S.; Ge, L.C.; Liu, H.; Chen, L.K.; Du, J.; Wang, H.S. Signals involved in the effects of bisphenol A (BPA) on proliferation and motility of Leydig cells: A comparative proteomic analysis. Toxicol. Res. 2016, 5, 1573–1584. [Google Scholar] [CrossRef]
- Warth, B.; Raffeiner, P.; Granados, A.; Huan, T.; Fang, M.; Forsberg, E.M.; Benton, H.P.; Goetz, L.; Johnson, C.H.; Siuzdak, G. Metabolomics Reveals that Dietary Xenoestrogens Alter Cellular Metabolism Induced by Palbociclib/Letrozole Combination Cancer Therapy. Cell Chem. Biol. 2018, 25, 291–300.e3. [Google Scholar] [CrossRef] [PubMed]
- Rudel, R.A.; Attfield, K.R.; Schifano, J.N.; Brody, J.G. Chemicals causing mammary gland tumors in animals signal new directions for epidemiology, chemicals testing, and risk assessment for breast cancer prevention. Cancer 2007, 109, 2635–2666. [Google Scholar] [CrossRef]
- Soto, A.M.; Sonnenschein, C.; Chung, K.L.; Fernandez, M.F.; Olea, N.; Olea Serrano, F. The E-SCREEN assay as a tool to identify estrogens: An update on estrogenic environmental pollutants. Environ. Health Perspect. 1995, 103, 113–122. [Google Scholar]
- Villalobos, M.; Olea, N.; Brotons, J.A.; Olea-Serrano, M.F.; de Almodovar, J.M.R.; Pedraza, V. The E-screen assay: A comparison of different MCF7 cell stocks. Environ. Health Perspect. 1995, 103, 844–850. [Google Scholar] [CrossRef]
- Sonneveld, E.; Jansen, H.J.; Riteco, J.A.C.; Brouwer, A.; van der Burg, B. Development of androgen- and estrogen-responsive bioassays members of a panel of human cell line-based highly selective steroid-responsive bioassays. Toxicol. Sci. 2005, 83, 136–148. [Google Scholar] [CrossRef] [PubMed]
- Bonefeld-Jorgensen, E.C.; Grünfeld, H.T.; Gjermandsen, I.M. Effect of pesticides on estrogen receptor transactivation in vitro: A comparison of stable transfected MVLN and transient transfected MCF-7 cells. Mol. Cell. Endocrinol. 2005, 244, 20–30. [Google Scholar] [CrossRef]
- Wong, S.P.; Li, J.; Shen, P.; Gong, Y.; Yap, P.S.; Yong, L.E. Ultrasensitive cell-based bioassay for the measurement of global estrogenic activity of flavonoid mixtures revealing additive, restrictive, and enhanced actions in binary and higher order combinations. Assay Drug Dev. Technol. 2007, 5, 355–362. [Google Scholar] [CrossRef]
- Lui, K.; Tamura, T.; Mori, T.; Zhou, D.; Chen, S. MCF-7aro/ERE, a novel cell line for rapid screening of aromatase inhibitors, ERα ligands and ERRα ligands. Biochem. Pharmacol. 2008, 76, 208–215. [Google Scholar] [CrossRef] [PubMed]
- Kanaya, N.; Nguyen, D.M.; Lu, H.; Wang, Y.Z.; Hsin, L.Y.; Petreas, M.; Nelson, D.; Guo, W.; Reynolds, P.; Synold, T.; et al. AroER tri-screen™ is a novel functional assay to estimate both estrogenic and estrogen precursor activity of chemicals or biological specimens. Breast Cancer Res. Treat. 2015, 151, 335–345. [Google Scholar] [CrossRef]
- Chen, S.; Hsieh, J.H.; Huang, R.; Sakamuru, S.; Hsin, L.Y.; Xia, M.; Shockley, K.R.; Auerbach, S.; Kanaya, N.; Lu, H.; et al. Cell-based high-throughput screening for aromatase inhibitors in the Tox21 10K library. Toxicol. Sci. 2015, 147, 446–457. [Google Scholar] [CrossRef] [PubMed]
- Pompili, L.; Porru, M.; Caruso, C.; Biroccio, A.; Leonetti, C. Patient-derived xenografts: A relevant preclinical model for drug development. J. Exp. Clin. Cancer Res. 2016, 35, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Hidalgo, M.; Amant, F.; Biankin, A.V.; Budinská, E.; Byrne, A.T.; Caldas, C.; Clarke, R.B.; de Jong, S.; Jonkers, J.; Mælandsmo, G.M.; et al. Patient-derived Xenograft models: An emerging platform for translational cancer research. Cancer Discov. 2014, 4, 998–1013. [Google Scholar] [CrossRef]
- Fichtner, I.; Rolff, J.; Soong, R.; Hoffmann, J.; Hammer, S.; Sommer, A.; Becker, M.; Merk, J. Establishment of patient-derived non-small cell lung cancer xenografts as models for the identification of predictive biomarkers. Clin. Cancer Res. 2008, 14, 6456–6468. [Google Scholar] [CrossRef]
- Derose, Y.S.; Wang, G.; Lin, Y.C.; Bernard, P.S.; Buys, S.S.; Ebbert, M.T.W.; Factor, R.; Matsen, C.; Milash, B.A.; Nelson, E.; et al. Tumor grafts derived from women with breast cancer authentically reflect tumor pathology, growth, metastasis and disease outcomes. Nat. Med. 2011, 17, 1514–1520. [Google Scholar] [CrossRef]
- Julien, S.; Merino-Trigo, A.; Lacroix, L.; Pocard, M.; Goeŕé, D.; Mariani, P.; Landron, S.; Bigot, L.; Nemati, F.; Dartigues, P.; et al. Characterization of a large panel of patient-derived tumor xenografts representing the clinical heterogeneity of human colorectal cancer. Clin. Cancer Res. 2012, 18, 5314–5328. [Google Scholar] [CrossRef]
- Dong, X.; Guan, J.; English, J.C.; Flint, J.; Yee, J.; Evans, K.; Murray, N.; MacAulay, C.; Ng, R.T.; Gout, P.W.; et al. Patient-derived first generation xenografts of non-small cell lung cancers: Promising tools for predicting drug responses for personalized chemotherapy. Clin. Cancer Res. 2010, 16, 1442–1451. [Google Scholar] [CrossRef]
- Bertotti, A.; Migliardi, G.; Galimi, F.; Sassi, F.; Torti, D.; Isella, C.; Corà, D.; di Nicolantonio, F.; Buscarino, M.; Petti, C.; et al. A molecularly annotated platform of patient-derived xenografts (“xenopatients”) identifies HER2 as an effective therapeutic target in cetuximab-resistant colorectal cancer. Cancer Discov. 2011, 1, 508–523. [Google Scholar] [CrossRef]
- Zhang, X.; Claerhout, S.; Prat, A.; Dobrolecki, L.E.; Petrovic, I.; Lai, Q.; Landis, M.D.; Wiechmann, L.; Schiff, R.; Giuliano, M.; et al. A renewable tissue resource of phenotypically stable, biologically and ethnically diverse, patient-derived human breast cancer xenograft models. Cancer Res. 2013, 73, 4885–4897. [Google Scholar] [CrossRef]
- Lillo, M.A.; Nichols, C.; Perry, C.; Runke, S.; Krutilina, R.; Seagroves, T.N.; Miranda-Carboni, G.A.; Krum, S.A. Methylparaben stimulates tumor initiating cells in ER+ breast cancer models. J. Appl. Toxicol. 2017, 37, 417–425. [Google Scholar] [CrossRef] [PubMed]
- Kanaya, N.; Bernal, L.; Chang, G.; Yamamoto, T.; Nguyen, D.; Wang, Y.Z.; Park, J.S.; Warden, C.; Wang, J.; Wu, X.; et al. Molecular mechanisms of polybrominated diphenyl ethers (BDE-47, BDE-100, and BDE-153) in human breast cancer cells and patient-derived xenografts. Toxicol. Sci. 2019, 169, 380–398. [Google Scholar] [CrossRef]
- Nakamura, H.; Wang, Y.; Kurita, T.; Adomat, H.; Cunha, G.R.; Wang, Y. Genistein increases epidermal growth factor receptor signaling and promotes tumor progression in advanced human prostate cancer. PLoS ONE 2011, 6, e20034. [Google Scholar] [CrossRef]
- Nakamura, H.; Wang, Y.; Xue, H.; Romanish, M.T.; Mager, D.L.; Helgason, C.D.; Wang, Y. Genistein versus ICI 182, 780: An ally or enemy in metastatic progression of prostate cancer. Prostate 2013, 73, 1747–1760. [Google Scholar] [CrossRef] [PubMed]
- Kimura, T.; Kiyota, H.; Nakata, D.; Masaki, T.; Kusaka, M.; Egawa, S. A novel androgen-dependent prostate cancer xenograft model derived from skin metastasis of a Japanese patient. Prostate 2009, 69, 1660–1667. [Google Scholar] [CrossRef]
- Mesmar, F.; Dai, B.; Ibrahim, A.; Hases, L.; Jafferali, M.H.; Augustine, J.J.; DiLorenzo, S.; Kang, Y.; Zhao, Y.; Wang, J.; et al. Clinical candidate and genistein analogue AXP107-11 has chemoenhancing functions in pancreatic adenocarcinoma through G protein-coupled estrogen receptor signaling. Cancer Med. 2019, 8, 7705–7719. [Google Scholar] [CrossRef]
- Mendes, N.; Carvalho, P.D.; Martins, F.; Mendonça, S.; Malheiro, A.R.; Ribeiro, A.; Carvalho, J.; Velho, S. Animal Models to Study Cancer and Its Microenvironment. In Tumor Microenvironment: The Main Driver of Metabolic Adaptation; Serpa, J., Ed.; Springer International Publishing: Cham, Switzerland, 2020; pp. 389–401. ISBN 978-3-030-34025-4. [Google Scholar]
- Day, C.P.; Merlino, G.; Van Dyke, T. Preclinical Mouse Cancer Models: A Maze of Opportunities and Challenges. Cell 2015, 163, 39–53. [Google Scholar] [CrossRef] [PubMed]
- Cohen, S.M. The relevance of experimental carcinogenicity studies to human safety. Curr. Opin. Toxicol. 2017, 3, 6–11. [Google Scholar] [CrossRef]
- Couteau, C.; Coiffard, L. Regulation no 1223/2009 on cosmetic products. Nouv. Dermatologiques 2010, 29, 217–221. [Google Scholar]
- Regev, A.; Teichmann, S.A.; Lander, E.S.; Amit, I.; Benoist, C.; Birney, E.; Bodenmiller, B.; Campbell, P.; Carninci, P.; Clatworthy, M.; et al. The human cell atlas. eLife 2017, 6, 1–30. [Google Scholar] [CrossRef]
- Schaum, N.; Karkanias, J.; Neff, N.F.; May, A.P.; Quake, S.R.; Wyss-Coray, T.; Darmanis, S.; Batson, J.; Botvinnik, O.; Chen, M.B.; et al. Single-cell transcriptomics of 20 mouse organs creates a Tabula Muris. Nature 2018, 562, 367–372. [Google Scholar]
- Kanaya, N.; Chang, G.; Wu, X.; Saeki, K.; Bernal, L.; Shim, H.J.; Wang, J.; Warden, C.; Yamamoto, T.; Li, J.; et al. Single-cell RNA-sequencing analysis of estrogen- and endocrine-disrupting chemical-induced reorganization of mouse mammary gland. Commun. Biol. 2019, 2, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Saeki, K.; Chang, G.; Kanaya, N.; Wu, X.; Wang, J.; Bernal, L.; Ha, D.; Neuhausen, S.L.; Chen, S. Mammary cell gene expression atlas links epithelial cell remodeling events to breast carcinogenesis. Commun. Biol. 2021, 4, 660. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.-J.; Tian, Y.; Li, M.-H.; Feng, Y.-Q.; Kong, L.; Zhang, F.-L.; Shen, W. Single-cell transcriptome dissection of the toxic impact of Di (2-ethylhexyl) phthalate on primordial follicle assembly. Theranostics 2021, 11, 4992–5009. [Google Scholar] [CrossRef] [PubMed]
- Chang, G.; Saeki, K.; Mori, H.; Chen, S. Environmental carcinogenesis at the single-cell level. Cancer Epidemiol. Biomark. Prev. 2020, 29, 1880–1886. [Google Scholar] [CrossRef] [PubMed]
- Stark, R.; Grzelak, M.; Hadfield, J. RNA sequencing: The teenage years. Nat. Rev. Genet. 2019, 20, 631–656. [Google Scholar] [CrossRef]
- Haque, A.; Engel, J.; Teichmann, S.A.; Lönnberg, T. A practical guide to single-cell RNA-sequencing for biomedical research and clinical applications. Genome Med. 2017, 9, 1–12. [Google Scholar] [CrossRef]
- Hammond, G.L. Diverse roles for sex hormone-binding globulin in reproduction. Biol. Reprod. 2011, 85, 431–441. [Google Scholar] [CrossRef]
- Déchaud, H.; Ravard, C.; Claustrat, F.; de la Perrière, A.B.; Pugeat, M. Xenoestrogen interaction with human sex hormone-binding globulin (hSHBG). Steroids 1999, 64, 328–334. [Google Scholar] [CrossRef]
- Sheikh, I.A.; Tayubi, I.A.; Ahmad, E.; Ganaie, M.A.; Bajouh, O.S.; AlBasri, S.F.; Abdulkarim, I.M.J.; Beg, M.A. Computational insights into the molecular interactions of environmental xenoestrogens 4-tert-octylphenol, 4-nonylphenol, bisphenol A (BPA), and BPA metabolite, 4-methyl-2, 4-bis (4-hydroxyphenyl) pent-1-ene (MBP) with human sex hormone-binding globulin. Ecotoxicol. Environ. Saf. 2017, 135, 284–291. [Google Scholar] [CrossRef]
- Zhou, W.; Liu, J.; Liao, L.; Han, S.; Liu, J. Effect of bisphenol A on steroid hormone production in rat ovarian theca-interstitial and granulosa cells. Mol. Cell Endocrinol. 2008, 283, 12–18. [Google Scholar] [CrossRef] [PubMed]
- Chen, S. Modulation of aromatase activity and expression by environmental chemicals. Front. Biosci. 2002, 7, 1712–1719. [Google Scholar] [CrossRef]
- Pino, A.M.; Valladares, L.E.; Palma, M.A.; Mancilla, A.M.; Yáñez, M.; Albala, C. Dietary isoflavones affect sex hormone-binding globulin levels in postmenopausal women. J. Clin. Endocrinol. Metab. 2000, 85, 2797–2800. [Google Scholar] [CrossRef] [PubMed]
- Low, Y.L.; Taylor, J.I.; Grace, P.B.; Dowsett, M.; Scollen, S.; Dunning, A.M.; Mulligan, A.A.; Welch, A.A.; Luben, R.N.; Khaw, K.T.; et al. Phytoestrogen exposure correlation with plasma estradiol in postmenopausal women in European Prospective Investigation of Cancer and Nutrition-Norfolk may involve diet-gene interactions. Cancer Epidemiol. Biomark. Prev. 2005, 14, 213–220. [Google Scholar]
- Low, Y.L.; Dunning, A.M.; Dowsett, M.; Folkerd, E.; Doody, D.; Taylor, J.; Bhaniani, A.; Luben, R.; Khaw, K.; Wareham, N.J.; et al. Phytoestrogen exposure is associated with circulating sex hormone levels in postmenopausal women and interact with ESR1 and NR1I2 gene variants. Cancer Epidemiol. Biomark. Prev. 2007, 16, 1009–1016. [Google Scholar] [CrossRef] [PubMed]
- van Duursen, M.B.M. Modulation of estrogen synthesis and metabolism by phytoestrogens in vitro and the implications for women’s health. Toxicol. Res. 2017, 6, 772–794. [Google Scholar] [CrossRef]
- Deluca, D.; Krazeisen, A.; Breitling, R.; Prehn, C.; Möller, G.; Adamski, J. Inhibition of 17beta-hydroxysteroid dehydrogenases by phytoestrogens: Comparison with other steroid metabolizing enzymes. J. Steroid. Biochem. Mol. Biol. 2005, 93, 285–292. [Google Scholar] [CrossRef]
- Kao, Y.C.; Zhou, C.; Sherman, M.; Laughton, C.A.; Chen, S. Molecular basis of the inhibition of human aromatase (estrogen synthetase) by flavone and isoflavone phytoestrogens: A site-directed mutagenesis study. Environ. Health Perspect. 1998, 106, 85–92. [Google Scholar] [CrossRef]
- Chen, S.; Oh, S.R.; Phung, S.; Hur, G.; Ye, J.J.; Kwok, S.L.; Shrode, G.E.; Belury, M.; Adams, L.S.; Williams, D. Anti-aromatase activity of phytochemicals in white button mushrooms (Agaricus bisporus). Cancer Res. 2006, 66, 12026–12034. [Google Scholar] [CrossRef]
- Wang, Y.; Gho, W.M.; Chan, F.; Chen, S.; Leung, L.K. The red clover (Trifolium pratense) isoflavone biochanin A inhibits aromatase activity and expression. Br. J. Nutr. 2008, 99, 303–310. [Google Scholar] [CrossRef]
- Kijima, I.; Phung, S.; Hur, G.; Kwok, S.L.; Chen, S. Grape seed extract is an aromatase inhibitor and a suppressor of aromatase expression. Cancer Res. 2006, 66, 5960–5967. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Chan, F.L.; Chen, S.; Leung, L.K. The plant polyphenol butein inhibits testosterone-induced proliferation in breast cancer cells expressing aromatase. Life Sci. 2005, 77, 39–51. [Google Scholar] [CrossRef] [PubMed]
- Tang, Z.-R.; Zhang, R.; Lian, Z.-X.; Deng, S.-L.; Yu, K. Estrogen-Receptor Expression and Function in Female Reproductive Disease. Cells 2019, 8, 1123. [Google Scholar] [CrossRef] [PubMed]
- Jonsson, P.; Katchy, A.; Williams, C. Support of a bi-faceted role of estrogen receptor β (ERβ) in ERα-positive breast cancer cells. Endocr. Relat. Cancer 2014, 21, 143–160. [Google Scholar] [CrossRef] [PubMed]
- Huang, B.; Warner, M.; Gustafsson, J.Å. Estrogen receptors in breast carcinogenesis and endocrine therapy. Mol. Cell Endocrinol. 2015, 418, 240–244. [Google Scholar] [CrossRef] [PubMed]
- Meerts, I.A.; Letcher, R.J.; Hoving, S.; Marsh, G.; Bergman, A.; Lemmen, J.G.; van der Burg, B.; Brouwer, A. In vitro estrogenicity of polybrominated diphenyl ethers, hydroxylated PDBEs, and polybrominated bisphenol A compounds. Environ. Health Perspect. 2001, 109, 399–407. [Google Scholar] [CrossRef]
- Watson, C.S.; Koong, L.; Jeng, Y.J.; Vinas, R. Xenoestrogen interference with nongenomic signaling actions of physiological estrogens in endocrine cancer cells. Steroids 2019, 142, 84–93. [Google Scholar] [CrossRef]
- Takayanagi, S.; Tokunaga, T.; Liu, X.; Okada, H.; Matsushima, A.; Shimohigashi, Y. Endocrine disruptor bisphenol A strongly binds to human estrogen-related receptor gamma (ERRgamma) with high constitutive activity. Toxicol. Lett. 2006, 167, 95–105. [Google Scholar] [CrossRef]
- Huang, B.; Luo, N.; Wu, X.; Xu, Z.; Wang, X.; Pan, X. The modulatory role of low concentrations of bisphenol A on tamoxifen-induced proliferation and apoptosis in breast cancer cells. Environ. Sci. Pollut. Res. Int. 2019, 26, 2353–2362. [Google Scholar] [CrossRef]
- Mueller, S.O.; Simon, S.; Chae, K.; Metzler, M.; Korach, K.S. Phytoestrogens and their human metabolites show distinct agonistic and antagonistic properties on estrogen receptor alpha (ERalpha) and ERbeta in human cells. Toxicol. Sci. 2004, 80, 14–25. [Google Scholar] [CrossRef]
- Jiang, H.; Fan, J.; Cheng, L.; Hu, P.; Liu, R. The anticancer activity of genistein is increased in estrogen receptor beta 1-positive breast cancer cells. Onco Targets Ther. 2018, 11, 8153–8163. [Google Scholar] [CrossRef]
- Christoforou, P.; Christopoulos, P.F.; Koutsilieris, M. The role of estrogen receptor β in prostate cancer. Mol. Med. 2014, 20, 427–434. [Google Scholar] [CrossRef]
- Lecomte, S.; Demay, F.; Ferrière, F.; Pakdel, F. Phytochemicals Targeting Estrogen Receptors: Beneficial Rather Than Adverse Effects? Int. J. Mol. Sci. 2017, 18, 1381. [Google Scholar] [CrossRef]
- Suetsugi, M.; Su, L.; Karlsberg, K.; Yuan, Y.C.; Chen, S. Flavone and isoflavone phytoestrogens are agonists of estrogen-related receptors. Mol. Cancer Res. 2003, 1, 981–991. [Google Scholar]
- Prossnitz, E.R.; Barton, M. The G-protein-coupled estrogen receptor GPER in health and disease. Nat. Rev. Endocrinol. 2011, 12, 715–726. [Google Scholar] [CrossRef]
- Viñas, R.; Jeng, Y.J.; Watson, C.S. Non-genomic effects of xenoestrogen mixtures. Int. J. Environ. Res. Public Health 2012, 9, 2694–2714. [Google Scholar] [CrossRef] [PubMed]
- Bulayeva, N.N.; Watson, C.S. Xenoestrogen-induced ERK-1 and ERK-2 activation via multiple membrane-initiated signaling pathways. Environ. Health Perspect. 2004, 112, 1481–1487. [Google Scholar] [CrossRef] [PubMed]
- Velloso, F.J.; Bianco, A.F.; Farias, J.O.; Torres, N.E.; Ferruzo, P.Y.; Anschau, V.; Jesus-Ferreira, H.C.; Chang, T.H.; Sogayar, M.C.; Zerbini, L.F.; et al. The crossroads of breast cancer progression: Insights into the modulation of major signaling pathways. Onco Targets Ther. 2017, 10, 5491–5524. [Google Scholar] [CrossRef]
- Chen, J.; Duan, Y.; Zhang, X.; Ye, Y.; Ge, B.; Chen, J. Genistein induces apoptosis by the inactivation of the IGF-1R/p-Akt signaling pathway in MCF-7 human breast cancer cells. Food Funct. 2015, 6, 995–1000. [Google Scholar] [CrossRef] [PubMed]
- Stolz, A.; Schönfelder, G.; Schneider, M.R. Endocrine Disruptors: Adverse Health Effects Mediated by EGFR? Trends Endocrinol. Metab. 2018, 29, 69–71. [Google Scholar] [CrossRef]
- Rothhammer, V.; Quintana, F.J. The aryl hydrocarbon receptor: An environmental sensor integrating immune responses in health and disease. Nat. Rev. Immunol. 2019, 19, 184–197. [Google Scholar] [CrossRef]
- Matthews, J.; Gustafsson, J.A. Estrogen receptor and aryl hydrocarbon receptor signaling pathways. Nucl. Recept. Signal. 2006, 4, e016. [Google Scholar] [CrossRef]
- Piasecka-Srader, J.; Sadowska, A.; Nynca, A.; Orlowska, K.; Jablonska, M.; Jablonska, O.; Petroff, B.K.; Ciereszko, R.E. The combined effects of 2,3,7,8-tetrachlorodibenzo-p-dioxin and the phytoestrogen genistein on steroid hormone secretion, AhR and ERβ expression and the incidence of apoptosis in granulosa cells of medium porcine follicles. J. Reprod. Dev. 2016, 62, 103–113. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Formosa, R.; Vassallo, J. The Complex Biology of the Aryl Hydrocarbon Receptor and Its Role in the Pituitary Gland. Horm. Cancer 2017, 8, 197–210. [Google Scholar] [CrossRef]
- Fanale, D.; Amodeo, V.; Caruso, S. The Interplay between Metabolism, PPAR Signaling Pathway, and Cancer. PPAR Res. 2017, 2017, 1830626. [Google Scholar] [CrossRef]
- Tyagi, S.; Gupta, P.; Saini, A.S.; Kaushal, C.; Sharma, S. The peroxisome proliferator-activated receptor: A family of nuclear receptors role in various diseases. J. Adv. Pharm. Technol. Res. 2011, 2, 236–240. [Google Scholar] [CrossRef]
- Riu, A.; Grimaldi, M.; le Maire, A.; Bey, G.; Phillips, K.; Boulahtouf, A.; Perdu, E.; Zalko, D.; Bourguet, W.; Balaguer, P. Peroxisome proliferator-activated receptor γ is a target for halogenated analogs of bisphenol A. Environ. Health Perspect. 2011, 119, 1227–1232. [Google Scholar] [CrossRef]
- Grygiel-Górniak, B. Peroxisome proliferator-activated receptors and their ligands: Nutritional and clinical implications—A review. Nutr. J. 2014, 13, 17. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Waltenberger, B.; Pferschy-Wenzig, E.M.; Blunder, M.; Liu, X.; Malainer, C.; Blazevic, T.; Schwaiger, S.; Rollinger, J.M.; Heiss, E.H.; et al. Natural product agonists of peroxisome proliferator-activated receptor gamma (PPARγ): A review. Biochem. Pharmacol. 2014, 92, 73–89. [Google Scholar] [CrossRef] [PubMed]
- Acharya, A.; Das, I.; Chandhok, D.; Saha, T. Redox regulation in cancer: A double-edged sword with therapeutic potential. Oxid. Med. Cell Longev. 2010, 3, 23–34. [Google Scholar] [CrossRef] [PubMed]
- Gassman, N.R. Induction of oxidative stress by bisphenol A and its pleiotropic effects. Environ. Mol. Mutagen. 2017, 58, 60–71. [Google Scholar] [CrossRef] [PubMed]
- Anet, A.; Olakkaran, S.; Purayil, A.K.; Puttaswamygowda, G.H. Bisphenol A induced oxidative stress mediated genotoxicity in Drosophila melanogaster. J. Hazard. Mater. 2019, 370, 42–53. [Google Scholar] [CrossRef] [PubMed]
- Lee, G.A.; Choi, K.C.; Hwang, K.A. Treatment with Phytoestrogens Reversed Triclosan and Bisphenol A-Induced Anti-Apoptosis in Breast Cancer Cells. Biomol. Ther. (Seoul) 2018, 26, 503–511. [Google Scholar] [CrossRef]
- Zafar, A.; Singh, S.; Naseem, I. Cu(II)-coumestrol interaction leads to ROS-mediated DNA damage and cell death: A putative mechanism for anticancer activity. J. Nutr. Biochem. 2016, 33, 15–27. [Google Scholar] [CrossRef]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef]
- Bussard, K.M.; Mutkus, L.; Stumpf, K.; Gomez-Manzano, C.; Marini, F.C. Tumor-associated stromal cells as key contributors to the tumor microenvironment. Breast Cancer Res. 2016, 18, 84. [Google Scholar] [CrossRef]
- Cavallo, F.; De Giovanni, C.; Nanni, P.; Forni, G.; Lollini, P.L. 2011: The immune hallmarks of cancer. Cancer Immunol. Immunother. 2011, 60, 319–326. [Google Scholar] [CrossRef] [PubMed]
- Grivennikov, S.I.; Greten, F.; Karin, M. Immunity, inflammation, and cancer. Cell 2010, 140, 883–899. [Google Scholar] [CrossRef]
- Nowak, K.; Jabłońska, E.; Ratajczak-Wrona, W. Immunomodulatory effects of synthetic endocrine disrupting chemicals on the development and functions of human immune cells. Environ. Int. 2019, 125, 350–364. [Google Scholar] [CrossRef]
- Medjakovic, S.; Mueller, M.; Jungbauer, A. Potential health-modulating effects of isoflavones and metabolites via activation of PPAR and AhR. Nutrients 2010, 2, 241–279. [Google Scholar] [CrossRef] [PubMed]
- Carter, A.B.; Misyak, S.A.; Hontecillas, R.; Bassaganya-Riera, J. Dietary modulation of inflammation-induced colorectal cancer through PPARγ. PPAR Res. 2009, 2009, 498352. [Google Scholar] [CrossRef] [PubMed]
- Notas, G.; Kampa, M.; Castanas, E. G Protein-Coupled Estrogen Receptor in Immune Cells and Its Role in Immune-Related Diseases. Front. Endocrinol. (Lausanne) 2020, 11, 579420. [Google Scholar] [CrossRef] [PubMed]
- Somasundaram, A.; Rothenberger, N.J.; Stabile, L.P. The Impact of Estrogen in the Tumor Microenvironment. Adv. Exp. Med. Biol. 2020, 1277, 33–52. [Google Scholar] [PubMed]
- Teixeira, D.; Marques, C.; Pestana, D.; Faria, A.; Norberto, S.; Calhau, C.; Monteiro, R. Effects of xenoestrogens in human M1 and M2 macrophage migration, cytokine release, and estrogen-related signaling pathways. Environ. Toxicol. 2015, 31, 1496–1509. [Google Scholar] [CrossRef]
- Curran, E.M.; Berghaus, L.J.; Vernetti, N.J.; Saporita, A.J.; Lubahn, D.B.; Estes, D.M. Natural killer cells express estrogen receptor-alpha and estrogen receptor-beta and can respond to estrogen via a non-estrogen receptor-alpha-mediated pathway. Cell Immunol. 2001, 214, 12–20. [Google Scholar] [CrossRef]
- Mace, T.A.; Ware, M.B.; King, S.A.; Loftus, S.; Farren, M.R.; McMichael, E.; Scoville, S.; Geraghty, C.; Young, G.; Carson, W.E.; et al. Soy isoflavones and their metabolites modulate cytokine-induced natural killer cell function. Sci. Rep. 2019, 9, 5068. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.; Li, B.; Li, Z.; Li, J.; Sun, S.; Sun, S. Cancer-associated adipocytes: Key players in breast cancer progression. J. Hematol. Oncol. 2019, 12, 95. [Google Scholar] [CrossRef]
- De Filippis, E.; Li, T.; Rosen, E.D. Exposure of adipocytes to bisphenol-A in vitro interferes with insulin action without enhancing adipogenesis. PLoS ONE 2018, 13, e0201122. [Google Scholar] [CrossRef]
- Pupo, M.; Pisano, A.; Lappano, R.; Santolla, M.F.; De Francesco, E.M.; Abonante, S.; Rosano, C.; Maggiolini, M. Bisphenol A induces gene expression changes and proliferative effects through GPER in breast cancer cells and cancer-associated fibroblasts. Environ. Health Perspect. 2012, 120, 1177–1182. [Google Scholar] [CrossRef]
- Hakkak, R.; Shaaf, S.; Jo, C.H.; Macleod, S.; Korourian, S. Effects of high-isoflavone soy diet vs. casein protein diet and obesity on DMBA-induced mammary tumor development. Oncol. Lett. 2011, 2, 29–36. [Google Scholar] [CrossRef]
- Montales, M.T.; Rahal, O.M.; Nakatani, H.; Matsuda, T.; Simmen, R.C. Repression of mammary adipogenesis by genistein limits mammosphere formation of human MCF-7 cells. J. Endocrinol. 2013, 218, 135–149. [Google Scholar] [CrossRef]
- Bernstein, L.; Ross, R.K. Endogenous hormones and breast cancer risk. Epidemiol. Rev. 1993, 15, 48–65. [Google Scholar] [CrossRef] [PubMed]
- Greenspan, L.C.; Lee, M.M. Endocrine disrupters and pubertal timing. Curr. Opin. Endocrinol. Diabetes Obes. 2018, 25, 49–54. [Google Scholar] [CrossRef]
- Santoro, N.; Randolph, J.F., Jr. Reproductive hormones and the menopause transition. Obstet. Gynecol. Clin. N. Am. 2011, 38, 455–466. [Google Scholar] [CrossRef]
- Prins, G.S. Endocrine disruptors and prostate cancer risk. Endocr. Relat. Cancer 2008, 15, 649–656. [Google Scholar] [CrossRef]
- Rietjens, I.M.C.M.; Louisse, J.; Beekmann, K. The potential health effects of dietary phytoestrogens. Br. J. Pharmacol. 2017, 174, 1263–1280. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.R.; Ko, N.Y.; Chen, K.H. Isoflavone Supplements for Menopausal Women: A Systematic Review. Nutrients 2019, 11, 2649. [Google Scholar] [CrossRef]
- Bilal, I.; Chowdhury, A.; Davidson, J.; Whitehead, S. Phytoestrogens and prevention of breast cancer: The contentious debate. World J. Clin. Oncol. 2014, 5, 705–712. [Google Scholar] [CrossRef] [PubMed]
- Rice, S.; Whitehead, S.A. Phytoestrogens and breast cancer--promoters or protectors? Endocr. Relat. Cancer 2006, 13, 995–1015. [Google Scholar] [CrossRef]
- Padmanabhan, V.; Song, W.; Puttabyatappa, M. Praegnatio Perturbatio-Impact of Endocrine-Disrupting Chemicals. Endocr. Rev. 2021, 42, 295–353. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.E.; Jung, H.W.; Lee, Y.J.; Lee, Y.A. Early-life exposure to endocrine-disrupting chemicals and pubertal development in girls. Ann. Pediatric Endocrinol. Metab. 2019, 24, 78–91. [Google Scholar] [CrossRef]
- Bhattacharya, P.; Keating, A.F. Impact of environmental exposures on ovarian function and role of xenobiotic metabolism during ovotoxicity. Toxicol. Appl. Pharmacol. 2012, 261, 227–235. [Google Scholar] [CrossRef]
- Gore, A.C.; Chappell, V.A.; Fenton, S.E.; Flaws, J.A.; Nadal, A.; Prins, G.S.; Toppari, J.; Zoeller, R.T. EDC-2: The Endocrine Society’s Second Scientific Statement on Endocrine-Disrupting Chemicals. Endocr. Rev. 2015, 36, E1–E150. [Google Scholar] [CrossRef]
- Legler, J.; Fletcher, T.; Govarts, E.; Porta, M.; Blumberg, B.; Heindel, J.J.; Trasande, L. Obesity, diabetes, and associated costs of exposure to endocrine-disrupting chemicals in the European Union. J. Clin. Endocrinol. Metab. 2015, 100, 1278–1288. [Google Scholar] [CrossRef] [PubMed]
- Rancière, F.; Lyons, J.G.; Loh, V.H.Y.; Botton, J.; Galloway, T.; Wang, T.; Shaw, J.E.; Magliano, D.J. Bisphenol A and the risk of cardiometabolic disorders: A systematic review with meta-analysis of the epidemiological evidence. Environ. Health 2015, 14, 46. [Google Scholar] [CrossRef]
- Kajta, M.; Wójtowicz, A.K. Impact of endocrine-disrupting chemicals on neural development and the onset of neurological disorders. Pharmacol. Rep. 2013, 65, 1632–1639. [Google Scholar] [CrossRef]
- Chiu, Y.H.; Mínguez-Alarcón, L.; Ford, J.B.; Keller, M.; Seely, E.W.; Messerlian, C.; Petrozza, J.; Williams, P.L.; Ye, X.; Calafat, A.M.; et al. Trimester-Specific Urinary Bisphenol A Concentrations and Blood Glucose Levels Among Pregnant Women from a Fertility Clinic. J. Clin. Endocrinol. Metab. 2017, 102, 1350–1357. [Google Scholar] [CrossRef]
- Bae, S.; Lim, Y.H.; Lee, Y.A.; Shin, C.H.; Oh, S.Y.; Hong, Y.C. Maternal Urinary Bisphenol A Concentration During Midterm Pregnancy and Children’s Blood Pressure at Age 4. Hypertension 2017, 69, 367–374. [Google Scholar] [CrossRef] [PubMed]
- Taylor, P.; Okosieme, O.E.; Murphy, R.; Hales, C.; Chiusano, E.; Maina, A.; Joomun, M.; Bestwick, J.P.; Smyth, P.; Paradice, R.; et al. Maternal perchlorate levels in women with borderline thyroid function during pregnancy and the cognitive development of their offspring: Data from the Controlled Antenatal Thyroid Study. J. Clin. Endocrinol. Metab. 2014, 99, 4291–4298. [Google Scholar] [CrossRef]
- Marchesini, G.R.; Meimaridou, A.; Haasnoot, W.; Meulenberg, E.; Albertus, F.; Mizuguchi, M.; Takeuchi, M.; Irth, H.; Murk, A.J. Biosensor discovery of thyroxine transport disrupting chemicals. Toxicol. Appl. Pharmacol. 2008, 232, 150–160. [Google Scholar] [CrossRef] [PubMed]
- Oulhote, Y.; Chevrier, J.; Bouchard, M.F. Exposure to Polybrominated Diphenyl Ethers (PBDEs) and Hypothyroidism in Canadian Women. J. Clin. Endocrinol. Metab. 2016, 101, 590–598. [Google Scholar] [CrossRef] [PubMed]
- Zaheer, K.; Akhtar, M.H. An updated review of dietary isoflavones: Nutrition, processing, bioavailability and impacts on human health. Crit. Rev. Food Sci. Nutr. 2017, 57, 1280–1293. [Google Scholar] [CrossRef]
- Shike, M.; Doane, A.S.; Russo, L.; Cabal, R.; Reis-Filho, J.S.; Gerald, W.; Cody, H.; Khanin, R.; Bromberg, J.; Norton, L. The Effects of Soy Supplementation on Gene Expression in Breast Cancer: A Randomized Placebo-Controlled Study. J. Natl. Cancer Inst. 2014, 106, 189. [Google Scholar] [CrossRef]
- Maskarinec, G.; Suzuki, S.; Pagano, I.S.; Morimoto, Y.; Franke, A.A.; Ehya, H. Cytology in Nipple Aspirate Fluid during a Randomized Soy Food Intervention among Premenopausal Women. Nutr. Cancer 2013, 65, 1116–1121. [Google Scholar] [CrossRef]
- Sen, C.; Morimoto, Y.; Heak, S.; Cooney, R.V.; Franke, A.A.; Maskarinec, G. Soy Foods and Urinary Isoprostanes: Results from a Randomized Study in Premenopausal Women. Food Funct. 2012, 3, 517. [Google Scholar] [CrossRef]
- McCann, S.E.; Edge, S.B.; Hicks, D.G.; Thompson, L.U.; Morrison, C.D.; Fetterly, G.; Andrews, C.; Clark, K.; Wilton, J.; Kulkarni, S. A Pilot Study Comparing the Effect of Flaxseed, Aromatase Inhibitor, and the Combination on Breast Tumor Biomarkers. Nutr. Cancer 2014, 66, 566–575. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.A.; Chatterton, R.T.; Michel, N.; Bryk, M.; Lee, O.; Ivancic, D.; Heinz, R.; Zalles, C.M.; Helenowski, I.B.; Jovanovic, B.D.; et al. Soy Isoflavone Supplementation for Breast Cancer Risk Reduction: A Randomized Phase II Trial. Cancer Prev. Res. (Phila) 2012, 5, 309–319. [Google Scholar] [CrossRef]
- Wu, A.H.; Spicer, D.; Garcia, A.; Tseng, C.-C.; Hovanessian-Larsen, L.; Sheth, P.; Martin, S.E.; Hawes, D.; Russell, C.; MacDonald, H.; et al. Double-Blind Randomized 12-Month Soy Intervention Had No Effects on Breast MRI Fibroglandular Tissue Density or Mammographic Density. Cancer Prev. Res. (Phila) 2015, 8, 942–951. [Google Scholar] [CrossRef]
- Zhu, W.; Qin, W.; Zhang, K.; Rottinghaus, G.E.; Chen, Y.-C.; Kliethermes, B.; Sauter, E.R. Trans-Resveratrol Alters Mammary Promoter Hypermethylation in Women at Increased Risk for Breast Cancer. Nutr. Cancer 2012, 64, 393–400. [Google Scholar] [CrossRef]
- Saghatelyan, T.; Tananyan, A.; Janoyan, N.; Tadevosyan, A.; Petrosyan, H.; Hovhannisyan, A.; Hayrapetyan, L.; Arustamyan, M.; Arnhold, J.; Rotmann, A.-R.; et al. Efficacy and Safety of Curcumin in Combination with Paclitaxel in Patients with Advanced, Metastatic Breast Cancer: A Comparative, Randomized, Double-Blind, Placebo-Controlled Clinical Trial. Phytomedicine 2020, 70, 153218. [Google Scholar] [CrossRef] [PubMed]
- Grainger, E.M.; Moran, N.E.; Francis, D.M.; Schwartz, S.J.; Wan, L.; Thomas-Ahner, J.; Kopec, R.E.; Riedl, K.M.; Young, G.S.; Abaza, R.; et al. A Novel Tomato-Soy Juice Induces a Dose-Response Increase in Urinary and Plasma Phytochemical Biomarkers in Men with Prostate Cancer. J. Nutr. 2019, 149, 26–35. [Google Scholar] [CrossRef] [PubMed]
- Hamilton-Reeves, J.M.; Banerjee, S.; Banerjee, S.K.; Holzbeierlein, J.M.; Thrasher, J.B.; Kambhampati, S.; Keighley, J.; Van Veldhuizen, P. Short-Term Soy Isoflavone Intervention in Patients with Localized Prostate Cancer: A Randomized, Double-Blind, Placebo-Controlled Trial. PLoS ONE 2013, 8, e68331. [Google Scholar] [CrossRef] [PubMed]
- Bosland, M.C.; Enk, E.; Schmoll, J.; Schlicht, M.J.; Randolph, C.; Deaton, R.J.; Xie, H.; Zeleniuch-Jacquotte, A.; Kato, I. Soy Protein Supplementation in Men Following Radical Prostatectomy: A 2-Year Randomized, Placebo-Controlled Clinical Trial. Am. J. Clin. Nutr. 2021, 113, 821–831. [Google Scholar] [CrossRef]
- Lazarevic, B.; Hammarström, C.; Yang, J.; Ramberg, H.; Diep, L.M.; Karlsen, S.J.; Kucuk, O.; Saatcioglu, F.; Taskèn, K.A.; Svindland, A. The Effects of Short-Term Genistein Intervention on Prostate Biomarker Expression in Patients with Localised Prostate Cancer before Radical Prostatectomy. Br. J. Nutr. 2012, 108, 2138–2147. [Google Scholar] [CrossRef] [PubMed]
- Saadipoor, A.; Razzaghdoust, A.; Simforoosh, N.; Mahdavi, A.; Bakhshandeh, M.; Moghadam, M.; Abdollahi, H.; Mofid, B. Randomized, Double-Blind, Placebo-Controlled Phase II Trial of Nanocurcumin in Prostate Cancer Patients Undergoing Radiotherapy: Nanocurcumin for Prostate Cancer Patients Undergoing Radiotherapy. Phytother. Res. 2019, 33, 370–378. [Google Scholar] [CrossRef]
- Greil, R.; Greil-Ressler, S.; Weiss, L.; Schönlieb, C.; Magnes, T.; Radl, B.; Bolger, G.T.; Vcelar, B.; Sordillo, P.P. A Phase 1 Dose-Escalation Study on the Safety, Tolerability and Activity of Liposomal Curcumin (LipocurcTM) in Patients with Locally Advanced or Metastatic Cancer. Cancer Chemother. Pharmacol. 2018, 82, 695–706. [Google Scholar] [CrossRef]
- Hejazi, J.; Rastmanesh, R.; Taleban, F.-A.; Molana, S.-H.; Hejazi, E.; Ehtejab, G.; Hara, N. Effect of Curcumin Supplementation during Radiotherapy on Oxidative Status of Patients with Prostate Cancer: A Double Blinded, Randomized, Placebo-Controlled Study. Nutr. Cancer 2016, 68, 77–85. [Google Scholar] [CrossRef]
- Quaas, A.M.; Kono, N.; Mack, W.J.; Hodis, H.N.; Felix, J.C.; Paulson, R.J.; Shoupe, D. Effect of Isoflavone Soy Protein Supplementation on Endometrial Thickness, Hyperplasia, and Endometrial Cancer Risk in Postmenopausal Women: A Randomized Controlled Trial. Menopause 2013, 20, 840–844. [Google Scholar] [CrossRef]
- Tuyaerts, S.; Rombauts, K.; Everaert, T.; Van Nuffel, A.M.T.; Amant, F. A Phase 2 Study to Assess the Immunomodulatory Capacity of a Lecithin-Based Delivery System of Curcumin in Endometrial Cancer. Front. Nutr. 2019, 5, 138. [Google Scholar] [CrossRef]
- Nguyen, A.V.; Martinez, M.; Stamos, M.J.; Moyer, M.P.; Planutis, K.; Hope, C.; Holcombe, R.F. Results of a Phase I Pilot Clinical Trial Examining the Effect of Plant-Derived Resveratrol and Grape Powder on Wnt Pathway Target Gene Expression in Colonic Mucosa and Colon Cancer. Cancer Manag. Res. 2009, 1, 25–37. [Google Scholar] [PubMed]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: Globocan Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Wogan, G.N.; Hecht, S.S.; Felton, J.S.; Conney, A.H.; Loeb, L.A. Environmental and chemical carcinogenesis. Semin. Cancer Biol. 2004, 14, 473–486. [Google Scholar] [CrossRef] [PubMed]
- Mbemi, A.; Khanna, S.; Njiki, S.; Yedjou, C.G.; Tchounwou, P.B. Impact of Gene-Environment Interactions on Cancer Development. Int. J. Environ. Res. Public Health 2020, 17, 8089. [Google Scholar] [CrossRef] [PubMed]
- Clavel, J. Progress in the epidemiological understanding of gene-environment interactions in major diseases: Cancer. Comptes Rendus Biol. 2007, 330, 306–317. [Google Scholar] [CrossRef] [PubMed]
- Vineis, P.; Kriebel, D. Causal models in epidemiology: Past inheritance and genetic future. Environ. Health 2006, 5, 21. [Google Scholar] [CrossRef]
- Goodspeed, A.; Heiser, L.M.; Gray, J.W.; Costello, J.C. Tumor-Derived Cell Lines as Molecular Models of Cancer Pharmacogenomics. Mol. Cancer Res. 2016, 14, 3–13. [Google Scholar] [CrossRef]
- Amato, R.; Pinelli, M.; D’Andrea, D.; Miele, G.; Nicodemi, M.; Raiconi, G.; Cocozza, S. A novel approach to simulate gene-environment interactions in complex diseases. BMC Bioinform. 2010, 11, 8. [Google Scholar] [CrossRef]
- Perrot-Applanat, M.; Kolf-Clauw, M.; Michel, C.; Beausoleil, C. Alteration of mammary gland development by bisphenol a and evidence of a mode of action mediated through endocrine disruption. Mol. Cell Endocrinol. 2018, 475, 29–53. [Google Scholar] [CrossRef]
- Wu, H.C.; Cohn, B.A.; Cirillo, P.M.; Santella, R.M.; Terry, M.B. DDT exposure during pregnancy and DNA methylation alterations in female offspring in the Child Health and Development Study. Reprod. Toxicol. 2020, 92, 138–147. [Google Scholar] [CrossRef]
- Ali, I.; Julin, B.; Glynn, A.; Högberg, J.; Berflund, M.; Johansson, J.; Andersson, S.; Andrén, O.; Giovannucci, E.; Wolk, A.; et al. Exposure to polychlorinated biphenyls and prostate cancer: Population-based prospective cohort and experimental studies. Carcinogenesis 2016, 37, 1144–1151. [Google Scholar] [CrossRef]
- Ho, S.M.; Tang, W.Y.; de Frausto, J.B.; Prins, G.S. Developmental exposure to estradiol and bisphenol A increases susceptibility to prostate carcinogenesis and epigenetically regulates phosphodiesterase type 4 variant 4. Cancer Res. 2006, 66, 5624–5632. [Google Scholar] [CrossRef] [PubMed]
- Prins, G.S.; Birch, L.; Tang, W.Y.; Ho, S.M. Developmental estrogen exposures predispose to prostate carcinogenesis with aging. Reprod. Toxicol. 2007, 23, 374–382. [Google Scholar] [CrossRef] [PubMed]
- Zeng, H.; Yang, Z.; Li, J.; Wen, Y.; Wu, Z.; Zheng, Y.; Yu, Y.; Xu, Y.; Gao, S.; Tan, F.; et al. Associations between female lung cancer risk and sex steroid hormones: A systematic review and meta-analysis of the worldwide epidemiological evidence on endogenous and exogenous sex steroid hormones. BMC Cancer 2021, 21, 690. [Google Scholar] [CrossRef]
- Wolf, L.A.; Terry, P.D.; Potter, J.D.; Bostick, R.M. Do factors related to endogenous and exogenous estrogens modify the relationship between obesity and risk of colorectal adenomas in women? Cancer Epidemiol. Biomark. Prev. 2007, 16, 676–683. [Google Scholar] [CrossRef][Green Version]
- Wang, Z.; Butler, L.M.; Wu, A.H.; Koh, W.; Jin, A.; Wang, R.; Yuan, J. Reproductive factors, hormone use and gastric cancer risk: The Singapore Chinese Health Study. Int. J. Cancer 2016, 138, 2837–2845. [Google Scholar] [CrossRef]
- Fucic, A.; Gamulin, M.; Ferencic, Z.; Rokotov, D.S.; Katic, J.; Bartonova, A.; Lovasic, I.B.; Merlo, D.F. Lung cancer and environmental chemical exposure: A review of our current state of knowledge with reference to the role of hormones and hormone receptors as an increased risk factor for developing lung cancer in man. Toxicol. Pathol. 2010, 38, 849–855. [Google Scholar] [CrossRef]
- Desmawati, D.; Sulastri, D. Phytoestrogens and Their Health Effect. Open Access Maced. J. Med. Sci. 2019, 7, 495–499. [Google Scholar] [CrossRef] [PubMed]
- Hilakivi-Clarke, L. Estrogens, BRCA1 and breast cancer. Cancer Res. 2000, 60, 4993–5000. [Google Scholar]
 ), cell apoptosis and death
reprogramming (
), cell apoptosis and death
reprogramming ( ), growth suppressor evading (
), growth suppressor evading ( ), genome instability and
mutation (
), genome instability and
mutation ( ), tumor inflammation-promoting
(
), tumor inflammation-promoting
( ), immune response destruction
(
), immune response destruction
( ), redox homeostasis
interrupting, and metabolic rewiring (
), redox homeostasis
interrupting, and metabolic rewiring ( ).
).
 ), cell apoptosis and death
reprogramming (
), cell apoptosis and death
reprogramming ( ), growth suppressor evading (
), growth suppressor evading ( ), genome instability and
mutation (
), genome instability and
mutation ( ), tumor inflammation-promoting
(
), tumor inflammation-promoting
( ), immune response destruction
(
), immune response destruction
( ), redox homeostasis
interrupting, and metabolic rewiring (
), redox homeostasis
interrupting, and metabolic rewiring ( ).
).
| Name | Source | Relative Binding Affinity | Biological Activity | Experimental Evidence | Public Health Implications | References | |
|---|---|---|---|---|---|---|---|
| ERα | ERβ | ||||||
| bisphenol A (BPA) | chemical used to manufacture polycarbonate plastics, epoxy resins, and added to other plastics; found in food containers, utensils, dental sealants, protective coatings, flame retardants, water supply pipes | 0.01 | 0.01 | disrupts ER activity by mimicking, enhancing, or inhibiting endogenous estrogen; directly impacts intracellular signal transduction ↑ ER mRNA | ↑ hyperplastic ducts ↑ ER+ cells ↑ PR+ cells ↑ cell proliferation Phosphorylation of AKT and ERK ↑ prostate cancer cell proliferation Aberrant development of prostate and urethra ↑ prostate tumor size AR antagonist ↑ SHBG | increased risk of breast, prostate, and uterine cancer no risk for ovarian cancer | [26,27,28,29] |
| dichlorodiphenyltrichloroethane (DDT) | pesticide; used to combat malaria, typhus, and other insect-borne human diseases | 0–0.01 | 0–0.02 | estrogenic activity | Accumulates in adipose tissue Stimulates uterine proliferation and impairs normal follicle development Inhibits PKA activation Alters gene expression and hormone synthesis. Inhibit PGE2 levels in ovaries | increased breast cancer risk | [30,31,32] |
| polychlorinated biphenyls (PCBs) | used as flame retardants; found in electrical equipment, construction materials, coatings, textiles, furniture padding, etc. | 0.01–3.4 | <0.01–7.2 | estrogenic/anti-estrogenic | ↓ cell growth ↓ proliferation ↓ AR activity ↑ competitive inhibition to AR ↑ uterus weight | increased breast cancer risk for certain PCBs | [36,37,38] |
| polybrominated diphenyl ethers (PBDEs) | used as flame retardants; found in electrical equipment, construction materials, coatings, textiles, furniture padding, etc. | 1.3–20 a | estrogenic activity | ↑ viability and proliferation of human breast, cervical, and ovarian cancer cells ↑ cell contact ↑ phosphorylation of PKCa and ERK1/2 proteins in tumor cells and in CHO cells | no clear association with breast cancer risk | [39,40,41,42,43] | |
| diethylstilbestrol (DES) | used to prevent miscarriage, premature labor, and pregnancy complications | 236 | 221 | hydrophobic interactions; potent transcriptional activator through genomic signaling | ↑ PI3 kinase signaling ↑ AKT phosphorylation ERRγ antagonist ↑ SRC1 ↑ SHBG ↓ LH, TSH, FSH, DHEA, testosterone, and E1 | vaginal cancer risk | [44,45] |
| methoxychlor (DMDT) | used to protect pets, crops, and livestock from pests such as mosquitoes, cockroaches, and other insects | <0.01 | <0.01 | ERα agonist ERβ antagonist anti-estrogen in ovaries | Inhibit estrogen binding to ER ↓ serum progesterone ↑ uterotrophic activity Impairs overall fertility | increased ovarian cancer risk | [33] |
| ethinyl estradiol (EE2) | ovulation inhibitor; used in hormonal contraceptives | 190 c | ↑ERRγ and RAGE expression primarily through Erα | ↑ cell proliferation but not as much as E2↑ | little/no breast cancer risk reduced risk for ovarian, endometrial, colorectal, and lymphatic/hematopoietic cancers | [46] | |
| phthalates | found in soft plastics used as packaging materials | N/A d | N/A d | competitive binding with E2 for ER | ↑ MCF7 cell proliferation and viability | increased breast cancer risk | [47,48] |
| nonylphenols | used in industrial processes and in consumer laundry detergents, personal hygiene, automotive, latex paints, and lawn care products | 0.0032–0.037 c | estrogen-like activity on ER+ breast cancer cells | ↑ prostate epithelial cell proliferation ↓ prostate cell viability Promotes cytoplasm-nucleus Translocation of ERα, but not ERβ | increased breast cancer risk | [49,50] | |
| parabens | used as preservatives in many foods such as beer, sauces, sodas, and cosmetics | 0.011–0.11 b | 0.011–0.123 b | ERRγ agonist | breast cancer cell proliferation ↑ tumor size Sulfotransferase inhibitor | increased breast cancer risk | [51,52] |
| Name | Source | Relative Binding Affinity | Biological Activity | Experimental Evidence | Public Health Implications | References | |
|---|---|---|---|---|---|---|---|
| ERα | ERβ | ||||||
| genistein (GEN) | soybeans and soy-containing products | 4 | 87 | [low]: estrogenic [high]: anti-estrogenic ↓ ERα protein/mRNa levels | ↑ apoptosis ↑ cell cycle arrest ↑ demethylation of tumor suppressor genes Inhibits ovarian cancer cell migration, invasion, and proliferation ↓ phosphorylation of PI3K and GSK3b RTK inhibitor DNA topoisomerase II inhibitor ER+ cell proliferation ↓ tumor associated macrophage ↓ proliferation VEGF inhibitor (angiogenesis) ↓ breast CSCs ↑ cell adhesion ↓ migration/invasion | breast and prostate cancer preventative decreased ovarian cancer risk | [53,54,55,56,57,58] |
| daidzein (DAI) | soybeans | 0.1 | 0.5 | anti-estrogenic in organs expressing more ERα estrogenic in ERβ-presenting organs | ↑ ERa expression/nuclear localization ↓ cell proliferation ↓ migration ↓ invasion Induces cell cycle arrest and apoptosis | endometrial cancer preventative | [59,60] |
| quercetin (QUE) | various fruits and vegetables such as apples, red grapes, onions, raspberries, honey, cherries, citrus fruits, green leafy vegetables, red wine, cappers, lovage, radish leaves, tea, cranberries, and peppers | 0.01 | 0.04 | estrogenic ↓ cytoplasmic ER levels ↑ tighter nuclear association to ER | ↑ antiproliferative ↓ mammospheres in breast cancer cells ↓ breast CSC characteristics ↓ EMT Regulates B-catenin signaling, leading to EMT inhibition [low]: ↑proliferation ↑ migration ↑ invasion ↓ apoptosis [high]: ↓cell growth ↓ metastatic process ↑ cell cycle arrest ↓ tumor volume | anti-cancer for breast cancer | [54,57,62,63,64,65,66,67,68] |
| apigenin (APE) | fruits and vegetables such as parsley chamomile, celery, vine-spinach, artichoke, oregano, red wine, and beer | 0.3 | 6 | ↓ ERα in uterus estrogenic/anti-estrogenic ↓ estradiol levels | [low]: ↑proliferation ↑ AKT phosphorylation ↑ invasion [high]: ↓proliferation ↓ AKT phosphorylation ↓ invasion ↑ apoptosis ↑ cell cycle arrest ↓ cell growth Inhibit MAPK | decreased breast, prostate, and ovarian cancer risk | [61,62,69,70,71,72,73,74] |
| resveratrol (RES) | Japanese knotweed grapes, wine, strawberries, and peanuts | 6.11–11.2 a | 4.7–15.66 a | ERRγ agonist | ↑ breast cancer cell proliferation ↑ tumor size Sulfotransferase inhibitor | increased breast cancer risk | [75,76,77,78,79] |
| myricetin (MYR) | vegetables, fruits, nuts, berries, tea, and red wine | N/A c | N/A c | Competitive binding to ER ERα agonist | Inhibits prostate cancer cell growth, key enzymes involved in the initiation and progression of cancer ↓ migration ↓ invasion ↓ adhesion ↓ tumor nodules ↓ MMP2 and MMP9 protein expression ↑ apoptosis CK2 inhibitor | decreased breast and prostate cancer risk | [80,81,82,83,84,85] |
| kaempferol (KPF) | tea, broccoli, apples, strawberries, beans, bee pollen, cabbage, capers, cauliflower, chia seeds, chives, cumin, moringa leaves, endive, fennel, and garlic | 0.1 | 3 | estrogenic activity ERα-dependent transcriptional activation activity | ↑ apoptosis ↓ cancer cell growth ↓ angiogenesis Preserve/protect cell viability ↓ migration ↓ MMP3 protein activity Inhibit VEGF release in breast cancer cells Reduced VEGF mRNA in ovarian cancer cells ↓ tumor growth/metastasis ↓ EMT ↑ cell cycle arrest Inhibits various cancer cell lines | decreased breast cancer risk | [86,87,88,89,90,91] |
| luteolin (LUT) | celery, peppermint, thyme, rosemary, oregano, artichoke, green pepper, and perilla leaf | N/A d | N/A d | Estrogenic | ↑ cell cycle arrest ↑ apoptosis ↓ proliferation Inhibit MAPK, EGFR, VEGF ↓ PSA ↓ aromatase ↓ ERK and FAK phosphorylation | anti-cancer for breast and prostate endometrial cancer risk | [92] |
| curcumin (CUR) | derived from the plant Curcuma longa; turmeric | N/A b | N/A b | ↓ ER expression | ↓ EMT and migration ability ↓ breast CSC population ↓ nuclear translocation of B-catenin (slug transactivation; restored E-cadherin expression) ↑ apoptosis ↑ cell cycle arrest ↑ senescence ↓ p53 Inhibits proliferation, migration, invasion, angiogenesis, and metastasis in breast cancer cells Interferes with osteoblast formation in prostate cancer cell line | anti-cancer | [93,94,95,96] |
| coumestrol (COU) | plants such as soybeans, clover, alfalfa sprouts, sunflower seeds, spinach, legumes, chickpeas, split peas, lima beans, and pinto beans | 20 | 140 | ↓ ERα protein/mRNA levels | Inhibits cell viability, cell growth, and proliferation ↑ Bax ↑ apoptosis ↑ cell cycle arrest ↑ ROS generation ↑ DNA damage ↑ ERK1/2 phosphorylation ↑ p53 proteins ↓ AKT phosphorylation | anti-cancer for breast and prostate cancers anti-tumor for ovarian, breast, lung, and cervical cancers decreased endometrial cancer risk | [98,99,100,101] |
| Identifier | Cancer Type/Prevention | Chemicals | Date | Participants/ Type of Study | Aims | Results |
|---|---|---|---|---|---|---|
| NCT00597532 [226] | Breast | Genistein + Daidzein | 8/2002–4/2016 | 140 women/ R P controlled study | To examine the effects of soy supplementation on breast cancer-related genes and pathways | Tumors- PRE vs POST = altered EXP of 21 out of 202 genes. ↑ FANCC & UGT2A1 EXP in TG vs. PG (p < 0.05) Over-EXP of FGFR2, E2F5, BUB1, CCNB2, MYBL2, CDK1, and CDC20 (p < 0.01) in tumors with high-genistein signature |
| NCT00513916 [232,[233] | Breast | Isoflavones | 7/2006–2/2012 | 82 multiethnic PR/ R, crossover ‡ | To study the effects of dietary soy on estrogens in breast fluid, blood, and urine samples from healthy women | High-soy diet resulted in a modest trend of a lower cytological subclass in breast epithelial cells ↑isoprostane excretion in high-soy diet (p = 0.02) |
| NCT00612560 [229] | Breast | Ground flaxseed (FS) ± anastrozole (AI) | 11/2007–4/2014 | 24 PO; 2 x 2 factorial R intervention | To examine the effect of flaxseed consumption compared to AI, and the effect of combined flaxseed and AI therapy on breast cancer treatment | ↓ serum steroid hormone DHEA w/ AI treatment (p = 0.009) PRE vs POST in FS + AI = ~40% ↓ EXP of estrogen receptorβ Lower Enterolactone excretion in FS + AI vs FS |
| NCT00290758 [230] | Breast | Mixed soy isoflavones | 1/2006–7/2009 | 126 women (≥ 25 years)/ R *B | To study how well genistein works in preventing breast cancer in women at high risk for breast cancer | ↑ Ki-67 labeling index within PR TG (p = 0.04) Within TG, ↑ EXP of 14/28 genes (p = 0.017–0.052), but no S changes in PG TG vs PG = ↑ ESR1, FAS, FOXA1, MYB (p = NS) |
| NCT01219075 [231] | Breast | Daidzein, genistein, glycitein | 7/2010–present | 85 women (30–75 years)/ D-B, R, P-controlled | To study soy isoflavones supplement in treating women at high risk or with breast cancer | NS differences in breast density area (p = 0.23) and mammogrpahic density % (p = 0.38) in TG vs PG |
| University of North Dakota School of Medicine and Health Sciences [232] | Breast | Trans- resveratrol | N/A | 39 women/D-B, R, P-controlled | To determine if trans-resveratrol had a dose-related effect on DNA methylation and prostaglandin expression in humans | ↑ levels of trans-resveratrol & resveratrol-glucuronide in serum = ↓ RASSF-1α methylation (p = 0.047) & ↓ PGE2 EXP in breast (p = 0.045) |
| National Center of Oncology, Yerevan, Armenia (Ministry of Health Republic of Armenia Registration No.: Nr 5592-17-02-23) [233] | Breast | Curcumin + Paclitaxel | 3/2017–9/2018 | 150 women (18–75 years)/ *, single-institution, R, P-controlled, D-B, parallel group, two-arm trial | To assess the efficacy and safety of intravenous curcumin infusion in combination with paclitaxel in patients with metastatic and advanced breast cancer | ↑ objective response rate (ORR) of TG vs PG (16 weeks after beginning treatment, p < 0.05; completed treatment, p < 0.01) 3 months after treatment, ↑ ORR TG vs. PG (p < 0.07) ↑ fatigue in TG vs. PG (p = 0.05), but the overall physical performance was significantly higher with curcumin than the placebo during treatment and at the end of follow-up |
| NCT00596895 [234] | Prostate | soy milk containing isoflavonoid | 11/2003–11/2007 | 20 men/ O-L, * nonrandomized trial | To evaluate the efficacy of isoflavone in patients with PSA recurrent prostate cancer after prior therapy. | Slope of PSA level (after vs. before study entry): ↓ in 6 men (p < 0.05), ↑ in 2 men (p < 0.05), and NS changes in 12 men A decline in the rise of serum PSA after the initiation of soy milk. |
| NCT01009736 [61] | Prostate | Tomato-soy juice | 1/2008–7/2009 | 60 men/ * dose-escalating | To study the side effects of tomato-soy juice and its effect on biomarkers in patients with prostate cancer undergoing prostatectomy | High TG vs PG, ↓prostate-specific antigen slope (p = 0.078) |
| NCT00255125 [235] | Prostate | Soy isoflavone capsules | 9/2005–10/2009 | 86 men (≥18 years)/ D-B, R, P-controlled | To evaluate the effects of soy isoflavone consumption on prostate specific antigen, hormone levels, total cholesterol, and apoptosis in men with localized prostate cancer. | TG vs PG in malignant prostate tissue = down-regulated 12 genes involved in cell cycle control and 9 genes involved in apoptosis No significant changes in serum total testosterone, free testosterone, total estrogen, estradiol, PSA, and total cholesterol |
| NCT00765479 [236] | Prostate | Soy protein isolate | 9/2011–7/2013 | 284 men (40–75 years)/ R, P-controlled | Secondary analysis of body weight, blood pressure, thyroid hormones, iron status, and clinical chemistry in a 2-y trial of soy protein supplementation in middle-aged to older men. | Soy supplementation did not affect body weight, blood pressure, serum total cholesterol, iron status parameters, calcium, phosphorus, and thyroid hormones. |
| NCT00546039 [237] | Prostate | Synthetic genistein | 4/2007–1/2009 | 47 Norwegian men/ * P-controlled, R, D-B | To evaluate safety and mechanisms of possible chemopreventive effects of synthetic genistein (BONISTEIN) in patients with localized prostate cancer undergoing laparoscopic radical prostatectomy | Genistein intervention significantly reduced the mRNA level of KLK4 in tumor cells (p = 0.033) and p27Kip1 In genistein intervention, no significant effects on proliferation-, cell cycle-, apoptosis-, or neuroendocrine biomarkers |
| NCT02724618 [238] | Prostate | Nanocurcumin | 3/2016–present | 64 men/ R, D-B, * P-controlled | To determine the efficacy of oral nanocurcumin in prostate cancer patients undergoing radiotherapy. | Nanocurcumin was well tolerated. No significant difference was found between two groups regarding tumor response. |
| NCT02138955 [239] | Prostate, Colon | Curcumin | 3/2014–6/2017 | 32 participants (18–85 years)/ ∞, single-center, O-L | To investigate the safety and tolerability of increasing doses of liposomal curcumin in patients with metastatic cancer | 300 mg/m2 liposomal curcumin over 6 h was the maximum tolerated dose, and a recommended starting dose for anti-cancer trials Anti-tumor activity was not detected |
| NCT01917890 [240] | Prostate | Curcumin | 3/2011–10/2013 | 40 men (50–80 years)/ R, D-B, P-controlled | To evaluate the effect of curcumin supplementation on oxidative status of patients with prostate cancer who undergo radiotherapy | In TG: ↓ activity of superoxide dismutase (SOD) (p = 0.026), and ↑ plasma total antioxidant capacity (TAC) (p = 0.014) ↓ PSA level in both TG and PG No significant differences in treatment outcomes were observed between TG and PG |
| NCT00118846 [241] | Endometria | Genistein, daidzein, glycitein, | 3/2004–3/2009 | 350 women (45-92 years)/ R, D-B, P-controlled | To determine whether long-term isoflavone soy protein (ISP) supplementation affects endometrial thickness and rates of endometrial hyperplasia and cancer in postmenopausal women | Soy-treated group did not significantly differ on the mean baseline or on-trial changes in endometrial thickness ISP has been found to predominantly act on the beta-type estrogen receptor because of its structure similar to 17β-estradiol and selective estrogen receptor modulator (SERM)-like activity. |
| NCT02017353 [242] | Endometrial | Curcumin Phytosome (CP) | 10/2013–10/2016 | 7 women (≥18 years)/ O-L, * non-randomized | To determine whether curcumin can inhibit tumor induced inflammation in patients with endometrial carcinoma. In addition, curcumin could possibly induce a better functioning of chemotherapy and a decrease in toxicity from chemotherapy. | In TG, downregulated MHC expression levels on leukocytes (p = 0.0313), frequency of monocytes (p= 0.0114), and ICOS expression by CD8+ T cells (p = 0.0002), but upregulated CD69 levels on CD16- NK cells (0.0313). |
| NCT00256334 [243] | Colon | Trans-resveratrol + quercetin | 7/2005–4/2009 | 11 participants (≥18 years)/∞ pilot, O-L | To evaluate the effects of a low dose of plant-derived resveratrol formulation and resveratrol-containing freeze-dried grape powder on Wnt signaling in the colon | Resveratrol did not inhibit Wnt pathway in colon cancer, but did inhibit Wnt pathway in normal colonic mucosa (p < 0.03) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, X.; Ha, D.; Yoshitake, R.; Chan, Y.S.; Sadava, D.; Chen, S. Exploring the Biological Activity and Mechanism of Xenoestrogens and Phytoestrogens in Cancers: Emerging Methods and Concepts. Int. J. Mol. Sci. 2021, 22, 8798. https://doi.org/10.3390/ijms22168798
Wang X, Ha D, Yoshitake R, Chan YS, Sadava D, Chen S. Exploring the Biological Activity and Mechanism of Xenoestrogens and Phytoestrogens in Cancers: Emerging Methods and Concepts. International Journal of Molecular Sciences. 2021; 22(16):8798. https://doi.org/10.3390/ijms22168798
Chicago/Turabian StyleWang, Xiaoqiang, Desiree Ha, Ryohei Yoshitake, Yin S. Chan, David Sadava, and Shiuan Chen. 2021. "Exploring the Biological Activity and Mechanism of Xenoestrogens and Phytoestrogens in Cancers: Emerging Methods and Concepts" International Journal of Molecular Sciences 22, no. 16: 8798. https://doi.org/10.3390/ijms22168798
APA StyleWang, X., Ha, D., Yoshitake, R., Chan, Y. S., Sadava, D., & Chen, S. (2021). Exploring the Biological Activity and Mechanism of Xenoestrogens and Phytoestrogens in Cancers: Emerging Methods and Concepts. International Journal of Molecular Sciences, 22(16), 8798. https://doi.org/10.3390/ijms22168798







