Protein Arginine Methyltransferase (PRMT) Inhibitors—AMI-1 and SAH Are Effective in Attenuating Rhabdomyosarcoma Growth and Proliferation in Cell Cultures
Abstract
:1. Introduction
2. Results
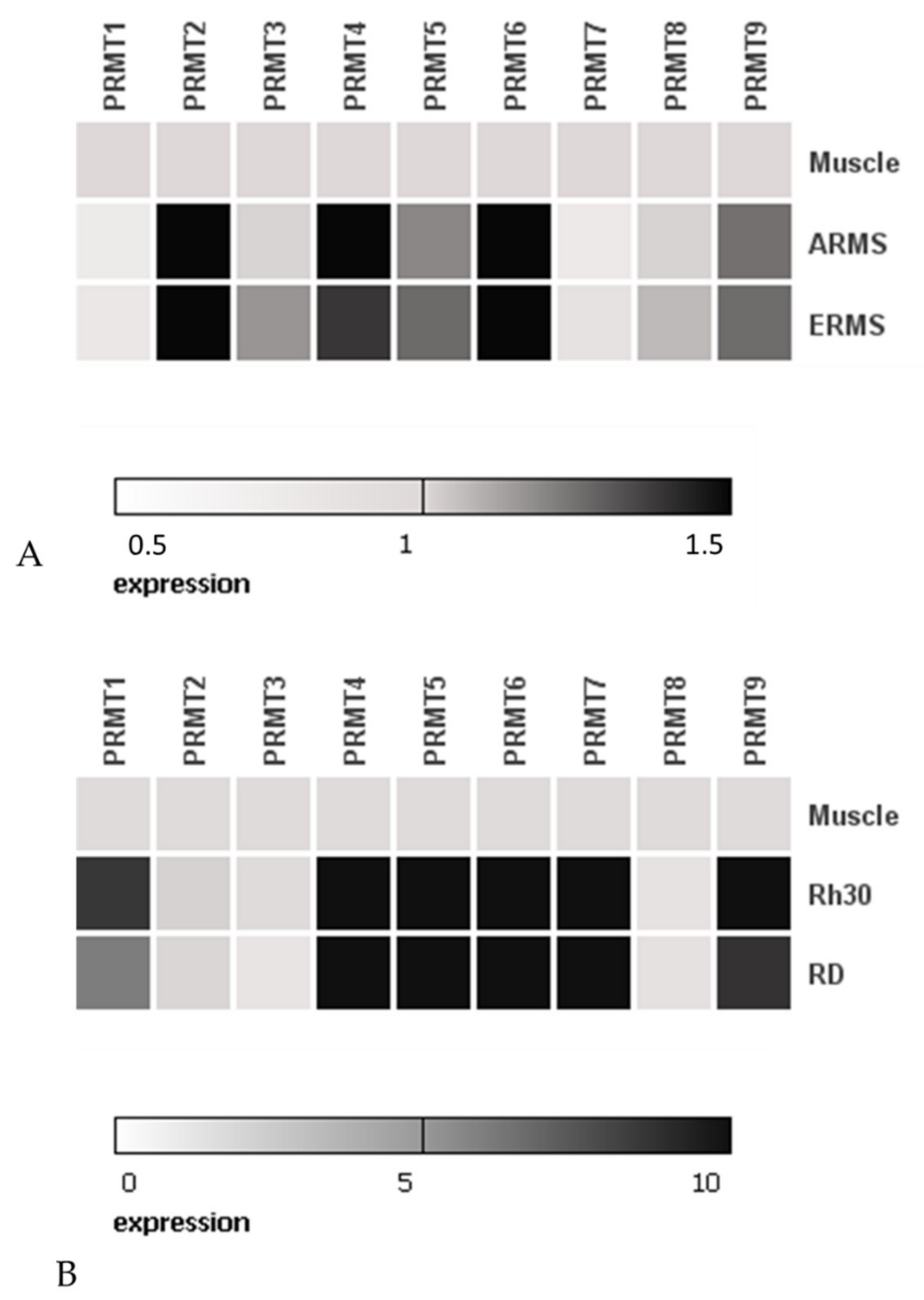
2.1. Rhabdomyosarcoma Cell Lines Show Increased Expression of Genes Encoding: PRMT1, PRMT4, PRMT5, PRMT6 and PRMT7
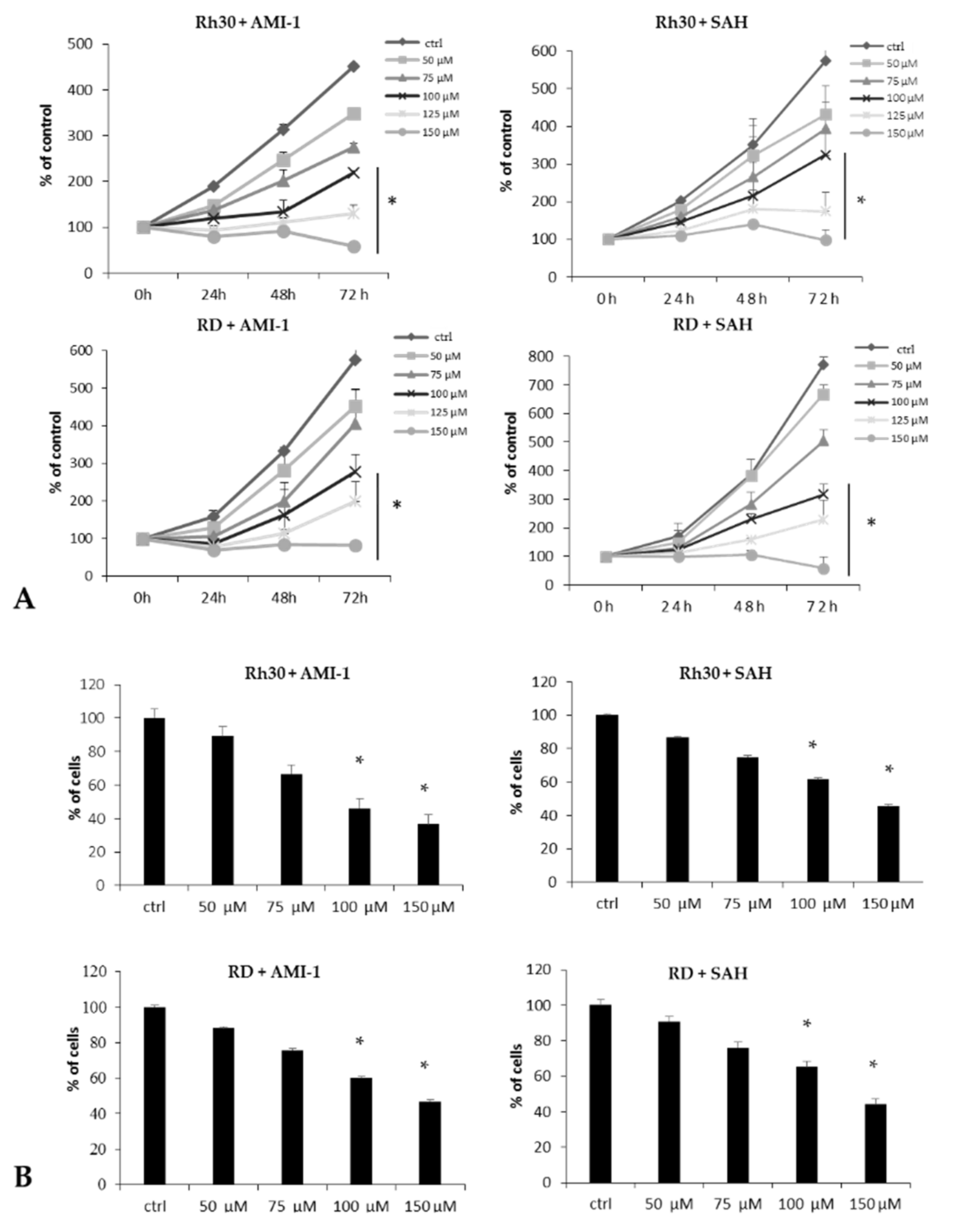
2.2. Inhibition of PRMT Activity Reduces the Viability, Proliferation and Clonogenicity of Rh30 and RD Cells
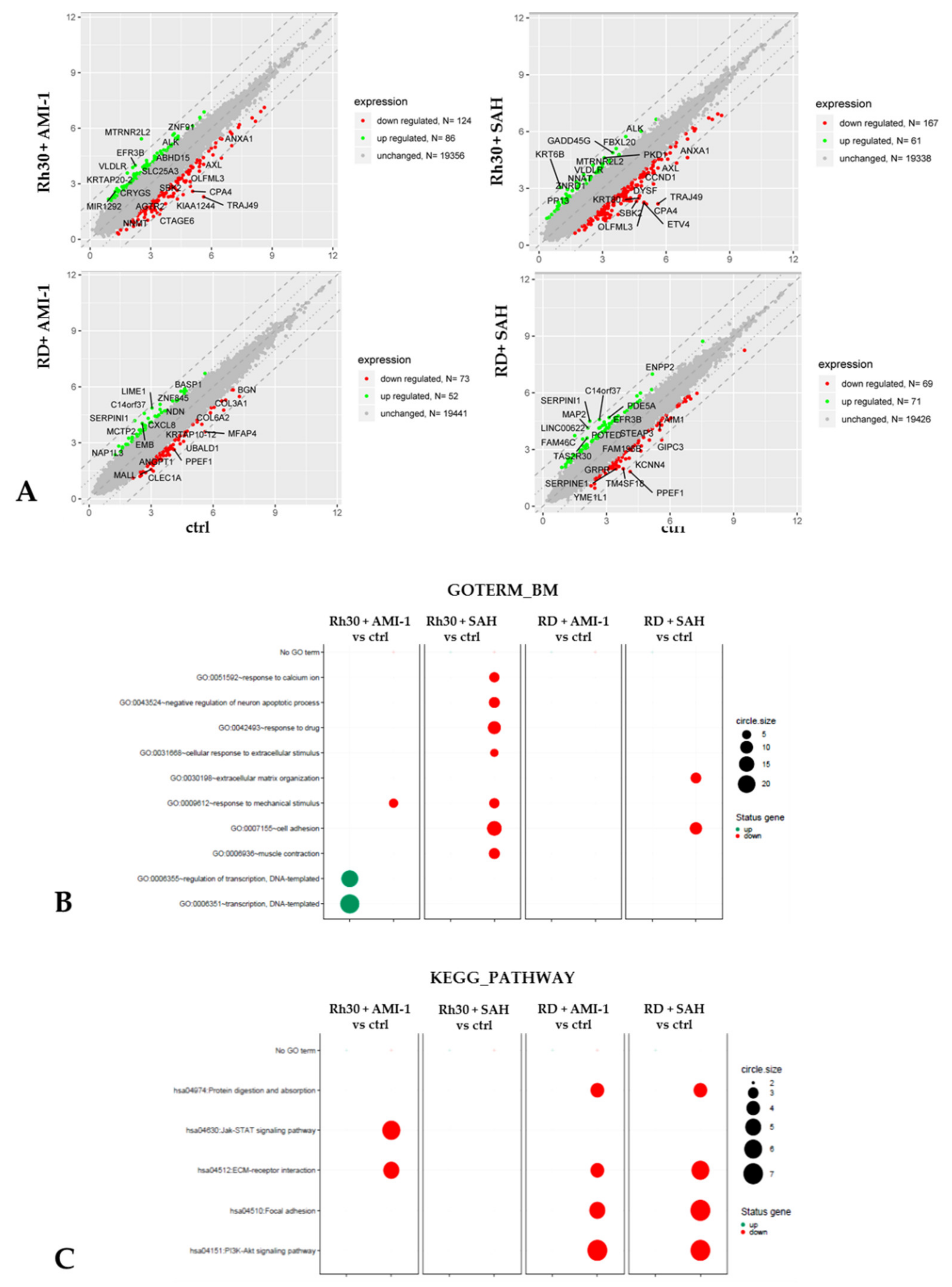
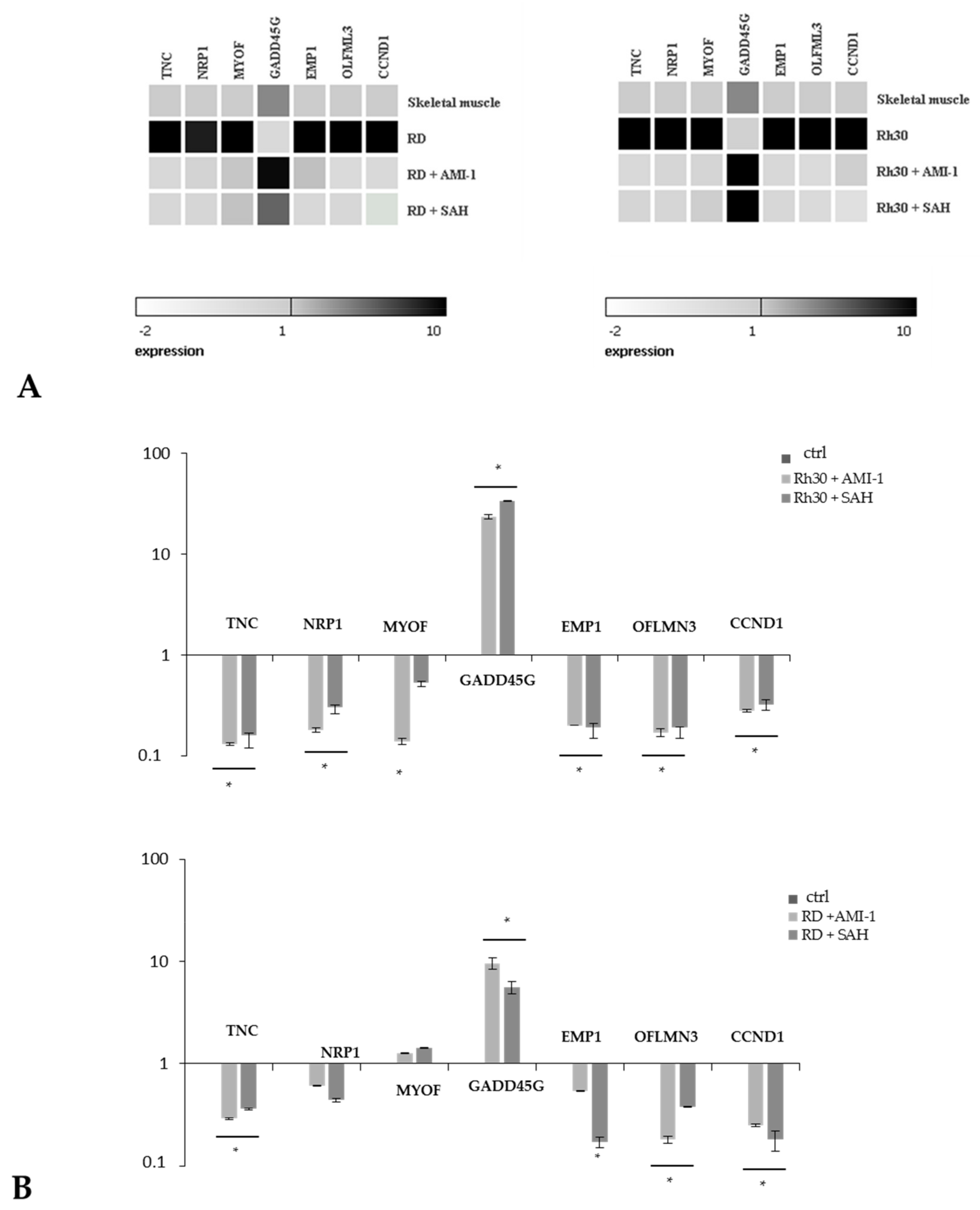
2.3. Changes in the Gene Expression Profile of RMS Cells after Exposure to AMI-1 and SAH
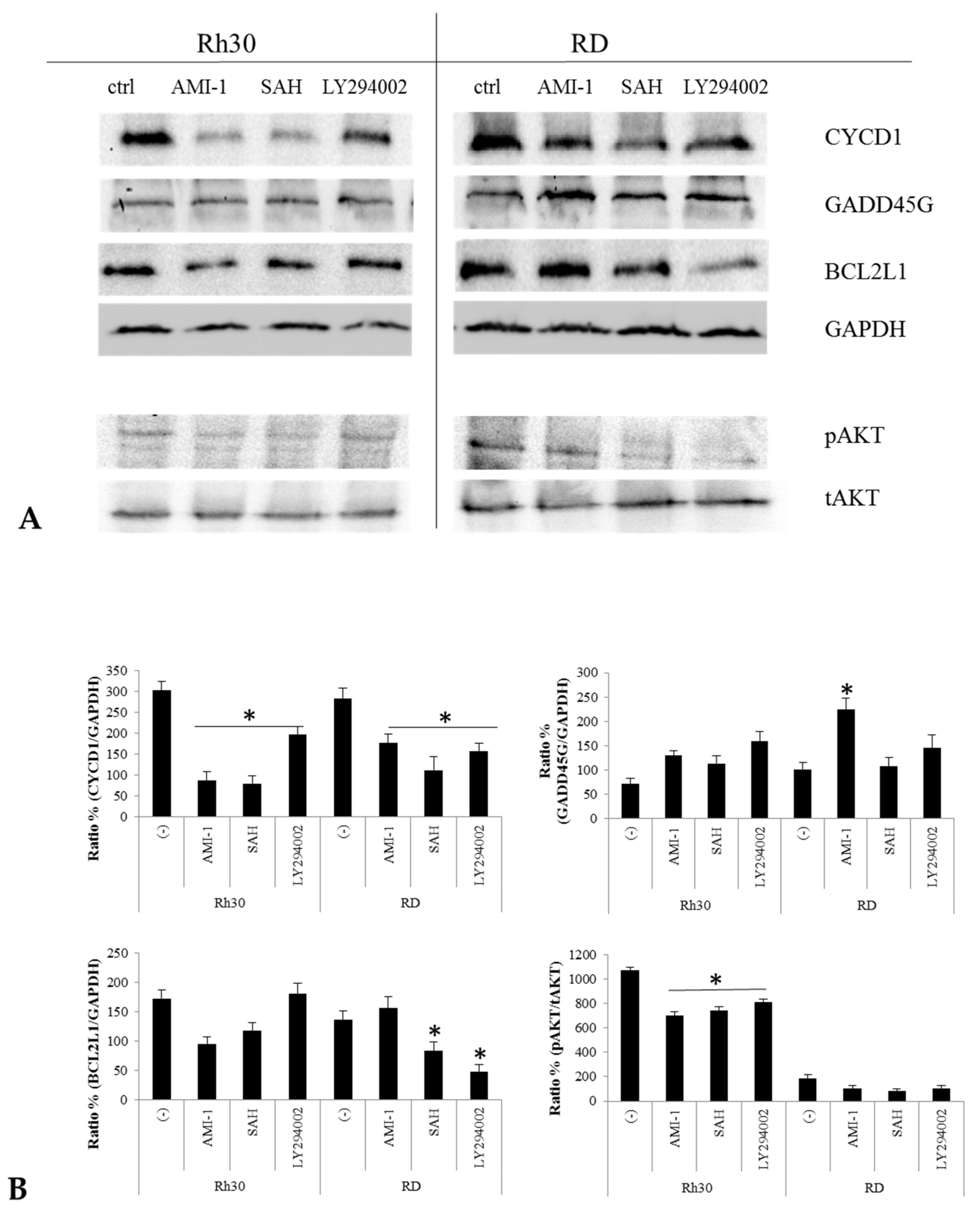
2.4. AMI-1 and SAH Decrease the Levels of Cyclin D1 and Bcl-xL and Increase the Level of GADD45G Protein by Negatively Regulating the PI3K-Akt Signalling Pathway
3. Discussion
4. Materials and Methods
4.1. Reagents
4.2. Cell Lines
4.3. Viability Test
4.4. Proliferation Test
4.5. Colony Formation Assay
4.6. Cell Cycle Analysis
4.7. Annexin V/PI Assays for Apoptosis
4.8. Oligonucleotide Microarrays
4.9. Real-Time Quantitative Reverse Transcription PCR (RQ-PCR)
4.10. Phosphorylation of Intracellular Pathway Proteins and Western Blotting
4.11. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| RMS | rhabdomyosarcoma |
| ARMS | alveolar RMS |
| ERMS | embryonal RMS |
| PRMT | protein arginine methyltransferase |
| AMI-1 | arginine methyltransferase inhibitor |
| SAH | S-adenosyl-l-homocysteine |
References
- Dagher, R.; Helman, L. Rhabdomyosarcoma: An overview. Oncologist 1999, 4, 34–44. [Google Scholar]
- Anderson, J.; Gordon, A.; Pritchard-Jones, K.; Shipley, J. Genes, chromosomes, and rhabdomyosarcoma. Genes Chromosom. Cancer 1999, 26, 275–285. [Google Scholar]
- Anderson, W.J.; Doyle, L.A. Updates from the 2020 World Health Organization Classification of Soft Tissue and Bone Tumours. Histopathology 2021, 78, 644–657. [Google Scholar]
- Salem, B.; Hofherr, S.; Turner, J.; Doros, L.; Smpokou, P. Childhood Rhabdomyosarcoma in Association With a RASopathy Clinical Phenotype and Mosaic Germline SOS1 Duplication. J. Pediatr. Hematol. Oncol. 2016, 38, e278–e282. [Google Scholar]
- Merlino, G.; Helman, L.J. Rhabdomyosarcoma—Working out the pathways. Oncogene 1999, 18, 5340–5348. [Google Scholar]
- Choi, J.H.; Ro, J.Y. The 2020 WHO Classification of Tumors of Soft Tissue: Selected Changes and New Entities. Adv. Anat. Pathol. 2021, 28, 44–58. [Google Scholar]
- Bhat, A.V.; Palanichamy Kala, M.; Rao, V.K.; Pignata, L.; Lim, H.J.; Suriyamurthy, S.; Chang, K.T.; Lee, V.K.; Guccione, E.; Taneja, R. Epigenetic Regulation of the PTEN-AKT-RAC1 Axis by G9a Is Critical for Tumor Growth in Alveolar Rhabdomyosarcoma. Cancer Res. 2019, 79, 2232–2243. [Google Scholar]
- Enβle, J.C.; Boedicker, C.; Wanior, M.; Vogler, M.; Knapp, S.; Fulda, S. Co-targeting of BET proteins and HDACs as a novel approach to trigger apoptosis in Rhabdomyosarcoma cells. Cancer Lett. 2018, 428, 160–172. [Google Scholar]
- Paulson, V.; Chandler, G.; Rakheja, D.; Galindo, R.L.; Wilson, K.; Amatruda, J.F.; Cameron, S. High-resolution array CGH identifies common mechanisms that drive embryonal rhabdomyosarcoma pathogenesis. Genes Chromosom. Cancer 2011, 50, 397–408. [Google Scholar]
- Williamson, D.; Lu, Y.J.; Gordon, T.; Sciot, R.; Kelsey, A.; Fisher, C.; Poremba, C.; Anderson, J.; Pritchard –Jones, K.; Shipley, J. Relationship between MYCN copy number and expression in rhabdomyosarcomas and correlation with adverse prognosis in the alveolar subtype. J. Clin. Oncol. 2005, 23, 880–888. [Google Scholar]
- Gryder, B.E.; Yohe, M.E.; Chou, H.C.; Zhang, X.; Marque, J.; Wachtel, M.; Shaefer, B.; Sen, N.; Song, Y.; Gualtieri, A.; et al. PAX3-FOXO1 Estabilishes Myogenic Super Enhancers and Confers BET Bromodomain Vulnerability. Cancer Discov. 2017, 7, 884–899. [Google Scholar]
- Sun, W.; Chatteriee, B.; Shem, J.F.; Patidar, R.; Song, Y.; Wang, Y.; Walker, R.L.; Pawel, B.R.; Linardic, C.M.; Houghton, P.; et al. Relationship of DNA methylation to mutational changes and transcriptional organization in fusion-positive and fusion negative rhabdomyosarcoma. Int. J. Cancer 2019, 144, 2707–2717. [Google Scholar]
- Pal, A.; Chiu, H.Y.; Taneja, R. Genetics, epigenetics and redox homeostasis in rhabdomyosarcoma: Emerging targets and therapeutics. Redox Biol. 2019, 25, 101124. [Google Scholar]
- Ludwig, A.K.; Zhang, P.; Cardoso, M.C. Modifiers and Readers of DNA Modifications and Their Impact on Genome Structure, Expression, and Stability in Disease. Front. Genet. 2016, 7, 115. [Google Scholar]
- Rodriguez-Parades, M.; Esteller, M. Cancer epigenetics reaches mainstream oncology. Nat. Med. 2011, 17, 330–339. [Google Scholar]
- Tarnowski, M.; Tkacz, M.; Czerewaty, M.; Poniewierska-Baran, A.; Grymuła, K.; Ratajczak, M.Z. 5-Azacytidine inhibits human rhabdomyosarcoma cell growth by downregulating insulin-like growth factor 2 expression and reactivating the H19 gene product miR-675, which negatively affects insulin-like growth factors and insulin signaling. Int. J. Oncol. 2015, 46, 2241–2250. [Google Scholar]
- Tarnowski, M.; Tkacz, M.; Kopytko, P.; Bujak, J.; Piotrowska, K.; Pawlik, A. Trichostatin A Inhibits Rhabdomyosarcoma Proliferation and Induces Differentiation through MyomiR Reactivation. Folia Biol. 2019, 65, 43–52. [Google Scholar]
- Litt, M.; Qiu, Y.; Huang, S. Histone arginine methylations: Their roles in chromatin dynamics and transcriptional regulation. Biosci. Rep. 2009, 2, 131–141. [Google Scholar]
- Bedford, M.T. The Family of Protein Arginine Methyltransferases. Enzymes 2006, 4, 31–50. [Google Scholar]
- Carlson, S.M.; Gozani, O. Emerging technologies to map the protein methylome. J. Mol. Biol. 2014, 426, 3350–3362. [Google Scholar]
- Gao, X.; Pan, W.S.; Dai, H.; Zhang, Y.; Wu, N.H.; Shen, Y.F. CARM1 activates myoegenin gene via PCAF in the early differentiation of TPA- induced rhabdomyosarcoma-derived cells. J. Cell Biochem. 2010, 110, 162–170. [Google Scholar]
- Blanc, R.S.; Richard, S. Arginine Methylation: The Coming of Age. Mol Cell 2017, 65, 8–24. [Google Scholar]
- Li, X.; Wang, C.; Jiang, H.; Luo, C. A patent review of arginine methyltransferase inhibitors (2010–2018). Expert Opin. Pat. 2019, 29, 97–114. [Google Scholar]
- Zhu, K.; Jiang, C.; Tao, H.; Liu, J.; Zhang, H.; Luo, C. Identification of a novel selective small-molecule inhibitor of protein arginine methyltransferase 5 (PRMT5) by virtual screening, resynthesis and biological evaluations. Bioorg. Med. Chem. Lett. 2018, 28, 1476–1483. [Google Scholar]
- Prabhu, L.; Chen, L.; Wei, H.; Demir, Ö.; Safa, A.; Zeng, L.; Amaro, R.E.; O’Neil, B.H.; Zhang, Z.Y.; Lu, T. Development of an AlphaLISA high throughput technique to screen for small molecule inhibitors targeting protein arginine methyltransferases. Mol. Biosyst. 2017, 21, 2509–2520. [Google Scholar]
- Baldwin, R.M.; Morettin, A.; Côté, J. Role of PRMTs in cancer: Could minor isoforms be leaving a mark? World J. Biol. Chem. 2014, 5, 115–129. [Google Scholar]
- Poulard, C.; Corbo, L.; Le Romancer, M. Protein arginine methylation/demethylation and cancer. Oncotarget 2016, 41, 67532–67550. [Google Scholar]
- Hadjikyriacou, A.; Yang, Y.; Espejo, A.; Bedford, M.T.; Clarke, S.G. Unique Features of Human Protein Arginine Methyltransferase 9 (PRMT9) and Its Substrate RNA Splicing Factor SF3B2. J. Biol. Chem. 2015, 290, 16723–16743. [Google Scholar]
- Zhang, B.; Dong, S.; Zhu, R.; Hu, C.; Hou, J.; Li, Y.; Zhao, Q.; Shao, X.; Bu, Q.; Li, H.; et al. Targeting protein arginine methyltransferase 5 inhibits colorectal cancer growth by decreasing arginine methylation of eIF4E and FGFR3. Oncotarget 2015, 26, 22799–22811. [Google Scholar]
- Kumar, B.; Yadav, A.; Brown, N.V.; Zhao, S.; Cipolla, M.J.; Wakely, P.E.; Schmitt, A.C.; Baiocchi, R.A.; Teknos, T.N.; Old, M. Nuclear PRMT5; cyclin D1 and IL-6 are associated with poor outcome in oropharyngeal squamous cell carcinoma patients and is in versely associated with p16-status. Oncotarget 2017, 8, 14847–14859. [Google Scholar]
- Nakai, K.; Xia, W.; Liao, H.W.; Saito, M.; Hung, M.C.; Yamaguchi, H. The role of PRMT1 in EGFR methylation and signaling in MDA-MB-468 triple-negative breast cancer cells. Breast Cancer 2018, 25, 74–80. [Google Scholar]
- Hernandez, S.; Dominiko, T. Novel Protein Arginine Methyltransferase 8 Isoform Is Essential for Cell Proliferation. J. Cell Biochem. 2016, 117, 2056–2066. [Google Scholar]
- Cheng, D.; Yadav, N.; King, R.W.; Swanson, M.S.; Weinstein, E.J.; Bedford, M.T. Small molecule regultors of protein argininę methyltransferases. J. Biol. Chem. 2004, 279, 23892–23899. [Google Scholar]
- Castellano, S.; Milite, C.; Ragno, R.; Simeoni, S.; Mia, A.; Limongeli, V.; Novellino, E.; Bauer, I.; Brosh, G.; Spannhoff, A.; et al. Design, synthesis and biological evaluation of carboxy analogues of argininę methyltransferase inhibitor 1 (AMI-1). Chem. Med. Chem. 2010, 5, 398–414. [Google Scholar]
- Fresno Vara, J.A.; Casado, E.; de Castro, J.; Ceias, P.; Belda-Iniesta, C.; Gonzalez-Baron, M. PI3K/Akt signaling pathway and cancer. Cancer Treat. Rev. 2004, 30, 193–204. [Google Scholar]
- De Luca, A.; Maiello, M.R.; D’Alessio, A.; Pergameno, M.; Normanno, N. The RAS/RAF/MEK/ERK and the PI3K/AKT signaling pathways: Role in cancer pathogenesis and implications for therapeutic approaches. Expert Opin. Targets 2012, 16, 17–27. [Google Scholar]
- Blackwell, E.; Ceman, S. Arginine methylation of RNA-binding proteins regulates cell function and differentiation. Mol. Reprod Dev. 2012, 3, 163–175. [Google Scholar]
- Sun, Q.; Fang, L.; Tang, X.; Lu, S.; Tamm, M.; Stolz, D.; Roth, M. TGF-β Upregulated Mitochondria Mass through the SMAD2/3→C/EBPβ→PRMT1 Signal Pathway in Primary Human Lung Fibroblasts. J. Immunol. 2019, 202, 37–47. [Google Scholar]
- Hye-Sook, H.; Dahee, C.; Seri, C.; Seung-Hoi, K. Roles of Protein Arginine Methyltransferases in the Control of Glucose Metabolism. Endocrinol. Metab. 2014, 29, 435–440. [Google Scholar]
- Carr, S.M.; Roworth, A.P.; Chan, C.; La Thangue, N.B. Post-translational control of transcription factors: Methylation ranks highly. FEBS J. 2015, 283, 4450–4465. [Google Scholar]
- Madea, M.; Hasegawa, H.; Sugiyama, M.; Hyodo, T.; Ito, S.; Chen, D.; Asano, E.; Masuda, A.; Hasegawa, Y.; Hamaguchi, M.; et al. Arginine methylation of ubiquitin-associated protein 2-like is required for the accurate distribution of chromosomes. FASEB J. 2016, 30, 312–323. [Google Scholar]
- Wang, S.M.; Dowhan, D.H.; Muscat, G.E.O. Epigenetic arginine methylation in breast cancer: Emerging therapeutic strategies. J. Mol. Endocrinol. 2019, 63, 223–237. [Google Scholar]
- Kleinschmidt, M.A.; de Graaf, P.; van Teeffeln, H.A.; Timmers, H.T. Cell cycle regulation by the PRMT6 arginine methyltransferase through repression of cyclin-dependent kinase inhibitors. PLoS ONE 2012. [Google Scholar] [CrossRef]
- Stopa, N.; Krebs, J.E.; Shechter, D. The PRMT5 argnine methyltransferase: Many roles in development, cancer and beyond. Cell Mol. Life Sci. 2015, 11, 2041–2059. [Google Scholar]
- Amano, Y.; Matsubara, D.; Yoshimoto, T.; Tamura, T.; Nishino, H.; Mori, Y.; Niki, T. Expression of protein arginine methyltransferase-5 in oral squamous cell carcinoma and its significance in epithelial-to-mesenchymal transition. Pathol. Int. 2018, 68, 359–366. [Google Scholar]
- Shailesh, H.; Zakaria, Z.Z.; Baiocchi, R.; Sif, S. Protein arginine methyltransferase 5 (PRMT5) dysregulation in cancer. Oncotarget 2018, 94, 36705–36718. [Google Scholar]
- Haghandish, N.; Baldwin, R.M.; Morettin, A.; Dawit, H.T.; Adhikary, H.; Masson, J.Y.; Mazroui, R.; Trinkle-Mulcahy, L.; Côté, J. PRMT7 methylates eukaryotic translation initiation factor 2α and regulates its role in stress granule formation. Mol. Biol. Cell 2019, 30, 778–793. [Google Scholar]
- Zhong, J.; Chen, Y.J.; Chen, L.; Shen, Y.Y.; Zhang, Q.H.; Yang, J.; Cao, R.X.; Zu, X.Y.; Wen, G.B. PRMT2β; a C-terminal PRMT2β, a C-terminal spilice variant of PRT2, inhibits the growth of breast cancer cells. Oncol. Rep. 2017, 38, 1303–1311. [Google Scholar]
- Zhang, B.; Dong, S.; Li, Z.; Lu, L.; Zhang, S.; Chen, X.; Cen, X.; Wu, Y. Targeting protein arginine methyltransferase 5 inhibits human hepatocellular carcinoma growth via the downregulation of beta-catenin. J. Transl. Med. 2015, 13. [Google Scholar]
- Shimura, T.; Noma, N.; Oikawa, T.; Ochiai, Y.; Kakuda, S.; Kuwahara, Y.; Takai, Y.; Takahashi, A.; Fukumoto, M. Activation of the AKT/cyclin D1/Cdk4 survival signaling pathway in radioresistant cancer stem cells. Oncogenesis 2012, 1, e12. [Google Scholar]
- Cen, L.; Hsieh, F.-C.; Lin, H.-J.; Chen, C.-S.; Qualman, S.J.; Lin, J. PDK-1/AKT pathway as a novel therapeutic target in rhabdomyosarcoma cells using OSU-03012 compound. Br. J. Cancer 2007, 97, 785–791. [Google Scholar]
- Renshaw, J.; Taylor, K.R.; Bishop, R.; Valenti, M.; De Haven Brandon, A.; Gowan, S.; Eccles, S.A.; Ruddle, R.R.; Johnson, L.D.; Raynaud, F.I.; et al. Dual blockade of the PI3K/AKT/mTOR (AZD8055) and RAS/MEK/ERK (AZD6244) pathways synergistically inhibits rhabdomyosarcoma cell growth in vitro and in vivo. Clin. Cancer Res. 2013, 19, 5940–5951. [Google Scholar]
- Zhang, B.; Chen, X.; Ge, S.; Peng, C.; Zhang, S.; Chen, X.; Liu, T.; Zhang, W. Arginine methyltransferase inhibitor-1 inhibits sarcoma viability in vitro and in vivo. Oncol. Lett. 2018, 16, 2161–2166. [Google Scholar]
- Hu, H.; Qian, K.; Ho, M.C.; Zheng, Y.G. Small Molecule Inhibtors of Protein Arginine Methyltransferases. Expert Opin. Investig. Drugs 2016, 25, 335–358. [Google Scholar]
- Siddigui, W.A.; Ahad, A.; Ahsan, H. The mystery of BCL2 family: Bcl2 proteins and apoptosis: An update. Arch. Toxicol. 2015, 89, 289–317. [Google Scholar]
- O’Hara, S.P.; Splinter, P.L.; Trussoni, C.E.; Guicciardi, E.; Splinter, N.P.; Al Suraih, M.S.; Nasser-Ghodsi, N.; Stollenwerk, D.; Gores, G.J.; La Russo, N.F. The transcription factor ETS1 promotes apoptosis resistance of senescent cholangiocytes by epigenetically up-regulating the apoptosis suppressor BCL2L1. J. Biol. Chem. 2019, 294, 18698–18713. [Google Scholar]
- Leverrier, Y.; Thomas, J.; Mathieu, A.-L.; Low, W.; Blanquer, B.; Marve, J. Role of PI3-kinase in Bcl-X induction and apoptosis inhibition mediated by IL-3 or IGF-1 in Baf-3 cells. Cell Death Differ. 1999, 6, 290–296. [Google Scholar]
- Tang, X.; Downes, C.P.; Whetton, A.D.; Owen-Lynch, P.J. Role of Phosphatidylinositol 3-Kinase and Specific Protein Kinase B Isoforms in the Suppression of Apoptosis Mediated by the Abelson Protein-tyrosine Kinase. J. Biol. Chem. 2000, 275, 13142–13148. [Google Scholar]
- Mak, S.K.; Kültz, D. Gadd45 Proteins Induce G2/M Arrest and Modulate Apoptosis in Kidney Cells Exposed to Hyperosmotic Stress. J. Biol. Chem. 2004, 279, 39075–39084. [Google Scholar]
- Lee, A.R.; Hong, K.; Choi, S.H.; Park, C.; Park, J.K.; Lee, J.I.; Bang, J.I.; Seol, D.W.; Lee, J.E.; Lee, D.R. Anti-apoptotic Regulation Contributes to the Successful Nuclear Reprogramming Using Cryopreserved Oocytes. Stem Cell Rep. 2019, 12, 545–556. [Google Scholar]
- Ying, J.; Srivastava, G.; Hsieh, W.S.; Gao, Z.; Murray, P.; Liao, S.K.; Ambinder, R.; Tao, Q. The Stress-Responsive Gene GADD45G Is a Functional Tumor Suppressor, with Its Response to Environmental Stresses Frequently Disrupted Epigenetically in MultipleTumors. Clin. Cancer Res. 2005, 11, 6442–6449. [Google Scholar]
- Auclair, Y.; Richard, S. The role of argininę methylation in the DNA damage response. DNA Repair 2013, 7, 459–465. [Google Scholar]
- Zhu, Q.S.; Ren, W.; Korchin, B.; Lahat, G.; Dicker, A.; Lu, Y.; Mills, G.; Pollock, R.E.; Lev, D. Soft tissue sarcoma cells are highly sensitive to AKT blockade: A role for p53-independent up-regulation of GADD45 alpha. Cancer Res. 2008, 68, 2895–2903. [Google Scholar]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Janisiak, J.; Kopytko, P.; Tkacz, M.; Rogińska, D.; Perużyńska, M.; Machaliński, B.; Pawlik, A.; Tarnowski, M. Protein Arginine Methyltransferase (PRMT) Inhibitors—AMI-1 and SAH Are Effective in Attenuating Rhabdomyosarcoma Growth and Proliferation in Cell Cultures. Int. J. Mol. Sci. 2021, 22, 8023. https://doi.org/10.3390/ijms22158023
Janisiak J, Kopytko P, Tkacz M, Rogińska D, Perużyńska M, Machaliński B, Pawlik A, Tarnowski M. Protein Arginine Methyltransferase (PRMT) Inhibitors—AMI-1 and SAH Are Effective in Attenuating Rhabdomyosarcoma Growth and Proliferation in Cell Cultures. International Journal of Molecular Sciences. 2021; 22(15):8023. https://doi.org/10.3390/ijms22158023
Chicago/Turabian StyleJanisiak, Joanna, Patrycja Kopytko, Marta Tkacz, Dorota Rogińska, Magdalena Perużyńska, Bogusław Machaliński, Andrzej Pawlik, and Maciej Tarnowski. 2021. "Protein Arginine Methyltransferase (PRMT) Inhibitors—AMI-1 and SAH Are Effective in Attenuating Rhabdomyosarcoma Growth and Proliferation in Cell Cultures" International Journal of Molecular Sciences 22, no. 15: 8023. https://doi.org/10.3390/ijms22158023
APA StyleJanisiak, J., Kopytko, P., Tkacz, M., Rogińska, D., Perużyńska, M., Machaliński, B., Pawlik, A., & Tarnowski, M. (2021). Protein Arginine Methyltransferase (PRMT) Inhibitors—AMI-1 and SAH Are Effective in Attenuating Rhabdomyosarcoma Growth and Proliferation in Cell Cultures. International Journal of Molecular Sciences, 22(15), 8023. https://doi.org/10.3390/ijms22158023





