Syndecan Transmembrane Domain Specifically Regulates Downstream Signaling Events of the Transmembrane Receptor Cytoplasmic Domain
Abstract
:1. Introduction
2. Results
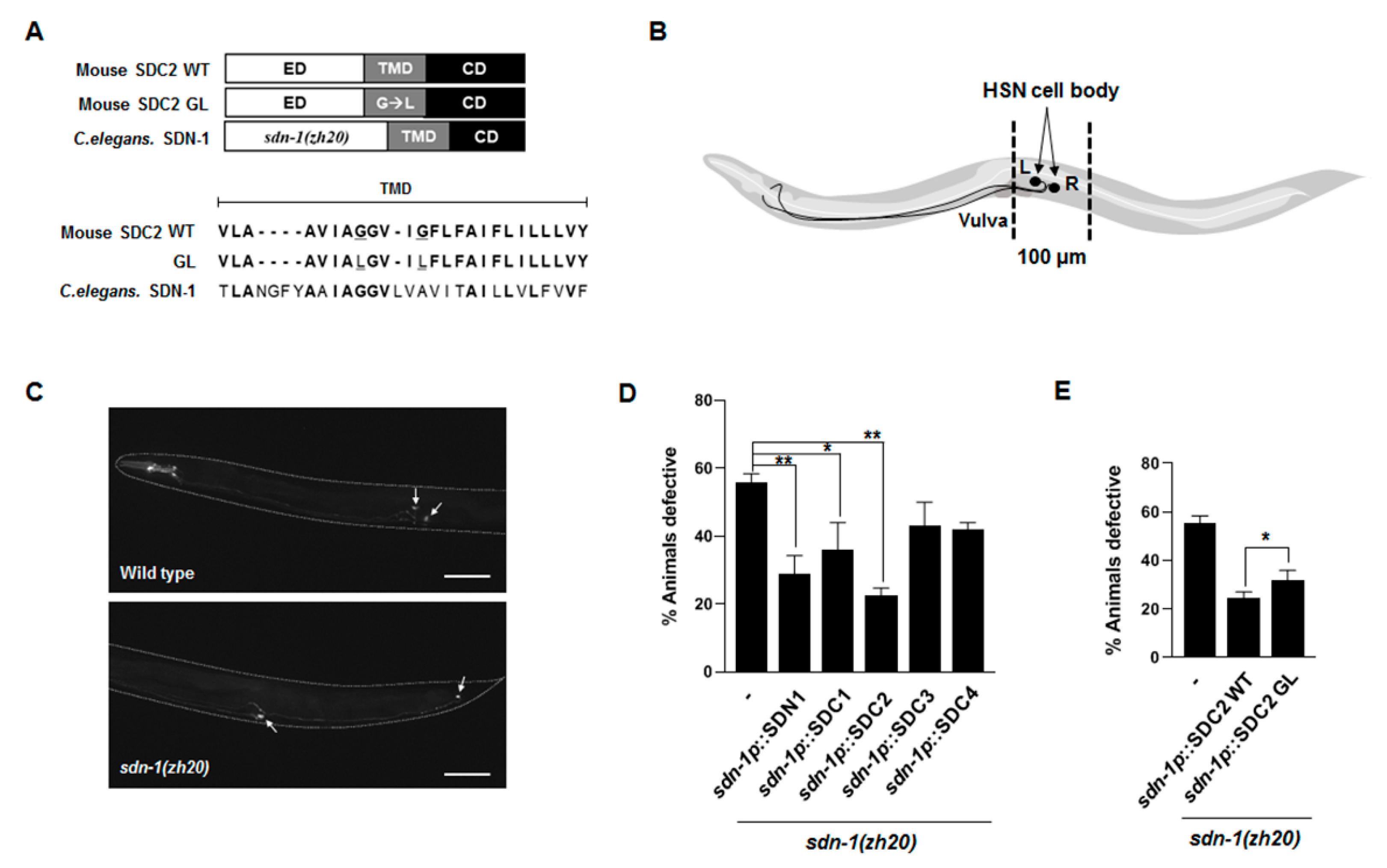
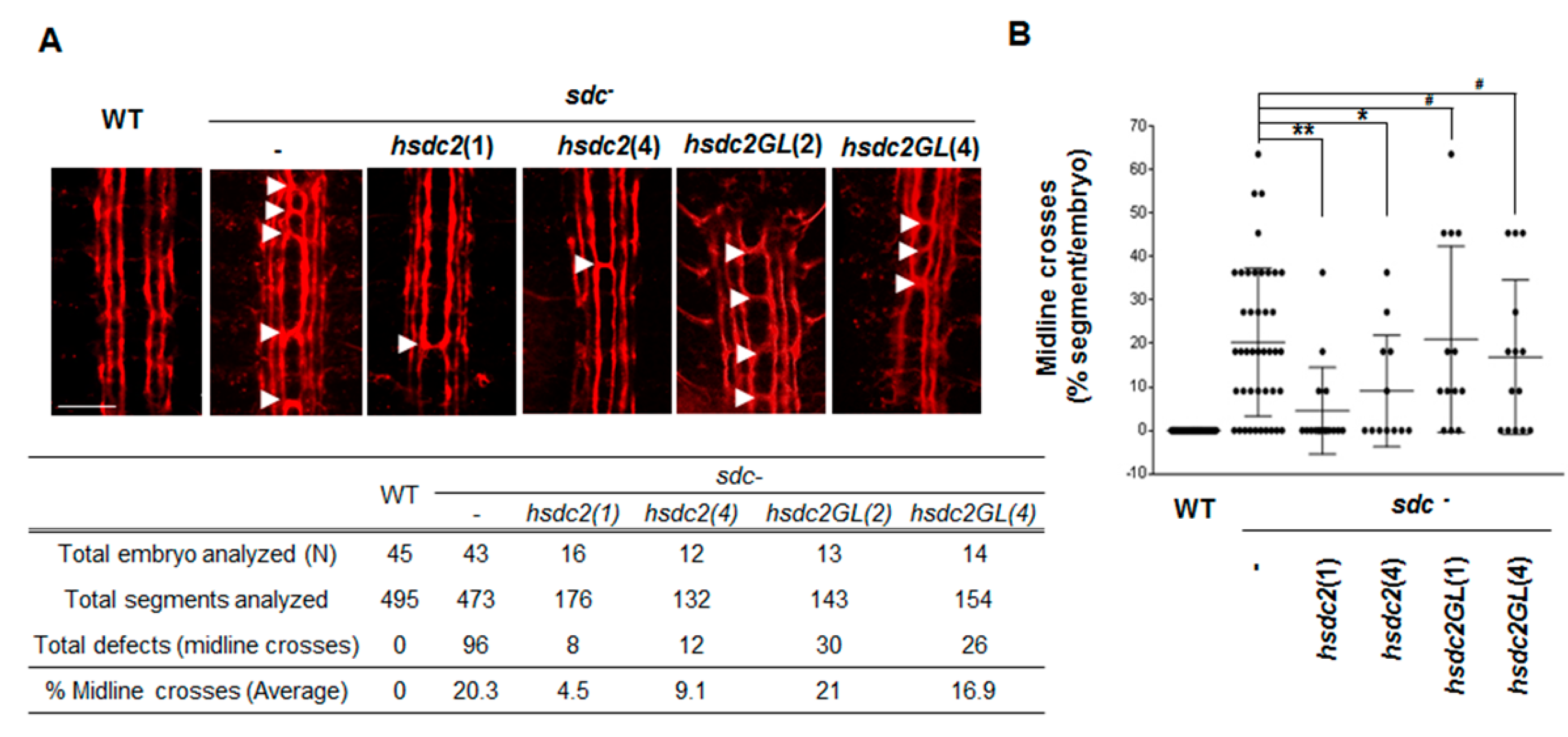
2.1. Transmembrane Domain Regulates Syndecan-2 Functions in Model Organisms
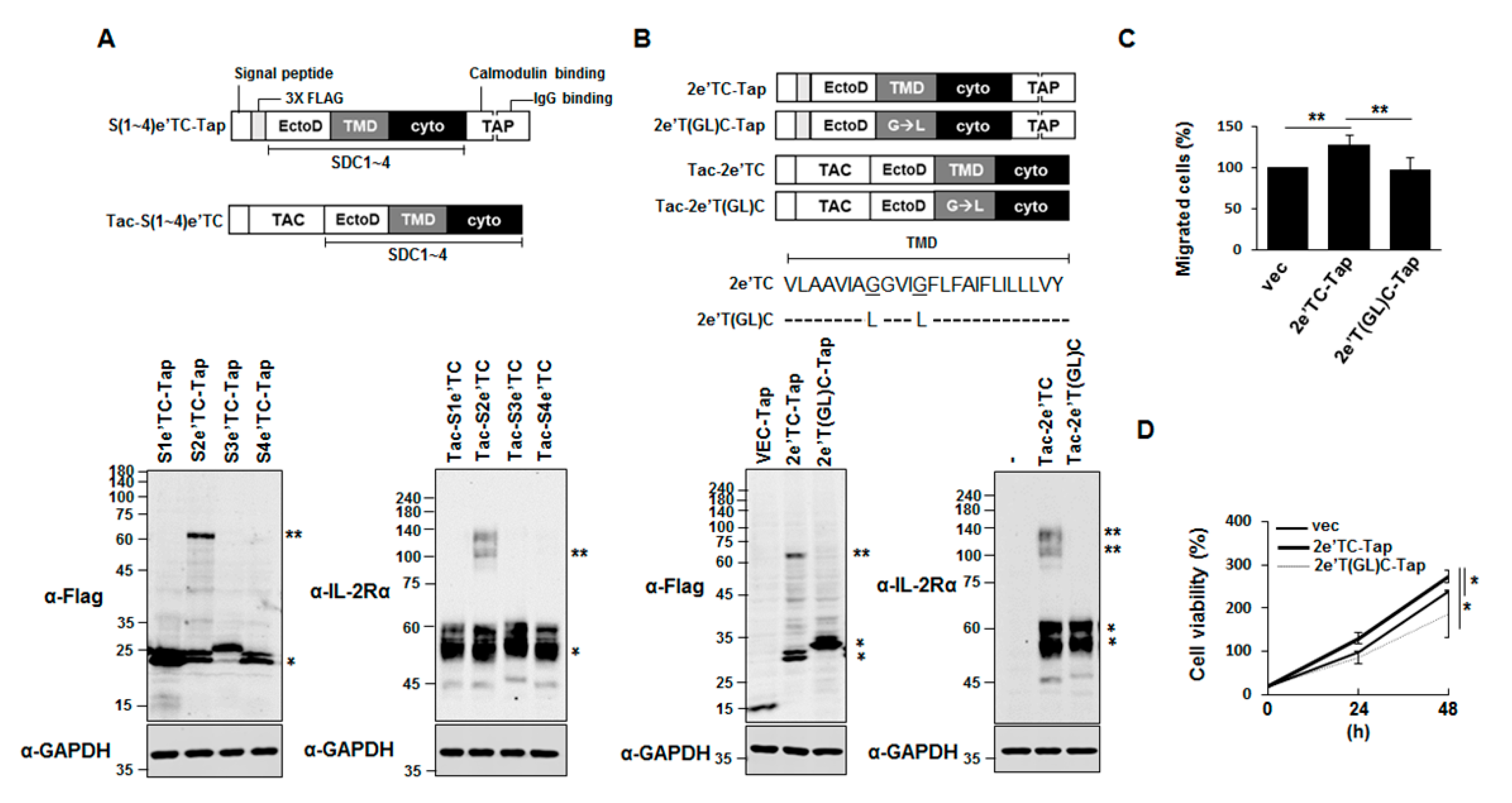
2.2. Syndecan-2 Transmembrane Domain Is Sufficient to Induce Dimer Formation of Chimeric Proteins
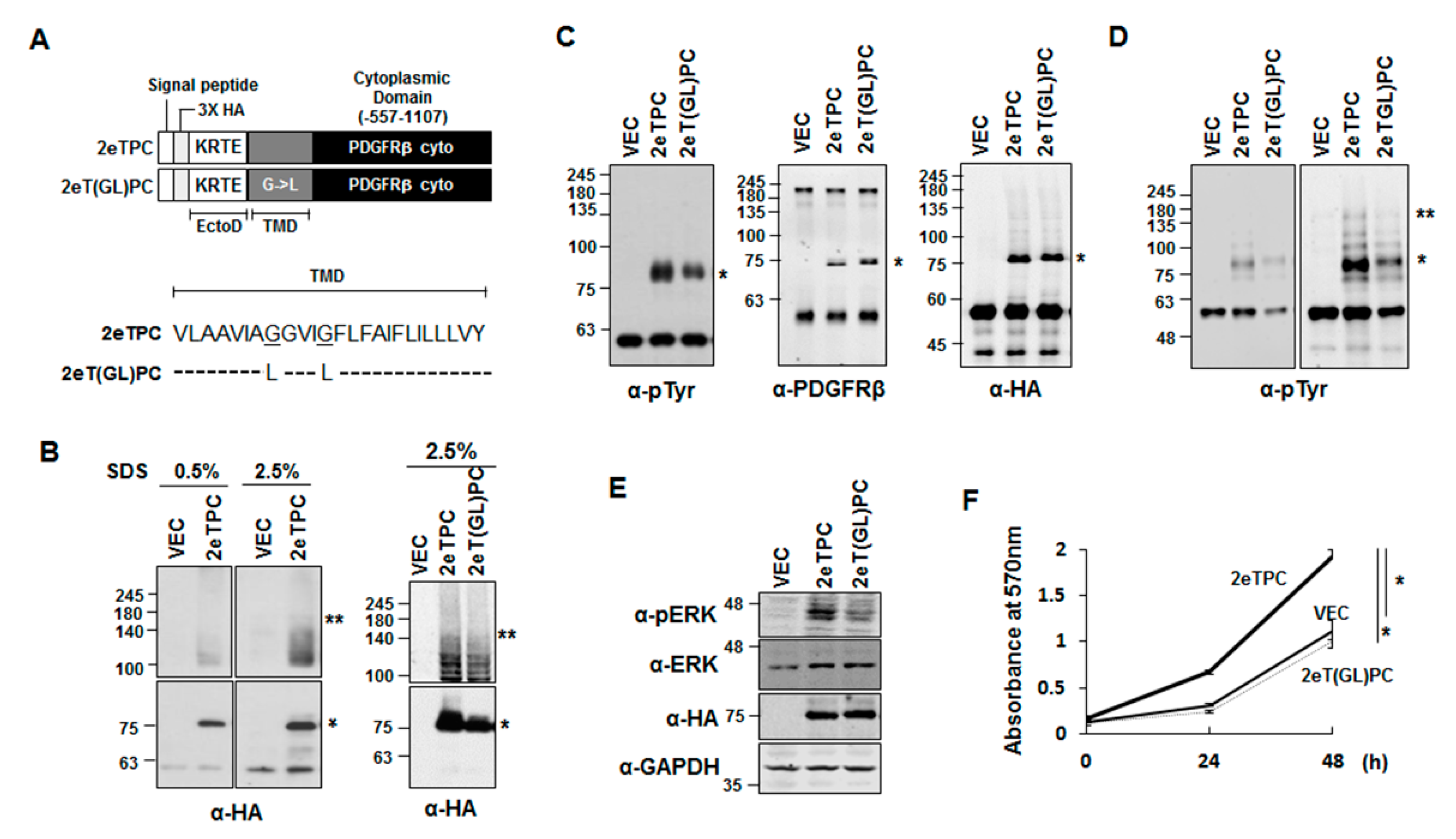
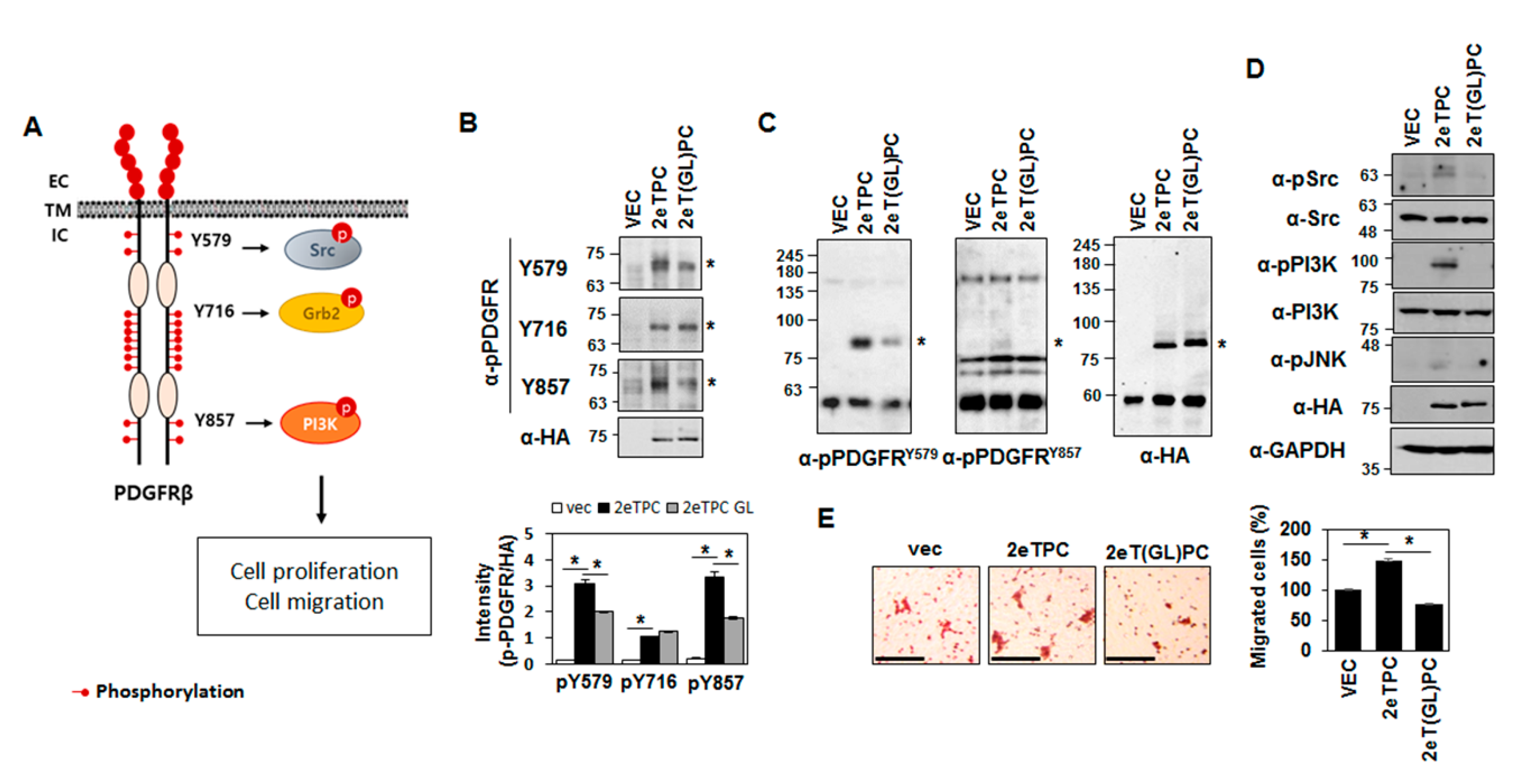
2.3. Transmembrane Domain-Induced Dimerization Regulates Phosphorylation of the Cytoplasmic Domain of a Syndecan-2-PDGFR Chimeric Protein
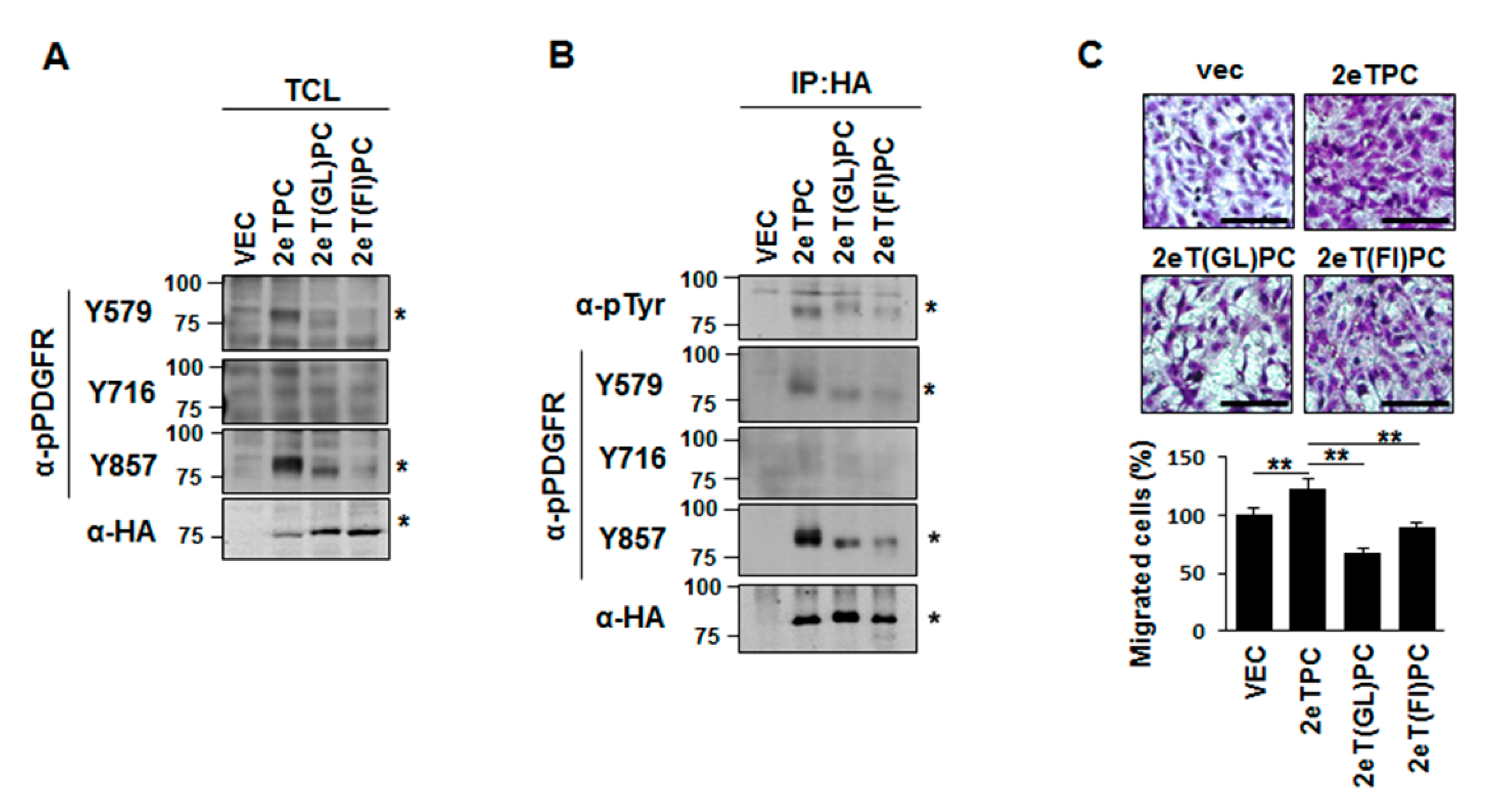
2.4. Transmembrane Domain-Induced Dimerization Specifically Regulates Phosphorylation on Tyr579 in the Cytoplasmic Domain of Syndecan-2-PDGF Receptor Chimera
3. Discussion
4. Materials and Methods
4.1. Antibodies
4.2. Plasmids
4.3. Cell Culture and Transfection
4.4. Cellular Fractionation, Immunoprecipitation, and Immunoblotting
4.5. Cell Proliferation Assay
4.6. Transwell Migration Assay
4.7. Functional Analysis of hsdc2 Mutation in DROSOPHILA
4.8. C. elegans Genetics and Transgenic Worms
4.9. HSN Cell Migration Assay
4.10. Statistical Analysis
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Locascio, L.E.; Donoghue, D.J. KIDs rule: Regulatory phosphorylation of RTKs. Trends Biochem. Sci. 2013, 38, 75–84. [Google Scholar] [CrossRef]
- Heldin, C.H.; Ostman, A.; Ronnstrand, L. Signal transduction via platelet-derived growth factor receptors. Biochim. Biophys. Acta 1998, 1378, F79–F113. [Google Scholar] [CrossRef]
- Tkachenko, E.; Rhodes, J.M.; Simons, M. Syndecans: New kids on the signaling block. Circ. Res. 2005, 96, 488–500. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Choi, S.; Lee, E.; Kwon, S.; Park, H.; Yi, J.Y.; Kim, S.; Han, I.O.; Yun, Y.; Oh, E.S. Transmembrane domain-induced oligomerization is crucial for the functions of syndecan-2 and syndecan-4. J. Biol. Chem. 2005, 280, 42573–42579. [Google Scholar] [CrossRef] [Green Version]
- Afratis, N.A.; Nikitovic, D.; Multhaupt, H.A.; Theocharis, A.D.; Couchman, J.R.; Karamanos, N.K. Syndecans—Key regulators of cell signaling and biological functions. FEBS J. 2017, 284, 27–41. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Woods, A.; Oh, E.S.; Couchman, J.R. Syndecan proteoglycans and cell adhesion. Matrix Biol. 1998, 17, 477–483. [Google Scholar] [CrossRef]
- Couchman, J.R. Syndecans: Proteoglycan regulators of cell-surface microdomains? Nat. Rev. Mol. Cell Biol. 2003, 4, 926–937. [Google Scholar] [CrossRef] [PubMed]
- Kwon, M.J.; Choi, Y.; Yun, J.H.; Lee, W.; Han, I.O.; Oh, E.S. A unique phenylalanine in the transmembrane domain strengthens homodimerization of the syndecan-2 transmembrane domain and functionally regulates syndecan-2. J. Biol. Chem. 2015, 290, 5772–5782. [Google Scholar] [CrossRef] [Green Version]
- Kwon, M.J.; Park, J.; Jang, S.; Eom, C.Y.; Oh, E.S. The Conserved Phenylalanine in the Transmembrane Domain Enhances Heteromeric Interactions of Syndecans. J. Biol. Chem. 2016, 291, 872–881. [Google Scholar] [CrossRef] [Green Version]
- Bocharov, E.V.; Lesovoy, D.M.; Pavlov, K.V.; Pustovalova, Y.E.; Bocharova, O.V.; Arseniev, A.S. Alternative packing of EGFR transmembrane domain suggests that protein-lipid interactions underlie signal conduction across membrane. Biochim. Biophys. Acta. 2016, 1858, 1254–1261. [Google Scholar] [CrossRef]
- Moriki, T.; Maruyama, H.; Maruyama, I.N. Activation of preformed EGF receptor dimers by ligand-induced rotation of the transmembrane domain. J. Mol. Biol. 2001, 311, 1011–1026. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.; Kim, S.; Lee, J.; Ko, S.G.; Lee, W.; Han, I.O.; Woods, A.; Oh, E.S. The oligomeric status of syndecan-4 regulates syndecan-4 interaction with alpha-actinin. Eur. J. Cell Biol. 2008, 87, 807–815. [Google Scholar] [CrossRef]
- Li, R.; Mitra, N.; Gratkowski, H.; Vilaire, G.; Litvinov, R.; Nagasami, C.; Weisel, J.W.; Lear, J.D.; DeGrado, W.F.; Bennett, J.S. Activation of integrin alphaIIbbeta3 by modulation of transmembrane helix associations. Science 2003, 300, 795–798. [Google Scholar] [CrossRef] [PubMed]
- Spring, J.; Paine-Saunders, S.E.; Hynes, R.O.; Bernfield, M. Drosophila syndecan: Conservation of a cell-surface heparan sulfate proteoglycan. Proc. Natl. Acad. Sci. USA 1994, 91, 3334–3338. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rhiner, C.; Gysi, S.; Frohli, E.; Hengartner, M.O.; Hajnal, A. Syndecan regulates cell migration and axon guidance in C. elegans. Development 2005, 132, 4621–4633. [Google Scholar] [CrossRef] [Green Version]
- Chanana, B.; Steigemann, P.; Jackle, H.; Vorbruggen, G. Reception of Slit requires only the chondroitin-sulphate-modified extracellular domain of Syndecan at the target cell surface. Proc. Natl. Acad. Sci. USA 2009, 106, 11984–11988. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, C.; Lau, T.L.; Ulmer, T.S.; Ginsberg, M.H. Interactions of platelet integrin alphaIIb and beta3 transmembrane domains in mammalian cell membranes and their role in integrin activation. Blood 2009, 113, 4747–4753. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hoch, R.V.; Soriano, P. Roles of PDGF in animal development. Development 2003, 130, 4769–4784. [Google Scholar] [CrossRef] [Green Version]
- Claesson-Welsh, L. Platelet-derived growth factor receptor signals. J. Biol. Chem. 1994, 269, 32023–32026. [Google Scholar] [CrossRef]
- Kazlauskas, A.; Cooper, J.A. Phosphorylation of the PDGF receptor beta subunit creates a tight binding site for phosphatidylinositol 3 kinase. EMBO J. 1990, 9, 3279–3286. [Google Scholar] [CrossRef]
- Sprang, S.R. G protein mechanisms: Insights from structural analysis. Annu. Rev. Biochem. 1997, 66, 639–678. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Scheerer, P.; Park, J.H.; Hildebrand, P.W.; Kim, Y.J.; Krauss, N.; Choe, H.W.; Hofmann, K.P.; Ernst, O.P. Crystal structure of opsin in its G-protein-interacting conformation. Nature 2008, 455, 497–502. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.; Kwon, M.J.; Lim, Y.; Yun, J.H.; Lee, W.; Oh, E.S. Trans-regulation of Syndecan Functions by Hetero-oligomerization. J. Biol. Chem. 2015, 290, 16943–16953. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arama, E.; Steller, H. Detection of apoptosis by terminal deoxynucleotidyl transferase-mediated dUTP nick-end labeling and acridine orange in Drosophila embryos and adult male gonads. Nat. Protoc. 2006, 1, 1725–1731. [Google Scholar] [CrossRef]
- Brenner, S. The genetics of Caenorhabditis elegans. Genetics 1974, 77, 71–94. [Google Scholar] [CrossRef]
- Sze, J.Y.; Zhang, S.; Li, J.; Ruvkun, G. The C. elegans POU-domain transcription factor UNC-86 regulates the tph-1 tryptophan hydroxylase gene and neurite outgrowth in specific serotonergic neurons. Development 2002, 129, 3901–3911. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hwang, J.; Jang, B.; Kim, A.; Lee, Y.; Lee, J.; Kim, C.; Kim, J.; Moon, K.M.; Kim, K.; Wagle, R.; et al. Syndecan Transmembrane Domain Specifically Regulates Downstream Signaling Events of the Transmembrane Receptor Cytoplasmic Domain. Int. J. Mol. Sci. 2021, 22, 7918. https://doi.org/10.3390/ijms22157918
Hwang J, Jang B, Kim A, Lee Y, Lee J, Kim C, Kim J, Moon KM, Kim K, Wagle R, et al. Syndecan Transmembrane Domain Specifically Regulates Downstream Signaling Events of the Transmembrane Receptor Cytoplasmic Domain. International Journal of Molecular Sciences. 2021; 22(15):7918. https://doi.org/10.3390/ijms22157918
Chicago/Turabian StyleHwang, Jisun, Bohee Jang, Ayoung Kim, Yejin Lee, Joonha Lee, Chungho Kim, Jinmahn Kim, Kyeong Min Moon, Kyuhyung Kim, Ram Wagle, and et al. 2021. "Syndecan Transmembrane Domain Specifically Regulates Downstream Signaling Events of the Transmembrane Receptor Cytoplasmic Domain" International Journal of Molecular Sciences 22, no. 15: 7918. https://doi.org/10.3390/ijms22157918
APA StyleHwang, J., Jang, B., Kim, A., Lee, Y., Lee, J., Kim, C., Kim, J., Moon, K. M., Kim, K., Wagle, R., Song, Y.-H., & Oh, E.-S. (2021). Syndecan Transmembrane Domain Specifically Regulates Downstream Signaling Events of the Transmembrane Receptor Cytoplasmic Domain. International Journal of Molecular Sciences, 22(15), 7918. https://doi.org/10.3390/ijms22157918






