In Search of Effective Anticancer Agents—Novel Sugar Esters Based on Polyhydroxyalkanoate Monomers
Abstract
:1. Introduction
2. Materials and Methods
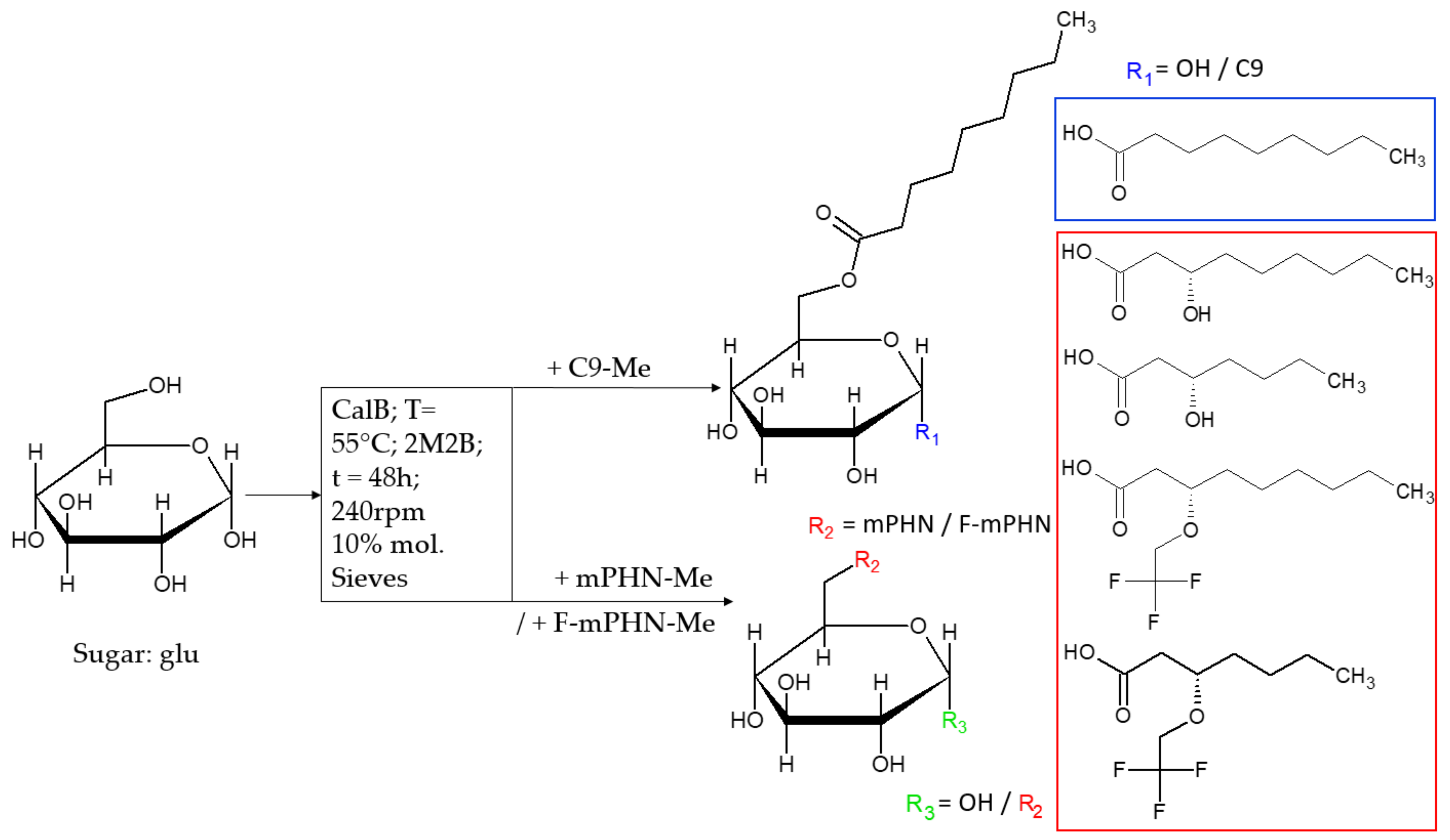
2.1. Preparation of Unmodified and Modified Polyhydroxynonanoate Monomers
2.2. Synthesis of Sugar Fatty Acid Esters (SFAE)
2.3. HPLC Analysis
2.4. Preparative HPLC
2.5. 1H NMR and 19F NMR Spectroscopy
2.6. IR Spectroscopy
2.7. Cell Cultures
2.8. MTT Cytotoxicity Assay
2.9. Fluorescent Staining
2.10. Transmigration Assay
2.11. Cell Structures Imaging
3. Results
3.1. Synthesis and Modification of Polyhydroxynonanoate Monomers (mPHN)
3.2. Structural Analysis of Synthesized Sugar Esters
3.3. MTT Assay Indicating Anti-Proliferative Properties of C9, mPHN and F-mPHN Based SFAE
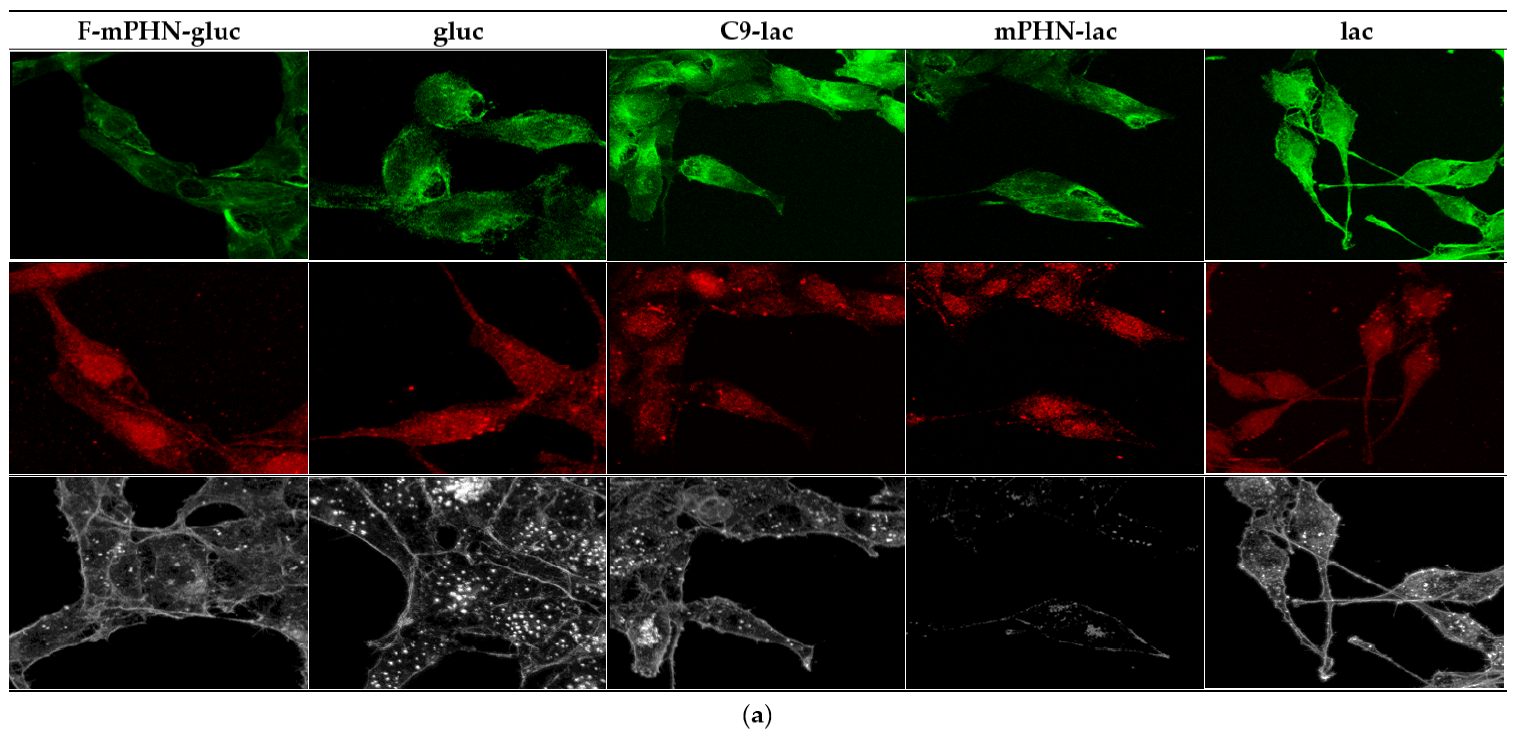
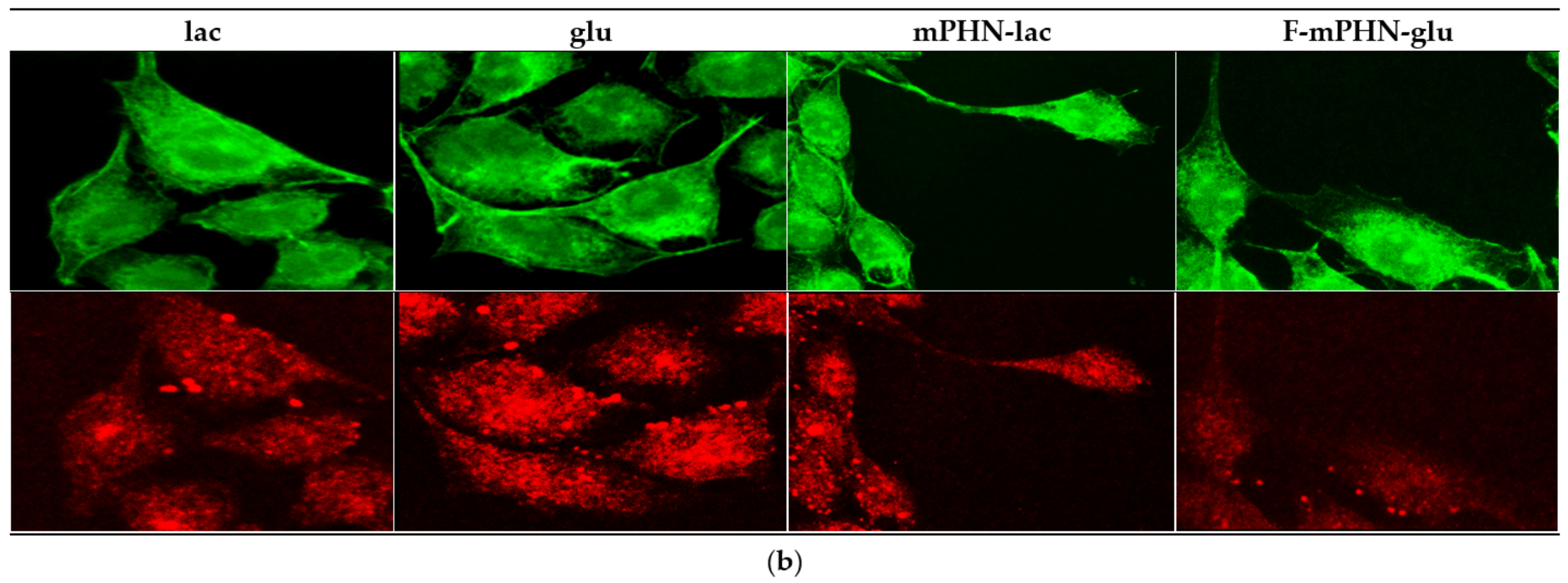
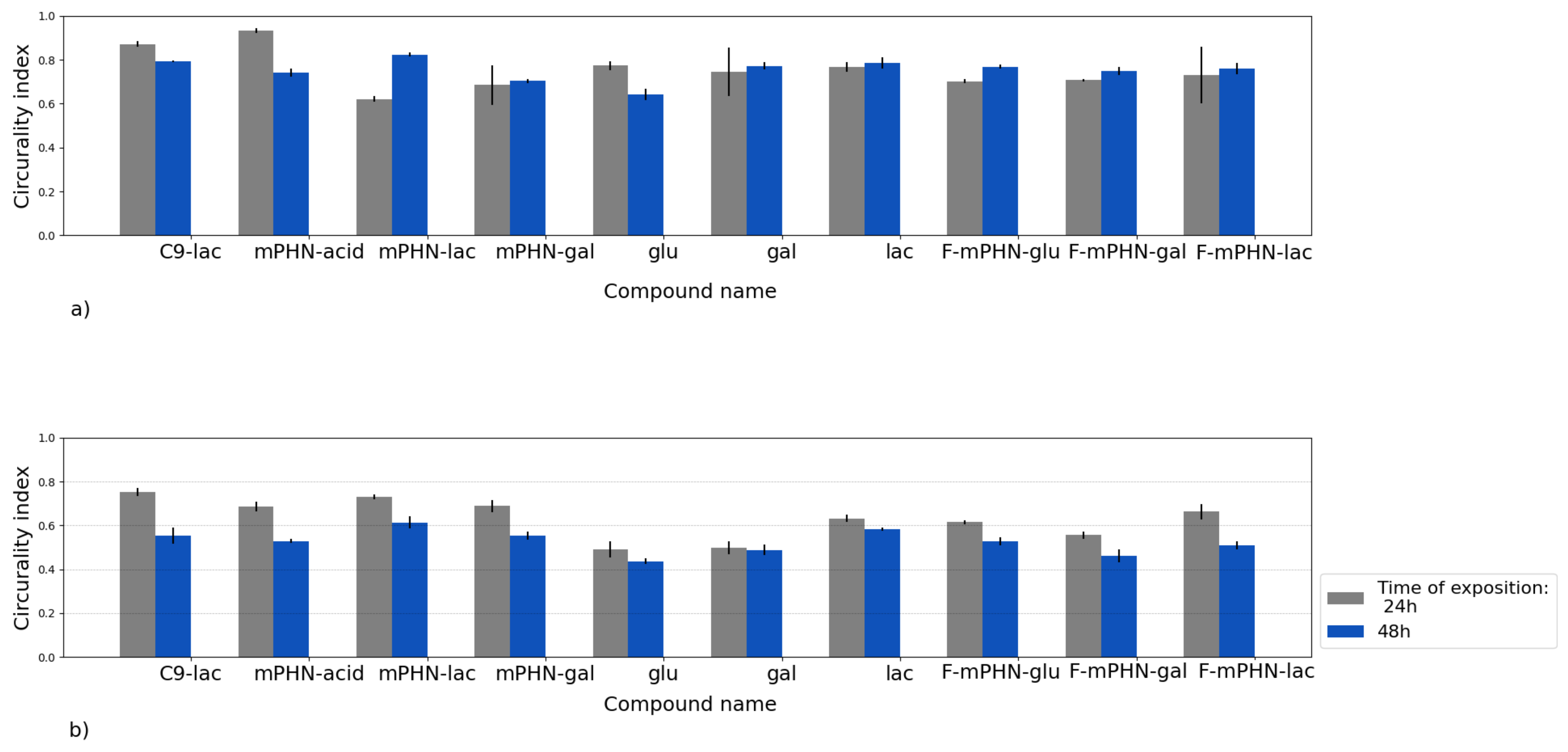
3.4. Cell Structures Imaging Indicate Reorganization of Intermediate Filaments in the Presence of a Selected SFAE
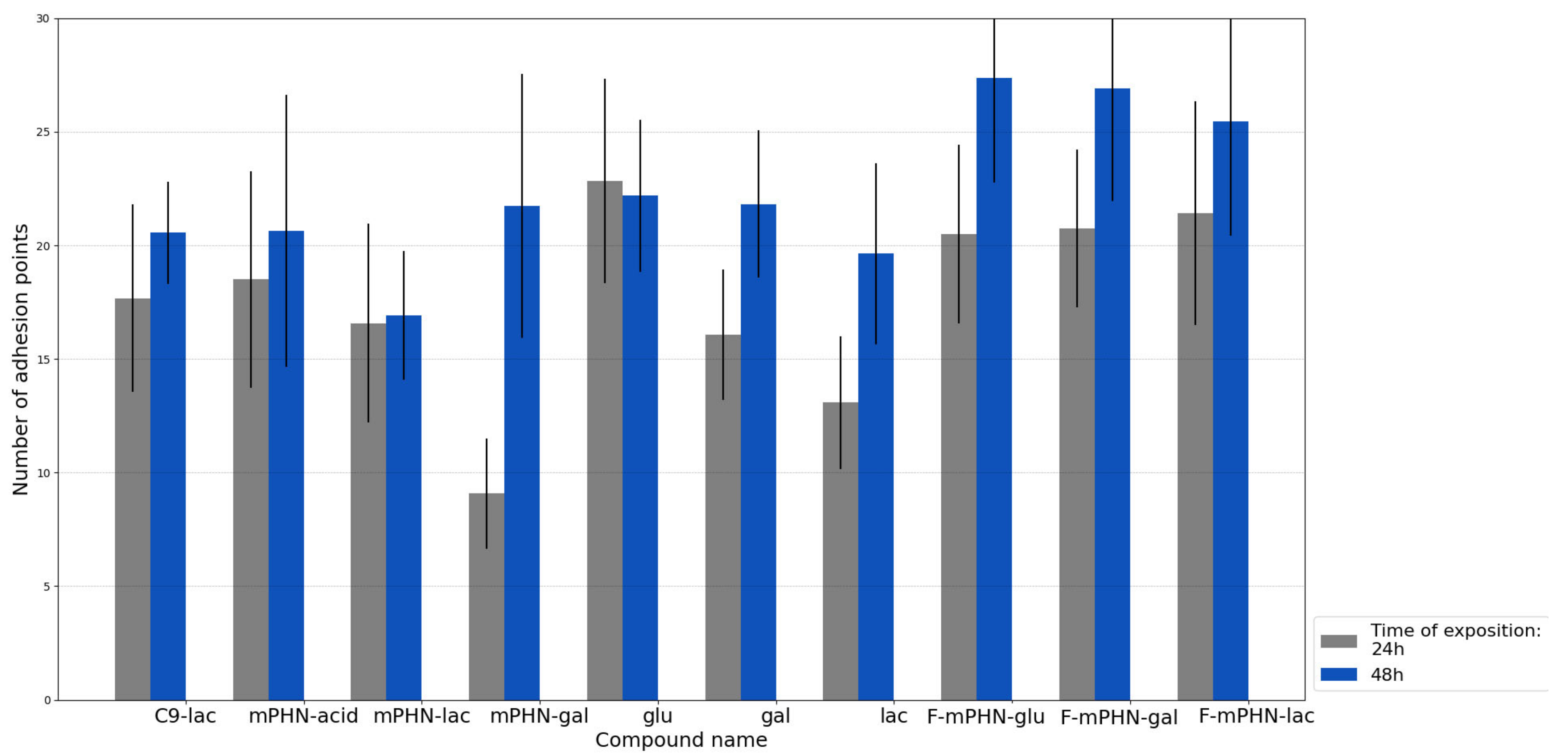
3.5. Inhibition of Transmigration Caused by C9, mPHN-lac and mPHN-glu
4. Discussion
5. Conclusions
6. Patents
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| C9 | nonanoic acid |
| C9-Me | nonanoic methyl ester |
| PHN | poly-(3R)-hydroxynonanoate-co-(3R)-hydroxyheptanoate |
| R3OH-C9 | (3R)-hydroxynonanoate |
| R3OH-C7 | (3R)-hydroxyheptanoate |
| mPHN | mixture of PHN monomers hydroxyacids: (3R)-hydroxynonanoate and (3R)-hydroxyheptanoate |
| mPHN-Me | mixture of PHN monomemers methyl esters: (3R)-hydroxynonanoate methyl esters |
| R3OH-C9-Me | (3R)-hydroxynonanoate methyl esters |
| R3OH-C7-Me | (3R)-hydroxyheptanoate methyl esters |
| F-mPHN-Me | mixture of fluorinated PHN monomers: (3R)-3-(2,2,2-trifluoroethoxy)nonanoate methyl esters and (3R)-3-(2,2,2-trifluoroethoxy)heptanoate methyl esters |
| glu | α-D-glucopiranose |
| gal | β-d-galactopyranose |
| lac | β-D-Galactopyranosyl-(1→4)-D-glucopiranose |
| C9-glu | glucose nonanoic ester |
| C9-gal | galactose nonanoic ester |
| C9-lac | lactose nonanoic ester |
| mPHN-glu | mixture of (3R)-hydroxynonanoate glucose esters and (3R)-hydroxyheptanoate glucose esters |
| mPHN-gal | mixture of (3R)-hydroxynonanoate galactose esters and (3R)-hydroxyheptanoate galactose esters |
| mPHN-lac | mixture of (3R)-hydroxynonanoate lactose esters and (3R)-hydroxyheptanoate lactose esters |
| F-mPHN-glu | mixture of (3R)-3-(2,2,2-trifluoroethoxy)nonanoate glucose esters and (3R)-3-(2,2,2-trifluoroethoxy)heptanoate glucose esters |
| F-mPHN-gal | mixture of (3R)-3-(2,2,2-trifluoroethoxy)nonanoate galactose esters and (3R)-3-(2,2,2-trifluoroethoxy)heptanoate galactose esters |
| F-mPHN-lac | mixture of (3R)-3-(2,2,2-trifluoroethoxy)nonanoate lactose esters and (3R)-3-(2,2,2-trifluoroethoxy)heptanoate lactose esters |
References
- WHO Cancer. 2021. Available online: https://www.who.int/health-topics/cancer (accessed on 15 March 2021).
- Miller, K.D.; Nogueira, L.; Mariotto, A.B.; Rowland, J.H.; Yabroff, K.R.; Alfano, C.M.; Jemal, A.; Kramer, J.L.; Siegel, R.L. Cancer treatment and survivorship statistics, 2019. CA Cancer J. Clin. 2019, 69, 363–385. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ferlay, J.; Colombet, M.; Soerjomataram, I.; Dyba, T.; Randi, G.; Bettio, M.; Gavin, A.; Visser, O.; Bray, F. Cancer incidence and mortality patterns in Europe: Estimates for 40 countries and 25 major cancers in 2018. Eur. J. Cancer 2018, 103, 356–387. [Google Scholar] [CrossRef] [PubMed]
- Babaei, N.; Salamci, M.U. Mixed therapy in cancer treatment for personalized drug administration using model reference adaptive control. Eur. J. Control 2019, 50, 117–137. [Google Scholar] [CrossRef]
- Rao, S.; Sclafani, F.; Eng, C.; Adams, R.A.; Guren, M.G.; Sebag-Montefiore, D.; Benson, A.; Bryant, A.; Peckitt, C.; Segelov, E.; et al. International rare cancers initiative multicenter randomized phase II trial of cisplatin and fluorouracil versus carboplatin and paclitaxel in advanced anal cancer: InterAAct. J. Clin. Oncol. 2020, 38, 2510–2518. [Google Scholar] [CrossRef] [PubMed]
- Meric-Bernstam, F.; Gonzalez-Angulo, A.M. Targeting the mTOR signaling network for cancer therapy. J. Clin. Oncol. 2009, 27, 2278–2287. [Google Scholar] [CrossRef] [PubMed]
- Kalia, V.C.; Patel, S.K.S.; Cho, B.-K.; Wood, T.K.; Lee, J.-K. Emerging applications of bacteria as antitumor agents. Semin. Cancer Biol. 2021. [Google Scholar] [CrossRef] [PubMed]
- Kovvasu, S.P.; Kunamaneni, P.; Joshi, R. Self-emulsifying Drug Delivery Systems and their Marketed Products: A Review. Asian J. Pharm. 2019, 13, 73–84. [Google Scholar]
- Staroń, J.; Dąbrowski, J.M.; Cichoń, E.; Guzik, M. Lactose esters: Synthesis and biotechnological applications. Crit. Rev. Biotechnol. 2018, 38, 245–258. [Google Scholar] [CrossRef]
- El-Laithy, H.M.; Shoukry, O.; Mahran, L.G. Novel sugar esters proniosomes for transdermal delivery of vinpocetine: Preclinical and clinical studies. Eur. J. Pharm. Biopharm. 2011, 77, 43–55. [Google Scholar] [CrossRef]
- Liang, M.Y.; Banwell, M.G.; Wang, Y.; Lan, P. Effect of Variations in the Fatty Acid Residue of Lactose Monoesters on Their Emulsifying Properties and Biological Activities. J. Agric. Food Chem. 2018, 66, 12594–12603. [Google Scholar] [CrossRef]
- Enayati, M.; Gong, Y.; Goddard, J.M.; Abbaspourrad, A. Synthesis and characterization of lactose fatty acid ester biosurfactants using free and immobilized lipases in organic solvents. Food Chem. 2018, 266, 508–513. [Google Scholar] [CrossRef]
- Marquis, R.E.; Clock, S.A.; Mota-Meira, M. Fluoride and organic weak acids as modulators of microbial physiology. FEMS Microbiol. Rev. 2003, 26, 493–510. [Google Scholar] [CrossRef]
- Lucarini, S.; Fagioli, L.; Campana, R.; Cole, H.; Duranti, A.; Baffone, W.; Vllasaliu, D.; Casettari, L. Unsaturated fatty acids lactose esters: Cytotoxicity, permeability enhancement and antimicrobial activity. Eur. J. Pharm. Biopharm. 2016, 107, 88–96. [Google Scholar] [CrossRef]
- Lucarini, S.; Fagioli, L.; Cavanagh, R.; Liang, W.; Perinelli, D.R.; Campana, M.; Stolnik, S.; Lam, J.K.W.; Casettari, L.; Duranti, A. Synthesis, structure–activity relationships and in vitro toxicity profile of lactose-based fatty acid monoesters as possible drug permeability enhancers. Pharmaceutics 2018, 10, 81. [Google Scholar] [CrossRef] [Green Version]
- Ferrer, M.; Perez, G.; Plou, F.J.; Castell, J.V.; Ballesteros, A. Antitumour activity of fatty acid maltotriose esters obtained by enzymatic synthesis. Biotechnol. Appl. Biochem. 2005, 42 Pt 1, 35–39. [Google Scholar] [CrossRef] [Green Version]
- Okabe, S.; Suganuma, M.; Tada, Y.; Ochiai, Y.; Sueoka, E.; Kohya, H.; Shibata, A.; Takahashi, M.; Mizutani, M.; Matsuzaki, T.; et al. Disaccharide esters screened for inhibition of tumor necrosis factor-α release are new anti-cancer agents. Jpn. J. Cancer Res. 1999, 90, 669–676. [Google Scholar] [CrossRef]
- Yanagita, R.C.; Kobashi, K.; Ogawa, C.; Ashida, Y.; Yamaashi, H.; Kawanami, Y. Anti-proliferative activity of 6-O-acyl-D-allose against the human leukemia MOLT-4F cell line. Biosci. Biotechnol. Biochem. 2014, 78, 190–194. [Google Scholar] [CrossRef]
- Kasuya, M.C.Z.; Ito, A.; Cusi, R.; Sato, T.; Hatanaka, K. Cellular uptake and saccharide chain elongation of “fluoro- amphiphilic” glycosides. Chem. Lett. 2005, 34, 856–857. [Google Scholar] [CrossRef]
- Yerien, D.E.; Bonesi, S.; Postigo, A. Fluorination methods in drug discovery. Org. Biomol. Chem. 2016, 14, 8398–8427. [Google Scholar] [CrossRef] [Green Version]
- Murphy, C.D.; Sandford, G. Recent advances in fluorination techniques and their anticipated impact on drug metabolism and toxicity. Expert Opin. Drug Metab. Toxicol. 2015, 11, 589–599. [Google Scholar] [CrossRef]
- Gál, B.; Bucher, C.; Burns, N.Z. Chiral alkyl halides: Underexplored motifs in medicine. Mar. Drugs 2016, 14, 206. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, J.; Gupta, K.; Aggarwal, S.; Aneja, R.; Chandra, R.; Panda, D.; Joshi, H.C. Brominated derivatives of noscapine are potent microtubule-interfering agents that perturb mitosis and inhibit cell proliferation. Mol. Pharmacol. 2003, 63, 799–807. [Google Scholar] [CrossRef] [PubMed]
- Isanbor, C.; Hagan, D.O. Fluorine in medicinal chemistry: A review of anti-cancer agents. J. Fluor. Chem. 2006, 127, 303–319. [Google Scholar] [CrossRef]
- Kirk, K.L. Fluorine in medicinal chemistry: Recent therapeutic applications of fluorinated small molecules. J. Fluor. Chem. 2006, 127, 1013–1029. [Google Scholar] [CrossRef]
- Ducki, S.; Forrest, R.; Hadfield, J.A.; Kendall, A.; Lawrence, N.J.; McGown, A.T.; Rennison, D. Potent antimitotic and cell growth inhibitory properties of substituted chalcones. Bioorganic Med. Chem. Lett. 1998, 8, 1051–1056. [Google Scholar] [CrossRef]
- Li, X.; Turánek, J.; Knötigová, P.; Kudláčková, H.; Mašek, J.; Parkin, S.; Rankin, S.E.; Knutson, B.L.; Lehmler, H.J. Hydrophobic tail length, degree of fluorination and headgroup stereochemistry are determinants of the biocompatibility of (fluorinated) carbohydrate surfactants. Colloids Surf. B Biointerfaces 2009, 73, 65–74. [Google Scholar] [CrossRef] [Green Version]
- Kalia, V.C.; Singh Patel, S.K.; Shanmugam, R.; Lee, J.K. Polyhydroxyalkanoates: Trends and advances toward biotechnological applications. Bioresour. Technol. 2021, 326, 124737. [Google Scholar] [CrossRef]
- Snoch, W.; Stępień, K.; Prajsnar, J.; Staroń, J.; Szaleniec, M.; Guzik, M. Influence of chemical modifications of polyhydroxyalkanoate-derived fatty acids on their antimicrobial properties. Catalysts 2019, 9, 510. [Google Scholar] [CrossRef] [Green Version]
- Sofińska, K.; Barbasz, J.; Witko, T.; Dryzek, J.; Haraźna, K.; Witko, M.; Kryściak-Czerwenka, J.; Guzik, M. Structural, topographical, and mechanical characteristics of purified polyhydroxyoctanoate polymer. J. Appl. Polym. Sci. 2019, 136, 136. [Google Scholar] [CrossRef]
- Guzik, M.; Witko, T.; Steinbüchel, A.; Wojnarowska, M.; Sołtysik, M.; Wawak, S. What Has Been Trending in the Research of Polyhydroxyalkanoates? A Systematic Review. Front. Bioeng. Biotechnol. 2020, 8, 35–39. [Google Scholar] [CrossRef]
- Witko, T.; Solarz, D.; Feliksiak, K.; Rajfur, Z.; Guzik, M. Cellular architecture and migration behavior of fibroblast cells on polyhydroxyoctanoate (PHO): A natural polymer of bacterial origin. Biopolymers 2019, 110, e23324. [Google Scholar] [CrossRef]
- Povea-Cabello, S.; Oropesa-Ávila, M.; de la Cruz-Ojeda, P.; Villanueva-Paz, M.; de la Mata, M.; Suárez-Rivero, J.M.; Álvarez-Córdoba, M.; Villalón-García, I.; Cotán, D.; Ybot-González, P.; et al. Dynamic reorganization of the cytoskeleton during apoptosis: The two coffins hypothesis. Int. J. Mol. Sci. 2017, 18, 2393. [Google Scholar] [CrossRef] [Green Version]
- Gourlay, C.; Ayscough, K. The actin cytoskeleton in ageing and apoptosis. FEMS Yeast Res. 2005, 5, 1193–1198. [Google Scholar] [CrossRef] [Green Version]
- Ankam, S.; Teo, B.K.K.; Pohan, G.; Ho, S.W.L.; Lim, C.K.; Yim, E.K.F. Temporal Changes in Nucleus Morphology, Lamin A/C and Histone Methylation During Nanotopography-Induced Neuronal Differentiation of Stem Cells. Front. Bioeng. Biotechnol. 2018, 6, 6. [Google Scholar] [CrossRef]
- Lennerz, J.; Schöchlin, M.; Weissinger, S.; Brandes, A.; Herrmann, M.; Möller, P. A nuclear circularity-based classifier for diagnostic distinction of desmoplastic from spindle cell melanoma in digitized histological images. J. Pathol. Inform. 2014, 5, 40. [Google Scholar] [CrossRef]
- Wang, B.; Hu, Q.; Wan, T.; Yang, F.; Cui, L.; Hu, S.; Gong, B.; Li, M.; Zheng, Q.C. Porous Lactose-Modified Chitosan Scaffold for Liver Tissue Engineering: Influence of Galactose Moieties on Cell Attachment and Mechanical Stability. Int. J. Polym. Sci. 2016, 2016, 2862738. [Google Scholar] [CrossRef]
- Carolina, N.; Carolina, N.; Hill, C.; Carolina, N. Cell Migration. Compr Physiol. 2015, 2, 2369–2392. [Google Scholar] [CrossRef]
- Liu, C.Y.; Lin, H.H.; Tang, M.J.; Wang, Y.K. Vimentin contributes to epithelial-mesenchymal transition ancer cell mechanics by mediating cytoskeletal organization and focal adhesion maturation. Oncotarget 2015, 6, 15966–15983. [Google Scholar] [CrossRef] [Green Version]
- Bachand, G.D.; Jain, R.; Ko, R.; Bouxsein, N.F.; Vandelinder, V. Inhibition of Microtubule Depolymerization by Osmolytes. Biomacromolecules 2018, 19, 2401–2408. [Google Scholar] [CrossRef]
- Upadhyaya, K.; Hamidullah; Singh, K.; Arun, A.; Shukla, M.; Srivastava, N.; Ashraf, R.; Sharma, A.; Mahar, R.; Shukla, S.K.; et al. Identification of gallic acid based glycoconjugates as a novel tubulin polymerization inhibitors. Org. Biomol. Chem. 2016, 14, 1338–1358. [Google Scholar] [CrossRef]
- Zhao, Y.; Liu, A.; Du, Y.; Cao, Y.; Zhang, E.; Zhou, Q.; Hai, H.; Zhen, Y.; Zhang, S. Effects of sucrose ester structures on liposome-mediated gene delivery. Acta Biomater. 2018, 72, 278–286. [Google Scholar] [CrossRef] [PubMed]
- Guan, Y.; Chen, H.; Zhong, Q. Nanoencapsulation of caffeic acid phenethyl ester in sucrose fatty acid esters to improve activities against cancer cells. J. Food Eng. 2019, 246, 125–133. [Google Scholar] [CrossRef]
- Richard, J. Challenges and opportunities in the delivery of cancer therapeutics. Ther. Deliv. 2011, 2, 107–121. [Google Scholar] [CrossRef] [PubMed]


| Ester | Monoester with C9 Chains [%] | Monoester with C7 Chains [%] | Diester C9 Chains [%] | Diester C7 Chains [%] | Diester C9C7 Chains [%] | Conversion after 48 h [%] |
|---|---|---|---|---|---|---|
| C9-glu | 57.8 | - | 42.2 | - | - | 17.8 |
| C9-gal | 99.9 | - | - | - | - | 17.8 |
| C9-lac | 56.2 | - | 43.8 | - | - | 10.4 |
| mPHN-glu | 88.1 | 0.7 | - | - | 11.2 | 42.5 |
| mPHN-gal | 38.6 | - | 18.8 | 42.7 | - | 24.3 |
| mPHN-lac | 59.4 | - | 40.6 | - | - | 12.6 |
| F-mPHN-glu | 0.4 | 8.5 | 76.0 | 10.3 | 4.6 | 18.9 |
| F-mPHN-gal | - | - | - | 90.9 | 9.1 | 10.3 |
| F-mPHN-lac | 86.6 | 0.2 | 0.5 | 12.7 | - | 11.0 |
| IC50 [10−3 mol L–1] | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Time [h] | C9-glu | C9-gal | C9-lac | PHN-glu | PHN-gal | PHN-lac | F-mPHN-glu | F-mPHN-gal | F-mPHN-lac | |
| DU145 | 24 | 1.32 | 1.56 | 0.92 | 0.93 | 0.75 | 0.16 | - | - | - |
| 72 | 0.66 | 0.78 | 0.84 | 0.54 | 0.50 | 0.13 | 0.10 | 0.10 | 0.08 | |
| 120 | 1.32 | 1.56 | 0.46 | 0.32 | 0.30 | 0.09 | 0.10 | 0.10 | 0.34 | |
| PNT2 | 24 | 2.49 | 2.39 | 1.71 | 1.51 | 1.26 | 0.42 | - | - | - |
| 72 | 1.11 | 1.23 | 0.92 | 1.31 | 0.61 | 0.20 | 0.93 | 1.18 | 1.16 | |
| 120 | 2.05 | 2.36 | 1.74 | 1.18 | 1.84 | 0.17 | 1.08 | 0.70 | 0.66 | |
| HTB140 | 24 | 1.47 | 1.66 | 0.62 | 1.77 | 0.97 | 0.64 | 0.06 | 0.16 | 0.19 |
| 72 | 1.32 | 1.56 | 0.92 | 1.20 | 1.22 | 0.28 | 0.09 | 0.16 | 0.22 | |
| 120 | 1.13 | 1.56 | 0.81 | 1.69 | 1.50 | 0.44 | 0.37 | 0.25 | 0.39 | |
| HaCAT | 72 | 1.32 | 1.56 | 0.92 | 1.43 | 1.20 | 0.38 | 0.73 | 0.58 | 0.64 |
| 120 | - | - | - | - | - | - | 0.80 | 0.95 | 0.67 | |
| HSF | 72 | 1.32 | 1.56 | 0.92 | 0.61 | 0.62 | 0.45 | - | - | - |
| 120 | - | - | - | 0.76 | 0.50 | 0.63 | 0.76 | 0.50 | 0.63 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Snoch, W.; Wnuk, D.; Witko, T.; Staroń, J.; Bojarski, A.J.; Jarek, E.; Plou, F.J.; Guzik, M. In Search of Effective Anticancer Agents—Novel Sugar Esters Based on Polyhydroxyalkanoate Monomers. Int. J. Mol. Sci. 2021, 22, 7238. https://doi.org/10.3390/ijms22137238
Snoch W, Wnuk D, Witko T, Staroń J, Bojarski AJ, Jarek E, Plou FJ, Guzik M. In Search of Effective Anticancer Agents—Novel Sugar Esters Based on Polyhydroxyalkanoate Monomers. International Journal of Molecular Sciences. 2021; 22(13):7238. https://doi.org/10.3390/ijms22137238
Chicago/Turabian StyleSnoch, Wojciech, Dawid Wnuk, Tomasz Witko, Jakub Staroń, Andrzej J. Bojarski, Ewelina Jarek, Francisco J. Plou, and Maciej Guzik. 2021. "In Search of Effective Anticancer Agents—Novel Sugar Esters Based on Polyhydroxyalkanoate Monomers" International Journal of Molecular Sciences 22, no. 13: 7238. https://doi.org/10.3390/ijms22137238
APA StyleSnoch, W., Wnuk, D., Witko, T., Staroń, J., Bojarski, A. J., Jarek, E., Plou, F. J., & Guzik, M. (2021). In Search of Effective Anticancer Agents—Novel Sugar Esters Based on Polyhydroxyalkanoate Monomers. International Journal of Molecular Sciences, 22(13), 7238. https://doi.org/10.3390/ijms22137238








