Low-Dose Acrolein, an Endogenous and Exogenous Toxic Molecule, Inhibits Glucose Transport via an Inhibition of Akt-Regulated GLUT4 Signaling in Skeletal Muscle Cells
Abstract
1. Introduction
2. Results
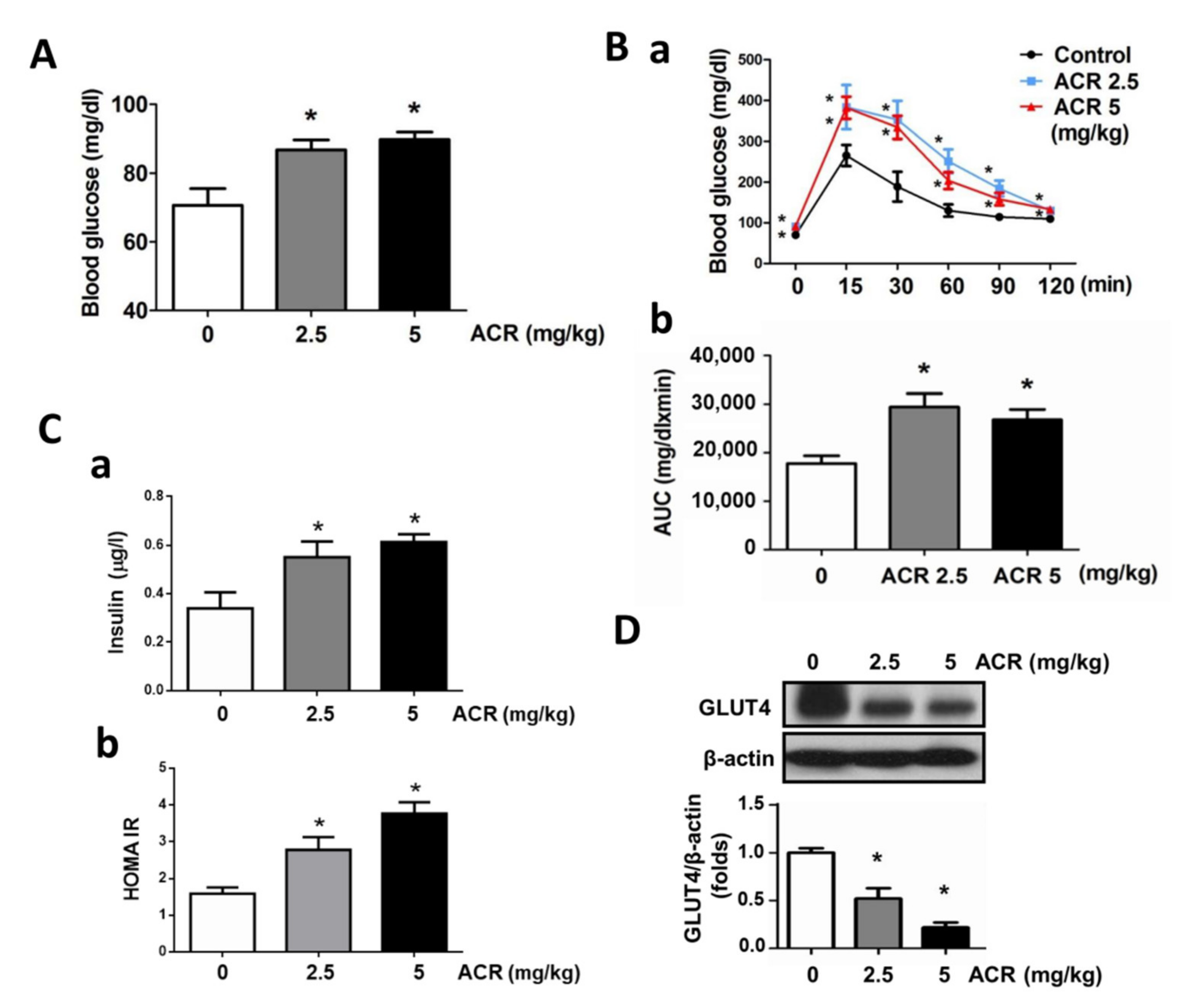
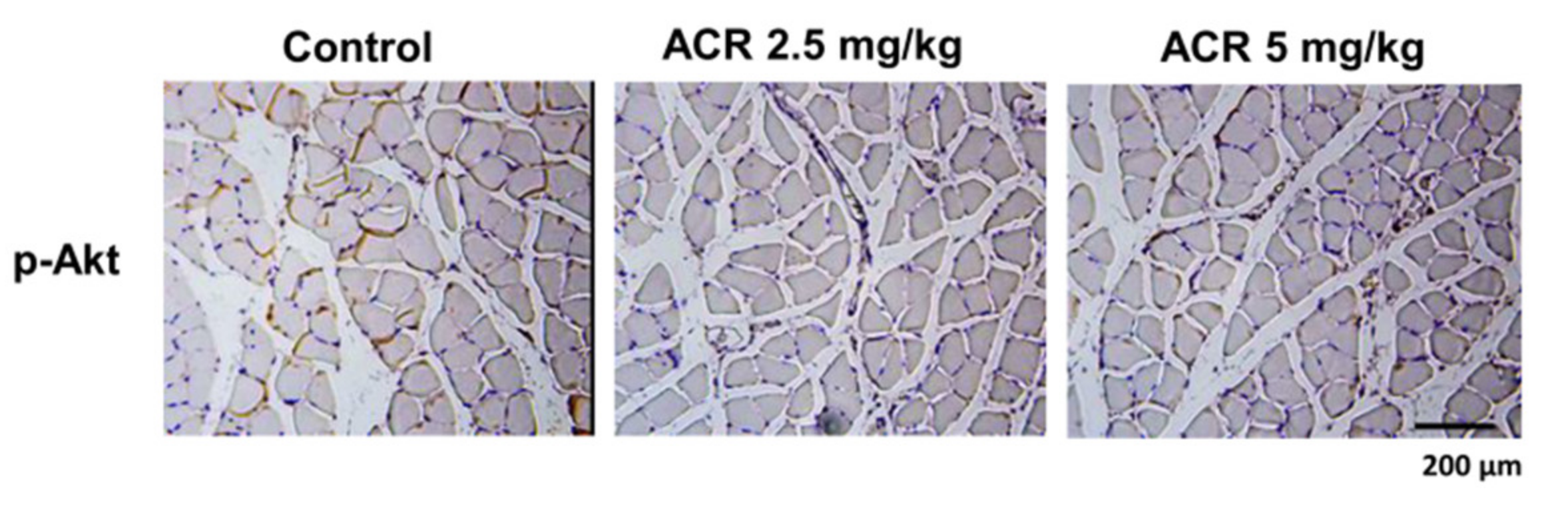
2.1. The In Vivo Effects of Acrolein on Blood Glucose and Muscle GLUT4 Expression in Mice
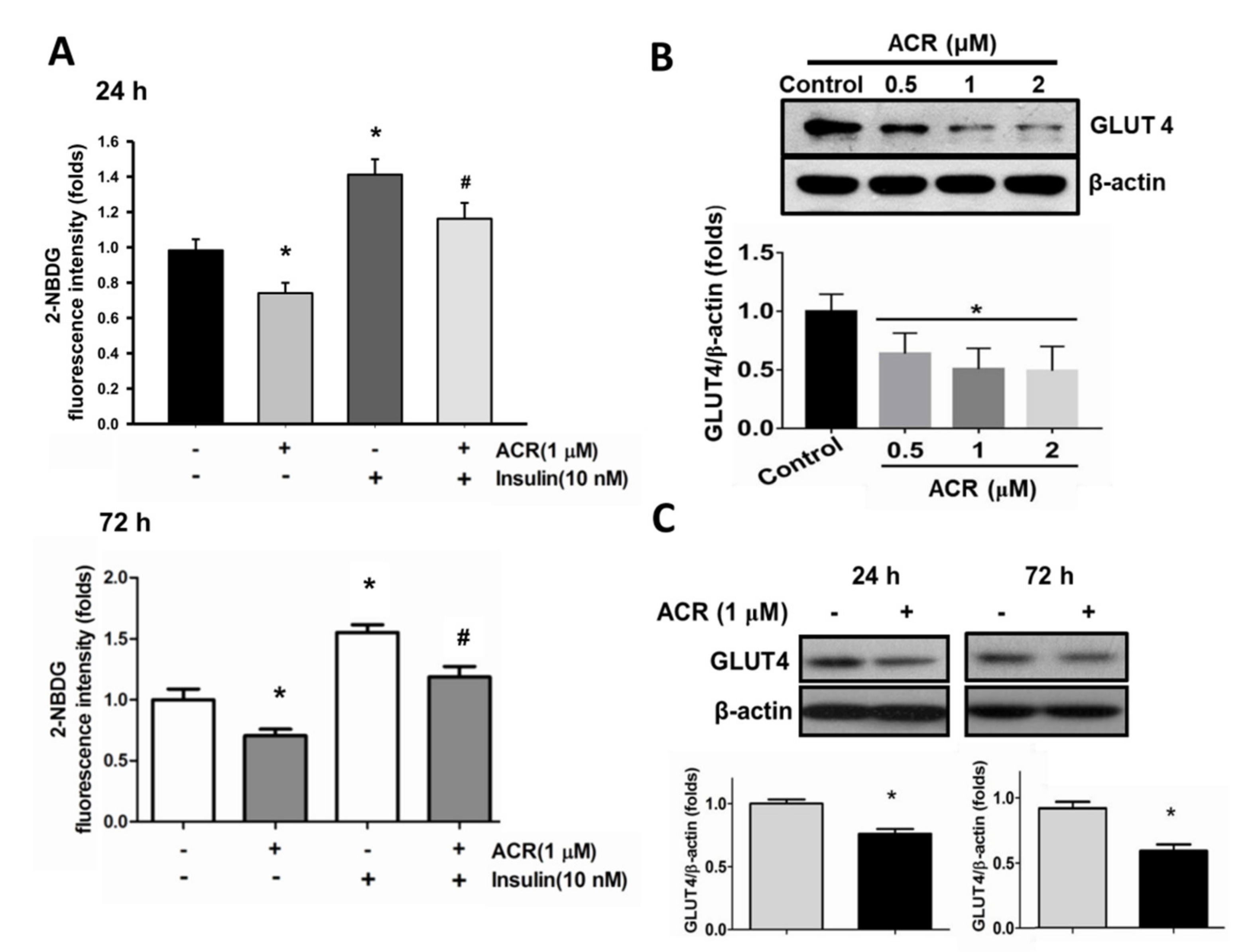
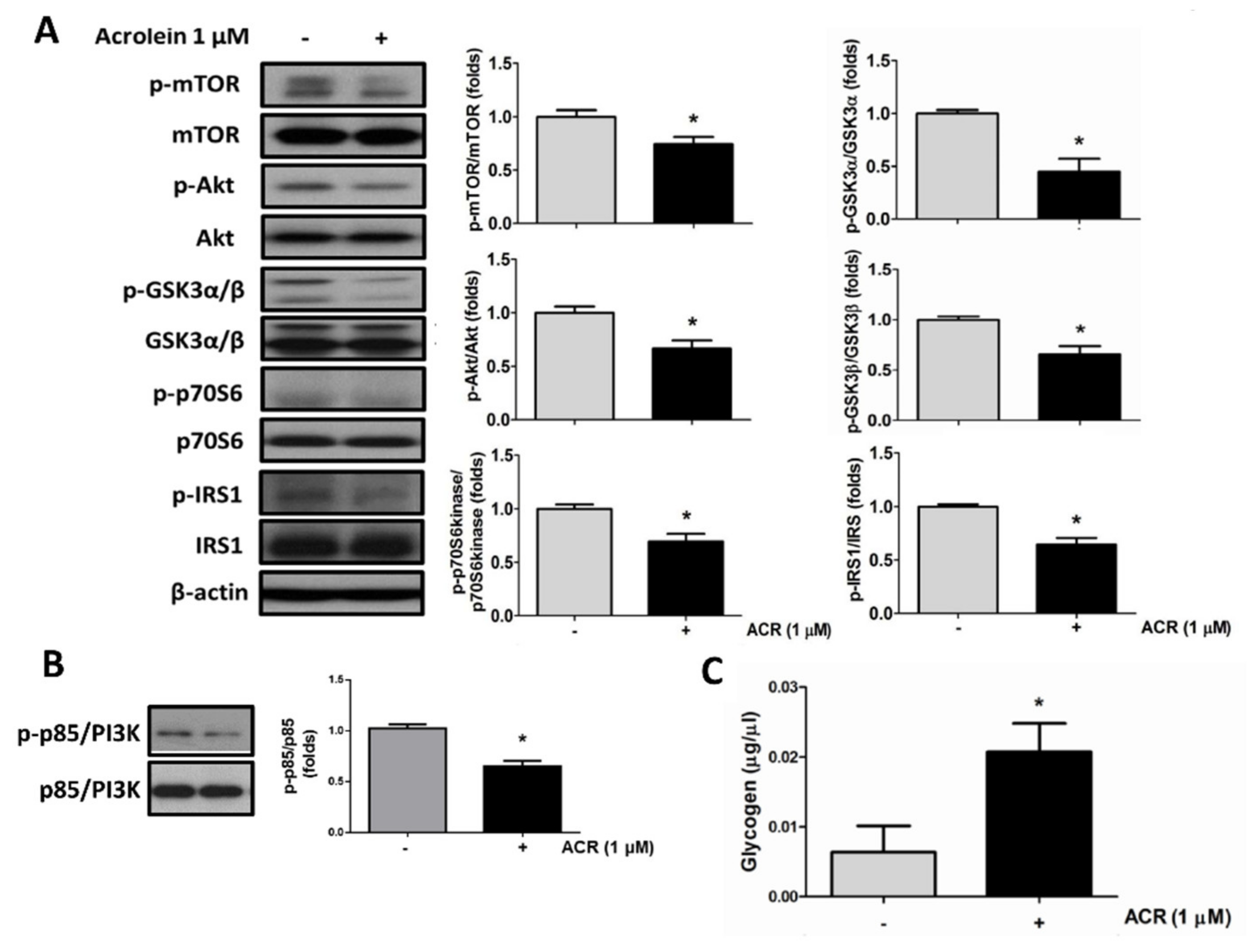
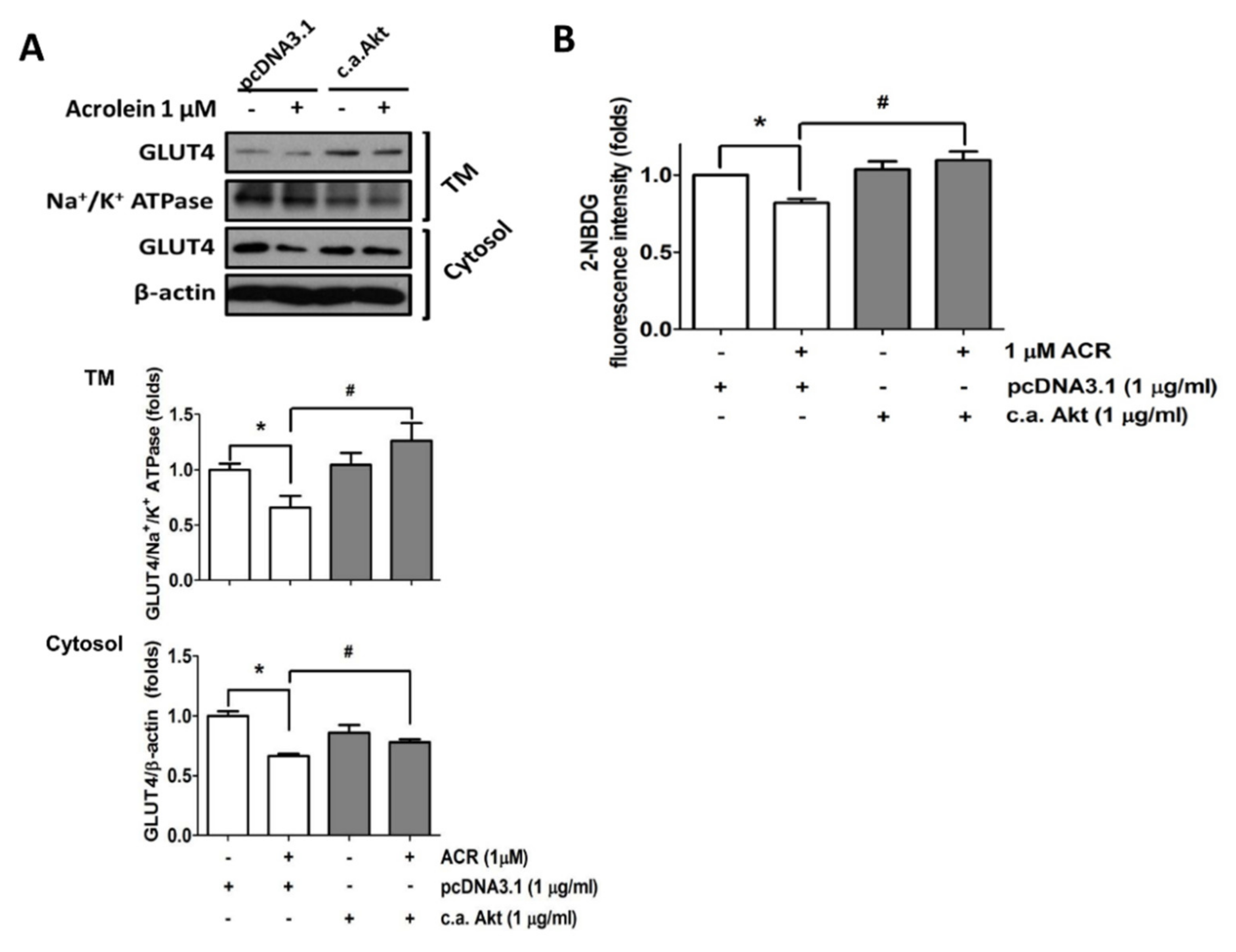
2.2. The In Vitro Effects of Acrolein on Glucose Metabolic Signaling in C2C12 Myotubes
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Blood Glucose Analysis and Oral Glucose Tolerance Test
4.3. Myoblasts and Myotube Formation
4.4. Glucose Transport Assay
4.5. Protein Extraction and Western Blot and Immunohistochemical Analysis
4.6. Transient Transfection
4.7. Determination of Glycogen Content
4.8. Statistics
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Klip, A. The many ways to regulate glucose transporter 4. Appl. Physiol. Nutr. Metab. 2009, 34, 481–487. [Google Scholar] [CrossRef] [PubMed]
- Morgan, B.J.; Chai, S.Y.; Albiston, A.L. GLUT4 associated proteins as therapeutic targets for diabetes. Recent Pat. Endocr. Metab. Immune Drug Discov. 2011, 5, 25–32. [Google Scholar] [CrossRef] [PubMed]
- Zanou, N.; Gailly, P. Skeletal muscle hypertrophy and regeneration: Interplay between the myogenic regulatory factors (MRFs) and insulin-like growth factors (IGFs) pathways. Cell Mol. Life Sci. 2013, 70, 4117–4130. [Google Scholar] [CrossRef] [PubMed]
- Mounier, R.; Théret, M.; Lantier, L.; Foretz, M.; Viollet, B. Expanding roles for AMPK in skeletal muscle plasticity. Trends Endocrinol. Metab. 2015, 26, 275–286. [Google Scholar] [CrossRef]
- Yin, H.; Price, F.; Rudnicki, M.A. Satellite cells and the muscle stem cell niche. Physiol. Rev. 2013, 93, 23–67. [Google Scholar] [CrossRef]
- Herman, M.A.; Kahn, B.B. Glucose transport and sensing in the maintenance of glucose homeostasis and metabolic harmony. J. Clin. Invest. 2006, 116, 1767–1775. [Google Scholar] [CrossRef]
- Nedachi, T.; Kanzaki, M. Regulation of glucose transporters by insulin and extracellular glucose in C2C12 myotubes. Am. J. Physiol. Endocrinol. Metab. 2006, 291, E817–E828. [Google Scholar] [CrossRef]
- Kanzaki, M.; Pessin, J.E. Insulin-stimulated GLUT4 translocation in adipocytes is dependent upon cortical actin remodeling. J. Biol. Chem. 2001, 276, 42436–42444. [Google Scholar] [CrossRef]
- Zierath, J.R.; He, L.; Gumà, A.; Odegoard Wahlström, E.; Klip, A.; Wallberg-Henriksson, H. Insulin action on glucose transport and plasma membrane GLUT4 content in skeletal muscle from patients with NIDDM. Diabetologia 1996, 39, 1180–1189. [Google Scholar] [CrossRef]
- Moghe, A.; Ghare, S.; Lamoreau, B.; Mohammad, M.; Barve, S.; McClain, C.; Joshi-Barve, S. Molecular mechanisms of acrolein toxicity: Relevance to human disease. Toxicol. Sci. 2015, 143, 242–255. [Google Scholar] [CrossRef]
- Chan, W.R.; Sidheswaran, M.; Sullivan, D.P.; Cohn, S.; Fisk, W.J. Cooking-related PM2.5 and acrolein measured in grocery stores and comparison with other retail types. Indoor Air 2015, 26, 489–500. [Google Scholar] [CrossRef]
- Grigsby, J.; Betts, B.; Vidro-Kotchan, E.; Culbert, R.; Tsin, A. A possible role of acrolein in diabetic retinopathy: Involvement of a VEGF/TGFbeta signaling pathway of the retinal pigment epithelium in hyperglycemia. Curr. Eye Res. 2012, 37, 1045–1053. [Google Scholar] [CrossRef]
- Suzuki, D.; Miyata, T. Carbonyl stress in the pathogenesis of diabetic nephropathy. Intern. Med. 1999, 38, 309–314. [Google Scholar] [CrossRef][Green Version]
- O’Toole, T.E.; Abplanalp, W.; Li, X.; Cooper, N.; Conklin, D.J.; Haberzettl, P.; Bhatnagar, A. Acrolein decreases endothelial cell migration and insulin sensitivity through induction of let-7a. Toxicol. Sci. 2014, 140, 271–282. [Google Scholar] [CrossRef]
- Feroe, A.G.; Attanasio, R.; Scinicariello, F. Acrolein metabolites, diabetes and insulin resistance. Environ. Res. 2016, 148, 1–6. [Google Scholar] [CrossRef]
- Chen, H.J.; Wang, C.C.; Chan, D.C.; Chiu, C.Y.; Yang, R.S.; Liu, S.H. Adverse effects of acrolein, a ubiquitous environmental toxicant, on muscle regeneration and mass. J. Cachexia Sarcopenia Muscle 2019, 10, 165–176. [Google Scholar] [CrossRef] [PubMed]
- Faroon, O.; Roney, N.; Taylor, J.; Ashizawa, A.; Lumpkin, M.H.; Plewak, D.J. Acrolein environmental levels and potential for human exposure. Toxicol. Ind. Health 2008, 24, 543–564. [Google Scholar] [CrossRef] [PubMed]
- Kehrer, J.P.; Biswal, S.S. The molecular effects of acrolein. Toxicol. Sci. 2000, 57, 6–15. [Google Scholar] [CrossRef] [PubMed]
- Igarashi, K.; Ueda, S.; Yoshida, K.; Kashiwagi, K. Polyamines in renal failure. Amino Acids 2006, 31, 477–483. [Google Scholar] [CrossRef]
- Kowalski, G.M.; Bruce, C.R. The regulation of glucose metabolism: Implications and considerations for the assessment of glucose homeostasis in rodents. Am. J. Physiol. Endocrinol. Metab. 2014, 307, E859–E871. [Google Scholar] [CrossRef]
- Iwata, M.; Suzuki, S.; Hayakawa, K.; Inoue, T.; Naruse, K. Uniaxial cyclic stretch increases glucose uptake into C2C12 myotubes through a signaling pathway independent of insulin-like growth factor-I. Horm. Metab. Res. 2009, 41, 16–22. [Google Scholar] [CrossRef]
- Karlsson, H.K.; Zierath, J.R. Insulin signaling and glucose transport in insulin resistant human skeletal muscle. Cell Biochem. Biophys. 2007, 48, 103–113. [Google Scholar] [CrossRef]
- Thiebaud, D.; Jacot, E.; DeFronzo, R.A.; Maeder, E.; Jequier, E.; Felber, J.P. The effect of graded doses of insulin on total glucose uptake, glucose oxidation, and glucose storage in man. Diabetes 1982, 31, 957–963. [Google Scholar] [CrossRef] [PubMed]
- Klip, A.; Sun, Y.; Chiu, T.T.; Foley, K.P. Signal transduction meets vesicle traffic: The software and hardware of GLUT4 translocation. Am. J. Physiol. Cell Physiol. 2014, 306, C879–C886. [Google Scholar] [CrossRef]
- McMahon, D.K.; Anderson, P.A.; Nassar, R.; Bunting, J.B.; Saba, Z.; Oakeley, A.E.; Malouf, N.N. C2C12 cells: Biophysical, biochemical, and immunocytochemical properties. Am. J. Physiol. 1994, 266, C1795–C1802. [Google Scholar] [CrossRef] [PubMed]
- Haga, Y.; Ishii, K.; Suzuki, T. N-glycosylation is critical for the stability and intracellular trafficking of glucose transporter GLUT4. J. Biol. Chem. 2011, 286, 31320–31327. [Google Scholar] [CrossRef]
- Ijuin, T.; Takenawa, T. Regulation of insulin signaling and glucose transporter 4 (GLUT4) exocytosis by phosphatidylinositol 3,4,5-trisphosphate (PIP3) phosphatase, skeletal muscle, and kidney enriched inositol polyphosphate phosphatase (SKIP). J. Biol. Chem. 2012, 287, 6991–6999. [Google Scholar] [CrossRef] [PubMed]
- Tremblay, F.; Lavigne, C.; Jacques, H.; Marette, A. Defective insulin-induced GLUT4 translocation in skeletal muscle of high fat-fed rats is associated with alterations in both Akt/protein kinase B and atypical protein kinase C (zeta/lambda) activities. Diabetes 2001, 50, 1901–1910. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Liu, G.; Guo, J.; Su, Z. The PI3K/AKT pathway in obesity and type 2 diabetes. Int. J. Biol. Sci. 2018, 14, 1483–1496. [Google Scholar] [CrossRef]
- Aguirre, V.; Werner, E.D.; Giraud, J.; Lee, Y.H.; Shoelson, S.E.; White, M.F. Phosphorylation of Ser307 in insulin receptor substrate-1 blocks interactions with the insulin receptor and inhibits insulin action. J. Biol. Chem. 2002, 277, 1517–1531. [Google Scholar] [CrossRef]
- Hançer, N.J.; Qiu, W.; Cherella, C.; Li, Y.; Copps, K.D.; White, M.F. Insulin and metabolic stress stimulate multisite serine/threonine phosphorylation of insulin receptor substrate 1 and inhibit tyrosine phosphorylation. J. Biol. Chem. 2014, 289, 12467–12484. [Google Scholar] [CrossRef]
- Copps, K.D.; Hancer, N.J.; Opare-Ado, L.; Qiu, W.; Walsh, C.; White, M.F. Irs1 serine 307 promotes insulin sensitivity in mice. Cell Metab. 2010, 11, 84–92. [Google Scholar] [CrossRef] [PubMed]
- Henriksen, E.J. Dysregulation of glycogen synthase kinase-3 in skeletal muscle and the etiology of insulin resistance and type 2 diabetes. Curr. Diabetes Rev. 2010, 6, 285–293. [Google Scholar] [CrossRef] [PubMed]
- Cross, D.A.; Alessi, D.R.; Cohen, P.; Andjelkovich, M.; Hemmings, B.A. Inhibition of glycogen synthase kinase-3 by insulin mediated by protein kinase B. Nature 1995, 378, 785–789. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.; Doble, B.W.; MacAulay, K.; Sinclair, E.M.; Drucker, D.J.; Woodgett, J.R. Tissue-specific role of glycogen synthase kinase 3β in glucose homeostasis and insulin action. Mol. Cell Biol. 2008, 28, 6314–6328. [Google Scholar] [CrossRef]
- Jensen, J.; Rustad, P.I.; Kolnes, A.J.; Lai, Y.C. The Role of Skeletal Muscle Glycogen Breakdown for Regulation of Insulin Sensitivity by Exercise. Front. Physiol. 2011, 2, 112. [Google Scholar] [CrossRef]
- Charron, M.J.; Katz, E.B.; Olson, A.L. GLUT4 gene regulation and manipulation. J. Biol. Chem. 1999, 274, 3253–3256. [Google Scholar] [CrossRef]
- Kim, Y.B.; Peroni, O.D.; Aschenbach, W.G.; Minokoshi, Y.; Kotani, K.; Zisman, A.; Kahn, C.R.; Goodyear, L.J.; Kahn, B.B. Muscle-specific deletion of the Glut4 glucose transporter alters multiple regulatory steps in glycogen metabolism. Mol. Cell Biol. 2005, 25, 9713–9723. [Google Scholar] [CrossRef]
- Murphy, S.D.; Porter, S. Effects of toxic chemicals on some adaptive liver enzymes, liver glycogen, and blood glucose in fasted rats. Biochem. Pharmacol. 1966, 15, 1665–1676. [Google Scholar] [CrossRef]
- Institute of Laboratory Animal Resources. Guide for the Care and Use of Laboratory Animals; National Academy Press: Washington, DC, USA, 2011. [Google Scholar]
- Zou, C.; Wang, Y.; Shen, Z. 2-NBDG as a fluorescent indicator for direct glucose uptake measurement. J. Biochem. Biophys. Methods 2005, 64, 207–215. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, C.-C.; Chen, H.-J.; Chan, D.-C.; Chiu, C.-Y.; Liu, S.-H.; Lan, K.-C. Low-Dose Acrolein, an Endogenous and Exogenous Toxic Molecule, Inhibits Glucose Transport via an Inhibition of Akt-Regulated GLUT4 Signaling in Skeletal Muscle Cells. Int. J. Mol. Sci. 2021, 22, 7228. https://doi.org/10.3390/ijms22137228
Wang C-C, Chen H-J, Chan D-C, Chiu C-Y, Liu S-H, Lan K-C. Low-Dose Acrolein, an Endogenous and Exogenous Toxic Molecule, Inhibits Glucose Transport via an Inhibition of Akt-Regulated GLUT4 Signaling in Skeletal Muscle Cells. International Journal of Molecular Sciences. 2021; 22(13):7228. https://doi.org/10.3390/ijms22137228
Chicago/Turabian StyleWang, Ching-Chia, Huang-Jen Chen, Ding-Cheng Chan, Chen-Yuan Chiu, Shing-Hwa Liu, and Kuo-Cheng Lan. 2021. "Low-Dose Acrolein, an Endogenous and Exogenous Toxic Molecule, Inhibits Glucose Transport via an Inhibition of Akt-Regulated GLUT4 Signaling in Skeletal Muscle Cells" International Journal of Molecular Sciences 22, no. 13: 7228. https://doi.org/10.3390/ijms22137228
APA StyleWang, C.-C., Chen, H.-J., Chan, D.-C., Chiu, C.-Y., Liu, S.-H., & Lan, K.-C. (2021). Low-Dose Acrolein, an Endogenous and Exogenous Toxic Molecule, Inhibits Glucose Transport via an Inhibition of Akt-Regulated GLUT4 Signaling in Skeletal Muscle Cells. International Journal of Molecular Sciences, 22(13), 7228. https://doi.org/10.3390/ijms22137228





