Molecular Characterization of TRPA Subfamily Genes and Function in Temperature Preference in Tuta absoluta (Meyrick) (Lepidoptera: Gelechiidae)
Abstract
:1. Introduction
2. Results
2.1. Cloning of TaPain and TaPyx
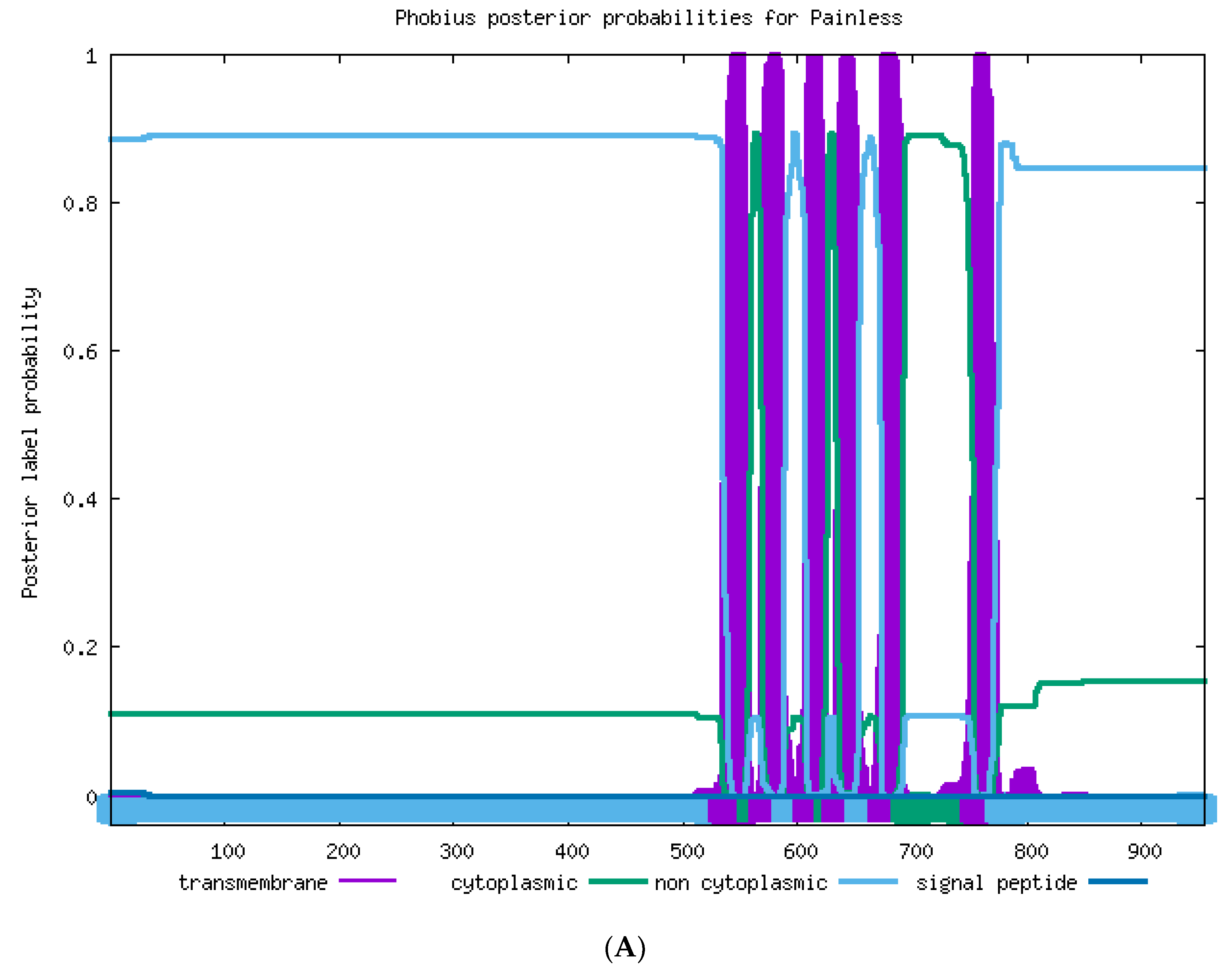
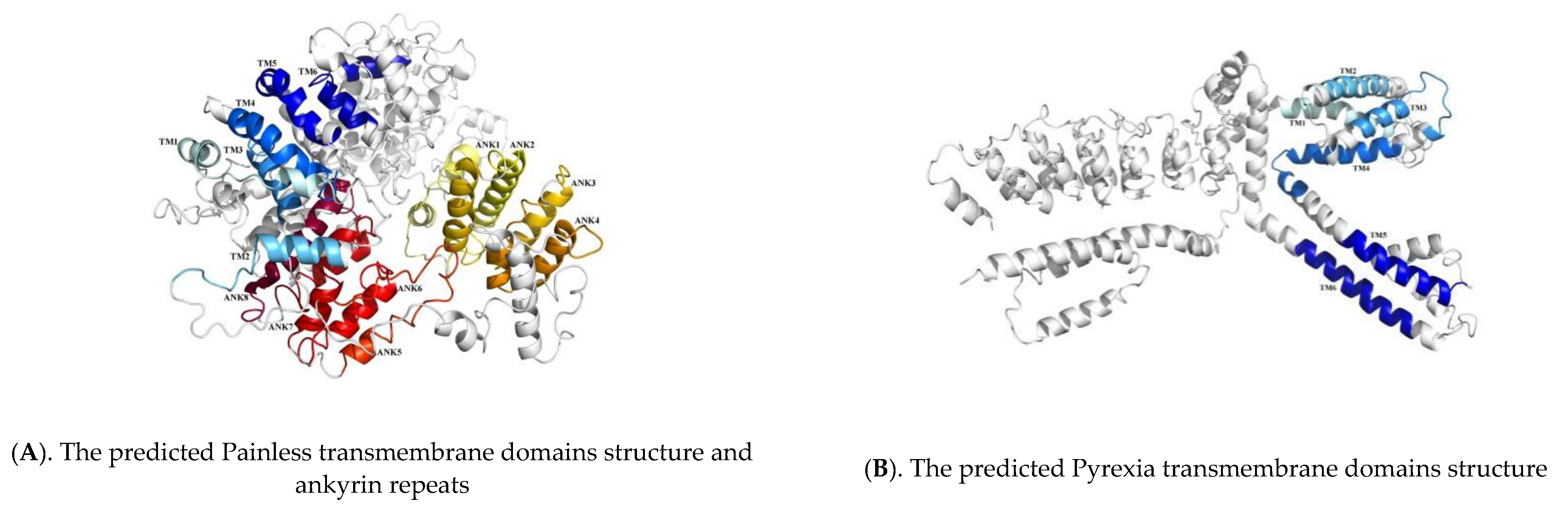
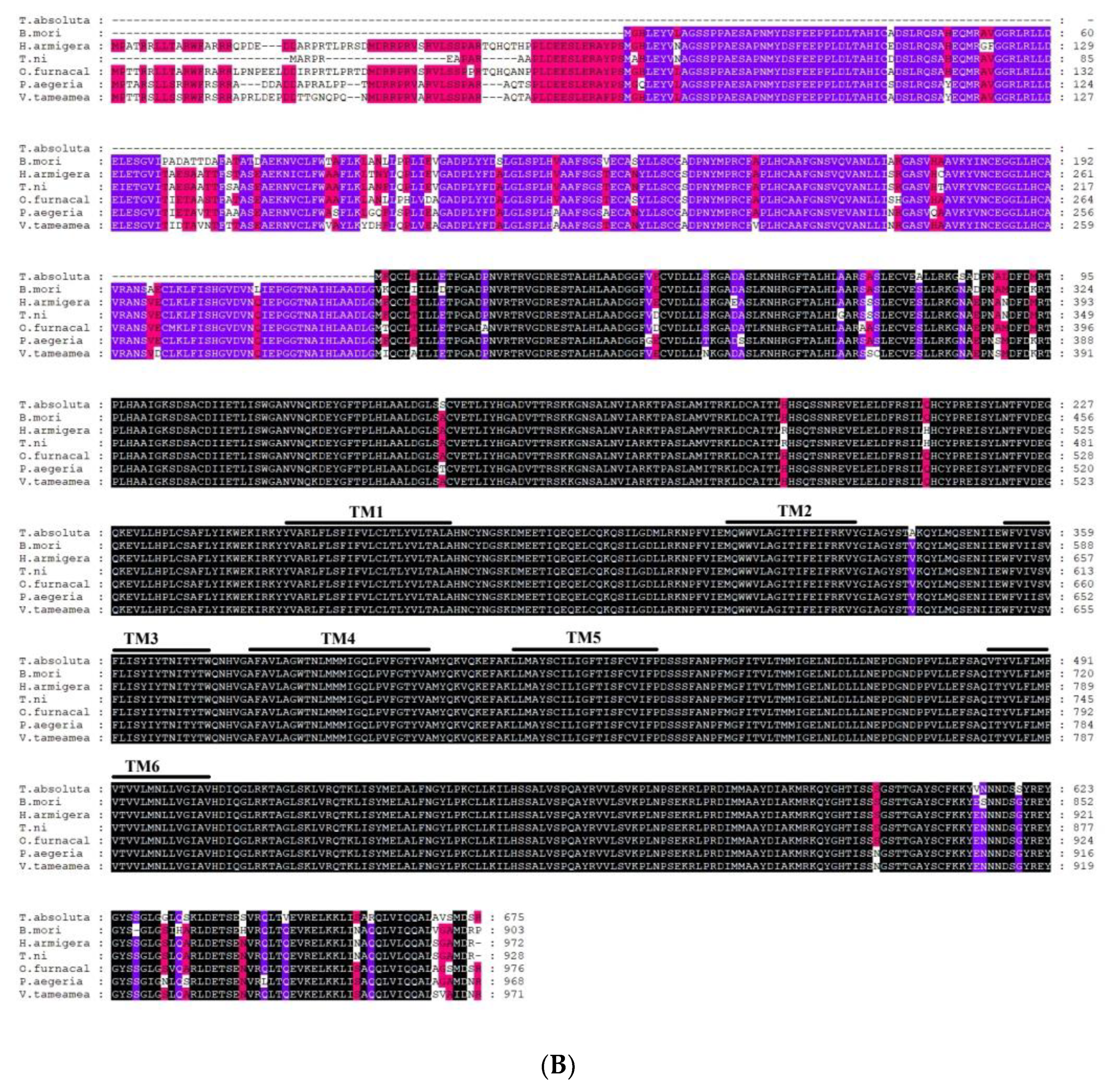
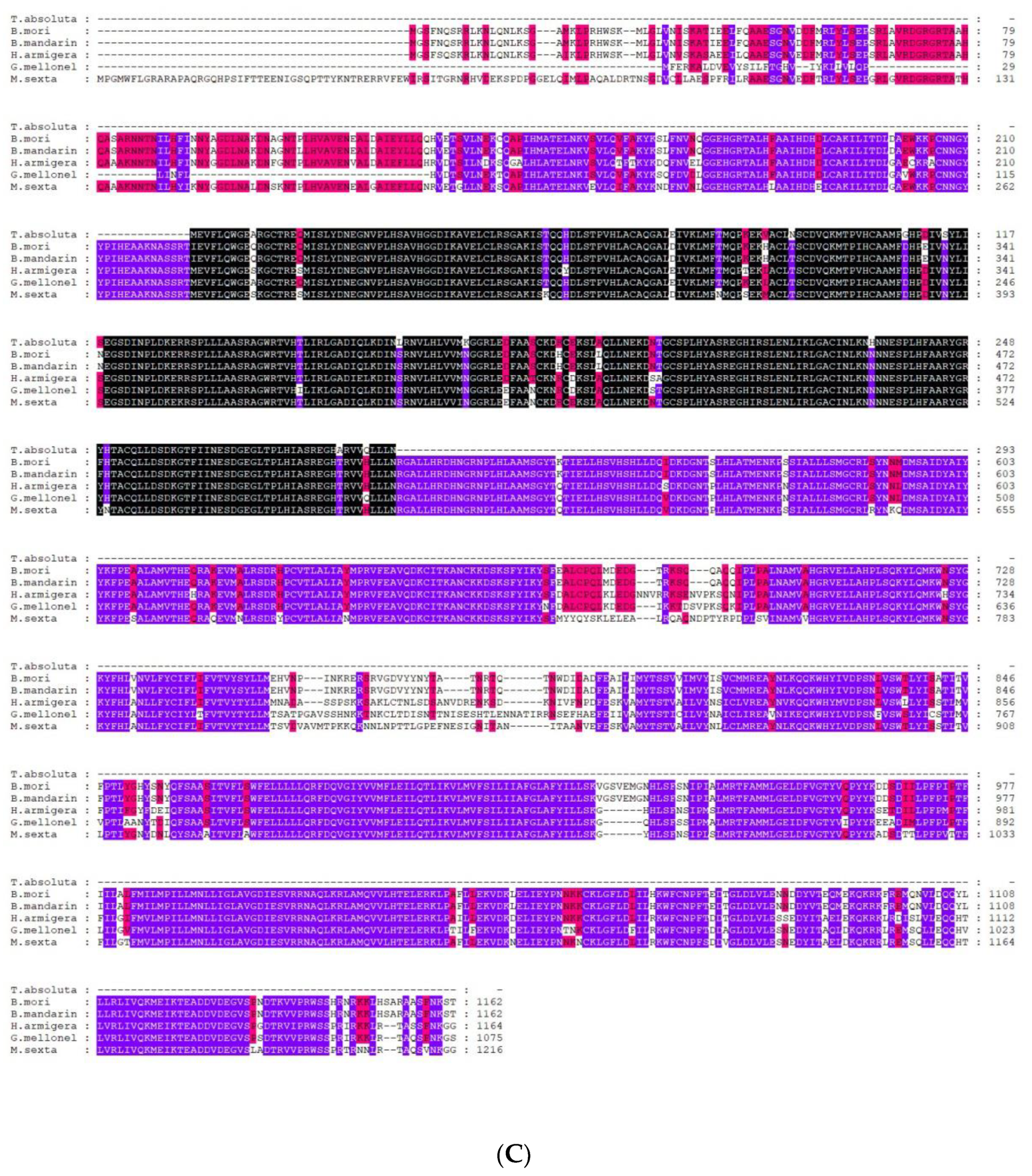
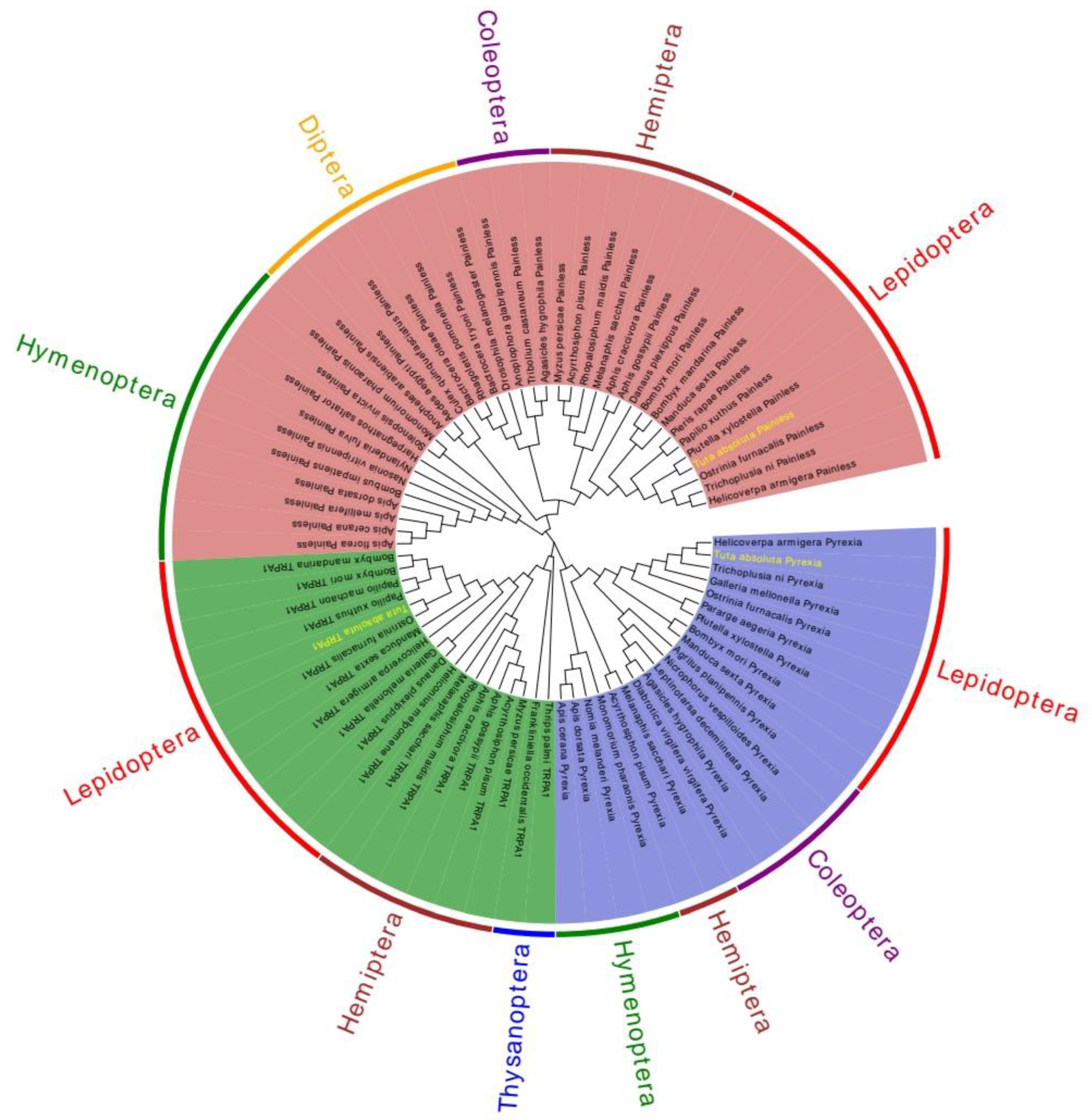
2.2. Sequence Analysis of TaTRPA1, TaPain, and TaPyx
2.3. Expression Profiles of TaTRPA1, TaPain, and TaPyx during Different Developmental Stages
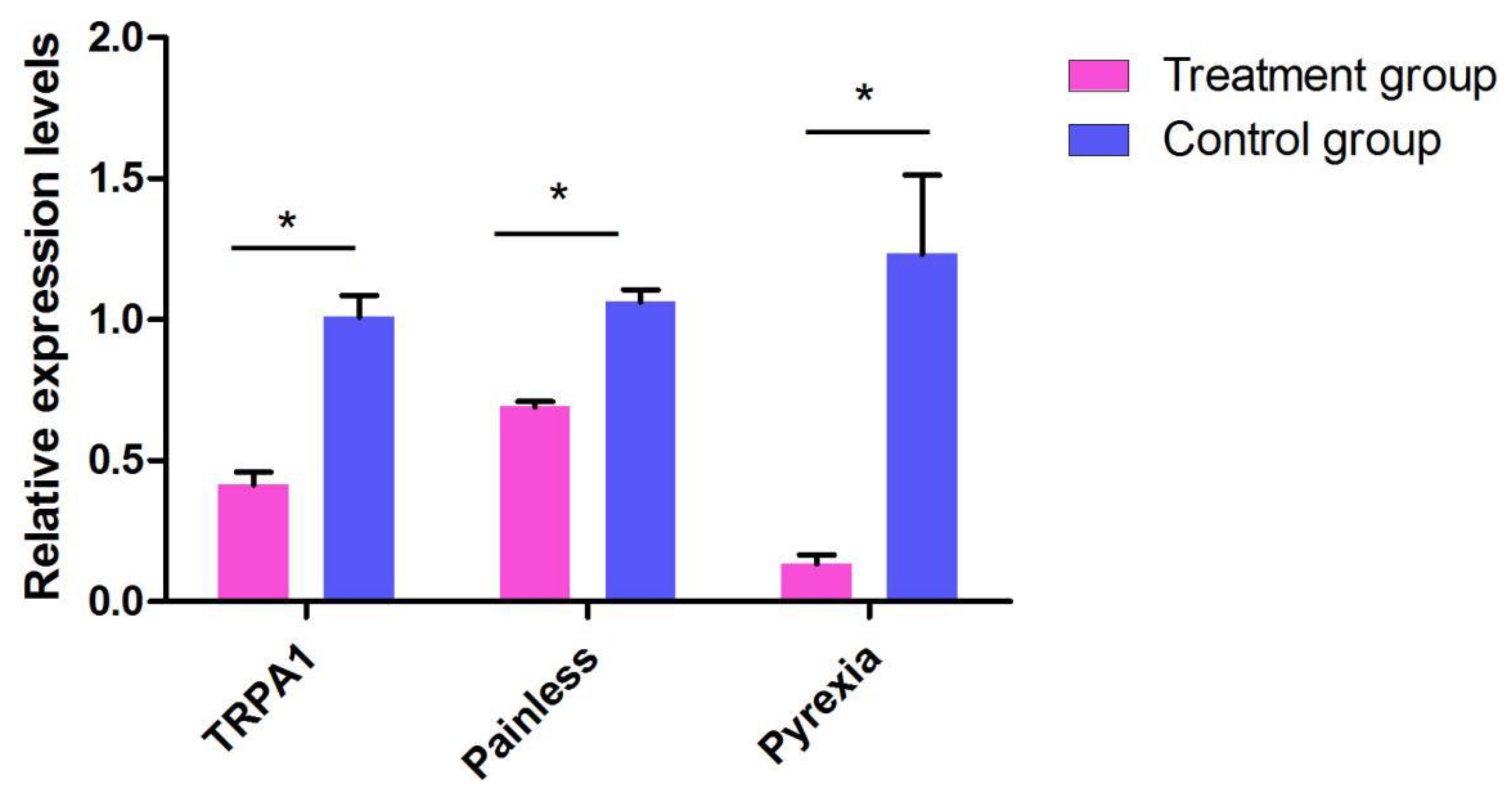
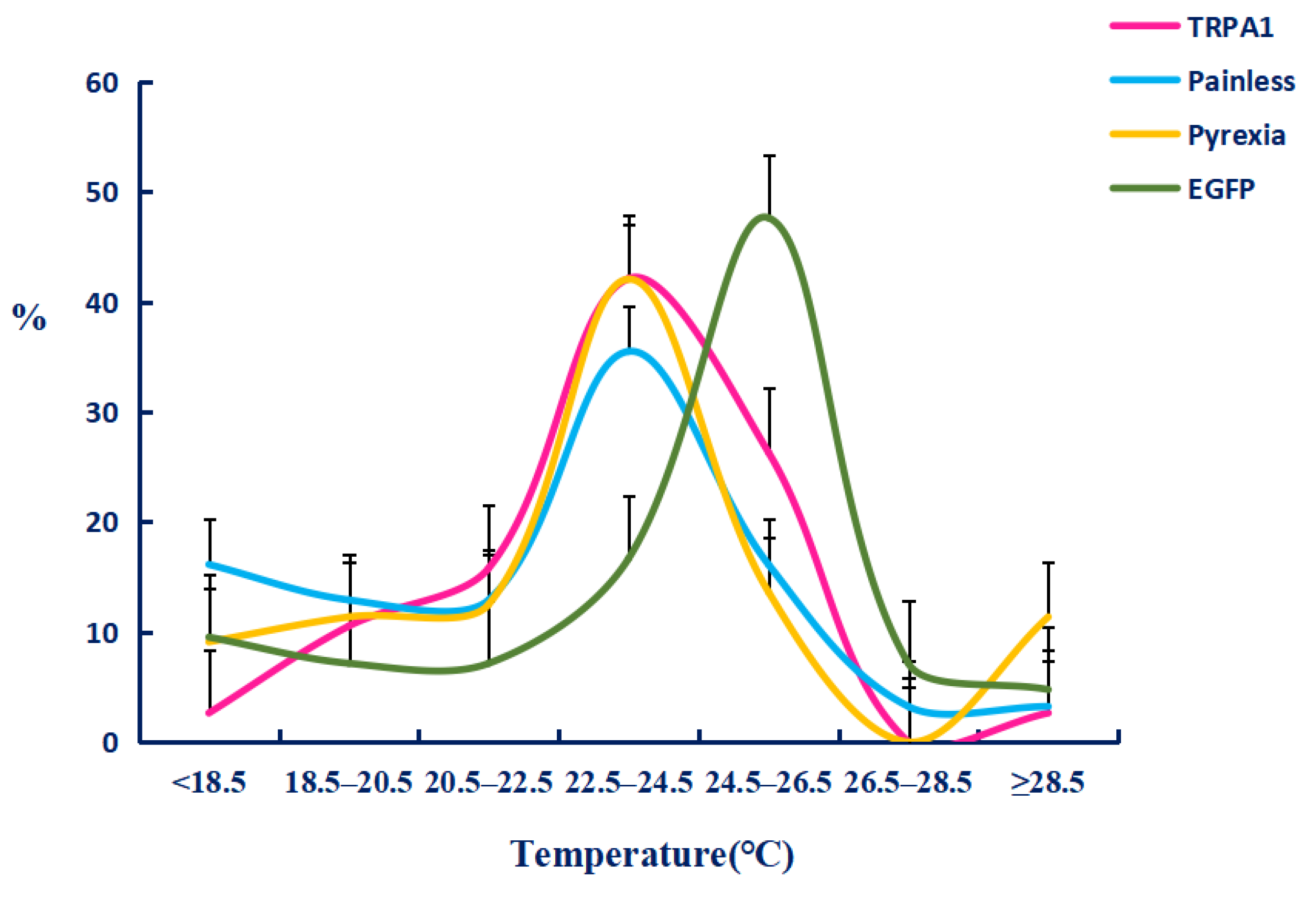
2.4. The Function of TaTRPA1, TaPain, and TaPyx Genes in Temperature Preference Behavior
3. Discussion
4. Materials and Methods
4.1. Insect Rearing and Host Plants
4.2. RNA Extraction and cDNA Synthesis
4.3. Cloning of the Three Genes of TRPA Family
4.4. Sequence Analysis of the Three TRPA Genes
4.5. Quantitative Real-Time PCR Analysis of Relative Expression Levels
4.6. Production of dsRNA Transcription Templates and Synthesis of dsRNA
4.7. RNA Interference (RNAi) and Detection
4.8. Preference Temperature Behavioral Assay
4.9. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Garcia, M.F.; Espul, J.C. Bioecology of the tomato moth (Scrobipalpula absoluta) in Mendoza, Argentine Republic. Rev. Investig. Agrop. 1982, 17, 135–146. [Google Scholar]
- Desneux, N.; Wajnberg, E.; Wyckhuys, K.A.G.; Burgio, G.; Arpaia, S.; Narváez-Vasquez, C.A.; González-Cabrera, J.; Ruescas, D.C.; Tabone, E.; Frandon, J.; et al. Biological invasion of European tomato crops by Tuta absoluta: Ecology, geographic expansion and prospects for biological control. J. Pest Sci. 2010, 83, 197–215. [Google Scholar] [CrossRef]
- Brévault, T.; Sylla, S.; Diatte, M.; Bernadas, G.; Diarra, K. Tuta absoluta Meyrick (Lepidoptera: Gelechiidae): A New Threat to Tomato Production in Sub-Saharan Africa. Afr. Entomol. 2014, 22, 441–444. [Google Scholar] [CrossRef]
- Chen, L.M.; Li, X.W.; He, T.J.; Li, P.J.; Liu, Y.; Zhou, S.X.; Wu, Q.C.; Chen, T.T.; Lu, Y.B.; Hou, Y.M. Comparative biochemical and transcriptome analyses in tomato and eggplant reveal their differential responses to Tuta absoluta infestation. Genomics 2021, 113, 2108–2121. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.F.; Ma, D.Y.; Wang, Y.S.H.; Gao, Y.H.; Liu, W.X.; Zhang, R.; Fu, W.J.; Xian, X.Q.; Wang, J.; Kuang, M.; et al. First report of the South American tomato leafminer, Tuta absoluta (Meyrick), in China. J. Integr. Agr. 2020, 19, 1912–1917. [Google Scholar] [CrossRef]
- Zhang, G.F.; Liu, W.X.; Wan, F.H.; Xian, X.Q.; Zhang, Y.B.; Guo, J.Y. Bioecology, damage and management of the tomato leafminer Tuta absoluta (Meyrick) (Lepidoptera: Gelechiidae) a world wide quarantine pest. J. Biosaf. 2018, 27, 155–163. [Google Scholar]
- Biondi, A.; Guedes, R.N.C.; Wan, F.H.; Desneux, N. Ecology, worldwide spread, and management of the invasive South American tomato pinworm, Tuta absoluta: Past, present, and future. Ann. Rev. Entom. 2017, 63, 239–258. [Google Scholar] [CrossRef] [PubMed]
- Desneux, N.; Luna, M.G.; Guillemaud, T.; Urbaneja, A. The invasive South American tomato pinworm, Tuta absoluta, continues to spread in Afro-Eurasia and beyond: The new threat to tomato world production. J. Pest Sci. 2011, 84, 403–408. [Google Scholar] [CrossRef]
- Campos, M.R.; Biondi, A.; Adiga, A.; Guedes, R.N.C.; Desneux, N. From the Western Palaearctic region to beyond: Tuta absoluta ten years after invading Europe. J. Pest Sci. 2017, 90, 787–796. [Google Scholar] [CrossRef]
- Mansour, R.; Brévault, T.; Chailleux, A.; Cherif, A.; Grissa-Lebdi, K.; Haddi, K.; Mohamed, S.A.; Nofemela, R.S.; Oke, A.; Sylla, S.; et al. Occurrence, biology, natural enemies and management of Tuta absoluta in Africa. Entom. Gen. 2018, 38, 83–111. [Google Scholar] [CrossRef]
- Gray, E.M. Thermal acclimation in a complex life cycle: The effects of larval and adult thermal conditions on metabolic rate and heat resistance in Culex pipiens (Diptera: Culicidae). J. Insect Physiol. 2013, 59, 1001–1007. [Google Scholar] [CrossRef]
- Overgaard, J.; Sørensen, J.G. Rapid thermal adaptation during field temperature variations in Drosophila melanogaster. Cryobiology 2008, 56, 159–162. [Google Scholar] [CrossRef]
- Bowler, K.; Terblanche, J.S. Insect thermal tolerance: What is the role of ontogeny, ageing and senescence. Biol. Rev. 2008, 83, 339–355. [Google Scholar] [CrossRef]
- Klepsatel, P.; Girish, T.N.; Dircksen, H.; Gáliková, M. Reproductive fitness of Drosophila is maximised by optimal developmental temperature. J. Exp. Biol. 2019, 222, jeb202184. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, D.; Li, X.W.; Ma, L.; Fu, K.Y.; Ding, X.H.; Guo, W.C.; Lü, Y.B. Effects of temperature on the growth, development and reproduction of the tomato leaf miner, Tuta absoluta (Lepidoptera: Gelechiidae). Acta Entomol. Sin. 2019, 62, 1417–1426. [Google Scholar]
- Tarusikirwa, V.L.; Mutamiswa, R.; Chidawanyika, F.; Nyamukondiwa, C. Cold hardiness of the South American tomato pinworm Tuta absoluta (Lepidoptera: Gelechiidae): Both larvae and adults are chill-usceptible. Pest Manag. Sci. 2020, 77, 184–193. [Google Scholar] [CrossRef]
- Tarusikirwa, V.L.; Mutamiswa, R.; English, S.; Chidawanyika, F.; Nyamukondiwa, C. Thermal plasticity in the invasive south American tomato pinworm Tuta absoluta (Meyrick) (Lepidoptera: Gelechiidae). J. Ther. Biol. 2020, 90, 102598. [Google Scholar] [CrossRef] [PubMed]
- Fry, F.E.J. Effects of the environment on animal activity. Univ. Toronto Stud. Biol. 1947, 55, 1–62. [Google Scholar]
- Huey, R.B.; Stevenson, R.D. Integrating thermal physiology and ecology of ectotherms: A discussion of approaches. Am. Zool. 1979, 19, 357–366. [Google Scholar] [CrossRef] [Green Version]
- Huey, R.B. Temperature, physiology, and the ecology of reptiles. In Biology of the Reptilia; Academic Press: New York, NY, USA, 1982; Volume 12, pp. 25–74. [Google Scholar]
- Hochachka, P.W.; Somero, G.N. Biochemical Adaptation, Mechanisms and Processes in Physiological Evolution; Oxford University Press: New York, NY, USA, 2002. [Google Scholar]
- Johnson, J.A.; Kelsch, S.W. Effects of evolutionary thermal environment on temperature-preference relationships in fishes. Environ. Biol. Fish. 1998, 53, 447–458. [Google Scholar] [CrossRef]
- Martin, T.L.; Huey, R.B. Why “suboptimal” is optimal: Jensen’s inequality and ecotherm thermal preferences. Am. Nat. 2008, 171, E102–E118. [Google Scholar] [CrossRef] [Green Version]
- Clusella-Trullas, S.; Blackburn, T.M.; Chown, S.L. Climatic predictors of temperature performance curve parameters in ectotherms imply complex responses to climate change. Am. Nat. 2011, 177, 738–751. [Google Scholar] [CrossRef] [Green Version]
- William, W.R.; Martha, E.C. Behavioral Thermoregulation and the “Final Preferendum” Paradigm. Am. Zool. 1979, 19, 211–224. [Google Scholar]
- Hagstrum, D.W.; Flinn, P.W.; Gaffney, J.J. Temperature gradient on Tribolium castaneum (Coleoptera: Tenebrionidae) adult dispersal in stored wheat. Environ. Entomol. 1998, 1, 123–129. [Google Scholar] [CrossRef]
- Fedorka, K.M.; Kutch, I.C.; Collins, L.; Musto, E. Cold temperature preference in bacterially infected Drosophila melanogaster improves survival but is remarkably suboptimal. J. Insect Physol. 2016, 93–94, 36–41. [Google Scholar] [CrossRef] [PubMed]
- Ji, S.X.; Wang, X.D.; Shen, X.N.; Liang, L.; Liu, W.X.; Wan, F.H.; Lü, Z.C. Using RNA Interference to Reveal the Function of Chromatin Remodeling Factor ISWI in Temperature Tolerance in Bemisia tabaci Middle East-Asia Minor 1 Cryptic Species. Insects 2020, 11, 113. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Crickenberger, S.; Hui, T.Y.; Yuan, F.L.; Bonebrake, T.C.; Williams, G.A. Preferred temperature of intertidal ectotherms: Broad patterns and methodological approaches. J. Therm. Biol. 2020, 87, 102468. [Google Scholar] [CrossRef]
- Buckley, L.B. Linking traits to energetics and population dynamics to predict lizard ranges in changing environments. Am. Nat. 2008, 171, E1–E19. [Google Scholar] [CrossRef] [Green Version]
- Buckley, L.B.; Urban, M.C.; Angilletta, M.J.; Crozier, L.G.; Rissler, L.J.; Sears, M.W. Can mechanism inform species’ distribution models? Ecol. Lett. 2010, 13, 1041–1054. [Google Scholar] [CrossRef]
- Monaco, C.J.; Wethey, D.S.; Gulledge, S.; Helmuth, B. Shore-level size gradients and thermal refuge use in the predatory sea star Pisaster ochraceus: The role of environmental stressors. Mar. Ecol. Prog. Ser. 2015, 539, 191–205. [Google Scholar] [CrossRef] [Green Version]
- Huey, R.B.; Slatkin, M. Cost and benefits of lizard thermoregulation. Q. Rev. Biol. 1976, 51, 363–384. [Google Scholar] [CrossRef] [PubMed]
- Hughes, N.F.; Grand, T.C. Physiological ecology meets the ideal-free distribution: Prediction the distribution of size-structured fish populations across temperature gradients. Environ. Biol. Fishes 2000, 59, 285–298. [Google Scholar] [CrossRef]
- Lampert, W.; McCauley, E.; Manly, B.F.J. Trade-offs in the vertical distribution of zooplankton: Ideal free distribution with costs? Proc. R. Soc. Lond. Ser. B Biol. Sci. 2003, 270, 765–773. [Google Scholar] [CrossRef]
- Fowler, M.A.; Montell, C. Drosophila TRP channels and animal behavior. Life Sci. 2012, 92, 394–403. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rogers, K. Transient Receptor Potential Channel. EB. 15 November 2018. Available online: https://www.britannica.com/science/transient-receptor-potential-channel (accessed on 30 June 2021).
- Ramsey, I.S.; Delling, M.; Clapham, D.E. An introduction to TRP channels. Annu. Rev. Physiol. 2006, 68, 619–647. [Google Scholar] [CrossRef] [Green Version]
- Venkatachalam, K.; Montell, C. TRP channels. Annu. Rev. Biochem. 2007, 76, 387–417. [Google Scholar] [CrossRef] [Green Version]
- Montell, C. The TRP superfamily of cation channels. Sci. Signal. 2005, 272, re3. [Google Scholar] [CrossRef] [Green Version]
- Montell, C.; Birnbaumer, L.; Flockerzi, V.; Bindels, R.J.; Bruford, E.A.; Caterina, M.J.; Clapham, D.E.; Harteneck, C.; Heller, S.; Julius, D.; et al. A unified nomenclature for the superfamily of TRP cation channels. Mol. Cell 2002, 9, 229–231. [Google Scholar] [CrossRef]
- Caterina, M.J.; Schumacher, M.A.; Tominaga, M.; Rosen, T.A.; Levine, J.D.; Julius, D. The capsaicin receptor: A heat-activated ion channel in the pain pathway. Nature 1997, 389, 816–824. [Google Scholar] [CrossRef]
- Lee, Y.; Lee, J.; Bang, S.; Hyun, S.; Kang, J.; Hong, S.T.; Bae, E.; Kaang, B.K.; Kim, J. Pyrexia is a new thermal transient receptor potential channel endowing tolerance to high temperatures in Drosophila melanogaster. Nat. Genet. 2005, 37, 305–310. [Google Scholar] [CrossRef]
- Neely, G.G.; Keene, A.C.; Duchek, P.; Chang, E.C.; Wang, Q.P.; Aksoy, Y.A.; Rosenzweig, M.; Costigan, M.; Woolf, C.J.; Garrity, P.A.; et al. TrpA1 regulates thermal nociception in Drosophila. PLoS ONE 2011, 6, e24343. [Google Scholar] [CrossRef]
- Tracey, W.D.; Wilson, R.I.; Laurent, G.; Benzer, S. painless, a Drosophila gene essential for nociception. Cell 2003, 113, 261–273. [Google Scholar] [CrossRef] [Green Version]
- Hwang, R.Y.; Stearns, N.A.; Tracey, W.D. The ankyrin repeat domain of the TRPA protein Painless is important for thermal nociception but not mechanical nociception. PLoS ONE 2012, 7, e30090. [Google Scholar] [CrossRef] [PubMed]
- Zhong, L.; Bellemer, A.; Yan, H.; Ken, H.; Jessica, R.; Hwang, R.Y.; Pitt, G.S.; Tracey, W.D. Thermosensory and non-thermosensory isoforms of Drosophila melanogaster TRPA1 reveal heat sensor domains of a thermoTRP channel. Cell Rep. 2012, 1, 43–55. [Google Scholar] [CrossRef] [Green Version]
- Sokabe, T.; Tsujiuchi, S.; Kadowaki, T.; Tominaga, M. Drosophila Painless is a Ca2+-requiring channel activated by noxious heat. J. Neurosci. 2008, 28, 9929–9938. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Patapoutian, A. TRP channels and thermosensation. Chem. Senses 2005, 30, i193–i194. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Castillo, K.; Diaz-Franulic, I.; Canan, J.; Gonzalez-Nilo, F.; Latorre, R. Thermally activated TRP channels: Molecular sensors for temperature detection. Phys. Biol. 2018, 15, 021001. [Google Scholar] [CrossRef]
- Lü, Z.C.; Li, Q.; Liu, W.X.; Wan, F.H. Transient Receptor Potential Is Essential for High Temperature Tolerance in Invasive Bemisia tabaci Middle East Asia Minor 1 Cryptic Species. PLoS ONE 2014, 9, e108428. [Google Scholar] [CrossRef]
- Ji, S.X.; Shen, X.N.; Liang, L.; Wang, X.D.; Liu, W.X.; Wan, F.H.; Lü, Z.C. Molecular characteristics and temperature tolerance function of the transient receptor potential in the native Bemisia tabaci AsiaII3 cryptic species. J. Integ. Agr. 2020, 19, 2746–2757. [Google Scholar] [CrossRef]
- Dai, T.M.; Wang, Y.S.; Liu, W.X.; Lü, Z.C.; Wan, F.H. Thermal discrimination and transgenerational temperature response in Bemisia tabaci Mediterranean (Hemiptera: Aleyrodidae): Putative involvement of the thermo-sensitive receptor BtTRPA. Environ. Entomol. 2018, 47, 204–209. [Google Scholar] [CrossRef] [PubMed]
- Tribolium Genome Sequencing Consortium; Richards, S. The genome of the model beetle and pest Tribolium castaneum. Nature 2008, 452, 949–955. [Google Scholar]
- Kim, H.G.; Margolies, D.; Park, Y. The roles of thermal transient receptor potential channels in thermotactic behavior and in thermal acclimation in the red flour beetle, Tribolium castaneum. J. Insect Physiol. 2015, 76, 47–55. [Google Scholar] [CrossRef]
- Vo, M.N.; Terrey, M.; Lee, J.W.; Roy, B.; Moresco, J.J.; Sun, L.; Fu, H.; Liu, Q.; Weber, T.G.; Yates, J.R., 3rd; et al. ANKRD16 prevents neuron loss caused by an editing-defective tRNA synthetase. Nature 2018, 557, 510–515. [Google Scholar] [CrossRef] [PubMed]
- Bork, P. Hundreds of ankyrin-like repeats in functionally diverse proteins: Mobile modules that cross phyla horizontally? Proteins 1993, 17, 363–374. [Google Scholar] [CrossRef] [PubMed]
- Fan, X.X.; Yang, L.; Xiang, B. Recent Research Progress of Ca2+ Permeable Channels. Prog. Biochem. Biophys. 2016, 43, 1129–1138. [Google Scholar]
- 59. Bubb, K.J.; Wen, H.; Panayiotou, C.M.; Finsterbusch, M.; Khan, F.J.; Chan, M.V.; Priestley, J.V.; Baker, M.D.; Ahluwalia, A. Activation of neuronal transient receptor potential vanilloid 1 channel underlies 20-hydroxyeicosatetraenoic acid-induced vasoactivity: Role for protein kinase A. Hypertension 2013, 62, 426–433. [Google Scholar] [CrossRef] [Green Version]
- Su, H.A.; Bai, X.; Zeng, T.; Lu, Y.Y.; Qi, Y.X. Identification, characterization and expression analysis of transient receptor potential channel genes in the oriental fruit fly. Bactrocera dorsalis. BMC Genom. 2018, 19, 674. [Google Scholar] [CrossRef] [Green Version]
- Bain, S.A.; Marshall, H.; de la Filia, A.G.; Laetsch, D.R.; Husnik, F.; Ross, L. Sex-specific expression and DNA methylation in a species with extreme sexual dimorphism and paternal genome elimination. Mol. Ecol. 2021, 1–17. [Google Scholar] [CrossRef]
- Chen, J.X.; Chen, J.; Lin, T. Identification and expression of dynamin-1-like protein gene from Monochamus alternatus. Jiangsu J. Agric. Sci. 2017, 33, 524–532. [Google Scholar]
- Zhang, Z.K.; Wu, S.Y.; Lei, Z.R. Cloning, Sequence Analysis and Expression Profile of an Odorant Binding Protein Gene in Western Flower Thrips (Frankliniella occidentalis). Sci. Agric. Sin. 2016, 49, 1106–1116. [Google Scholar]
- Sayeed, O.; Benzer, S. Behavioral genetics of thermosensation and hygrosensation in Drosophila. Proc. Natl. Acad. Sci. USA 1996, 93, 6079–6084. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fu, T.; Hull, J.J.; Yang, T.; Wang, G. Identification and functional characterization of four transient receptor potential ankyrin 1 variants in Apolygus lucorum (Meyer-Dür). Insect Mol. Biol. 2016, 25, 370–384. [Google Scholar] [CrossRef] [PubMed]
- Kwon, Y.; Shim, H.S.; Wang, X.; Montell, C. Control of thermotactic behavior via coupling of a TRP channel to a phospholipase C signaling cascade. Nat. Neurosci. 2008, 11, 871–873. [Google Scholar] [CrossRef] [PubMed]
- Dillon, M.E.; Liu, R.; Wang, G.; Huey, R.B. Disentangling thermal preference and the thermal dependence of movement in ectotherms. J. Therm. Biol. 2012, 37, 631–639. [Google Scholar] [CrossRef]
- Luo, J.; Shen, W.L.; Montell, C. TRPA1 mediates sensation of the rate of temperature change in Drosophila larvae. Nat. Neurosci. 2017, 20, 34–41. [Google Scholar] [CrossRef] [Green Version]
- Lee, Y. Contribution of Drosophila TRPA1-expressing neurons to circadian locomotor activity patterns. PLoS ONE 2013, 8, e85189. [Google Scholar] [CrossRef]
- Wolfgang, W.; Simoni, A.; Gentile, C.; Stanewsky, R. The Pyrexia transient receptor potential channel mediates circadian clock synchronization to low temperature cycles in Drosophila melanogaster. Proc. Biol. Sci. 2013, 280, 20130959. [Google Scholar] [CrossRef] [Green Version]
- Sato, A.; Sokabe, T.; Kashio, M.; Yasukochi, Y.; Tominaga, M.; Shiomi, K. Embryonic thermosensitive TRPA1 determines transgenerational diapause phenotype of the silkworm, Bombyx mori. Proc. Natl. Acad. Sci. USA 2014, 111, E1249–E1255. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, C.; Zwiebel, L.J. Molecular characterization of larval peripheral thermosensory responses of the malaria vector mosquito Anopheles gambiae. PLoS ONE 2013, 8, e72595. [Google Scholar] [CrossRef] [Green Version]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Pfaffl, M.W. A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res. 2001, 29, e45. [Google Scholar] [CrossRef] [PubMed]
- Head, L.M.; Tang, X.; Hayley, S.E.; Goda, T.; Umezaki, Y.; Chang, E.C.; Leslie, J.R.; Fujiwara, M.; Garrity, P.A.; Hamada, F.N. The influence of light on temperature preference in Drosophila. Curr. Biol. 2015, 25, 1063–1068. [Google Scholar] [CrossRef] [PubMed] [Green Version]





| Gene Name | Primer Name | Primer Sequence (5´→3´) | Amplicon Length/bp |
|---|---|---|---|
| Primers for full-length gene amplification | |||
| TRPA1 | TRPA1-F277 | GCGGTGGAGTTGTGCTT | 515 |
| TRPA1-R791 | CGGCTGGCGTAATGTAG | ||
| Painless | Pain-F139 | AGTGCTTGTGGACAGTGTT | 1488 |
| Pain-R1626 | CTAGCGAGTTATTTGGGTAA | ||
| Pain-F1456 | GCGTCGGACTACGAAATA | 1314 | |
| Pain-R2769 | GCAGAAAGTGAGGAAACAA | ||
| Pain-F2546 | ACAAGAGATAAGAGCGGATG | 727 | |
| Pain-F3272 | GTGGAAGGCAGTTAGAAGTA | ||
| Pyrexia | Pyx-F1 | CGAGGTGCTTCAGTTCA | 1250 |
| Pyx-R1250 | CTCACGATTACAAACCATTC | ||
| Pyx-F867 | AGTGCTTTTACATCCG | 1460 | |
| Pyx- R2326 | GACATCAACAATAGGGT | ||
| Primers for qPCR | |||
| TRPA1 | TRPA1-QF123 | ACACGAAGCAGCCAAAAACG | 183 (for RNAi) |
| TRPA1-QR305 | GCTCCGGACCTCAAGCACAA | ||
| TRPA1-QF675 | AGGAGGGAGGTTGGAAGAC | 115 (for expression) | |
| TRPA1-QR789 | GCTGGCGTAATGTAGAGGC | ||
| Painless | Pain-QF2749 | GTTTGTTTCCTCACTTTCTGCC | 132 |
| Pain-QR2880 | ATCCACCGCCTTCTATATCCTC | ||
| Pyrexia | Pyx-QF1821 | TTCTTCTGCCCTTGTCTCACC | 135 |
| Pyx-QR1955 | CCGTATTGTTTCCGCATTTTG | ||
| RpL5 | RpL5-QF | CAGTCGTCGAGCCAGCAACA | 129 bp |
| RpL5-QR | TCCCGCATTGAAGGAGACCA | ||
| Primers for dsRNA synthesis | |||
| TRPA1 | TRPA1-DF277 | taatacgactcactatagggGCGGTGGAGTTGTGCTT | 515 |
| TRPA1-DR791 | taatacgactcactatagggCGGCTGGCGTAATGTAG | ||
| Painless | Pain-DF1456 | taatacgactcactatagggGCGTCGGACTACGAAATA | 303 |
| Pain-DR1758 | taatacgactcactatagggTCAGAAGTGGTCGCAAAT | ||
| Pyrexia | Pyx-DF1281 | taatacgactcactatagggCATCACGTACACATGGCAAAA | 335 |
| Pyx-DR1615 | taatacgactcactatagggAGAACTCAAGTAAAACGGGGG | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, X.-D.; Lin, Z.-K.; Ji, S.-X.; Bi, S.-Y.; Liu, W.-X.; Zhang, G.-F.; Wan, F.-H.; Lü, Z.-C. Molecular Characterization of TRPA Subfamily Genes and Function in Temperature Preference in Tuta absoluta (Meyrick) (Lepidoptera: Gelechiidae). Int. J. Mol. Sci. 2021, 22, 7157. https://doi.org/10.3390/ijms22137157
Wang X-D, Lin Z-K, Ji S-X, Bi S-Y, Liu W-X, Zhang G-F, Wan F-H, Lü Z-C. Molecular Characterization of TRPA Subfamily Genes and Function in Temperature Preference in Tuta absoluta (Meyrick) (Lepidoptera: Gelechiidae). International Journal of Molecular Sciences. 2021; 22(13):7157. https://doi.org/10.3390/ijms22137157
Chicago/Turabian StyleWang, Xiao-Di, Ze-Kai Lin, Shun-Xia Ji, Si-Yan Bi, Wan-Xue Liu, Gui-Fen Zhang, Fang-Hao Wan, and Zhi-Chuang Lü. 2021. "Molecular Characterization of TRPA Subfamily Genes and Function in Temperature Preference in Tuta absoluta (Meyrick) (Lepidoptera: Gelechiidae)" International Journal of Molecular Sciences 22, no. 13: 7157. https://doi.org/10.3390/ijms22137157
APA StyleWang, X.-D., Lin, Z.-K., Ji, S.-X., Bi, S.-Y., Liu, W.-X., Zhang, G.-F., Wan, F.-H., & Lü, Z.-C. (2021). Molecular Characterization of TRPA Subfamily Genes and Function in Temperature Preference in Tuta absoluta (Meyrick) (Lepidoptera: Gelechiidae). International Journal of Molecular Sciences, 22(13), 7157. https://doi.org/10.3390/ijms22137157







