Cyanine Dyes for Photo-Thermal Therapy: A Comparison of Synthetic Liposomes and Natural Erythrocyte-Based Carriers
Abstract
1. Introduction
2. Results and Discussion
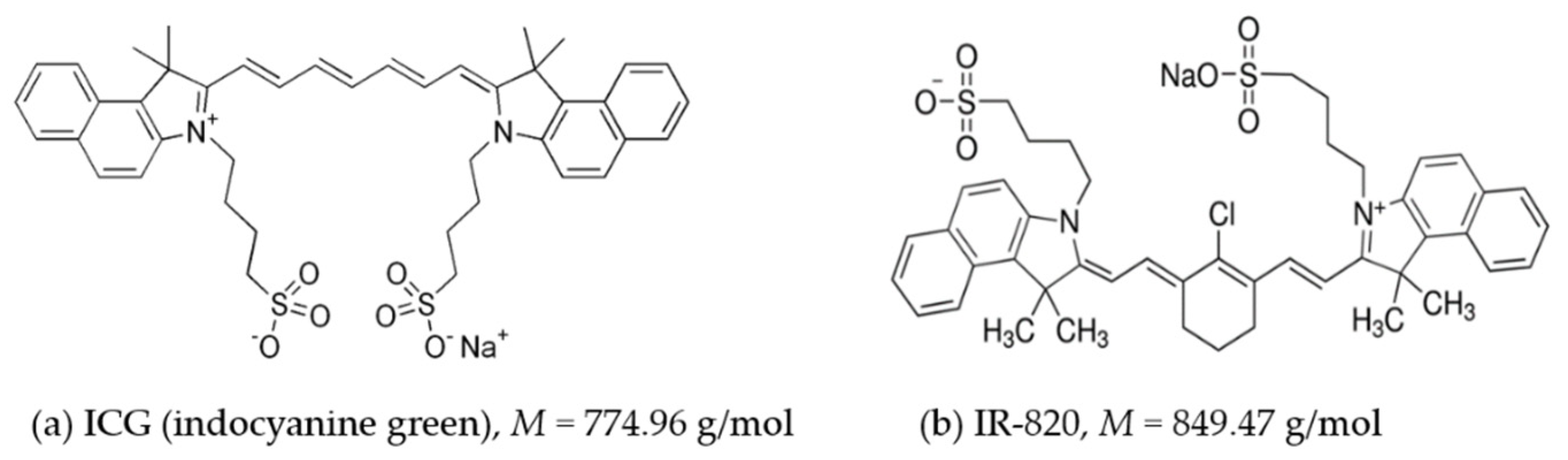
2.1. Light Absorption Properties of ICG and IR-820 in Different Solvents
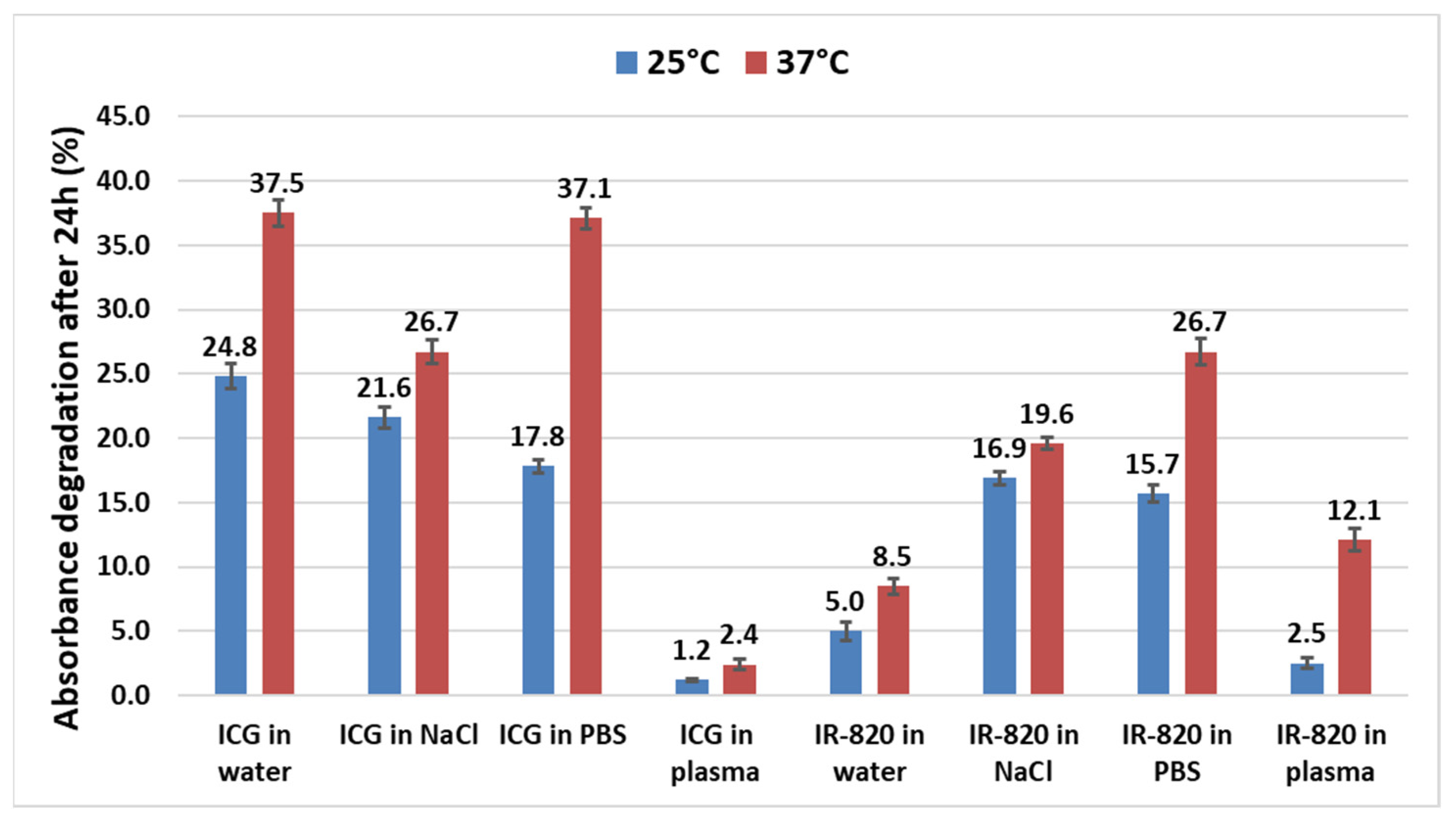
2.2. Temperature-Stability Study of ICG and IR-820 in Different Solvents
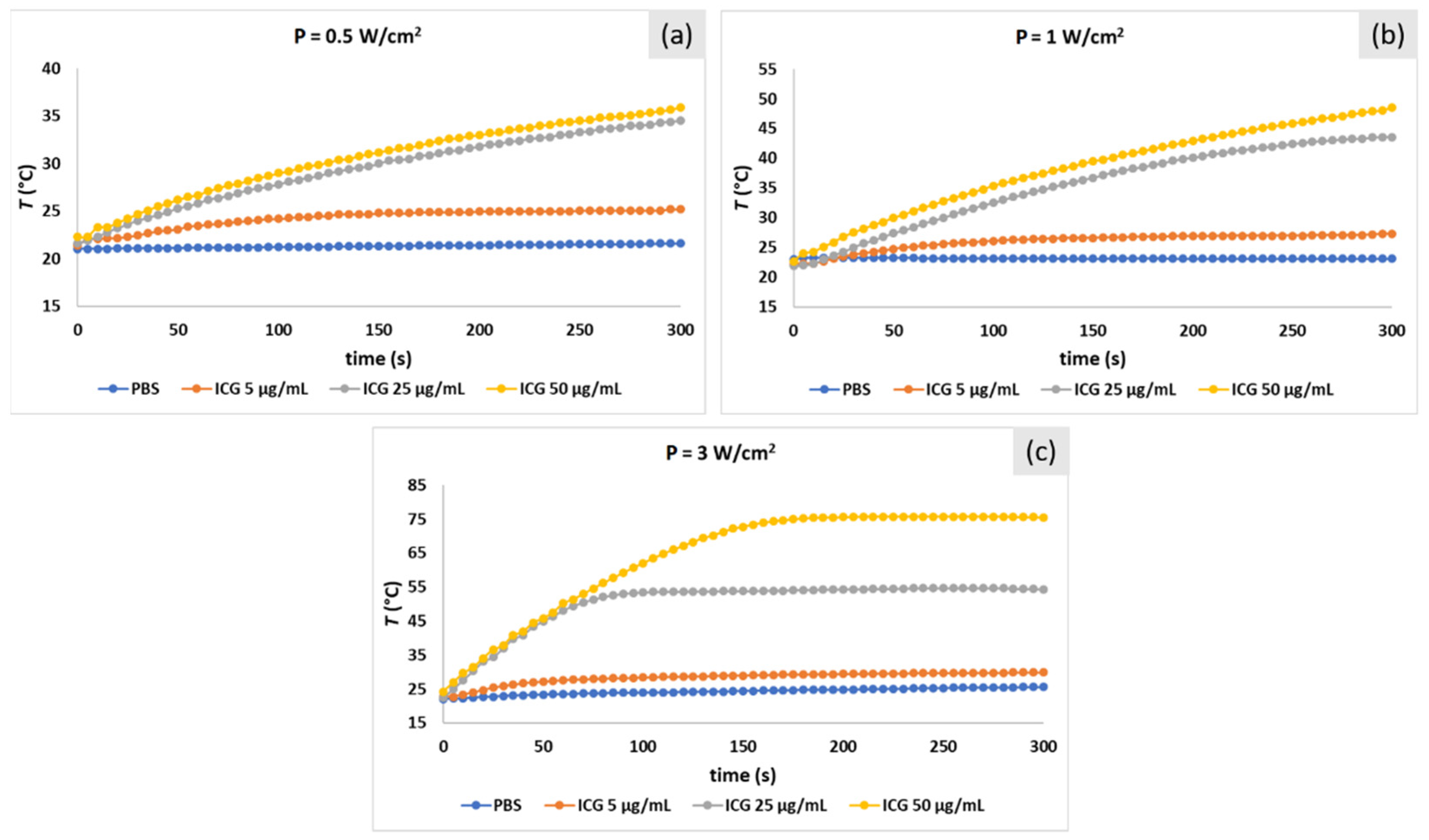
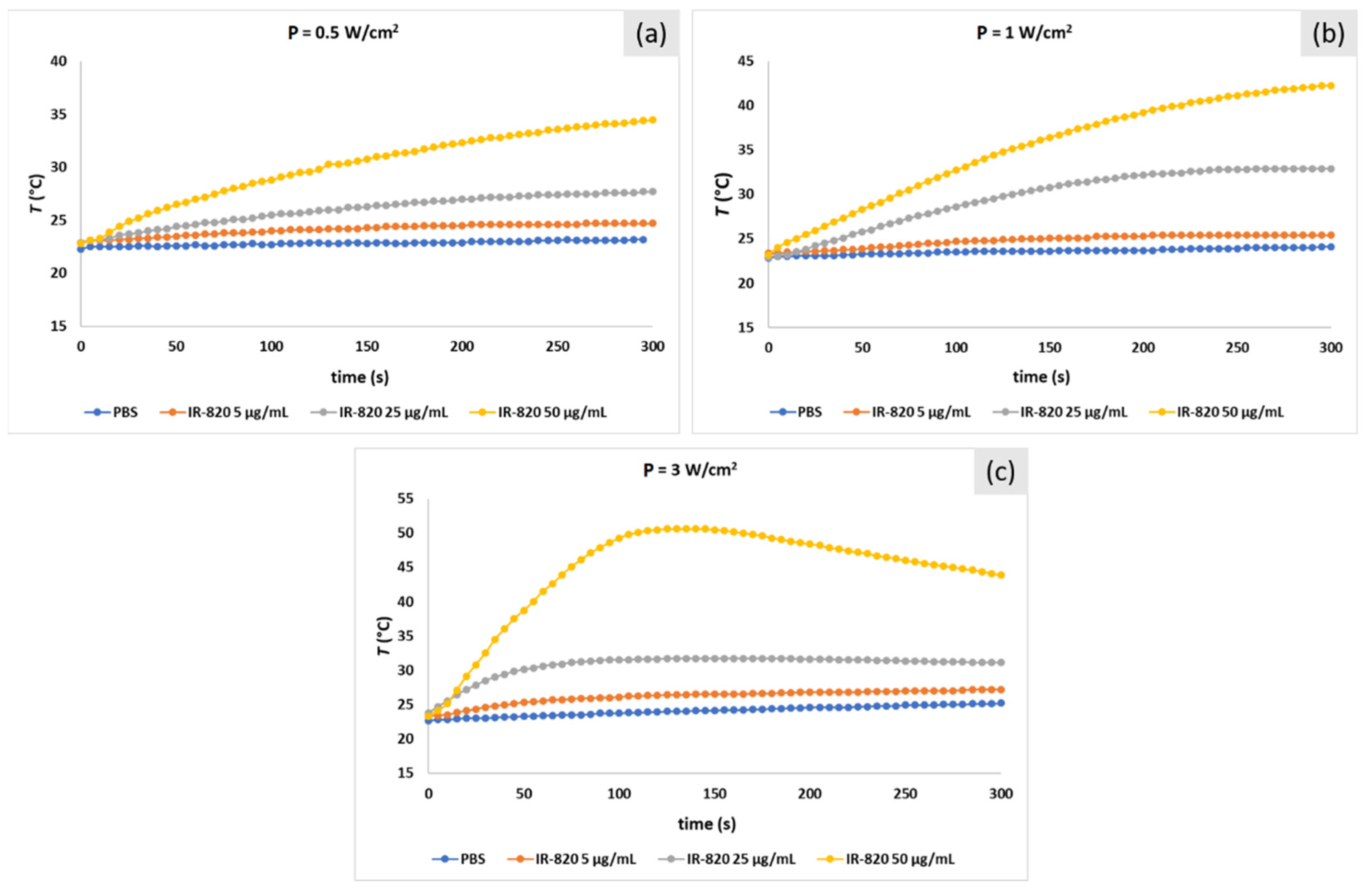
2.3. Photo-Stability Study of ICG and IR-820 Solutions in PBS
2.4. Preparation of ICG- and IR-820-Loaded Carriers
2.5. Temperature Stability of Dye-Loaded Liposomes and EMVs
2.6. Photo-Thermal Effect of ICG- and IR-820-Loaded Liposomes and EMVs
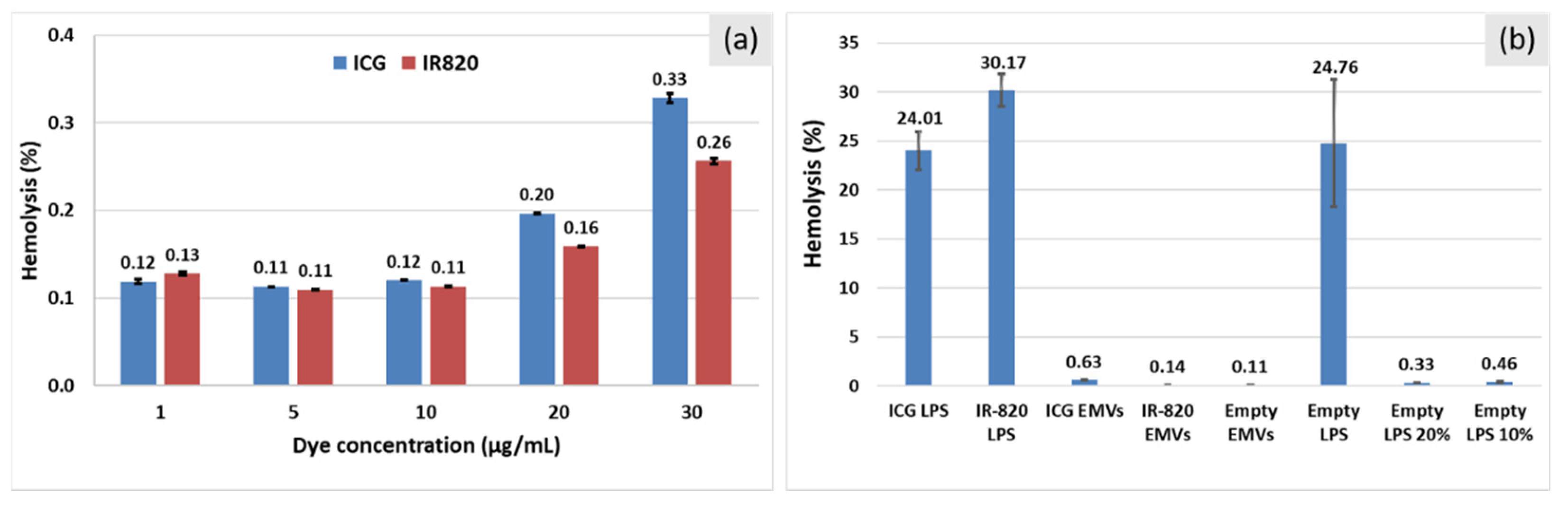
2.7. Hemolysis Study
3. Materials and Methods
3.1. Chemicals
3.2. Preparation of Dye-Loaded Liposomes
3.3. Preparation of Dye-Loaded EMVs
3.4. Purification of Dye-Loaded EMVs/Liposomes and Encapsulation Efficiency
3.5. Hydrodynamic Diameter and Zeta Potential Measurements
3.6. Absorption Spectra of IR-820 and ICG Solutions
3.7. Temperature Stability of IR-820 and ICG Solutions
3.8. Photo-Stability of IR-820 and ICG Solutions
3.9. Hemotoxicity Study
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Herrmann, J.; Bodmeier, R. Degradation Kinetics of Somatostatin in Aqueous Solution. Drug Dev. Ind. Pharm. 2003, 29, 1027–1033. [Google Scholar] [CrossRef] [PubMed]
- Holzer, W.; Mauerer, M.; Penzkofer, A.; Szeimies, R.M.; Abels, C.; Landthaler, M.; Bäumler, W. Photostability and thermal stability of indocyanine green. J. Photochem. Photobiol. B Biol. 1998, 47, 155–164. [Google Scholar] [CrossRef]
- Kraft, J.C.; Ho, R.J.Y. Interactions of indocyanine green and lipid in enhancing near-infrared fluorescence properties: The basis for near-infrared imaging in vivo. Biochemistry 2014, 53, 1275–1283. [Google Scholar] [CrossRef]
- Alander, J.T.; Kaartinen, I.; Laakso, A.; Pätilä, T.; Spillmann, T.; Tuchin, V.V.; Venermo, M.; Välisuo, P. A Review of indocyanine green fluorescent imaging in surgery. Int. J. Biomed. Imaging 2012, 2012, 940585. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Yang, F.; Jiang, G.; Wang, J. Applications of indocyanine green based near-infrared fluorescence imaging in thoracic surgery. J. Thorac. Dis. 2016, 8, S738–S743. [Google Scholar] [CrossRef] [PubMed]
- Qi, C.; Zhang, H.; Chen, Y.; Su, S.; Wang, X.F.; Huang, X.Q.; Fang, C.; Li, B.; Xia, X.M.; He, P. Effectiveness and safety of indocyanine green fluorescence imaging-guided hepatectomy for liver tumors: A systematic review and first meta-analysis. Photodiagn. Photodyn. Ther. 2019, 28, 346–353. [Google Scholar] [CrossRef]
- Shirata, C.; Kaneko, J.; Inagaki, Y.; Kokudo, T.; Sato, M.; Kiritani, S.; Akamatsu, N.; Arita, J.; Sakamoto, Y.; Hasegawa, K.; et al. Near-infrared photothermal/photodynamic therapy with indocyanine green induces apoptosis of hepatocellular carcinoma cells through oxidative stress. Sci. Rep. 2017, 7, 13958. [Google Scholar] [CrossRef]
- Engel, E.; Schraml, R.; Maisch, T.; Kobuch, K.; Konig, B.; Szeimies, R.M.; Hillenkamp, J.; Baümler, W.; Vasold, R. Light-induced decomposition of indocyanine green. Investig. Ophthalmol. Vis. Sci. 2008, 49, 1777–1783. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Fernandez, A.; Manchanda, R.; Lei, T.; Carvajal, D.A.; Tang, Y.; Kazmi, S.Z.R.; McGoron, A.J. Comparative study of the optical and heat generation properties of IR820 and indocyanine green. Mol. Imaging 2012, 11, 99–113. [Google Scholar] [CrossRef]
- Food and Drug Administration Product Insert: Indocyanine Green (IC-GreenTM). Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2006/011525s017lbl.pdf (accessed on 7 June 2021).
- Narayanan, R.; Kenney, M.C.; Kamjoo, S.; Trinh, T.H.T.; Seigel, G.M.; Resende, G.P.; Kuppermann, B.D. Toxicity of indocyanine green (ICG) in combination with light on retinal pigment epithelial cells and neurosensory retinal cells. Curr. Eye Res. 2005, 30, 471–478. [Google Scholar] [CrossRef]
- Bäumler, W.; Abels, C.; Karrer, S.; Weiß, T.; Messmann, H.; Landthaler, M.; Szeimies, R.-M. Photo-oxidative killing of human colonic cancer cells using indocyanine green and infrared light. Br. J. Cancer 1999, 80, 360–363. [Google Scholar] [CrossRef]
- Conceição, D.S.; Ferreira, D.P.; Vieira Ferreira, L.F. Photochemistry and cytotoxicity evaluation of heptamethinecyanine near infrared (NIR) dyes. Int. J. Mol. Sci. 2013, 14, 18557–18571. [Google Scholar] [CrossRef]
- Yoon, H.J.; Lee, H.S.; Lim, J.Y.; Park, J.H. Liposomal Indocyanine Green for Enhanced Photothermal Therapy. ACS Appl. Mater. Interfaces 2017, 9, 5683–5691. [Google Scholar] [CrossRef] [PubMed]
- Su, W.; He, M.; Xing, J.; Zhong, Y.; Li, Z. Facile synthesis of porous bifunctional Fe3O4@Y2O3:Ln nanocomposites using carbonized ferrocene as templates. RSC Adv. 2013, 3, 25970. [Google Scholar] [CrossRef]
- Phua, K.K.L.; Boczkowski, D.; Dannull, J.; Pruitt, S.; Leong, K.W.; Nair, S.K. Whole Blood Cells Loaded with Messenger RNA as an Anti-Tumor Vaccine. Adv. Healthc. Mater. 2014, 3, 837–842. [Google Scholar] [CrossRef]
- D’Alessandro, A.; Righetti, P.G.; Zolla, L. The red blood cell proteome and interactome: An update. J. Proteome Res. 2010, 9, 144–163. [Google Scholar] [CrossRef] [PubMed]
- Oldenborg, P.A.; Zheleznyak, A.; Fang, Y.F.; Lagenaur, C.F.; Gresham, H.D.; Lindberg, F.P. Role of CD47 as a marker of self on red blood cells. Science 2000, 288, 2051–2054. [Google Scholar] [CrossRef] [PubMed]
- Bahmani, B.; Bacon, D.; Anvari, B. Erythrocyte-derived photo-theranostic agents: Hybrid nano-vesicles containing indocyanine green for near infrared imaging and therapeutic applications. Sci. Rep. 2013, 3, 2180. [Google Scholar] [CrossRef] [PubMed]
- Burns, J.M.; Vankayala, R.; Mac, J.T.; Anvari, B. Erythrocyte-Derived Theranostic Nanoplatforms for Near Infrared Fluorescence Imaging and Photodestruction of Tumors. ACS Appl. Mater. Interfaces 2018, 10, 27621–27630. [Google Scholar] [CrossRef]
- Ryu, J.H.; Lee, S.; Son, S.; Kim, S.H.; Leary, J.F.; Choi, K.; Kwon, I.C. Theranostic nanoparticles for future personalized medicine. J. Control. Release 2014, 190, 477–484. [Google Scholar] [CrossRef]
- Weigand, R.; Rotermund, F.; Penzkofer, A. Aggregation dependent absorption reduction of indocyanine green. J. Phys. Chem. A 1997, 101, 7729–7734. [Google Scholar] [CrossRef]
- Jung, B.; Vullev, V.I.; Anvari, B. Revisiting indocyanine green: Effects of serum and physiological temperature on absorption and fluorescence characteristics. IEEE J. Sel. Top. Quantum Electron. 2014, 20, 7000409. [Google Scholar] [CrossRef]
- Emerson, E.S. The geometrical structure and absorption spectrum of a cyanine dye aggregate. J. Phys. Chem. 1967, 71, 2396–2403. [Google Scholar] [CrossRef]
- Zhou, J.F.; Chin, M.P.; Schafer, S.A. Aggregation and Degradation of Indocyanine Green. In Proceedings SPIE 2128, Laser Surgery: Advanced Characterization, Therapeutics, and Systems IV; SPIE Society of Photo-Optical Instrumentation Engineers: Bellingham, WA, USA, 1994; Volume 2128, pp. 495–505. [Google Scholar] [CrossRef]
- Saxena, V.; Sadoqi, M.; Shao, J. Degradation kinetics of indocyanine green in aqueous solution. J. Pharm. Sci. 2003, 92, 2090–2097. [Google Scholar] [CrossRef] [PubMed]
- Mindt, S.; Karampinis, I.; John, M.; Neumaier, M.; Nowak, K. Stability and degradation of indocyanine green in plasma, aqueous solution and whole blood. Photochem. Photobiol. Sci. 2018, 17, 1189–1196. [Google Scholar] [CrossRef]
- Gorka, A.P.; Nani, R.R.; Schnermann, M.J. Cyanine polyene reactivity: Scope and biomedical applications. Org. Biomol. Chem. 2015, 13, 7584–7598. [Google Scholar] [CrossRef]
- Saxena, V.; Sadoqi, M.; Shao, J. Enhanced photo-stability, thermal-stability and aqueous-stability of indocyanine green in polymeric nanoparticulate systems. J. Photochem. Photobiol. B Biol. 2004, 74, 29–38. [Google Scholar] [CrossRef]
- Lee, Y.-H.; Lai, Y.-H. Synthesis, Characterization, and Biological Evaluation of Anti-HER2 Indocyanine Green-Encapsulated PEG-Coated PLGA Nanoparticles for Targeted Phototherapy of Breast Cancer Cells. PLoS ONE 2016, 11, e0168192. [Google Scholar] [CrossRef] [PubMed]
- Ehrenberg, B.; Anderson, J.L.; Foote, C.S. Kinetics and Yield of Singlet Oxygen Photosensitized by Hypericin in Organic and Biological Media. Photochem. Photobiol. 1998, 68, 135–140. [Google Scholar] [CrossRef] [PubMed]
- Hackbarth, S.; Pfitzner, S.; Guo, L.; Ge, J.; Wang, P.; Röder, B. Singlet Oxygen Kinetics in Polymeric Photosensitizers. J. Phys. Chem. C 2018, 122, 12071–12076. [Google Scholar] [CrossRef]
- Yaseen, M.A.; Yu, J.; Wong, M.S.; Anvari, B. Stability assessment of indocyanine green within dextran-coated mesocapsules by absorbance spectroscopy. J. Biomed. Opt. 2007, 12, 064031. [Google Scholar] [CrossRef][Green Version]
- Yeroslavsky, G.; Umezawa, M.; Okubo, K.; Nigoghossian, K.; Thi Kim Dung, D.; Miyata, K.; Kamimura, M.; Soga, K. Stabilization of indocyanine green dye in polymeric micelles for NIR-II fluorescence imaging and cancer treatment. Biomater. Sci. 2020, 8, 2245–2254. [Google Scholar] [CrossRef]
- Yeroslavsky, G.; Umezawa, M.; Okubo, K.; Nigoghossian, K.; Dung, D.T.K.; Kamimura, M.; Soga, K. Photostabilization of indocyanine green dye by energy transfer in phospholipid-peg micelles. J. Photopolym. Sci. Technol. 2019, 32, 115–121. [Google Scholar] [CrossRef]
- Valcourt, D.M.; Dang, M.N.; Day, E.S. IR820-loaded PLGA nanoparticles for photothermal therapy of triple-negative breast cancer. J. Biomed. Mater. Res. Part A 2019, 107, 1702–1712. [Google Scholar] [CrossRef] [PubMed]
- Banno, B.; Ickenstein, L.M.; Chiu, G.N.C.; Bally, M.B.; Thewalt, J.; Brief, E.; Wasan, E.K. The functional roles of poly(ethylene glycol)-lipid and lysolipid in the drug retention and release from lysolipid-containing thermosensitive liposomes in vitro and in vivo. J. Pharm. Sci. 2010, 99, 2295–2308. [Google Scholar] [CrossRef]
- Needham, D.; Zhelev, D.V. Lysolipid exchange with lipid vesicle membranes. Ann. Biomed. Eng. 1995, 23, 287–298. [Google Scholar] [CrossRef] [PubMed]
- Kostevšek, N.; Cheung, C.C.L.L.; Serša, I.; Kreft, M.E.; Monaco, I.; Franchini, M.C.; Vidmar, J.; Al-Jamal, W.T.; Comes Franchini, M.; Vidmar, J.; et al. Magneto-liposomes as MRI contrast agents: A systematic study of different liposomal formulations. Nanomaterials 2020, 10, 889. [Google Scholar] [CrossRef]
- Chen, W.R.; Adams, R.L.; Higgins, A.K.; Bartels, K.E.; Nordquist, R.E. Photothermal effects on murine mammary tumors using indocyanine green and an 808-nm diode laser: An in vivo efficacy study. Cancer Lett. 1996, 98, 169–173. [Google Scholar] [CrossRef]
- Su, F.Y.; Chen, J.; Son, H.N.; Kelly, A.M.; Convertine, A.J.; West, T.E.; Skerrett, S.J.; Ratner, D.M.; Stayton, P.S. Polymer-augmented liposomes enhancing antibiotic delivery against intracellular infections. Biomater. Sci. 2018, 6, 1976–1985. [Google Scholar] [CrossRef]

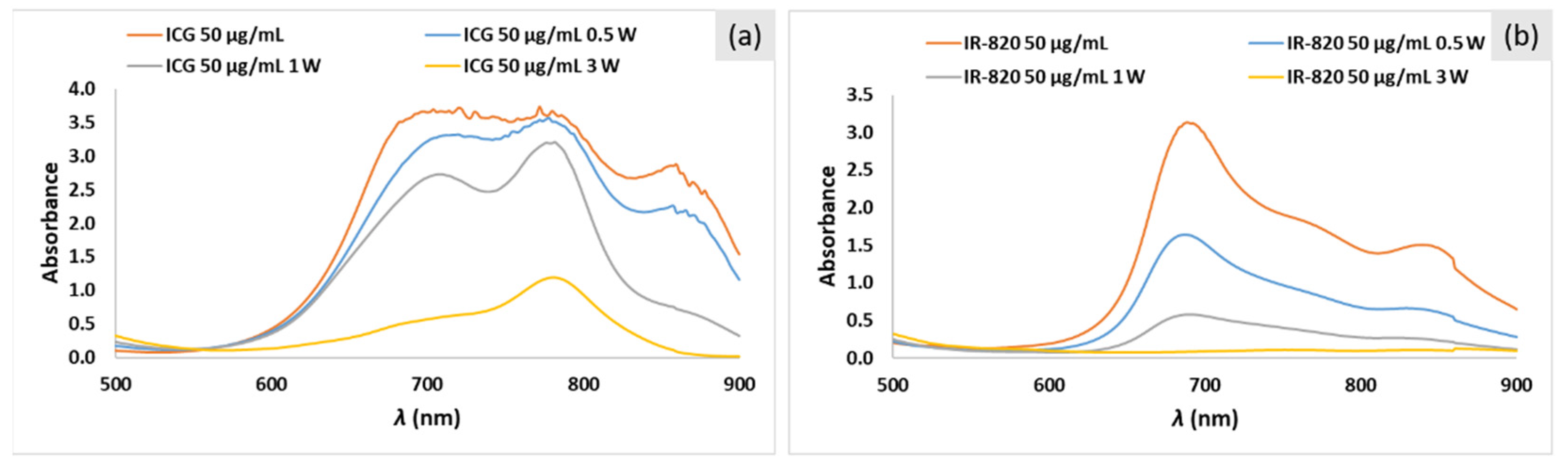
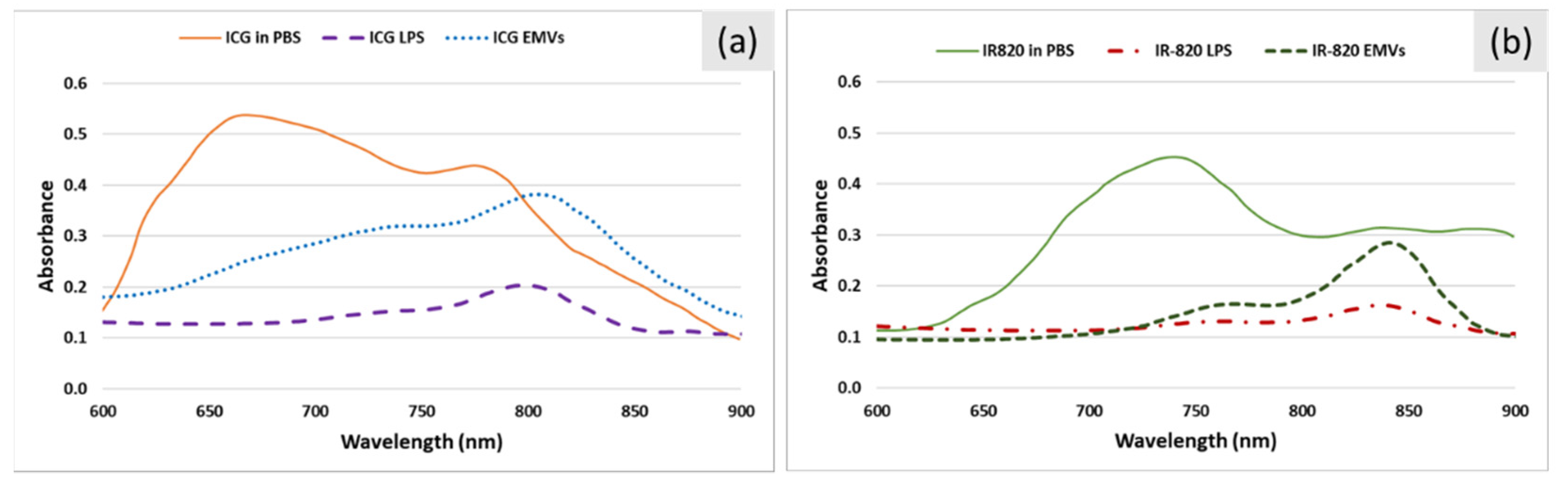
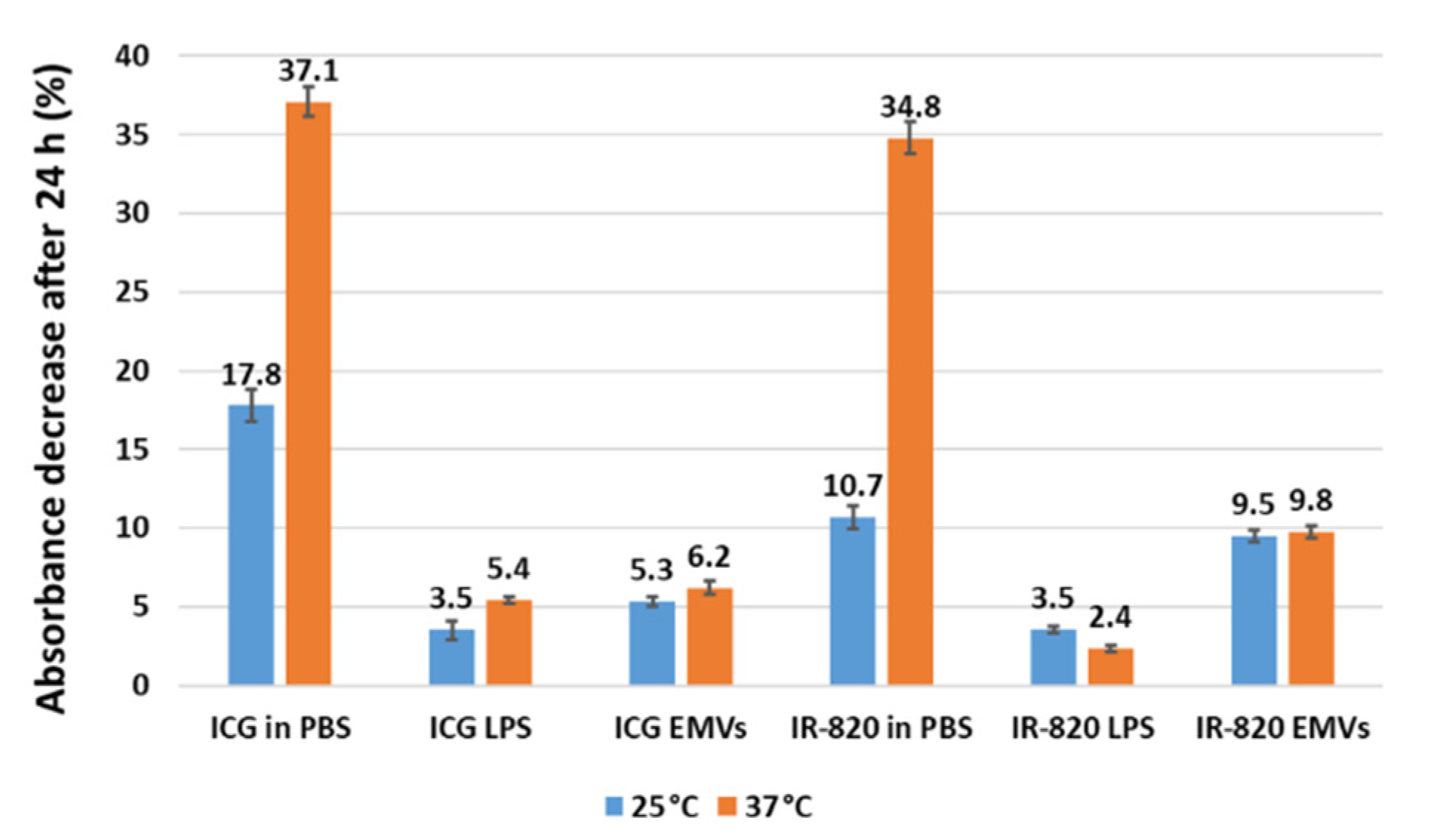
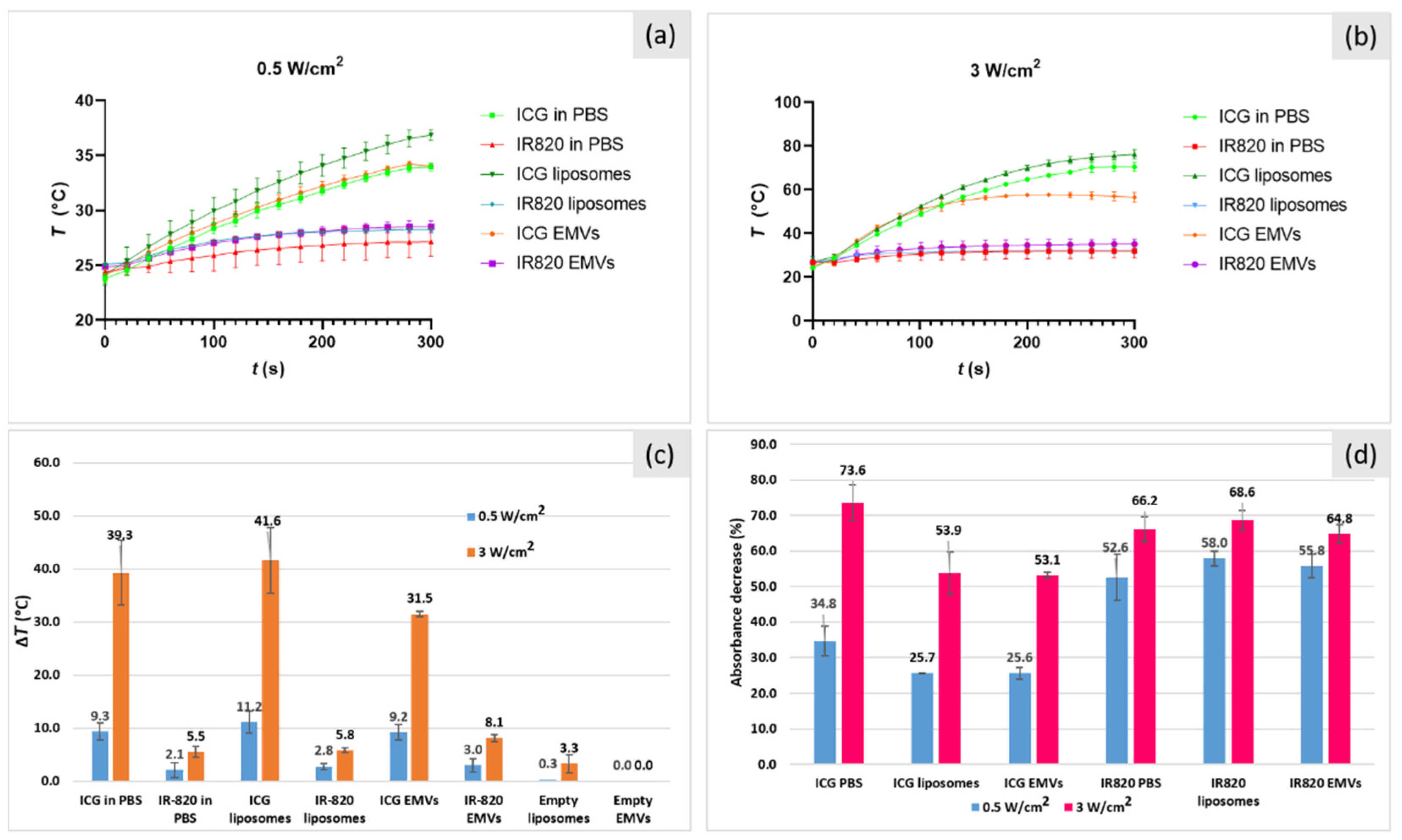
| Concentration (μg/mL) | P (W/cm2) | Absorbance before Irradiation | Absorbance after Irradiation | % Degradation | ΔT (°C) |
|---|---|---|---|---|---|
| 5 | 0.5 | 0.748 | 0.295 | 60.5 | 3.8 |
| 5 | 1.0 | 0.748 | 0.237 | 68.3 | 5.1 |
| 5 | 3.0 | 0.748 | 0.145 | 80.6 | 7.5 |
| 25 | 0.5 | 2.326 | 1.884 | 19.0 | 13.2 |
| 25 | 1.0 | 2.326 | 1.398 | 39.9 | 21.8 |
| 25 | 3.0 | 2.326 | 0.375 | 83.9 | 31.1 |
| 50 | 0.5 | 3.678 | 3.567 | 3.0 | 12.8 |
| 50 | 1.0 | 3.678 | 3.197 | 13.1 | 24.2 |
| 50 | 3.0 | 3.678 | 1.182 | 67.9 | 50.7 |
| Concentration (μg/mL) | P (W/cm2) | Absorbance before Irradiation | Absorbance after Irradiation | % Degradation | ΔT (°C) |
|---|---|---|---|---|---|
| 5 | 0.5 | 0.160 | 0.070 | 56.3 | 1.8 |
| 5 | 1.0 | 0.160 | 0.056 | 64.8 | 2.0 |
| 5 | 3.0 | 0.160 | 0.036 | 77.4 | 3.4 |
| 25 | 0.5 | 0.491 | 0.150 | 69.5 | 4.8 |
| 25 | 1.0 | 0.491 | 0.117 | 76.1 | 10.0 |
| 25 | 3.0 | 0.491 | 0.095 | 80.7 | 8.0 |
| 50 | 0.5 | 1.410 | 0.655 | 53.5 | 11.7 |
| 50 | 1.0 | 1.410 | 0.271 | 80.8 | 19.0 |
| 50 | 3.0 | 1.410 | 0.093 | 93.4 | 27.3 |
| Sample | Size (nm) | PDI | EE (%) |
|---|---|---|---|
| Empty LPS | 115 ± 1 | 0.087 ± 0.050 | - |
| Empty EMVs | 234 ± 4 | 0.238 ± 0.011 | - |
| ICG LPS | 126 ± 3 | 0.171 ± 0.083 | 45 ± 3 |
| ICG-EMVs | 215 ± 5 | 0.198 ± 0.075 | 57 ± 2 |
| IR-820 LPS | 116 ± 2 | 0.142 ± 0.091 | 46 ± 4 |
| IR-820 EMVs | 260 ± 4 | 0.229 ± 0.081 | 53 ± 3 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Della Pelle, G.; Delgado López, A.; Salord Fiol, M.; Kostevšek, N. Cyanine Dyes for Photo-Thermal Therapy: A Comparison of Synthetic Liposomes and Natural Erythrocyte-Based Carriers. Int. J. Mol. Sci. 2021, 22, 6914. https://doi.org/10.3390/ijms22136914
Della Pelle G, Delgado López A, Salord Fiol M, Kostevšek N. Cyanine Dyes for Photo-Thermal Therapy: A Comparison of Synthetic Liposomes and Natural Erythrocyte-Based Carriers. International Journal of Molecular Sciences. 2021; 22(13):6914. https://doi.org/10.3390/ijms22136914
Chicago/Turabian StyleDella Pelle, Giulia, Andrea Delgado López, Marina Salord Fiol, and Nina Kostevšek. 2021. "Cyanine Dyes for Photo-Thermal Therapy: A Comparison of Synthetic Liposomes and Natural Erythrocyte-Based Carriers" International Journal of Molecular Sciences 22, no. 13: 6914. https://doi.org/10.3390/ijms22136914
APA StyleDella Pelle, G., Delgado López, A., Salord Fiol, M., & Kostevšek, N. (2021). Cyanine Dyes for Photo-Thermal Therapy: A Comparison of Synthetic Liposomes and Natural Erythrocyte-Based Carriers. International Journal of Molecular Sciences, 22(13), 6914. https://doi.org/10.3390/ijms22136914







