Targeting Telomere Biology in Acute Lymphoblastic Leukemia
Abstract
1. Introduction
2. Patients and Methods
2.1. Study Design
2.2. Patients
2.3. Measurement of Telomere Length
2.4. Measurement of Telomerase Activity
2.5. Ex Vivo Assays Testing the Effects of Imetelstat on Primary B-ALL Cells
2.6. Statistical Analyses
3. Results
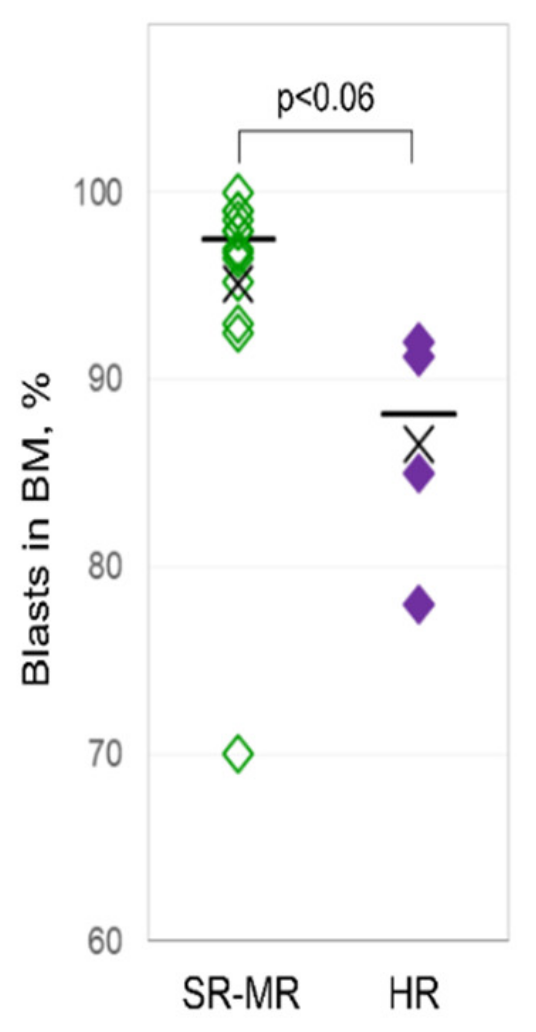
3.1. Characteristics of Patients
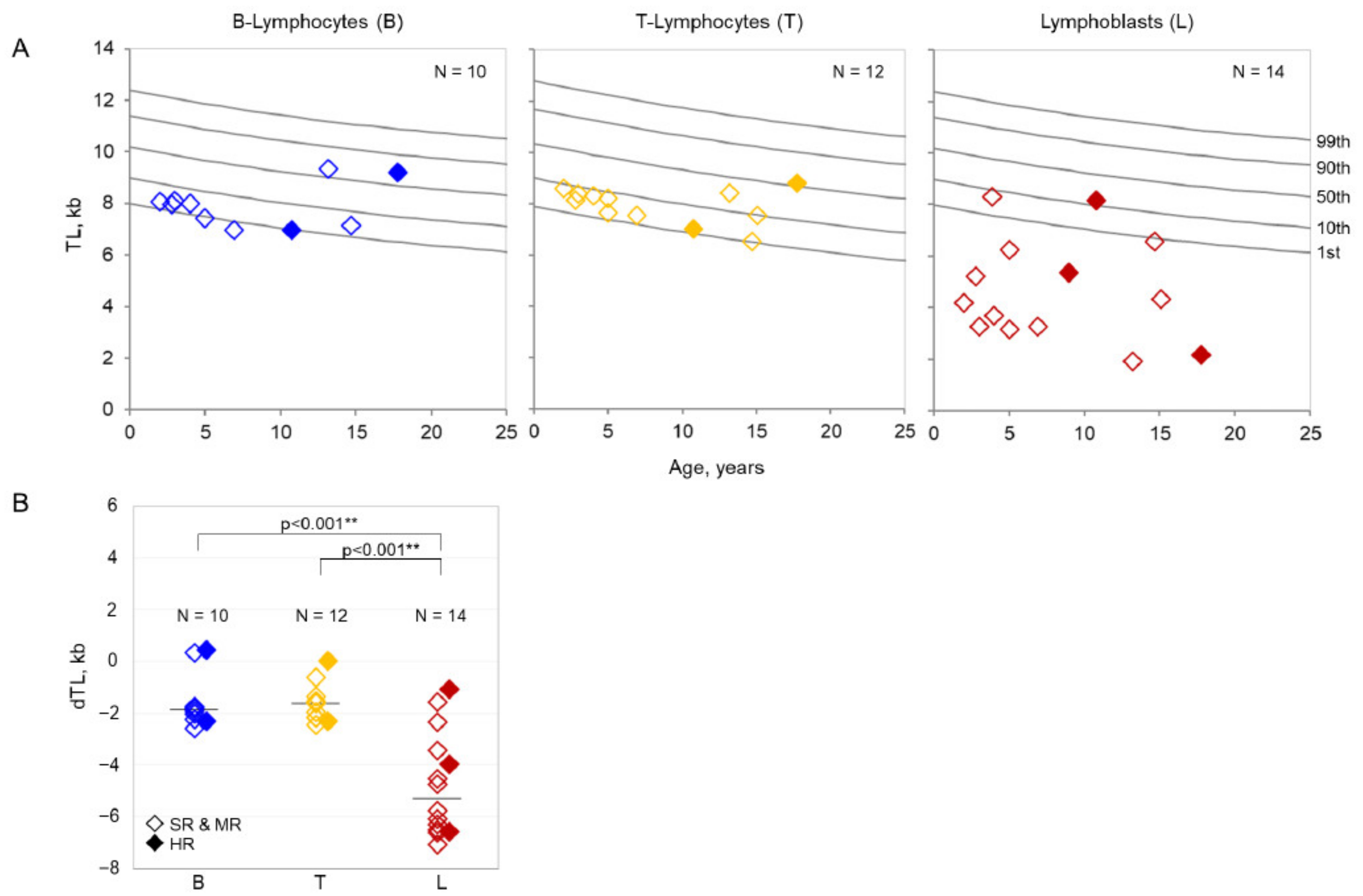
3.2. Telomere Length
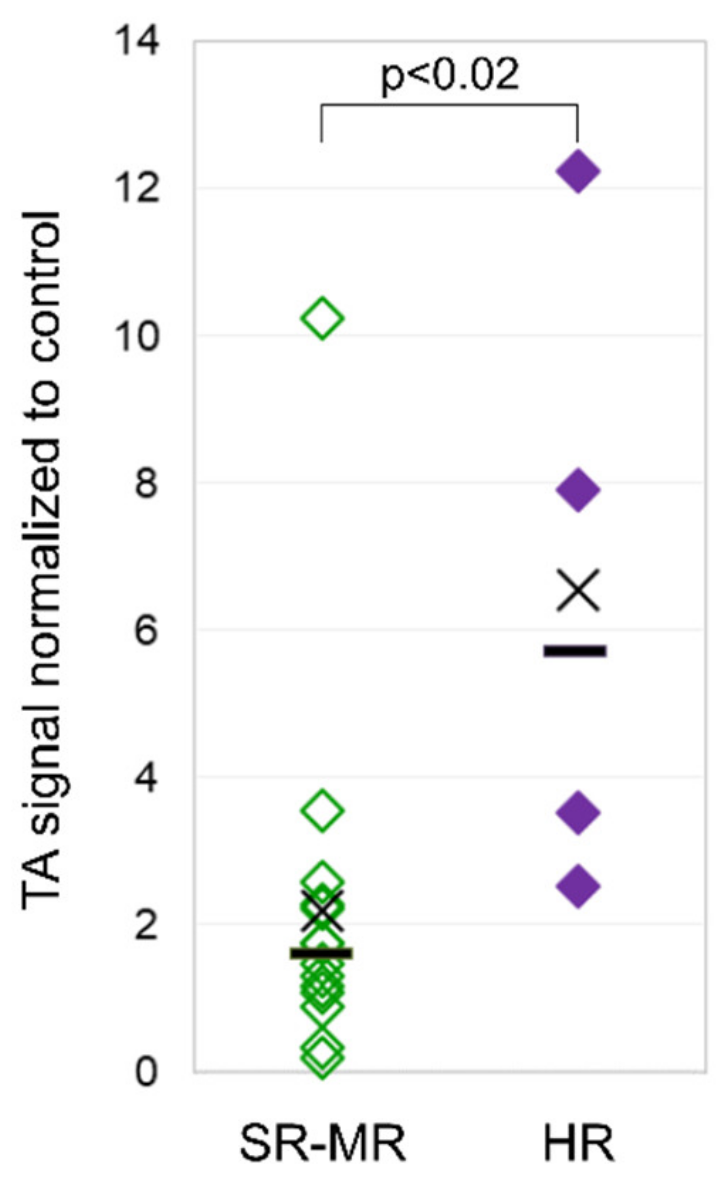
3.3. Telomerase Activity
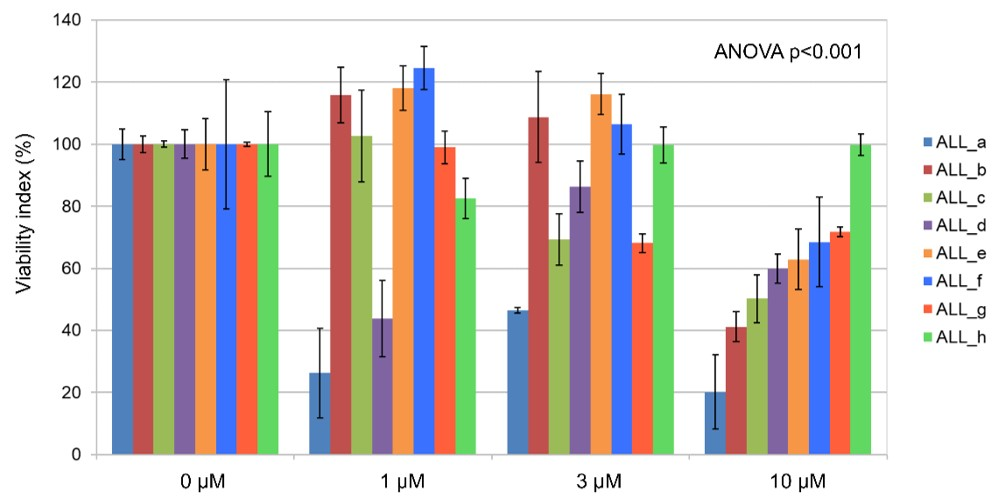
3.4. Ex Vivo Effects of Imetelstat on Primary B-ALL Cells
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Inaba, H.; Mullighan, C.G. Pediatric acute lymphoblastic leukemia. Haematologica 2020. [Google Scholar] [CrossRef]
- Terwilliger, T.; Abdul-Hay, M. Acute lymphoblastic leukemia: A comprehensive review and 2017 update. Blood Cancer J. 2017, 7, e577. [Google Scholar] [CrossRef] [PubMed]
- Moorman, A.V. New and emerging prognostic and predictive genetic biomarkers in B-cell precursor acute lymphoblastic leukemia. Haematologica 2016, 101, 407–416. [Google Scholar] [CrossRef]
- Bassan, R.; Pavoni, C.; Intermesoli, T.; Spinelli, O.; Tosi, M.; Audisio, E.; Marmont, F.; Cattaneo, C.; Borlenghi, E.; Cortelazzo, S.; et al. Updated risk-oriented strategy for acute lymphoblastic leukemia in adult patients 18-65 years: NILG ALL 10/07. Blood Cancer J 2020, 10, 119. [Google Scholar] [CrossRef] [PubMed]
- Shay, J.W. Role of Telomeres and Telomerase in Aging and Cancer. Cancer Discov. 2016, 6, 584–593. [Google Scholar] [CrossRef]
- Srinivas, N.; Rachakonda, S.; Kumar, R. Telomeres and Telomere Length: A General Overview. Cancers 2020, 12, 558. [Google Scholar] [CrossRef]
- Adamson, D.J.; King, D.J.; Haites, N.E. Significant telomere shortening in childhood leukemia. Cancer Genet. Cytogenet. 1992, 61, 204–206. [Google Scholar] [CrossRef]
- Engelhardt, M.; Ozkaynak, M.F.; Drullinsky, P.; Sandoval, C.; Tugal, O.; Jayabose, S.; Moore, M.A. Telomerase activity and telomere length in pediatric patients with malignancies undergoing chemotherapy. Leukemia 1998, 12, 13–24. [Google Scholar] [CrossRef]
- Hiyama, E.; Hiyama, K. Telomerase as tumor marker. Cancer Lett. 2003, 194, 221–233. [Google Scholar] [CrossRef]
- Tabori, U.; Dome, J.S. Telomere biology of pediatric cancer. Cancer Investig. 2007, 25, 197–208. [Google Scholar] [CrossRef]
- Kim, M.; Oh, B.; Kim, T.Y.; Yoon, S.S.; Kim, S.Y.; Hwang, S.M.; Lee, D.S. Elevated telomerase activity in essential thrombocythemia with extreme thrombocytosis. Clin. Biochem. 2014, 47, 389–392. [Google Scholar] [CrossRef]
- Bruedigam, C.; Lane, S.W. Telomerase in hematologic malignancies. Curr. Opin. Hematol. 2016, 23, 346–353. [Google Scholar] [CrossRef] [PubMed]
- Bruedigam, C.; Bagger, F.O.; Heidel, F.H.; Paine Kuhn, C.; Guignes, S.; Song, A.; Austin, R.; Vu, T.; Lee, E.; Riyat, S.; et al. Telomerase inhibition effectively targets mouse and human AML stem cells and delays relapse following chemotherapy. Cell Stem Cell 2014, 15, 775–790. [Google Scholar] [CrossRef] [PubMed]
- Baerlocher, G.M.; Oppliger Leibundgut, E.; Ottmann, O.G.; Spitzer, G.; Odenike, O.; McDevitt, M.A.; Roth, A.; Daskalakis, M.; Burington, B.; Stuart, M.; et al. Telomerase Inhibitor Imetelstat in Patients with Essential Thrombocythemia. N. Engl. J. Med. 2015, 373, 920–928. [Google Scholar] [CrossRef]
- Steensma, D.P.; Fenaux, P.; Van Eygen, K.; Raza, A.; Santini, V.; Germing, U.; Font, P.; Diez-Campelo, M.; Thepot, S.; Vellenga, E.; et al. Imetelstat Achieves Meaningful and Durable Transfusion Independence in High Transfusion-Burden Patients With Lower-Risk Myelodysplastic Syndromes in a Phase II Study. J. Clin. Oncol. 2021, 39, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Mascarenhas, J.; Komrokji, R.S.; Cavo, M.; Martino, B.; Niederwieser, D.; Reiter, A.; Scott, B.L.; Baer, M.R.; Hoffman, R.; Odenike, O.; et al. Imetelstat Is Effective Treatment for Patients with Intermediate-2 or High-Risk Myelofibrosis Who Have Relapsed on or Are Refractory to Janus Kinase Inhibitor Therapy: Results of a Phase 2 Randomized Study of Two Dose Levels. Blood 2018, 132. [Google Scholar] [CrossRef]
- Schultz, K.R.; Pullen, D.J.; Sather, H.N.; Shuster, J.J.; Devidas, M.; Borowitz, M.J.; Carroll, A.J.; Heerema, N.A.; Rubnitz, J.E.; Loh, M.L.; et al. Risk- and response-based classification of childhood B-precursor acute lymphoblastic leukemia: A combined analysis of prognostic markers from the Pediatric Oncology Group (POG) and Children’s Cancer Group (CCG). Blood 2007, 109, 926–935. [Google Scholar] [CrossRef]
- Baerlocher, G.M.; Vulto, I.; de Jong, G.; Lansdorp, P.M. Flow cytometry and FISH to measure the average length of telomeres (flow FISH). Nat. Protoc. 2006, 1, 2365–2376. [Google Scholar] [CrossRef]
- Mender, I.; Shay, J.W. Telomerase Repeated Amplification Protocol (TRAP). Bio-Protocol 2015, 5. [Google Scholar] [CrossRef]
- Makrilia, N.; Kollias, A.; Manolopoulos, L.; Syrigos, K. Cell adhesion molecules: Role and clinical significance in cancer. Cancer Investig. 2009, 27, 1023–1037. [Google Scholar] [CrossRef]
- Windisch, R.; Pirschtat, N.; Kellner, C.; Chen-Wichmann, L.; Lausen, J.; Humpe, A.; Krause, D.S.; Wichmann, C. Oncogenic deregulation of cell adhesion molecules in leukemia. Cancers 2019, 11, 311. [Google Scholar] [CrossRef]
- Roth, A.; de Beer, D.; Nuckel, H.; Sellmann, L.; Duhrsen, U.; Durig, J.; Baerlocher, G.M. Significantly shorter telomeres in T-cells of patients with ZAP-70+/CD38+ chronic lymphocytic leukaemia. Br. J. Haematol. 2008, 143, 383–386. [Google Scholar] [CrossRef]
- Malaska, J.; Sklenickova, M.; Krejci, K.; Fajkusova, L.; Bajer, M.; Hrstkova, H.; Fajkus, J. Telomerase activity and expression and telomere analysis in situ in the course of treatment of childhood leukemias. Blood Cells Mol. Dis. 2000, 26, 534–539. [Google Scholar] [CrossRef]
- Franco, S.; Ozkaynak, M.F.; Sandoval, C.; Tugal, O.; Jayabose, S.; Engelhardt, M.; Moore, M.A. Telomere dynamics in childhood leukemia and solid tumors: A follow-up study. Leukemia 2003, 17, 401–410. [Google Scholar] [CrossRef]
- Januszkiewicz, D.; Wysoki, J.; Lewandowski, K.; Pernak, M.; Nowicka, K.; Rembowska, J.; Nowak, J. Lack of correlation between telomere length and telomerase activity and expression in leukemic cells. Int. J. Mol. Med. 2003, 12, 935–938. [Google Scholar] [CrossRef] [PubMed]
- Kleideiter, E.; Bangerter, U.; Schwab, M.; Boukamp, P.; Koscielniak, E.; Klotz, U.; Greil, J. Telomeres and telomerase in paediatric patients with T-cell acute lymphoblastic leukaemia (T-ALL). Leukemia 2005, 19, 296–298. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Jones, C.H.; Pepper, C.; Baird, D.M. Telomere dysfunction and its role in haematological cancer. Br. J. Haematol. 2012, 156, 573–587. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Xiao, H.; Zhang, X.; Wang, C.; Huang, H. The role of telomeres and telomerase in hematologic malignancies and hematopoietic stem cell transplantation. J. Hematol. Oncol. 2014, 7, 61. [Google Scholar] [CrossRef]
- Baerlocher, G.M.; Mak, J.; Roth, A.; Rice, K.S.; Lansdorp, P.M. Telomere shortening in leukocyte subpopulations from baboons. J. Leukoc. Biol. 2003, 73, 289–296. [Google Scholar] [CrossRef]
- Guilleret, I.; Yan, P.; Grange, F.; Braunschweig, R.; Bosman, F.T.; Benhattar, J. Hypermethylation of the human telomerase catalytic subunit (htert) gene correlates with telomerase activity. Int. J. Cancer 2002, 101, 335–341. [Google Scholar] [CrossRef] [PubMed]
- Borssén, M.; Cullman, I.; Norén-Nyström, U.; Sundström, C.; Porwit, A.; Forestier, E.; Roos, G. hTERT promoter methylation and telomere length in childhood acute lymphoblastic leukemia: Associations with immunophenotype and cytogenetic subgroup. Exp. Hematol. 2011, 39, 1144–1151. [Google Scholar] [CrossRef] [PubMed]
- Qian, H.; Chen, X.; Liu, S.; Wen, R.; Wen, F. Methylation of cdkn2b cpg islands is associated with upregulated telomerase activity in children with acute lymphoblastic leukemia. Oncol. Lett. 2017, 13. [Google Scholar] [CrossRef][Green Version]
- Fernandez-Marcelo, T.; Gomez, A.; Pascua, I.; de Juan, C.; Head, J.; Hernando, F.; Jarabo, J.R.; Calatayud, J.; Torres-Garcia, A.J.; Iniesta, P. Telomere length and telomerase activity in non-small cell lung cancer prognosis: Clinical usefulness of a specific telomere status. J. Exp. Clin. Cancer Res. 2015, 34, 78. [Google Scholar] [CrossRef]
- Tatsumoto, N.; Hiyama, E.; Murakami, Y.; Imamura, Y.; Shay, J.W.; Matsuura, Y.; Yokoyama, T. High telomerase activity is an independent prognostic indicator of poor outcome in colorectal cancer. Clin. Cancer Res. 2000, 6, 2696–2701. [Google Scholar]
- Sellmann, L.; de Beer, D.; Bartels, M.; Opalka, B.; Nuckel, H.; Duhrsen, U.; Durig, J.; Seifert, M.; Siemer, D.; Kuppers, R.; et al. Telomeres and prognosis in patients with chronic lymphocytic leukaemia. Int. J. Hematol. 2011, 93, 74–82. [Google Scholar] [CrossRef] [PubMed]
- Ackermann, S.; Cartolano, M.; Hero, B.; Welte, A.; Kahlert, Y.; Roderwieser, A.; Bartenhagen, C.; Walter, E.; Gecht, J.; Kerschke, L.; et al. A mechanistic classification of clinical phenotypes in neuroblastoma. Science 2018, 362, 1165–1170. [Google Scholar] [CrossRef] [PubMed]
- Verstovsek, S.; Manshouri, T.; Smith, F.O.; Giles, F.J.; Cortes, J.; Estey, E.; Kantarjian, H.; Keating, M.; Jeha, S.; Albitar, M. Telomerase activity is prognostic in pediatric patients with acute myeloid leukemia: Comparison with adult acute myeloid leukemia. Cancer 2003, 97, 2212–2217. [Google Scholar] [CrossRef]
- Mascarenhas, J.K.R.; Cavo, M.; Martino, B.; Niederwieser, D.; Reiter, A.; Scott, B.L.; Baer, M.R.; Hoffman, R.; Odenike, O.; Bussolari, J.; et al. Telomerase Activity, Telomere Length and hTERT Expression Correlate with Clinical Outcomes in Higher-Risk Myelofibrosis (MF) Relapsed/Refractory (R/R) to Janus Kinase Inhibitor Treated with Imetelstat. In Proceedings of the 62nd ASH Annual Meeting, San Diego, CA, USA, 6 December 2020; American Society of Hematology: Washington, DC, USA, 2020. [Google Scholar]
- Messinger, Y.; Gaynon, P.; Raetz, E.; Hutchinson, R.; Dubois, S.; Glade-Bender, J.; Sposto, R.; van der Giessen, J.; Eckroth, E.; Bostrom, B.C. Phase I study of bortezomib combined with chemotherapy in children with relapsed childhood acute lymphoblastic leukemia (ALL): A report from the therapeutic advances in childhood leukemia (TACL) consortium. Pediatr. Blood Cancer 2010, 55, 254–259. [Google Scholar] [CrossRef]
- Messinger, Y.H.; Gaynon, P.S.; Sposto, R.; van der Giessen, J.; Eckroth, E.; Malvar, J.; Bostrom, B.C.; Therapeutic Advances in Childhood, L.; Lymphoma, C. Bortezomib with chemotherapy is highly active in advanced B-precursor acute lymphoblastic leukemia: Therapeutic Advances in Childhood Leukemia & Lymphoma (TACL) Study. Blood 2012, 120, 285–290. [Google Scholar] [CrossRef]
| Characteristics at Diagnosis of ALL | Overall (n = 18) | Non-High Risk (n = 14) | High Risk (n = 4) |
| Age, median years (range) | 6.5 (2.2–17.8) | 5.6 (2.2–15.1) | 9.9 (5.8–17.8) |
| Sex (f/m) | 7/11 | 6/8 | 1/3 |
| Hematology | |||
| Leukocyte count in PB (G/L) median (range) | 9.1 (0.8–75.9) | 7.7 (0.8–75.9) | 21.9 (5.7–32.3) |
| Lymphocyte count in PB (G/L) median (range) | 2.0 (0.6–8.1) | 2.0 (0.6–8.1) | 2.0 (0.7–2.3) |
| Lymphoblast count in PB (G/L) median (range) | 5.9 (0.0–35.1) | 4.1 (0.0–35.1) | 17.1 (3.2–27.8) |
| Lymphoblast proportion in PB (%) median (range) | 68 (0–90) | 64 (0–90) | 79 (56–87) |
| Lymphoblast proportion in BM (%) median (range) | 97 (70–100) | 98 (70–100) | 88 (78–92) |
| Cytogenetics | |||
| t(12;21)/ETV6-RUNX1 | 6 | 6 | 0 |
| Hyperdiploidy | 4 | 4 | 0 |
| Deletion 9p + other aberration | 3 | 3 | 0 |
| t(1;19)/TCF3-PXB1 | 2 | 2 | 0 |
| Other | 3 | 0 | 3 |
| Treatment response | |||
| Blast count in PB day 8 (G/L) median (range) | 0.028 (0–0.600) | 0.028 (0–0.500) | 0.181 (0.003–0.600) |
| FACS-MRD in BM day 15 (%) median (range) | 0.30 (0–14.18) | 0.30 (0–5.40) | 5.63 (0.03–14.18) |
| PCR-MRD BM day 33 (log) median (range) | 1.0 × 10−6 (neg. −1.0 × 10−3) | 1.0 × 10−6 (neg. −2.0 × 10−4) | 7.0 × 10−4 (neg. −1.0 × 10−3) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Karow, A.; Haubitz, M.; Oppliger Leibundgut, E.; Helsen, I.; Preising, N.; Steiner, D.; Dantonello, T.M.; Ammann, R.A.; Roessler, J.; Kartal-Kaess, M.; et al. Targeting Telomere Biology in Acute Lymphoblastic Leukemia. Int. J. Mol. Sci. 2021, 22, 6653. https://doi.org/10.3390/ijms22136653
Karow A, Haubitz M, Oppliger Leibundgut E, Helsen I, Preising N, Steiner D, Dantonello TM, Ammann RA, Roessler J, Kartal-Kaess M, et al. Targeting Telomere Biology in Acute Lymphoblastic Leukemia. International Journal of Molecular Sciences. 2021; 22(13):6653. https://doi.org/10.3390/ijms22136653
Chicago/Turabian StyleKarow, Axel, Monika Haubitz, Elisabeth Oppliger Leibundgut, Ingrid Helsen, Nicole Preising, Daniela Steiner, Tobias M. Dantonello, Roland A. Ammann, Jochen Roessler, Mutlu Kartal-Kaess, and et al. 2021. "Targeting Telomere Biology in Acute Lymphoblastic Leukemia" International Journal of Molecular Sciences 22, no. 13: 6653. https://doi.org/10.3390/ijms22136653
APA StyleKarow, A., Haubitz, M., Oppliger Leibundgut, E., Helsen, I., Preising, N., Steiner, D., Dantonello, T. M., Ammann, R. A., Roessler, J., Kartal-Kaess, M., Röth, A., & Baerlocher, G. M. (2021). Targeting Telomere Biology in Acute Lymphoblastic Leukemia. International Journal of Molecular Sciences, 22(13), 6653. https://doi.org/10.3390/ijms22136653







