Levels of Circulating PD-L1 Are Decreased in Patients with Resectable Cholangiocarcinoma
Abstract
1. Introduction
2. Results
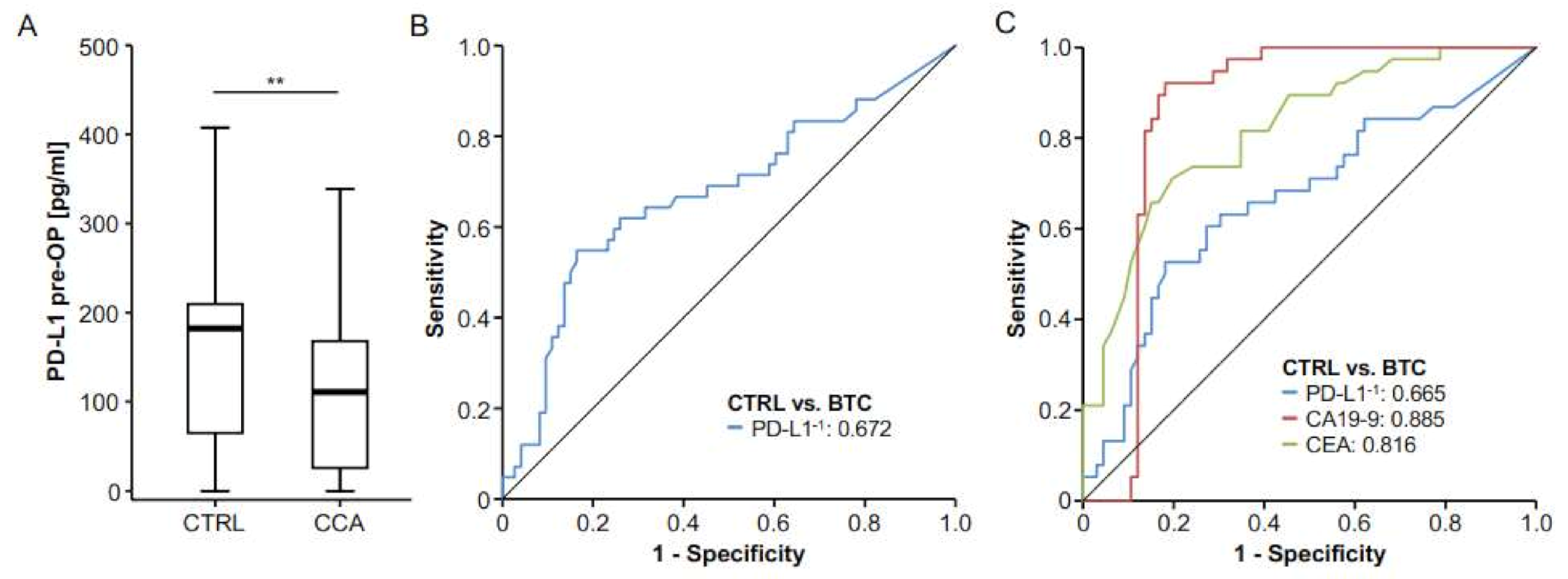
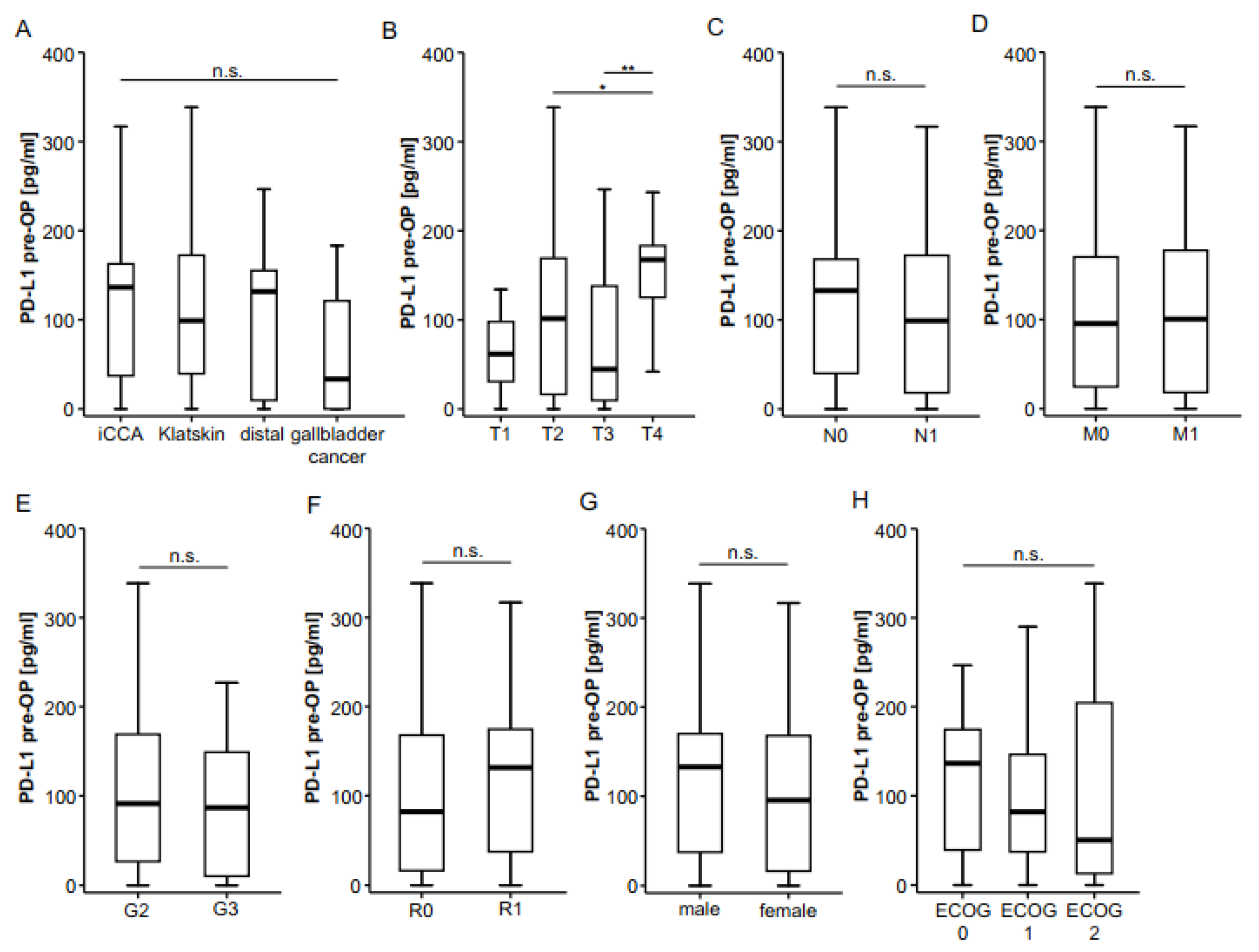
2.1. Concentrations of Circulating Preoperative PD-L1 Are Decreased in Patients with BTC at Diagnosis
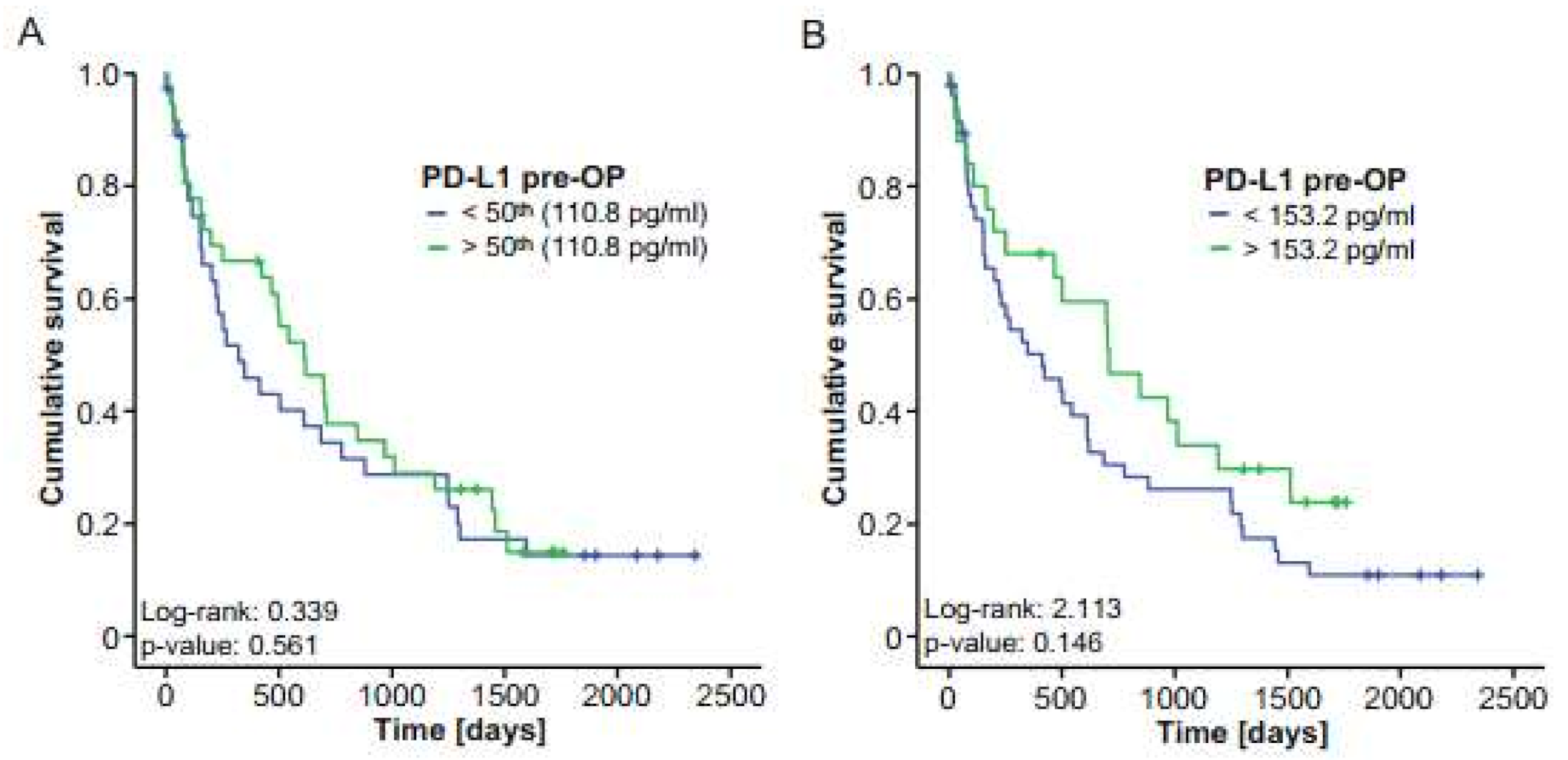
2.2. Association of Preoperative PD-L1 Serum Concentrations and Patients’ Survival
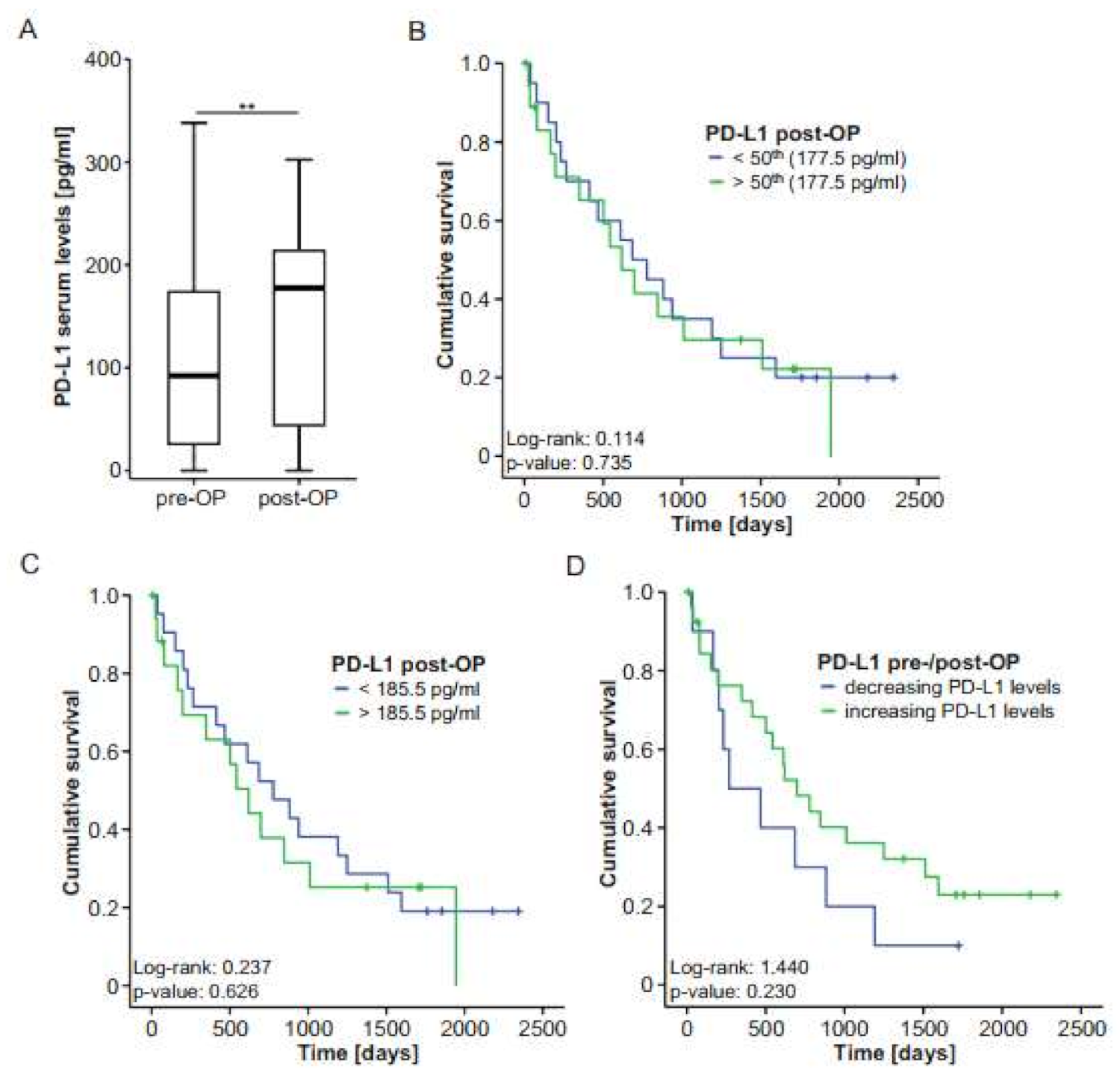
2.3. Postoperative PD-L1 Concentrations Are Elevated in Patients with BTC After Tumor Resection
3. Discussion
4. Patients and Methods
4.1. Study Design, Data Collection and Patient Characteristics
4.2. Determination of PD-L1 Serum Concentrations by ELISA
4.3. Measurements of Cytokine Serum Levels
4.4. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Razumilava, N.; Gores, G.J. Cholangiocarcinoma. Lancet 2014, 383, 2168–2179. [Google Scholar] [CrossRef]
- Adeva, J.; Sangro, B.; Salati, M.; Edeline, J.; La Casta, A.; Bittoni, A.; Berardi, R.; Bruix, J.; Valle, J.W. Medical treatment for cholangiocarcinoma. Liver Int. 2019, 39, 123–142. [Google Scholar] [CrossRef] [PubMed]
- Sano, T.; Shimada, K.; Sakamoto, Y.; Ojima, H.; Esaki, M.; Kosuge, T. Prognosis of perihilar cholangiocarcinoma: Hilar bile duct cancer versus intrahepatic cholangiocarcinoma involving the hepatic hilus. Ann. Surg. Oncol. 2007, 15, 590–599. [Google Scholar] [CrossRef] [PubMed]
- Loosen, S.H.; Vucur, M.; Trautwein, C.; Roderburg, C.; Luedde, T. Circulating biomarkers for cholangiocarcinoma. Dig. Dis. 2018, 36, 281–288. [Google Scholar] [CrossRef] [PubMed]
- Berretta, M.; Cavaliere, C.; Alessandrini, L.; Stanzione, B.; Facchini, G.; Balestreri, L.; Perin, T.; Canzonieri, V. Serum and tissue markers in hepatocellular carcinoma and cholangiocarcinoma: Clinical and prognostic implications. Oncotarget 2016, 8, 14192–14220. [Google Scholar] [CrossRef]
- Macias, R.I.R.; Kornek, M.; Rodrigues, P.M.; Paiva, N.A.; Castro, R.E.; Urban, S.; Pereira, S.P.; Cadamuro, M.; Rupp, C.; Loosen, S.H.; et al. Diagnostic and prognostic biomarkers in cholangiocarcinoma. Liver Int. 2019, 39, 108–122. [Google Scholar] [CrossRef]
- Najafian, N.; Khoury, S.J. T cell costimulatory pathways: Blockade for autoimmunity. Expert Opin. Biol. Ther. 2003, 3, 227–236. [Google Scholar] [CrossRef]
- Blank, C.; Gajewski, T.F.; Mackensen, A. Interaction of PD-L1 on tumor cells with PD-1 on tumor-specific T cells as a mechanism of immune evasion: Implications for tumor immunotherapy. Cancer Immunol. Immunother. 2005, 54, 307–314. [Google Scholar] [CrossRef]
- Patel, S.P.; Kurzrock, R. PD-L1 expression as a predictive biomarker in cancer immunotherapy. Mol. Cancer Ther. 2015, 14, 847–856. [Google Scholar] [CrossRef]
- Zhu, X.; Lang, J. Soluble PD-1 and PD-L1: Predictive and prognostic significance in cancer. Oncotarget 2017, 8, 97671–97682. [Google Scholar] [CrossRef]
- Wang, L.; Wang, H.; Chen, H.; Wang, W.-D.; Chen, X.-Q.; Geng, Q.-R.; Xia, Z.-J.; Lu, Y. Serum levels of soluble programmed death ligand 1 predict treatment response and progression free survival in multiple myeloma. Oncotarget 2015, 6, 41228–41236. [Google Scholar] [CrossRef]
- Qu, Y.; Wang, H.; Liu, H.; Sun, X.; Li, J.; Yu, H. Molecular mechanism of expression changes of immunological indexes of PD-1/sPD-L1 after radiotherapy in nonsmall cell lung cancer. BioMed Res. Int. 2021, 2021, 1–8. [Google Scholar] [CrossRef]
- Krafft, U.; Olah, C.; Reis, H.; Kesch, C.; Darr, C.; Grünwald, V.; Tschirdewahn, S.; Hadaschik, B.; Horvath, O.; Kenessey, I.; et al. High serum PD-L1 levels are associated with poor survival in urothelial cancer patients treated with chemotherapy and immune checkpoint inhibitor therapy. Cancers 2021, 13, 2548. [Google Scholar] [CrossRef]
- Mair, M.J.; Ilhan-Mutlu, A.; Pajenda, S.; Kiesel, B.; Wöhrer, A.; Widhalm, G.; Dieckmann, K.; Marosi, C.; Wagner, L.; Preusser, M.; et al. Circulating PD-L1 levels change during bevacizumab-based treatment in recurrent glioma. Cancer Immunol. Immunother. 2021, 1–8. [Google Scholar] [CrossRef]
- Fanale, D.; Incorvaia, L.; Badalamenti, G.; De Luca, I.; Algeri, L.; Bonasera, A.; Corsini, L.; Brando, C.; Russo, A.; Iovanna, J.; et al. Prognostic role of plasma PD-1, PD-L1, pan-BTN3As and BTN3A1 in patients affected by metastatic gastrointestinal stromal tumors: Can immune checkpoints act as a sentinel for short-term survival? Cancers 2021, 13, 2118. [Google Scholar] [CrossRef]
- Kushlinskii, N.E.; Gershtein, E.S.; Chang, V.L.; Korotkova, E.A.; Alferov, A.A.; Kontorshchikov, M.M.; Sokolov, N.Y.; Karamysheva, E.I.; Ognerubov, N.A.; Stilidi, I.S. Prognostic significance of soluble forms of immune checkpoint PD-1/PDL1 receptor and ligand in blood plasma of gastric cancer patients. Klin. Lab. Diagn. 2021, 66, 139–146. [Google Scholar] [CrossRef]
- Park, H.; Bang, J.-H.; Nam, A.-R.; Park, J.E.; Jin, M.H.; Bang, Y.-J.; Oh, D.-Y. Prognostic implications of soluble programmed death-ligand 1 and its dynamics during chemotherapy in unresectable pancreatic cancer. Sci. Rep. 2019, 9, 1–8. [Google Scholar] [CrossRef]
- Kruger, S.; Legenstein, M.-L.; Rösgen, V.; Haas, M.; Modest, D.P.; Westphalen, C.B.; Ormanns, S.; Kirchner, T.; Heinemann, V.; Holdenrieder, S.; et al. Serum levels of soluble programmed death protein 1 (sPD-1) and soluble programmed death ligand 1 (sPD-L1) in advanced pancreatic cancer. OncoImmunology 2017, 6, e1310358. [Google Scholar] [CrossRef]
- Shigemori, T.; Toiyama, Y.; Okugawa, Y.; Yamamoto, A.; Yin, C.; Narumi, A.; Ichikawa, T.; Ide, S.; Shimura, T.; Fujikawa, H.; et al. Soluble PD-L1 expression in circulation as a predictive marker for recurrence and prognosis in gastric cancer: Direct comparison of the clinical burden between tissue and serum PD-L1 expression. Ann. Surg. Oncol. 2018, 26, 876–883. [Google Scholar] [CrossRef]
- Kong, P.; Wang, J.; Song, Z.; Liu, S.; He, W.; Jiang, C.; Xie, Q.; Yang, L.; Xia, X.; Xia, L. Circulating lymphocytes, PD-L1 expression on tumor-infiltrating lymphocytes, and survival of colorectal cancer patients with different mismatch repair gene status. J. Cancer 2019, 10, 1745–1754. [Google Scholar] [CrossRef]
- Das, S.; Sarrou, E.; Podgrabinska, S.; Cassella, M.; Mungamuisk, S.K.; Feirt, N.; Gordon, R.; Nagi, C.S.; Wang, Y.; Entenberg, D.; et al. Tumor cell entry into the lymph node is controlled by CCL1 chemokine expressed by lymph node lymphatic sinuses. J. Exp. Med. 2013, 210, 1509–1528. [Google Scholar] [CrossRef] [PubMed]
- Hong, C.H.; Lin, S.H.; Lee, C.H. CCL21 induces mTOR-dependent MALAT1 expression, leading to cell migration in cutaneous T-cell lymphoma. In Vivo 2019, 33, 793–800. [Google Scholar] [CrossRef]
- Lin, Z.-Y.; Chuang, Y.-H.; Chuang, W.-L. Cancer-associated fibroblasts up-regulate CCL2, CCL26, IL6 and LOXL2 genes related to promotion of cancer progression in hepatocellular carcinoma cells. Biomed. Pharmacother. 2012, 66, 525–529. [Google Scholar] [CrossRef] [PubMed]
- Deng, X.; Tu, Z.; Xiong, M.; Tembo, K.; Zhou, L.; Liu, P.; Pan, S.; Xiong, J.; Yang, X.; Leng, J.; et al. Wnt5a and CCL25 promote adult T-cell acute lymphoblastic leukemia cell migration, invasion and metastasis. Oncotarget 2017, 8, 39033–39047. [Google Scholar] [CrossRef]
- Valle, J.W.; Kelley, R.K.; Nervi, B.; Oh, D.-Y.; Zhu, A.X. Biliary tract cancer. Lancet 2021, 397, 428–444. [Google Scholar] [CrossRef]
- Lapitz, A.; Arbelaiz, A.; O’Rourke, C.J.; Lavin, J.L.; La Casta, A.; Ibarra, C.; Jimeno, J.P.; Santos-Laso, A.; Izquierdo-Sanchez, L.; Krawczyk, M.; et al. Patients with cholangiocarcinoma present specific RNA profiles in serum and urine extracellular vesicles mirroring the tumor expression: Novel liquid biopsy biomarkers for disease diagnosis. Cells 2020, 9, 721. [Google Scholar] [CrossRef] [PubMed]
- Haanen, J.B.; Robert, C. Immune checkpoint inhibitors. Immuno-Oncology 2015, 42, 55–66. [Google Scholar] [CrossRef]
- Bashir, B.; Wilson, M.A. Novel immunotherapy combinations. Curr. Oncol. Rep. 2019, 21, 96. [Google Scholar] [CrossRef]
- Alley, E.W.; Lopez, J.; Santoro, A.; Morosky, A.; Saraf, S.; Piperdi, B.; van Brummelen, E. Clinical safety and activity of pembrolizumab in patients with malignant pleural mesothelioma (KEYNOTE-028): Preliminary results from a non-randomised, open-label, phase 1b trial. Lancet Oncol. 2017, 18, 623–630. [Google Scholar] [CrossRef]
- Tessier-Cloutier, B.; Kalloger, S.E.; Al-Kandari, M.; Milne, K.; Gao, D.; Nelson, B.H.; Renouf, D.J.; Sheffield, B.S.; Schaeffer, D.F. Programmed cell death ligand 1 cut-point is associated with reduced disease specific survival in resected pancreatic ductal adenocarcinoma. BMC Cancer 2017, 17, 1–10. [Google Scholar] [CrossRef]
- Corrigan, L.; Lowery, M. Ivosidenib for the treatment of isocitrate dehydrogenase-1 mutant cholangiocarcinoma. Expert Rev. Gastroenterol. Hepatol. 2021, 15, 475–481. [Google Scholar] [CrossRef]
- Abou-Alfa, G.K.; Macarulla, T.; Javle, M.M.; Kelley, R.K.; Lubner, S.J.; Adeva, J.; Cleary, J.M.; Catenacci, D.V.; Borad, M.J.; Bridgewater, J.; et al. Ivosidenib in IDH1-mutant, chemotherapy-refractory cholangiocarcinoma (ClarIDHy): A multicentre, randomised, double-blind, placebo-controlled, phase 3 study. Lancet Oncol. 2020, 21, 796–807. [Google Scholar] [CrossRef]
- Abou-Alfa, G.K.; Sahai, V.; Hollebecque, A.; Vaccaro, G.; Melisi, D.; Al-Rajabi, R.; Paulson, A.S.; Borad, M.J.; Gallinson, D.; Murphy, A.G.; et al. Pemigatinib for previously treated, locally advanced or metastatic cholangiocarcinoma: A multicentre, open-label, phase 2 study. Lancet Oncol. 2020, 21, 671–684. [Google Scholar] [CrossRef]
- Saborowski, A.; Lehmann, U.; Vogel, A. FGFR inhibitors in cholangiocarcinoma: What’s now and what’s next? Ther. Adv. Med. Oncol. 2020, 12. [Google Scholar] [CrossRef]
- Bekaii-Saab, T.S.; Valle, J.W.; Van Cutsem, E.; Rimassa, L.; Furuse, J.; Ioka, T.; Melisi, D.; Macarulla, T.; Bridgewater, J.; Wasan, H.; et al. FIGHT-302: First-line pemigatinib vs gemcitabine plus cisplatin for advanced cholangiocarcinoma with FGFR2 rearrangements. Futur. Oncol. 2020, 16, 2385–2399. [Google Scholar] [CrossRef]
- Marin, J.J.G.; Prete, M.G.; Lamarca, A.; Tavolari, S.; Landa-Magdalena, A.; Brandi, G.; Segatto, O.; Vogel, A.; Macias, R.I.R.; Rodrigues, P.M.; et al. Current and novel therapeutic opportunities for systemic therapy in biliary cancer. Br. J. Cancer 2020, 123, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Loosen, S.H.; Tacke, F.; Püthe, N.; Binneboesel, M.; Wiltberger, G.; Alizai, P.H.; Kather, J.N.; Paffenholz, P.; Ritz, T.; Koch, A.; et al. High baseline soluble urokinase plasminogen activator receptor (suPAR) serum levels indicate adverse outcome after resection of pancreatic adenocarcinoma. Carcinogenesis 2019, 40, 947–955. [Google Scholar] [CrossRef] [PubMed]
- Loosen, S.H.; Roderburg, C.; Kauertz, K.L.; Pombeiro, I.; Leyh, C.; Benz, F.; Vucur, M.; Longerich, T.; Koch, A.; Braunschweig, T.; et al. Elevated levels of circulating osteopontin are associated with a poor survival after resection of cholangiocarcinoma. J. Hepatol. 2017, 67, 749–757. [Google Scholar] [CrossRef]
- Koch, A.; Voigt, S.; Kruschinski, C.; Sanson, E.; Dückers, H.; Horn, A.; Yagmur, E.; Zimmermann, H.; Trautwein, C.; Tacke, F. Circulating soluble urokinase plasminogen activator receptor is stably elevated during the first week of treatment in the intensive care unit and predicts mortality in critically ill patients. Crit. Care 2011, 15, R63. [Google Scholar] [CrossRef] [PubMed]
| Parameter | PD-L1 Pre-OP | |
|---|---|---|
| rS | p-Value | |
| Sodium | −0.162 | 0.173 |
| Potassium | 0.146 | 0.222 |
| Leukocytes | −0.073 | 0.543 |
| Thrombocytes | −0.055 | 0.649 |
| AST | 0.068 | 0.568 |
| ALT | 0.086 | 0.550 |
| Bilirubin | 0.085 | 0.480 |
| ALP | 0.030 | 0.805 |
| GGT | 0.040 | 0.740 |
| CRP | 0.126 | 0.300 |
| Creatinine | −0.092 | 0.440 |
| Characteristic | |
|---|---|
| Healthy controls | 42 |
| BTC patients | 73 |
| Sex (%): | |
| male–female | 52.8–47.2 |
| Age (years, median and range) | 68.0 (37–84) |
| BMI (kg/m2, median and range) | 25.99 (19.15–46.36) |
| BTC characteristics (%): | |
| T1-T2-T3-T4 | 4.5–40.9–33.3–21.2 |
| N0-N1 | 41.9–58.1 |
| M0-M1 | 81.2–18.8 |
| G2-G3 | 58.5–41.5 |
| R0-R1 | 61.0–39.0 |
| Tumor localization (%): | |
| intrahepatic CCA | 34.2 |
| perihilar CCA | 37.0 |
| distal | 17.8 |
| gallbladder carcinoma | 11.0 |
| Clinical performance status (%): | |
| ECOG 0-1-2 | 52.1-38.0-9.0 |
| Laboratory parameters of BTC patients (median and range): | |
| WBC (cells/nL) | 7.9 (2.9–21.6) |
| CRP (mg/L) | 18.2 (0.0–230.0) |
| AST (U/L) | 47.0 (18.0–1587.0) |
| ALT (U/L) | 46.0 (10.0–1097.0) |
| GGT (U/L) | 348 (36.0–2015.0) |
| ALP (U/L) | 229.0 (53.0–1055.0) |
| Bilirubin (mg/dL) | 1.0 (0.24–21.49) |
| Creatinine (mg/dL) | 0.82 (0.43–1.9) |
| Haemoglobin (g/L) | 12.5 (7.8–16.2) |
| Platelets (cells/nL) | 275.0 (75.0–931.0) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Roderburg, C.; Loosen, S.H.; Bednarsch, J.; Alizai, P.H.; Roeth, A.A.; Schmitz, S.M.; Vucur, M.; Luedde, M.; Paffenholz, P.; Tacke, F.; et al. Levels of Circulating PD-L1 Are Decreased in Patients with Resectable Cholangiocarcinoma. Int. J. Mol. Sci. 2021, 22, 6569. https://doi.org/10.3390/ijms22126569
Roderburg C, Loosen SH, Bednarsch J, Alizai PH, Roeth AA, Schmitz SM, Vucur M, Luedde M, Paffenholz P, Tacke F, et al. Levels of Circulating PD-L1 Are Decreased in Patients with Resectable Cholangiocarcinoma. International Journal of Molecular Sciences. 2021; 22(12):6569. https://doi.org/10.3390/ijms22126569
Chicago/Turabian StyleRoderburg, Christoph, Sven H. Loosen, Jan Bednarsch, Patrick H. Alizai, Anjali A. Roeth, Sophia M. Schmitz, Mihael Vucur, Mark Luedde, Pia Paffenholz, Frank Tacke, and et al. 2021. "Levels of Circulating PD-L1 Are Decreased in Patients with Resectable Cholangiocarcinoma" International Journal of Molecular Sciences 22, no. 12: 6569. https://doi.org/10.3390/ijms22126569
APA StyleRoderburg, C., Loosen, S. H., Bednarsch, J., Alizai, P. H., Roeth, A. A., Schmitz, S. M., Vucur, M., Luedde, M., Paffenholz, P., Tacke, F., Trautwein, C., Ulmer, T. F., Neumann, U. P., & Luedde, T. (2021). Levels of Circulating PD-L1 Are Decreased in Patients with Resectable Cholangiocarcinoma. International Journal of Molecular Sciences, 22(12), 6569. https://doi.org/10.3390/ijms22126569









