MUC1: Structure, Function, and Clinic Application in Epithelial Cancers
Abstract
1. Introduction
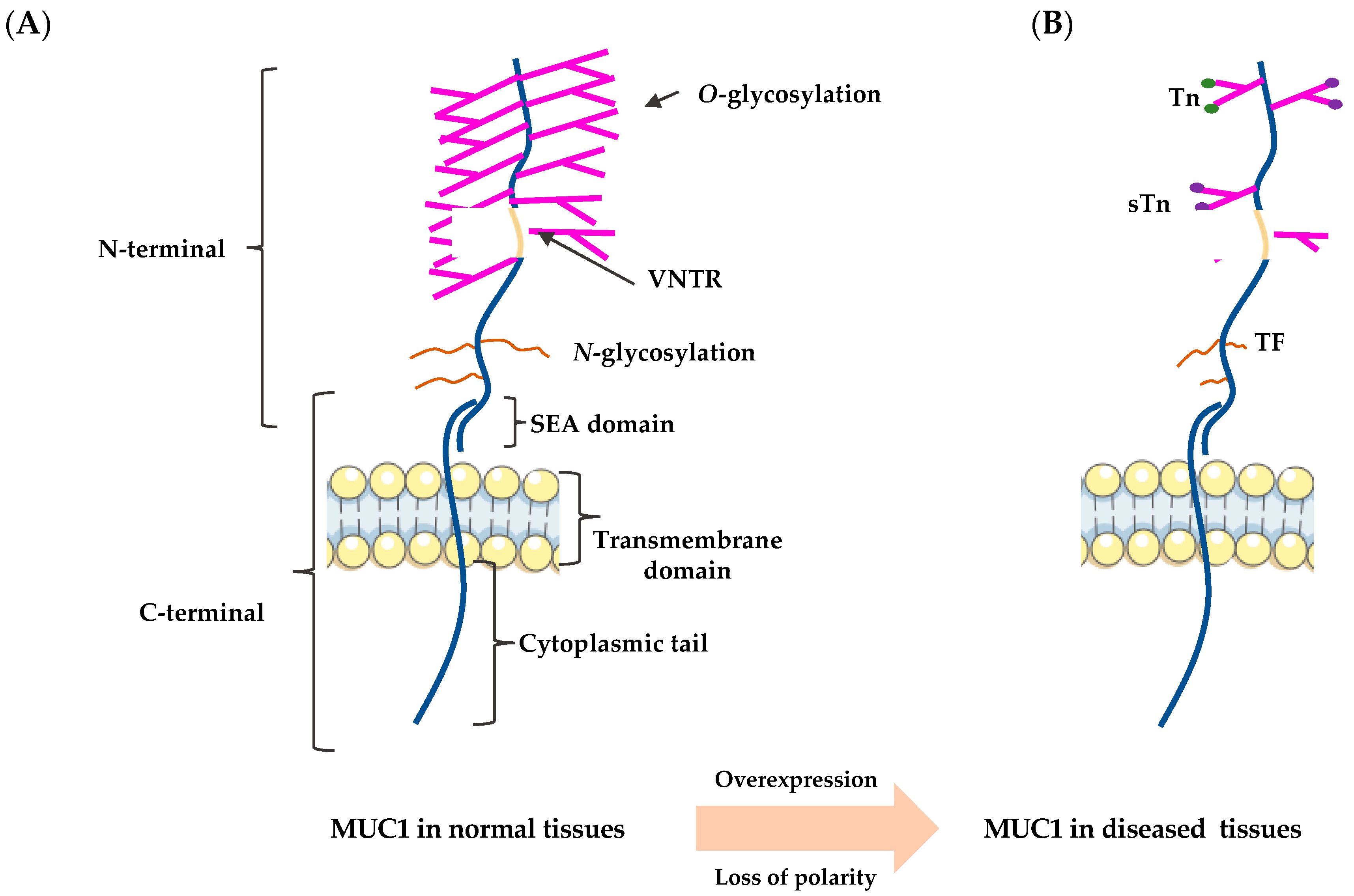
2. The Structure of MUC1
2.1. The N-Terminus
2.2. The C-Terminus
3. Function of MUC1 in Normal Tissues
4. Function of MUC1 in Cancer Tissues
4.1. Pro- or Anti-Inflammatory Role
4.2. Pharmacodynamic Inhibitors
4.3. MUC1 Promotes Migration and Invasion of Cancers
4.4. MUC1 Inhibits Cancer Cell Growth and Apoptosis
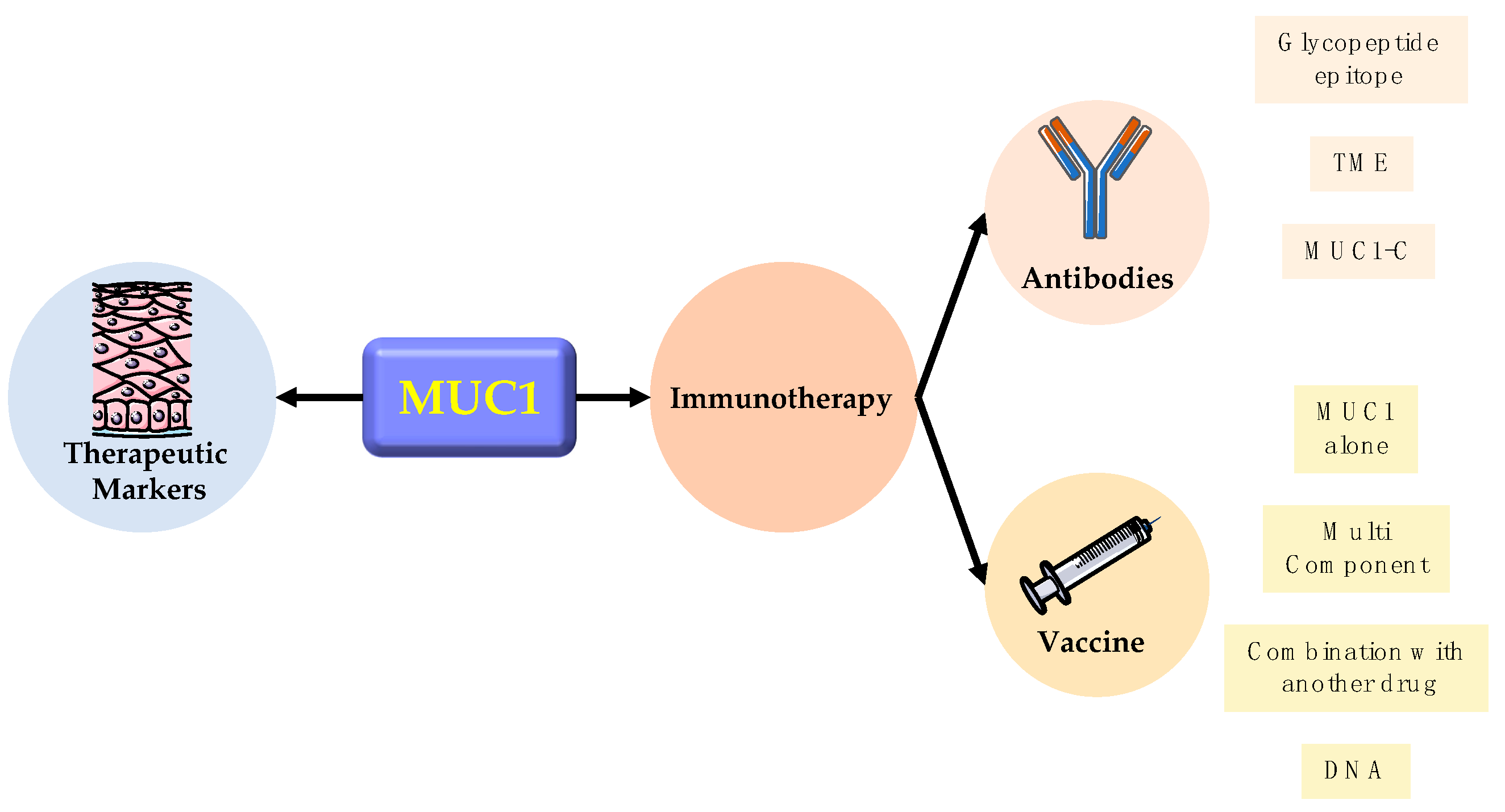
5. Clinic Application
5.1. Therapeutic Marker
5.2. Immunotherapy
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
Abbreviations
| 5-FU | fluorouracil |
| ABC | ATP-binding cassette |
| AP-1 | activator protein 1 |
| CLP | cercal ligation and puncture |
| CRB3 | Crumbs homolog-3 |
| CTL | cytotoxic T lymphocyte |
| DA | dopamine |
| DC | dendritic cell |
| EGFR | epidermal growth factor receptor |
| EMT | epithelial-mesenchymal transition |
| FLT3 | fms-like tyrosine kinase 3 |
| GSK3β | glycogen synthase kinase 3β |
| HCC | hepatocellular carcinoma |
| HIF | hypoxia-inducible factor |
| IFN-g | interferon-g |
| JAK | Janus kinase |
| JNK | c-Jun N-terminal kinase |
| LLC | Lewis lung cancer |
| LPS | lipopolysaccharide |
| MAPK | mitogen-activated protein kinase |
| MS | multiple sclerosis |
| MSI | microsatellite instability |
| mTOR | mammalian target of rapamycin |
| MUC1 | Mucin1 |
| MUC1-C | C-terminal subunit |
| MUC1-N | N-terminal subunit |
| NF | nuclear factor |
| NPs | nanoparticles |
| NRP1 | neuropilin-1 |
| PD-L1 | Programmed death-ligand 1 |
| PDGF | platelet-derived growth factor |
| PI3K | phosphoinositide 3-kinase |
| PKCδ | protein kinase C delta |
| PMIP | inhibitory peptide |
| SEA | sperm protein-enterokinase-agarin |
| STAT | signal transducer and activator of transcription |
| TACAs | tumor-associated carbohydrate antigens |
| TF or T | Thomsen-Friedenreich |
| TGF-β | transforming growth factor β |
| TLR | Toll-like receptor |
| TMB | as tumor mutational burden |
| TNBC | triple-negative breast cancer |
| TRs | tandem repeats |
| TβRI | TGF-β type I receptor |
| VEGF | vascular endothelial growth factor |
| VNTRs | variable number tandem repeat |
| YAP | yes-associated protein |
| ZEB1 | zinc-finger E-box-binding homeobox 1 |
References
- Nath, S.; Mukherjee, P. MUC1: A multifaceted oncoprotein with a key role in cancer progression. Trends Mol. Med. 2014, 20, 332–342. [Google Scholar] [CrossRef] [PubMed]
- Bose, M.; Mukherjee, P. Microbe-MUC1 Crosstalk in Cancer-Associated Infections. Trends Mol. Med. 2020, 26, 324–336. [Google Scholar] [CrossRef] [PubMed]
- Gao, T.; Cen, Q.; Lei, H. A review on development of MUC1-based cancer vaccine. Biomed. Pharmacother. 2020, 132, 110888. [Google Scholar] [CrossRef] [PubMed]
- Carson, D.D. The cytoplasmic tail of MUC1: A very busy place. Sci. Signal. 2008, 1, pe35. [Google Scholar] [CrossRef]
- Kufe, D.W. Mucins in cancer: Function, prognosis and therapy. Nat. Rev. Cancer 2009, 9, 874–885. [Google Scholar] [CrossRef] [PubMed]
- Joshi, S.; Kumar, S.; Bafna, S.; Rachagani, S.; Wagner, K.U.; Jain, M.; Batra, S.K. Genetically engineered mucin mouse models for inflammation and cancer. Cancer Metastasis Rev. 2015, 34, 593–609. [Google Scholar] [CrossRef] [PubMed]
- Van Putten, J.P.M.; Strijbis, K. Transmembrane Mucins: Signaling Receptors at the Intersection of Inflammation and Cancer. J. Innate Immun. 2017, 9, 281–299. [Google Scholar] [CrossRef]
- Ballester, B.; Milara, J.; Cortijo, J. The role of mucin 1 in respiratory diseases. Eur. Respir. Rev. 2021, 30. [Google Scholar] [CrossRef]
- Dhar, P.; McAuley, J. The Role of the Cell Surface Mucin MUC1 as a Barrier to Infection and Regulator of Inflammation. Front. Cell. Infect. Microbiol. 2019, 9, 117. [Google Scholar] [CrossRef]
- Schroeder, J.A.; Thompson, M.C.; Gardner, M.M.; Gendler, S.J. Transgenic MUC1 interacts with epidermal growth factor receptor and correlates with mitogen-activated protein kinase activation in the mouse mammary gland. J. Biol. Chem. 2001, 276, 13057–13064. [Google Scholar] [CrossRef]
- Duraisamy, S.; Ramasamy, S.; Kharbanda, S.; Kufe, D. Distinct evolution of the human carcinoma-associated transmembrane mucins, MUC1, MUC4 AND MUC16. Gene 2006, 373, 28–34. [Google Scholar] [CrossRef]
- Ahmad, R.; Raina, D.; Trivedi, V.; Ren, J.; Rajabi, H.; Kharbanda, S.; Kufe, D. MUC1 oncoprotein activates the IkappaB kinase beta complex and constitutive NF-kappaB signalling. Nat. Cell Biol. 2007, 9, 1419–1427. [Google Scholar] [CrossRef]
- Inagaki, Y.; Gao, J.; Song, P.; Kokudo, N.; Nakata, M.; Tang, W. Clinicopathological utility of sialoglycoconjugates in diagnosing and treating colorectal cancer. World J. Gastroenterol. 2014, 20, 6123–6132. [Google Scholar] [CrossRef]
- Gebregiworgis, T.; Purohit, V.; Shukla, S.K.; Tadros, S.; Chaika, N.V.; Abrego, J.; Mulder, S.E.; Gunda, V.; Singh, P.K.; Powers, R. Glucose Limitation Alters Glutamine Metabolism in MUC1-Overexpressing Pancreatic Cancer Cells. J. Proteome Res. 2017, 16, 3536–3546. [Google Scholar] [CrossRef]
- Alam, M.; Bouillez, A.; Tagde, A.; Ahmad, R.; Rajabi, H.; Maeda, T.; Hiraki, M.; Suzuki, Y.; Kufe, D. MUC1-C Represses the Crumbs Complex Polarity Factor CRB3 and Downregulates the Hippo Pathway. Mol. Cancer Res. 2016, 14, 1266–1276. [Google Scholar] [CrossRef]
- Gibier, J.B.; Hemon, B.; Fanchon, M.; Gaudelot, K.; Pottier, N.; Ringot, B.; Van Seuningen, I.; Glowacki, F.; Cauffiez, C.; Blum, D.; et al. Dual role of MUC1 mucin in kidney ischemia-reperfusion injury: Nephroprotector in early phase, but pro-fibrotic in late phase. Biochim. Biophys. Acta Mol. Basis Dis. 2017, 1863, 1336–1349. [Google Scholar] [CrossRef]
- Marczynski, M.; Winkeljann, B.; Lieleg, O. Advances in Mucin Biopolymer Research: Purification, Characterization, and Applications. In Biopolymers for Biomedical and Biotechnological Applications; Wiley: Hoboken, NJ, USA, 2021; pp. 181–208. [Google Scholar] [CrossRef]
- Kasprzak, A.; Adamek, A. Mucins: The Old, the New and the Promising Factors in Hepatobiliary Carcinogenesis. Int. J. Mol. Sci. 2019, 20, 1288. [Google Scholar] [CrossRef]
- Carroll-Portillo, A.; Lin, H.C. Exploring Mucin as Adjunct to Phage Therapy. Microorganisms 2021, 9, 509. [Google Scholar] [CrossRef]
- Hattrup, C.L.; Gendler, S.J. Structure and function of the cell surface (tethered) mucins. Annu. Rev. Physiol. 2008, 70, 431–457. [Google Scholar] [CrossRef]
- Syrkina, M.S.; Maslakova, A.A.; Potashnikova, D.M.; Veiko, V.P.; Vassetzky, Y.S.; Rubtsov, M.A. Dual Role of the Extracellular Domain of Human Mucin MUC1 in Metastasis. J. Cell. Biochem. 2017, 118, 4002–4011. [Google Scholar] [CrossRef] [PubMed]
- Gendler, S.; Taylor-Papadimitriou, J.; Duhig, T.; Rothbard, J.; Burchell, J. A highly immunogenic region of a human polymorphic epithelial mucin expressed by carcinomas is made up of tandem repeats. J. Biol. Chem. 1988, 263, 12820–12823. [Google Scholar] [CrossRef]
- Patton, S.; Gendler, S.J.; Spicer, A.P. The epithelial mucin, MUC1, of milk, mammary gland and other tissues. Biochim. Biophys. Acta 1995, 1241, 407–423. [Google Scholar] [CrossRef]
- Khodabakhsh, F.; Merikhian, P.; Eisavand, M.R.; Farahmand, L. Crosstalk between MUC1 and VEGF in angiogenesis and metastasis: A review highlighting roles of the MUC1 with an emphasis on metastatic and angiogenic signaling. Cancer Cell Int. 2021, 21, 200. [Google Scholar] [CrossRef] [PubMed]
- Levitin, F.; Stern, O.; Weiss, M.; Gil-Henn, C.; Ziv, R.; Prokocimer, Z.; Smorodinsky, N.I.; Rubinstein, D.B.; Wreschner, D.H. The MUC1 SEA Module Is a Self-cleaving Domain. J. Biol. Chem. 2005, 280, 33374–33386. [Google Scholar] [CrossRef]
- Ahmad, R.; Raina, D.; Joshi, M.D.; Kawano, T.; Ren, J.; Kharbanda, S.; Kufe, D. MUC1-C oncoprotein functions as a direct activator of the nuclear factor-kappaB p65 transcription factor. Cancer Res. 2009, 69, 7013–7021. [Google Scholar] [CrossRef]
- Kufe, D.W. MUC1-C oncoprotein as a target in breast cancer: Activation of signaling pathways and therapeutic approaches. Oncogene 2013, 32, 1073–1081. [Google Scholar] [CrossRef]
- Radziejewska, I.; Borzym-Kluczyk, M.; Leszczynska, K. Luteolin alters MUC1 extracellular domain, sT antigen, ADAM-17, IL-8, IL-10 and NF-kappaB expression in Helicobacter pylori-infected gastric cancer CRL-1739 cells: A preliminary study. Biomed. Rep. 2021, 14, 19. [Google Scholar] [CrossRef]
- Beckwith, D.M.; Cudic, M. Tumor-associated O-glycans of MUC1: Carriers of the glyco-code and targets for cancer vaccine design. Semin. Immunol. 2020, 47, 101389. [Google Scholar] [CrossRef]
- Singh, R.; Bandyopadhyay, D. MUC1: A target molecule for cancer therapy. Cancer Biol. Ther. 2007, 6, 481–486. [Google Scholar] [CrossRef]
- Sousa, A.M.; Rei, M.; Freitas, R.; Ricardo, S.; Caffrey, T.; David, L.; Almeida, R.; Hollingsworth, M.A.; Santos-Silva, F. Effect of MUC1/beta-catenin interaction on the tumorigenic capacity of pancreatic CD133(+) cells. Oncol. Lett. 2016, 12, 1811–1817. [Google Scholar] [CrossRef]
- Martinez-Saez, N.; Peregrina, J.M.; Corzana, F. Principles of mucin structure: Implications for the rational design of cancer vaccines derived from MUC1-glycopeptides. Chem. Soc. Rev. 2017, 46, 7154–7175. [Google Scholar] [CrossRef]
- Wang, S.; You, L.; Dai, M.; Zhao, Y. Mucins in pancreatic cancer: A well-established but promising family for diagnosis, prognosis and therapy. J. Cell. Mol. Med. 2020, 24, 10279–10289. [Google Scholar] [CrossRef]
- Ligtenberg, M.J.; Buijs, F.; Vos, H.L.; Hilkens, J. Suppression of cellular aggregation by high levels of episialin. Cancer Res. 1992, 52, 2318–2324. [Google Scholar]
- Wei, X.; Xu, H.; Kufe, D. Human mucin 1 oncoprotein represses transcription of the p53 tumor suppressor gene. Cancer Res. 2007, 67, 1853–1858. [Google Scholar] [CrossRef]
- Ilkovitch, D.; Carrio, R.; Lopez, D.M. Mechanisms of antitumor and immune-enhancing activities of MUC1/sec, a secreted form of mucin-1. Immunol. Res. 2013, 57, 70–80. [Google Scholar] [CrossRef]
- Kashyap, B.; Kullaa, A.M. Regulation of mucin 1 expression and its relationship with oral diseases. Arch. Oral Biol. 2020, 117, 104791. [Google Scholar] [CrossRef]
- Marczynski, M.; Jiang, K.; Blakeley, M.; Srivastava, V.; Vilaplana, F.; Crouzier, T.; Lieleg, O. Structural Alterations of Mucins Are Associated with Losses in Functionality. Biomacromolecules 2021, 22, 1600–1613. [Google Scholar] [CrossRef]
- Martinez-Carrasco, R.; Argüeso, P.; Fini, M.E. Membrane-associated mucins of the human ocular surface in health and disease. Ocul. Surf. 2021, in press Epub ahead of print. [Google Scholar] [CrossRef]
- Kosmerl, E.; Rocha-Mendoza, D.; Ortega-Anaya, J.; Jiménez-Flores, R.; García-Cano, I. Improving Human Health with Milk Fat Globule Membrane, Lactic Acid Bacteria, and Bifidobacteria. Microorganisms 2021, 9, 341. [Google Scholar] [CrossRef]
- Brayman, M.; Thathiah, A.; Carson, D.D. MUC1: A multifunctional cell surface component of reproductive tissue epithelia. Reprod. Biol. Endocrinol. 2004, 2, 4. [Google Scholar] [CrossRef]
- Hikita, S.T.; Kosik, K.S.; Clegg, D.O.; Bamdad, C. MUC1* mediates the growth of human pluripotent stem cells. PLoS ONE 2008, 3, e3312. [Google Scholar] [CrossRef]
- Johansson, M.E.; Sjovall, H.; Hansson, G.C. The gastrointestinal mucus system in health and disease. Nat. Rev. Gastroenterol. Hepatol. 2013, 10, 352–361. [Google Scholar] [CrossRef]
- Yen, J.H.; Xu, S.; Park, Y.S.; Ganea, D.; Kim, K.C. Higher susceptibility to experimental autoimmune encephalomyelitis in Muc1-deficient mice is associated with increased Th1/Th17 responses. Brain Behav. Immun. 2013, 29, 70–81. [Google Scholar] [CrossRef]
- Wang, Y.M.; Ji, R.; Chen, W.W.; Huang, S.W.; Zheng, Y.J.; Yang, Z.T.; Qu, H.P.; Chen, H.; Mao, E.Q.; Chen, Y.; et al. Paclitaxel alleviated sepsis-induced acute lung injury by activating MUC1 and suppressing TLR-4/NF-kappaB pathway. Drug Des. Dev. Ther. 2019, 13, 3391–3404. [Google Scholar] [CrossRef]
- Wang, Y.M.; Qi, X.; Gong, F.C.; Chen, Y.; Yang, Z.T.; Mao, E.Q.; Chen, E.Z. Protective and predictive role of Mucin1 in sepsis-induced ALI/ARDS. Int. Immunopharmacol. 2020, 83, 106438. [Google Scholar] [CrossRef]
- Cascio, S.; Finn, O.J. Intra- and Extra-Cellular Events Related to Altered Glycosylation of MUC1 Promote Chronic Inflammation, Tumor Progression, Invasion, and Metastasis. Biomolecules 2016, 6, 39. [Google Scholar] [CrossRef]
- Haddon, L.; Hugh, J. MUC1-mediated motility in breast cancer: A review highlighting the role of the MUC1/ICAM-1/Src signaling triad. Clin. Exp. Metastasis 2015, 32, 393–403. [Google Scholar] [CrossRef]
- Rajabi, H.; Kufe, D. MUC1-C Oncoprotein Integrates a Program of EMT, Epigenetic Reprogramming and Immune Evasion in Human Carcinomas. Biochim. Biophys. Acta Rev. Cancer 2017, 1868, 117–122. [Google Scholar] [CrossRef]
- Kalra, A.V.; Campbell, R.B. Mucin overexpression limits the effectiveness of 5-FU by reducing intracellular drug uptake and antineoplastic drug effects in pancreatic tumours. Eur. J. Cancer 2009, 45, 164–173. [Google Scholar] [CrossRef]
- Jin, W.; Liao, X.; Lv, Y.; Pang, Z.; Wang, Y.; Li, Q.; Liao, Y.; Ye, Q.; Chen, G.; Zhao, K.; et al. MUC1 induces acquired chemoresistance by upregulating ABCB1 in EGFR-dependent manner. Cell Death Dis. 2017, 8, e2980. [Google Scholar] [CrossRef]
- Gunda, V.; Souchek, J.; Abrego, J.; Shukla, S.K.; Goode, G.D.; Vernucci, E.; Dasgupta, A.; Chaika, N.V.; King, R.J.; Li, S.; et al. MUC1-Mediated Metabolic Alterations Regulate Response to Radiotherapy in Pancreatic Cancer. Clin. Cancer Res. 2017, 23, 5881–5891. [Google Scholar] [CrossRef] [PubMed]
- Shukla, S.K.; Purohit, V.; Mehla, K.; Gunda, V.; Chaika, N.V.; Vernucci, E.; King, R.J.; Abrego, J.; Goode, G.D.; Dasgupta, A.; et al. MUC1 and HIF-1alpha Signaling Crosstalk Induces Anabolic Glucose Metabolism to Impart Gemcitabine Resistance to Pancreatic Cancer. Cancer Cell 2017, 32, 71–87. [Google Scholar] [CrossRef] [PubMed]
- Yi, F.T.; Lu, Q.P. Mucin 1 promotes radioresistance in hepatocellular carcinoma cells through activation of JAK2/STAT3 signaling. Oncol. Lett. 2017, 14, 7571–7576. [Google Scholar] [CrossRef] [PubMed]
- Caffrey, T.; Sagar, S.; Thomas, D.; Lewallen, M.E.; Hollingsworth, M.A.; Radhakrishnan, P. The glycoprotein mucin-1 negatively regulates GalNAc transferase 5 expression in pancreatic cancer. FEBS Lett. 2019, 593, 2751–2761. [Google Scholar] [CrossRef]
- Malinda, R.R.; Zeeberg, K.; Sharku, P.C.; Ludwig, M.Q.; Pedersen, L.B.; Christensen, S.T.; Pedersen, S.F. TGFbeta Signaling Increases Net Acid Extrusion, Proliferation and Invasion in Panc-1 Pancreatic Cancer Cells: SMAD4 Dependence and Link to Merlin/NF2 Signaling. Front. Oncol. 2020, 10, 687. [Google Scholar] [CrossRef]
- Maupin, K.A.; Sinha, A.; Eugster, E.; Miller, J.; Ross, J.; Paulino, V.; Keshamouni, V.G.; Tran, N.; Berens, M.; Webb, C.; et al. Glycogene expression alterations associated with pancreatic cancer epithelial-mesenchymal transition in complementary model systems. PLoS ONE 2010, 5, e13002. [Google Scholar] [CrossRef]
- Heldin, C.H.; Vanlandewijck, M.; Moustakas, A. Regulation of EMT by TGFbeta in cancer. FEBS Lett. 2012, 586, 1959–1970. [Google Scholar] [CrossRef]
- Grover, P.; Nath, S.; Nye, M.D.; Zhou, R.; Ahmad, M.; Mukherjee, P. SMAD4-independent activation of TGF-β signaling by MUC1 in a human pancreatic cancer cell line. Oncotarget 2018, 9, 6897–6910. [Google Scholar] [CrossRef]
- Wang, J.; Liu, G.; Li, Q.; Wang, F.; Xie, F.; Zhai, R.; Guo, Y.; Chen, T.; Zhang, N.; Ni, W.; et al. Mucin1 promotes the migration and invasion of hepatocellular carcinoma cells via JNK-mediated phosphorylation of Smad2 at the C-terminal and linker regions. Oncotarget 2015, 6, 19264–19278. [Google Scholar] [CrossRef]
- Zhao, P.; Meng, M.; Xu, B.; Dong, A.; Ni, G.; Lu, L. Decreased expression of MUC1 induces apoptosis and inhibits migration in pancreatic cancer PANC-1 cells via regulation of Slug pathway. Cancer Biomark. 2017, 20, 469–476. [Google Scholar] [CrossRef]
- Roy, L.D.; Sahraei, M.; Subramani, D.B.; Besmer, D.; Nath, S.; Tinder, T.L.; Bajaj, E.; Shanmugam, K.; Lee, Y.Y.; Hwang, S.I.; et al. MUC1 enhances invasiveness of pancreatic cancer cells by inducing epithelial to mesenchymal transition. Oncogene 2011, 30, 1449–1459. [Google Scholar] [CrossRef]
- Xu, H.L.; Zhao, X.; Zhang, K.M.; Tang, W.; Kokudo, N. Inhibition of KL-6/MUC1 glycosylation limits aggressive progression of pancreatic cancer. World J. Gastroenterol. 2014, 20, 12171–12181. [Google Scholar] [CrossRef]
- Sahraei, M.; Roy, L.D.; Curry, J.M.; Teresa, T.L.; Nath, S.; Besmer, D.; Kidiyoor, A.; Dalia, R.; Gendler, S.J.; Mukherjee, P. MUC1 regulates PDGFA expression during pancreatic cancer progression. Oncogene 2012, 31, 4935–4945. [Google Scholar] [CrossRef]
- Bozkaya, G.; Korhan, P.; Cokaklı, M.; Erdal, E.; Sağol, O.; Karademir, S.; Korch, C.; Atabey, N. Cooperative interaction of MUC1 with the HGF/c-Met pathway during hepatocarcinogenesis. Mol. Cancer 2012, 11, 64. [Google Scholar] [CrossRef]
- Woo, J.K.; Choi, Y.; Oh, S.H.; Jeong, J.H.; Choi, D.H.; Seo, H.S.; Kim, C.W. Mucin 1 enhances the tumor angiogenic response by activation of the AKT signaling pathway. Oncogene 2012, 31, 2187–2198. [Google Scholar] [CrossRef]
- Kitamoto, S.; Yokoyama, S.; Higashi, M.; Yamada, N.; Takao, S.; Yonezawa, S. MUC1 enhances hypoxia-driven angiogenesis through the regulation of multiple proangiogenic factors. Oncogene 2013, 32, 4614–4621. [Google Scholar] [CrossRef]
- Zhou, R.; Curry, J.M.; Roy, L.D.; Grover, P.; Haider, J.; Moore, L.J.; Wu, S.T.; Kamesh, A.; Yazdanifar, M.; Ahrens, W.A.; et al. A novel association of neuropilin-1 and MUC1 in pancreatic ductal adenocarcinoma: Role in induction of VEGF signaling and angiogenesis. Oncogene 2016, 35, 5608–5618. [Google Scholar] [CrossRef]
- Bouillez, A.; Rajabi, H.; Pitroda, S.; Jin, C.; Alam, M.; Kharbanda, A.; Tagde, A.; Wong, K.K.; Kufe, D. Inhibition of MUC1-C Suppresses MYC Expression and Attenuates Malignant Growth in KRAS Mutant Lung Adenocarcinomas. Cancer Res. 2016, 76, 1538–1548. [Google Scholar] [CrossRef]
- Besmer, D.M.; Curry, J.M.; Roy, L.D.; Tinder, T.L.; Sahraei, M.; Schettini, J.; Hwang, S.I.; Lee, Y.Y.; Gendler, S.J.; Mukherjee, P. Pancreatic ductal adenocarcinoma mice lacking mucin 1 have a profound defect in tumor growth and metastasis. Cancer Res. 2011, 71, 4432–4442. [Google Scholar] [CrossRef]
- Li, Q.; Wang, F.; Liu, G.; Yuan, H.; Chen, T.; Wang, J.; Xie, F.; Zhai, R.; Wang, F.; Guo, Y.; et al. Impact of Mucin1 knockdown on the phenotypic characteristics of the human hepatocellular carcinoma cell line SMMC-7721. Oncol. Rep. 2014, 31, 2811–2819. [Google Scholar] [CrossRef]
- Chen, Q.; Li, D.; Ren, J.; Li, C.; Xiao, Z.X. MUC1 activates JNK1 and inhibits apoptosis under genotoxic stress. Biochem. Biophys. Res. Commun. 2013, 440, 179–183. [Google Scholar] [CrossRef]
- Senapati, S.; Das, S.; Batra, S.K. Mucin-interacting proteins: From function to therapeutics. Trends Biochem. Sci. 2010, 35, 236–245. [Google Scholar] [CrossRef]
- Wang, J.; Ni, W.H.; Hu, K.B.; Zhai, X.Y.; Xie, F.; Jie, J.; Zhang, N.N.; Jiang, L.N.; Yuan, H.Y.; Tai, G.X. Targeting MUC1 and JNK by RNA interference and inhibitor inhibit the development of hepatocellular carcinoma. Cancer Sci. 2017, 108, 504–511. [Google Scholar] [CrossRef]
- Stroopinsky, D.; Kufe, D.; Avigan, D. MUC1 in hematological malignancies. Leuk. Lymphoma 2016, 57, 2489–2498. [Google Scholar] [CrossRef]
- Deng, J.; Wang, L.; Chen, H.; Li, L.; Ma, Y.; Ni, J.; Li, Y. The role of tumour-associated MUC1 in epithelial ovarian cancer metastasis and progression. Cancer Metastasis Rev. 2013, 32, 535–551. [Google Scholar] [CrossRef] [PubMed]
- Wen, R.; Gao, F.; Zhou, C.J.; Jia, Y.B. Polymorphisms in mucin genes in the development of gastric cancer. World J. Gastrointest. Oncol. 2015, 7, 328–337. [Google Scholar] [CrossRef] [PubMed]
- Kesari, M.V.; Gaopande, V.L.; Joshi, A.R.; Babanagare, S.V.; Gogate, B.P.; Khadilkar, A.V. Immunohistochemical study of MUC1, MUC2 and MUC5AC in colorectal carcinoma and review of literature. Indian J. Gastroenterol. 2015, 34, 63–67. [Google Scholar] [CrossRef] [PubMed]
- Hazgui, M.; Weslati, M.; Boughriba, R.; Ounissi, D.; Bacha, D.; Bouraoui, S. MUC1 and MUC5AC implication in Tunisian colorectal cancer patients. Turk. J. Med. Sci. 2021, 51, 309–318. [Google Scholar] [CrossRef]
- Wang, S.; You, L.; Dai, M.; Zhao, Y. Quantitative assessment of the diagnostic role of mucin family members in pancreatic cancer: A meta-analysis. Ann. Transl. Med. 2021, 9, 192. [Google Scholar] [CrossRef]
- Xu, F.; Liu, F.; Zhao, H.; An, G.; Feng, G. Prognostic Significance of Mucin Antigen MUC1 in Various Human Epithelial Cancers: A Meta-Analysis. Medicine (Baltimore) 2015, 94, e2286. [Google Scholar] [CrossRef]
- Wang, H.; Wu, T.; Li, M.; Tao, Y. Recent advances in nanomaterials for colorimetric cancer detection. J. Mater. Chem. B 2021, 9, 921–938. [Google Scholar] [CrossRef]
- Rashid, S.; Nawaz, M.H.; Rehman, I.u.; Hayat, A.; Marty, J.L. Dopamine/mucin-1 functionalized electro-active carbon nanotubes as a probe for direct competitive electrochemical immunosensing of breast cancer biomarker. Sens. Actuators B Chem. 2021, 330. [Google Scholar] [CrossRef]
- Maleki, F.; Rezazadeh, F.; Varmira, K. MUC1-Targeted Radiopharmaceuticals in Cancer Imaging and Therapy. Mol. Pharm. 2021. [Google Scholar] [CrossRef]
- Li, R.; An, Y.; Jin, T.; Zhang, F.; He, P. Detection of MUC1 protein on tumor cells and their derived exosomes for breast cancer surveillance with an electrochemiluminescence aptasensor. J. Electroanal. Chem. 2021, 882. [Google Scholar] [CrossRef]
- Yousefi, M.; Dehghani, S.; Nosrati, R.; Zare, H.; Evazalipour, M.; Mosafer, J.; Tehrani, B.S.; Pasdar, A.; Mokhtarzadeh, A.; Ramezani, M. Aptasensors as a new sensing technology developed for the detection of MUC1 mucin: A review. Biosens. Bioelectron. 2019, 130, 1–19. [Google Scholar] [CrossRef]
- Taylor-Papadimitriou, J.; Burchell, J.M.; Graham, R.; Beatson, R. Latest developments in MUC1 immunotherapy. Biochem. Soc. Trans. 2018, 46, 659–668. [Google Scholar] [CrossRef]
- Haurum, J.S.; Arsequell, G.; Lellouch, A.C.; Wong, S.Y.; Dwek, R.A.; McMichael, A.J.; Elliott, T. Recognition of carbohydrate by major histocompatibility complex class I-restricted, glycopeptide-specific cytotoxic T lymphocytes. J. Exp. Med. 1994, 180, 739–744. [Google Scholar] [CrossRef]
- Danielczyk, A.; Stahn, R.; Faulstich, D.; Loffler, A.; Marten, A.; Karsten, U.; Goletz, S. PankoMab: A potent new generation anti-tumour MUC1 antibody. Cancer Immunol. Immunother. 2006, 55, 1337–1347. [Google Scholar] [CrossRef]
- Bouillez, A.; Adeegbe, D.; Jin, C.; Hu, X.; Tagde, A.; Alam, M.; Rajabi, H.; Wong, K.K.; Kufe, D. MUC1-C promotes the suppressive immune microenvironment in non-small cell lung cancer. Oncoimmunology 2017, 6, e1338998. [Google Scholar] [CrossRef]
- Kim, M.J.; Choi, J.R.; Tae, N.; Wi, T.M.; Kim, K.M.; Kim, D.H.; Lee, E.S. Novel Antibodies Targeting MUC1-C Showed Anti-Metastasis and Growth-Inhibitory Effects on Human Breast Cancer Cells. Int. J. Mol. Sci. 2020, 21, 3258. [Google Scholar] [CrossRef]
- Maeda, T.; Hiraki, M.; Jin, C.; Rajabi, H.; Tagde, A.; Alam, M.; Bouillez, A.; Hu, X.; Suzuki, Y.; Miyo, M.; et al. MUC1-C Induces PD-L1 and Immune Evasion in Triple-Negative Breast Cancer. Cancer Res. 2018, 78, 205–215. [Google Scholar] [CrossRef]
- Panchamoorthy, G.; Jin, C.; Raina, D.; Bharti, A.; Yamamoto, M.; Adeebge, D.; Zhao, Q.; Bronson, R.; Jiang, S.; Li, L.; et al. Targeting the human MUC1-C oncoprotein with an antibody-drug conjugate. JCI Insight 2018, 3. [Google Scholar] [CrossRef]
- Detappe, A.; Mathieu, C.; Jin, C.; Agius, M.P.; Diringer, M.C.; Tran, V.L.; Pivot, X.; Lux, F.; Tillement, O.; Kufe, D.; et al. Anti-MUC1-C Antibody-Conjugated Nanoparticles Potentiate the Efficacy of Fractionated Radiation Therapy. Int. J. Radiat. Oncol. Biol. Phys. 2020, 108, 1380–1389. [Google Scholar] [CrossRef]
- Wu, G.; Kim, D.; Kim, J.N.; Park, S.; Maharjan, S.; Koh, H.; Moon, K.; Lee, Y.; Kwon, H.J. A Mucin1 C-terminal Subunit-directed Monoclonal Antibody Targets Overexpressed Mucin1 in Breast Cancer. Theranostics 2018, 8, 78–91. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.; Maharjan, S.; Kim, D.; Kim, J.N.; Park, B.K.; Koh, H.; Moon, K.; Lee, Y.; Kwon, H.J. A Novel Monoclonal Antibody Targets Mucin1 and Attenuates Growth in Pancreatic Cancer Model. Int. J. Mol. Sci. 2018, 19, 2004. [Google Scholar] [CrossRef] [PubMed]
- Hisatsune, A.; Nakayama, H.; Kawasaki, M.; Horie, I.; Miyata, T.; Isohama, Y.; Kim, K.C.; Katsuki, H. Anti-MUC1 antibody inhibits EGF receptor signaling in cancer cells. Biochem. Biophys. Res. Commun. 2011, 405, 377–381. [Google Scholar] [CrossRef] [PubMed]
- Nabavinia, M.S.; Gholoobi, A.; Charbgoo, F.; Nabavinia, M.; Ramezani, M.; Abnous, K. Anti-MUC1 aptamer: A potential opportunity for cancer treatment. Med. Res. Rev. 2017, 37, 1518–1539. [Google Scholar] [CrossRef]
- North, S.A.; Graham, K.; Bodnar, D.; Venner, P. A Pilot Study of the Liposomal MUC1 Vaccine BLP25 in Prostate Specific Antigen Failures After Radical Prostatectomy. J. Urol. 2006, 176, 91–95. [Google Scholar] [CrossRef]
- Hossain, M.K.; Wall, K.A. Immunological Evaluation of Recent MUC1 Glycopeptide Cancer Vaccines. Vaccines 2016, 4, 25. [Google Scholar] [CrossRef]
- Wu, X.; McFall-Boegeman, H.; Rashidijahanabad, Z.; Liu, K.; Pett, C.; Yu, J.; Schorlemer, M.; Ramadan, S.; Behren, S.; Westerlind, U.; et al. Synthesis and immunological evaluation of the unnatural beta-linked mucin-1 Thomsen-Friedenreich conjugate. Org. Biomol. Chem. 2021, 19, 2448–2455. [Google Scholar] [CrossRef]
- McDonald, D.M.; Hanna, C.C.; Ashhurst, A.S.; Corcilius, L.; Byrne, S.N.; Payne, R.J. Synthesis of a Self-Adjuvanting MUC1 Vaccine via Diselenide-Selenoester Ligation-Deselenization. ACS Chem. Biol. 2018, 13, 3279–3285. [Google Scholar] [CrossRef]
- Liu, L.; Wang, Y.; Miao, L.; Liu, Q.; Musetti, S.; Li, J.; Huang, L. Combination Immunotherapy of MUC1 mRNA Nano-vaccine and CTLA-4 Blockade Effectively Inhibits Growth of Triple Negative Breast Cancer. Mol. Ther. 2018, 26, 45–55. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, Z.; Yu, F.; Li, M.; Zhu, H.; Wang, K.; Meng, M.; Zhao, W. The Adjuvant of alpha-Galactosylceramide Presented by Gold Nanoparticles Enhances Antitumor Immune Responses of MUC1 Antigen-Based Tumor Vaccines. Int. J. Nanomed. 2021, 16, 403–420. [Google Scholar] [CrossRef]
- Curry, J.M.; Besmer, D.M.; Erick, T.K.; Steuerwald, N.; Das Roy, L.; Grover, P.; Rao, S.; Nath, S.; Ferrier, J.W.; Reid, R.W.; et al. Indomethacin enhances anti-tumor efficacy of a MUC1 peptide vaccine against breast cancer in MUC1 transgenic mice. PLoS ONE 2019, 14, e0224309. [Google Scholar] [CrossRef]
- Lakshminarayanan, V.; Thompson, P.; Wolfert, M.A.; Buskas, T.; Bradley, J.M.; Pathangey, L.B.; Madsen, C.S.; Cohen, P.A.; Gendler, S.J.; Boons, G.J. Immune recognition of tumor-associated mucin MUC1 is achieved by a fully synthetic aberrantly glycosylated MUC1 tripartite vaccine. Proc. Natl. Acad. Sci. USA 2012, 109, 261–266. [Google Scholar] [CrossRef]
- Rong, Y.; Jin, D.; Wu, W.; Lou, W.; Wang, D.; Kuang, T.; Ni, X.; Qin, X. Induction of protective and therapeutic anti-pancreatic cancer immunity using a reconstructed MUC1 DNA vaccine. BMC Cancer 2009, 9, 191. [Google Scholar] [CrossRef]
- Sugiura, D.; Aida, S.; Denda-Nagai, K.; Takeda, K.; Kamata-Sakurai, M.; Yagita, H.; Irimura, T. Differential effector mechanisms induced by vaccination with MUC1 DNA in the rejection of colon carcinoma growth at orthotopic sites and metastases. Cancer Sci. 2008, 99, 2477–2484. [Google Scholar] [CrossRef]
- Nagai, K.; Adachi, T.; Harada, H.; Eguchi, S.; Sugiyama, H.; Miyazaki, Y. Dendritic Cell-based Immunotherapy Pulsed with Wilms Tumor 1 Peptide and Mucin 1 as an Adjuvant Therapy for Pancreatic Ductal Adenocarcinoma After Curative Resection: A Phase I/IIa Clinical Trial. Anticancer Res. 2020, 40, 5765–5776. [Google Scholar] [CrossRef]
- Pourjafar, M.; Samadi, P.; Saidijam, M. MUC1 antibody-based therapeutics: The promise of cancer immunotherapy. Immunotherapy 2020, 12, 1269–1286. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, W.; Zhang, Z.; Zhang, S.; Zhu, P.; Ko, J.K.-S.; Yung, K.K.-L. MUC1: Structure, Function, and Clinic Application in Epithelial Cancers. Int. J. Mol. Sci. 2021, 22, 6567. https://doi.org/10.3390/ijms22126567
Chen W, Zhang Z, Zhang S, Zhu P, Ko JK-S, Yung KK-L. MUC1: Structure, Function, and Clinic Application in Epithelial Cancers. International Journal of Molecular Sciences. 2021; 22(12):6567. https://doi.org/10.3390/ijms22126567
Chicago/Turabian StyleChen, Wenqing, Zhu Zhang, Shiqing Zhang, Peili Zhu, Joshua Ka-Shun Ko, and Ken Kin-Lam Yung. 2021. "MUC1: Structure, Function, and Clinic Application in Epithelial Cancers" International Journal of Molecular Sciences 22, no. 12: 6567. https://doi.org/10.3390/ijms22126567
APA StyleChen, W., Zhang, Z., Zhang, S., Zhu, P., Ko, J. K.-S., & Yung, K. K.-L. (2021). MUC1: Structure, Function, and Clinic Application in Epithelial Cancers. International Journal of Molecular Sciences, 22(12), 6567. https://doi.org/10.3390/ijms22126567




