Activity of CdTe Quantum-Dot-Tagged Superoxide Dismutase and Its Analysis in Capillary Electrophoresis
Abstract
1. Introduction
2. Results
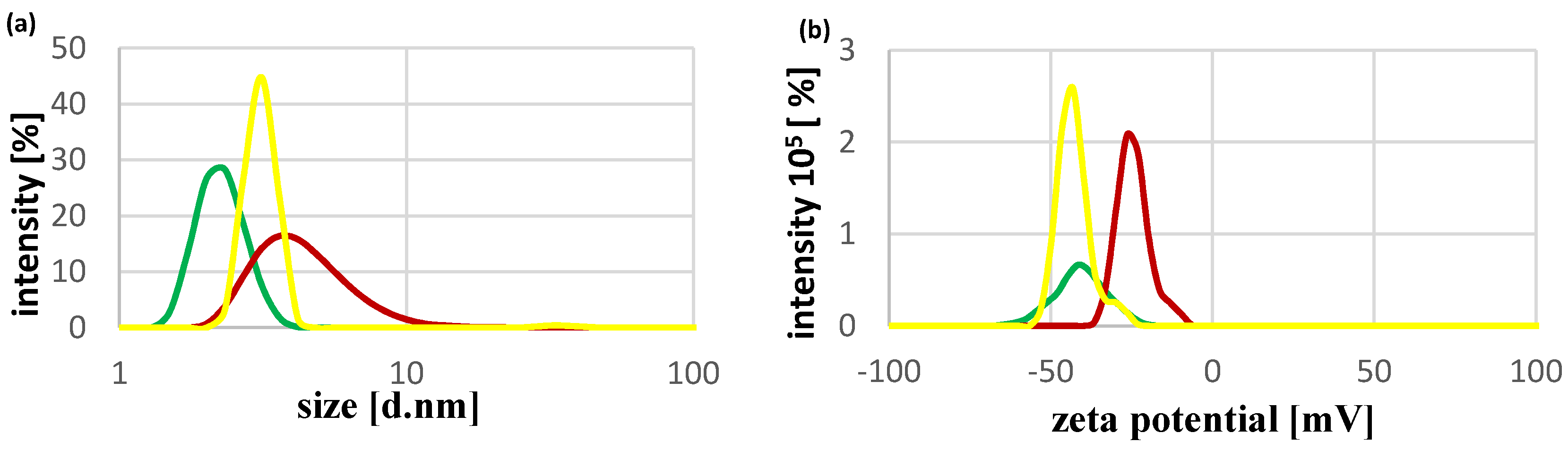
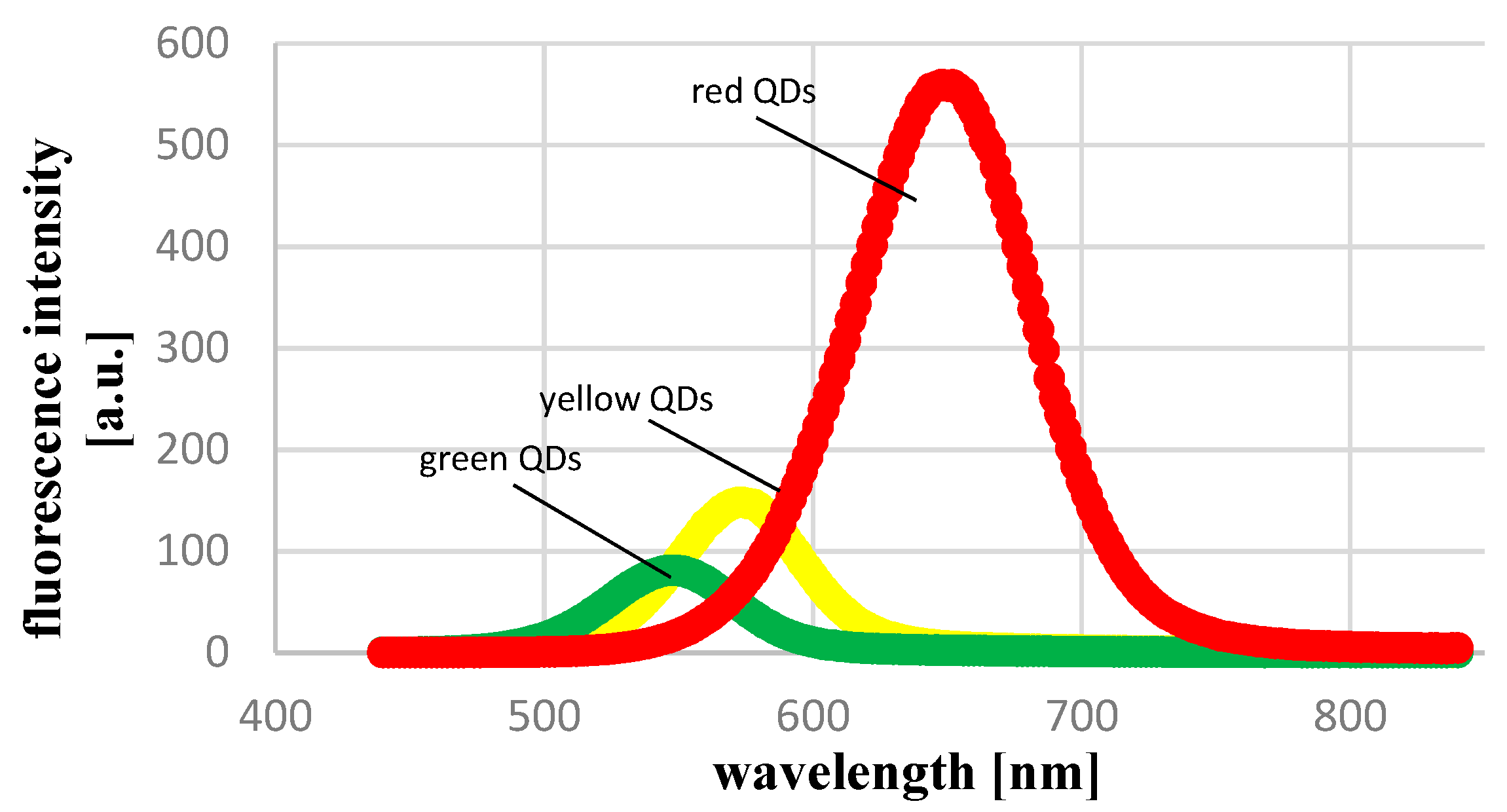
2.1. Physicochemical Characteristics of CdTe QDs
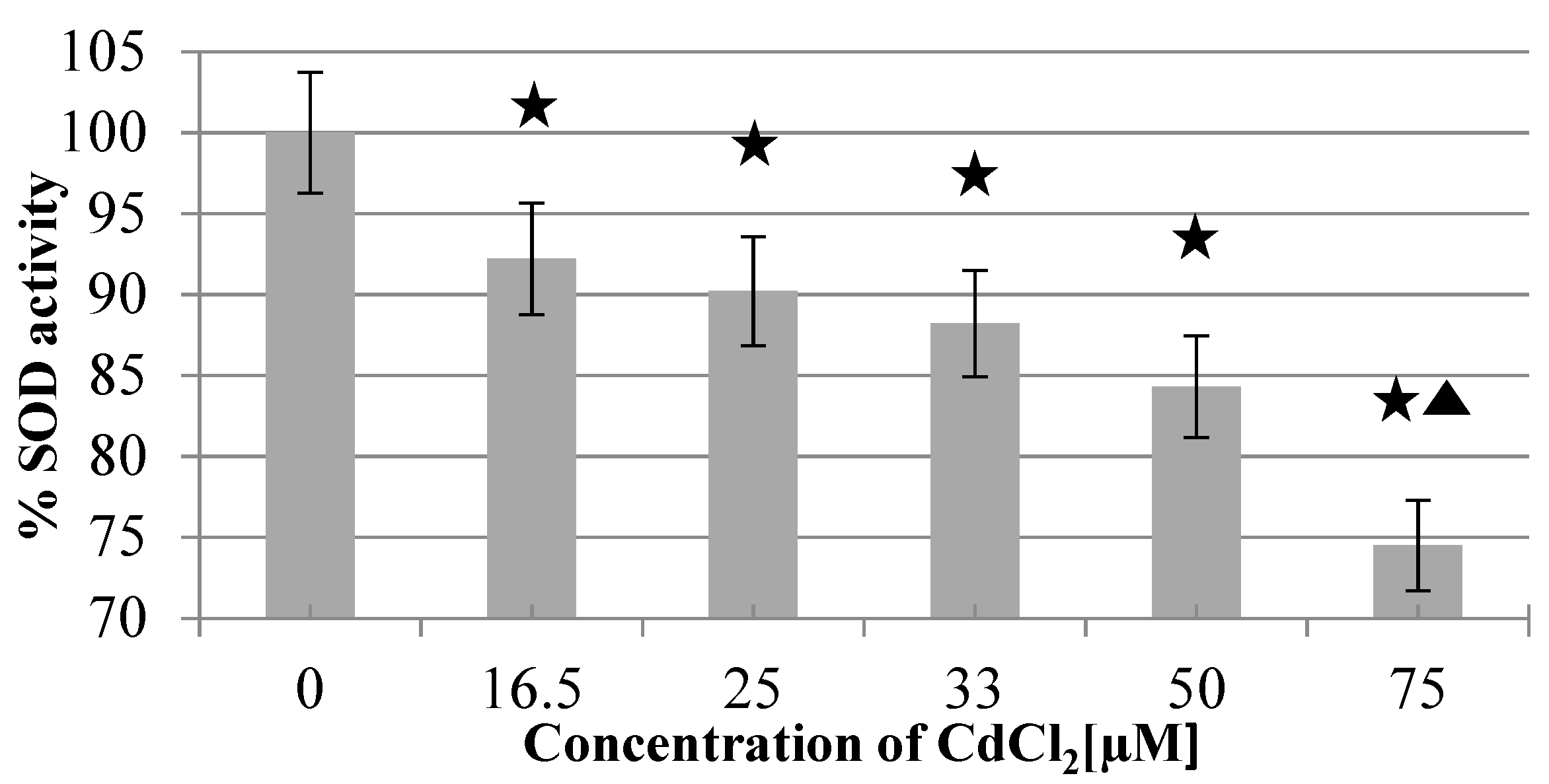
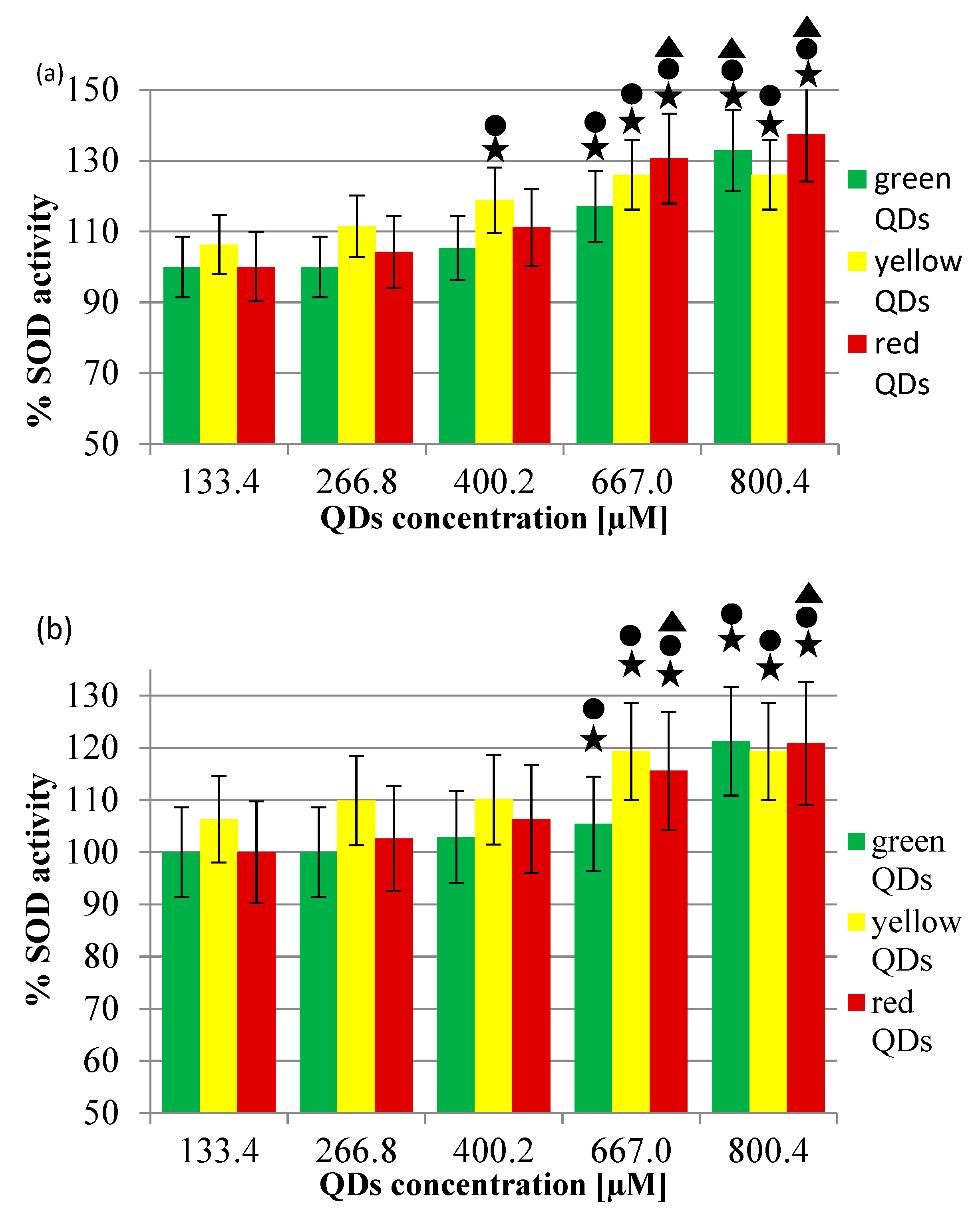
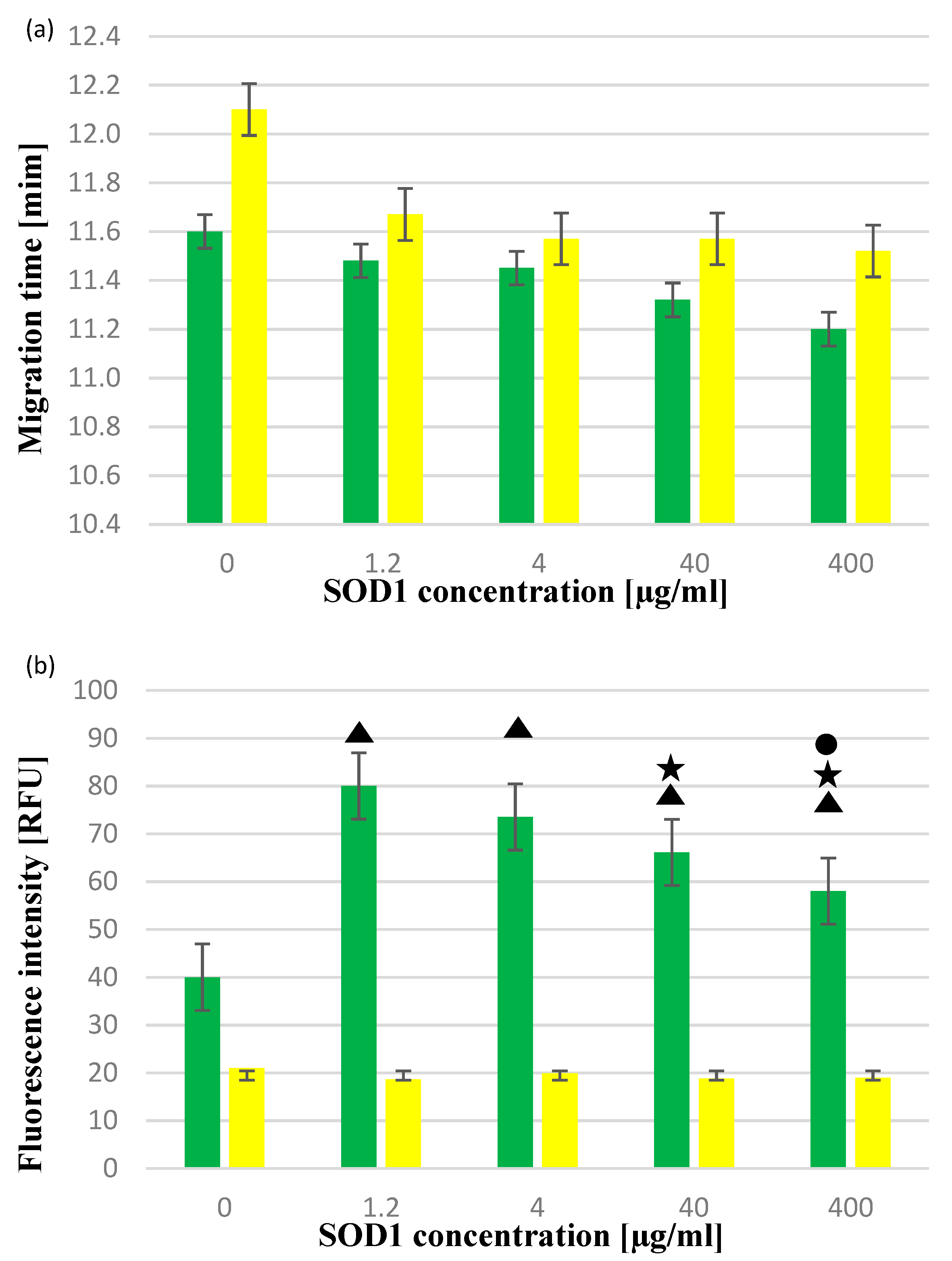
2.2. The Activity of SOD1 in the Presence of CdCl2 or CdTe QDs
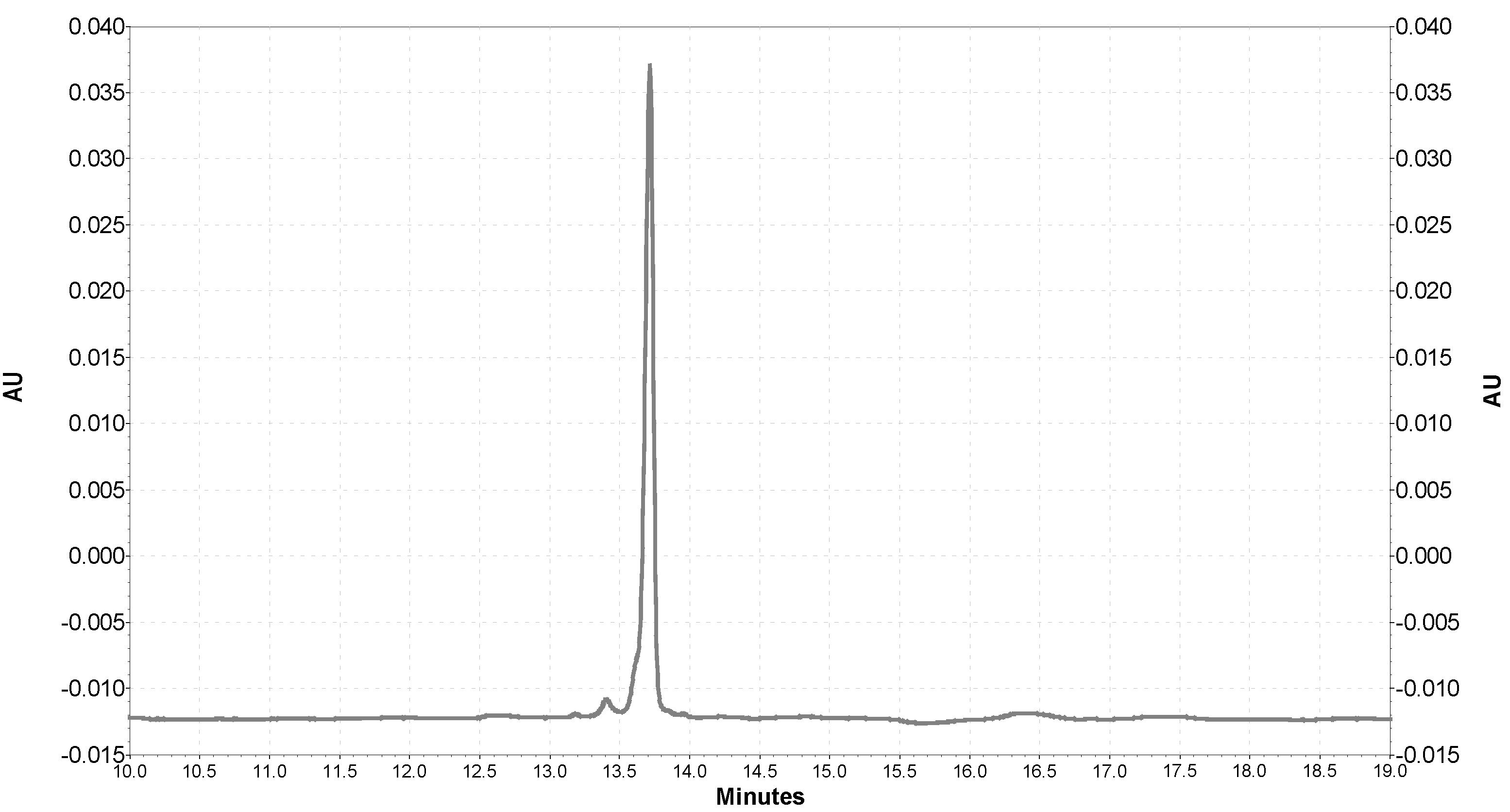
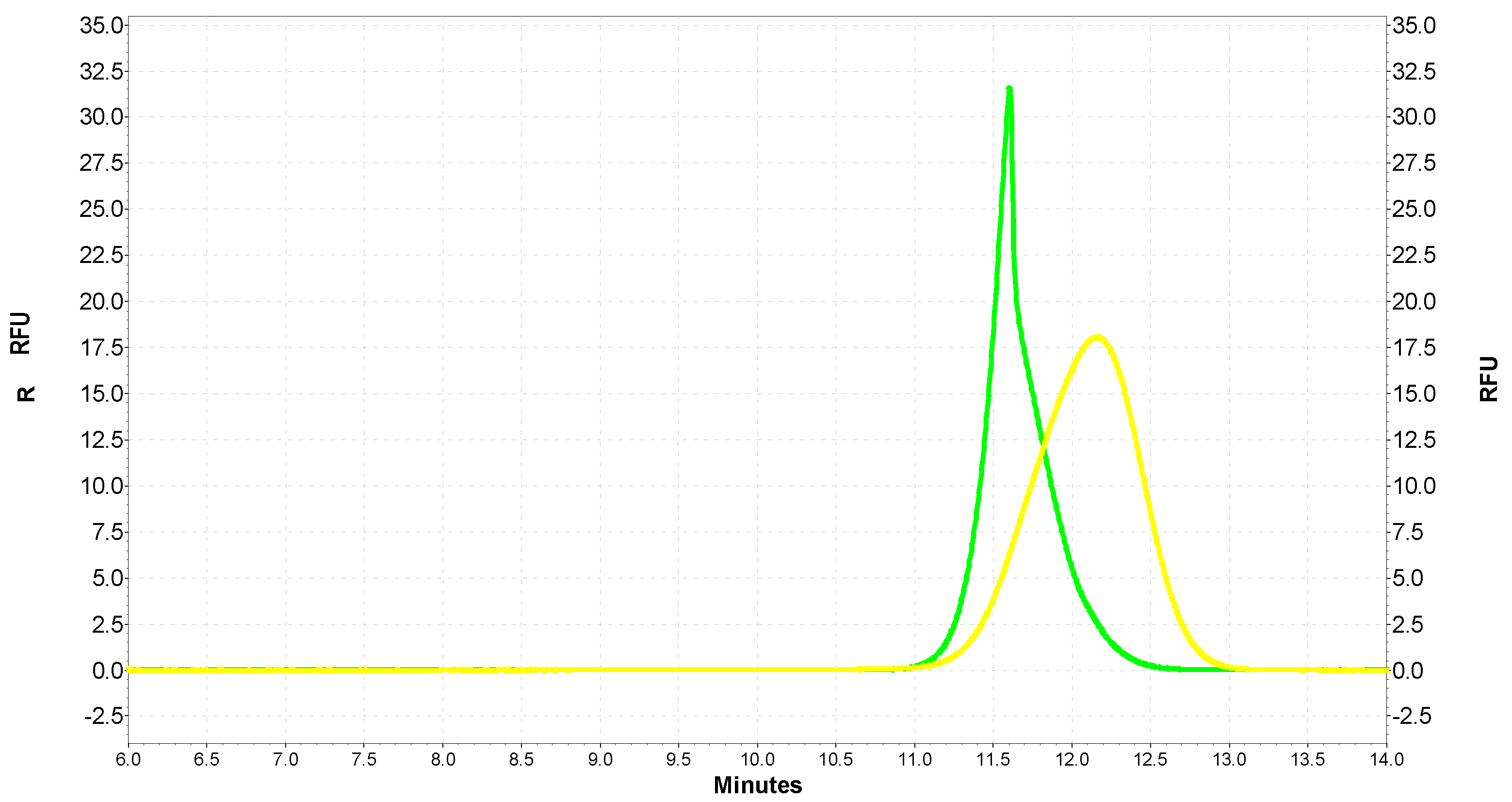
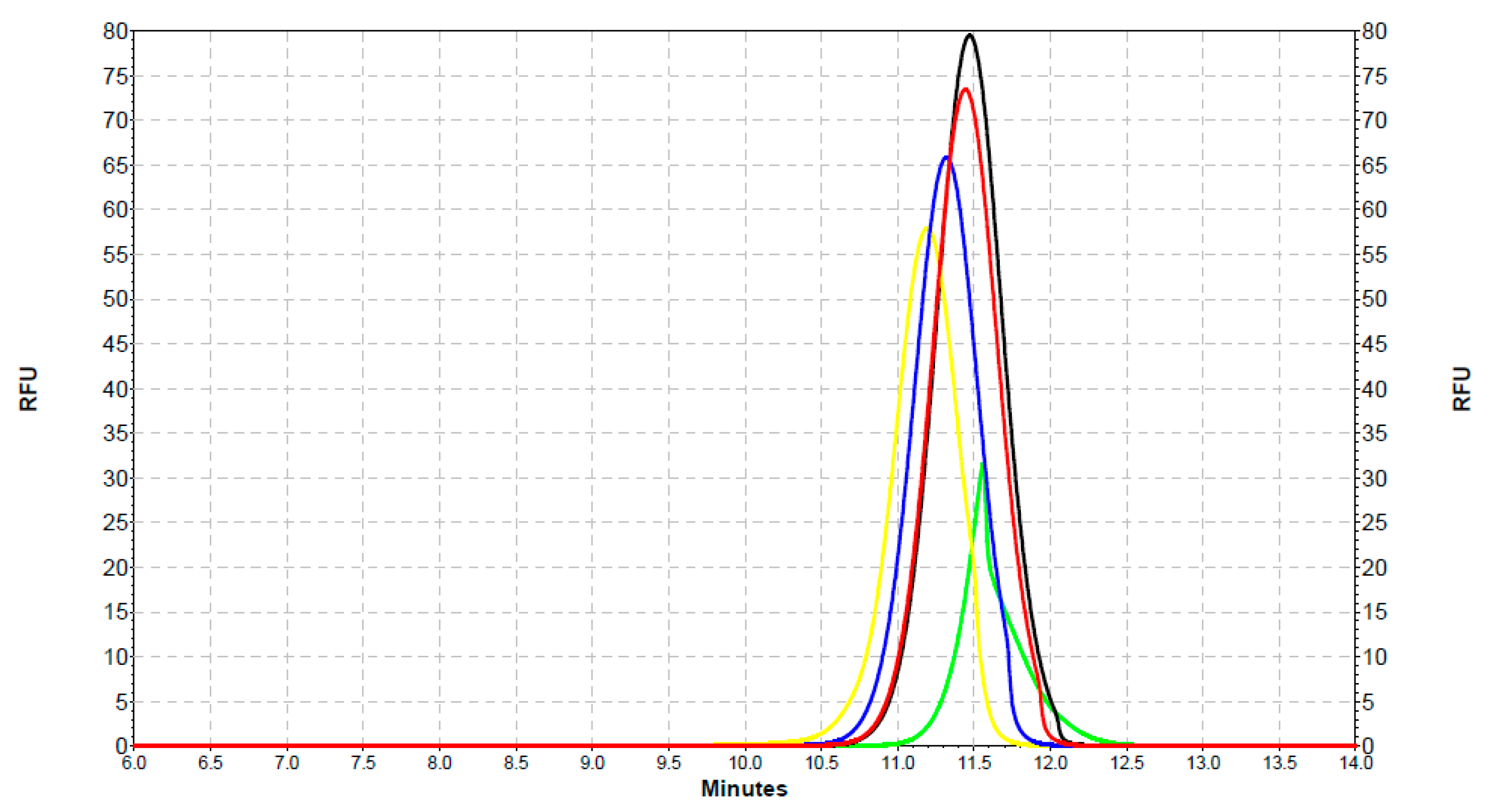
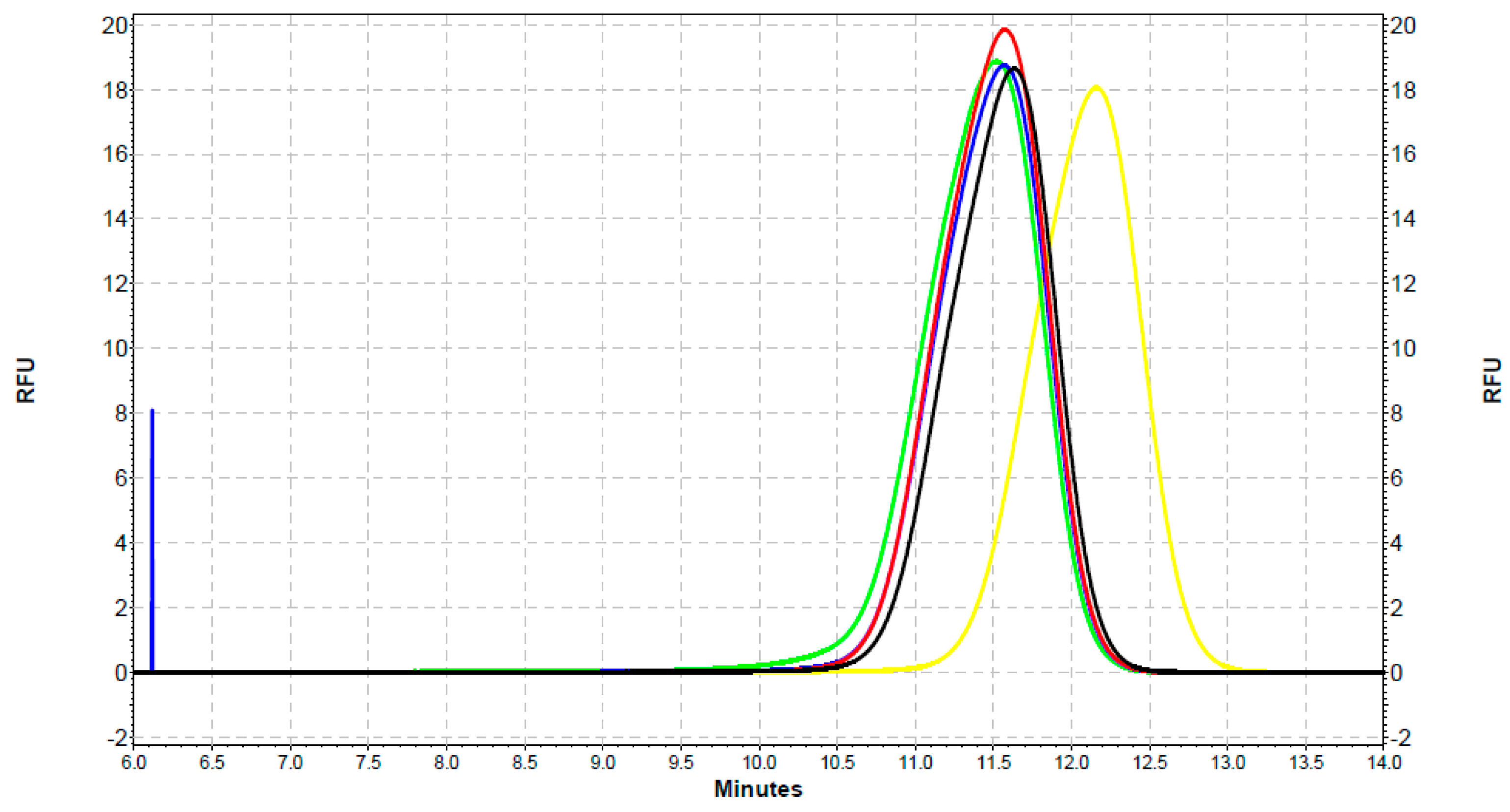
2.3. Analysis of the Interaction of SOD1 with QDs with the Use of Capillary Electrophoresis
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Methods
4.2.1. Synthesis of CdTe QDs and SOD–QDs Complexes Preparation
4.2.2. Dynamic Light Scattering
4.2.3. Fluorescence Spectra Measurement
4.2.4. Assaying for SOD1 Activity
4.2.5. Capillary Electrophoresis
4.2.6. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Chan, W.C.; Maxwell, D.J.; Gao, X.; Bailey, R.E.; Han, M.; Nie, S. Luminescent quantum dots for multiplexed biological detection and imaging. Curr. Opin. Biotechnol. 2002, 13, 40–46. [Google Scholar] [CrossRef]
- Nirmal, M.; Brus, L. Luminescence photophysics in semiconductor nanocrystals. Acc. Chem. Res. 1999, 32, 407–414. [Google Scholar] [CrossRef]
- Alivisatos, A.P. Semiconductor clusters, nanocrystals, and quantum dots. Science 1996, 271, 933–937. [Google Scholar] [CrossRef]
- Olkhovets, A.; Hsu, R.-C.; Lipovskii, A.; Wise, F.W. Size-dependent temperature variation of the energy gap in lead-salt quantum dots. Phys. Rev. Lett. 1998, 81, 3539–3542. [Google Scholar] [CrossRef]
- Leatherdale, C.A.; Woo, W.-K.; Mikulec, F.V.; Bawendi, M.G. On the absorption cross section of cdse nanocrystal quantum dots. J. Phys. Chem. B. 2002, 106, 7619–7622. [Google Scholar] [CrossRef]
- Michalet, X.; Pinaud, F.F.; Bentolila, L.A.; Tsay, J.M.; Doose, J.; Li, J.J.; Sundaresan, G.; Wu, A.M.; Gambhir, S.S.; Weiss, S. Quantum dots for live cells, in vivo imaging, and diagnostics. Science 2005, 28, 538–544. [Google Scholar] [CrossRef]
- Wang, G.; Li, Z.; Ma, N. Next-generation DNA-functionalized quantum dots as biological sensors. ACS Chem. Biol. 2018, 13, 1705–1713. [Google Scholar] [CrossRef]
- Inoshita, T.; Sakaki, H. Density of states and phonon-induced relaxation of electrons in semiconductor quantum dots. Phys. Rev. B. 1997, 56, 4355–4358. [Google Scholar] [CrossRef]
- Hoshino, A.; Fujioka, K.; Oku, T.; Suga, M.; Sasaki, Y.F.; Ohta, T.; Yasuhara, M.; Suzuki, K.; Yamamoto, K. Physicochemical properties and cellular toxicity of nanocrystal quantum dots depend on their surface modification. Nano Lett. 2004, 4, 2163–2169. [Google Scholar] [CrossRef]
- Mashinchian, O.; Johari-Ahar, M.; Ghaemi, B.; Rashidi, M.; Barar, J.; Omidi, Y. Impacts of quantum dots in molecular detection and bioimaging of cancer. Bioimpacts 2014, 4, 149–166. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.; Cao, Z.; Wu, M.; Lu, C. Intracellular tracking of single native molecules with electroporation-delivered quantum dots. Anal. Chem. 2014, 86, 11403–11409. [Google Scholar] [CrossRef]
- Lisse, D.; Richter, C.P.; Drees, C.; Birkholz, O.; You, C.; Rampazzo, E.; Piehler, J. Monofunctional stealth nanoparticle for unbiased single molecule tracking inside living cells. Nano Lett. 2014, 14, 2189–2195. [Google Scholar] [CrossRef] [PubMed]
- Komatsuzaki, A.; Ohyanagi, T.; Tsukasaki, Y.; Miyanaga, Y.; Ueda, M.; Jin, T. Compact halo-ligand-conjugated quantum dots for multicolored single-molecule imaging of overcrowding GPCR proteins on cell membranes. Small 2015, 11, 1396–1401. [Google Scholar] [CrossRef] [PubMed]
- Varela, J.A.; Dupuis, J.P.; Etchepare, L.; Espana, A.; Cognet, L.; Groc, L. Targeting neurotransmitter receptors with nanoparticles in vivo allows single-molecule tracking in acute brain slices. Nat. Commun. 2016, 7, 10947. [Google Scholar] [CrossRef] [PubMed]
- Resch-Genger, U.; Grabolle, M.; Cavaliere-Jaricot, S.; Nitschke, R.; Nann, T. Quantum dots versus organic dyes as fluorescent labels. Nat. Methods 2008, 5, 763–775. [Google Scholar] [CrossRef] [PubMed]
- Stroh, M.; Zimmer, J.P.; Duda, D.G.; Levchenko, T.S.; Cohen, K.S.; Brown, E.B.; Scadden, D.T.; Torchilin, V.P.; Bawendi, M.G.; Fukumura, D.; et al. Quantum dots spectrally distinguish multiple species within the tumor milieu in vivo. Nat. Med. 2005, 11, 678–682. [Google Scholar] [CrossRef]
- Gao, X.; Cui, Y.; Levenson, R.M.; Chung, L.W.; Nie, S. In vivo cancer targeting and imaging with semiconductor quantum dots. Nat. Biotechnol. 2004, 22, 969–976. [Google Scholar] [CrossRef] [PubMed]
- Levene, M.J.; Dombeck, D.A.; Kasischke, K.A.; Molloy, R.P.; Webb, W.W. In vivo multiphoton microscopy of deep brain tissue. J. Neurophysiol. 2004, 91, 1908–1912. [Google Scholar] [CrossRef] [PubMed]
- Akerman, M.E.; Chan, W.C.; Laakkonen, P.; Bhatia, S.N.; Ruoslahti, E. Nanocrystal targeting in vivo. Proc. Natl. Acad. Sci. USA 2002, 99, 12617–12621. [Google Scholar] [CrossRef]
- Voura, E.B.; Jaiswal, J.K.; Mattoussi, H.; Simon, S.M. Tracking metastatic tumor cell extravasation with quantum dot nanocrystals and fluorescence emission-scanning microscopy. Nat. Med. 2004, 10, 993–998. [Google Scholar] [CrossRef] [PubMed]
- Drbohlavova, J.; Adam, V.; Kizek, R.; Hubalek, J. Quantum dots—Characterization, preparation and usage in biological systems. Int. J. Mol. Sci. 2009, 10, 656–673. [Google Scholar] [CrossRef] [PubMed]
- Stanisavljevic, M.; Krizkova, S.; Vaculovicova, M.; Kizek, R.; Adam, V. Quantum dots-fluorescence resonance energy transfer-based nanosensors and their application. Biosens. Bioelectron. 2015, 74, 562–574. [Google Scholar] [CrossRef]
- Manabe, N.; Hoshino, A.; Liang, Y.; Goto, T.; Kato, N.; Yamamoto, K. Quantum dot as a drug tracer in vivo. IEEE Trans. NanoBiosci. 2006, 5, 263–267. [Google Scholar] [CrossRef] [PubMed]
- Bagalkot, V.; Zhang, L.; Levy-Nissenbaum, E.; Jon, S.; Kantoff, P.W.; Langer, O.C. Quantum dot−aptamer conjugates for synchronous cancer imaging, therapy, and sensing of drug delivery based on bi-fluorescence resonance energy transfer. Nano Lett. 2007, 7, 3065–3070. [Google Scholar] [CrossRef] [PubMed]
- Zrazhevskiy, P.; Sena, M.; Gao, X. Designing multifunctional quantum dots for bioimaging, detection, and drug delivery. Chem. Soc. Rev. 2010, 39, 4326–4354. [Google Scholar] [CrossRef]
- Probst, C.E.; Zrazhevskiy, P.; Bagalkot, V.; Gao, X. Quantum dots as a platform for nanoparticle drug delivery vehicle design. Adv. Drug Deliv. Rev. 2013, 65, 703–718. [Google Scholar] [CrossRef]
- Zhang, L.; Shang, L.; Dong, S. Sensitive and selective determination of Cu2+ by electrochemiluminescence of CdTe quantum dots. Electrochem. Commun. 2008, 10, 1452–1454. [Google Scholar] [CrossRef]
- Chen, S.; Zhang, X.; Zhang, Q.; Hou, X.; Zhou, Q.; Yan, J.; Tan, W. CdSe quantum dots decorated by mercaptosuccinic acid as fluorescence probe for Cu2+. J. Lumin. 2011, 131, 947–951. [Google Scholar] [CrossRef]
- Dutta, P.; Saikia, D.; Adhikary, N.C.; Sarma, N.S. Macromolecular systems with MSA-capped CdTe and CdTe/ZnS core/shell quantum dots as superselective and ultrasensitive optical sensors for picric acid Explosive. ACS Appl. Mater. Interfaces 2015, 7, 24778–24790. [Google Scholar] [CrossRef] [PubMed]
- Yuan, J.; Guo, W.; Yin, J.; Wang, E. Glutathione-capped CdTe quantum dots for the sensitive detection of glucose. Talanta 2009, 77, 1858–1863. [Google Scholar] [CrossRef]
- Yuan, J.; Guo, W.; Wang, E. Utilizing a CdTe quantum dots−enzyme hybrid system for the determination of both phenolic compounds and hydrogen peroxide. Anal. Chem. 2008, 80, 1141–1145. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Sun, X.; Lao, J.; He, H.; Cheng, T.; Wang, M.; Wang, S.; Huang, F. Multifunctional graphene quantum dots for simultaneous targeted cellular imaging and drug delivery. Colloids Surf. B Biointerfaces 2014, 122, 638–644. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.-X.; Zhu, B.-J. The research and applications of quantum dots as nano-carriers for targeted drug delivery and cancer therapy. Nanoscale Res. Lett. 2016, 11, 207. [Google Scholar] [CrossRef] [PubMed]
- Lewandowski, Ł.; Kepinska, M.; Milnerowicz, H. Inhibition of copper-zinc superoxide dismutase activity by selected environmental xenobiotics. Environ. Toxicol. Pharmacol. 2018, 58, 105–113. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Cui, E.; Liu, R. Molecular mechanism of copper-zinc superoxide dismutase activity change exposed to N-acetyl-L-cysteine-capped CdTe quantum dots-induced oxidative damage in mouse primary hepatocytes and nephrocytes. Environ. Sci. Pollut. Res. 2015, 22, 18267–18277. [Google Scholar] [CrossRef]
- McCord, J.M.; Fridovich, I. Superoxide Dismutase. An enzymic function for erythrocuprein (hemocuprein). J. Biol. Chem. 1969, 244, 6049–6055. [Google Scholar] [CrossRef]
- Fukai, T.; Ushio-Fukai, M. Superoxide dismutases: Role in redox signaling, vascular function, and diseases. Antioxid. Redox. Signal. 2011, 15, 1583–1606. [Google Scholar] [CrossRef]
- Lewandowski, Ł.; Kepinska, M.; Milnerowicz, H. The copper-zinc superoxide dismutase activity in selected diseases. Eur. J. Clin. Invest. 2019, 49, e13036. [Google Scholar] [CrossRef]
- Younus, H. Therapeutic potentials of superoxide dismutase. Int. J. Health Sci. 2018, 12, 88–93. [Google Scholar]
- Samia, A.C.; Chen, X.; Burda, C. Semiconductor Quantum Dots for Photodynamic Therapy. J. Am. Chem. Soc. 2003, 125, 15736–15737. [Google Scholar] [CrossRef]
- Ipe, B.I.; Lehnig, M.; Niemeyer, C.M. On the generation of free radical species from quantum dots. Small 2005, 1, 706–709. [Google Scholar] [CrossRef]
- Lai, L.; Lin, C.; Xu, Z.-Q.; Han, X.-L.; Tian, F.-F.; Mei, P.; Li, D.-W.; Ge, Y.-S.; Jiang, F.-L.; Zhang, Y.-Z.; et al. Spectroscopic studies on the interactions between CdTe quantum dots coated with different ligands and human serum albumin. Spectrochim. Acta Part. A Mol. Biomol. Spectrosc. 2012, 97, 366–376. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.; Qiu, H.; Lu, S.; Zhu, F.; Xiao, Q. Study on the molecular interaction of graphene quantum dots with human serum albumin: Combined spectroscopic and electrochemical approaches. J. Hazard. Mater. 2015, 285, 18–26. [Google Scholar] [CrossRef]
- Shukla, G.S.; Hussain, T.; Srivastava, R.S.; Chandra, S.V. Glutathione peroxidase and catalase in liver, kidney, testis and brain regions of rats following cadmium exposure and subsequent withdrawal. Ind. Health 1989, 27, 59–69. [Google Scholar] [CrossRef]
- Roméo, M.; Bennani, N.; Gnassia-Barelli, M.; Lafaurie, M.; Girard, J.P. Cadmium and copper display different responses towards oxidative stress in the kidney of the sea bass Dicentrarchus labrax. Aquat. Toxicol. 2000, 48, 185–194. [Google Scholar] [CrossRef]
- Pruell, R.J.; Engelhardt, F.R. Liver cadmium uptake, catalase inhibition and cadmium thionein production in the killifish (Fundulus Heteroclitus) induced by experimental cadmium exposure. Mar. Environ. Res. 1980, 3, 101–111. [Google Scholar] [CrossRef]
- Jamall, I.S.; Crispin Smith, J. Effects of cadmium on glutathione peroxidase, superoxide dismutase, and lipid peroxidation in the rat heart: A possible mechanism of cadmium cardiotoxicity. Toxicol. Appl. Pharmacol. 1985, 80, 33–42. [Google Scholar] [CrossRef]
- Huang, Y.-H.; Shih, C.-M.; Huang, C.-J.; Lin, C.-M.; Chou, C.-M.; Tsai, M.-L.; Liu, T.P.; Chiu, J.-F.; Chen, C.-T. Effects of cadmium on structure and enzymatic activity of Cu,Zn-SOD and oxidative status in neural cells. J. Cell Biochem. 2006, 98, 577–589. [Google Scholar] [CrossRef]
- Zhu, J.Y.; Chan, K.M. Mechanism of cadmium-induced cytotoxicity on the ZFL zebrafish liver cell line. Metallomics 2012, 4, 1064–1076. [Google Scholar] [CrossRef] [PubMed]
- Banni, M.; Chouchene, L.; Said, K.; Kerkeni, A.; Messaoudi, I. Mechanisms underlying the protective effect of zinc and selenium against cadmium-induced oxidative stress in zebrafish Danio rerio. Biometals 2011, 24, 981–992. [Google Scholar] [CrossRef] [PubMed]
- Nyyssönen, K.; Porkkala-Sarataho, E.; Kaikkonen, J.; Salonen, J.T. Ascorbate and urate are the strongest determinants of plasma antioxidative capacity and serum lipid resistance to oxidation in Finnish men. Atherosclerosis 1997, 130, 223–233. [Google Scholar] [CrossRef]
- Wang, G.; Su, X.; Yang, S.; Jia, Y.; Li, D. The double-effect mechanism between Fe3O4 nanoparticles and MSA-capped CdTe QDs. J. Lumin. 2012, 132, 2505–2511. [Google Scholar] [CrossRef]
- Yu, X.; Liu, J.; Zuo, S.; Yu, Y.; Cai, K.; Yang, R. Application of mercaptosuccinic acid capped CdTe quantum dots for latent fingermark development. Forensic Sci. Int. 2013, 231, 125–130. [Google Scholar] [CrossRef]
- Guszpit, E.; Krizkova, S.; Kepinska, M.; Rodrigo, M.A.; Milnerowicz, H.; Kopel, P.; Kizek, R. Fluorescence-tagged metallothionein with CdTe quantum dots analyzed by the chip-CE technique. J. Nanopart. Res. 2015, 17, 423. [Google Scholar] [CrossRef][Green Version]
- Wang, J.; Zhang, H.; Zhang, T.; Zhang, R.; Liu, R.; Chen, Y. Molecular mechanism on cadmium-induced activity changes of catalase and superoxide dismutase. Int. J. Biol. Macromol. 2015, 77, 59–67. [Google Scholar] [CrossRef] [PubMed]
- Zeng, G.-M.; Chen, A.-W.; Chen, G.-Q.; Hu, X.-J.; Guan, S.; Shang, C.; Lu, L.-H.; Zou, Z.-J. Responses ofp chrysosporium to toxic pollutants: Physiological flux, oxidative stress, and detoxification. Environ. Sci. Technol. 2012, 46, 7818–7825. [Google Scholar] [CrossRef]
- Derfus, A.M.; Chan, W.C.; Bhatia, S.N. Probing the cytotoxicity of semiconductor quantum dots. Nano Lett. 2004, 4, 11–18. [Google Scholar] [CrossRef]
- Lu, Z.; Li, C.M.; Bao, H.; Qiao, Y.; Toh, Y.; Yang, X. Mechanism of antimicrobial activity of CdTe quantum dots. Langmuir 2008, 24, 5445–5452. [Google Scholar] [CrossRef]
- Singh, B.R.; Singh, B.N.; Khan, W.; Singh, H.B.; Naqvi, A.H. ROS-mediated apoptotic cell death in prostate cancer LNCaP cells induced by biosurfactant stabilized CdS quantum dots. Biomaterials 2012, 33, 5753–5767. [Google Scholar] [CrossRef]
- Stanisavjevic, M.; Chomoucka, J.; Dostalova, S.; Krizkova, S.; Vaculovicova, M.; Adam, V.; Rene, K. Interactions between CdTe quantum dots and DNA revealed by capillary electrophoresis with laser-induced fluorescence detection. Electrophoresis 2014, 35, 2587–2592. [Google Scholar] [CrossRef]
- Tang, T.; Deng, J.; Zhang, M.; Shi, G.; Zhou, T. Quantum dot- DNA aptamer conjugates coupled with capillary electrophoresis: A universal strategy for radiometric detection of organosphorus pesticides. Talanta 2016, 146, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Weng, J.; Sang, F.; Song, X.; Cao, C.; Ren, J. Characterization of quantum dot bioconjugates by capillary electrophoresis with laser-induced fluorescent detection. J. Chromatogr. A 2006, 1131, 251–254. [Google Scholar] [CrossRef] [PubMed]
- Matczuk, M.; Legat, J.; Timerbaev, A.R.; Jarosz, M. A sensitive and versatile method for characterization of protein- mediated transformations of quantum dots. Analyst 2016, 141, 2574–2580. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Guan, L.-Y.; Li, Y.-Q.; Lin, S.; Zhang, M.-Z.; Chen, J.; Ma, Z.-Y.; Zhao, Y.-D. Characterization of CdTe/CdSe quantum dots- transferrin fluorescent probes for cellular labeling. Anal. Chim. Acta 2012, 741, 86–92. [Google Scholar] [CrossRef] [PubMed]
- Janu, L.; Stanisavljevic, M.; Krizkova, S.; Sobrova, P.; Vaculovicova, M.; Kizek, R.; Adam, V. Electrophoretic study of peptide-mediated quantum dot-human immunoglobulin bioconjugation. Electrophoresis 2013, 34, 2725–2732. [Google Scholar] [CrossRef] [PubMed]
- Duan, J.; Song, L.; Zhan, J. One-pot synthesis of highly luminescent CdTe quantum dots by microwave irradiation reduction and their Hg 2+ -sensitive properties. Nano Res. 2009, 2, 61–68. [Google Scholar] [CrossRef]
- Misra, H.; Fridovich, I. The role of superoxide anion in the autooxidation of epinephrine and a simple assay for superoxide dismutase. J. Biol. Chem. 1972, 247, 3170–3175. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zaręba, N.; Lewandowski, Ł.; Kunachowicz, D.; Kizek, R.; Kepinska, M. Activity of CdTe Quantum-Dot-Tagged Superoxide Dismutase and Its Analysis in Capillary Electrophoresis. Int. J. Mol. Sci. 2021, 22, 6156. https://doi.org/10.3390/ijms22116156
Zaręba N, Lewandowski Ł, Kunachowicz D, Kizek R, Kepinska M. Activity of CdTe Quantum-Dot-Tagged Superoxide Dismutase and Its Analysis in Capillary Electrophoresis. International Journal of Molecular Sciences. 2021; 22(11):6156. https://doi.org/10.3390/ijms22116156
Chicago/Turabian StyleZaręba, Natalia, Łukasz Lewandowski, Dominika Kunachowicz, Rene Kizek, and Marta Kepinska. 2021. "Activity of CdTe Quantum-Dot-Tagged Superoxide Dismutase and Its Analysis in Capillary Electrophoresis" International Journal of Molecular Sciences 22, no. 11: 6156. https://doi.org/10.3390/ijms22116156
APA StyleZaręba, N., Lewandowski, Ł., Kunachowicz, D., Kizek, R., & Kepinska, M. (2021). Activity of CdTe Quantum-Dot-Tagged Superoxide Dismutase and Its Analysis in Capillary Electrophoresis. International Journal of Molecular Sciences, 22(11), 6156. https://doi.org/10.3390/ijms22116156






