microRNAs in the Regulation of Melanogenesis
Abstract
1. Introduction
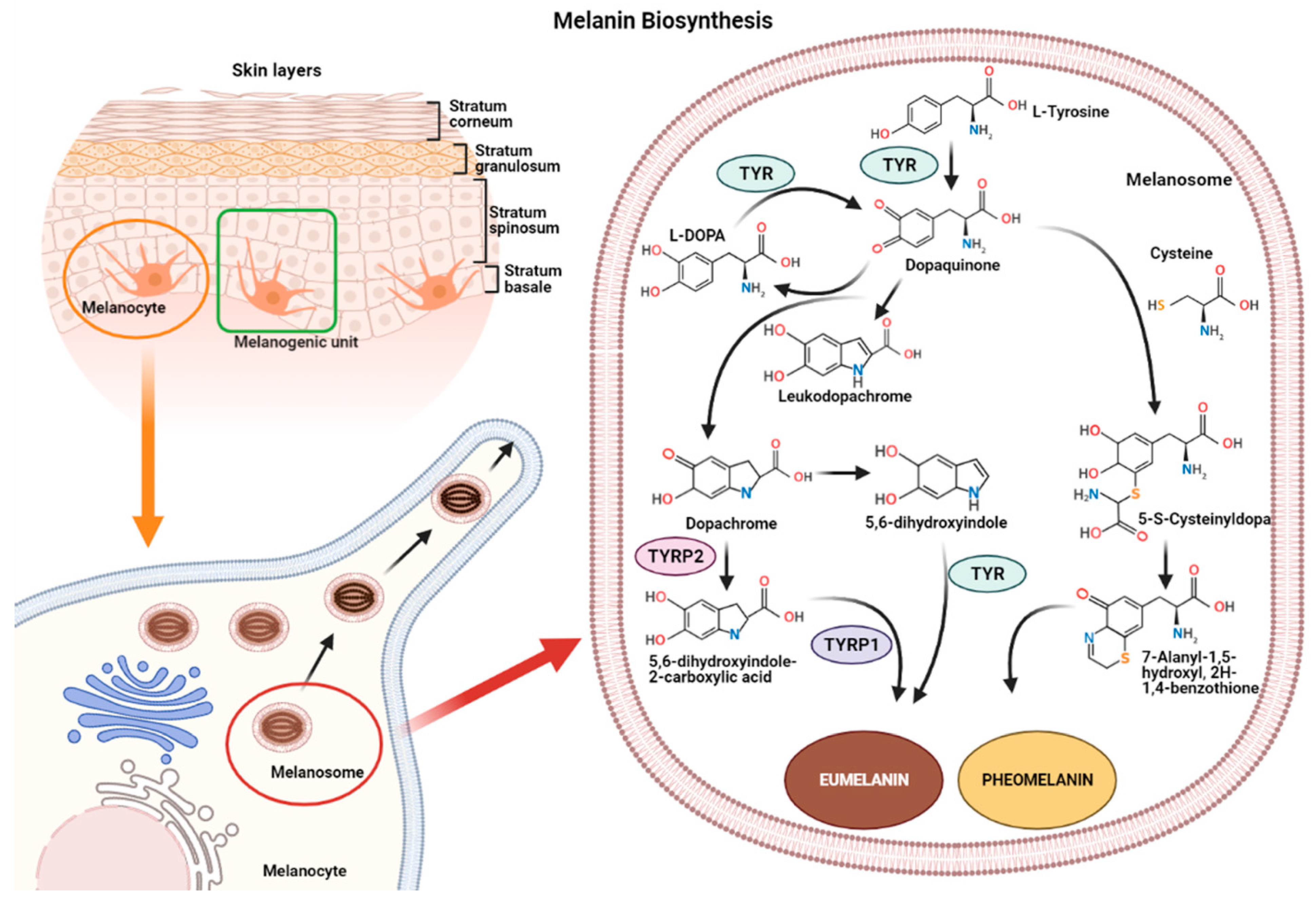
2. Melanin Biosynthesis
- (1)
- In reactions leading to eumelanin production, dopaquinone undergoes intramolecular cyclization to produce leukodopachrome (cyclodopa). Cyclodopa undergoes redox exchange with another molecule of dopaquinone to form dopachrome and DOPA [21]. The dopachrome downstream process is branched in two ways. The first leads to the formation of 5,6-dihydroxyindole-2-carboxylic acid (DHICA) through TYRP-2 intervention and then into eumelanin by TYRP-1 conversion. The second leads to the conversion of dopachrome into 5,6-dihydroxyindole (DHI) and then into eumelanin involving TYR. At the end of this reaction, black-brownish eumelanin is formed.
- (2)
- In reactions leading to pheomelanin production, in the presence of cystein or glutathione, dopaquinone can be converted into 5-S-cysteinyldopa, or glutathionyldopa, which is then converted into quinoline and finally polymerized into red-yellow pheomelanin.
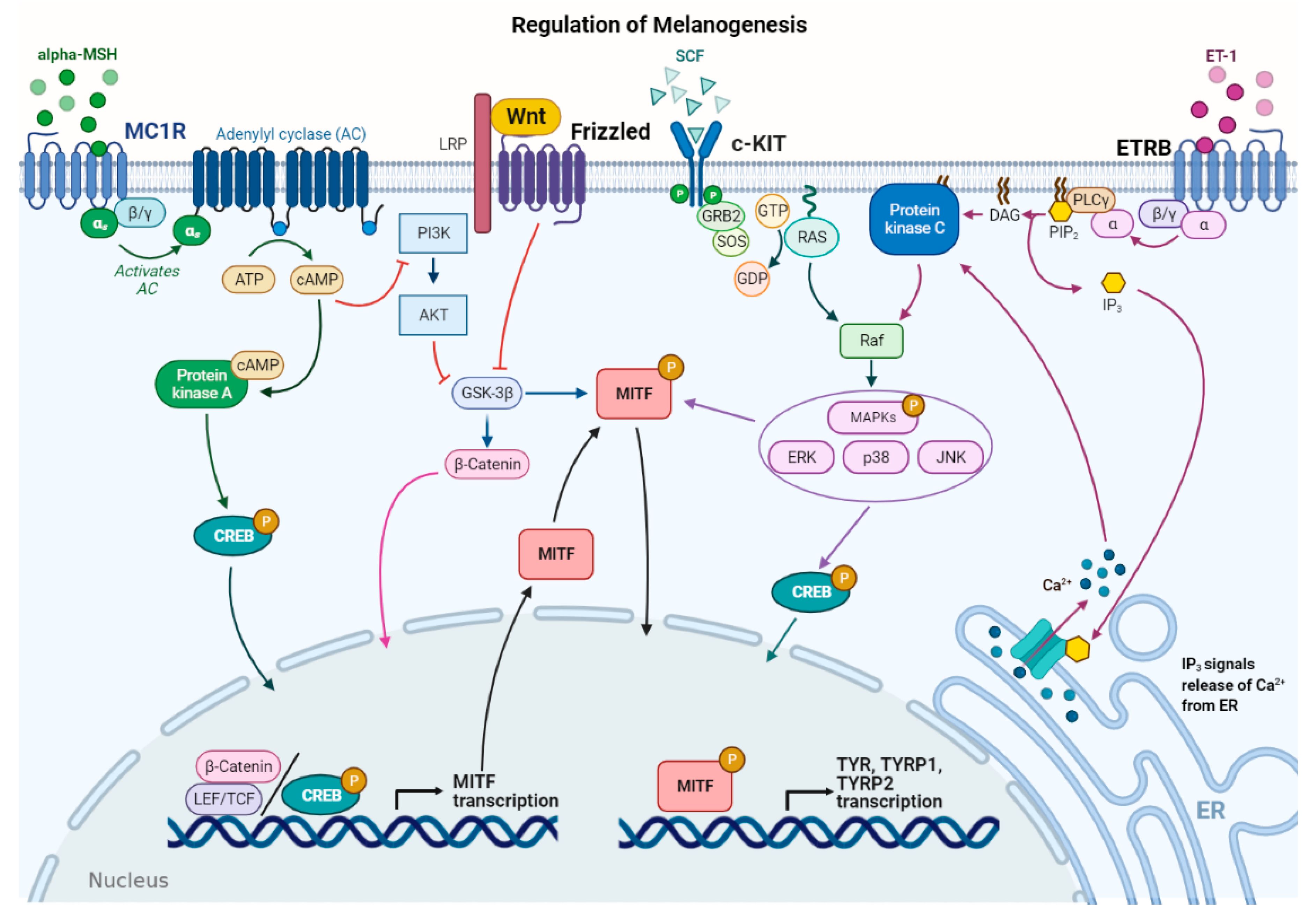
3. Melanogenesis Regulation
4. miRNA Activities and Identification
4.1. miRNA Regulating MITF
4.2. miRNAs Regulating Other Genes in Melanogenesis
| miRNA | Cell Model | Target Gene | Effect on Melanogenesis | Ref. |
|---|---|---|---|---|
| miR-434-5p | Mouse skin, human skin cell cultures | TYR | Negative | [65] |
| miR-330-5p | Melanoma cells, normal human melanocytes | TYR | Negative | [56,67] |
| miR-203 | Keratinocytes exposed to UV | Kinesin Superfamily Protein 5b | Positive | [67] |
| miR-3196 | Keratinocytes exposed to UV | Unknown target gene | Positive | [67] |
| miR-21a-5p | Human melanocytes | SOX5 | Positive | [68] |
| miR-145 | Murine melan-a melanocytes | Myo5a | Negative | [56] |
| miR-380-3p | Alpaca melanocytes | SOX6 | Negative | [56] |
| miR-200c | Normal human epidermal keratinocytes (NHEK) | SOX1 | Positive | [69] |
| miR-27a-3p | Alpaca and Mouse melanocytes | Wnt3a | Negative | [54,70] |
| miR-379 | Alpaca melanocytes | IGF1R | Negative | [71] |
| miR-143-5p | Human melanocytes | Myo5a | Negative | [72] |
| miR-143-5p | Alpaca melanocytes | TAK1 | Negative | [73] |
| miR-125b | WM266-4 human melanoma cells, MNT1 human melanoma cells | SH3BP4 | Negative | [60] |
4.3. miRNA Regulated by UV Rays
4.4. Common miRNAs in Melanogenesis and Melanomagenesis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| 3′-UTR | three prime untranslated region |
| 3D-MHE model | three-dimensional tissue culture model of human epidermis |
| ACTH | adrenocorticotrophic hormone |
| AKT | protein kinase B |
| bFGF | basic fibroblast growth factor |
| bHLH-ZIP | basic helix-loop-helix leucine zipper |
| cAMP | cyclic adenosine monophosphate |
| CDK2 | cyclin-dependent kinase 2 |
| CREB | cAMP-responsive element binding protein |
| DAG | diacyglycerol |
| DCT | dopachrome tautomerase |
| ERK1/2 | extracellular regulated protein kinases1/2 |
| ETRB | endothelin type B receptor |
| Fscn1 | fascin1 |
| FZD | frizzled receptor |
| GPCRs | G protein-coupled receptors |
| GSK3β | glycogen synthase kinase 3β |
| HGF | hepatocyte growth factor |
| IGF-1R | insulin-like growth factor receptor I |
| IL-6 | interleukin-6 |
| IP3 | inositol triphosphate |
| KGF | keratinocyte growth factor |
| L-DOPA | dihydroxyphenylalanine |
| LED | light emitting diode |
| LEF-1 | lymphoid enhancer-binding factor 1 |
| LEF-TCF | lymphoid enhancing factor-1/T-cell factor |
| lncRNA | long non-coding RNAs |
| MAPK | mitogen-activated protein kinase |
| MC1R | melanocortin 1 receptor |
| MEK1 | mitogen-activated protein kinase 1 |
| miRNAs | microRNAs |
| MITF | microphthalmia-associated transcription factor |
| MLPH | melanophilin |
| MYO5A | myosin VA |
| NHEK | normal human epidermal keratinocytes |
| NHEM | normal human epidermal melanocytes |
| NO | nitric oxide |
| OTC | skin organotypic culture |
| PAX3 | paired box family of transcription box 3 |
| PGE2 | prostaglandin E2 |
| PI3K | phosphoinositide-3 kinase |
| Pig-1 | immortalized human epidermal melanocytes |
| PIP2 | phosphatidylinositol 4,5-bisphosphate |
| PKA | protein kinase A |
| PLCγ | activated phospholipase Cγ |
| POMC | proopiomelanocortin |
| RISC | RNA-Induced Silencing Complex |
| RNA | ribonucleic acid |
| ROS | reactive oxygen species |
| RSK | ribosomal S6 kinase |
| SCF | stem cell factor |
| SH3BP4 | SRC homology 3 domain-binding protein 4 |
| SOX | sex-determining region Y-box |
| SOX1 | sex determining region Y-box 1 |
| SOX10 | sex determining region Y-box 10 |
| SOX5 | sex determining region Y-box 5 |
| SOX6 | sex determining region Y-box 6 |
| SOX9 | sex determining region Y-box |
| STAT3 | signal transducer and activator of transcription 3 |
| STTMs | short tandem target mimic |
| TAK1 | TGF-β-activated kinase 1 |
| TGF-β | transforming growth factor-β |
| TYR | tyrosinase |
| TYRP-1 | tyrosinase-related protein 1 |
| TYRP-2 | tyrosinase-related protein 2 |
| UV | ultraviolet |
| UVA | ultraviolet A |
| UVB | ultraviolet B |
| WNT3A | Wnt family member 3A |
| α-MSH | α-melanocyte stimulating hormone |
References
- D’Orazio, J.; Jarrett, S.; Amaro-Ortiz, A.; Scott, T. UV Radiation and the Skin. Int. J. Mol. Sci. 2013, 14, 2222. [Google Scholar] [CrossRef]
- Miller, A.J.; Mihm, M.C. Melanoma. N. Engl. J. Med. 2006, 355, 51–65. [Google Scholar] [CrossRef]
- Nguyen, N.T.; Fisher, D.E. MITF and UV Responses in Skin: From Pigmentation to Addiction. Pigment. Cell Melanoma Res. 2019, 32, 224–236. [Google Scholar] [CrossRef]
- Hida, T.; Kamiya, T.; Kawakami, A.; Ogino, J.; Sohma, H.; Uhara, H.; Jimbow, K. Elucidation of Melanogenesis Cascade for Identifying Pathophysiology and Therapeutic Approach of Pigmentary Disorders and Melanoma. Int. J. Mol. Sci. 2020, 21, 6129. [Google Scholar] [CrossRef] [PubMed]
- Lee, A.-Y. Recent Progress in Melasma Pathogenesis. Pigment Cell Melanoma Res. 2015, 28, 648–660. [Google Scholar] [CrossRef] [PubMed]
- Fu, C.; Chen, J.; Lu, J.; Yi, L.; Tong, X.; Kang, L.; Pei, S.; Ouyang, Y.; Jiang, L.; Ding, Y.; et al. Roles of Inflammation Factors in Melanogenesis (Review). Mol. Med. Rep. 2020, 21, 1421–1430. [Google Scholar] [CrossRef] [PubMed]
- Pillaiyar, T.; Manickam, M.; Jung, S.-H. Recent Development of Signaling Pathways Inhibitors of Melanogenesis. Cell Signal. 2017, 40, 99–115. [Google Scholar] [CrossRef]
- Madireddy, S.; Crane, J.S. Hypopigmented Macules. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2021. [Google Scholar]
- D’Mello, S.A.N.; Finlay, G.J.; Baguley, B.C.; Askarian-Amiri, M.E. Signaling Pathways in Melanogenesis. Int. J. Mol. Sci. 2016, 17, 1144. [Google Scholar] [CrossRef]
- Yamaguchi, Y.; Passeron, T.; Watabe, H.; Yasumoto, K.; Rouzaud, F.; Hoashi, T.; Hearing, V.J. The Effects of Dickkopf 1 on Gene Expression and Wnt Signaling by Melanocytes: Mechanisms Underlying Its Suppression of Melanocyte Function and Proliferation. J. Investig. Dermatol. 2007, 127, 1217–1225. [Google Scholar] [CrossRef]
- Serre, C.; Busuttil, V.; Botto, J.-M. Intrinsic and Extrinsic Regulation of Human Skin Melanogenesis and Pigmentation. Int. J. Cosmet. Sci. 2018, 40, 328–347. [Google Scholar] [CrossRef]
- Horsburgh, S.; Fullard, N.; Roger, M.; Degnan, A.; Todryk, S.; Przyborski, S.; O’Reilly, S. MicroRNAs in the Skin: Role in Development, Homoeostasis and Regeneration. Clin. Sci. 2017, 131, 1923–1940. [Google Scholar] [CrossRef] [PubMed]
- Yamada, T.; Hasegawa, S.; Iwata, Y.; Arima, M.; Kobayashi, T.; Numata, S.; Nakata, S.; Sugiura, K.; Akamatsu, H. UV Irradiation-Induced DNA Hypomethylation around WNT1 Gene: Implications for Solar Lentigines. Exp. Dermatol. 2019, 28, 723–729. [Google Scholar] [CrossRef] [PubMed]
- Jiang, L.; Huang, J.; Hu, Y.; Lei, L.; Ouyang, Y.; Long, Y.; Li, H.; Li, S.; Yang, L.; Yang, Y.; et al. Identification of the CeRNA Networks in α-MSH-Induced Melanogenesis of Melanocytes. Aging 2020, 13, 2700–2726. [Google Scholar] [CrossRef] [PubMed]
- Lanzillotti, C.; De Mattei, M.; Mazziotta, C.; Taraballi, F.; Rotondo, J.C.; Tognon, M.; Martini, F. Long Non-Coding RNAs and MicroRNAs Interplay in Osteogenic Differentiation of Mesenchymal Stem Cells. Front. Cell Dev. Biol. 2021, 9, 646032. [Google Scholar] [CrossRef] [PubMed]
- Neagu, M.; Constantin, C.; Cretoiu, S.M.; Zurac, S. MiRNAs in the Diagnosis and Prognosis of Skin Cancer. Front. Cell Dev. Biol. 2020, 8, 71. [Google Scholar] [CrossRef] [PubMed]
- Slominski, A.; Wortsman, J.; Plonka, P.M.; Schallreuter, K.U.; Paus, R.; Tobin, D.J. Hair Follicle Pigmentation. J. Investig. Dermatol. 2005, 124, 13–21. [Google Scholar] [CrossRef]
- Slominski, A.; Tobin, D.J.; Shibahara, S.; Wortsman, J. Melanin Pigmentation in Mammalian Skin and Its Hormonal Regulation. Physiol. Rev. 2004, 84, 1155–1228. [Google Scholar] [CrossRef]
- Nordlund, J.J. The Melanocyte and the Epidermal Melanin Unit: An Expanded Concept. Dermatol. Clin. 2007, 25, 271–281. [Google Scholar] [CrossRef]
- Schallreuter, K.U.; Kothari, S.; Chavan, B.; Spencer, J.D. Regulation of Melanogenesis—Controversies and New Concepts. Exp. Dermatol. 2008, 17, 395–404. [Google Scholar] [CrossRef] [PubMed]
- Land, E.J.; Ramsden, C.A.; Riley, P.A. Quinone Chemistry and Melanogenesis. Methods Enzym. 2004, 378, 88–109. [Google Scholar] [CrossRef]
- Del Bino, S.; Ito, S.; Sok, J.; Nakanishi, Y.; Bastien, P.; Wakamatsu, K.; Bernerd, F. Chemical Analysis of Constitutive Pigmentation of Human Epidermis Reveals Constant Eumelanin to Pheomelanin Ratio. Pigment Cell Melanoma Res. 2015, 28, 707–717. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.; Choi, H.; Joo, K.-M.; Kim, D.; Lee, T.R. Manassantin B Inhibits Melanosome Transport in Melanocytes by Disrupting the Melanophilin-Myosin Va Interaction. Pigment. Cell Melanoma Res. 2012, 25, 765–772. [Google Scholar] [CrossRef]
- Wakamatsu, K.; Graham, A.; Cook, D.; Thody, A.J. Characterisation of ACTH Peptides in Human Skin and Their Activation of the Melanocortin-1 Receptor. Pigment Cell Res. 1997, 10, 288–297. [Google Scholar] [CrossRef] [PubMed]
- Tagashira, H.; Miyamoto, A.; Kitamura, S.-I.; Tsubata, M.; Yamaguchi, K.; Takagaki, K.; Imokawa, G. UVB Stimulates the Expression of Endothelin B Receptor in Human Melanocytes via a Sequential Activation of the P38/MSK1/CREB/MITF Pathway Which Can Be Interrupted by a French Maritime Pine Bark Extract through a Direct Inactivation of MSK1. PLoS ONE 2015, 10, e0128678. [Google Scholar] [CrossRef] [PubMed]
- Imokawa, G. Melanocyte Activation Mechanisms and Rational Therapeutic Treatments of Solar Lentigos. Int. J. Mol. Sci. 2019, 20, 3666. [Google Scholar] [CrossRef]
- Lin, J.Y.; Fisher, D.E. Melanocyte Biology and Skin Pigmentation. Nature 2007, 445, 843–850. [Google Scholar] [CrossRef]
- Roberts, D.W.; Newton, R.A.; Beaumont, K.A.; Helen Leonard, J.; Sturm, R.A. Quantitative Analysis of MC1R Gene Expression in Human Skin Cell Cultures. Pigment Cell Res. 2006, 19, 76–89. [Google Scholar] [CrossRef]
- Mosca, S.; Cardinali, G.; Flori, E.; Briganti, S.; Bottillo, I.; Mileo, A.M.; Maresca, V. The PI3K Pathway Induced by AMSH Exerts a Negative Feedback on Melanogenesis and Contributes to the Release of Pigment. Pigment Cell Melanoma Res. 2021, 34, 72–88. [Google Scholar] [CrossRef]
- Levy, C.; Khaled, M.; Fisher, D.E. MITF: Master Regulator of Melanocyte Development and Melanoma Oncogene. Trends Mol. Med. 2006, 12, 406–414. [Google Scholar] [CrossRef]
- Vachtenheim, J.; Borovanský, J. “Transcription Physiology” of Pigment Formation in Melanocytes: Central Role of MITF. Exp. Dermatol. 2010, 19, 617–627. [Google Scholar] [CrossRef] [PubMed]
- Ohbayashi, N.; Fukuda, M. Recent Advances in Understanding the Molecular Basis of Melanogenesis in Melanocytes. F1000Research 2020, 9. [Google Scholar] [CrossRef]
- Hsiao, J.J.; Fisher, D.E. The Roles of Microphthalmia-Associated Transcription Factor and Pigmentation in Melanoma. Arch. Biochem. Biophys. 2014, 563, 28–34. [Google Scholar] [CrossRef]
- Khaled, M.; Larribere, L.; Bille, K.; Aberdam, E.; Ortonne, J.-P.; Ballotti, R.; Bertolotto, C. Glycogen Synthase Kinase 3beta Is Activated by CAMP and Plays an Active Role in the Regulation of Melanogenesis. J. Biol. Chem. 2002, 277, 33690–33697. [Google Scholar] [CrossRef]
- Saha, B.; Singh, S.K.; Sarkar, C.; Bera, R.; Ratha, J.; Tobin, D.J.; Bhadra, R. Activation of the Mitf Promoter by Lipid-Stimulated Activation of P38-Stress Signalling to CREB. Pigment Cell Res. 2006, 19, 595–605. [Google Scholar] [CrossRef]
- Wang, Y.; Viennet, C.; Robin, S.; Berthon, J.-Y.; He, L.; Humbert, P. Precise Role of Dermal Fibroblasts on Melanocyte Pigmentation. J. Dermatol. Sci 2017, 88, 159–166. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, A.K.; Funasaka, Y.; Slominski, A.; Ermak, G.; Hwang, J.; Pawelek, J.M.; Ichihashi, M. Production and Release of Proopiomelanocortin (POMC) Derived Peptides by Human Melanocytes and Keratinocytes in Culture: Regulation by Ultraviolet B. Biochim. Biophys. Acta 1996, 1313, 130–138. [Google Scholar] [CrossRef]
- Cui, R.; Widlund, H.R.; Feige, E.; Lin, J.Y.; Wilensky, D.L.; Igras, V.E.; D’Orazio, J.; Fung, C.Y.; Schanbacher, C.F.; Granter, S.R.; et al. Central Role of P53 in the Suntan Response and Pathologic Hyperpigmentation. Cell 2007, 128, 853–864. [Google Scholar] [CrossRef]
- Lee, R.C.; Feinbaum, R.L.; Ambros, V. The C. Elegans Heterochronic Gene Lin-4 Encodes Small RNAs with Antisense Complementarity to Lin-14. Cell 1993, 75, 843–854. [Google Scholar] [CrossRef]
- Finotti, A.; Fabbri, E.; Lampronti, I.; Gasparello, J.; Borgatti, M.; Gambari, R. MicroRNAs and Long Non-Coding RNAs in Genetic Diseases. Mol. Diagn. Ther. 2019, 23, 155–171. [Google Scholar] [CrossRef]
- Rotondo, J.C.; Mazzoni, E.; Bononi, I.; Tognon, M.; Martini, F. Association Between Simian Virus 40 and Human Tumors. Front. Oncol. 2019, 9, 670. [Google Scholar] [CrossRef]
- Pradhan, A.K.; Bhoopathi, P.; Talukdar, S.; Scheunemann, D.; Sarkar, D.; Cavenee, W.K.; Das, S.K.; Emdad, L.; Fisher, P.B. MDA-7/IL-24 Regulates the MiRNA Processing Enzyme DICER through Downregulation of MITF. Proc. Natl. Acad. Sci. USA 2019, 116, 5687–5692. [Google Scholar] [CrossRef]
- Correia de Sousa, M.; Gjorgjieva, M.; Dolicka, D.; Sobolewski, C.; Foti, M. Deciphering MiRNAs’ Action through MiRNA Editing. Int. J. Mol. Sci. 2019, 20, 6249. [Google Scholar] [CrossRef] [PubMed]
- Huang, W. MicroRNAs: Biomarkers, Diagnostics, and Therapeutics. Methods Mol. Biol. 2017, 1617, 57–67. [Google Scholar] [CrossRef]
- Mazziotta, C.; Lanzillotti, C.; Iaquinta, M.R.; Taraballi, F.; Torreggiani, E.; Rotondo, J.C.; Otòn-Gonzalez, L.; Mazzoni, E.; Frontini, F.; Bononi, I.; et al. MicroRNAs Modulate Signaling Pathways in Osteogenic Differentiation of Mesenchymal Stem Cells. Int. J. Mol. Sci. 2021, 22, 2362. [Google Scholar] [CrossRef] [PubMed]
- Iaquinta, M.R.; Lanzillotti, C.; Mazziotta, C.; Bononi, I.; Frontini, F.; Mazzoni, E.; Oton-Gonzalez, L.; Rotondo, J.C.; Torreggiani, E.; Tognon, M.; et al. The Role of MicroRNAs in the Osteogenic and Chondrogenic Differentiation of Mesenchymal Stem Cells and Bone Pathologies. Theranostics 2021, in press. [Google Scholar] [CrossRef] [PubMed]
- Nedaeinia, R.; Manian, M.; Jazayeri, M.H.; Ranjbar, M.; Salehi, R.; Sharifi, M.; Mohaghegh, F.; Goli, M.; Jahednia, S.H.; Avan, A.; et al. Circulating Exosomes and Exosomal MicroRNAs as Biomarkers in Gastrointestinal Cancer. Cancer Gene. Ther. 2017, 24, 48–56. [Google Scholar] [CrossRef]
- O’Brien, J.; Hayder, H.; Zayed, Y.; Peng, C. Overview of MicroRNA Biogenesis, Mechanisms of Actions, and Circulation. Front. Endocrinol. 2018, 9, 402. [Google Scholar] [CrossRef]
- Wawrzyniak, O.; Zarębska, Ż.; Rolle, K.; Gotz-Więckowska, A. Circular and Long Non-Coding RNAs and Their Role in Ophthalmologic Diseases. Acta Biochim. Pol. 2018, 65, 497–508. [Google Scholar] [CrossRef]
- Grillone, K.; Riillo, C.; Scionti, F.; Rocca, R.; Tradigo, G.; Guzzi, P.H.; Alcaro, S.; Di Martino, M.T.; Tagliaferri, P.; Tassone, P. Non-Coding RNAs in Cancer: Platforms and Strategies for Investigating the Genomic “Dark Matter”. J. Exp. Clin. Cancer Res. 2020, 39, 117. [Google Scholar] [CrossRef]
- Mione, M.; Bosserhoff, A. MicroRNAs in Melanocyte and Melanoma Biology. Pigment Cell Melanoma Res. 2015, 28, 340–354. [Google Scholar] [CrossRef]
- Itoh, T.; Fukatani, K.; Nakashima, A.; Suzuki, K. MicroRNA-141-3p and MicroRNA-200a-3p Regulate α-Melanocyte Stimulating Hormone-Stimulated Melanogenesis by Directly Targeting Microphthalmia-Associated Transcription Factor. Sci. Rep. 2020, 10, 2149. [Google Scholar] [CrossRef]
- Chen, T.; Zhao, B.; Liu, Y.; Wang, R.; Yang, Y.; Yang, L.; Dong, C. MITF-M Regulates Melanogenesis in Mouse Melanocytes. J. Dermatol. Sci. 2018, 90, 253–262. [Google Scholar] [CrossRef]
- Tian, X.; Jiang, J.; Fan, R.; Wang, H.; Meng, X.; He, X.; He, J.; Li, H.; Geng, J.; Yu, X.; et al. Identification and Characterization of MicroRNAs in White and Brown Alpaca Skin. BMC Genom. 2012, 13, 555. [Google Scholar] [CrossRef]
- Zhu, Z.; He, J.; Jia, X.; Jiang, J.; Bai, R.; Yu, X.; Lv, L.; Fan, R.; He, X.; Geng, J.; et al. MicroRNA-25 Functions in Regulation of Pigmentation by Targeting the Transcription Factor MITF in Alpaca (Lama Pacos) Skin Melanocytes. Domest. Anim. Endocrinol. 2010, 38, 200–209. [Google Scholar] [CrossRef]
- Liu, X.; Du, B.; Zhang, P.; Zhang, J.; Zhu, Z.; Liu, B.; Fan, R. MiR-380-3p Regulates Melanogenesis by Targeting SOX6 in Melanocytes from Alpacas (Vicugna Pacos). BMC Genom. 2019, 20, 962. [Google Scholar] [CrossRef]
- Dong, C.; Wang, H.; Xue, L.; Dong, Y.; Yang, L.; Fan, R.; Yu, X.; Tian, X.; Ma, S.; Smith, G.W. Coat Color Determination by MiR-137 Mediated down-Regulation of Microphthalmia-Associated Transcription Factor in a Mouse Model. RNA 2012, 18, 1679–1686. [Google Scholar] [CrossRef]
- Bemis, L.T.; Chen, R.; Amato, C.M.; Classen, E.H.; Robinson, S.E.; Coffey, D.G.; Erickson, P.F.; Shellman, Y.G.; Robinson, W.A. MicroRNA-137 Targets Microphthalmia-Associated Transcription Factor in Melanoma Cell Lines. Cancer Res. 2008, 68, 1362–1368. [Google Scholar] [CrossRef] [PubMed]
- Valadi, H.; Ekström, K.; Bossios, A.; Sjöstrand, M.; Lee, J.J.; Lötvall, J.O. Exosome-Mediated Transfer of MRNAs and MicroRNAs Is a Novel Mechanism of Genetic Exchange between Cells. Nat. Cell Biol. 2007, 9, 654–659. [Google Scholar] [CrossRef]
- Kim, N.-H.; Choi, S.-H.; Kim, C.-H.; Lee, C.H.; Lee, T.R.; Lee, A.-Y. Reduced MiR-675 in Exosome in H19 RNA-Related Melanogenesis via MITF as a Direct Target. J. Investig. Dermatol. 2014, 134, 1075–1082. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Zhang, J.; Wang, W.; Cheung, F.W.; Lu, Y.; Ng, C.; Kung, H.; Liu, W. MicroRNA-218 Inhibits Melanogenesis by Directly Suppressing Microphthalmia-Associated Transcription Factor Expression. RNA Biol. 2014, 11, 732–741. [Google Scholar] [CrossRef] [PubMed]
- Du, B.; Liu, X.; Khan, A.; Wan, S.; Guo, X.; Xue, J.; Fan, R. MiRNA-183∼96∼182 Regulates Melanogenesis, Cell Proliferation and Migration in B16 Cells. Acta Histochem. 2020, 122, 151508. [Google Scholar] [CrossRef]
- Yang, Y.; Wei, X.; Bai, J.; Huang, M.; Hao, T.; Hao, Y.; Wang, Y.; Li, C. MicroRNA-340 Is Involved in Ultraviolet B-Induced Pigmentation by Regulating the MITF/TYRP1 Axis. J. Int. Med. Res. 2020, 48, 300060520971510. [Google Scholar] [CrossRef]
- Goswami, S.; Tarapore, R.S.; Poenitzsch Strong, A.M.; TeSlaa, J.J.; Grinblat, Y.; Setaluri, V.; Spiegelman, V.S. MicroRNA-340-Mediated Degradation of Microphthalmia-Associated Transcription Factor (MITF) MRNA Is Inhibited by Coding Region Determinant-Binding Protein (CRD-BP). J. Biol. Chem. 2015, 290, 384–395. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.T.; Chen, J.S.; Chang, D.C.; Lin, S.-L. Mir-434-5p Mediates Skin Whitening and Lightening. Clin. Cosmet. Investig. Dermatol. 2008, 1, 19–35. [Google Scholar] [CrossRef][Green Version]
- Rambow, F.; Bechadergue, A.; Saintigny, G.; Morizot, F.; Mahé, C.; Larue, L. MiR-330-5p Targets Tyrosinase and Induces Depigmentation. J. Investig. Dermatol. 2014, 134, 2846–2849. [Google Scholar] [CrossRef]
- Lo Cicero, A.; Delevoye, C.; Gilles-Marsens, F.; Loew, D.; Dingli, F.; Guéré, C.; André, N.; Vié, K.; van Niel, G.; Raposo, G. Exosomes Released by Keratinocytes Modulate Melanocyte Pigmentation. Nat. Commun. 2015, 6, 7506. [Google Scholar] [CrossRef]
- Aguennouz, M.; Guarneri, F.; Oteri, R.; Polito, F.; Giuffrida, R.; Cannavò, S.P. Serum Levels of MiRNA-21-5p in Vitiligo Patients and Effects of MiRNA-21-5p on SOX5, Beta-Catenin, CDK2 and MITF Protein Expression in Normal Human Melanocytes. J. Dermatol. Sci. 2021, 101, 22–29. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Wang, D.; Wang, X.; Mao, Y.; Xu, Z.; Sun, Y.; Mei, X.; Song, J.; Shi, W. Down-Regulation of Exosomal MiR-200c Derived from Keratinocytes in Vitiligo Lesions Suppresses Melanogenesis. J. Cell Mol. Med. 2020, 24, 12164–12175. [Google Scholar] [CrossRef]
- Zhao, Y.; Wang, P.; Meng, J.; Ji, Y.; Xu, D.; Chen, T.; Fan, R.; Yu, X.; Yao, J.; Dong, C. MicroRNA-27a-3p Inhibits Melanogenesis in Mouse Skin Melanocytes by Targeting Wnt3a. Int. J. Mol. Sci. 2015, 16, 921. [Google Scholar] [CrossRef]
- Liu, B.; Zhang, J.; Hu, S.; Qi, S.; Jia, Q.; Yang, W.; Yang, S.; Ji, K.; Liu, X.; Dong, C.; et al. MicroRNA-379 Mediates Pigmentation, Migration and Proliferation of Melanocytes by Targeting the Insulin-like Growth Factor 1 Receptor. Exp. Dermatol. 2020, 29, 467–476. [Google Scholar] [CrossRef] [PubMed]
- Alves, C.P.; Yokoyama, S.; Goedert, L.; Pontes, C.L.S.; Sousa, J.F.; Fisher, D.E.; Espreafico, E.M. MYO5A Gene Is a Target of MITF in Melanocytes. J. Investig. Dermatol. 2017, 137, 985–989. [Google Scholar] [CrossRef] [PubMed]
- Ji, K.; Zhang, P.; Zhang, J.; Fan, R.; Liu, Y.; Yang, S.; Hu, S.; Liu, X.; Dong, C. MicroRNA 143-5p Regulates Alpaca Melanocyte Migration, Proliferation and Melanogenesis. Exp. Dermatol. 2018, 27, 166–171. [Google Scholar] [CrossRef]
- Dynoodt, P.; Mestdagh, P.; Van Peer, G.; Vandesompele, J.; Goossens, K.; Peelman, L.J.; Geusens, B.; Speeckaert, R.M.; Lambert, J.L.W.; Van Gele, M.J.L. Identification of MiR-145 as a Key Regulator of the Pigmentary Process. J. Investig. Dermatol. 2013, 133, 201–209. [Google Scholar] [CrossRef]
- Harris, M.L.; Baxter, L.L.; Loftus, S.K.; Pavan, W.J. Sox Proteins in Melanocyte Development and Melanoma. Pigment Cell Melanoma Res. 2010, 23, 496–513. [Google Scholar] [CrossRef]
- Van Gele, M.; Geusens, B.; Schmitt, A.-M.; Aguilar, L.; Lambert, J. Knockdown of Myosin Va Isoforms by RNAi as a Tool to Block Melanosome Transport in Primary Human Melanocytes. J. Investig. Dermatol. 2008, 128, 2474–2484. [Google Scholar] [CrossRef] [PubMed]
- Qi, S.; Liu, B.; Zhang, J.; Liu, X.; Dong, C.; Fan, R. Knockdown of MicroRNA-143-5p by STTM Technology Affects Eumelanin and Pheomelanin Production in Melanocytes. Mol. Med. Rep. 2019, 20, 2649–2656. [Google Scholar] [CrossRef]
- Kim, K.-H.; Bin, B.-H.; Kim, J.; Dong, S.E.; Park, P.J.; Choi, H.; Kim, B.J.; Yu, S.J.; Kang, H.; Kang, H.H.; et al. Novel Inhibitory Function of MiR-125b in Melanogenesis. Pigment Cell Melanoma Res. 2014, 27, 140–144. [Google Scholar] [CrossRef]
- Jayanthy, A.; Setaluri, V. Light-Regulated MicroRNAs. Photochem. Photobiol. 2015, 91, 163–172. [Google Scholar] [CrossRef] [PubMed]
- Guo, L.; Huang, Z.-X.; Chen, X.-W.; Deng, Q.-K.; Yan, W.; Zhou, M.-J.; Ou, C.-S.; Ding, Z.-H. Differential Expression Profiles of MicroRNAs in NIH3T3 Cells in Response to UVB Irradiation. Photochem. Photobiol. 2009, 85, 765–773. [Google Scholar] [CrossRef] [PubMed]
- Jian, Q.; An, Q.; Zhu, D.; Hui, K.; Liu, Y.; Chi, S.; Li, C. MicroRNA 340 Is Involved in UVB-Induced Dendrite Formation through the Regulation of RhoA Expression in Melanocytes. Mol. Cell Biol. 2014, 34, 3407–3420. [Google Scholar] [CrossRef] [PubMed]
- Jiang, S.; Yu, X.; Dong, C. MiR-137 Affects Melanin Synthesis in Mouse Melanocyte by Repressing the Expression of c-Kit and Tyrp2 in SCF/c-Kit Signaling Pathway. Biosci. Biotechnol. Biochem. 2016, 80, 2115–2121. [Google Scholar] [CrossRef]
- Cha, H.J.; Kim, O.-Y.; Lee, G.T.; Lee, K.S.; Lee, J.H.; Park, I.-C.; Lee, S.-J.; Kim, Y.R.; Ahn, K.J.; An, I.-S.; et al. Identification of Ultraviolet B Radiation-induced MicroRNAs in Normal Human Dermal Papilla Cells. Mol. Med. Rep. 2014, 10, 1663–1670. [Google Scholar] [CrossRef]
- Melnik, B.C.; John, S.M.; Carrera-Bastos, P.; Schmitz, G. MicroRNA-21-Enriched Exosomes as Epigenetic Regulators in Melanomagenesis and Melanoma Progression: The Impact of Western Lifestyle Factors. Cancers 2020, 12, 2111. [Google Scholar] [CrossRef]
- Wang, P.; Zhao, Y.; Fan, R.; Chen, T.; Dong, C. MicroRNA-21a-5p Functions on the Regulation of Melanogenesis by Targeting Sox5 in Mouse Skin Melanocytes. Int. J. Mol. Sci. 2016, 17, 959. [Google Scholar] [CrossRef] [PubMed]
- Lin, K.-Y.; Chen, C.-M.; Lu, C.-Y.; Cheng, C.-Y.; Wu, Y.-H. Regulation of MiR-21 Expression in Human Melanoma via UV-Ray-Induced Melanin Pigmentation. Environ. Toxicol. 2017, 32, 2064–2069. [Google Scholar] [CrossRef]
- Wäster, P.; Eriksson, I.; Vainikka, L.; Öllinger, K. Extracellular Vesicles Released by Melanocytes after UVA Irradiation Promote Intercellular Signaling via MiR21. Pigment Cell Melanoma Res. 2020, 33, 542–555. [Google Scholar] [CrossRef]
- Zhou, B.; Xu, Y.; Permatasari, F.; Liu, W.; Li, W.; Guo, X.; Huang, Q.; Guo, Z.; Luo, D. Characterization of the MiRNA Profile in UVB-Irradiated Normal Human Keratinocytes. Exp. Dermatol. 2012, 21, 317–319. [Google Scholar] [CrossRef] [PubMed]
- Kraemer, A.; Chen, I.-P.; Henning, S.; Faust, A.; Volkmer, B.; Atkinson, M.J.; Moertl, S.; Greinert, R. UVA and UVB Irradiation Differentially Regulate MicroRNA Expression in Human Primary Keratinocytes. PLoS ONE 2013, 8, e83392. [Google Scholar] [CrossRef]
- Jin, S.; Chen, L.; Xu, Z.; Xing, X.; Zhang, C.; Xiang, L. 585 Nm Light-Emitting Diodes Inhibit Melanogenesis through Upregulating H19/MiR-675 Axis in LEDs-Irradiated Keratinocytes by Paracrine Effect. J. Dermatol. Sci. 2020, 98, 102–108. [Google Scholar] [CrossRef] [PubMed]
- Sabarimurugan, S.; Royam, M.M.; Das, A.; Das, S.; Gothandam, K.M.; . Jayaraj, R. Systematic Review and Meta-Analysis of the Prognostic Significance of MiRNAs in Melanoma Patients. Mol. Diagn. Ther. 2018, 22, 653–669. [Google Scholar] [CrossRef]
- Möller, K.; Sigurbjornsdottir, S.; Arnthorsson, A.O.; Pogenberg, V.; Dilshat, R.; Fock, V.; Brynjolfsdottir, S.H.; Bindesboll, C.; Bessadottir, M.; Ogmundsdottir, H.M.; et al. MITF Has a Central Role in Regulating Starvation-Induced Autophagy in Melanoma. Sci. Rep. 2019, 9, 1055. [Google Scholar] [CrossRef]
- Varrone, F.; Caputo, E. The MiRNAs Role in Melanoma and in Its Resistance to Therapy. Int. J. Mol. Sci. 2020, 21, 878. [Google Scholar] [CrossRef]
- Tang, H.; Xu, X.; Xiao, W.; Liao, Y.; Xiao, X.; Li, L.; Li, K.; Jia, X.; Feng, H. Silencing of MicroRNA-27a Facilitates Autophagy and Apoptosis of Melanoma Cells through the Activation of the SYK-dependent MTOR Signaling Pathway. J. Cell. Biochem. 2019, 120, 13262–13274. [Google Scholar] [CrossRef]
- Melnik, B.C. MiR-21: An Environmental Driver of Malignant Melanoma? J. Transl. Med. 2015, 13. [Google Scholar] [CrossRef]
- Takano, K.; Hachiya, A.; Murase, D.; Tanabe, H.; Kasamatsu, S.; Takahashi, Y.; Moriwaki, S.; Hase, T. Quantitative Changes in the Secretion of Exosomes from Keratinocytes Homeostatically Regulate Skin Pigmentation in a Paracrine Manner. J. Dermatol. 2020, 47, 265–276. [Google Scholar] [CrossRef]
- Liu, Y.; Xue, L.; Gao, H.; Chang, L.; Yu, X.; Zhu, Z.; He, X.; Geng, J.; Dong, Y.; Li, H.; et al. Exosomal MiRNA Derived from Keratinocytes Regulates Pigmentation in Melanocytes. J. Dermatol. Sci. 2019, 93, 159–167. [Google Scholar] [CrossRef] [PubMed]
- Brahmbhatt, H.D.; Gupta, R.; Gupta, A.; Rastogi, S.; Misri, R.; Mobeen, A.; Ghosh, A.; Kothari, P.; Sitaniya, S.; Scaria, V.; et al. The Long Noncoding RNA MALAT1 Suppresses MiR-211 to Confer Protection from Ultraviolet-Mediated DNA Damage in Vitiligo Epidermis by Upregulating Sirtuin 1. Br. J. Dermatol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Randall, M.J.; Jüngel, A.; Rimann, M.; Wuertz-Kozak, K. Advances in the Biofabrication of 3D Skin in Vitro: Healthy and Pathological Models. Front. Bioeng. Biotechnol. 2018, 6, 154. [Google Scholar] [CrossRef] [PubMed]
- Gerloff, D.; Sunderkötter, C.; Wohlrab, J. Importance of MicroRNAs in Skin Oncogenesis and Their Suitability as Agents and Targets for Topical Therapy. Skin Pharmacol. Physiol. 2020, 33, 270–279. [Google Scholar] [CrossRef] [PubMed]
| miRNA | Cell Model | Target Gene | Effect on Melanogenesis | Ref. |
|---|---|---|---|---|
| miR-25 | Alpaca melanocytes | MITF | Negative | [56] |
| miR508-3p | Alpaca melanocytes | MITF | Negative | [56] |
| miR-137 | Alpaca melanocytes | MITF | Negative | [58] |
| miR-675 | Melanocytes of melasma patients, keratinocytes of melasma | MITF | Negative | [5,59,60] |
| miR-218 | Melan-a murine melanocytes, human skin OTC | MITF | Negative | [61] |
| miR-183 | B16 melanoma cells | MITF | Negative | [62] |
| miR-340 | Human epidermal melanocytes (Pig-I) | MITF | Negative | [63,64] |
| miR-200a-3p | B16-4A5 melanoma cells | MITF | Negative | [52] |
| miR-148a-3p | B16-4A5 melanoma cells | MITF | Negative | [52] |
| miR-141-3p | B16-4A5 melanoma cells | MITF | Negative | [52] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hushcha, Y.; Blo, I.; Oton-Gonzalez, L.; Mauro, G.D.; Martini, F.; Tognon, M.; Mattei, M.D. microRNAs in the Regulation of Melanogenesis. Int. J. Mol. Sci. 2021, 22, 6104. https://doi.org/10.3390/ijms22116104
Hushcha Y, Blo I, Oton-Gonzalez L, Mauro GD, Martini F, Tognon M, Mattei MD. microRNAs in the Regulation of Melanogenesis. International Journal of Molecular Sciences. 2021; 22(11):6104. https://doi.org/10.3390/ijms22116104
Chicago/Turabian StyleHushcha, Yekatsiaryna, Irene Blo, Lucia Oton-Gonzalez, Giulia Di Mauro, Fernanda Martini, Mauro Tognon, and Monica De Mattei. 2021. "microRNAs in the Regulation of Melanogenesis" International Journal of Molecular Sciences 22, no. 11: 6104. https://doi.org/10.3390/ijms22116104
APA StyleHushcha, Y., Blo, I., Oton-Gonzalez, L., Mauro, G. D., Martini, F., Tognon, M., & Mattei, M. D. (2021). microRNAs in the Regulation of Melanogenesis. International Journal of Molecular Sciences, 22(11), 6104. https://doi.org/10.3390/ijms22116104








