The BAG2 and BAG6 Genes Are Involved in Multiple Abiotic Stress Tolerances in Arabidopsis Thaliana
Abstract
1. Introduction
2. Results
2.1. BAG2 and BAG6 Expression during Arabidopsis Development
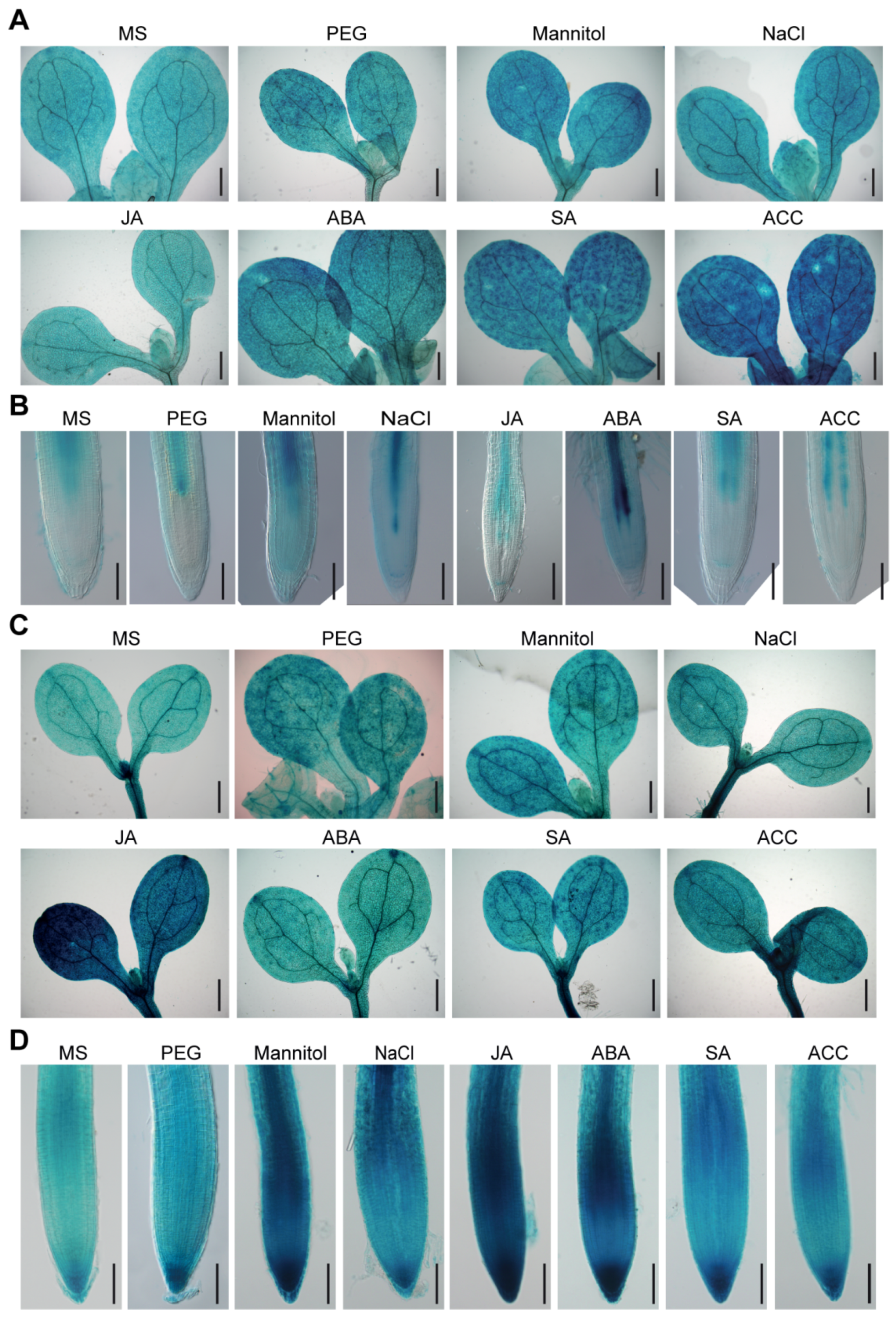
2.2. BAG2 and BAG6 Expression Responses to Abiotic Stress and Plant Hormones
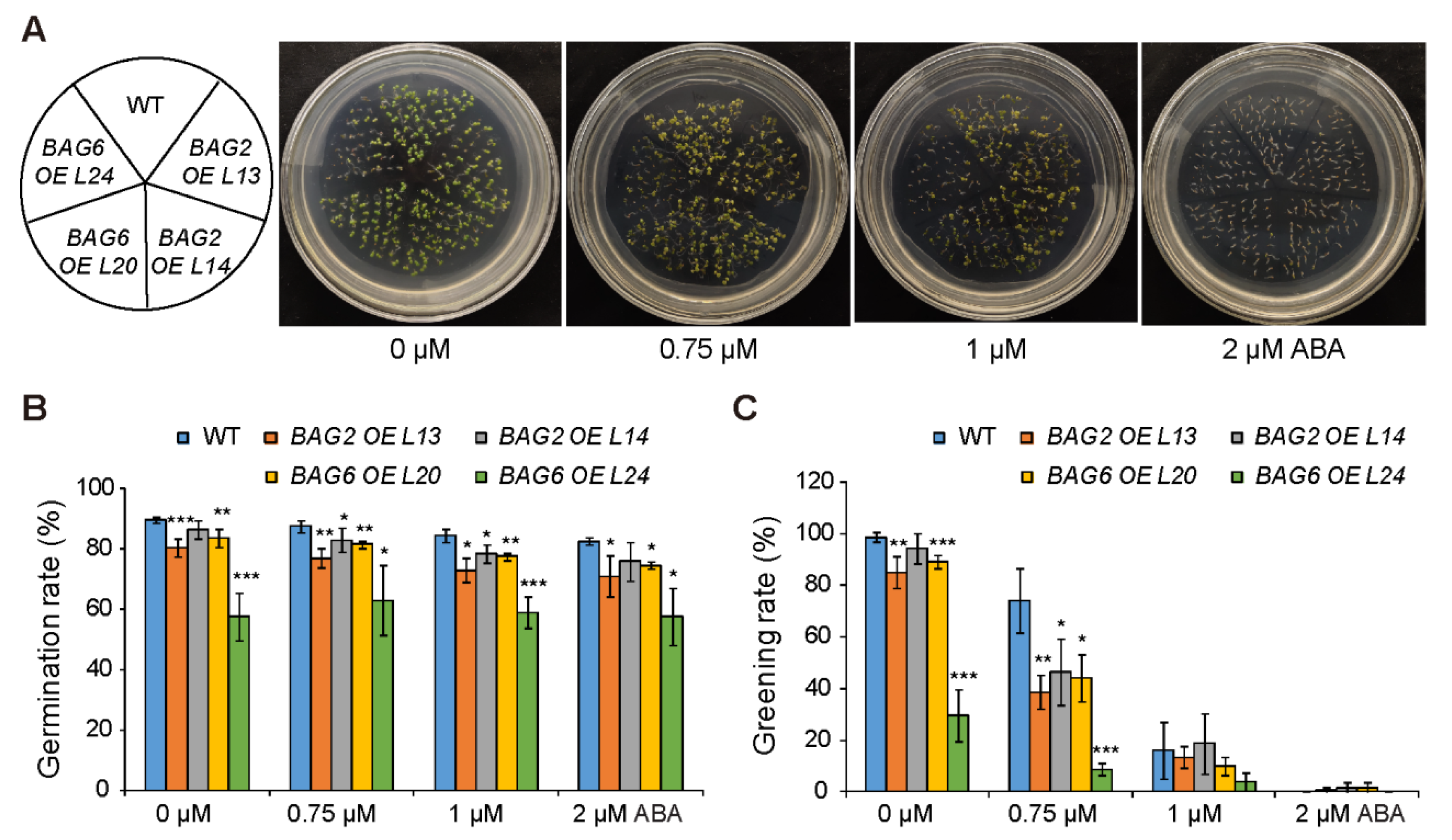
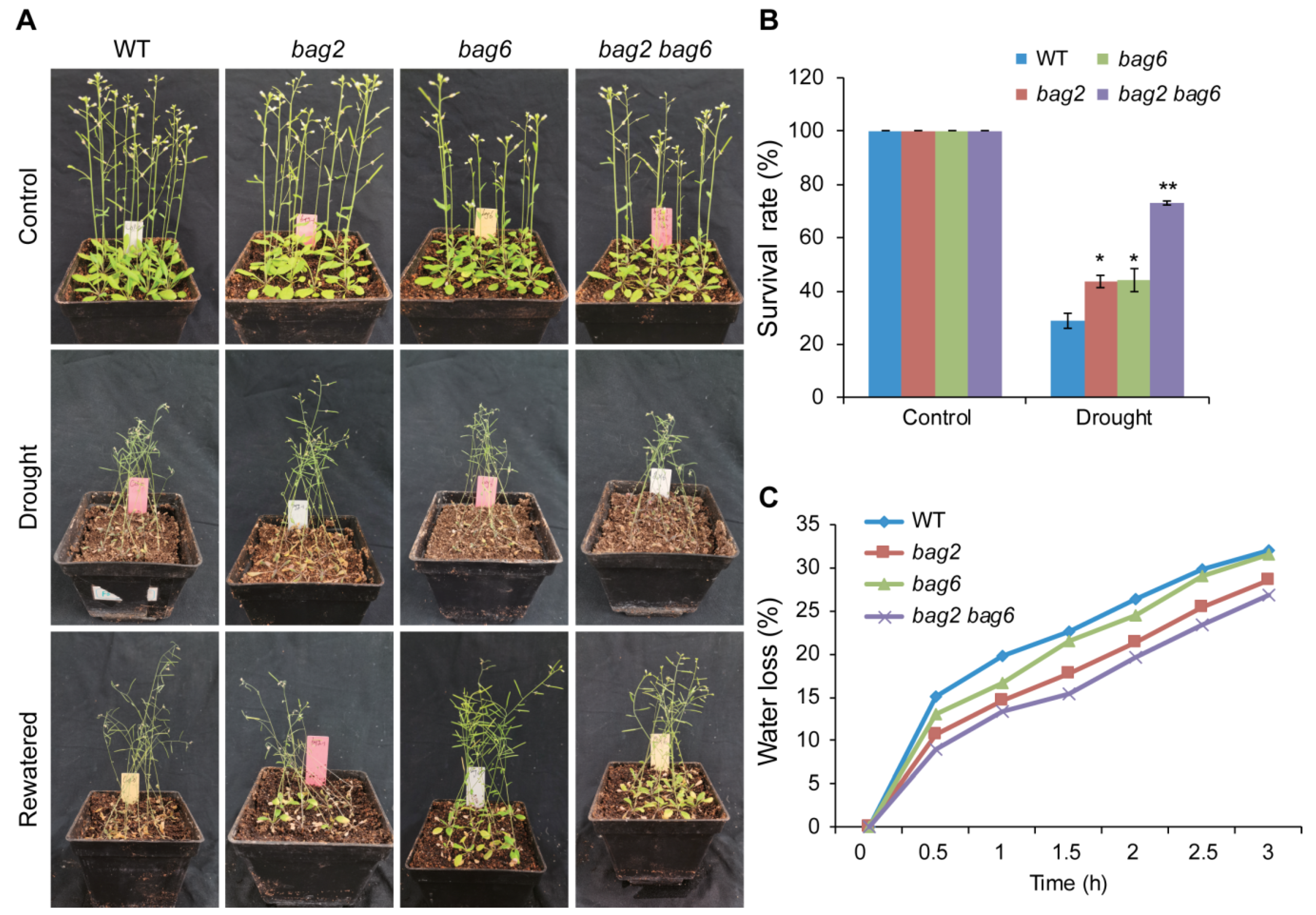
2.3. BAG2 and BAG6 Genes Are Involved in Arabidopsis Responses to ABA and Drought Treatment
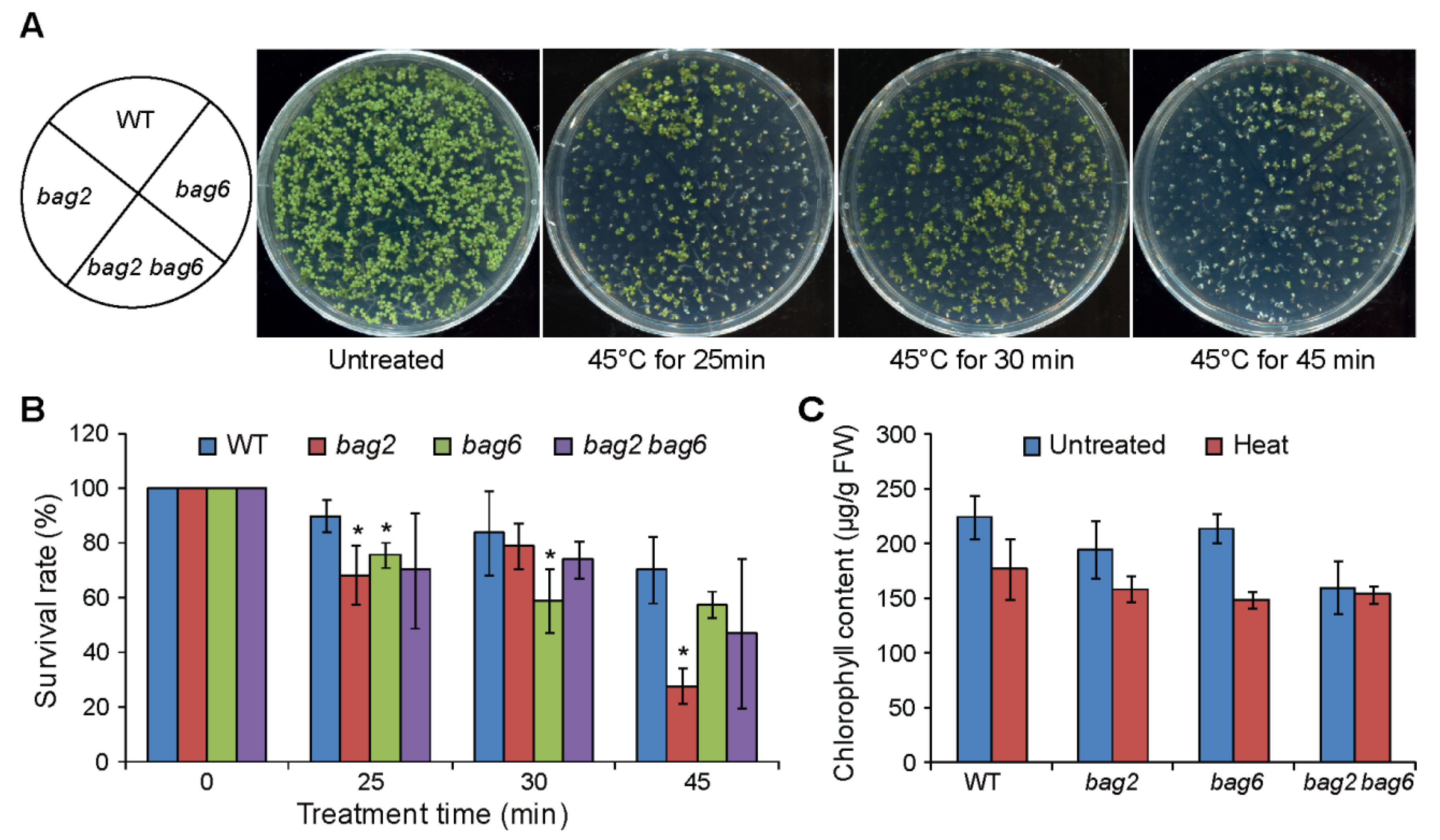
2.4. Mutation of BAG2 and BAG6 in Arabidopsis Compromises Tolerance to Heat Stress
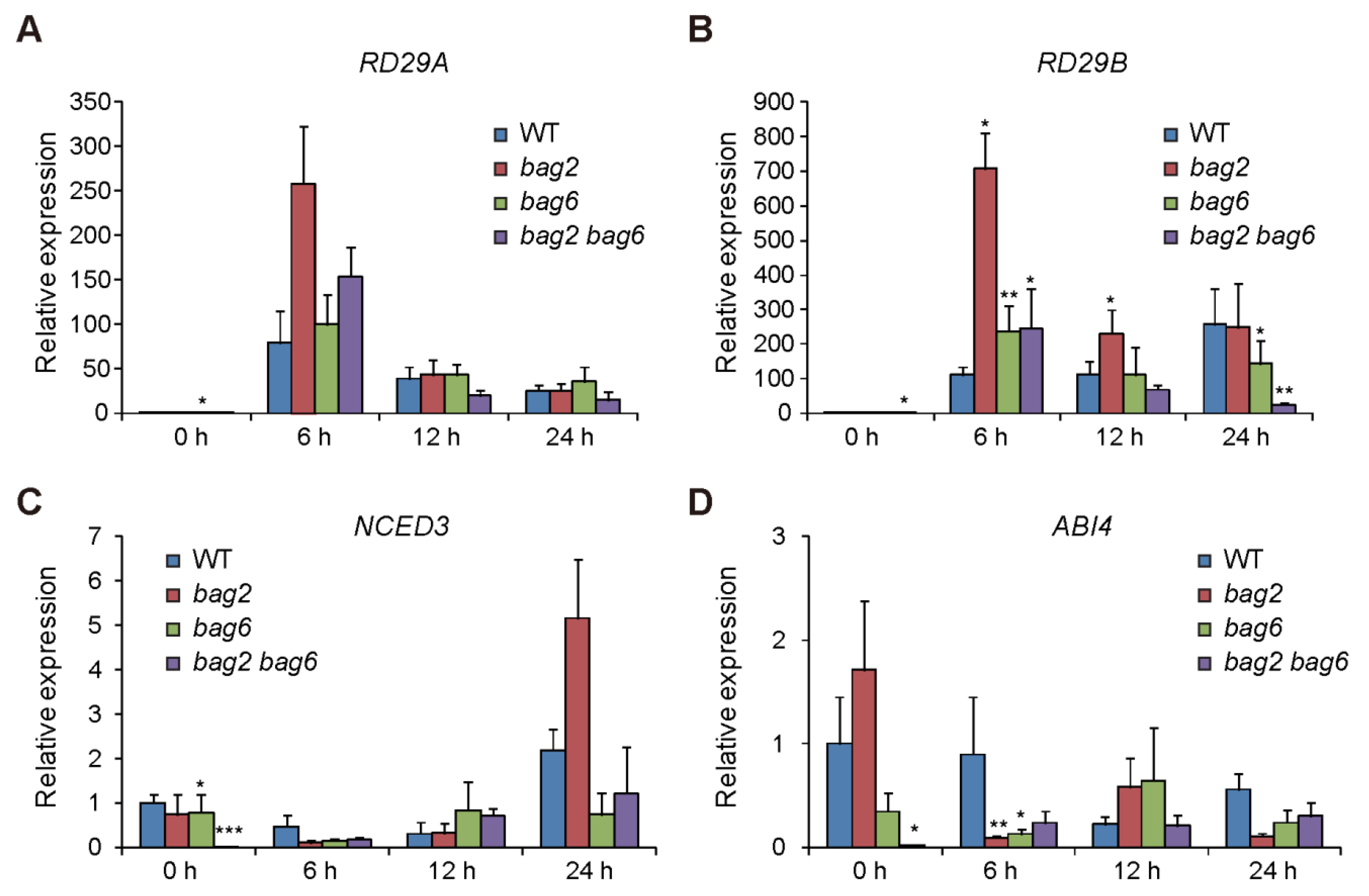
2.5. Stress- and ABA-Related Genes Are Differentially Regulated in the bag2, bag6, and bag2 bag6 Mutants Compared to WT
2.6. ROS Accumulation in WT and Mutant Plants
3. Discussion
4. Materials and Methods
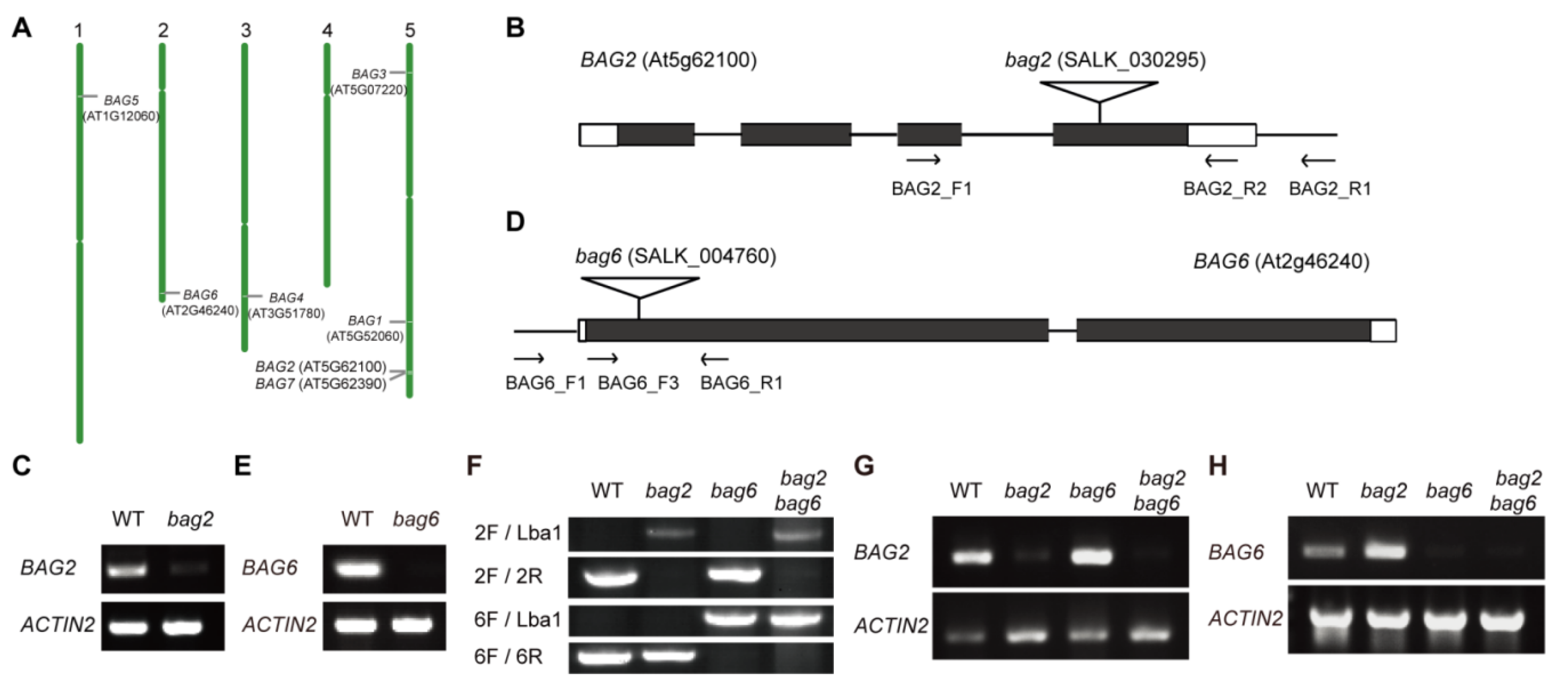
4.1. Plant Materials and Growth Conditions
4.2. Vector Construction and Generation of Transgenic Plants
4.3. RT-PCR Analysis
4.4. GUS Histochemical Staining
4.5. Drought and ABA Treatments
4.6. Heat Treatment and Measurement of Chlorophyll Content
4.7. DAB and NBT Staining
4.8. Hormone Treatment
4.9. RT-qPCR
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| BAG | Bcl-2 associated athanogene |
| ABA | Abscisic acid |
| ACC | 1-aminocyclopropane-1-carboxylic acid |
| JA | Jasmonate |
| SA | Salicylic acid |
| GA | Gibberellic acid |
| WT | Wild type |
| GUS | β-glucuronidase |
| PCD | programmed cell death |
| ROS | reactive oxygen species |
References
- Takayama, S.; Sato, T.; Krajewski, S.; Kochel, K.; Irie, S.; Millan, J.A.; Reed, J.C. Cloning and functional analysis of BAG-1: A novel Bcl-2-binding protein with anti-cell death activity. Cell 1995, 80, 279–284. [Google Scholar] [CrossRef]
- Antoku, K.; Maser, R.S.; Scully, W.J., Jr.; Delach, S.M.; Johnson, D.E. Isolation of Bcl-2 binding proteins that exhibit homology with BAG-1 and suppressor of death domains protein. Biochem. Biophys. Res. Commun. 2001, 286, 1003–1010. [Google Scholar] [CrossRef] [PubMed]
- Doong, H.; Vrailas, A.; Kohn, E.C. What’s in the ‘BAG’?—A functional domain analysis of the BAG-family proteins. Cancer Lett. 2002, 188, 25–32. [Google Scholar] [CrossRef]
- Fang, S.; Li, L.; Cui, B.; Men, S.; Shen, Y.; Yang, X. Structural insight into plant programmed cell death mediated by BAG proteins in Arabidopsis thaliana. Acta Crystallogr. D Biol. Crystallogr. 2013, 69, 934–945. [Google Scholar] [CrossRef]
- Takayama, S.; Bimston, D.N.; Matsuzawa, S.; Freeman, B.C.; Aime-Sempe, C.; Xie, Z.; Morimoto, R.I.; Reed, J.C. BAG-1 modulates the chaperone activity of Hsp70/Hsc70. EMBO J. 1997, 16, 4887–4896. [Google Scholar] [CrossRef] [PubMed]
- Kawahara, H.; Minami, R.; Yokota, N. BAG6/BAT3: Emerging roles in quality control for nascent polypeptides. J. Biochem. 2013, 153, 147–160. [Google Scholar] [CrossRef] [PubMed]
- Kapadia, M.; De Snoo, M.L.; Kalia, L.V.; Kalia, S.K. Regulation of Parkin-dependent mitophagy by Bcl-2-associated athanogene (BAG) family members. Neural. Regen. Res. 2021, 16, 684–685. [Google Scholar]
- Jain, S.; Wiemann, P.; Thill, E.; Williams, B.; Keller, N.P.; Kabbage, M. A Bcl-2 associated athanogene (bagA) modulates sexual development and secondary metabolism in the filamentous fungus Aspergillus nidulans. Front. Microbiol. 2018, 9, 1316. [Google Scholar] [CrossRef] [PubMed]
- Yan, J.; He, C.; Zhang, H. The BAG-family proteins in Arabidopsis thaliana. Plant Sci. 2003, 165, 1–7. [Google Scholar] [CrossRef]
- Doukhanina, E.V.; Chen, S.; Van der Zalm, E.; Godzik, A.; Reed, J.; Dickman, M.B. Identification and functional characterization of the BAG protein family in Arabidopsis thaliana. J. Biol. Chem. 2006, 281, 18793–18801. [Google Scholar] [CrossRef]
- Kabbage, M.; Dickman, M.B. The BAG proteins: A ubiquitous family of chaperone regulators. Cell. Mol. Life Sci. 2008, 65, 1390–1402. [Google Scholar] [CrossRef] [PubMed]
- Castro, A.; Saavedra, L.; Ruibal, C.; Lascano, R.; Vidal, S. Genome-wide identification, characterization and expression analysis of the Bcl-2 associated athanogene (BAG) gene family in Physcomitrium patens. BioRxiv 2020. [Google Scholar] [CrossRef]
- Nguyen, P.; Hess, K.; Smulders, L.; Le, D.; Briseno, C.; Chavez, C.M.; Nikolaidis, N. Origin and evolution of the human Bcl2-associated athanogene-1 (BAG-1). Int. J. Mol. Sci. 2020, 21, 9701. [Google Scholar] [CrossRef] [PubMed]
- Thanthrige, N.; Jain, S.; Bhowmik, S.D.; Ferguson, B.J.; Kabbage, M.; Mundree, S.; Williams, B. Centrality of BAGs in plant PCD, stress responses, and host defense. Trends Plant Sci. 2020, 25, 1131–1140. [Google Scholar] [CrossRef]
- Li, L.; Xing, Y.; Chang, D.; Fang, S.; Cui, B.; Li, Q.; Wang, X.; Guo, S.; Yang, X.; Men, S.; et al. CaM/BAG5/Hsc70 signaling complex dynamically regulates leaf senescence. Sci. Rep. 2016, 6, 31889. [Google Scholar] [CrossRef] [PubMed]
- Ren, K.; Feng, L.; Sun, S.; Zhuang, X. Plant mitophagy in comparison to mammals: What is still missing? Int. J. Mol. Sci. 2021, 27, 1236. [Google Scholar] [CrossRef] [PubMed]
- Nawkar, G.M.; Maibam, P.; Park, J.H.; Woo, S.G.; Kim, C.Y.; Lee, S.Y.; Kang, C.H. In silico study on Arabidopsis BAG gene expression in response to environmental stresses. Protoplasma 2017, 254, 409–421. [Google Scholar] [CrossRef]
- Lee, D.W.; Kim, S.J.; Oh, Y.J.; Choi, B.; Lee, J.; Hwang, I. Arabidopsis BAG1 functions as a cofactor in Hsc70-mediated proteasomal degradation of unimported plastid proteins. Mol. Plant 2016, 9, 1428–1431. [Google Scholar] [CrossRef]
- Fu, C.; Hou, Y.; Ge, J.; Zhang, L.; Liu, X.; Huo, P.; Liu, J. Increased fes1a thermotolerance is induced by BAG6 knockout. Plant Mol. Biol. 2019, 100, 73–82. [Google Scholar] [CrossRef] [PubMed]
- Williams, B.; Kabbage, M.; Britt, R.; Dickman, M.B. AtBAG7, an Arabidopsis Bcl-2–associated athanogene, resides in the endoplasmic reticulum and is involved in the unfolded protein response. Proc. Natl. Acad. Sci. USA 2010, 107, 6088–6093. [Google Scholar] [CrossRef]
- You, Q.; Zhai, K.; Yang, D.; Yang, W.; Wu, J.; Liu, J.; Pan, W.; Wang, J.; Zhu, X.; Jian, Y.; et al. An E3 ubiquitin ligase-BAG protein module controls plant innate immunity and broad-spectrum disease resistance. Cell Host Microbe 2016, 20, 758–769. [Google Scholar] [CrossRef] [PubMed]
- Locascio, A.; Marqués, M.C.; García-Martínez, G.; Corratgé-Faillie, C.; Andrés-Colás, N.; Rubio, L.; Fernández, J.A.; Véry, A.A.; Mulet, J.M.; Yenush, L. BCL2-ASSOCIATED ATHANOGENE4 regulates the KAT1 potassium channel and controls stomatal movement. Plant Physiol. 2019, 181, 1277–1294. [Google Scholar] [CrossRef] [PubMed]
- Ren, Z.H.; Gao, J.; Li, L.G.; Cai, X.L.; Huang, W.; Chao, D.Y.; Zhu, M.Z.; Wang, Z.Y.; Luan, S.; Lin, H.X. A rice quantitative trait locus for salt tolerance encodes a sodium transporter. Nat. Genet. 2005, 37, 1141–1146. [Google Scholar] [CrossRef]
- Wang, J.; Nan, N.; Li, N.; Liu, Y.; Wang, T.J.; Hwang, I.; Liu, B.; Xu, Z.Y. A DNA methylation reader-chaperone regulator-transcription factor complex activates OsHKT1;5 expression during salinity stress. Plant Cell 2020, 32, 3535–3558. [Google Scholar] [CrossRef] [PubMed]
- Fu, S.; Li, L.; Kang, H.; Yang, X.; Men, S.; Shen, Y. Chronic mitochondrial calcium elevation suppresses leaf senescence. Biochem. Biophys. Res. Commun. 2017, 487, 672–677. [Google Scholar] [CrossRef] [PubMed]
- Kang, C.H.; Jung, W.Y.; Kang, Y.H.; Kim, J.Y.; Kim, D.G.; Jeong, J.C.; Baek, D.W.; Jin, J.B.; Lee, J.Y.; Kim, M.O.; et al. AtBAG6, a novel calmodulin-binding protein, induces programmed cell death in yeast and plants. Cell Death Differ. 2006, 13, 84–95. [Google Scholar] [CrossRef]
- Li, Y.; Kabbage, M.; Liu, W.; Dickman, M.B. Aspartyl protease-mediated cleavage of BAG6 is necessary for autophagy and fungal resistance in plants. Plant Cell 2016, 28, 233–247. [Google Scholar] [CrossRef]
- Echevarría-Zomeño, S.; Fernández-Calvino, L.; Castro-Sanz, A.B.; López, J.A.; Vázquez, J.; Castellano, M.M. Dissecting the proteome dynamics of the early heat stress response leading to plant survival or death in Arabidopsis. Plant Cell Environ. 2016, 39, 1264–1278. [Google Scholar] [CrossRef]
- Zhang, J.X.; Wang, C.; Yang, C.Y.; Wang, J.Y.; Chen, L.; Bao, X.M.; Zhao, Y.X.; Zhang, H.; Liu, J. The role of Arabidopsis AtFes1A in cytosolic Hsp70 stability and abiotic stress tolerance. Plant J. 2010, 62, 539–548. [Google Scholar] [CrossRef] [PubMed]
- Fu, C.; Zhang, J.; Liu, X.; Yang, W.; Yu, H.; Liu, J. AtFes1A is essential for highly efficient molecular chaperone function in Arabidopsis. J. Plant Biol. 2015, 59, 366–373. [Google Scholar] [CrossRef]
- Li, Y.; Williams, B.; Dickman, M. Arabidopsis B-cell lymphoma2 (Bcl-2)-associated athanogene 7 (BAG7)-mediated heat tolerance requires translocation, sumoylation and binding to WRKY 29. New Phytol. 2017, 214, 695–705. [Google Scholar] [CrossRef]
- Pan, Y.J.; Liu, L.; Lin, Y.C.; Zu, Y.G.; Li, L.P.; Tang, Z.H. Ethylene antagonizes salt-induced growth retardation and cell death process via transcriptional controlling of ethylene-, BAG- and senescence-associated genes in Arabidopsis. Front. Plant Sci. 2016, 7, 696. [Google Scholar] [CrossRef]
- Yamaguchi-Shinozaki, K.; Shinozaki, K. Characterization of the expression of a desiccation-responsive rd29 gene of Arabidopsis thaliana and analysis of its promoter in transgenic plants. Mol. Gen. Genet. 1993, 236, 331–340. [Google Scholar] [CrossRef]
- Yamaguchi-Shinozaki, K.; Shinozaki, K. A novel cis-acting element in an Arabidopsis gene is involved in responsiveness to drought, low-temperature, or high-salt stress. Plant Cell 1994, 6, 251–264. [Google Scholar]
- Narusaka, Y.; Nakashima, K.; Shinwari, Z.K.; Sakuma, Y.; Furihata, T.; Abe, H.; Narusaka, M.; Shinozaki, K.; Yamaguchi-Shinozaki, K. Interaction between two cis-acting elements, ABRE and DRE, in ABA-dependent expression of Arabidopsis rd29A gene in response to dehydration and high-salinity stresses. Plant J. 2003, 34, 137–148. [Google Scholar] [CrossRef] [PubMed]
- Nakashima, K.; Fujita, Y.; Katsura, K.; Maruyama, K.; Narusaka, Y.; Seki, M.; Shinozaki, K.; Yamaguchi-Shinozaki, K. Transcriptional regulation of ABI3- and ABA-responsive genes including RD29B and RD29A in seeds, germinating embryos, and seedlings of Arabidopsis. Plant Mol. Biol. 2006, 60, 51–68. [Google Scholar] [CrossRef] [PubMed]
- Iuchi, S.; Kobayashi, M.; Taji, T.; Naramoto, M.; Seki, M.; Kato, T.; Tabata, S.; Kakubari, Y.; Yamaguchi-Shinozaki, K.; Shinozaki, K. Regulation of drought tolerance by gene manipulation of 9-cis-epoxycarotenoid dioxygenase, a key enzyme in abscisic acid biosynthesis in Arabidopsis. Plant J. 2001, 27, 325–333. [Google Scholar] [CrossRef] [PubMed]
- Soderman, E.M.; Brocard, I.M.; Lynch, T.J.; Finkelstein, R.R. Regulation and function of the arabidopsis ABA-insensitive4 gene in seed and abscisic acid response signaling networks. Plant Physiol. 2000, 124, 1752–1765. [Google Scholar] [CrossRef]
- Kumpf, R.P.; Nowack, M.K. The root cap: A short story of life and death. J. Exp. Bot. 2015, 66, 5651–5662. [Google Scholar] [CrossRef]
- Hellmann, E.; Ko, D.; Ruonala, R.; Helariutta, Y. Plant vascular tissues-connecting tissue comes in all shapes. Plants 2018, 7, 109. [Google Scholar] [CrossRef]
- Bao, S.; Shen, G.; Li, G.; Liu, Z.; Arif, M.; Wei, Q.; Men, S. The Arabidopsis nucleoporin NUP1 is essential for megasporogenesis and early stages of pollen development. Plant Cell Rep. 2019, 38, 59–74. [Google Scholar] [CrossRef] [PubMed]
- Clough, S.J.; Bent, A.F. Floral dip: A simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J. 1998, 16, 735–743. [Google Scholar] [CrossRef]
- Liu, H.; Liu, B.; Chen, X.; Zhu, H.; Zou, C.; Men, S. AUX1 acts upstream of PIN2 in regulating root gravitropism. Biochem. Biophys. Res. Commun. 2018, 507, 433–436. [Google Scholar] [CrossRef] [PubMed]
- Seo, K.I.; Lee, J.H.; Nezames, C.D.; Zhong, S.; Song, E.; Byun, M.O.; Deng, X.W. ABD1 is an Arabidopsis DCAF substrate receptor for CUL4-DDB1–based E3 ligases that acts as a negative regulator of abscisic acid signaling. Plant Cell 2014, 26, 695–711. [Google Scholar] [CrossRef] [PubMed]
- Arnon, D.I. Copper enzymes in isolated chloroplasts. Polyphenoloxidase in Beta vulgaris. Plant Physiol. 1949, 24, 1–15. [Google Scholar] [CrossRef]
- Yang, L.; Wang, S.; Sun, L.; Ruan, M.; Li, S.; He, R.; Zhang, W.; Liang, C.; Wang, X.; Bi, Y. Involvement of G6PD5 in ABA response during seed germination and root growth in Arabidopsis. BMC Plant Biol. 2019, 19, 44. [Google Scholar] [CrossRef]
- Wang, M.; Li, P.; Ma, Y.; Nie, X.; Grebe, M.; Men, S. Membrane sterol composition in Arabidopsis thaliana affects root elongation via auxin biosynthesis. Int. J. Mol. Sci. 2021, 22, 437. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Li, Z.; Yang, Z.; Leshem, Y.; Shen, Y.; Men, S. Mitochondrial heat shock cognate protein 70 contributes to auxinmediated embryo development. Plant. Physiol. 2021. [Google Scholar] [CrossRef] [PubMed]




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Arif, M.; Li, Z.; Luo, Q.; Li, L.; Shen, Y.; Men, S. The BAG2 and BAG6 Genes Are Involved in Multiple Abiotic Stress Tolerances in Arabidopsis Thaliana. Int. J. Mol. Sci. 2021, 22, 5856. https://doi.org/10.3390/ijms22115856
Arif M, Li Z, Luo Q, Li L, Shen Y, Men S. The BAG2 and BAG6 Genes Are Involved in Multiple Abiotic Stress Tolerances in Arabidopsis Thaliana. International Journal of Molecular Sciences. 2021; 22(11):5856. https://doi.org/10.3390/ijms22115856
Chicago/Turabian StyleArif, Muhammad, Zitong Li, Qiong Luo, Luhua Li, Yuequan Shen, and Shuzhen Men. 2021. "The BAG2 and BAG6 Genes Are Involved in Multiple Abiotic Stress Tolerances in Arabidopsis Thaliana" International Journal of Molecular Sciences 22, no. 11: 5856. https://doi.org/10.3390/ijms22115856
APA StyleArif, M., Li, Z., Luo, Q., Li, L., Shen, Y., & Men, S. (2021). The BAG2 and BAG6 Genes Are Involved in Multiple Abiotic Stress Tolerances in Arabidopsis Thaliana. International Journal of Molecular Sciences, 22(11), 5856. https://doi.org/10.3390/ijms22115856





