Characterization of an Endolysin Targeting Clostridioides difficile That Affects Spore Outgrowth
Abstract
1. Introduction
2. Results and Discussion
2.1. In Silico Analysis of C. difficile and Phages Infecting C. difficile in Search of Lytic Enzymes
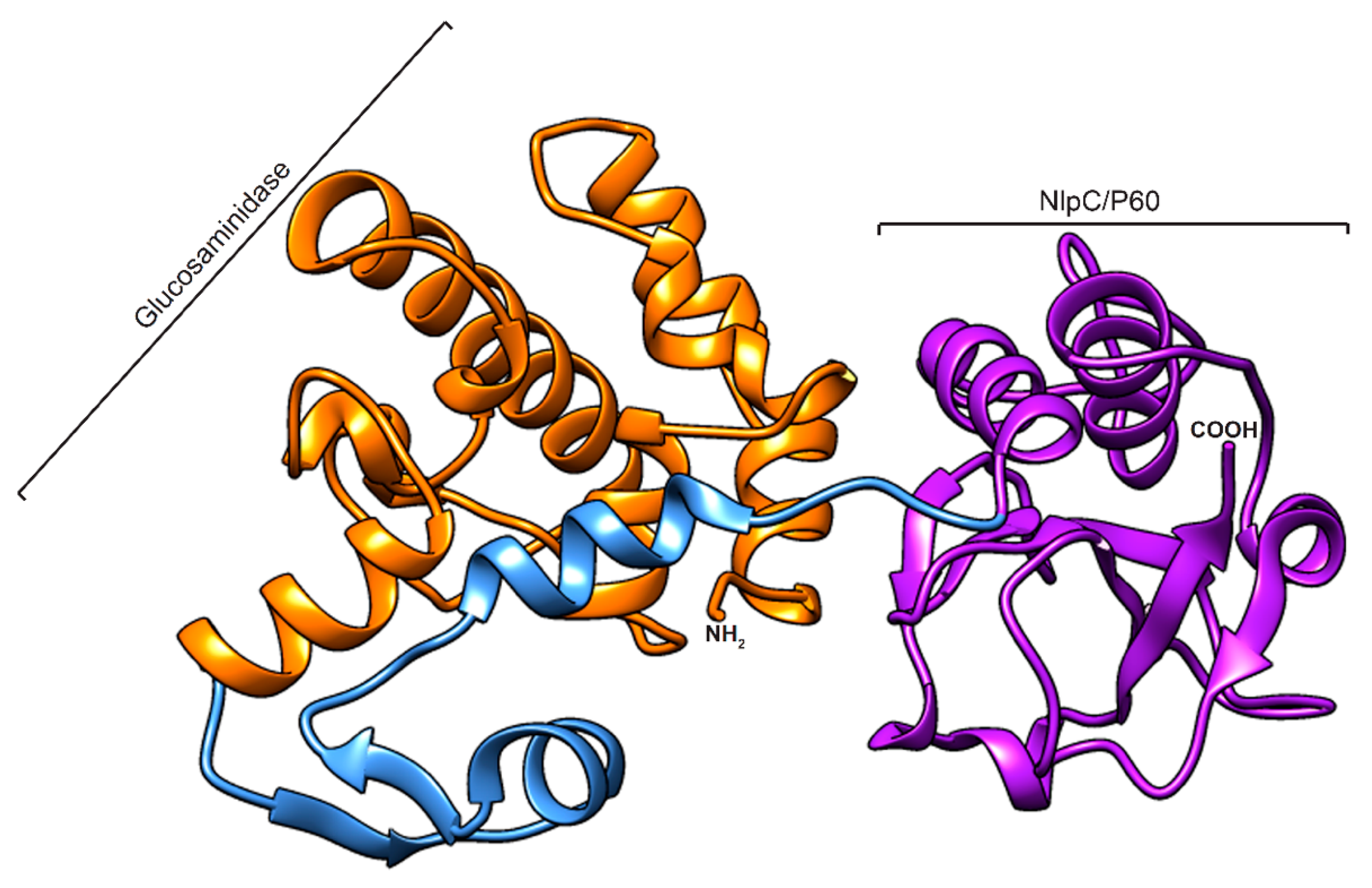
2.2. Homology Modelling
2.3. Expression and Lytic Activity of Endolysins
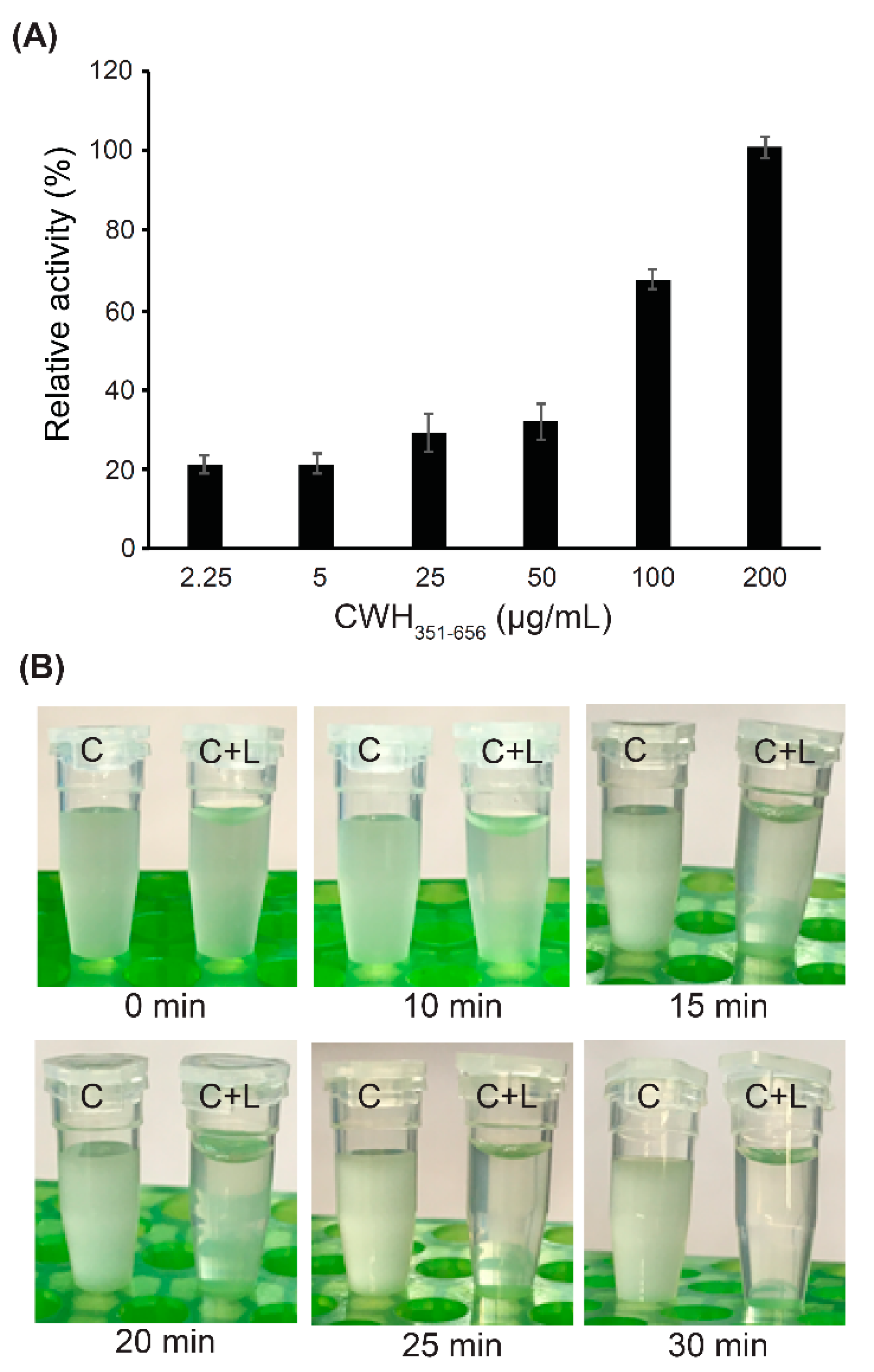
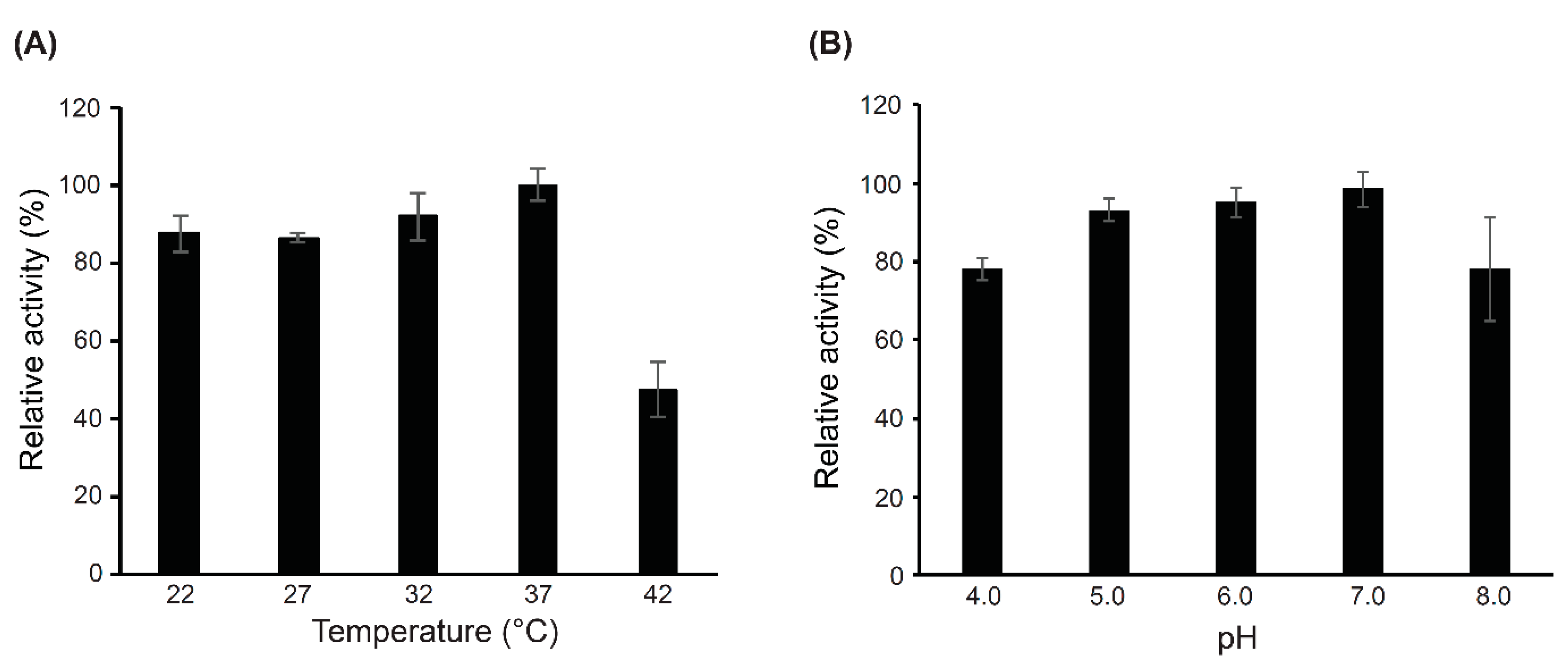
2.4. Characterization of the Catalytic Domains (CWH351—656) of Cell Wall Hydrolase (CWH)
2.5. Host Specificity of Full-Length CWH and CWH351—656
2.6. Optical Microscopy of Lytic Activity
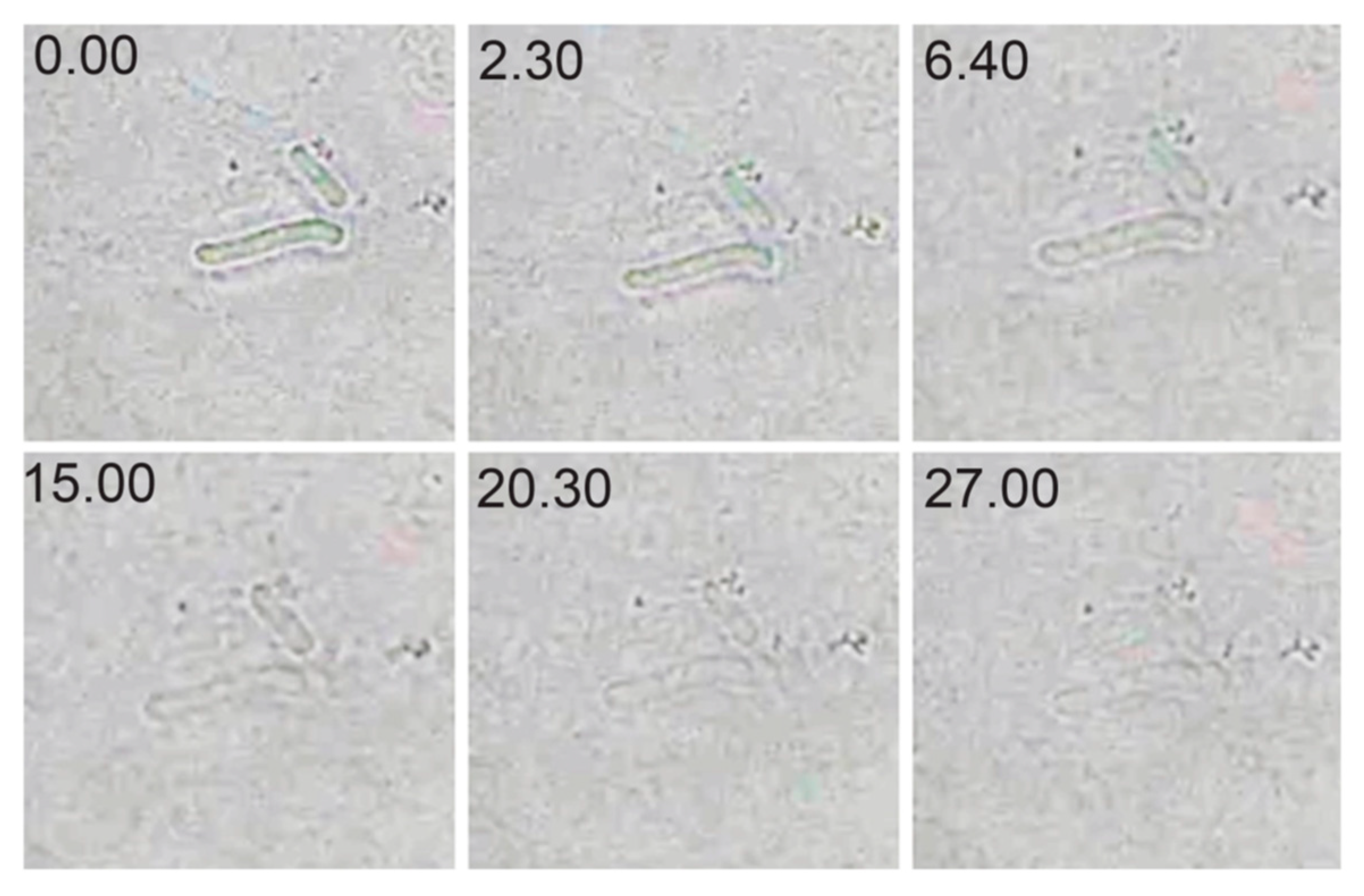
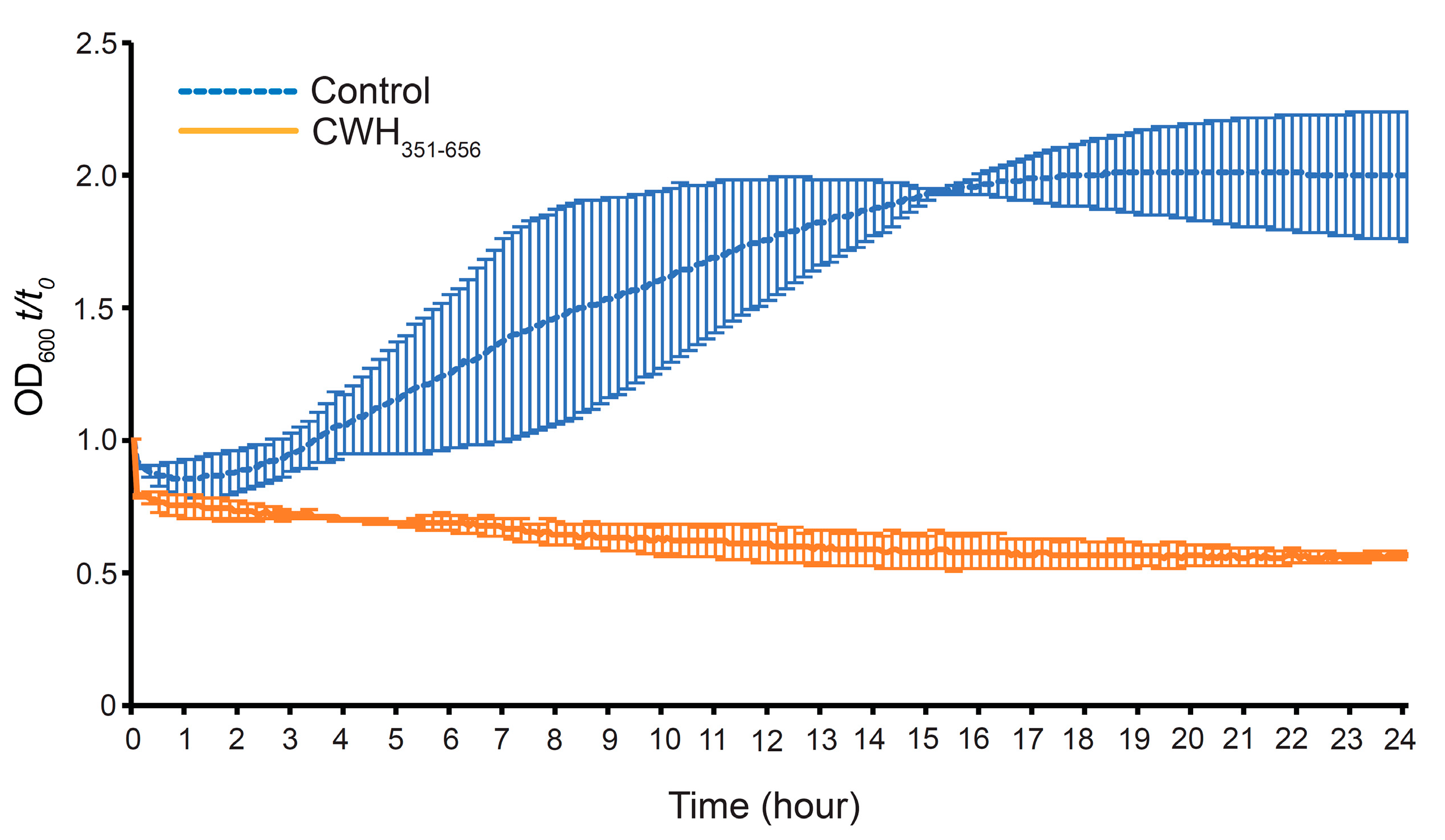
2.7. Effect of CWH351—656 on C. difficile Spore Outgrowth
3. Materials and Methods
3.1. Bacterial Strains and Growth Conditions
3.2. Cloning, Expression, and Purification of CWH and Its Subdomains
3.3. Structural Modelling of CWH351—656
3.4. Lytic Activity Assays
3.5. Optical Microscopy of Lytic Activity
3.6. C. difficile Spore Preparation
3.7. Effect of CWH351—656 on C. difficile Spore Germination and Outgrowth
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Weese, J.S. Clostridium difficile in food: Innocent bystander or serious threat? Clin. Microbiol. Infect. 2010, 16, 3–10. [Google Scholar] [CrossRef]
- Kuijper, E.J.; Coignard, B.; Tüll, P. Emergence of Clostridium difficile-associated disease in North America and Europe. Clin. Microbiol. Infect. 2006, 12, 2–18. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention. Antibiotic Resistance Threats in the United States; U.S. Department of Health and Human Services: Atlanta, GA, USA, 2019. [CrossRef]
- Guh, A.Y.; Mu, Y.; Winston, L.G.; Johnston, H.; Olson, D.; Farley, M.M.; Wilson, L.E.; Holzbauer, S.M.; Phipps, E.C.; Dumyati, G.K.; et al. Trends in U.S. Burden of Clostridioides difficile Infection and Outcomes. N. Engl. J. Med. 2020, 382, 1320–1330. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Prabhu, V.S.; Marcella, S.W. Attributable Healthcare Resource Utilization and Costs for Patients with Primary and Recurrent Clostridium difficile Infection in the United States. Clin. Infect. Dis. 2018, 66, 1326–1332. [Google Scholar] [CrossRef]
- Bouza, E. Consequences of Clostridium difficile infection: Understanding the healthcare burden. Clin. Microbiol. Infect. 2012, 18, 5–12. [Google Scholar] [CrossRef]
- Vaishnavi, C. Clinical spectrum & pathogenesis of Clostridium difficile associated diseases. Indian J. Med Res. 2010, 131, 487–499. [Google Scholar]
- McDonald, L.C.; Gerding, D.N.; Johnson, S.; Bakken, J.S.; Carroll, K.C.; Coffin, S.E.; Dubberke, E.R.; Garey, K.W.; Gould, C.V.; Kelly, C.; et al. Clinical Practice Guidelines for Clostridium difficile Infection in Adults and Children: 2017 Update by the Infectious Diseases Society of America (IDSA) and Society for Healthcare Epidemiology of America (SHEA). Clin. Infect. Dis. 2018, 66, e1–e48. [Google Scholar] [CrossRef] [PubMed]
- Khanna, S. Defending against a difficult clostridioides with a vaccine. Lancet Infect. Dis. 2021, 21, 157–158. [Google Scholar] [CrossRef]
- Deakin, L.J.; Clare, S.; Fagan, R.P.; Dawson, L.F.; Pickard, D.J.; West, M.R.; Wren, B.W.; Fairweather, N.F.; Dougan, G.; Lawley, T.D. The Clostridium difficile spo0AGene Is a Persistence and Transmission Factor. Infect. Immun. 2012, 80, 2704–2711. [Google Scholar] [CrossRef]
- Ma, G.K.; Brensinger, C.M.; Wu, Q.; Lewis, J.D. Increasing Incidence of Multiply Recurrent Clostridium difficile Infection in the United States. Ann. Intern. Med. 2017, 167, 152–158. [Google Scholar] [CrossRef]
- Leffler, D.A.; Lamont, J.T. Treatment of Clostridium Difficile-Associated Disease. Gastroenterology 2009, 136, 1899–1912. [Google Scholar] [CrossRef]
- Monaghan, T.; Boswell, T.; Mahida, Y.R. Recent advances in Clostridium difficile-associated disease. Postgrad. Med. J. 2009, 85, 152–162. [Google Scholar] [CrossRef]
- Abt, M.C.; McKenney, P.T.; Pamer, E.G. Clostridium difficile colitis: Pathogenesis and host defence. Nat. Rev. Genet. 2016, 14, 609–620. [Google Scholar] [CrossRef]
- Venugopal, A.A.; Johnson, S. Fidaxomicin: A Novel Macrocyclic Antibiotic Approved for Treatment of Clostridium difficile Infection. Clin. Infect. Dis. 2012, 54, 568–574. [Google Scholar] [CrossRef]
- Bartsch, S.M.; Umscheid, C.A.; Fishman, N.; Lee, B.Y. Is Fidaxomicin Worth the Cost? An Economic Analysis. Clin. Infect. Dis. 2013, 57, 555–561. [Google Scholar] [CrossRef]
- Rohlke, F.; Stollman, N. Fecal microbiota transplantation in relapsing Clostridium difficile infection. Ther. Adv. Gastroenterol. 2012, 5, 403–420. [Google Scholar] [CrossRef]
- Kalakuntla, A.S.; Nalakonda, G.; Nalakonda, K.; Pidikiti, C.V.; Aasim, S.A. Probiotics and Clostridium difficile: A Review of Dysbiosis and the Rehabilitation of Gut Microbiota. Cureus 2019, 11, e5063. [Google Scholar] [CrossRef] [PubMed]
- O’Horo, J.C.; Jindai, K.; Kunzer, B.; Safdar, N. Treatment of recurrent Clostridium difficile infection: A systematic review. Infection 2013, 42, 43–59. [Google Scholar] [CrossRef]
- Abedon, S.T.; Kuhl, S.J.; Blasdel, B.G.; Kutter, E.M. Phage treatment of human infections. Bacteriophage 2011, 1, 66–85. [Google Scholar] [CrossRef]
- Nilsson, A.S. Phage therapy—constraints and possibilities. Upsala J. Med. Sci. 2014, 119, 192–198. [Google Scholar] [CrossRef] [PubMed]
- Loc-Carrillo, C.; Abedon, S.T. Pros and cons of phage therapy. Bacteriophage 2011, 1, 111–114. [Google Scholar] [CrossRef]
- Hargreaves, K.R.; Clokie, M.R.J. Clostridium difficile phages: Still difficult? Front. Microbiol. 2014, 5, 184. [Google Scholar] [CrossRef]
- Zucca, M.; Scutera, S.; Savoia, D. Novel avenues for Clostridium difficile infection drug discovery. Expert Opin. Drug Discov. 2013, 8, 459–477. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Zhang, Y.; Dong, K.; Kuo, C.J.; Li, C.; Zhu, Y.Q.; Qin, J.; Li, Q.T.; Chang, Y.F.; Guo, X.; et al. Isolation and Characterization of the Novel Phage JD032 and Global Transcriptomic Response during JD032 Infection of Clostridioides difficile Ribotype 078. mSystems 2020, 5, e00017–e00020. [Google Scholar] [CrossRef]
- Nale, J.Y.; Spencer, J.; Hargreaves, K.R.; Buckley, A.M.; Trzepiński, P.; Douce, G.R.; Clokie, M.R.J. Bacteriophage Combinations Significantly Reduce Clostridium difficile GrowthIn Vitroand Proliferation In Vivo. Antimicrob. Agents Chemother. 2016, 60, 968–981. [Google Scholar] [CrossRef] [PubMed]
- Nale, J.Y.; Redgwell, T.A.; Millard, A.; Clokie, M.R.J. Efficacy of an Optimised Bacteriophage Cocktail to Clear Clostridium difficile in a Batch Fermentation Model. Antibiotics 2018, 7, 13. [Google Scholar] [CrossRef]
- Schmelcher, M.; Donovan, D.M.; Loessner, M.J. Bacteriophage endolysins as novel antimicrobials. Futur. Microbiol. 2012, 7, 1147–1171. [Google Scholar] [CrossRef] [PubMed]
- Nelson, D.; Loomis, L.; Fischetti, V.A. Prevention and elimination of upper respiratory colonization of mice by group A streptococci by using a bacteriophage lytic enzyme. Proc. Natl. Acad. Sci. USA 2001, 98, 4107–4112. [Google Scholar] [CrossRef]
- Peng, Z.; Wang, S.; Gide, M.; Zhu, D.; Patabendige, H.M.L.W.; Li, C.; Cai, J.; Sun, X. A Novel Bacteriophage Lysin-Human Defensin Fusion Protein Is Effective in Treatment of Clostridioides difficile Infection in Mice. Front. Microbiol. 2019, 9, 3234. [Google Scholar] [CrossRef] [PubMed]
- Mehta, K.K.; Paskaleva, E.E.; Wu, X.; Grover, N.; Mundra, R.V.; Chen, K.; Zhang, Y.; Yang, Z.; Feng, H.; Dordick, J.S.; et al. Newly identified bacteriolytic enzymes that target a wide range of clinical isolates of Clostridium difficile. Biotechnol. Bioeng. 2016, 113, 2568–2576. [Google Scholar] [CrossRef]
- Mondal, S.I.; Draper, L.A.; Ross, R.P.; Hill, C. Bacteriophage endolysins as a potential weapon to combat Clostridioides difficile infection. Gut Microbes. 2020, 12, e1813533. [Google Scholar] [CrossRef]
- Mayer, M.J.; Narbad, A.; Gasson, M.J. Molecular Characterization of a Clostridium difficile Bacteriophage and Its Cloned Biologically Active Endolysin. J. Bacteriol. 2008, 190, 6734–6740. [Google Scholar] [CrossRef]
- Mayer, M.J.; Garefalaki, V.; Spoerl, R.; Narbad, A.; Meijers, R. Structure-Based Modification of a Clostridium difficile-Targeting Endolysin Affects Activity and Host Range. J. Bacteriol. 2011, 193, 5477–5486. [Google Scholar] [CrossRef]
- Wang, Q.; Euler, C.W.; Delaune, A.; Fischetti, V.A. Using a Novel Lysin To Help Control Clostridium difficile Infections. Antimicrob. Agents Chemother. 2015, 59, 7447–7457. [Google Scholar] [CrossRef] [PubMed]
- Meessen-Pinard, M.; Sekulovic, O.; Fortier, L.C. Evidence of In Vivo Prophage Induction during Clostridium difficile Infection. Appl. Environ. Microbiol. 2012, 78, 7662–7670. [Google Scholar] [CrossRef]
- Boudry, P.; Semenova, E.; Monot, M.; Datsenko, K.A.; Lopatina, A.; Sekulovic, O.; Ospina-Bedoya, M.; Fortier, L.C.; Severinov, K.; Dupuy, B.; et al. Function of the CRISPR-Cas System of the Human Pathogen Clostridium difficile. mBio 2015, 6, e01112-15. [Google Scholar] [CrossRef]
- Marchler-Bauer, A.; Bo, Y.; Han, L.; He, J.; Lanczycki, C.J.; Lu, S.; Chitsaz, F.; Derbyshire, M.K.; Geer, R.C.; Gonzales, N.R.; et al. CDD/SPARCLE: Functional classification of proteins via subfamily domain architectures. Nucleic Acids Res. 2017, 45, D200–D203. [Google Scholar] [CrossRef]
- El-Gebali, S.; Mistry, J.; Bateman, A.; Eddy, S.R.; Luciani, A.; Potter, S.C.; Qureshi, M.; Richardson, L.J.; Salazar, G.A.; Smart, A.; et al. The Pfam protein families database in 2019. Nucleic Acids Res. 2018, 47, D427–D432. [Google Scholar] [CrossRef]
- Gabler, F.; Nam, S.; Till, S.; Mirdita, M.; Steinegger, M.; Söding, J.; Lupas, A.N.; Alva, V. Protein Sequence Analysis Using the MPI Bioinformatics Toolkit. Curr. Protoc. Bioinform. 2020, 72, e108. [Google Scholar] [CrossRef]
- Darzentas, N. Circoletto: Visualizing sequence similarity with Circos. Bioinformatics 2010, 26, 2620–2621. [Google Scholar] [CrossRef]
- Porter, C.J.; Schuch, R.; Pelzek, A.J.; Buckle, A.M.; McGowan, S.; Wilce, M.C.; Rossjohn, J.; Russell, R.; Nelson, D.; Fischetti, V.A.; et al. The 1.6 Å Crystal Structure of the Catalytic Domain of PlyB, a Bacteriophage Lysin Active Against Bacillus anthracis. J. Mol. Biol. 2007, 366, 540–550. [Google Scholar] [CrossRef]
- Low, L.Y.; Yang, C.; Perego, M.; Osterman, A.; Liddington, R.C. Structure and Lytic Activity of a Bacillus anthracis Prophage Endolysin. J. Biol. Chem. 2005, 280, 35433–35439. [Google Scholar] [CrossRef]
- Horgan, M.; O’Flynn, G.; Garry, J.; Cooney, J.; Coffey, A.; Fitzgerald, G.F.; Ross, R.P.; McAuliffe, O. Phage Lysin LysK Can Be Truncated to Its CHAP Domain and Retain Lytic Activity against Live Antibiotic-Resistant Staphylococci. Appl. Environ. Microbiol. 2008, 75, 872–874. [Google Scholar] [CrossRef]
- Cheng, Q.; Fischetti, V.A. Mutagenesis of a bacteriophage lytic enzyme PlyGBS significantly increases its antibacterial activity against group B streptococci. Appl. Microbiol. Biotechnol. 2007, 74, 1284–1291. [Google Scholar] [CrossRef]
- Meroueh, S.O.; Bencze, K.Z.; Hesek, D.; Lee, M.; Fisher, J.F.; Stemmler, T.L.; Mobashery, S. Three-dimensional structure of the bacterial cell wall peptidoglycan. Proc. Natl. Acad. Sci. USA 2006, 103, 4404–4409. [Google Scholar] [CrossRef]
- Low, L.Y.; Yang, C.; Perego, M.; Osterman, A.; Liddington, R. Role of Net Charge on Catalytic Domain and Influence of Cell Wall Binding Domain on Bactericidal Activity, Specificity, and Host Range of Phage Lysins. J. Biol. Chem. 2011, 286, 34391–34403. [Google Scholar] [CrossRef]
- Shang, X.; Nelson, D.C. Contributions of Net Charge on the PlyC Endolysin CHAP Domain. Antibiotics 2019, 8, 70. [Google Scholar] [CrossRef]
- Gilmer, D.B.; Schmitz, J.E.; Euler, C.W.; Fischetti, V.A. Novel Bacteriophage Lysin with Broad Lytic Activity Protects against Mixed Infection by Streptococcus pyogenes and Methicillin-Resistant Staphylococcus aureus. Antimicrob. Agents Chemother. 2013, 57, 2743–2750. [Google Scholar] [CrossRef]
- Schleifer, K.H.; Kandler, O. Peptidoglycan types of bacterial cell walls and their taxonomic implications. Bacteriol. Rev. 1972, 36, 407–477. [Google Scholar] [CrossRef]
- Cummins, C.S.; Johnson, J.L. Taxonomy of the Clostridia: Wall Composition and DNA Homologies in Clostridium butyricum and Other Butyric Acid-producing Clostridia. J. Gen. Microbiol. 1971, 67, 33–46. [Google Scholar] [CrossRef]
- Barbut, F.; Menuet, D.; Verachten, M.; Girou, E. Comparison of the Efficacy of a Hydrogen Peroxide Dry-Mist Disinfection System and Sodium Hypochlorite Solution for Eradication of Clostridium difficile Spores. Infect. Control. Hosp. Epidemiol. 2009, 30, 507–514. [Google Scholar] [CrossRef]
- Burns, D.A.; Heap, J.T.; Minton, N.P. Clostridium difficile spore germination: An update. Res. Microbiol. 2010, 161, 730–734. [Google Scholar] [CrossRef]
- Berman, H.M.; Westbrook, J.; Feng, Z.; Gilliland, G.; Bhat, T.N.; Weissig, H.; Shindyalov, I.N.; Bourne, P.E. The Protein Data Bank. Nucleic Acids Res. 2000, 28, 235–242. [Google Scholar] [CrossRef]
- Xu, D.; Zhang, Y. Improving the Physical Realism and Structural Accuracy of Protein Models by a Two-Step Atomic-Level Energy Minimization. Biophys. J. 2011, 101, 2525–2534. [Google Scholar] [CrossRef]
- Laskowski, R.; MacArthur, M.W.; Moss, D.S.; Thornton, J. PROCHECK: A program to check the stereochemical quality of protein structures. J. Appl. Crystallogr. 1993, 26, 283–291. [Google Scholar] [CrossRef]
- Colovos, C.; Yeates, T.O. Verification of protein structures: Patterns of nonbonded atomic interactions. Protein Sci. 1993, 2, 1511–1519. [Google Scholar] [CrossRef]
- Pettersen, E.F.; Goddard, T.D.; Huang, C.C.; Couch, G.S.; Greenblatt, D.M.; Meng, E.C.; Ferrin, T.E. UCSF Chimera—A visualization system for exploratory research and analysis. J. Comput. Chem. 2004, 25, 1605–1612. [Google Scholar] [CrossRef]
- Westbye, A.; Fogg, P.; Beatty, J. Endolysin Expression, Purification and Activity Determination by Zymography. Bio-protocol 2014, 4, e1208. [Google Scholar] [CrossRef]
- Theriot, C.M.; Koenigsknecht, M.J., Jr.; Carlson, P.E.; Hatton, G.E.; Nelson, A.M.; Li, B.; Huffnagle, G.B.; Li, J.Z.; Young, V.B. Antibiotic-induced shifts in the mouse gut microbiome and metabolome increase susceptibility to Clostridium difficile infection. Nat. Commun. 2014, 5, 3114. [Google Scholar] [CrossRef]
- Perez, J.; Springthorpe, V.S.; A Sattar, S. Clospore: A Liquid Medium for Producing High Titers of Semi-purified Spores of Clostridium difficile. J. AOAC Int. 2011, 94, 618–626. [Google Scholar] [CrossRef]
- Paredes-Sabja, D.; Bond, C.; Carman, R.J.; Setlow, P.; Sarker, M.R. Germination of spores of Clostridium difficile strains, including isolates from a hospital outbreak of Clostridium difficile-associated disease (CDAD). Microbiology 2008, 154, 2241–2250. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Allen, C.A.; Babakhani, F.; Sears, P.; Nguyen, L.; Sorg, J.A. Both Fidaxomicin and Vancomycin Inhibit Outgrowth of Clostridium difficile Spores. Antimicrob. Agents Chemother. 2013, 57, 664–667. [Google Scholar] [CrossRef] [PubMed]


| Clostridioides Strains | Lysin Activity # | |
|---|---|---|
| CWH | CWH351—656 | |
| Clostridioides difficile 43255 | +++ | +++ |
| Clostridioides difficile APC 1401 | ++ | +++ |
| Clostridioides difficile 630 | ++ | +++ |
| Clostridioides difficile APC 1412 | +++ | +++ |
| Clostridioides difficile 28196 | +++ | +++ |
| Non-Clostridioides Strains | ||
| Bacillus cereus | + | ++ |
| Clostridium perfringens | – | – |
| Clostridium scindens | + | ++ |
| Clostridium symbiosum | – | – |
| Enterococcus faecalis | – | + |
| Enterococcus faecium | – | – |
| Lactobacillus paracasei | – | – |
| Lactococcus lactis | – | – |
| Listeria innocua | – | + |
| Listeria monocytogenes | – | – |
| Staphylococcus aureus | – | – |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mondal, S.I.; Akter, A.; Draper, L.A.; Ross, R.P.; Hill, C. Characterization of an Endolysin Targeting Clostridioides difficile That Affects Spore Outgrowth. Int. J. Mol. Sci. 2021, 22, 5690. https://doi.org/10.3390/ijms22115690
Mondal SI, Akter A, Draper LA, Ross RP, Hill C. Characterization of an Endolysin Targeting Clostridioides difficile That Affects Spore Outgrowth. International Journal of Molecular Sciences. 2021; 22(11):5690. https://doi.org/10.3390/ijms22115690
Chicago/Turabian StyleMondal, Shakhinur Islam, Arzuba Akter, Lorraine A. Draper, R. Paul Ross, and Colin Hill. 2021. "Characterization of an Endolysin Targeting Clostridioides difficile That Affects Spore Outgrowth" International Journal of Molecular Sciences 22, no. 11: 5690. https://doi.org/10.3390/ijms22115690
APA StyleMondal, S. I., Akter, A., Draper, L. A., Ross, R. P., & Hill, C. (2021). Characterization of an Endolysin Targeting Clostridioides difficile That Affects Spore Outgrowth. International Journal of Molecular Sciences, 22(11), 5690. https://doi.org/10.3390/ijms22115690






